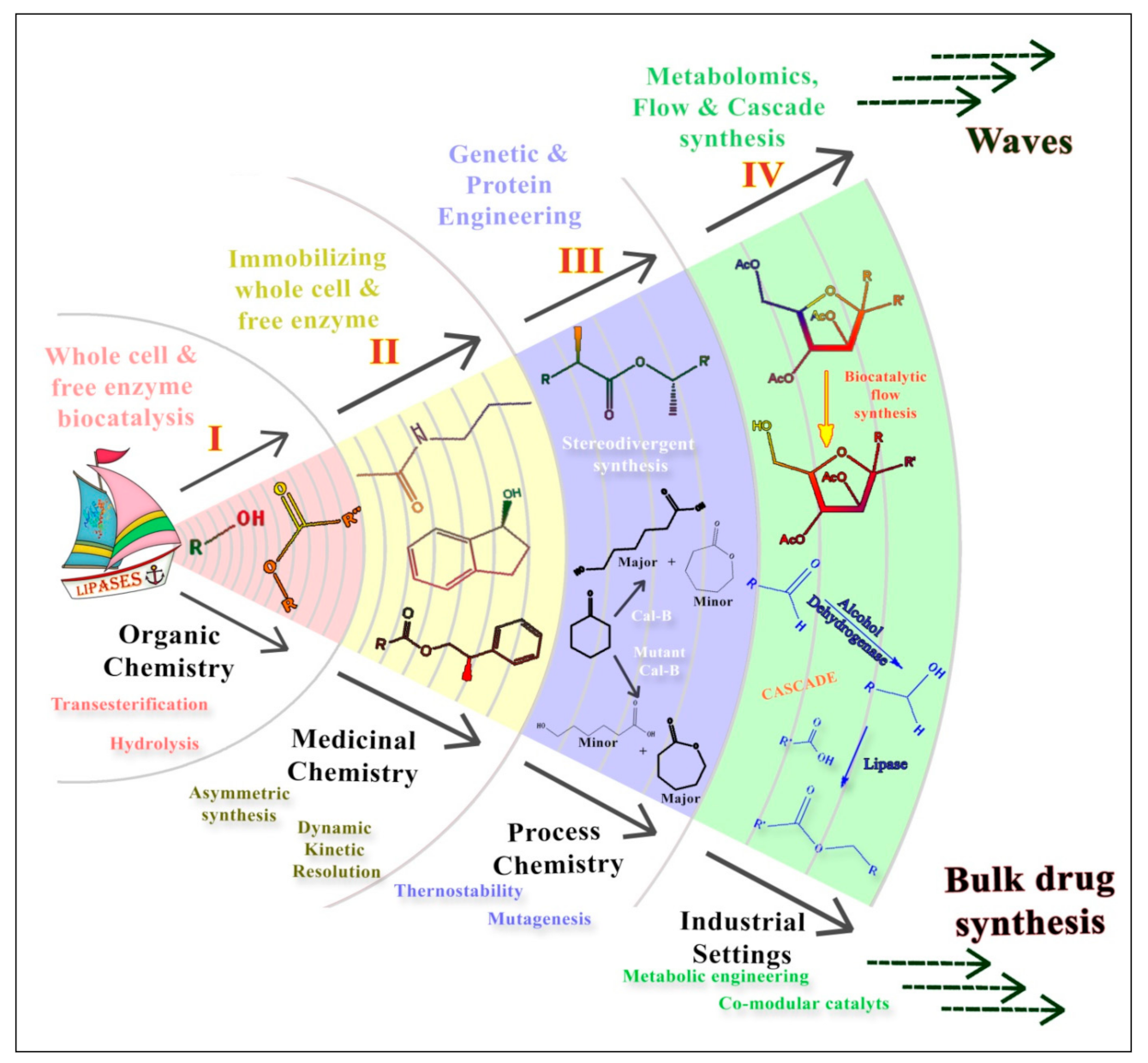
Diversifying Arena of Drug Synthesis: In the Realm of Lipase Mediated Waves of Biocatalysis
Abstract
:1. Introduction
2. Lipases: A Versatile Manifesto
3. Second Wave of Lipase Mediated Drug Synthesis
4. Third Wave of Lipase Mediated Drug Synthesis
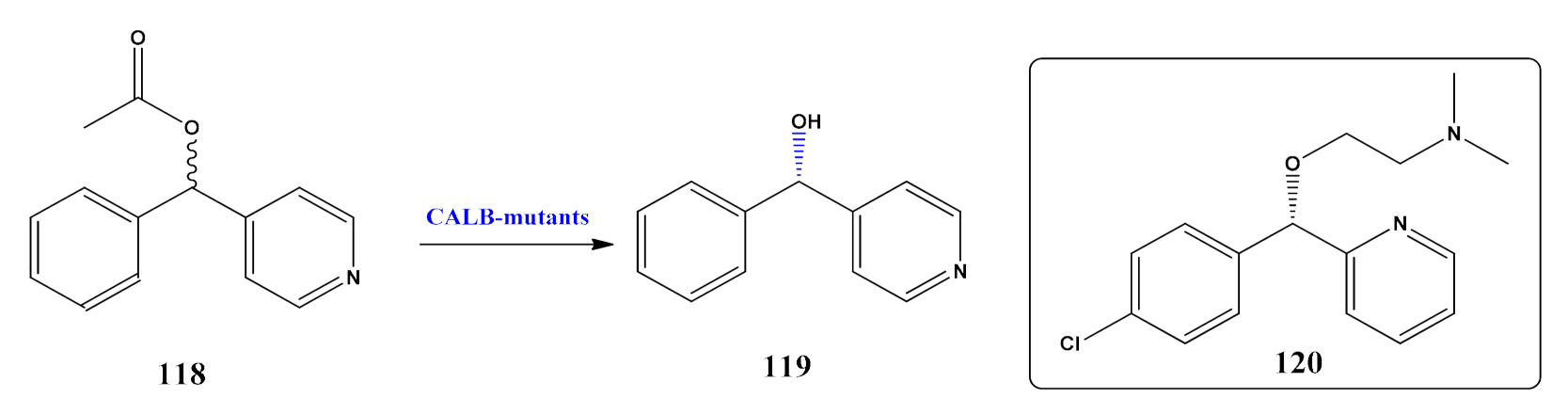
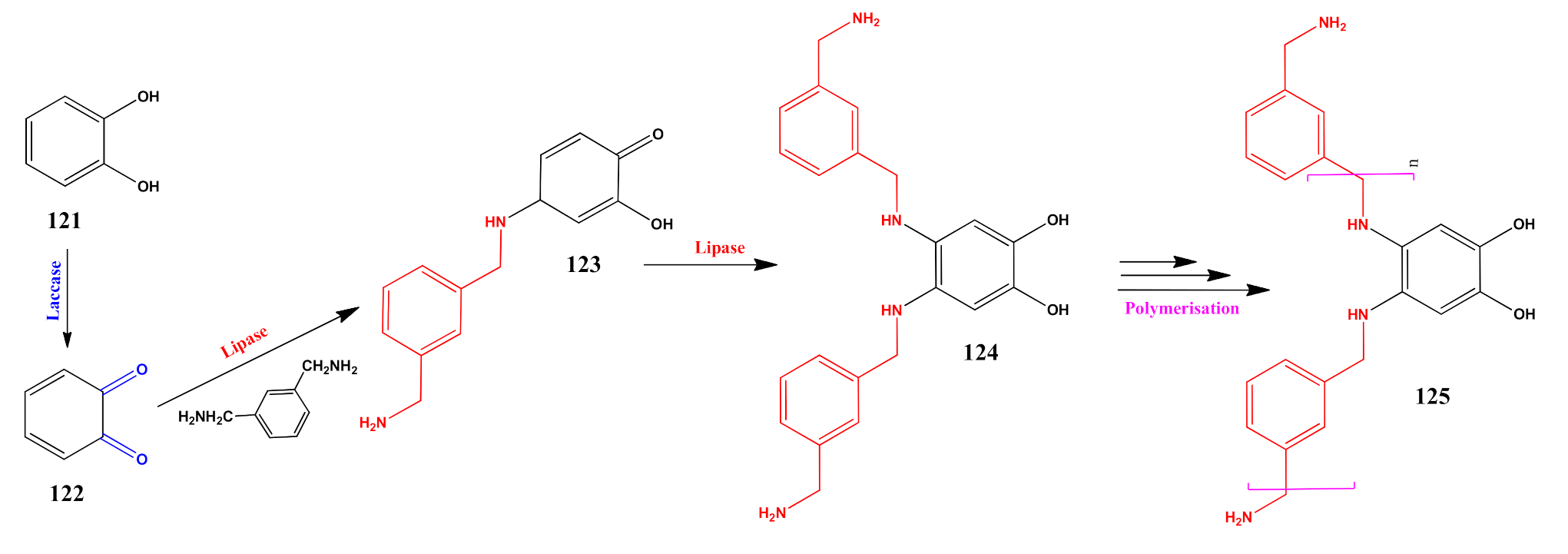
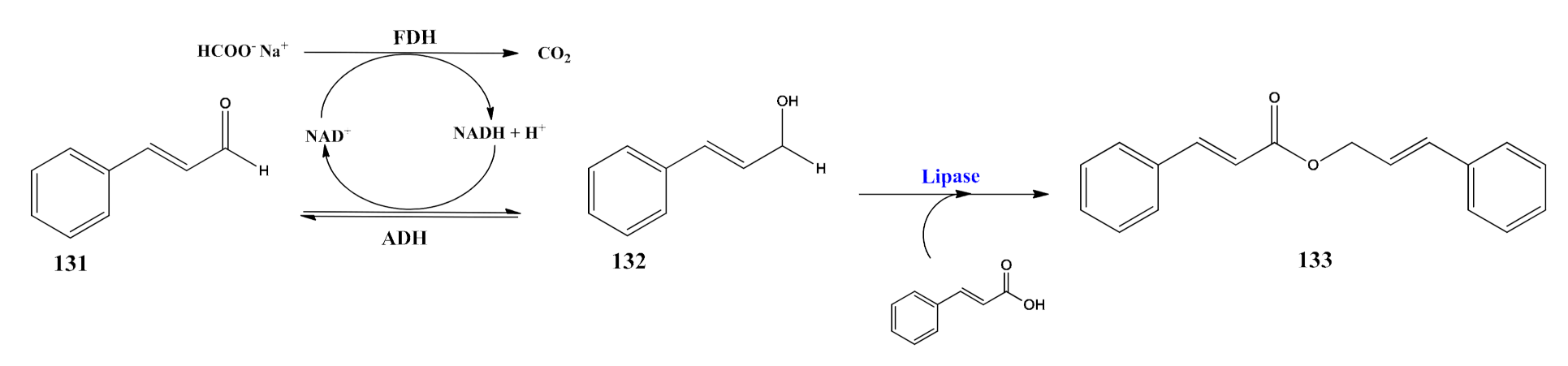
5. Burgeoning Fourth Wave of Biocatalysis
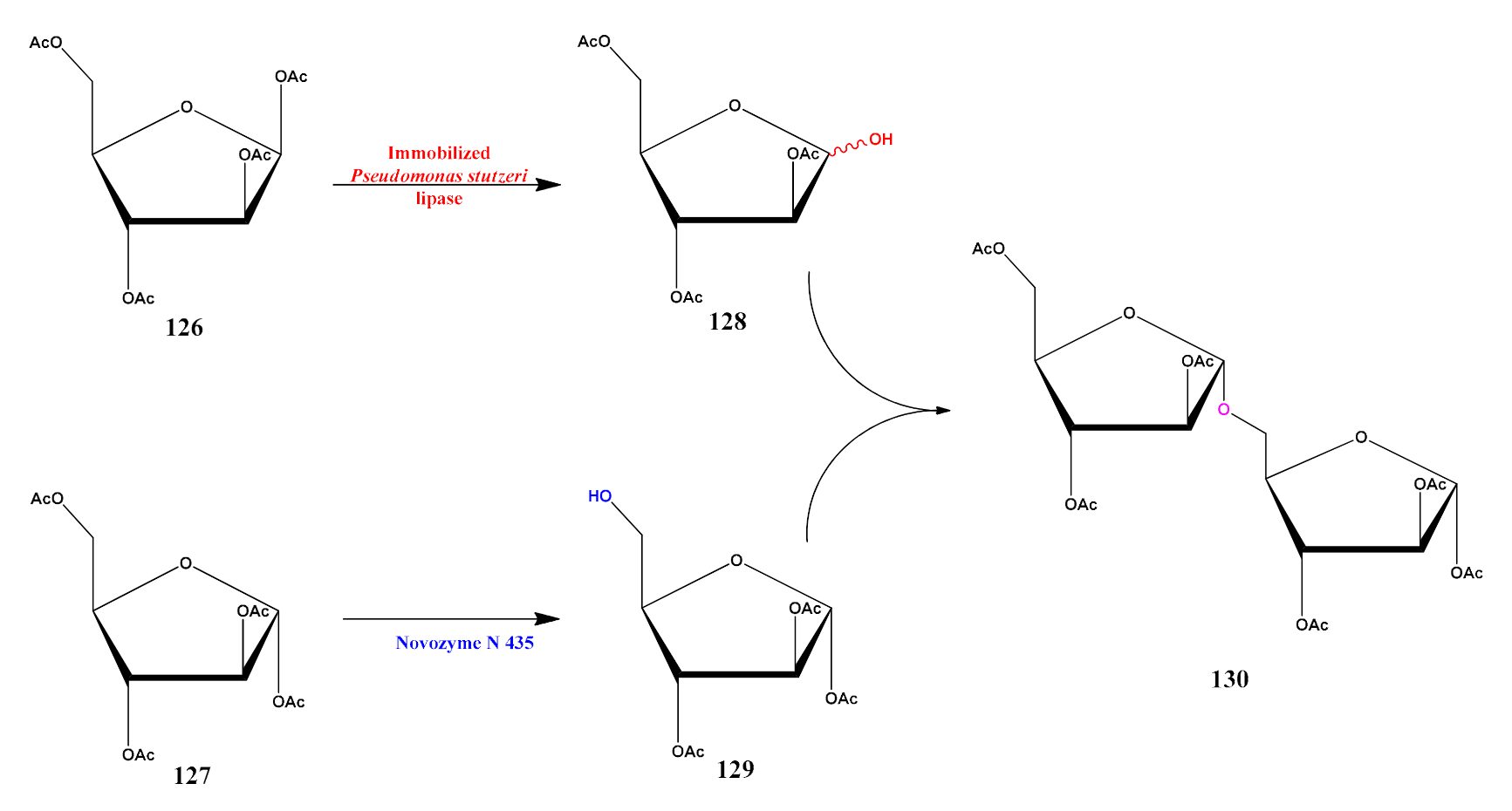
6. Lipase Mediated Late-Stage Functionalization (LSF)
7. Conclusions and Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bornscheuer, U.T.; Huisman, G.W.; Kazlauskas, R.J.; Lutz, S.; Moore, J.C.; Robins, K. Engineering the third wave of biocatalysis. Nature 2012, 485, 185–194. [Google Scholar] [CrossRef]
- Cui, J.; Zhao, Y.; Liu, R.; Zhong, C.; Jia, S. Surfactant-activated lipase hybrid nanoflowers with enhanced enzymatic performance. Sci. Rep. 2016, 6, 27928. [Google Scholar] [CrossRef] [Green Version]
- Suo, H.; Xu, L.; Xu, C.; Qiu, X.; Chen, H.; Huang, H.; Hu, Y. Graphene oxide nanosheets shielding of lipase immobilized on magnetic composites for the improvement of enzyme stability. ACS Sustain. Chem. Eng. 2019, 7, 4486–4494. [Google Scholar] [CrossRef]
- De Simone, A.; Hoesl, M.G.; Budisa, N. Engineering Lipases with an Expanded Genetic Code; WILEY-VHC: Weinheim, Germany, 2016; pp. 3–12. [Google Scholar]
- Sarmah, N.; Revathi, D.; Sheelu, G.; Yamuna Rani, K.; Sridhar, S.; Mehtab, V.; Sumana, C. Recent advances on sources and industrial applications of lipases. Biotechnol. Prog. 2018, 34, 5–28. [Google Scholar] [CrossRef]
- Anobom, C.D.; Pinheiro, A.S.; De-Andrade, R.A.; Aguieiras, E.C.; Andrade, G.C.; Moura, M.V.; Almeida, R.V.; Freire, D.M. From structure to catalysis: Recent developments in the biotechnological applications of lipases. BioMed Res. Int. 2014, 2014, 684506. [Google Scholar] [CrossRef] [Green Version]
- Fotiadou, R.; Patila, M.; Hammami, M.A.; Enotiadis, A.; Moschovas, D.; Tsirka, K.; Spyrou, K.; Giannelis, E.P.; Avgeropoulos, A.; Paipetis, A.; et al. Development of effective lipase-hybrid nanoflowers enriched with carbon and magnetic nanomaterials for biocatalytic transformations. Nanomaterials 2019, 9, 808. [Google Scholar] [CrossRef] [Green Version]
- Du, X.; Liu, X.; Li, Y.; Wu, C.; Wang, X.; Xu, P. Efficient biocatalyst by encapsulating lipase into nanoporous gold. Nanoscale Res. Lett. 2013, 8, 180. [Google Scholar] [CrossRef] [Green Version]
- Altosaar, I.; Giband, M.; Schernthaner, J.P.; Tanchak, M.A.; Sardana, R.K.; Potier, B. Strategies and tactics for cloning genes, coding for lipase, from higher plants. In Recent Advances in Biotechnology; Springer: Dordrecht, The Netherlands, 1992; pp. 373–381. [Google Scholar]
- Mohammadi, M.; Sepehrizadeh, Z.; Ebrahim-Habibi, A.; Shahverdi, A.R.; Faramarzi, M.A.; Setayesh, N. Enhancing activity and thermostability of lipase A from Serratia marcescens by site-directed mutagenesis. Enzym. Microb. Technol. 2016, 93–94, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Riva, S. 1983–2013: The long wave of biocatalysis. Trends Biotechnol. 2013, 31, 120–121. [Google Scholar] [CrossRef]
- Dwivedee, B.P.; Soni, S.; Sharma, M.; Bhaumik, J.; Laha, J.K.; Banerjee, U.C. Promiscuity of lipase-catalyzed reactions for organic synthesis: A recent update. ChemistrySelect 2018, 3, 2441–2466. [Google Scholar] [CrossRef]
- UniProt Knowledgebase for Enzymes. Enzyme Classification 3.-.-.-. UniProt. Available online: https://www.uniprot.org/uniprot/?query=ec:3.-.-.-%20reviewed:yes (accessed on 14 September 2021).
- Simeó, Y.; Sinisterra, J.V.; Alcántara, A.R. Regioselective enzymatic acylation of pharmacologically interesting nucleosides in 2-methyltetrahydrofuran, a greener substitute for THF. Green Chem. 2009, 11, 855–862. [Google Scholar] [CrossRef]
- Goswami, A.; Kissick, T.P. Enzymatic desymmetrization of dimethyl cylcohex-4-ene-cis-1, 2-dicarboxylate to (1 S, 2 R)-2-(methoxycarbonyl) cyclohex-4-ene-1-carboxylic acid. Org. Process. Res. Dev. 2009, 13, 483–488. [Google Scholar] [CrossRef]
- Palocci, C.; Falconi, M.; Chronopoulou, L.; Cernia, E. Lipase-catalyzed regioselective acylation of tritylglycosides in supercritical carbon dioxide. J. Supercrit. Fluids 2008, 45, 88–93. [Google Scholar] [CrossRef]
- de María, P.D.; de Gonzalo, G.; Alcántara, A.R. Biocatalysis as useful tool in asymmetric synthesis: An assessment of recently granted patents (2014–2019). Catalysts 2019, 9, 802. [Google Scholar] [CrossRef] [Green Version]
- Reetz, M.T. Biocatalysis in organic chemistry and biotechnology: Past, present, and future. J. Am. Chem. Soc. 2013, 135, 12480–12496. [Google Scholar] [CrossRef]
- Kapoor, M.; Gupta, M.N. Lipase promiscuity and its biochemical applications. Process. Biochem. 2012, 47, 555–569. [Google Scholar] [CrossRef]
- Gandhi, N.N. Applications of lipase. J. Am. Oil Chem. Soc. 1997, 74, 621–634. [Google Scholar] [CrossRef]
- Bora, L.; Gohain, D.; Das, R. Recent advances in production and biotechnological applications of thermostable and alkaline bacterial lipases. J. Chem. Technol. Biotechnol. 2013, 88, 1959–1970. [Google Scholar] [CrossRef]
- Kapoor, M.; Majumder, A.B.; Mukherjee, J.; Gupta, M.N. Decarboxylative aldol reaction catalysed by lipases and a protease in organic co-solvent mixtures and nearly anhydrous organic solvent media. Biocatal. Biotransform. 2012, 30, 399–408. [Google Scholar] [CrossRef]
- Kannan, K.; Mukherjee, J.; Gupta, M.N. Immobilization of a lipase on mesocellular foam of silica for biocatalysis in low-water-containing organic solvents. Chem. Lett. 2014, 43, 1064–1066. [Google Scholar] [CrossRef]
- Roy, I.; Mukherjee, J.; Gupta, M.N. High activity preparations of lipases and proteases for catalysis in low water containing organic solvents and ionic liquids. In Immobilization of Enzymes and Cells. Methods in Molecular Biology (Methods and Protocols); Humana Press: Totowa, NJ, USA, 2013; Volume 1051, pp. 275–284. [Google Scholar]
- Malhotra, D.; Mukherjee, J.; Gupta, M.N. Lipase catalyzed transesterification of castor oil by straight chain higher alcohols. J. Biosci. Bioeng. 2015, 119, 280–283. [Google Scholar] [CrossRef]
- Sharma, R.; Chisti, Y.; Banerjee, U.C. Production, purification, characterization, and applications of lipases. Biotechnol. Adv. 2001, 19, 627–662. [Google Scholar] [CrossRef] [Green Version]
- Bornscheuer, U.T.; Kazlauskas, R.J. Hydrolases in Organic Synthesis: Regio-and Stereoselective Biotransformations, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2006; 355p. [Google Scholar]
- Chandra, P.; Enespa Singh, R.; Arora, P.K. Microbial lipases and their industrial applications: A comprehensive review. Microb. Cell Fact. 2020, 19, 169. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dhar, K.; Kanwar, S.S.; Arora, P.K. Lipase catalysis in organic solvents: Advantages and applications. Biol. Proced. Online 2016, 18, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Derewenda, U.; Brzozowski, A.M.; Lawson, D.M.; Derewenda, Z.S. Catalysis at the interface: The anatomy of a conformational change in a triglyceride lipase. Biochemistry 1992, 31, 1532–1541. [Google Scholar] [CrossRef]
- Madan, B.; Mishra, P. Directed evolution of Bacillus licheniformis lipase for improvement of thermostability. Biochem. Eng. J. 2014, 91, 276–282. [Google Scholar] [CrossRef]
- Li, G.; Fang, X.; Su, F.; Chen, Y.; Xu, L.; Yan, Y. Enhancing the thermostability of Rhizomucor miehei lipase with a limited screening library by rational-design point mutations and disulfide bonds. Appl. Environ. Microbiol. 2018, 84, e02129-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foley, A.M.; Gavin, D.P.; Joniec, I.; Maguire, A.R. Impact of variation of the acyl group on the efficiency and selectivity of the lipase-mediated resolution of 2-phenylalkanols. Tetrahedron Asymmetry 2017, 28, 1144–1153. [Google Scholar] [CrossRef]
- Dwivedee, B.P.; Soni, S.; Laha, J.K.; Banerjee, U.C. Self assembly through sonication: An expeditious and green approach for the synthesis of organic-inorganic hybrid nanopetals and their application as biocatalyst. ChemNanoMat 2018, 4, 670–681. [Google Scholar] [CrossRef]
- Bansode, S.R.; Rathod, V.K. An intensified technique for lipase catalysed amide synthesis. Chem. Eng. Process. Process Intensif. 2019, 143, 107605. [Google Scholar] [CrossRef]
- Wang, X.P.; Zhou, P.F.; Li, Z.G.; Yang, B.; Hollmann, F.; Wang, Y.H. Engineering a lipase B from Candida antactica with efficient perhydrolysis performance by eliminating its hydrolase activity. Sci. Rep. 2017, 7, 44599. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Cen, Y.; Singh, W.; Fan, J.; Wu, L.; Lin, X.; Zhou, J.; Huang, M.; Reetz, M.T.; Wu, Q. Stereodivergent protein engineering of a lipase to access all possible stereoisomers of chiral esters with two stereocenters. J. Am. Chem. Soc. 2019, 141, 7934–7945. [Google Scholar] [CrossRef] [PubMed]
- Bavaro, T.; Pinto, A.; Dall’Oglio, F.; Hernáiz, M.J.; Morelli, C.F.; Zambelli, P.; Micheli, C.D.; Conti, P.; Tamborini, L.; Terreni, M. Flow-based biocatalysis: Application to peracetylated arabinofuranosyl-1,5-arabinofuranose synthesis. Process Biochem. 2018, 72, 112–118. [Google Scholar] [CrossRef]
- Engelmann, C.; Johannsen, J.; Waluga, T.; Fieg, G.; Liese, A.; Bubenheim, P. A Multi-enzyme cascade for the production of high-value aromatic compounds. Catalysts 2020, 10, 1216. [Google Scholar] [CrossRef]
- Fernandez-Lafuente, R.; Armisén, P.; Sabuquillo, P.; Fernández-Lorente, G.; Guisán, J.M. Immobilization of lipases by selective adsorption on hydrophobic supports. Chem. Phys. Lipids 1998, 93, 185–197. [Google Scholar] [CrossRef]
- Kurtovic, I.; Nalder, T.D.; Cleaver, H.; Marshall, S.N. Immobilisation of Candida rugosa lipase on a highly hydrophobic support: A stable immobilised lipase suitable for non-aqueous synthesis. Biotechnol. Rep. 2020, 28, e00535. [Google Scholar] [CrossRef] [PubMed]
- Shuai, W.; Das, R.K.; Naghdi, M.; Brar, S.K.; Verma, M. A review on the important aspects of lipase immobilization on nanomaterials. Biotechnol. Appl. Biochem. 2017, 64, 496–508. [Google Scholar] [CrossRef]
- Ortiz, C.; Ferreira, M.L.; Barbosa, O.; dos Santos, J.C.; Rodrigues, R.C.; Berenguer-Murcia, Á.; Briand, L.E.; Fernandez-Lafuente, R. Novozym 435: The “perfect” lipase immobilized biocatalyst? Catal. Sci. Technol. 2019, 9, 2380–2420. [Google Scholar] [CrossRef] [Green Version]
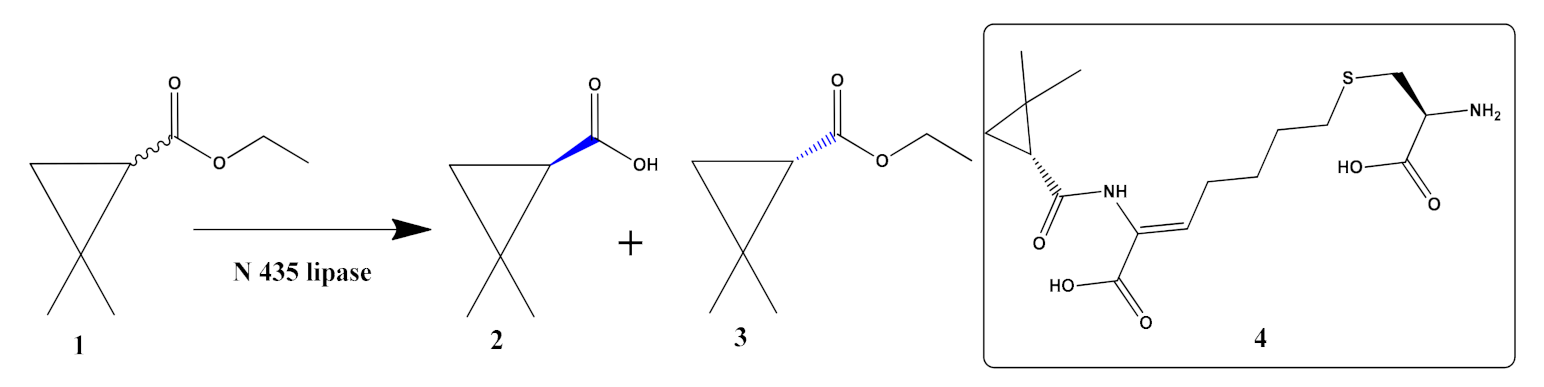
- Liang, F.; Huang, J.; He, J.; Wang, P. Improved enantioselective hydrolysis of racemic ethyl-2, 2-dimethylcyclopropanecarboxylate catalyzed by modified Novozyme 435. Biotechnol. Bioprocess Eng. 2012, 17, 952–958. [Google Scholar] [CrossRef]
- Sakulsombat, M.; Vongvilai, P.; Ramström, O. Efficient asymmetric synthesis of 1-cyano-tetrahydroisoquinolines from lipase dual activity and opposite enantioselectivities in α-Aminonitrile resolution. Chemistry 2014, 20, 11322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blank, N.; Opatz, T. Enantioselective synthesis of tetrahydroprotoberberines and bisbenzylisoquinoline alkaloids from a deprotonated α-aminonitrile. J. Org. Chem. 2011, 76, 9777–9784. [Google Scholar] [CrossRef] [PubMed]
- Sontakke, J.; Yadav, G. Microwave assisted synthesis of ethyl 2-(4-aminophenyl) acetate using Novozyme 435. CCAT 2014, 3, 27–34. [Google Scholar] [CrossRef]
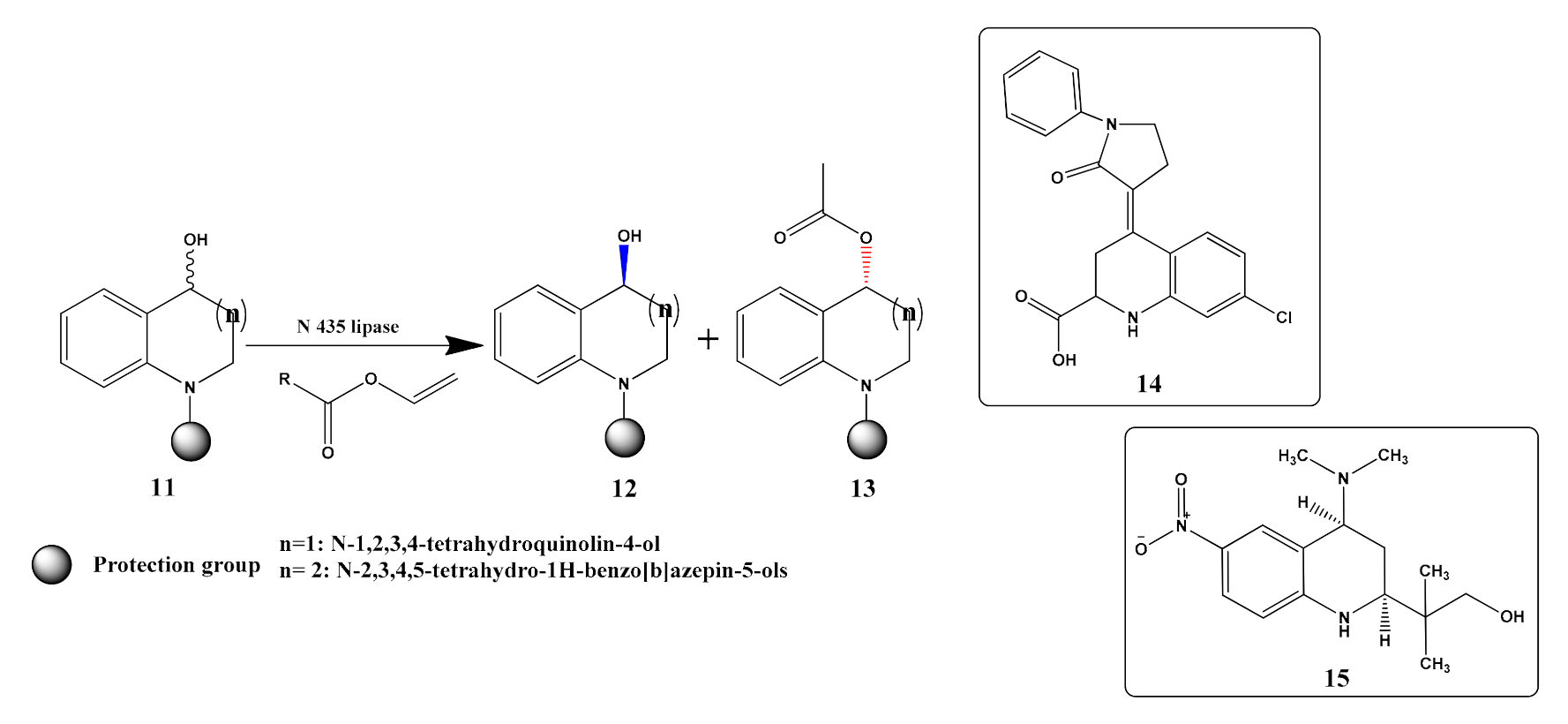
- Zhou, X.; Zheng, D.; Cui, B.; Han, W.; Chen, Y. Novozyme 435 lipase mediated enantioselective kinetic resolution: A facile method for the synthesis of chiral tetrahydroquinolin-4-ol and tetrahydro-1H-benzo[b]azepin-5-ol derivatives. Tetrahedron 2015, 71, 4738–4744. [Google Scholar] [CrossRef]
- Banks, A.; Breen, G.F.; Caine, D.; Carey, J.S.; Drake, C.; Forth, M.A.; Gladwin, A.; Guelfi, S.; Hayes, J.F.; Maragni, P.; et al. Process development and scale up of a glycine antagonist. Org. Process Res. Dev. 2009, 13, 1130–1140. [Google Scholar] [CrossRef]
- Nagata, N.; Miyakawa, M.; Amano, S.; Furuya, K.; Yamamoto, N.; Nejishima, H.; Inoguchi, K. Tetrahydroquinolines as a novel series of nonsteroidal selective androgen receptor modulators: Structural requirements for better physicochemical and biological properties. Bioorganic Med. Chem. Lett. 2011, 21, 6310–6313. [Google Scholar] [CrossRef]
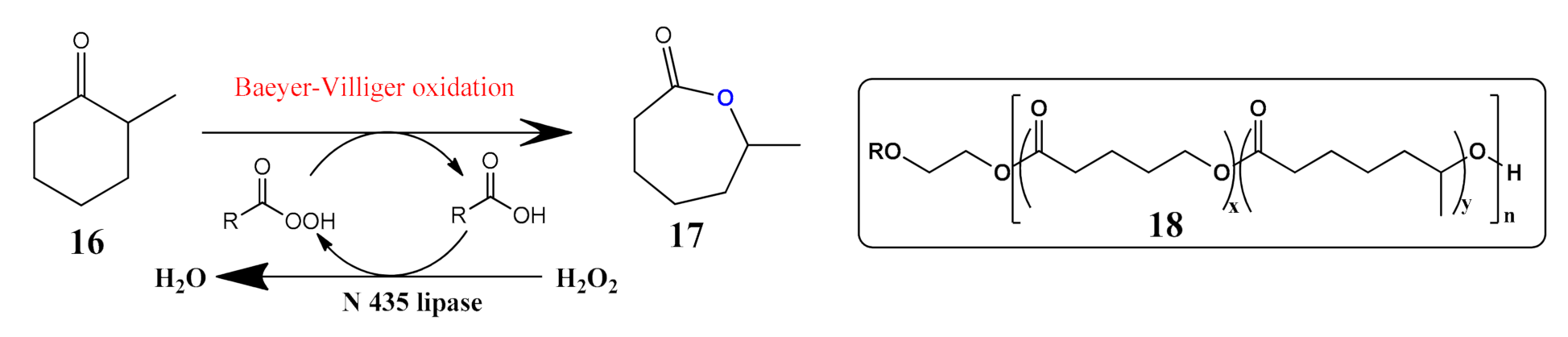
- Drożdż, A.; Erfurt, K.; Bielas, R.; Chrobok, A. Chemo-enzymatic Baeyer–Villiger oxidation in the presence of Candida antarctica lipase B and ionic liquids. New J. Chem. 2015, 39, 1315–1321. [Google Scholar] [CrossRef]
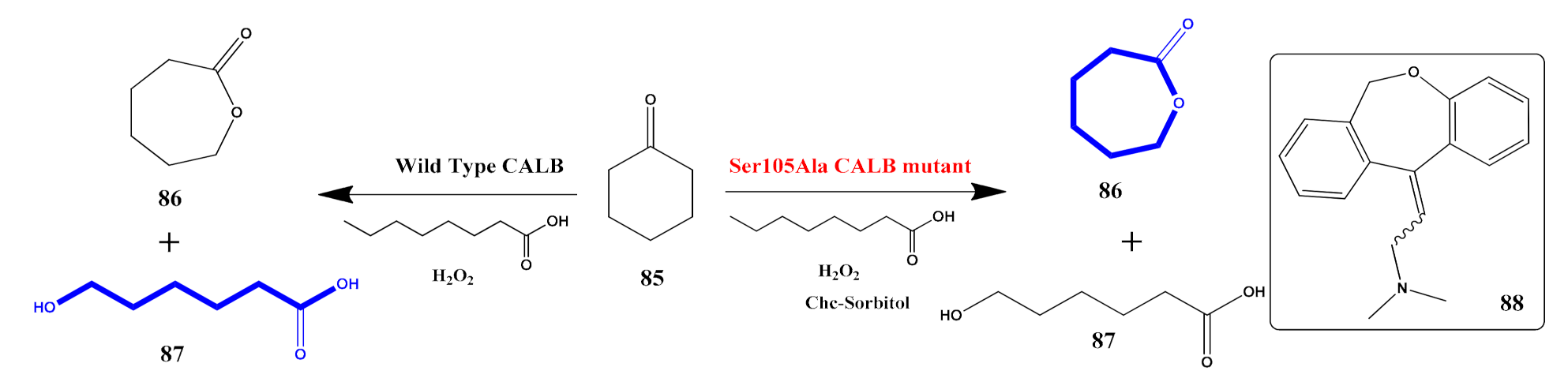
- Duale, K.; Latos, P.; Chrobok, A.; Domiński, A.; Maksymiak, M.M.; Adamus, G.; Kowalczuk, M. Towards Advances in Molecular Understanding of Boric Acid Biocatalyzed Ring-Opening (Co) Polymerization of δ-Valerolactone in the Presence of Ethylene Glycol as an Initiator. Molecules 2021, 26, 4859. [Google Scholar] [CrossRef]
- Jiang, C.; Cheng, G. Synergistic effect of Pd/C and Novozyme 435 on the dynamic kinetic resolution of 1, 1, 1-trifluoroisopropylamine. Chem. Eng. Commun. 2016, 203, 1222–1226. [Google Scholar] [CrossRef]
- Onyeagusi, C.I.; Malcolmson, S.J. Strategies for the Catalytic Enantioselective Synthesis of α-Trifluoromethyl Amines. ACS Catal. 2020, 10, 12507–12536. [Google Scholar] [CrossRef]
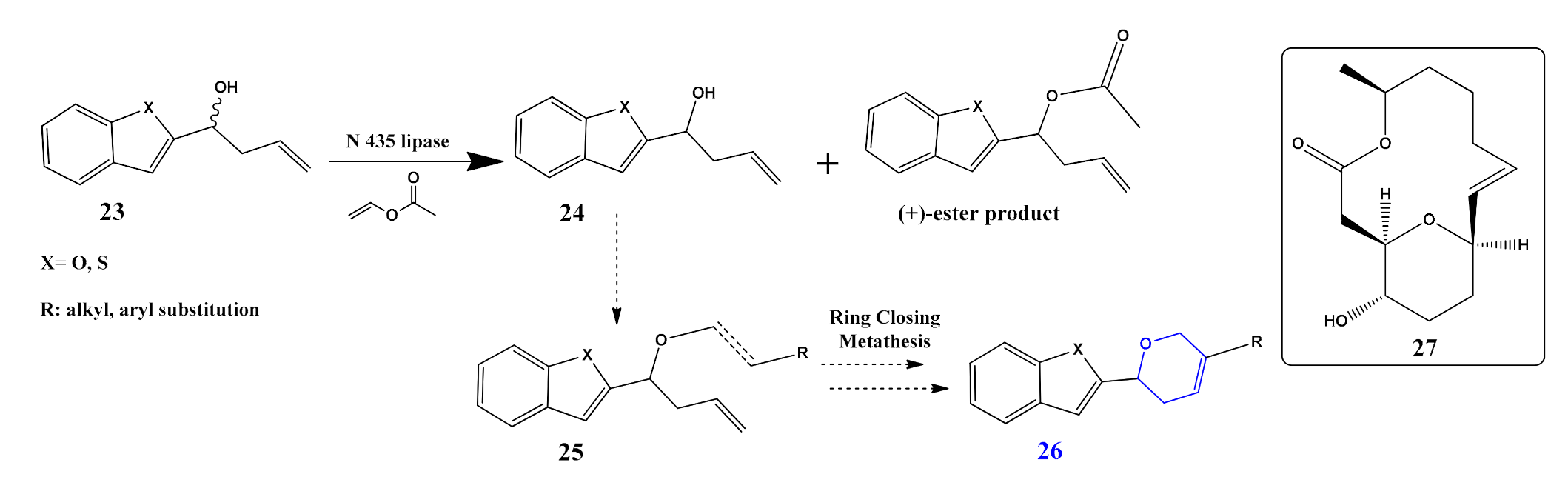
- Büyükadalı, N.N.; Aslan, N.; Gümüş, S.; Gümüş, A. Stereoselective synthesis of benzofuran and benzothiophene substituted dihydropyran derivatives via ring closing metathesis. Tetrahedron Asymmetry 2016, 27, 954–959. [Google Scholar] [CrossRef]
- Lambu, M.R.; Kumar, S.; Yousuf, S.K.; Sharma, D.K.; Hussain, A.; Kumar, A.; Malik, F.; Mukherjee, D. Medicinal chemistry of dihydropyran-based medium ring macrolides related to aspergillides: Selective inhibition of Pi3kα. J. Med. Chem. 2013, 56, 6122–6135. [Google Scholar] [CrossRef]
- Reddy, Y.N.; Kumari, T.N.; Thota, P.; Jyothi, P.; Gupta, A.K. Chemoenzymatic total synthesis of cryptocaryalactone natural products. Tetrahedron Lett. 2018, 59, 160–162. [Google Scholar] [CrossRef]
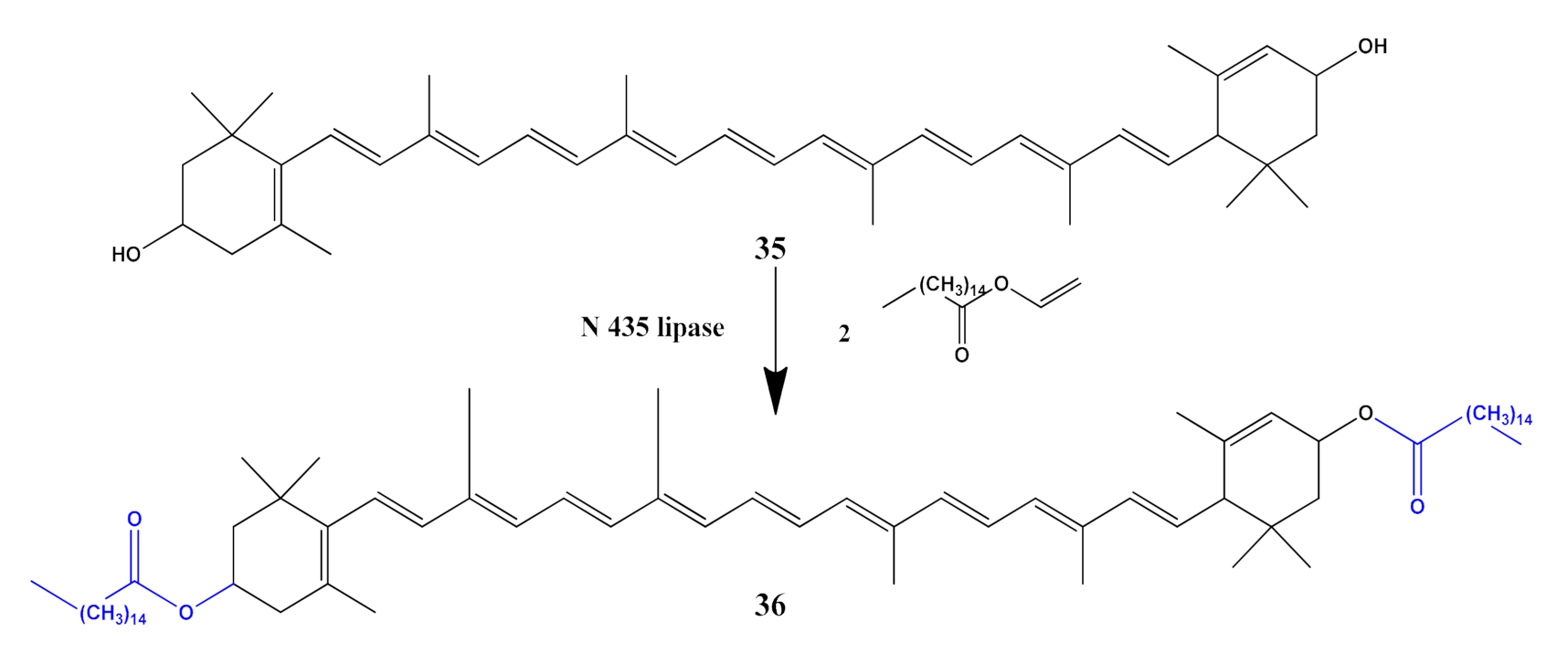
- Wang, R.; Hou, M.; Zhang, Y.; Ge, J.; Liu, Z. Enzymatic synthesis of lutein dipalmitate in organic solvents. Catal. Lett. 2015, 145, 995–999. [Google Scholar] [CrossRef]
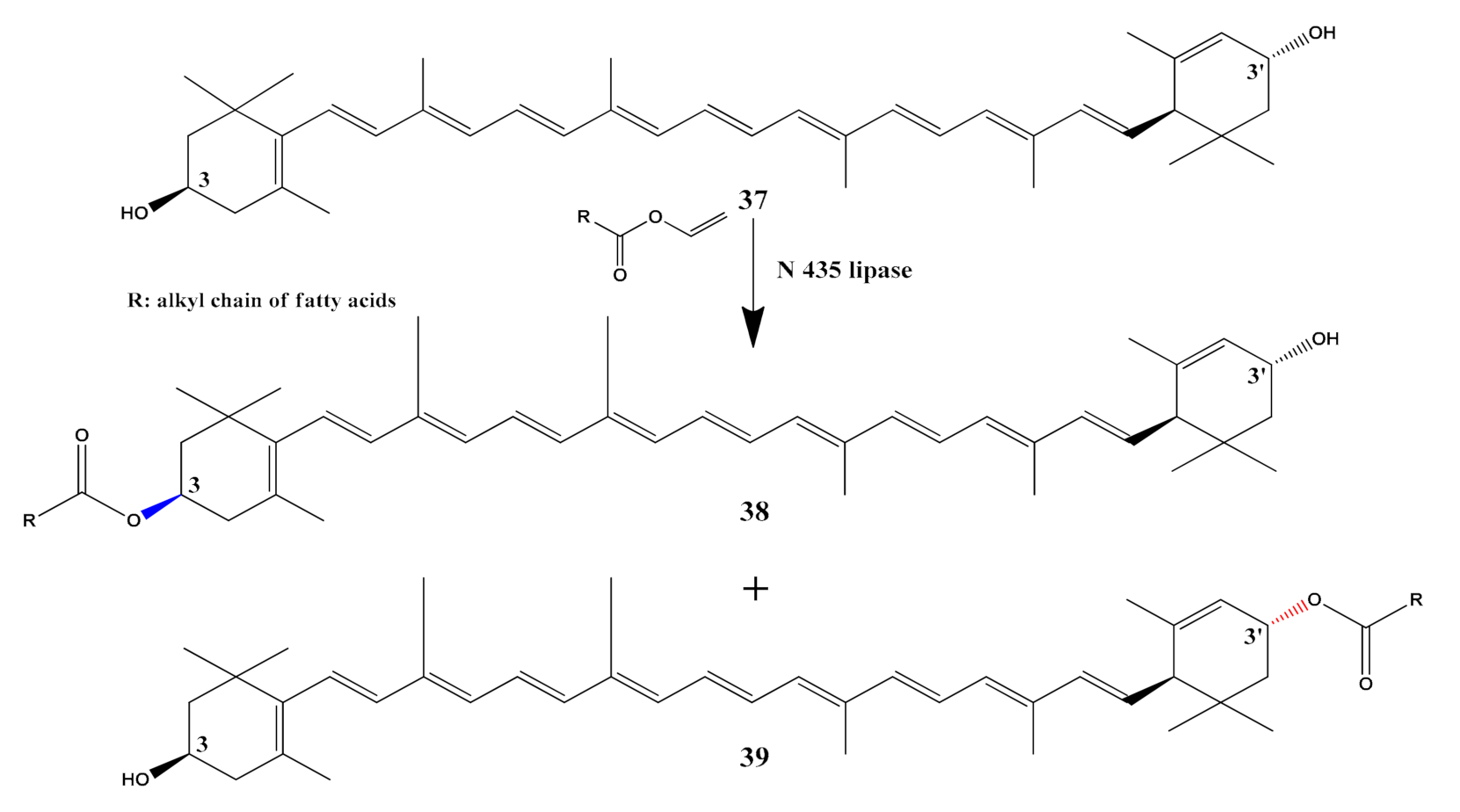
- Lujan-Montelongo, J.A.; Mendoza-Figueroa, H.L.; Silva-Cuevas, C.; Sánchez-Chávez, A.C.; Polindara-García, L.A.; Oliveros-Cruz, S.; Torres-Cardona, M.D. Highly regioselective enzymatic synthesis of lutein-3-monoesters. Tetrahedron Lett. 2018, 59, 4096–4101. [Google Scholar] [CrossRef]
- Dwivedee, B.P.; Soni, S.; Laha, J.K.; Banerjee, U.C. Facile immobilization of Pseudomonas fluorescens lipase on polyaniline nanofibers (PANFs-PFL): A route to develop robust nanobiocatalyst. Int. J. Biol. Macromol. 2018, 119, 8–14. [Google Scholar] [CrossRef]
- Viñambres, M.; Filice, M.; Marciello, M. Modulation of the catalytic properties of lipase B from Candida antarctica by immobilization on tailor-made magnetic iron oxide nanoparticles: The key role of nanocarrier surface engineering. Polymers 2018, 10, 615. [Google Scholar] [CrossRef] [Green Version]
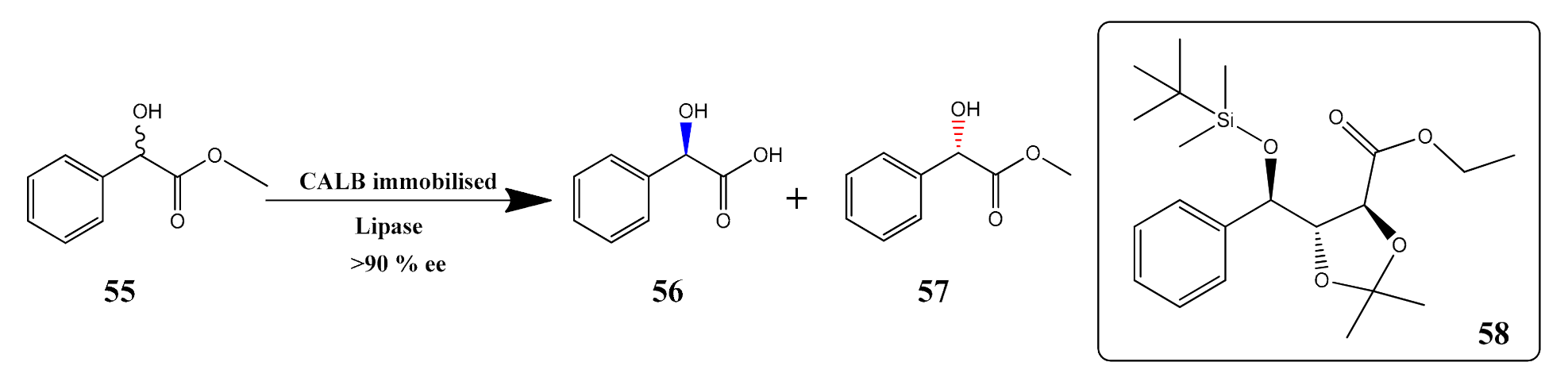
- Martínková, L.; Křen, V. Biocatalytic production of mandelic acid and analogues: A review and comparison with chemical processes. Appl. Microbiol. Biotechnol. 2018, 102, 3893–3900. [Google Scholar] [CrossRef]
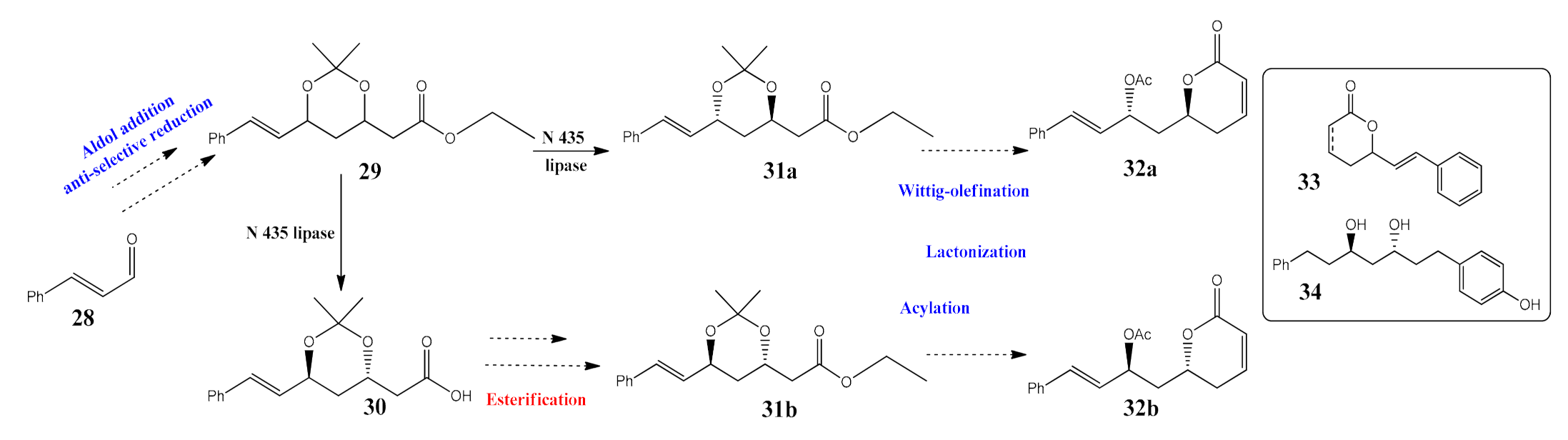
- Surivet, J.P.; Vatèle, J.M. Total synthesis of antitumor Goniothalamus styryllactones. Tetrahedron 1999, 55, 13011–13028. [Google Scholar] [CrossRef]
- Du, Y.; Gao, J.; Kong, W.; Zhou, L.; Ma, L.; He, Y.; Huang, Z.; Jiang, Y. Enzymatic synthesis of glycerol carbonate using a lipase immobilized on magnetic organosilica nanoflowers as a catalyst. ACS Omega 2018, 3, 6642–6650. [Google Scholar] [CrossRef] [Green Version]
- Ou, G.; He, B.; Yuan, Y. Lipases are soluble and active in glycerol carbonate as a novel biosolvent. Enzym. Microb. Technol. 2011, 49, 167–170. [Google Scholar] [CrossRef]
- Feng, Z.; Xu, B. Inspiration from the mirror: D-amino acid containing peptides in biomedical approaches. Biomol. Concepts 2016, 7, 179–187. [Google Scholar] [CrossRef] [PubMed]
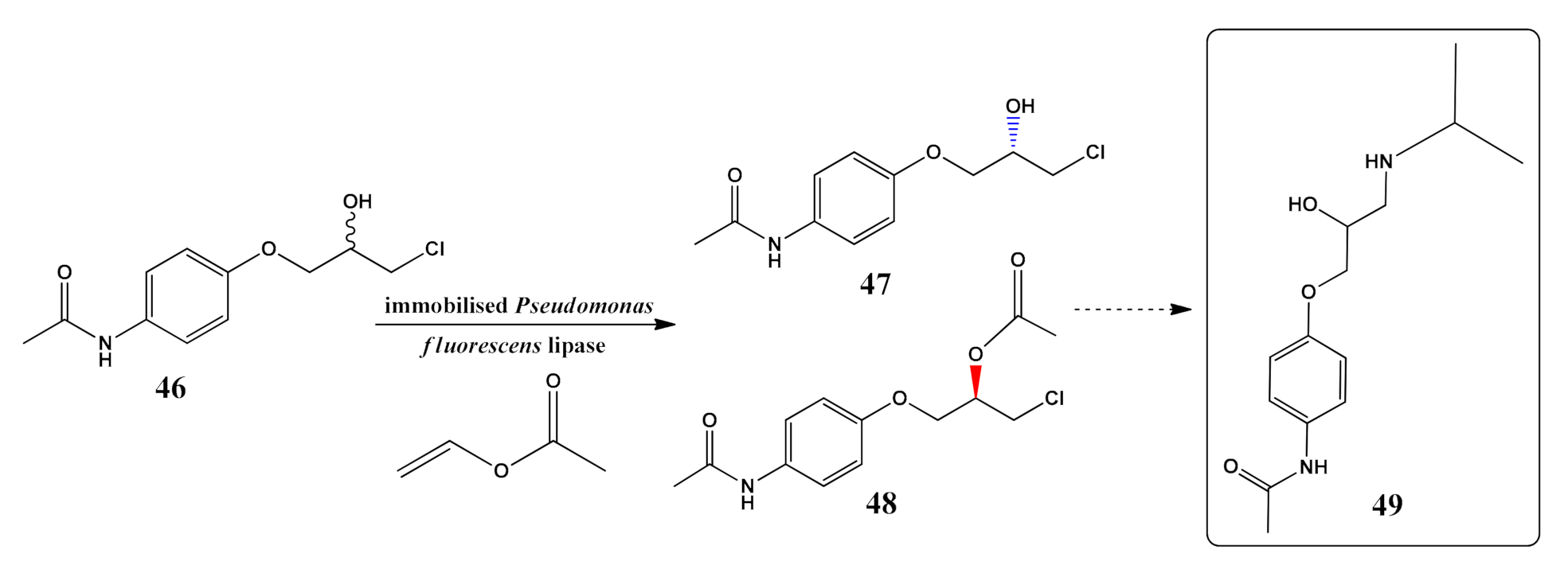
- Dwivedee, B.P.; Soni, S.; Bhimpuria, R.; Laha, J.K.; Banerjee, U.C. Tailoring a robust and recyclable nanobiocatalyst by immobilization of Pseudomonas fluorescens lipase on carbon nanofiber and its application in synthesis of enantiopure carboetomidate analogue. Int. J. Biol. Macromol. 2019, 133, 1299–1310. [Google Scholar] [CrossRef]
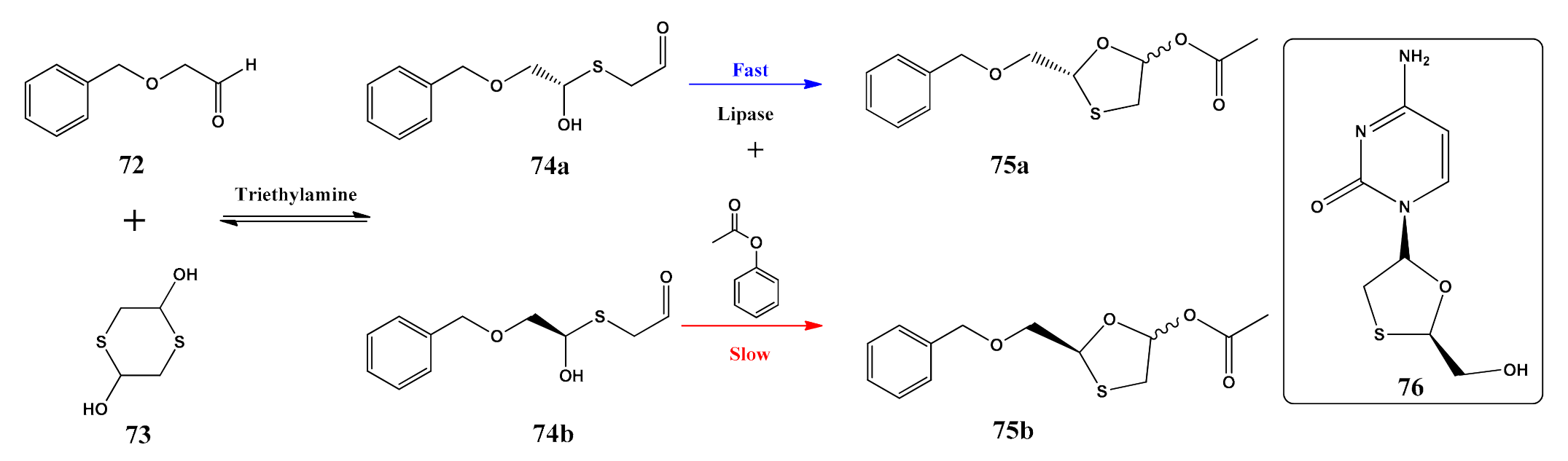
- Zhang, Y.; Sun, Y.; Tang, H.; Zhao, Q.; Ren, W.; Lv, K.; Yang, F.; Wang, F.; Liu, J. One-Pot Enzymatic Synthesis of Enantiopure 1,3-Oxathiolanes Using Trichosporon laibachii Lipase and the Kinetic Model. Org. Process Res. Dev. 2020, 24, 579–587. [Google Scholar] [CrossRef]
- Soni, S.; Dwivedee, B.P.; Banerjee, U.C. Tailoring a stable and recyclable nanobiocatalyst by immobilization of surfactant treated Burkholderia cepacia lipase on polyaniline nanofibers for biocatalytic application. Int. J. Biol. Macromol. 2020, 161, 573–586. [Google Scholar] [CrossRef] [PubMed]
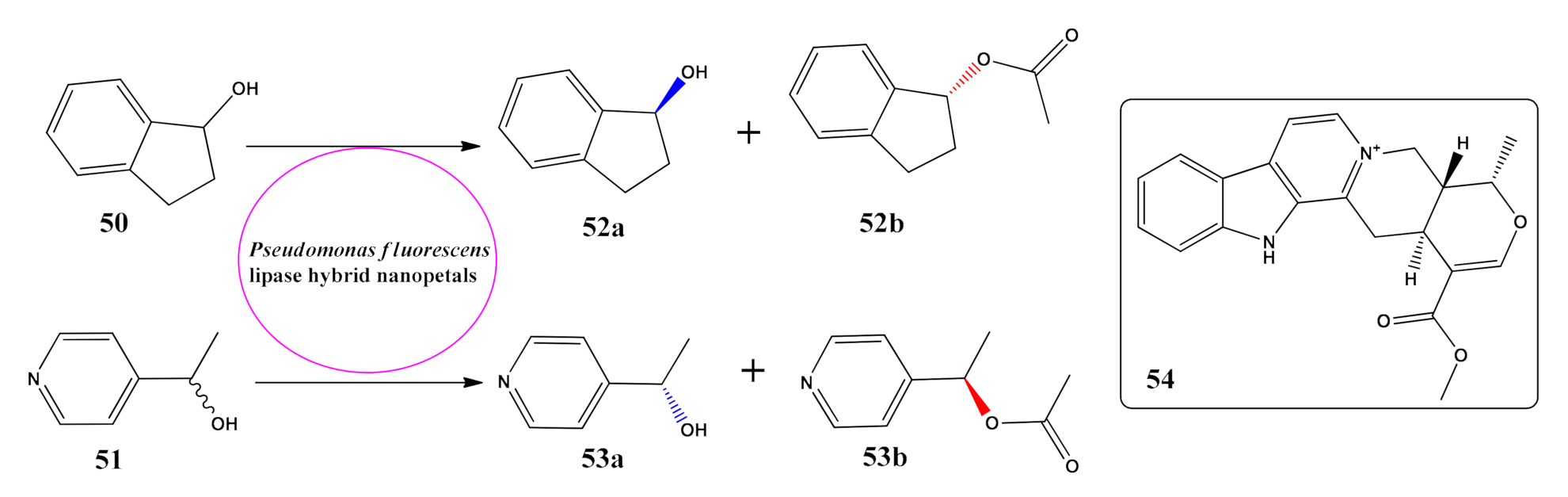
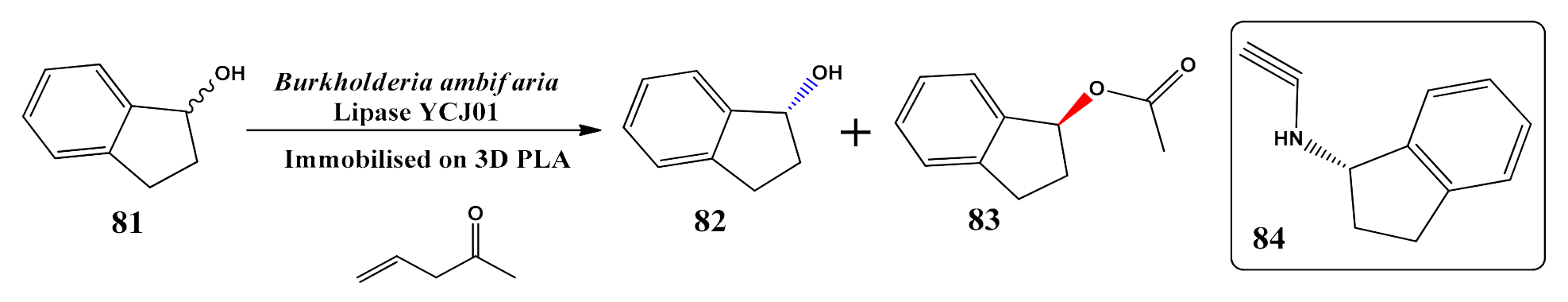
- Zhang, J.; Gao, B.; Lv, K.; Kumissay, L.; Wu, B.; Chu, J.; He, B. Specific immobilization of lipase on functionalized 3D printing scaffolds via enhanced hydrophobic interaction for efficient resolution of racemic 1-indanol. Biochem. Biophys. Res. Commun. 2021, 546, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Bansode, S.R.; Rathod, V.K. An investigation of lipase catalysed sonochemical synthesis: A review. Ultrason. Sonochem. 2017, 38, 503–529. [Google Scholar] [CrossRef]
- Woodley, J.M. New frontiers in biocatalysis for sustainable synthesis. Curr. Opin. Green Sustain. Chem. 2020, 21, 22–26. [Google Scholar] [CrossRef]
- Kazlauskas, R.J.; Bornscheuer, U.T. Finding better protein engineering strategies. Nat. Chem. Biol. 2009, 5, 526–529. [Google Scholar] [CrossRef]
- Bornscheuer, U.T. Alteration of lipase properties by protein engineering methods. Oléagineux Corps Gras Lipides 2008, 15, 184–188. [Google Scholar] [CrossRef] [Green Version]
- Otten, L.G.; Hollmann, F.; Arends, I.W.C.E. Enzyme engineering for enantioselectivity: From trial-and-error to rational design? Trends Biotechnol. 2010, 28, 46–54. [Google Scholar] [CrossRef]
- Reetz, M.T.; Carballeira, J.D. Iterative saturation mutagenesis (ISM) for rapid directed evolution of functional enzymes. Nat. Protoc. 2007, 2, 891–903. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, M.; Sudoh, T.; Kawamura, T.; Yamamoto, Y. A lactone-fused cyclohexadiene as a versatile platform for diversified synthesis of 5,6,5-tricyclic scaffolds. Org. Biomol. Chem. 2015, 13, 5862–5866. [Google Scholar] [CrossRef]
- Nortcliffe, A.; Moody, C.J. Seven-membered ring scaffolds for drug discovery: Access to functionalised azepanes and oxepanes through diazocarbonyl chemistry. Bioorganic Med. Chem. 2015, 23, 2730–2735. [Google Scholar] [CrossRef] [PubMed]
- Xia, B.; Xu, J.; Xiang, Z.; Cen, Y.; Hu, Y.; Lin, X.; Wu, Q. Stereoselectivity-Tailored, Metal-Free Hydrolytic Dynamic Kinetic Resolution of Morita–Baylis–Hillman Acetates Using an Engineered Lipase–Organic Base Cocatalyst. ACS Catal. 2017, 7, 4542–4549. [Google Scholar] [CrossRef]
- Zuckerman, D.S.; Woerpel, K.A. Synthesis of Enantiopure Triols from Racemic Baylis–Hillman Adducts Using a Diastereoselective Peroxidation Reaction. Org. Lett. 2020, 22, 9075–9080. [Google Scholar] [CrossRef] [PubMed]
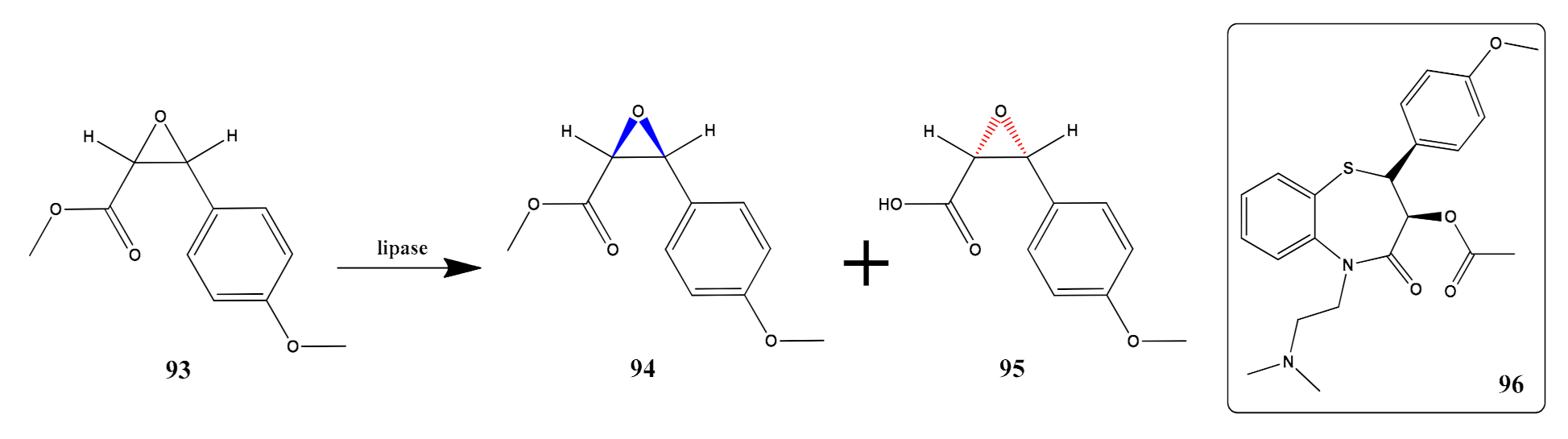
- Chen, K.-C.; Zheng, M.-M.; Pan, J.; Li, C.-X.; Xu, J.-H. Protein engineering and homologous expression of Serratia marcescens lipase for efficient synthesis of a pharmaceutically relevant chiral epoxyester. Appl. Biochem. Biotechnol. 2017, 183, 543–554. [Google Scholar] [CrossRef]
- Yang, B.; Wang, H.; Song, W.; Chen, X.; Liu, J.; Luo, Q.; Liu, L. Engineering of the conformational dynamics of lipase to increase enantioselectivity. ACS Catal. 2017, 7, 7593–7599. [Google Scholar] [CrossRef]
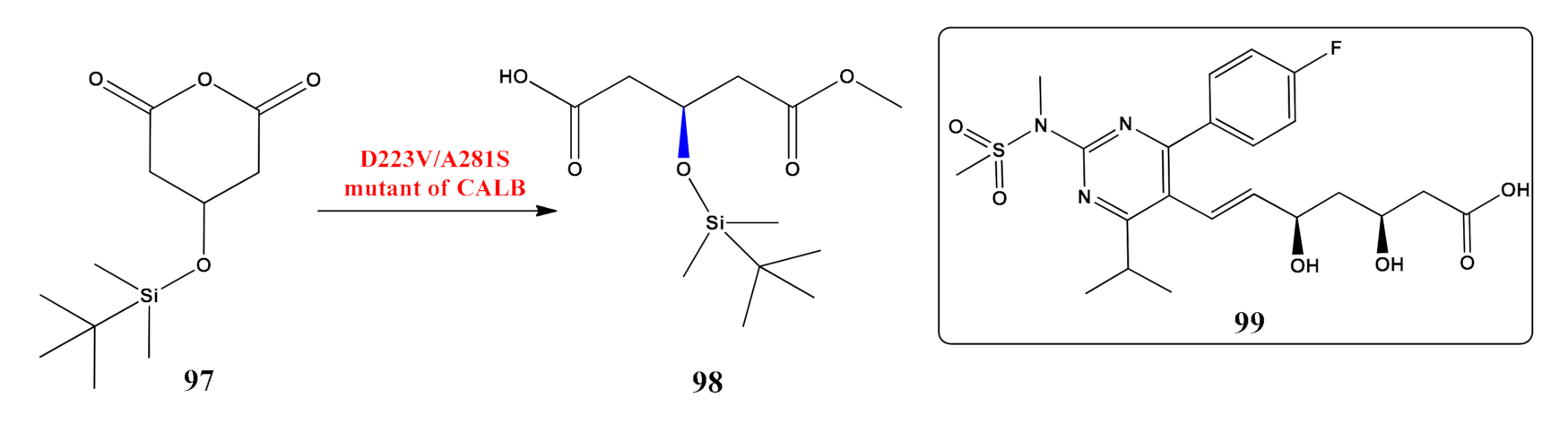
- Wang, H.; Li, Z.; Yu, X.; Chen, R.; Chen, X.; Liu, L. Green synthesis of (R)-3-TBDMSO glutaric acid methyl monoester using Novozym 435 in non-aqueous media. RSC Adv. 2015, 5, 75160–75166. [Google Scholar] [CrossRef]
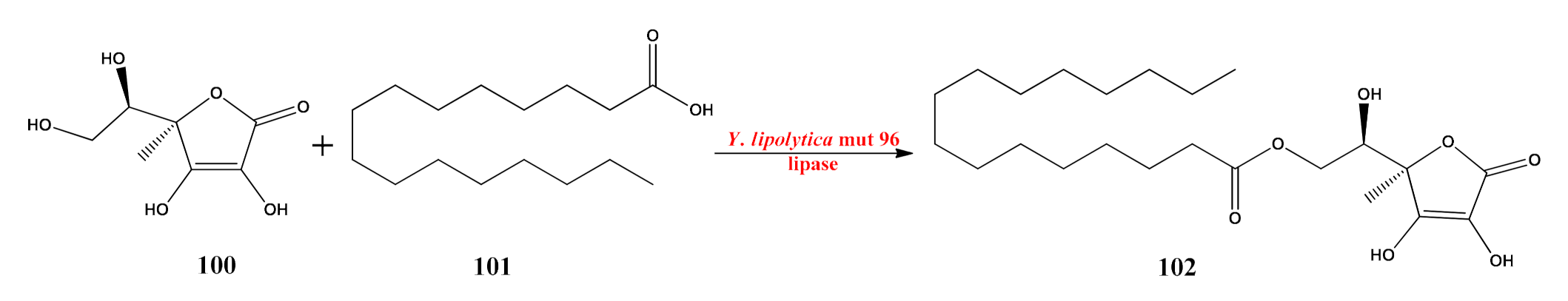
- Ping, L.; Yuan, X.; Zhang, M.; Chai, Y.; Shan, S. Improvement of extracellular lipase production by a newly isolated Yarrowia lipolytica mutant and its application in the biosynthesis of L-ascorbyl palmitate. Int. J. Biol. Macromol. 2018, 106, 302–311. [Google Scholar] [CrossRef] [PubMed]
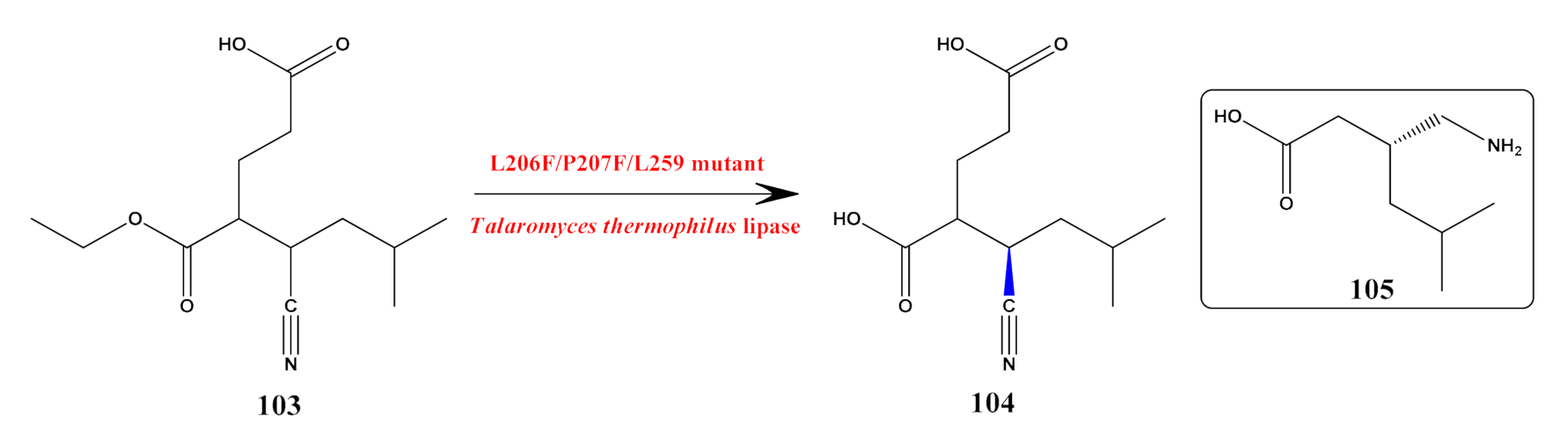
- Ding, X.; Zheng, R.-C.; Tang, X.-L.; Zheng, Y.-G. Engineering of Talaromyces thermophilus lipase by altering its crevice-like binding site for highly efficient biocatalytic synthesis of chiral intermediate of Pregablin. Bioorganic Chem. 2018, 77, 330–338. [Google Scholar] [CrossRef]
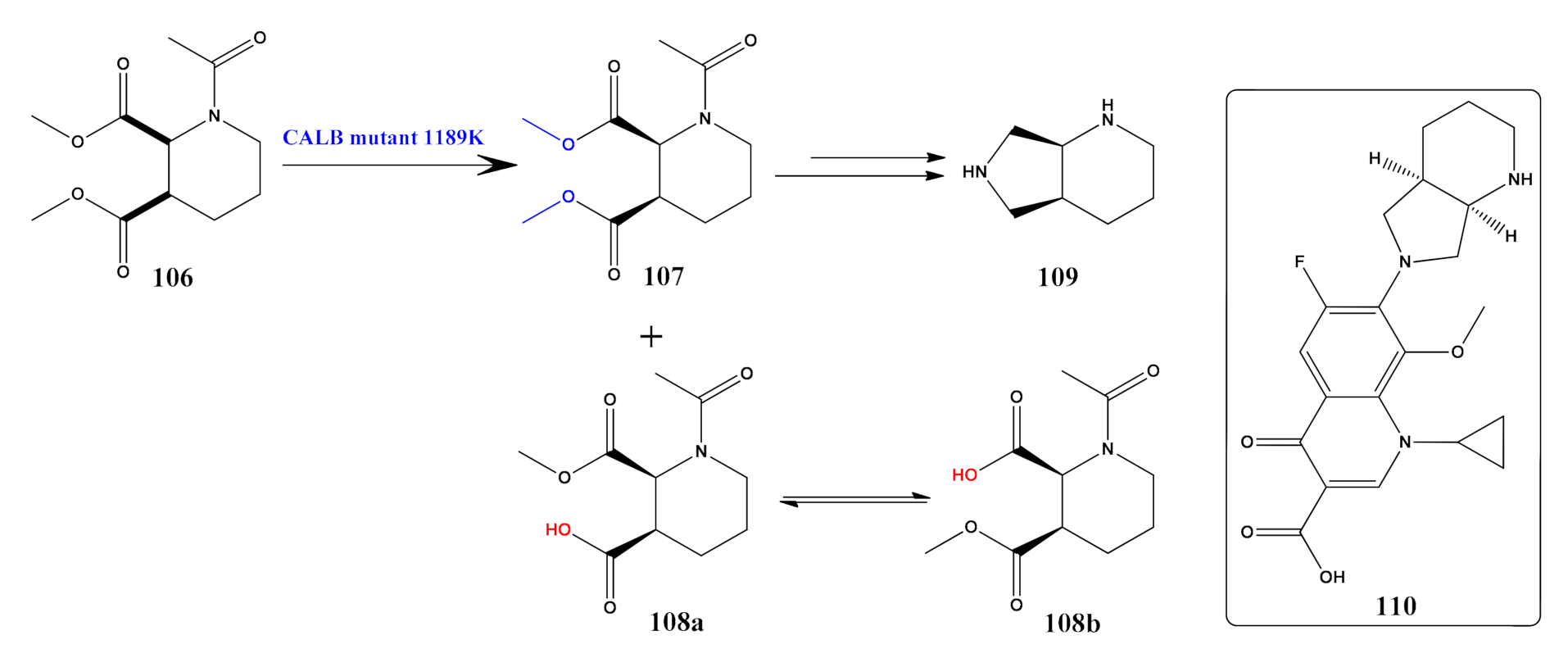
- Shen, J.W.; Qi, J.M.; Zhang, X.J.; Liu, Z.Q.; Zheng, Y.G. Significantly increased catalytic activity of Candida antarctica lipase B for the resolution of cis-(±)-dimethyl 1-acetylpiperidine-2, 3-dicarboxylate. Catal. Sci. Technol. 2018, 8, 4718–4725. [Google Scholar] [CrossRef]
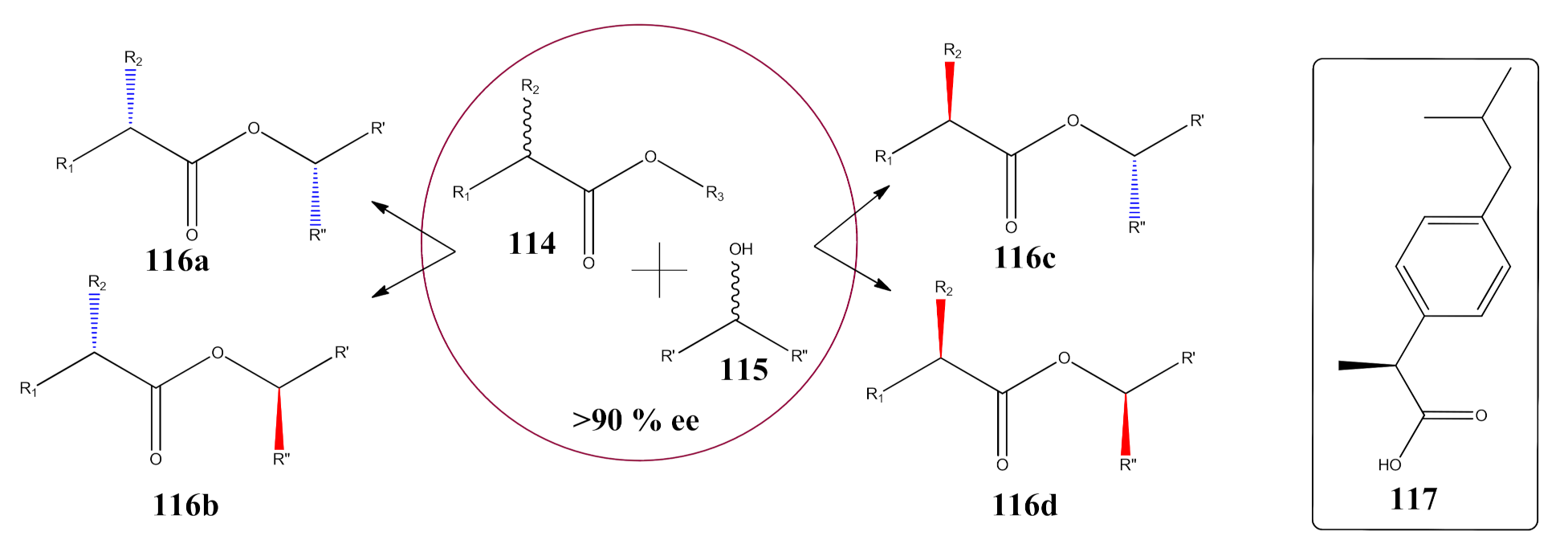
- Cen, Y.; Li, D.; Xu, J.; Wu, Q.; Wu, Q.; Lin, X. Highly focused library-based engineering of Candida antarctica lipase B with (S)-selectivity towards sec-alcohols. Adv. Synth. Catal. 2019, 361, 126–134. [Google Scholar] [CrossRef]
- Barbieri, C.; Caruso, E.; D’Arrigo, P.; Fantoni, G.P.; Servi, S. Chemo-enzymatic synthesis of (R)-and (S)-3, 4-dichlorophenylbutanolide intermediate in the synthesis of Sertraline. Tetrahedron Asymmetry 1999, 10, 3931–3937. [Google Scholar] [CrossRef]
- Li, D.Y.; Lou, Y.J.; Xu, J.; Chen, X.Y.; Lin, X.F.; Wu, Q. Electronic Effect-Guided Rational Design of Candida antarctica Lipase B for Kinetic Resolution Towards Diarylmethanols. Adv. Synth. Catal. 2021, 363, 1867–1872. [Google Scholar] [CrossRef]
- Bornscheuer, U.T. The fourth wave of biocatalysis is approaching. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2018, 376, 20170063. [Google Scholar] [CrossRef]
- Britton, J.; Majumdar, S.; Weiss, G.A. Continuous flow biocatalysis. Chem. Soc. Rev. 2018, 47, 5891–5918. [Google Scholar] [CrossRef] [PubMed]
- Ricca, E.; Brucher, B.; Schrittwieser, J.H. Multi-enzymatic cascade reactions: Overview and perspectives. Adv. Synth. Catal. 2011, 353, 2239–2262. [Google Scholar] [CrossRef]
- Scheibel, D.M.; Gitsov, I. Unprecedented enzymatic synthesis of perfectly structured alternating copolymers via ‘green’ reaction cocatalyzed by laccase and lipase compartmentalized within supramolecular complexes. Biomacromolecules 2019, 20, 927–936. [Google Scholar] [CrossRef]
- Ruwizhi, N.; Aderibigbe, B.A. Cinnamic acid derivatives and their biological efficacy. Int. J. Mol. Sci. 2020, 21, 5712. [Google Scholar] [CrossRef] [PubMed]
- Börgel, J.; Ritter, T. Late-stage functionalization. Chem 2020, 6, 1877–1887. [Google Scholar] [CrossRef]
- Cernak, T.; Dykstra, K.D.; Tyagarajan, S.; Vachal, P.; Krska, S.W. The Medicinal Chemist’s toolbox for late stage functionalization of drug-like molecules. Chem. Soc. Rev. 2016, 45, 546–576. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, G.A.; Oki, Y.; Akai, S. Lipase-Catalyzed Dynamic Kinetic Resolution of C1-and C2-Symmetric Racemic Axially Chiral 2, 2′-Dihydroxy-1, 1′-biaryls. Angew. Chem. Int. Ed. 2018, 57, 10278–10282. [Google Scholar] [CrossRef] [PubMed]











Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verma, S.; Choudhary, R.N.; Kanadje, A.P.; Banerjee, U.C. Diversifying Arena of Drug Synthesis: In the Realm of Lipase Mediated Waves of Biocatalysis. Catalysts 2021, 11, 1328. https://doi.org/10.3390/catal11111328
Verma S, Choudhary RN, Kanadje AP, Banerjee UC. Diversifying Arena of Drug Synthesis: In the Realm of Lipase Mediated Waves of Biocatalysis. Catalysts. 2021; 11(11):1328. https://doi.org/10.3390/catal11111328
Chicago/Turabian StyleVerma, Sahil, Rahul Narayanlal Choudhary, Akash Prakash Kanadje, and Uttam Chand Banerjee. 2021. "Diversifying Arena of Drug Synthesis: In the Realm of Lipase Mediated Waves of Biocatalysis" Catalysts 11, no. 11: 1328. https://doi.org/10.3390/catal11111328
APA StyleVerma, S., Choudhary, R. N., Kanadje, A. P., & Banerjee, U. C. (2021). Diversifying Arena of Drug Synthesis: In the Realm of Lipase Mediated Waves of Biocatalysis. Catalysts, 11(11), 1328. https://doi.org/10.3390/catal11111328