Eco-Friendly Cotton/Linen Fabric Treatment Using Aqueous Ozone and Ultraviolet Photolysis
Abstract
1. Introduction
2. Results
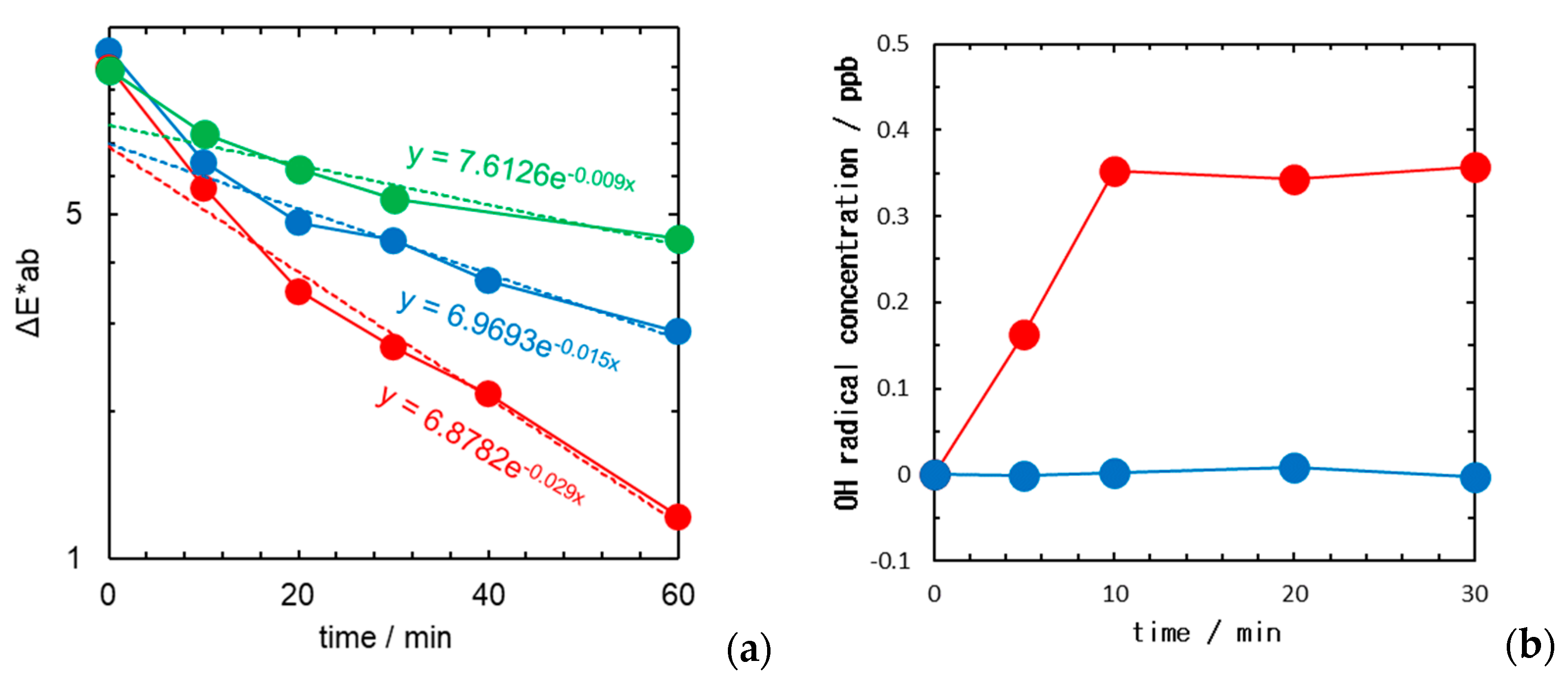
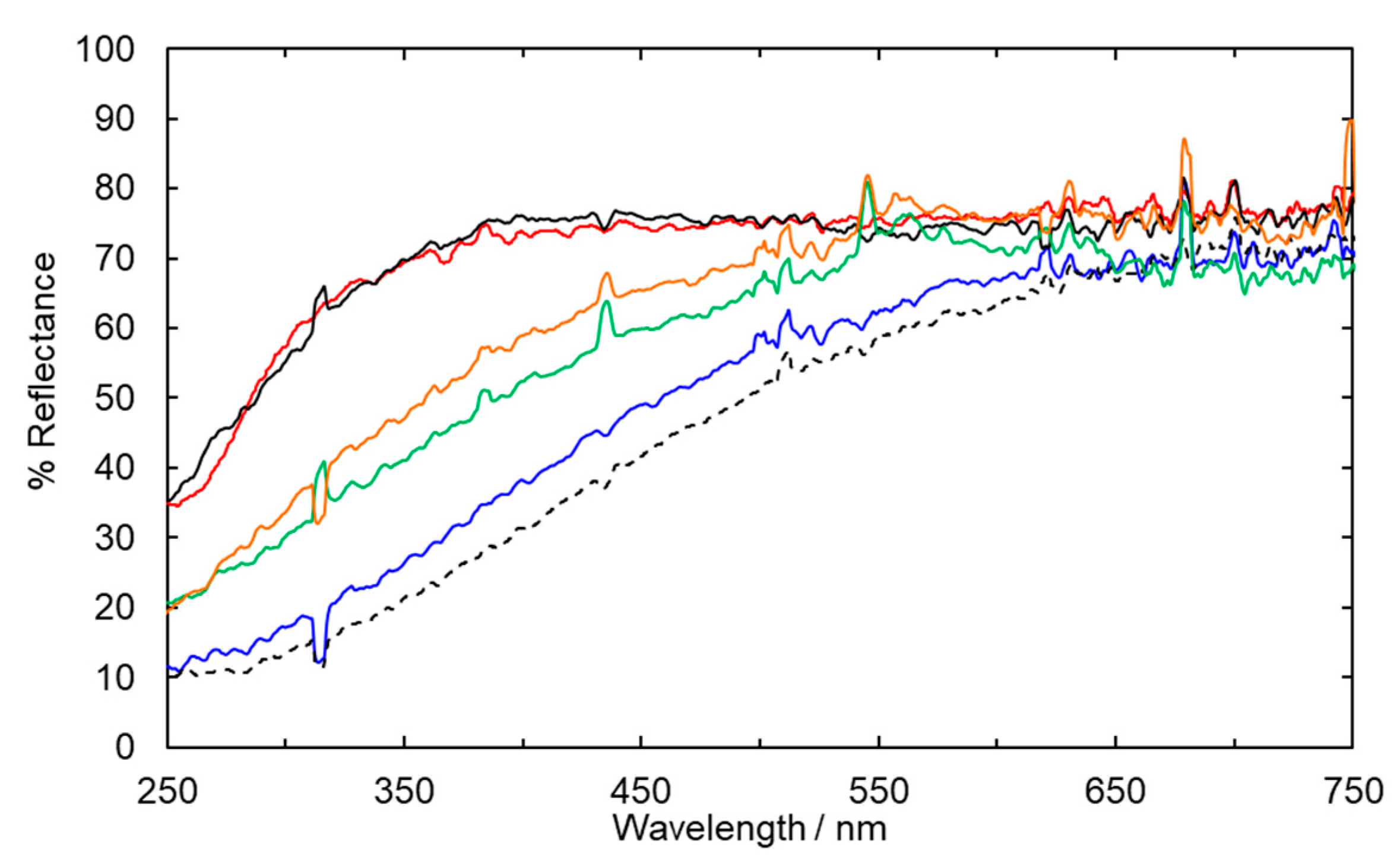
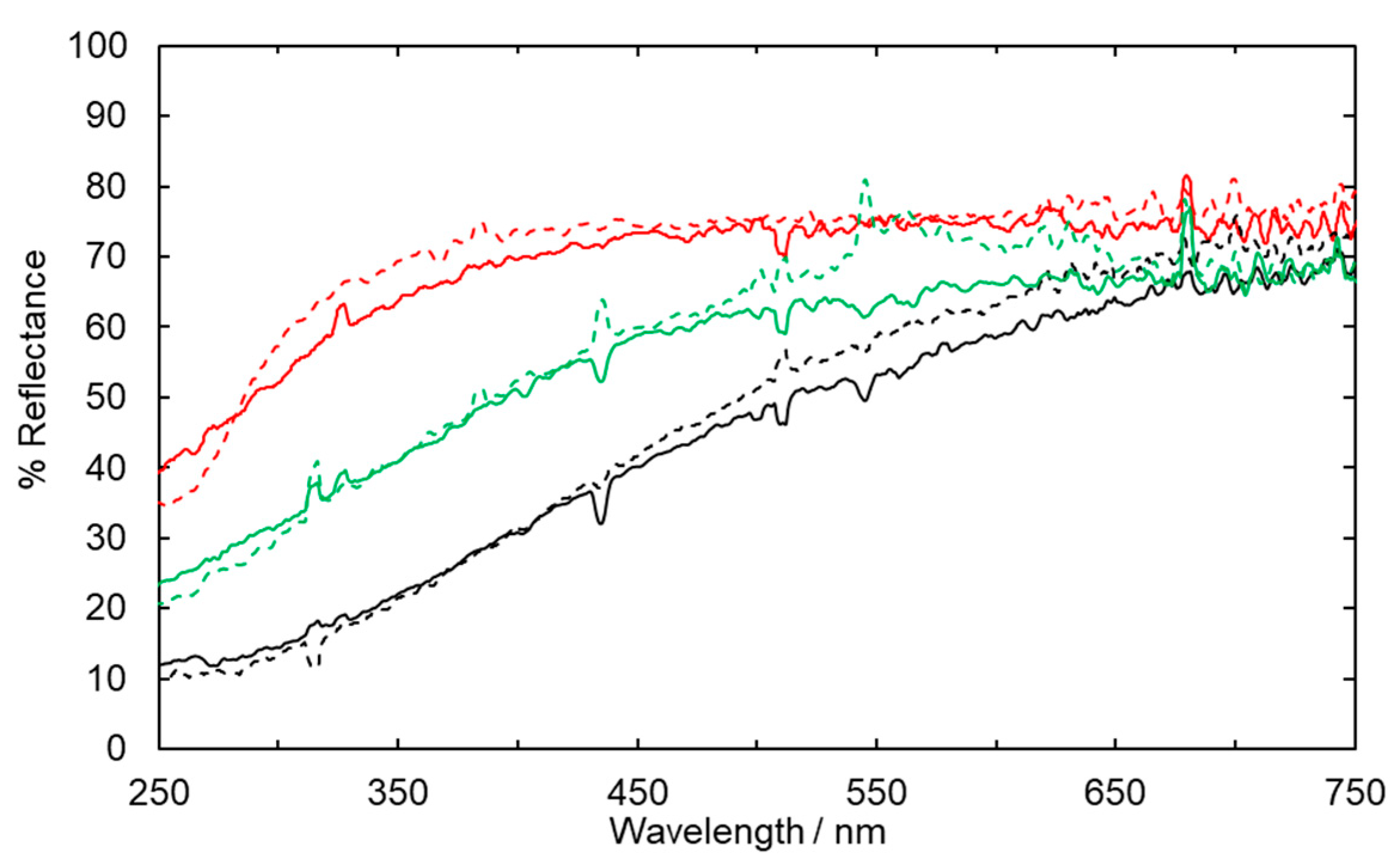
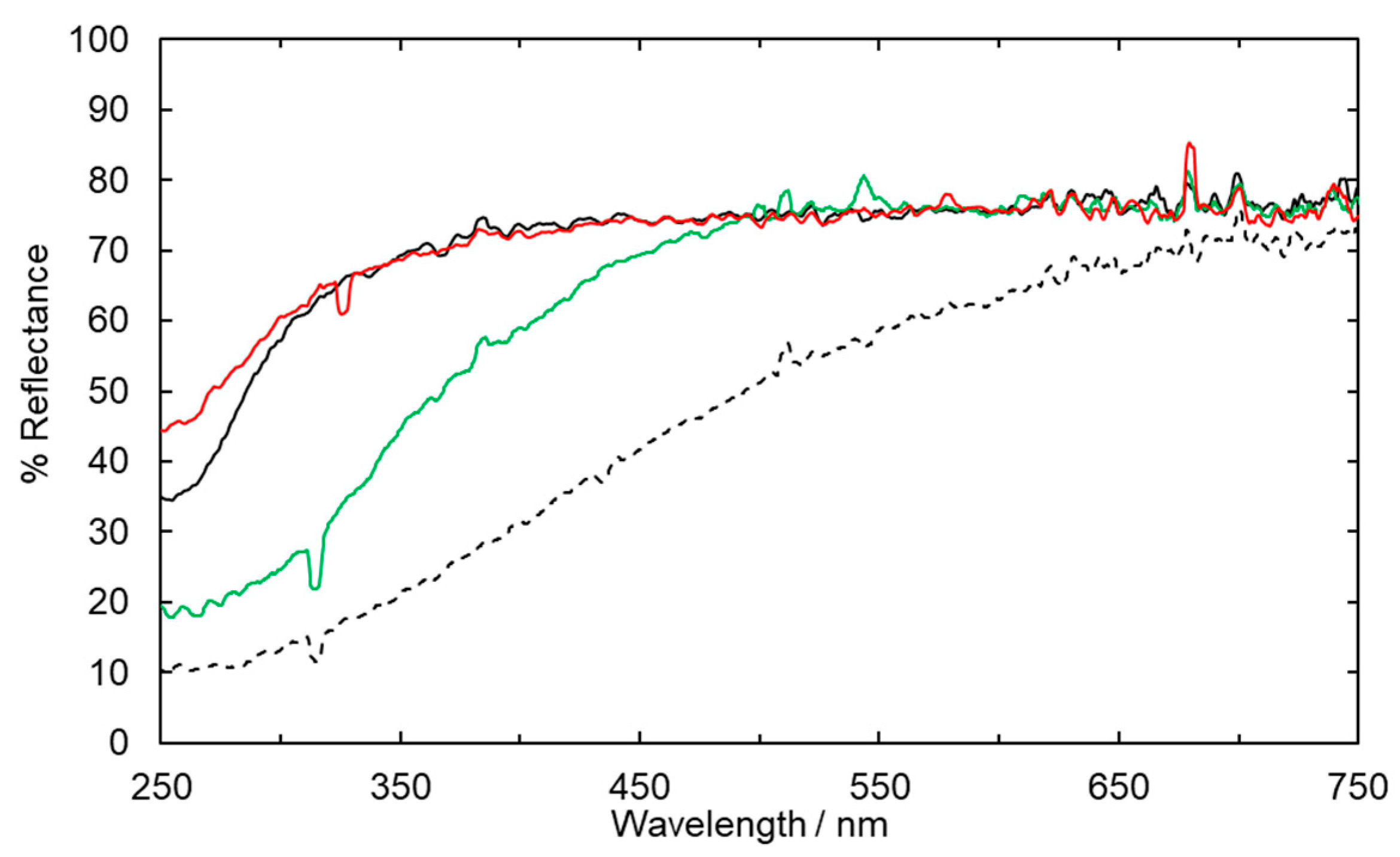
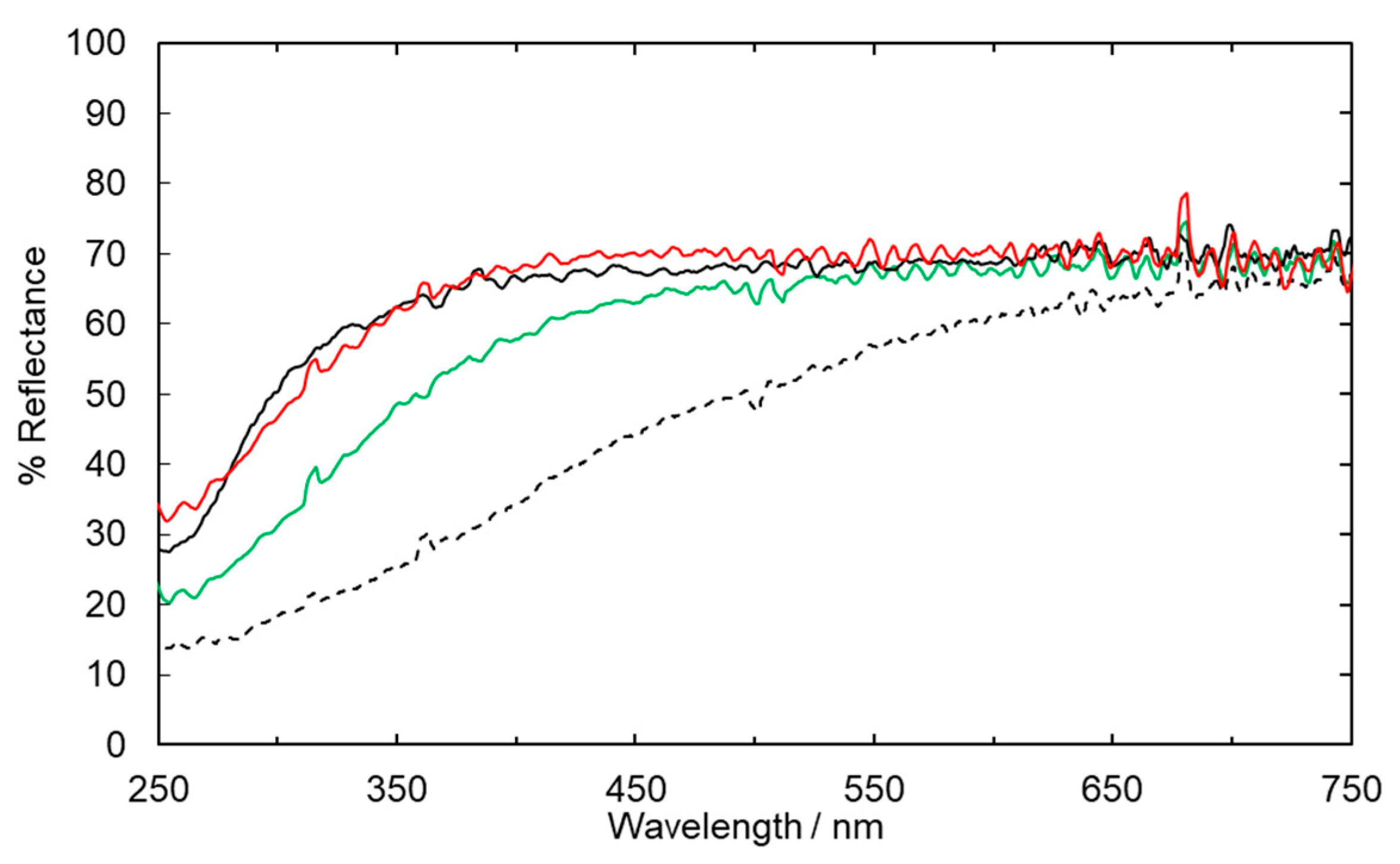
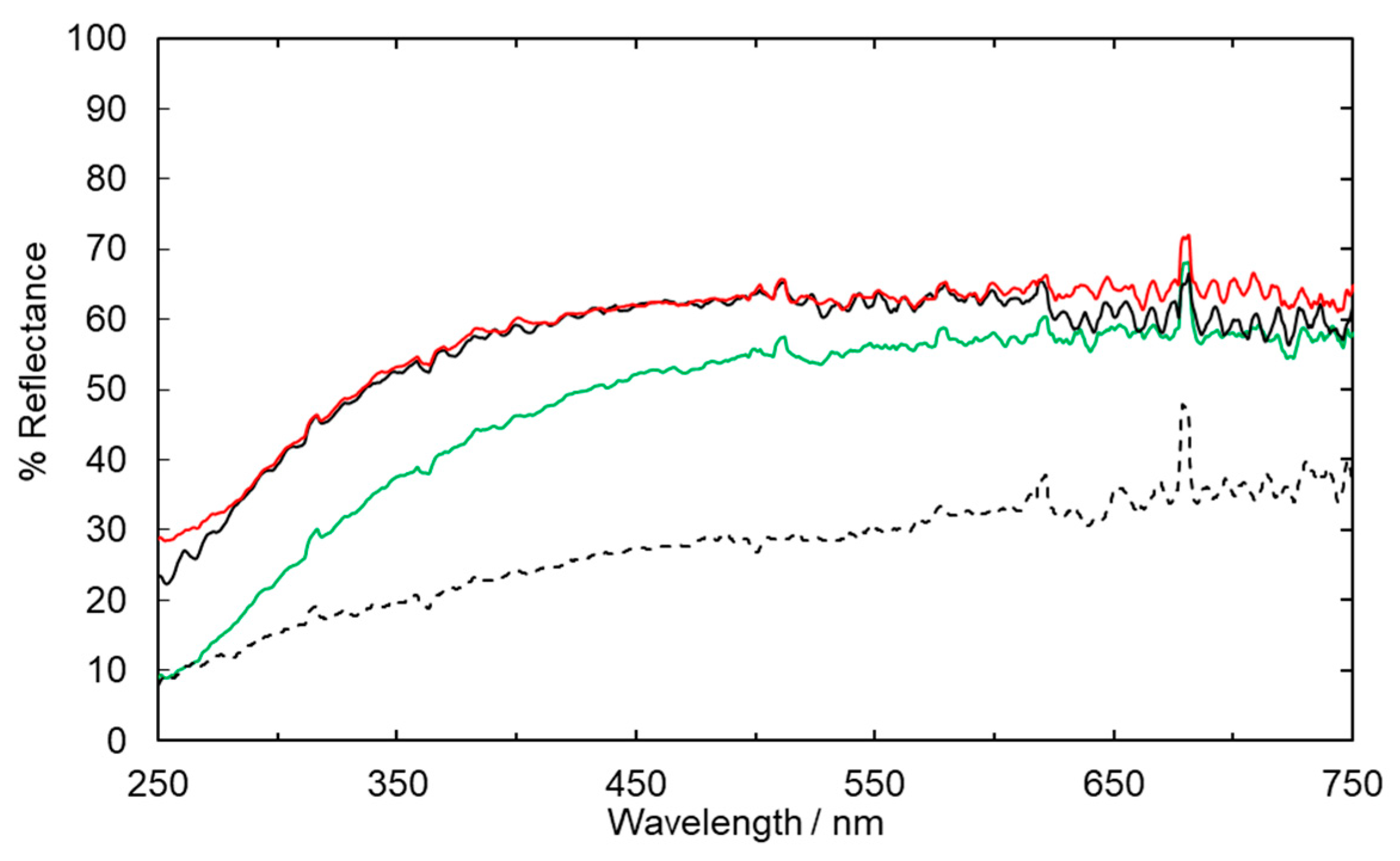
2.1. Fabric Bleaching Using Aqueous Ozone under UV Irradiation
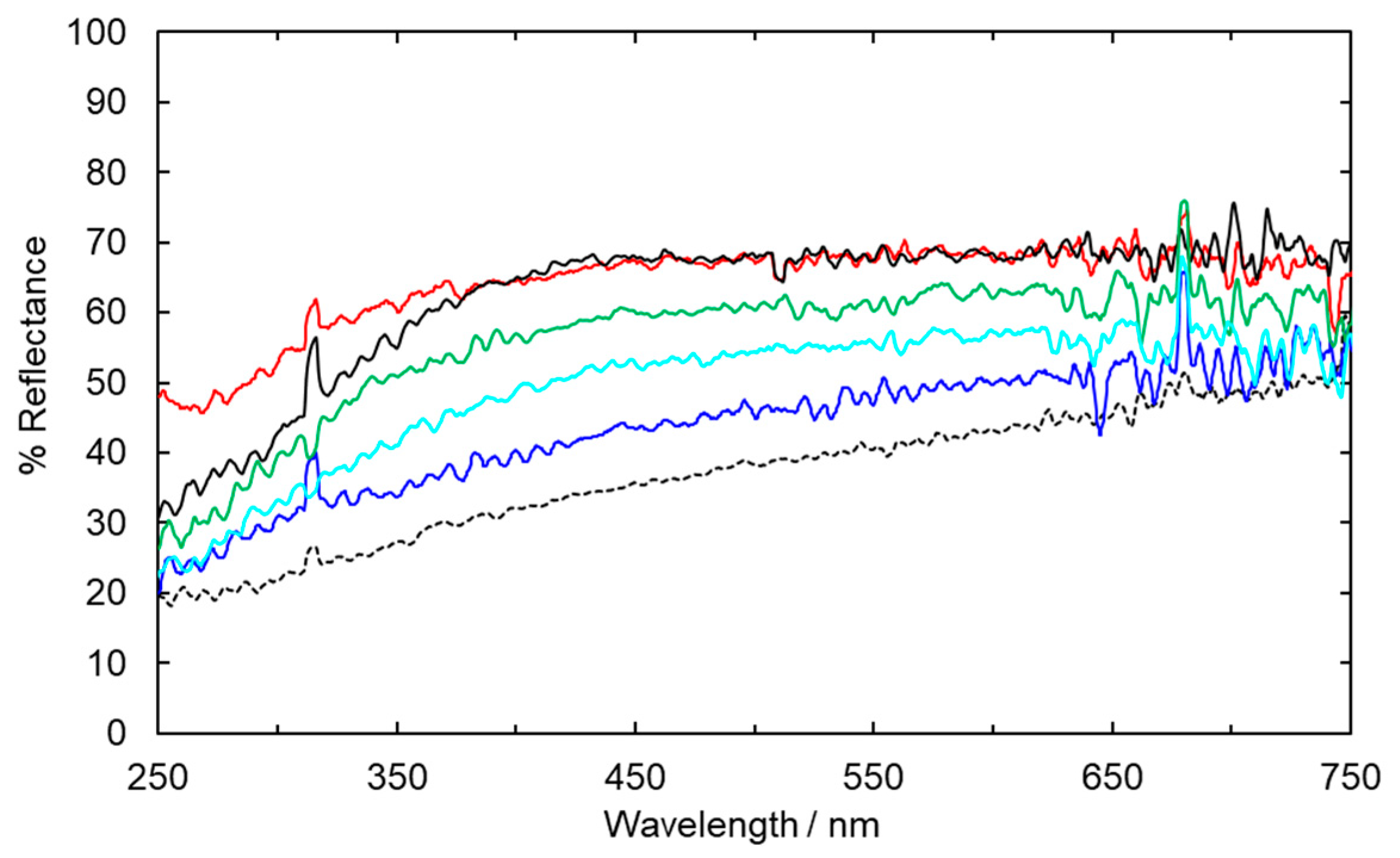
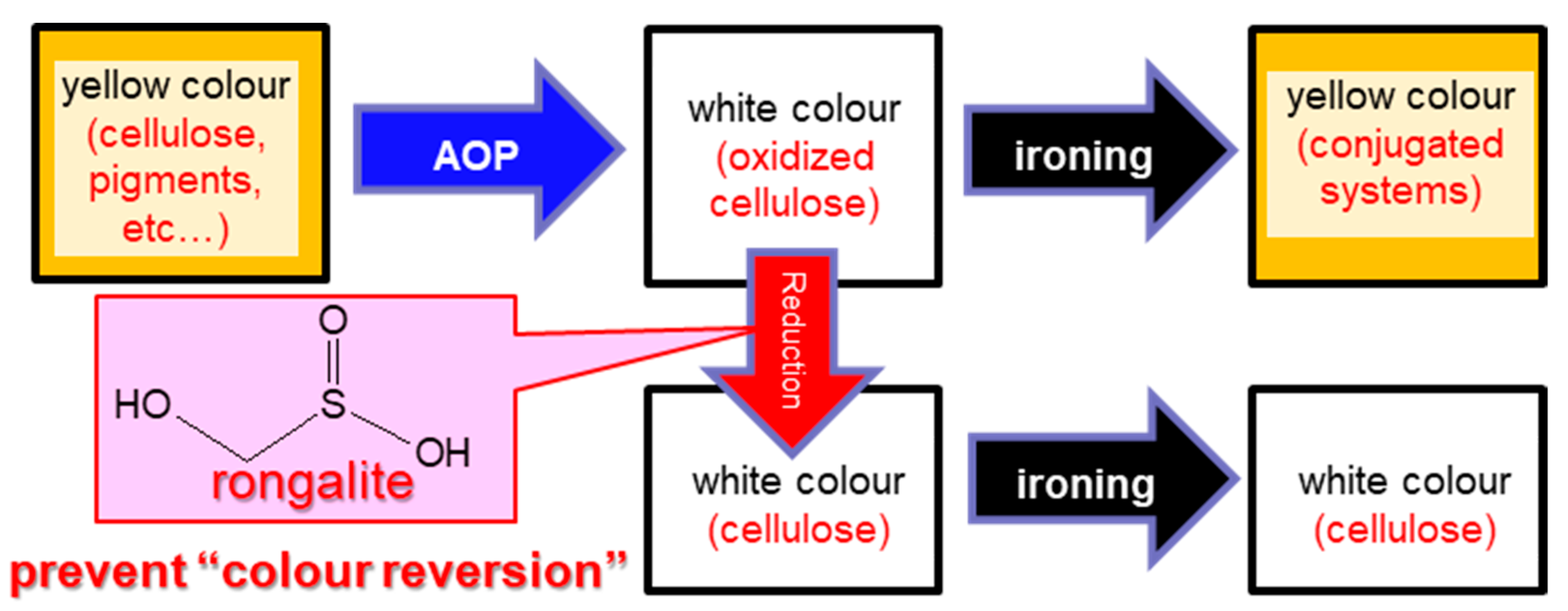
2.2. Suppression of Colour Reversion Using Rongalite Treatment
3. Discussion
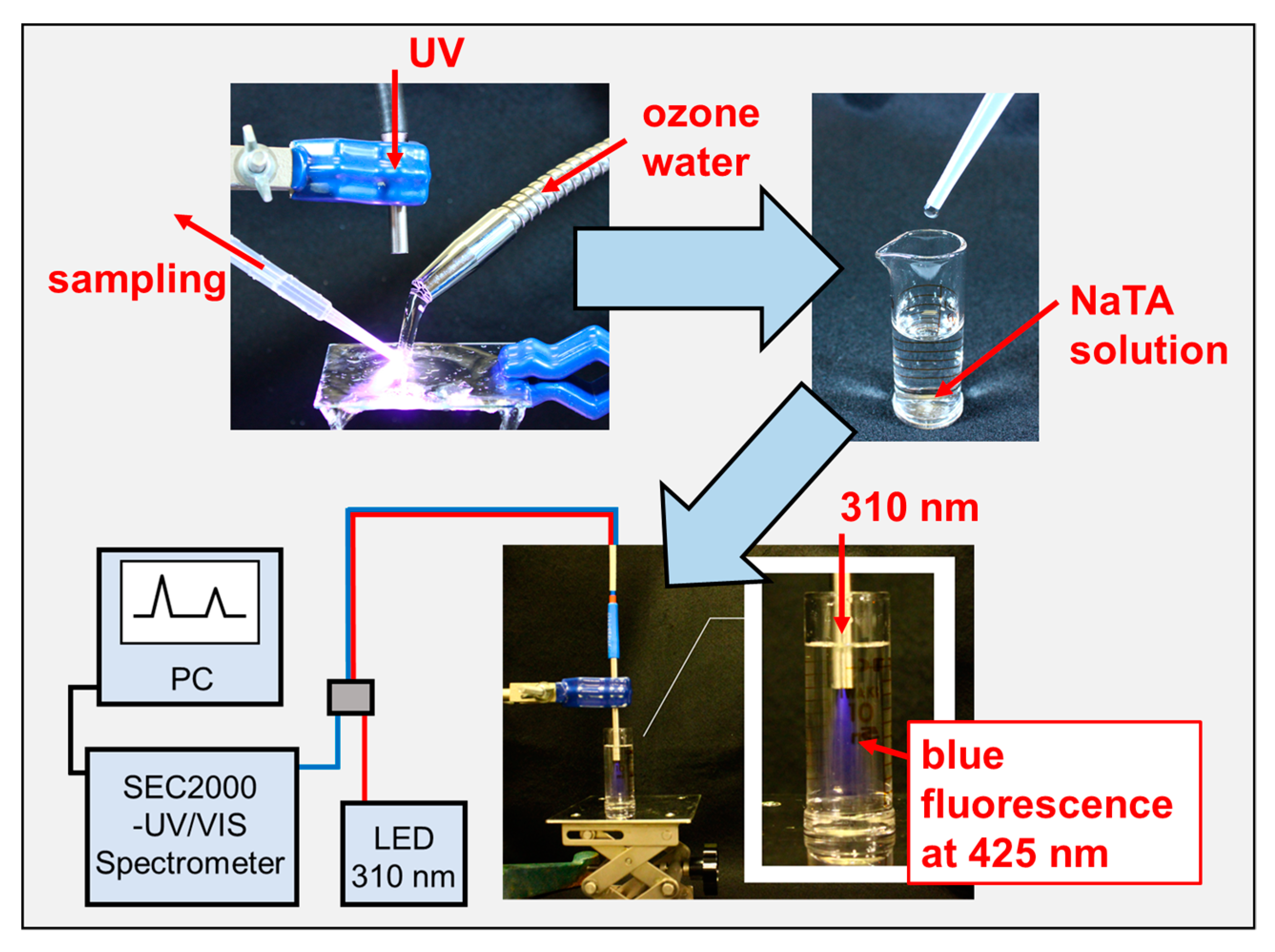
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abdel-Halim, E.S. An effective redox system for bleaching cotton cellulose. Carbohydr. Polym. 2012, 90, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Hebeish, A.; Hashem, M.; Shaker, N.; Ramadan, M.; El-Sadek, B.; Hady, M.A. New development for combined bioscouring and bleaching of cotton-based fabrics. Carbohydr. Polym. 2009, 78, 961–972. [Google Scholar] [CrossRef]
- Lim, S.-H.; Gürsoy, N.Ç.; Hauser, P.; Hinks, D. Performance of a new cationic bleach activator on a hydrogen peroxide bleaching system. Coloration Technol. 2004, 120, 114–118. [Google Scholar] [CrossRef]
- Shafie, A.E.; Fouda, M.M.G.; Hashem, M. One-step process for bio-scouring and peracetic acid bleaching of cotton fabric. Carbohydr. Polym. 2009, 78, 302–308. [Google Scholar] [CrossRef]
- Yao, Y.; Kubota, Y.; Murakami, T.; Ochiai, T.; Ishiguro, H.; Nakata, K.; Fujishima, A. Electrochemical Inactivation Kinetics of Boron-Doped Diamond Electrode on Waterborne Pathogens. J. Water Health 2011, 9, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, T.; Tago, S.; Hayashi, M.; Hirota, K.; Kondo, T.; Satomura, K.; Fujishima, A. Boron-doped diamond powder (BDDP)-based polymer composites for dental treatment using flexible pinpoint electrolysis unit. Electrochem. Commun. 2016, 68, 49–53. [Google Scholar] [CrossRef]
- Ouchi, A. Photochemical Reactions and their Application to Textile Processing. Oleoscience 2015, 15, 455–460. [Google Scholar] [CrossRef]
- Abdel-Halim, E.S.; Al-Deyab, S.S. One-step bleaching process for cotton fabrics using activated hydrogen peroxide. Carbohydr. Polym. 2013, 92, 1844–1849. [Google Scholar] [CrossRef]
- Li, Q.; Ni, L.; Wang, J.; Quan, H.; Zhou, Y. Establishing an ultrasound-assisted activated peroxide system for efficient and sustainable scouring-bleaching of cotton/spandex fabric. Ultrason. Sonochem. 2020, 68, 105220. [Google Scholar] [CrossRef]
- Perincek, S.; Bahtiyari, I.; Korlu, A.; Duran, K. New Techniques in Cotton Finishing. Text. Res. J. 2009, 79, 121–128. [Google Scholar] [CrossRef]
- Perincek, S.D.; Duran, K.; Korlu, A.E.; Bahtiyari, İ.M. An Investigation in the Use of Ozone Gas in the Bleaching of Cotton Fabrics. Ozone Sci. Eng. 2007, 29, 325–333. [Google Scholar] [CrossRef]
- Piccoli, H.H.; Ulson de Souza, A.A.; Ulson de Souza, S.M.A.G. Bleaching of Knitted Cotton Fabric Applying Ozone. Ozone Sci. Eng. 2015, 37, 170–177. [Google Scholar] [CrossRef]
- Hashem, M.; Taleb, M.A.; El-Shall, F.N.; Haggag, K. New prospects in pretreatment of cotton fabrics using microwave heating. Carbohydr. Polym. 2014, 103, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Farooq, A.; Ali, S.; Abbas, N.; Fatima, G.A.; Ashraf, M.A. Comparative performance evaluation of conventional bleaching and enzymatic bleaching with glucose oxidase on knitted cotton fabric. J. Clean. Prod. 2013, 42, 167–171. [Google Scholar] [CrossRef]
- He, Y.; Wang, X.; Xu, J.; Yan, J.; Ge, Q.; Gu, X.; Jian, L. Application of integrated ozone biological aerated filters and membrane filtration in water reuse of textile effluents. Bioresour. Technol. 2013, 133, 150–157. [Google Scholar] [CrossRef]
- Buffle, M.-O.; Schumacher, J.; Meylan, S.; Jekel, M.; von Gunten, U. Ozonation and Advanced Oxidation of Wastewater: Effect of O3 Dose, pH, DOM and HO•-Scavengers on Ozone Decomposition and HO• Generation. Ozone Sci. Eng. 2006, 28, 247–259. [Google Scholar] [CrossRef]
- Hayashi, H.; Akamine, S.; Ichiki, R.; Kanazawa, S. Comparison of OH radical concentration generated by underwater discharge using two methods. Int. J. Plasma Environ. Sci. Technol. 2016, 10, 24–28. [Google Scholar]
- Kanazawa, S.; Kawano, H.; Watanabe, S.; Furuki, T.; Akamine, S.; Ichiki, R.; Ohkubo, T.; Kocik, M.; Mizeraczyk, J. Observation of OH radicals produced by pulsed discharges on the surface of a liquid. Plasma Sources Sci. Technol. 2011, 20, 034010. [Google Scholar] [CrossRef]
- Sharma, V.K.; Graham, N.J.D. Oxidation of Amino Acids, Peptides and Proteins by Ozone: A Review. Ozone Sci. Eng. 2010, 32, 81–90. [Google Scholar] [CrossRef]
- Ikehata, K.; Jodeiri Naghashkar, N.; Gamal El-Din, M. Degradation of Aqueous Pharmaceuticals by Ozonation and Advanced Oxidation Processes: A Review. Ozone: Sci. Eng. 2006, 28, 353–414. [Google Scholar] [CrossRef]
- Matsumi, Y.; Comes, F.J.; Hancock, G.; Hofzumahaus, A.; Hynes, A.J.; Kawasaki, M.; Ravishankara, A.R. Quantum yields for production of O(1D) in the ultraviolet photolysis of ozone: Recommendation based on evaluation of laboratory data. J. Geophys. Res. Atmos. 2002, 107, 1–12. [Google Scholar] [CrossRef]
- Attri, P.; Kim, Y.H.; Park, D.H.; Park, J.H.; Hong, Y.J.; Uhm, H.S.; Kim, K.-N.; Fridman, A.; Choi, E.H. Generation mechanism of hydroxyl radical species and its lifetime prediction during the plasma-initiated ultraviolet (UV) photolysis. Sci. Rep. 2015, 5, 9332. [Google Scholar] [CrossRef] [PubMed]
- Goswami, K.K. Bleaching of linen (Linum usitatissimum). Indian J. Fibre Texlie Res. 1993, 18, 82–86. [Google Scholar]
- Yui, Y.; Tanaka, C.; Isogai, A. Functionalization of Cottton Fabrics by TEMPO-Mediated Oxidation. SENI GAKKAISHI 2013, 69, 222–228. [Google Scholar] [CrossRef]
- Arooj, F.; Ahmad, N.; Shaikh, I.A.; Chaudhry, M.N. Application of ozone in cotton bleaching with multiple reuse of a water bath. Text. Res. J. 2013, 84, 527–538. [Google Scholar] [CrossRef]
- Japanese Standards Association. JIS Z 8722 Methods of Colour Measurement-Reflecting and Transmitting Objects; Japanese Standards Association: Tokyo, Japan, 2009. [Google Scholar]
- Ochiai, T.; Aoki, D.; Saito, H.; Akutsu, Y.; Nagata, M. Analysis of Adsorption and Decomposition of Odour and Tar Components in Tobacco Smoke on Non-Woven Fabric-Supported Photocatalysts. Catalysts 2020, 10, 304. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamada, K.; Ochiai, T.; Tsuchida, Y.; Miyano, K.; Ishikawa, Y.; Nagura, T.; Kimura, N. Eco-Friendly Cotton/Linen Fabric Treatment Using Aqueous Ozone and Ultraviolet Photolysis. Catalysts 2020, 10, 1265. https://doi.org/10.3390/catal10111265
Hamada K, Ochiai T, Tsuchida Y, Miyano K, Ishikawa Y, Nagura T, Kimura N. Eco-Friendly Cotton/Linen Fabric Treatment Using Aqueous Ozone and Ultraviolet Photolysis. Catalysts. 2020; 10(11):1265. https://doi.org/10.3390/catal10111265
Chicago/Turabian StyleHamada, Kengo, Tsuyoshi Ochiai, Yasuyuki Tsuchida, Kyohei Miyano, Yosuke Ishikawa, Toshinari Nagura, and Noritaka Kimura. 2020. "Eco-Friendly Cotton/Linen Fabric Treatment Using Aqueous Ozone and Ultraviolet Photolysis" Catalysts 10, no. 11: 1265. https://doi.org/10.3390/catal10111265
APA StyleHamada, K., Ochiai, T., Tsuchida, Y., Miyano, K., Ishikawa, Y., Nagura, T., & Kimura, N. (2020). Eco-Friendly Cotton/Linen Fabric Treatment Using Aqueous Ozone and Ultraviolet Photolysis. Catalysts, 10(11), 1265. https://doi.org/10.3390/catal10111265






