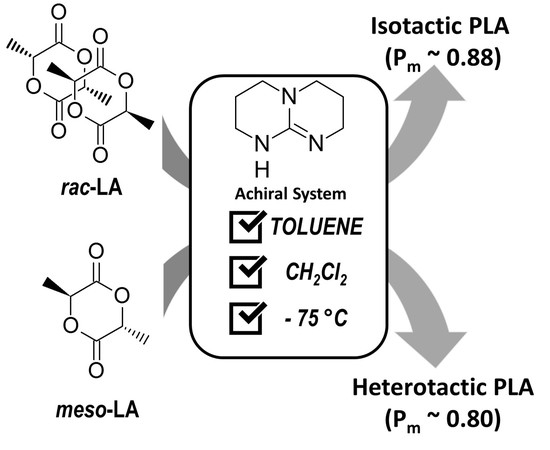
Stereoselective ROP of rac- and meso-Lactides Using Achiral TBD as Catalyst
Abstract
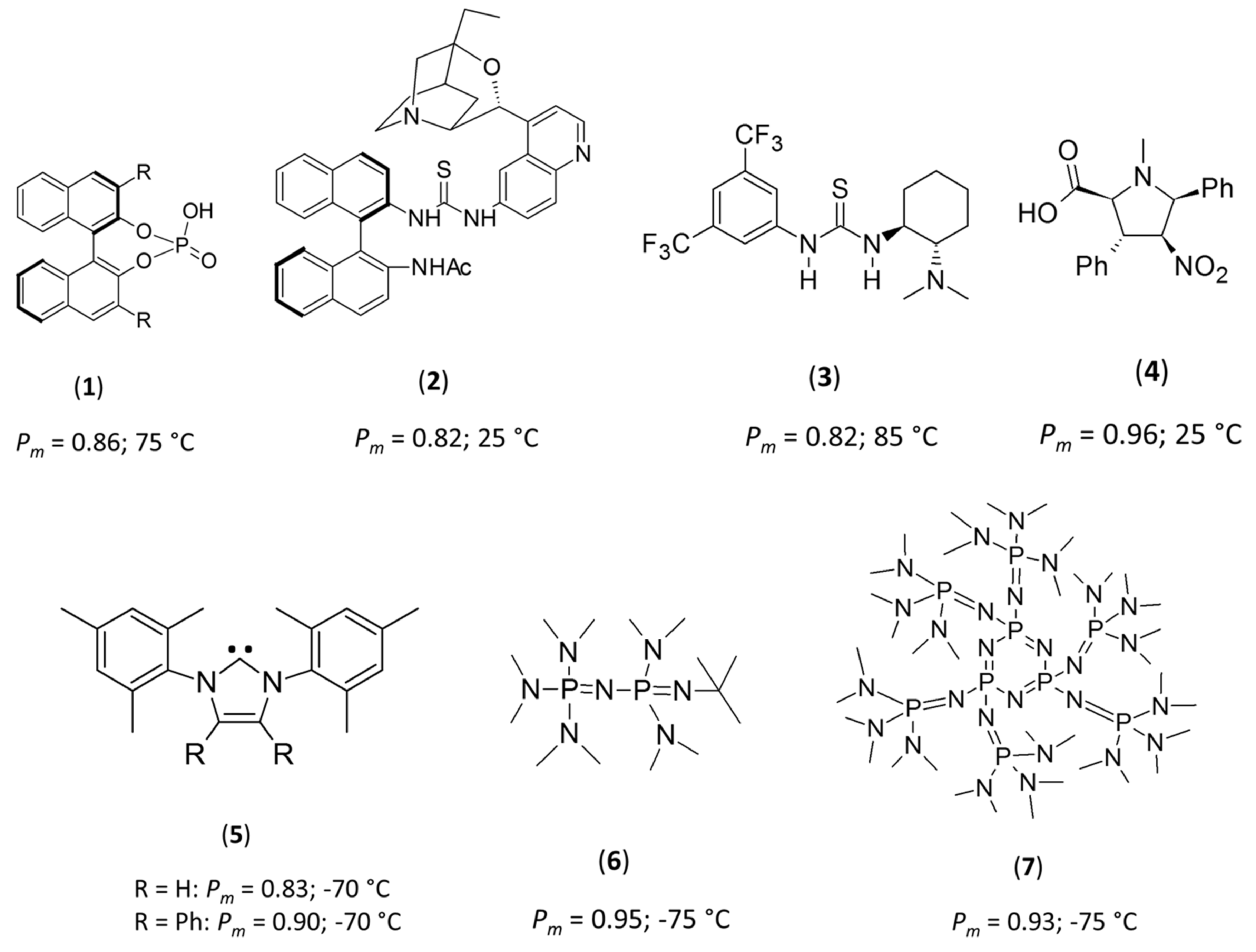
1. Introduction
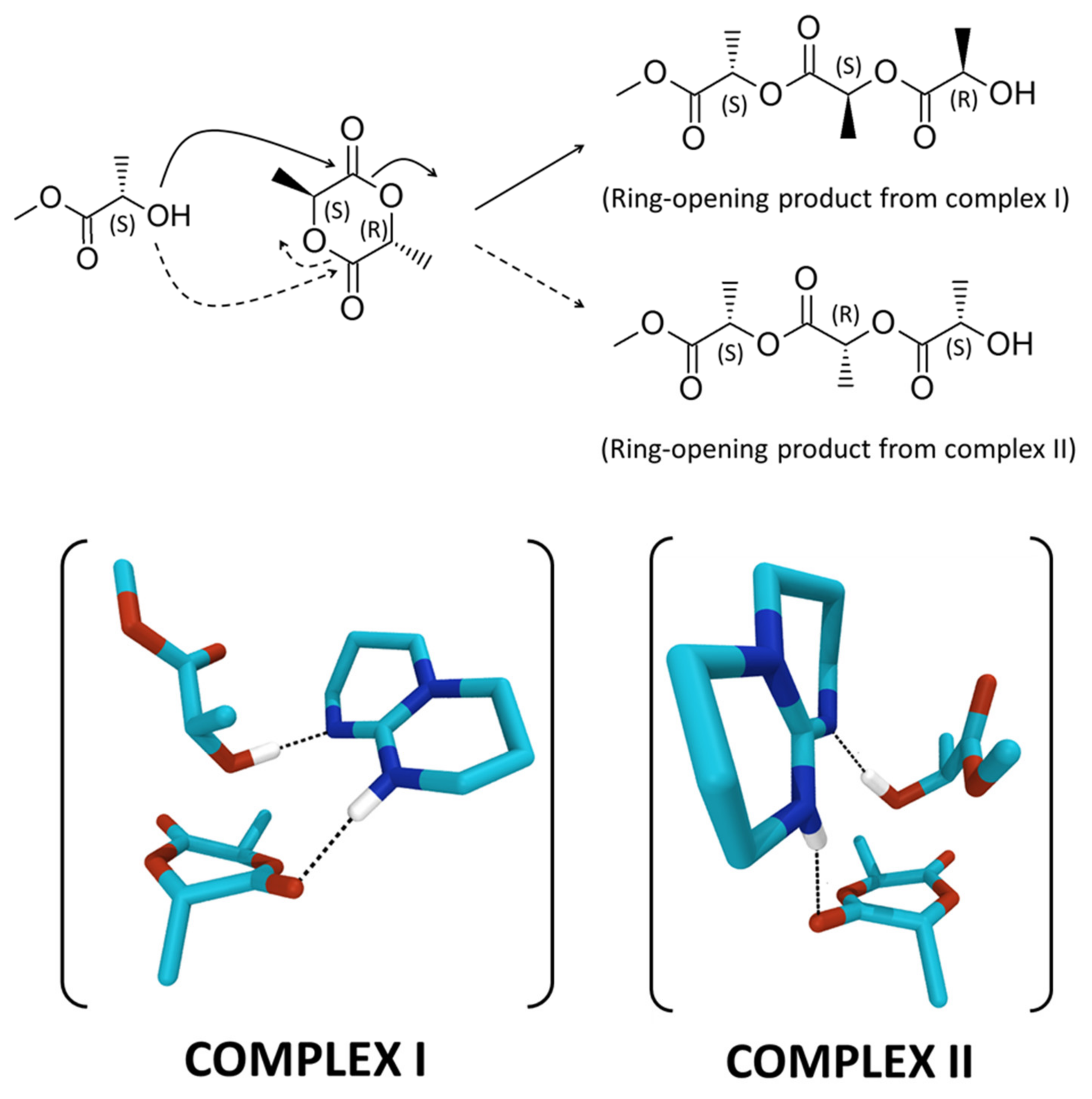
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Characterization Techniques
3.3. Protocol of Rac-LA Polymerization
3.4. Theoretical Investigations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Okada, M. Chemical Synthesis of Biodegradable Polymers. Prog. Polym. Sci. 2002, 27, 87–133. [Google Scholar] [CrossRef]
- Thomas, C.M. Stereocontrolled Ring-Opening Polymerization of Cyclic Esters: Synthesis of New Polyester Microstructures. Chem. Soc. Rev. 2010, 39, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Spassky, N.; Pluta, C.; Simic, V.; Thiam, M.; Wisniewski, M. Stereochemical Aspects of the Controlled Ring-Opening Polymerization of Chiral Cyclic Esters. Macromol. Symp. 1998, 128, 39–51. [Google Scholar] [CrossRef]
- Ovitt, T.M.; Coates, G.W. Stereochemistry of Lactide Polymerization with Chiral Catalysts: New Opportunities for Stereocontrol Using Polymer Exchange Mechanisms. J. Am. Chem. Soc. 2002, 124, 1316–1326. [Google Scholar] [CrossRef] [PubMed]
- Worch, J.C.; Prydderch, H.; Jimaja, S.; Bexis, P.; Becker, M.L.; Dove, A.P. Stereochemical enhancement of polymer properties. Nat. Rev. Chem. 2019, 3, 514–535. [Google Scholar] [CrossRef]
- Stanford, M.J.; Dove, A.P. Stereocontrolled Ring-Opening Polymerisation of Lactide. Chem. Soc. Rev. 2010, 39, 486–494. [Google Scholar] [CrossRef]
- Dijkstra, P.J.; Du, H.; Feijen, J. Single site catalysts for stereoselective ring-opening polymerization of lactide. Polym. Chem. 2011, 2, 520–527. [Google Scholar] [CrossRef]
- Ottou, W.N.; Sardon, H.; Mecerreyes, D.; Vignolle, J.; Taton, D. Update and challenges in organo-mediated polymerization reactions. Prog. Polym. Sci. 2016, 56, 64–115. [Google Scholar] [CrossRef]
- Jensen, T.R.; Breyfogle, L.E.; Hillmyer, M.A.; Tolman, W.B. Stereoselective polymerization of D,L-lactide using N-heterocyclic carbene based compounds. Chem. Comm. 2004, 10, 2504–2505. [Google Scholar] [CrossRef]
- Dove, A.P.; Li, H.; Pratt, R.C.; Lohmeijer, B.G.G.; Culkin, D.A.; Waymouth, R.M.; Hedrick, J.L. Stereoselective Polymerization of Rac- and Meso-Lactide Catalyzed by Sterically Encumbered N-Heterocyclic Carbenes. Chem. Commun. 2006, 27, 2881. [Google Scholar] [CrossRef]
- Pratt, R.C.; Lohmeijer, B.G.G.; Long, D.A.; Lundberg, P.N.P.; Dove, A.P.; Li, H.; Wade, C.G.; Waymouth, R.M.; Hedrick, J.L. Exploration, Optimization, and Application of Supramolecular Thiourea- Amine Catalysts for the Synthesis of Lactide (Co)Polymers. Macromolecules 2006, 39, 7863–7871. [Google Scholar] [CrossRef]
- Zhang, L.; Nederberg, F.; Messman, J.M.; Pratt, R.C.; Hedrick, J.L.; Wade, C.G. Organocatalytic Stereoselective Ring-Opening Polymerization of Lactide with Dimeric Phosphazene Bases. J. Am. Chem. Soc. 2007, 129, 12610–12611. [Google Scholar] [CrossRef] [PubMed]
- Miyake, G.M.; Chen, E.Y.X. Cinchona Alkaloids as Stereoselective Organocatalysts for the Partial Kinetic Resolution Polymerization of Rac-Lactide. Macromolecules 2011, 44, 4116–4124. [Google Scholar] [CrossRef]
- Makiguchi, K.; Yamanaka, T.; Kakuchi, T.; Terada, M.; Satoh, T. Binaphthol-Derived Phosphoric Acids as Efficient Chiral Organocatalysts for the Enantiomer-Selective Polymerization of Rac-Lactide. Chem. Commun. 2014, 50, 2883–2885. [Google Scholar] [CrossRef]
- Zhu, J.B.; Chen, E.Y.X. From Meso-Lactide to Isotactic Polylactide: Epimerization by B/N Lewis Pairs and Kinetic Resolution by Organic Catalysts. J. Am. Chem. Soc. 2015, 137, 12506–12509. [Google Scholar] [CrossRef]
- Sanchez-Sanchez, A.; Rivilla, I.; Agirre, M.; Basterretxea, A.; Etxeberria, A.; Veloso, A.; Sardon, H.; Mecerreyes, D.; Cossío, F.P. Enantioselective Ring-Opening Polymerization of Rac-Lactide Dictated by Densely Substituted Amino Acids. J. Am. Chem. Soc. 2017, 139, 4805–4814. [Google Scholar] [CrossRef]
- Liu, S.; Li, H.; Zhao, N.; Li, Z. Stereoselective Ring-Opening Polymerization of Rac -Lactide Using Organocatalytic Cyclic Trimeric Phosphazene Base. ACS Macro Lett. 2018, 7, 624–628. [Google Scholar] [CrossRef]
- Orhan, B.; Tschan, M.J.-L.; Wirotius, A.-L.; Dove, A.P.; Coulembier, O.; Taton, D. Isoselective Ring-Opening Polymerization of rac-Lactide from Chiral Takemoto’s Organocatalysts: Elucidation of Stereocontrol. ACS Macro Lett. 2018, 7, 1413–1419. [Google Scholar] [CrossRef]
- Lim, J.Y.C.; Yuntawattana, N.; Beer, P.D.; Williams, C.K. Isoselective Lactide Ring Opening Polymerisation using [2]Rotaxane Catalysts. Angew. Chem. Int. Ed. 2019, 58, 6007–6011. [Google Scholar] [CrossRef]
- Lohmeijer, B.G.G.; Pratt, R.C.; Leibfarth, F.; Logan, J.W.; Long, D.A.; Dove, A.P.; Nederberg, F.; Choi, J.; Wade, C.; Waymouth, R.M.; et al. Guanidine and Amidine Organocatalysts for Ring-Opening Polymerization of Cyclic Esters. Macromolecules 2006, 39, 8574–8583. [Google Scholar] [CrossRef]
- Pratt, R.C.; Lohmeijer, B.G.G.; Long, D.A.; Waymouth, R.M.; Hedrick, J.L. Triazabicyclodecene: A Simple Bifunctional Organocatalyst for Acyl Transfer and Ring-Opening Polymerization of Cyclic Esters. J. Am. Chem. Soc. 2006, 128, 4556–4557. [Google Scholar] [CrossRef] [PubMed]
- Baran, J.; Duda, A.; Kowalski, A.; Szymanski, R.; Penczek, S. Intermolecular chain transfer to polymer with chain scission: General treatment and determination of kp/ktr in L,L-lactide polymerization. Macromol. Rapid Commun. 1997, 18, 325–333. [Google Scholar] [CrossRef]
- Chuma, A.; Horn, H.W.; Swope, W.C.; Pratt, R.C.; Zhang, L.; Lohmeijer, B.G.G.; Wade, C.G.; Waymouth, R.M.; Hedrick, J.L.; Rice, L.E. The Reaction Mechanism for the Organocatalytic Ring-Opening Polymerization of l-Lactide Using a Guanidine-Based Catalyst: Hydrogen-Bonded or Covalently Bound? J. Am. Chem. Soc. 2008, 130, 6749–6754. [Google Scholar] [CrossRef] [PubMed]
- Simon, L.; Goodman, J.M. The Mechanism of TBD-Catalyzed Ring-Opening Polymerization of Cyclic Esters. J. Org. Chem. 2007, 72, 9656–9662. [Google Scholar] [CrossRef]
- Nifant’ev, I.; Ivchenko, P. DFT Modeling of Organocatalytic Ring-Opening Polymerization of Cyclic Esters: A Crucial Role of Proton Exchange and Hydrogen Bonding. Polymers 2019, 11, 2078. [Google Scholar] [CrossRef]

| Entry | [rac-LA]0 (M) | [rac-LA]0/ [BnOH]0/ [TBD]0 | t (min) | Conv. 1 (%) | Mn2 (g.mol−1) | ĐM2 | Pm3 | Tg4 (°C) | Tm4 (°C) | ΔHm (J.g−1) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.08 | 100:1:0.1 | 50 | 17 | 2600 | 1.09 | 0.84 | 44 | 157 | 18.9 |
| 2 | 0.08 | 100:1:0.1 | 120 | 30 | 4200 | 1.09 | 0.80 | 48 | 155 | 16.0 |
| 3 | 0.22 | 100:1:1 | 20 | 42 | 7000 | 1.15 | 0.85 | 52 | 144 | 26.6 |
| 4 | 0.22 | 100:1:1 | 30 | 77 | 11,000 | 1.25 | 0.85 | 51 | 141 | 8.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moins, S.; Hoyas, S.; Lemaur, V.; Orhan, B.; Delle Chiaie, K.; Lazzaroni, R.; Taton, D.; Dove, A.P.; Coulembier, O. Stereoselective ROP of rac- and meso-Lactides Using Achiral TBD as Catalyst. Catalysts 2020, 10, 620. https://doi.org/10.3390/catal10060620
Moins S, Hoyas S, Lemaur V, Orhan B, Delle Chiaie K, Lazzaroni R, Taton D, Dove AP, Coulembier O. Stereoselective ROP of rac- and meso-Lactides Using Achiral TBD as Catalyst. Catalysts. 2020; 10(6):620. https://doi.org/10.3390/catal10060620
Chicago/Turabian StyleMoins, Sébastien, Sébastien Hoyas, Vincent Lemaur, Beste Orhan, Kayla Delle Chiaie, Roberto Lazzaroni, Daniel Taton, Andrew P. Dove, and Olivier Coulembier. 2020. "Stereoselective ROP of rac- and meso-Lactides Using Achiral TBD as Catalyst" Catalysts 10, no. 6: 620. https://doi.org/10.3390/catal10060620
APA StyleMoins, S., Hoyas, S., Lemaur, V., Orhan, B., Delle Chiaie, K., Lazzaroni, R., Taton, D., Dove, A. P., & Coulembier, O. (2020). Stereoselective ROP of rac- and meso-Lactides Using Achiral TBD as Catalyst. Catalysts, 10(6), 620. https://doi.org/10.3390/catal10060620