Spark Ablation for the Fabrication of PEM Water Electrolysis Catalyst-Coated Membranes
Abstract
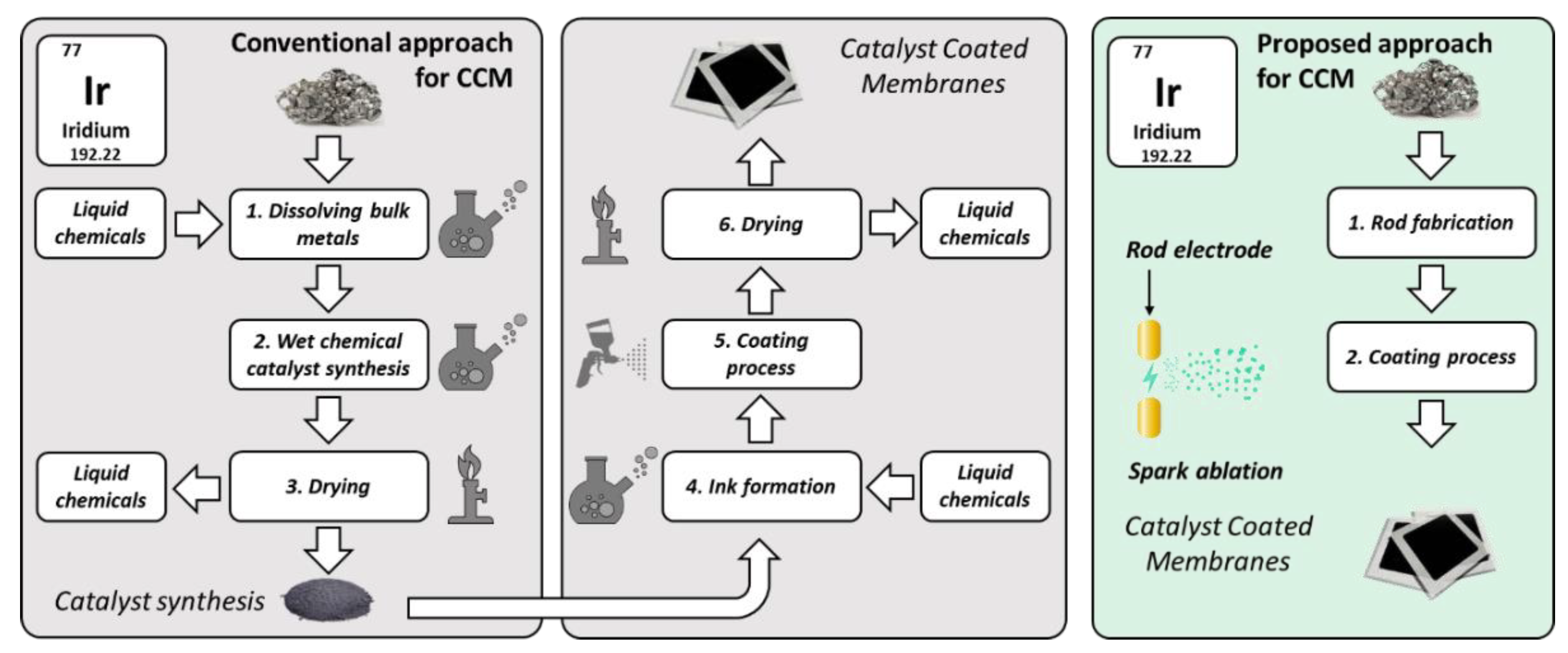
1. Introduction
2. Results and Discussion
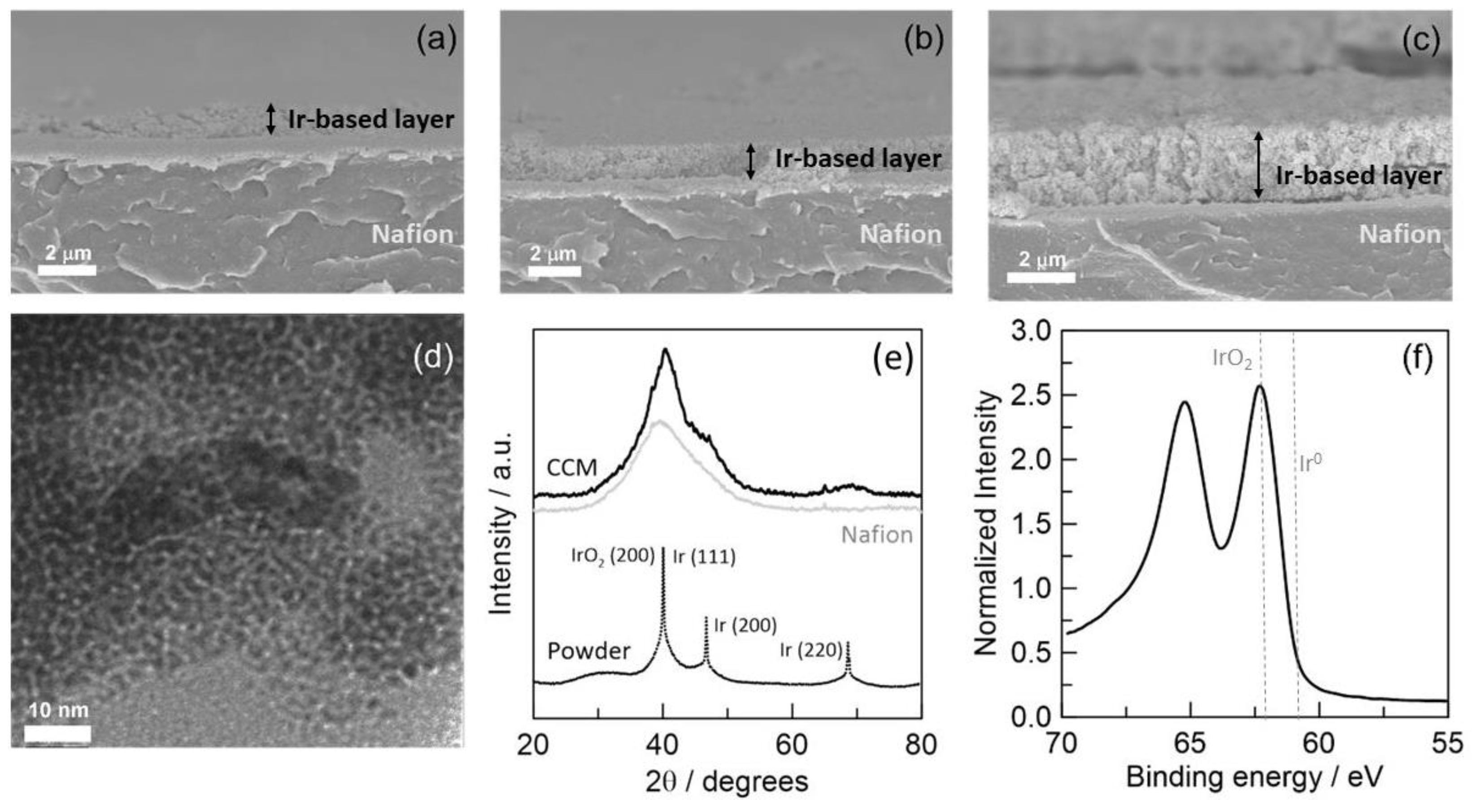
2.1. Material Characterization
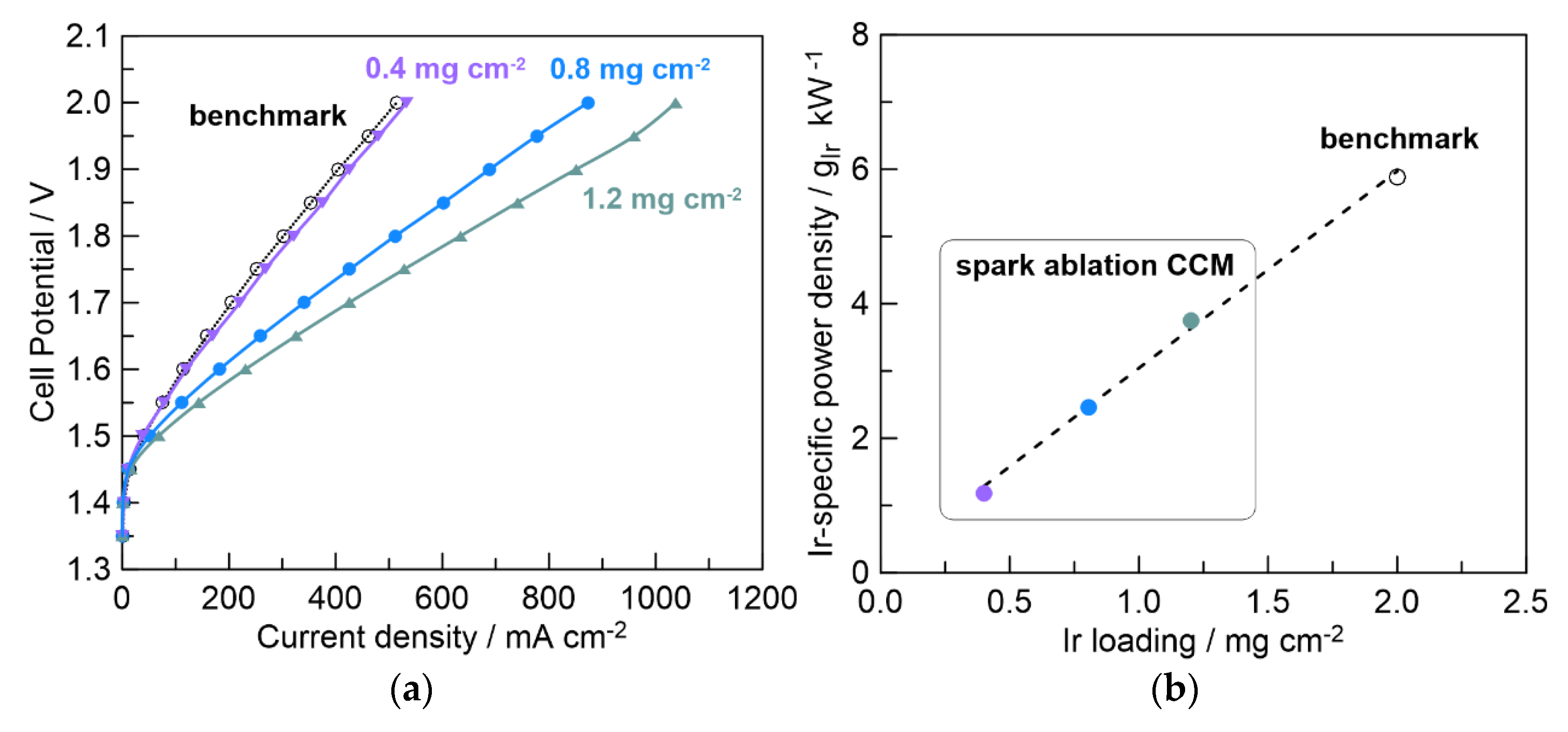
2.2. Electrocatalytic Activity of Spark-Ablation Anode-Coated CCMs
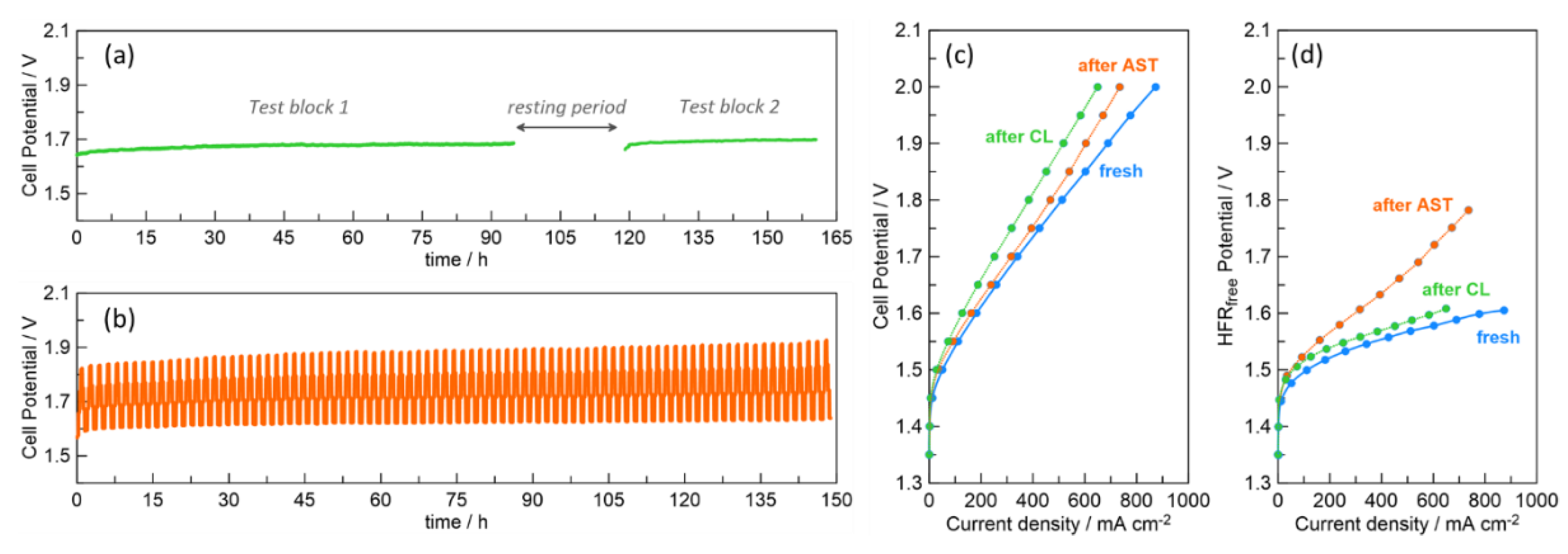
2.3. Durability of Spark-Ablation Anode-Coated CCMs
2.4. Performance of Full CCMs Fabricated with Spark Ablation
2.5. Other Practical Considerations
3. Materials and Methods
3.1. Fabrication of Catalyst-Coated Membranes (CCMs) with Spark Ablation
3.2. Physicochemical Characterization
3.3. Catalyst Loading
3.4. Fabrication of Final Membrane Electrode Assemblies
3.5. Polarization Measurements
3.6. Durability Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taibi, E.; Miranda, R.; Vanhoudt, W.; Winkel, T.; Lanoix, J.-C.; Barth, F. Hydrogen from Renewable Power: Technology Outlook for the Energy Transition; International Renewable Energy Agency: Abu Dhabi, United Arab Emirates, 2018. [Google Scholar]
- Schmidt, O.; Gambhir, A.; Staffell, I.; Hawkes, A.; Nelson, J.; Few, S. Future cost and performance of water electrolysis: An expert elicitation study. Int. J. Hydrogen Energy 2017, 42, 30470–30492. [Google Scholar] [CrossRef]
- Sapountzi, F.M.; Gracia, J.M.; Weststrate, C.J.; Fredriksson, H.O.A.; Niemantsverdriet, J.W. Electrocatalysts for the generation of hydrogen, oxygen and synthesis gas. Prog. Energy Combust. Sci. 2017, 58, 1–35. [Google Scholar] [CrossRef]
- Babic, U.; Suermann, M.; Büchi, F.N.; Gubler, L.; Schmidt, T.J. Critical REVIEW—Identifying Critical gaps for polymer electrolyte water electrolysis development. J. Electrochem. Soc. 2017, 164, F387–F399. [Google Scholar] [CrossRef]
- Shiva Kumar, S.; Himabindu, V. Hydrogen production by PEM water electrolysis—A review. Mater. Sci. Energy Technol. 2019, 2, 442–454. [Google Scholar] [CrossRef]
- Bernt, M.; Hartig-Weiß, A.; Tovini, M.F.; El-Sayed, H.A.; Schramm, C.; Schröter, J.; Gebauer, C.; Gasteiger, H.A. Current challenges in catalyst development for PEM water electrolyzers. Chemie Ingenieur Technik 2020, 92, 31–39. [Google Scholar] [CrossRef]
- International Renewable Energy Agency. Green Hydrogen Cost Reduction Scaling Up Electrolysers to Meet The 1.5°C Climate Goal; International Renewable Energy Agency: Abu Dhabi, United Arab Emirates, 2020. [Google Scholar]
- Holzapfel, P.K.R.; Bühler, M.; Escalera-López, D.; Bierling, M.; Speck, F.D.; Mayrhofer, K.J.J.; Cherevko, S.; Pham, C.V.; Thiele, S. Fabrication of a robust PEM water electrolyzer based on non-noble metal cathode catalyst: [Mo3S13]2− clusters anchored to n-doped carbon nanotubes. Small 2020, 16, 2003161. [Google Scholar] [CrossRef]
- Ng, J.W.D.; Hellstern, T.R.; Kibsgaard, J.; Hinckley, A.C.; Benck, J.D.; Jaramillo, T.F. Polymer electrolyte membrane electrolyzers utilizing non-precious mo-based hydrogen evolution catalysts. ChemSusChem 2015, 8, 3512–3519. [Google Scholar] [CrossRef]
- Sarno, M.; Ponticorvo, E. High hydrogen production rate on RuS2@MoS2 hybrid nanocatalyst by PEM electrolysis. Int. J. Hydrogen Energy 2019, 44, 4398–4405. [Google Scholar] [CrossRef]
- Morozan, A.; Johnson, H.; Roiron, C.; Genay, G.; Aldakov, D.; Ghedjatti, A.; Nguyen, C.T.; Tran, P.D.; Kinge, S.; Artero, V. Nonprecious bimetallic iron–molybdenum sulfide electrocatalysts for the hydrogen evolution reaction in proton exchange membrane electrolyzers. ACS Catal. 2020, 10, 14336–14348. [Google Scholar] [CrossRef]
- Mo, J.; Stefanov, B.I.; Lau, T.H.M.; Chen, T.; Wu, S.; Wang, Z.; Gong, X.Q.; Wilkinson, I.; Schmid, G.; Tsang, S.C.E. Superior performance of Ag over Pt for hydrogen evolution reaction in water electrolysis under high overpotentials. ACS Appl. Energy Mater. 2019, 2, 1221–1228. [Google Scholar] [CrossRef]
- King, L.A.; Hubert, M.A.; Capuano, C.; Manco, J.; Danilovic, N.; Valle, E.; Hellstern, T.R.; Ayers, K.; Jaramillo, T.F. A non-precious metal hydrogen catalyst in a commercial polymer electrolyte membrane electrolyser. Nat. Nanotechnol. 2019, 14, 1071–1074. [Google Scholar] [CrossRef] [PubMed]
- Brito, J.; Restivo, J.; Sousa, J.P.S.; Spera, N.C.M.; Falcão, D.S.; Rocha, A.; Pinto, A.M.F.R.; Pereira, M.F.R.; Soares, O.S.G.P.; Sadykov, A.; et al. Implementation of transition metal phosphides as Pt-free catalysts for PEM water electrolysis. Energies 2022, 15, 1821. [Google Scholar] [CrossRef]
- Sapountzi, F.M.; Orlova, E.D.; Sousa, J.P.S.; Salonen, L.M.; Lebedev, O.I.; Zafeiropoulos, G.; Tsampas, M.N.; Niemantsverdriet, H.J.W.; Kolen’ko, Y.V. FeP Nanocatalyst with Preferential [010] Orientation Boosts the Hydrogen Evolution Reaction in Polymer-Electrolyte Membrane Electrolyzer. Energy Fuels 2020, 34, 6423–6429. [Google Scholar] [CrossRef]
- Owens-Baird, B.; Xu, J.; Petrovykh, D.Y.; Bondarchuk, O.; Ziouani, Y.; González-Ballesteros, N.; Yox, P.; Sapountzi, F.M.; Niemantsverdriet, H.; Kolen’ko, Y.V.; et al. NiP2: A story of two divergent polymorphic multifunctional materials. Chem. Mater. 2019, 31, 3407–3418. [Google Scholar] [CrossRef]
- Bernt, M.; Siebel, A.; Gasteiger, H.A. Analysis of voltage losses in PEM water electrolyzers with low platinum group metal loadings. J. Electrochem. Soc. 2018, 165, F305–F314. [Google Scholar] [CrossRef]
- Li, L.; Wang, P.; Shao, Q.; Huang, X. Recent progress in advanced electrocatalyst design for acidic oxygen evolution reaction. Adv. Mater. 2021, 33, 2004243. [Google Scholar] [CrossRef]
- Barrios Jimenez, A.M.; Burkhardt, U.; Cardoso-Gil, R.; Höfer, K.; Altendorf, S.G.; Schlögl, R.; Grin, Y.; Antonyshyn, I. HF2B2IR5: A self-optimizing catalyst for the oxygen evolution reaction. ACS Appl. Energy Mater. 2020, 3, 11042–11052. [Google Scholar] [CrossRef]
- Evans, T.A.; Choi, K.S. Electrochemical synthesis and investigation of stoichiometric, phase—Pure CoSb2O6 and MnSb2O6 electrodes for the oxygen evolution reaction in acidic media. ACS Appl. Energy Mater. 2020, 3, 5563–5571. [Google Scholar] [CrossRef]
- Yang, L.; Yu, G.; Ai, X.; Yan, W.; Duan, H.; Chen, W.; Li, X.; Wang, T.; Zhang, C.; Huang, X.; et al. Efficient oxygen evolution electrocatalysis in acid by a perovskite with face-sharing IrO6 octahedral dimers. Nat. Commun. 2018, 9, 5236. [Google Scholar] [CrossRef]
- Lemoine, K.; Gohari-Bajestani, Z.; Moury, R.; Terry, A.; Guiet, A.; Grenèche, J.M.; Hémon-Ribaud, A.; Heidary, N.; Maisonneuve, V.; Kornienko, N.; et al. Amorphous iron-manganese oxyfluorides, promising catalysts for oxygen evolution reaction under acidic media. ACS Appl. Energy Mater. 2021, 4, 1173–1181. [Google Scholar] [CrossRef]
- Sun, X.; Xu, K.; Fleischer, C.; Liu, X.; Grandcolas, M.; Strandbakke, R.; Bjørheim, T.; Norby, T.; Chatzitakis, A. Earth-abundant electrocatalysts in proton exchange membrane electrolyzers. Catalysts 2018, 8, 657. [Google Scholar] [CrossRef]
- Minke, C.; Suermann, M.; Bensmann, B.; Hanke-Rauschenbach, R. Is iridium demand a potential bottleneck in the realization of large-scale PEM water electrolysis? Int. J. Hydrogen Energy 2021, 46, 23581–23590. [Google Scholar] [CrossRef]
- Zhao, S.; Stocks, A.; Rasimick, B.; More, K.; Xu, H. Highly active, durable dispersed iridium nanocatalysts for PEM water electrolyzers. J. Electrochem. Soc. 2018, 165, F82–F89. [Google Scholar] [CrossRef]
- Oakton, E.; Lebedev, D.; Povia, M.; Abbott, D.F.; Fabbri, E.; Fedorov, A.; Nachtegaal, M.; Copéret, C.; Schmidt, T.J. IrO2 -TiO2: A high-surface-area, active, and stable electrocatalyst for the oxygen evolution reaction. ACS Catal. 2017, 7, 2346–2352. [Google Scholar] [CrossRef]
- Neophytides, S.G.; Murase, K.; Zafeiratos, S.; Papakonstantinou, G.; Paloukis, F.E.; Krstajic, N.V.; Jaksic, M.M. Composite hypo-hyper-d-intermetallic and interionic phases as supported interactive electrocatalysts. J. Phys. Chem. B 2006, 110, 3030–3042. [Google Scholar] [CrossRef]
- Ma, L.; Sui, S.; Zhai, Y. Preparation and characterization of Ir/TiC catalyst for oxygen evolution. J. Power Sources 2008, 177, 470–477. [Google Scholar] [CrossRef]
- Karimi, F.; Peppley, B.A. Metal carbide and oxide supports for iridium-based oxygen evolution reaction electrocatalysts for polymer electrolyte membrane water electrolysis. Electrochim. Acta 2017, 246, 654–670. [Google Scholar] [CrossRef]
- Lebedev, D.; Ezhov, R.; Heras-Domingo, J.; Comas-Vives, A.; Kaeffer, N.; Willinger, M.; Solans-Monfort, X.; Huang, X.; Pushkar, Y.; Copéret, C. Atomically dispersed iridium on indium tin oxide efficiently catalyzes water oxidation. ACS Cent. Sci. 2020, 6, 1189–1198. [Google Scholar] [CrossRef]
- Hegge, F.; Lombeck, F.; Cruz Ortiz, E.; Bohn, L.; von Holst, M.; Kroschel, M.; Hübner, J.; Breitwieser, M.; Strasser, P.; Vierrath, S. Efficient and stable low iridium loaded anodes for PEM water electrolysis made possible by nanofiber interlayers. ACS Appl. Energy Mater. 2020, 3, 8276–8284. [Google Scholar] [CrossRef]
- Papaderakis, A.; Pliatsikas, N.; Prochaska, C.; Vourlias, G.; Patsalas, P.; Tsiplakides, D.; Balomenou, S.; Sotiropoulos, S. Oxygen evolution at IrO2 Shell–Ir−Ni core electrodes prepared by galvanic replacement. J. Phys. Chem. C 2016, 120, 19995–20005. [Google Scholar] [CrossRef]
- Bele, M.; Jovanovič, P.; Marinko, Ž.; Drev, S.; Šelih, V.S.; Kovač, J.; Gaberšček, M.; Koderman Podboršek, G.; Dražić, G.; Hodnik, N.; et al. Increasing the oxygen-evolution reaction performance of nanotubular titanium oxynitride-supported Ir nanoparticles by a strong metal–support interaction. ACS Catal. 2020, 10, 13688–13700. [Google Scholar] [CrossRef]
- Nong, H.N.; Oh, H.; Reier, T.; Willinger, E.; Willinger, M.; Petkov, V.; Teschner, D.; Strasser, P. Oxide-supported IrNiOx core–shell particles as efficient, cost-effective, and stable catalysts for electrochemical water splitting. Angew. Chem. Int. Ed. 2015, 54, 2975–2979. [Google Scholar] [CrossRef] [PubMed]
- Lewinski, K.A.; van der Vliet, D.; Luopa, S.M. NSTF advances for PEM electrolysis—The effect of alloying on activity of NSTF electrolyzer catalysts and performance of NSTF based PEM electrolyzers. ECS Trans. 2015, 69, 893–917. [Google Scholar] [CrossRef]
- Alia, S.M.; Shulda, S.; Ngo, C.; Pylypenko, S.; Pivovar, B.S. Iridium-based nanowires as highly active, oxygen evolution reaction electrocatalysts. ACS Catal. 2018, 8, 2111–2120. [Google Scholar] [CrossRef]
- Kim, Y.J.; Lim, A.; Kim, J.M.; Lim, D.; Chae, K.H.; Cho, E.N.; Han, H.J.; Jeon, K.U.; Kim, M.; Lee, G.H.; et al. Highly efficient oxygen evolution reaction via facile bubble transport realized by three-dimensionally stack-printed catalysts. Nat. Commun. 2020, 11, 4921. [Google Scholar] [CrossRef]
- Cheng, J.; Yang, J.; Kitano, S.; Juhasz, G.; Higashi, M.; Sadakiyo, M.; Kato, K.; Yoshioka, S.; Sugiyama, T.; Yamauchi, M.; et al. Impact of Ir-valence control and surface nanostructure on oxygen evolution reaction over a highly efficient Ir–TiO2 nanorod catalyst. ACS Catal. 2019, 9, 6974–6986. [Google Scholar] [CrossRef]
- Oh, H.-S.; Nong, H.N.; Reier, T.; Gliech, M.; Strasser, P. Oxide-supported Ir nanodendrites with high activity and durability for the oxygen evolution reaction in acid PEM water electrolyzers. Chem. Sci. 2015, 6, 3321–3328. [Google Scholar] [CrossRef]
- Ouimet, R.J.; Ebaugh, T.A.; Mirshekari, G.; Bliznakov, S.; Bonville, L.J.; Maric, R. Current status on the manufacturing of nanomaterials for proton exchange membrane energy systems by vapor-based processes. Energy Fuels 2021, 35, 1933–1956. [Google Scholar] [CrossRef]
- Ayers, K.E.; Renner, J.N.; Danilovic, N.; Wang, J.X.; Zhang, Y.; Maric, R.; Yu, H. Pathways to Ultra-low platinum group metal catalyst loading in proton exchange membrane electrolyzers. Catal. Today 2016, 262, 121–132. [Google Scholar] [CrossRef]
- Laube, A.; Hofer, A.; Ressel, S.; Chica, A.; Bachmann, J.; Struckmann, T. PEM water electrolysis cells with catalyst coating by atomic layer deposition. Int. J. Hydrogen Energy 2021, 46, 38972–38982. [Google Scholar] [CrossRef]
- Sapountzi, F.M.; Divane, S.C.; Papaioannou, E.I.; Souentie, S.; Vayenas, C.G. The role of nafion content in sputtered IrO2 based anodes for low temperature PEM water electrolysis. J. Electroanal. Chem. 2011, 662, 116–122. [Google Scholar] [CrossRef]
- Slavcheva, E.; Radev, I.; Bliznakov, S.; Topalov, G.; Andreev, P.; Budevski, E. Sputtered iridium oxide films as electrocatalysts for water splitting via PEM electrolysis. Electrochim. Acta 2007, 52, 3889–3894. [Google Scholar] [CrossRef]
- Labou, D.; Slavcheva, E.; Schnakenberg, U.; Neophytides, S. Performance of laboratory polymer electrolyte membrane hydrogen generator with sputtered iridium oxide anode. J. Power Sources 2008, 185, 1073–1078. [Google Scholar] [CrossRef]
- Schmidt-Ott, A. Spark Ablation, Building Blocks for Nanotechnology; Schmidt-Ott, A., Ed.; Jenny Stanford Publishing: New York, NY, USA, 2020. [Google Scholar] [CrossRef]
- Pfeiffer, T.V.; Feng, J.; Schmidt-Ott, A. New developments in spark production of nanoparticles. Adv. Powder Technol. 2014, 25, 56–70. [Google Scholar] [CrossRef]
- Van Ginkel, H.J.; Romijn, J.; Vollebregt, S.; Zhang, G.Q. High Step Coverage Interconnects by Printed Nanoparticles. In Proceedings of the 2021 23rd European Microelectronics and Packaging Conference & Exhibition (EMPC), Virtual Conference, 13–16 September 2021; pp. 1–4. [Google Scholar] [CrossRef]
- Aghajani, S.; Accardo, A.; Tichem, M. Tunable photoluminescence and SERS behaviour of additively manufactured Au nanoparticle patterns. RSC Adv. 2021, 11, 16849–16859. [Google Scholar] [CrossRef] [PubMed]
- Aghajani, S.; Accardo, A.; Tichem, M. Aerosol direct writing and thermal tuning of copper nanoparticle patterns as surface-enhanced raman scattering sensors. ACS Appl. Nano Mater. 2020, 3, 5665–5675. [Google Scholar] [CrossRef]
- Becker, R.; Weber, K.; Pfeiffer, T.V.; van Kranendonk, J.; Schouten, K.J. A scalable high-throughput deposition and screening setup relevant to industrial electrocatalysis. Catalysts 2020, 10, 1165. [Google Scholar] [CrossRef]
- Rajan, Z.S.H.S.; Binninger, T.; Kooyman, P.J.; Susac, D.; Mohamed, R. Organometallic Chemical deposition of crystalline iridium oxide nanoparticles on antimony-doped tin oxide support with high-performance for the oxygen evolution reaction. Catalysis Sci. Technol. 2020, 10, 3938–3948. [Google Scholar] [CrossRef]
- Freakley, S.J.; Ruiz-Esquius, J.; Morgan, D.J. The X-Ray photoelectron spectra of Ir, IrO2 and IrCl3 revisited. Surf. Interface Anal. 2017, 49, 794–799. [Google Scholar] [CrossRef]
- Bender, G.; Carmo, M.; Smolinka, T.; Gago, A.; Danilovic, N.; Mueller, M.; Ganci, F.; Fallisch, A.; Lettenmeier, P.; Friedrich, K.A.; et al. Initial approaches in benchmarking and round robin testing for proton exchange membrane water electrolyzers. Int. J. Hydrogen Energy 2019, 44, 9174–9187. [Google Scholar] [CrossRef]
- Tan, X.; Shen, J.; Semagina, N.; Secanell, M. Decoupling structure-sensitive deactivation mechanisms of Ir/IrOx electrocatalysts toward oxygen evolution reaction. J. Catal. 2019, 371, 57–70. [Google Scholar] [CrossRef]
- Papakonstantinou, G.; Algara-Siller, G.; Teschner, D.; Vidaković-Koch, T.; Schlögl, R.; Sundmacher, K. Degradation study of a proton exchange membrane water electrolyzer under dynamic operation conditions. Appl. Energy 2020, 280, 115911. [Google Scholar] [CrossRef]
- Siracusano, S.; Baglio, V.; van Dijk, N.; Merlo, L.; Aricò, A.S. Enhanced performance and durability of low catalyst loading PEM water electrolyser based on a short-side chain perfluorosulfonic ionomer. Appl. Energy 2017, 192, 477–489. [Google Scholar] [CrossRef]
- Scofield, J.H. Hartree-slater subshell photoionization cross-sections at 1254 and 1487 EV. J. Electron Spectrosc. Relat. Phenom. 1976, 8, 129–137. [Google Scholar] [CrossRef]
- Frensch, S.H.; Fouda-Onana, F.; Serre, G.; Thoby, D.; Araya, S.S.; Kær, S.K. Influence of the operation mode on PEM water electrolysis degradation. Int. J. Hydrogen Energy 2019, 44, 29889–29898. [Google Scholar] [CrossRef]
- Dhawan, H.; Secanell, M.; Semagina, N. State-of-the-art iridium-based catalysts for acidic water electrolysis: A minireview of wet-chemistry synthesis methods: Preparation routes for active and durable iridium catalysts. Johns. Matthey Technol. Rev. 2021, 65, 247–262. [Google Scholar] [CrossRef]
- Xie, M.; Chu, T.; Wang, T.; Wan, K.; Yang, D.; Li, B.; Ming, P.; Zhang, C. Preparation, performance and challenges of catalyst layer for proton exchange membrane fuel cell. Membranes 2021, 11, 879. [Google Scholar] [CrossRef]
- Tsotridis, G.; Pilenga, A. EU Harmonised Protocols for Testing of Low Temperature Water Electrolysers; European Comission: Brussels, Belgium, 2021. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sapountzi, F.M.; Lavorenti, M.; Vrijburg, W.; Dimitriadou, S.; Tyburska-Pueschel, B.; Thüne, P.; Niemantsverdriet, H.; Pfeiffer, T.V.; Tsampas, M.N. Spark Ablation for the Fabrication of PEM Water Electrolysis Catalyst-Coated Membranes. Catalysts 2022, 12, 1343. https://doi.org/10.3390/catal12111343
Sapountzi FM, Lavorenti M, Vrijburg W, Dimitriadou S, Tyburska-Pueschel B, Thüne P, Niemantsverdriet H, Pfeiffer TV, Tsampas MN. Spark Ablation for the Fabrication of PEM Water Electrolysis Catalyst-Coated Membranes. Catalysts. 2022; 12(11):1343. https://doi.org/10.3390/catal12111343
Chicago/Turabian StyleSapountzi, Foteini M., Marek Lavorenti, Wilbert Vrijburg, Sofia Dimitriadou, Beata Tyburska-Pueschel, Peter Thüne, Hans Niemantsverdriet, Tobias V. Pfeiffer, and Mihalis N. Tsampas. 2022. "Spark Ablation for the Fabrication of PEM Water Electrolysis Catalyst-Coated Membranes" Catalysts 12, no. 11: 1343. https://doi.org/10.3390/catal12111343
APA StyleSapountzi, F. M., Lavorenti, M., Vrijburg, W., Dimitriadou, S., Tyburska-Pueschel, B., Thüne, P., Niemantsverdriet, H., Pfeiffer, T. V., & Tsampas, M. N. (2022). Spark Ablation for the Fabrication of PEM Water Electrolysis Catalyst-Coated Membranes. Catalysts, 12(11), 1343. https://doi.org/10.3390/catal12111343