Precursor Effect on Mn-Fe-Ce/TiO2 Catalysts for Selective Catalytic Reduction of NO with NH3 at Low Temperatures
Abstract
:1. Introduction
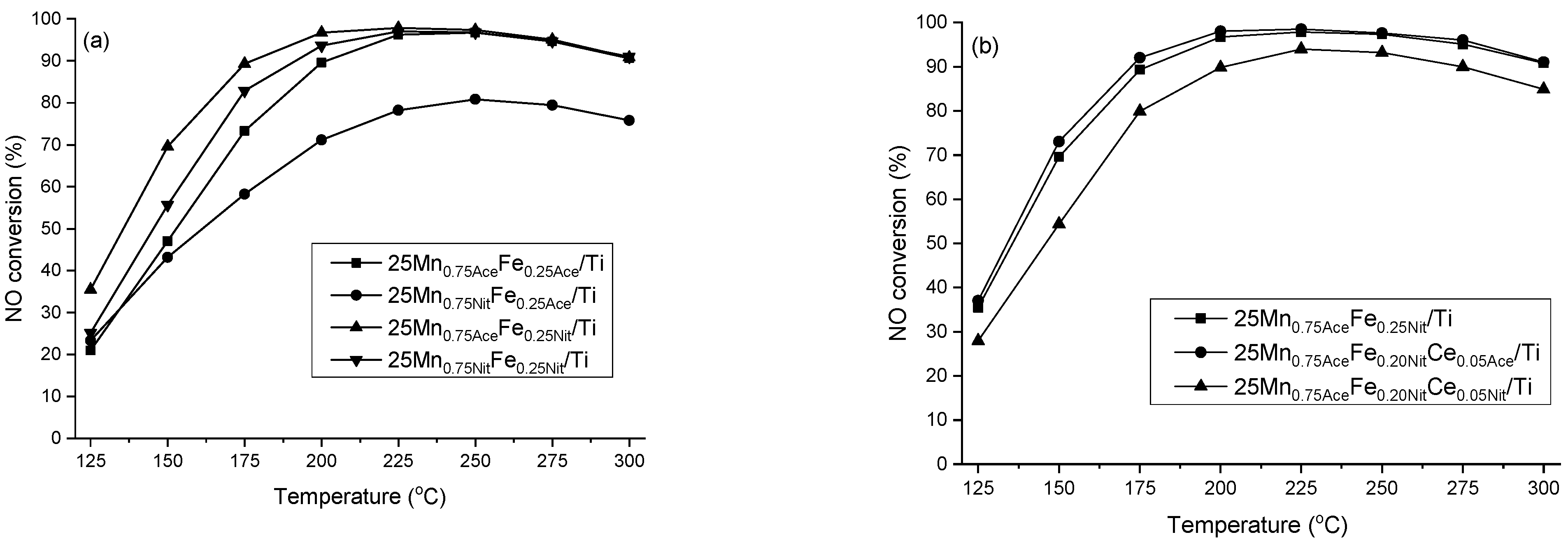
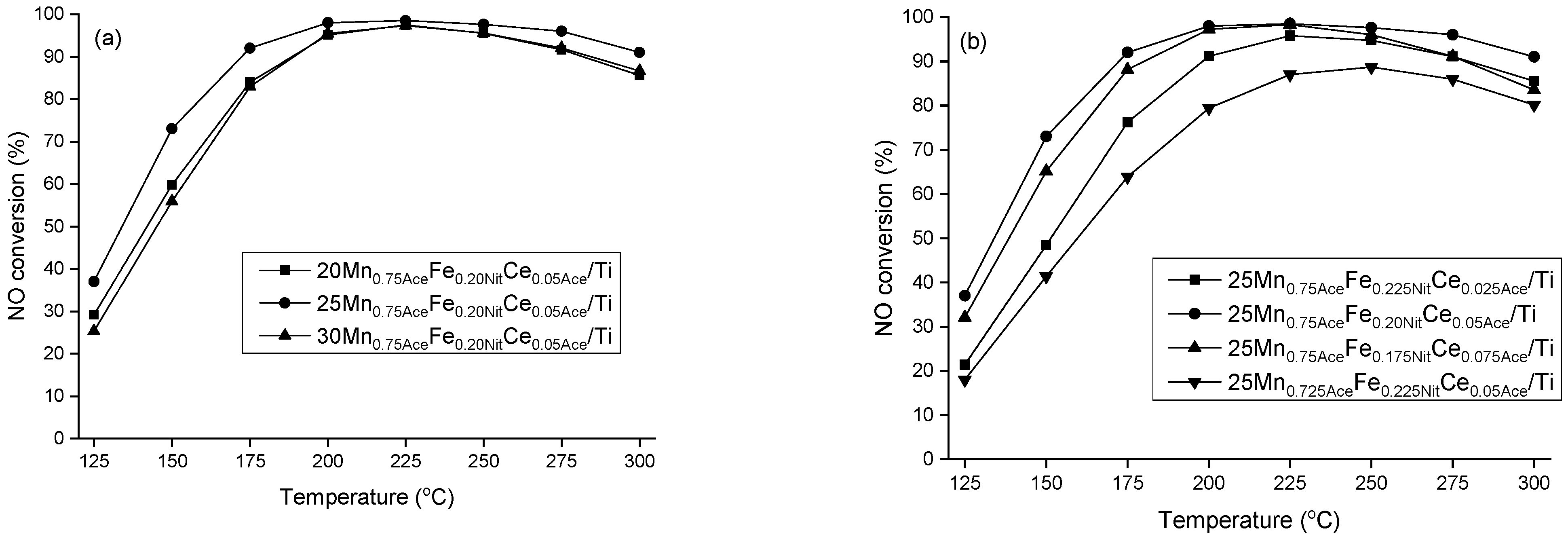
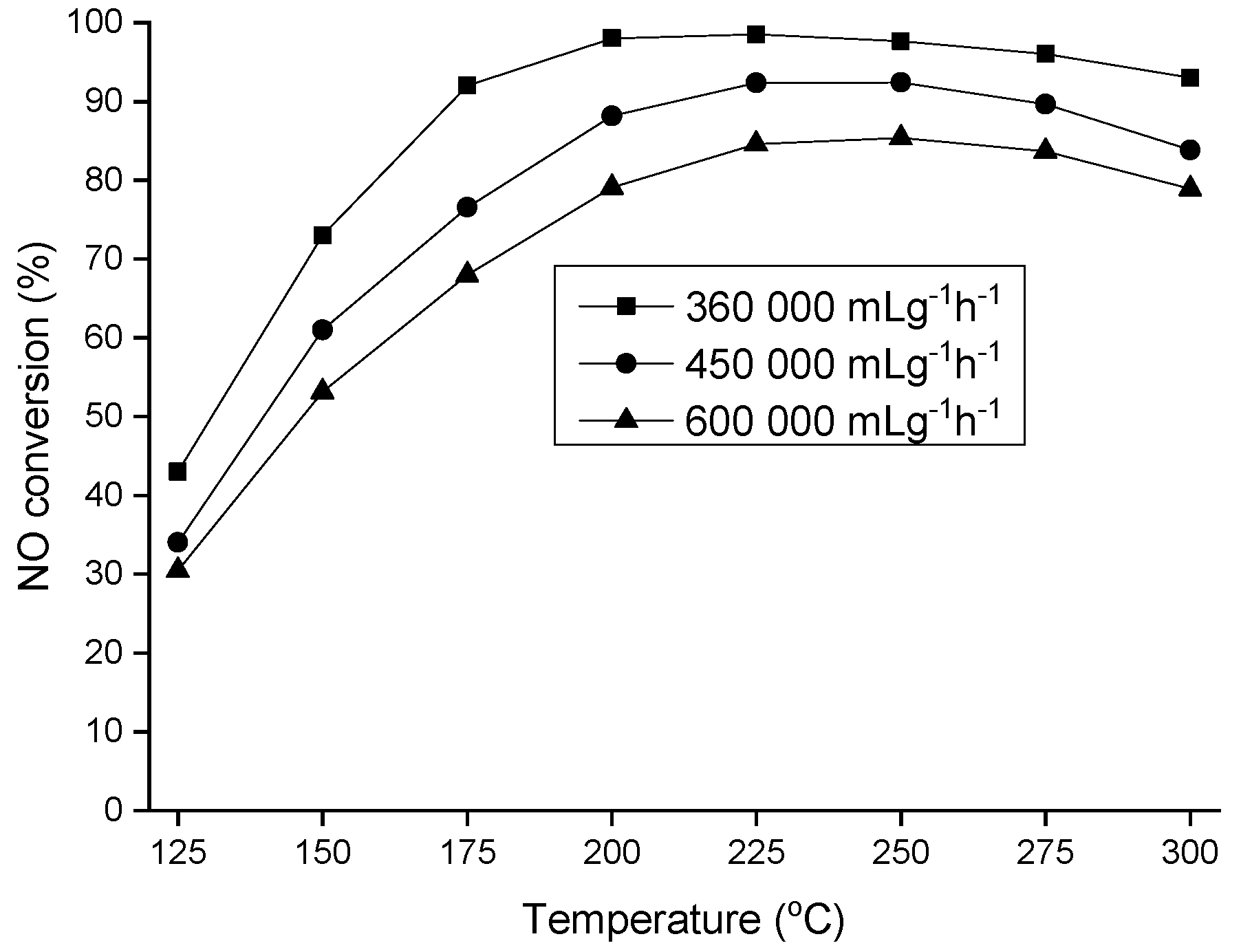
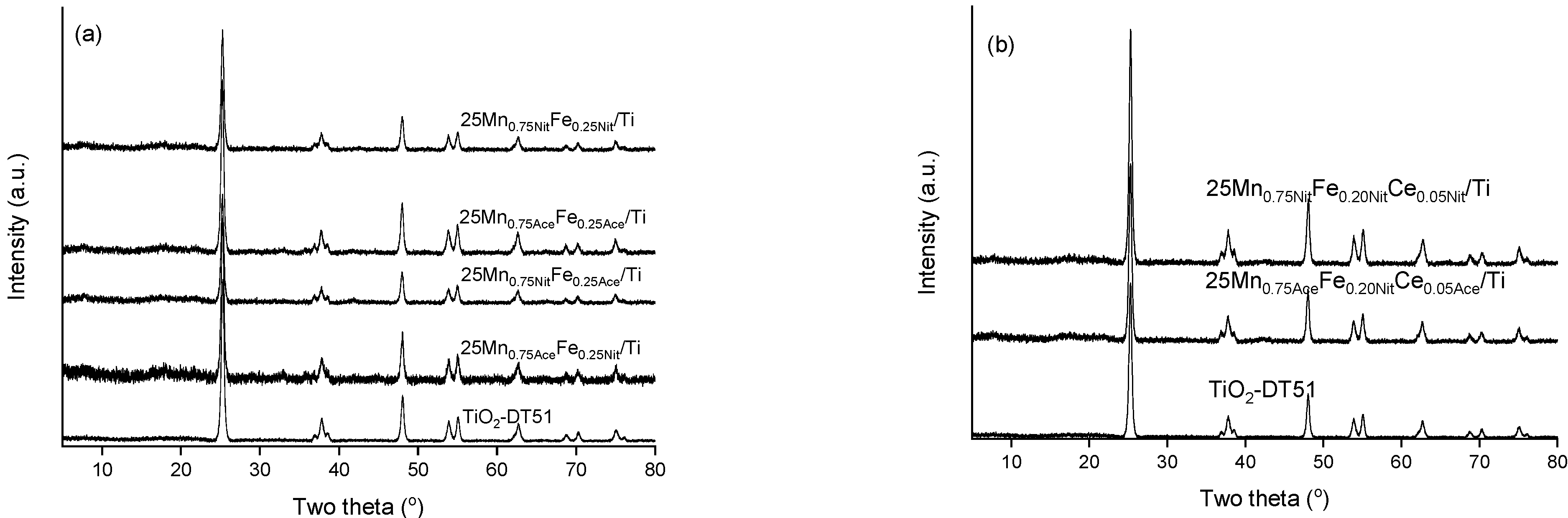
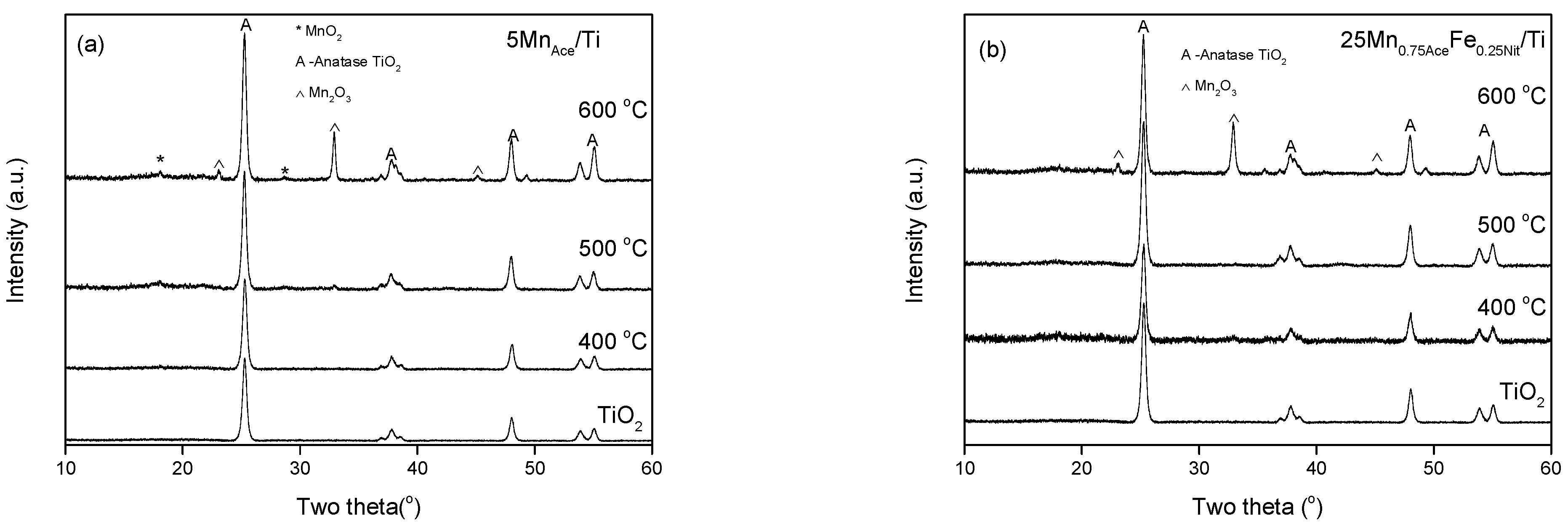
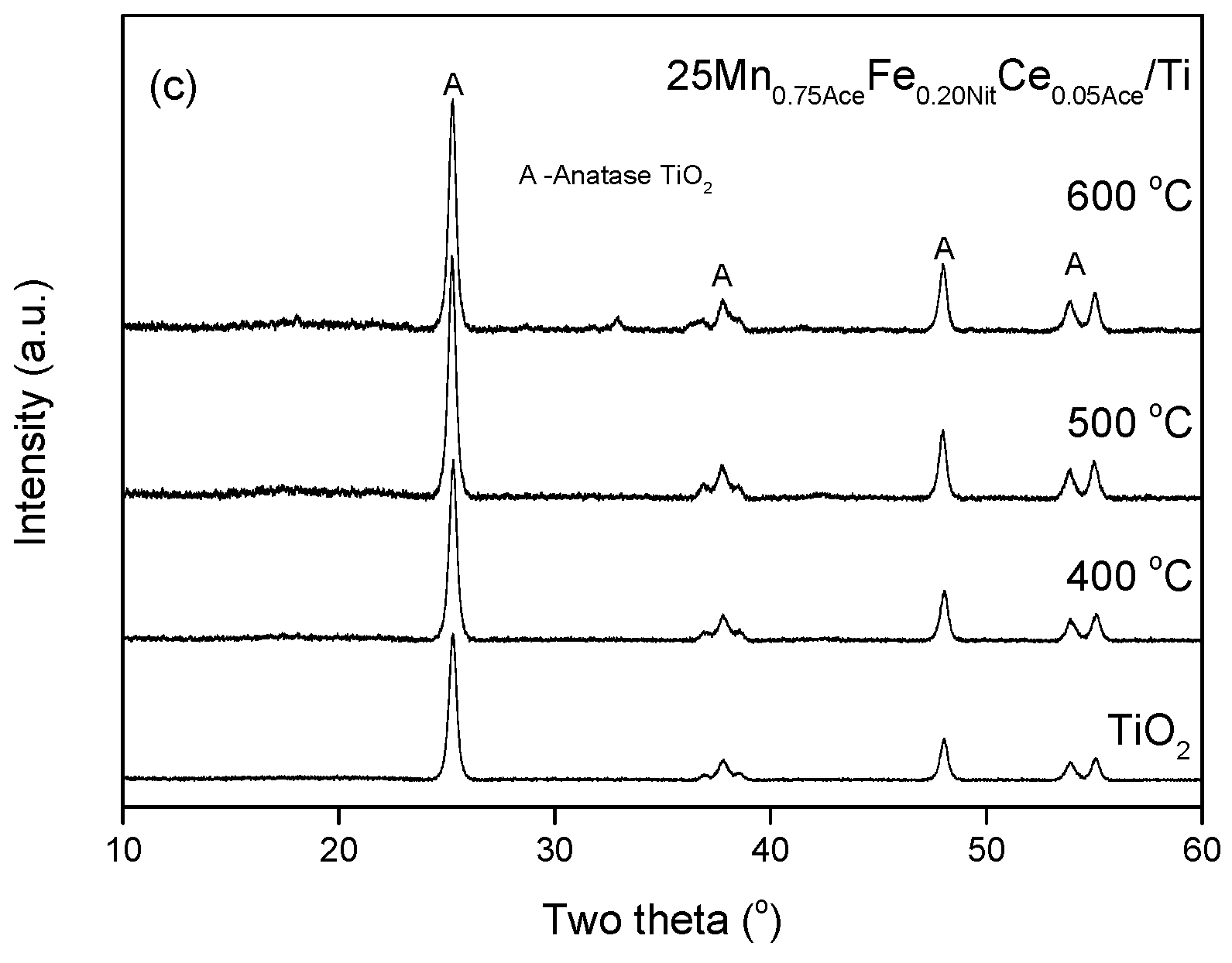
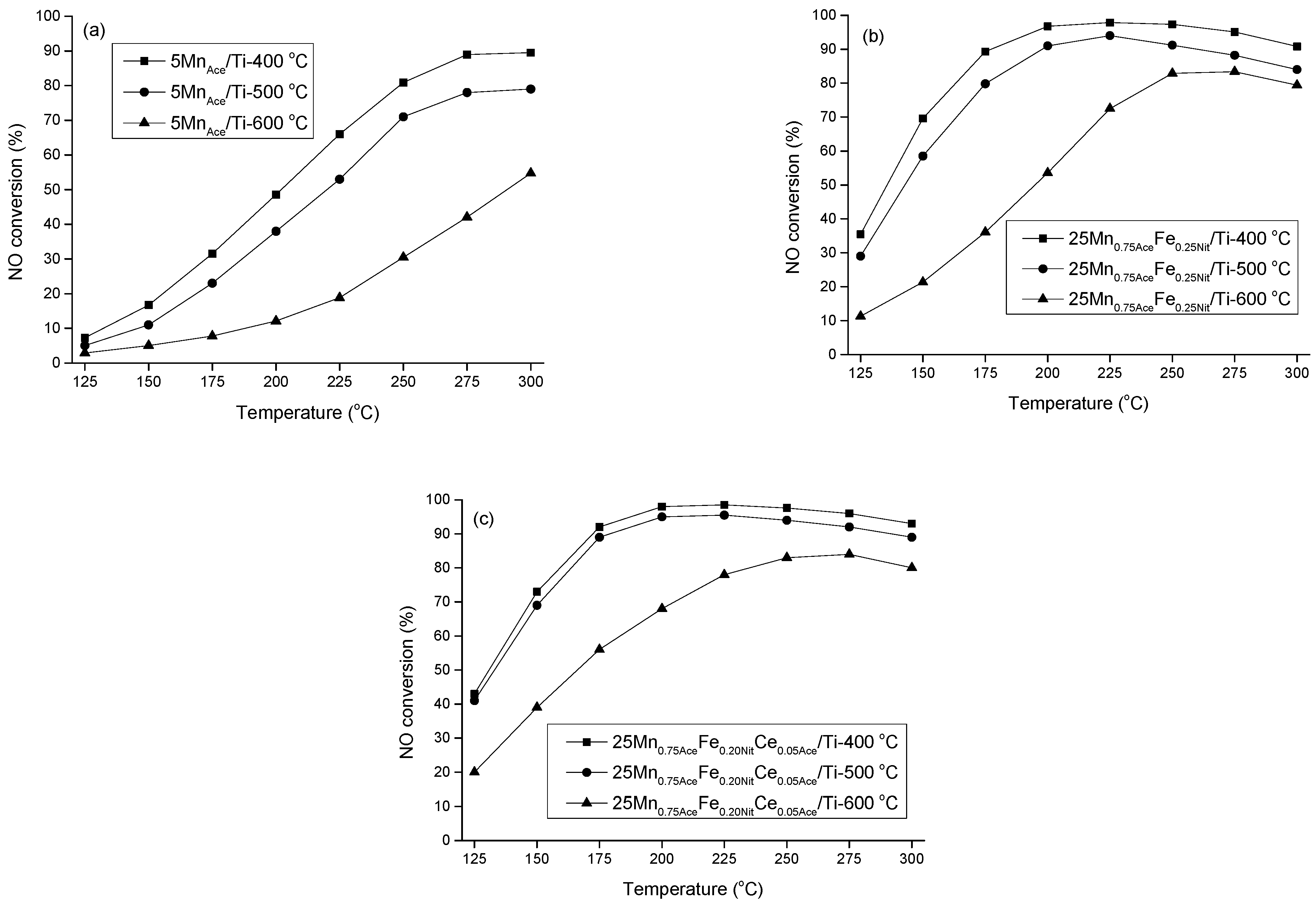
2. Results and Discussion
3. Experimental
3.1. Catalyst Synthesis
3.2. Catalyst Characterization
3.2.1. X-ray Powder Diffraction
3.2.2. Nitrogen Physisorption
3.2.3. Chemisorption
3.2.4. X-ray Photoemission Spectroscopy
3.3. Catalytic Activity Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bosch, H.; Janssen, F. Formation and control of nitrogen oxides. Catal. Today 1988, 2, 369–379. [Google Scholar]
- Parvulescu, V.I.; Grange, P.; Delmon, B. Catalytic removal of NO. Catal. Today 1998, 46, 233–316. [Google Scholar] [CrossRef]
- Busca, G.; Lietti, L.; Ramis, G.; Berti, F. Chemical and mechanistic aspects of the selective catalytic reduction of NOx by ammonia over oxide catalysts: A review. Appl. Catal. B 1998, 18, 1–36. [Google Scholar] [CrossRef]
- Alemany, L.J.; Lietti, L.; Ferlazzo, N.; Forzatti, P.; Busca, G.; Giamello, E.; Bregani, F. Reactivity and physicochemical characterization of V2O5-WO3/TiO2 De-NOx catalysts. J. Catal. 1995, 155, 117–130. [Google Scholar]
- Heck, R.M. Catalytic abatement of nitrogen oxides-stationary applications. Catal. Today 1999, 53, 519–523. [Google Scholar] [CrossRef]
- Due-Hansen, J.; Boghosian, S.; Kustov, A.; Fristrup, P.; Tsilomelekis, G.; Ståhl, K.; Christensen, C.H.; Fehrmann, R. Vanadia-based SCR catalysts supported on tungstated and sulfated zirconia: Influence of doping with potassium. J. Catal. 2007, 251, 459–473. [Google Scholar] [CrossRef]
- Putluru, S.S.R.; Jensen, A.D.; Riisager, A.; Fehrmann, R. Heteropoly acid promoted V2O5/TiO2 catalysts for NO abatement with ammonia in alkali containing flue gases. Catal. Sci. Technol 2011, 1, 631–637. [Google Scholar] [CrossRef]
- Putluru, S.S.R.; Kristensen, S.B.; Due-Hansen, J.; Riisager, A.; Fehrmann, R. Alternative alkali resistant deNOx catalysts. Catal. Today 2012, 184, 192–196. [Google Scholar] [CrossRef]
- Singoredjo, L.; Korver, R.; Kapteijn, F.; Moulijn, J. Alumina Supported Manganese Oxides for the Low-Temperature Selective Catalytic Reduction of Nitric-Oxide with Ammonia. Appl. Catal. B Environ. 1992, 1, 297–316. [Google Scholar] [CrossRef]
- Smirniotis, P.G.; Peńa, D.A.; Uphade, B.S. Low-temperature selective catalytic reduction (SCR) of NO with NH3 by using Mn, Cr, and Cu oxides supported on Hombikat TiO2. Angew. Chem. Int. Ed. 2001, 40, 2479–2482. [Google Scholar] [CrossRef]
- Long, R.Q.; Yang, R.T.; Chang, R. Low temperature selective catalytic reduction (SCR) of NO with NH3 over Fe-Mn based catalysts. Chem. Commun. 2002, 5, 452–453. [Google Scholar] [CrossRef]
- Wu, Z.; Jiang, B.; Liu, Y. Effect of transition metals addition on the catalyst for manganese/titania for low-temperature selective catalytic reduction of nitric oxide with ammonia. Appl. Catal. B 2008, 79, 347–355. [Google Scholar] [CrossRef]
- Roy, S.; Viswanath, B.; Hegde, M.S.; Madras, G. Low-temperature selective catalytic reduction of NO with NH3 over Ti0.9M0.1O2-δ (M= Cr, Mn, Fe, Co, Cu). J. Phys. Chem. C 2008, 112, 6002–6012. [Google Scholar] [CrossRef]
- Qi, G.; Yang, R.T. Low-temperature selective catalytic reduction of NO with NH3 over iron and manganese oxides supported on titania. Appl. Catal. B Environ. 2003, 44, 217–225. [Google Scholar] [CrossRef]
- Wu, S.; Yao, X.; Zhang, L.; Cao, Y.; Zou, W.; Li, L.; Ma, K.; Tang, C.; Gao, F.; Dong, L. Improved low temperature NH3-SCR performance of FeMnTiOx mixed oxide with CTAB-assisted synthesis. Chem. Commun. 2015, 51, 3470–3473. [Google Scholar] [CrossRef]
- Qi, G.; Yang, R.T.; Chang, R. MnOx-CeO2 mixed oxides prepared by co-precipitation for selective catalytic reduction of NO with NH3 at low temperatures. Appl. Catal. B Environ. 2004, 51, 93–106. [Google Scholar] [CrossRef]
- Shang, T.; Hui, S.; Niu, Y.; Liang, L.; Liu, C.; Wang, D. Effect of the addition of Ce to MnOx/Ti catalyst on reduction of N2O in low-temperature SCR. Asia Pac. J. Chem. Eng. 2014, 9, 810–817. [Google Scholar] [CrossRef]
- Jin, R.; Liu, Y.; Wang, Y.; Cen, W.; Wu, Z.; Wang, H.; Weng, X. The role of cerium in the improved SO2 tolerance for NO reduction with NH3 over Mn-Ce/TiO2 catalyst at low temperature. Appl. Catal. B Environ. 2014, 148–149, 582–588. [Google Scholar] [CrossRef]
- Putluru, S.S.R.; Schill, L.; Jensen, A.D.; Siret, B.; Tabaries, F.; Fehrmann, R. Mn/TiO2 and Mn-Fe/TiO2 catalysts synthesized by deposition precipitation-promising for selective catalytic reduction of NO with NH3 at low temperatures. Appl. Catal. B Environ. 2015, 165, 628–635. [Google Scholar] [CrossRef]
- Kapteijn, F.; Vanlangeveld, A.D.; Moulijn, J.A.; Andreini, A.; Vuurman, M.A.; Turek, A.M.; Jehng, J.M.; Wachs, I.E. Alumina-Supported Manganese Oxide Catalysts. J. Catal. 1994, 150, 94–104. [Google Scholar] [CrossRef]
- Li, J.; Chen, J.; Ke, R.; Luo, C.; Hao, J. Effects of precursors on the surface Mn species and the activities for NO reduction over MnOx/TiO2 catalysts. Catal. Commun. 2007, 8, 1896–1900. [Google Scholar] [CrossRef]
- Peña, D.A.; Uphade, B.S.; Smirniotis, P.G. TiO2-supported metal oxide catalysts for low-temperature selective catalytic reduction of NO with NH3: I. Evaluation and characterization of first row transition metals. J. Catal. 2004, 221, 421–431. [Google Scholar] [CrossRef]
- Huang, Z.; Gu, X.; Wen, W.; Hu, P.; Makkee, M.; Lin, H.; Kapteijn, F.; Tang, X. A “smart” hollandite deNOx catalyst: Self-protection against alkali poisoning. Angew. Chem. Int. Ed. 2013, 52, 660–664. [Google Scholar] [CrossRef]
- Smirniotis, P.G.; Sreekanth, P.M.; Peña, D.A.; Jenkins, R.G. Manganese oxide catalysts supported on TiO2, Al2O3, and SiO2: A comparison for low-temperature SCR of NO with NH3. Ind. Eng. Chem. Res. 2006, 45, 6436–6443. [Google Scholar] [CrossRef]
- Thirupathi, B.; Smirniotis, P.G. Co-doping a metal (Cr, Fe, Co, Ni, Cu, Zn, Ce, and Zr) on Mn/TiO2 catalyst and its effect on the selective reduction of NO with NH3 at low-temperatures. Appl. Catal. B Environ. 2011, 110, 195–206. [Google Scholar] [CrossRef]
- Kang, M.; Park, E.D.; Kim, J.M.; Yie, J.E. Manganese oxide catalysts for NOx reduction with NH3 at low temperatures. Appl. Catal. A Gen. 2007, 327, 261–269. [Google Scholar] [CrossRef]
- Tang, X.; Hao, J.; Xu, W.; Li, J. Low temperature selective catalytic reduction of NOx with NH3 over amorphous MnOx catalysts prepared by three methods. Catal. Commun. 2007, 8, 329–334. [Google Scholar] [CrossRef]
- Iwasaki, M.; Shinjoh, H. A comparative study of “standard”, “fast” and “nO2” SCR reactions over Fe/zeolite catalyst. Appl. Catal. A Gen. 2010, 390, 71–77. [Google Scholar] [CrossRef]
- Shen, B.; Liu, T.; Zhao, N.; Yang, X.; Deng, L. Iron-doped Mn-Ce/TiO2 catalyst for low temperature selective catalytic reduction of NO with NH3. J. Environ. Sci. 2010, 22, 1447–1454. [Google Scholar] [CrossRef]



| 2.3 vol.% H2O | 10 vol.% H2O | ||||
|---|---|---|---|---|---|
| Catalyst | NO Conv. (%) | N2O (ppm) | Sel. N2O (%) | N2O (ppm) | Sel. N2O (%) |
| 25Mn0.75AceFe0.25Nit/Ti | 69.6 | 35 | 5.0 | 0 | 0 |
| 25Mn0.75AceFe0.20NitCe0.05Ace/Ti | 73.0 | 15 | 2.1 | 0 | 0 |
| 25Mn0.75AceFe0.20NitCe0.05Nit/Ti | 54.4 | 20 | 3.7 | 0 | 0 |
| Catalyst | Surface Area | H2 Consumption | NO oxidation to NO2 |
|---|---|---|---|
| (m2/g) | (µmol/g) | (%) * | |
| 25Mn0.75AceFe0.25Nit/Ti | 95 | 4120 | 60 |
| 25Mn0.75NitFe0.25Nit/Ti | 91 | 4004 | 58 |
| 25Mn0.75NitFe0.25Nit/Ti | 100 | 3996 | 46 |
| 25Mn0.75NitFe0.25Ace/Ti | 100 | 3972 | 42 |
| 25Mn0.75AceFe0.20NitCe0.05Ace/Ti | 102 | 5040 | 66 |
| 25Mn0.75AceFe0.20NitCe0.05Nit/Ti | 96 | 4907 | 56 |
| Catalyst | Ti | Mn | Fe | Ce | Mn/Fe | Ot | Oα/Ot |
|---|---|---|---|---|---|---|---|
| 25Mn0.75AceFe0.25Nit/Ti | 14.5 | 8.8 | 4.1 | -- | 2.1 | 72.6 | 50.1 |
| 25Mn0.75AceFe0.20NitCe0.05Ace/Ti | 12.0 | 9.5 | 7.3 | 2.2 | 1.3 | 69.0 | 83.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Putluru, S.S.R.; Schill, L.; Jensen, A.D.; Siret, B.; Tabaries, F.; Fehrmann, R. Precursor Effect on Mn-Fe-Ce/TiO2 Catalysts for Selective Catalytic Reduction of NO with NH3 at Low Temperatures. Catalysts 2021, 11, 259. https://doi.org/10.3390/catal11020259
Putluru SSR, Schill L, Jensen AD, Siret B, Tabaries F, Fehrmann R. Precursor Effect on Mn-Fe-Ce/TiO2 Catalysts for Selective Catalytic Reduction of NO with NH3 at Low Temperatures. Catalysts. 2021; 11(2):259. https://doi.org/10.3390/catal11020259
Chicago/Turabian StylePutluru, Siva Sankar Reddy, Leonhard Schill, Anker Degn Jensen, Bernard Siret, Frank Tabaries, and Rasmus Fehrmann. 2021. "Precursor Effect on Mn-Fe-Ce/TiO2 Catalysts for Selective Catalytic Reduction of NO with NH3 at Low Temperatures" Catalysts 11, no. 2: 259. https://doi.org/10.3390/catal11020259
APA StylePutluru, S. S. R., Schill, L., Jensen, A. D., Siret, B., Tabaries, F., & Fehrmann, R. (2021). Precursor Effect on Mn-Fe-Ce/TiO2 Catalysts for Selective Catalytic Reduction of NO with NH3 at Low Temperatures. Catalysts, 11(2), 259. https://doi.org/10.3390/catal11020259








