Transition Metal B-Site Substitutions in LaAlO3 Perovskites Reorient Bio-Ethanol Conversion Reactions
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization of the Catalysts
2.1.1. Phase and Textural Properties
2.1.2. Acid-Base Properties
2.1.3. H2-TPR Experiments
2.2. Catalytic Tests
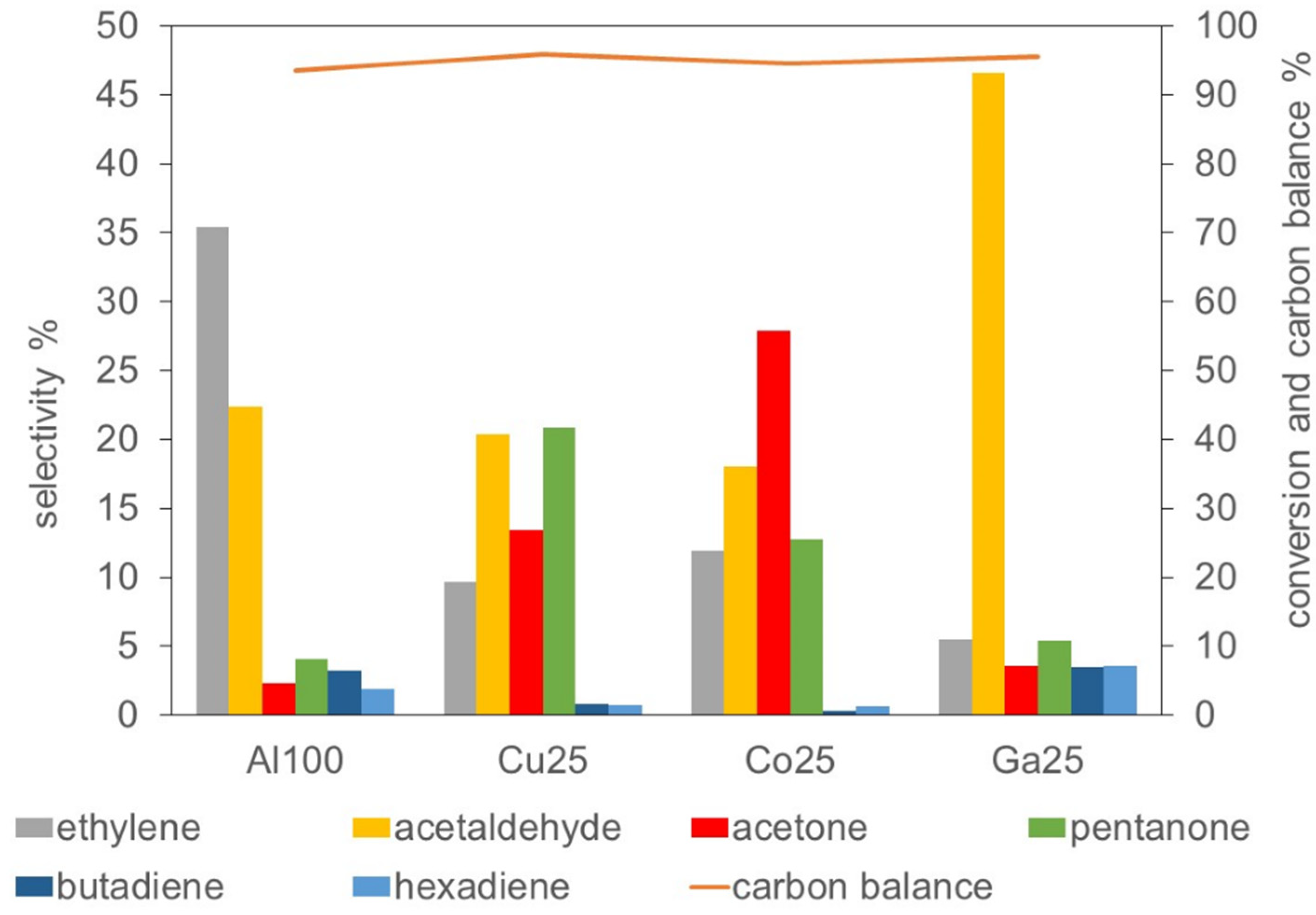
2.2.1. Overview of the Catalytic Activity
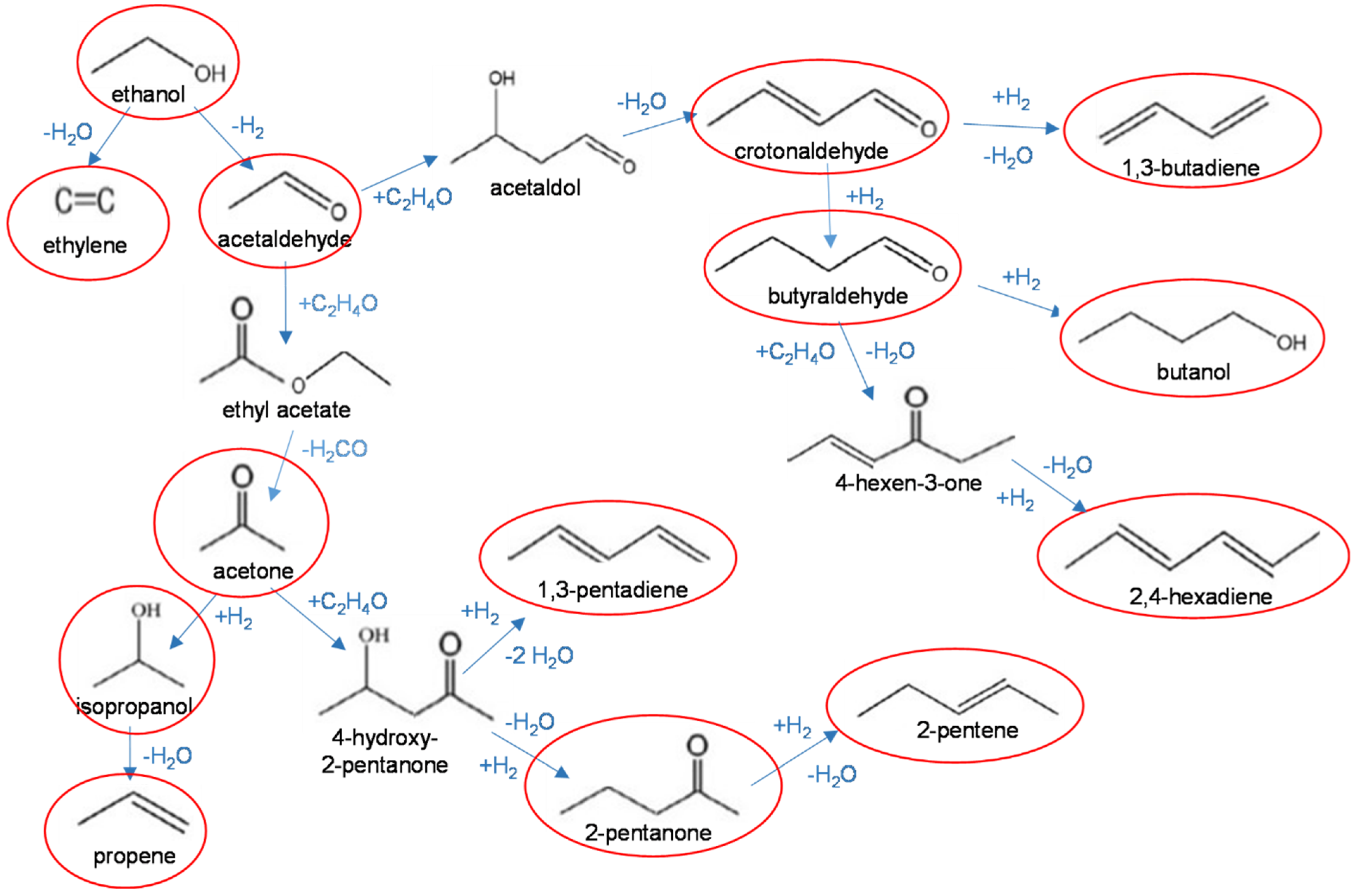
2.2.2. Primary Reactions of Ethanol
2.2.3. Acetaldehyde Reactivity
2.2.4. Acetone Reactions
2.2.5. Diolefins Formation by Self-Condensation of Acetaldehyde
2.2.6. Correlations between Activity and Composition of the Catalysts
3. Materials and Methods
3.1. Preparation of Catalysts
3.2. Characterisation of Catalysts
3.3. Catalytic Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ethanol. Available online: https://www.essentialchemicalindustry.org/chemicals/ethanol.html (accessed on 19 September 2020).
- IEA. Renewables 2019. Available online: https://webstore.iea.org/download/direct/2854?fileName=Renewables_2019.pdf (accessed on 29 January 2021).
- Lemos, P.; Mesquita, F. Future of Global Bioethanol: An Appraisal of Results, Risk and Uncertainties. In Global Bioethanol; Elsevier BV: Amsterdam, The Netherlands, 2016; pp. 221–237. [Google Scholar]
- Compare and Contrast Transport Biofuels Policies. Available online: https://www.ieabioenergy.com/wp-content/uploads/2020/03/IEA-Bioenergy-Task-39-Implementation-Agendas-Final-Draft-Feb-4-2020.pdf (accessed on 29 January 2021).
- Available online: https://www.world-grain.com/articles/13635-global-ethanol-production-could-drop-to-2013-levels (accessed on 22 January 2021).
- Takei, T.; Iguchi, N.; Haruta, M. Synthesis of Acetoaldehyde, Acetic Acid, and Others by the Dehydrogenation and Oxidation of Ethanol. Catal. Surv. Asia 2011, 15, 80–88. [Google Scholar] [CrossRef]
- Angelici, C.; Weckhuysen, B.M.; Bruijnincx, P.C.A. Chemocatalytic Conversion of Ethanol into Butadiene and Other Bulk Chemicals. ChemSusChem 2013, 6, 1595–1614. [Google Scholar] [CrossRef]
- Sun, J.; Wang, Y. Recent Advances in Catalytic Conversion of Ethanol to Chemicals. ACS Catal. 2014, 4, 1078–1090. [Google Scholar] [CrossRef]
- Garbarino, G.; Pampararo, G.; Phung, T.K.; Riani, P.; Busca, G. Heterogeneous Catalysis in (Bio)Ethanol Conversion to Chemicals and Fuels: Thermodynamics, Catalysis, Reaction Paths, Mechanisms and Product Selectivities. Energies 2020, 13, 3587. [Google Scholar] [CrossRef]
- Zhang, M.; Yu, Y. Dehydration of Ethanol to Ethylene. Ind. Eng. Chem. Res. 2013, 52, 9505–9514. [Google Scholar] [CrossRef]
- Freitas, I.; Damyanova, S.; Oliveira, D.; Marques, C.; Bueno, J. Effect of Cu Content on the Surface and Catalytic Properties of Cu/ZrO2 Catalyst for Ethanol Dehydrogenation. J. Mol. Catal. A Chem. 2014, 381, 26–37. [Google Scholar] [CrossRef]
- Wang, Q.-N.; Shi, L.; Lu, A.-H. Highly Selective Copper Catalyst Supported on Mesoporous Carbon for the Dehydrogenation of Ethanol to Acetaldehyde. ChemCatChem 2015, 7, 2846–2852. [Google Scholar] [CrossRef]
- Garbarino, G.; Riani, P.; García, M.V.; Finocchio, E.; Escribano, V.S.; Busca, G. A Study of Ethanol Dehydrogenation to Acetaldehyde over Copper/Zinc Aluminate Catalysts. Catal. Today 2020, 354, 167–175. [Google Scholar] [CrossRef]
- Marcu, I.-C.; Tanchoux, N.; Fajula, F.; Tichit, D. Catalytic Conversion of Ethanol into Butanol over M–Mg–Al Mixed Oxide Catalysts (M = Pd, Ag, Mn, Fe, Cu, Sm, Yb) Obtained from LDH Precursors. Catal. Lett. 2012, 143, 23–30. [Google Scholar] [CrossRef]
- Scalbert, J.; Thibault-Starzyk, F.; Jacquot, R.; Morvan, D.; Meunier, F. Ethanol Condensation to Butanol at High Temperatures over a Basic Heterogeneous Catalyst: How Relevant Is Acetaldehyde Self-Aldolization? J. Catal. 2014, 311, 28–32. [Google Scholar] [CrossRef]
- Yusoff, M.N.A.M.; Zulkifli, N.W.M.; Masum, B.M.; Masjuki, H.H. Feasibility of Bioethanol and Biobutanol as Transportation Fuel in Spark-Ignition Engine: A Review. RSC Adv. 2015, 5, 100184–100211. [Google Scholar] [CrossRef]
- Sun, Z.; Vasconcelos, A.C.; Bottari, G.; Stuart, M.C.A.; Bonura, G.; Cannilla, C.; Frusteri, F.; Barta, K. Efficient Catalytic Conversion of Ethanol to 1-Butanol via the Guerbet Reaction over Copper-and Nickel-Doped Porous. ACS Sustain. Chem. Eng. 2017, 5, 1738–1746. [Google Scholar] [CrossRef]
- Mazzoni, R.; Cesari, C.; Zanotti, V.; Lucarelli, C.; Tabanelli, T.; Puzzo, F.; Passarini, F.; Neri, E.; Marani, G.; Prati, R.; et al. Catalytic Biorefining of Ethanol from Wine Waste to Butanol and Higher Alcohols: Modeling the Life Cycle Assessment and Process Design. ACS Sustain. Chem. Eng. 2019, 7, 224–237. [Google Scholar] [CrossRef]
- Benito, P.; Vaccari, A.; Antonetti, C.; Licursi, D.; Schiarioli, N.; Rodriguez-Castellón, E.; Galletti, A.M.R. Tunable Copper-Hydrotalcite Derived Mixed Oxides for Sustainable Ethanol Condensation to N-Butanol in Liquid Phase. J. Clean. Prod. 2019, 209, 1614–1623. [Google Scholar] [CrossRef]
- Jones, M.D.; Keir, C.G.; Di Iulio, C.; Robertson, R.A.M.; Williams, C.V.; Apperley, D.C. Investigations into the Conversion of Ethanol into 1,3-Butadiene. Catal. Sci. Technol. 2011, 1, 267–272. [Google Scholar] [CrossRef]
- Sushkevich, V.L.; Ivanova, I.I.; Ordomsky, V.V.; Taarning, E. Design of a Metal-Promoted Oxide Catalyst for the Selective Synthesis of Butadiene from Ethanol. ChemSusChem 2014, 7, 2527–2536. [Google Scholar] [CrossRef]
- Chieregato, A.; Ochoa, J.V.; Bandinelli, C.; Fornasari, G.; Cavani, F.; Mella, M. On the Chemistry of Ethanol on Basic Oxides: Revising Mechanisms and Intermediates in the Lebedev and Guerbet reactions. ChemSusChem 2014, 8, 377–388. [Google Scholar] [CrossRef]
- Baylon, R.A.; Sun, J.; Wang, Y. Conversion of Ethanol to 1,3-Butadiene Over Na doped Zn Zr O Mixed Metal Oxides. Catal. Today 2016, 259, 446–452. [Google Scholar] [CrossRef]
- Ochoa, J.V.; Bandinelli, C.; Vozniuk, O.; Chieregato, A.; Malmusi, A.; Recchi, C.; Cavani, F. An Analysis of the Chemical, Physical and Reactivity Features of MgO–SiO2 Catalysts for Butadiene Synthesis with the Lebedev Process. Green Chem. 2015, 18, 1653–1663. [Google Scholar] [CrossRef]
- Ochoa, J.V.; Malmusi, A.; Recchi, C.; Cavani, F. Understanding the Role of Gallium as a Promoter of Magnesium Silicate Catalysts for the Conversion of Ethanol into Butadiene. ChemCatChem 2017, 9, 2128–2135. [Google Scholar] [CrossRef]
- Inui, K.; Kurabayashi, T.; Sato, S. Direct Synthesis of Ethyl Acetate from Ethanol Carried Out under Pressure. J. Catal. 2002, 212, 207–215. [Google Scholar] [CrossRef]
- Santacesaria, E.; Carotenuto, G.; Tesser, R.; Di Serio, M. Ethanol Dehydrogenation to Ethyl Acetate by Using Copper and Copper Chromite Catalysts. Chem. Eng. J. 2012, 179, 209–220. [Google Scholar] [CrossRef]
- Murthy, R.; Patnaik, P.; Sidheswaran, P.; Jayamani, M. Conversion of Ethanol to Acetone over Promoted Iron Oxide Catalysis. J. Catal. 1988, 109, 298–302. [Google Scholar] [CrossRef]
- Rodrigues, C.P.; Zonetti, P.C.; Silva, C.G.; Gaspar, A.B.; Appel, L.G. Chemicals from Ethanol—the Acetone One-Pot Synthesis. Appl. Catal. A Gen. 2013, 458, 111–118. [Google Scholar] [CrossRef]
- He, D.; Ding, Y.; Chen, W.; Lu, Y.; Luo, H. One-Step Synthesis of 2-Pentanone from Ethanol over K-Pd/MnOx-ZrO2-ZnO catalyst. J. Mol. Catal. A Chem. 2005, 226, 89–92. [Google Scholar] [CrossRef]
- Iwamoto, M. Selective Catalytic Conversion of Bio-Ethanol to Propene: A Review of Catalysts and Reaction Pathways. Catal. Today 2015, 242, 243–248. [Google Scholar] [CrossRef]
- Xue, F.; Miao, C.; Yue, Y.; Hua, W.; Gao, Z. Direct Conversion of Bio-Ethanol to Propylene in High Yield over the Composite of in 2O3 and Zeolite Beta. Green Chem. 2017, 19, 5582–5590. [Google Scholar] [CrossRef]
- Polo-Garzon, F.; Yang, S.-Z.; Fung, V.; Foo, G.S.; Bickel, E.E.; Chisholm, M.F.; Jiang, D.-E.; Wu, Z. Controlling Reaction Selectivity through the Surface Termination of Perovskite Catalysts. Angew. Chem. Int. Ed. 2017, 56, 9820–9824. [Google Scholar] [CrossRef]
- Mitchell, R.H. Perovskites Modern and Ancient; Almaz Press: Thunder Bay, ON, Canada, 2002. [Google Scholar]
- Peña, M.A.; Fierro, J.L.G. Chemical Structures and Performance of Perovskite Oxides. Chem. Rev. 2001, 101, 1981–2018. [Google Scholar] [CrossRef]
- Tilley, R.J.D. Perovskites Structure-Property Relationships; Wiley: Chichester, UK, 2016; ISBN 0791118935668. [Google Scholar]
- Zheng, Y.-S.; Zhang, M.; Li, Q.; Zhu, Y.-A.; Sui, Z.J.; Chen, D.; Zhou, X.-G. Electronic Origin of Oxygen Transport Behavior in La-Based Perovskites: A Density Functional Theory Study. J. Phys. Chem. C 2018, 123, 275–290. [Google Scholar] [CrossRef]
- Zhu, H.; Zhang, P.; Dai, S. Recent Advances of Lanthanum-Based Perovskite Oxides for Catalysis. ACS Catal. 2015, 5, 6370–6385. [Google Scholar] [CrossRef]
- Tejuca, L.G.; Fierro, J.L.G. Properties and Applications of Perovskite-Type Oxides; Taylor and Francis: Boca Raton, FL, USA, 1993; ISBN 0-8247-8786-2. [Google Scholar]
- Suntivich, J.; May, K.J.; Gasteiger, H.A.; Goodenough, J.B.; Shao-Horn, Y. A Perovskite Oxide Optimized for Oxygen Evolution Catalysis from Molecular Orbital Principles. Science 2011, 334, 1383–1385. [Google Scholar] [CrossRef]
- Wang, W.; Tadé, M.O.; Shao, Z. Research Progress of Perovskite Materials in Photocatalysis- and Photovoltaics-Related Energy Conversion and Environmental Treatment. Chem. Soc. Rev. 2015, 44, 5371–5408. [Google Scholar] [CrossRef]
- Granger, P.; Parvulescu, V.I.; Kaliaguine, S.; Prellier, W. Perovskites and Related Mixed Oxides-Concepts and Applications; Wiley-VCH: Weinheim, Germany, 2016; ISBN 978-3-527-33763-7. [Google Scholar]
- Grabowska, E. Selected Perovskite Oxides: Characterization, Preparation and Photocatalytic Properties—A Review. Appl. Catal. B Environ. 2016, 186, 97–126. [Google Scholar] [CrossRef]
- Zhang, G.; Liu, G.; Wang, L.; Irvine, J.T.S. Inorganic Perovskite Photocatalysts for Solar Energy Utilization. Chem. Soc. Rev. 2016, 45, 5951–5984. [Google Scholar] [CrossRef]
- Correa-Baena, J.-P.; Saliba, M.; Buonassisi, T.; Grätzel, M.; Abate, A.; Tress, W.; Hagfeldt, A. Promises and Challenges of Perovskite Solar Cells. Science 2017, 358, 739–744. [Google Scholar] [CrossRef]
- Hwang, J.; Rao, R.R.; Giordano, L.; Katayama, Y.; Yu, Y.; Shao-Horn, Y. Perovskites in Catalysis and Electrocatalysis. Science 2017, 358, 751–756. [Google Scholar] [CrossRef]
- Zhu, J.; Li, H.; Zhong, L.; Xiao, P.; Xu, X.; Yang, X.; Zhao, Z.; Li, J. Perovskite Oxides: Preparation, Characterizations, and Applications in Heterogeneous Catalysis. ACS Catal. 2014, 4, 2917–2940. [Google Scholar] [CrossRef]
- Royer, S.; Duprez, D.; Can, F.; Courtois, X.; Batiot-Dupeyrat, C.; Laassiri, S.; Alamdari, H. Perovskites as Substitutes of Noble Metals for Heterogeneous Catalysis: Dream or Reality. Chem. Rev. 2014, 114, 10292–10368. [Google Scholar] [CrossRef]
- Ezbiri, M.; Allen, K.M.; Gàlvez, M.E.; Michalsky, R.; Steinfeld, A. Design Principles of Perovskites for Thermochemical Oxygen Separation. ChemSusChem 2015, 8, 1966–1971. [Google Scholar] [CrossRef]
- Deng, H.; Lin, L.; Liu, S. Catalysis of Cu-Doped Co-Based Perovskite-Type Oxide in Wet Oxidation of Lignin to Produce Aromatic Aldehydes. Energy Fuels 2010, 24, 4797–4802. [Google Scholar] [CrossRef]
- Zhang, J.; Tan, D.; Meng, Q.; Weng, X.; Wu, Z. Structural Modification of LaCoO3 Perovskite for Oxidation Reactions: The Synergistic Effect of Ca2+ and Mg2+ Co-Substitution on Phase Formation and Catalytic Performance. Appl. Catal. B Environ. 2015, 172–173, 18–26. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Łamacz, A.; Beaunier, P.; Czajkowska, S.; Domanski, M.; Krztoń, A.; Van Le, T.; Djéga-Mariadassou, G. Partial Oxidation of Methane over Bifunctional Catalyst I. In Situ Formation of Ni0/La2O3 During Temperature Programmed POM Reaction over LaNiO3 Perovskite. Appl. Catal. B Environ. 2014, 152–153, 360–369. [Google Scholar] [CrossRef]
- Irusta, S.; Pina, M.; Menendez, M.; Santamaría, J. Catalytic Combustion of Volatile Organic Compounds over La-Based Perovskites. J. Catal. 1998, 179, 400–412. [Google Scholar] [CrossRef]
- Onrubia-Calvo, J.A.; Pereda-Ayo, B.; De-La-Torre, U.; González-Velasco, J.R. Key Factors in Sr-Doped LaBO3 (B = Co or Mn) Perovskites for NO Oxidation in Efficient Diesel Exhaust Purification. Appl. Catal. B Environ. 2017, 213, 198–210. [Google Scholar] [CrossRef]
- Jabłońska, M.; Palkovits, R. Perovskite-Based Catalysts for the Control of Nitrogen Oxide Emissions from Diesel Engines. Catal. Sci. Technol. 2019, 9, 2057–2077. [Google Scholar] [CrossRef]
- Wu, Y.; Li, D.; Lu, J.; Xie, S.; Dong, L.; Fan, M.; Li, B. LaMnO3-La2CuO4 Two-Phase Synergistic System with Broad Active Window in Nox Efficient Reduction. Mol. Catal. 2020, 493, 111111. [Google Scholar] [CrossRef]
- Martinovic, F.; Tran, Q.N.; Deorsola, F.A.; Bensaid, S.; Palkovits, R.; Paulus, W.; Bonelli, B.; Di Renzo, F.; Pirone, R. SO2 Deactivation Mechanism of NO Oxidation and Regeneration of the LaCoO3 Perovskite. Catal. Sci. Technol. 2020, 10, 2193–2202. [Google Scholar] [CrossRef]
- Truong, T.G.; Rotonnelli, B.; Rieu, M.; Viricelle, J.-P.; Kalaitzidou, I.; Marinha, D.; Burel, L.; Caravaca, A.; Vernoux, P.; Kaper, H. Catalytic and Electrochemical Properties of Ag Infiltrated Perovskite Coatings for Propene Deep Oxidation. Catalysis 2020, 10, 729. [Google Scholar] [CrossRef]
- Russo, N.; Furfori, S.; Fino, D.; Saracco, G.; Specchia, V. Lanthanum Cobaltite Catalysts for Diesel Soot Combustion. Appl. Catal. B Environ. 2008, 83, 85–95. [Google Scholar] [CrossRef]
- Tran, Q.N.; Martinovic, F.; Ceretti, M.; Esposito, S.; Bonelli, B.; Paulus, W.; Di Renzo, F.; Deorsola, F.A.; Bensaid, S.; Pirone, R. Co-Doped LaAlO3 Perovskite Oxide for NOx-Assisted Soot Oxidation. Appl. Catal. A Gen. 2020, 589, 117304. [Google Scholar] [CrossRef]
- Escalona, N.; Aranzaez, W.; Leiva, K.; Martinez, N.; Pecchi, G. NI Nanoparticles Prepared from Ce Substituted LaNiO3 for the Guaiacol Conversion. Appl. Catal. A Gen. 2014, 481, 1–10. [Google Scholar] [CrossRef]
- Chen, M.-Y.; Zada, B.; Fu, Y. Perovskite Type Oxide-Supported Ni Catalysts for the Production of 2,5-Dimethylfuran from Biomass-Derived 5-Hydroxymethylfurfural. Green Chem. 2016, 18, 3858–3866. [Google Scholar] [CrossRef]
- Lin, K.-H.; Wang, C.-B.; Chien, S.-H. Catalytic Performance of Steam Reforming of Ethanol at Low Temperature over LaNiO3 Perovskite. Int. J. Hydrogen Energy 2013, 38, 3226–3232. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, L.; Jiang, B.; Wang, K.; Tang, D.; Dou, B. An Intelligent Oxygen Carrier of La−Sr NiO−for Hydrogen Production by Chemical Looping Reforming of Ethanol. Int. J. Hydrogen Energy 2017, 42, 17102–17111. [Google Scholar] [CrossRef]
- Tesquet, G.; Faye, J.; Hosoglu, F.; Mamede, A.-S.; Dumeignil, F.; Capron, M. Ethanol Reactivity over La1+x FeO3+δ Perovskites. Appl. Catal. A: Gen. 2016, 511, 141–148. [Google Scholar] [CrossRef]
- Chen, R.-K.; Yu, T.-F.; Wu, M.-X.; Tzeng, T.-W.; Chung, P.-W.; Lin, Y.-C. The Aldolization Nature of Mn4+-Nonstoichiometric Oxygen Pair Sites of Perovskite-Type LaMnO3 in the Conversion of Ethanol. ACS Sustain. Chem. Eng. 2018, 6, 11949–11958. [Google Scholar] [CrossRef]
- Yu, T.-F.; Chang, C.-W.; Chung, P.-W.; Lin, Y.-C. Unsupported and Silica-Supported Perovskite-Type Lanthanum Manganite and Lanthanum Ferrite in the Conversion of Ethanol. Fuel Process. Technol. 2019, 194, 106117. [Google Scholar] [CrossRef]
- Polo-Garzon, F.; Wu, Z. Acid–Base Catalysis over Perovskites: A Review. J. Mater. Chem. A 2018, 6, 2877–2894. [Google Scholar] [CrossRef]
- Church, J.S.; Cant, N.W.; Trimm, D.L. Stabilisation of Aluminas by Rare Earth and Alkaline Earth Ions. Appl. Catal. A Gen. 1993, 101, 105–116. [Google Scholar] [CrossRef]
- Yamamoto, T.; Hatsui, T.; Matsuyama, T.; Tanaka, T.; Funabiki, T. Structures and Acid−Base Properties of La/Al2O3Role of La Addition to Enhance Thermal Stability of γ-Al2O3. Chem. Mater. 2003, 15, 4830–4840. [Google Scholar] [CrossRef]
- Garbarino, G.; Wang, C.; Valsamakis, I.; Chitsazan, S.; Riani, P.; Finocchio, E.; Flytzani-Stephanopoulos, M.; Busca, G. Acido-Basicity of Lanthana/Alumina Catalysts and Their Activity in Ethanol Conversion. Appl. Catal. B Environ. 2017, 200, 458–468. [Google Scholar] [CrossRef]
- Klimkiewicz, R.; Trawczyński, J. Secondary Ketonization of Primary Alcohol over Lamn-Based Mixed Oxides with Perovskite-like Structure. Appl. Catal. A Gen. 2009, 360, 199–204. [Google Scholar] [CrossRef]
- Zou, H.; Ge, X.; Shen, J. Surface Acidity and Basicity of γ-Al2O3 Doped with K+ and La3+ and Calcined at Elevated Temperatures. Thermochim. Acta 2003, 397, 81–86. [Google Scholar] [CrossRef]
- Delmastro, A.; Geobaldo, F.; Vallino, M.; Abbattista, F. Solid State Equilibria in the System Al2O3-La2O3-Cr2O3: Reactivity Catalyst/Support. J. Eur. Ceram. Soc. 1998, 18, 607–611. [Google Scholar] [CrossRef]
- Foo, G.S.; Polo-Garzon, F.; Fung, V.; Jiang, D.-E.; Overbury, S.H.; Wu, Z. Acid–Base Reactivity of Perovskite Catalysts Probed via Conversion of 2-Propanol over Titanates and Zirconates. ACS Catal. 2017, 7, 4423–4434. [Google Scholar] [CrossRef]
- Gervasini, A. Acidity and Basicity of Metal Oxide Surfaces II. Determination by Catalytic Decomposition of Isopropanol. J. Catal. 1991, 131, 190–198. [Google Scholar] [CrossRef]
- Aramendía, M.; Borau, V.; Jimenez, C.; Marinas, J.; Porras, A.; Urbano, F. Magnesium Oxides as Basic Catalysts for Organic Processes. J. Catal. 1996, 161, 829–838. [Google Scholar] [CrossRef]
- Hattori, H.; Ono, Y. Catalysts and Catalysis for Acid–Base Reactions. In Metal Oxides in Heterogeneous Catalysis; Elsevier BV: Amsterdam, The Netherlands, 2018; pp. 133–209. [Google Scholar]
- Enriquez, E.; Chen, A.; Harrell, Z.; Dowden, P.; Koskelo, N.; Roback, J.; Janoschek, M.; Chen, C.; Jia, Q. Oxygen Vacancy-Tuned Physical Properties in Perovskite Thin Films with Multiple B-site Valance States. Sci. Rep. 2017, 7, 46184. [Google Scholar] [CrossRef]
- Sis, L.B.; Wirtz, G.P.; Sorenson, S.C. Structure and Properties of Reduced LaCoO3. J. Appl. Phys. 1973, 44, 5553–5559. [Google Scholar] [CrossRef]
- Yang, X.; Yang, L.; Fan, W.; Lin, H. Effect of Redox Properties of LaCoO3 Perovskite Catalyst on Production of Lactic Acid from Cellulosic Biomass. Catal. Today 2016, 269, 56–64. [Google Scholar] [CrossRef]
- Olusola, O.J.; Sudip, M. Temperature Programme Reduction (TPR) Studies of Cobalt Phases in-Alumina Supported Cobalt Catalysts. J. Pet. Technol. Altern. Fuels 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Dewilde, J.F.; Czopinski, C.J.; Bhan, A. Ethanol Dehydration and Dehydrogenation on γ-Al2O3: Mechanism of Acetaldehyde Formation. ACS Catal. 2014, 4, 4425–4433. [Google Scholar] [CrossRef]
- Malmusi, A.; Ochoa, J.V.; Tabanelli, T.; Basile, F.; Lucarelli, C.; Agnoli, S.; Carraro, F.; Granozzi, G.; Cavani, F. Ethanol Aerobic and Anaerobic Oxidation with FeVO4 and V2O5 Catalysts. Appl. Catal. A Gen. 2019, 570, 139–147. [Google Scholar] [CrossRef]
- Ogata, Y.; Kawasaki, A.; Kishi, I. Kinetics of the Tischenko Reaction of Acetaldehyde with Aluminium Isopropoxide. Tetrahedron 1967, 23, 825–830. [Google Scholar] [CrossRef]
- Ogata, Y.; Kawasaki, A. Alkoxide Transfer from Aluminium Alkoxide to Aldehyde in the Tishchenko Reaction. Tetrahedron 1969, 25, 929–935. [Google Scholar] [CrossRef]
- Tanabe, K. The Conversion of Benzaldehyde into Benzyl Benzoate with Alkaline Earth Metal Oxide Catalysts. J. Catal. 1974, 35, 247–255. [Google Scholar] [CrossRef]
- Seki, T.; Nakajo, T.; Onaka, M. The Tishchenko Reaction: A Classic and Practical Tool for Ester Synthesis. Chem. Lett. 2006, 35, 824–829. [Google Scholar] [CrossRef]
- Bonne, M.; Bion, N.; Pailloux, F.; Valange, S.; Royer, S.; Tatibouët, J.-M.; Duprez, D. Improved Oxygen Mobility in Nanosized Mixed-Oxide Particles Synthesized Using a Simple Nanocasting Route. Chem. Commun. 2008, 4504–4506. [Google Scholar] [CrossRef]
- Zonetti, P.C.; Celnik, J.; Letichevsky, S.; Gaspar, A.B.; Appel, L.G. Chemicals from Ethanol—The Dehydrogenative Route of the Ethyl Acetate One-Pot Synthesis. J. Mol. Catal. A Chem. 2011, 334, 29–34. [Google Scholar] [CrossRef]
- Carotenuto, G.; Tesser, R.; Di Serio, M.; Santacesaria, E. Kinetic Study of Ethanol Dehydrogenation to Ethyl Acetate Promoted by a Copper/Copper-Chromite Based Catalyst. Catal. Today 2013, 203, 202–210. [Google Scholar] [CrossRef]
- Colley, S.W.; Tabatabaei, J.; Waugh, K.C.; Wood, M.A. The Detailed Kinetics and Mechanism of Ethyl Ethanoate Synthesis over a Cu/Cr2O3 Catalyst. J. Catal. 2005, 236, 21–33. [Google Scholar] [CrossRef]
- Tosoni, S.; Pacchioni, G. Acetic Acid Ketonization on Tetragonal Zirconia: Role of Surface Reduction. J. Catal. 2016, 344, 465–473. [Google Scholar] [CrossRef]
- Hattori, H. Heterogeneous Basic Catalysis. Chem. Rev. 1995, 95, 537–558. [Google Scholar] [CrossRef]
- Xu, M.; Gines, M.J.; Hilmen, A.-M.; Stephens, B.L.; Iglesia, E. Isobutanol and Methanol Synthesis on Copper Catalysts Supported on Modified Magnesium Oxide. J. Catal. 1997, 171, 130–147. [Google Scholar] [CrossRef]
- Vargas, D.C.; Salazar, S.; Mora, J.R.; Van Geem, K.M.; Streitwieser, D.A. Experimental and Theoretical Study of the Thermal Decomposition of Ethyl Acetate during Fast Pyrolysis. Chem. Eng. Res. Des. 2020, 157, 153–161. [Google Scholar] [CrossRef]
- Mizuno, S.; Kurosawa, M.; Tanaka, M.; Iwamoto, M. One-Path and Selective Conversion of Ethanol to Propene on Scandium-modified Indium Oxide Catalysts. Chem. Lett. 2012, 41, 892–894. [Google Scholar] [CrossRef]
- Hayashi, F.; Iwamoto, M. Yttrium-Modified Ceria as a Highly Durable Catalyst for the Selective Conversion of Ethanol to Propene and Ethene. ACS Catal. 2012, 3, 14–17. [Google Scholar] [CrossRef]
- Matheus, C.R.V.; Chagas, L.H.; Gonzalez, G.G.; Aguiar, E.F.S.; Appel, L.G. Synthesis of Propene from Ethanol: A Mechanistic Study. ACS Catal. 2018, 8, 7667–7678. [Google Scholar] [CrossRef]
- Valsamakis, I.; Garbarino, G. Lanthanum-Based Catalysts for (Bio)Ethanol Conversion: Effect of Preparation Method on Catalytic Performance—Hard Tem-Plating versus Hydrolysis. J. Chem. Technol. Biotechnol. 2020, 6627, 1–6627. [Google Scholar] [CrossRef]
- Vozniuk, O.; Bazzo, C.; Albonetti, S.; Tanchoux, N.; Bosselet, F.; Millet, J.-M.M.; Di Renzo, F.; Cavani, F. Structural Changes of Binary/Ternary Spinel Oxides During Ethanol Anaerobic Decomposition. ChemCatChem 2017, 9, 2219–2230. [Google Scholar] [CrossRef]
- Jagadeesh, R.V.; Junge, H.; Pohl, M.-M.; Radnik, J.; Brückner, A.; Beller, M. Selective Oxidation of Alcohols to Esters Using Heterogeneous Co3O4–NC Catalysts under Mild Conditions. J. Am. Chem. Soc. 2013, 135, 10776–10782. [Google Scholar] [CrossRef]
- Izzo, L.; Tabanelli, T.; Cavani, F.; Vàsquez, P.B.; Lucarelli, C.; Mella, M.; Blair, P. The Competition between Dehydrogenation and Dehydration Reactions for Primary and Secondary Alcohols over Gallia: Unravelling the Effects of Molecular and Electronic Structure via a Two-Pronged Theoretical/Experimental Approach. Catal. Sci. Technol. 2020, 10, 3433–3449. [Google Scholar] [CrossRef]




| Catalyst | Cell Size a/Å | DScherrer/nm | ||||
|---|---|---|---|---|---|---|
| Calcined at 700 °C | after Catalysis | after H2-TPR | Calcined at 700 °C | after Catalysis | after H2-TPR | |
| Al100 | 3.793 | 3.794 | 3.792 | 30 | 29 | 35 |
| Co25 | 3.794 | 3.794 | 3.796 | 29 | 29 | 21 |
| Ga25 | 3.816 | 3.815 | 3.816 | 29 | 31 | 31 |
| Cu25 | 3.811 | 3.811 | 3.805 | 29 | 28 | 30 |
| Catalyst | SBET m2 g−1 | Vpore cm3 g−1 | Dave nm |
|---|---|---|---|
| Al100 | 14.6 | 0.07 | 29 |
| Co25 | 11.6 | 0.04 | 17 |
| Ga25 | 14.7 | 0.05 | 20 |
| Cu25 | 16.1 | 0.07 | 28 |
| Catalyst | Conversion % | Carbon Balance % | Selectivity % | |
|---|---|---|---|---|
| Acetone | Propylene | |||
| Al100 | 67.8 | 95.4 | 74.8 | 25.2 |
| Co25 | 98.8 | 98.7 | 93.8 | 6.2 |
| Ga25 | 97.2 | 91.5 | 96.5 | 3.5 |
| Catalyst | Al100 | Co25 | Ga25 |
|---|---|---|---|
| conversion % | 19.9 | 21.0 | 37.1 |
| C balance % | 97.1 | 96.1 | 94.4 |
| selectivity | |||
| ethylene | 1.4 | 0.9 | 1.7 |
| acetaldehyde | 5.0 | 8.2 | 11.7 |
| ethanol | 44.0 | 42.0 | 36.1 |
| acetone | 25.7 | 27.9 | 23.8 |
| isopropanol | 0.2 | 0.2 | 1.1 |
| pentanone | 0.2 | 0.7 | 3.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, Q.N.; Gimello, O.; Tanchoux, N.; Ceretti, M.; Albonetti, S.; Paulus, W.; Bonelli, B.; Di Renzo, F. Transition Metal B-Site Substitutions in LaAlO3 Perovskites Reorient Bio-Ethanol Conversion Reactions. Catalysts 2021, 11, 344. https://doi.org/10.3390/catal11030344
Tran QN, Gimello O, Tanchoux N, Ceretti M, Albonetti S, Paulus W, Bonelli B, Di Renzo F. Transition Metal B-Site Substitutions in LaAlO3 Perovskites Reorient Bio-Ethanol Conversion Reactions. Catalysts. 2021; 11(3):344. https://doi.org/10.3390/catal11030344
Chicago/Turabian StyleTran, Quang Nguyen, Olinda Gimello, Nathalie Tanchoux, Monica Ceretti, Stefania Albonetti, Werner Paulus, Barbara Bonelli, and Francesco Di Renzo. 2021. "Transition Metal B-Site Substitutions in LaAlO3 Perovskites Reorient Bio-Ethanol Conversion Reactions" Catalysts 11, no. 3: 344. https://doi.org/10.3390/catal11030344
APA StyleTran, Q. N., Gimello, O., Tanchoux, N., Ceretti, M., Albonetti, S., Paulus, W., Bonelli, B., & Di Renzo, F. (2021). Transition Metal B-Site Substitutions in LaAlO3 Perovskites Reorient Bio-Ethanol Conversion Reactions. Catalysts, 11(3), 344. https://doi.org/10.3390/catal11030344










