Ni-CeO2/C Catalysts with Enhanced OSC for the WGS Reaction
Abstract
:1. Introduction
2. Results and Discussion
2.1. Catalysts Composition and Textural Properties
| Sample | SBET(m2/g) | Vmicro(cm3/g) | Vmeso(cm3/g) | Ni (wt.%) * | CeO2 (wt.%)* |
|---|---|---|---|---|---|
| C | 1487 | 0.52 | 0.62 | -- | -- |
| 20CeO2/C | 1083 | 0.37 | 0.47 | -- | 22.2 |
| CeO2 | 101 | 0.04 | 0.07 | -- | -- |
| Ni20CeO2/C | 807 | 0.28 | 0.34 | 12.7 | 21.6 |
| Ni/CeO2 | 70 | 0.03 | 0.04 | 14.1 | -- |
2.2. OSCC and OSC
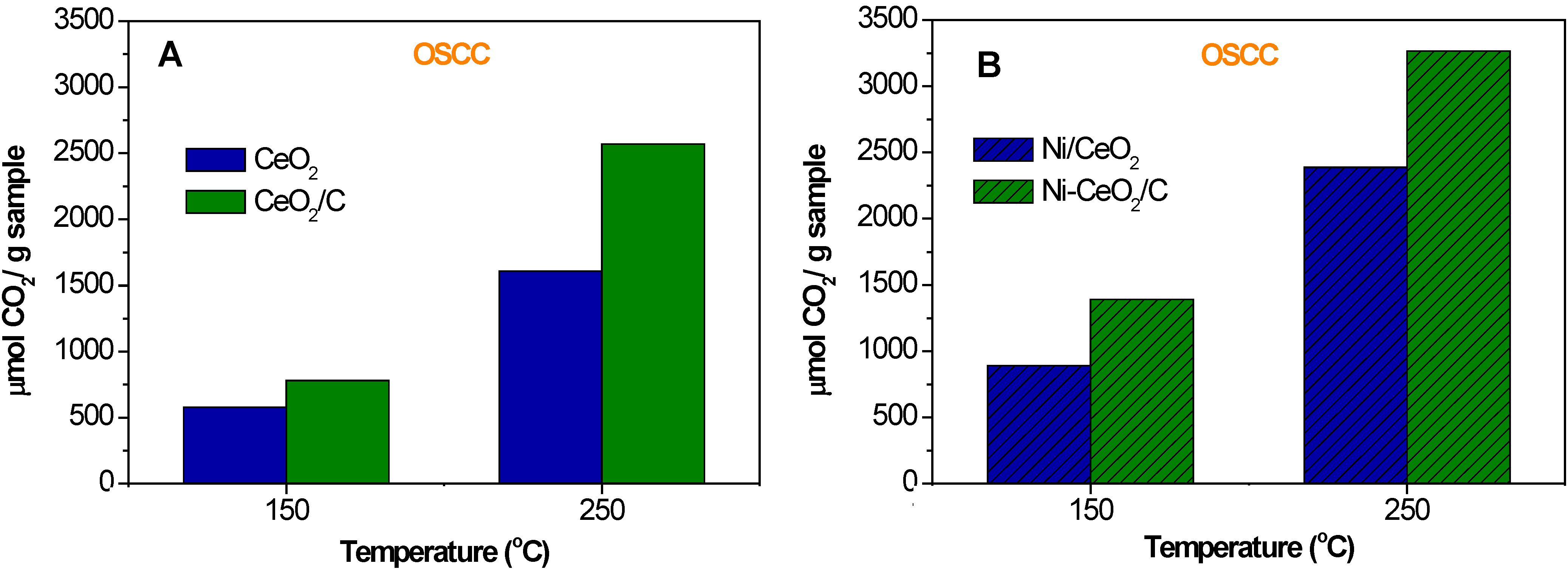
| Sample | OSC150 °C | OSC250 °C |
|---|---|---|
| CeO2 | 10 | 180 |
| CeO2/C | 25 | 255 |
| Ni/CeO2 | 410 | 726 |
| Ni-CeO2/C | 620 | 850 |
2.3. WGS Behavior
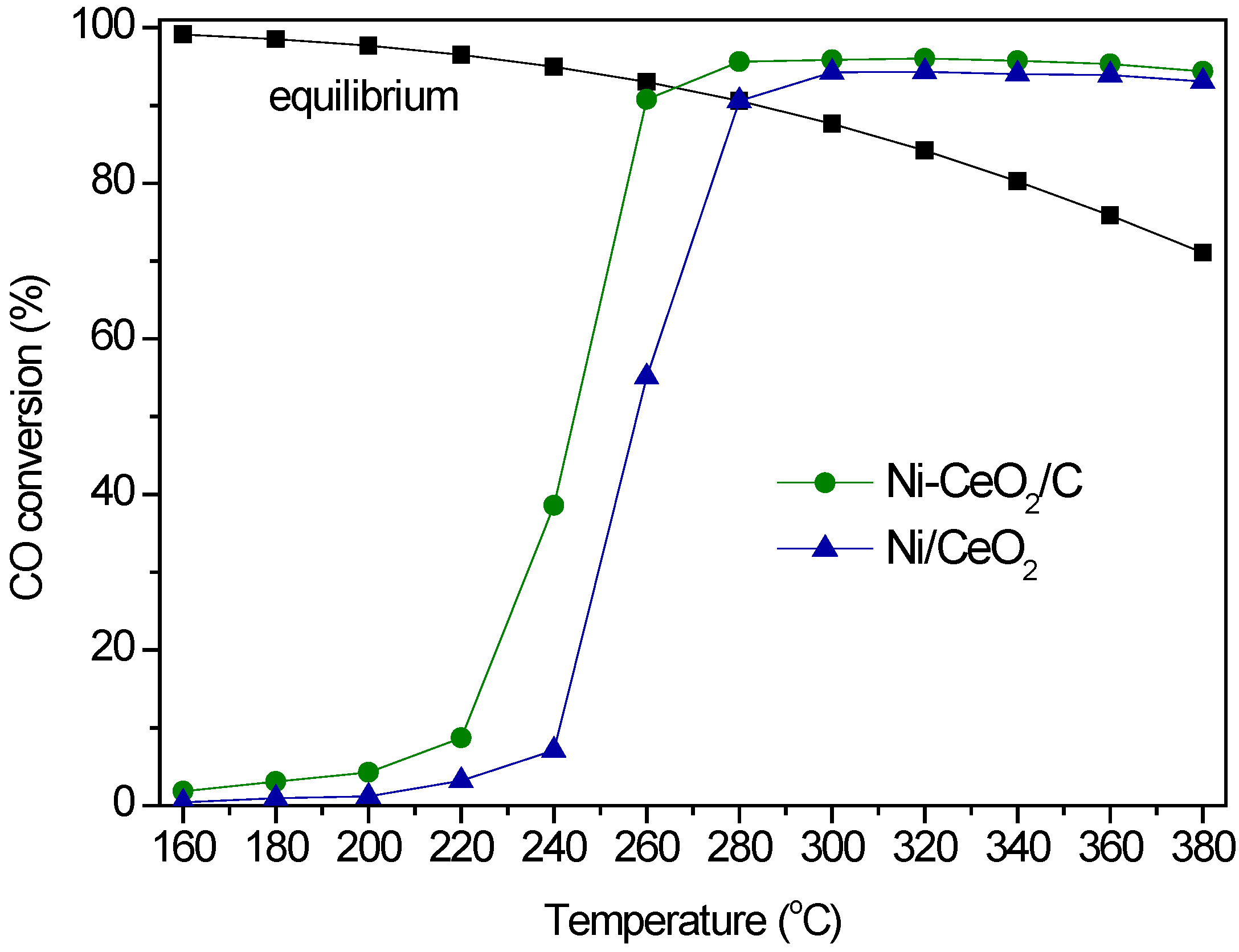
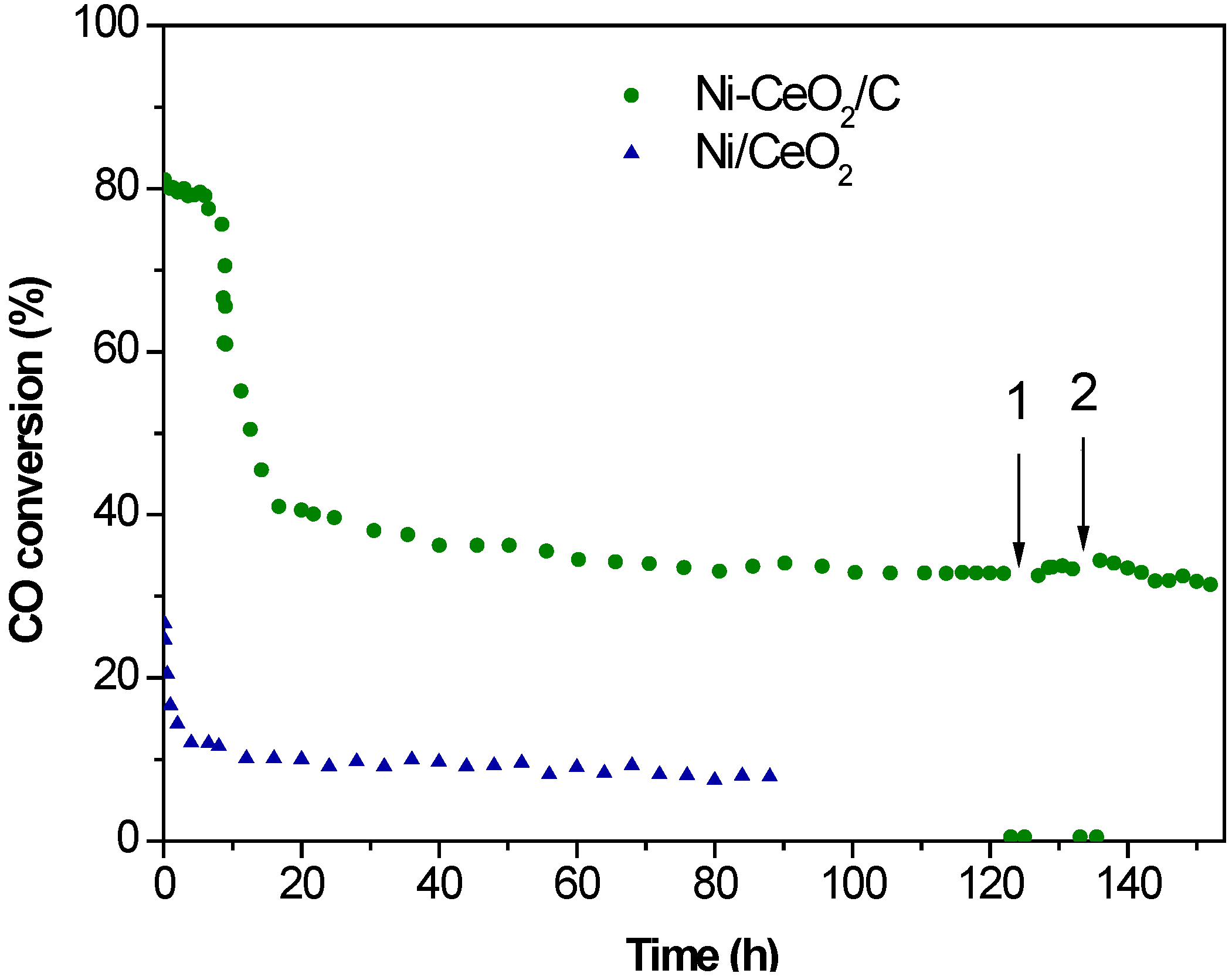
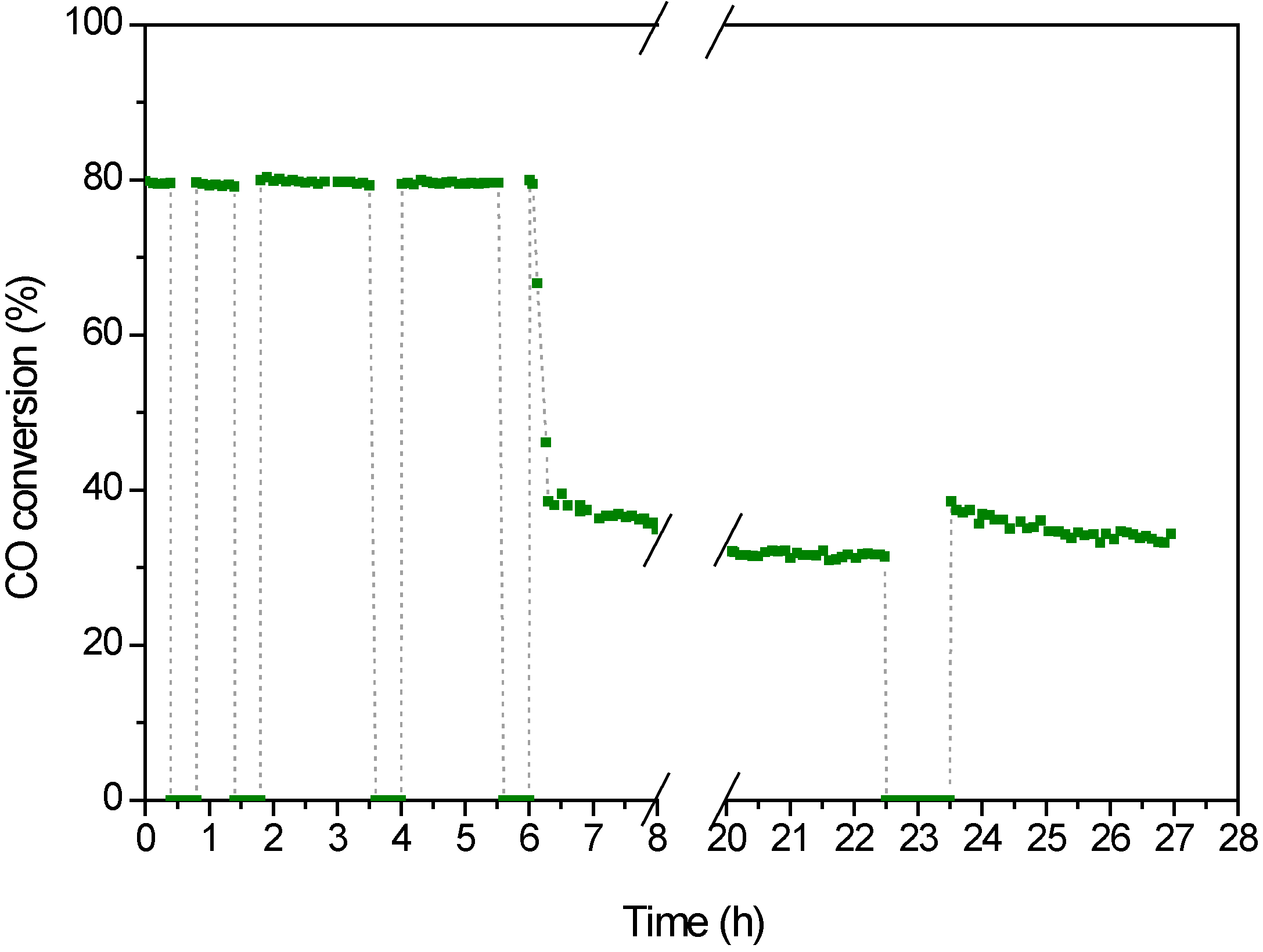

3. Experimental Section
3.1 Catalysts Preparation
3.2 Textural Properties
3.3 Elemental Analysis
3.4 OSCC and OSC Experiments
3.5 TEM
3.6 WGS Catalytic Tests
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ladebeck, J.R.; Wagner, J.P. Catalyst development for water-gas shift. In Handbook of Fuel Cells-Fundamentals, Technology and Applications; Vielstich, W., Lamm, A., Gasteiger, H.A., Eds.; Wiley, New York, NY, USA, 2003; Volume 3, Chapter 16, Part 2; pp. 190–201. [Google Scholar]
- Ratnasamy, C.; Wagner, J.P. Water-gas shift catalysis. Catal. Rev. Sci. Eng. 2009, 51, 325–440. [Google Scholar] [CrossRef]
- Deng, W.; De Jesus, J.; Saltsburg, H.; Flytzani Stephanopoulos, M. Low-content gold-ceria catalysts for the water-gas shift and preferential CO oxidation reactions. Appl. Catal. A 2005, 291, 126–135. [Google Scholar] [CrossRef]
- Summers, J.C.; Ausen, S.A. Interaction of cerium oxide with noble metals. J. Catal. 1979, 58, 131–143. [Google Scholar] [CrossRef]
- Andreeva, D.; Idakiev, V.; Tabakova, T.; Ilieva, L.; Falaras, P.; Bourlinos, A.; Travlos, A. Low-temperature water-gas shift reaction over Au/CeO2 catalysts. Catal. Today 2002, 72, 51–57. [Google Scholar] [CrossRef]
- Colussi, S.; Katta, L.; Amoroso, F.; Farrauto, R.J.; Trovarelli, A. Ceria-based palladium zinc catalysts as promising materials for water gas shift reaction. Catal. Commun. 2014, 47, 63–66. [Google Scholar] [CrossRef]
- Reina, T.R.; Ivanova, S.; Centeno, M.A.; Odriozola, J.A. Low-temperature CO oxidation on multicomponent gold based catalysts. Front. Chem. 2013, 12, 1–9. [Google Scholar]
- Serrano-Ruiz, J.C.; Sepúlveda-Escribano, A.; Rodríguez-Reinoso, F.; Duprez, D. Pt-Sn catalysts supported on highly dispersed ceria on carbon. J. Mol. Catal. A 2007, 268, 227–234. [Google Scholar] [CrossRef]
- Barrio, L.; Kubacka, A.; Zhou, G.; Estrella, M.; Martínez-Arias, A.; Hanson, J.C. Unusual physical and chemical properties of Ni in Ce1−xNixO2−y oxides: structural characterization and catalytic activity for the water-gas shift reaction. J. Phys. Chem. C 2010, 114, 12689–12697. [Google Scholar]
- Jacobs, G.; Chenu, E.; Patterson, P.M.; Williams, L.; Sparks, D.; Thomas, G.; Davis, B.H. Water-gas shift: comparative screening of metal promoters for metal/ceria systems and role of the metal. Appl. Catal. A 2004, 258, 203–214. [Google Scholar] [CrossRef]
- Li, Y.; Fu, Q.; Flytzani-Stephanopoulos, M. Low-temperature water-gas shift reaction over Cu- and Ni-loaded cerium oxide catalysts. Appl. Catal. B 2000, 27, 179–191. [Google Scholar] [CrossRef]
- Ilinich, O.; Ruettinger, W.; Liu, X.; Farrauto, R. Cu-Al2O3-CuAl2O4 water-gas shift catalyst for hydrogen production in fuel cell applications: Mechanism of deactivation under start-stop operating conditions. J. Catal. 2007, 247, 112–118. [Google Scholar] [CrossRef]
- Pastor-Pérez, L.; Buitrago-Sierra, R.; Sepúlveda-Escribano, A. CeO2-promoted Ni/activated carbon catalysts for the water-gas shift (WGS) reaction. Int. J. Hydrogen Energy 2014, 39, 17589–17599. [Google Scholar] [CrossRef]
- Kacimi, S.; Barbier, J.; Taha, R.; Duprez, D. Oxygen storage capacity of promoted Rh/CeC2 catalysts. Exceptional behaviour of RhCu/CeO2. Catal. Lett. 1993, 22, 343–350. [Google Scholar] [CrossRef]
- Reina, T.R.; Ivanova, S.; Delgado, J.J.; Ivanov, I.; Tabakova, T.; Idakiev, V.; Centeno, M.A.; Odriozola, J.A. Viabilty of Au/CeO2-ZnO/Al2O3 catalysts for pure hydrogen production by the Water-Gas Shift reaction. ChemCatChem 2014, 6, 1401–1409. [Google Scholar]
- Senanayake, S.D.; Rodriguez, J.A.; Stacchiola, D. Electronic metalsupport interactions and the production of hydrogen through the water-gas shift reaction and ethanol steam reforming: Fundamental studies with well-defined model catalysts. J. Phys. Chem. C 2012, 116, 9544–9549. [Google Scholar] [CrossRef]
- Lin, J.H.; Guliants, V.V. Hydrogen production through water-gas shift reaction over supported Cu, Ni, and Cu-Ni nanoparticles catalysts prepared from metal colloids. ChemCatChem 2012, 4, 1611–1121. [Google Scholar] [CrossRef]
- Lin, J.H.; Biswas, P.; Guliants, V.V.; Misture, S. Hydrogen production by water-gas shift reaction over bimetallic Cu-Ni catalysts supported on La-doped mesoporous ceria. Appl. Catal. A 2010, 387, 87–94. [Google Scholar] [CrossRef]
- Burch, R. Gold catalysts for pure hydrogen production in the water-gas shift reaction: Activity, structure and reaction mechanism. Phys. Chem. 2006, 47, 5483–5500. [Google Scholar]
- Wang, T.; Porosoff, M.D.; Chen, J.G. Effects of oxide supports on the water-gas shift reaction over Pt-Ni bimetallic catalysts: Activity and methanation inhibition. Catal. Today 2014, 15, 61–69. [Google Scholar] [CrossRef]
- Liu, X.; Ruettinger, W.; Xu, X.; Farrauto, R. Deactivation of Pt/CeO2 water-gas shift catalysts due to shutdown/startup modes for fuel cell applications. Appl. Catal. B 2005, 56, 69–75. [Google Scholar] [CrossRef]
- Senanayake, S.D.; Evans, J.; Agnoli, S.; Barrio, L.; Chen, T.L.; Hrbek, J.; Rodriguez, J.A. Water-gas shift and CO methanation reactions over NiCeO2(111) catalysts. Top. Catal. 2011, 54, 34–41. [Google Scholar] [CrossRef]
- Bunluesin, T.; Gorte, R.J.; Graham, G.W. Studies of the water-gas shift reaction on ceria-supported Pt, Pd, and Rh: implications for oxygen-storage properties. Appl. Catal. B 1998, 15, 107–114. [Google Scholar] [CrossRef]
- Gonzalez-De la Cruz, V.M.; Holgado, J.P.; Pereñíguez, R.; Caballero, A. Morphology changes induced by strong metal-support interaction on a Ni-ceria catalytic system. J. Catal. 2008, 257, 307–314. [Google Scholar] [CrossRef]
- Serrano-Ruiz, J.C.; Ramos-Fernandez, E.V.; Silvestre-Albero, J.; Sepúlveda-Escribano, A.; Rodríguez-Reinoso, F. Preparation and characterization of CeO2 highly dispersed on activated carbon. Mater. Res. Bull. 2008, 43, 1850–1857. [Google Scholar] [CrossRef]
- Royer, S.; Duprez, D. Catalytic oxidation of carbon monoxide over transition metal oxides. ChemCatChem 2011, 3, 24–65. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pastor-Pérez, L.; Reina, T.R.; Ivanova, S.; Centeno, M.Á.; Odriozola, J.A.; Sepúlveda-Escribano, A. Ni-CeO2/C Catalysts with Enhanced OSC for the WGS Reaction. Catalysts 2015, 5, 298-309. https://doi.org/10.3390/catal5010298
Pastor-Pérez L, Reina TR, Ivanova S, Centeno MÁ, Odriozola JA, Sepúlveda-Escribano A. Ni-CeO2/C Catalysts with Enhanced OSC for the WGS Reaction. Catalysts. 2015; 5(1):298-309. https://doi.org/10.3390/catal5010298
Chicago/Turabian StylePastor-Pérez, Laura, Tomás Ramírez Reina, Svetlana Ivanova, Miguel Ángel Centeno, José Antonio Odriozola, and Antonio Sepúlveda-Escribano. 2015. "Ni-CeO2/C Catalysts with Enhanced OSC for the WGS Reaction" Catalysts 5, no. 1: 298-309. https://doi.org/10.3390/catal5010298
APA StylePastor-Pérez, L., Reina, T. R., Ivanova, S., Centeno, M. Á., Odriozola, J. A., & Sepúlveda-Escribano, A. (2015). Ni-CeO2/C Catalysts with Enhanced OSC for the WGS Reaction. Catalysts, 5(1), 298-309. https://doi.org/10.3390/catal5010298










