Abstract
Catalytic hydrotreatment (HT) is one of the most important refining steps in the actual petroleum-based refineries for the production of fuels and chemicals, and it will play also a crucial role for the development of biomass-based refineries. In fact, the utilization of HT processes for the upgrading of biomass and/or lignocellulosic residues aimed to the production of synthetic fuels and chemical intermediates represents a reliable strategy to reduce both carbon dioxide emissions and fossil fuels dependence. At this regard, the catalytic hydrotreatment of oils obtained from either thermochemical (e.g., pyrolysis) or physical (e.g., vegetable seeds pressing) processes allows to convert biomass-derived oils into a biofuel with properties very similar to conventional ones (so-called drop-in biofuels). Similarly, catalytic hydro-processing also may have a key role in the valorization of other biorefinery streams, such as lignocellulose, for the production of high-added value chemicals. This review is focused on recent hydrotreatment developments aimed to stabilizing the pyrolytic oil from biomasses. A particular emphasis is devoted on the catalyst formulation, reaction pathways, and technologies.
1. Introduction
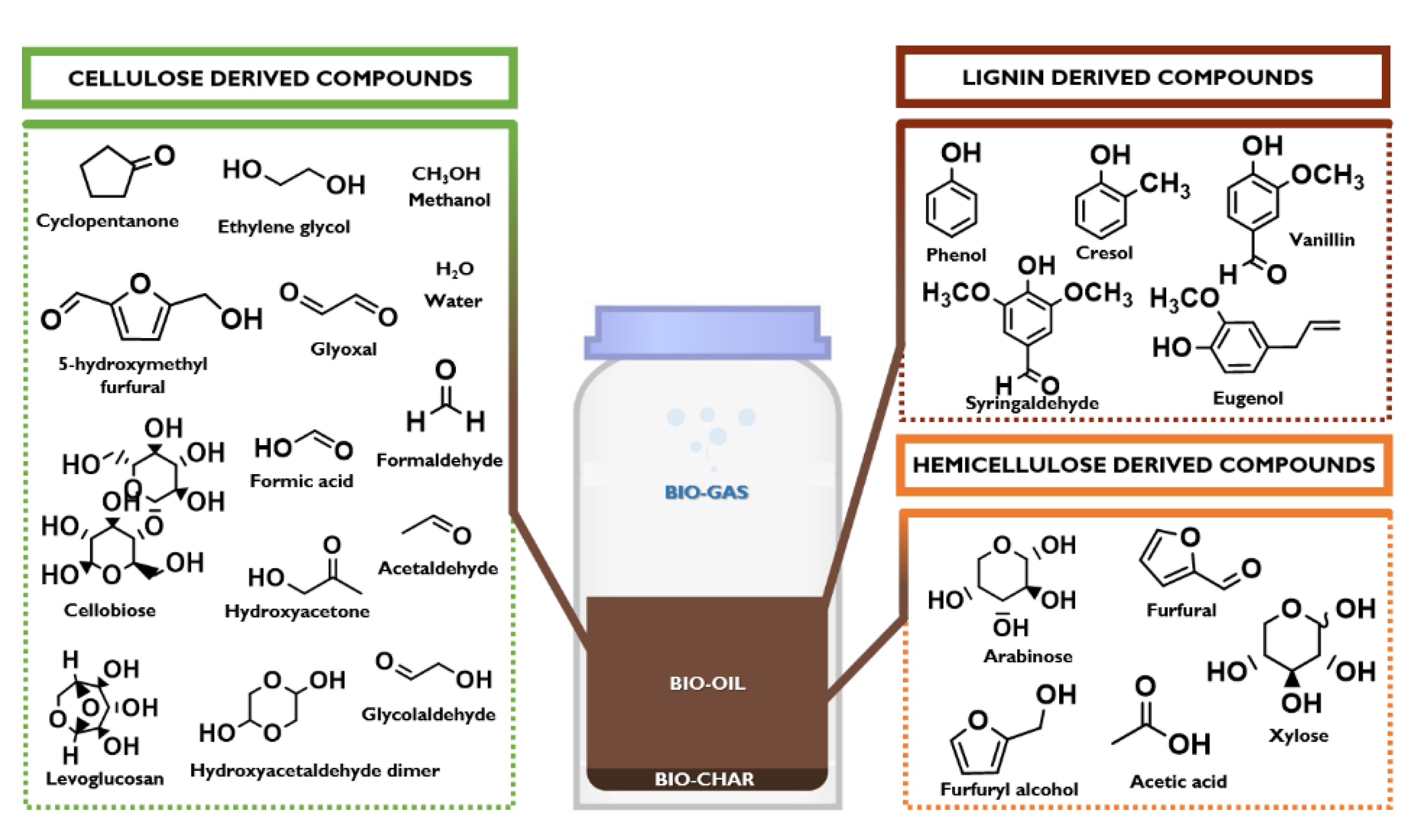
In a green and sustainable perspective, the world is moving from a strong fossil fuels’ dependence to a consistent use of renewable feedstocks. In this view, Anastas and Green proposed in 1998 “the 12 principles of green chemistry” [1], where a particular attention was also given to (second and third generation) transportation biofuels, chemicals, commodities, and pharmaceuticals directly produced from biomass in modern biorefineries [2,3,4,5,6]. This transition is given not only by the matured awareness that fossil resources are running out, but it is mostly accelerated by the United Nation decision to adopt the 2030 Agenda for Sustainable Development, a program action of 17 ambitious goals (SDGs) and 169 targets aimed to eradicate the poverty, to protect the planet, and to ensure the prosperity for all [7]. Biomasses, that currently supply about 80% of global renewable energy and a low-emissions character, represent a unique sustainable pathway to successfully address SDGs [1,7,8]. Among several technologies that can use biomass waste as the feedstock to produce energy fuels, power, heat, and various high value-added chemicals [9,10,11,12,13,14], an interesting example is the use of lignocellulose (plant based biomasses mainly composed of cellulose, hemicellulose, and lignin) and microalgae (biomasses with high protein and carbohydrate content characterized by the absence of lignin) for the production of bio-oil that can be used as intermediate for the production of liquid bio-fuels [15]. Bio-oil is a dark brown-red colored liquid, with a characteristic smell of smoke and a chemical composition strictly related to the biomass feedstocks containing a wide number of unique compounds generated from the rapid quenching of pyrolytic fragments of lignocellulose [16]. Figure 1 shows the main compounds present in the bio-oil: an aqueous solution of several products derived from the fragmentation of cellulose and hemicellulose and from the depolymerization of lignin. The mixture consists of various organic compounds (20–30 wt%), water (19–20 wt%), water-soluble oligomers (WS, also known as pyrolityc humin), and water-insoluble oligomers (WIS, also known as pyrolityc lignin) (43–59 wt%) that can be efficiently used for several applications, such as drop-in fuel, production of chemicals, and various carbon-based materials [17,18,19,20,21,22].
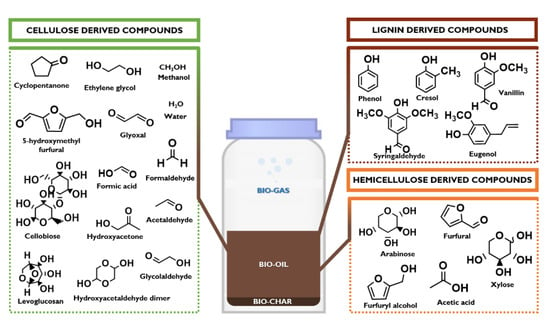
Figure 1.
A simplified chemical composition of bio-oil: main lignocellulose-derived compounds.
Conventionally, bio-oil is produced by using a high energy demanding multistep process, such as pyrolysis (fast or slow, thermal or thermo-catalytic) and hydrothermal liquefaction (HTL). These thermochemical processes are conducted in the absence of oxygen and at high reaction temperature with the aim to allow the decomposition/depolymerisation of lignocellulose and microalgae into a bio-oil liquid (the major product), solid (bio-char), and gaseous products (CO2, CO, CH4, H2) (bio-syngas [15,23,24]. Bio-char can access applications in several fields (e.g., soil amendment in agriculture, chemical sensing, adsorbent material in wastewater remediation) or combusted to recover energy for the pyrolysis stage [25], while bio-syngas may be directly utilized for many energy uses (e.g., electricity generation, fuel for transport, cooking fuel, feedstock for fuel cells) [26]. HTL processes were developed to improve the efficiency of direct thermal decomposition methods and differ from the pyrolysis for the adoption of lower reaction temperatures and for the presence of a homogeneous or heterogeneous catalyst by applying water and simple aliphatic (e.g., methanol, ethanol, and 2-propanol) alcohols used as such or in combination as reaction solvents. However, bio-oils arising from these two processes cannot be directly used as drop-in fuels in conventional engines due to problems related to the presence of a common limiting feature, the high oxygen content of biomass otherwise responsible of chemical unfavorable properties of bio-oil (high acidity, high viscosity, thermal and chemical instability) [16].
Thus, a biorefinery process in which biomass is first converted in bio-oil by pyrolytic or HTL step followed by an oxygen removal stage represents a most promising approach for the production of biofuels and chemicals.
To this regard, in order to mitigate the oxygen content and to improve the bio-oil properties for practical use, some catalytic approach (catalytic cracking, hydrodeoxygenation HDO, etc.), based on a thermal-catalytic treatment of biomass (hydrotreatment or hydrotreating process), come to help. Among them, one of the most promising strategies is the catalytic hydrodeoxygenation (HDO) that allows the reduction of oxygen content providing, at the same time, the highest C-atom efficiency.
Although this process allows to obtain bio-oil in a high yield, the formation of a variable quantity of coke remains a problem to be solved. In this context, it was reported that the presence of a suitable catalyst in a two-step biorefinery process can reduce the formation of coke by improving, at the same time, the bio-oil properties [27,28]. The first step (or stabilization step) permits the transformation of carbonyl and carboxyl functional groups into alcohols promoted by noble metals catalysts (Pt, Ru, and Pd) in a temperature range between 100 and 300 °C. The second step is conducted between 350 and 400 °C and is driven by sulphide conventional catalysts that allows to completely remove oxygen species.
This review aims to provide a brief overview on recent advances in the catalytic hydrogenation process of bio-oil arising from thermal treatment of lignocellulosic biomass and microalgae, highlighting progresses made in terms of enhancing catalyst efficient activity for upgrade bio-oil HDO.
2. Bio-Oil Proprieties
Bio-oil is the main product of biomass pyrolysis. Historical documents report that this process was already used in ancient Egypt to prepare sealants for boats and ointments. In the 18th century, wood distillation provided compounds such as soluble tar, pitch, creosote oil, as well as chemical and non-condensable gases. Interest in biomass pyrolysis was revived in the 1980s, when the process was perfected to have a high yield of liquid compound [29]. The pyrolysis process carried out with a temperature between 400 °C and 600 °C and varying the residence time and heating rate, the product distribution changes. To maximize the process in term of liquid yield, the fast pyrolysis at ~500 °C is usually preferred, advantageously producing a liquid yield up to three times larger than the conventional and slow pyrolysis [30].
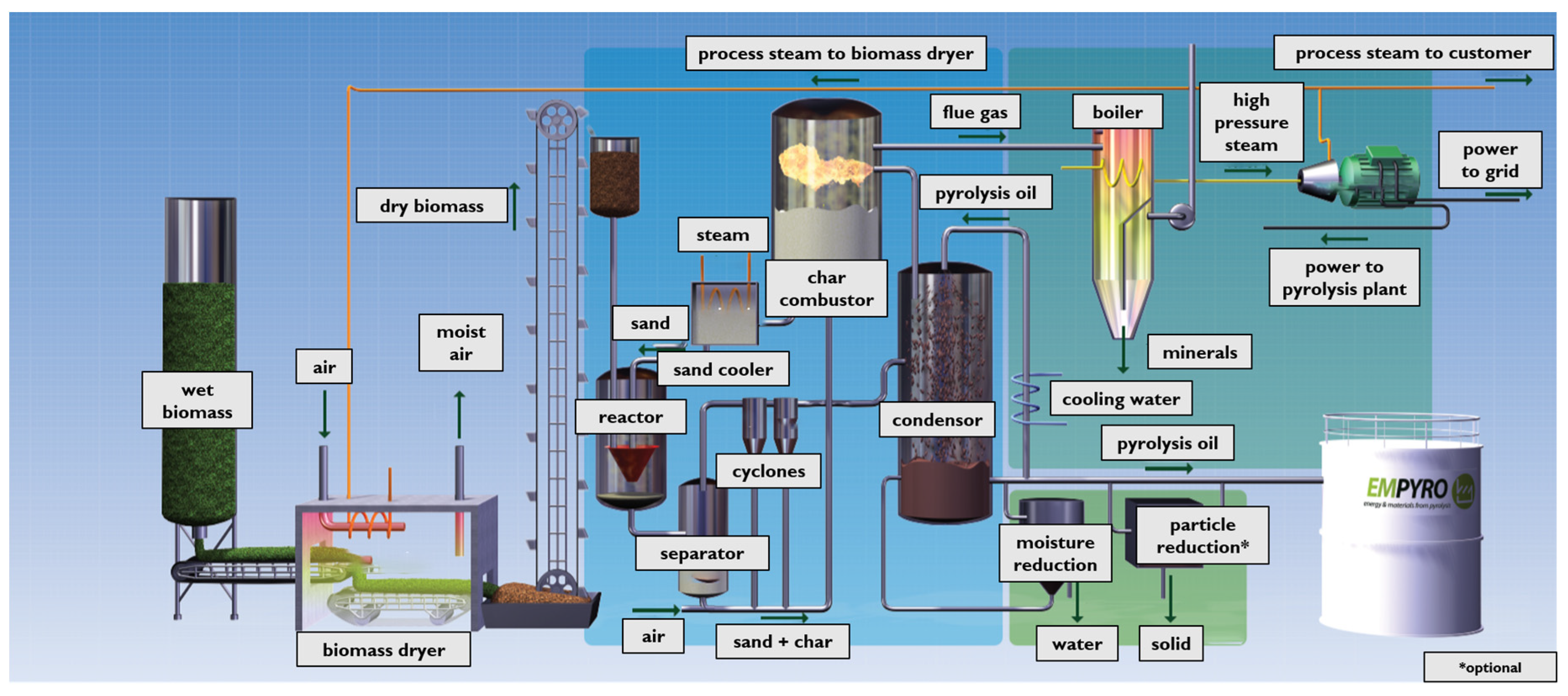
As an example, Figure 2 shows the flow diagram of the BTG Bi-oliquids BV pyrolysis plant [31]. The first part of the plant consists in a drying unit where biomass from different origin (for example, wood, rice husk, bagasse, sludge, tobacco, energy crops, palm-oil residues, straw, olive stone residues, chicken manure) is dried to decrease the water content. The dry biomass, in presence of a hot carrier (sand), is then converted in a fluidized bed reactor into pyrolysis oil, gas, and char. After that, the products and the sand are separated from the vapor/gas phase by a series of cyclones. Then, the char and sand fraction is moved to a fluid bed combustor, where the char is used to heat the sand recycled in the fluidized bed. The vapor/gas phase is instead quenched by re-circulated oil to divide the bio-oil from the incondensable gases, where the latter are captured as high-pressure steam and utilized in a steam turbine system.
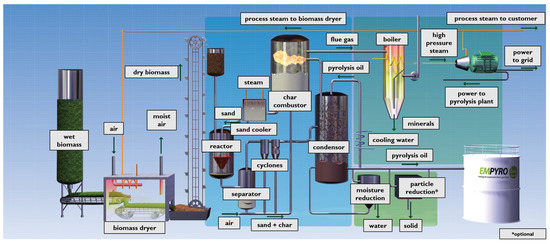
Figure 2.
Pyrolysis plant. With permission from BTG Bioliquids BTG bioliquids BV [31].
Furthermore, recent researchers are focusing their attention to microalgae as feedstock for fast pyrolytic reaction [32]. Microalgae are classified as third-generation biofuel due to their fast growth cycle and high lipid content (~50%), easily converted in fuels. Moreover, microalgae do not require arable land and are adaptable at different water sources, including wastewater. The pyrolysis of microalgae is usually carried out in presence of a catalyst, such as zeolites, aluminosilicates, transitional metal-loaded zeolites, MOFs, silica gel [33,34,35]. The pyrolysis process for microalgae may be performed as (i) one-pot step process, where microalgae and catalyst are mixed together (ii) or a double-step process, the pyrolysis vapors from microalgae are swept over a catalyst at a specific temperature [36,37].
The fast pyrolysis of biomass produces hundreds of different compounds (Table 1), where their composition depends of the cellulose, hemicellulose, lignin, and extractive content in the respective feedstock. The influence of biomass composition on bio-oils composition can be appreciated from the variability of the bio-oils elemental composition reported in Table 2, where the C content can vary from 39% (pine sawdust) to about 60% (beech wood) under the same pyrolysis conditions. Furthermore, the operating conditions of the fast pyrolysis influence the bio-oil composition [38,39,40].

Table 1.
Bio-oil composition.

Table 2.
Feedstock composition updated from [35].
3. Catalytic Hydrogenation of BIO-Oil
In refineries, the hydrogenation reactions are common operations to limit the presence of oxygen, nitrogen, sulphur, olefins, and aromatics. The reaction is generally catalyzed by molybdenum together with Ni or Co supported by γAl2O3. The operating conditions depend on the type of feed: LHSV 0.2 to 8.0, H2 circulation from 50 to 675 Nm3/m3, H2 pressure between 14 and 138 bar, and temperatures between 290 and 470 °C [55]. Actually, there are not industrial processes for HDO of bio-oil, but several catalysts have been tested from noble metals to Ni and Co, in presence of acid supports such as Al2O3 and SiO2, or C, in the temperature range 150–500 °C, pressure range between 2 and 200 bar [47]. In this section, recent advances on catalysts for HDO of bio-oil are summarized. Furthermore, technical aspects of emergent technologies (e.g., membrane reactors) for hydroprocessing are also discussed.
3.1. Catalysts
Hydro-processing is conventionally catalyzed in presence of metals from group VIII, such as nickel, palladium, and platinum [56]. Furthermore, group VIB metals (tungsten and molybdenum) have also been used for oxygen removal, since they are resistant to attack by oxygen, acids, and alkalis [57,58]. According to Masel [59], hydrogen is reactive in the surfaces of Co, Ni, Ru, Rh, Pd, Os, Ir, Pt as well as on Sc, Ti, V, Y, Zr, Nb, Mo, La, Hf, Ta, W, Cr, Mn, Fe, Tc, and Re. A slower uptake of hydrogen was observed with Cu [59]. Some authors increased the catalyst activity adding a second metal in order to promote an efficient adsorption of hydrogen at low temperature [60,61]. The most used supports were alumina-silica, carbon, titania (rutile), and zirconia (monoclinic form). Activated carbon is a well-known high-surface area (typically ~1000 m2/g) support material, which has been shown to be stable in hot water processing environments; rutile titania and monoclinic zirconia have lesser surface area (typically 30–80 m2/g) but have also demonstrated their utility as catalytic metal support and have been used in the hot water processing environment [62,63,64]. A possible pathway for upgrading bio-oils is represented by hydrogenation reactions in liquid phase, with the conversion of aldehydes, ketones, sugars, phenols, etc., in more stable alcohols. In order to improve the conversion of the bio-oils compounds and enhance the selectivity on desired products, several catalysts have been studied (Table 3). Interesting is the work of Wei et al. [65], where Pt over different ceria-zirconia supports were evaluated for the hydrogenation of cinnamaldehyde at 10 bar and 60 °C, obtaining a conversion in the range of 60–95%.

Table 3.
Hydrogenation reaction.
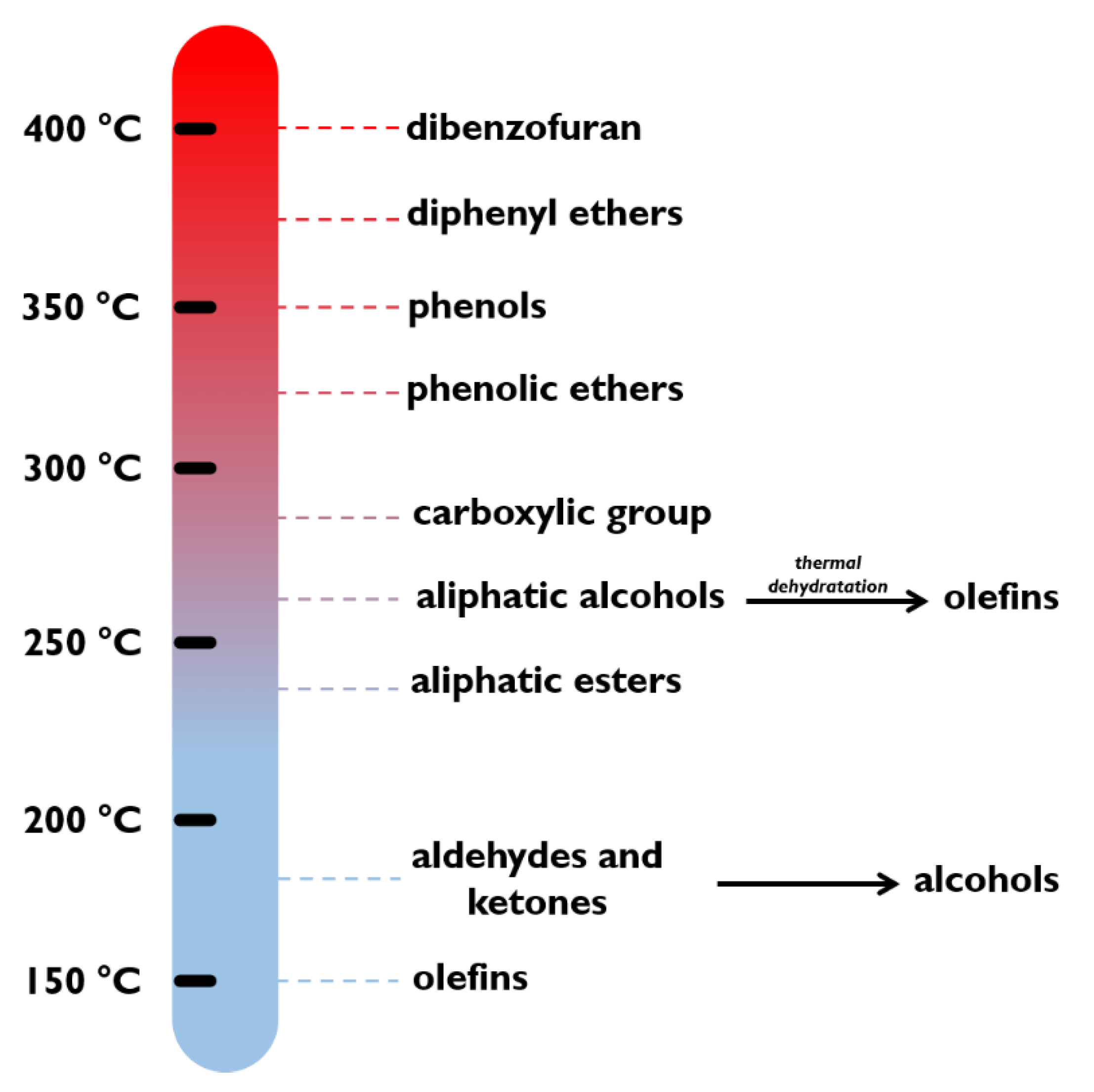
Liao et al. [77] used CeO as support with different metals (Ni, Co, and Cu) for the hydrogenation in liquid phase of maleic anhydride at 50 bar and 210 °C, converting all the reactant after 60, 180, and 420 min, for Ni, Co, and Cu, respectively. Elliott al. [78] elaborated a reactivity scale of hydrogenation of different organic compounds in presence of CoMo and NiMo sulphided catalysts (see Figure 3) based on literature work [79]. Olefins, aldehydes, and ketones were hydrogenated at low temperatures as low as 150–200 °C, while the alcohols at 250–300 °C. Carboxylic and phenolic ethers reacted at around 300 °C.
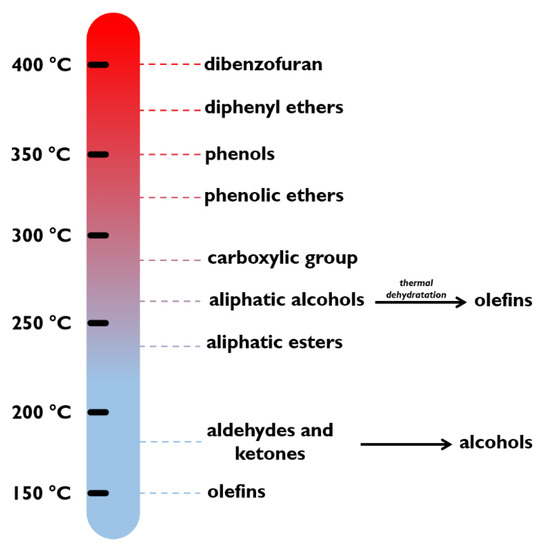
Figure 3.
Reactivity scale of organic compounds under hydrotreatment conditions. Adapted with permission from [78].
Recently, copper catalysts have attracted much attention for the conversion of glycerol to propylene glycol because of their intrinsic ability to selectively cleave the C-O bonds in glycerol rather than the C-C bonds. To increase the activity of Cu metal, Cu-based catalysts such as Cu-Cr, Cu-Al, and Cu-Mg have been developed to promote the hydrogenolysis reaction. Bienholz et al. prepared a highly dispersed silica-supported copper catalyst (Cu/SiO2) using an ion-exchange method and achieved 100% glycerol conversion with 87% propylene glycol selectivity at optimum conditions of 5 mL/h of 40 wt% aqueous glycerol solution, 255 °C, and 300 mL/min of H2 at 15 bar [80]. Liu’s group studied the glycerol hydrogenolysis over Ru-Cu catalysts supported on different support materials including SiO2, Al2O3, NaY zeolite, TiO2, ZrO2, and HY zeolite. The best activity was observed for Ru-Cu/ZrO2 with 100% glycerol conversion and 78.5% propylene glycol selectivity. The high activity of this catalyst was attributed to the synergistic effect of Ru in the catalyst related to hydrogen spill-over, while the high selectivity was attributed mainly to the low acidity of the support and the Cu amount [81].The HDO of the Water soluble fraction of Bio-Oil (WBO) at different temperatures (220, 270, and 310 °C) at 190 bar, using 5 wt% Ru/C catalyst, was studied by de Miguel Mercader et al. [82], where the recovery of carbon in oil phase increased from 16.3 wt% to 38.5 wt%, when the temperature was increased from 220 to 310 °C. In another study, several lignin model compounds (phenol, m-cresol, anisole, guaiacol, and diphenyl ether) were tested for HDO reactions in presence of MoO3 at atmospheric pressure and temperature between 150 and 250 °C [83]. The authors noted that, according to the bond dissociation energy, the highest catalytic reactivity was obtained with diphenyl ether, but important carburization phenomena have been noted onto the catalyst surface.
Bagnato et al. [84] prepared by impregnation technique a series of monometallic and bimetallic metal catalysts in which the zirconia was doped with Pd and not noble metals (Cu and Fe), characterized, and their performances studied in term of conversion and selectivity for key bio-compounds. Vanillin was completely converted after 80 min at 100 °C and 50 bar, in presence of PdFe/ZrO2. Meanwhile, the PdFe reached the conversion of 65.5% and 20% for furfural to furfuryl alcohol and glucose to sorbitol (74% selectivity), respectively.
The authors noted that the bimetallic catalyst was able to improve the conversion than the monometallic, mainly due the adsorption mechanism onto the catalytic surface: the not noble metal favoured the bonding to the aldehyde group, while the noble metal favoured the hydrogen molecule adsorption.
Bergem et al. [67] investigated the HDO of a model WBO using Ru/TiO2 and Ru/C catalysts in a packet bed reactor (PBR) at a temperature between 100 and 140 °C, ~62 bar. A completed conversion was noted already a 100 °C for compounds such as acetone, acetaldehyde, propionaldehyde, 2-propen-1-ol, 1-hydroxy-2-butanone, 3-hydroxy-2-butanone, 2-hexanone, and 2-furanone. Other compounds such as furfural and hydroxyacetaldehyde required elevate temperature (>140 °C) for converting completely. Furthermore, the authors observed a decrement of catalyst activity, about 25% after 90 h, due at acid leaching. Sanna et al. [85] studied the HDO of a real WBO in presence of Ru/C and Pt/C catalysts in a two-stage continuous reactor. In the first stage, the reaction was carried out in presence of Ru/C catalyst at 125 °C, while in the second stage, it was carried out at a temperature between 200 and 250 °C with Pt/C, at 50 and 100 bar, and different weight hourly space velocities from 0.75 to 6 h−1. During the first low temperature stage, the unstable bio-oil functionalities were stabilized into alcohols, where the main products were ethylene glycol, propylene glycol, and sorbitol, losing 7% of carbon as gas and solid phase. Furthermore, the catalyst showed a constant activity for about 80 h. In the second-high temperature stage, 45% of the carbon was converted in gasoline blend stocks and C2 to C6 diols.
3.2. Kinetic Mechanism
The reactions involved during the hydrotreating of bio-oil have been widely studied [86,87,88], as shown below:
In the following section, the reaction mechanisms of some of the most representative bio-oil compounds will be discussed.
3.2.1. Phenol
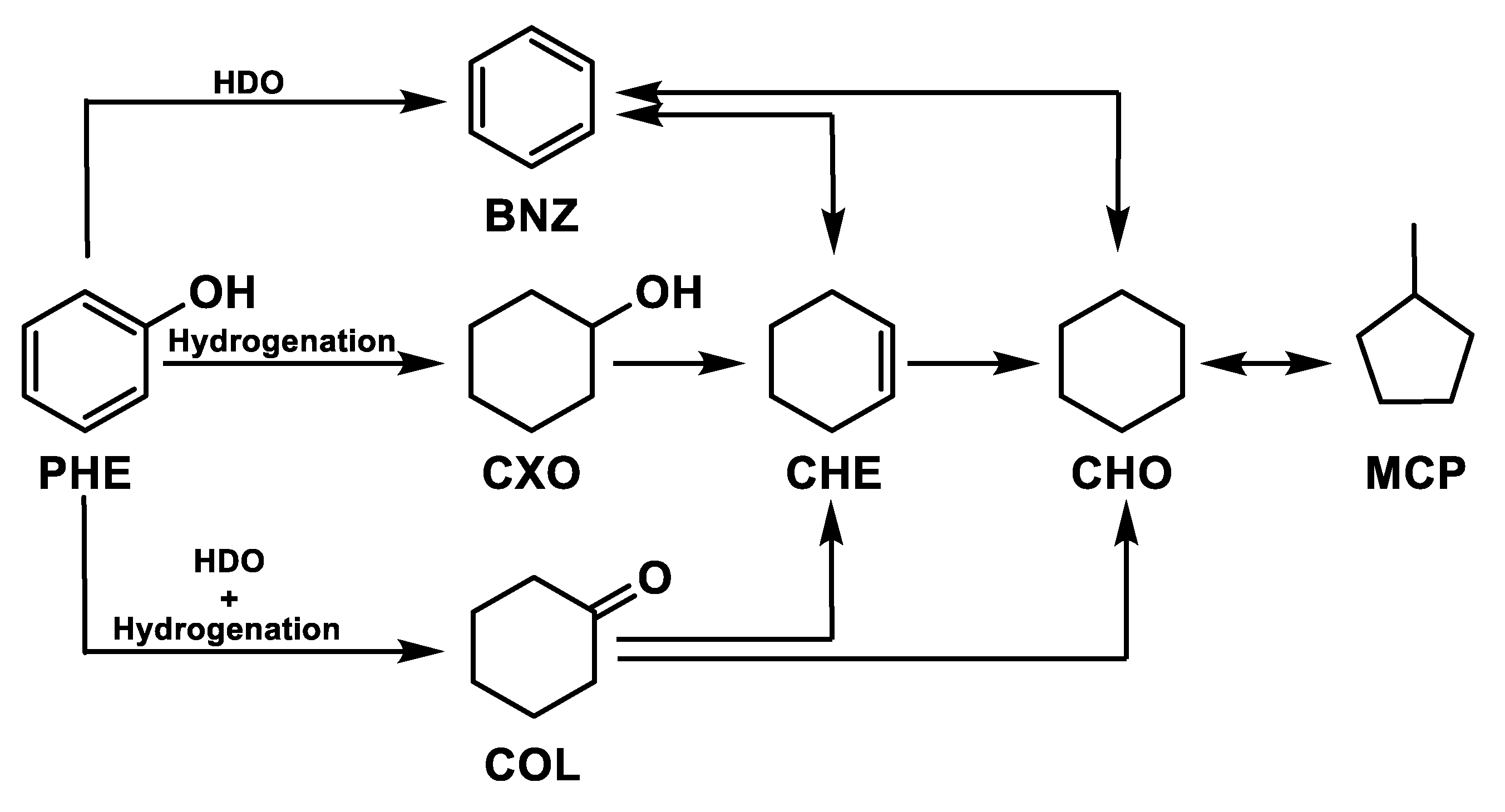
The phenol hydrogenation has been widely studied [89,90,91,92,93]. The reaction pathways are shown in Figure 4, where hydrogen reacts with the phenol (PHE) attacking the hydroxyl group to produce benzene with subsequent production of cyclohexene (CHE) and cyclohexane (CHO). Another reaction pathway of the aromatic ring is the formation of cyclohexanol (CXO) with consecutive hydrogenation in cyclohexene and cyclohexane. A further reaction pathway is represented by the formation of cyclohexanone (COL) with subsequent cyclohexanol hydrogenation in cyclohexene and cyclohexane. Finally, methylcyclopentane (MCP) can be produced by isomerization reaction.
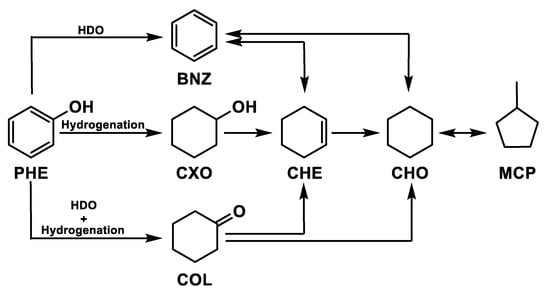
Figure 4.
Phenol hydrogenation pathways.
3.2.2. Guaiacol
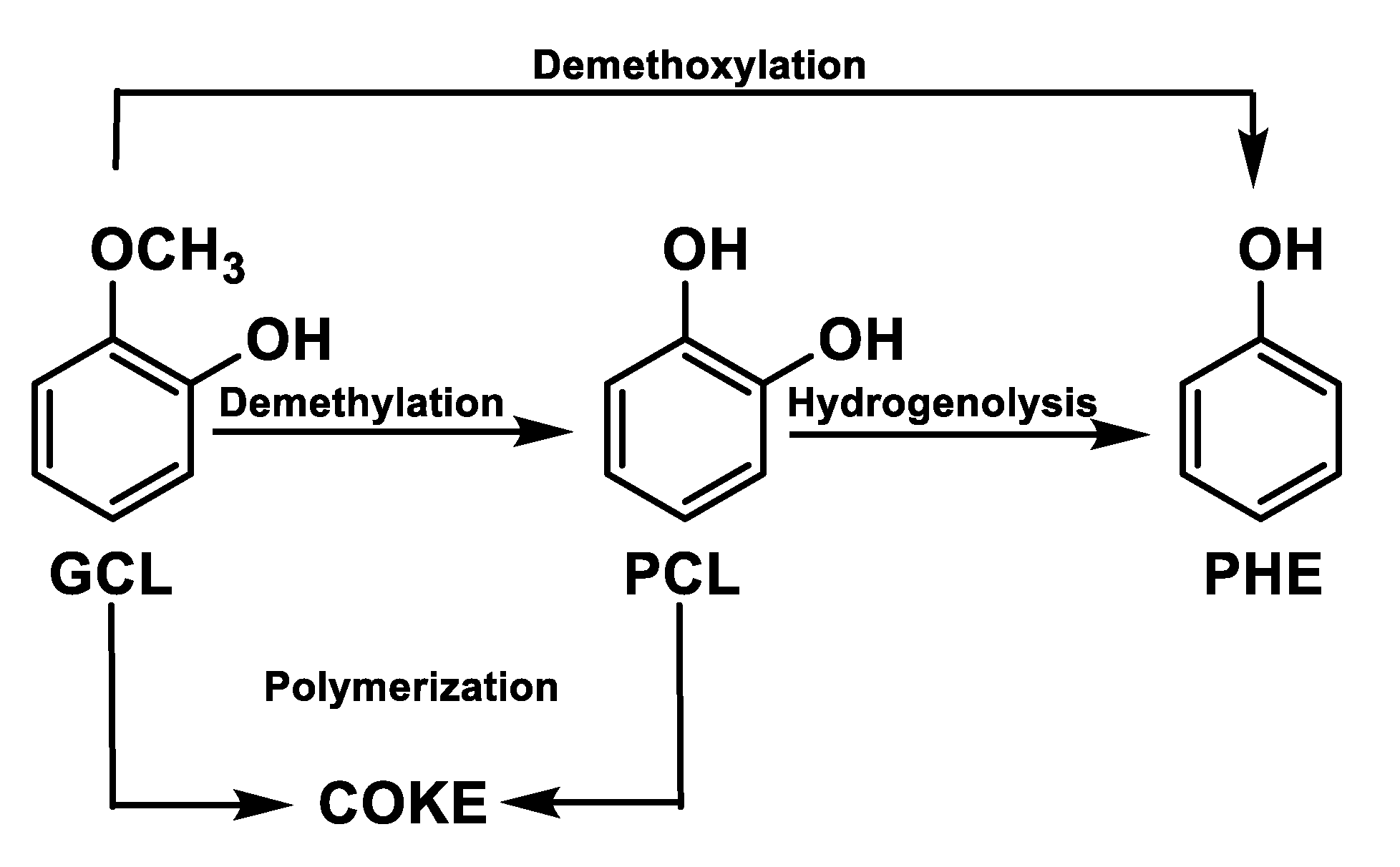
Another representative compound in bio-oil is guaiacol that reacts forming phenol [39,40,41,42,43] via two paths: (1) direct demethoxylation; and (2) indirect reaction through demethylation to catechol with subsequent hydrogenation of the latter compound. The undesired polymerisation of guaiacol (GCL) (Figure 5) leads to coke formation.
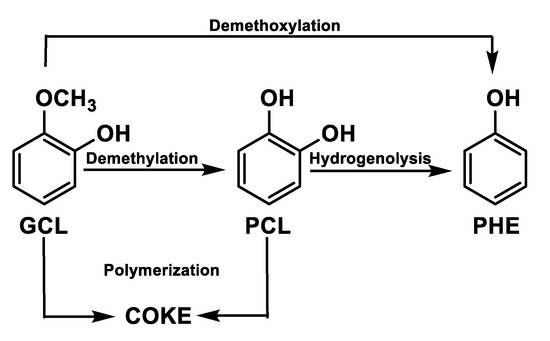
Figure 5.
Guaiacol reaction path.
Bindwal et al. [94] proposed a kinetic rate for the hydrogenation of guaiacol in 1,2 cycloexanediol in presence of 5% Ru/C catalyst according to the Langmuir–Hinshe–wood–Hougen–Watson (LHHW) model. The authors, according to the experimental data obtained, identified the limitation step for the reaction taking place on the catalyst surface, assuming the dissociative adsorption of H2. The reaction rate was described by the following equation:
where CB CH2 are the molar concentration of guaiacol and hydrogen, respectively, k3,a the kinetic constant, KB and KH2 are the adsorption constant of guaiacol and hydrogen.
3.2.3. Levoglucosan
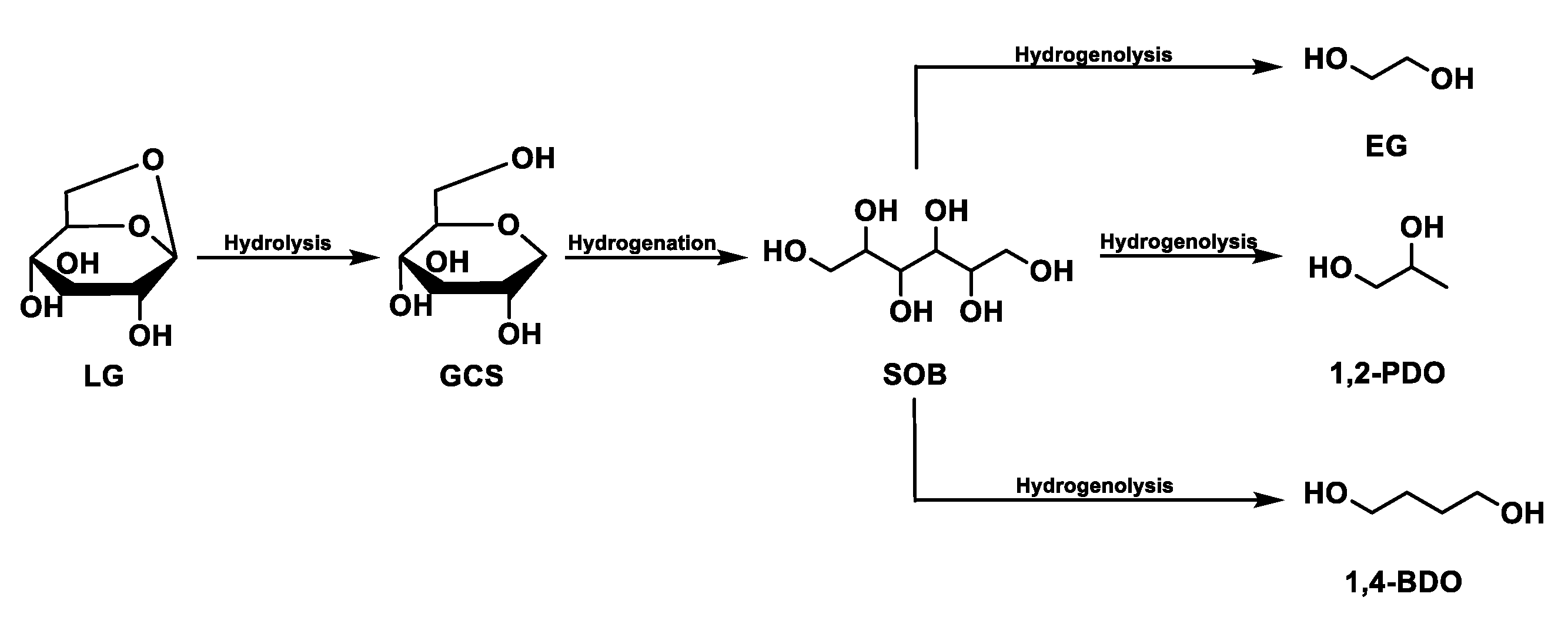
The hydrolysis of levoglucosan has been studied in a solution of water and in the presence of Ru/C [95]. The path involves the production of glucose (hydrolysis reaction) with subsequent hydrogenation into sorbitol. Finally, ethylene glycol, 1,2-Propanediol, and 1,4-Butanediol are produced by the hydrogenation of sorbitol (Figure 6).
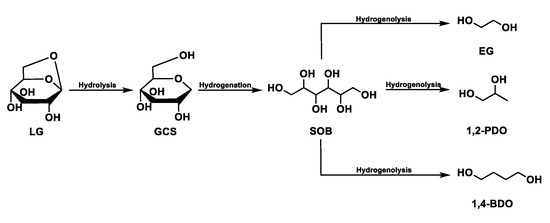
Figure 6.
Levoglucosan reaction path.
Bindwal et al. [95] proposed a kinetic rate for the hydrogenation in presence of Ru/C, where the H2 and levoglucosan (LG) chemisorbed and dissociated on the surface catalyst are as follows:
represented by the following equation:
3.2.4. Other Compounds
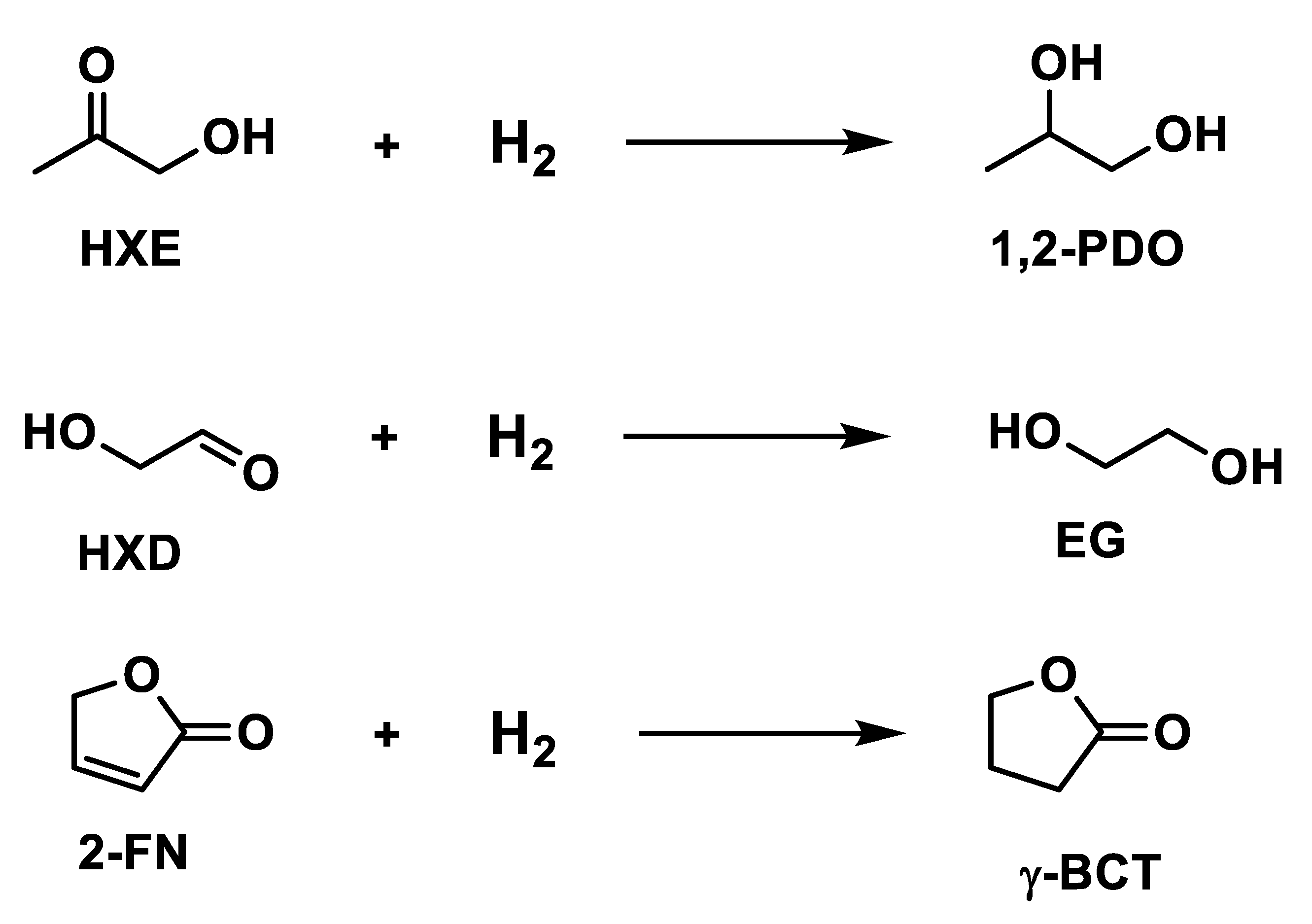
Bindwal et al. [94] studied the kinetics rate of other compounds using 5% Ru/C catalyst to convert hydroxycetone, hydroxyacetaldehyde and 2-furanone in 1,2 propanediol, ethylene glycol and γ-butyrolactone, according to the reactions in Figure 7.
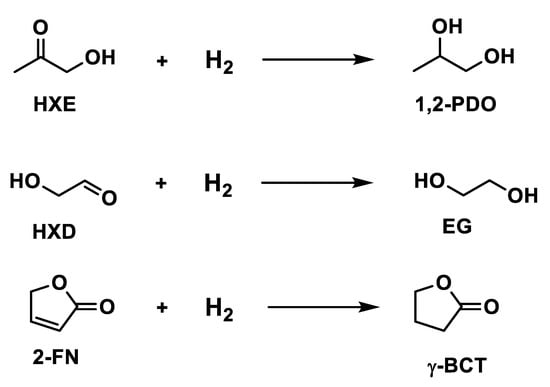
Figure 7.
Hydrogenation of hydroxyacetone, hydroxyacetaldehyde, and 2-furanone.
The authors proposed different kinetics rates varying the limitation step and the possibility to have an atomic or molecular H2 adsorption.
The kinetics rates hypothesized were validated experimentally confirming that the reactions are surface-reaction limited in presence of dissociative adsorption of H2. The equation for the kinetics rate were
Zhang et al. [96,97] described the reaction kinetics by dividing the products as Light oil ranged from 36 °C to 250 °C, heavy oil from 250 °C to 450 °C, vapors, water, and coke. They assumed a series of parallel reactions with a first-order kinetics in presence of CoMo/γAl2O3 catalyst.
Furthermore, Sheu et al. [98] divided the bio-oil into six groups (heavy non-volatiles, light non-volatile, phenols, aromatics, alkanes, Coke + H2O + Outlet Gases) and used three different catalysts (Pt/Al2O3/SiO2, CoMo/γ-Al2O3, and Ni-W/γ-Al2O3) to study the hydrogenation of bio-oil. Moreover, the authors proposed a reaction pathway by series-parallels of first-order reactions.
ki is a kinetic constant and depend of the temperature and pressure by
where ki0 and ni are the parameters of the reaction and the catalysts used.
3.3. Reactor Technologies
3.3.1. Conventional Reactors
The hydrogenation reaction is largely used in refinery to convert the heavy oil fraction into light hydrocarbons. The existing process have been based on the following reactors: fixed beds (FBRs), moving beds (MBRs), and expanded or ebullated beds (EBRs). The main difference among the reactors involves the transport phenomena and some technical details.
The FBRs are the main reactor systems used commercially and used for hydrogenating light hydrocarbon mixture such as naphtha and middle distillate. The FBRs are designed for operating in an adiabatic condition. The reactor is divided into three catalytic zones separated to an inert material (ceramic balls), the liquid and gas stream through the first catalytic bed. The output fed exchange heat by the inert bed and subsequently quenched adding fresh gas reactant and then fed inlet of the second catalytic bed. The output of the second reactor is cooled again by the inert bed and by quenching. EBR reactors have been also used to hydrogenate feeds such as vacuum residue.
The EBRs are used for heavy feeds with a large amount of metals and asphaltenes, where the liquid and gas streams are fed from bottom expanding and mixing the catalyst bed, reducing the pressure drop effect. In the output of the catalytic bed the hydrogen not reacted is recycled, while the liquid products are recovered by a flash unit.
3.3.2. Membrane Reactor
The main disadvantage of hydrogenation reaction is represented by mass transport limitation, because the reaction takes place in contact with the gaseous, liquid, and solid phase. The system has to have a high operating pressure, improving the gas solubility into the liquid system and high temperature to advantage the kinetic, but at the same time, the H2 solubility decreases under those conditions. A membrane reactor (MR) is an operation unit to produce new species by chemical reaction and separation process in a single equipment [99]. The use of MRs can reduce the process footprint, since the plant will be more compact and can result in lower investment costs, improving the economics of the process [100,101].
In a MBR, the fresh catalyst is fed from the top and trough the reactor, while the reactant stream is fed from the bottom. Afterthought, the products leave the MBR and the deactivated catalyst is sent to the regenerator reactor, where the coke deposition is burned and the activated catalyst returns to MBR.
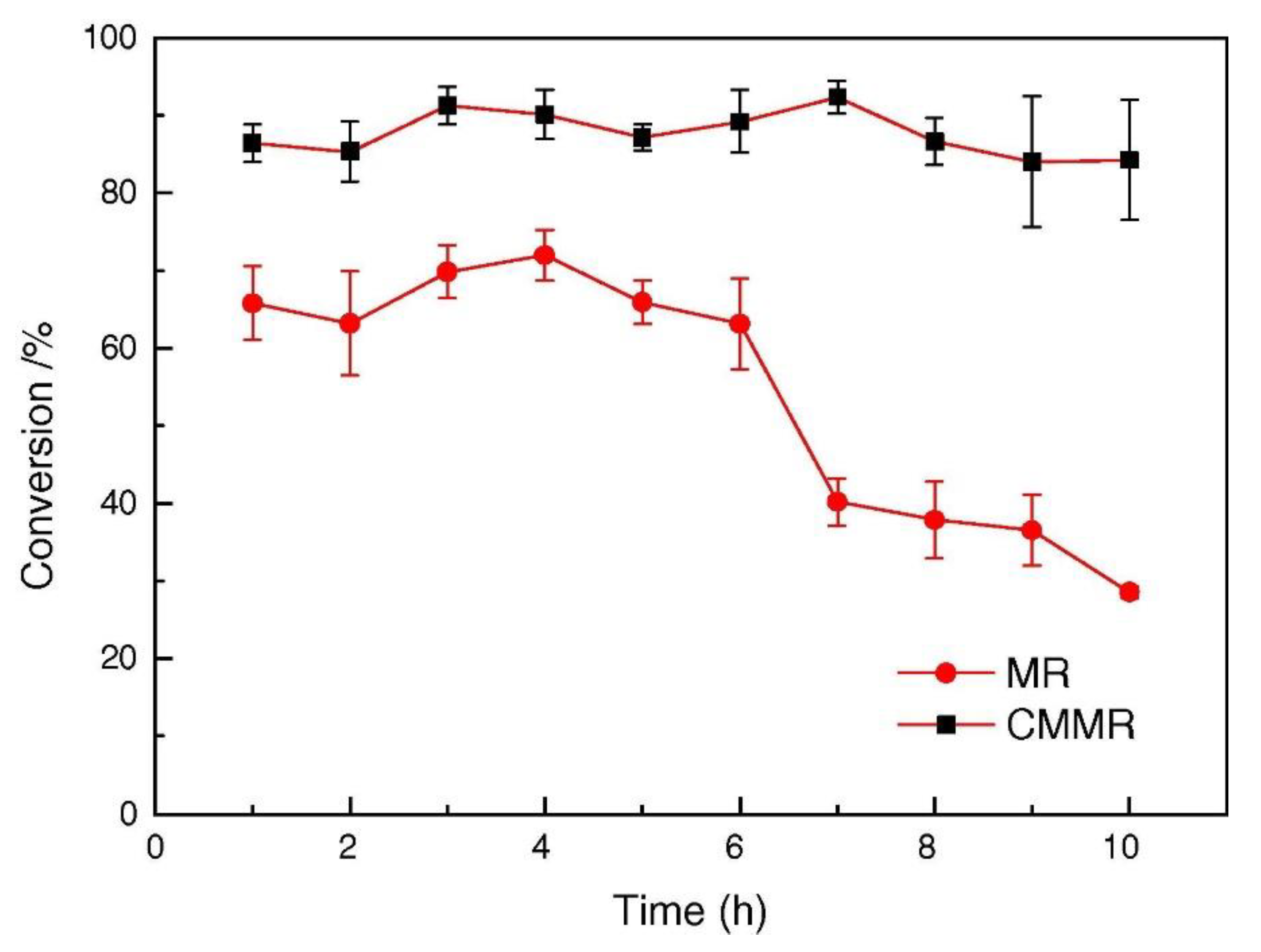
One of the features of the MRs is to act as a contactor between the three phases during HDO reaction. Furthermore, the membrane can have catalytic activity chancing the product distribution as reported by Liu et al. [102], who compared packed bed MR and catalytic MR for the hydrogenation of nitrobenzene in presence of Pd/γ-Al2O3 catalyst. The CMMR showed best performance in term of conversion and catalytic stability (~85% for 10 h) as shown in Figure 8.
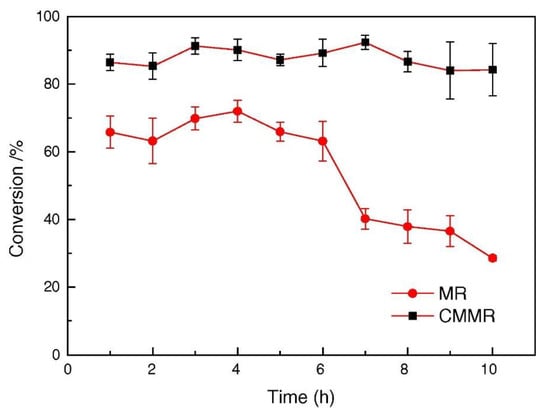
Figure 8.
Comparison between catalytic membrane reactor (CMR) and packed bed membrane reactor (PBMR) [102].
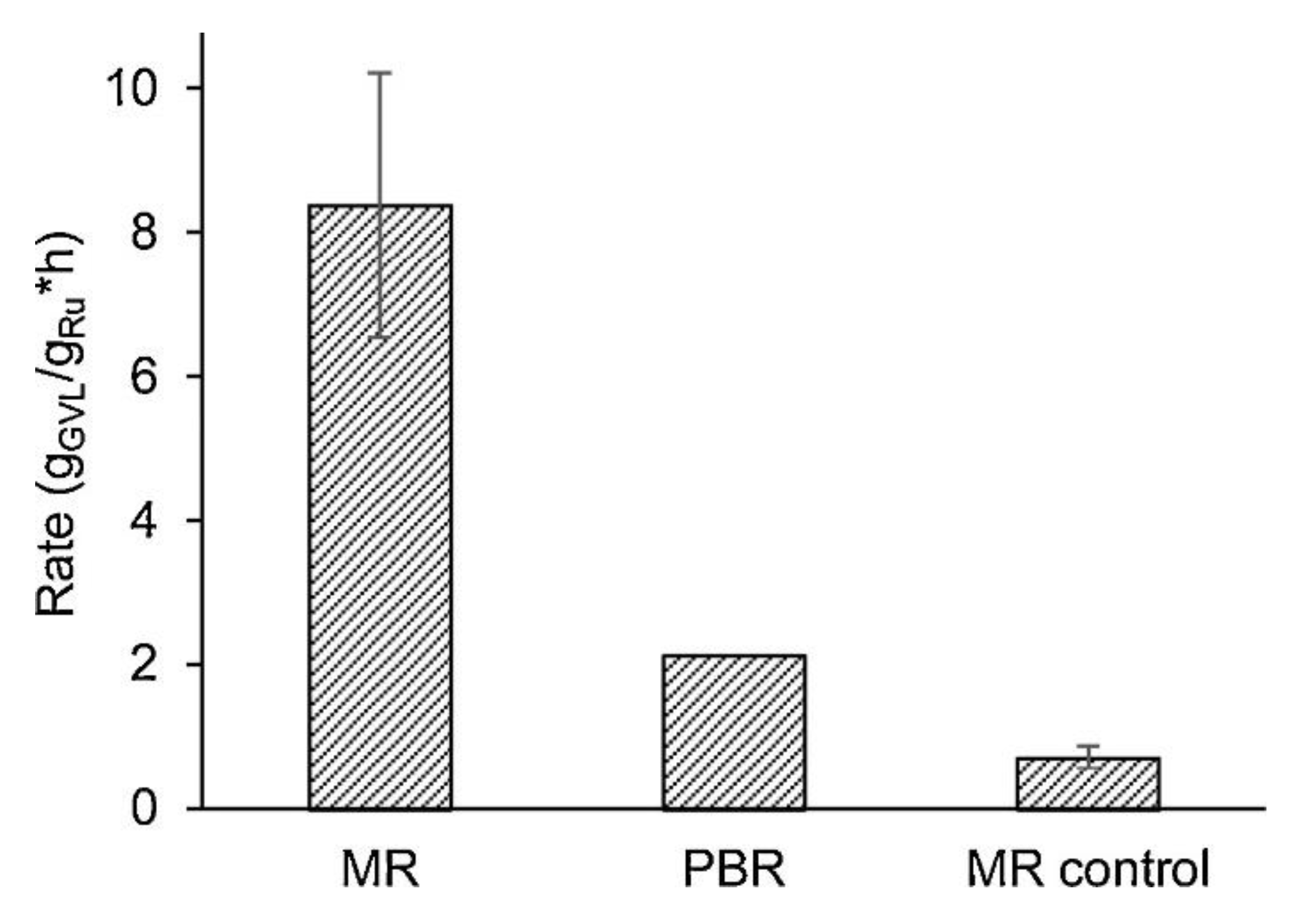
Liu et al. [103] studied the selectivity hydrogenation of butadiene in butene at 40 °C and 10 bar by catalytic membrane reactors (CMRs), obtaining a butene selectivity higher than 99% and butadiene concentration in the output stream lower than 10 ppm. Another example of hydrogenation reaction is reported in Table 4. Despite the increasing interest in catalytic membranes, the HDO of bio-oil in MR is a novelty, since in literature there is only one article [104] available on the topic, where the authors used a MR for the hydrogenation of levulinic acid (compound present in bio-oils) by a porous expanded polytetrafluoroethylene (ePTFE) membrane with Ru catalyst particles. Moreover, the same membranes were coated only in one side with a dense Matrimid layer, which was used to control the hydrogen flux through the membrane. The reaction was studied in a temperature and reaction pressure between 40 and 90 °C and 0.7 and 5.6 bar, respectively. Furthermore, the authors compared the result obtained with a PBR as shown in Figure 9, where the kinetic rate is presented as ratio of gamma-valerolactone product (g/h) over grams of Ru. In particular, the MR without the Matrimid layer obtained the best performance (four times more than PBR) with a conversion of 0.0065%, while the MR with the control layer (Matrimid) showed a kinetic rate two times less than the PBR.

Table 4.
MR for hydrogenation reaction.
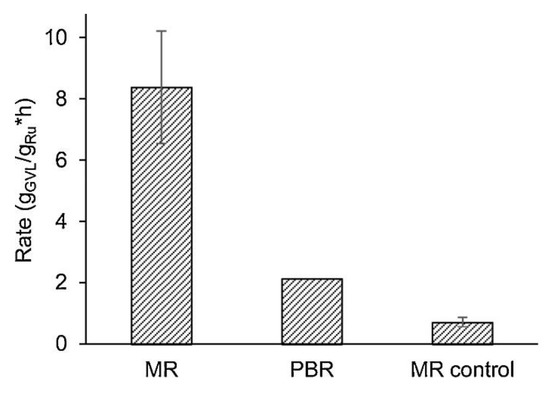
Figure 9.
Kinetic rate of hydrogenation of levulonic acid using membrane reactors (MRs) and a packet bed reactor (PBR) [104].
Recent studies have emphasized the functionality of MR to be able to achieve a TOF equal to 48,000 h−1 for the partial hydrogenation of furfural in presence of Ru–polyethersulfone (PES) catalytic membrane at 70 °C and 7 bar [105].
4. Concluding Remarks and Future Outlook
The valorization of biomass and residues for the production of liquid fuels by both thermochemical (e.g., pyrolysis) or physical (e.g., pressing) methods has attracted a great attention from both scientific and technological point of view. In fact, the utilization of vegetable raw materials for the production of synthetic chemical intermediates and hydrocarbons is considered one of the most investigated strategies aimed at reducing both the carbon dioxide emissions and the dependence on fossils fuels.
In this review, we summarize the main aspects related to pyrolysis and to the properties of the obtained bio-oils, focusing great attention to the hydrotreatment process alternatives for converting the pyrolysis bio-oil into drop-in fuel.
Research studies usually are focused on the pyrolysis of a well-defined biomass, while a limited number of papers are devoted to biomass residues with variable composition. The former approach is useful for understanding the complex mechanism involved during the pyrolysis and the effect of process parameters on reaction pathways, while the latter is of paramount importance for developing the technology at a pilot and demonstrative scale. In fact, the compositional variations in the feedstock modify both yield and composition of bio-oil, and this aspect has a significant impact on the viability of the process. As previously mentioned, biomass availability at a low cost is one of the biggest challenges of biorefinery. Therefore, more effort should be put on the experimental investigation at a pilot and demonstrative scale on the production of bio-oil from biomass residues with a large compositional variability. The research is also focused on the role of biomass pre-treatment on bio-oil quality. In particular, physical, chemical, and thermal methods may be adopted. As an example, the modification of size and shape of biomass particles has an effect on heat transfer with an impact on bio-oil quality. Whereas, the reduction of hemicellulose by a thermal method, such as torrefaction, decreases the amount of organic acids, acetals, and water in the bio-oil, with a positive impact on bio-oil stability, but with a higher inorganics content. The amount of inorganics may be reduced by physical pre-treatment such as biomass washing with water or acids. Unfortunately, there is a lack of information and knowledge about the economic feasibility of the biomass pre-treatment methods. Another important aspect that should be investigated in more detail is the stability of produced bio-oil. In fact, the bio-oil is a complex mixture containing water and both polar and nonpolar organics that cause several reactions, e.g., oligomerization condensation and dehydration, with aging of the bio-oil and formation of a more complex multiphase systems. The addition of alcohols, such as methanol, usually improves stability, homogeneity, and viscosity of bio-oil. Further research on bio-oil stabilization is needed to address technical issues during bio-oil storage and processing.
This review aims to summarize recent advances on the conversion to pyrolysis bio-oil into drop-in fuels by catalytic hydrogenation. In this regard, the research efforts should be better focused on (i) catalytic assessment of novel catalysts, and (ii) experimental investigation at pilot and demonstrative scale of hydrotreatment of real bio-oil. Concerning the first point, several metals and metals supported over moderately acid solids have been investigated. Ni-Mo or Co-Mo bimetallic systems supported over gamma-alumina are the most investigated catalysts for hydrotreatment, since they are well-known catalysts for hydroprocessing oil-derived streams, i.e., hydrodesulphurization. In these systems, Mo represents the active phase for the removal of heteroatoms, while Ni or Co acts as promoters for the hydrogenation step. Several alternative catalysts have been studied mostly for the hydrogenation of model compounds, whose catalytic behavior is in part discussed in this review. For instance, different metals and different supports have been studied, while a less attention was paid to the design of innovative hybrid systems, where the catalytic functionalities requested by the process, e.g., redox, acids, are carefully tuned with the aim to improve catalyst effectiveness. In this regard, research should be also devoted to the study of reaction mechanism as a function of surface properties of the catalysts. This approach has brought advances in other fields, such as hydrogenation of carbon dioxide to synthetic fuels, and it may be useful for a better understanding of catalysis of hydrogenation of bio-oils.
Nevertheless, bio-oil strongly differs from typical crude oil derived streams, due to the presence of a large amount of oxygenated compounds, e.g., carboxylic acids, phenols, aldehydes, ketones, sugars, and water. For this reason, the physic-chemical features of the catalyst for hydrotreatment of bio-oil should be carefully tuned as a function of bio-oil composition. On the contrary, a limited number of studies were carried out on the hydrotreatment of real pyrolysis bio-oil. In that case, the number of variables and the issues strongly increase. As an example, the presence of unsaturated oligomers in real bio-oil may lead to the formation of coke with deactivation of the catalyst. Therefore, future focus should be on the separation of bio-oil fractions in order to assess the most suitable bio-oil cut for hydroprocessing. In fact, the presence of oligomers inevitably causes a large amount of coke formation with catalyst deactivation. Of course, the presence of an additional step between pyrolysis and hydrotreatment has a significant effect on the process costs. The experimental investigation of hydrotreatment of bio-oil fractions at an either pilot or demonstrative scale may push towards more research in the field of bio-oil pre-treatments, as well as address also challenges, such as hydrogen consumption. In fact, most of the papers on hydrotreatment of bio-oil or bio-oil models are focused on product yield and quality, but it is difficult to find quantitative information on hydrogen consumption, which is usually used in large excess. As in the case of pyrolysis step, investigations at scales larger than laboratory of hydrogenation steps may surely provide quantitative data useful for viability studies on the production of drop-in fuels from biomass via pyrolysis.
Author Contributions
Conceptualization and organization: G.B., E.P. and E.C.; Writing—original draft preparation: G.B., A.S., E.P. and E.C.; Writing—review and editing: E.P.; All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| BNZ | Benzene |
| CHE | Cyclohexene |
| CHO | Cyclohexanol |
| CMR | Catalytic membrane reactor |
| COL | Cyclohexanone |
| CXO | Cyclohexanol |
| Ea | Activation energy |
| EBR | Ebullated bed reactor |
| EG | Ethylene glycol |
| γ-BCT | γ-Butyrolactone |
| GCL | Guaiacol |
| GCS | Glucose |
| HDO | Hydrodeoxygenation reaction |
| HD | Hydrotreating |
| HTL | Hydrothermal liquefaction |
| HXD | Hydroxyacetaldehyde |
| HXE | Hydroxyacetone |
| k | Kinetic rate |
| k0 | Pre-exponential number |
| LG | Levoglucosan |
| LHHW | Langmuir–Hinshelwood–Hougen–Watson |
| LHSV | Liquid hourly space velocity |
| MR | Membrane reactor |
| MBR | Moving bed reactor |
| MCP | Methylcyclopentane |
| n | Kinetic order |
| PBR | Packet bed reactor |
| PBMR | Packed bed membrane reactor |
| PCL | Pyrocatechol |
| PHE | Phenol |
| SOB | Sorbitol |
| TEA | Techno-economical assessment |
| TOF | Turnover of frequency |
| WBO | Water soluble bio-oil fraction |
| 1,2-PDO | 1,2-Propanediol |
| 1,4-BDO | 1,4-Butanediol |
| 2-FN | 2-Furanone |
References
- Anastas, P.; Eghbali, N. Green Chemistry: Principles and Practice. Chem. Soc. Rev. 2010, 39, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Ragauskas, A.J.; Williams, C.K.; Davison, B.H.; Britovsek, G.; Cairney, J.; Eckert, C.A.; Frederick, W.J.; Hallett, J.P.; Leak, D.J.; Liotta, C.L.; et al. The Path Forward for Biofuels and Biomaterials. Science 2006, 311, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Paone, E.; Tabanelli, T.; Mauriello, F. The rise of lignin biorefinery. Curr. Opin. Green Sustain. Chem. 2020, 24, 1–6. [Google Scholar] [CrossRef]
- Paone, E.; Beneduci, A.; Corrente, G.A.; Malara, A.; Mauriello, F. Hydrogenolysis of aromatic ethers under lignin-first conditions. Mol. Catal. 2020, 497, 111228. [Google Scholar] [CrossRef]
- Malara, A.; Paone, E.; Frontera, P.; Bonaccorsi, L.; Panzera, G.; Mauriello, F. Sustainable exploitation of coffee silverskin in water remediation. Sustainability 2020, 10, 3547. [Google Scholar] [CrossRef]
- Xu, C.; Paone, E.; Rodríguez-Padrón, D.; Luque, R.; Mauriello, F. Recent catalytic routes for the preparation and the upgrading of biomass derived furfural and 5-hydroxymethylfurfural. ChemSocRev 2020, 49, 4273–4306. [Google Scholar] [CrossRef]
- Transforming Our World: The 2030 Agenda for Sustainable Development. Available online: https://sustainabledevelopment.un.org/post2015/transformingourworld (accessed on 11 January 2021).
- Carlos, R.M.; Khang, D.B. Characterization of biomass energy projects in Southeast Asia. Biomass Bioenergy 2008, 32, 525–532. [Google Scholar] [CrossRef]
- Paone, E.; Espro, C.; Pietropaolo, R.; Mauriello, F. Selective arene production from transfer hydrogenolysis of benzyl phenyl ether promoted by a co-precipitated Pd/Fe3O4 catalyst. Catal. Sci. Technol. 2016, 6, 7937–7941. [Google Scholar] [CrossRef]
- Mauriello, F.; Paone, E.; Pietropaolo, R.; Balu, A.M.; Luque, R. Catalytic transfer hydrogenolysis of lignin-derived aromatic ethers promoted by bimetallic Pd/Ni systems. ACS Sustain. Chem. Eng. 2018, 6, 9269–9276. [Google Scholar] [CrossRef]
- Espro, C.; Gumina, B.; Paone, E.; Mauriello, F. Upgrading lignocellulosic biomasses: Hydrogenolysis of platform derived molecules promoted by heterogeneous Pd-Fe catalysts. Catalysts 2017, 7, 78. [Google Scholar] [CrossRef]
- Espro, C.; Gumina, B.; Szumelda, T.; Paone, E.; Mauriello, F. Catalytic transfer hydrogenolysis as an effective tool for the reductive upgrading of cellulose, hemicellulose, lignin, and their derived molecules. Catalysts 2018, 8, 313. [Google Scholar] [CrossRef]
- Mauriello, F.; Ariga-Miwa, H.; Paone, E.; Pietropaolo, R.; Takakusagi, S.; Asakura, K. Transfer hydrogenolysis of aromatic ethers promoted by the bimetallic Pd/Co catalyst. Catal. Today 2020, 357, 511–517. [Google Scholar] [CrossRef]
- Malara, A.; Paone, E.; Bonaccorsi, L.; Mauriello, F.; Macario, A.; Frontera, P. Pd/Fe3O4 Nanofibers for the Catalytic Conversion of Lignin-Derived Benzyl Phenyl Ether under Transfer Hydrogenolysis Conditions. Catalysts 2020, 10, 20. [Google Scholar] [CrossRef]
- Srifa, A.; Chaiwat, W.; Pitakjakpipop, P.; Anutrasakda, W.; Faungnawakij, K. Chapter 6—Advances in bio-oil production and upgrading technologies. In Sustainable Bioenergy; Elsevier: Amsterdam, The Netherlands, 2019; pp. 167–198. [Google Scholar]
- Han, Y.; Gholizadeh, M.; Tran, C.-C.; Kaliaguine, S.; Li, C.-Z.; Olarte, M.; Garcia-Perez, M. Hydrotreatment of pyrolysis bio-oil: A review. Fuel Process. Technol. 2019, 195, 106140. [Google Scholar] [CrossRef]
- Tabanelli, T.; Paone, E.; Blair Vásquez, P.; Pietropaolo, R.; Cavani, F.; Mauriello, F. Transfer Hydrogenation of Methyl and Ethyl Levulinate Promoted by a ZrO2 Catalyst: Comparison of Batch vs Continuous Gas-Flow Conditions. ACS Sustain. Chem. Eng. 2019, 7, 9937–9947. [Google Scholar] [CrossRef]
- Fasolini, A.; Cucciniello, R.; Paone, E.; Mauriello, F.; Tabanelli, T. Short Overview on the Hydrogen Production Via Aqueous Phase Reforming (APR) of Cellulose, C6-C5 Sugars and Polyols. Catalysts 2019, 9, 917. [Google Scholar] [CrossRef]
- Xu, C.; Paone, E.; Rodríguez-Padrón, D.; Luque, R.; Mauriello, F. Reductive catalytic routes towards sustainable production of hydrogen, fuels and chemicals from biomass derived polyols. Renew. Sustain. Energy Rev. 2020, 127, 109852. [Google Scholar] [CrossRef]
- Weldekidan, H.; Strezov, V.; Town, G. Review of solar energy for biofuel extraction. Renew. Sustain. Energy Rev. 2018, 88, 184–192. [Google Scholar] [CrossRef]
- Pistone, A.; Espro, C. Current trends on turning biomass wastes into carbon materials for electrochemical sensing and rechargeable battery applications. Curr. Opin. Green Sustain. Chem. 2020, 26, 100374. [Google Scholar] [CrossRef]
- Hu, X.; Gholizadeh, M. Progress of the applications of bio-oil. Renew. Sustain. Energy Rev. 2020, 134, 110124. [Google Scholar] [CrossRef]
- Bagnato, G.; Boulet, F.; Sanna, A. Effect of Li-LSX zeolite, NiCe/Al2O3 and NiCe/ZrO2 on the production of drop-in bio-fuels by pyrolysis and hydrotreating of Nannochloropsis and isochrysis microalgae. Energy 2019, 179, 199–213. [Google Scholar] [CrossRef]
- Kumar, R.; Strezova, V.; Weldekidan, H.; He, J.; Singh, S.; Kan, T.; Dastjerdi, B. Lignocellulose biomass pyrolysis for bio-oil production: A review of biomass pre-treatment methods for production of drop-in fuels. Renew. Sustain. Energy Rev. 2020, 123, 109763. [Google Scholar] [CrossRef]
- Fang, J.; Zhan, L.; Ok, Y.S.; Gao, B. Minireview of potential applications of hydrochar derived from hydrothermal carbonization of biomass. J. Ind. Eng. Chem. 2018, 57, 15–21. [Google Scholar] [CrossRef]
- Ullah Khan, I.; Othman, M.H.D.; Hashim, H.; Matsuura, T.; Ismail, A.F.; Rezaei-DashtArzhandi, M.; Wan Azelee, I. Biogas as a renewable energy fuel—A review of biogas upgrading, utilisation and storage. Energy Convers. Manag. 2017, 150, 277–294. [Google Scholar] [CrossRef]
- Baker, E.G.; Elliott, D.C. Catalytic Hydrotreating of Biomass-Derived Oils, in Pyrolysis Oils from Biomass. ACS Symp. Ser. 1988, 21, 228–240. [Google Scholar]
- Bagnato, G.S.; Sanna, A. Process and Techno-Economic Analysis for Fuel and Chemical Production by Hydrodeoxygenation of Bio-Oil. Catalysts 2019, 9, 1021. [Google Scholar] [CrossRef]
- Lima, S.; Fernandes, A.; Antunes, M.M.; Pillinger, M.; Ribeiro, F.; Valente, A.A. Dehydration of Xylose into Furfural in the Presence of Crystalline Microporous Silicoaluminophosphates. Catal. Lett. 2010, 135, 41–47. [Google Scholar] [CrossRef]
- Demirbas, A. Thermochemical Processes. In Biorefineries: For Biomass Upgrading Facilities. Green Energy and Technology; Springer: London, UK, 2010; pp. 135–192. [Google Scholar]
- BTG Bioliquids BV. 2020. Available online: https://www.btg-bioliquids.com (accessed on 11 January 2021).
- Li, F.; Srivatsa, S.C.; Bhattacharya, S. A review on catalytic pyrolysis of microalgae to high-quality bio-oil with low oxygeneous and nitrogenous compounds. Renew. Sustain. Energy Rev. 2019, 108, 481–497. [Google Scholar] [CrossRef]
- Furimsky, E. Catalytic hydrodeoxygenation. Appl. Catal. A Gen. 2000, 199, 147–190. [Google Scholar] [CrossRef]
- Fisk, C.A.; Morgan, T.; Ji, Y.; Crocker, M.; Crofcheck, C.; Lewis, S.A. Bio-oil upgrading over platinum catalysts using in situ generated hydrogen. Appl. Catal. A Gen. 2009, 358, 150–156. [Google Scholar] [CrossRef]
- Li, P.; Chen, X.; Wang, X.; Shao, J.; Lin, G.; Yang, H.; Yang, Q.; Chen, H. Catalytic Upgrading of Fast Pyrolysis Products with Fe-, Zr-, and Co-Modified Zeolites Based on Pyrolyzer–GC/MS Analysis. Energy Fuels 2017, 31, 3979–3986. [Google Scholar] [CrossRef]
- Dong, T.; Wang, J.; Miao, C.; Zheng, Y.; Chen, S. Two-step in situ biodiesel production from microalgae with high free fatty acid content. Bioresour. Technol. 2013, 136, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Zainan, N.H.; Srivatsa, S.C.; Bhattacharya, S. Catalytic pyrolysis of microalgae Tetraselmis suecica and characterization study using in situ Synchrotron-based Infrared Microscopy. Fuel 2015, 161, 345–354. [Google Scholar] [CrossRef]
- Demirbas, A. The influence of temperature on the yields of compounds existing in bio-oils obtained from biomass samples via pyrolysis. Fuel Process. Technol. 2007, 88, 591–597. [Google Scholar] [CrossRef]
- Venderbosch, R.H.; Ardiyanti, A.R.; Wildschut, J.; Oasmaa, A.; Heeres, H.J. Stabilization of biomass-derived pyrolysis oils. J. Chem. Technol. Biotechnol. 2010, 85, 674–686. [Google Scholar] [CrossRef]
- Oasmaa, A.; Solantausta, Y.; Arpiainen, V.; Kuoppala, E.; Sipilä, K. Fast pyrolysis bio-oils from wood and agricultural residues. Energy Fuels 2010, 2, 1380–1388. [Google Scholar] [CrossRef]
- Wildschut, J.; Mahfud, F.H.; Venderbosch, R.H.; Heeres, H.J. Hydrotreatment of Fast Pyrolysis Oil Using Heterogeneous Noble-Metal Catalysts. Ind. Eng. Chem. Res. 2009, 48, 10324–10334. [Google Scholar] [CrossRef]
- App, A.R.; Khromova, S.A.; Venderbosch, R.H.; Yakovlev, V.A.; Heeres, H.J. Catalytic hydrotreatment of fast-pyrolysis oil using non-sulfided bimetallic Ni-Cu catalysts on a δ-Al2O3 support. Appl. Catal. B 2012, 117–118, 105–117. [Google Scholar]
- Lu, Q.; Yang, X.-L.; Zhu, X.-F. Analysis on chemical and physical properties of bio-oil pyrolyzed from rice husk. J. Anal. Appl. Pyrolysis 2008, 82, 191–198. [Google Scholar] [CrossRef]
- Wildschut, J.; Iqbal, M.; Mahfud, F.H.; Cabrera, I.M.; Venderbosch, R.H.; Heeres, H.J. Insights in the hydrotreatment of fast pyrolysis oil using a ruthenium on carbon catalyst. Energy Environ. Sci. 2010, 3, 962–970. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hart, T.R.; Neuenschwande, G.G.; Rotness, L.J.; Olarte, M.V.; Zacher, A.H.; Solantausta, Y. Catalytic Hydroprocessing of Fast Pyrolysis Bio-oil from Pine Sawdust. Energy Fuels 2012, 26, 3891–3896. [Google Scholar] [CrossRef]
- Elliott, D.C.; Neuenschwander, G.G. Liquid Fuels by Low-Severity Hydrotreating of Biocrude. In Developments in Thermochemical Biomass Conversion: Volume 1/Volume 2; Bridgwater, A.V., Boocock, D.G.B., Eds.; Springer: Dordrecht, The Netherlands, 1997; pp. 611–621. [Google Scholar]
- Patel, M.; Kumar, A. Production of renewable diesel through the hydroprocessing of lignocellulosic biomass-derived bio-oil: A review. Renew. Sustain. Energy Rev. 2016, 58, 1293–1307. [Google Scholar] [CrossRef]
- Zhang, S.; Yan, Y.; Li, T.; Ren, Z. Upgrading of liquid fuel from the pyrolysis of biomass. Bioresour. Technol. 2005, 96, 545–550. [Google Scholar] [CrossRef] [PubMed]
- López, R.; Fernández, C.; Gómez, X.; Martínez, O.; Sánchez, M.E. Thermogravimetric analysis of lignocellulosic and microalgae biomasses and their blends during combustion. J. Therm. Anal. 2013, 114, 295–305. [Google Scholar] [CrossRef]
- Liu, H.; Chen, Y.; Yang, H.; Gentili, F.G.; Söderlind, U.; Wang, X.; Zhang, W.; Chen, H. Hydrothermal carbonization of natural microalgae containing a high ash content. Fuel 2019, 249, 441–448. [Google Scholar] [CrossRef]
- Plis, A.; Lasek, J.; Skawińska, A. Kinetic analysis of the combustion process of Nannochloropsis gaditana microalgae based on thermogravimetric studies. J. Anal. Appl. Pyrolysis 2017, 127, 109–119. [Google Scholar] [CrossRef]
- Miao, X.; Wu, Q. High yield bio-oil production from fast pyrolysis by metabolic controlling of Chlorella protothecoides. J. Biotechnol. 2004, 110, 85–93. [Google Scholar] [CrossRef]
- Jena, U.; Das, K.C. Comparative Evaluation of Thermochemical Liquefaction and Pyrolysis for Bio-Oil Production from Microalgae. Energy Fuels 2011, 25, 5472–5482. [Google Scholar] [CrossRef]
- Guo, Q.; Wu, M.; Wang, K.; Zhang, L.; Xu, X. Catalytic Hydrodeoxygenation of Algae Bio-oil over Bimetallic Ni–Cu/ZrO2 Catalysts. Ind. Eng. Chem. Res. 2015, 54, 890–899. [Google Scholar] [CrossRef]
- Gruia, A. Hydrotreating. In Handbook of Petroleum Processing; Jones, D.S.J.S., Pujado, P.R., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 321–354. [Google Scholar]
- Jacobson, K.; Maheria, K.C.; Kumar Dalai, A. Bio-oil valorization: A review. Renew. Sustain. Energy Rev. 2013, 23, 91–106. [Google Scholar] [CrossRef]
- Mauchausse, C.; Kural, E.; Trimm, D.L.; Cant, N.W. Optimization of tungsten-based catalysts for the hydrotreatment of coal-derived liquids. Fuel 1992, 71, 203–209. [Google Scholar] [CrossRef]
- Bulushev, D.A.; Ross, J.R.H. Catalysis for conversion of biomass to fuels via pyrolysis and gasification: A review. Catal. Today 2011, 171, 1–13. [Google Scholar] [CrossRef]
- Masel, R.I. Principles of Adsorption and Reaction on Solid Surfaces; Wiley: New York, NY, USA; Chichester, UK, 1996. [Google Scholar]
- Ptushinskiĭ, Y.G. Low-temperature adsorption of gases on metal surfaces (Review). Low Temp. Phys. 2004, 30, 1–26. [Google Scholar] [CrossRef]
- Ferrin, P.; Kandoi, S.; Nilekar, A.U.; Mavrikakis, M. Hydrogen adsorption, absorption and diffusion on and in transition metal surfaces: A DFT study. Surf. Sci. 2012, 606, 679–689. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hu, J.; Hart, T.R.; Neuenschwander, G.G. Palladium Catalyzed Hydrogenation of Bio-Oils and Organic Compounds. U.S. Patent 7,425,657, 16 September 2008. [Google Scholar]
- Mortensen, P.M.; Grunwaldt, J.D.; Jensen, P.A.; Jensen, A.D. Screening of Catalysts for Hydrodeoxygenation of Phenol as a Model Compound for Bio-oil. ACS Catal. 2013, 3, 1774–1785. [Google Scholar] [CrossRef]
- Ardiyanti, A.R.; Khromova, S.A.; Venderbosch, R.H.; Yakovlev, V.A.; Melián-Cabrera, I.V.; Heeres, H.J. Catalytic hydrotreatment of fast pyrolysis oil using bimetallic Ni–Cu catalysts on various supports. Appl. Catal. A 2012, 449, 121–130. [Google Scholar] [CrossRef]
- Wei, S.; Zhao, Y.; Fan, G.; Yang, L.; Li, F. Structure-dependent selective hydrogenation of cinnamaldehyde over high-surface-area CeO2-ZrO2 composites supported Pt nanoparticles. Chem. Eng. J. 2017, 322, 234–245. [Google Scholar] [CrossRef]
- Zhou, M.; Liu, P.; Wang, K.; Xu, J.; Jiang, J. Catalytic hydrogenation and one step hydrogenation-esterification to remove acetic acid for bio-oil upgrading: Model reaction study. Catal. Sci. Technol. 2016, 6, 7783–7792. [Google Scholar] [CrossRef]
- Bergem, H.; Xu, R.; Brown, R.C.; Huber, G.W. Low temperature aqueous phase hydrogenation of the light oxygenate fraction of bio-oil over supported ruthenium catalysts. Green Chem. 2017, 19, 3252–3262. [Google Scholar] [CrossRef]
- Yuan, Q.; Zhang, D.; van Haandel, L.; Ye, F.; Xue, T.; Hensen, E.J.M.; Guan, Y. Selective liquid phase hydrogenation of furfural to furfuryl alcohol by Ru/Zr-MOFs. J. Mol. Catal. A Chem. 2015, 406, 58–64. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, Y.; Du, B.; Nasaruddin, R.R.; Chen, T.; Xie, J. Golden Carbon Nanotube Membrane for Continuous Flow Catalysis. Ind. Eng. Chem. Res. 2017, 56, 2999–3007. [Google Scholar] [CrossRef]
- Fiorio, J.L.; López, N.; Rossi, L.M. Gold–Ligand-Catalyzed Selective Hydrogenation of Alkynes into cis-Alkenes via H2 Heterolytic Activation by Frustrated Lewis Pairs. ACS Catal. 2017, 7, 2973–2980. [Google Scholar] [CrossRef]
- Takeda, Y.; Tamura, M.; Nakagawa, Y.; Okumura, K.; Tomishige, K. Characterization of Re–Pd/SiO2 Catalysts for Hydrogenation of Stearic Acid. ACS Catal. 2015, 5, 7034–7047. [Google Scholar] [CrossRef]
- Raj, K.J.A.; Prakash, M.G.; Mahalakshmy, R.; Elangovan, T.; Viswanathan, B. Liquid phase hydrogenation of crotanaldehyde over nickel supported on titania. J. Mol. Catal. A Chem. 2013, 366, 92–98. [Google Scholar]
- Fulajtárova, K.; Soták, T.; Hronec, M.; Vávra, I.; Dobročka, E.; Omastová, M. Aqueous phase hydrogenation of furfural to furfuryl alcohol over Pd–Cu catalysts. Appl. Catal. A 2015, 502, 78–85. [Google Scholar] [CrossRef]
- Wang, C.; Guo, Z.; Yang, Y.; Chang, J.; Borgna, A. Hydrogenation of Furfural as Model Reaction of Bio-Oil Stabilization under Mild Conditions Using Multiwalled Carbon Nanotube (MWNT)-Supported Pt Catalysts. Ind. Eng. Chem. Res. 2014, 53, 11284–11291. [Google Scholar] [CrossRef]
- Ota, N.; Tamura, M.; Nakagawa, Y.; Okumura, K.; Tomishige, K. Performance, Structure, and Mechanism of ReOx–Pd/CeO2 Catalyst for Simultaneous Removal of Vicinal OH Groups with H2. ACS Catal. 2016, 6, 3213–3226. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Tamura, R.; Tamura, M.; Tomishige, K. Combination of supported bimetallic rhodium–molybdenum catalyst and cerium oxide for hydrogenation of amide. Sci. Technol. Adv. Mater. 2015, 16, 014901. [Google Scholar] [CrossRef]
- Liao, X.; Zhang, Y.; Guo, J.; Zhao, L.; Hill, M.; Jiang, Z.; Zhao, Y. The Catalytic Hydrogenation of Maleic Anhydride on CeO2−δ-Supported Transition Metal Catalysts. Catalysts 2017, 7, 272. [Google Scholar] [CrossRef]
- Elliott, D.C. Historical developments in hydroprocessing bio-oils. Energy Fuels 2007, 21, 1792–1815. [Google Scholar] [CrossRef]
- Laurent, E.; Delmon, B. Study of the hydrodeoxygenation of carbonyl, car☐ylic and guaiacyl groups over sulfided CoMo/γ-Al2O3 and NiMo/γ-Al2O3 catalysts: I. Catalytic reaction schemes. Appl. Catal. A 1994, 109, 77–96. [Google Scholar] [CrossRef]
- Bienholz, A.; Schwab, F.; Claus, P. Hydrogenolysis of glycerol over a highly active CuO/ZnO catalyst prepared by an oxalate gel method: Influence of solvent and reaction temperature on catalyst deactivation. Green Chem. 2010, 12, 290–295. [Google Scholar] [CrossRef]
- Liu, H.; Liang, S.; Jiang, T.; Han, B.; Zhou, Y. Hydrogenolysis of Glycerol to 1,2-Propanediol over Ru–Cu Bimetals Supported on Different Supports. CLEAN Soil Air Water 2012, 40, 318–324. [Google Scholar] [CrossRef]
- de Miguel Mercader, F.; Groeneveld, M.J.; Kersten, S.R.A.; Geantet, C.; Toussaint, G.; Way, N.W.J.; Schaverien, C.J.; Hogendoorn, K.J.A. Hydrodeoxygenation of pyrolysis oil fractions: Process understanding and quality assessment through co-processing in refinery units. Energy Environ. Sci. 2011, 4, 985–997. [Google Scholar] [CrossRef]
- Prasomsri, T.; Shetty, M.; Murugappan, K.; Román-Leshkov, Y. Insights into the catalytic activity and surface modification of MoO3 during the hydrodeoxygenation of lignin-derived model compounds into aromatic hydrocarbons under low hydrogen pressures. Energy Environ. Sci. 2014, 7, 2660–2669. [Google Scholar] [CrossRef]
- Bagnato, G.; Signoretto, M.; Pizzolitto, C.; Menegazzo, F.; Xi, X.; ten Brink, G.H.; Kooi, B.J.; Heeres, H.J.; Sanna, A. Hydrogenation of Biobased Aldehydes to Monoalcohols Using Bimetallic Catalysts. ACS Sustain. Chem. Eng. 2020, 8, 11994–12004. [Google Scholar] [CrossRef]
- Sanna, A.; Vispute, T.P.; Huber, G.W. Hydrodeoxygenation of the aqueous fraction of bio-oil with Ru/C and Pt/C catalysts. Appl. Catal. B 2015, 165, 446–456. [Google Scholar] [CrossRef]
- Oasmaa, A.; Kuoppala, E.; Solantausta, Y. Fast Pyrolysis of Forestry Residue. 2. Physicochemical Composition of Product Liquid. Energy Fuels 2003, 17, 433–443. [Google Scholar] [CrossRef]
- Oasmaa, A.; Kuoppala, E.; Gust, S.; Solantausta, Y. Fast Pyrolysis of Forestry Residue. 1. Effect of Extractives on Phase Separation of Pyrolysis Liquids. Energy Fuels 2003, 17, 1–12. [Google Scholar] [CrossRef]
- Adjaye, J.D.; Bakhshi, N.N. Production of hydrocarbons by catalytic upgrading of a fast pyrolysis bio-oil. Part II: Comparative catalyst performance and reaction pathways. Fuel Process. Technol. 1995, 45, 185–202. [Google Scholar] [CrossRef]
- De la Puente, G.; Gil, A.; Pis, J.J.; Grange, P. Effects of Support Surface Chemistry in Hydrodeoxygenation Reactions over CoMo/Activated Carbon Sulfided Catalysts. Langmuir 1999, 15, 5800–5806. [Google Scholar] [CrossRef]
- Gutierrez, A.; Kaila, R.K.; Honkela, M.L.; Slioor, R.; Krause, A.O.I. Hydrodeoxygenation of guaiacol on noble metal catalysts. Catal. Today 2009, 147, 239–246. [Google Scholar] [CrossRef]
- Bui, V.N.; Laurenti, D.; Afanasiev, P.; Geantet, C. Hydrodeoxygenation of guaiacol with CoMo catalysts. Part I: Promoting effect of cobalt on HDO selectivity and activity. Appl. Catal. B 2011, 101, 239–245. [Google Scholar] [CrossRef]
- Centeno, A.; Laurent, E.; Delmon, B. Influence of the Support of CoMo Sulfide Catalysts and of the Addition of Potassium and Platinum on the Catalytic Performances for the Hydrodeoxygenation of Carbonyl, Carboxyl, and Guaiacol-Type Molecules. J. Catal. 1995, 154, 288–298. [Google Scholar] [CrossRef]
- Ferrari, M.; Delmon, B.; Grange, P. Influence of the active phase loading in carbon supported molybdenum–cobalt catalysts for hydrodeoxygenation reactions. Microporous Mesoporous Mater. 2002, 56, 279–290. [Google Scholar] [CrossRef]
- Bindwal, A.B.; Bari, A.H.; Vaidya, P.D. Kinetics of low temperature aqueous-phase hydrogenation of model bio-oil compounds. Chem. Eng. J. 2012, 207–208, 725–733. [Google Scholar] [CrossRef]
- Bindwal, A.B.; Vaidya, P.D. Kinetics of Aqueous-Phase Hydrogenation of Levoglucosan over Ru/C Catalyst. Ind. Eng. Chem. Res. 2013, 52, 17781–17789. [Google Scholar] [CrossRef]
- Zhang, S.; Yan, Y.; Li, T.; Ren, Z. Lumping Kinetic Model for Hydrotreating of Bio-oil from the Fast Pyrolysis of Biomass. Energy Sources Part A 2009, 31, 639–645. [Google Scholar] [CrossRef]
- Zhang, S.-P. Study of Hydrodeoxygenation of Bio-Oil from the Fast Pyrolysis of Biomass. Energy Sources 2003, 25, 57–65. [Google Scholar]
- Sheu, Y.-H.E.; Anthony, R.G.; Soltes, E.J. Kinetic studies of upgrading pine pyrolytic oil by hydrotreatment. Fuel Process. Technol. 1988, 19, 31–50. [Google Scholar] [CrossRef]
- Bagnato, G.; Iulianelli, A.; Sanna, A.; Basile, A. Glycerol Production and Transformation: A Critical Review with Particular Emphasis on Glycerol Reforming Reaction for Producing Hydrogen in Conventional and Membrane Reactors. Membranes 2017, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Iulianelli, A.; Longo, T.; Basile, A. Methanol steam reforming reaction in a Pd–Ag membrane reactor for CO-free hydrogen production. Int. J. Hydrogen Energy 2008, 33, 5583–5588. [Google Scholar] [CrossRef]
- Iulianelli, A.; Longo, T.; Basile, A. CO-free hydrogen production by steam reforming of acetic acid carried out in a Pd–Ag membrane reactor: The effect of co-current and counter-current mode. Int. J. Hydrogen Energy 2008, 33, 4091–4096. [Google Scholar] [CrossRef]
- Liu, M.; Zhu, X.; Chen, R.; Liao, Q.; Feng, H.; Li, L. Catalytic membrane microreactor with Pd/γ-Al2O3 coated PDMS film modified by dopamine for hydrogenation of nitrobenzene. Chem. Eng. J. 2016, 301, 35–41. [Google Scholar] [CrossRef]
- Liu, C.; Xu, Y.; Liao, S.; Yu, D. Mono- and bimetallic catalytic hollow- fiber reactors for the selective hydrogenation of butadiene in 1-butene. Appl. Catal. A Gen. 1998, 172, 23–29. [Google Scholar] [CrossRef]
- Stanford, J.P.; Soto, M.C.; Pfromm, P.H.; Rezac, M.E. Aqueous phase hydrogenation of levulinic acid using a porous catalytic membrane reactor. Catal. Today 2016, 268, 19–28. [Google Scholar] [CrossRef]
- Bagnato, G.; Figoli, A.; Ursino, C.; Galiano, F.; Sanna, A. A novel Ru–polyethersulfone (PES) catalytic membrane for highly efficient and selective hydrogenation of furfural to furfuryl alcohol. J. Mater. Chem. A 2018, 6, 4955–4965. [Google Scholar] [CrossRef]
- Liguori, F.; Barbaro, P.; Giordano, C.; Sawa, H. Partial hydrogenation reactions over Pd-containing hybrid inorganic/polymeric catalytic membranes. Appl. Catal. A Gen. 2013, 459, 81–88. [Google Scholar] [CrossRef]
- Aran, H.C.; Klooster, H.; Jani, J.M.; Wessling, M.; Lefferts, L.; Lammertink, R.G.H. Influence of geometrical and operational parameters on the performance of porous catalytic membrane reactors. Chem. Eng. J. 2012, 207–208, 814–821. [Google Scholar] [CrossRef]
- Bottino, A.; Capannelli, G.; Comite, A.; Del Borghi, A.; Di Felice, R. Catalytic ceramic membrane in a three-phase reactor for the competitive hydrogenation–isomerisation of methylenecyclohexane. Sep. Purif. Technol. 2004, 34, 239–245. [Google Scholar] [CrossRef]
- Bottino, A.; Capannelli, G.; Comite, A.; Di Felice, R. Polymeric and ceramic membranes in three-phase catalytic membrane reactors for the hydrogenation of methylenecyclohexane. Desalination 2002, 144, 411–416. [Google Scholar] [CrossRef]
- Bengtson, G.; Fritsch, D. Catalytic membrane reactor for the selective hydrogenation of edible oil: Platinum versus palladium catalyst. Desalination 2006, 200, 666–667. [Google Scholar] [CrossRef]
- Liguori, F.; Barbaro, P.; Sawa, H. Continuous flow hydrogenation reactions by Pd catalysts onto hybrid ZrO2/PVA materials. Appl. Catal. A Gen. 2014, 488, 58–65. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).