The Impact of Pressure and Hydrocarbons on NOx Abatement over Cu- and Fe-Zeolites at Pre-Turbocharger Position
Abstract
1. Introduction
2. Results and Discussion
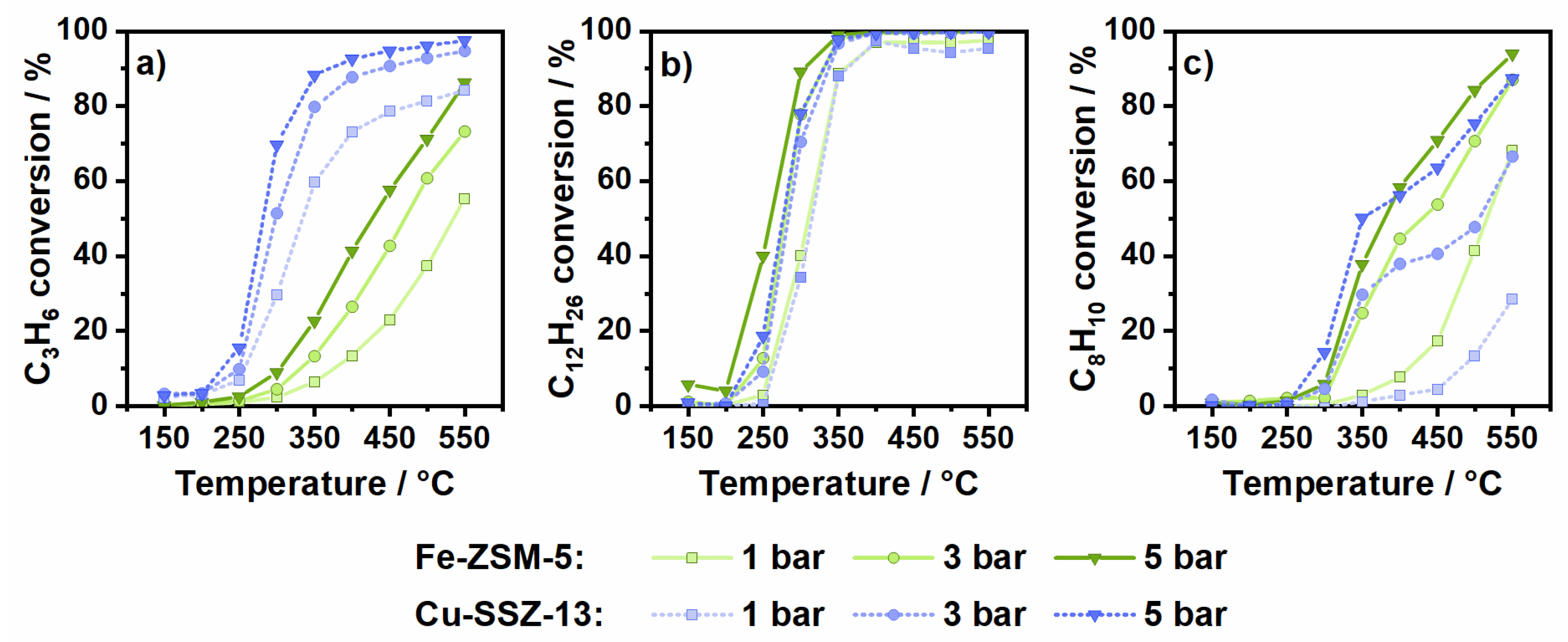
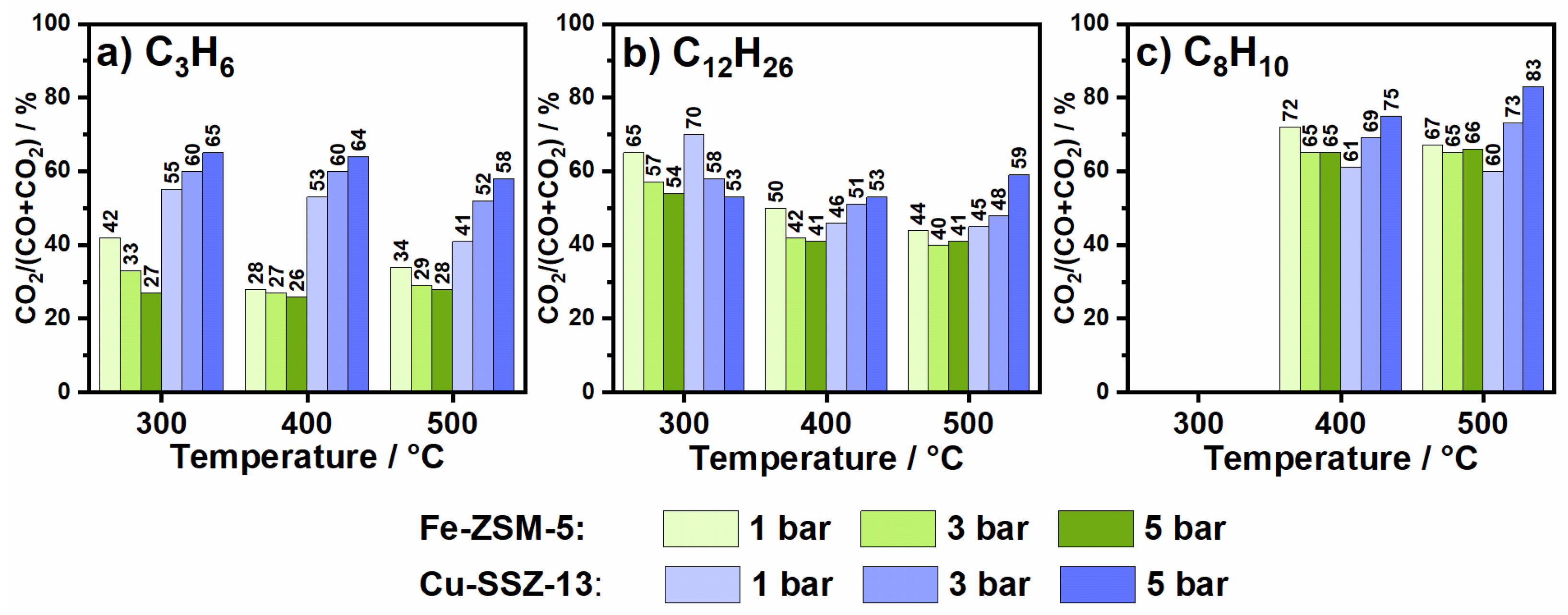
2.1. Hydrocarbon Oxidation at Elevated Pressure in the Presence of Oxygen
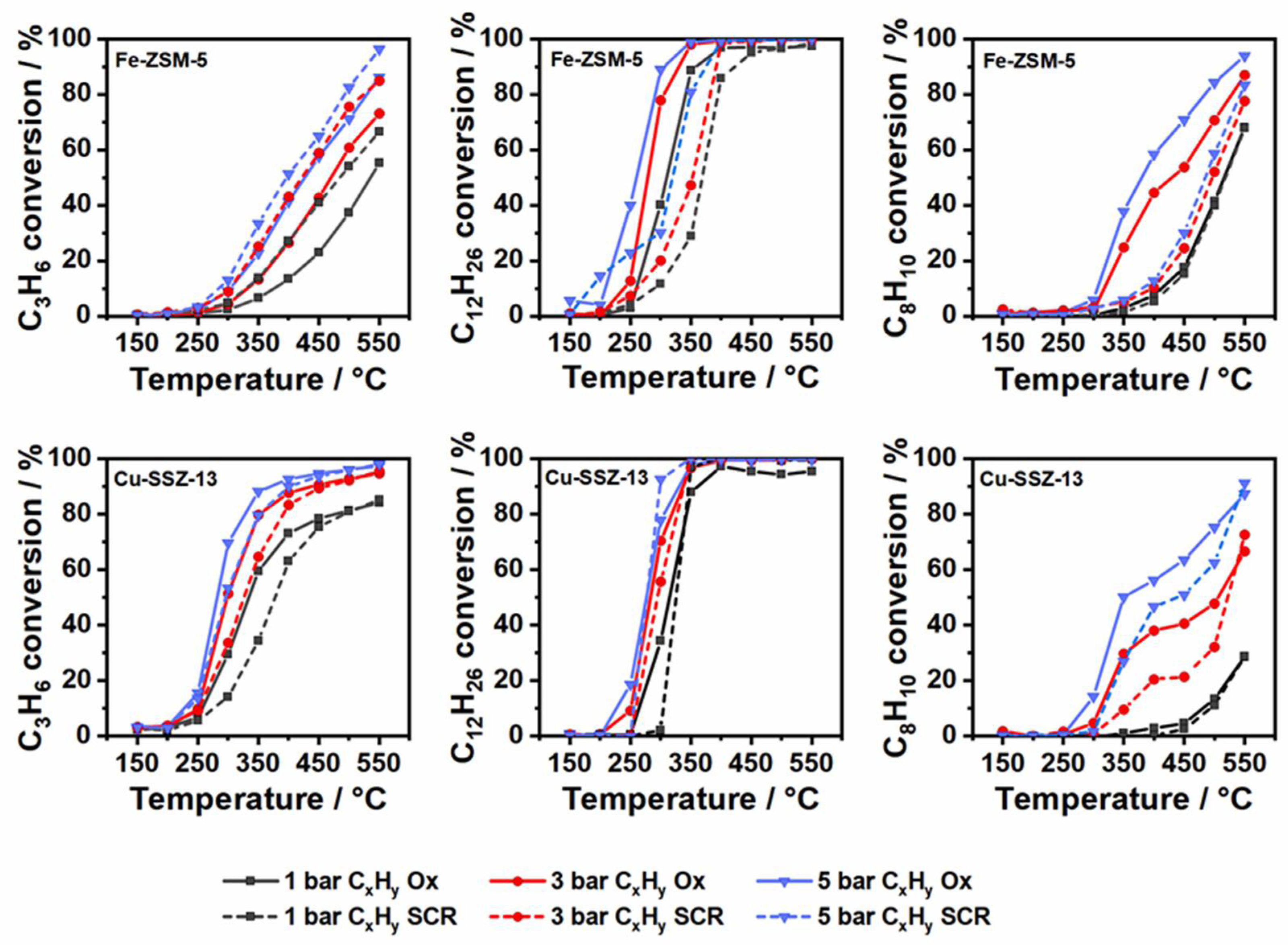
2.2. Hydrocarbon Oxidation at Elevated Pressure in a Standard SCR Gas Mixture
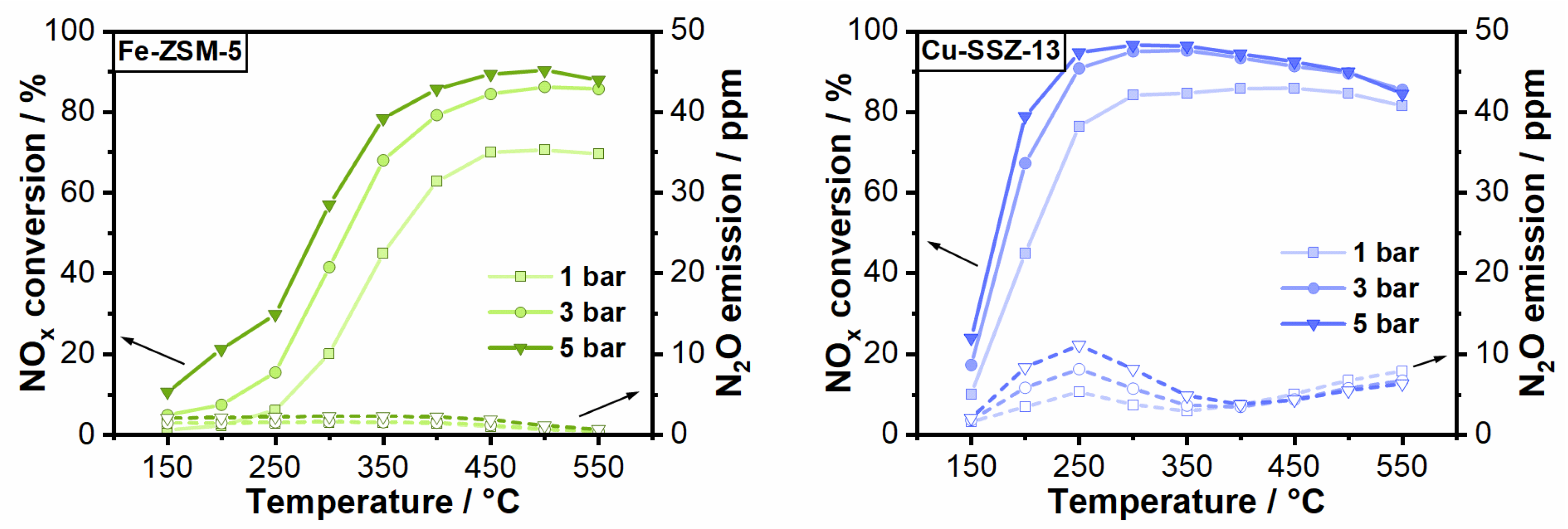
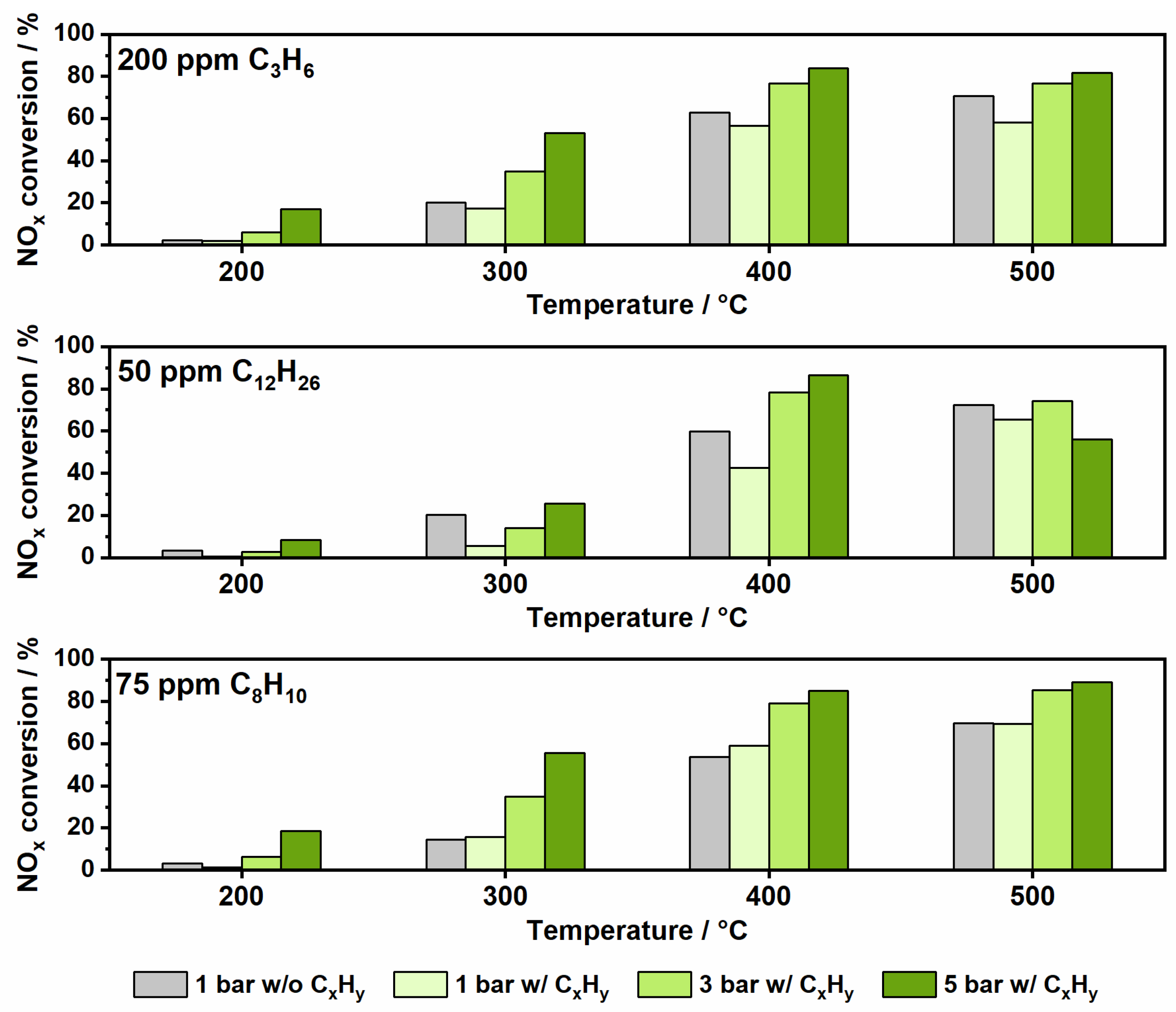
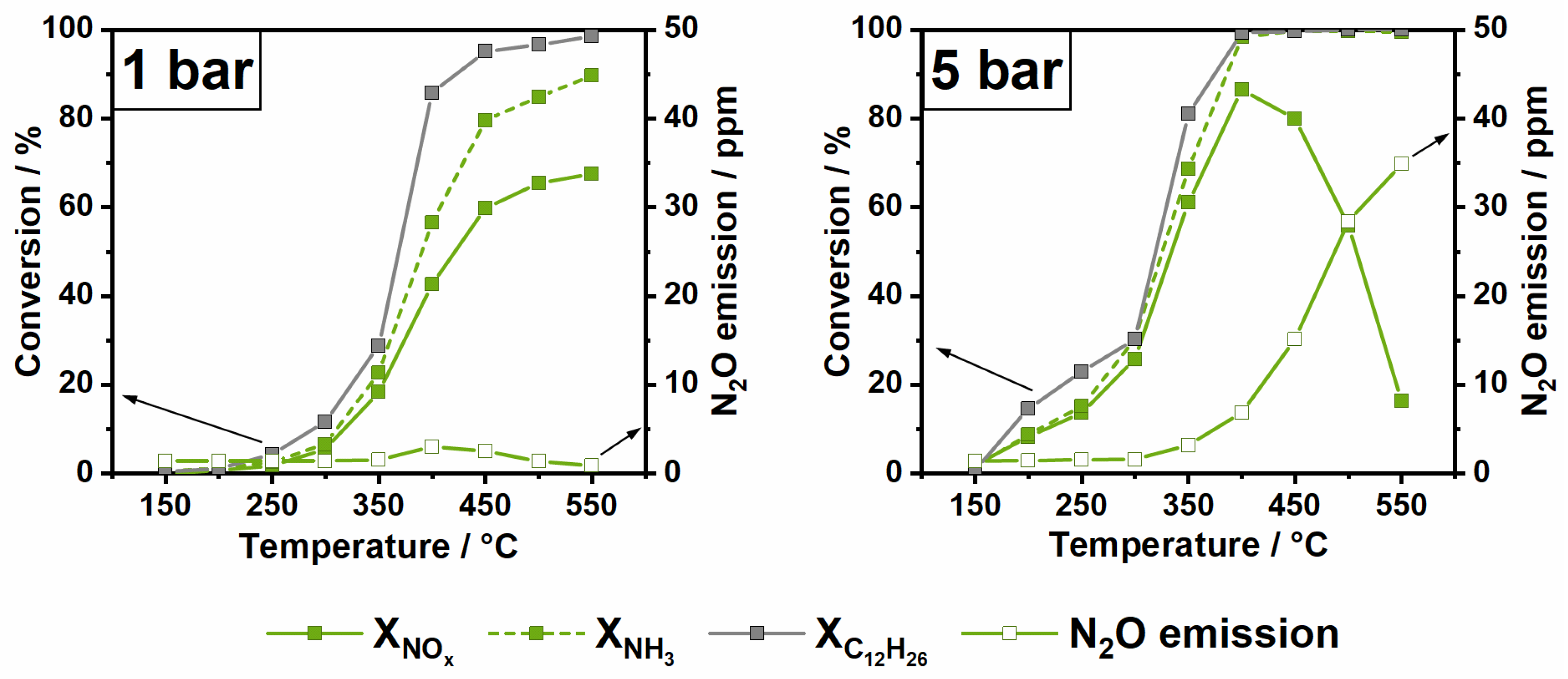
2.3. Impact of Elevated Pressure and Hydrocarbon Presence on NOx Conversion
3. Materials and Methods
3.1. Catalyst Preparation
3.2. Catalyst Characterization
3.3. Test Bench and Procedure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Group, C. Worldwide Emission Standards and Related Regulations: Passenger Cars/Light and Medium Duty Vehicles. Available online: https://www.continental-automotive.com/getattachment/8f2dedad-b510-4672-a005-3156f77d1f85/EMISSIONBOOKLET_2019.pdf (accessed on 28 September 2020).
- Kampa, M.; Castanas, E. Human health effects of air pollution. Environ. Pollut. 2008, 151, 362–367. [Google Scholar] [CrossRef]
- Rani, B.; Singh, U.; Chuhan, A.K.; Sharma, D.; Maheshwari, R. Photochemical Smog Pollution and Its Mitigation Measures. J. Adv. Sci. Res. 2011, 2, 28–33. [Google Scholar]
- Peel, J.L.; Haeuber, R.; Garcia, V.; Russell, A.G.; Neas, L. Impact of nitrogen and climate change interactions on ambient air pollution and human health. Biogeochemistry 2013, 114, 121–134. [Google Scholar] [CrossRef]
- Likens, G.E.; Wright, R.F.; Galloway, J.N.; Butler, T.J. Acid Rain. Sci. Am. 1979, 241, 43–51. [Google Scholar] [CrossRef]
- Maiboom, A.; Tauzia, X.; Hétet, J.-F. Experimental study of various effects of exhaust gas recirculation (EGR) on combustion and emissions of an automotive direct injection diesel engine. Energy 2008, 33, 22–34. [Google Scholar] [CrossRef]
- Mendez, S.; Thirouard, B. Using Multiple Injection Strategies in Diesel Combustion: Potential to Improve Emissions, Noise and Fuel Economy Trade-Off in Low-CR Engines. SAE Int. J. Fuels Lubr. 2008, 1, 662–674. [Google Scholar] [CrossRef]
- Nova, I.; Tronconi, E. Urea-SCR Technology for deNOx after Treatment Of Diesel Exhausts; Springer: Berlin/Heidelberg, Germany, 2014; Volume 5. [Google Scholar]
- Guan, B.; Zhan, R.; Lin, H.; Huang, Z. Review of state of the art technologies of selective catalytic reduction of NOx from diesel engine exhaust. Appl. Therm. Eng. 2014, 66, 395–414. [Google Scholar] [CrossRef]
- Shan, W.; Song, H. Catalysts for the selective catalytic reduction of NOx with NH3 at low temperature. Catal. Sci. Technol. 2015, 5, 4280–4288. [Google Scholar] [CrossRef]
- Yadav, D.; Prasad, R. Low Temperature de-NOx Technology-a Challenge for Vehicular Exhaust and its Remedation: An Overview. Procedia Technol. 2016, 24, 639–644. [Google Scholar] [CrossRef][Green Version]
- Zammit, M.; DiMaggio, C.L.; Kim, C.H.; Lambert, C.; Muntean, G.G.; Peden, C.H.; Parks, J.E.; Howden, K. Future Automotive Aftertreatment Solutions: The 150 °C Challenge Workshop Report. U.S. Drive Report 2013. [Google Scholar] [CrossRef]
- Presti, M.; Scheeder, A.; Brück, R. Motornahe Abgasnachbehandlung im Nutzfahrzeug: Eine Lösung für CARB 2020 NOx? Continental Emitec GmbH—8th Emission Control Dresden. 2016. Available online: http://wordpress.emission-control-dresden.de/8-emission-control-2016/ (accessed on 6 March 2021).
- Günter, T.; Pesek, J.; Schäfer, K.; Bertótiné Abai, A.; Casapu, M.; Deutschmann, O.; Grunwaldt, J.-D. Cu-SSZ-13 as pre-turbine NOx-removal-catalyst: Impact of pressure and catalyst poisons. Appl. Catal. B Environ. 2016, 198, 548–557. [Google Scholar] [CrossRef]
- Rammelt, T.; Torkashvand, B.; Hauck, C.; Böhm, J.; Gläser, R.; Deutschmann, O. Nitric Oxide Reduction of Heavy-Duty Diesel Off-Gas by NH3-SCR in Front of the Turbocharger. Emiss. Control Sci. Technol. 2017, 3, 275–288. [Google Scholar] [CrossRef]
- Kröcher, O.; Elsener, M.; Bothien, M.-R.; Dölling, W. SCR vor Turbolader—Einfluss des Drucks auf die NOx-Reduktion. MTZ—Mot. Z. 2014, 75, 68–73. [Google Scholar] [CrossRef]
- Christensen, S.R.; Hansen, B.B.; Pedersen, K.H.; Thøgersen, J.R.; Jensen, A.D. Selective Catalytic Reduction of NOx over V2O5-WO3-TiO2 SCR Catalysts—A Study at Elevated Pressure for Maritime Pre-turbine SCR Configuration. Emiss. Control Sci. Technol. 2019, 5, 263–278. [Google Scholar] [CrossRef]
- Jayat, F.; Pace, L.; Konieczny, R. Vorturboladerkatalysatoren_-_Anforderungen aus aufladetechnischer Sicht sowie zukünftiger, innovativer Abgasnachbehandlungskonzepte, Emitec GmbH. 2017. Available online: https://docplayer.org/33955352-Vorturboladerkatalysatoren-anforderungen-aus-aufladetechnischer-sicht-sowie-zukuenftiger-innovativer-abgasnachbehandlungskonzepte.html (accessed on 6 March 2021).
- Serrano, J.R.; Guardiola, C.; Piqueras, P.; Angiolini, E. Analysis of the Aftertreatment Sizing for Pre-Turbo DPF and DOC Exhaust Line Configurations; 2014-01-1498; SAE Technical Paper: Warrendale, PA, USA, 2014. [Google Scholar] [CrossRef]
- Subramaniam, M.N.; Hayes, C.; Tomazic, D.; Downey, M.; Bruestle, C. Pre-Turbo Aftertreatment Position for Large Bore Diesel Engines—Compact & Cost-Effective Aftertreatment with a Fuel Consumption Advantage. SAE Int. J. Eng. 2011, 4, 106–116. [Google Scholar] [CrossRef]
- Liu, C.-H.; Giewont, K.; Toops, T.J.; Walker, E.A.; Horvatits, C.; Kyriakidou, E.A. Non-catalytic gas phase NO oxidation in the presence of decane. Fuel 2021, 286, 119388. [Google Scholar] [CrossRef]
- Hazlett, M.J.; Epling, W.S. Coupled Heterogeneous and Homogeneous Hydrocarbon Oxidation Reactions in Model Diesel Oxidation Catalysts. Emiss. Control Sci. Technol. 2017, 3, 5–17. [Google Scholar] [CrossRef]
- Zengel, D.; Stehle, M.; Deutschmann, O.; Casapu, M.; Grunwaldt, J.-D. Impact of gas phase reactions and catalyst poisons on the NH3-SCR activity of a V2O5-WO3/TiO2 catalyst at pre-turbine position. Appl. Catal. B Environ. 2021, 119991. [Google Scholar] [CrossRef]
- Starokon, E.; Vedyagin, A.; Pirutko, L.; Mishakov, I. Oxidation of CO and hydrocarbons with molecular oxygen over Fe–ZSM-5 zeolite. J. Porous Mater. 2015, 22, 521–527. [Google Scholar] [CrossRef]
- Torkashvand, B.; Gremminger, A.; Valchera, S.; Casapu, M.; Grunwaldt, J.-D.; Deutschmann, O. The Impact of Pre-Turbine Catalyst Placement on Methane Oxidation in Lean-Burn Gas Engines: An Experimental and Numerical Study; 2017-01-1019; SAE Technical Paper: Warrendale, PA, USA, 2017. [Google Scholar] [CrossRef]
- Zheng, Y.; Harold, M.P.; Luss, D. Effects of CO, H2 and C3H6 on Cu-SSZ-13 catalyzed NH3-SCR. Catal. Today 2016, 264, 44–54. [Google Scholar] [CrossRef]
- Vakhrushin, P.A.; Vishnetskaya, M.V.; Kokorin, A.I. The oxidation of dodecane on a vanadium-molybdenum catalyst. Russ. J. Phys. Chem. B 2012, 6, 169–172. [Google Scholar] [CrossRef]
- Vishnetskaya, M.V.; Vakhrushin, P.A. Oxidation of dodecane on transition metal oxides. Russ. J. Phys. Chem. A 2012, 86, 1664–1668. [Google Scholar] [CrossRef]
- Luo, J.-Y.; Yezerets, A.; Henry, C.; Hess, H.; Kamasamudram, K.; Chen, H.-Y.; Epling, W.S. Hydrocarbon Poisoning of Cu-Zeolite SCR Catalysts; 2012-01-1096; SAE Technical Paper: Warrendale, PA, USA, 2012. [Google Scholar] [CrossRef]
- Kurman, M.S.; Natelson, R.H.; Cernansky, N.P.; Miller, D.L. Speciation of the reaction intermediates from n-dodecane oxidation in the low temperature regime. Proc. Combust. Inst. 2011, 33, 159–166. [Google Scholar] [CrossRef]
- Becker, L.; Förster, H. Investigations of coke deposits formed during deep oxidation of benzene over Pd and Cu exchanged Y-type zeolites. Appl. Catal. A Gen. 1997, 153, 31–41. [Google Scholar] [CrossRef]
- Saleh, R.Y.; Wachs, I.E. Reaction network and kinetics of o-xylene oxidation to phthalic anhydride over V2O5/TiO2 (anatase) catalysts. Appl. Catal. 1987, 31, 87–98. [Google Scholar] [CrossRef]
- Andersson, S.L.T. Reaction networks in the catalytic vapor-phase oxidation of toluene and xylenes. J. Catal. 1986, 98, 138–149. [Google Scholar] [CrossRef]
- Wachs, I.E.; Saleh, R.Y.; Chan, S.S.; Chersich, C.C. The interaction of vanadium pentoxide with titania (anatase): Part I. Effect on o-xylene oxidation to phthalic anhydride. Appl. Catal. 1985, 15, 339–352. [Google Scholar] [CrossRef]
- Schaefgen, J.R. The pyrolysis of p-xylene. J. Polym. Sci. 1955, 15, 203–219. [Google Scholar] [CrossRef]
- Zengel, D.; Koch, P.; Torkashvand, B.; Grunwaldt, J.-D.; Casapu, M.; Deutschmann, O. Emission of Toxic HCN During NOx Removal by Ammonia SCR in the Exhaust of Lean-Burn Natural Gas Engines. Angew. Chem. Int. Ed. 2020, 59, 14423–14428. [Google Scholar] [CrossRef] [PubMed]
- Ngo, A.B.; Vuong, T.H.; Atia, H.; Bentrup, U.; Kondratenko, V.A.; Kondratenko, E.V.; Rabeah, J.; Ambruster, U.; Brückner, A. Effect of Formaldehyde in Selective Catalytic Reduction of NO x by Ammonia (NH3-SCR) on a Commercial V2O5-WO3/TiO2 Catalyst under Model Conditions. Environ. Sci. Technol. 2020, 54, 11753–11761. [Google Scholar] [CrossRef]
- Elsener, M.; Nuguid, R.J.G.; Kröcher, O.; Ferri, D. HCN production from formaldehyde during the selective catalytic reduction of NOx with NH3 over V2O5/WO3-TiO2. Appl. Catal. B Environ. 2021, 281, 119462. [Google Scholar] [CrossRef]
- Shi, X.; Liu, F.; Xie, L.; Shan, W.; He, H. NH3-SCR Performance of Fresh and Hydrothermally Aged Fe-ZSM-5 in Standard and Fast Selective Catalytic Reduction Reactions. Environ. Sci. Technol. 2013, 47, 3293–3298. [Google Scholar] [CrossRef]
- Kröcher, O.; Devadas, M.; Elsener, M.; Wokaun, A.; Söger, N.; Pfeifer, M.; Demel, Y.; Mussmann, L. Investigation of the selective catalytic reduction of NO by NH3 on Fe-ZSM5 monolith catalysts. Appl. Catal. B Environ. 2006, 66, 208–216. [Google Scholar] [CrossRef]
- Li, J.; Zhu, R.; Cheng, Y.; Lambert, C.K.; Yang, R.T. Mechanism of Propene Poisoning on Fe-ZSM-5 for Selective Catalytic Reduction of NOx with Ammonia. Environ. Sci. Technol. 2010, 44, 1799–1805. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, J.; Cheng, Y.; Lambert, C.K.; Fu, L. Propene Poisoning on Three Typical Fe-zeolites for SCR of NOx with NH3: From Mechanism Study to Coating Modified Architecture. Environ. Sci. Technol. 2012, 46, 1747–1754. [Google Scholar] [CrossRef]
- Heo, I.; Sung, S.; Park, M.B.; Chang, T.S.; Kim, Y.J.; Cho, B.K.; Hong, S.B.; Choung, J.W.; Nam, I.-S. Effect of Hydrocarbon on DeNOx Performance of Selective Catalytic Reduction by a Combined Reductant over Cu-Containing Zeolite Catalysts. ACS Catal. 2019, 9, 9800–9812. [Google Scholar] [CrossRef]
- Ma, L.; Su, W.; Li, Z.; Li, J.; Fu, L.; Hao, J. Mechanism of propene poisoning on Cu-SSZ-13 catalysts for SCR of NOx with NH3. Catal. Today 2015, 245, 16–21. [Google Scholar] [CrossRef]
- Ye, Q.; Wang, L.; Yang, R.T. Activity, propene poisoning resistance and hydrothermal stability of copper exchanged chabazite-like zeolite catalysts for SCR of NO with ammonia in comparison to Cu/ZSM-5. Appl. Catal. A Gen. 2012, 427–428, 24–34. [Google Scholar] [CrossRef]
- Jabłońska, M.; Delahay, G.; Kruczała, K.; Błachowski, A.; Tarach, K.A.; Brylewska, K.; Petitto, C.; Góra-Marek, K. Standard and Fast Selective Catalytic Reduction of NO with NH3 on Zeolites Fe-BEA. J. Physical Chem. C 2016, 120, 16831–16842. [Google Scholar] [CrossRef]
- Günter, T.; Carvalho, H.W.; Doronkin, D.E.; Sheppard, T.; Glatzel, P.; Atkins, A.J.; Rudolph, J.; Jacob, C.R.; Casapu, M.; Grunwaldt, J.D. Structural snapshots of the SCR reaction mechanism on Cu-SSZ-13. Chem. Commun. (Camb) 2015, 51, 9227–9230. [Google Scholar] [CrossRef]

| C3H6 Ox. | HCHO | CH3CHO | C2H4 | C3H6 | ||||||||
| Temperature | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar |
| 300 °C (Fe) | 3 | 6 | 12 | 0 | 1 | 2 | 0 | 0 | 0 | - | - | - |
| 400 °C (Fe) | 16 | 33 | 43 | 2 | 3 | 5 | 1 | 2 | 3 | - | - | - |
| 500 °C (Fe) | 24 | 36 | 32 | 2 | 3 | 4 | 2 | 4 | 5 | - | - | - |
| 300 °C (Cu) | 13 | 34 | 59 | 5 | 5 | 4 | 0 | 0 | 1 | - | - | - |
| 400 °C (Cu) | 45 | 36 | 35 | 5 | 2 | 1 | 9 | 9 | 7 | - | - | - |
| 500 °C (Cu) | 34 | 16 | 9 | 3 | 1 | 1 | 18 | 11 | 7 | - | - | - |
| C12H26 Ox. | HCHO | CH3CHO | C2H4 | C3H6 | ||||||||
| Temperature | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar |
| 300 °C (Fe) | 6 | 18 | 32 | 12 | 26 | 27 | 1 | 2 | 2 | 2 | 2 | 2 |
| 400 °C (Fe) | 52 | 70 | 67 | 16 | 6 | 2 | 9 | 8 | 6 | 5 | 2 | 1 |
| 500 °C (Fe) | 28 | 23 | 14 | 2 | 0 | 0 | 11 | 8 | 5 | 5 | 1 | 0 |
| 300 °C (Cu) | 3 | 7 | 13 | 6 | 16 | 23 | 0 | 1 | 1 | 0 | 0 | 0 |
| 400 °C (Cu) | 25 | 18 | 15 | 20 | 11 | 8 | 7 | 5 | 3 | 2 | 1 | 0 |
| 500 °C (Cu) | 19 | 4 | 2 | 12 | 4 | 2 | 15 | 4 | 1 | 3 | 0 | 0 |
| C8H10 Ox. | HCHO | CH3CHO | C2H4 | C3H6 | ||||||||
| Temperature | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar |
| 300 °C (Fe) | 1 | 1 | 2 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 2 |
| 400 °C (Fe) | 3 | 7 | 9 | 0 | 3 | 2 | 0 | 0 | 0 | 1 | 18 | 25 |
| 500 °C (Fe) | 7 | 11 | 10 | 2 | 1 | 0 | 0 | 0 | 0 | 0 | 2 | 2 |
| 300 °C (Cu) | 0 | 0 | 1 | 0 | 1 | 2 | 0 | 0 | 0 | 0 | 0 | 4 |
| 400 °C (Cu) | 0 | 1 | 1 | 1 | 6 | 4 | 0 | 0 | 0 | 1 | 10 | 17 |
| 500 °C (Cu) | 0 | 1 | 1 | 0 | 4 | 2 | 0 | 0 | 0 | 0 | 6 | 9 |
| StdSCR-C3H6 | HCHO | CH3CHO | C2H4 | HCN | ||||||||
| Temperature | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar |
| 300 °C (Fe) | 3 | 7 | 11 | 1 | 4 | 3 | 0 | 0 | 0 | 2 | 3 | 4 |
| 400 °C (Fe) | 19 | 35 | 44 | 3 | 3 | 3 | 1 | 2 | 2 | 19 | 19 | 14 |
| 500 °C (Fe) | 21 | 26 | 23 | 2 | 4 | 3 | 2 | 3 | 3 | 29 | 29 | 13 |
| 300 °C (Cu) | 2 | 12 | 28 | 0 | 2 | 3 | 0 | 0 | 1 | 7 | 10 | 8 |
| 400 °C (Cu) | 19 | 28 | 23 | 5 | 4 | 1 | 5 | 7 | 7 | 28 | 9 | 4 |
| 500 °C (Cu) | 23 | 13 | 8 | 3 | 0 | 0 | 16 | 11 | 6 | 5 | 0 | 0 |
| StdSCR-C12H26 | HCHO | CH3CHO | C2H4 | HCN | ||||||||
| Temperature | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar |
| 300 °C (Fe) | 1 | 3 | 6 | 2 | 3 | 6 | 0 | 0 | 0 | 1 | 1 | 3 |
| 400 °C (Fe) | 43 | 39 | 32 | 15 | 9 | 6 | 4 | 3 | 3 | 22 | 20 | 13 |
| 500 °C (Fe) | 17 | 11 | 4 | 3 | 1 | 1 | 9 | 6 | 2 | 37 | 4 | 0 |
| 300 °C (Cu) | 1 | 8 | 13 | 1 | 0 | 21 | 0 | 0 | 1 | 0 | 0 | 0 |
| 400 °C (Cu) | 28 | 20 | 17 | 20 | 24 | 8 | 8 | 5 | 3 | 11 | 2 | 1 |
| 500 °C (Cu) | 20 | 5 | 2 | 13 | 6 | 1 | 14 | 5 | 1 | 2 | 0 | 0 |
| StdSCR-C8H10 | HCHO | CH3CHO | C3H6 | HCN | ||||||||
| Temperature | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar | 1 bar | 3 bar | 5 bar |
| 300 °C (Fe) | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 400 °C (Fe) | 1 | 3 | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 |
| 500 °C (Fe) | 4 | 6 | 7 | 0 | 0 | 1 | 0 | 0 | 0 | 9 | 5 | 3 |
| 300 °C (Cu) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 400 °C (Cu) | 0 | 1 | 2 | 0 | 3 | 6 | 0 | 2 | 9 | 0 | 0 | 0 |
| 500 °C (Cu) | 1 | 2 | 4 | 0 | 4 | 6 | 1 | 2 | 6 | 0 | 0 | 0 |
| Fe-ZSM-5 | Cu-SSZ-13 | |
|---|---|---|
| Amount of ion exchanged metal [wt.%] | 1.5 | 1.7 |
| Surface area [m2 g−1] | 410 | 770 |
| Pore volume [mL g−1] | 0.2 | 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zengel, D.; Barth, S.; Casapu, M.; Grunwaldt, J.-D. The Impact of Pressure and Hydrocarbons on NOx Abatement over Cu- and Fe-Zeolites at Pre-Turbocharger Position. Catalysts 2021, 11, 336. https://doi.org/10.3390/catal11030336
Zengel D, Barth S, Casapu M, Grunwaldt J-D. The Impact of Pressure and Hydrocarbons on NOx Abatement over Cu- and Fe-Zeolites at Pre-Turbocharger Position. Catalysts. 2021; 11(3):336. https://doi.org/10.3390/catal11030336
Chicago/Turabian StyleZengel, Deniz, Simon Barth, Maria Casapu, and Jan-Dierk Grunwaldt. 2021. "The Impact of Pressure and Hydrocarbons on NOx Abatement over Cu- and Fe-Zeolites at Pre-Turbocharger Position" Catalysts 11, no. 3: 336. https://doi.org/10.3390/catal11030336
APA StyleZengel, D., Barth, S., Casapu, M., & Grunwaldt, J.-D. (2021). The Impact of Pressure and Hydrocarbons on NOx Abatement over Cu- and Fe-Zeolites at Pre-Turbocharger Position. Catalysts, 11(3), 336. https://doi.org/10.3390/catal11030336








