Recent Advances in the Catalytic Treatment of Volatile Organic Compounds: A Review Based on the Mixture Effect
Abstract
:1. Introduction
2. Effect of Mixture on VOCs Oxidation
2.1. VOC in Mixture
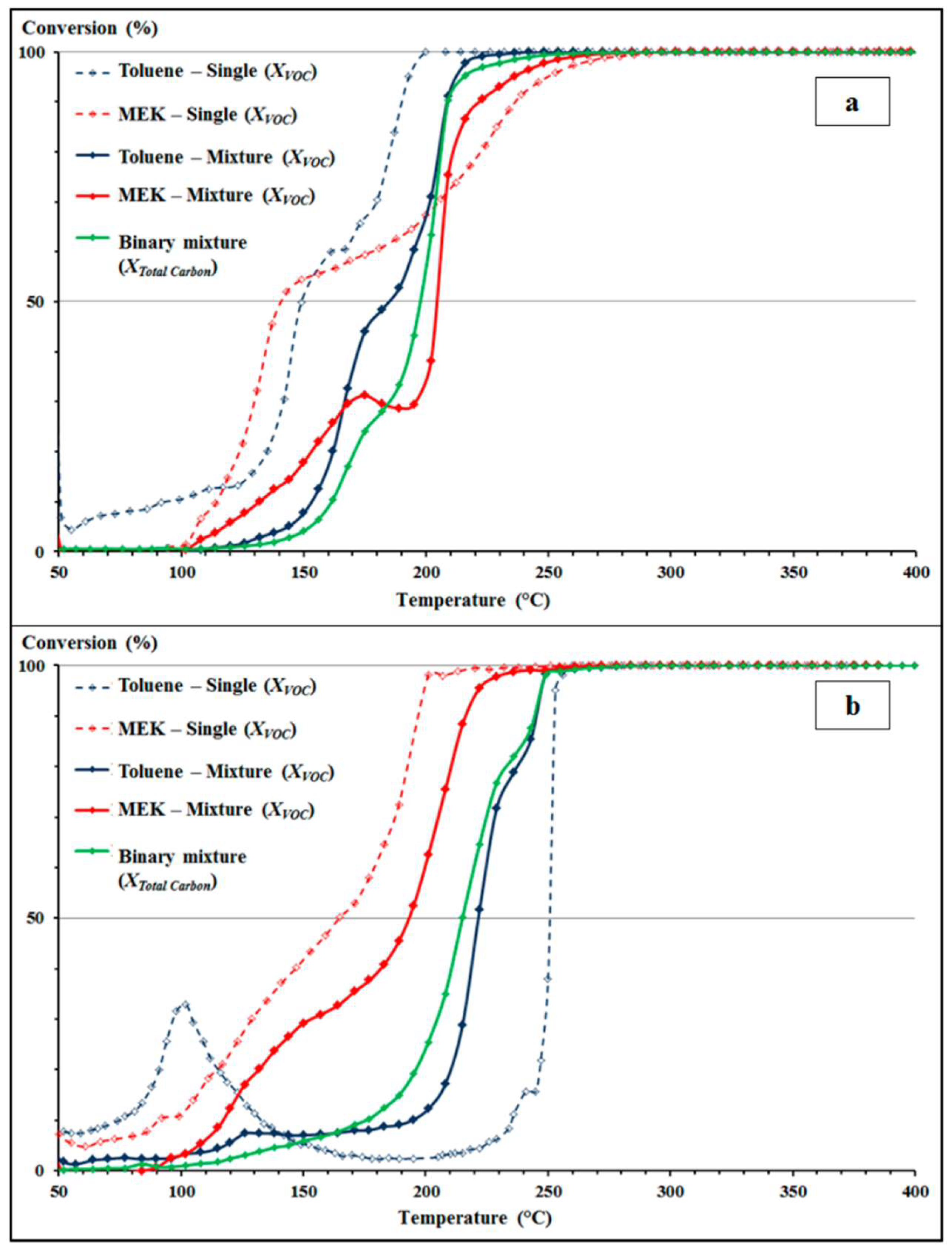
2.2. Toluene in Mixture
2.3. Benzene in Mixture
2.4. Oxi-Derivated in Mixture
2.4.1. With Oxi-Derivated VOCs
2.4.2. With Hydrocarbon VOCs
2.5. Hydrocarbons in Mixture
2.6. Conclusion
3. Effect of Moisture
3.1. Moisture Effect over HCHO Oxidation
3.1.1. Adsorption Competition between Water and HCHO
3.1.2. Beneficial Effect of the Water
- Storage of HCHO as CO form by oxidation at the surface of Pt;
- Oxidation step of CO possible by external O2 or Pt–O sites to form CO2.
- HCHO adsorption on birnessite favored by hydrogen bonds between HCHO and surface hydroxyl;
- Adsorbed HCHO oxidation into formate or carbonate by hydroxyl;
- Regeneration of surface hydroxyl via the reaction between active oxygen and water vapor (O2−, O− + H2O → 2 –OH);
- Carbonate and formate desorption into CO2 stimulated by the adsorption competition with water.
3.2. Moisture Effect over Toluene Oxidation
3.3. Conclusion
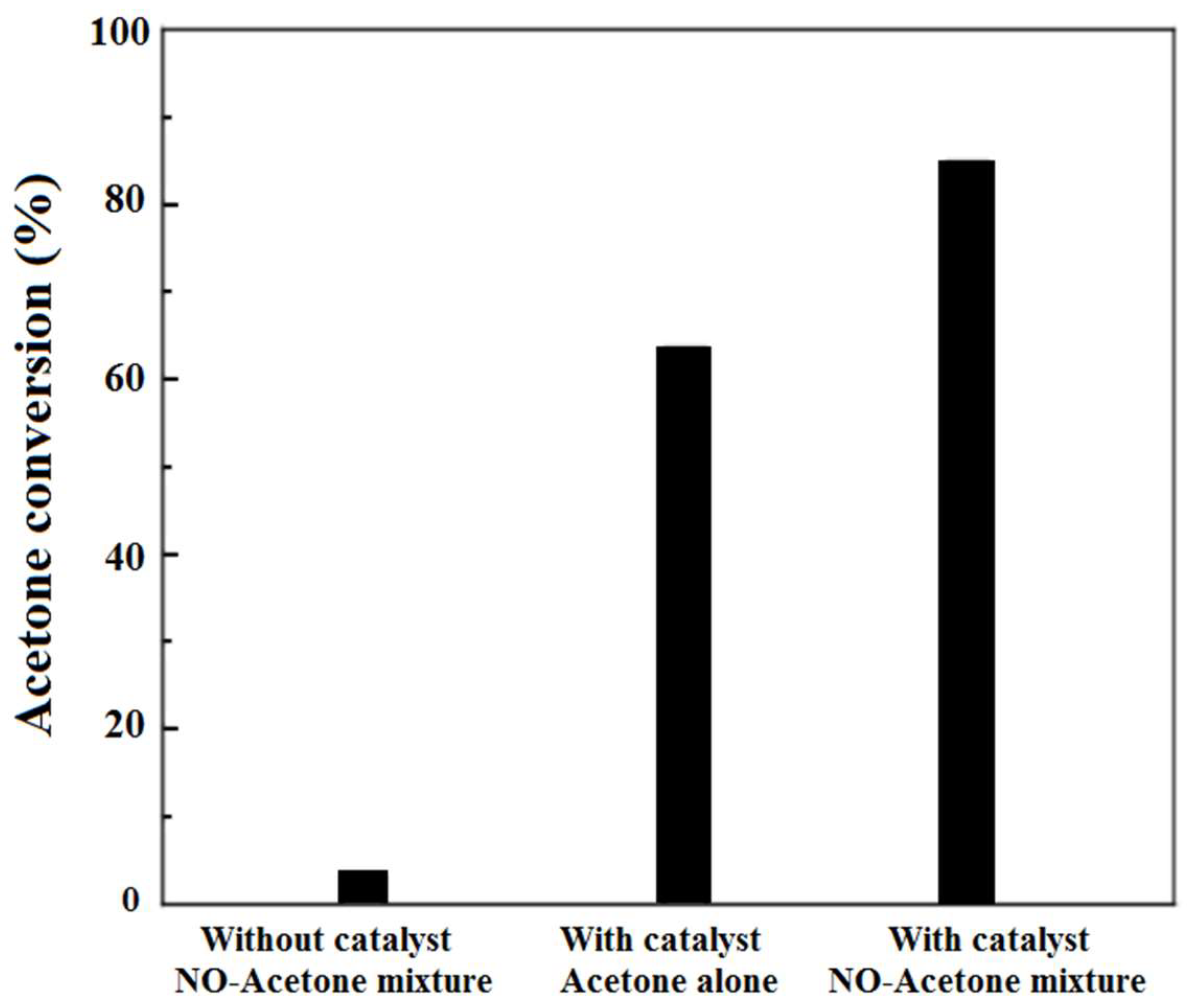
4. VOCs Oxidation in Presence of NOX
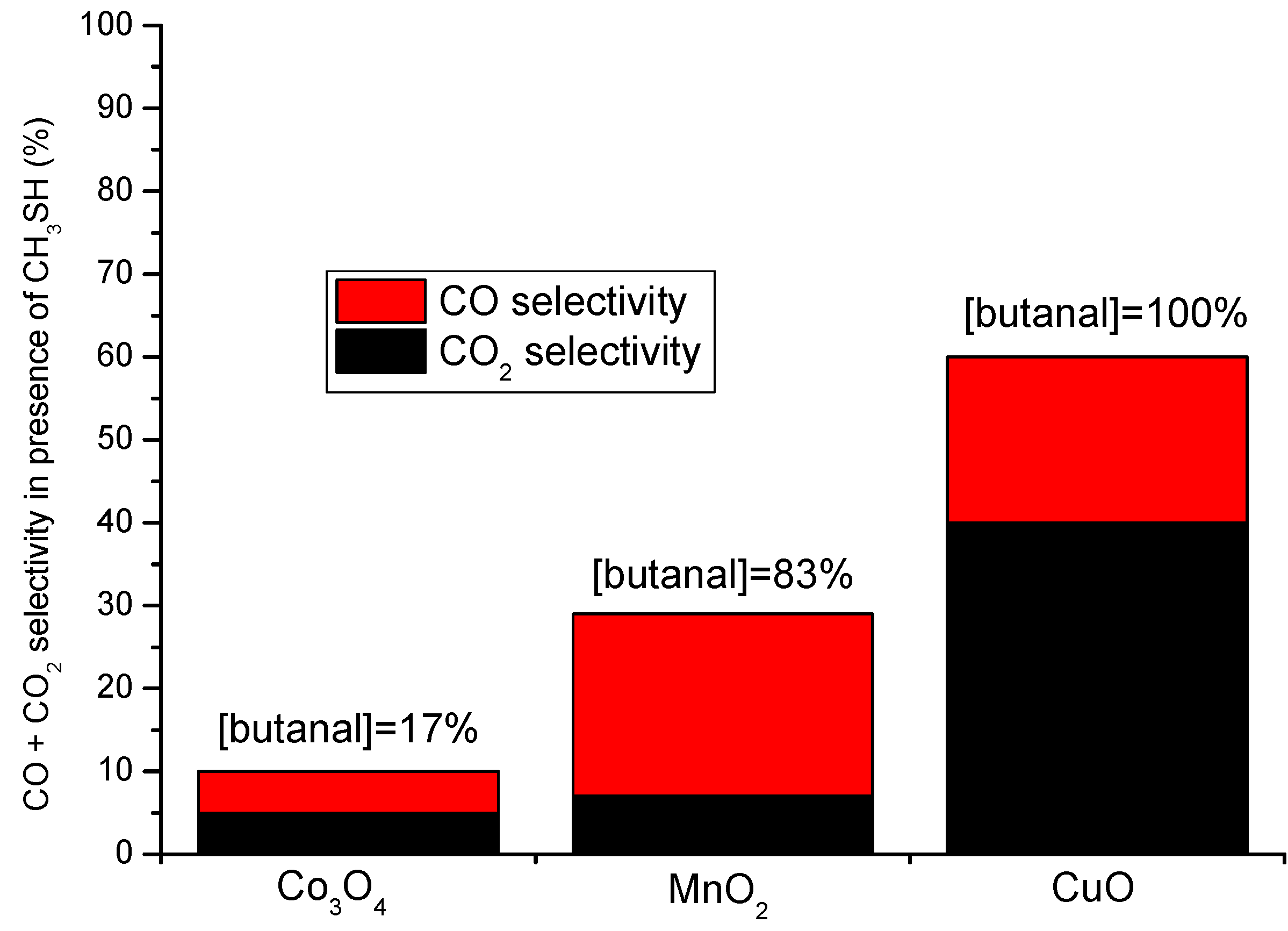
5. VOCs Oxidation in Presence of Sulfur
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Le Cloirec, P. Les Composés Organiques Volatiles dans l’Environnement; Technique et Documentation/Lavoisier; Lavoisier Bookseller: Paris, France, 1998; ISBN 2-7430-0232-8. [Google Scholar]
- World Health Organization. Indoor Air Quality: Organic Pollutants: Report on a World Health Organization Meeting, Berlin (West), 23–27 August 1987; World Health Organization, Regional Office for Europe: Copenhagen, Denmark, 1989. [Google Scholar]
- Directive, E.U. Directive 2004/42/EC of the European Parliament and of the council of 21 April 2004 on the limitation of emissions of volatile organic compounds due to the use of organic solvents in certain paints and varnishes and vehicle refinishing products and amending Directive 1999/13/EC4. Off. J. Eur. Union L 2014, 143, 87–96. [Google Scholar]
- Derwent, R.G.; Jenkin, M.E.; Saunders, S.M.; Pilling, M.J. Photochemical ozone creation potentials for organic compounds in northwest Europe calculated with a master chemical mechanism. Atmos. Environ. 1998, 32, 2429–2441. [Google Scholar] [CrossRef]
- O’Malley, A.; Hodnett, B.K. The influence of volatile organic compound structure on conditions required for total oxidation. Catal. Today 1999, 54, 31–38. [Google Scholar] [CrossRef]
- Huang, H.; Xu, Y.; Feng, Q.; Leung, D.Y.C. Low temperature catalytic oxidation of volatile organic compounds: A review. Catal. Sci. Technol. 2015, 5, 2649–2669. [Google Scholar] [CrossRef]
- Ivanova, S.; Pérez, A.; Centeno, M.Á.; Odriozola, J.A. Structured Catalysts for Volatile Organic Compound Removal. In New and Future Developments in Catalysis; Elsevier: Amsterdam, The Netherlands, 2013; pp. 233–256. ISBN 9780444538703. [Google Scholar]
- He, C.; Cheng, J.; Zhang, X.; Douthwaite, M.; Pattisson, S.; Hao, Z. Recent Advances in the Catalytic Oxidation of Volatile Organic Compounds: A Review Based on Pollutant Sorts and Sources. Chem. Rev. 2019, 119, 4471–4568. [Google Scholar] [CrossRef]
- Krishnamurthy, A.; Adebayo, B.; Gelles, T.; Rownaghi, A.; Rezaei, F. Abatement of gaseous volatile organic compounds: A process perspective. Catal. Today 2020, 350, 100–119. [Google Scholar] [CrossRef]
- Gelles, T.; Krishnamurthy, A.; Adebayo, B.; Rownaghi, A.; Rezaei, F. Abatement of gaseous volatile organic compounds: A material perspective. Catal. Today 2020, 350, 3–18. [Google Scholar] [CrossRef]
- Liotta, L.F. Catalytic oxidation of volatile organic compounds on supported noble metals. Appl. Catal. B Environ. 2010, 100, 403–412. [Google Scholar] [CrossRef]
- Guo, Y.; Wen, M.; Li, G.; An, T. Recent advances in VOC elimination by catalytic oxidation technology onto various nanoparticles catalysts: A critical review. Appl. Catal. B Environ. 2021, 281, 119447. [Google Scholar] [CrossRef]
- Brunet, J.; Genty, E.; Barroo, C.; Cazier, F.; Poupin, C.; Siffert, S.; Thomas, D.; De Weireld, G.; de Bocarmé, T.V.; Cousin, R. The CoAlCeO mixed oxide: An alternative to palladium-based catalysts for total oxidation of industrial VOCs. Catalysts 2018, 8, 64. [Google Scholar] [CrossRef] [Green Version]
- Genty, E.; Brunet, J.; Poupin, C.; Ojala, S.; Siffert, S.; Cousin, R. Influence of CO addition on the toluene total oxidation over Co based mixed oxide catalysts. Appl. Catal. B Environ. 2019, 247, 163–172. [Google Scholar] [CrossRef]
- Xia, Y.; Xia, L.; Liu, Y.; Yang, T.; Deng, J.; Dai, H. Concurrent catalytic removal of typical volatile organic compound mixtures over Au-Pd/α-MnO2 nanotubes. J. Environ. Sci. 2018, 64, 276–288. [Google Scholar] [CrossRef]
- Musialik-Piotrowska, A.; Syczewska, K. Combustion of volatile organic compounds in two-component mixtures over monolithic perovskite catalysts. Catal. Today 2000, 59, 269–278. [Google Scholar] [CrossRef]
- Santos, V.P.; Pereira, M.F.R.; Órfão, J.J.M.; Figueiredo, J.L. Mixture effects during the oxidation of toluene, ethyl acetate and ethanol over a cryptomelane catalyst. J. Hazard. Mater. 2011, 185, 1236–1240. [Google Scholar] [CrossRef]
- Blasin-Aubé, V.; Belkouch, J.; Monceaux, L. General study of catalytic oxidation of various VOCs over La0.8Sr0.2MnO3+x perovskite catalyst—influence of mixture. Appl. Catal. B Environ. 2003, 43, 175–186. [Google Scholar] [CrossRef]
- He, C.; Li, P.; Cheng, J.; Hao, Z.P.; Xu, Z.P. A comprehensive study of deep catalytic oxidation of benzene, toluene, ethyl acetate, and their mixtures over Pd/ZSM-5 catalyst: Mutual effects and kinetics. Water Air Soil Pollut. 2010, 209, 365–376. [Google Scholar] [CrossRef]
- Aguero, F.N.; Barbero, B.P.; Gambaro, L.; Cadús, L.E. Catalytic combustion of volatile organic compounds in binary mixtures over MnOx/Al2O3 catalyst. Appl. Catal. B Environ. 2009, 91, 108–112. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, P.; Zheng, K.; Wang, C.; Liu, Y.; Dai, H.; Wang, C.; Hsi, H.C.; Deng, J. Size effect, mutual inhibition and oxidation mechanism of the catalytic removal of a toluene and acetone mixture over TiO2 nanosheet-supported Pt nanocatalysts. Appl. Catal. B Environ. 2020, 274, 118963. [Google Scholar] [CrossRef]
- Ordóñez, S.; Bello, L.; Sastre, H.; Rosal, R.; Díez, F.V. Kinetics of the deep oxidation of benzene, toluene, n-hexane and their binary mixtures over a platinum on γ-alumina catalyst. Appl. Catal. B Environ. 2002, 38, 139–149. [Google Scholar] [CrossRef]
- Grbic, B.; Radic, N.; Terlecki-Baricevic, A. Kinetics of deep oxidation of n-hexane and toluene over Pt/Al2O3 catalysts: Oxidation of mixture. Appl. Catal. B Environ. 2004, 50, 161–166. [Google Scholar] [CrossRef]
- Genty, E.; Dib, H.; Brunet, J.; Poupin, C.; Siffert, S.; Cousin, R. Effect of Ce Addition on MgAl Mixed Oxides for the Total Oxidation of CO and Toluene. Top. Catal. 2019, 62, 397–402. [Google Scholar] [CrossRef]
- Piumetti, M.; Fino, D.; Russo, N. Mesoporous manganese oxides prepared by solution combustion synthesis as catalysts for the total oxidation of VOCs. Appl. Catal. B Environ. 2015, 163, 277–287. [Google Scholar] [CrossRef]
- Santos, V.P.; Carabineiro, S.A.C.; Tavares, P.B.; Pereira, M.F.R.; Órfão, J.J.M.; Figueiredo, J.L. Oxidation of CO, ethanol and toluene over TiO2 supported noble metal catalysts. Appl. Catal. B Environ. 2010, 99, 198–205. [Google Scholar] [CrossRef]
- Burgos, N.; Paulis, M.; Mirari Antxustegi, M.; Montes, M. Deep oxidation of VOC mixtures with platinum supported on Al2O3/Al monoliths. Appl. Catal. B Environ. 2002, 38, 251–258. [Google Scholar] [CrossRef]
- Kim, S.C.; Shim, W.G. Catalytic combustion of VOCs over a series of manganese oxide catalysts. Appl. Catal. B Environ. 2010, 98, 180–185. [Google Scholar] [CrossRef]
- Becker, L.; Förster, H. Oxidative decomposition of benzene and its methyl derivatives catalyzed by copper and palladium ion-exchanged Y-type zeolites. Appl. Catal. B Environ. 1998, 17, 43–49. [Google Scholar] [CrossRef]
- Mazzarino, I.; Barresi, A.A. Catalytic combustion of VOC mixtures in a monolithic reactor. Catal. Today 1993, 17, 335–347. [Google Scholar] [CrossRef]
- Colman Lerner, J.E.; Peluso, M.A.; Porta, A.; Thomas, H.J.; Sambeth, J.E. Catalytic removal of a mixture of volatile organic compounds present in indoor air at various work sites over Pt, MnOx and Pt/MnOx supported monoliths. React. Kinet. Mech. Catal. 2015, 114, 395–407. [Google Scholar] [CrossRef]
- Barakat, T.; Rooke, J.C.; Cousin, R.; Lamonier, J.-F.; Giraudon, J.-M.; Su, B.-L.; Siffert, S. Investigation of the elimination of VOC mixtures over a Pd-loaded V-doped TiO2 support. New J. Chem. 2014, 38, 2066–2074. [Google Scholar] [CrossRef]
- Papaefthimiou, P.; Ioannides, T.; Verykios, X.E. Performance of doped Pt/TiO2 (W6+) catalysts for combustion of volatile organic compounds (VOCS). Appl. Catal. B Environ. 1998, 15, 75–92. [Google Scholar] [CrossRef]
- Zuhairi Abdullah, A.; Bakar, M.Z.A.; Bhatia, S. A Kinetic Study of Catalytic Combustion of Ethyl Acetate and Benzene in Air Stream over Cr-ZSM-5 Catalyst. Ind. Eng. Chem. Res. 2003, 42, 6059–6067. [Google Scholar] [CrossRef]
- Gangwal, S.; Ramanathan, K.; Caffrey, P.; Mullins, M.; Spivey, J. Mixture Effects in the Catalytic Oxidation of VOCs in Air; EPA United States Environmental Protection Agency: Washington, DC, USA, 1998.
- Papaefthimiou, P.; Ioannides, T.; Verykios, X. Combustion of non-halogenated volatile organic compounds over group VIII metal catalysts. Appl. Catal. B Environ. 1997, 13, 175–184. [Google Scholar] [CrossRef]
- Gangwal, S.K.; Mullins, M.E.; Spivey, J.J.; Caffrey, P.R.; Tichenor, B.A. Kinetics and Selectivity of Deep Catalytic Oxidation of n-Hexane and Benzene. Appl. Catal. 1988, 36, 231–247. [Google Scholar] [CrossRef]
- Soares, O.S.G.P.; Órfão, J.J.M.; Figueiredo, J.L.; Pereira, M.F.R. Oxidation of mixtures of ethyl acetate and butyl acetate over cryptomelane and the effect of water vapor. Environ. Prog. Sustain. Energy 2016, 35, 1324–1329. [Google Scholar] [CrossRef]
- Tsou, J.; Magnoux, P.; Guisnet, M.; Órfão, J.J.M.; Figueiredo, J.L. Catalytic oxidation of volatile organic compounds: Oxidation of methyl-isobutyl-ketone over Pt/zeolite catalysts. Appl. Catal. B Environ. 2005, 57, 117–123. [Google Scholar] [CrossRef]
- Tabakova, T.; Kolentsova, E.; Dimitrov, D.; Ivanov, K.; Manzoli, M.; Venezia, A.M.; Karakirova, Y.; Petrova, P.; Nihtianova, D.; Avdeev, G. CO and VOCs Catalytic Oxidation Over Alumina Supported Cu–Mn Catalysts: Effect of Au or Ag Deposition. Top. Catal. 2017, 60, 110–122. [Google Scholar] [CrossRef]
- Cullis, C.F.; Keene, D.E.; Trimm, D.L. Studies of the partial oxidation of methane over heterogeneous catalysts. J. Catal. 1970, 19, 378–385. [Google Scholar] [CrossRef]
- Delimaris, D.; Ioannides, T. VOC oxidation over MnOx-CeO2 catalysts prepared by a combustion method. Appl. Catal. B Environ. 2008, 84, 303–312. [Google Scholar] [CrossRef]
- Yao, Y.F.Y. The oxidation of hydrocarbons and CO over metal oxides. II. α-Cr2O3. J. Catal. 1973, 28, 139–149. [Google Scholar] [CrossRef]
- Andersson, S.L.T. Reaction networks in the catalytic vapor-phase oxidation of toluene and xylenes. J. Catal. 1986, 98, 138–149. [Google Scholar] [CrossRef]
- Karuppiah, J.; Linga Reddy, E.; Manoj Kumar Reddy, P.; Ramaraju, B.; Karvembu, R.; Subrahmanyam, C. Abatement of mixture of volatile organic compounds (VOCs) in a catalytic non-thermal plasma reactor. J. Hazard. Mater. 2012, 237, 283–289. [Google Scholar] [CrossRef]
- Spivey, J.J.; Bryant, P.A. Hydroisomerization of n-C5 and n-C6 Mixtures on Zeolite Catalysts. Ind. Eng. Chem. Process Des. Dev. 1982, 21, 750–760. [Google Scholar] [CrossRef]
- Kikuchi, R.; Maeda, S.; Sasaki, K.; Wennerström, S.; Eguchi, K. Low-temperature methane oxidation over oxide-supported Pd catalysts: Inhibitory effect of water vapor. Appl. Catal. A Gen. 2002, 232, 23–28. [Google Scholar] [CrossRef]
- Pan, H.; Xu, M.; Li, Z.; Huang, S.; He, C. Catalytic combustion of styrene over copper based catalyst: Inhibitory effect of water vapor. Chemosphere 2009, 76, 721–726. [Google Scholar] [CrossRef]
- Lahousse, C.; Bernier, A.; Grange, P.; Delmon, B.; Papaefthimiou, P.; Ioannides, T.; Verykios, X. Evaluation of γ-MnO2 as a VOC removal catalyst: Comparison with a noble metal catalyst. J. Catal. 1998, 178, 214–225. [Google Scholar] [CrossRef]
- Wang, Z.; Pei, J.; Zhang, J. Catalytic oxidization of indoor formaldehyde at room temperature—Effect of operation conditions. Build. Environ. 2013, 65, 49–57. [Google Scholar] [CrossRef]
- Sidheswaran, M.A.; Destaillats, H.; Sullivan, D.P.; Larsen, J.; Fisk, W.J. Quantitative room-temperature mineralization of airborne formaldehyde using manganese oxide catalysts. Appl. Catal. B Environ. 2011, 107, 34–41. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Li, Y.; Huang, X.; Xu, Y.; Zhu, H.; Wang, J.; Shen, W. MnOx-CeO2 mixed oxide catalysts for complete oxidation of formaldehyde: Effect of preparation method and calcination temperature. Appl. Catal. B Environ. 2006, 62, 265–273. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, P.; Li, J.; Jiang, C.; Yunus, R.; Kim, J. Room-Temperature Oxidation of Formaldehyde by Layered Manganese Oxide: Effect of Water. Environ. Sci. Technol. 2015, 49, 12372–12379. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, F.; Zhai, Y.; Ariga, H.; Yi, N.; Liu, Y.; Asakura, K.; Flytzani-Stephanopoulos, M.; He, H. Alkali-metal-promoted Pt/TiO2 opens a more efficient pathway to formaldehyde oxidation at ambient temperatures. Angew. Chem. Int. Ed. 2012, 51, 9628–9632. [Google Scholar] [CrossRef]
- Huang, H.; Leung, D.Y.C. Complete elimination of indoor formaldehyde over supported Pt catalysts with extremely low Pt content at ambient temperature. J. Catal. 2011, 280, 60–67. [Google Scholar] [CrossRef]
- Huang, H.; Ye, X.; Huang, H.; Zhang, L.; Leung, D.Y.C. Mechanistic study on formaldehyde removal over Pd/TiO2 catalysts: Oxygen transfer and role of water vapor. Chem. Eng. J. 2013, 230, 73–79. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, S.; Crocker, M.; Zhu, X.; Chen, B.; Xie, J.; Shi, C.; Ma, D. An energy-efficient catalytic process for the tandem removal of formaldehyde and benzene by metal/HZSM-5 catalysts. Catal. Sci. Technol. 2015, 5, 4968–4972. [Google Scholar] [CrossRef]
- An, N.; Yu, Q.; Liu, G.; Li, S.; Jia, M.; Zhang, W. Complete oxidation of formaldehyde at ambient temperature over supported Pt/Fe2O3 catalysts prepared by colloid-deposition method. J. Hazard. Mater. 2011, 186, 1392–1397. [Google Scholar] [CrossRef]
- Chen, B.B.; Zhu, X.B.; Crocker, M.; Wang, Y.; Shi, C. FeOx-supported gold catalysts for catalytic removal of formaldehyde at room temperature. Appl. Catal. B Environ. 2014, 154–155, 73–81. [Google Scholar] [CrossRef]
- Wang, J.; Yunus, R.; Li, J.; Li, P.; Zhang, P.; Kim, J. In situ synthesis of manganese oxides on polyester fiber for formaldehyde decomposition at room temperature. Appl. Surf. Sci. 2015, 357, 787–794. [Google Scholar] [CrossRef]
- Wang, J.; Li, J.; Jiang, C.; Zhou, P.; Zhang, P.; Yu, J. The effect of manganese vacancy in birnessite-type MnO2 on room-temperature oxidation of formaldehyde in air. Appl. Catal. B Environ. 2017, 204, 147–155. [Google Scholar] [CrossRef]
- Wang, J.; Li, D.; Li, P.; Zhang, P.; Xu, Q.; Yu, J. Layered manganese oxides for formaldehyde-oxidation at room temperature: The effect of interlayer cations. RSC Adv. 2015, 5, 100434–100442. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, X.; Crocker, M.; Chen, B.; Shi, C. A comparative study of the catalytic oxidation of HCHO and CO over Mn0.75Co2.25O4 catalyst: The effect of moisture. Appl. Catal. B Environ. 2014, 160–161, 542–551. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, G.; Zhang, P. Layered birnessite-type MnO2 with surface pits for enhanced catalytic formaldehyde oxidation activity. J. Mater. Chem. A 2017, 5, 5719–5725. [Google Scholar] [CrossRef]
- Rochard, G.; Ciotonea, C.; Ungureanu, A.; Giraudon, J.-M.; Royer, S.; Lamonier, J.-F. MnOx loaded mesoporous silica for the catalytic oxidation of formaldehyde. Effect of the melt infiltration conditions on the activity—stability behavior. ChemCatChem 2019, 12, 1664–1675. [Google Scholar] [CrossRef]
- Chen, H.; Rui, Z.; Ji, H. Monolith-Like TiO2 Nanotube Array Supported Pt Catalyst for HCHO Removal under Mild Conditions. Ind. Eng. Chem. Res. 2014, 53, 7629–7636. [Google Scholar] [CrossRef]
- Heidinger, B.; Royer, S.; Giraudon, J.-M.; Gardoll, O.; Alamdari, H.; Lamonier, J.-F. Reactive Grinding synthesis of La(Sr,Ce)CoO3 and their properties in toluene catalytic total oxidation. ChemCatChem 2020, 12, 2271–2282. [Google Scholar] [CrossRef]
- Hu, J.; Li, W.B.; Liu, R.F. Highly efficient copper-doped manganese oxide nanorod catalysts derived from CuMnO hierarchical nanowire for catalytic combustion of VOCs. Catal. Today 2018, 314, 147–153. [Google Scholar] [CrossRef]
- Xie, S.; Deng, J.; Liu, Y.; Zhang, Z.; Yang, H.; Jiang, Y.; Arandiyan, H.; Dai, H.; Au, C.T. Excellent catalytic performance, thermal stability, and water resistance of 3DOM Mn2O3-supported Au-Pd alloy nanoparticles for the complete oxidation of toluene. Appl. Catal. A Gen. 2015, 507, 82–90. [Google Scholar] [CrossRef]
- Li, X.; Wang, L.; Xia, Q.; Liu, Z.; Li, Z. Catalytic oxidation of toluene over copper and manganese based catalysts: Effect of water vapor. Catal. Commun. 2011, 14, 15–19. [Google Scholar] [CrossRef]
- Heinen, A.W.; Peters, J.A.; Bekkum, H. Van Competitive adsorption of water and toluene on modified activated carbon supports. Appl. Catal. A Gen. 2000, 194, 193–202. [Google Scholar] [CrossRef]
- Zhu, J.; Andersson, S.L.T. Effect of water on the catalytic oxidation of toluene over vanadium oxide catalysts. Appl. Catal. 1989, 53, 251–262. [Google Scholar] [CrossRef]
- Chen, L.; Liao, Y.; Xin, S.; Song, X.; Liu, G.; Ma, X. Simultaneous removal of NO and volatile organic compounds (VOCs) by Ce/Mo doping-modified selective catalytic reduction (SCR) catalysts in denitrification zone of coal-fired flue gas. Fuel 2020, 262, 116485. [Google Scholar] [CrossRef]
- Li, G.; Shen, K.; Wang, L.; Zhang, Y.; Yang, H.; Wu, P.; Wang, B.; Zhang, S. Synergistic degradation mechanism of chlorobenzene and NO over the multi-active center catalyst: The role of NO2, Brønsted acidic site, oxygen vacancy. Appl. Catal. B Environ. 2021, 286, 119865. [Google Scholar] [CrossRef]
- Xiao, C.X.; Yan, N.; Zou, M.; Hou, S.C.; Kou, Y.; Liu, W.; Zhang, S. NO2-catalyzed deep oxidation of methanol: Experimental and theoretical studies. J. Mol. Catal. A Chem. 2006, 252, 202–211. [Google Scholar] [CrossRef]
- Mrad, R.; Aissat, A.; Cousin, R.; Courcot, D.; Siffert, S. Catalysts for NOx selective catalytic reduction by hydrocarbons (HC-SCR). Appl. Catal. A Gen. 2015, 504, 542–548. [Google Scholar] [CrossRef]
- Motak, M.; Kuterasiński, Ł.; Da Costa, P.; Samojeden, B. Catalytic activity of layered aluminosilicates for VOC oxidation in the presence of NOx. Comptes Rendus Chim. 2015, 18, 1106–1113. [Google Scholar] [CrossRef]
- Karthik, M.; Lin, L.Y.; Bai, H. Bifunctional mesoporous Cu-Al-MCM-41 materials for the simultaneous catalytic abatement of NOx and VOCs. Microporous Mesoporous Mater. 2009, 117, 153–160. [Google Scholar] [CrossRef]
- Aissat, A.; Courcot, D.; Cousin, R.; Siffert, S. VOCs removal in the presence of NOx on Cs-Cu/ZrO2 catalysts. Catal. Today 2011, 176, 120–125. [Google Scholar] [CrossRef]
- Mrad, R.; Cousin, R.; Saliba, N.A.; Tidahy, L.; Siffert, S. Degradation of VOCs and NOx over Mg(Cu)-AlFe mixed oxides derived from hydrotalcite-like compounds. Comptes Rendus Chim. 2015, 18, 351–357. [Google Scholar] [CrossRef]
- Bertinchamps, F.; Treinen, M.; Blangenois, N.; Mariage, E.; Gaigneaux, E.M. Positive effect of NOx on the performances of VOx/TiO2-based catalysts in the total oxidation abatement of chlorobenzene. J. Catal. 2005, 230, 493–498. [Google Scholar] [CrossRef]
- Debecker, D.P.; Bertinchamps, F.; Blangenois, N.; Eloy, P.; Gaigneaux, E.M. On the impact of the choice of model VOC in the evaluation of V-based catalysts for the total oxidation of dioxins: Furan vs. chlorobenzene. Appl. Catal. B Environ. 2007, 74, 223–232. [Google Scholar] [CrossRef]
- Fan, C.; Li, K.; Peng, Y.; Duan, R.; Hu, F.; Jing, Q.; Chen, J.; Li, J. Fe-Doped α-MnO2 nanorods for the catalytic removal of NOx and chlorobenzene: The relationship between lattice distortion and catalytic redox properties. Phys. Chem. Chem. Phys. 2019, 21, 25880–25888. [Google Scholar] [CrossRef]
- Shao, J.; Wang, Z.; Liu, P.; Lin, F.; Zhu, Y.; He, Y.; Cen, K. Interplay effect on simultaneous catalytic oxidation of NOx and toluene over different crystal types of MnO2 catalysts. Proc. Combust. Inst. 2020, 38, 5433–5441. [Google Scholar] [CrossRef]
- Pope, D.; Walker, D.S.; Moss, R.L. Evaluation of platinum-honeycomb catalysts for the destructive oxidation of low concentrations of odorous compounds in air. Atmos. Environ. 1978, 12, 1921–1927. [Google Scholar] [CrossRef]
- Heyes, C.J.; Irwin, J.G.; Johnson, H.A.; Moss, R.L. Catalytic Oxidation of Organic Air Pollutants. Part 1. Single Metal Oxide Catalysts. J. Chem. Technol. Biotechnol. 1982, 32, 1025–1033. [Google Scholar] [CrossRef]
- Zagoruiko, A.N.; Mokrinskii, V.V.; Veniaminov, S.A.; Noskov, A.S. On the performance stability of the MnOx/Al2O3 catalyst for VOC incineration under forced adsorption-catalytic cycling conditions. J. Environ. Chem. Eng. 2017, 5, 5850–5856. [Google Scholar] [CrossRef]
- Shao, J.; Lin, F.; Wang, Z.; Liu, P.; Tang, H.; He, Y.; Cen, K. Low temperature catalytic ozonation of toluene in flue gas over Mn-based catalysts: Effect of support property and SO2/water vapor addition. Appl. Catal. B Environ. 2020, 266, 118662. [Google Scholar] [CrossRef]
- Laitinen, T.; Ojala, S.; Cousin, R.; Koivikko, N.; Poupin, C.; El Assal, Z.; Aho, A.; Keiski, R.L. Activity, selectivity, and stability of vanadium catalysts in formaldehyde production from emissions of volatile organic compounds. J. Ind. Eng. Chem. 2020, 83, 375–386. [Google Scholar] [CrossRef]
- Koivikko, N.; Laitinen, T.; Mouammine, A.; Ojala, S.; Keiski, R.L. Catalytic activity studies of Vanadia/Silica–Titania catalysts in SVOC partial oxidation to formaldehyde: Focus on the catalyst composition. Catalysts 2018, 8, 56. [Google Scholar] [CrossRef] [Green Version]
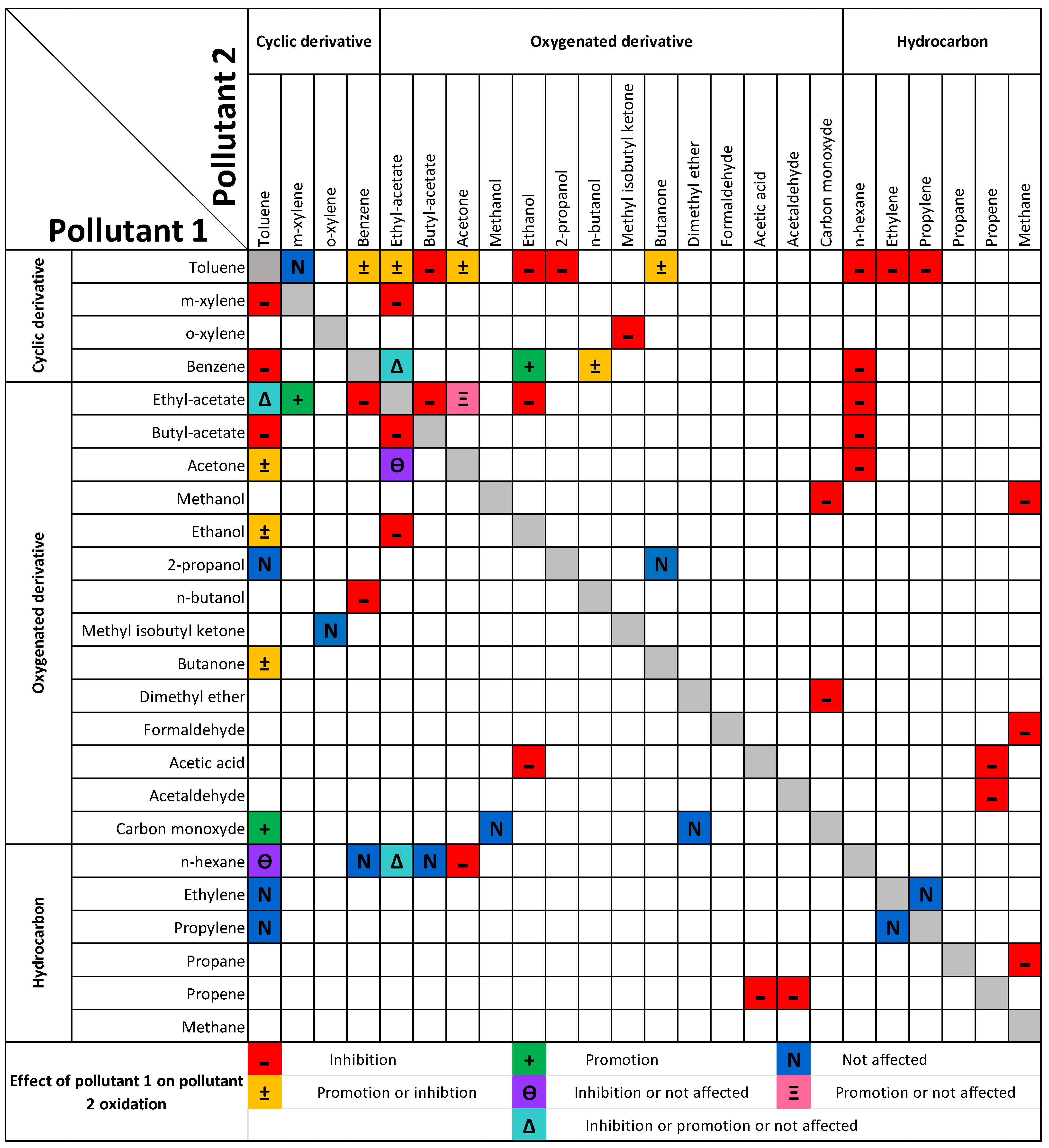
| Pollutant 1 | Pollutant 2 | Effect 1 on 2 | Effect 2 on 1 |
|---|---|---|---|
| Toluene | m-xylene | Not affected [15] | Inhibition [15] |
| Ethyl-acetate | Inhibition [16,17,18,19]/Promotion [20] | Not affected [17]/Inhibition [16,18,20]/Promotion [19] | |
| Acetone | Inhibition [16,21]/Promotion [18] | Inhibition [16,21]/Promotion [18] | |
| Butyl acetate | Inhibition [16] | Inhibition [16] | |
| n-hexane | Inhibition [22,23] | Not affected [22]/Inhibition [23] | |
| CO | - | Promotion [14,24] | |
| Ethylene | Inhibition [25] | Not affected [25] | |
| Propylene | Inhibition [25] | Not affected [25] | |
| Ethanol | Inhibition [17,20,26] | Promotion [17]/Inhibition [20,26] | |
| 2-propanol | Inhibition [27] | Not affected [27] | |
| Benzene | Inhibition [19,22,28]/Promotion [29,30] | Inhibition [19,22,28] | |
| Butanone | Inhibition [13,31]/Promotion [32] | Inhibition [13,31]/Promotion [13] | |
| Benzene | Ethyl acetate | Not affected [33]/Promotion [34,35]/Inhibition [19] | Inhibition [19,33,34] |
| n-butanol | Promotion [36]/Inhibition [36] | Inhibition [35] | |
| n-hexane | Inhibition [22,35,37] | Not affected [22,37] | |
| Ethanol | Promotion [30] | - | |
| Ethyl acetate | Acetone | Promotion [15]/Not affected [15] | Not affected [15]/Inhibition [15] |
| m-xylene | Promotion [15] | Inhibition [15] | |
| n-hexane | Inhibition [16,18,35] | Not affected [16]/Inhibition [18]/Promotion [35] | |
| Ethanol | Inhibition [17,20] | Inhibition [17,20] | |
| Butyl acetate | Inhibition [38] | Inhibition [38] | |
| Methyl isobutyl ketone | o-xylene | Not affected [39] | Inhibition [39] |
| Butanone | 2-propanol | - | Not affected [27] |
| n-hexane | Acetone | Inhibition [16] | Inhibition [16] |
| Butyl acetate | Not affected [16] | Inhibition [16] | |
| Carbon monoxide | Methanol | Not affected [40] | Inhibition [40] |
| Dimethyl ether | Not affected [40] | Inhibition [40] | |
| Methane | Propane | - | Inhibition [41] |
| Formaldehyde | - | Inhibition [41] | |
| Methanol | - | Inhibition [41] | |
| Ethanol | Acetic acid | - | Inhibition [42] |
| Propene | Acetic acid | Inhibition [43] | Inhibition [43] |
| Acetaldehyde | Inhibition [43] | Inhibition [43] | |
| Ethylene | Propylene | Not affected [25] | Not affected [25] |
| Compound | T50 (°C) | T90 (°C) |
|---|---|---|
| Toluene | 250 | 360 |
| With acetone | 280 | 410 |
| With ethyl acetate | 275 | 400 |
| With butyl acetate | 265 | 400 |
| n-hexane | 255 | 425 |
| With acetone | 270 | >500 |
| With ethyl acetate | 290 | 500 |
| With butyl acetate | 270 | 500 |
| Acetone | 200 | 235 |
| With toluene | 230 | 300 |
| With n-hexane | <200 | 250 |
| Ethyl acetate | 205 | 300 |
| With toluene | 250 | 345 |
| With n-hexane | 215 | 300 |
| Butyl acetate | <200 | 280 |
| With toluene | 200 | 325 |
| With n-hexane | 200 | 280 |
| Catalyst | Conditions | Water Content in Relative Humidity | Activity | Ref. |
|---|---|---|---|---|
| 0.5 Pd/γ-Al2O3 | GHSV = 125,000 h−1 T = 23–25 °C Activities are measured after 15 h | 20% | 50% | [50] |
| 30% | 50% | |||
| 40% | 45% | |||
| 49% | 43% | |||
| 61% | 44% | |||
| 80% | 40% | |||
| 87% | 37% | |||
| Fe2O3-MnO2 | 20% | 6% | ||
| 50% | 5% | |||
| 80% | 5% | |||
| CuO-MnO2 | 20% | 15% | ||
| 50% | 20% | |||
| 74% | 22% | |||
| MnO2 | [HCHO] = 150–200 ppm Face velocity = 50 cm s−1 Activities are measured after 100 h | >90% | ~80% | [51] |
| 25–30% | 85–90% | |||
| MnOx-CeO2 | GHSV = 21,000 mL·gcat−1 h−1 [HCHO] = 580 ppm T = 100 °C | 0% | 100% | [52] |
| 92% | 95% | |||
| MnO2 Birnessite | GHSV = 180,000 h−1 [HCHO] = 10 ppm mcata = 0.05 g Rate flow = 300 mL·min−1 T = 25 °C | 0% | - | [53] |
| 33% | 85% | |||
| 65% | 85% | |||
| 92% | 65% | |||
| 1% Pt/TiO2 1% Na—1% Pt/TiO2 2% Na—1% Pt/TiO2 | GHSV = 120,000 h−1 [HCHO] = 600 ppm Flow rate = 50 mL·min−1 T = 25 °C | 50% | 19% 98% 100% | [54] |
| 0.1% Pt/TiO2 | GHSV = 80,000 h−1 [HCHO] = 10 ppm T = 25 °C Activities are measured after 10 h | 0% | 45.2% | [55] |
| 25% | ~100% | |||
| 50% | ~100% | |||
| 75% | ~97% | |||
| 97.5% | 95.6% | |||
| 0.1% Pd-TiO2/DP | GHSV = 120,000 mL·gcat−1 h−1 [HCHO] = 10 ppm mcata = 0.5 g Rate flow = 1 L min−1 T = Room temperature Activities are measured after 10 h | 0% | 54.5% | [56] |
| 25% | 78.5% | |||
| 50% | >95% | |||
| 75% | >95% | |||
| 97.5% | >95% | |||
| HZ | [HCHO] = 70 ppm T = 25 °C | 0 | <0.1 | [57] |
| 50% | <0.1 | |||
| Pt/HZ | 0 | 60 | ||
| 50% | 100 | |||
| Pd/HZ | 0 | 20 | ||
| 50% | 96 | |||
| Ag/HZ | 0 | <0.1 | ||
| 50% | <0.1 | |||
| Pt/SiO2 | 0 | 90 | ||
| 50% | 100 | |||
| Pt/Fe2O3 | GHSV = 60,000 h−1 [HCHO] = 400 ppm mcata = 0.2 g T = 25 °C | 0% | 85% | [58] |
| 3%(water vapor) | 100% | |||
| Au/FeOx-C200 | GHSV = 34,000 h−1 [HCHO] = 80 ppm mcata = 0.19 g Rate flow = 100 mL min−1 T = 25 °C Activities are expressed in µmol·s−1·gAu.s−1 | 0% | 0.21 | [59] |
| 25% | 7.55 | |||
| 50% | 10.78 | |||
| 75% | 9.7 | |||
| δ-MnO2/PET | GHSV = 17,000 h−1 [HCHO] = 0.6 mg·m−3 mcata = 0.5 g Flow rate = 1 L·min−1 T = 25 °C Activities are measured after 10 h | 0% | 65% | [60] |
| 30% | 75% | |||
| 50% | 94% | |||
| 80% | 89% | |||
| MnO2 birnessite | [HCHO] = 40 ppm mcata = 0.1 g Flow rate = 200 mL·min−1 T = 30–140 °C | 80% | T50 = 53 °C | [61] |
| K-Birnessite | GHSV = 1,200,000 h−1 [HCHO] = 200 ppm mcata = 50 mg T = room temperature Activities are measured after 10 h | 48% | 40% | [62] |
| Mg-Birnessite | 0% | |||
| Ca-Birnessite | 30% | |||
| Fe-Birnessite | 5% | |||
| Mn0.75Co2.25O4 | GHSV = 60,000 h−1 [HCHO] = 80 ppm mcata = 0.150 g Flow rate = 100 mL/min T = 70 °C Activities are measured after 30 h | 0% | 30% | [63] |
| 50% | 70% | |||
| MnO2 | GHSV = 240,000 mL·g−1.h−1 [HCHO] = 480 ppm | 48% | T50 = 85 °C T90 = 110 °C | [64] |
| MnO2-P1 | T50 = 74 °C T90 = 97 °C | |||
| MnO2-P2 | T50 = 62 °C T90 = 86 °C | |||
| MnO2/SBA-15 | GHSV = 30,000 h−1 [HCHO] = 100 ppm mcata = 0.1 g T = 130 °C Activities are expressed in relative activity after 60 h | 50% | 0.9 | [65] |
| Mn3O4/SBA-15 | 0.5 |
| Catalyst | Conditions | Water Content | Activity | Ref. | |
|---|---|---|---|---|---|
| La0.9SR0.1CoO3 | 200 mg—[Toluene] = 1000 ppm in air—230 °C | 0 | 93% | Toluene conversion into CO2 | [67] |
| 50% R.H. | 58% | ||||
| CuMnO | 180 mg—[Toluene] = 1000 ppm in air | 0 | 100% | Toluene conversion at 230 °C | [68] |
| 2.3 vol% | 97% | ||||
| 0 | 96.6% | Toluene conversion at 220 °C | |||
| 2.3 vol% | 86.9% | ||||
| 0 | 62.5% | Toluene conversion at 210 °C | |||
| 2.3 vol% | 58.3% | ||||
| Au/Mn2O3 | 50 mg—[Toluene] = 1000 ppm in air—33.4 mL·min−1—40 000 mL·g−1·h−1 | 0 | 260 °C | 90% Toluene conversion | [69] |
| 1 vol% | 280 °C | ||||
| Pd/Mn2O3 | 0 | 235 °C | |||
| 1 vol% | 240 °C | ||||
| Au-Pd/Mn2O3 | 0 | 165 °C | |||
| 0.5 vol% | 160 °C | ||||
| 1 vol% | 150 °C | ||||
| 2 vol% | 160 °C | ||||
| 5 vol% | 180 °C | ||||
| Cu-Mn/γ-Al2O3 | [Toluene] = 1200 ppm in air—15,000 h−1 | 0 | 225 °C | 90% Toluene conversion | [70] |
| 3.8 vol% | 270 °C | ||||
| Cu-Mn/TiO2 | 0 | 225 °C | |||
| 3.8 vol% | 255 °C | ||||
| Cu-Mn/cordierite | 0 | 255 °C | |||
| 3.8 vol% | 280 °C | ||||
| V2O5/TiO2 | P toluene = 0.0128 atm in air—240 °C | 0 | 0.2 | mmol.gcat−1·h−1 after 200 min | [72] |
| 0.2 atm P H2O | 1.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rochard, G.; Olivet, L.; Tannous, M.; Poupin, C.; Siffert, S.; Cousin, R. Recent Advances in the Catalytic Treatment of Volatile Organic Compounds: A Review Based on the Mixture Effect. Catalysts 2021, 11, 1218. https://doi.org/10.3390/catal11101218
Rochard G, Olivet L, Tannous M, Poupin C, Siffert S, Cousin R. Recent Advances in the Catalytic Treatment of Volatile Organic Compounds: A Review Based on the Mixture Effect. Catalysts. 2021; 11(10):1218. https://doi.org/10.3390/catal11101218
Chicago/Turabian StyleRochard, Guillaume, Lilian Olivet, Mariebelle Tannous, Christophe Poupin, Stéphane Siffert, and Renaud Cousin. 2021. "Recent Advances in the Catalytic Treatment of Volatile Organic Compounds: A Review Based on the Mixture Effect" Catalysts 11, no. 10: 1218. https://doi.org/10.3390/catal11101218
APA StyleRochard, G., Olivet, L., Tannous, M., Poupin, C., Siffert, S., & Cousin, R. (2021). Recent Advances in the Catalytic Treatment of Volatile Organic Compounds: A Review Based on the Mixture Effect. Catalysts, 11(10), 1218. https://doi.org/10.3390/catal11101218







