Advanced Treatment of Phosphorus Pesticide Wastewater Using an Integrated Process of Coagulation and Ozone Catalytic Oxidation
Abstract
:1. Introduction
2. Results
2.1. Coagulation Treatment of Biochemical Tailwater from Phosphorus-Containing Pesticide Wastewater
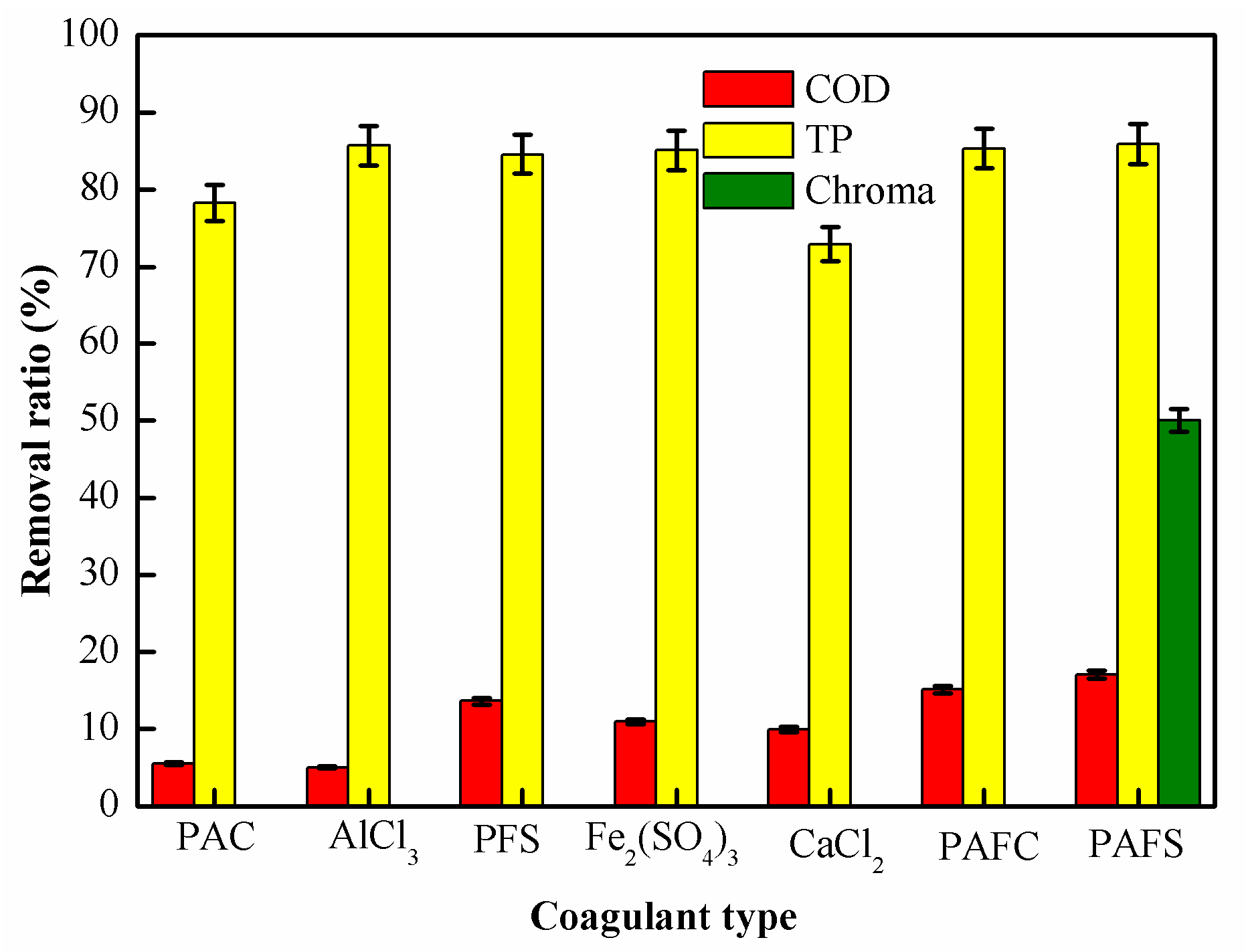
2.1.1. Effect of Coagulant Type on Coagulation Performance
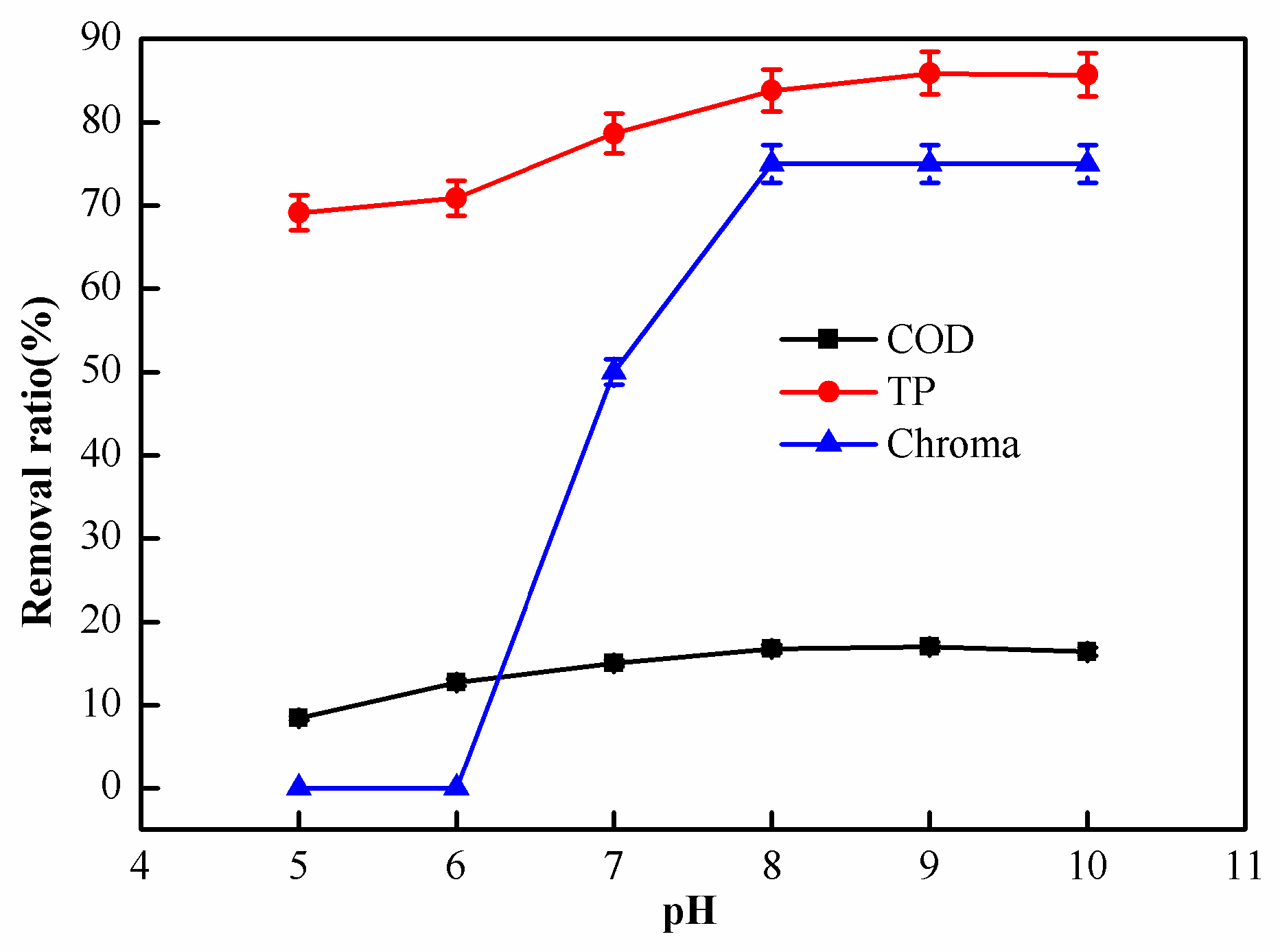
2.1.2. Effect of pH on Coagulation Performance
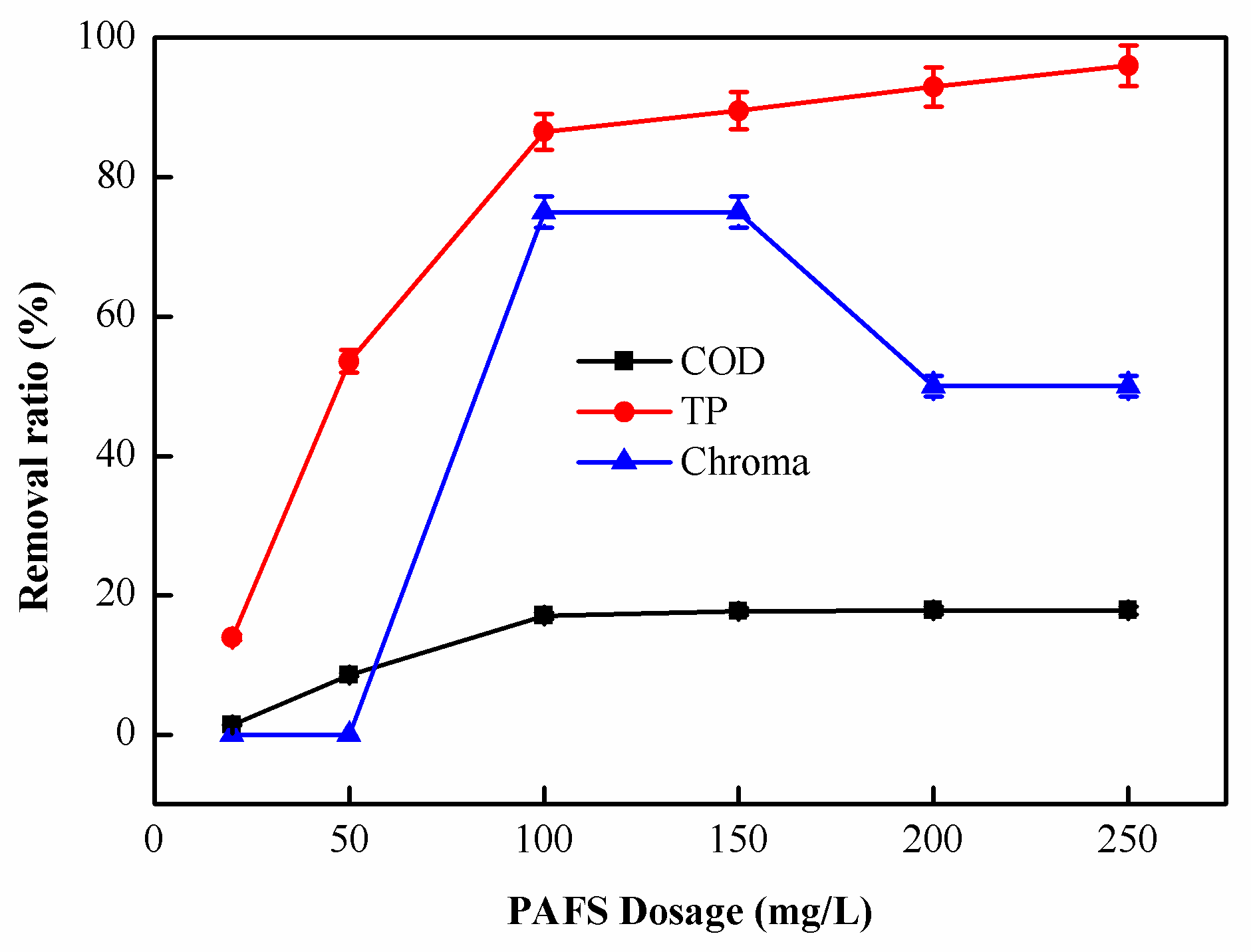
2.1.3. Effect of PAFS Dosage on Coagulation Performance
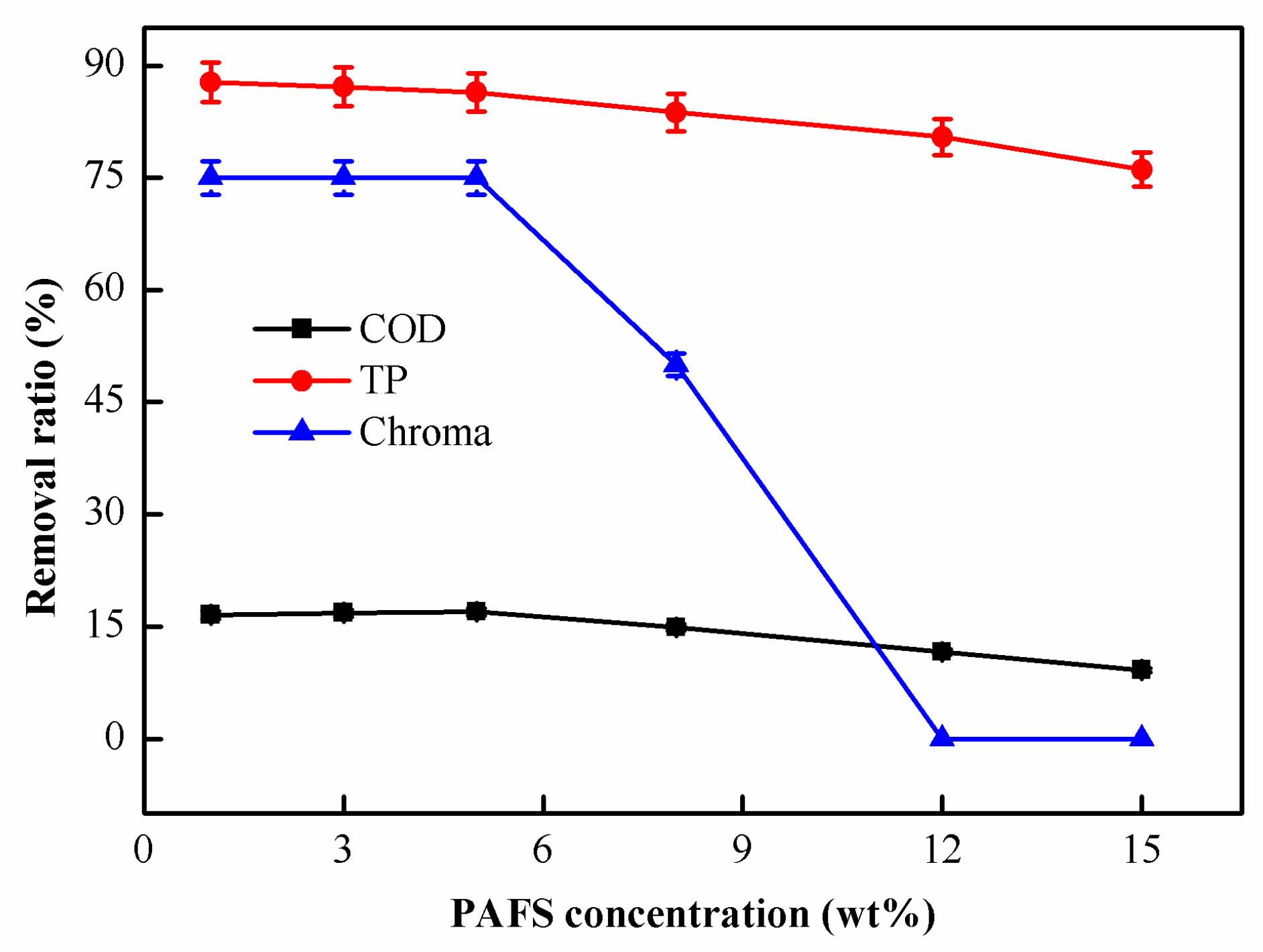
2.1.4. Effect of PAFS Concentration on Coagulation Performance
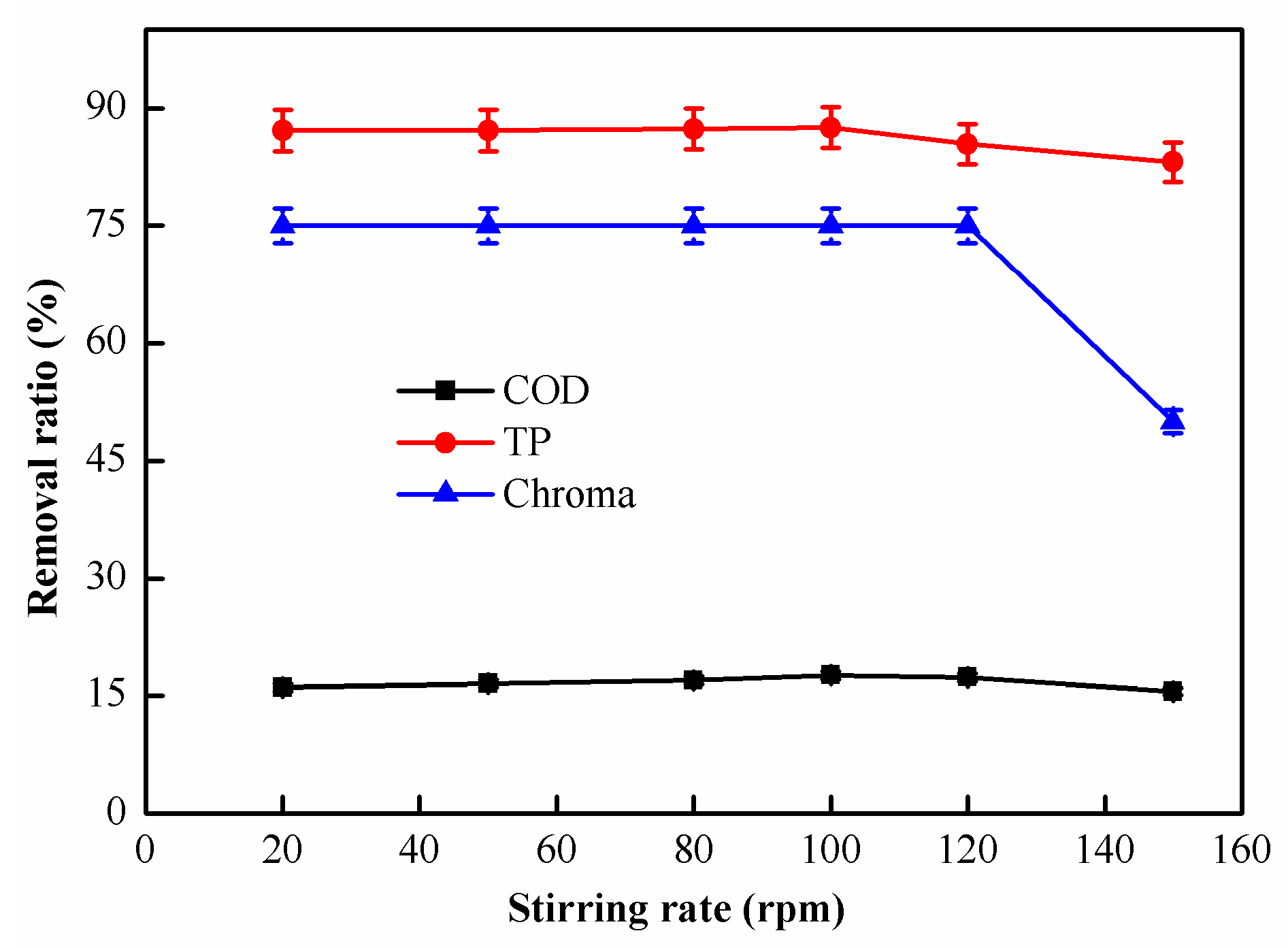
2.1.5. Effect of Stirring Speed on Coagulation Performance
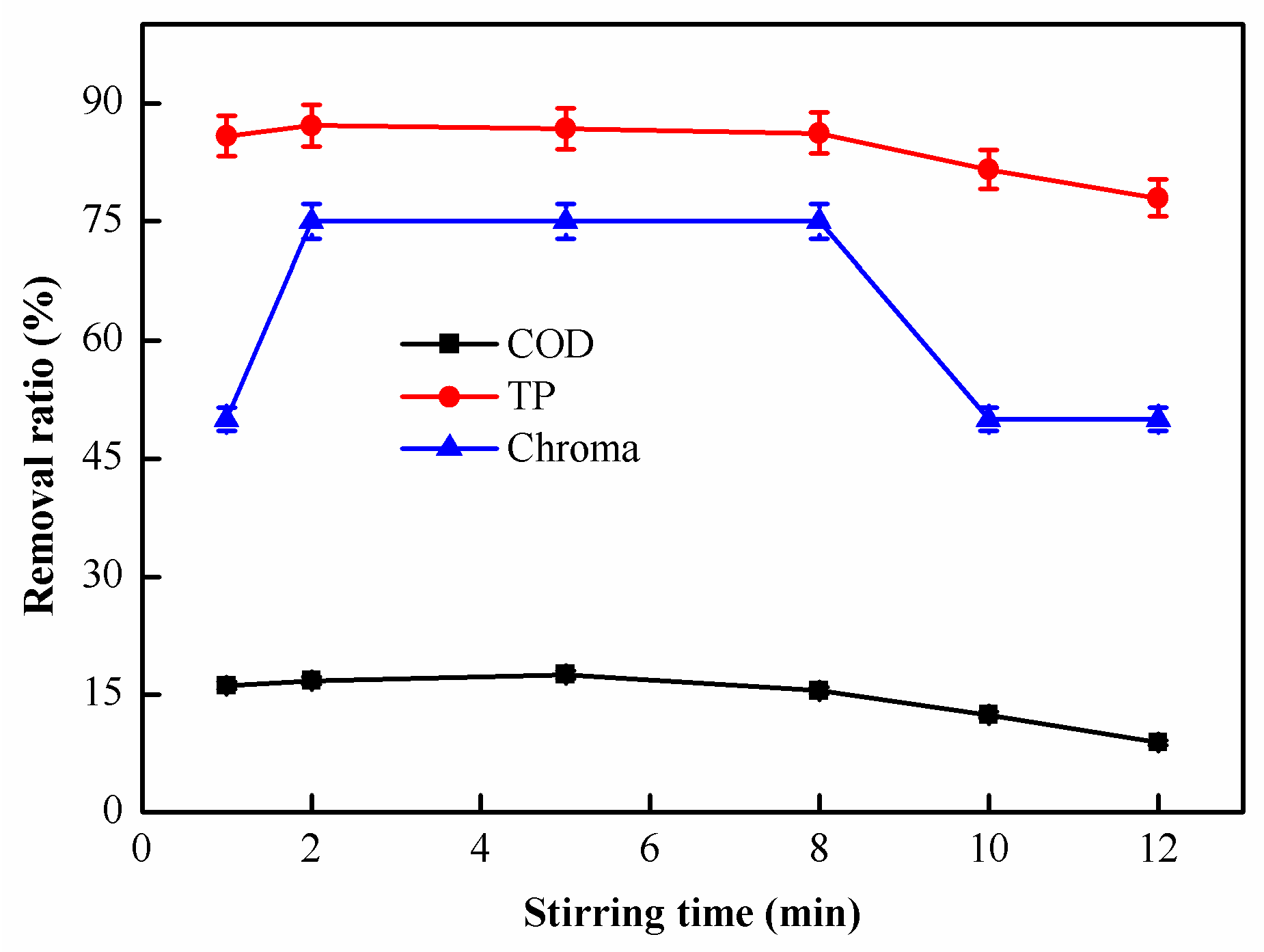
2.1.6. Effect of Stirring Time on Coagulation Performance
2.2. Ozone Catalytic Oxidation Treatment of Biochemical Tailwater from Phosphorus-Containing Pesticide Wastewater
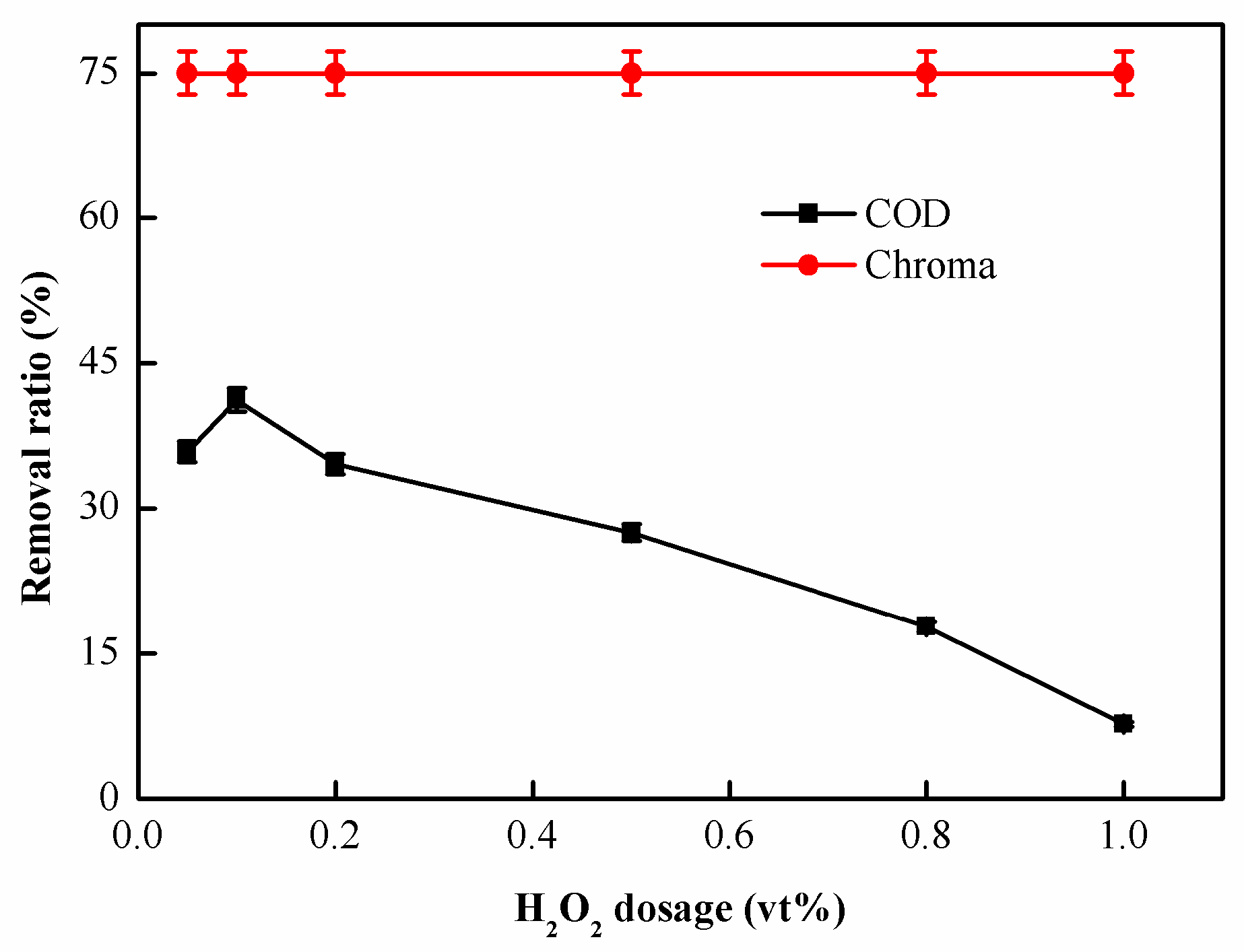
2.2.1. Effect of H2O2 Dosage on Catalytic Oxidation Performance
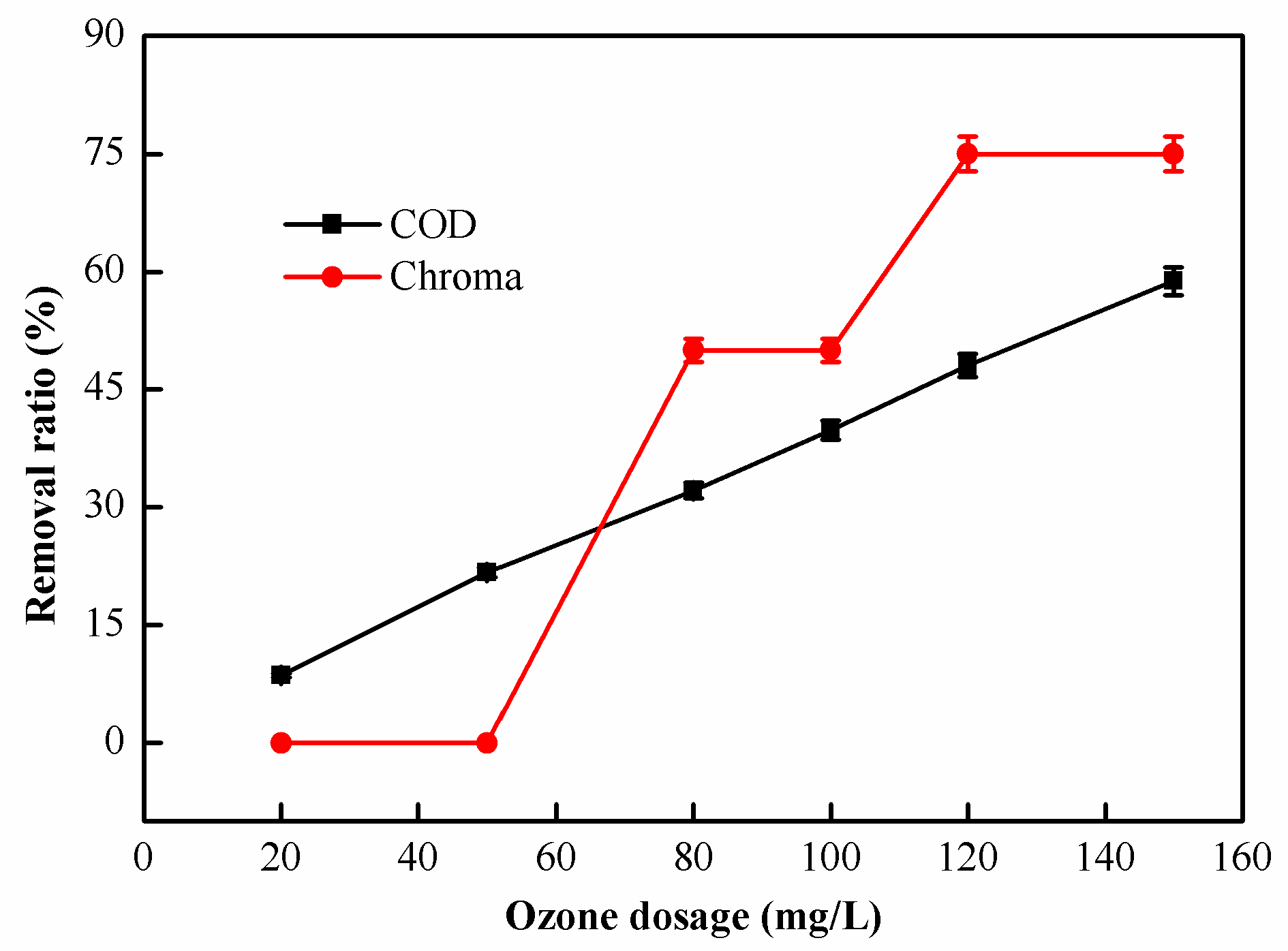
2.2.2. Effect of O3 Dosage on Catalytic Oxidation Performance
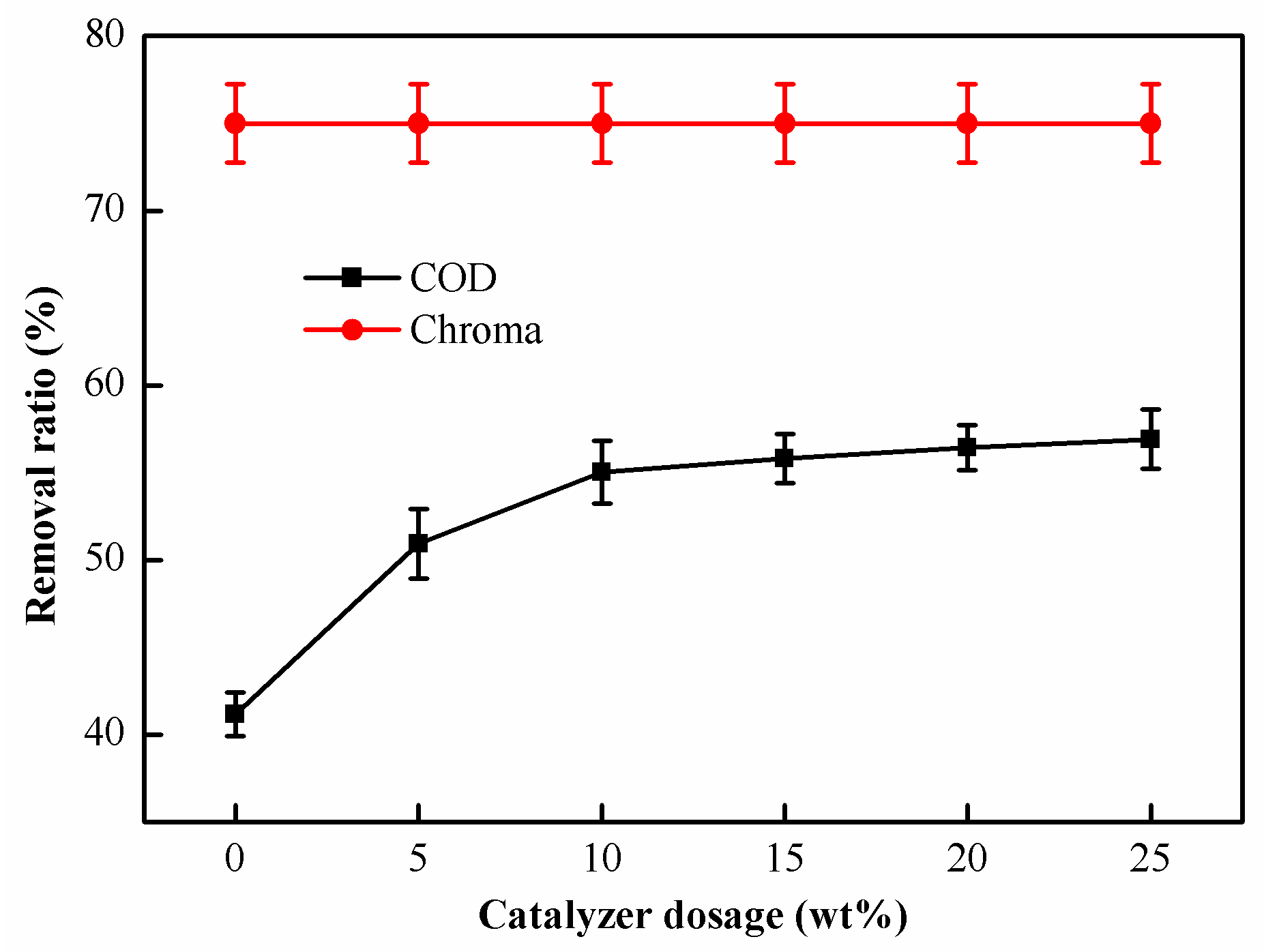
2.2.3. Effect of Catalyst Dosage on Catalytic Oxidation Performance
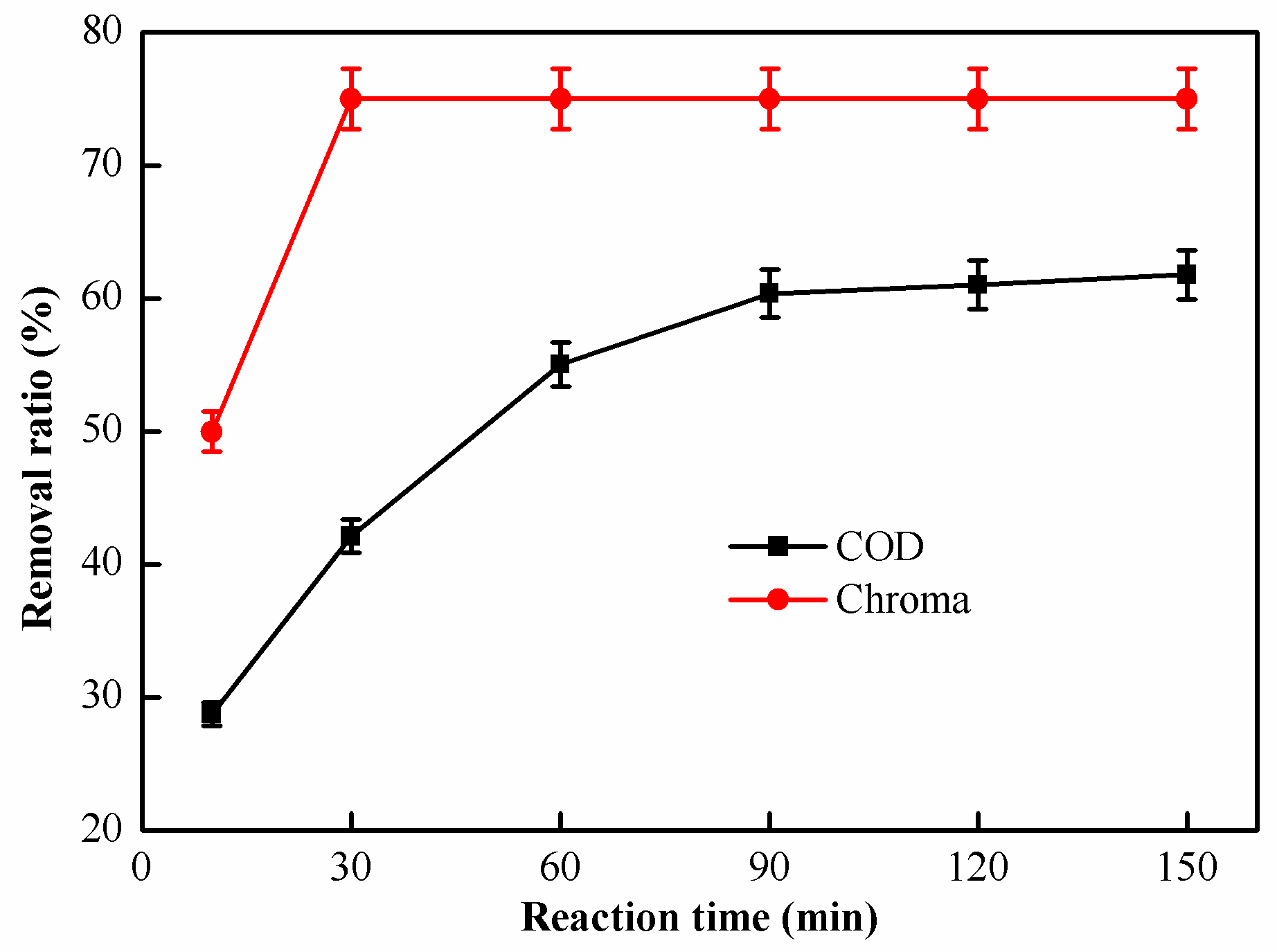
2.2.4. Effect of Reaction Time on Catalytic Oxidation Performance
3. Materials and Methods
3.1. Materials
3.2. Coagulation and Catalytic Oxidation Experiment
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Altinok, I.; Capkin, E.; Karahan, S.; Boran, M. Effects of water quality and fish size on toxicity of methiocarb, a carbamate pesticide, to rainbow trout. Environ. Toxicol. Pharmacol. 2006, 22, 20–26. [Google Scholar] [CrossRef]
- Fischer, A.; Wezel, A.P.; Hollender, J.; Cornelissen, E.; Hofman, R.; van der Hoek, J.P. Development and application of relevance and reliability criteria for water treatment removal efficiencies of chemicals of emerging concern. Water Res. 2019, 161, 274–287. [Google Scholar] [CrossRef] [PubMed]
- Asif, M.B.; Fida, Z.; Tufail, A.; van de Merwe, J.P.; Leusch, F.; Pramanik, B.K.; Price, W.E.; Hai, F.I. Persulfate oxidation-assisted membrane distillation process for micropollutant degradation and membrane fouling control. Sep. Purif. Technol. 2019, 222, 321–331. [Google Scholar] [CrossRef]
- Kanan, S.; Moyet, M.A.; Arthur, R.B.; Patterson, H.H. Recent advances on TiO2-based photocatalysts toward the degradation of pesticides and major organic pollutants from water bodies. Catal. Rev. 2020, 62, 1–65. [Google Scholar] [CrossRef]
- Barba, S.; Carvela, M.; Villasenor, J.; Rodrigo, M.A.; Canizares, P. Fixed-bed biological barrier coupled with electrokinetics for the in situ electrobioremediation of 2,4-dichlorophenoxyacetic acid polluted soil. J. Chem. Technol. Biotechnol. 2019, 94, 2684–2692. [Google Scholar] [CrossRef]
- Ul Haq, A.; Usman, M.; Muneer, M.; Maqbool, T.; Khosa, M.; Abbas, N. Performance and mechanism of removal of atrazine pesticide from aqueous media utilizing pumpkin seeds shell powder. Desalin. Water Treat. 2019, 160, 229–239. [Google Scholar] [CrossRef]
- Sun, W.; Sun, Y.; Zhu, H.; Zheng, H. Catalytic activity and evaluation of Fe-Mn@Bt for ozonizing coal chemical biochemical tail water. Sep. Purif. Technol. 2020, 239, 116524. [Google Scholar] [CrossRef]
- Sun, W.; Sun, Y.; Shah, K.J.; Zheng, H.; Ma, B. Electrochemical degradation of oxytetracycline by Ti-Sn-Sb/γ-Al2O3 three-dimensional electrodes. J. Environ. Manag. 2019, 241, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Pasupuleti, R.R.; Tsai, P.C.; Ponnusamy, V.K. A fast and sensitive analytical procedure for monitoring of synthetic pyrethroid pesticides’ metabolites in environmental water samples. Microchem. J. 2019, 148, 355–363. [Google Scholar] [CrossRef]
- Ramya, K.; Vasudevan, N. Performance evaluation of ETP from pesticide manufacturing industry by using WWQI and multivariate statistical analysis. Environ. Sci. Pollut. Res. 2019, 26, 20595–20609. [Google Scholar] [CrossRef]
- Ramon, F.; Lull, C. Legal measures to prevent and manage soil contamination and to increase food safety for consumer health: The case of Spain. Environ. Pollut. 2019, 250, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Edwards, Q.A.; Sultana, T.; Kulikov, S.M.; Garner-O’Neale, L.D.; Metcalfe, C.D. Micropollutants related to human activity in groundwater resources in Barbados, West Indies. Sci. Total Environ. 2019, 671, 76–82. [Google Scholar] [CrossRef]
- Aydin, S.; Aydin, M.E.; Beduk, F.; Ulvi, A. Removal of antibiotics from aqueous solution by using magnetic Fe3O4/red mud-nanoparticles. Sci. Total Environ. 2019, 670, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Yasar, A.; Ghaffar, A.; Mahfooz, Y.; Tabinda, A.B.; Mehmood, A. Comparative performance evaluation of ozone oxidation and coagulation for the treatment of electroplating wastewater. Desalin. Water Treat. 2021, 230, 268–275. [Google Scholar] [CrossRef]
- Zheng, H.; Sun, Y.; Zhu, C.; Guo, J.; Zhao, C.; Liao, Y.; Guan, Q. UV-initiated polymerization of hydrophobically associating cationic flocculants: Synthesis, characterization, and dewatering properties. Chem. Eng. J. 2013, 234, 318–326. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, L.; Jin, X.; Liu, L.; Zhang, Y.; Ni, Q.; Yao, J. Synthesis of hydrophobically modified cellulose-based flocculant and its application in treatments of kaolin suspension and machining wastewater. Cellulose 2017, 24, 5639–5647. [Google Scholar] [CrossRef]
- Zhang, F.; Wu, K.; Zhou, H.; Hu, Y.; Sergei, P.; Wu, H.; Wei, C. Ozonation of aqueous phenol catalyzed by biochar produced from sludge obtained in the treatment of coking wastewater. J. Environ. Manag. 2018, 224, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Ren, M.; Zhu, C.; Xu, Y.; Zheng, H.; Xiao, X.; Wu, H.; Xia, T.; You, Z. UV-Initiated graft copolymerization of cationic Chitosan-Based flocculants for treatment of zinc Phosphate-Contaminated wastewater. Ind. Eng. Chem. Res. 2016, 55, 10025–10035. [Google Scholar] [CrossRef]
- Sun, Y.; Li, P.; Zheng, H.; Zhao, C.; Xiao, X.; Xu, Y.; Sun, W.; Wu, H.; Ren, M. Electrochemical treatment of chloramphenicol using Ti-Sn/γ-Al2O3 particle electrodes with a three-dimensional reactor. Chem. Eng. J. 2017, 308, 1233–1242. [Google Scholar] [CrossRef]
- Chen, W.; Zheng, H.; Zhai, J.; Wang, Y.; Xue, W.; Tang, X.; Zhang, Z.; Sun, Y. Characterization and coagulation-flocculation performance of a composite coagulant: Poly-ferric-aluminum-silicate-sulfate. Desalin. Water Treat. 2015, 56, 1776–1786. [Google Scholar] [CrossRef]
- Sun, Y.; Zhu, C.; Zheng, H.; Sun, W.; Xu, Y.; Xiao, X.; You, Z.; Liu, C. Characterization and coagulation behavior of polymeric aluminum ferric silicate for high-concentration oily wastewater treatment. Chem. Eng. Res. Des. 2017, 119, 23–32. [Google Scholar] [CrossRef]
- Sun, Y.; Zhu, C.; Sun, W.; Xu, Y.; Xiao, X.; Zheng, H.; Wu, H.; Liu, C. Plasma-initiated polymerization of chitosan-based CS-g-P(AM-DMDAAC) flocculant for the enhanced flocculation of low-algal-turbidity water. Carbohydr. Polym. 2017, 164, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Danesh, S.; Oleszkiewicz, J.A. Volatile fatty acid production and uptake in biological nutrient removal systems with process separation. Water Environ. Res. 1997, 69, 1106–1111. [Google Scholar] [CrossRef]
- Piazza, G.J.; Lora, J.H.; Wayman, L.I.; Garcia, R.A. Removal of lignin from straw spent pulping liquor using synthetic cationic and biobased flocculants. Sep. Purif. Technol. 2017, 188, 348–357. [Google Scholar] [CrossRef]
- Konduri, M.K.R.; Fatehi, P. Influence of pH and ionic strength on flocculation of clay suspensions with cationic xylan copolymer. Colloids Surf. A 2017, 530, 20–32. [Google Scholar] [CrossRef]
- Lu, X.; Xu, Y.; Sun, W.; Sun, Y.; Zheng, H. UV-initiated synthesis of a novel chitosan-based flocculant with high flocculation efficiency for algal removal. Sci. Total Environ. 2017, 609, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z. The flocculation mechanism and treatment of oily wastewater by flocculation. Water Sci. Technol. 2017, 76, 2630–2637. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Ma, X.; Li, X.; Fan, J.; Chen, Q.; Liu, X.; Pan, J. Preparation of a cationic polyacrylamide (CPAM) and its flocculation performance in the environmental estrogen removal and separation. Desalin. Water Treat. 2017, 100, 231–242. [Google Scholar] [CrossRef]
- You, Z.; Zhuang, C.; Sun, Y.; Zhang, S.; Zheng, H. Efficient removal of TiO2 nanoparticles by enhanced flocculation–coagulation. Ind. Eng. Chem. Res. 2019, 58, 14528–14537. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, H.; Sun, Y.; Chiang, P.; Sun, W.; Xu, Y.; Zheng, H.; Shah, K.J. Characterization and sludge dewatering performance evaluation of the photo-initiated cationic flocculant PDD. J. Taiwan Inst. Chem. Eng. 2018, 93, 253–262. [Google Scholar] [CrossRef]
- Zhang, Z.; Nong, H.; Zhao, L.; Zhang, H. A particle size distribution analysis method to quantitatively evaluate coagulation of fine particles in aqueous solutions. Part. Sci. Technol. 2018, 36, 771–777. [Google Scholar] [CrossRef]
- Chen, L.; Sun, Y.; Sun, W.; Shah, K.J.; Xu, Y.; Zheng, H. Efficient cationic flocculant MHCS-g-P(AM-DAC) synthesized by UV-induced polymerization for algae removal. Sep. Purif. Technol. 2019, 210, 10–19. [Google Scholar] [CrossRef]
- Sun, Y.; Shah, K.J.; Sun, W.; Zheng, H. Performance evaluation of chitosan-based flocculants with good pH resistance and high heavy metals removal capacity. Sep. Purif. Technol. 2019, 215, 208–216. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, A.; Pan, S.; Sun, W.; Zhu, C.; Shah, K.J.; Zheng, H. Novel chitosan-based flocculants for chromium and nickle removal in wastewater via integrated chelation and flocculation. J. Environ. Manag. 2019, 248, 109241. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wang, J.; Xu, S.; Lin, K.A.; Tong, S. Oxygen vacancy of CeO2 improved efficiency of H2O2/O3 for the degradation of acetic acid in acidic solutions. Sep. Purif. Technol. 2018, 207, 92–98. [Google Scholar] [CrossRef]
- Ahmadi, M.; Rahmani, H.; Takdastan, A.; Jaafarzadeh, N.; Mostoufi, A. A novel catalytic process for degradation of bisphenol a from aqueous solutions: A synergistic effect of nano-Fe3O4@Alg-Fe on O3/H2O2. Process Saf. Environ. 2016, 104, 413–421. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, S.; Sun, W.; Zhu, S.; Zheng, H. Flocculation activity and evaluation of chitosan-based flocculant CMCTS-g-P(AM-CA) for heavy metal removal. Sep. Purif. Technol. 2020, 241, 116737. [Google Scholar] [CrossRef]
- Peng, R.; Shen, T.; Tong, S.; Ma, C. An effective heterogeneous Ozone-Based advanced oxidation process in acidic solution—Ti-MCM-41/H2O2/O3. Ozone-Sci. Eng. 2016, 38, 194–202. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, W.; Shah, K.J.; Chiang, P.; Zheng, H. Characterization and flocculation evaluation of a novel carboxylated chitosan modified flocculant by UV initiated polymerization. Carbohydr. Polym. 2019, 208, 213–220. [Google Scholar] [CrossRef]
- Liang, J.; Ning, X.; Sun, J.; Song, J.; Hong, Y.; Cai, H. An integrated permanganate and ozone process for the treatment of textile dyeing wastewater: Efficiency and mechanism. J. Clean. Prod. 2018, 204, 12–19. [Google Scholar] [CrossRef]
- Liu, C.; Chen, X.; Zhang, J.; Zhou, H.; Zhang, L.; Guo, Y. Advanced treatment of bio-treated coal chemical wastewater by a novel combination of microbubble catalytic ozonation and biological process. Sep. Purif. Technol. 2018, 197, 295–301. [Google Scholar] [CrossRef]
- Sun, W.; Zhou, S.; Sun, Y.; Xu, Y. Synthesis and evaluation of cationic flocculant P(DAC-PAPTAC-AM) for flocculation of coal chemical wastewater. J. Environ. Sci.-China 2021, 99, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Lumeau, J.; Glebova, L.; Glebov, L.B. Absorption and scattering in photo-thermo-refractive glass induced by UV-exposure and thermal development. Opt. Mater. 2014, 36, 621–627. [Google Scholar] [CrossRef] [Green Version]
- Raeisivand, S.; Sadeghi, M.; Hemati, S.; Fadaei, A.; Sedehi, M.; Sadeghi, A.; Hoseini, S.M. Photocatalytic degradation of catechol in aqueous solutions: A comparison between UV/Fe2O3 and Fe2O3/sunlight processes. Desalin. Water Treat. 2019, 154, 340–346. [Google Scholar] [CrossRef]
- Li, X.; Chen, W.; Ma, L.; Huang, Y.; Wang, H. Characteristics and mechanisms of catalytic ozonation with Fe-shaving-based catalyst in industrial wastewater advanced treatment. J. Clean. Prod. 2019, 222, 174–181. [Google Scholar] [CrossRef]
| Index | Characteristic | pH | COD (mg/L) | TP (mg/L) | Chroma (time) |
|---|---|---|---|---|---|
| Value | Light yellow, transparent, and no pungent odor | 8.1 | 78.6 | 5.2 | 32.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, S.; Chen, L.; Zhao, M.; Liu, A.; Wang, M.; Sun, Y. Advanced Treatment of Phosphorus Pesticide Wastewater Using an Integrated Process of Coagulation and Ozone Catalytic Oxidation. Catalysts 2022, 12, 103. https://doi.org/10.3390/catal12010103
Cao S, Chen L, Zhao M, Liu A, Wang M, Sun Y. Advanced Treatment of Phosphorus Pesticide Wastewater Using an Integrated Process of Coagulation and Ozone Catalytic Oxidation. Catalysts. 2022; 12(1):103. https://doi.org/10.3390/catal12010103
Chicago/Turabian StyleCao, Shengping, Lei Chen, Minyan Zhao, Ankang Liu, Mingxiu Wang, and Yongjun Sun. 2022. "Advanced Treatment of Phosphorus Pesticide Wastewater Using an Integrated Process of Coagulation and Ozone Catalytic Oxidation" Catalysts 12, no. 1: 103. https://doi.org/10.3390/catal12010103
APA StyleCao, S., Chen, L., Zhao, M., Liu, A., Wang, M., & Sun, Y. (2022). Advanced Treatment of Phosphorus Pesticide Wastewater Using an Integrated Process of Coagulation and Ozone Catalytic Oxidation. Catalysts, 12(1), 103. https://doi.org/10.3390/catal12010103







