Comparing Sustainable and Conventional Methods for Synthesizing Copper Oxide Nanoparticles: Implications for Hydrogen Evolution
Abstract
1. Introduction
2. Analysis and Interpretation of Results
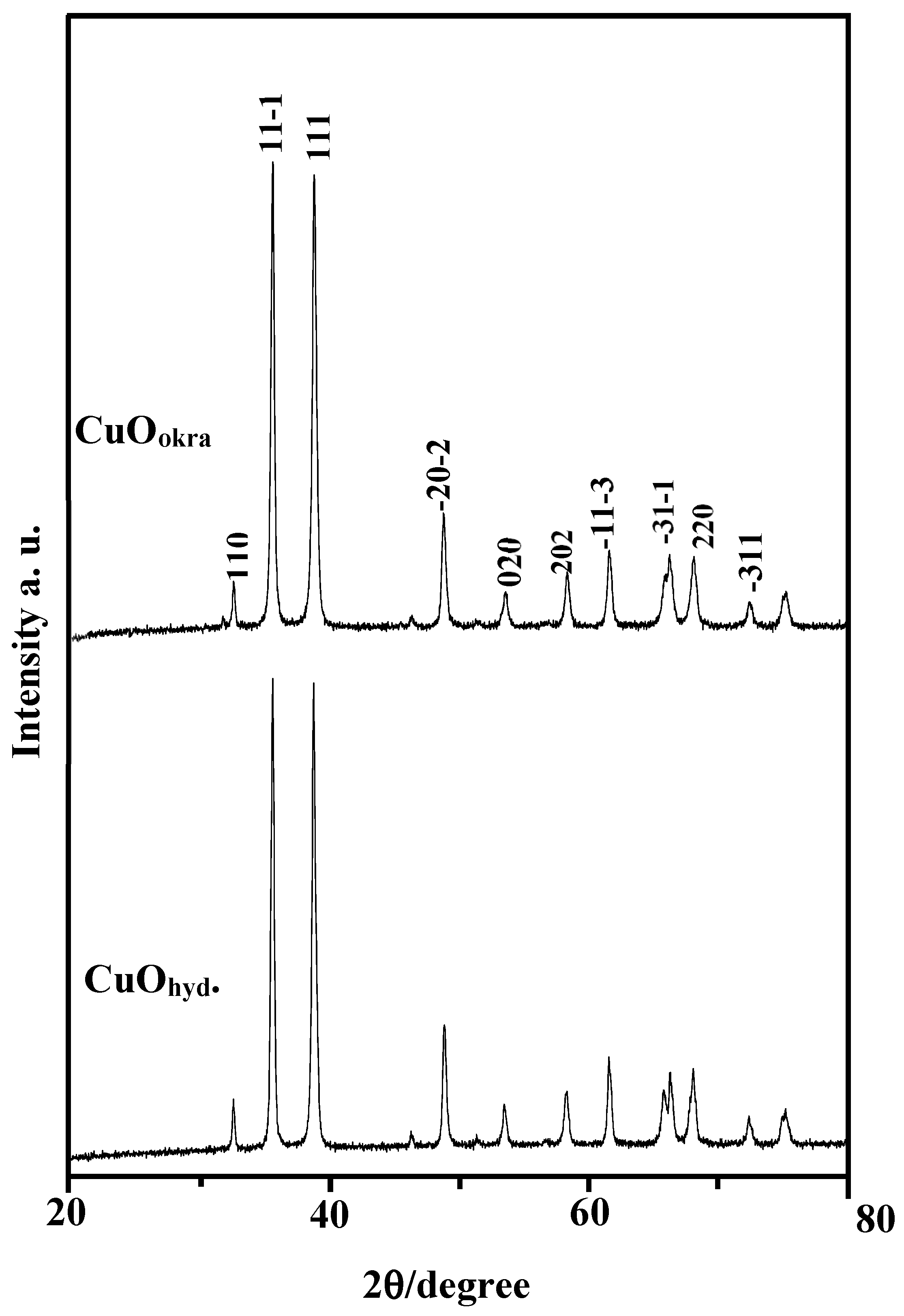
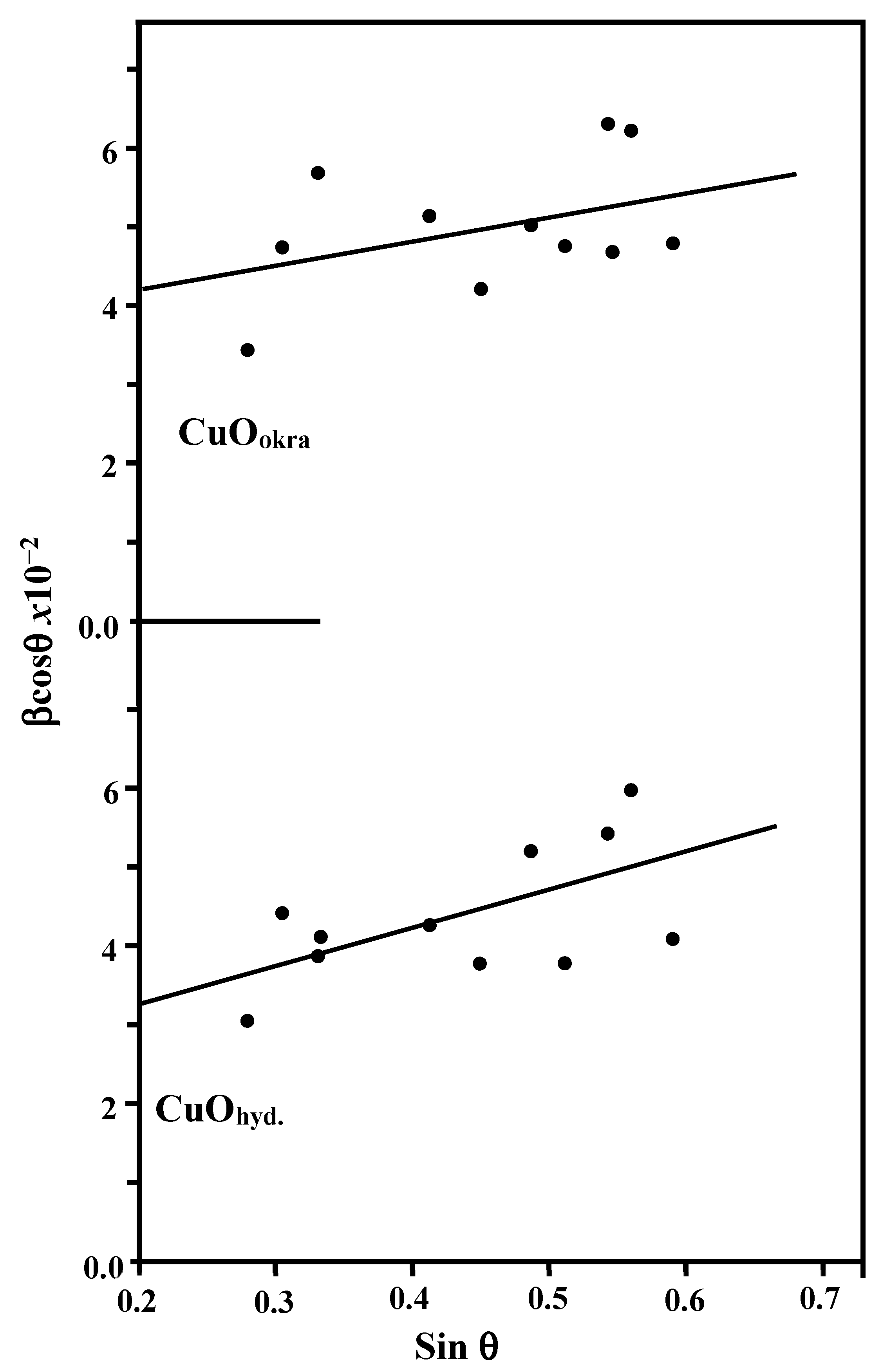
2.1. XRD
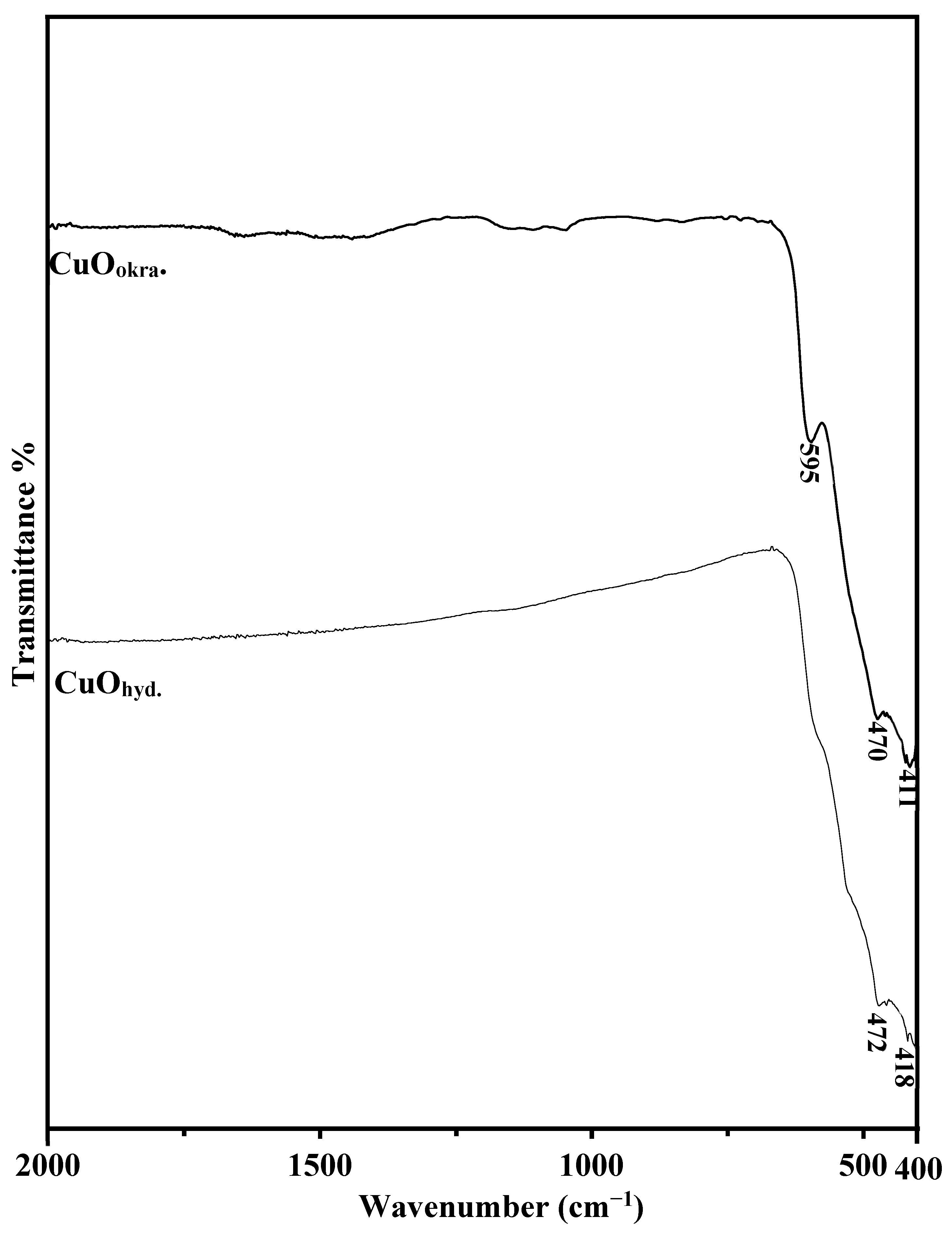
2.2. ATR-FTIR Study
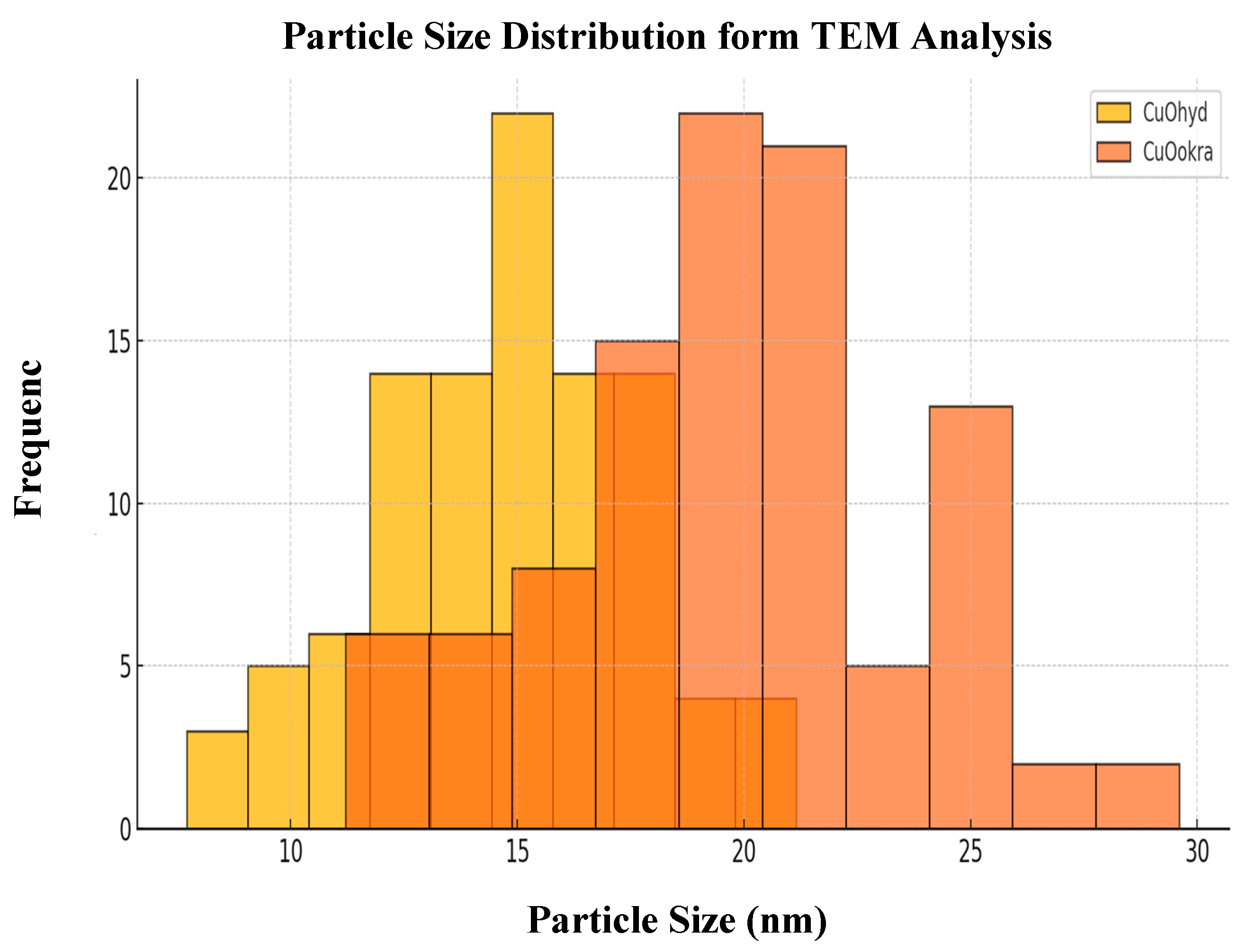
2.3. Morphological Analysis (FESEM and HRTEM)
2.4. Surface Texturing
2.5. Hydrogen Evolution
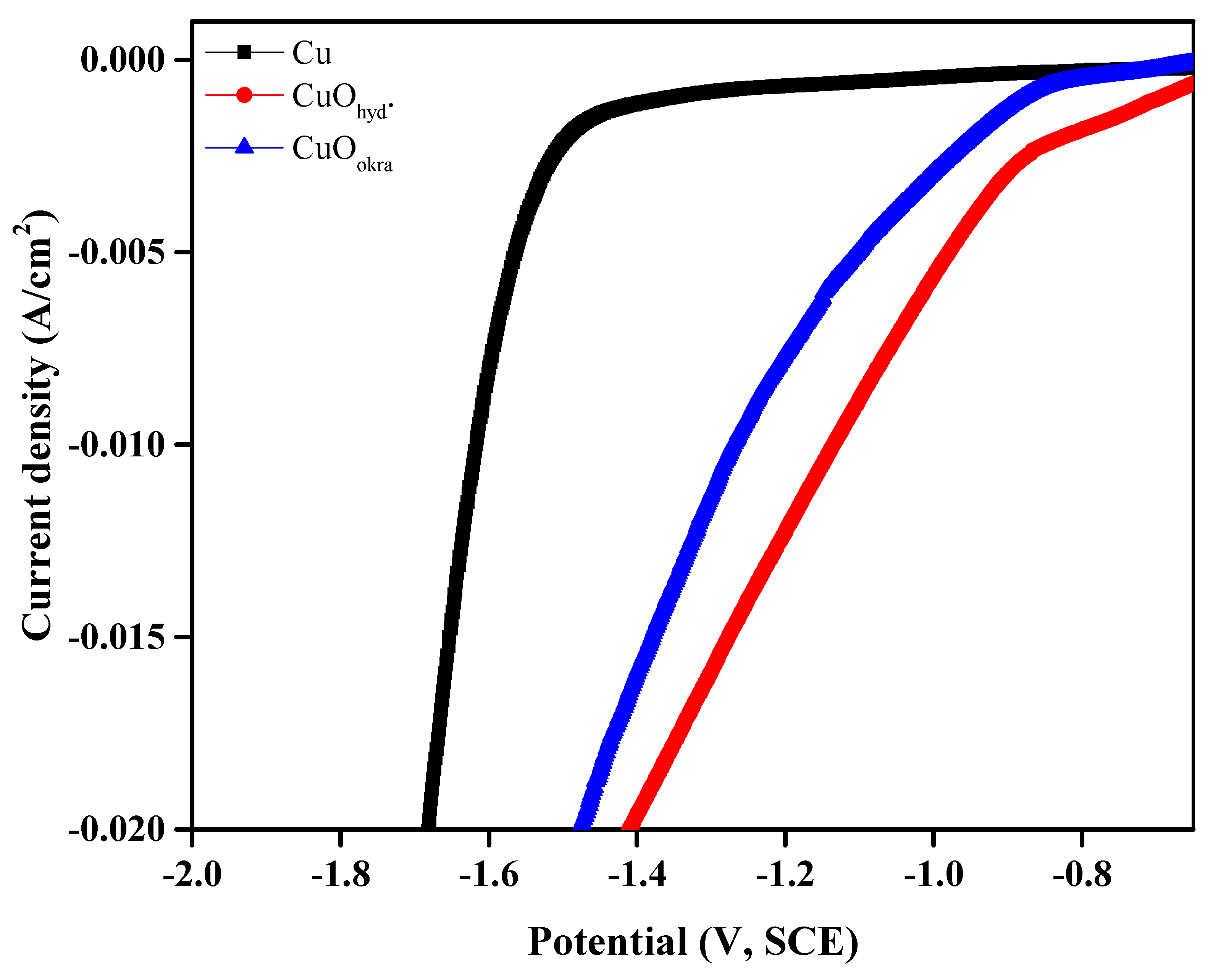
2.5.1. Cathodic Polarization Analysis
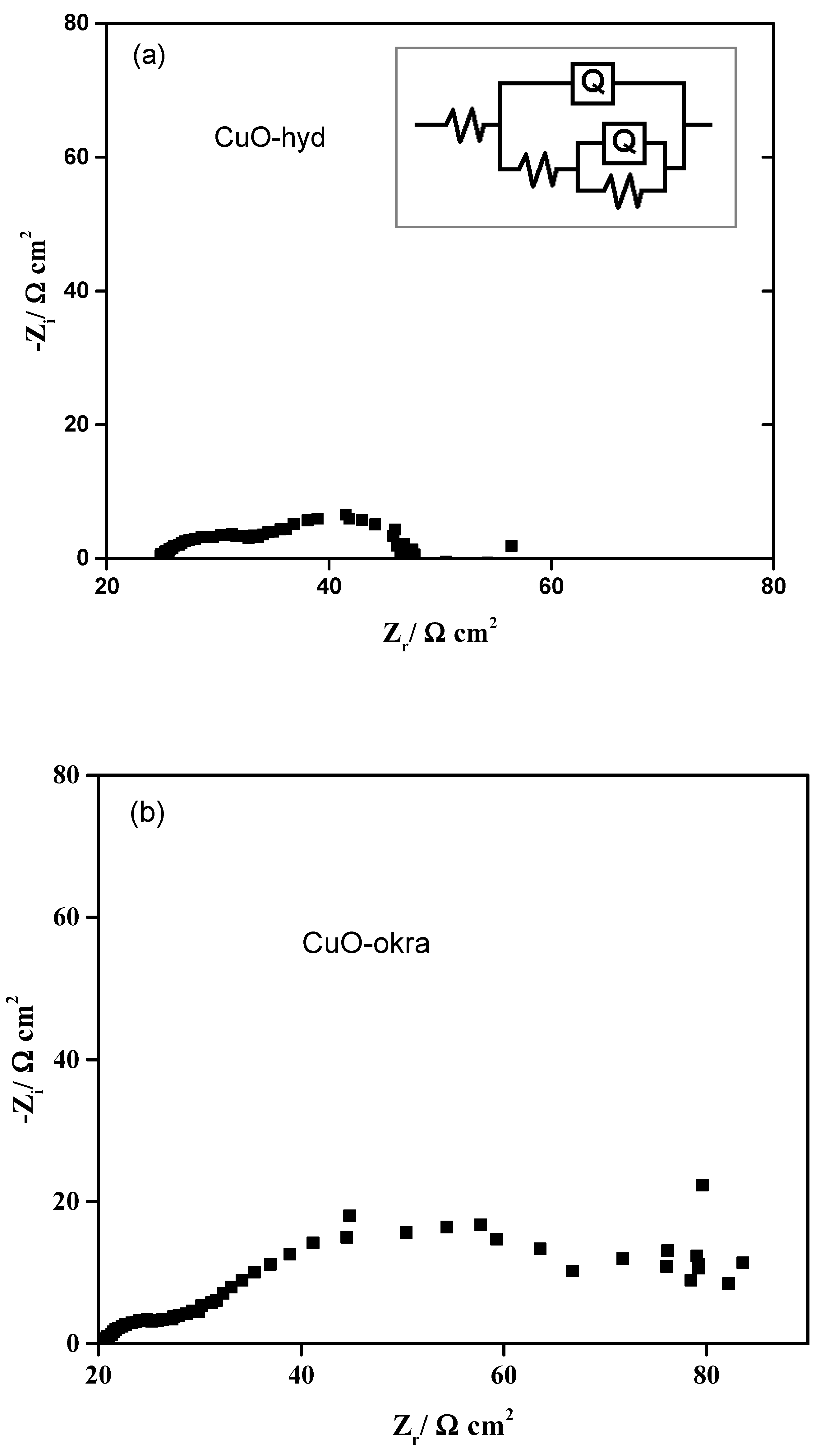
2.5.2. Electrochemical Impedance Spectroscopy, EIS
3. Experimental
3.1. Materials
3.1.1. Extraction Process of Okra Fruit
3.1.2. Green Synthesis of CuO Nanoparticles
3.1.3. Fabrication of CuO NPs Using Hydrazine Hydrate
3.2. Working Electrode Preparation and Electrochemical Measurement
3.3. Structural and Morphological Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ye, L.; Wen, Z. Self-supported three-dimensional Cu/Cu2O–CuO/rGO nanowire array electrodes for an efficient hydrogen evolution reaction. Chem. Commun. 2018, 54, 6388–6391. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Li, T.-T.; Qian, J.; Hu, Y.; Guo, F.; Zheng, Y.-Q. Self-supported hierarchical CuOx@Co3O4 heterostructures as efficient bifunctional electrocatalysts for water splitting. J. Mater. Chem. A 2018, 6, 14431–14439. [Google Scholar] [CrossRef]
- Ifkovits, Z.P.; Evans, J.M.; Meier, M.C.; Papadantonakis, K.M.; Lewis, N.S. Decoupled electrochemical water-splitting systems: A review and perspective. Energy Environ. Sci. 2021, 14, 4740–4759. [Google Scholar] [CrossRef]
- Sirisomboonchai, S.; Li, X.; Kitiphatpiboon, N.; Channoo, R.; Li, S.; Ma, Y.; Kongparakul, S.; Samart, C.; Abudula, A.; Guan, G. Fabrication of CuOx nanowires@NiMnOx nanosheets core@shell-type electrocatalysts: Crucial roles of defect modification and valence states for overall water electrolysis. J. Mater. Chem. A 2020, 8, 16463–16476. [Google Scholar] [CrossRef]
- Li, L.; Wang, P.; Shao, Q.; Huang, X. Metallic nanostructures with low dimensionality for electrochemical water splitting. Chem. Soc. Rev. 2020, 49, 3072–3106. [Google Scholar] [CrossRef]
- Yin, K.; Chao, Y.; Lv, F.; Tao, L.; Zhang, W.; Lu, S.; Li, M.; Zhang, Q.; Gu, L.; Li, H.; et al. One Nanometer PtIr Nanowires as High-Efficiency Bifunctional Catalysts for Electrosynthesis of Ethanol into High Value-Added Multicarbon Compound Coupled with Hydrogen Production. J. Am. Chem. Soc. 2021, 143, 10822–10827. [Google Scholar] [CrossRef]
- Yuan, C.-Z.; Hui, K.S.; Yin, H.; Zhu, S.; Zhang, J.; Wu, X.-L.; Hong, X.; Zhou, W.; Fan, X.; Bin, F.; et al. Regulating Intrinsic Electronic Structures of Transition-Metal-Based Catalysts and the Potential Applications for Electrocatalytic Water Splitting. ACS Mater. Lett. 2021, 3, 752–780. [Google Scholar] [CrossRef]
- Li, W.; Wang, C.; Lu, X. Integrated transition metal and compounds with carbon nanomaterials for electrochemical water splitting. J. Mater. Chem. A 2021, 9, 3786–3827. [Google Scholar] [CrossRef]
- Gao, M.; Sheng, W.; Zhuang, Z.; Fang, Q.; Gu, S.; Jiang, J.; Yan, Y. Efficient Water Oxidation Using Nanostructured α-Nickel-Hydroxide as an Electrocatalyst. J. Am. Chem. Soc. 2014, 136, 7077–7084. [Google Scholar] [CrossRef]
- Hutchings, G.S.; Zhang, Y.; Li, J.; Yonemoto, B.T.; Zhou, X.; Zhu, K.; Jiao, F. In Situ Formation of Cobalt Oxide Nanocubanes as Efficient Oxygen Evolution Catalysts. J. Am. Chem. Soc. 2015, 137, 4223–4229. [Google Scholar] [CrossRef]
- Li, Y.; Wang, J.; Tian, X.; Ma, L.; Dai, C.; Yang, C.; Zhou, Z. Carbon doped molybdenum disulfide nanosheets stabilized on graphene for the hydrogen evolution reaction with high electrocatalytic ability. Nanoscale 2016, 8, 1676–1683. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Zhou, Y.; Chen, Y.; Li, P.; Liu, Q.; Wang, J. Ultrafine Molybdenum Carbide Nanoparticles Composited with Carbon as a Highly Active Hydrogen-Evolution Electrocatalyst. Angew. Chem. Int. Ed. 2015, 54, 14723–14727. [Google Scholar] [CrossRef] [PubMed]
- Javid-Naderi, M.J.; Sabouri, Z.; Jalili, A.; Zarrinfar, H.; Samarghandian, S.; Darroudi, M. Green synthesis of copper oxide nanoparticles using okra (Abelmoschus esculentus) fruit extract and assessment of their cytotoxicity and photocatalytic applications. Environ. Technol. Innov. 2023, 32, 103300. [Google Scholar] [CrossRef]
- Negrescu, A.M.; Killian, M.S.; Raghu, S.N.V.; Schmuki, P.; Mazare, A.; Cimpean, A. Metal Oxide Nanoparticles: Review of Synthesis, Characterization and Biological Effects. J. Funct. Biomater. 2022, 13, 274. [Google Scholar] [CrossRef]
- Okoye, P.; Azi, S.; Qahtan, T.; Owolabi, T.; Saleh, T. Synthesis, properties, and applications of doped and undoped CuO and Cu2O nanomaterials. Mater. Today Chem. 2023, 30, 101513. [Google Scholar] [CrossRef]
- Dantas, A.P.; Raimundo, R.A.; Neto, P.F.; Lopes, C.M.; Santos, J.R.; Loureiro, F.J.; Pereira, T.O.; Morales, M.A.; Medeiros, E.S.; Macedo, D.A. Copper oxide nanofibers obtained by solution blow spinning as catalysts for oxygen evolution reaction. Ceram. Int. 2024, 50, 13034–13045. [Google Scholar] [CrossRef]
- Seehra, M.; Punnoose, A. Particle size dependence of exchange-bias and coercivity in CuO nanoparticles. Solid State Commun. 2003, 128, 299–302. [Google Scholar] [CrossRef]
- Vineesh, T.V.; Yarmiayev, V.; Zitoun, D. Tailoring the electrochemical hydrogen evolution activity of Cu3P through oxophilic surface modification. Electrochem. Commun. 2020, 113, 106691. [Google Scholar] [CrossRef]
- Rheinländer, P.J.; Herranz, J.; Durst, J.; Gasteiger, H.A. Kinetics of the Hydrogen Oxidation/Evolution Reaction on Polycrystalline Platinum in Alkaline Electrolyte Reaction Order with Respect to Hydrogen Pressure. J. Electrochem. Soc. 2014, 161, F1448–F1457. [Google Scholar] [CrossRef]
- Khandaker, J.I. Hydrothermal synthesis of CuO nanoparticles and a study on property variation with synthesis temperature. J. Appl. Fundam. Sci. 2020, 6, 52. [Google Scholar]
- Shui, A.; Zhu, W.; Xu, L.; Qin, D.; Wang, Y. Green sonochemical synthesis of cupric and cuprous oxides nanoparticles and their optical properties. Ceram. Int. 2013, 39, 8715–8722. [Google Scholar] [CrossRef]
- Rangel, W.M.; Santa, R.A.A.B.; Riella, H.G. A facile method for synthesis of nanostructured copper (II) oxide by coprecipitation. J. Mater. Res. Technol. 2020, 9, 994–1004. [Google Scholar] [CrossRef]
- Patel, M.; Mishra, S.; Verma, R.; Shikha, D. Synthesis of ZnO and CuO nanoparticles via Sol gel method and its characterization by using various technique. Discov. Mater. 2022, 2, 1. [Google Scholar] [CrossRef]
- Thakur, N.; Anu; Kumar, K.; Kumar, A. Effect of (Ag, Zn) co-doping on structural, optical and bactericidal properties of CuO nanoparticles synthesized by a microwave-assisted method. Dalton Trans. 2021, 50, 6188–6203. [Google Scholar] [CrossRef]
- Murugan, B.; Rahman, M.Z.; Fatimah, I.; Anita Lett, J.; Annaraj, J.; Kaus, N.H.M.; Al-Anber, M.A.; Sagadevan, S. Green synthesis of CuO nanoparticles for biological applications. Inorg. Chem. Commun. 2023, 155, 111088. [Google Scholar] [CrossRef]
- Moroda, M.D.; Deressa, T.L.; Tiwikrama, A.H.; Chala, T.F. Green synthesis of copper oxide nanoparticles using Rosmarinus officinalis leaf extract and evaluation of its antimicrobial activity. Next Mater. 2024, 7, 100337. [Google Scholar] [CrossRef]
- Pawar, S.M.; Patil, S.S.; Sonawane, K.D.; More, V.B.; Patil, P.S. Hydrothermally synthesized copper oxide nanoparticles: Rietveld analysis and antimicrobial studies. Surf. Interfaces 2024, 51, 104598. [Google Scholar] [CrossRef]
- Nzilu, D.M.; Madivoli, E.S.; Makhanu, D.S.; Wanakai, S.I.; Kiprono, G.K.; Kareru, P.G. Green synthesis of copper oxide nanoparticles and its efficiency in degradation of rifampicin antibiotic. Sci. Rep. 2023, 13, 14030. [Google Scholar] [CrossRef]
- Rehman, S.; Shad, N.A.; Sajid, M.M.; Ali, K.; Javed, Y.; Jamil, Y.; Sajjad, M.; Nawaz, A.; Sharma, S.K. Tuning Structural and Optical Properties of Copper Oxide Nanomaterials by Thermal Heating and Its Effect on Photocatalytic Degradation of Congo Red Dye. J. Chem. Chem. Eng. 2022, 41, 1549–1560. [Google Scholar]
- Williamson, G.K.; Smallman, R.E., III. Dislocation densities in some annealed and cold-worked metals from measurements on the X-ray debye-scherrer spectrum. Philos. Mag. 1956, 1, 34–46. [Google Scholar] [CrossRef]
- Berra, D.; Laouini, S.E.; Benhaoua, B.; Ouahrani, M.R.; Berrani, D.; Rahal, A. Green synthesis of copper oxide nanoparticles by Pheonix dactylifera L leaves extract. J. Nanomater. Biostructures 2018, 13, 1231–1238. [Google Scholar]
- Bin Mobarak, M.; Hossain, S.; Chowdhury, F.; Ahmed, S. Synthesis and characterization of CuO nanoparticles utilizing waste fish scale and exploitation of XRD peak profile analysis for approximating the structural parameters. Arab. J. Chem. 2022, 15, 104117. [Google Scholar] [CrossRef]
- Huang, G.; Zhu, Y. Synthesis and photoactivity enhancement of ZnWO4 photocatalysts doped with chlorine. CrystEngComm 2012, 14, 8076–8082. [Google Scholar] [CrossRef]
- Jaihindh, D.P.; Anand, P.; Chen, R.-S.; Yu, W.-Y.; Wong, M.-S.; Fu, Y.-P. Cl-doped CuO for electrochemical hydrogen evolution reaction and tetracycline photocatalytic degradation. J. Environ. Chem. Eng. 2023, 11, 109852. [Google Scholar] [CrossRef]
- Jiang, N.; Tang, Q.; Sheng, M.; You, B.; Jiang, D.; Sun, Y. Nickel sulfides for electrocatalytic hydrogen evolution under alkaline conditions: A case study of crystalline NiS, NiS2, and Ni3S2 nanoparticles. Catal. Sci. Technol. 2016, 6, 1077–1084. [Google Scholar] [CrossRef]
- Hanan, A.; Shu, D.; Aftab, U.; Cao, D.; Laghari, A.J.; Solangi, M.Y.; Abro, M.I.; Nafady, A.; Vigolo, B.; Tahira, A.; et al. Co2FeO4@rGO composite: Towards trifunctional water splitting in alkaline media. Int. J. Hydrogen Energy 2022, 47, 33919–33937. [Google Scholar] [CrossRef]
- Yang, S.; Wen, H.; Liu, Z.; Zhai, J.; Yu, Y.; Li, K.; Huang, Z.; Sun, D. Engineering double sulfur-vacancy in CoS1.097@MoS2 Core-shell heterojunctions for hydrogen evolution in a wide pH range. Inorg. Chem. 2023, 62, 17401–17408. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Vrubel, H.; Bensimon, M.; Hu, X. Easily prepared dinickel phosphide (Ni2P) nanoparticles as an efficient and robust electrocatalyst for hydrogen evolution. Phys. Chem. Chem. Phys. 2014, 16, 5917–5921. [Google Scholar] [CrossRef] [PubMed]
- Man, H.-W.; Tsang, C.-S.; Li, M.-M.-J.; Mo, J.; Huang, B.; Lee, L.Y.S.; Leung, Y.-C.; Wong, K.-Y.; Tsang, S.C.E. Transition metal-doped nickel phosphide nanoparticles as electro-and photocatalysts for hydrogen generation reactions. Appl. Catal. B 2019, 242, 186–193. [Google Scholar] [CrossRef]
- Meshkian, R.; Dahlqvist, M.; Lu, J.; Wickman, B.; Halim, J.; Thornberg, J.; Tao, Q.; Li, S.; Intikhab, S.; Snyder, J. W-Based Atomic Laminates and Their 2D Derivative W1.33C MXene with Vacancy Ordering. Adv. Mater. 2018, 30, 1706409. [Google Scholar] [CrossRef]
- Hanan, A.; Ahmed, M.; Lakhan, M.N.; Shar, A.H.; Cao, D.; Asif, A.; Ali, A.; Gul, M. Novel rGO@Fe3O4 nanostructures: An active electrocatalyst for hydrogen evolution reaction in alkaline media. J. Indian Chem. Soc. 2022, 99, 100442. [Google Scholar] [CrossRef]
- Belhadj, H.; Messaoudi, Y.; Khelladi, M.R.; Azizi, A. A facile synthesis of metal ferrites (MFe2O4, M = Co, Ni, Zn, Cu) as effective electrocatalysts toward electrochemical hydrogen evolution reaction. Int. J. Hydrogen Energy 2022, 47, 20129–20137. [Google Scholar] [CrossRef]
- Birry, L.; Lasia, A. Studies of the Hydrogen Evolution Reaction on Raney Nickel—Molybdenum Electrodes. J. Appl. Electrochem. 2004, 34, 735–749. [Google Scholar] [CrossRef]
- Badawy, W.; Nady, H.; Negem, M. Cathodic hydrogen evolution in acidic solutions using electrodeposited nano-crystalline Ni–Co cathodes. Int. J. Hydrogen Energy 2014, 39, 10824–10832. [Google Scholar] [CrossRef]
- Krstajic, N.V.; Jovic, V.D.; Gajic-Krstajic, L.; Jovic, B.M.; Antozzi, A.L.; Martelli, G.N. Electrodeposition of Ni–Mo alloy coatings and their characterization as cathodes for hydrogen evolution in sodium hydroxide solution. Int. J. Hydrogen Energy 2008, 33, 3676–3687. [Google Scholar] [CrossRef]
- Hu, H.; Qiao, M.; Pei, Y.; Fan, K.; Li, H.; Zong, B. Kinetics of hydrogen evolution in alkali leaching of rapidly quenched Ni–Al alloy. Appl. Catal. A 2003, 252, 173–183. [Google Scholar] [CrossRef]
- Los, P.; Lasia, A.; Ménard, H.; Brossard, L. Impedance studies of porous lanthanum-phosphate-bonded nickel electrodes in concentrated sodium hydroxide solution. J. Electroanal. Chem. 1993, 360, 101–118. [Google Scholar] [CrossRef]
- Solmaz, R.; Kardaş, G. Hydrogen evolution and corrosion performance of NiZn coatings. Energy Convers. Manag. 2007, 48, 583–591. [Google Scholar] [CrossRef]
- Shervedani, R.K.; Mardam, A.R. Kinetics of hydrogen evolution reaction on nanocrystalline electrodeposited Ni62Fe35C3 cathode in alkaline solution by electrochemical impedance spectroscopy. Electrochim. Acta 2007, 53, 426–433. [Google Scholar] [CrossRef]



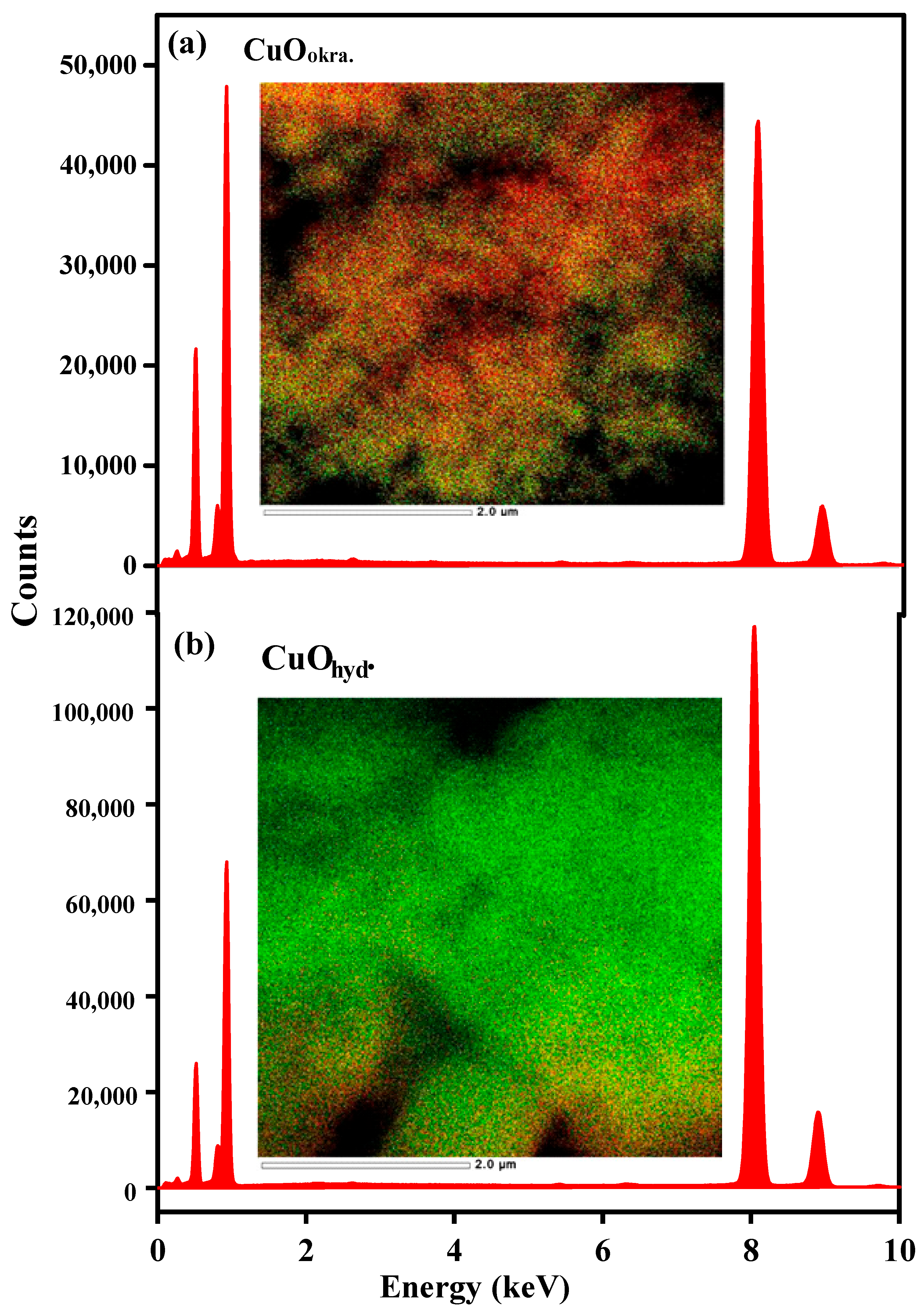
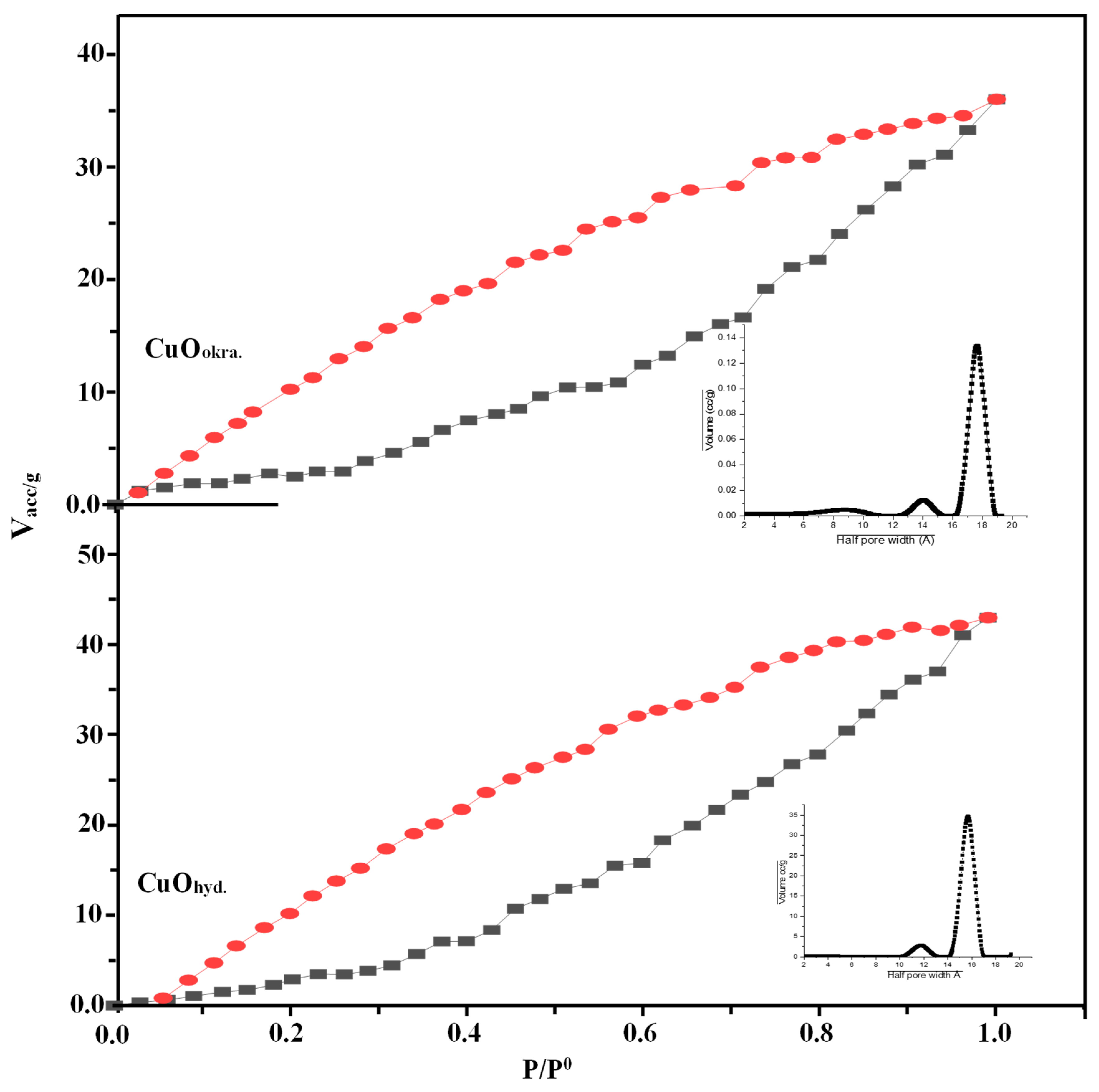
| Samples | XRD Data | Surface Texture | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a (Å) | b (Å) | c (Å) | β (°) | Volume (Å3) | DShr (nm) | DWH (nm) | ɛ ×10−4 | (δ) (g/cm3) ×10−6 | Ref. | SBET (m2g−1) | r— (Å) | Vp Total (cm3 g−1) | |
| CuOhyd | 4.686 | 3.427 | 5.133 | 99.52 | 81.296 | 34.4 | 56.8 | 10.973 | 3.10181 | This work | 46.21 | 17.69 | 0.0801 |
| CuOokra | 4.690 | 3.427 | 5.133 | 99.65 | 81.334 | 34.8 | 39.8 | 8.306 | 6.3031 | 32.18 | 17.83 | 0.0592 | |
| JCPDS cards 01-077-7717 | 4.685 | 3.423 | 5.132 | 99.52 | 81.17 | 6.51 | 32 | ||||||
| CuO NPs (waste fish) | 4.689 | 3.469 | 5.115 | 99.309 | 82.10 | 41.5 | 35.11 | 10.7 | 6.43 | 33 | |||
| CuO NPs (co-precipitation) | 4.700 | 3.408 | 5.105 | 99.52 | 80.644 | 18.81 | 20.92 | 11.4 | 6.55 | 34 | |||
| Materials | η20 (mV, RHE) | i1.3/ mA cm−2 | i1.4/ mA cm−2 | i1.5/ mA cm−2 |
|---|---|---|---|---|
| CuOhyd | 343 | 15.9 | 19.7 | 23.5 |
| CuOokra | 408 | 11.3 | 16 | 21.7 |
| Materials | Overpotential (mV vs. RHE), η10 | Reference |
|---|---|---|
| Ni3S2/NF | 335 | 35 |
| Co2FeO4@rGO | 320 | 36 |
| CoS1.097@MoS2 | 139 | 37 |
| Ni2P | 250 (η20) | 38 |
| NiFeP | 690 | 39 |
| W1.33C | 320 | 40 |
| rGO-Fe3O4 | 300 | 41 |
| CoFe2O4 | 270 | 42 |
| CuOhyd | 343 (η20) | Present work |
| CuOokra | 408 (η20) | Present work |
| Materials | Rs (Ω) | R1 (Ω cm2) | Q1/Yo (Ω−1 cm−2 sn2 | R2 (Ω cm2) | Q2/Yo (Ω−1 cm−2 sn2 |
|---|---|---|---|---|---|
| CuOhyd | 24.91 | 9.446 | 0.0002358 | 164.7 | 0.003125 |
| CuOokra | 19.86 | 23.93 | 0.004329 | 49.76 | 0.00135 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alenezy, E.K.; Ali, I.O.; Hashem, N.; Salama, T.M. Comparing Sustainable and Conventional Methods for Synthesizing Copper Oxide Nanoparticles: Implications for Hydrogen Evolution. Catalysts 2025, 15, 823. https://doi.org/10.3390/catal15090823
Alenezy EK, Ali IO, Hashem N, Salama TM. Comparing Sustainable and Conventional Methods for Synthesizing Copper Oxide Nanoparticles: Implications for Hydrogen Evolution. Catalysts. 2025; 15(9):823. https://doi.org/10.3390/catal15090823
Chicago/Turabian StyleAlenezy, Ebtsam Khalefah, Ibraheem Othman Ali, Nady Hashem, and Tarek Mohamed Salama. 2025. "Comparing Sustainable and Conventional Methods for Synthesizing Copper Oxide Nanoparticles: Implications for Hydrogen Evolution" Catalysts 15, no. 9: 823. https://doi.org/10.3390/catal15090823
APA StyleAlenezy, E. K., Ali, I. O., Hashem, N., & Salama, T. M. (2025). Comparing Sustainable and Conventional Methods for Synthesizing Copper Oxide Nanoparticles: Implications for Hydrogen Evolution. Catalysts, 15(9), 823. https://doi.org/10.3390/catal15090823






