Fundamentals of Gas Diffusion Electrodes and Electrolysers for Carbon Dioxide Utilisation: Challenges and Opportunities
Abstract
1. Introduction
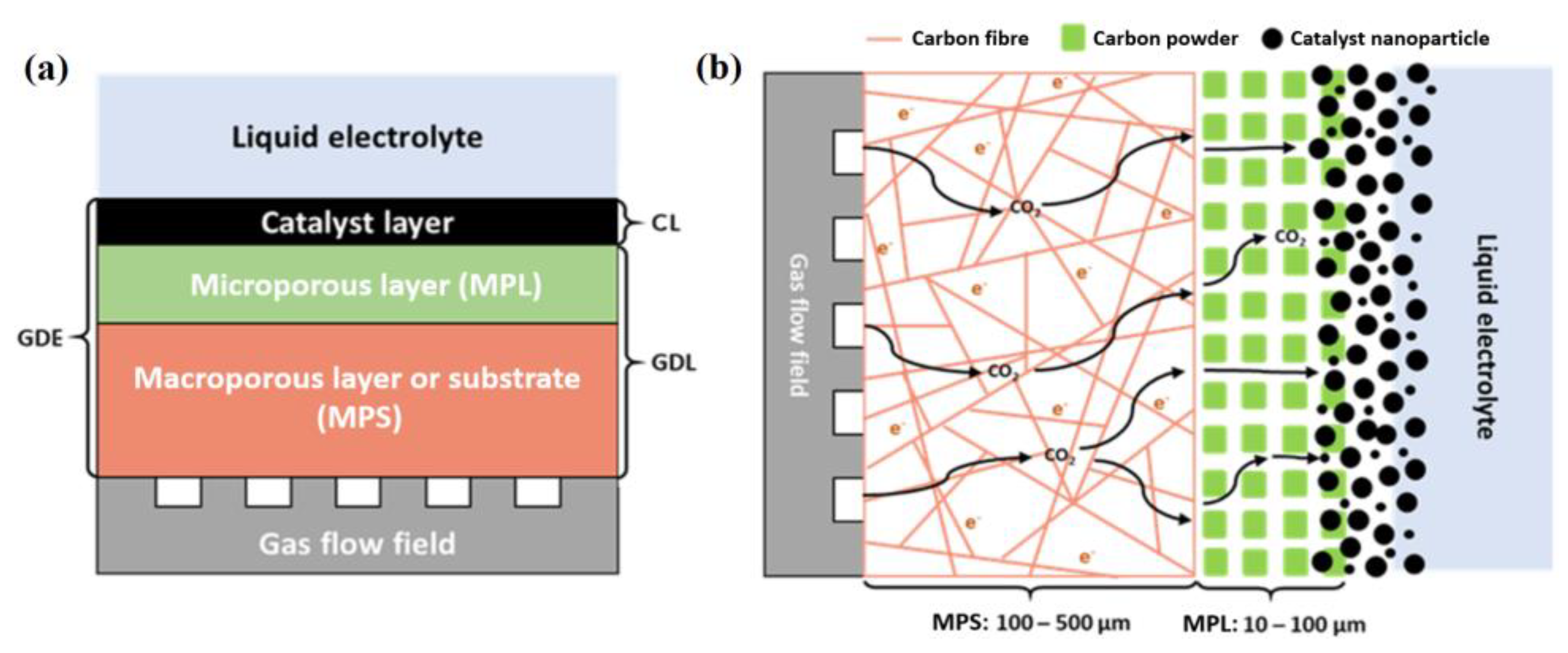
2. Gas Diffusion Electrodes
2.1. Macroporous Layer or Support (MPS)
2.2. Microporous Layer (MPL)
3. Aqueous-Fed and Gas-Fed Electrolysers
4. Carbon Dioxide Electrolysers
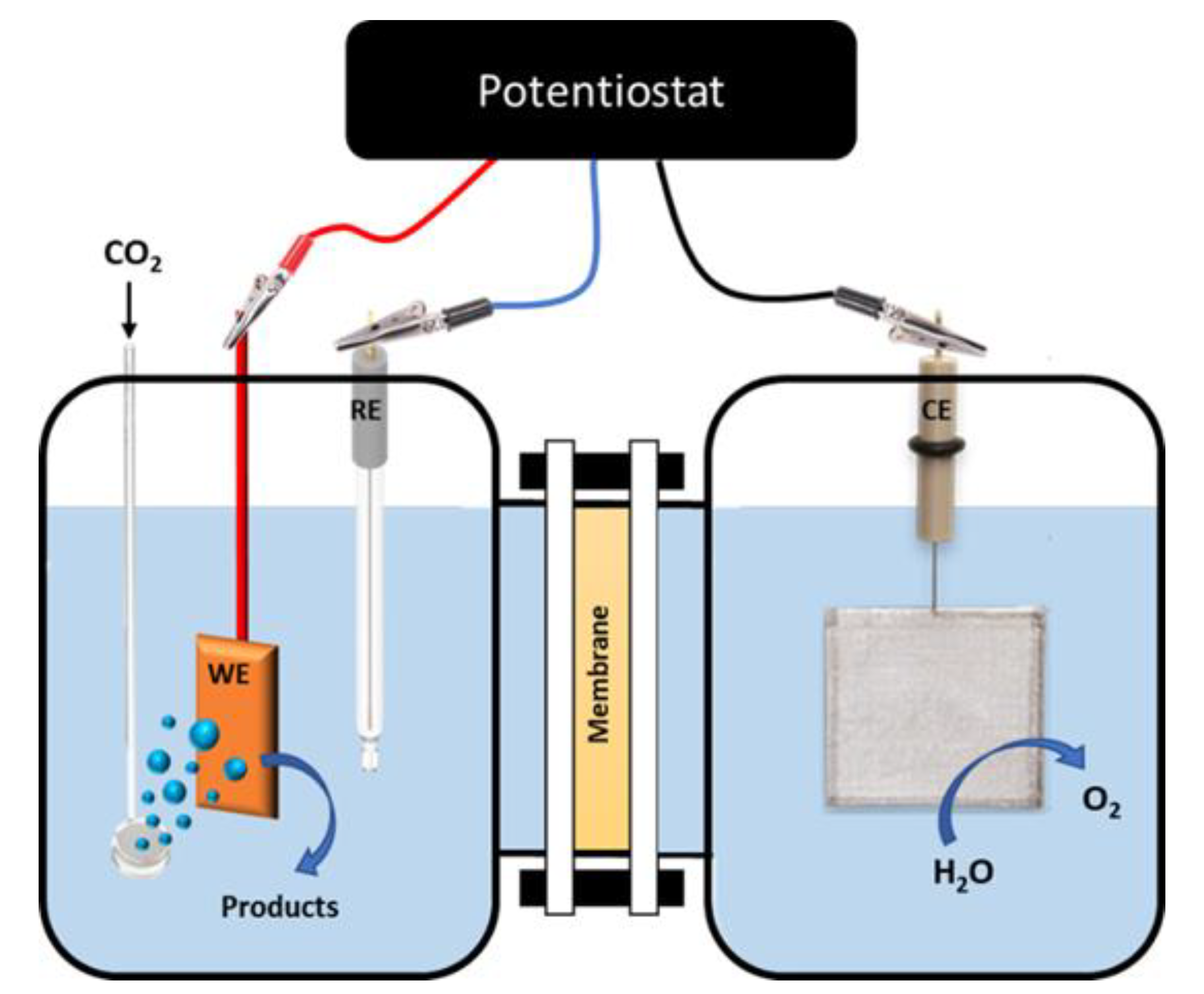
4.1. H-Cell Reactors
4.2. Flow Cell Reactors
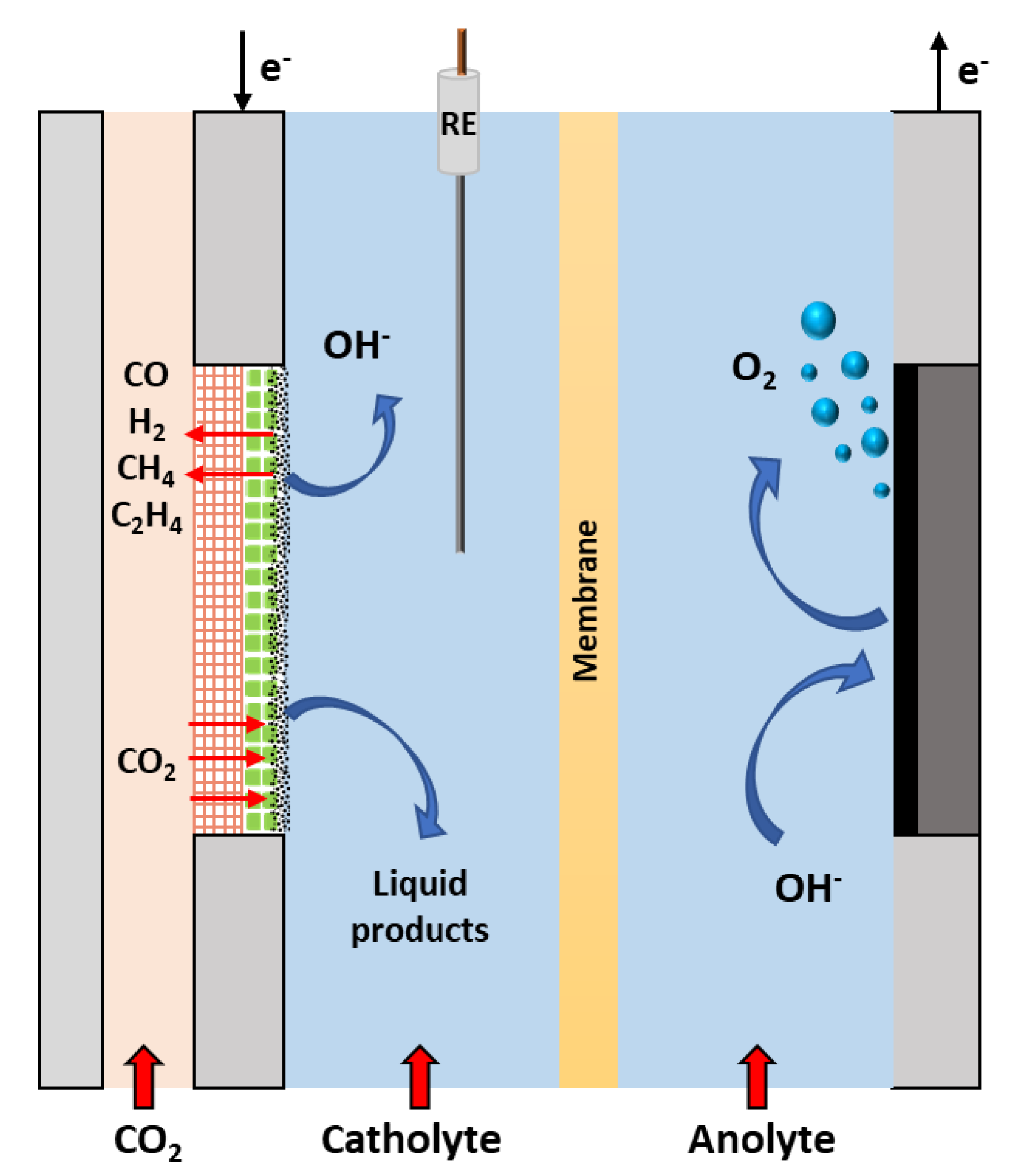
4.2.1. Basic Gas-Fed Flow Electrolysers
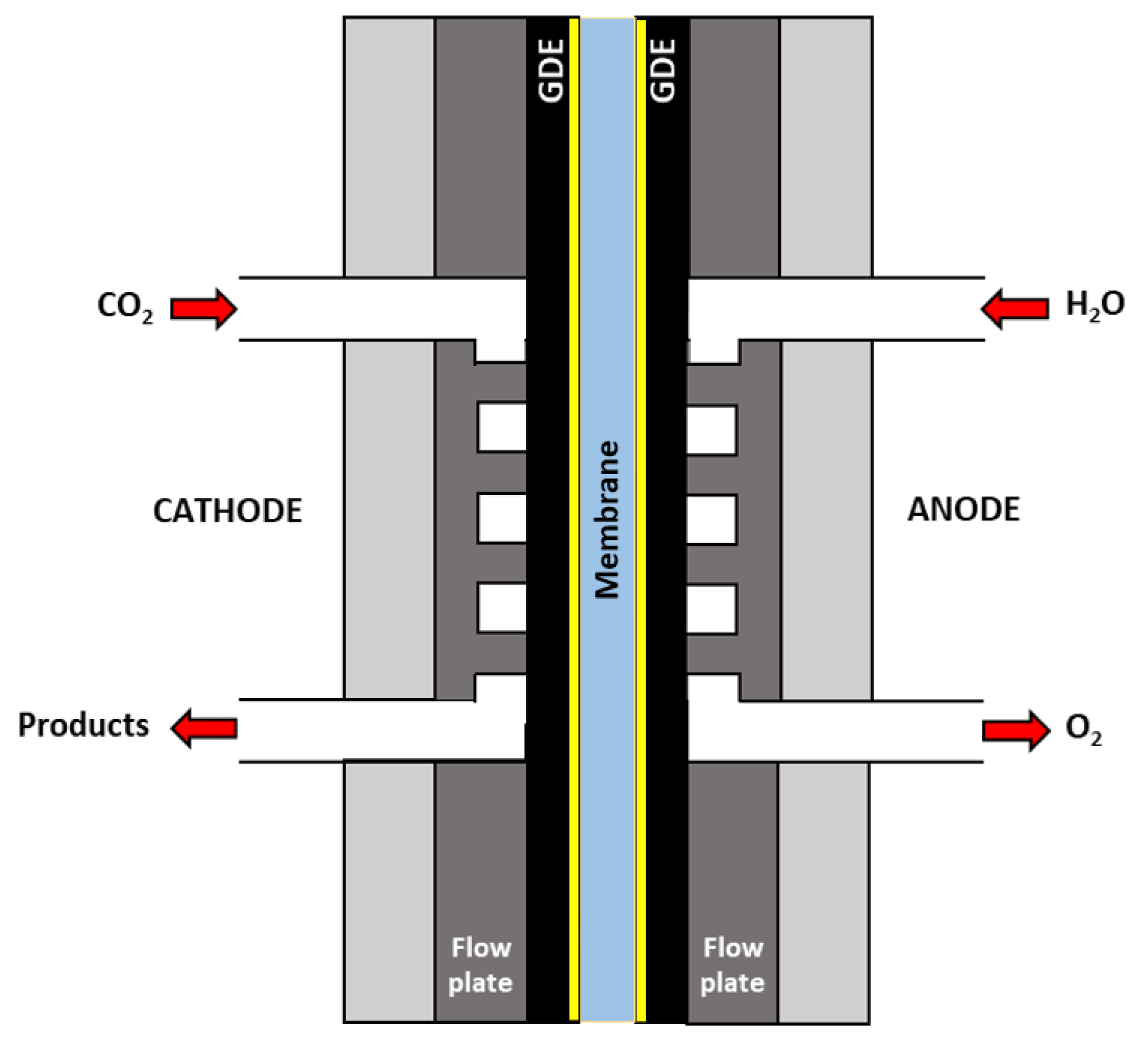
4.2.2. Polymer Electrolyte Membrane Electrolysers
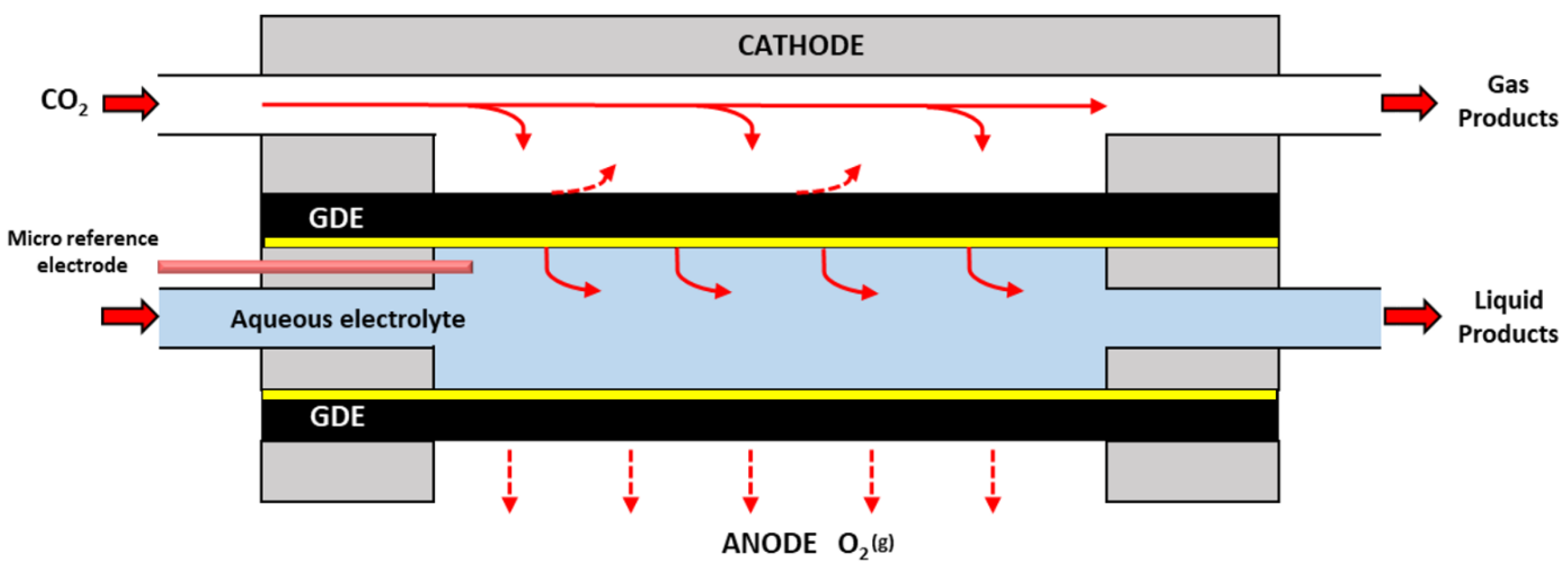
4.2.3. Microfluidic Electrolysers
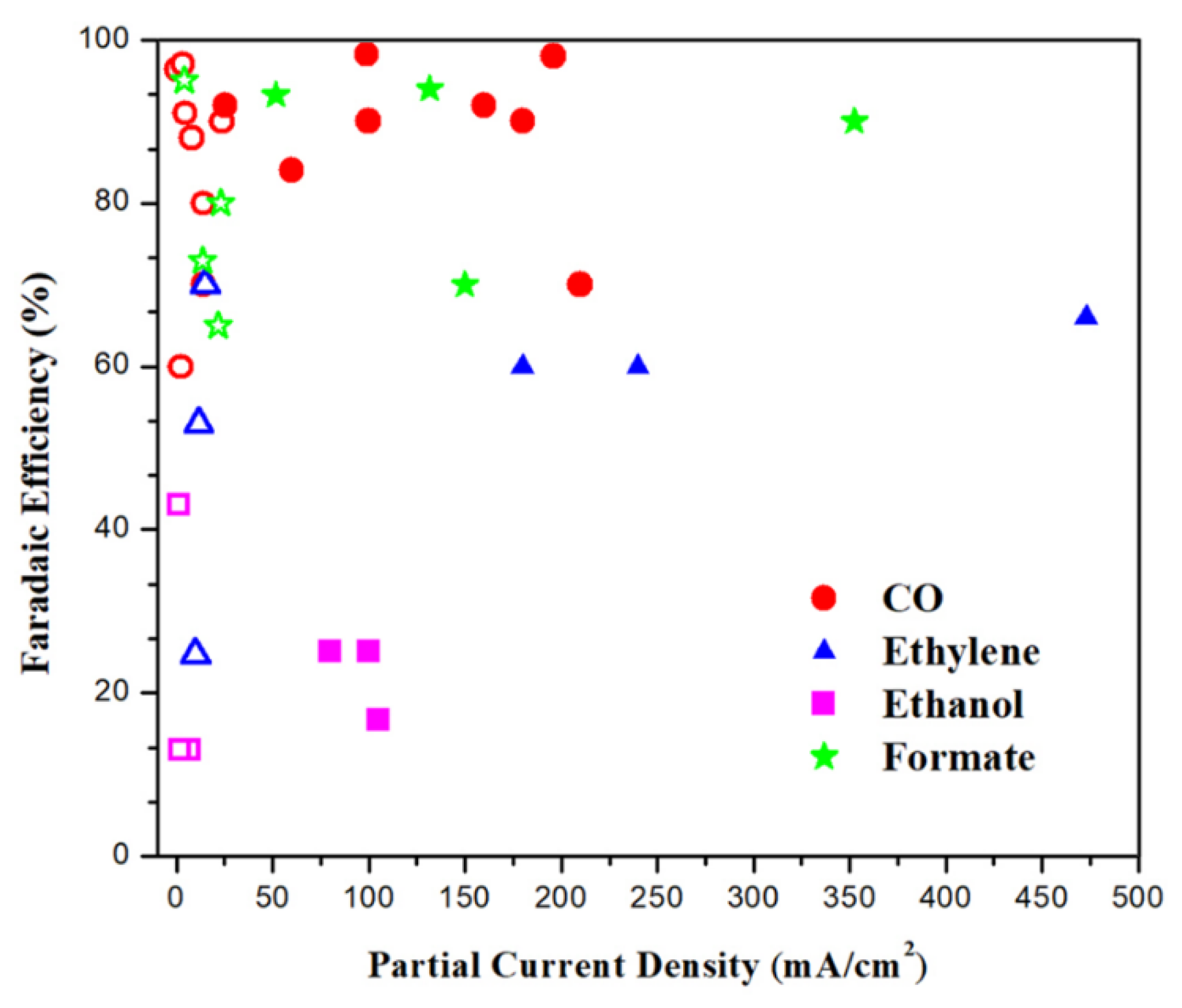
4.2.4. Comparison of Reactor Configurations
4.2.5. The Role of Phases in Reactor Operation
5. Failures and Challenges in CO2 Flow Electrolysers
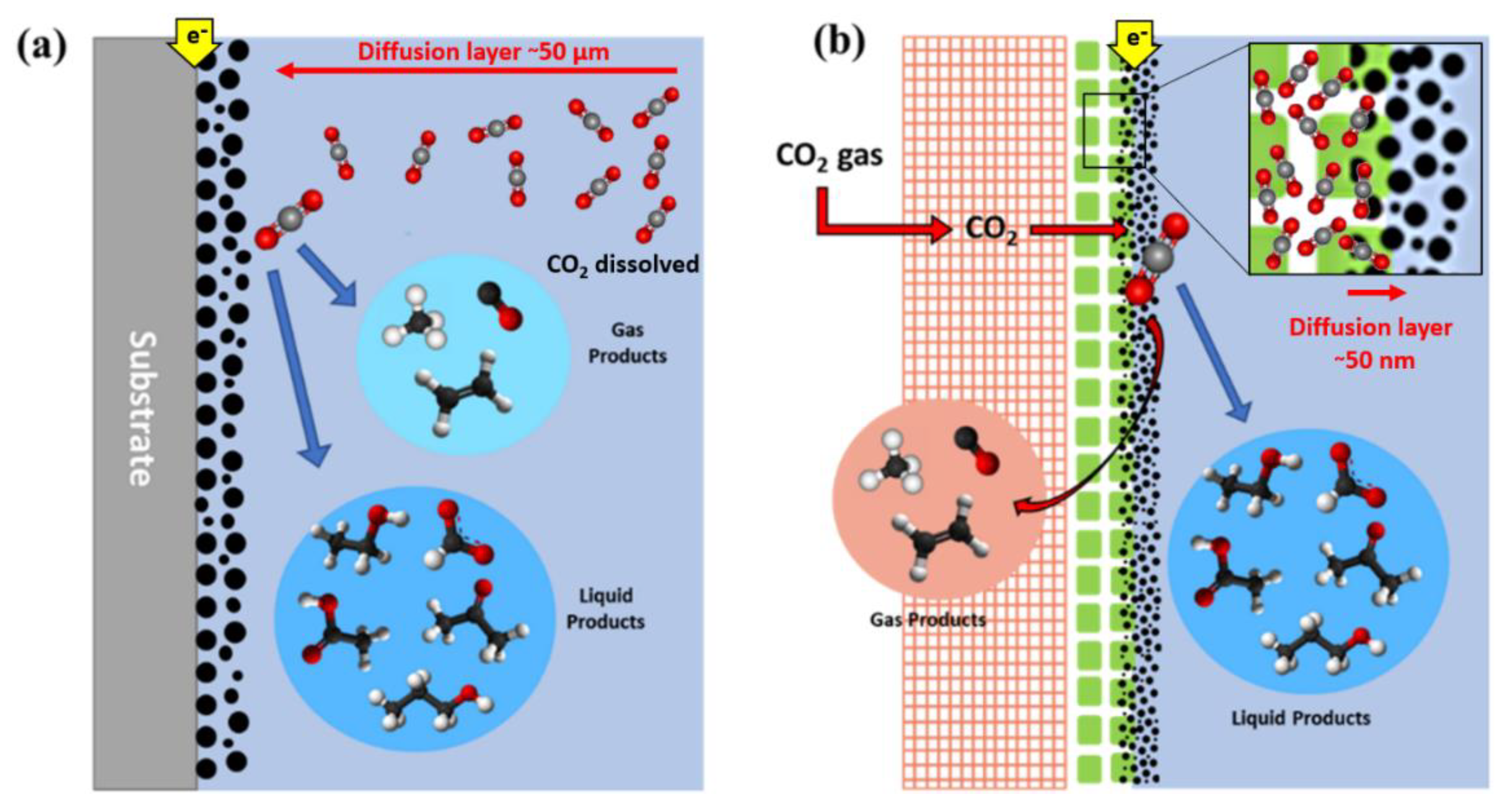
5.1. Components of Gas Diffusion Electrodes
5.2. Stability and Degradation of Electrolyser Components
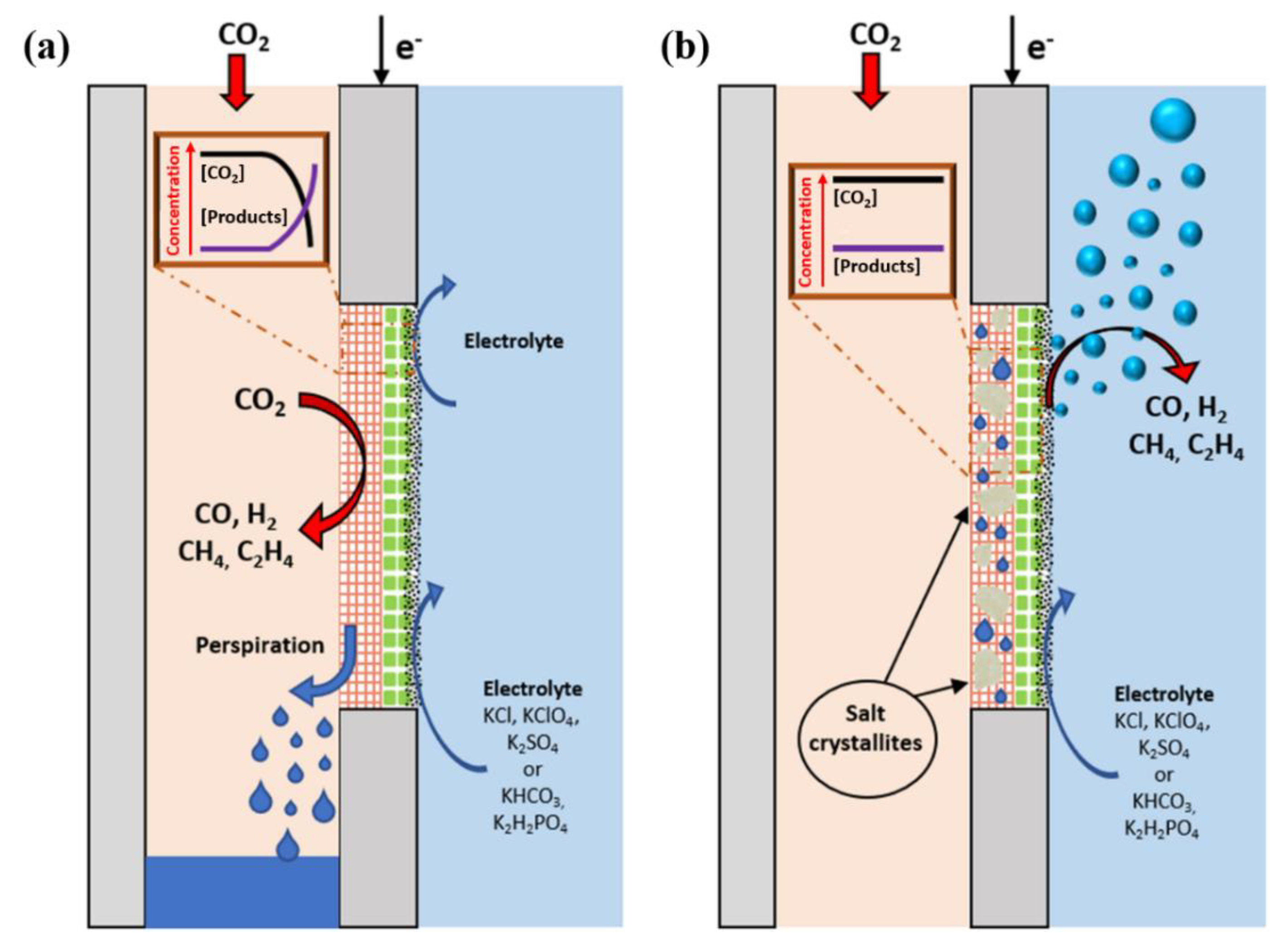
5.3. Electrolyte Carbonation
5.4. Flow Configurations
5.4.1. Challenges in Flow-Through Configuration
5.4.2. Challenges in Flow-By Configuration
6. Opportunities for Improvement of CO2 Electrolysis
7. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Masson-Delmotte, V.P.; Zhai, H.-O.P.; Roberts, D.; Skea, J.; Shukla, P.R.; Pirani, A.; Moufouma-Okia, W.C.; Péan, R.P.; Connors, S.; Matthews, J.B.R.; et al. Global Warming of 1.5 C An IPCC Special Report on the Impacts of Global Warming of 1.5 C Above Pre-Industrial Levels and Related Global Greenhouse Gas. Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change; Intergovernmental Panel on Climate Change, World Meteorological Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Cuéllar-Franca, R.M.; Azapagic, A. Carbon capture, storage and utilisation technologies: A critical analysis and comparison of their life cycle environmental impacts. J. CO2 Util. 2015, 9, 82–102. [Google Scholar] [CrossRef]
- Bujnicki, J.; Dykstra, P.; Fortunato, E.; Heuer, R.-D.; Keskitalo, C.; Nurse, P. Novel Carbon Capture and Utilization Technologies; European Commission, Publications Office of the European Union: Luxembourg, 2018. [Google Scholar]
- Olivier, J.G.; Peters, J.A.; Janssens-Maenhout, G. Trends in Global CO2 Emissions: 2012 Report; PBL Netherlands Environmental Assessment Agency: The Hague, The Netherlands, 2012. [Google Scholar]
- Olivier, J.G.J.; Peters, J.A.H.W. Trends in Global CO2 and Total Greenhouse Gas Emissions: 2018 Report; PBL Netherlands Environmental Assessment Agency: The Hague, The Netherlands, 2018. [Google Scholar]
- Artz, J.; Müller, T.E.; Thenert, K.; Kleinekorte, J.; Meys, R.; Sternberg, A.; Bardow, A.; Leitner, W. Sustainable conversion of carbon dioxide: An integrated review of catalysis and life cycle assessment. Chem. Rev. 2018, 118, 434–504. [Google Scholar] [CrossRef]
- Whipple, D.T.; Kenis, P.J.A. Prospects of CO2 utilization via direct heterogeneous electrochemical reduction. J. Phys. Chem. Lett. 2010, 1, 3451–3458. [Google Scholar] [CrossRef]
- El Mekawy, A.; Hegab, H.M.; Mohanakrishna, G.; Elbaz, A.F.; Bulut, M.; Pant, D. Technological advances in CO2 conversion electro-biorefinery: A step toward commercialization. Bioresour. Technol. 2016, 215, 357–370. [Google Scholar] [CrossRef]
- Nitopi, S.; Bertheussen, E.; Scott, S.B.; Liu, X.; Engstfeld, A.K.; Horch, S.; Seger, B.; Stephens, I.E.L.; Chan, K.; Hahn, C.; et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 2019, 119, 7610–7672. [Google Scholar] [CrossRef]
- Raciti, D.; Wang, C. Recent advances in CO2 reduction electrocatalysis on copper. ACS Energy Lett. 2018, 3, 1545–1556. [Google Scholar] [CrossRef]
- Perry, S.C.; Leung, P.-K.; Wang, L.; de León, C.P. Developments on carbon dioxide reduction: Their promise, achievements and challenges. Curr. Opin. Electrochem. 2020. [Google Scholar] [CrossRef]
- Du, C.; Wang, X.; Chen, W.; Feng, S.; Wen, J.; Wu, Y.A. CO2 transformation to multicarbon products by photocatalysis and electrocatalysis. Mater. Today Adv. 2020, 6, 100071. [Google Scholar] [CrossRef]
- Singh, M.R.; Clark, E.L.; Bell, A.T. Effects of electrolyte, catalyst, and membrane composition and operating conditions on the performance of solar-driven electrochemical reduction of carbon dioxide. PCCP 2015, 17, 18924–18936. [Google Scholar] [CrossRef] [PubMed]
- Durst, J.; Rudnev, A.; Dutta, A.; Fu, Y.; Herranz, J.; Kaliginedi, V.; Kuzume, A.; Permyakova, A.A.; Paratcha, Y.; Broekmann, P.; et al. Electrochemical CO2 Reduction—A critical view on fundamentals, materials and applications. CHIMIA 2015, 69, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Mathur, V.K.; Crawford, J. Fundamentals of Gas Diffusion Layers in PEM Fuel Cells. In Recent Trends in Fuel Cell Science and Technology; Basu, S., Ed.; Springer: New York, NY, USA, 2007; pp. 116–128. [Google Scholar]
- Inaba, M.; Jensen, A.W.; Sievers, G.W.; Escudero-Escribano, M.; Zana, A.; Arenz, M. Benchmarking high surface area electrocatalysts in a gas diffusion electrode: Measurement of oxygen reduction activities under realistic conditions. Energy Environ. Sci. 2018, 11, 988–994. [Google Scholar] [CrossRef]
- Pan, C.; Li, Q.; Jensen, J.O.; He, R.; Cleemann, L.N.; Nilsson, M.S.; Bjerrum, N.J.; Zeng, Q. Preparation and operation of gas diffusion electrodes for high-temperature proton exchange membrane fuel cells. J. Power Sources 2007, 172, 278–286. [Google Scholar] [CrossRef]
- Birdja, Y.Y.; Pérez-Gallent, E.; Figueiredo, M.C.; Göttle, A.J.; Calle-Vallejo, F.; Koper, M.T.M. Advances and challenges in understanding the electrocatalytic conversion of carbon dioxide to fuels. Nat. Energy 2019, 4, 732–745. [Google Scholar] [CrossRef]
- Verma, S.; Kim, B.; Jhong, H.-R.M.; Ma, S.; Kenis, P.J.A. A Gross-Margin Model for Defining Technoeconomic Benchmarks in the Electroreduction of CO2. ChemSusChem 2016, 9, 1972–1979. [Google Scholar] [CrossRef]
- Spurgeon, J.M.; Kumar, B. A comparative technoeconomic analysis of pathways for commercial electrochemical CO2 reduction to liquid products. Energy Environ. Sci. 2018, 11, 1536–1551. [Google Scholar] [CrossRef]
- Li, X.; Anderson, P.; Jhong, H.-R.M.; Paster, M.; Stubbins, J.F.; Kenis, P.J.A. Greenhouse gas emissions, energy efficiency, and cost of synthetic fuel production using electrochemical CO2 conversion and the Fischer–Tropsch process. Energy Fuels 2016, 30, 5980–5989. [Google Scholar] [CrossRef]
- Murata, K.; Tanaka, H.; Ishii, K. Electrochemical reduction of CO2 by a gas-diffusion electrode composed of fac-Re(diimine)(CO)3Cl and carbon nanotubes. J. Phys. Chem. C 2019, 123, 12073–12080. [Google Scholar] [CrossRef]
- Bajracharya, S.; Vanbroekhoven, K.; Buisman, C.J.N.; Pant, D.; Strik, D.P.B.T.B. Application of gas diffusion biocathode in microbial electrosynthesis from carbon dioxide. Environ. Sci. Pollut. Res. 2016, 23, 22292–22308. [Google Scholar] [CrossRef]
- Higgins, D.; Hahn, C.; Xiang, C.; Jaramillo, T.F.; Weber, A.Z. Gas-diffusion electrodes for carbon dioxide reduction: A new paradigm. ACS Energy Lett. 2019, 4, 317–324. [Google Scholar] [CrossRef]
- Park, S.; Lee, J.-W.; Popov, B.N. A review of gas diffusion layer in PEM fuel cells: Materials and designs. Int. J. Hydrogen Energy 2012, 37, 5850–5865. [Google Scholar] [CrossRef]
- Park, S.; Popov, B.N. Effect of a GDL based on carbon paper or carbon cloth on PEM fuel cell performance. Fuel 2011, 90, 436–440. [Google Scholar] [CrossRef]
- Khodabakhshi, S.; Fulvio, P.F.; Andreoli, E. Carbon black reborn: Structure and chemistry for renewable energy harnessing. Carbon 2020, 162, 604–649. [Google Scholar] [CrossRef]
- Mathias, M.F.; Roth, J.; Fleming, J.; Lehnert, W. Diffusion media materials and characterisation. In Handbook of Fuel Cells; Vielstich, W., Lamm, A., Gasteiger, H.A., Yokokawa, H., Eds.; Wiley: New York, NY, USA, 2010. [Google Scholar]
- Cabasso, I.; Yuan, Y.; Xu, X. Gas Diffusion Electrodes Based on Poly (Vinylidene Fluoride) Carbon Blends. U.S. Patent No. 5,783,325, 21 July 1998. [Google Scholar]
- MacLeod, E.N. Wet Proofed Conductive Current Collector for the Electrochemical Cells. U.S. Patent No. 4,215,183, 29 July 1980. [Google Scholar]
- Lim, C.; Wang, C.Y. Effects of hydrophobic polymer content in GDL on power performance of a PEM fuel cell. Electrochim. Acta 2004, 49, 4149–4156. [Google Scholar] [CrossRef]
- Park, G.-G.; Sohn, Y.-J.; Yang, T.-H.; Yoon, Y.-G.; Lee, W.-Y.; Kim, C.-S. Effect of PTFE contents in the gas diffusion media on the performance of PEMFC. J. Power Sources 2004, 131, 182–187. [Google Scholar] [CrossRef]
- Bevers, D.; Rogers, R.; von Bradke, M. Examination of the influence of PTFE coating on the properties of carbon paper in polymer electrolyte fuel cells. J. Power Sources 1996, 63, 193–201. [Google Scholar] [CrossRef]
- Zhang, F.-Y.; Advani, S.G.; Prasad, A.K. Performance of a metallic gas diffusion layer for PEM fuel cells. J. Power Sources 2008, 176, 293–298. [Google Scholar] [CrossRef]
- Fushinobu, K.; Takahashi, D.; Okazaki, K. Micromachined metallic thin films for the gas diffusion layer of PEFCs. J. Power Sources 2006, 158, 1240–1245. [Google Scholar] [CrossRef]
- Luo, W.; Zhang, J.; Li, M.; Züttel, A. Boosting CO production in electrocatalytic CO2 reduction on highly porous Zn catalysts. ACS Catal. 2019, 9, 3783–3791. [Google Scholar] [CrossRef]
- Park, S.B.; Park, Y.-I. Fabrication of gas diffusion layer (GDL) containing microporous layer using flourinated ethylene prophylene (FEP) for proton exchange membrane fuel cell (PEMFC). Int. J. Precis. Eng. Manuf. 2012, 13, 1145–1151. [Google Scholar] [CrossRef]
- Nam, J.H.; Lee, K.-J.; Hwang, G.-S.; Kim, C.-J.; Kaviany, M. Microporous layer for water morphology control in PEMFC. Int. J. Heat Mass Trans. 2009, 52, 2779–2791. [Google Scholar] [CrossRef]
- Kitahara, T.; Konomi, T.; Nakajima, H. Microporous layer coated gas diffusion layers for enhanced performance of polymer electrolyte fuel cells. J. Power Sources 2010, 195, 2202–2211. [Google Scholar] [CrossRef]
- Jhong, H.-R.M.; Brushett, F.R.; Kenis, P.J.A. The effects of catalyst layer deposition methodology on electrode performance. Adv. Energy Mater. 2013, 3, 589–599. [Google Scholar] [CrossRef]
- Park, S.; Lee, J.-W.; Popov, B.N. Effect of carbon loading in microporous layer on PEM fuel cell performance. J. Power Sources 2006, 163, 357–363. [Google Scholar] [CrossRef]
- Antolini, E.; Passos, R.R.; Ticianelli, E.A. Effects of the carbon powder characteristics in the cathode gas diffusion layer on the performance of polymer electrolyte fuel cells. J. Power Sources 2002, 109, 477–482. [Google Scholar] [CrossRef]
- Nam, J.H.; Kaviany, M. Effective diffusivity and water-saturation distribution in single- and two-layer PEMFC diffusion medium. Int. J. Heat Mass Trans. 2003, 46, 4595–4611. [Google Scholar] [CrossRef]
- Weber, A.Z.; Newman, J. Effects of microporous layers in polymer electrolyte fuel cells. J. Electrochem. Soc. 2005, 152, A677–A688. [Google Scholar] [CrossRef]
- Jordan, L.R.; Shukla, A.K.; Behrsing, T.; Avery, N.R.; Muddle, B.C.; Forsyth, M. Effect of diffusion-layer morphology on the performance of polymer electrolyte fuel cells operating at atmospheric pressure. J. Appl. Electrochem. 2000, 30, 641–646. [Google Scholar] [CrossRef]
- Jordan, L.R.; Shukla, A.K.; Behrsing, T.; Avery, N.R.; Muddle, B.C.; Forsyth, M. Diffusion layer parameters influencing optimal fuel cell performance. J. Power Sources 2000, 86, 250–254. [Google Scholar] [CrossRef]
- Kong, C.S.; Kim, D.-Y.; Lee, H.-K.; Shul, Y.-G.; Lee, T.-H. Influence of pore-size distribution of diffusion layer on mass-transport problems of proton exchange membrane fuel cells. J. Power Sources 2002, 108, 185–191. [Google Scholar] [CrossRef]
- Ismail, M.S.; Damjanovic, T.; Ingham, D.B.; Pourkashanian, M.; Westwood, A. Effect of polytetrafluoroethylene-treatment and microporous layer-coating on the electrical conductivity of gas diffusion layers used in proton exchange membrane fuel cells. J. Power Sources 2010, 195, 2700–2708. [Google Scholar] [CrossRef]
- Lee, H.-K.; Park, J.-H.; Kim, D.-Y.; Lee, T.-H. A study on the characteristics of the diffusion layer thickness and porosity of the PEMFC. J. Power Sources 2004, 131, 200–206. [Google Scholar] [CrossRef]
- Lin, G.; Nguyen, T.V. Effect of Thickness and Hydrophobic Polymer content of the gas diffusion layer on electrode flooding level in a PEMFC. J. Electrochem. Soc. 2005, 152, A1942. [Google Scholar] [CrossRef]
- Paganin, V.; Ticianelli, E.; Gonzalez, E.R. Development and electrochemical studies of gas diffusion electrodes for polymer electrolyte fuel cells. J. Appl. Electrochem. 1996, 26, 297–304. [Google Scholar] [CrossRef]
- Kim, B.; Hillman, F.; Ariyoshi, M.; Fujikawa, S.; Kenis, P.J.A. Effects of composition of the micro porous layer and the substrate on performance in the electrochemical reduction of CO2 to CO. J. Power Sources 2016, 312, 192–198. [Google Scholar] [CrossRef]
- Wang, Q.; Dong, H.; Yu, H.; Yu, H. Enhanced performance of gas diffusion electrode for electrochemical reduction of carbon dioxide to formate by adding polytetrafluoroethylene into catalyst layer. J. Power Sources 2015, 279, 1–5. [Google Scholar] [CrossRef]
- Song, T.J.; Song, H.; Kim, B.; Oh, J. Towards higher rate electrochemical CO2 conversion: From liquid-phase to gas-phase systems. Catalysts 2019, 9, 224. [Google Scholar] [CrossRef]
- Lee, M.-Y.; Park, K.T.; Lee, W.; Lim, H.; Kwon, Y.; Kang, S. Current achievements and the future direction of electrochemical CO2 reduction: A short review. Crit. Rev. Environ. Sci. Technol. 2020, 50, 769–815. [Google Scholar] [CrossRef]
- Hou, L.; Yan, J.; Takele, L.; Wang, Y.; Yan, X.; Gao, Y. Current progress of metallic and carbon-based nanostructure catalysts towards the electrochemical reduction of CO2. Inorg. Chem. Front. 2019, 6, 3363–3380. [Google Scholar] [CrossRef]
- Qiao, J.; Liu, Y.; Hong, F.; Zhang, J. A review of catalysts for the electroreduction of carbon dioxide to produce low-carbon fuels. Chem. Soc. Rev. 2014, 43, 631–675. [Google Scholar] [CrossRef]
- Burdyny, T.; Smith, W.A. CO2 reduction on gas-diffusion electrodes and why catalytic performance must be assessed at commercially-relevant conditions. Energy Environ. Sci. 2019, 12, 1442–1453. [Google Scholar] [CrossRef]
- Li, M.; Garg, S.; Chang, X.; Ge, L.; Li, L.; Konarova, M.; Rufford, T.E.; Rudolph, V.; Wang, G. Toward excellence of transition metal-based catalysts for CO2 electrochemical reduction: An overview of strategies and rationales. Small Methods 2020. [Google Scholar] [CrossRef]
- Weekes, D.M.; Salvatore, D.A.; Reyes, A.; Huang, A.; Berlinguette, C.P. Electrolytic CO2 reduction in a flow cell. Acc. Chem. Res. 2018, 51, 910–918. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.M. Trends in Electrochemistry and Corrosion at the Beginning of the 21st Century; Edicions Universitat Barcelona: Barcelona, Spain, 2004; Volume 20. [Google Scholar]
- Lucile, F.; Cézac, P.; Contamine, F.; Serin, J.-P.; Houssin, D.; Arpentinier, P. Solubility of carbon dioxide in water and aqueous solution containing sodium hydroxide at temperatures from (293.15 to 393.15) K and pressure up to 5 MPa: Experimental measurements. J. Chem. Eng. Data 2012, 57, 784–789. [Google Scholar] [CrossRef]
- Cook, R.L.; MacDuff, R.C.; Sammells, A.F. High rate gas phase CO2 reduction to ethylene and methane using gas diffusion electrodes. J. Electrochem. Soc. 1990, 137, 607. [Google Scholar] [CrossRef]
- Dinh, C.-T.; Burdyny, T.; Kibria, M.G.; Seifitokaldani, A.; Gabardo, C.M.; García de Arquer, F.P.; Kiani, A.; Edwards, J.P.; De Luna, P.; Bushuyev, O.S.; et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 2018, 360, 783. [Google Scholar] [CrossRef]
- Jiang, K.; Sandberg, R.B.; Akey, A.J.; Liu, X.; Bell, D.C.; Nørskov, J.K.; Chan, K.; Wang, H. Metal ion cycling of Cu foil for selective C–C coupling in electrochemical CO2 reduction. Nat. Catal. 2018, 1, 111–119. [Google Scholar] [CrossRef]
- Kim, D.; Kley, C.S.; Li, Y.; Yang, P. Copper nanoparticle ensembles for selective electroreduction of CO2 to C2–C3 products. PNAS 2017, 114, 10560–10565. [Google Scholar] [CrossRef]
- Cave, E.R.; Montoya, J.H.; Kuhl, K.P.; Abram, D.N.; Hatsukade, T.; Shi, C.; Hahn, C.; Nørskov, J.K.; Jaramillo, T.F. Electrochemical CO2 reduction on Au surfaces: Mechanistic aspects regarding the formation of major and minor products. PCCP 2017, 19, 15856–15863. [Google Scholar] [CrossRef]
- Zheng, X.; De Luna, P.; García de Arquer, F.P.; Zhang, B.; Becknell, N.; Ross, M.B.; Li, Y.; Banis, M.N.; Li, Y.; Liu, M.; et al. Sulfur-modulated tin sites enable highly selective electrochemical reduction of CO2 to formate. Joule 2017, 1, 794–805. [Google Scholar] [CrossRef]
- Feaster, J.T.; Shi, C.; Cave, E.R.; Hatsukade, T.; Abram, D.N.; Kuhl, K.P.; Hahn, C.; Nørskov, J.K.; Jaramillo, T.F. Understanding selectivity for the electrochemical reduction of carbon dioxide to formic acid and carbon monoxide on metal electrodes. ACS Catal. 2017, 7, 4822–4827. [Google Scholar] [CrossRef]
- Kottakkat, T.; Klingan, K.; Jiang, S.; Jovanov, Z.P.; Davies, V.H.; El-Nagar, G.A.M.; Dau, H.; Roth, C. Electrodeposited AgCu foam catalysts for enhanced reduction of CO2 to CO. ACS Appl. Mater. Interfaces 2019, 11, 14734–14744. [Google Scholar] [CrossRef] [PubMed]
- Barasa, G.O.; Yu, T.; Lu, X.; Zhou, X.; Wang, H.; Qian, L.; Yu, Y.; Liu, L.; Lei, P. Electrochemical training of nanoporous Cu-In catalysts for efficient CO2-to-CO conversion and high durability. Electrochim. Acta 2019, 295, 584–590. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, L.; Shi, L.; Yang, T.; Niu, D.; Hu, S.; Zhang, X. Enhancing CO2 electroreduction on nanoporous silver electrode in the presence of halides. Electrochim. Acta 2019, 313, 561–569. [Google Scholar] [CrossRef]
- Geng, Z.; Cao, Y.; Chen, W.; Kong, X.; Liu, Y.; Yao, T.; Lin, Y. Regulating the coordination environment of Co single atoms for achieving efficient electrocatalytic activity in CO2 reduction. Appl. Catal. B 2019, 240, 234–240. [Google Scholar] [CrossRef]
- Peng, X.; Chen, Y.; Mi, Y.; Zhuo, L.; Qi, G.; Ren, J.; Qiu, Y.; Liu, X.; Luo, J. Efficient Electroreduction CO2 to CO over MnO2 Nanosheets. Inorg. Chem. 2019, 58, 8910–8914. [Google Scholar] [CrossRef]
- Liu, K.; Wang, J.; Shi, M.; Yan, J.; Jiang, Q. Simultaneous achieving of high faradaic efficiency and CO partial current density for CO2 reduction via robust, noble-metal-free Zn nanosheets with favorable adsorption energy. Adv. Energy Mater. 2019, 9, 1900276. [Google Scholar] [CrossRef]
- Choi, J.; Kim, J.; Wagner, P.; Gambhir, S.; Jalili, R.; Byun, S.; Sayyar, S.; Lee, Y.M.; MacFarlane, D.R.; Wallace, G.G.; et al. Energy efficient electrochemical reduction of CO2 to CO using a three-dimensional porphyrin/graphene hydrogel. Energy Environ. Sci. 2019, 12, 747–755. [Google Scholar] [CrossRef]
- Bejtka, K.; Zeng, J.; Sacco, A.; Castellino, M.; Hernández, S.; Farkhondehfal, M.A.; Savino, U.; Ansaloni, S.; Pirri, C.F.; Chiodoni, A. Chainlike mesoporous SnO2 as a well-performing catalyst for electrochemical CO2 reduction. ACS Appl. Energy Mater. 2019, 2, 3081–3091. [Google Scholar] [CrossRef]
- Jia, L.; Yang, H.; Deng, J.; Chen, J.; Zhou, Y.; Ding, P.; Li, L.; Han, N.; Li, Y. Copper-bismuth bimetallic microspheres for selective electrocatalytic reduction of CO2 to formate. Chin. J. Chem. 2019, 37, 497–500. [Google Scholar] [CrossRef]
- Zhang, Z.; Ahmad, F.; Zhao, W.; Yan, W.; Zhang, W.; Huang, H.; Ma, C.; Zeng, J. Enhanced electrocatalytic reduction of CO2 via chemical coupling between indium oxide and reduced graphene oxide. Nano Lett. 2019, 19, 4029–4034. [Google Scholar] [CrossRef]
- Kim, Y.E.; Lee, W.; Youn, M.H.; Jeong, S.K.; Kim, H.J.; Park, J.C.; Park, K.T. Leaching-resistant SnO2/γ-Al2O3 nanocatalyst for stable electrochemical CO2 reduction into formate. J. Ind. Eng. Chem. 2019, 78, 73–78. [Google Scholar] [CrossRef]
- Ning, H.; Wang, X.; Wang, W.; Mao, Q.; Yang, Z.; Zhao, Q.; Song, Y.; Wu, M. Cubic Cu2O on nitrogen-doped carbon shells for electrocatalytic CO2 reduction to C2H4. Carbon 2019, 146, 218–223. [Google Scholar] [CrossRef]
- Kim, J.; Choi, W.; Park, J.W.; Kim, C.; Kim, M.; Song, H. Branched copper oxide nanoparticles induce highly selective ethylene production by electrochemical carbon dioxide reduction. J. Am. Chem. Soc. 2019, 141, 6986–6994. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Yang, M.-P.; Hu, Q.-L.; Li, S.-M.; Wang, H.; Lu, J.-X. Cu/TiO2 nanoparticles modified nitrogen-doped graphene as a highly efficient catalyst for the selective electroreduction of CO2 to different alcohols. J. CO2 Util. 2018, 24, 334–340. [Google Scholar] [CrossRef]
- Jeon, H.S.; Kunze, S.; Scholten, F.; Roldan Cuenya, B. Prism-shaped Cu nanocatalysts for electrochemical CO2 reduction to ethylene. ACS Catal. 2018, 8, 531–535. [Google Scholar] [CrossRef]
- Hoang, T.T.H.; Verma, S.; Ma, S.; Fister, T.T.; Timoshenko, J.; Frenkel, A.I.; Kenis, P.J.A.; Gewirth, A.A. Nanoporous copper–silver alloys by additive-controlled electrodeposition for the selective electroreduction of CO2 to ethylene and ethanol. J. Am. Chem. Soc. 2018, 140, 5791–5797. [Google Scholar] [CrossRef]
- Zhuang, T.-T.; Liang, Z.-Q.; Seifitokaldani, A.; Li, Y.; De Luna, P.; Burdyny, T.; Che, F.; Meng, F.; Min, Y.; Quintero-Bermudez, R.; et al. Steering post-C–C coupling selectivity enables high efficiency electroreduction of carbon dioxide to multi-carbon alcohols. Nat. Catal. 2018, 1, 421–428. [Google Scholar] [CrossRef]
- Lv, J.-J.; Jouny, M.; Luc, W.; Zhu, W.; Zhu, J.-J.; Jiao, F. A highly porous copper electrocatalyst for carbon dioxide reduction. Adv. Mat. 2018, 30, 1803111. [Google Scholar] [CrossRef]
- Verma, S.; Hamasaki, Y.; Kim, C.; Huang, W.; Lu, S.; Jhong, H.-R.M.; Gewirth, A.A.; Fujigaya, T.; Nakashima, N.; Kenis, P.J.A. Insights into the low overpotential electroreduction of CO2 to CO on a supported gold catalyst in an alkaline flow electrolyzer. ACS Energy Lett. 2018, 3, 193–198. [Google Scholar] [CrossRef]
- Haas, T.; Krause, R.; Weber, R.; Demler, M.; Schmid, G. Technical photosynthesis involving CO2 electrolysis and fermentation. Nat. Catal. 2018, 1, 32–39. [Google Scholar] [CrossRef]
- Lu, X.; Leung, D.Y.C.; Wang, H.; Xuan, J. A high performance dual electrolyte microfluidic reactor for the utilization of CO2. Appl. Energy 2017, 194, 549–559. [Google Scholar] [CrossRef]
- Yang, H.; Kaczur, J.J.; Sajjad, S.D.; Masel, R.I. Electrochemical conversion of CO2 to formic acid utilizing Sustainion™ membranes. J. CO2 Util. 2017, 20, 208–217. [Google Scholar] [CrossRef]
- García de Arquer, F.P.; Bushuyev, O.S.; De Luna, P.; Dinh, C.-T.; Seifitokaldani, A.; Saidaminov, M.I.; Tan, C.-S.; Quan, L.N.; Proppe, A.; Kibria, M.G.; et al. 2D metal oxyhalide-derived catalysts for efficient CO2 electroreduction. Adv. Mater. 2018, 30, 1802858. [Google Scholar] [CrossRef] [PubMed]
- Sajjad, S.D.; Gao, Y.; Liu, Z.; Yang, H.; Masel, R. Tunable-high performance Sustainion™ anion exchange membranes for electrochemical applications. ECS Trans. 2017, 77, 1653–1656. [Google Scholar] [CrossRef]
- Lee, W.; Kim, Y.E.; Youn, M.H.; Jeong, S.K.; Park, K.T. Catholyte-free electrocatalytic CO2 reduction to formate. Angew. Chem. Int. Ed. 2018, 57, 6883–6887. [Google Scholar] [CrossRef]
- Li, J.; Chen, G.; Zhu, Y.; Liang, Z.; Pei, A.; Wu, C.-L.; Wang, H.; Lee, H.R.; Liu, K.; Chu, S.; et al. Efficient electrocatalytic CO2 reduction on a three-phase interface. Nat. Catal. 2018, 1, 592–600. [Google Scholar] [CrossRef]
- Jhong, H.-R.M.; Tornow, C.E.; Kim, C.; Verma, S.; Oberst, J.L.; Anderson, P.S.; Gewirth, A.A.; Fujigaya, T.; Nakashima, N.; Kenis, P.J.A. Gold Nanoparticles on polymer-wrapped carbon nanotubes: An Efficient and selective catalyst for the electroreduction of CO2. ChemPhysChem 2017, 18, 3274–3279. [Google Scholar] [CrossRef]
- Ma, S.; Liu, J.; Sasaki, K.; Lyth, S.M.; Kenis, P.J.A. Carbon foam decorated with silver nanoparticles for electrochemical CO2 conversion. Energy Technol. 2017, 5, 861–863. [Google Scholar] [CrossRef]
- Del Castillo, A.; Alvarez-Guerra, M.; Solla-Gullón, J.; Sáez, A.; Montiel, V.; Irabien, A. Sn nanoparticles on gas diffusion electrodes: Synthesis, characterization and use for continuous CO2 electroreduction to formate. J. CO2 Utiliz. 2017, 18, 222–228. [Google Scholar] [CrossRef]
- Kibria, M.G.; Dinh, C.-T.; Seifitokaldani, A.; De Luna, P.; Burdyny, T.; Quintero-Bermudez, R.; Ross, M.B.; Bushuyev, O.S.; García de Arquer, F.P.; Yang, P.; et al. A Surface reconstruction route to high productivity and selectivity in CO2 electroreduction toward C2+ hydrocarbons. Adv. Mat. 2018, 30, 1804867. [Google Scholar] [CrossRef]
- Jouny, M.; Luc, W.; Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 2018, 57, 2165–2177. [Google Scholar] [CrossRef]
- Chen, C.; Khosrowabadi Kotyk, J.F.; Sheehan, S.W. Progress toward commercial application of electrochemical carbon dioxide reduction. Chem 2018, 4, 2571–2586. [Google Scholar] [CrossRef]
- Marken, F.; Fermin, D. Electrochemical Reduction of Carbon Dioxide: Overcoming the Limitations of Photosynthesis; Marken, F., Fermin, D., Eds.; Royal Society of Chemistry: London, UK, 2018. [Google Scholar]
- Jhong, H.-R.M.; Ma, S.; Kenis, P.J.A. Electrochemical conversion of CO2 to useful chemicals: Current status, remaining challenges, and future opportunities. Curr. Opin. Chem. Eng. 2013, 2, 191–199. [Google Scholar] [CrossRef]
- Vennekoetter, J.-B.; Sengpiel, R.; Wessling, M. Beyond the catalyst: How electrode and reactor design determine the product spectrum during electrochemical CO2 reduction. Chem. Eng. J. 2019, 364, 89–101. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, S.; Barreras, F.; Pastor, E.; Lázaro, M.J. Electrochemical reactors for CO2 reduction: From acid media to gas phase. Int. J. Hydrogen Energy 2016, 41, 19756–19765. [Google Scholar] [CrossRef]
- Endrődi, B.; Bencsik, G.; Darvas, F.; Jones, R.; Rajeshwar, K.; Janáky, C. Continuous-flow electroreduction of carbon dioxide. Prog. Energy Combust. Sci. 2017, 62, 133–154. [Google Scholar] [CrossRef]
- Merino-Garcia, I.; Alvarez-Guerra, E.; Albo, J.; Irabien, A. Electrochemical membrane reactors for the utilisation of carbon dioxide. Chem. Eng. J. 2016, 305, 104–120. [Google Scholar] [CrossRef]
- Liang, S.; Altaf, N.; Huang, L.; Gao, Y.; Wang, Q. Electrolytic cell design for electrochemical CO2 reduction. J. CO2 Util. 2020, 35, 90–105. [Google Scholar] [CrossRef]
- Hori, Y.; Kikuchi, K.; Suzuki, S. Production of CO and CH4 in electrochemical reduction of CO2 at metal electrodes in aqueous hydrogencarbonate solution. Chem. Lett 1985, 14, 1695–1698. [Google Scholar] [CrossRef]
- Ahn, S.; Klyukin, K.; Wakeham, R.J.; Rudd, J.A.; Lewis, A.R.; Alexander, S.; Carla, F.; Alexandrov, V.; Andreoli, E. Poly-Amide modified copper foam electrodes for enhanced electrochemical reduction of carbon dioxide. ACS Catal. 2018, 8, 4132–4142. [Google Scholar] [CrossRef]
- Andreoli, E.; Annibaldi, V.; Rooney, D.A.; Breslin, C.B. Electrochemical fabrication of copper-based hybrid microstructures and mechanism of formation of related hierarchical structures on polypyrrole films. J. Phys. Chem. C 2011, 115, 20076–20083. [Google Scholar] [CrossRef]
- Kazimierska, E.; Andreoli, E.; Barron, A.R. Understanding the effect of carbon nanotube functionalization on copper electrodeposition. J. Appl. Electrochem. 2019, 49, 731–741. [Google Scholar] [CrossRef]
- Verma, S.; Lu, S.; Kenis, P.J.A. Co-electrolysis of CO2 and glycerol as a pathway to carbon chemicals with improved technoeconomics due to low electricity consumption. Nat. Energy 2019, 4, 466–474. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, S.; Zhu, X.; Yang, W. Electrochemical reduction of CO2 in solid oxide electrolysis cells. J. Energy Chem. 2017, 26, 593–601. [Google Scholar] [CrossRef]
- Küngas, R. Review—Electrochemical CO2 reduction for CO production: Comparison of low- and high-temperature electrolysis technologies. J. Electrochem. Soc. 2020, 167, 044508. [Google Scholar] [CrossRef]
- Kopljar, D.; Inan, A.; Vindayer, P.; Wagner, N.; Klemm, E. Electrochemical reduction of CO2 to formate at high current density using gas diffusion electrodes. J. Appl. Electrochem. 2014, 44, 1107–1116. [Google Scholar] [CrossRef]
- Kibria, M.G.; Edwards, J.P.; Gabardo, C.M.; Dinh, C.-T.; Seifitokaldani, A.; Sinton, D.; Sargent, E.H. Electrochemical CO2 reduction into chemical feedstocks: From mechanistic electrocatalysis models to system design. Adv. Mater. 2019, 31, 1807166. [Google Scholar] [CrossRef]
- Zhang, J.; Luo, W.; Züttel, A. Crossover of liquid products from electrochemical CO2 reduction through gas diffusion electrode and anion exchange membrane. J. Catal. 2020, 385, 140–145. [Google Scholar] [CrossRef]
- Möller, T.; Ju, W.; Bagger, A.; Wang, X.; Luo, F.; Ngo Thanh, T.; Varela, A.S.; Rossmeisl, J.; Strasser, P. Efficient CO2 to CO electrolysis on solid Ni–N–C catalysts at industrial current densities. Energy Environ. Sci. 2019, 12, 640–647. [Google Scholar] [CrossRef]
- Gabardo, C.M.; Seifitokaldani, A.; Edwards, J.P.; Dinh, C.-T.; Burdyny, T.; Kibria, M.G.; O’Brien, C.P.; Sargent, E.H.; Sinton, D. Combined high alkalinity and pressurization enable efficient CO2 electroreduction to CO. Energy Environ. Sci. 2018, 11, 2531–2539. [Google Scholar] [CrossRef]
- Jeanty, P.; Scherer, C.; Magori, E.; Wiesner-Fleischer, K.; Hinrichsen, O.; Fleischer, M. Upscaling and continuous operation of electrochemical CO2 to CO conversion in aqueous solutions on silver gas diffusion electrodes. J. CO2 Utiliz. 2018, 24, 454–462. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Z.; Lee, H.; Mao, J.; Grimes, C.A.; Liu, C.; Zhang, M.; Lu, Z.; Chen, Y.; Feng, S.P. Efficient electroreduction of CO2 to CO by Ag-decorated S-doped g-C3N4/CNT nanocomposites at industrial scale current density. Mater. Today Phys. 2020, 12, 100176. [Google Scholar] [CrossRef]
- Lu, X.; Wu, Y.; Yuan, X.; Huang, L.; Wu, Z.; Xuan, J.; Wang, Y.; Wang, H. High-performance electrochemical CO2 reduction cells based on non-noble metal catalysts. ACS Energy Lett. 2018, 3, 2527–2532. [Google Scholar] [CrossRef]
- Lu, X.; Jiang, Z.; Yuan, X.; Wu, Y.; Malpass-Evans, R.; Zhong, Y.; Liang, Y.; McKeown, N.B.; Wang, H. A bio-inspired O2-tolerant catalytic CO2 reduction electrode. Sci. Bull. 2019, 64, 1890–1895. [Google Scholar] [CrossRef]
- Irtem, E.; Andreu, T.; Parra, A.; Hernández-Alonso, M.D.; García-Rodríguez, S.; Riesco-García, J.M.; Penelas-Pérez, G.; Morante, J.R. Low-energy formate production from CO2 electroreduction using electrodeposited tin on GDE. J. Mat. Chem. A 2016, 4, 13582–13588. [Google Scholar] [CrossRef]
- Ávila-Bolívar, B.; García-Cruz, L.; Montiel, V.; Solla-Gullón, J. Electrochemical reduction of CO2 to formate on easily prepared carbon-supported bi nanoparticles. Molecules 2019, 24, 2032. [Google Scholar] [CrossRef]
- Hoang, T.T.H.; Ma, S.; Gold, J.I.; Kenis, P.J.A.; Gewirth, A.A. Nanoporous copper films by additive-controlled electrodeposition: CO2 reduction catalysis. ACS Catal. 2017, 7, 3313–3321. [Google Scholar] [CrossRef]
- Nam, D.-H.; Bushuyev, O.S.; Li, J.; De Luna, P.; Seifitokaldani, A.; Dinh, C.-T.; García de Arquer, F.P.; Wang, Y.; Liang, Z.; Proppe, A.H.; et al. Metal–organic frameworks mediate cu coordination for selective CO2 electroreduction. J. Am. Chem. Soc. 2018, 140, 11378–11386. [Google Scholar] [CrossRef]
- De Luna, P.; Quintero-Bermudez, R.; Dinh, C.-T.; Ross, M.B.; Bushuyev, O.S.; Todorović, P.; Regier, T.; Kelley, S.O.; Yang, P.; Sargent, E.H. Catalyst electro-redeposition controls morphology and oxidation state for selective carbon dioxide reduction. Nat. Catal. 2018, 1, 103–110. [Google Scholar] [CrossRef]
- De Gregorio, G.L.; Burdyny, T.; Loiudice, A.; Iyengar, P.; Smith, W.A.; Buonsanti, R. Facet-Dependent Selectivity of Cu catalysts in electrochemical CO2 reduction at commercially viable current densities. ACS Catal. 2020, 10, 4854–4862. [Google Scholar] [CrossRef]
- Albo, J.; Irabien, A. Cu2O-loaded gas diffusion electrodes for the continuous electrochemical reduction of CO2 to methanol. J. Catal. 2016, 343, 232–239. [Google Scholar] [CrossRef]
- Ren, D.; Gao, J.; Pan, L.; Wang, Z.; Luo, J.; Zakeeruddin, S.M.; Hagfeldt, A.; Grätzel, M. Atomic layer deposition of ZnO on CuO enables selective and efficient electroreduction of carbon dioxide to liquid fuels. Angew. Chem. Int. Ed. 2019, 131, 15178–15182. [Google Scholar] [CrossRef]
- Duarte, M.; De Mot, B.; Hereijgers, J.; Breugelmans, T. Electrochemical reduction of CO2: Effect of convective CO2 supply in gas diffusion electrodes. ChemElectroChem 2019, 6, 5596–5602. [Google Scholar] [CrossRef]
- Burchardt, T. An evaluation of electrocatalytic activity and stability for air electrodes. J. Power Sources 2004, 135, 192–197. [Google Scholar] [CrossRef]
- Nwabara, U.O.; Cofell, E.R.; Verma, S.; Negro, E.; Kenis, P.J.A. Durable cathodes and electrolyzers for the efficient aqueous electrochemical reduction of CO2. ChemSusChem 2020, 13, 855–875. [Google Scholar] [CrossRef]
- Oener, S.Z.; Ardo, S.; Boettcher, S.W. Ionic Processes in water electrolysis: The role of ion-selective membranes. ACS Energy Lett. 2017, 2, 2625–2634. [Google Scholar] [CrossRef]
- Aeshala, L.M.; Uppaluri, R.; Verma, A. Electrochemical conversion of CO2 to fuels: Tuning of the reaction zone using suitable functional groups in a solid polymer electrolyte. PCCP 2014, 16, 17588–17594. [Google Scholar] [CrossRef]
- Jiménez, C.; García, J.; Camarillo, R.; Martínez, F.; Rincón, J. Electrochemical CO2 reduction to fuels using Pt/CNT catalysts synthesized in supercritical medium. Energy Fuels 2017, 31, 3038–3046. [Google Scholar] [CrossRef]
- Narayanan, S.R.; Haines, B.; Soler, J.; Valdez, T.I. Electrochemical conversion of carbon dioxide to formate in alkaline polymer electrolyte membrane cells. J. Electrochem. Soc. 2011, 158, A167. [Google Scholar] [CrossRef]
- Salvatore, D.A.; Weekes, D.M.; He, J.; Dettelbach, K.E.; Li, Y.C.; Mallouk, T.E.; Berlinguette, C.P. Electrolysis of gaseous CO2 to CO in a flow cell with a bipolar membrane. ACS Energy Lett. 2018, 3, 149–154. [Google Scholar] [CrossRef]
- Kutz, R.B.; Chen, Q.; Yang, H.; Sajjad, S.D.; Liu, Z.; Masel, I.R. Sustainion imidazolium-functionalized polymers for carbon dioxide electrolysis. Energy Technol. 2017, 5, 929–936. [Google Scholar] [CrossRef]
- Ren, S.; Joulié, D.; Salvatore, D.; Torbensen, K.; Wang, M.; Robert, M.; Berlinguette, C.P. Molecular electrocatalysts can mediate fast, selective CO2 reduction in a flow cell. Science 2019, 365, 367. [Google Scholar] [CrossRef] [PubMed]
- Endrődi, B.; Kecsenovity, E.; Samu, A.; Darvas, F.; Jones, R.V.; Török, V.; Danyi, A.; Janáky, C. Multilayer electrolyzer stack converts carbon dioxide to gas products at high pressure with high efficiency. ACS Energy Lett. 2019, 4, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Hou, P.; Wang, X.; Wang, Z.; Li, W.; Kang, P. Carbon nanotubes with rich pyridinic nitrogen for gas phase CO2 electroreduction. Appl. Catal. B 2019, 250, 347–354. [Google Scholar] [CrossRef]
- Lee, J.; Lim, J.; Roh, C.-W.; Whang, H.S.; Lee, H. Electrochemical CO2 reduction using alkaline membrane electrode assembly on various metal electrodes. J. CO2 Util. 2019, 31, 244–250. [Google Scholar] [CrossRef]
- Jiang, K.; Siahrostami, S.; Zheng, T.; Hu, Y.; Hwang, S.; Stavitski, E.; Peng, Y.; Dynes, J.; Gangisetty, M.; Su, D.; et al. Isolated Ni single atoms in graphene nanosheets for high-performance CO2 reduction. Energy Environ. Sci. 2018, 11, 893–903. [Google Scholar] [CrossRef]
- Díaz-Sainz, G.; Alvarez-Guerra, M.; Solla-Gullón, J.; García-Cruz, L.; Montiel, V.; Irabien, A. Catalyst coated membrane electrodes for the gas phase CO2 electroreduction to formate. Cat. Today 2018. [Google Scholar] [CrossRef]
- Machunda, R.L.; Ju, H.; Lee, J. Electrocatalytic reduction of CO2 gas at Sn based gas diffusion electrode. Curr. Appl. Phys. 2011, 11, 986–988. [Google Scholar] [CrossRef]
- Lee, S.; Ju, H.; Machunda, R.; Uhm, S.; Lee, J.K.; Lee, H.J.; Lee, J. Sustainable production of formic acid by electrolytic reduction of gaseous carbon dioxide. J. Mat. Chem. A 2015, 3, 3029–3034. [Google Scholar] [CrossRef]
- Bitar, Z.; Fecant, A.; Trela-Baudot, E.; Chardon-Noblat, S.; Pasquier, D. Electrocatalytic reduction of carbon dioxide on indium coated gas diffusion electrodes—Comparison with indium foil. Appl. Catal. B 2016, 189, 172–180. [Google Scholar] [CrossRef]
- Machunda, R.L.; Lee, J.; Lee, J. Microstructural surface changes of electrodeposited Pb on gas diffusion electrode during electroreduction of gas-phase CO2. Surf. Interface Anal. 2010, 42, 564–567. [Google Scholar] [CrossRef]
- Merino-Garcia, I.; Albo, J.; Irabien, A. Tailoring gas-phase CO2 electroreduction selectivity to hydrocarbons at Cu nanoparticles. Nanotechnology 2017, 29, 014001. [Google Scholar] [CrossRef] [PubMed]
- Birdja, Y.Y.; Vos, R.E.; Wezendonk, T.A.; Jiang, L.; Kapteijn, F.; Koper, M.T.M. Effects of substrate and polymer encapsulation on CO2 electroreduction by immobilized indium(iii) protoporphyrin. ACS Catal. 2018, 8, 4420–4428. [Google Scholar] [CrossRef] [PubMed]
- Whipple, D.T.; Finke, E.C.; Kenis, P.J.A. Microfluidic reactor for the electrochemical reduction of carbon dioxide: The effect of pH. Electrochem. Solid State Lett. 2010, 13, B109. [Google Scholar] [CrossRef]
- Ma, S.; Sadakiyo, M.; Luo, R.; Heima, M.; Yamauchi, M.; Kenis, P.J.A. One-step electrosynthesis of ethylene and ethanol from CO2 in an alkaline electrolyzer. J. Power Sources 2016, 301, 219–228. [Google Scholar] [CrossRef]
- Hereijgers, J.; Ottevaere, H.; Breugelmans, T.; De Malsche, W. Membrane deflection in a flat membrane microcontactor: Experimental study of spacer features. J. Membr. Sci. 2016, 504, 153–161. [Google Scholar] [CrossRef]
- Verma, S.; Lu, X.; Ma, S.; Masel, R.I.; Kenis, P.J.A. The effect of electrolyte composition on the electroreduction of CO2 to CO on Ag based gas diffusion electrodes. PCCP 2016, 18, 7075–7084. [Google Scholar] [CrossRef]
- Jhong, H.-R.M.; Tornow, C.E.; Smid, B.; Gewirth, A.A.; Lyth, S.M.; Kenis, P.J.A. A nitrogen-doped carbon catalyst for electrochemical CO2 conversion to CO with high selectivity and current density. ChemSusChem 2017, 10, 1094–1099. [Google Scholar] [CrossRef]
- Ma, S.; Luo, R.; Gold, J.I.; Yu, A.Z.; Kim, B.; Kenis, P.J.A. Carbon nanotube containing Ag catalyst layers for efficient and selective reduction of carbon dioxide. J. Mater. Chem. A 2016, 4, 8573–8578. [Google Scholar] [CrossRef]
- Liang, C.; Kim, B.; Yang, S.; Yang, L.; Francisco Woellner, C.; Li, Z.; Vajtai, R.; Yang, W.; Wu, J.; Kenis, P.J.A.; et al. High efficiency electrochemical reduction of CO2 beyond the two-electron transfer pathway on grain boundary rich ultra-small SnO2 nanoparticles. J. Mat. Chem. A 2018, 6, 10313–10319. [Google Scholar] [CrossRef]
- Lu, X.; Leung, D.Y.C.; Wang, H.; Maroto-Valer, M.M.; Xuan, J. A pH-differential dual-electrolyte microfluidic electrochemical cells for CO2 utilization. Renew. Energy 2016, 95, 277–285. [Google Scholar] [CrossRef]
- Wu, J.; Ma, S.; Sun, J.; Gold, J.I.; Tiwary, C.; Kim, B.; Zhu, L.; Chopra, N.; Odeh, I.N.; Vajtai, R.; et al. A metal-free electrocatalyst for carbon dioxide reduction to multi-carbon hydrocarbons and oxygenates. Nat. Commun. 2016, 7, 13869. [Google Scholar] [CrossRef] [PubMed]
- Paidar, M.; Fateev, V.; Bouzek, K. Membrane electrolysis—History, current status and perspective. Electrochim. Acta 2016, 209, 737–756. [Google Scholar] [CrossRef]
- Greenblatt, J.B.; Miller, D.J.; Ager, J.W.; Houle, F.A.; Sharp, I.D. The technical and energetic challenges of separating (photo) electrochemical carbon dioxide reduction products. Joule 2018, 2, 381–420. [Google Scholar] [CrossRef]
- Ghosh, S.; Garapati, M.S.; Ghosh, A.; Sundara, R. Nonprecious catalyst for three-phase contact in a proton exchange membrane CO2 conversion full cell for efficient electrochemical reduction of carbon dioxide. ACS Appl. Mater. Interfaces 2019, 11, 40432–40442. [Google Scholar] [CrossRef]
- Wu, J.; Sharma, P.P.; Harris, B.H.; Zhou, X.-D. Electrochemical reduction of carbon dioxide: IV dependence of the Faradaic efficiency and current density on the microstructure and thickness of tin electrode. J. Power Sources 2014, 258, 189–194. [Google Scholar] [CrossRef]
- Yamamoto, T.; Tryka, D.A.; Hashimoto, K.; Fujishima, A.; Okawa, M. Electrochemical reduction of CO2 in micropores. In Studies in Surface Science and Catalysis; Inui, T., Anpo, M., Izui, K., Yanagida, S., Yamaguchi, T., Eds.; Elsevier: Amsterdam, The Netherlands, 1998; Volume 114, pp. 585–588. [Google Scholar]
- Weng, L.-C.; Bell, A.T.; Weber, A.Z. Modeling gas-diffusion electrodes for CO2 reduction. PCCP 2018, 20, 16973–16984. [Google Scholar] [CrossRef]
- Singh, M.R.; Kwon, Y.; Lum, Y.; Ager, J.W.; Bell, A.T. Hydrolysis of Electrolyte cations enhances the electrochemical reduction of CO2 over Ag and Cu. J. Am. Chem. Soc. 2016, 138, 13006–13012. [Google Scholar] [CrossRef]
- Resasco, J.; Chen, L.D.; Clark, E.; Tsai, C.; Hahn, C.; Jaramillo, T.F.; Chan, K.; Bell, A.T. Promoter effects of alkali metal cations on the electrochemical reduction of carbon dioxide. J. Am. Chem. Soc. 2017, 139, 11277–11287. [Google Scholar] [CrossRef]
- Leonard, M.E.; Clarke, L.E.; Forner-Cuenca, A.; Brown, S.M.; Brushett, F.R. Investigating electrode flooding in a flowing electrolyte, gas-fed carbon dioxide electrolyzer. ChemSusChem 2020, 13, 400–411. [Google Scholar] [CrossRef]
- Bidault, F.; Brett, D.J.L.; Middleton, P.H.; Brandon, N.P. Review of gas diffusion cathodes for alkaline fuel cells. J. Power Sources 2009, 187, 39–48. [Google Scholar] [CrossRef]
- Owejan, J.P.; Owejan, J.E.; Gu, W.; Trabold, T.A.; Tighe, T.W.; Mathias, M.F. Water transport mechanisms in PEMFC gas diffusion layers. J. Electrochem. Soc. 2010, 157, B1456. [Google Scholar] [CrossRef]
- Gostick, J.T.; Ioannidis, M.A.; Fowler, M.W.; Pritzker, M.D. On the role of the microporous layer in PEMFC operation. Electrochem. Commun. 2009, 11, 576–579. [Google Scholar] [CrossRef]
- Garg, S.; Li, M.; Weber, A.Z.; Ge, L.; Li, L.; Rudolph, V.; Wang, G.; Rufford, T.E. Advances and challenges in electrochemical CO2 reduction processes: An engineering and design perspective looking beyond new catalyst materials. J. Mater. Chem. A 2020, 8, 1511–1544. [Google Scholar] [CrossRef]
- Ikeda, S.; Ito, T.; Azuma, K.; Ito, K.; Noda, H. Electrochemical mass reduction of carbon dioxide using cu-loaded gas diffusion electrodes I. Preparation of electrode and reduction products. Denki Kagaku 1995, 63, 303–309. [Google Scholar] [CrossRef]
- Li, A.; Wang, H.; Han, J.; Liu, L. Preparation of a Pb loaded gas diffusion electrode and its application to CO2 electroreduction. Front. Chem. Sci. Eng. 2012, 6, 381–388. [Google Scholar] [CrossRef]
- Katsounaros, I.; Ipsakis, D.; Polatides, C.; Kyriacou, G. Efficient electrochemical reduction of nitrate to nitrogen on tin cathode at very high cathodic potentials. Electrochim. Acta 2006, 52, 1329–1338. [Google Scholar] [CrossRef]
- Bard, A.J.; Parsons, R.; Jordan, J. International Union of Pure and Applied Chemistry. In Standard Potentials in Aqueous Solution; M. Dekker: New York, NY, USA, 1985. [Google Scholar]
- Ikemiya, N.; Natsui, K.; Nakata, K.; Einaga, Y. Long-term continuous conversion of CO2 to formic acid using boron-doped diamond electrodes. ACS Sustain. Chem. Eng. 2018, 6, 8108–8112. [Google Scholar] [CrossRef]
- Guilminot, E.; Corcella, A.; Chatenet, M.; Maillard, F.; Charlot, F.; Berthomé, G.; Iojoiu, C.; Sanchez, J.Y.; Rossinot, E.; Claude, E. Membrane and active layer degradation upon PEMFC steady-state operation. J. Electrochem. Soc. 2007, 154, B1106. [Google Scholar] [CrossRef]
- Park, J.; Oh, H.; Ha, T.; Lee, Y.I.; Min, K. A review of the gas diffusion layer in proton exchange membrane fuel cells: Durability and degradation. Appl. Energy 2015, 155, 866–880. [Google Scholar] [CrossRef]
- Kang, J.; Kim, J. Membrane electrode assembly degradation by dry/wet gas on a PEM fuel cell. Int. J. Hydrogen Energy 2010, 35, 13125–13130. [Google Scholar] [CrossRef]
- Keith, D.W.; Holmes, G.; Angelo, D.S.; Heidel, K. A process for capturing CO2 from the atmosphere. J. Joule 2018, 2, 1573–1594. [Google Scholar] [CrossRef]
- Schulz, K.G.; Riebesell, U.; Rost, B.; Thoms, S.; Zeebe, R.E. Determination of the rate constants for the carbon dioxide to bicarbonate inter-conversion in pH-buffered seawater systems. Mar. Chem. 2006, 100, 53–65. [Google Scholar] [CrossRef]
- Rumble, J.R. CRC Handbook of Chemistry and Physics; CRC Press: London, UK, 2017. [Google Scholar]
- Dinh, C.-T.; García de Arquer, F.P.; Sinton, D.; Sargent, E.H. High Rate, selective, and stable electroreduction of CO2 to CO in basic and neutral media. ACS Energy Lett. 2018, 3, 2835–2840. [Google Scholar] [CrossRef]
- Todorova, T.K.; Schreiber, M.W.; Fontecave, M. Mechanistic Understanding of CO2 reduction reaction (CO2RR) toward multicarbon products by heterogeneous copper-based catalysts. ACS Catal. 2020, 10, 1754–1768. [Google Scholar] [CrossRef]
- Kim, B.; Ma, S.; Molly Jhong, H.-R.; Kenis, P.J.A. Influence of dilute feed and pH on electrochemical reduction of CO2 to CO on Ag in a continuous flow electrolyzer. Electrochim. Acta 2015, 166, 271–276. [Google Scholar] [CrossRef]
- De Mot, B.; Hereijgers, J.; Duarte, M.; Breugelmans, T. Influence of flow and pressure distribution inside a gas diffusion electrode on the performance of a flow-by CO2 electrolyzer. Chem. Eng. J. 2019, 378, 122224. [Google Scholar] [CrossRef]
- Beni, G.; Hackwood, S. Electro-wetting displays. Appl. Phys. Lett. 1981, 38, 207–209. [Google Scholar] [CrossRef]
- Santamaria, A.D.; Das, P.K.; MacDonald, J.C.; Weber, A.Z. Liquid-water interactions with gas-diffusion-layer surfaces. J. Electrochem. Soc. 2014, 161, F1184–F1193. [Google Scholar] [CrossRef]
- Weng, L.-C.; Bell, A.T.; Weber, A.Z. Towards membrane-electrode assembly systems for CO2 reduction: A modeling study. Energy Environ. Sci. 2019, 12, 1950–1968. [Google Scholar] [CrossRef]
- Torbensen, K.; Joulié, D.; Ren, S.; Wang, M.; Salvatore, D.; Berlinguette, C.P.; Robert, M. Molecular catalysts boost the rate of electrolytic CO2 reduction. ACS Energy Lett. 2020, 5, 1512–1518. [Google Scholar] [CrossRef]
- Duan, Y.; Luebke, D.R.; Pennline, H.W.; Li, B.; Janik, M.J.; Halley, J.W. Ab Initio thermodynamic study of the CO2 capture properties of potassium carbonate sesquihydrate, K2CO3·1.5H2O. J. Phys. Chem. C 2012, 116, 14461–14470. [Google Scholar] [CrossRef]
- Veran-Tissoires, S.; Marcoux, M.; Prat, M. Discrete salt crystallization at the surface of a porous medium. Phys. Rev. Lett. 2012, 108, 054502. [Google Scholar] [CrossRef] [PubMed]
- Shahidzadeh-Bonn, N.; Desarnaud, J.; Bertrand, F.; Chateau, X.; Bonn, D. Damage in porous media due to salt crystallization. Phys. Rev. E 2010, 81, 066110. [Google Scholar] [CrossRef]
- Shapoval, G.S.; Pud, A.A.; Zamotayev, P.V.; Kachan, A.A. Degradation of some carbon-chain polymers by electrochemical reduction. Polym. Sci. (USSR) 1985, 27, 2427–2431. [Google Scholar] [CrossRef]
- Rolla, A.; Sadkowski, A.; Wild, J.; Zóltowski, P. On the problem of ageing of carbon—Air electrodes in alkaline electrolytes. J. Power Sources 1980, 5, 189–196. [Google Scholar] [CrossRef]
- Lamminen, J. Preparation of Air Electrodes and Long Run Tests. J. Electrochem. Soc. 1991, 138, 905. [Google Scholar] [CrossRef]
- Al-Saleh, M.A.; Gültekin, S.; Al-Zakri, A.S.; Celiker, H. Effect of carbon dioxide on the performance of Ni/PTFE and Ag/PTFE electrodes in an alkaline fuel cell. J. Appl. Electrochem. 1994, 24, 575–580. [Google Scholar] [CrossRef]
- Naughton, M.S.; Brushett, F.R.; Kenis, P.J.A. Carbonate resilience of flowing electrolyte-based alkaline fuel cells. J. Power Sources 2011, 196, 1762–1768. [Google Scholar] [CrossRef]
- Perry, S.C.; Gateman, S.M.; Malpass-Evans, R.; McKeown, N.; Wegener, M.; Nazarovs, P.; Mauzeroll, J.; Wang, L.; Ponce de León, C. Polymers with intrinsic microporosity (PIMs) for targeted CO2 reduction to ethylene. Chemosphere 2020, 248, 125993. [Google Scholar] [CrossRef]
- Sánchez, O.G.; Birdja, Y.Y.; Bulut, M.; Vaes, J.; Breugelmans, T.; Pant, D. Recent advances in industrial CO2 electroreduction. Curr. Opin. Green Sustain. Chem. 2019, 16, 47–56. [Google Scholar] [CrossRef]
- Edwards, J.P.; Xu, Y.; Gabardo, C.M.; Dinh, C.-T.; Li, J.; Qi, Z.; Ozden, A.; Sargent, E.H.; Sinton, D. Efficient electrocatalytic conversion of carbon dioxide in a low-resistance pressurized alkaline electrolyzer. Appl. Energy 2020, 261, 114305. [Google Scholar] [CrossRef]
- Gabardo, C.M.; O’Brien, C.P.; Edwards, J.P.; McCallum, C.; Xu, Y.; Dinh, C.-T.; Li, J.; Sargent, E.H.; Sinton, D. Continuous carbon dioxide electroreduction to concentrated multi-carbon products using a membrane electrode assembly. Joule 2019, 3, 2777–2791. [Google Scholar] [CrossRef]
- García de Arquer, F.P.; Dinh, C.-T.; Ozden, A.; Wicks, J.; McCallum, C.; Kirmani, A.R.; Nam, D.-H.; Gabardo, C.; Seifitokaldani, A.; Wang, X.; et al. CO2 electrolysis to multicarbon products at activities greater than 1 A cm−2. Science 2020, 367, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Z.; Dinh, C.-T.; Li, J.; Ozden, A.; Golam Kibria, M.; Seifitokaldani, A.; Tan, C.-S.; Gabardo, C.M.; Luo, M.; et al. Catalyst synthesis under CO2 electroreduction favours faceting and promotes renewable fuels electrosynthesis. Nat. Catal. 2020, 3, 98–106. [Google Scholar] [CrossRef]
- Wang, X.; Xu, A.; Li, F.; Hung, S.-F.; Nam, D.-H.; Gabardo, C.M.; Wang, Z.; Xu, Y.; Ozden, A.; Rasouli, A.S.; et al. Efficient methane electrosynthesis enabled by tuning local CO2 availability. J. Am. Chem. Soc. 2020, 142, 3525–3531. [Google Scholar] [CrossRef]
- Zhang, J.; Luo, W.; Züttel, A. Self-supported copper-based gas diffusion electrodes for CO2 electrochemical reduction. J. Mater. Chem. A 2019, 7, 26285–26292. [Google Scholar] [CrossRef]
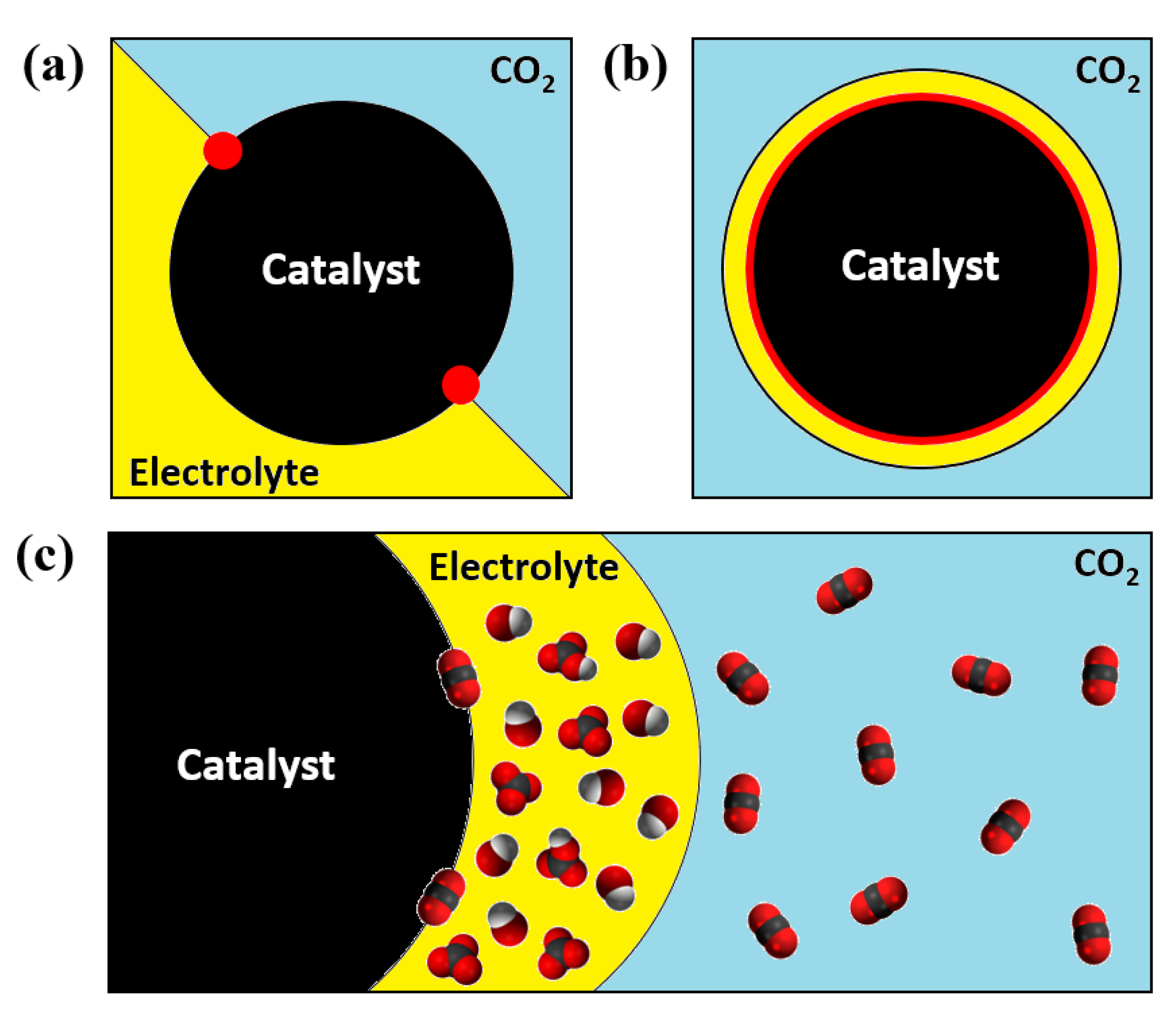
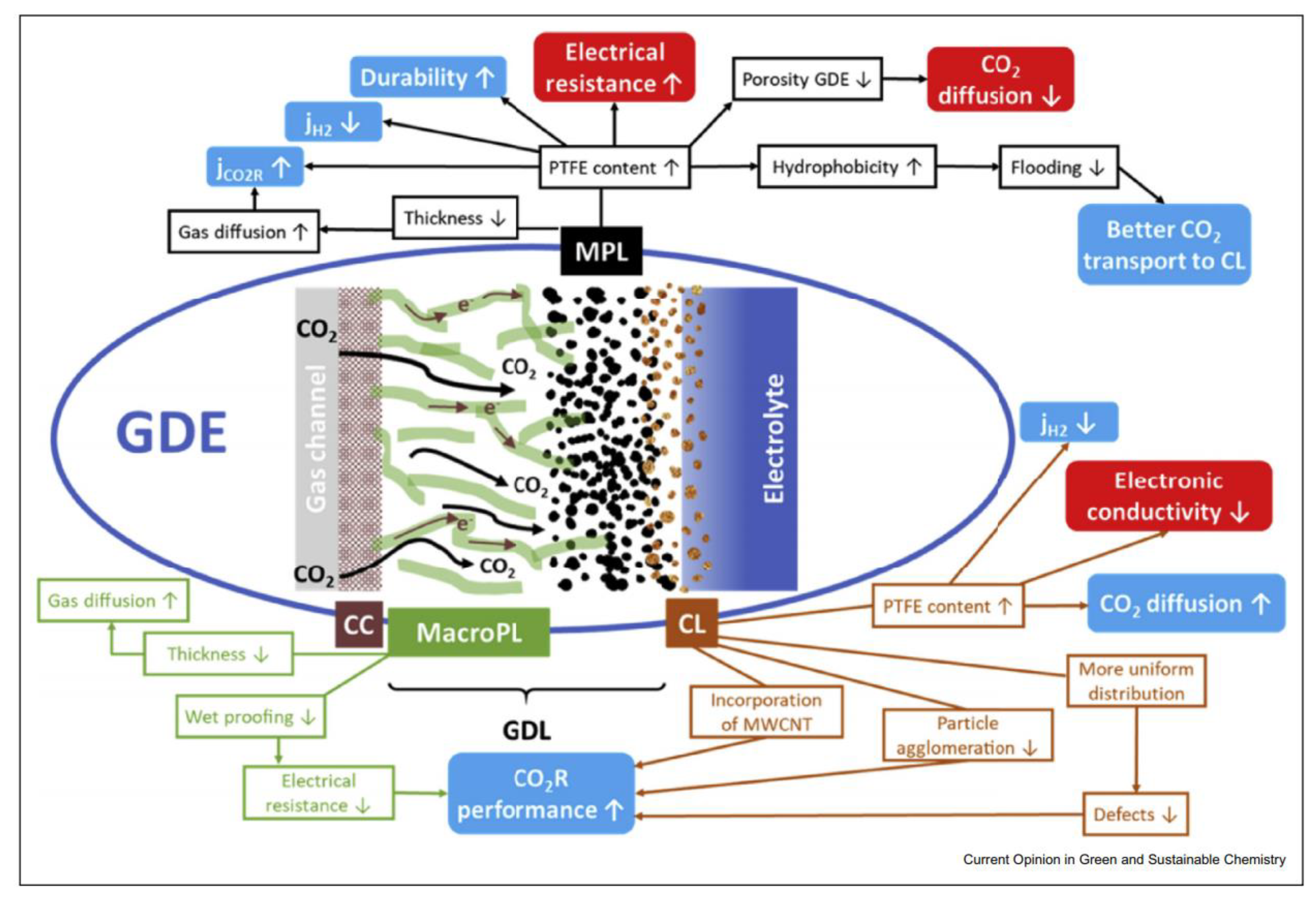
| Main Product | Catalyst | Membrane Type | GDL Type | FE (%) | Current Density (mA/cm2) | Stability | Reference |
|---|---|---|---|---|---|---|---|
| CO | Ni-N-C | AEM (Selemion AMV) | Freudenberg C2 | ~85 | 700 | 20 h | [119] |
| CO | Ag | AEM (FAA-3PK-130) | Sigracet 39BC | >42 | 300 | 10 h | [120] |
| CO | Ag | CEM (Nafion 117) | Covestro | ~60 | 150 | 600 h | [121] |
| CO | Ag-S-C3N4/CNT | CEM (Nafion) | Hesen HCP120 | >80 | ~300 | 24 h | [122] |
| CO | CoPc-CN/CNT | AEM (Selemion DSV) | Toray, TGPH-120 | 94 | 33 | 10 h | [123] |
| CO | CoOx/CNT | AEM (Selemion DSV) | Toray, TGPH-120 | ~76 | 27.3 | 18 h | [124] |
| Formate | Sn | CEM (Nafion 117) | Sigracet GDL 35BC | ~90 | 200 | 5 h | [116] |
| Formate | Sn | CEM (Nafion 117) | Toray TGP-H-60 | 71 | 8.13 | 6 h | [125] |
| Formate | Sn | CEM (Nafion 117) | Toray, TGPH-90 | ~70 | 200 | n.a. | [98] |
| Formate | Bi | CEM (Nafion 112) | Toray, TGPH-120 | >93 | 10 | 70 h | [126] |
| Formate | SnO2/CNT | AEM (Selemion DSV) | Toray, TGPH-120 | 83 | 236 | 10 h | [123] |
| Ethylene | CuDAT-wire | AEM (FAP-375-PP) | Sigracet 35 BC | 40 | ~90 | 8 h | [127] |
| Ethylene | Cu | AEM | Freudenberg | ~60 | 400 | n.a. | [99] |
| Ethylene | Cu-MOF | AEM | Sigracet | 45 | 262 | 140 min | [128] |
| Ethylene | Cu (ERD) | AEM | Freudenberg | 38 | 450 | 1 h | [129] |
| Ethylene | Cu | AEM (FAB-PK-130) | Sigracet BC39 | ~60 | 300 | 6 h | [130] |
| Methanol | Cu2O/ZnO | CEM (Nafion 117) | Toray, TGP-H-60 | 27.5 | 10 | 20 h | [131] |
| Ethanol | Cu2S/Cu-V | AEM | n.a. | ~25 | 400 | 150 min | [86] |
| Ethanol | CuZn | AEM (FKS-50) | Sigracet 38 BC | ~40 | 200 | 10 h | [132] |
| Ethanol | CuDAT-wire | AEM (Fumatech FAP-375-PP) | Sigracet 35 BC | ~20 | ~80 | 8 h | [127] |
| Main Product | Catalyst | Membrane Type | FE (%) | Current Density (mA/cm2) | Stability | Reference |
|---|---|---|---|---|---|---|
| CO | Ag | BPM (Fumatech) | 50 | 200 | 24 h | [140] |
| CO | Ag | AEM (Sustanion) | >90 | 200 | 4380 h | [141] |
| CO | CoPc | AEM (Sustanion) | >95 | 175 | 8 h | [142] |
| CO | Ag | AEM (Sustanion) | ~95 | >250 | 8 h | [143] |
| CO | NCNTs | CEM (Nafion 117) | >94.5 | 22 | 40 h | [144] |
| CO | Pb | AEM (Sustanion) | 96.7 | 202 | 10 h | [145] |
| CO | Ni GNS | AEM (PSMIM) | 97 | 50 | 20 h | [146] |
| Formate | Sn | AEM (Sustanion) | 94 | 140 | 550 h | [91] |
| Formate | Sn | CEM (Nafion 117) | ~50 | 45 | n.a. | [147] |
| Formate | Sn | CEM (Nafion 115) | 93.3 | 41.5 | 48 h | [94] |
| Formate | Sn | CEM (Nafion 117) | 18 | 2 | 1 h | [148] |
| Formate | Sn | CEM (Nafion 115) | >5 | 5 | 10 h | [149] |
| Formate | In | CEM (Nafion 117) | 45 | 6.2 | ~8 h | [150] |
| Formate | Pb | CEM (Nafion 117) | 65 | 46 | 1 h | [151] |
| Ethylene | Cu | CEM (Nafion 117) | 92.8 | 7.5 | 45 min | [152] |
| Ethane | Cu2O | AEM (QPEI/PVA/KOH) | 17 | 4.81 | 25 min | [137] |
| Main Product | Catalyst | Membrane Type | GDL Type | FE (%) | Current Density (mA/cm2) | Stability | Reference |
|---|---|---|---|---|---|---|---|
| CO | Ag | n.a. | Sigracet 35 BC | >97 | ~340 | n.a. | [157] |
| CO | Au/PyPBI/MWNT | n.a. | Sigracet 35 BC | ~90 | ~60 | 26 h | [96] |
| CO | Ag/Carbon Foam | n.a. | Sigracet 35 BC | 84 | ~50 | n.a. | [97] |
| CO | CN/MWNT | n.a. | Sigracet 35 BC | 98 | ~90 | Several hours | [158] |
| CO | Ag/MWNT | n.a. | Sigracet 35 BC | 95 | 350 | 7 min | [159] |
| CO | Au/PyPBI/MWNT | AEM (FAA-3-PK-75) | Sigracet 35 BC | ~98 | 158 | 8 h | [88] |
| Formate | Sn2O | AEM (Fumatech) | Sigracet 35 BC | 64 | 145 | n.a. | [160] |
| Formate | BiOBr | n.a. | Freudenberg | 90 | 200 | 65 h | [92] |
| Formate | Pb | n.a. | Hesen, HCP120 | 95.6 | 143 | n.a. | [161] |
| Ethylene | NGQD | n.a. | Sigracet 35 BC | 31 | ~100 | n.a. | [162] |
| Ethylene | CuAg wire | AEM (FAP-375-PP) | Sigracet 35 BC | ~60 | ~300 | n.a. | [85] |
| Ethylene | Cu | AEM | Sigracet 35 BC | ~46 | ~200 | 4 h | [155] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernandez-Aldave, S.; Andreoli, E. Fundamentals of Gas Diffusion Electrodes and Electrolysers for Carbon Dioxide Utilisation: Challenges and Opportunities. Catalysts 2020, 10, 713. https://doi.org/10.3390/catal10060713
Hernandez-Aldave S, Andreoli E. Fundamentals of Gas Diffusion Electrodes and Electrolysers for Carbon Dioxide Utilisation: Challenges and Opportunities. Catalysts. 2020; 10(6):713. https://doi.org/10.3390/catal10060713
Chicago/Turabian StyleHernandez-Aldave, Sandra, and Enrico Andreoli. 2020. "Fundamentals of Gas Diffusion Electrodes and Electrolysers for Carbon Dioxide Utilisation: Challenges and Opportunities" Catalysts 10, no. 6: 713. https://doi.org/10.3390/catal10060713
APA StyleHernandez-Aldave, S., & Andreoli, E. (2020). Fundamentals of Gas Diffusion Electrodes and Electrolysers for Carbon Dioxide Utilisation: Challenges and Opportunities. Catalysts, 10(6), 713. https://doi.org/10.3390/catal10060713






