Biocatalysis in the Swiss Manufacturing Environment
Abstract
1. Biocatalysis in Switzerland
1.1. The Chemical and Pharmaceutical Industry
- An increasing number of companies are considering using enzymes in their chemical synthesis approaches every year.
- Enzyme steps are typically considered opportunistically if the enzyme is available in commercial quantities.
- For companies without in-house fermentation facilities and prior biocatalysis experience, it is perceived that only hydrolytic enzymes are readily available in industrial quantities for manufacturing purposes.
- Industrial interviewees signaled the need for a broader enzyme toolbox containing ready-to-use enzymes.
- Unlike our European neighbors, there are no Swiss CDMOs (Contract Development and Manufacturing Organizations) and service companies offering support in enzyme R&D, piloting and production.
- Academia tends to have a more positive view of the industrial applicability of biocatalysis than industry.
- Besides the pharmaceutical industry, the fragrance and flavor industry should play a leading role and drive innovation in biocatalysis.
| Companies |
| Aschem GmbH. Swiss fine chemical SME (Small Medium Enterprise) founded in 2018 by a Chinese entrepreneur focusing on the design, synthesis, and production of building blocks for drug discovery and services involving nanomaterials. Example of a Swiss company having strong connections to China relying on Chinese partners for R&D and production. The strong ties are manifested in the use of Chinese characters in the company logo. |
| AVA Biochem. AVA Biochem is a private company located in Muttenz, subsidiary of AVA-CO2 (former Avalon Industries AG) in Zug. The company produces the furan-based platform chemical 5-hydroxymethylfurfural (5-HMF) from renewable carbohydrates in ton quantities. AVA-Biochem uses a thermo-chemical process for the conversion of waste biomass, which is not competing with the food supply chain. 5-HMF is the starting material for the production of furan dicarboxylic methyl ester or polyethylene furanoate. The company currently supplies ton quantities out of its Muttenz pilot plant but is presently investing in manufacturing facilities in Europe. |
| AZAD. Azad Pharma AG is a private company from Schaffhausen established in 2000 to handle Cilag’s API portfolio. The small company focuses on small molecules that are difficult to synthesize. Together with local applied universities, AZAD developed new synthetic routes to established and recently approved drugs, without infringing existing patents. Process development is managed from Schaffhausen with CDMOs in Europe, Taiwan, India, or China. |
| Axpo. The production and distribution of energy is the main business of this public company. In addition, Axpo is also Switzerland’s most important producer of renewable energy. Axpo Biomass operates 15 fermentation plants (SST solid state fermentation) recycling biowaste from more than 2500 customers. The company advertises that up to 1000 kWh energy and 900 kg of natural fertilizer can be produced with 1 ton of biowaste. Axpo Compogas AG offers disposal and recycling services for organic wastes. |
| Bachem. Founded in 1971, this public company specializes in the development and production of peptides, with sales of 314 million in 2019 and a total of 1200 employees. The company carried out ~150 cGMP projects targeting new chemical entities (NCEs). As of 2019, the synthesis and production of oligonucleotides is a new service added to the portfolio of the company. Bachem plans to increase sales to CHF 500 million within the next three years, CHF 100 million of which should be contributed by the new oligonucleotide business. |
| BASF Schweiz AG. Ciba Specialty Chemicals was acquired and integrated into the BASF organization in 2009 and renamed BASF Schweiz AG in March 2010. Ciba was one of the founding members of the Swiss Industrial Biocatalysis Consortium (SIBC). BASF Schweiz has nine sites counting 1600 employees. The site Schweizerhalle focuses on the development of new products and improves existing processes. At the time, Ciba applied biocatalytic steps for the production of specialty monomers by transesterification and employed lipase-catalyzed resolutions. |
| Biosynth Carbosynth. Biosynth Carbosynth was founded in 1965 and today employs approximately 500 employees. Headquartered in Switzerland, this private company operates in Switzerland, UK, Slovakia, and China, offering a wide range of products such as biochemicals, chemical intermediates, APIs, and natural products for the life sciences. They are a niche API manufacturer and operate reactors between 50 L and 2500 L in Switzerland, including CMO services. Enzymatic reactions are possible. |
| Cerbios-Pharma SA. 100% family-owned company founded in 1980, headquartered in Barbegno-Lugano with 132 employees generating sales of CHF 26 million. Cerbios offers third party CDMO services for APIs, HPAPIs, Biologics, and ADCs. For the latter, Cerbios, AGC Biologics, and Oncotec formed a collaboration called Proveo in 2015 to provide services for antibody drug conjugates. Cerbios’ facilities are located in Barbengo-Lugano (TI) and in Couvet (NE) under the name of GMT SA Fine Chemicals. Growth of Cerbios was mainly driven by the chemical divisions’ activity. Biocatalytic steps are evaluated in API process development, such as in the case of a process for the formylation of tetrahydrofolic acid with a transferase. |
| Cilag. Cilag (Chemical Industrial Laboratory) started activities in 1936 and became a part of the Johnson & Johnson family. Today, it is a unit of Janssen, the pharmaceutical division of Johnson & Johnson. Cilag is one of the larger Swiss manufacturer of small molecule APIs and drug substances with about 1200 employees. The company focuses on difficult to synthesize molecules, both from a process as well as intellectual property point of view. The company has shifted away from large scale manufacturing in Switzerland, instead concentrating on products with a higher value added, such as compounds requiring high containment installations. |
| Corden Pharma. Corden Pharma is a full-service CDMO with 1570 employees, which was founded in 1980. Since 2006, Cordon Pharma has been part of the cGMP pharma service and manufacturing platform of the International Chemical Investors Group (ICIG) with sites in Europe and the US. The facility in Liestal (reaction vessels up to 2.5 m3) offers process development and cGMP manufacturing for small molecule APIs, intermediates, and chiral building blocks, with special expertise in synthetic lipids, carbohydrates, and conjugates thereof. Biocatalysis is not a strategic direction at Corden Pharma, although a few enzymatic processes have been carried out in the Liestal facility. The parent company ICIG recently acquired a former Sandoz site in Germany with a total fermenter volume of 3000 m3 currently producing non cGMP food and feed enzymes. |
| Clariant. Clariant is a public company founded 1995 as a spin-off from the chemical business of Sandoz during the formation of Novartis. Clariant has 18,000 employees generating group sales of CHF 6.62 billion. Clariant’s life science related products include personal care chemicals, crops, and the biocides business. In 2011 Clariant acquired Süd-Chemie AG and their Sunliquid® process and plant in Straubing (Bavaria) for the production of cellulosic ethanol from agricultural waste streams, i.e., from non-food sources. The associated know-how can also be used for other bio-based chemicals such as propylene, isobutylene, or ethylene oxide. Liquebeet® is a sugar beet-based carbon and energy source for fermentations. |
| DOTTIKON EXCLUSIV SYNTHESIS AG. Founded in 1913, the company manufactures exclusive APIs, high-quality intermediates, and performance chemicals. DOTTIKON is employing a one-site strategy and is currently in the midst of a major capacity expansion. DOTTIKON ES HOLDING AG, the parent company, is listed in the main segment of the SIX Swiss Exchange. With over 600 employees in Dottikon/Switzerland and sales of CHF 175 million (2019/20), DOTTIKON is a strategic development and manufacturing partner providing full CDMO services with know-how in hazardous reactions, high-pressure processes, low-temperature and flow chemistry. A wide spectrum of reaction conditions and OEBs (occupational exposure bands) down to 1 µg/m3 in reactors from 100 to 12,500 L allow them to synthesize different types of small molecules. |
| DSM. Royal DSM Global Dutch company is active in nutrition, health, and sustainable living. The company counts 23,000 employees and generates approximately EUR 10 billion in sales. Through the acquisition of the former Roche vitamin and fine chemicals business, DSM Nutritional Product has three manufacturing sites in Switzerland. DSM Nutritional Products headquarters and R&D center are located in Kaiseraugst, Sisseln, with 1000 employees, producing vitamin E, vitamin A, vitamin K, astaxanthin, folic acids, and other products. DSM Lalden, with 160 employees, produces precursors used in vitamin production. With the acquisition of the Swiss SME Pentpharm in 2007 DSM acquired expertise in API proteins from snake and other natural sources. Biotechnology (biosynthesis and biocatalysis) is a key technology for DSM, a SIBC founding member. With the divestment of stake in Patheon, custom manufacturing is not an activity of DSM anymore. |
| Evolva. Evolva is a Swiss biotech company focused on the research, development, and commercialization of nature-based ingredients using engineered bacteria and yeast. The public SME counts approximately 70 employees. The company received US EPA registration for nootkatone (NootkaShieldTM), a next-generation protection against insect-borne diseases, in August 2020. Other products include several formulations of resveratrol, a compound associated with health benefits. After launching the cold-water dispersible resveratrol powder, Veri-SperseTM, in 2019, Evolva followed up with another formulation, named Veri-te AquaTM, a soluble resveratrol that was developed based on a market need, especially in functional beverages. Evolva plans to launch another product, EVE-X157/Z4, under its flavor and fragrance sector by the end of 2020. The first commercial-scale production of EVERSWEETTM, a non-artificial zero-calories stevia sweetener, was launched in November 2019 at Cargill’s fermentation facility in Blair, Nebraska (USA). |
| Firmenich. Founded in 1895, Firmenich is the world’s largest privately-owned fragrance and flavor company, recording a revenue of CHF 3.9 billion for the fiscal year 2019. Firmenich has 7600 employees and invests 10% of its turnover in R&D. The company is leading in its industry regarding biotechnological manufacturing principles. CLEARWOOD®, AMBROX®, Z11, or DreamwoodTM are examples of perfumery ingredients using biotechnological manufacturing principles. |
| Givaudan. Founded in 1895, this global flavor and fragrance company employs 12,000 people and generated CHF 6.6 billion in 2019. Givaudan uses organic chemical synthesis and biotechnological means for ingredient production. It has established a strong biotech R&D to implement biotechnological manufacturing principles, among them the patented application of laccases or the use of squalene hopene cyclase for the production of (-)-Ambrox. Givaudan also operates a marine biotechnology center of excellence in Brittany for the development of novel algae-based cosmetic products. In addition, the F&F company has strengthened its biotechnology sector with a number of acquisitions, including the French company Soliance, which specializes in microbial and microalgae product sourcing. Givaudan is a founding member of the SIBC. |
| Huntsman. Huntsman is a US multinational manufacturer of chemical products for consumers and industry. In 2006, Huntsman purchased the CIBA textile effect business of Ciba Specialty Chemicals in Basel with 4200 employees. The Huntsman division “Advanced Materials” operates a manufacturing site in Monthey (Valais). Plans to merge Clariant and Huntsman failed in 2017 due to an activist investor who acquired a quarter of Clariant shares to stop the deal. |
| Inofea. Established in 2011 and incorporated in early 2014 in Basel, this SME has developed a new enzyme platform (enzzen®-enzymes) to supply more stable enzymes to industry, which would also be suitable for continuous processing. The enzyme stabilization is achieved via a proprietary immobilization technique and tailor-made shield, an enabling technology offered to customers. |
| Lonza. Lonza is a public company founded in 1897, which counts approximately 15,400 employees today. Lonza is Switzerland’s largest CMO. The company operates globally and generated sales of CHF 5.9 billion in 2019. Lonza was one of first companies applying biotechnology in chemical synthesis in the early 1980s to obtain optically active intermediates and enantiomerically pure drugs and continues to use biocatalysis opportunistically in chemicals synthesis. Today, Lonza is the largest CMO manufacturer of monoclonal antibodies. The chemical division “Specialty Ingredients” is to be sold, while the biopharmaceuticals business is massively expanding through the IbexTM project located in Visp. Lonza is a founding member of the SIBC. |
| Nestlé. Nestlé was formed in 1905 by the merger of the Anglo-Swiss Milk Company with Farine Lactée of Henri Nestlé. Nestlé employed 291,000 people and generated sales of CHF 92.6 billion in 2019. The Nestlé R&D has expertise in biotransformation which are harnessed in several of its R&D and Product Technology Centres. Biotechnology is an important manufacturing tool for Nestlé as showcased by the production of MAGGI Liquid Seasoning, which is based on wheat gluten fermentation. Enzyme technology is applied in extraction processes for a chocolate malt beverage or for cereal products. Nestlé also uses probiotics in many products. Nestlé acquired Aimmune Therapeutics to gain a leading position in food allergy prevention and treatment. |
| Nitrochemie. Swiss-German conglomerate between the Ruag MRO Holding and Rheinmetall AG. Among other activities, the company carries out custom manufacturing and process development for chemical intermediates. |
| Novartis. Figures among the top three pharmaceutical companies in the world together with Swiss competitor Roche. Novartis uses biotechnological and biocatalytic methods for small and large molecule production and drug metabolite production. Novartis was an early adopter of biocatalysis in drug manufacturing and is a founding member of the SIBC. |
| Merck. Merck is a science and technology company with the business sectors healthcare, life science, and performance materials generating sales of EUR 16 billion with 57,000 employees. The origin of the company goes back to 1668 when Friedrich J. Merck founded a pharmacy in Darmstadt. Merck is active since 1929 in Switzerland with nine sites, including five manufacturing sites with a total of 2370 employees. In 2006, Merck acquired Serono for the production of biotech-based medicines, followed by the acquisition of the life science company Sigma-Aldrich in 2015. The Buchs site with 450 employees was originally the site of the Swiss fine chemical company Fluka, founded in 1950 by the Swiss pharmaceutical companies Ciba, Geigy, and Hoffmann-La Roche. Biocatalysis has always been an important technology for analytical and manufacturing applications at the Buchs site. Sigma-Aldrich is a SIBC founding member. |
| Perseo. Based on its enzzen®-technology, Inofea AG founded Perseo Pharma AG in 2019, a pharma product development company with a focus on therapeutic enzymes for the treatment of cancer, gastrointestinal, and rare genetic diseases. |
| Proveo. Collaboration between Cerbios, AGC Biologics and Oncotec since 2015 to provide services for antibody drug conjugates. |
| Roche. Roche belongs to the top five global pharmaceutical companies. The company uses, among others, biotechnological and biocatalytic methods for small and large molecule production. Biocatalysis work is dedicated to process R&D and used for the generation of drug metabolites. Especially the production of chiral intermediates via enzymatic steps renders biocatalysis an attractive alternative to classical organic chemistry. Another focus is the replacement of metal catalysts to avoid toxicology problems due to heavy metal contaminations. Enzyme production (fermentation) and strain development is outsourced to contract research organizations (CROs) but can be produced in house on lab scale. A broad range of enzymes are used. Founding member of the SIBC. |
| Siegfried. Siegfried is a public company, which was founded in 1873, generating sales of CHF 833 million in 2019 with 2200 employees. Siegfried is the second largest CMO in Switzerland for the synthesis of drug substances. Siegfried has nine sites, six in Europe, two in the US, and one in China. The Swiss sites are Evionnaz and the headquarters in Zofingen, with a local production capacity of 350 m3. Siegfried uses enzyme catalysis opportunistically if the desired enzymes are available in the required quantities. Siegfried owns a former BASF site in Minden, Germany with 1458 m3 fermentation volume, in which caffeine, theophylline, ephedrine, and pseudoephedrine were produced by Knoll AG, later BASF. This site has decades of experience with yeast-mediated synthesis of e.g., ephedrine. |
| Solvias. Private company founded 1999 with headquarters in Basel counting more than 500 employees. Solvias is a provider of integrated solutions in analytical services. In addition, it carries out chemical development including route-scouting services and cGMP custom synthesis of chiral and complex molecules in kg amounts for clinical trials. The company combines high-throughput experimentation with a catalyst library of more than 500 ligands for heterogenous catalytic transformations for asymmetric hydrogenations, C–X couplings, and asymmetric C–C bond formations. |
| SpiroChem AG. Founded in 2011 as an ETH Zurich spin-off with headquarters in Basel, the company successfully acts as a CRO for drug design and proprietary process chemistry. SpiroChem AG employs 50 FTEs working on high-value-added molecular building blocks for the lead optimization of candidates in drug discovery. Introduction of enzymatic transformations in chemical synthesis routes is not excluded but the current focus is to remain a leader in synthetic chemistry. |
| SSE. Founded in 1894, the public company Société Suisse des Éxplosifs SA diversified in the early 1970s. SSE invested in a new production unit for fine chemicals and founded Valsynthèse SA in 1983, offering custom pilot and commercial production with hazardous chemical reactions such as nitration or phosgenation (COCl2). The SSE group generated sales of CHF 132 million with 650 employees in 2018. The business segment, Fine Chemicals, including the contract manufacturing activities, was recently separated from the explosives business. SSE is a medium-size player among the Swiss CMOs for organic chemicals. |
| Swissfillon. Swissfillon AG is one of the few Swiss CMOs able to offer aseptic fill and finish with expertise for drug product manufacturing, including primary packaging selection, process development, and secondary packaging for pharmaceutical and biotech companies. Founded in 2013 in Visp (Valais, Switzerland) as a spinoff of the pharma engineering company pixon engineering AG, Swissfillon operates flexible, highly automated, and fully cGMP compliant filling lines for vials, syringes, and cartridges from 1l to 200l bulk batch size. Swissfillon also has expertise related to regulatory requirements for the drug product in combination with the medical device such as, for example, injectables for ophthalmic applications. |
| Syngenta. The agro-divisions of Astra Zeneca and Novartis formed the global agrochemistry player Syngenta in 2000. In 2016, ChemChina made a successful takeover offer of USD 43 billion to the Syngenta shareholders, and by summer 2017, ChemChina owned 98% of Syngenta. In 2020, Syngenta AG, Adama (based in Israel) and the agricultural businesses of Sinochem became a single entity, the Syngenta Group, headquartered in Switzerland, with a turnover of USD 23 billion. In the 2019 annual report, Syngenta AG reported sales of USD 13.6 billion generated with 28,000 employees resulting in an EBITDA (Earnings before interest, taxes, depreciation and amortization) of 22%. Syngenta possesses six sites Switzerland, including the headquarter in Basel, the Monthey site producing crop protection products, the Kaisten site for the production of an herbicide precursor, the Münchwilen site dedicated to development activities, and Stein, one of the three main R&D sites. Recently, Syngenta’s presence in Switzerland was reinforced by the acquisition of a new production site in Muttenz. Syngenta uses biocatalytic methods for discovery, route scouting, and small molecules production. Syngenta is a founding member of the Swiss Industrial Biocatalysis Consortium (SIBC). |
| SynphaBase. Established in 2000 in Pratteln, SynphaBase is a spin-off of the University of Applied Sciences in Muttenz/Basel. SynphaBase is a custom research and development organization offering route scouting in process chemistry, including multistep synthesis from mg to metric tons by working with other custom manufacturers. The company uses primarily organic chemical synthesis, although enzymatic catalysis has occasionally been tested to facilitate procedures. |
| Vifor. The origins of Vifor Pharma date back to 1872. The company grew over the years through merger and acquisition and has several manufacturing sites in Switzerland. Ettingen and Villars-sur-Glâne focus on the manufacture of small molecules while OM Pharma has a focus on biotechnological manufacturing. The site in the canton of Fribourg (Villars-sur-Glâne) offers contract development and manufacturing service for organic chemical synthesis. |
| VioChemicals. Established in 2001, VioChemicals is a CMO for specialty chemicals, intermediates and APIs produced by chemical synthesis. VioChemicals uses cGMP manufacturing facilities in China and India, while their R&D center is located in Greece. |
| Organizations |
| CCBIO. Competence center of Biocatalysis of the Zürich Applied University (ZHAW). Establishes biocatalysis as a complementary method to organic synthesis by expanding the biocatalytic toolbox through algorithm- and automation-assisted enzyme engineering and gene mining. |
| FIT (Farma Industria Ticino). Founded in 1980 as a private organization with 30 member companies, which have a combined workforce of 2900 employees and an annual turnover of CHF 1.7 billion. The organization represents the chemical pharmaceutical industry, which is an important economic pillar in the Swiss canton of Ticino. |
| SATW. The working group “Biotechnology” of the Swiss Academy of Engineering Sciences focuses on products that are relevant for the Swiss economic space, such as small molecule pharmaceuticals, intermediates, fine chemicals, biochemicals, and other specialties. A joint SBA/SATW initiative currently aims to consolidate the Swiss biotechnology sector. |
| SBA. Swiss Biotech Association represents the interest of the biotechnology industry, currently predominantly representing the biopharmaceutical industry. A joint SBA/SATW initiative currently aims to consolidate the Swiss biotechnology sector. |
| SIBC. Swiss Industrial Biocatalysis Consortium was founded in 2004 to help overcome the limitations of applying enzymes in organic synthesis. Current members include Innosyn, Givaudan, Lonza, Merck, Novartis, Roche, and Syngenta. Four associations support the SIBC activities: SBA, scienceindustries, SATW and SKB. |
| SKB. Swiss Coordination Committee for Biotechnology is an umbrella organization encouraging the development of a sustainable biotechnology sector. |
1.2. Trends and New Advances
- Production relocation of APIs and final formulation to countries with cheap labor, notably China and India. In particular, the production of small molecule APIs and off patent drugs and formulations was affected in this way.
- Pharma companies concentrated their production to a small number of sites in countries with low wages (and often lower environmental standards). The number of production sites was further reduced through mergers and acquisitions.
- The transfer of processes and technologies led to a loss in manufacturing know-how in Western countries.
2. Current Status of Biocatalysis
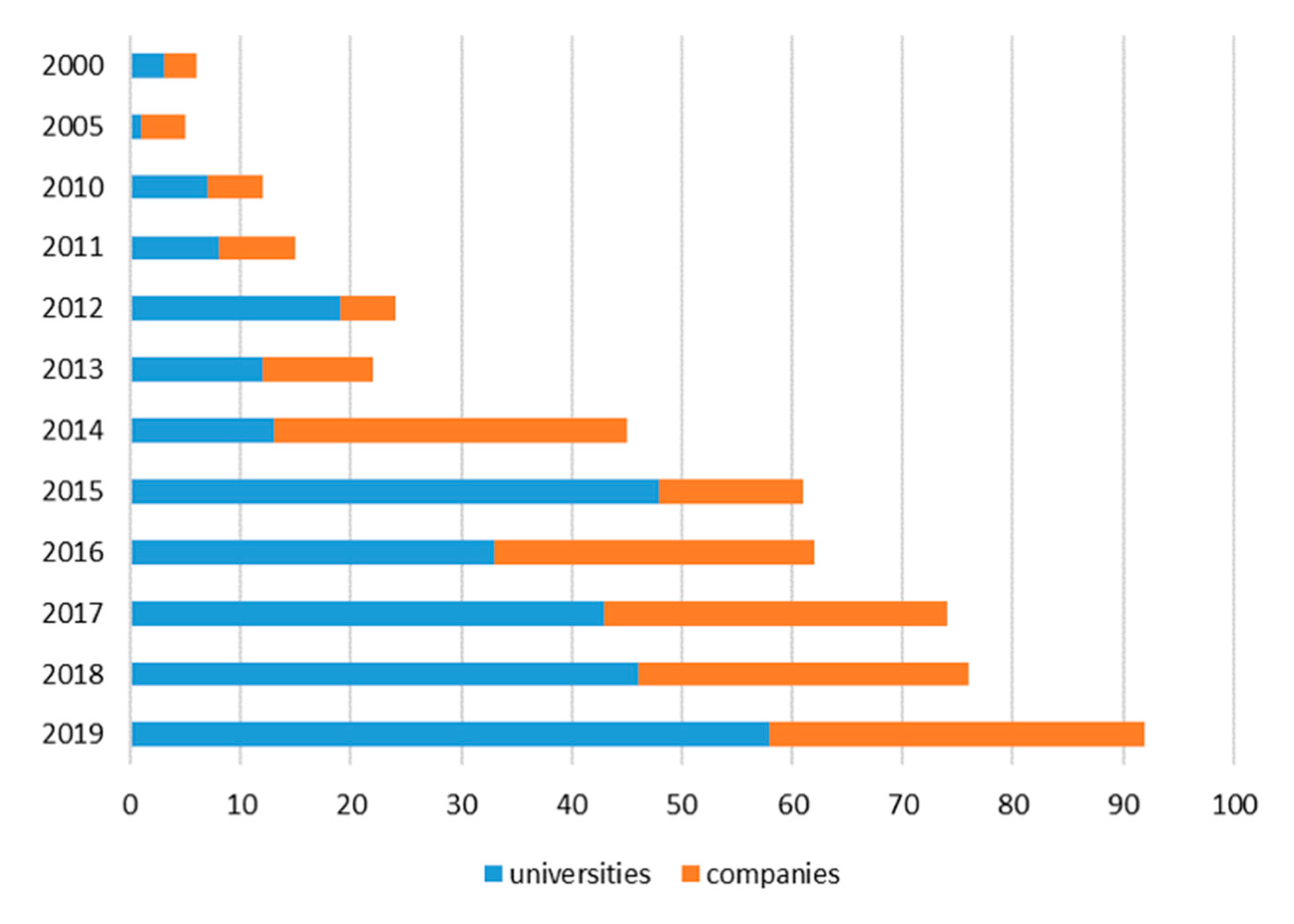
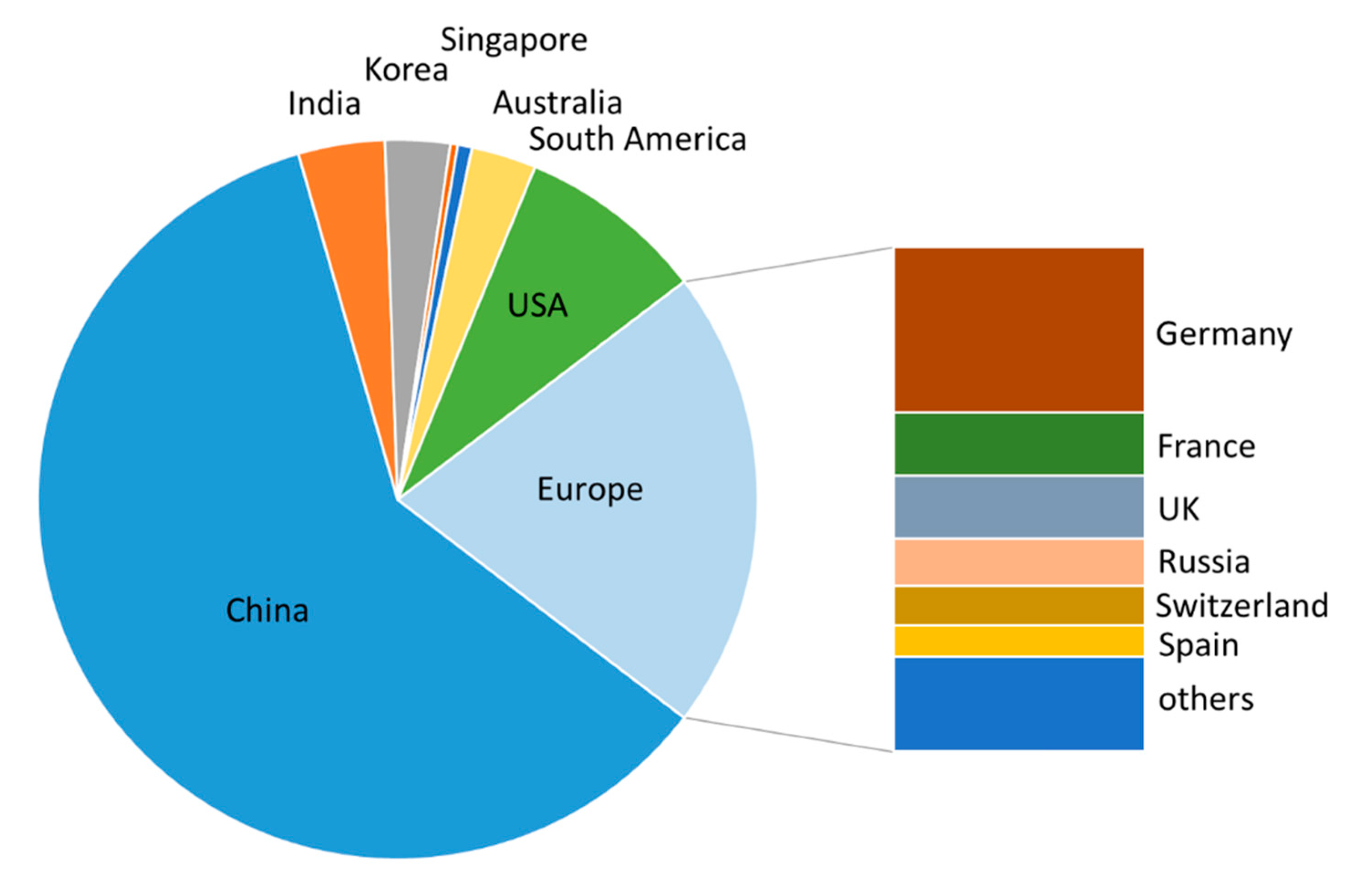
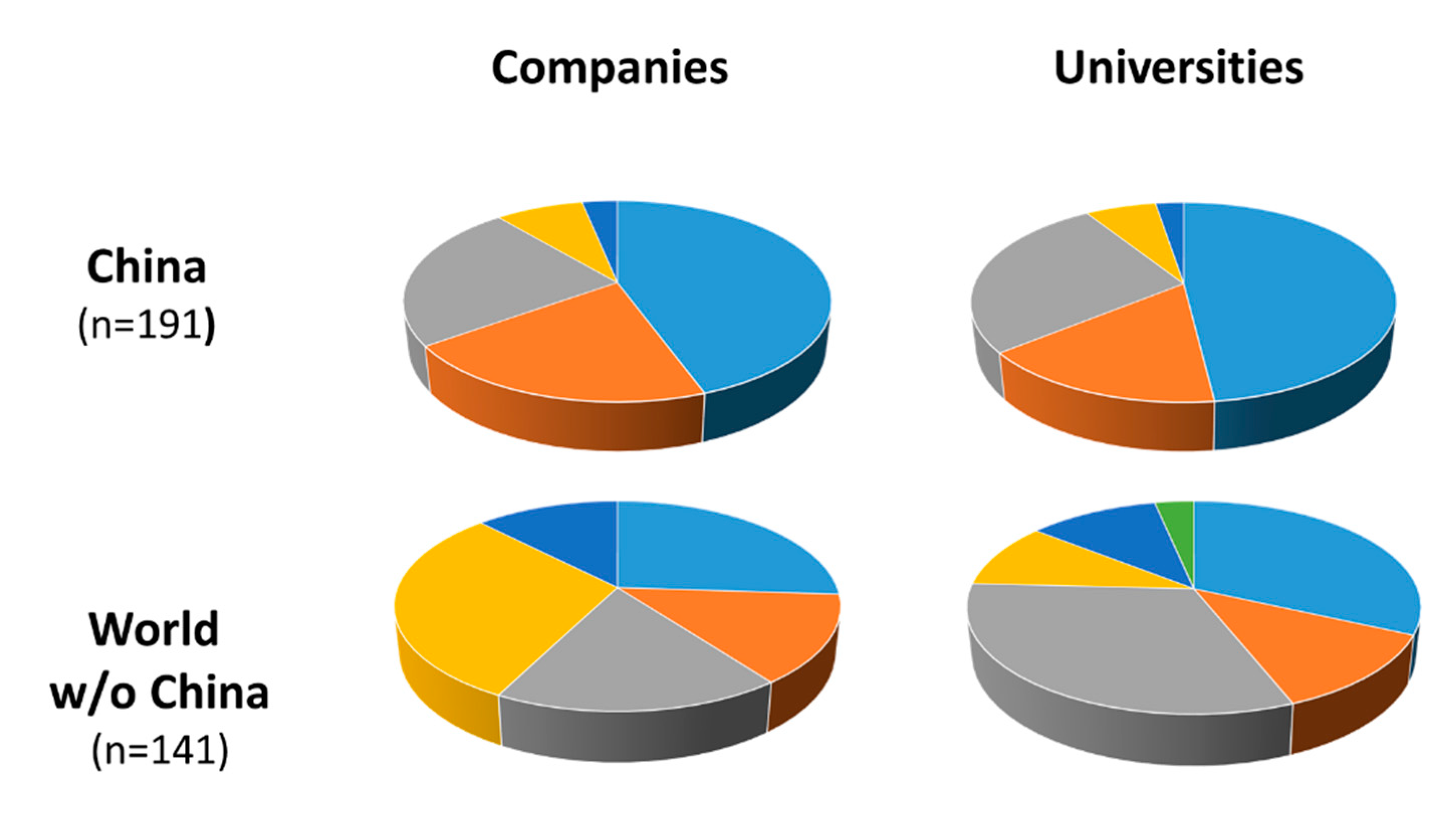
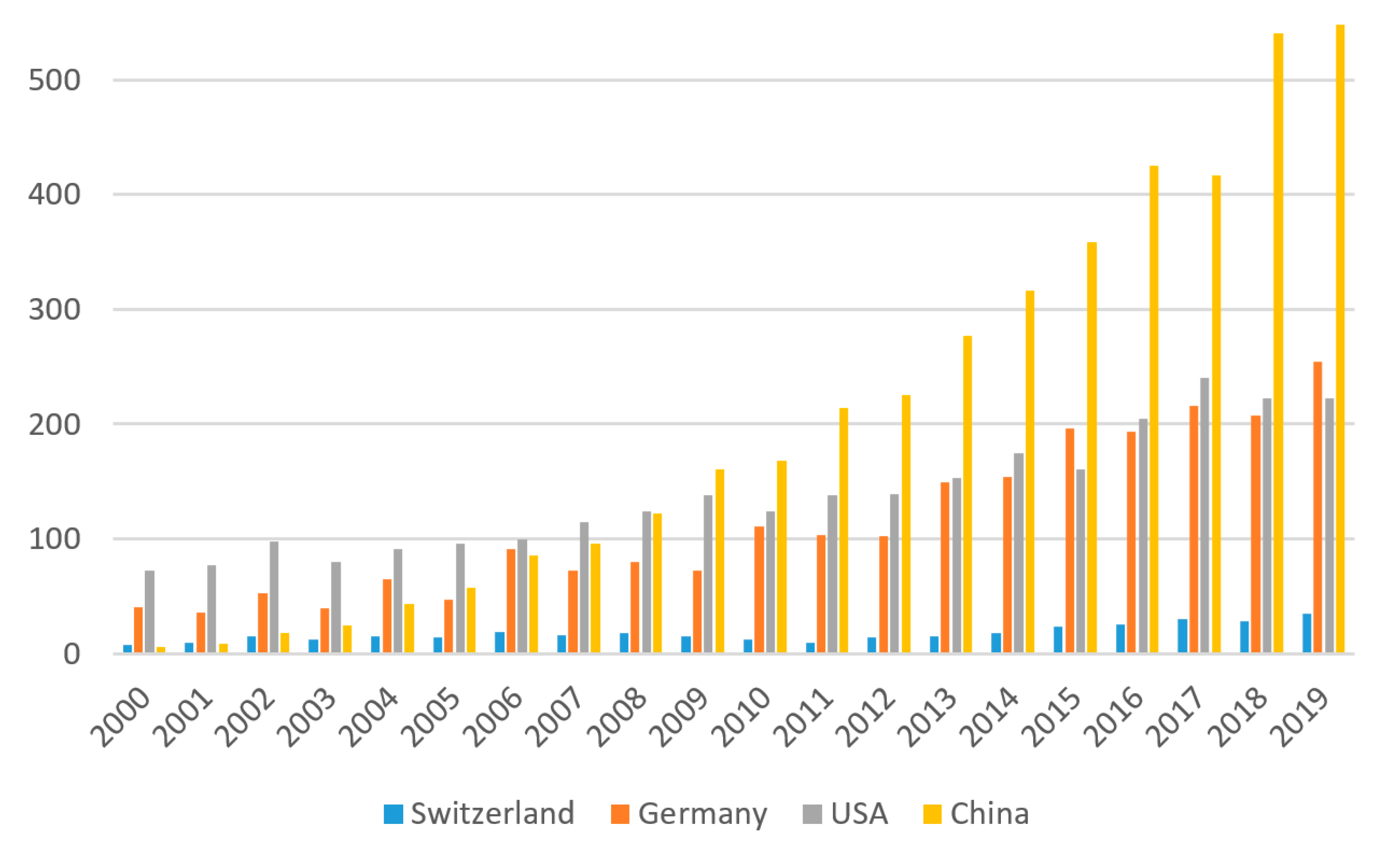
2.1. Patenting Activity
2.2. Biocatalytic Trends in Swiss Academic Research
2.2.1. Screening
2.2.2. Enzyme Cascades
2.2.3. Novel Enzyme Toolboxes
2.2.4. Biopolymers
2.2.5. Immobilization and Flow Chemistry
2.3. Biocatalytic Trends in Swiss industrial research
- The use of enzymatic transformations is no longer considered independently but biocatalysis is increasingly being used in concert with organic chemistry and flow chemistry to yield highly productive manufacturing routes.
- The biotechnological production of fine chemicals, specifically in the flavor and fragrance sector, allows the manufacture of complex olfactory molecules.
- In collaboration with academic groups, the exploration of industrially not yet fully established enzyme families allows to broaden the accessible molecular space.
2.3.1. Combining Biocatalysis with Transition-Metal Catalysis and Flow Chemistry
2.3.2. Producing Fine Chemicals via Biotechnological Means
2.3.3. Exploring the Biocatalytic Space
3. Opportunities
3.1. Self-Sustainable Development and Production of Critical Drugs in Developed Countries—Biocatalysis Impact
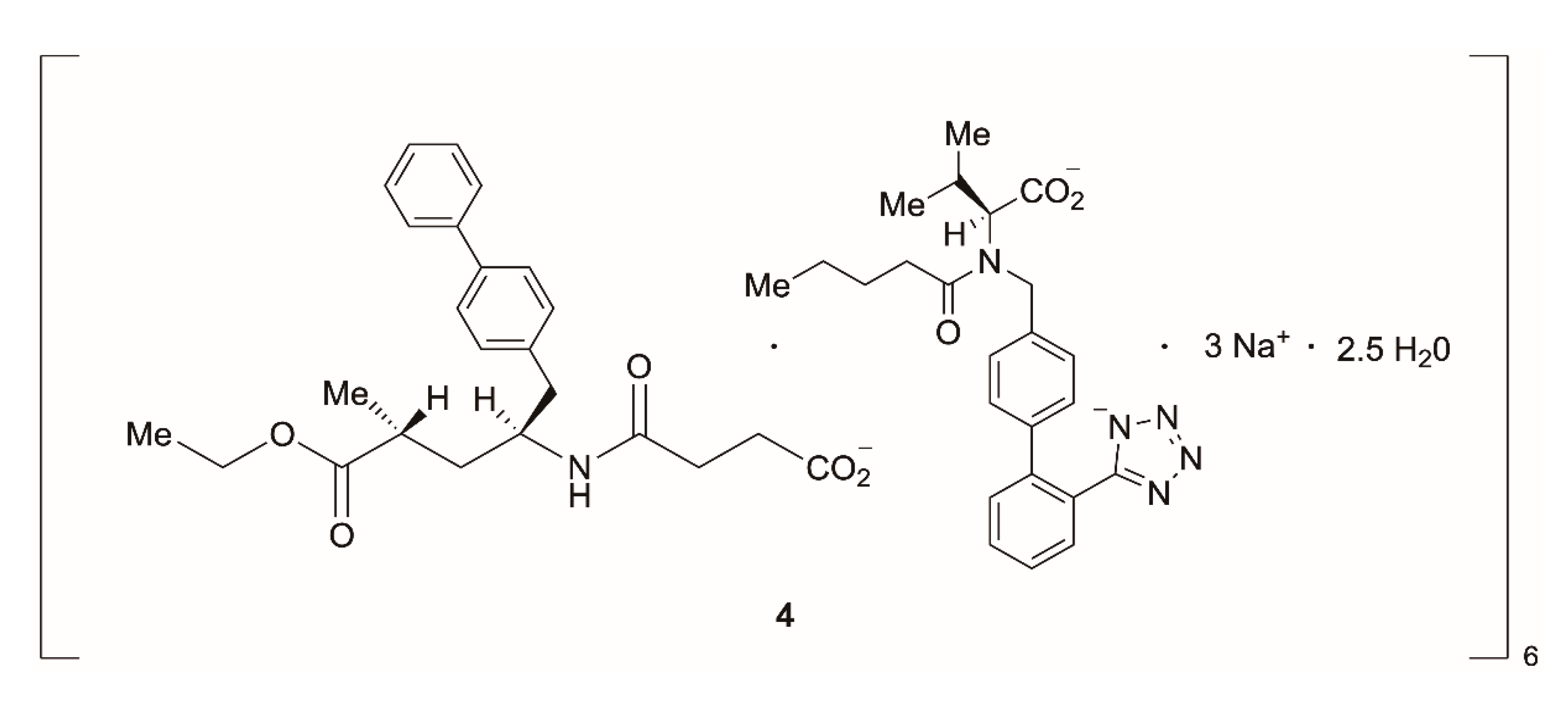
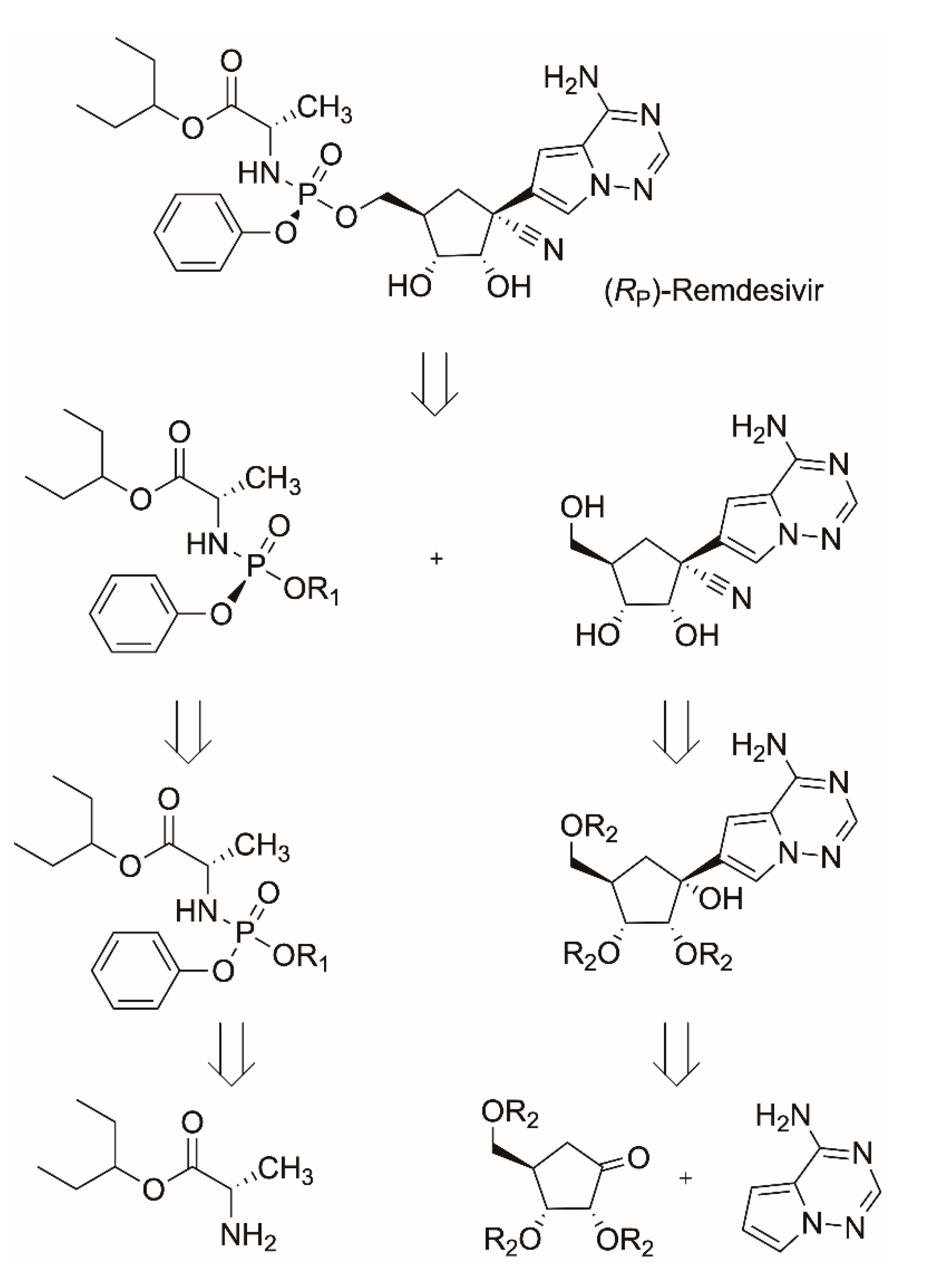
3.1.1. Remdesivir
3.1.2. Vincristine
3.1.3. Vitamin K1
3.2. Collaborations between Industry and Academia and Education
3.2.1. Education in Switzerland
3.2.2. Facilitating Academic–Industrial Collaborations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Switzerland Seen as Top Test Market for Europe. Available online: https://www.swissinfo.ch/eng/switzerland-seen-as-top-test-market-for-europe/3039956 (accessed on 11 November 2020).
- Buller, R.H.K.; Mirata, M.; Meyer, H.-P. An appreciation of Biocatalysis in the Swiss Manufacturing Environment. In Biocatalysis: An Industrial Perspective; de Gonzalo, G., Domínguez de Maria, P., Eds.; Royal Society of Chemistry: Cambridge, UK, 2018; pp. 1–43. [Google Scholar]
- Swiss Biotech Report 2020. Available online: https://www.swissbiotech.org/report/ (accessed on 21 August 2020).
- The Importance of the Pharmaceutical Industry for Switzerland. Available online: https://www.bak-economics.com/fileadmin/documents/reports/BAK_Economics_Polynomics_Interpharma_Bedeutungsstudie_e.pdf (accessed on 28 August 2020).
- Meyer, H.P.; Werbitzky, O. Development of Swiss Biotechnology Beyond the Biopharmaceutical Sector. In memoriam Prof. Dr. Oreste Ghisalba (1946–2018). Chimia 2020, 74, 345–359. [Google Scholar] [CrossRef] [PubMed]
- Antibiotic Development: The 10 × 20 Initiative. Available online: https://www.idsocety.org; https://www.idsociety.org/policy--advocacy/antimicrobial-resistance/antibiotic-development-the-10-x-20-initiative/ (accessed on 28 August 2020).
- AMR Action Fund. Available online: https://amractionfund.com/ (accessed on 4 September 2020).
- Reporting Federal Office for National Economic Supply FONES. Available online: https://www.bwl.admin.ch/bwl/en/home/themen/heilmittel/meldestelle.html (accessed on 2 December 2020).
- Drugshortage.ch. Available online: https://www.drugshortage.ch/ (accessed on 31 July 2020).
- WHO Model Lists of Essential Medicines. Available online: https://www.who.int/groups/expert-committee-on-selection-and-use-of-essential-medicines/essential-medicines-lists (accessed on 13 November 2020).
- 11 Wu, S.; Snajdrova, R.; Moore, J.C.; Baldenius, K.; Bornscheuer, U.T. Biocatalysis: Enzymatic Synthesis for Industrial Applications. Angew. Chem. Int. Ed. 2020, 59, 2–54. [Google Scholar] [CrossRef]
- Prud’homme, D. China’s shifting patent landscape and State-led patenting strategy. J. Intellect. Prop. Law Pract. 2015, 10, 619–625. [Google Scholar] [CrossRef]
- Notice of the State Intellectual Property Office on Issuing the National Patent Development Strategy (2011–2020). Available online: http://www.lawinfochina.com/Display.aspx?Lib=law&Id=8611&keyword (accessed on 20 November 2020).
- Debon, A.; Pott, M.; Obexer, R.; Green, A.P.; Friedrich, L.; Griffiths, A.D.; Hilvert, D. Ultrahigh-throughput screening enables efficient single-round oxidase remodelling. Nat. Catal. 2019, 2, 740–747. [Google Scholar] [CrossRef]
- Femmer, C.; Bechtold, M.; Held, M.; Panke, S. In vivo directed enzyme evolution in nanoliter reactors with antimetabolite selection. Metab. Eng. 2020, 59, 15–23. [Google Scholar] [CrossRef]
- Liao, C.S.; Seebeck, F.P. S-adenosylhomocysteine as a methyl transfer catalyst in biocatalytic methylation reactions. Nat. Catal. 2019, 2, 696–701. [Google Scholar] [CrossRef]
- Liao, C.S.; Seebeck, F.P. Asymmetric beta-Methylation of L- and D-alpha-Amino Acids by a Self-Contained Enzyme Cascade. Angew. Chem. Int. Ed. 2020, 59, 7184–7187. [Google Scholar] [CrossRef]
- Wu, S.K.; Zhou, Y.; Gerngross, D.; Jeschek, M.; Ward, T.R. Chemo-enzymatic cascades to produce cycloalkenes from bio-based resources. Nat. Commun. 2019, 10, 5060:1–5060:9. [Google Scholar] [CrossRef]
- Hayashi, T.; Ligibel, M.; Sager, E.; Voss, M.; Hunziker, J.; Schroer, K.; Snajdrova, R.; Buller, R. Evolved Aliphatic Halogenases Enable Regiocomplementary C-H Functionalization of Pharmaceutically Relevant. Angew. Chem. Int. Ed. 2019, 58, 18535–18539. [Google Scholar] [CrossRef]
- Amstutz, V.; Hanik, N.; Pott, J.; Utsunomia, C.; Zinn, M. Tailored biosynthesis of polyhydroxyalkanoates in chemostat cultures. In Enzymatic Polymerizations; Bruns, N., Loos, K., Eds.; Academic Press Ltd.-Elsevier Science Ltd.: London, UK, 2019; Volume 627, pp. 99–123. [Google Scholar]
- Hanik, N.; Utsunomia, C.; Guebitz, G.; Zinn, M. All about blocks... and copolymers thereof—Current and future playgrounds for polyhydroxyalkanoates. New Biotechnol. 2018, 44, S106. [Google Scholar] [CrossRef]
- Briand, M.L.; Gebleux, R.; Richina, F.; Correro, M.R.; Grether, Y.; Dudal, Y.; Braga-Lagache, S.; Heller, M.; Beerli, R.R.; Grawunder, U.; et al. Partially shielded enzymes capable of processing large protein substrates. Chem. Commun. 2020, 56, 5170–5173. [Google Scholar] [CrossRef] [PubMed]
- Padrosa, D.R.; Benitez-Mateos, A.I.; Calvey, L.; Paradisi, F. Cell-free biocatalytic syntheses of L-pipecolic acid: A dual strategy approach and process intensification in flow. Green Chem. 2020, 22, 5310–5316. [Google Scholar] [CrossRef]
- Contente, M.L.; Tamborini, L.; Molinari, F.; Paradisi, F. Aromas flow: Eco-friendly, continuous, and scalable preparation of flavour esters. J. Flow Chem. 2020, 10, 235–240. [Google Scholar] [CrossRef]
- Han, C.; Savage, S.; Al-Sayah, M.; Yajima, H.; Remarchuk, T.; Reents, R.; Wirz, B.; Iding, H.; Bachmann, S.; Fantasia, S.M.; et al. Asymmetric Synthesis of Akt Kinase Inhibitor Ipatasertib. Org. Lett. 2017, 19, 4806–4809. [Google Scholar] [CrossRef]
- Swiss Chemical Society. Available online: https://polycoll.ch/index.php?option=com_content&view=category&layout=blog&id=91&Itemid=590&lang=en (accessed on 2 December 2020).
- Gu, X.; Zhao, J.; Chen, L.; Li, Y.; Yu, B.; Tian, X.; Min, Z.; Xu, S.; Gu, H.; Sun, J.; et al. Application of Transition-Metal Catalysis, Biocatalysis, and Flow Chemistry as State-of-the-Art Technologies in the Synthesis of LCZ696. J. Org. Chem. 2020, 85, 6844–6853. [Google Scholar] [CrossRef]
- Novick, S.J.; Dellas, N.; Alvizo, O.; Garcia, R.D.; Ching, C.; Entwistle, D. Engineered Transaminase Polypeptides for Industrial Biocatalysis. U.S. Patent No. WO 2018/231462 A1, 20 December 2018. [Google Scholar]
- Eichhorn, E.; Locher, E.; Guillemer, S.; Wahler, D.; Fourage, L.; Schilling, B. Biocatalytic Process for (−)-Ambrox Production Using Squalene Hopene Cyclase. Adv. Synth. Catal. 2018, 360, 2339–2351. [Google Scholar] [CrossRef]
- Armanino, N.; Charpentier, J.; Flachsmann, F.; Goeke, A.; Liniger, M.; Kraft, P. What’s Hot, What’s Not: The Trends of the Past 20 Years in the Chemistry of Odorants. Angew. Chem. Int. Ed. 2020, 59, 16310–16344. [Google Scholar] [CrossRef]
- Schalk, M.; Pastore, L.; Mirata, M.A.; Khim, S.; Schouwey, M.; Deguerry, F.; Pineda, V.; Rocci, L.; Daviet, L. Toward a Biosynthetic Route to Sclareol and Amber Odorants. J. Am. Chem. Soc. 2012, 134, 18900–18903. [Google Scholar] [CrossRef]
- Weber, N.; Hatsch, A.; Labagnere, L.; Heider, H. Production of (S)-2-aminobutyric acid and (S)-2-aminobutanol in Saccharomyces cerevisiae. Microb. Cell Fact. 2017, 16, 51:1–51:17. [Google Scholar] [CrossRef]
- Heath, R.S.; Birmingham, W.R.; Thompson, M.P.; Taglieber, A.; Daviet, L.; Turner, N.J. An Engineered Alcohol Oxidase for the Oxidation of Primary Alcohols. ChemBioChem 2019, 20, 276–281. [Google Scholar] [CrossRef]
- Ligibel, M.; Moore, C.; Bruccoleri, R.; Snajdrova, R. Identification and application of threonine aldolase for synthesis of valuable alpha-amino, beta-hydroxy-building blocks. Biochim. Biophys. Acta-Proteins Proteom. 2020, 1868, 140323:1–140323:8. [Google Scholar] [CrossRef]
- Schober, M.; MacDermaid, C.; Ollis, A.A.; Chang, S.; Khan, D.; Hosford, J.; Latham, J.; Ihnken, L.A.F.; Brown, M.J.B.; Fuerst, D.; et al. Chiral synthesis of LSD1 inhibitor GSK2879552 enabled by directed evolution of an imine reductase. Nat. Catal. 2019, 2, 909–915. [Google Scholar] [CrossRef]
- Mitsukura, K.; Suzuki, M.; Shinoda, S.; Kuramoto, T.; Yoshida, T.; Nagasawa, T. Purification and Characterization of a Novel (R)-Imine Reductase from Streptomyces sp. GF3587. Biosci. Biotechnol. Biochem. 2011, 75, 1778–1782. [Google Scholar] [CrossRef] [PubMed]
- Grogan, G.; Turner, N.J. InspIRED by Nature: NADPH-Dependent Imine Reductases (IREDs) as Catalysts for the Preparation of Chiral Amines. Chem. Eur. J. 2016, 22, 1900–1907. [Google Scholar] [CrossRef]
- Grogan, G. Synthesis of chiral amines using redox biocatalysis. Curr. Opin. Chem. Biol. 2018, 43, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Wetzl, D.; Berrera, M.; Sandon, N.; Fishlock, D.; Ebeling, M.; Müller, M.; Hanlon, S.; Wirz, B.; Iding, H. Expanding the Imine Reductase Toolbox by Exploring the Bacterial Protein-Sequence Space. ChemBioChem 2015, 16, 1749–1756. [Google Scholar] [CrossRef]
- Wetzl, D.; Gand, M.; Ross, A.; Müller, H.; Matzel, P.; Hanlon, S.P.; Müller, M.; Wirz, B.; Höhne, M.; Iding, H. Asymmetric Reductive Amination of Ketones Catalyzed by Imine Reductases. ChemCatChem 2016, 8, 2023–2026. [Google Scholar] [CrossRef]
- Wohlgemuth, R. Horizons of Systems Biocatalysis and Renaissance of Metabolite Synthesis. Biotechnol. J. 2018, 13, 1700620:1–1700620:14. [Google Scholar] [CrossRef]
- Gauss, D.; Schönenberger, B.; Molla, G.S.; Kinfu, B.M.; Chow, J.; Liese, A.; Streit, W.R.; Wohlgemuth, R. Biocatalytic Phosphorylation of Metabolites. In Applied Biocatalysis: From Fundamental Science to Industrial Applications; Hilterhaus, L., Liese, A., Kettling, U., Antranikian, G., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2016; pp. 147–177. [Google Scholar] [CrossRef]
- Hardt, N.; Kinfu, B.M.; Chow, J.; Schoenenberger, B.; Streit, W.R.; Obkircher, M.; Wohlgemuth, R. Biocatalytic Asymmetric Phosphorylation Catalyzed by Recombinant Glycerate-2-Kinase. ChemBioChem 2017, 18, 1518–1522. [Google Scholar] [CrossRef]
- Matsumi, R.; Hellriegel, C.; Schoenenberger, B.; Milesi, T.; van der Oost, J.; Wohlgemuth, R. Biocatalytic asymmetric phosphorylation of mevalonate. RSC Adv. 2014, 4, 12989–12994. [Google Scholar] [CrossRef]
- Schoenenberger, B.; Wszolek, A.; Milesi, T.; Brundiek, H.; Obkircher, M.; Wohlgemuth, R. Synthesis of N-omega-Phospho-(L)-arginine by Biocatalytic Phosphorylation of (L)-Arginine. ChemCatChem 2017, 9, 121–126. [Google Scholar] [CrossRef]
- Schoenenberger, B.; Wszolek, A.; Meier, R.; Brundiek, H.; Obkircher, M.; Wohlgemuth, R. Recombinant AroL-Catalyzed Phosphorylation for the Efficient Synthesis of Shikimic Acid 3-Phosphate. Biotechnol. J. 2018, 13, 1700529:1–1700529:8. [Google Scholar] [CrossRef] [PubMed]
- Gauss, D.; Sanchez-Moreno, I.; Oroz-Guinea, I.; Garcia-Junceda, E.; Wohlgemuth, R. Phosphorylation Catalyzed by Dihydroxyacetone Kinase. Eur. J. Org. Chem. 2018, 23, 2892–2895. [Google Scholar] [CrossRef]
- Wohlgemuth, R.; Liese, A.; Streit, W. Biocatalytic Phosphorylations of Metabolites: Past, Present, and Future. Trends Biotechnol. 2017, 35, 452–465. [Google Scholar] [CrossRef]
- Wohlgemuth, R. Key advances in biocatalytic phosphorylations in the last two decades–Biocatalytic syntheses in vitro and biotransformations in-vivo (in humans). Biotechnol. J. 2020, in press. [Google Scholar] [CrossRef]
- Matsubara, K.; Koehling, R.; Schoenenberger, B.; Kouril, T.; Esser, D.; Braesen, C.; Siebers, B.; Wohlgemuth, R. One-step synthesis of 2-keto-3-deoxy-D-gluconate by biocatalytic dehydration of D-gluconate. J. Biotechnol. 2014, 191, 69–77. [Google Scholar] [CrossRef]
- Krevet, S.; Shen, L.; Bohnen, T.; Schoenenberger, B.; Meier, R.; Obkircher, M.; Bangert, K.; Koehling, R.; Allenspach, E.; Wohlgemuth, R.; et al. Enzymatic Synthesis of 2-Keto-3-Deoxy-6-Phosphogluconate by the 6-Phosphogluconate-Dehydratase From Caulobacter crescentus. Front. Bioeng. Biotechnol. 2020, 8, 185:1–185:10. [Google Scholar] [CrossRef]
- Shen, L.; Kohlhaas, M.; Enoki, J.; Meier, R.; Schoenenberger, B.; Wohlgemuth, R.; Kourist, R.; Niemeyer, F.; van Niekerk, D.; Braesen, C.; et al. A combined experimental and modelling approach for the Weimberg pathway optimisation. Nat. Commun. 2020, 11, 1098:1–1098:13. [Google Scholar] [CrossRef]
- Vogel, M.A.K.; Burger, H.; Schlager, N.; Meier, R.; Schonenberger, B.; Bisschops, T.; Wohlgemuth, R. Highly efficient and scalable chemoenzymatic syntheses of (R)- and (S)-lactaldehydes. React. Chem. Eng. 2016, 1, 156–160. [Google Scholar] [CrossRef]
- Schoenenberger, B.; Wszolek, A.; Meier, R.; Brundiek, H.; Obkircher, M.; Wohlgemuth, R. Biocatalytic asymmetric Michael addition reaction of L-arginine to fumarate for the green synthesis of N-(((4S)-4-amino-4-carboxy-butyl amino) iminomethyl)-L-aspartic acid lithium salt (L-argininosuccinic acid lithium salt). RSC Adv. 2017, 7, 48952–48957. [Google Scholar] [CrossRef]
- Fryszkowska, A.; Devine, P.N. Biocatalysis in drug discovery and development. Curr. Opin. Chem. Biol. 2020, 55, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Hohmann, E.; Chu, H.Y.; Luetkemeyer, A.; Kline, S.; et al. Remdesivir for the Treatment of Covid-19—Final Report. N. Eng. J. Med. 2020, 383, 1813–1826. [Google Scholar] [CrossRef] [PubMed]
- Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment. Available online: https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-issues-emergency-use-authorization-potential-covid-19-treatment (accessed on 10 November 2020).
- Warren, T.K.; Jordan, R.; Lo, M.K.; Ray, A.S.; Mackman, R.L.; Soloveva, V.; Siegel, D.; Perron, M.; Bannister, R.; Hui, H.C.; et al. Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature 2016, 531, 381–385. [Google Scholar] [CrossRef]
- Al Bujuq, N. Methods of Synthesis of Remdesivir, Favipiravir, Hydroxychloroquine, and Chloroquine: Four Small Molecules Repurposed for Clinical Trials during the Covid-19 Pandemic. Synthesis-Stuttgart 2020, 52, 3735–3750. [Google Scholar] [CrossRef]
- Liang, C.; Tian, L.; Liu, Y.; Hui, N.; Qiao, G.; Li, H.; Shi, Z.; Tang, Y.; Zhang, D.; Xie, X.; et al. A promising antiviral candidate drug for the COVID-19 pandemic: A mini-review of remdesivir. Eur. J. Med. Chem. 2020, 201, 112527:1–112527:15. [Google Scholar] [CrossRef] [PubMed]
- Bigley, A.N.; Narindoshvili, T.; Raushel, F.M. A Chemoenzymatic Synthesis of the (RP)-Isomer of the Antiviral Prodrug Remdesivir. Biochemistry 2020, 59, 3038–3043. [Google Scholar] [CrossRef] [PubMed]
- Paymode, D.J.; Cardoso, F.S.P.; Agrawal, T.; Tomlin, J.W.; Cook, D.W.; Burns, J.M.; Stringham, R.W.; Sieber, J.D.; Gupton, B.F.; Snead, D.R. Expanding Access to Remdesivir via an Improved Pyrrolotriazine Synthesis: Supply Centered Synthesis. Org. Lett. 2020, 22, 7656–7661. [Google Scholar] [CrossRef]
- Xue, F.; Zhou, X.; Zhou, R.; Zhou, X.; Xiao, D.; Gu, E.; Guo, X.; Xiang, J.; Wang, K.; Yang, L.; et al. Improvement of the C-glycosylation Step for the Synthesis of Remdesivir. Org. Process Res. Dev. 2020, 24, 1772–1777. [Google Scholar] [CrossRef]
- Vieira, T.; Stevens, A.C.; Chtchemelinine, A.; Gao, D.; Badalov, P.; Heumann, L. Development of a Large-Scale Cyanation Process Using Continuous Flow Chemistry En Route to the Synthesis of Remdesivir. Org. Process Res. Dev. 2020, 24, 2113–2121. [Google Scholar] [CrossRef]
- Schooley, R.T.; Carlin, A.F.; Beadle, J.R.; Valiaeva, N.; Zhang, X.-Q.; Garretson, A.F.; Smith, V.I.; Murphy, J.; Hostetler, K.Y. Rethinking Remdesivir: Synthesis of Lipid Prodrugs that Substantially Enhance Anti-Coronavirus Activity. bioRxiv 2020. [Google Scholar] [CrossRef]
- Kumar, A. Vincristine and vinblastine: A review. Int. J. Med. Pharm. Sci. 2016, 6, 23–30. [Google Scholar]
- Kuboyama, T.; Yokoshima, S.; Tokuyama, H.; Fukuyama, T. Stereocontrolled total synthesis of (+)-vincristine. Proc. Nat. Acad. Sci. USA 2004, 101, 11966–11970. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Colby, D.A.; Seto, S.; Va, P.; Tam, A.; Kakei, H.; Rayl, T.J.; Hwang, I.; Boger, D.L. Total Synthesis of Vinblastine, Vincristine, Related Natural Products, and Key Structural Analogues. J. Am. Chem. Soc. 2009, 131, 4904–4916. [Google Scholar] [CrossRef]
- Arya, S.S.; Rookes, J.E.; Cahill, D.M.; Lenka, S.K. Next-generation metabolic engineering approaches towards development of plant cell suspension cultures as specialized metabolite producing biofactories. Biotechnol. Adv. 2020, 45, 107635:1–107635:19. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Liao, P.; Nile, S.H.; Georgiev, M.I.; Kai, G. Biotechnological Exploration of Transformed Root Culture for Value-Added Products. Trends in Biotechnol. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Casini, A.; Chang, F.-Y.; Eluere, R.; King, A.M.; Young, E.M.; Dudley, Q.M.; Karim, A.; Pratt, K.; Bristol, C.; Forget, A.; et al. A Pressure Test to Make 10 Molecules in 90 Days: External Evaluation of Methods to Engineer Biology. J. Am. Chem. Soc. 2018, 140, 4302–4316. [Google Scholar] [CrossRef]
- Langlois, N.; Gueritte, F.; Langlois, Y.; Potier, P. Application of a modification of the Polonovski reaction to the synthesis of vinblastine-type alkaloids. J. Am. Chem. Soc. 1976, 98, 7017–7024. [Google Scholar] [CrossRef]
- Kutney, J.P.; Hibino, T.; Jahngen, E.; Okutani, T.; Ratcliffe, A.H.; Treasurywala, A.M.; Wunderly, S. Total synthesis of indole and dihydroindole alkaloids. IX. Studies on the synthesis of bisindole alkaloids in the vinblastine-vincristine series. The biogenetic approach. Helv. Chim. Acta 1976, 59, 2858–2882. [Google Scholar] [CrossRef]
- Gotoh, H.; Sears, J.E.; Eschenmoser, A.; Boger, D.L. New Insights into the Mechanism and an Expanded Scope of the Fe(III)-Mediated Vinblastine Coupling Reaction. J. Am. Chem. Soc. 2012, 134, 13240–13243. [Google Scholar] [CrossRef][Green Version]
- Boon, B.A.; Boger, D.L. Triarylaminium Radical Cation Promoted Coupling of Catharanthine with Vindoline: Diastereospecific Synthesis of Anhydrovinblastine and Reaction Scope. J. Am. Chem. Soc. 2019, 141, 14349–14355. [Google Scholar] [CrossRef]
- Sottomayor, M.; Lopez-Serrano, M.; DiCosmo, F.; Barcelo, A.R. Purification and characterization of alpha-3′,4′-anhydrovinblastine synthase (peroxidase-like) from Catharanthus roseus (L) G. Don. FEBS Lett. 1998, 428, 299–303. [Google Scholar] [CrossRef]
- Hamada, H.; Nakazawa, K. Biotransformation of vinblastine to vincristine by cell-suspension cultures of catharanthus-roseus. Biotechnol. Lett. 1991, 13, 805–806. [Google Scholar] [CrossRef]
- Kumar, A.; Ahmad, A. Biotransformation of vinblastine to vincristine by the endophytic fungus Fusarium oxysporum isolated from Catharanthus roseus. Biocatal. Biotransform. 2013, 31, 89–93. [Google Scholar] [CrossRef]
- Caputi, L.; Franke, J.; Farrow, S.C.; Chung, K.; Payne, R.M.E.; Trinh-Don, N.; Dang, T.-T.T.; Carqueijeiro, I.S.T.; Koudounas, K.; de Bernonville, T.D.; et al. Missing enzymes in the biosynthesis of the anticancer drug vinblastine in Madagascar periwinkle. Science 2018, 360, 1235–1238. [Google Scholar] [CrossRef] [PubMed]
- Bonrath, W.; Netscher, T. Catalytic processes in vitamins synthesis and production. Appl. Catal. A-Gen. 2005, 280, 55–73. [Google Scholar] [CrossRef]
- Netscher, T.; Bonrath, W.; Bendik, I.; Zimmermann, J.P.; Weber, F.; Rüttimann, A. Vitamins, 5. Vitamin K. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2005; pp. 1–25. [Google Scholar] [CrossRef]
- Gross, J.; Cho, W.K.; Lezhneva, L.; Falk, J.; Krupinska, K.; Shinozaki, K.; Seki, M.; Herrmann, R.G.; Meurer, J. A plant locus essential for phylloquinone (vitamin K-1) biosynthesis originated from a fusion of four eubacterial genes. J. Biol. Chem. 2006, 281, 17189–17196. [Google Scholar] [CrossRef]
- Tarento, T.D.C.; McClure, D.D.; Talbot, A.M.; Regtop, H.L.; Biffin, J.R.; Valtchev, P.; Dehghani, F.; Kavanagh, J.M. A potential biotechnological process for the sustainable production of vitamin K-1. Crit. Rev. Biotechnol. 2019, 39, 1–19. [Google Scholar] [CrossRef]
- Liu, J.-M.; Solem, C.; Jensen, P.R. Harnessing biocompatible chemistry for developing improved and novel microbial cell factories. Microb. Biotechnol. 2019, 13, 54–66. [Google Scholar] [CrossRef]
- Tarento, T.D.C.; McClure, D.D.; Dehghani, F.; Kavanagh, J.M. Pilot-scale production of phylloquinone (vitamin K-1) using a bubble column photo-bioreactor. Biochem. Eng. J. 2019, 150, 107243:1–107243:6. [Google Scholar] [CrossRef]
- Hecht, K. Innovation in Biocatalysis—A Swiss Network Project Coordinated by the Competence Center for Biocatalysis (CCBIO). Chimia 2017, 71, 866–871. [Google Scholar] [CrossRef]
- NCCR Master in Chemical Biology. Available online: https://nccr-chembio.ch/education-training/masters/ (accessed on 3 November 2020).
- Major Biological Chemistry. Available online: https://ethz.ch/content/dam/ethz/special-interest/biol/department/BiologicalChemistry.pdf (accessed on 3 November 2020).
- Biochemie—Chemische Biologie. Available online: https://ethz.ch/content/dam/ethz/special-interest/chab/chab-dept/studies/documents/C_CI/WL_BSc_BCB_okt2020.pdf (accessed on 3 November 2020).
- INNOSUISSE. Available online: https://www.innosuisse.ch/inno/en/home.html (accessed on 9 November 2020).
- BRIDGE. Available online: https://www.bridge.ch/en/ (accessed on 9 November 2020).
- Hardy, M.A.; Wright, B.A.; Bachman, J.L.; Boit, T.B.; Haley, H.M.S.; Knapp, R.R.; Lusi, R.F.; Okada, T.; Tona, V.; Garg, N.K.; et al. Treating a Global Health Crisis with a Dose of Synthetic Chemistry. ACS Cent. Sci. 2020, 6, 1017–1030. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.P.; Brown, M.J.B.; Diaz-Rodriguez, A.; Lloyd, R.C.; Roiban, G.D. Biocatalysis: A Pharma Perspective. Adv. Synth. Catal. 2019, 361, 2421–2432. [Google Scholar] [CrossRef]
- Wohlgemuth, R. Building Bridges between Biotechnology and Chemistry—Oreste Ghisalba’s Pioneering Activities, Publications and Programs. Chimia 2020, 74, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Wohlgemuth, R. Biocatalysis—Key enabling tools from biocatalytic one-step and multi-step reactions to biocatalytic total synthesis. N. Biotechnol. 2020, 60, 113–123. [Google Scholar] [CrossRef]





| Product | Application | Structure | Comment |
|---|---|---|---|
| Adenine | Starting material for e.g., Tenovir |  | Periodic shortage resulting in spiking prices; produced by fermentation. |
| Benzodiazepin | Psychoactive drug (sedative, anxiolytic) | 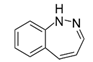 | First synthesized by Hoffmann-La Roche in the 1950s. |
| Dextran | Treatment of hypovolemia (decreased volume of circulating blood plasma) |  | Low molecular dextran or α-D-1,6- polyisomaltose produced by fermentation with Leuconostoc mesenteroides. A shortage occurred because of quality issues with cheap material from India and China. |
| Imidazole | Topical formulations for fungal infections | 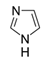 | - |
| Levothyroxine (Euthyrox®) | Thyroid hormone | 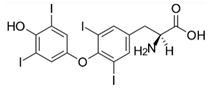 | - |
| Lamotrigine | Anti-epileptic drug |  | Approved since 1993. |
| Midazolam (Dormicum®) | Psychoactive drug | 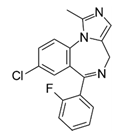 | Short-acting benzodiazepine derivative with an imidazole structure. This relaxant is used during intubations and its scarcity was a problem during the first COVID-19 wave (Midazolam, Remifentanil and Profol) |
| Mefenamic acid (Ponstan®) | Nonsteroidal anti-inflammatory drug | 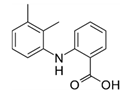 | Recall because of lead and lithium impurities found in the drug formulations. |
| Melphalan (Alkeran®) | Cytostatic drug |  | Example of an indispensable drug which was periodically not available. |
| Paracetamol (Dafalgan®) | Non-opioid analgesic | 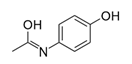 | Panic buying sprees during the first wave of the COVID-19 pandemic caused shortages. |
| Piperacillin/Tazobactam | Combination of a broad spectrum β-lactam antibiotic (top) with a β-lactamase inhibitor (bottom) | 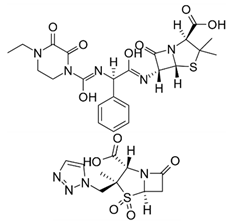 | Semisynthetic ampicillin derived antibiotic/penicillanic acid sulfone derivative. An explosion in a Chinese factory interrupted supplies. |
| Propofol | Parenteral anesthetic | 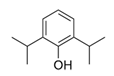 | Relaxant used for intubations during the first COVID-19 pandemic wave. |
| Remdesivir | Antiviral | 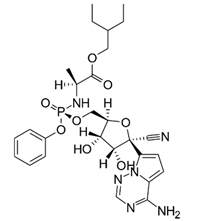 | ATP analogue; described to work against Ebola and tested against the SARS-CoV-2 virus. |
| Remifentanil | Short acting synthetic opioid analgesic |  | Relaxant used for intubations during the first COVID-19 pandemic wave. |
| Rifampicin | Broad spectrum antibiotic | 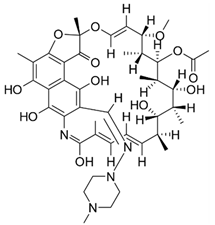 | Semisynthetic antibiotic derived from Amylatopsis rifamycinia; macrocyclic antibiotic. |
| Felbamate (Taloxa®) | Anti-epileptic drug | 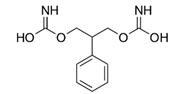 | - |
| Lorazepam (Temesta®) | Sedative | 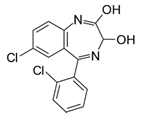 | - |
| Oxytocin (Syntocinon®) | Gynecological drug | 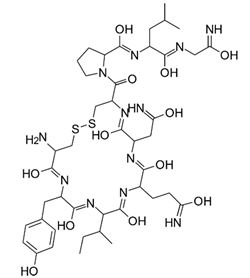 | Cyclic nonapeptide used for the improvement of uterine contractions. |
| Vincristine | Anti-cancer drug | 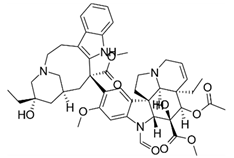 | Alkaloid isolated from the periwinkle Catharanthus roseus; inexpensive drug; Teva stopped production for the USA. |
| Vitamin K1 | Fat-soluble vitamin | 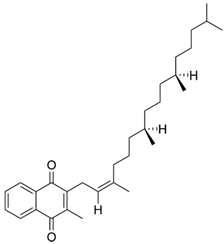 | Required in blood clotting; due to shortages much more expensive Vitamin K1 formulations had to be administered. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hecht, K.; Meyer, H.-P.; Wohlgemuth, R.; Buller, R. Biocatalysis in the Swiss Manufacturing Environment. Catalysts 2020, 10, 1420. https://doi.org/10.3390/catal10121420
Hecht K, Meyer H-P, Wohlgemuth R, Buller R. Biocatalysis in the Swiss Manufacturing Environment. Catalysts. 2020; 10(12):1420. https://doi.org/10.3390/catal10121420
Chicago/Turabian StyleHecht, Katrin, Hans-Peter Meyer, Roland Wohlgemuth, and Rebecca Buller. 2020. "Biocatalysis in the Swiss Manufacturing Environment" Catalysts 10, no. 12: 1420. https://doi.org/10.3390/catal10121420
APA StyleHecht, K., Meyer, H.-P., Wohlgemuth, R., & Buller, R. (2020). Biocatalysis in the Swiss Manufacturing Environment. Catalysts, 10(12), 1420. https://doi.org/10.3390/catal10121420