A Three-Step Process for the Bioconversion of Whey Permeate into a Glucose-Free D-Tagatose Syrup
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Composition of Whey Permeate
2.2. Hydrolysis of Whey Permeate by β-Galactosidase from Bifidobacterium bifidus
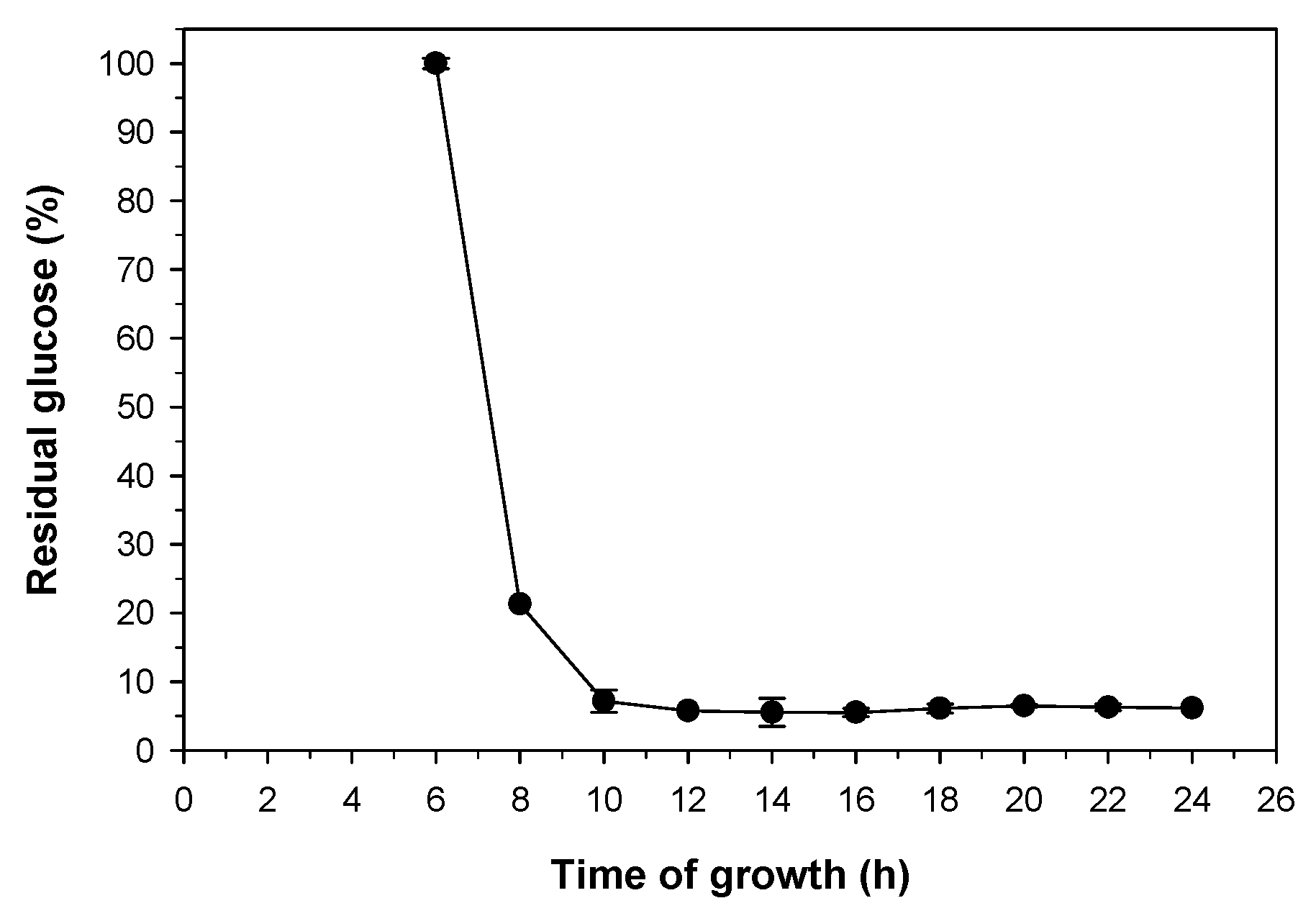
2.3. Study of the Growth Time and Concentration of Pichia pastoris for Elimination of D-Glucose
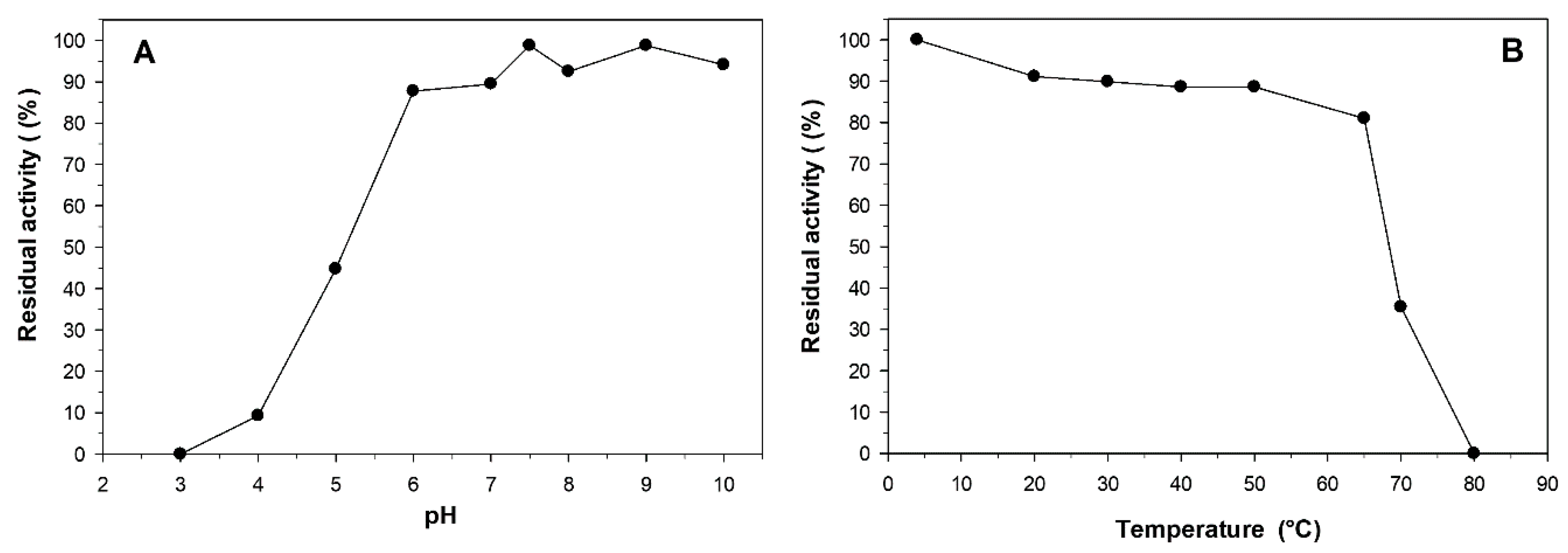
2.4. Effect of pH and Temperature on the Stability of L-Arabinose Isomerase
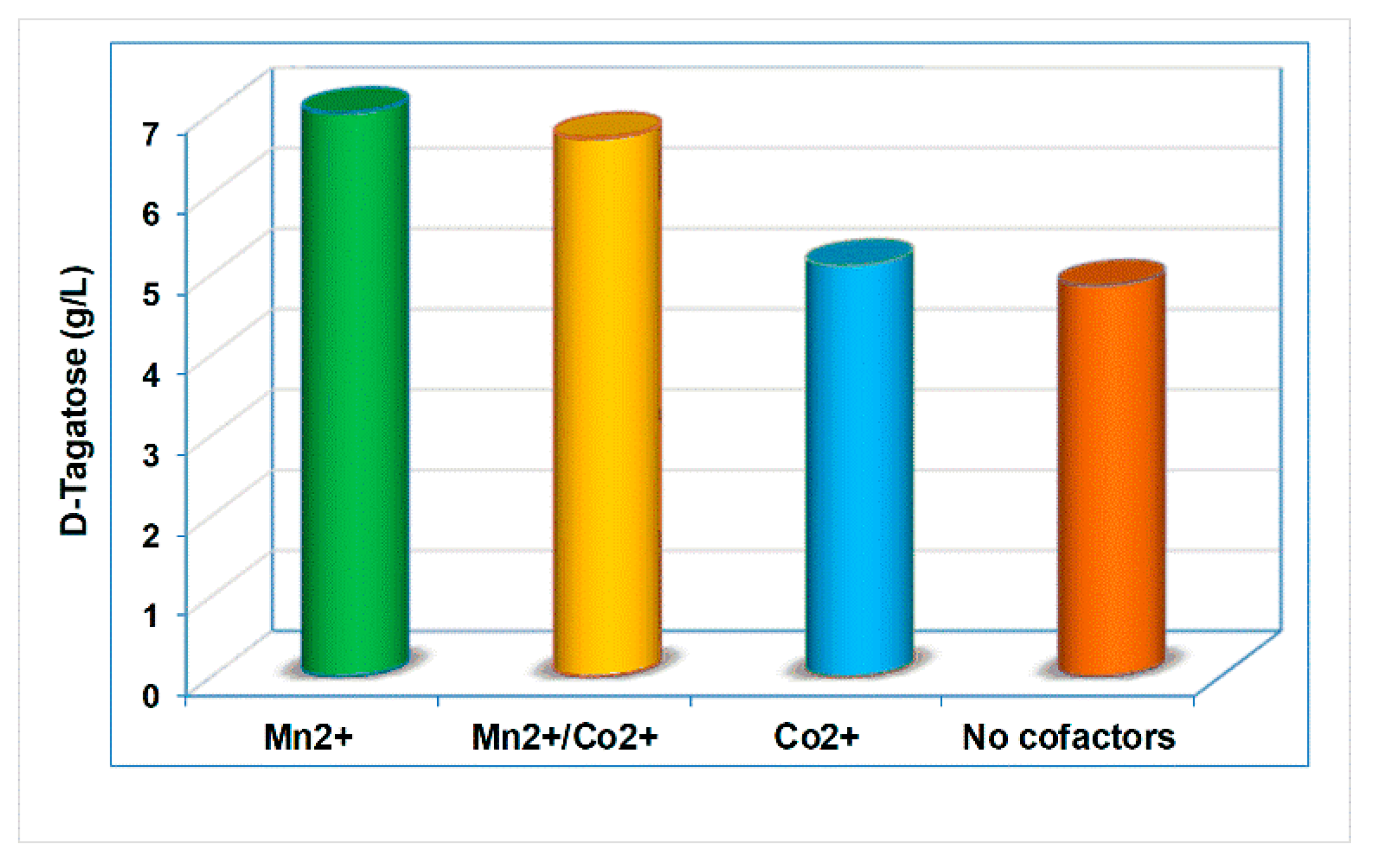
2.5. Effect of Cofactors, pH and Temperature on the Activity of L-Arabinose Isomerase
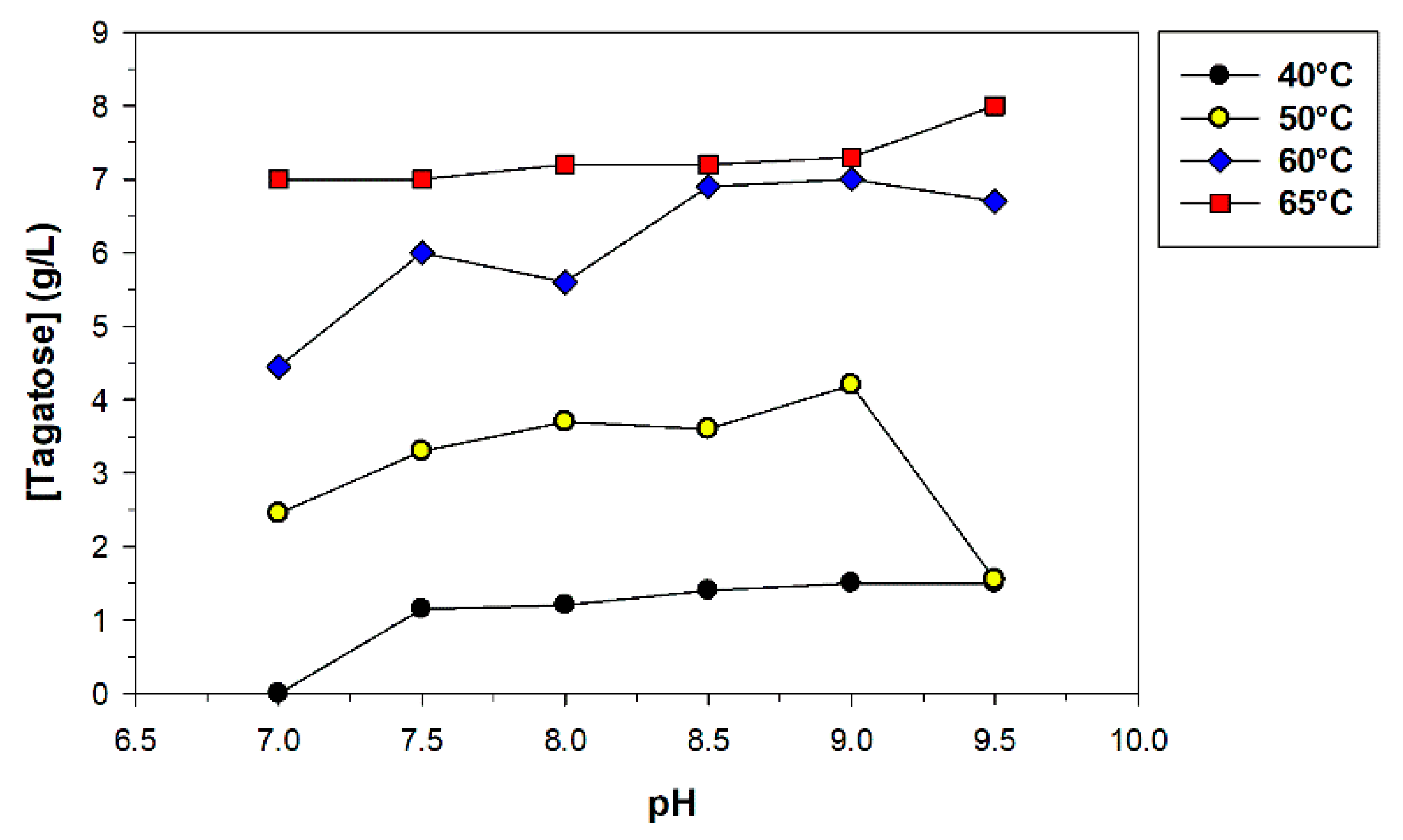
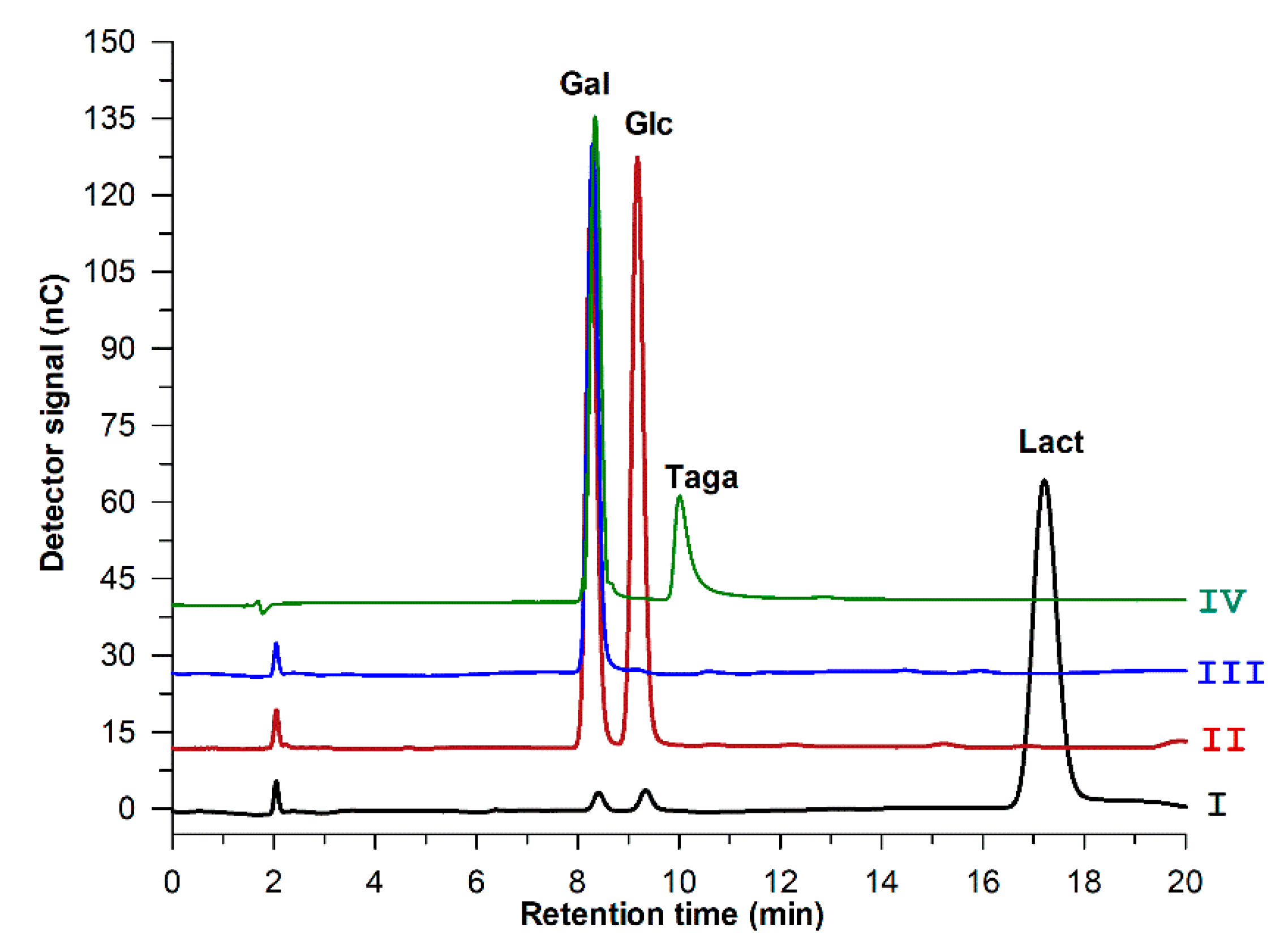
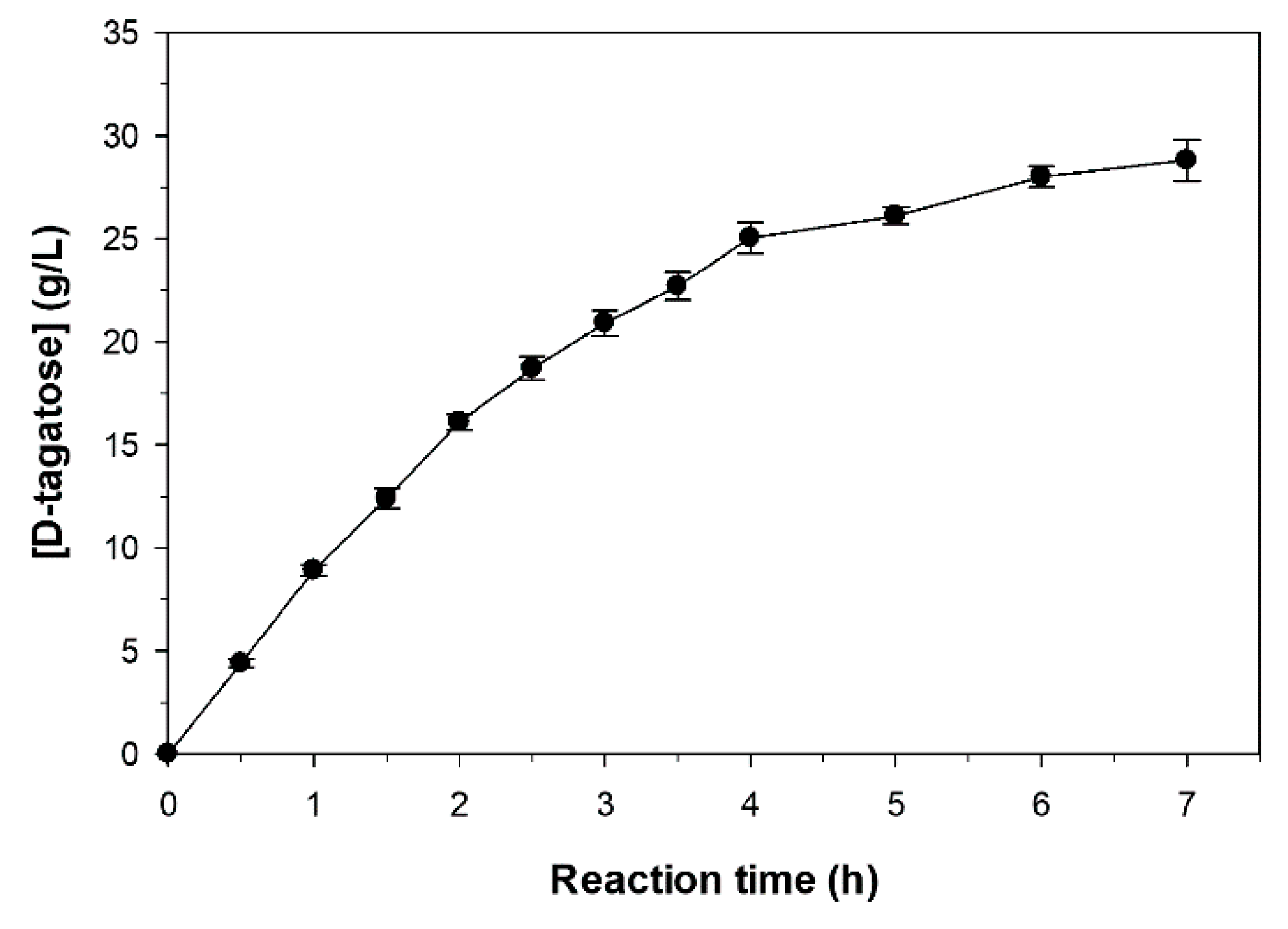
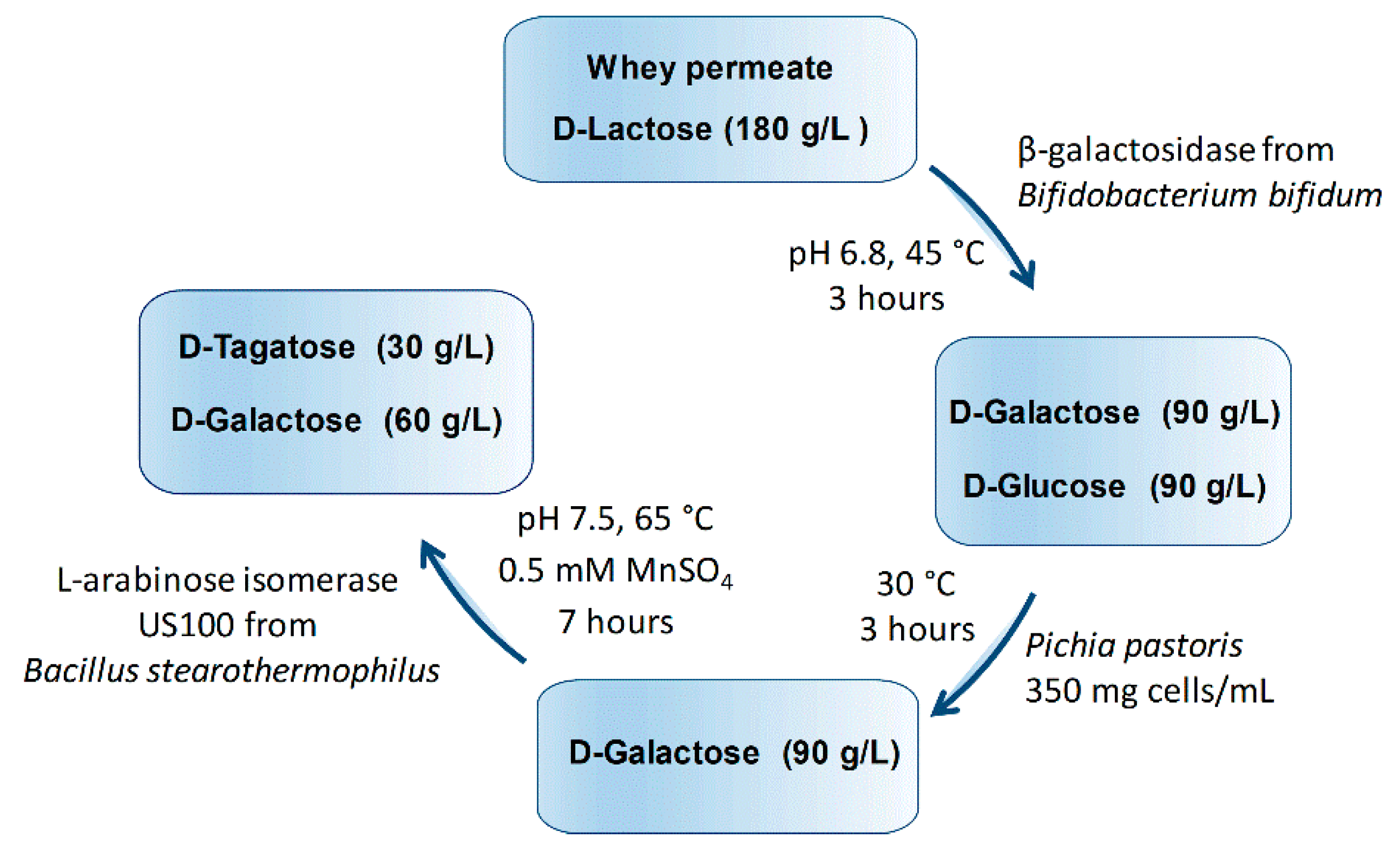
2.6. Sequential Biotransformation of Whey Permeate into D-Tagatose
3. Materials and Methods
3.1. Enzymes and Reagents
3.2. Elemental Analysis of Whey Permeate using Inductive Coupled Plasma Mass Spectrometry
3.3. β-Galactosidase Activity Assay
3.4. Lactose Hydrolysis in Whey by Bifidobacterium Bifidum β-Galactosidase
3.5. Effect of Growth Time and Concentration of Pichia pastoris on the Elimination of D-Glucose
3.6. L-Arabinose Isomerase Activity Assay
3.7. Stability of L-Arabinose Isomerase
3.8. Effect of Metals, pH and Temperature on the Activity of L-Arabinose Isomerase
3.9. Sequential Biotransformation of Whey Permeate into D-Tagatose
3.10. HPAEC-PAD Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Torrico, D.D.; Tam, J.; Fuentes, S.; Viejo, C.G.; Dunshea, F.R. D-tagatose as a sucrose substitute and its effect on the physico-chemical properties and acceptability of strawberry-flavored yogurt. Foods 2019, 8, 256. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.K. Tagatose: Properties, applications, and biotechnological processes. Appl. Microbiol. Biotechnol. 2007, 76, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Von Rymon Lipinski, G.-W. Sweeteners. In Biotechnology of Food and Feed Additives; Zorn, H., Czermak, P., Eds.; Springer: Berlin, Germany, 2014; pp. 1–28. [Google Scholar] [CrossRef]
- Acevedo, W.; Capitaine, C.; Rodríguez, R.; Araya-Durán, I.; González-Nilo, F.; Pérez-Correa, J.R.; Agosin, E. Selecting optimal mixtures of natural sweeteners for carbonated soft drinks through multi-objective decision modeling and sensory validation. J. Sens. Stud. 2018, 33, 6. [Google Scholar] [CrossRef]
- Levin, G.V. Tagatose, the new GRAS sweetener and health product. J. Med. Food 2002, 5, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Chikkerur, J.; Roy, S.C.; Dhali, A.; Kolte, A.P.; Sridhar, M.; Samanta, A.K. Tagatose as a potential nutraceutical: Production, properties, biological roles, and applications. J. Food Sci. 2018, 83, 2699–2709. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Arraez, S.; Ferrer, C.; Capella, J.V.; Ortolá, M.D.; Castelló, M.L. Development of lemon marmalade formulated with new sweeteners (isomaltulose and tagatose): Effect on antioxidant, rheological and optical properties. J. Food Process Eng. 2017, 40, e12371. [Google Scholar] [CrossRef]
- Manzo, R.M.; Antunes, A.S.L.M.; de Sousa Mendes, J.; Hissa, D.C.; Gonҫalves, L.R.B.; Mammarella, E.J. Biochemical characterization of heat-tolerant recombinant L-arabinose isomerase from Enterococcus faecium DBFIQ E36 strain with feasible applications in D-tagatose production. Mol. Biotechnol. 2019, 61, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.X.; Wen, P.; Wang, Y.; Ouyang, D.M.; Wang, D.; Chen, Y.Z.; Song, Y.; Deng, J.; Sun, Y.M.; Wang, H. The constipation-relieving property of D-tagatose by modulating the composition of gut microbiota. Int. J. Mol. Sci. 2019, 20, 5721. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, I.; Fogelfeld, L. Tagatose: From a sweetener to a new diabetic medication? Expert Opin. Investig. Drugs 2010, 19, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Levin, G.V.; Donner, T.W. Tagatose, a new antidiabetic and obesity control drug. Diabetes Obes. Metab. 2008, 10, 109–134. [Google Scholar] [CrossRef] [PubMed]
- Nagata, Y.; Mizuta, N.; Kanasaki, A.; Tanaka, K. Rare sugars, D-allulose, D-tagatose and D-sorbose, differently modulate lipid metabolism in rats. J. Sci. Food Agric. 2018, 98, 2020–2026. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Metzger, L.E.; Martínez-Monteagudo, S.I. One-pot synthesis of sweetening syrup from lactose. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Drabo, P.; Delidovich, I. Catalytic isomerization of galactose into tagatose in the presence of bases and Lewis acids. Catal. Commun. 2018, 107, 24–28. [Google Scholar] [CrossRef]
- Freimund, S.; Huwig, A.; Giffhorn, F.; Köpper, S. Convenient chemo-enzymatic synthesis of D-tagatose. J. Carbohydr. Chem. 1996, 15, 115–120. [Google Scholar] [CrossRef]
- Sha, F.; Zheng, Y.; Chen, J.; Chen, K.; Cao, F.; Yan, M.; Ouyang, P. D-Tagatose manufacture through bio-oxidation of galactitol derived from waste xylose mother liquor. Green Chem. 2018, 20, 2382–2391. [Google Scholar] [CrossRef]
- Bober, J.R.; Nair, N.U. Galactose to tagatose isomerization at moderate temperatures with high conversion and productivity. Nat. Commun. 2019, 10, 4548. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.J.; Patel, A.T.; Akhani, R.; Dedania, S.; Patel, D.H. Bioproduction of D-tagatose from D-galactose using phosphoglucose isomerase from Pseudomonas aeruginosa PAO1. Appl. Biochem. Biotechnol. 2016, 179, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, S.S.; Singh, R.; Kang, Y.C.; Zhao, H.; Lee, J.K. Cloning and characterization of a galactitol 2-dehydrogenase from Rhizobium legumenosarum and its application in D-tagatose production. Enzyme Microb. Technol. 2014, 58–59, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Boudebbouze, S.; Maguin, E.; Rhimi, M. Bacterial L-arabinose isomerases: Industrial application for D-tagatose production. Recent Pat. DNA Gene Seq. 2011, 5, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Chouayekh, H.; Bejar, W.; Rhimi, M.; Jelleli, K.; Mseddi, M.; Bejar, S. Characterization of an L-arabinose isomerase from the Lactobacillus plantarum NC8 strain showing pronounced stability at acidic pH. FEMS Microbiol. Lett. 2007, 277, 260–267. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, M.; Melo, V.M.M.; Hissa, D.C.; Manzo, R.M.; Mammarella, E.J.; Antunes, A.S.L.M.; García, J.L.; Pessela, B.C.; Gonçalves, L.R.B. One-step immobilization and stabilization of a recombinant Enterococcus faecium DBFIQ E36 L-arabinose isomerase for D-tagatose synthesis. Appl. Biochem. Biotechnol. 2019, 188, 310–325. [Google Scholar] [CrossRef] [PubMed]
- Du, M.; Zhao, D.; Cheng, S.; Sun, D.; Chen, M.; Gao, Z.; Zhang, C. Towards efficient enzymatic conversion of D-galactose to D-tagatose: Purification and characterization of L-arabinose isomerase from Lactobacillus brevis. Bioprocess Biosyst. Eng. 2019, 42, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Mei, W.; Wang, L.; Zang, Y.; Zheng, Z.; Ouyang, J. Characterization of an L-arabinose isomerase from Bacillus coagulans NL01 and its application for D-tagatose production. BMC Biotechnol. 2016, 16, 55. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.K.; Hong, M.G.; Chang, P.S.; Lee, B.H.; Yoo, S.H. Biochemical properties of L-arabinose isomerase from Clostridium hylemonae to produce D-tagatose as a functional sweetener. PLoS ONE 2018, 13, e0196099. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Mei, W.; Xia, M.; He, Q.; Ouyang, J. Rational DESIGN of Bacillus coagulans NL01 L-arabinose isomerase and use of its F279I variant in D-tagatose production. J. Agric. Food Chem. 2017, 65, 4715–4721. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, S.; Xu, H.; Wu, L.; Xu, Z.; Liu, J.; Feng, X. Efficient production of D-tagatose using a food-grade surface display system. J. Agric. Food Chem. 2014, 62, 6756–6762. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; An, Y.; Yun, J.; Yang, M.; Magocha, T.A.; Zhu, J.; Xue, Y.; Qi, Y.; Hossain, Z.; Sun, W.; et al. Enhanced D-tagatose production by spore surface-displayed L-arabinose isomerase from isolated Lactobacillus brevis PC16 and biotransformation. Bioresour. Technol. 2018, 247, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.W.; Hyeon, J.E.; Shin, S.K.; Han, S.O. Trienzymatic complex system for isomerization of agar-derived D-galactose into D-tagatose as a low-calorie sweetener. J. Agric. Food Chem. 2020, 68, 3195–3202. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zabed, H.M.; Yun, J.; Yuan, J.; Zhang, Y.; Wang, Y.; Qi, X. Two-stage biosynthesis of D-tagatose from milk whey powder by an engineered Escherichia coli strain expressing L-arabinose isomerase from Lactobacillus plantarum. Bioresour. Technol. 2020, 305, 123010. [Google Scholar] [CrossRef] [PubMed]
- Illanés, A. Whey upgrading by enzyme biocatalysis. Electron. J. Biotechnol. 2011, 14, 9. [Google Scholar] [CrossRef]
- Zheng, Z.; Xie, J.; Liu, P.; Li, X.; Ouyang, J. Elegant and efficient biotransformation for dual production of D-tagatose and bioethanol from cheese whey powder. J. Agric. Food Chem. 2019, 67, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Chen, J.; Jensen, P.R.; Solem, C. Sweet as sugar—Efficient conversion of lactose into sweet sugars using a novel whole-cell catalyst. J. Agric. Food Chem. 2019, 67, 6257–6262. [Google Scholar] [CrossRef] [PubMed]
- Torres, P.; Batista-Viera, F. Production of D-tagatose and D-fructose from whey by co-immobilized enzymatic system. Mol. Catal. 2019, 463, 99–109. [Google Scholar] [CrossRef]
- Nath, A.; Verasztó, B.; Basak, S.; Koris, A.; Kovács, Z.; Vatai, G. Synthesis of lactose-derived nutraceuticals from dairy waste whey—A review. Food Bioprocess Technol. 2016, 9, 16–48. [Google Scholar] [CrossRef]
- Jayamuthunagai, J.; Srisowmeya, G.; Chakravarthy, M.; Gautam, P. D-Tagatose production by permeabilized and immobilized Lactobacillus plantarum using whey permeate. Bioresour. Technol. 2017, 235, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Wanarska, M.; Kur, J. A method for the production of D-tagatose using a recombinant Pichia pastoris strain secreting β-D-galactosidase from Arthrobacter chlorophenolicus and a recombinant L-arabinose isomerase from Arthrobacter sp. 22c. Microb. Cell Fact. 2012, 11, 113. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Xu, Z.; Tang, B.; Li, S.; Gao, J.; Chi, B.; Xu, H. Construction and co-expression of polycistronic plasmids encoding thermophilic L-arabinose isomerase and hyperthermophilic β-galactosidase for single-step production of D-tagatose. Biochem. Eng. J. 2016, 109, 28–34. [Google Scholar] [CrossRef]
- Zhan, Y.; Xu, Z.; Li, S.; Liu, X.; Xu, L.; Feng, X.; Xu, H. Coexpression of β-D-galactosidase and L-arabinose isomerase in the production of D-tagatose: A functional sweetener. J. Agric. Food Chem. 2014, 62, 2412–2417. [Google Scholar] [CrossRef] [PubMed]
- Plou, F.J.; Polaina, J.; Sanz-Aparicio, J.; Fernandez-Lobato, M. β-Galactosidases for lactose hydrolysis and galactooligosaccharide synthesis. In Microbial Enzyme Technology in Food Applications; Ray, R.C., Rosell, C.M., Eds.; CRC Press: Boca Raton, FL, USA, 2016; pp. 123–146. [Google Scholar]
- Jørgensen, F.; Hansen, O.C.; Stougaard, P. Enzymatic conversion of D-galactose to D-tagatose: Heterologous expression and characterisation of a thermostable L-arabinose isomerase from Thermoanaerobacter mathranii. Appl. Microbiol. Biotechnol. 2004, 64, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Santibáñez, L.; Fernández-Arrojo, L.; Guerrero, C.; Plou, F.J.; Illanes, A. Removal of lactose in crude galacto-oligosaccharides by β-galactosidase from Kluyveromyces lactis. J. Mol. Catal. B Enzym. 2016, 133, 85–91. [Google Scholar] [CrossRef]
- Füreder, V.; Rodriguez-Colinas, B.; Cervantes, F.; Fernandez-Arrojo, L.; Poveda, A.; Jimenez-Barbero, J.; Ballesteros, A.O.; Plou, F.J. Selective synthesis of galactooligosaccharides containing β(1→3) linkages with β-galactosidase from Bifidobacterium bifidum (Saphera). J. Agric. Food Chem. 2020, 68, 4930–4938. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.A.; Lee, S.L.; Chou, C.C. Enzymatic production of galactooligosaccharides by beta-galactosidase from Bifidobacterium longum BCRC 15708. J. Agric. Food Chem. 2007, 55, 2225–2230. [Google Scholar] [CrossRef] [PubMed]
- Goulas, A.; Tzortzis, G.; Gibson, G.R. Development of a process for the production and purification of alpha- and beta-galactooligosaccharides from Bifobacterium bifidum NCIMB 41171. Int. Dairy J. 2007, 17, 648–656. [Google Scholar] [CrossRef]
- Mattanovich, D.; Graf, A.; Stadlmann, J.; Dragosits, M.; Redl, A.; Maurer, M.; Kleinheinz, M.; Sauer, M.; Altmann, F.; Gasser, B. Genome, secretome and glucose transport highlight unique features of the protein production host Pichia pastoris. Microb. Cell Fact. 2009, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Ávila-Fernández, Á.; Galicia-Lagunas, N.; Rodríguez-Alegría, M.E.; Olvera, C.; López-Munguía, A. Production of functional oligosaccharides through limited acid hydrolysis of agave fructans. Food Chem. 2011, 129, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Potvin, G.; Zhang, Z.; Defela, A.; Lam, H. Screening of alternative carbon sources for recombinant protein production in Pichia pastoris. Int. J. Chem. React. Eng. 2016, 14, 251–257. [Google Scholar] [CrossRef]
- Rhimi, M.; Bejar, S. Cloning, purification and biochemical characterization of metallic-ions independent and thermoactive L-arabinose isomerase from the Bacillus stearothermophilus US100 strain. Biochim. Biophys. Acta Gen. Subj. 2006, 1760, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Rhimi, M.; Aghajari, N.; Juy, M.; Chouayekh, H.; Maguin, E.; Haser, R.; Bejar, S. Rational design of Bacillus stearothermophilus US100 L-arabinose isomerase: Potential applications for D-tagatose production. Biochimie 2009, 91, 650–653. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-J.; Lee, D.-W.; Choe, E.-A.; Hong, Y.-H.; Kim, S.-B.; Kim, B.-C.; Pyun, Y.-R. Characterization of a Characterization of a thermoacidophilic L-arabinose isomerase from Alicyclobacillus acidocaldarius: Role of Lys-269 in pH optimum. Appl. Environ. Microbiol. 2005, 71, 7888. [Google Scholar] [CrossRef] [PubMed]
- Rhimi, M.; Juy, M.; Aghajari, N.; Haser, R.; Bejar, S. Probing the essential catalytic residues and substrate affinity in the thermoactive Bacillus stearothermophilus US100 L-arabinose isomerase by site-directed mutagenesis. J. Bacteriol. 2007, 189, 3556–3563. [Google Scholar] [CrossRef] [PubMed]
- Rhimi, M.; Messaoud, E.B.; Borgi, M.A.; khadra, K.B.; Bejar, S. Co-expression of L-arabinose isomerase and D-glucose isomerase in E. coli and development of an efficient process producing simultaneously D-tagatose and D-fructose. Enzyme Microb. Technol. 2007, 40, 1531–1537. [Google Scholar] [CrossRef]
- Zuluaga, J.; Rodríguez, N.; Rivas-Ramirez, I.; De La Fuente, V.; Rufo, L.; Amils, R. An improved semiquantitative method for elemental analysis of plants using inductive coupled plasma mass spectrometry. Biol. Trace Elem. Res. 2011, 144, 1302–1317. [Google Scholar] [CrossRef] [PubMed]
- Britton, H.T.S.; Robinson, R.A. Universal buffer solutions and the dissociation constant of veronal. J. Chem. Soc. 1931, 1456–1462. [Google Scholar] [CrossRef]

| Analyte | Concentration (ppm) |
|---|---|
| Na | 390 |
| Mg | 98 |
| K | 1492 |
| Ca | 829 |
| Ti | 0.35 |
| Fe | 0.18 |
| Zn | 1.1 |
| Rb | 1.6 |
| Sr | 0.45 |
| Mo | 0.1 |
| Temperature (°C) | Residual lactose (%) a | |||||
|---|---|---|---|---|---|---|
| 30 min | 60 min | 90 min | 120 min | 150 min | 180 min | |
| 30 | 89.3 | 81.5 | 76.6 | 65.5 | 57.1 | 24.7 |
| 40 | 73.4 | 45.6 | 39.7 | 36.9 | 21.3 | 18.7 |
| 45 | 76.3 | 31.7 | 29.8 | 26.1 | 10.7 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cervantes, F.V.; Neifar, S.; Merdzo, Z.; Viña-Gonzalez, J.; Fernandez-Arrojo, L.; Ballesteros, A.O.; Fernandez-Lobato, M.; Bejar, S.; Plou, F.J. A Three-Step Process for the Bioconversion of Whey Permeate into a Glucose-Free D-Tagatose Syrup. Catalysts 2020, 10, 647. https://doi.org/10.3390/catal10060647
Cervantes FV, Neifar S, Merdzo Z, Viña-Gonzalez J, Fernandez-Arrojo L, Ballesteros AO, Fernandez-Lobato M, Bejar S, Plou FJ. A Three-Step Process for the Bioconversion of Whey Permeate into a Glucose-Free D-Tagatose Syrup. Catalysts. 2020; 10(6):647. https://doi.org/10.3390/catal10060647
Chicago/Turabian StyleCervantes, Fadia V., Sawssan Neifar, Zoran Merdzo, Javier Viña-Gonzalez, Lucia Fernandez-Arrojo, Antonio O. Ballesteros, Maria Fernandez-Lobato, Samir Bejar, and Francisco J. Plou. 2020. "A Three-Step Process for the Bioconversion of Whey Permeate into a Glucose-Free D-Tagatose Syrup" Catalysts 10, no. 6: 647. https://doi.org/10.3390/catal10060647
APA StyleCervantes, F. V., Neifar, S., Merdzo, Z., Viña-Gonzalez, J., Fernandez-Arrojo, L., Ballesteros, A. O., Fernandez-Lobato, M., Bejar, S., & Plou, F. J. (2020). A Three-Step Process for the Bioconversion of Whey Permeate into a Glucose-Free D-Tagatose Syrup. Catalysts, 10(6), 647. https://doi.org/10.3390/catal10060647







