Simplified Method to Optimize Enzymatic Esters Syntheses in Solvent-Free Systems: Validation Using Literature and Experimental Data
Abstract
1. Introduction
2. Results
2.1. SER Validation from Literature and Experimental Data
2.2. Thermodynamics and Kinetics Aspects
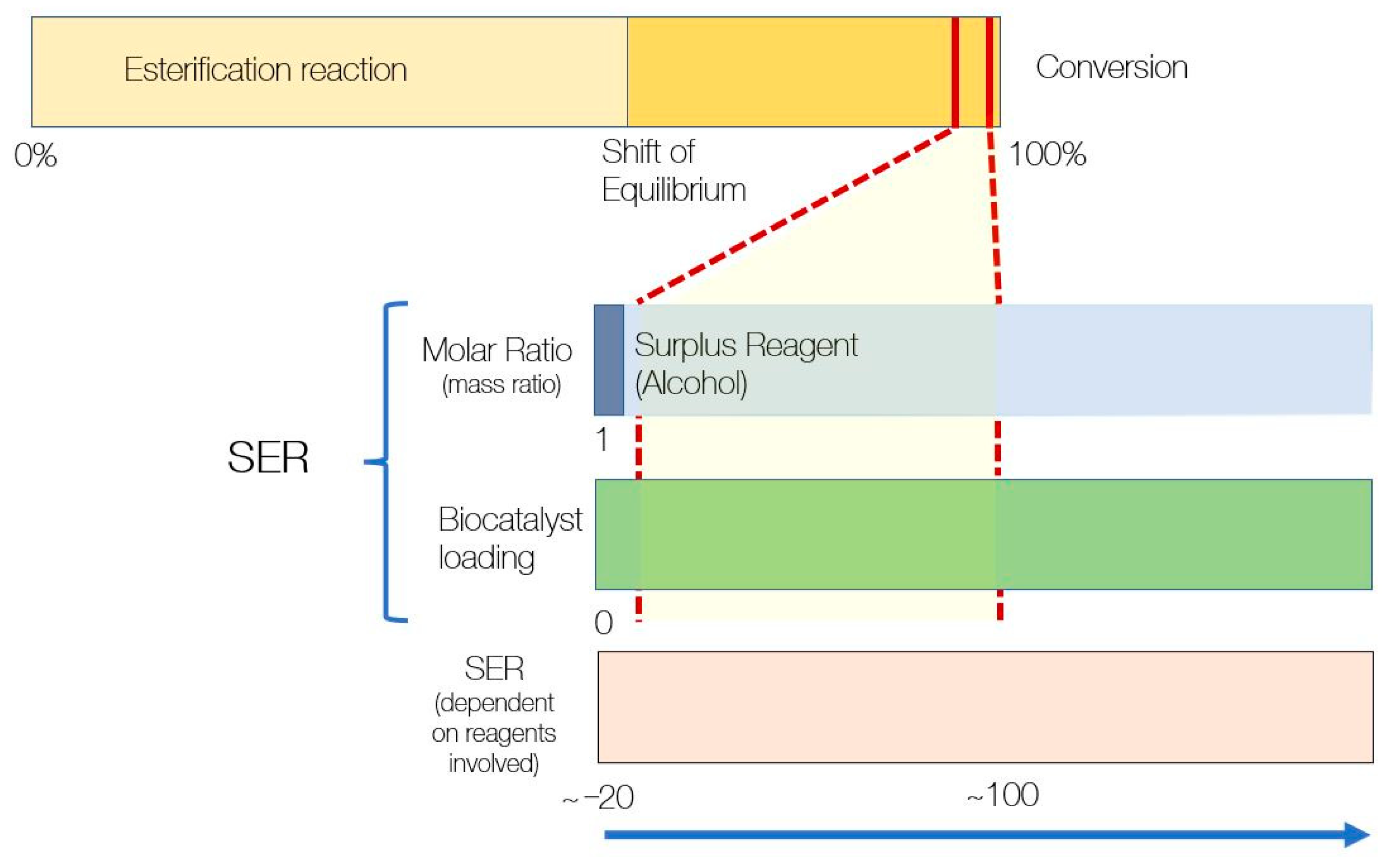
2.3. Handling SER for Design Experiments
2.4. Limitations of SER
3. Materials and Methods
3.1. Data Obtained from the Literature
3.2. Data Obtained Experimentally
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sandoval, G.; Condoret, J.S.; Monsan, P.; Marty, A. Esterification by Immobilized Lipase in Solvent-Free Media: Kinetic and Thermodynamic Arguments. Biotechnol. Bioeng. 2002, 78, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Yahya, A.R.M.; Anderson, W.A.; Moo-young, M. Ester synthesis in lipase catalyzed reactions. Enzym. Microb. Technol. 1998, 23, 438–450. [Google Scholar] [CrossRef]
- Chowdhury, A.; Mitra, D. A kinetic study on the Novozyme 435-catalyzed esterification of free fatty acids with octanol to produce octyl esters. Biotechnol. Prog. 2015, 31, 1494–1499. [Google Scholar] [CrossRef] [PubMed]
- Kuperkar, V.V.; Lade, V.G.; Prakash, A.; Rathod, V.K. Synthesis of isobutyl propionate using immobilized lipase in a solvent free system: Optimization and kinetic studies. J. Mol. Catal. B Enzym. 2014, 99, 143–149. [Google Scholar] [CrossRef]
- Vadgama, R.N.; Odaneth, A.A.; Lali, A.M. Green synthesis of isopropyl tetradecanoate in novel single phase medium Part I: Batch optimization studies. Biotechnol. Rep. 2015, 8, 133–137. [Google Scholar] [CrossRef]
- Sousa, R.R.; Pazutti, L.V.B.; Dalmaso, G.Z.L.; Siqueira, D.F.; Silva, A.S.; Ferreira-Leitão, V.S. A practical approach to obtain high yield lipase-mediated synthesis of octyl octanoate with Novozym 435. Biocatal. Biotransform. 2020, 38, 293–303. [Google Scholar] [CrossRef]
- Sose, M.T.; Bansode, S.R.; Rathod, V.K. Solvent free lipase catalyzed synthesis of butyl octanoate. J. Chem. Sci. 2017, 129, 1755–1760. [Google Scholar] [CrossRef]
- Aljawish, A.; Heuson, E.; Bigan, M.; Froidevaux, R. Lipase catalyzed esterification of formic acid in solvent and solvent-free systems. Biocatal. Agric. Biotechnol. 2019, 20, 101221. [Google Scholar] [CrossRef]
- Parikh, D.T.; Lanjekar, K.J.; Rathod, V.K. Kinetics and thermodynamics of lipase catalysed synthesis of propyl decanoate. Biotechnol. Lett. 2019, 41, 1163–1175. [Google Scholar] [CrossRef]
- Adlercreutz, P. Immobilisation and application of lipases in organic media. Chem. Soc. Rev. 2013, 42, 6406–6436. [Google Scholar] [CrossRef]
- Sheldon, R.A.; van Pelt, S. Enzyme immobilisation in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef]
- Thangaraj, B.; Solomon, P.R. Immobilization of Lipases—A Review. Part I: Enzyme Immobilization. ChemBioEng Rev. 2019, 6, 157–166. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzym. Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Galan, C.; Berenguer-Murcia, Á.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- Barbosa, O.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Rodrigues, R.C.; Fernandez-Lafuente, R. Strategies for the one-step immobilization-purification of enzymes as industrial biocatalysts. Biotechnol. Adv. 2015, 33, 435–456. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Lafuente, R. Lipase from Thermomyces lanuginosus: Uses and prospects as an industrial biocatalyst. J. Mol. Catal. B Enzym. 2010, 62, 197–212. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Fernandez-Lafuente, R. Lipase from Rhizomucor miehei as a biocatalyst in fats and oils modification. J. Mol. Catal. B Enzym. 2010, 66, 15–32. [Google Scholar] [CrossRef]
- Khan, N.R.; Rathod, V.K. Enzyme catalyzed synthesis of cosmetic esters and its intensification: A review. Process Biochem. 2015, 50, 1793–1806. [Google Scholar] [CrossRef]
- Ortiz, C.; Ferreira, M.L.; Barbosa, O.; Dos Santos, J.C.S.; Rodrigues, R.C.; Berenguer-Murcia, Á.; Briand, L.E.; Fernandez-Lafuente, R. Novozym 435: The “perfect” lipase immobilized biocatalyst? Catal. Sci. Technol. 2019, 9, 2380–2420. [Google Scholar] [CrossRef]
- Nielsen, P.M.; Brask, J.; Fjerbaek, L. Enzymatic biodiesel production: Technical and economical considerations. Eur. J. Lipid Sci. Technol. 2008, 110, 692–700. [Google Scholar] [CrossRef]
- Szczesna Antczak, M.; Kubiak, A.; Antczak, T.; Bielecki, S. Enzymatic biodiesel synthesis—Key factors affecting efficiency of the process. Renew. Energy 2009, 34, 1185–1194. [Google Scholar] [CrossRef]
- Pourzolfaghar, H.; Abnisa, F.; Daud, W.M.A.W.; Aroua, M.K. A review of the enzymatic hydroesterification process for biodiesel production. Renew. Sustain. Energy Rev. 2016, 61, 245–257. [Google Scholar] [CrossRef]
- Neta, N.S.; Teixeira, J.A.; Rodrigues, L.R. Sugar Ester Surfactants: Enzymatic Synthesis and Applications in Food Industry. Crit. Rev. Food Sci. Nutr. 2015, 55, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, R.; Anbumathi, P.; Viruthagiri, T. Lipase applications in food industry. Indian J. Biotechnol. 2007, 6, 141–158. [Google Scholar]
- Hasenhuettl, G.L. Synthesis and commercial preparation of food emulsifiers. In Food Emulsifiers and Their Applications, 2nd ed.; Springer: Cham, Switzerlands, 2008; pp. 11–37. [Google Scholar] [CrossRef]
- Ansorge-Schumacher, M.B.; Thum, O. Immobilised lipases in the cosmetics industry. Chem. Soc. Rev. 2013, 42, 6475–6490. [Google Scholar] [CrossRef]
- Bolina, I.C.A.; Gomes, R.A.B.; Mendes, A.A. Biolubricant Production from Several Oleaginous Feedstocks Using Lipases as Catalysts: Current Scenario and Future Perspectives. Bioenergy Res. 2021. [Google Scholar] [CrossRef]
- Cecilia, J.A.; Plata, D.B.; Saboya, R.M.A.; de Luna, F.M.T.; Cavalcante, C.L.; Rodríguez-Castellón, E. An overview of the biolubricant production process: Challenges and future perspectives. Processes 2020, 8, 257. [Google Scholar] [CrossRef]
- Straathof, A.J.J. Transformation of biomass into commodity chemicals using enzymes or cells. Chem. Rev. 2014, 114, 1871–1908. [Google Scholar] [CrossRef]
- Halling, P.J. Solvent selection for biocatalysis in mainly organic systems: Predictions of effects on equilibrium position. Biotechnol. Bioeng. 1990, 35, 691–701. [Google Scholar] [CrossRef]
- Kasche, V. Mechanism and yields in enzyme catalysed equilibrium and kinetically controlled synthesis of β-lactam antibiotics, peptides and other condensation products. Enzym. Microb. Technol. 1986, 8, 4–16. [Google Scholar] [CrossRef]
- Kasche, V.; Haufler, U.; Riechmann, L. Equilibrium and Kinetically Controlled Synthesis with Enzymes: Semisynthesis of Penicillins and Peptides. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1987; Volume 136, pp. 280–292. ISBN 9780121820367. [Google Scholar]
- Halling, P.J. Thermodynamic predictions for biocatalysis in nonconventional media: Theory, tests, and recommendations for experimental design and analysis. Enzym. Microb. Technol. 1994, 16, 178–206. [Google Scholar] [CrossRef]
- Hari Krishna, S.; Karanth, N.G. Lipases and Lipase-Catalyzed Esterification Reactions in Nonaqueous Media. Catal. Rev. 2002, 44, 499–591. [Google Scholar] [CrossRef]
- Castillo, E.; Torres-Gavillan, A.; Sandoval, G.; Marty, A. Thermodynamical Methods for Optimization of Lipase-Catalyzed Reactions. In Lipases and Phospholipases; Humana Press: Totowa, NJ, USA, 2012; Volume 861, pp. 383–400. [Google Scholar]
- Colombié, S.; Tweddell, R.J.; Condoret, J.S.; Marty, A. Water activity control: A way to improve the efficiency of continuous lipase esterification. Biotechnol. Bioeng. 1998, 60, 362–368. [Google Scholar] [CrossRef]
- Castillo, E.; Dossat, V.; Marty, A.; Stéphane Condoret, J.; Combes, D. The role of silica gel in lipase-catalyzed esterification reactions of high-polar substrates. JAOCS J. Am. Oil Chem. Soc. 1997, 74, 77–85. [Google Scholar] [CrossRef]
- Dossat, V.; Combes, D.; Marty, A. Continuous enzymatic transesterification of high oleic sunflower oil in a packed bed reactor: Influence of the glycerol production. Enzym. Microb. Technol. 1999, 25, 194–200. [Google Scholar] [CrossRef]
- Marty, A.; Dossat, V.; Condoret, J.S. Continuous operation of lipase-catalyzed reactions in nonaqueous solvents: Influence of the production of hydrophilic compounds. Biotechnol. Bioeng. 1997, 56, 232–237. [Google Scholar] [CrossRef]
- Séverac, E.; Galy, O.; Turon, F.; Pantel, C.A.; Condoret, J.S.; Monsan, P.; Marty, A. Selection of CalB immobilization method to be used in continuous oil transesterification: Analysis of the economical impact. Enzym. Microb. Technol. 2011, 48, 61–70. [Google Scholar] [CrossRef]
- Martins, A.B.; Schein, M.F.; Friedrich, J.L.R.; Fernandez-Lafuente, R.; Ayub, M.A.Z.; Rodrigues, R.C. Ultrasound-assisted butyl acetate synthesis catalyzed by Novozym 435: Enhanced activity and operational stability. Ultrason. Sonochem. 2013, 20, 1155–1160. [Google Scholar] [CrossRef]
- Paludo, N.; Alves, J.S.; Altmann, C.; Ayub, M.A.Z.; Fernandez-Lafuente, R.; Rodrigues, R.C. The combined use of ultrasound and molecular sieves improves the synthesis of ethyl butyrate catalyzed by immobilized Thermomyces lanuginosus lipase. Ultrason. Sonochem. 2014, 22, 89–94. [Google Scholar] [CrossRef]
- Martins, A.B.; Friedrich, J.L.R.; Cavalheiro, J.C.; Garcia-Galan, C.; Barbosa, O.; Ayub, M.A.Z.; Fernandez-Lafuente, R.; Rodrigues, R.C. Improved production of butyl butyrate with lipase from Thermomyces lanuginosus immobilized on styrene-divinylbenzene beads. Bioresour. Technol. 2013, 134, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Fallavena, L.P.; Antunes, F.H.F.; Alves, J.S.; Paludo, N.; Ayub, M.A.Z.; Fernandez-Lafuente, R.; Rodrigues, R.C. Ultrasound technology and molecular sieves improve the thermodynamically controlled esterification of butyric acid mediated by immobilized lipase from Rhizomucor miehei. RSC Adv. 2014, 4, 8675–8681. [Google Scholar] [CrossRef]
- Graebin, N.G.; Martins, A.B.; Lorenzoni, A.S.G.; Garcia-Galan, C.; Fernandez-Lafuente, R.; Ayub, M.A.Z.; Rodrigues, R.C. Immobilization of lipase B from Candida antarctica on porous styrene-divinylbenzene beads improves butyl acetate synthesis. Biotechnol. Prog. 2012, 28, 406–412. [Google Scholar] [CrossRef]
- Poppe, J.K.; Garcia-Galan, C.; Matte, C.R.; Fernandez-Lafuente, R.; Rodrigues, R.C.; Ayub, M.A.Z. Optimization of synthesis of fatty acid methyl esters catalyzed by lipase B from Candida antarctica immobilized on hydrophobic supports. J. Mol. Catal. B Enzym. 2013, 94, 51–56. [Google Scholar] [CrossRef]
- Alves, J.S.; Garcia-Galan, C.; Schein, M.F.; Silva, A.M.; Barbosa, O.; Ayub, M.A.Z.; Fernandez-Lafuente, R.; Rodrigues, R.C. Combined effects of ultrasound and immobilization protocol on butyl acetate synthesis catalyzed by CALB. Molecules 2014, 19, 9562–9576. [Google Scholar] [CrossRef]
- Martins, A.B.; Graebin, N.G.; Lorenzoni, A.S.G.; Fernandez-Lafuente, R.; Ayub, M.A.Z.; Rodrigues, R.C. Rapid and high yields of synthesis of butyl acetate catalyzed by Novozym 435: Reaction optimization by response surface methodology. Process Biochem. 2011, 46, 2311–2316. [Google Scholar] [CrossRef]
- Aguieiras, E.C.G.; Ribeiro, D.S.; Couteiro, P.P.; Bastos, C.M.B.; de Queiroz, D.S.; Parreira, J.M.; Langone, M.A.P. Investigation of the Reuse of Immobilized Lipases in Biodiesel Synthesis: Influence of Different Solvents in Lipase Activity. Appl. Biochem. Biotechnol. 2016, 179, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Mulalee, S.; Srisuwan, P.; Phisalaphong, M. Influences of operating conditions on biocatalytic activity and reusability of Novozym 435 for esterification of free fatty acids with short-chain alcohols: A case study of palm fatty acid distillate. Chin. J. Chem. Eng. 2015, 23, 1851–1856. [Google Scholar] [CrossRef]
- Serrano-Arnaldos, M.; Montiel, M.C.; Ortega-Requena, S.; Máximo, F.; Bastida, J. Development and economic evaluation of an eco-friendly biocatalytic synthesis of emollient esters. Bioprocess Biosyst. Eng. 2019, 43, 495–505. [Google Scholar] [CrossRef]
- Aguieiras, E.C.G.; de Barros, D.S.N.; Sousa, H.; Fernandez-Lafuente, R.; Freire, D.M.G. Influence of the raw material on the final properties of biodiesel produced using lipase from Rhizomucor miehei grown on babassu cake as biocatalyst of esterification reactions. Renew. Energy 2017, 113, 112–118. [Google Scholar] [CrossRef]
- Alves, M.D.; Cren, É.C.; Mendes, A.A. Kinetic, thermodynamic, optimization and reusability studies for the enzymatic synthesis of a saturated wax ester. J. Mol. Catal. B Enzym. 2016, 133, S377–S387. [Google Scholar] [CrossRef]
- Bhavsar, K.V.; Yadav, G.D. Process intensification by microwave irradiation in immobilized-lipase catalysis in solvent-free synthesis of ethyl valerate. Mol. Catal. 2018, 461, 34–39. [Google Scholar] [CrossRef]
- Bhavsar, K.V.; Yadav, G.D. Microwave assisted solvent-free synthesis of n-butyl propionate by immobilized lipase as catalyst. Biocatal. Agric. Biotechnol. 2018, 14, 264–269. [Google Scholar] [CrossRef]
- Garcia, T.; Sanchez, N.; Martinez, M.; Aracil, J. Enzymatic synthesis of fatty esters Part II. Optimization studies. Enzym. Microb. Technol. 1999, 25, 591–597. [Google Scholar] [CrossRef]
- Hari Krishna, S.; Divakar, S.; Prapulla, S.G.; Karanth, N.G. Enzymatic synthesis of isoamyl acetate using immobilized lipase from Rhizomucor miehei. J. Biotechnol. 2001, 87, 193–201. [Google Scholar] [CrossRef]
- Chang, S.-W.; Shaw, J.-F.; Yang, K.-H.; Shih, I.-L.; Hsieh, C.-H.; Shieh, C.-J. Optimal lipase-catalyzed formation of hexyl laurate. Green Chem. 2005, 7, 547. [Google Scholar] [CrossRef]
- Güvenç, A.; Kapucu, N.; Kapucu, H.; Aydoǧan, Ö.; Mehmetoǧlu, Ü. Enzymatic esterification of isoamyl alcohol obtained from fusel oil: Optimization by response surface methodolgy. Enzym. Microb. Technol. 2007, 40, 778–785. [Google Scholar] [CrossRef]
- Mahapatra, P.; Kumari, A.; Kumar, V. Enzymatic synthesis of fruit flavor esters by immobilized lipase from Rhizopus oligosporus optimized with response surface methodology. J. Mol. Catal. B Enzym. 2009, 60, 57–63. [Google Scholar] [CrossRef]
- Richetti, A.; Leite, S.G.F.; Antunes, O.A.C.; Souza, A.L.F. De Optimization of 2-ethylhexyl palmitate Production Using Lipozyme RM IM as Catalyst in a Solvent-Free System. Appl. Biochem. Biotechnol. 2010, 160, 2498–2508. [Google Scholar] [CrossRef]
- Barros, D.; Azevedo, A.A.; Cabral, J.; Fonseca, L. Optimization Of Flavor Esters Synthesis By Fusarium Solani Pisi Cutinase. J. Food Biochem. 2012, 36, 275–284. [Google Scholar] [CrossRef]
- Rahman, I.N.A.; Manan, F.M.A.; Marzuki, N.H.C.; Mahat, N.A.; Attan, N.; Keyon, A.S.A.; Jamalis, J.; Aboul-Enein, H.Y.; Wahab, R.A. A statistical approach for optimizing the high yield green production of the flavor ester butyl butyrate. J. Teknol. 2017, 79, 141–151. [Google Scholar] [CrossRef][Green Version]
- Jaiswal, K.S.; Rathod, V.K. Acoustic cavitation promoted lipase catalysed synthesis of isobutyl propionate in solvent free system: Optimization and kinetic studies. Ultrason. Sonochem. 2018, 40, 727–735. [Google Scholar] [CrossRef]
- Serrano-Arnaldos, M.; Bastida, J.; Máximo, F.; Ortega-Requena, S.; Montiel, C. One-Step Solvent-Free Production of a Spermaceti Analogue Using Commercial Immobilized Lipases. ChemistrySelect 2018, 3, 748–752. [Google Scholar] [CrossRef]
- Santos, J.C.; Bueno, T.; Rós, P.C.M.; de Castro, H.F. Lipase-catalyzed Synthesis of Butyl Esters by Direct Esterification in Solvent-Free System. J. Chem. Technol. Biotechnol. 2007, 82, 956–961. [Google Scholar] [CrossRef]
- Karra-chaâbouni, M.; Ghamgui, H.; Bezzine, S.; Rekik, A.; Gargouri, Y. Production of flavour esters by immobilized Staphylococcus simulans lipases in a solvent-free system. Process Biochem. 2006, 41, 1692–1698. [Google Scholar] [CrossRef]
- Rocha, J.M.S.; Gil, M.H.; Garcia, F.A.P. Optimisation of the enzymatic synthesis of n-octyl oleate with immobilised lipase in the absence of solvents. J. Chem. Technol. Biotechnol. 1999, 74, 607–612. [Google Scholar] [CrossRef]
- Dai, W.C.; Chiu, S.J.; Huang, D.Y.; Juan, H.Y.; Chen, C.Y.; Chen, S.S.; Su, C.H.; Li, S.Y. Lipase-catalyzed synthesis of butyl propionate in solvent-free system: Optimization by response surface methodology. J. Taiwan Inst. Chem. Eng. 2014, 45, 2233–2237. [Google Scholar] [CrossRef]
- Sousa, R.R.; Costa, M.; Pinto, C.; Cristina, E.; Aguieiras, G.; Cipolatti, E.P.; Andrade, E.; Ayla, M.; Ana, S.; Pinto, J.C.; et al. Comparative performance and reusability studies of lipases on syntheses of octyl esters with an economic approach. Bioprocess Biosyst. Eng. 2021. [Google Scholar] [CrossRef] [PubMed]
- Ghamgui, H.; Karra-Chaabouni, M.; Gargouri, Y. 1-Butyl oleate synthesis by immobilized lipase from Rhizopus oryzae: A comparative study between n-hexane and solvent-free system. Enzym. Microb. Technol. 2004, 35, 355–363. [Google Scholar] [CrossRef]
- Ben Salah, R.; Ghamghui, H.; Miled, N.; Mejdoub, H.; Gargouri, Y. Production of butyl acetate ester by lipase from novel strain of Rhizopus oryzae. J. Biosci. Bioeng. 2007, 103, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Ghamgui, H.; Karra-chaabouni, M.; Bezzine, S.; Miled, N.; Gargouri, Y. Production of isoamyl acetate with immobilized Staphylococcus simulans lipase in a solvent-free system. Enzym. Microb. Technol. 2006, 38, 788–794. [Google Scholar] [CrossRef]
- Gawas, S.D.; Jadhav, S.V.; Rathod, V.K. Solvent Free Lipase Catalysed Synthesis of Ethyl Laurate: Optimization and Kinetic Studies. Appl. Biochem. Biotechnol. 2016, 180, 1428–1445. [Google Scholar] [CrossRef] [PubMed]
- Yadav, G.D.; Thorat, P.A. Microwave assisted lipase catalyzed synthesis of isoamyl myristate in solvent-free system. J. Mol. Catal. B Enzym. 2012, 83, 16–22. [Google Scholar] [CrossRef]
- Pereira, G.N.; Holz, J.P.; Giovannini, P.P.; Oliveira, J.V.; de Oliveira, D.; Lerin, L.A. Enzymatic esterification for the synthesis of butyl stearate and ethyl stearate. Biocatal. Agric. Biotechnol. 2018, 16, 373–377. [Google Scholar] [CrossRef]
- Goldberg, M.; Thomas, D.; Legoy, M.D. Water activity as a key parameter of synthesis reactions: The example of lipase in biphasic (liquid/solid) media. Enzym. Microb. Technol. 1990, 12, 976–981. [Google Scholar] [CrossRef]
- Nordblad, M.; Adlercreutz, P. Effects of acid concentration and solvent choice on enzymatic acrylation by Candida antarctica lipase B. J. Biotechnol. 2008, 133, 127–133. [Google Scholar] [CrossRef]
- Bélafi-Bakó, K.; Badr, A.K.; Ehrenstein, U.; Gubicza, L. Kinetics of Ethyl Acetate Formation by Lipase in Organic Solvent and Solvent-Free System. Chem. Pap. 2003, 57, 278–281. [Google Scholar]
- Flores, M.V.; Sewalt, J.J.W.; Janssen, A.E.M.; Van Der Padt, A. The Nature of Fatty Acid Modifies the Equilibrium Position in the Esterification Catalyzed by Lipase. Biotechnol. Bioeng. 2000, 67, 364–371. [Google Scholar] [CrossRef]
- Paiva, A.L.; Balcão, V.M.; Malcata, F.X. Kinetic and mechanisms of reactions catalyzed by immobilized lipases. Enzym. Microb. Technol. 2000, 27, 187–204. [Google Scholar] [CrossRef]
- Foresti, M.L.; Pedernera, M.; Ferreira, M.L.; Bucalá, V. Kinetic modeling of enzymatic ethyl oleate synthesis carried out in biphasic systems. Appl. Catal. A Gen. 2008, 334, 65–72. [Google Scholar] [CrossRef]
- Lopresto, G.C.; Calabrò, V.; Woodley, J.M.; Tufvesson, P. Enzymatic kinetic study on the enzymatic esterification of octanoic acid and hexanol by immobilized Candida antarctica lipase B. J. Mol. Catal. B. Enzym. 2014, 110, 64–71. [Google Scholar] [CrossRef]
- Eggers, D.K.; Blanch, H.W.; Prausnitz, J.M. Extractive catalysis: Solvent effects on equilibria of enzymatic reactions in two-phase systems. Enzym. Microb. Technol. 1989, 11, 84–89. [Google Scholar] [CrossRef]
- Valivety, R.H.; Johnston, G.A.; Suckling, C.J.; Halling, P.J. Solvent effects on biocatalysis in organic systems: Equilibrium position and rates of lipase catalyzed esterification. Biotechnol. Bioeng. 1991, 38, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Janseen, A.E.M.; Van der Padt, A.; Van Sonsbeek, H.M.; Van’t Riet, K. The effect of organic solvents on the equilibrium position of enzymatic acylglycerol synthesis. Biotechnol. Bioeng. 1993, 41, 95–103. [Google Scholar] [CrossRef]
- Janssen, A.E.M.; Van Der Padt, A.; Van’t Riet, K. Solvent Effects on Lipase-Catalyzed Esterification of Glycerol and Fatty Acids. Biotechnol. Bioeng. 1993, 42, 953–962. [Google Scholar] [CrossRef]
- Güvenç, A.; Kapucu, N.; Mehmetoǧlu, Ü. The production of isoamyl acetate using immobilized lipases in a solvent-free system. Process Biochem. 2002, 38, 379–386. [Google Scholar] [CrossRef]
- Bucalá, V.; Briozzo, M.; Foresti, M.L.; Ferreira, M.L.; Trubiano, G.; Bottini, S. Influence of liquid-liquid equilibria on the modelling of solvent-free ethyl oleate synthesis. In Proceedings of the 2nd Mercosur Congress on Chemical Engineering, Rio de Janeiro, Brazil, 14–18 August 2005; pp. 1–10. [Google Scholar]
- Garcia, S.; Vidinha, P.; Arvana, H.; Gomes Da Silva, M.D.R.; Ferreira, M.O.; Cabral, J.M.S.; Macedo, E.A.; Harper, N.; Barreiros, S. Cutinase activity in supercritical and organic media: Water activity, solvation and acid-base effects. J. Supercrit. Fluids 2005, 35, 62–69. [Google Scholar] [CrossRef][Green Version]
- Garcia, T.; Sanchez, N.; Martinez, M.; Aracil, J. Enzymatic synthesis of fatty esters. Part I. Kinetic approach. Enzym. Microb. Technol. 1999, 25, 584–590. [Google Scholar] [CrossRef]
- Martins, A.B.; Da Silva, A.M.; Schein, M.F.; Garcia-Galan, C.; Záchia Ayub, M.A.; Fernandez-Lafuente, R.; Rodrigues, R.C. Comparison of the performance of commercial immobilized lipases in the synthesis of different flavor esters. J. Mol. Catal. B Enzym. 2014, 105, 18–25. [Google Scholar] [CrossRef]
- Barbosa, O.; Ortiz, C.; Torres, R.; Fernandez-Lafuente, R. Effect of the immobilization protocol on the properties of lipase B from Candida antarctica in organic media: Enantiospecifc production of atenolol acetate. J. Mol. Catal. B Enzym. 2011, 71, 124–132. [Google Scholar] [CrossRef]
- Fernández-Lorente, G.; Betancor, L.; Carrascosa, A.V.; Palomo, J.M.; Guisan, J.M. Modulation of the selectivity of immobilized lipases by chemical and physical modifications: Release of omega-3 fatty acids from fish oil. JAOCS J. Am. Oil Chem. Soc. 2012, 89, 97–102. [Google Scholar] [CrossRef]
- Foresti, M.L.; Ferreira, M.L. Solvent-free ethyl oleate synthesis mediated by lipase from Candida antarctica B adsorbed on polypropylene powder. Catal. Today 2005, 107–108, 23–30. [Google Scholar] [CrossRef]
- Condoret, J.S.; Vankan, S.; Joulia, X.; Marty, A. Prediction of water adsorption curves for heterogeneous biocatalysis in organic and supercritical solvents. Chem. Eng. Sci. 1997, 52, 213–220. [Google Scholar] [CrossRef]
- Eby, J.M.; Peretti, S.W. Performance in synthetic applications of a yeast surface display-based biocatalyst. RSC Adv. 2015, 5, 30425–30432. [Google Scholar] [CrossRef]
- Tacias-Pascacio, V.G.; Virgen-Ortíz, J.J.; Jiménez-Pérez, M.; Yates, M.; Torrestiana-Sanchez, B.; Rosales-Quintero, A.; Fernandez-Lafuente, R. Evaluation of different lipase biocatalysts in the production of biodiesel from used cooking oil: Critical role of the immobilization support. Fuel 2017, 200, 1–10. [Google Scholar] [CrossRef]
- Khan, N.R.; Rathod, V.K. Microwave mediated lipase-catalyzed synthesis of n-butyl palmitate and thermodynamic studies. Biocatal. Agric. Biotechnol. 2020, 29, 101741. [Google Scholar] [CrossRef]
- Baum, S.; Mueller, J.J.; Hilterhaus, L.; Eckstein, M.; Thum, O.; Liese, A. The bubble column reactor: A novel reactor type for cosmetic esters. In Applied Biocatalysis: From Fundamental Science to Industrial Applications; Wiley: Hoboken, NJ, USA, 2016; pp. 343–366. [Google Scholar] [CrossRef]
- Sousa, R.R.; Silva, A.S.; Fernandez-Lafuente, R.; Ferreira-Leitão, V.S. Solvent-free esterifications mediated by immobilized lipases: A review from thermodynamic and kinetic perspectives. Catal Sci. Technol. 2021, 11, 5696–5711. [Google Scholar] [CrossRef]


| Acid | Alcohol | Lipase | Immobilization Support | T (°C) | Conversion Response Reported (%) | SER Calculated Range | SER with the Highest Conversion | Ref. |
|---|---|---|---|---|---|---|---|---|
| Good level of agreement | ||||||||
| Metanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 40 | 55.0–93.0% | 35 to 39 | 36 (93.0%) | [8] |
| Metanoic Acid | n-Octanol | Novozym 435® | Lewatit VP OC 1600 | 40 | - | - | 35 (92.0%) | [8] |
| Propionic Acid | Isobutanol | Fermase CALB 10000 | Polyglycidemethaacrylate | 60 | 71.8–94.2% | 0 to 20 | 13 (94.2%) *** | [65] |
| Propionic Acid | Isobutanol | Novozym 435® | Lewatit VP OC 1600 | 40 | 63.8–92.5% | 0 to 30 | 20 (92.5%) | [4] |
| Butanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 37 | 41.0–48.6% | −3 to −1 | −1 (48.6%) * | [67] |
| Pentanoic Acid | Ethanol | Lipase from S. simulans | CaCO3 | 37 | 29.0–51.0% | −20 to −4 | −4 (51.0%) ** | [68] |
| Pentanoic Acid | Ethanol | Novozym 435® | Lewatit VP OC 1600 | 50 | 40.0–69.0% | −50 to −21 | −33 (69.0%) *** | [55] |
| Octanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 37 | - | - | −4 (38.0%) * | [67] |
| Octanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 50 | 85.0–89.0% | −24 to 27 | 27 (89.0%) | Data not published |
| Octanoic Acid | n-Butanol | Lipozyme RM IM® | Duolite ES 562 | 50 | 68.2–89.0% | −12 to 14 | 14 (89.0%) | Data not published |
| Octanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 60 | 76.5–92.5% | −14 to 9 | 0.5 (92.5%) | [7] |
| Octanoic Acid | n-Octanol | Novozym 435® | Lewatit VP OC 1600 | 65 | 74.7–96.1% | −19 to 207 | 9 (96.1%) | [6] |
| Octanoic Acid | n-Octanol | Lipozyme RM IM® | Duolite ES 562 | 65 | 19.6–99.0% | −10 to 171 | 4 (99.0%) | Data not published |
| Decanoic Acid | n-Propanol | Fermase CALB 10,000 | Polyglycidemethaacrylate | 60 | 19.0–83.8% | −96 to 8 | 1 (83.8%) | [9] |
| Dodecanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 37 | 28.0–35.0% | −10 to −3 | −10 (35.0%) | [67] |
| Dodecanoic Acid | Hexadecan-1-ol | Lipozyme RM IM® | Duolite ES 562 | 70 | 67.0–98.1% | 4 to 32 | 4 (98.1%) | [66] |
| Tetradecanoic Acid | Isopropanol | Novozym 435® | Lewatit VP OC 1600 | 60 | 7.0–87.7% | 7 to 60 | 15 (87.7%) | [5] |
| Tetradecanoic Acid | Hexadecan-1-ol | Lipozyme RM IM® | Duolite ES 562 | 70 | 65.9–97.3% | 4 to 33 | 4 (97.3%) | [66] |
| Hexadecanoic Acid | Hexadecan-1-ol | Lipozyme RM IM® | Duolite ES 562 | 70 | 61.8–97.1% | 4 to 33 | 4 (97.1%) | [66] |
| Octadecanoic Acid | Hexadecan-1-ol | Lipozyme RM IM® | Duolite ES 562 | 70 | 60.5–95.8% | 4 to 33 | 4 (95.8%) | [66] |
| Octadec-9-enoic Acid | n-Octanol | Lipase from R. miehei | Poly(ehtylene)-g-co-hydroxyethyl methaacrylate | 37 | - | - | 56 (82.0%) | [69] |
| Intermediate level of agreement | ||||||||
| Propionic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 45 | 81.2–92.7% | 100 to 600 | 100 (92.7%) | [70] |
| Propionic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 60 | 35.0–92.0% | 39 to 160 | 85 (92.0%) *** | [56] |
| Butanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 53.9 | - | - | 91 (99.6%) | [64] |
| Decanoic Acid | n-Octanol | Novozym 435® | Lewatit VP OC 1600 | 50 | 92.0–96.0% | −1 to 0 | −0.4 (96.0%) | [71] |
| DecanoicAcid | n-Octanol | Lipozyme RM IM® | Duolite ES 562 | 50 | 88.8–95.1% | −1 to 0 | −0.2 (95.1%) | [71] |
| Dodecanoic Acid | n-Octanol | Novozym 435® | Lewatit VP OC 1600 | 50 | 93.9–96.3% | −10 to −1.5 | −1.5 (96.3%) | [71] |
| Dodecanoic Acid | n-Octanol | Lipozyme RM IM® | Duolite ES 562 | 50 | 86.7–97.7% | −10 to −1.5 | −4 (97.7%) | [71] |
| Octadec-9-enoic Acid | n-Butanol | Lipase from R. oryzae | CaCO3 | 37 | 18.0–81.0% | −54 to −5 | −5 (81.0%) | [72] |
| Low level of agreement | ||||||||
| Etanoic Acid | n-Butanol | Lipase from R. oryzae | Celite 545 | 37 | 12.0–61.0% | −7.5 to 9 | 1 (61.0%) ** | [73] |
| Etanoic Acid | Isopentanol | Lipase from S. simulans | CaCO3 | 37 | 2.0–64.0% | −26 to 35 | −7 (64.0%) ** | [74] |
| Etanoic Acid | Isopentanol | Novozym 435® | Lewatit VP OC 1600 | 30 | 46.7–68.4% | 3 to 10 | 3 (68.4%) | [60] |
| Etanoic Acid | n-Hexanol | Lipase from S. simulans | CaCO3 | 37 | 21.0–43.0% | 3 to 14 | 3 (43.0%) ** | [68] |
| Dodecanoic Acid | Ethanol | Fermase CALB 10,000 | Polyglycidemethaacrylate | 60 | 67.0–92.4% | −31 to −2 | −15 (92.4%) | [75] |
| TetradecanoicAcid | Isopentanol | Novozym 435® | Lewatit VP OC 1600 | 60 | 82.0–97.0% | −197 to 17 | −99 (97.0%) *** | [76] |
| Hexadecanoic Acid | Isopropanol | Novozym 435® | Lewatit VP OC 1600 | 75 | 33.5–88.0% | −35 to −10 | −10 (88.0%) | [57] |
| Octadecanoic Acid | Ethanol | Novozym 435® | Lewatit VP OC 1600 | 60 | 66.0–92.0% | −65 to 27 | −19 (92.0%) | [77] |
| Octadecanoic Acid | n-Butanol | Novozym 435® | Lewatit VP OC 1600 | 60 | 61.0–92.0% | −74 to 104 | −48 (92.0%) | [77] |
| Octadec-9-enoic Acid | Ethanol | Lipozyme® | Duolite A568 | 40 | 72.0–99.0% | −41 to −4 | −41 (99.0%) | [1] |
| Reaction Condition | Initial Condition of the Media | Effect on Biocatalyst | Condition on Equilibrium | Possible Optimization Path |
|---|---|---|---|---|
| Large stoichiometric excess of a highly polar reagent | Possible strip-off of essential water on enzymes pH acid (if exceeding reagent is the acid) Low log P; low viscosity | Possible reduction in the enzymatic activity | Accumulation of the water on the organic phase pH slightly acid Low log P; increased viscosity | Adoption of high biocatalyst loadings Control of the water activity Fractioned additions of the exceeding reagent |
| Discrete excess of a highly polar reagent | pH slightly acid (if the surplus reagent is the acid) Limited solubility/poor solvation (if limiting reagent is non-polar) Intermediate log P; intermediate viscosity (if limiting reagent have a long-chain) | - | pH neutral (or slightly acid) Intermediate log P; considerable viscosity | Possibility of adopting reduced biocatalyst loadings |
| Discrete excess of a non-polar reagent | pH slightly acid (if the surplus reagent is the acid) Limited solubility/poor solvation (if limiting reagent is highly-polar) Intermediate log P; intermediate viscosity (if limiting reagent have a long-chain) | - | pH neutral (or slightly acid) Intermediate log P; considerable viscosity | Possibility of adopting reduced biocatalyst loadings |
| Large stoichiometric excess of a non-polar reagent | pH acid (or slightly acid) High log P; high viscosity (if surplus reagent have a long-chain) | Possible reduction in the enzymatic activity | Accumulation of the water on catalytic phase pH neutral (or slightly acid) High log P; high viscosity | Adoption of high biocatalyst loadings Control of the water activity Increase the temperature |
| Acid:Alcohol Molar Ratio | Biocatalyst Loading (% wt/wt Acid) | Biocatalyst Loading (% wt/wt Total) | SER | Conversion (%) |
|---|---|---|---|---|
| 1:1 | 0.7 | 0.5 | −97 | 19% |
| 1:1 | 1.3 | 1.0 | −48 | 47% |
| 1:1 | 2.7 | 2.0 | −24 | 59% |
| 1:1 | 4.0 | 3.0 | −16 | 62% |
| 1:2 | 3.4 | 2.0 | −9 | 71% |
| 1:3 | 4.1 | 2.0 | 1 | 83% |
| 1:4 | 4.8 | 2.0 | 8 | 78% |
| 2:1 | 2.3 | 2.0 | −35 | 36% |
| 3:1 | 2.2 | 2.0 | −40 | 24% |
| Synthesis (Product) | Pentyl Propionate | Ethyl Hexadecanoate | Dodecyl Hexanoate |
|---|---|---|---|
| Enzyme (commercial lipase) | Novozym 435® (7000 PLU) | Novozym 435® (7000 PLU) | Novozym 435® (7000 PLU) |
| Temperature | 60 °C | 70 °C | 60 °C |
| Initial quantity of acid | 1.85 g (0.025 mol) | 1.85 g (0.0072 mol) | 1.85 g (0.016 mol) |
| Minimum biocatalyst loading | 0.037 g (260 PLU) | 0.019 g (135 PLU) | 0.037 g (260 PLU) |
| Maximum biocatalyst loading | 0.065 g (455 PLU) | 0.037 g (260 PLU) | 0.065 g (455 PLU) |
| SER and the respective reaction conditions | SER 18 Molar ratio 1:1.1/Bioc. loading 2.0% | SER −43 Molar ratio 1:3.2/Bioc. loading 1.0% | SER 27 Molar ratio 1:0.9/Bioc. loading 2.0% |
| SER 24 Molar ratio 1:1.5/Bioc. loading 3.5% | SER −30 Molar ratio 1:3.9/Bioc. loading 1.0% | SER 38 Molar ratio 1:1.1/Bioc. loading 2.0% | |
| SER 39 Molar ratio 1:2/Bioc. loading 2.0% | SER −18 Molar ratio 1:3.6/Bioc. loading 2.0% | SER 42 Molar ratio 1:1.4/Bioc. loading 3.5% | |
| SER 42 Molar ratio 1:1.5/Bioc. loading 2.0% | SER −1 Molar ratio 1:5.4/Bioc. loading 2.0% | SER 68 Molar ratio 1:1.7/Bioc. loading 3.5% | |
| SER 74 Molar ratio 1:3/Bioc. loading 3.5% | SER 4 Molar ratio 1:6/Bioc. loading 2.0% | SER 75 Molar ratio 1:2/Bioc. loading 3.5% |
| Ethyl Dodecanoate | Butyl Dodecanoate | Octyl Dodecanoate | Butyl Octanoate | Butyl Decanoate | |||||
|---|---|---|---|---|---|---|---|---|---|
| SER | Conversion | SER | Conversion | SER | Conversion | SER | Conversion | SER | Conversion |
| 13 (1:5.2) (1.5%) | 89.0% | 8 (1:3) (1.5%) | 88.9% | 10 (1:1.7) (1.5%) | 90.7% | 1 (1:1.9) (1.5%) | 87.4% | 7 (1:2.6) (1.5%) | 89.5% |
| 19 (1:6.4) (2.5%) | 85.2% | 27 (1:3.7) (2.5%) | 92.1% | 16 (1:2.1) (2.5%) | 90.9% | 13 (1:2.7) (2.5%) | 90.3% | 20 (1:3.5) (2.5%) | 91.9% |
| Advantages |
| Predictability of reaction’ s behavior |
| Easy to handle |
| Reduced number of experiments to achieve high yields |
| Offer a range of reaction conditions that achieve high yields |
| Possibility to reduce the biocatalyst loading keeping high yields |
| Disadvantages |
| One SER number may be associated with more than one reaction condition |
| Optimum range may be shifted depending on the nature of the reagents |
| Temperature effects are not considered |
| Differences in molar ratio of reagents are not considered |
| Differences in esterification activity of lipases are not considered |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousa, R.R.d.; Silva, A.S.d.; Fernandez-Lafuente, R.; Ferreira-Leitão, V.S. Simplified Method to Optimize Enzymatic Esters Syntheses in Solvent-Free Systems: Validation Using Literature and Experimental Data. Catalysts 2021, 11, 1357. https://doi.org/10.3390/catal11111357
Sousa RRd, Silva ASd, Fernandez-Lafuente R, Ferreira-Leitão VS. Simplified Method to Optimize Enzymatic Esters Syntheses in Solvent-Free Systems: Validation Using Literature and Experimental Data. Catalysts. 2021; 11(11):1357. https://doi.org/10.3390/catal11111357
Chicago/Turabian StyleSousa, Ronaldo Rodrigues de, Ayla Sant’Ana da Silva, Roberto Fernandez-Lafuente, and Viridiana Santana Ferreira-Leitão. 2021. "Simplified Method to Optimize Enzymatic Esters Syntheses in Solvent-Free Systems: Validation Using Literature and Experimental Data" Catalysts 11, no. 11: 1357. https://doi.org/10.3390/catal11111357
APA StyleSousa, R. R. d., Silva, A. S. d., Fernandez-Lafuente, R., & Ferreira-Leitão, V. S. (2021). Simplified Method to Optimize Enzymatic Esters Syntheses in Solvent-Free Systems: Validation Using Literature and Experimental Data. Catalysts, 11(11), 1357. https://doi.org/10.3390/catal11111357







