Waste Animal Bones as Catalysts for Biodiesel Production; A Mini Review
Abstract
1. Introduction
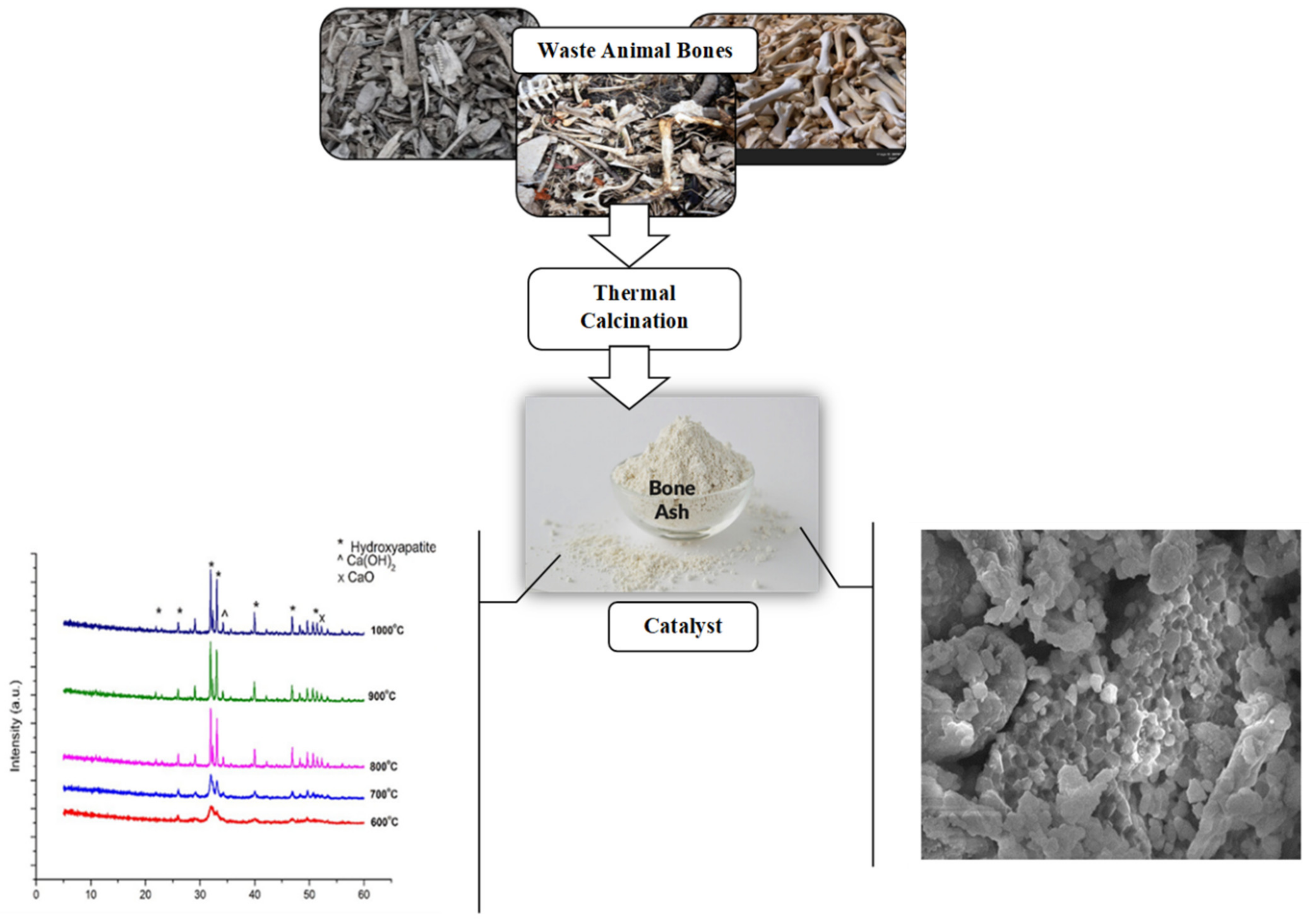
2. Sources of Bone Waste
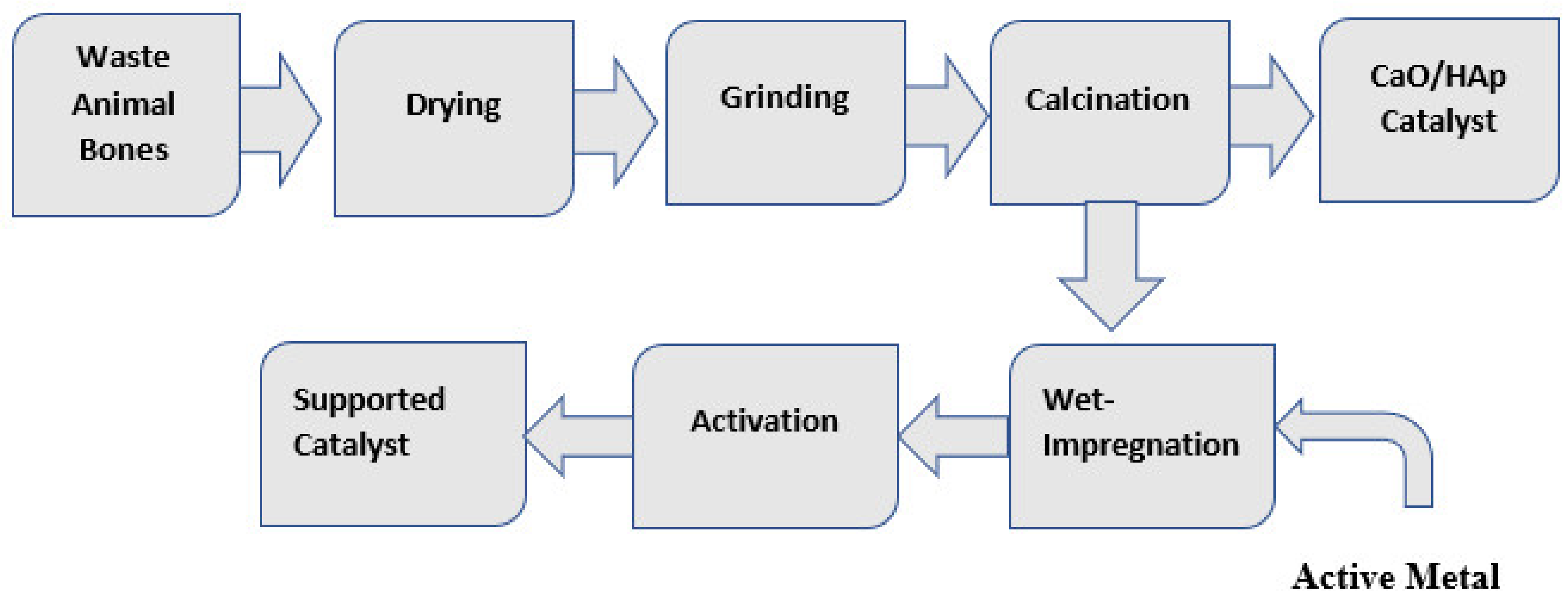
3. Catalyst Preparation
3.1. Effect of Calcination Temperature on Bones Derived Catalysts
3.2. Effect of Calcination Temperature on Surface Area of Bones Derived Catalysts
3.3. Effect of Calcination Temperature on Total Basicity of Bones Derived Catalysts
4. Diverse Animal Sourced Bone-Based Catalysts and Their Biodiesel Yields
4.1. Reaction Parameters Study
4.2. Elemental Composition of Waste Animal Bones
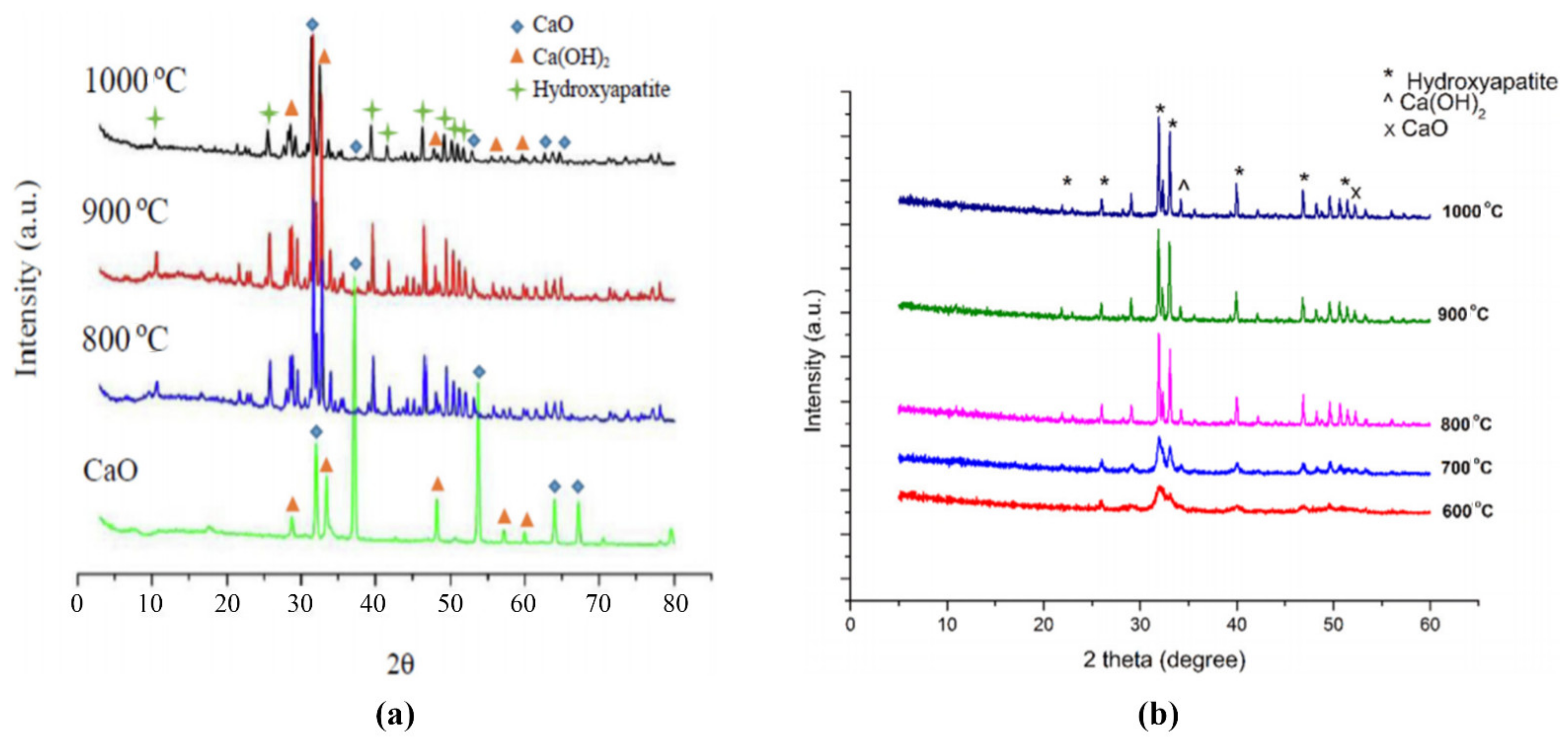
4.3. XRD Analysis of Bones Derived Catalysts
4.4. Reaction Mechanism
4.5. Bones Derived Catalyst Reusability
4.6. Heterogeneously Catalyzed Technology in Biodiesel Production
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wan, L.; Liu, H.; Skala, D. Biodiesel production from soybean oil in subcritical methanol using MnCO3/ZnO as catalyst. Appl. Catal. B Environ. 2014, 152, 352–359. [Google Scholar] [CrossRef]
- Soudagar, M.E.M.; Mujtaba, M.A.; Safaei, M.R.; Afzal, A.; Ahmed, W.; Banapurmath, N.R.; Hossain, N.; Bashir, S.; Badruddin, I.A.; Goodarzi, M.; et al. Effect of Sr@ZnO nanoparticles and Ricinus communis biodiesel-diesel fuel blends on modified CRDI diesel engine characteristics. Energy 2021, 215, 119094. [Google Scholar] [CrossRef]
- Khan, H.M.; Ali, C.H.; Iqbal, T.; Yasin, S.; Sulaiman, M.; Mahmood, H.; Raashid, M.; Pasha, M.; Mu, B. Current scenario and potential of biodiesel production from waste cooking oil in Pakistan: An overview. Chin. J. Chem. Eng. 2019, 27, 2238–2250. [Google Scholar] [CrossRef]
- Mujtaba, M.; Cho, H.M.; Masjuki, H.; Kalam, M.; Ong, H.; Gul, M.; Harith, M.; Yusoff, M. Critical review on sesame seed oil and its methyl ester on cold flow and oxidation stability. Energy Rep. 2020, 6, 40–54. [Google Scholar] [CrossRef]
- Shan, R.; Chen, G.; Yan, B.; Shi, J.; Liu, C. Porous CaO-based catalyst derived from PSS-induced mineralization for biodiesel production enhancement. Energy Convers. Manag. 2015, 106, 405–413. [Google Scholar] [CrossRef]
- Su, F.; Guo, Y. Advancements in solid acid catalysts for biodiesel production. Green Chem. 2014, 16, 2934–2957. [Google Scholar] [CrossRef]
- Luque, R.; Lovett, J.C.; Datta, B.; Clancy, J.; Campelo, J.M.; Romero, A.A. Biodiesel as feasible petrol fuel replacement: A multidisciplinary overview. Energy Environ. Sci. 2010, 3, 1706–1721. [Google Scholar] [CrossRef]
- Kumar, M.; Sharma, M.P. Selection of potential oils for biodiesel production. Renew. Sustain. Energy Rev. 2016, 56, 1129–1138. [Google Scholar] [CrossRef]
- Semwal, S.; Arora, A.K.; Badoni, R.P.; Tuli, D.K. Biodiesel production using heterogeneous catalysts. Bioresour. Technol. 2011, 102, 2151–2161. [Google Scholar] [CrossRef]
- Mujtaba, M.; Masjuki, H.; Kalam, M.; Ong, H.C.; Gul, M.; Farooq, M.; Soudagar, M.E.M.; Ahmed, W.; Harith, M.; Yusoff, M. Ultrasound-assisted process optimization and tribological characteristics of biodiesel from palm-sesame oil via response surface methodology and extreme learning machine-Cuckoo search. Renew. Energy 2020, 158, 202–214. [Google Scholar] [CrossRef]
- Amani, H.; Ahmad, Z.; Hameed, B. Highly active alumina-supported Cs-Zr mixed oxide catalysts for low-temperature transesterification of waste cooking oil. Appl. Catal. A Gen. 2014, 487, 16–25. [Google Scholar] [CrossRef]
- Lee, A.F.; Bennett, J.A.; Manayil, J.C.; Wilson, K. Heterogeneous catalysis for sustainable biodiesel production via esterification and transesterification. Chem. Soc. Rev. 2014, 43, 7887–7916. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, Z.; Xu, Y.; Liu, Q.; Qian, G. CaFeAl mixed oxide derived heterogeneous catalysts for transesterification of soybean oil to biodiesel. Bioresour. Technol. 2015, 190, 438–441. [Google Scholar] [CrossRef]
- Paterson, G.; Issariyakul, T.; Baroi, C.; Bassi, A.; Dalai, A. Ion-exchange resins as catalysts in transesterification of triolein. Catal. Today 2013, 212, 157–163. [Google Scholar] [CrossRef]
- Nisar, J.; Razaq, R.; Farooq, M.; Iqbal, M.; Khan, R.A.; Sayed, M.; Shah, A.; ur Rahman, I. Enhanced biodiesel production from Jatropha oil using calcined waste animal bones as catalyst. Renew. Energy 2017, 101, 111–119. [Google Scholar] [CrossRef]
- Bennett, J.A.; Wilson, K.; Lee, A.F. Catalytic applications of waste derived materials. J. Mater. Chem. A 2016, 4, 3617–3637. [Google Scholar] [CrossRef]
- Balakrishnan, M.; Batra, V.; Hargreaves, J.; Pulford, I. Waste materials-Catalytic opportunities: An overview of the application of large scale waste materials as resources for catalytic applications. Green Chem. 2011, 13, 16–24. [Google Scholar] [CrossRef]
- Corro, G.; Sánchez, N.; Pal, U.; Bañuelos, F. Biodiesel production from waste frying oil using waste animal bone and solar heat. Waste Manag. 2016, 47, 105–113. [Google Scholar] [CrossRef]
- Lohri, C.R.; Camenzind, E.J.; Zurbrügg, C. Financial sustainability in municipal solid waste management—Costs and revenues in Bahir Dar, Ethiopia. Waste Manag. 2014, 34, 542–552. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Xu, C.; Li, B. Application of waste eggshell as low-cost solid catalyst for biodiesel production. Bioresour. Technol. 2009, 100, 2883–2885. [Google Scholar] [CrossRef]
- Gorji, A. Animal renewable waste resource as catalyst in biodiesel production. J. Biodivers. Environ. Sci. 2015, 7, 36–49. [Google Scholar]
- Boro, J.; Deka, D.; Thakur, A.J. A review on solid oxide derived from waste shells as catalyst for biodiesel production. Renew. Sustain. Energy Rev. 2012, 16, 904–910. [Google Scholar] [CrossRef]
- Lee, A.F.; Wilson, K. Recent developments in heterogeneous catalysis for the sustainable production of biodiesel. Catal. Today 2015, 242, 3–18. [Google Scholar] [CrossRef]
- Piccirillo, C.; Silva, M.; Pullar, R.; Da Cruz, I.B.; Jorge, R.; Pintado, M.; Castro, P.M. Extraction and characterisation of apatite-and tricalcium phosphate-based materials from cod fish bones. Mater. Sci. Eng. C 2013, 33, 103–110. [Google Scholar] [CrossRef]
- Shan, R.; Shi, J.; Yan, B.; Chen, G.; Yao, J.; Liu, C. Transesterification of palm oil to fatty acids methyl ester using K2CO3/palygorskite catalyst. Energy Convers. Manag. 2016, 116, 142–149. [Google Scholar] [CrossRef]
- Taleb, M.A.; Mamouni, R.; Saffaj, N.; Mouna, A.; Taha, M.; Benlhachemi, A.; Bakiz, B.; Ezahri, M.; Villain, S. Animal Bone Meal as new recyclable and ecological catalyst for the oximes Synthesis in solvent-Free Conditions. J. Mater. Environ. Sci. 2016, 7, 4580–4588. [Google Scholar]
- Haider, A.; Haider, S.; Han, S.S.; Kang, I.-K. Recent advances in the synthesis, functionalization and biomedical applications of hydroxyapatite: A review. RSC Adv. 2017, 7, 7442–7458. [Google Scholar] [CrossRef]
- Karmakar, B.; Halder, G. Progress and future of biodiesel synthesis: Advancements in oil extraction and conversion technologies. Energy Convers. Manag. 2019, 182, 307–339. [Google Scholar] [CrossRef]
- Gharby, S.; Harhar, H.; Bouzoubaa, Z.; Asdadi, A.; El Yadini, A.; Charrouf, Z. Chemical characterization and oxidative stability of seeds and oil of sesame grown in Morocco. J. Saudi Soc. Agric. Sci. 2017, 16, 105–111. [Google Scholar] [CrossRef]
- Perego, C.; Villa, P. Catalyst preparation methods. Catal. Today 1997, 34, 281–305. [Google Scholar] [CrossRef]
- Crucianelli, M.; Bizzarri, B.M.; Saladino, R. SBA-15 Anchored Metal Containing Catalysts in the Oxidative Desulfurization Process. Catalysts 2019, 9, 984. [Google Scholar] [CrossRef]
- Meschini, R.; D’Eliseo, D.; Filippi, S.; Bertini, L.; Bizzarri, B.M.; Botta, L.; Saladino, R.; Velotti, F. Tyrosinase-treated hydroxytyrosol-enriched olive vegetation waste with increased antioxidant activity promotes autophagy and inhibits the inflammatory response in human THP-1 monocytes. J. Agric. Food. Chem. 2018, 66, 12274–12284. [Google Scholar] [CrossRef]
- Abdullah, S.H.Y.S.; Hanapi, N.sH.M.; Azid, A.; Umar, R.; Juahir, H.; Khatoon, H.; Endut, A. A review of biomass-derived heterogeneous catalyst for a sustainable biodiesel production. Renew. Sustain. Energy Rev. 2017, 70, 1040–1051. [Google Scholar] [CrossRef]
- Obadiah, A.; Swaroopa, G.A.; Kumar, S.V.; Jeganathan, K.R.; Ramasubbu, A. Biodiesel production from palm oil using calcined waste animal bone as catalyst. Bioresour. Technol. 2012, 116, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Oopathum, C.; Weeramongkhonlert, V.; Smith, C.B.; Chaveanghong, S.; Ketwong, P.; Boonyuen, S. Transesterification of soybean oil using bovine bone waste as new catalyst. Bioresour. Technol. 2013, 143, 686–690. [Google Scholar] [CrossRef]
- Roschat, W.; Kacha, M.; Yoosuk, B.; Sudyoadsuk, T.; Promarak, V. Biodiesel production based on heterogeneous process catalyzed by solid waste coral fragment. Fuel 2012, 98, 194–202. [Google Scholar] [CrossRef]
- Farooq, M.; Ramli, A.; Naeem, A. Biodiesel production from low FFA waste cooking oil using heterogeneous catalyst derived from chicken bones. Renew. Energy 2015, 76, 362–368. [Google Scholar] [CrossRef]
- AlSharifi, M.; Znad, H. Development of a lithium based chicken bone (Li-Cb) composite as an efficient catalyst for biodiesel production. Renew. Energy 2019, 136, 856–864. [Google Scholar] [CrossRef]
- Thitsartarn, W.; Kawi, S. An active and stable CaO–CeO 2 catalyst for transesterification of oil to biodiesel. Green Chem. 2011, 13, 3423–3430. [Google Scholar] [CrossRef]
- Chen, G.; Shan, R.; Shi, J.; Liu, C.; Yan, B. Biodiesel production from palm oil using active and stable K doped hydroxyapatite catalysts. Energy Convers. Manag. 2015, 98, 463–469. [Google Scholar] [CrossRef]
- Ghanei, R.; Dermani, R.K.; Salehi, Y.; Mohammadi, M. Waste animal bone as support for CaO impregnation in catalytic biodiesel production from vegetable oil. Waste Biomass Valorization 2016, 7, 527–532. [Google Scholar] [CrossRef]
- Buasri, A.; Inkaew, T.; Kodephun, L.; Yenying, W.; Loryuenyong, V. Natural hydroxyapatite (NHAp) derived from pork bone as a renewable catalyst for biodiesel production via microwave irradiation. In Key Engineering Materials; Trans Tech Publications Ltd: Stafa-Zurich, Switzerland, 2015; pp. 216–220. [Google Scholar]
- Ansari Samani, M.; Hosseinzdeh Samani, B.; Lotfalian, A.; Rostami, S.; Najafi, G.; Fayyazi, E.; Mamat, R. The feasibility and optimization of biodiesel production from Celtis australis L. oil using chicken bone catalyst and ultrasonic waves. Biofuels 2020, 11, 513–521. [Google Scholar] [CrossRef]
- Tan, Y.H.; Abdullah, M.O.; Kansedo, J.; Mubarak, N.M.; San Chan, Y.; Nolasco-Hipolito, C. Biodiesel production from used cooking oil using green solid catalyst derived from calcined fusion waste chicken and fish bones. Renew. Energy 2019, 139, 696–706. [Google Scholar] [CrossRef]
- Chingakham, C.; Tiwary, C.; Sajith, V. Waste animal bone as a novel layered heterogeneous catalyst for the transesterification of biodiesel. Catal. Lett. 2019, 149, 1100–1110. [Google Scholar] [CrossRef]
- Madhu, D.; Singh, B.; Sharma, Y.C. Studies on application of fish waste for synthesis of high quality biodiesel. RSC Adv. 2014, 4, 31462–31468. [Google Scholar] [CrossRef]
- Chakraborty, R.; Bepari, S.; Banerjee, A. Application of calcined waste fish (Labeo rohita) scale as low-cost heterogeneous catalyst for biodiesel synthesis. Bioresour. Technol. 2011, 102, 3610–3618. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, R.; RoyChowdhury, D. Fish bone derived natural hydroxyapatite-supported copper acid catalyst: Taguchi optimization of semibatch oleic acid esterification. Chem. Eng. J. 2013, 215, 491–499. [Google Scholar] [CrossRef]
- Ali, C.H.; Asif, A.H.; Iqbal, T.; Qureshi, A.S.; Kazmi, M.A.; Yasin, S.; Danish, M.; Mu, B.-Z. Improved transesterification of waste cooking oil into biodiesel using calcined goat bone as a catalyst. Energy Sources Part A Recovery Util. Environ. Eff. 2018, 40, 1076–1083. [Google Scholar] [CrossRef]
- Khan, H.M.; Iqbal, T.; Ali, C.H.; Javaid, A.; Cheema, I.I. Sustainable biodiesel production from waste cooking oil utilizing waste ostrich (Struthio camelus) bones derived heterogeneous catalyst. Fuel 2020, 277, 118091. [Google Scholar] [CrossRef]
- Jazie, A.A.; Pramanik, H.; Sinha, A. Transesterification of peanut and rapeseed oils using waste of animal bone as cost effective catalyst. Mater. Renew. Sustain. Energy 2013, 2, 1–10. [Google Scholar] [CrossRef][Green Version]
- Rahman, M. Valorization of harmful algae E. compressa for biodiesel production in presence of chicken waste derived catalyst. Renew. Energy 2018, 129, 132–140. [Google Scholar] [CrossRef]
- Volli, V.; Purkait, M.K.; Shu, C.-M. Preparation and characterization of animal bone powder impregnated fly ash catalyst for transesterification. Sci. Total Environ. 2019, 669, 314–321. [Google Scholar] [CrossRef]
- AlSharifi, M.; Znad, H. Transesterification of waste canola oil by lithium/zinc composite supported on waste chicken bone as an effective catalyst. Renew. Energy 2020, 151, 740–749. [Google Scholar] [CrossRef]
- Chinglenthoiba, C.; Das, A.; Vandana, S. Enhanced biodiesel production from waste cooking palm oil, with NaOH-loaded Calcined fish bones as the catalyst. Environ. Sci. Pollut. Res. 2020, 27, 15925–15930. [Google Scholar] [CrossRef]
- Ayodeji, A.A.; Blessing, I.E.; Rasheed, B.; Modupe, O.E.; Ajibola, O.; Oluwabunmi, A.G.; Ojo, F.S. Production of biodiesel from soybean oil using calcium oxide and cow bone as catalysts. Mater. Focus 2018, 7, 542–548. [Google Scholar] [CrossRef]
- Singh, V.; Sharma, Y.C. Low cost guinea fowl bone derived recyclable heterogeneous catalyst for microwave assisted transesterification of Annona squamosa L. seed oil. Energy Convers. Manag. 2017, 138, 627–637. [Google Scholar] [CrossRef]
- Kowthaman, C.N.; Varadappan, A.M.S. Synthesis, characterization, and optimization of Schizochytrium biodiesel production using Na+-doped nanohydroxyapatite. Int. J. Energy Res. 2019, 43, 3182–3200. [Google Scholar] [CrossRef]
- Sulaiman, S.; Jamaludin, N.F.A.; Kabbashi, N.A. Development of CaO/PVA catalyst from fish bone for biodiesel production. Bull. Chem. React. Eng. Catal. 2019, 14, 153–157. [Google Scholar] [CrossRef]
- Chakraborty, R.; Das, S.; Bhattacharjee, S.K. Optimization of biodiesel production from Indian mustard oil by biological tri-calcium phosphate catalyst derived from turkey bone ash. Clean Technol. Environ. Policy 2015, 17, 455–463. [Google Scholar] [CrossRef]
- Satraidi, H.; Prasetyaningrum, A.; Ningrum, A.S.; Dewi, R.O.N. Development of heterogeneous catalyst from chicken bone and catalytic testing for biodiesel with simultaneous processing. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Semarang, Indonesia, 7–8 September 2018; p. 012125. [Google Scholar]
- Suwannasom, P.; Sriraksa, R.; Tansupo, P.; Ruangviriyachai, C. Optimization of biodiesel production from waste cooking oil using waste bone as a catalyst. Energy Sources Part A Recovery Util. Environ. Eff. 2016, 38, 3221–3228. [Google Scholar] [CrossRef]
- Yan, B.; Zhang, Y.; Chen, G.; Shan, R.; Ma, W.; Liu, C. The utilization of hydroxyapatite-supported CaO-CeO2 catalyst for biodiesel production. Energy Convers. Manag. 2016, 130, 156–164. [Google Scholar] [CrossRef]
- Mamo, T.T.; Mekonnen, Y.S. Microwave-Assisted Biodiesel Production from Microalgae, Scenedesmus Species, Using Goat Bone–Made Nano-catalyst. Appl. Biochem. Biotechnol. 2019, 1–16. [Google Scholar] [CrossRef]
- Anand, M.; Deepanraj, B.; Ranjitha, J.; Noor, M. Biodiesel Production from Mixed Elengi and Pongamia Oil using Calcined Waste Animal Bone as a Novel Heterogeneous Catalyst. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Chennai, India, 20–21 February 2020; p. 012063. [Google Scholar]
- Chaveanghong, S.; Smith, S.M.; Smith, C.B.; Luengnaruemitchai, A.; Boonyuen, S. Simultaneous transesterification and esterification of acidic oil feedstocks catalyzed by heterogeneous tungsten loaded bovine bone under mild conditions. Renew. Energy 2018, 126, 156–162. [Google Scholar] [CrossRef]
- Lim, B.P.; Maniam, G.P.; Hamid, S. Biodiesel from adsorbed waste oil on spent bleaching clay using CaO as a heterogeneous catalyst. Eur. J. Sci. Res. 2009, 33, 347–357. [Google Scholar]
- Mutreja, V.; Singh, S.; Ali, A. Potassium impregnated nanocrystalline mixed oxides of La and Mg as heterogeneous catalysts for transesterification. Renew. Energy 2014, 62, 226–233. [Google Scholar] [CrossRef]
- Xie, W.; Zhao, L. Heterogeneous CaO–MoO3–SBA-15 catalysts for biodiesel production from soybean oil. Energy Convers. Manag. 2014, 79, 34–42. [Google Scholar] [CrossRef]
- Thinnakorn, K.; Tscheikuna, J. Biodiesel production via transesterification of palm olein using sodium phosphate as a heterogeneous catalyst. Appl. Catal. A Gen. 2014, 476, 26–33. [Google Scholar] [CrossRef]
- Mujtaba, M.; Masjuki, H.; Kalam, M.; Noor, F.; Farooq, M.; Ong, H.C.; Gul, M.; Soudagar, M.E.M.; Bashir, S.; Rizwanul Fattah, I. Effect of Additivized Biodiesel Blends on Diesel Engine Performance, Emission, Tribological Characteristics, and Lubricant Tribology. Energies 2020, 13, 3375. [Google Scholar] [CrossRef]
- Yusoff, M.N.A.M.; Zulkifli, N.W.M.; Sukiman, N.L.; Chyuan, O.H.; Hassan, M.H.; Hasnul, M.H.; Zulkifli, M.S.A.; Abbas, M.M.; Zakaria, M.Z. Sustainability of Palm Biodiesel in Transportation: A Review on Biofuel Standard, Policy and International Collaboration Between Malaysia and Colombia. Bioenergy Res. 2020. [Google Scholar] [CrossRef]
- Razzaq, L.; Imran, S.; Anwar, Z.; Farooq, M.; Abbas, M.M.; Mehmood Khan, H.; Asif, T.; Amjad, M.; Soudagar, M.E.M.; Shaukat, N.J.E. Maximising Yield and Engine Efficiency Using Optimised Waste Cooking. Oil. Biodiesel. 2020, 13, 5941. [Google Scholar]
- Joschek, S.; Nies, B.; Krotz, R.; Göpferich, A. Chemical and physicochemical characterization of porous hydroxyapatite ceramics made of natural bone. Biomaterials 2000, 21, 1645–1658. [Google Scholar] [CrossRef]
- Haberko, K.; Bućko, M.M.; Brzezińska-Miecznik, J.; Haberko, M.; Mozgawa, W.; Panz, T.; Pyda, A.; Zarębski, J. Natural hydroxyapatite—Its behaviour during heat treatment. J. Eur. Ceram. Soc. 2006, 26, 537–542. [Google Scholar] [CrossRef]
- Zhu, Q.; Ablikim, Z.; Chen, T.; Cai, Q.; Xia, J.; Jiang, D.; Wang, S. The preparation and characterization of HA/β-TCP biphasic ceramics from fish bones. Ceram. Int. 2017, 43, 12213–12220. [Google Scholar] [CrossRef]
- Akindoyo, J.O.; Ghazali, S.; Beg, M.D.; Jeyaratnam, N. Characterization and elemental quantification of natural hydroxyapatite produced from cow bone. Chem. Eng. Technol. 2019, 42, 1805–1815. [Google Scholar] [CrossRef]
- Bee, S.-L.; Mariatti, M.; Ahmad, N.; Yahaya, B.; Hamid, Z.A. Effect of the calcination temperature on the properties of natural hydroxyapatite derived from chicken bone wastes. Mater. Today Proc. 2019, 16, 1876–1885. [Google Scholar] [CrossRef]
- Ayodeji, A.A.; Blessing, I.E.; Sunday, F.O. Data on calcium oxide and cow bone catalysts used for soybean biodiesel production. Data Brief 2018, 18, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; He, H.; Wang, Y.; Zhu, S. Transesterification of soybean oil to biodiesel using SrO as a solid base catalyst. Catal. Commun. 2007, 8, 1107–1111. [Google Scholar] [CrossRef]
- Bezerra Mota Gomes Arruda, T.; Arruda Rodrigues, F.E.; Duarte Arruda, D.T.; Pontes Silva Ricardo, N.M.; Barbosa Dantas, M.; Costa de Araújo, K. Chromatography, spectroscopy and thermal analysis of oil and biodiesel of sesame ( Sesamum indicum )—An alternative for the Brazilian Northeast. Ind. Crop. Prod. 2016, 91, 264–271. [Google Scholar] [CrossRef]
- Islam, F.; Gill, R.A.; Ali, B.; Farooq, M.A.; Xu, L.; Najeeb, U.; Zhou, W. Sesame. In Breeding Oilseed Crops for Sustainable Production; Academic Press: Cambridge, MA, USA, 2016; pp. 135–147. [Google Scholar]
| Animal bone | Catalyst Synthesis | Catalyst | Feedstock | Reaction Conditions (M/O, Catalyst wt%, Time, Temperature) | Yield | References |
|---|---|---|---|---|---|---|
| Waste animal bone | Calcined at 900 °C and soaked in KOH solution | HAp/KCaPO4 | Jatropha oil | 9, 6, 180, 70 ± 3 | 96.1 | [15] |
| Sheep bone | Calcined at 800 °C | HAp | Palm oil | 18, 20, 240, 65 | Y = 96.78 | [34] |
| Chicken bone | Calcined at 900 °C for 4 h | β -Ca3(PO4)2 | Low FFA WCO | 15, 5 g, 240, 65 | Y = 89.33 | [37] |
| Pig bone | Calcined at 900 °C for 4 h, 30 wt.% K2CO3 loaded, and calcined at 600 °C | K/HAp | Palm oil | 9, 8, 90, 65 | Y = 96.4 | [40] |
| Bovine bone | Calcined at 750 °C for 6 h | CaO | Soybean oil | 6, 8, 180, 65 | Y = 97 | [35] |
| Fish bone | Calcined at 900 °C for 2 h | H2SO4, β -Ca3(PO4)2 | WFO | 6.5, 1.5, 120, 55 | Y > 96 | [46] |
| Fish bone | Calcined at 997.42 C for 2 h | β -Ca3(PO4)2 | Soybean oil | 6.27, 1.01, 300, 70 | Y = 97.73 | [47] |
| Fish bone | 50 wt.% Cu(NO3)25H2O impregnated, 90 °C freeze drying temperature, and calcined at 500 °C for 4 h | Cu/HAp | Oleic acid | /,/, 60, 70 | C = 91.86 | [48] |
| Goat bone | Calcined at 800 °C soaked in KOH | K/CaO | WCO | 9, 6, 300, 65 | Y = 84 | [49] |
| Ostrich bone | Calcined at 900 °C for 4 h | HAp | WCO | 15, 5, 240, 60 | Y = 90.56 | [50] |
| Waste animal bone | Calcined at 900 °C for 2 h | HAp | Peanut oil Rapeseed oil | 20, 18, 240, 60 | Y = 94 Y = 96 | [51] |
| Chicken bone | Calcined at 800 °C for 4 h | β -Ca3(PO4)2 | E. compressa algal oil | 9, 5, 180, 65 | Y = 94 | [52] |
| Pork bone | Calcined at 900 °C for 2 h | β -Ca3(PO4)2 | Jatropha Curcas oil | 18, 4, 5, 800 W | Y = 94 | [42] |
| Sheep bone | 10 mass% of calcined animal bone powder loaded catalyst on fly ash through wet impregnation and calcined at 900 °C for 2 h | β -Ca3(PO4)2 | Mustard oil | 5.5, 10, 360, 65 | Y = 90.4 o | [53] |
| Chicken bones | Li/Zn loaded chicken bones and calcined at 850 °C for 4 h | Li/Zn-chicken bones | Waste canola oil | 18, 4, 210, 60 | Y = 98 | [54] |
| Fish bone | NaOH loaded over bone calcined by a hydrothermal process at 200 °C for 12 h. | Na/HAp | Waste palm oil | 9, 2.5, 90, 65 | Y = 98 | [55] |
| Cow bone | Calcined at 800 °C | HAp | Soybean oil | 9, 15, 180, 55 | Y = 92.2 | [56] |
| Guinea fowl bone | Calcined at 900 °C for 5 h | β -Ca3(PO4)2 | Annona squamosa L. seed oil | 18, 4, 20, 65, 800 W | Y = 95.82 | [57] |
| Cow bone | Calcined cow bones (900 °C for 5 h) and impregnated with sodium nitrate by wetness impregnation method | Sodium-supported nHAP | Schizochytrium oil | 12, 9.5, 121,/ | Y = 96 | [58] |
| Fish bone | Calcined at 900 °C for 4 h and supported on polyvinyl alcohol (PVA) | CaO/PVA | Palm oil | 20, 10, 180, 65 | Y = 80.4 | [59] |
| Sheep bone | Calcined at 600 °C for 8 h | CaO/HAp | Canola oil | 12, 5, 300, 60 | C = 95.18 | [41] |
| Turkey bone | Calcined at 909.4 °C for 4 h | β -Ca3(PO4)2 | Mustard oil | 9.9, 4.97,/, 65 | Y = 91.2 | [60] |
| Chicken bone | Calcined at 800 °C for 3 h | Ca-Fe2O3 | WCO | 6, 3, 180, 65 | Y = 22 | [61] |
| Waste bovine bone | Calcined at 750 °C for 4 h | WCO | 15.49, 6.42, 128.67,/ | Y = 97.59 | [62] | |
| Pig bone | Calcined at 1000 °C for 4 h and CaO-CeO2/HAp catalysts were prepared using wet impregnation technique | 30%CaO-CeO2/HAp−650 | Palm oil | 9, 11, 180, 65 | Y = 91.84 | [63] |
| Fusion of chicken and fish bone | Calcined for 4 h in air environment to 1000 °C at a heating rate of 3 °C/min | CaO/HAp | UCO | 10, 1.98 w/v, 92.4, 65 | Y = 89.5 | [44] |
| Cow bone | Calcined at 800 °C for 4 h for FFA esterification reaction | HAp | WFO | 12, 10, 240, 70 | C = 96 | [18] |
| Chicken bone | Calcined at 900 °C for 4 h | HAp | Celtis australis L. oil | 10.45, 5.69, 32.08,/(Ultrasonic) | Y = 95.1 | [43] |
| Goat bone | Calcined at 900 °C for 3 h | CaO | Algae oil | 11, 2, 180, 60 | Y = 92 | [64] |
| Goat bone | Calcined at 800 °C for 2 hand refluxed in distilled water for 24 h and recalcined at 900 °C for 2 h | CaO | Mixed Elengi and Pongamia Oil | 60 mL/200, 0.5, 240,/ | Y = 82.3 | [65] |
| Bovine bone | Loadings of 12-tungstophosphoric acid (TPA) were varied 117 (0–30 wt.%), with the specimen gained in each case subjected to calcination at 750 °C for 6 h | Tungsten/HAp | WCO | 6, 8, 300, 100 | Y = 98.9 | [66] |
| Elements | Calcined Fish Bone (Salmo salar) [76] | Calcined Cow Bone [77] | Calcined Chicken Bone [78] | Calcined Bovine Bone [79] | Calcined Ostrich Bone [50] | Calcined Sheep Bone [53] |
|---|---|---|---|---|---|---|
| (900 °C) | (950 °C) | (900 °C) | (800 °C) | (900 °C) | (900 °C) | |
| Ca | 38.43 | 27.71 | 53.58 | 64.92 | 60.0 | 29.79 |
| P | 19.08 | 16.39 | 40.99 | 31.53 | 12.8 | 9.2 |
| Mg | 0.43 | 0.31 | 0.813 | 1.36 | 0.8 | 0.48 |
| Na | 0.30 | 0.31 | 0.196 | 1.91 | 0.4 | - |
| K | 0.08 | 0.04 | 0.028 | 0.091 | 0.01 | 5.22 |
| Sr | 0.08 | 0.02 | 0.053 | 0.086 | 0.1 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, F.; Alshahrani, S.; Abbas, M.M.; Khan, H.M.; Jamil, A.; Yaqoob, H.; Soudagar, M.E.M.; Imran, M.; Ahmad, M.; Munir, M. Waste Animal Bones as Catalysts for Biodiesel Production; A Mini Review. Catalysts 2021, 11, 630. https://doi.org/10.3390/catal11050630
Hussain F, Alshahrani S, Abbas MM, Khan HM, Jamil A, Yaqoob H, Soudagar MEM, Imran M, Ahmad M, Munir M. Waste Animal Bones as Catalysts for Biodiesel Production; A Mini Review. Catalysts. 2021; 11(5):630. https://doi.org/10.3390/catal11050630
Chicago/Turabian StyleHussain, Fayaz, Saad Alshahrani, Muhammad Mujtaba Abbas, Haris Mahmood Khan, Asif Jamil, Haseeb Yaqoob, Manzoore Elahi M. Soudagar, Muhammad Imran, Mushtaq Ahmad, and Mamoona Munir. 2021. "Waste Animal Bones as Catalysts for Biodiesel Production; A Mini Review" Catalysts 11, no. 5: 630. https://doi.org/10.3390/catal11050630
APA StyleHussain, F., Alshahrani, S., Abbas, M. M., Khan, H. M., Jamil, A., Yaqoob, H., Soudagar, M. E. M., Imran, M., Ahmad, M., & Munir, M. (2021). Waste Animal Bones as Catalysts for Biodiesel Production; A Mini Review. Catalysts, 11(5), 630. https://doi.org/10.3390/catal11050630









