1,10-Phenanthroline-Iron Complex-Derived Fe-N-C Electrocatalysts: Enhanced Oxygen Reduction Activity and Stability Through Synthesis Tuning
Abstract
1. Introduction
2. Results and Discussion
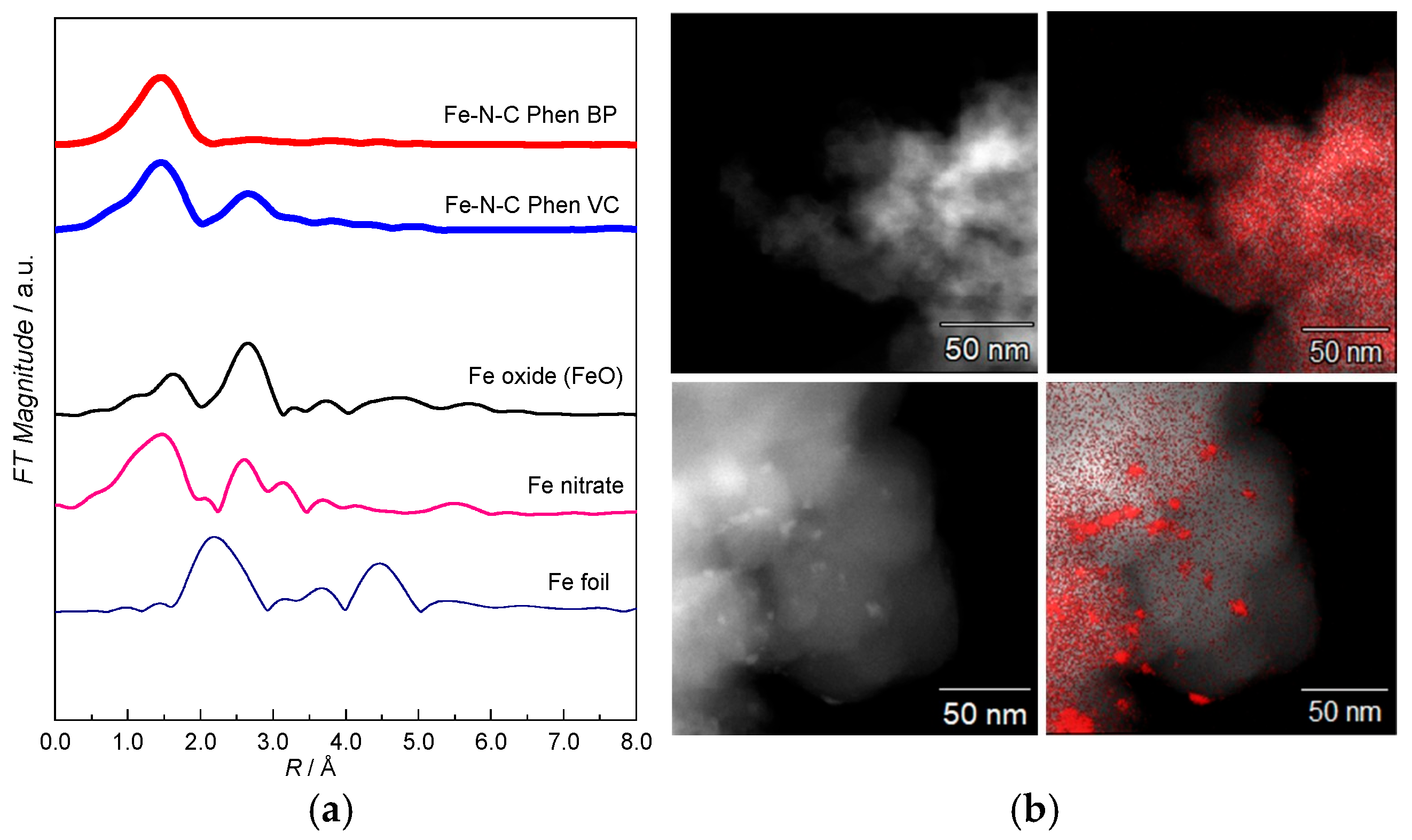
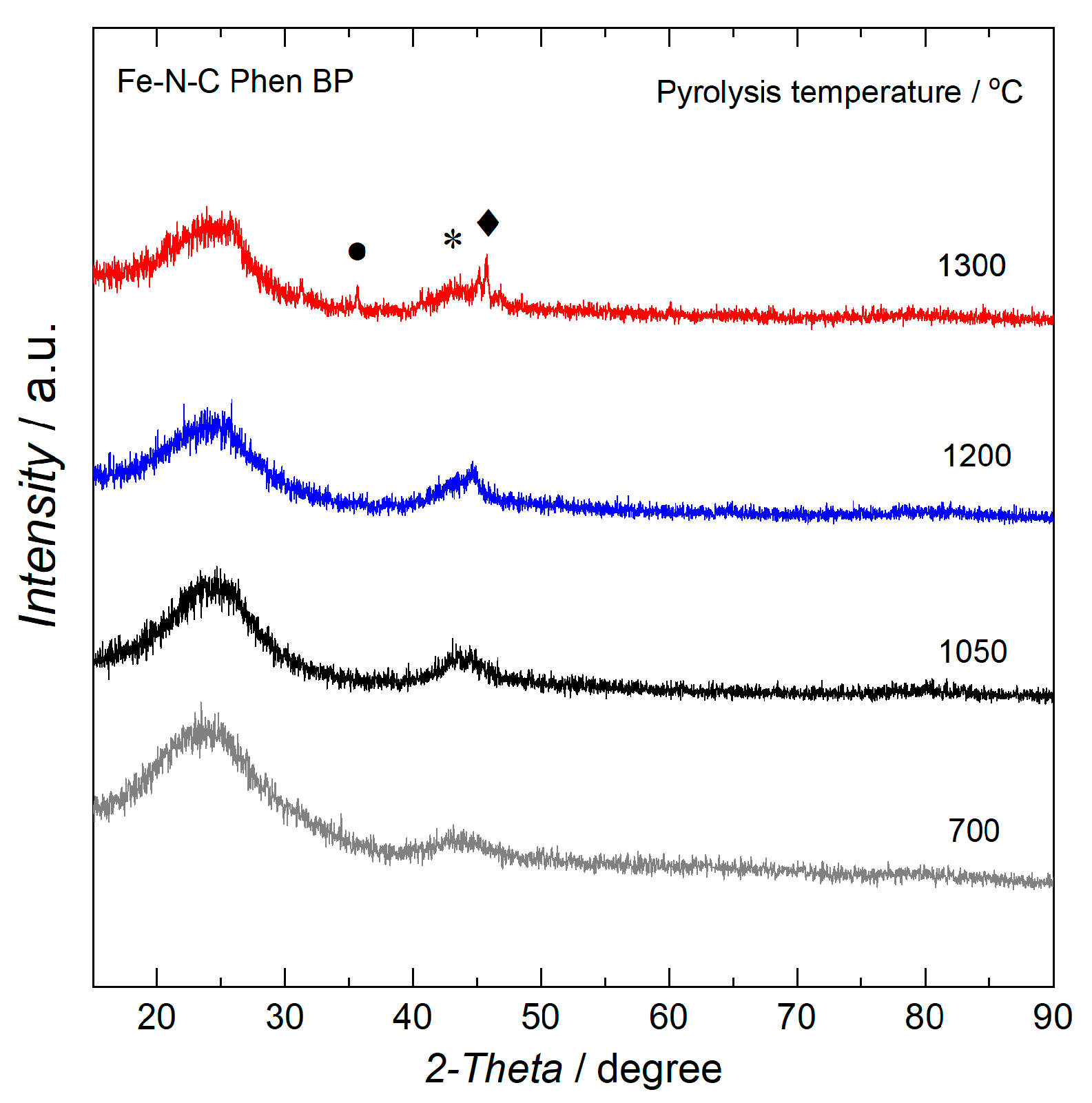
2.1. Characterization
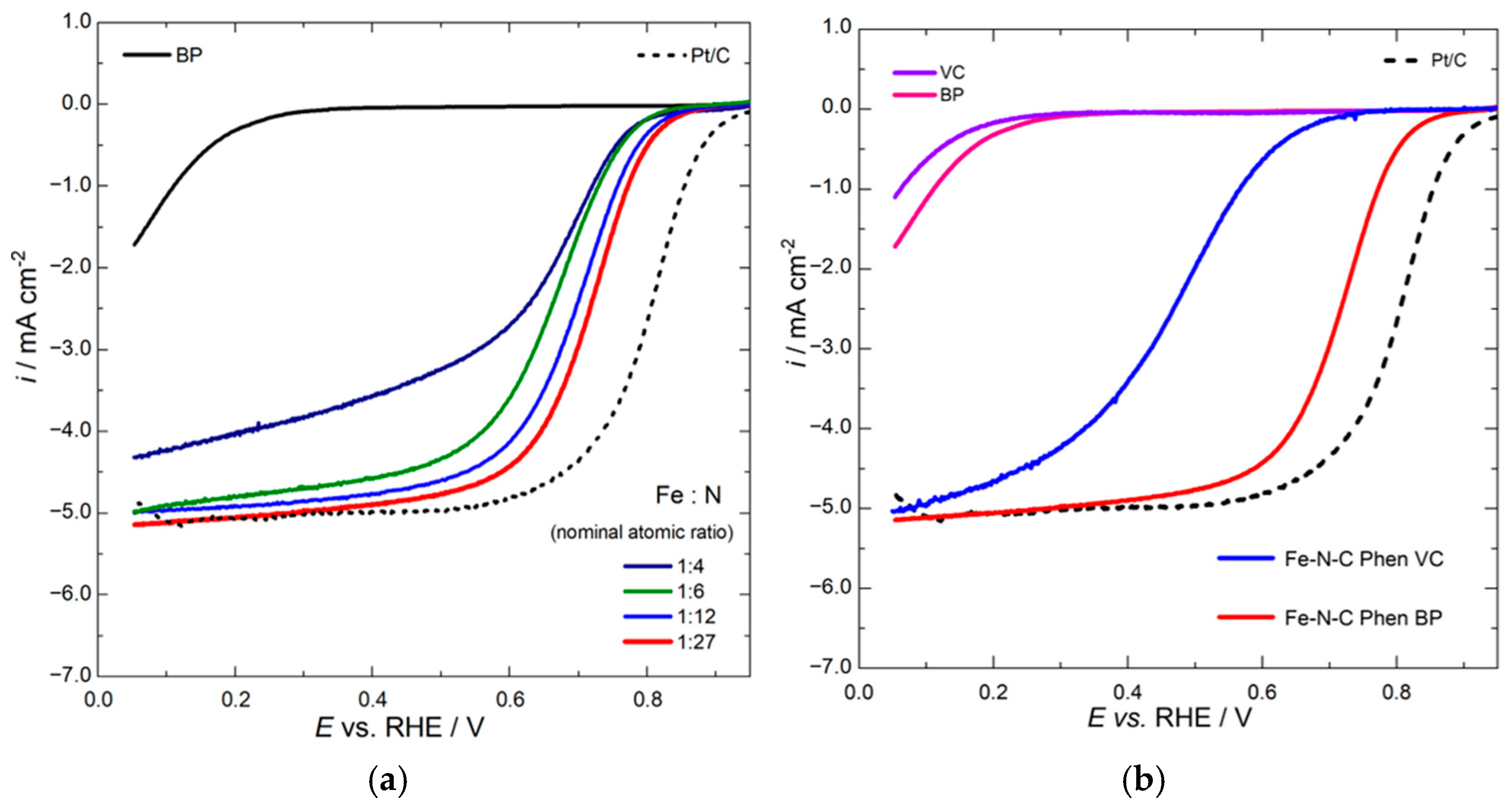
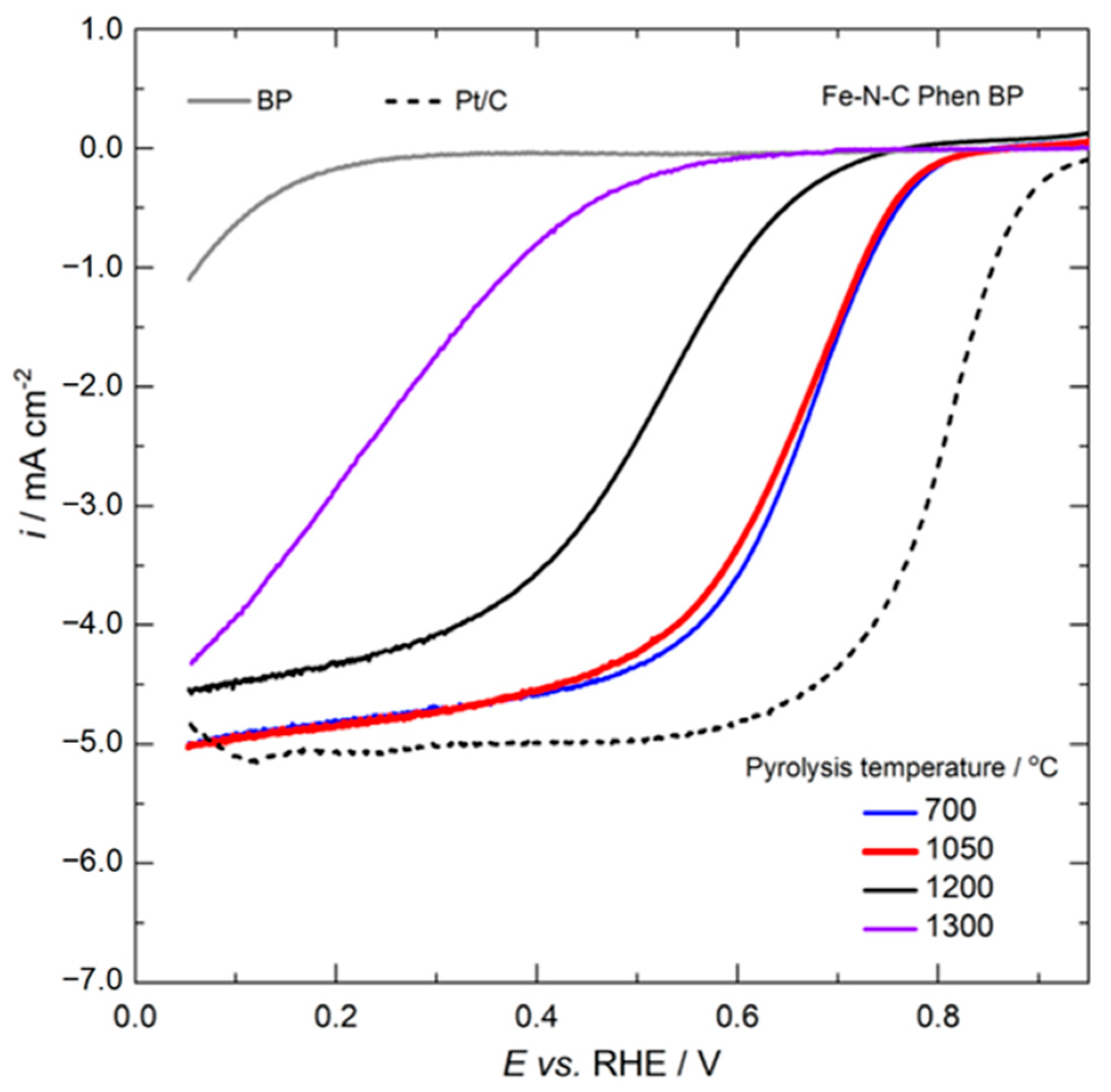
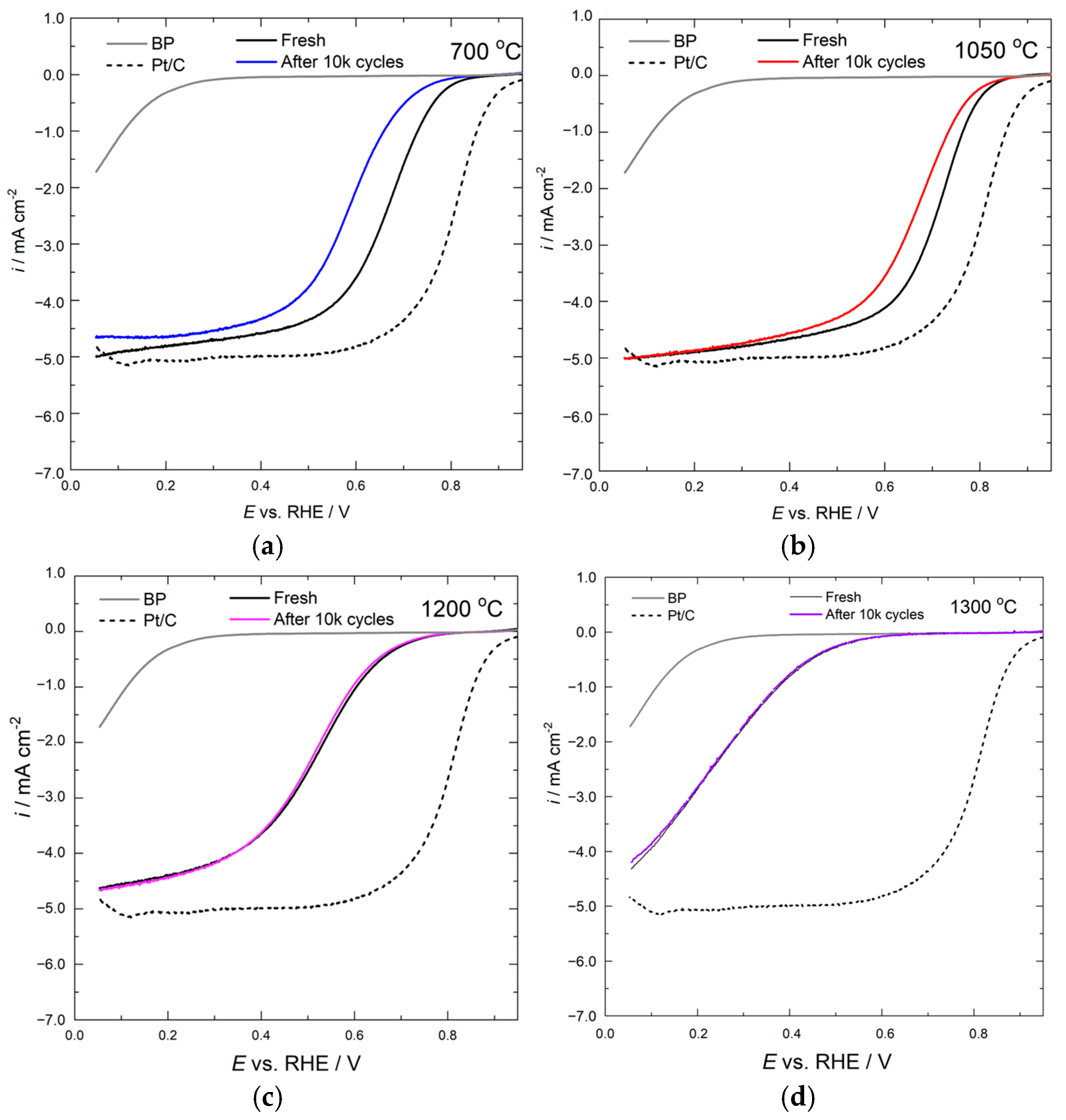
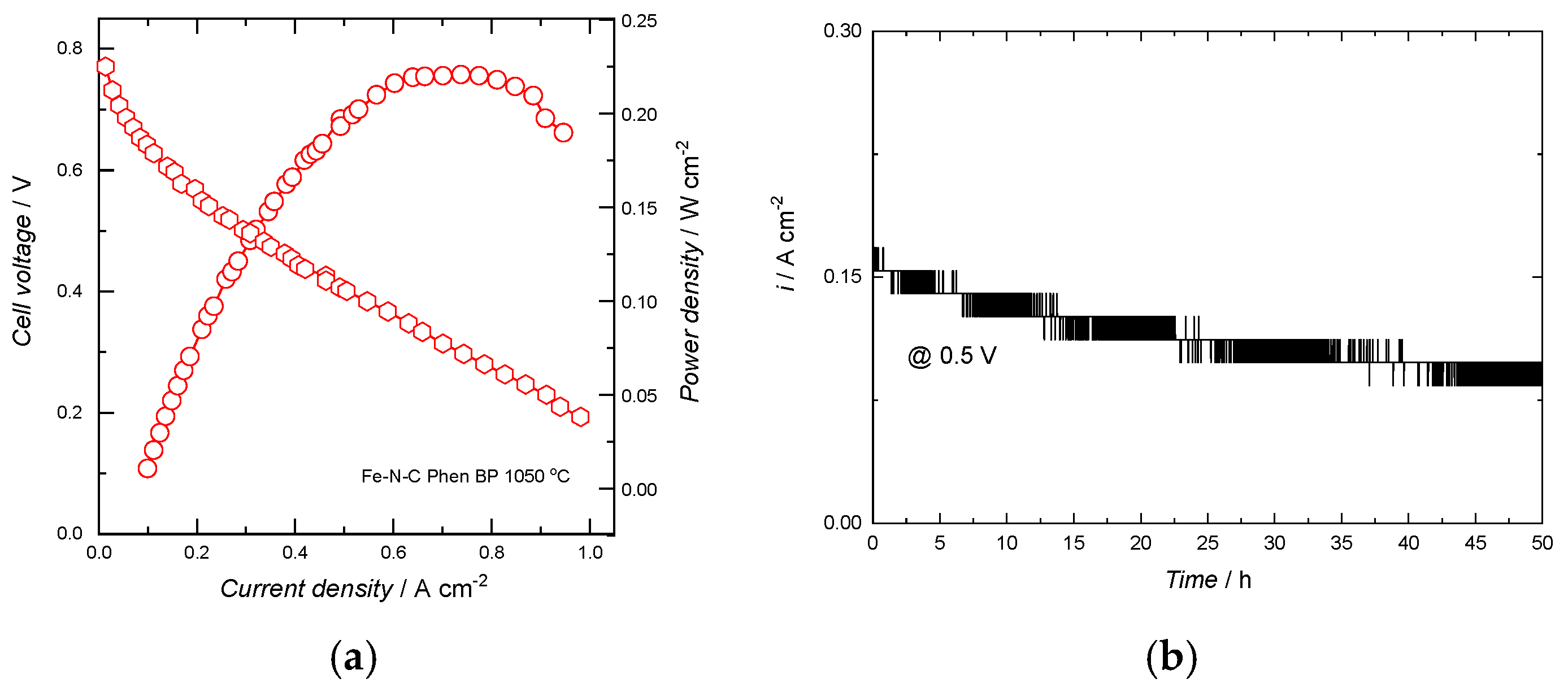
2.2. Electrochemical Results
3. Experimental
3.1. Synthesis of Electrocatalysts
3.2. Physical Characterization
3.3. Electrochemical Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dubau, L.; Castanheira, L.; Maillard, F.; Chatenet, M.; Lottin, O.; Maranzana, G.; Dillet, J.; Lamibrac, A.; Perrin, J.C.; Moukheiber, E.; et al. A Review of PEM Fuel Cell Durability: Materials Degradation, Local Heterogeneities of Aging and Possible Mitigation Strategies. Wiley Interdiscip. Rev. Energy Environ. 2014, 3, 540–560. [Google Scholar] [CrossRef]
- Lopes, P.P.; Li, D.; Lv, H.; Wang, C.; Tripkovic, D.; Zhu, Y.; Schimmenti, R.; Daimon, H.; Kang, Y.; Snyder, J.; et al. Eliminating Dissolution of Platinum-Based Electrocatalysts at the Atomic Scale. Nat. Mater. 2020, 19, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Mehmood, A.; Ali, B.; Nam, K.W.; Kucernak, A. Oxygen Reduction Reaction Activity in Non-Precious Single-Atom (M-N/C) Catalysts—Contribution of Metal and Carbon/Nitrogen Framework-Based Sites. ACS Catal. 2023, 13, 6661–6674. [Google Scholar] [CrossRef]
- He, Y.; Guo, H.; Hwang, S.; Yang, X.; He, Z.; Braaten, J.; Karakalos, S.; Shan, W.; Wang, M.; Zhou, H.; et al. Single Cobalt Sites Dispersed in Hierarchically Porous Nanofiber Networks for Durable and High-Power PGM-Free Cathodes in Fuel Cells. Adv. Mater. 2020, 32, 2003577. [Google Scholar] [CrossRef]
- Banham, D.; Kishimoto, T.; Zhou, Y.; Sato, T.; Bai, K.; Ozaki, J.I.; Imashiro, Y.; Ye, S. Critical Advancements in Achieving High Power and Stable Nonprecious Metal Catalyst-Based MEAs for Real-World Proton Exchange Membrane Fuel Cell Applications. Sci. Adv. 2018, 4, eaar7180. [Google Scholar] [CrossRef]
- Kumar, K.; Gairola, P.; Lions, M.; Ranjbar-Sahraie, N.; Mermoux, M.; Dubau, L.; Zitolo, A.; Jaouen, F.; Maillard, F. Physical and Chemical Considerations for Improving Catalytic Activity and Stability of Non-Precious-Metal Oxygen Reduction Reaction Catalysts. ACS Catal. 2018, 8, 11264–11276. [Google Scholar] [CrossRef]
- Ahluwalia, R.K.; Wang, X.; Osmieri, L.; Peng, J.-K.; Cetinbas, C.F.; Park, J.; Myers, D.J.; Chung, H.T.; Neyerlin, K.C. Stability of Atomically Dispersed Fe-N-C ORR Catalyst in Polymer Electrolyte Fuel Cell Environment. J. Electrochem. Soc. 2021, 168, 024513. [Google Scholar] [CrossRef]
- Cui, P.; Zhao, L.; Long, Y.; Dai, L.; Hu, C. Carbon-Based Electrocatalysts for Acidic Oxygen Reduction Reaction. Angew. Chem. Int. Ed. 2023, 62, e202218269. [Google Scholar] [CrossRef]
- Zhao, Y.; Yin, P.; Yang, Y.; Wang, R.; Gong, C.; Li, J.; Guo, J.; Wang, Q.; Ling, T. Converting Fe-N-C Single-atom Catalyst to a New FeNxSey Cluster Catalyst for Proton-exchange Membrane Fuel Cells. Angew. Chem. Int. Ed. 2025, 64, e202419501. [Google Scholar] [CrossRef]
- Ramaswamy, N.; Mukerjee, S. Influence of Inner- and Outer-Sphere Electron Transfer Mechanisms During Electrocatalysis of Oxygen Reduction in Alkaline Media. J. Phys. Chem. C 2011, 115, 18015–18026. [Google Scholar] [CrossRef]
- Yang, N.; Peng, L.; Li, L.; Li, J.; Liao, Q.; Shao, M.; Wei, Z. Theoretically Probing the Possible Degradation Mechanisms of an FeNC Catalyst During the Oxygen Reduction Reaction. Chem. Sci. 2021, 12, 12476–12484. [Google Scholar] [CrossRef] [PubMed]
- Chenitz, R.; Kramm, U.I.; Lefèvre, M.; Glibin, V.; Zhang, G.; Sun, S.; Dodelet, J.P. A Specific Demetalation of Fe-N4 Catalytic Sites in the Micropores of NC_Ar + NH3 is at the Origin of the Initial Activity Loss of the Highly Active Fe/N/C Catalyst Used for the Reduction of Oxygen in PEM Fuel Cells. Energy Environ. Sci. 2018, 11, 365–382. [Google Scholar] [CrossRef]
- Rauf, M.; Di Zhao, Y.; Wang, Y.C.; Zheng, Y.P.; Chen, C.; Yang, X.D.; Zhou, Z.Y.; Sun, S.G. Insight into the Different ORR Catalytic Activity of Fe/N/C Between Acidic and Alkaline Media: Protonation of Pyridinic Nitrogen. Electrochem. Commun. 2016, 73, 71–74. [Google Scholar] [CrossRef]
- Choi, C.H.; Lim, H.-K.; Chon, G.; Chung, M.W.; Altin, A.; Sahraie, N.R.; Sougrati, M.-T.; Stievano, L.; Oh, H.S.; Park, E.S.; et al. The Achilles’ Heel of Iron-Based Catalysts During Oxygen Reduction in an Acidic Medium. Energy Environ. Sci. 2018, 11, 3176–3182. [Google Scholar] [CrossRef]
- Bai, J.; Zhao, T.; Xu, M.; Mei, B.; Yang, L.; Shi, Z.; Zhu, S.; Wang, Y.; Jiang, Z.; Zhao, J.; et al. Monosymmetric Fe-N4 Sites Enabling Durable Proton Exchange Membrane Fuel Cell Cathode by Chemical Vapor Modification. Nat. Commun. 2024, 15, 4219. [Google Scholar] [CrossRef]
- Na, G.; Hwang, W.; Shin, H.; Park, S.; Park, J.E.; Lee, J.; Shin, Y.; Choi, H.; Shim, J.; Yeom, K.; et al. Durable and Active Nitrogen-Coordinated Iron Single-Atom Catalyst for Proton Exchange Membrane Fuel Cells Through Carbon Encapsulation. Adv. Energy Mater. 2024, 14, 2400565. [Google Scholar] [CrossRef]
- Zhang, L.; Dong, Y.; Li, L.; Shi, Y.; Zhang, Y.; Wei, L.; Dong, C.L.; Lin, Z.; Su, J. Concurrently Boosting Activity and Stability of Oxygen Reduction Reaction Catalysts via Judiciously Crafting Fe-Mn Dual Atoms for Fuel Cells. Nanomicro Lett. 2025, 17, 88. [Google Scholar] [CrossRef]
- Ye, W.; Chen, S.; Lin, Y.; Yang, L.; Chen, S.; Zheng, X.; Qi, Z.; Wang, C.; Long, R.; Chen, M.; et al. Precisely Tuning the Number of Fe Atoms in Clusters on N-Doped Carbon Toward Acidic Oxygen Reduction Reaction. Chem 2019, 5, 2865–2878. [Google Scholar] [CrossRef]
- He, Y.; Liu, S.; Priest, C.; Shi, Q.; Wu, G. Atomically Dispersed Metal–Nitrogen–Carbon Catalysts for Fuel Cells: Advances in Catalyst Design, Electrode Performance, and Durability Improvement. Chem. Soc. Rev. 2020, 49, 3484–3524. [Google Scholar] [CrossRef]
- Sahraie, N.R.; Kramm, U.I.; Steinberg, J.; Zhang, Y.; Thomas, A.; Reier, T.; Paraknowitsch, J.P.; Strasser, P. Quantifying the Density and Utilization of Active Sites in Non-Precious Metal Oxygen Electroreduction Catalysts. Nat. Commun. 2015, 6, 8618. [Google Scholar] [CrossRef]
- Yang, H.; Shang, L.; Zhang, Q.; Shi, R.; Waterhouse, G.I.N.; Gu, L.; Zhang, T. A Universal Ligand Mediated Method for Large Scale Synthesis of Transition Metal Single Atom Catalysts. Nat. Commun. 2019, 10, 4585. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Yoo, J.M.; Ahn, C.-Y.; Jang, J.-H.; Son, Y.J.; Shin, H.; Kang, J.; Kang, Y.S.; Yoo, S.J.; Lee, K.-S.; et al. Rational Generation of Fe-Nx Active Sites in Fe-N-C Electrocatalysts Facilitated by Fe-N Coordinated Precursors for the Oxygen Reduction Reaction. ChemCatChem 2019, 11, 5982–5988. [Google Scholar] [CrossRef]
- Woo, J.; Yang, S.Y.; Sa, Y.J.; Choi, W.Y.; Lee, M.H.; Lee, H.W.; Shin, T.J.; Kim, T.Y.; Joo, S.H. Promoting Oxygen Reduction Reaction Activity of Fe-N/C Electrocatalysts by Silica-Coating-Mediated Synthesis for Anion-Exchange Membrane Fuel Cells. Chem. Mater. 2018, 30, 6684–6701. [Google Scholar] [CrossRef]
- Li, X.; Li, P.; Yang, J.; Xie, L.; Wang, N.; Lei, H.; Zhang, C.; Zhang, W.; Lee, Y.M.; Zhang, W.; et al. A Cobalt (II) Porphyrin with a Tethered Imidazole for Efficient Oxygen Reduction and Evolution Electrocatalysis. J. Energy Chem. 2023, 76, 617–621. [Google Scholar] [CrossRef]
- Snitkoff, R.Z.; Levy, N.; Ozery, I.; Ruthstein, S.; Elbaz, L. Imidazole Decorated Reduced Graphene Oxide: A Biomimetic Ligand for Selective Oxygen Reduction Electrocatalysis with Metalloporphyrins. Carbon 2019, 143, 223–229. [Google Scholar] [CrossRef]
- Du, C.; Gao, Y.; Chen, H.; Li, P.; Zhu, S.; Wang, J.; He, Q.; Chen, W. A Cu and Fe Dual-Atom Nanozyme Mimicking Cytochrome C Oxidase to Boost the Oxygen Reduction Reaction. J. Mater. Chem. A 2020, 8, 16994–17001. [Google Scholar] [CrossRef]
- Mamtani, K.; Singh, D.; Tian, J.; Millet, J.M.M.; Miller, J.T.; Co, A.C.; Ozkan, U.S. Evolution of N-Coordinated Iron-Carbon (FeNC) Catalysts and their Oxygen Reduction (ORR) Performance in Acidic Media at Various Stages of Catalyst Synthesis: An Attempt at Benchmarking. Catal. Lett. 2016, 146, 1749–1770. [Google Scholar] [CrossRef]
- Yin, S.; Yi, H.; Liu, M.; Yang, J.; Yang, S.; Zhang, B.W.; Chen, L.; Cheng, X.; Huang, H.; Huang, R.; et al. An In Situ Exploration of How Fe/N/C Oxygen Reduction Catalysts Evolve During Synthesis under Pyrolytic Conditions. Nat. Commun. 2024, 15, 6229. [Google Scholar] [CrossRef]
- Guan, D.; Xu, H.; Huang, Y.-C.; Jing, C.; Tsujimoto, Y.; Xu, X.; Lin, Z.; Tang, J.; Wang, Z.; Sun, X.; et al. Operando Studies Redirect Spatiotemporal Restructuration of Model Coordinated Oxides in Electrochemical Oxidation. Adv. Mater. 2025, 37, 2413073. [Google Scholar] [CrossRef]
- Qian, Z.; Hu, Z.; Zhang, Z.; Li, Z.; Dou, M.; Wang, F. Out-of-Plane FeII-N4 Moiety Modified Fe-N Co-Doped Porous Carbons as High-Performance Electrocatalysts for the Oxygen Reduction Reaction. Catal. Sci. Technol. 2017, 7, 4017–4023. [Google Scholar] [CrossRef]
- Zou, D.; Yi, Y.; Song, Y.; Guan, D.; Xu, M.; Ran, R.; Wang, W.; Zhou, W.; Shao, Z. The BaCe0.16Y0.04Fe0.8O3−δ Nanocomposite: A New High-Performance Cobalt-Free Triple-Conducting Cathode for Protonic Ceramic Fuel Cells Operating at Reduced Temperatures. J. Mater. Chem. A 2022, 10, 5381. [Google Scholar] [CrossRef]
- Xia, D.; Tang, X.; Dai, S.; Ge, R.; Rykov, A.; Wang, J.; Huang, T.-H.; Wang, K.-W.; Wei, Y.; Zhang, K.; et al. Ultrastable Fe-N-C Fuel Cell Electrocatalysts by Eliminating Non-Coordinating Nitrogen and Regulating Coordination Structures at High Temperatures. Adv. Mater. 2023, 35, 2204474. [Google Scholar] [CrossRef] [PubMed]
- Teixeira Santos, K.; Kumar, K.; Dubau, L.; Ge, H.; Berthon-Fabry, S.; Vasconcellos, C.S.A.; Lima, F.H.B.; Asset, T.; Atanassov, P.; Saveleva, V.A.; et al. Spontaneous Aerobic Ageing of Fe-N-C Materials and Consequences on Oxygen Reduction Reaction Kinetics. J. Power Sources 2023, 564, 232829. [Google Scholar] [CrossRef]
- Guo, W.; Pan, M.; Xie, Q.; Fan, H.; Luo, L.; Jing, Q.; Shen, Y.; Yan, Y.; Liu, M.; Wang, Z. Achieving pH-Universal Oxygen Electrolysis via Synergistic Density and Coordination Tuning Over Biomass-Derived Fe Single-Atom Catalyst. Nat. Commun. 2025, 16, 2920. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasconcellos, C.S.A.; Galiote, N.A.; Khan, N.; Paredes-Salazar, E.A.; Souza, M.L.; Sasaki, K.; Li, M.; Lima, F.H.B. 1,10-Phenanthroline-Iron Complex-Derived Fe-N-C Electrocatalysts: Enhanced Oxygen Reduction Activity and Stability Through Synthesis Tuning. Catalysts 2025, 15, 821. https://doi.org/10.3390/catal15090821
Vasconcellos CSA, Galiote NA, Khan N, Paredes-Salazar EA, Souza ML, Sasaki K, Li M, Lima FHB. 1,10-Phenanthroline-Iron Complex-Derived Fe-N-C Electrocatalysts: Enhanced Oxygen Reduction Activity and Stability Through Synthesis Tuning. Catalysts. 2025; 15(9):821. https://doi.org/10.3390/catal15090821
Chicago/Turabian StyleVasconcellos, Carlos S. A., Nelson A. Galiote, Nadeem Khan, Enrique A. Paredes-Salazar, Maykon L. Souza, Kotaro Sasaki, Meng Li, and Fabio H. B. Lima. 2025. "1,10-Phenanthroline-Iron Complex-Derived Fe-N-C Electrocatalysts: Enhanced Oxygen Reduction Activity and Stability Through Synthesis Tuning" Catalysts 15, no. 9: 821. https://doi.org/10.3390/catal15090821
APA StyleVasconcellos, C. S. A., Galiote, N. A., Khan, N., Paredes-Salazar, E. A., Souza, M. L., Sasaki, K., Li, M., & Lima, F. H. B. (2025). 1,10-Phenanthroline-Iron Complex-Derived Fe-N-C Electrocatalysts: Enhanced Oxygen Reduction Activity and Stability Through Synthesis Tuning. Catalysts, 15(9), 821. https://doi.org/10.3390/catal15090821








