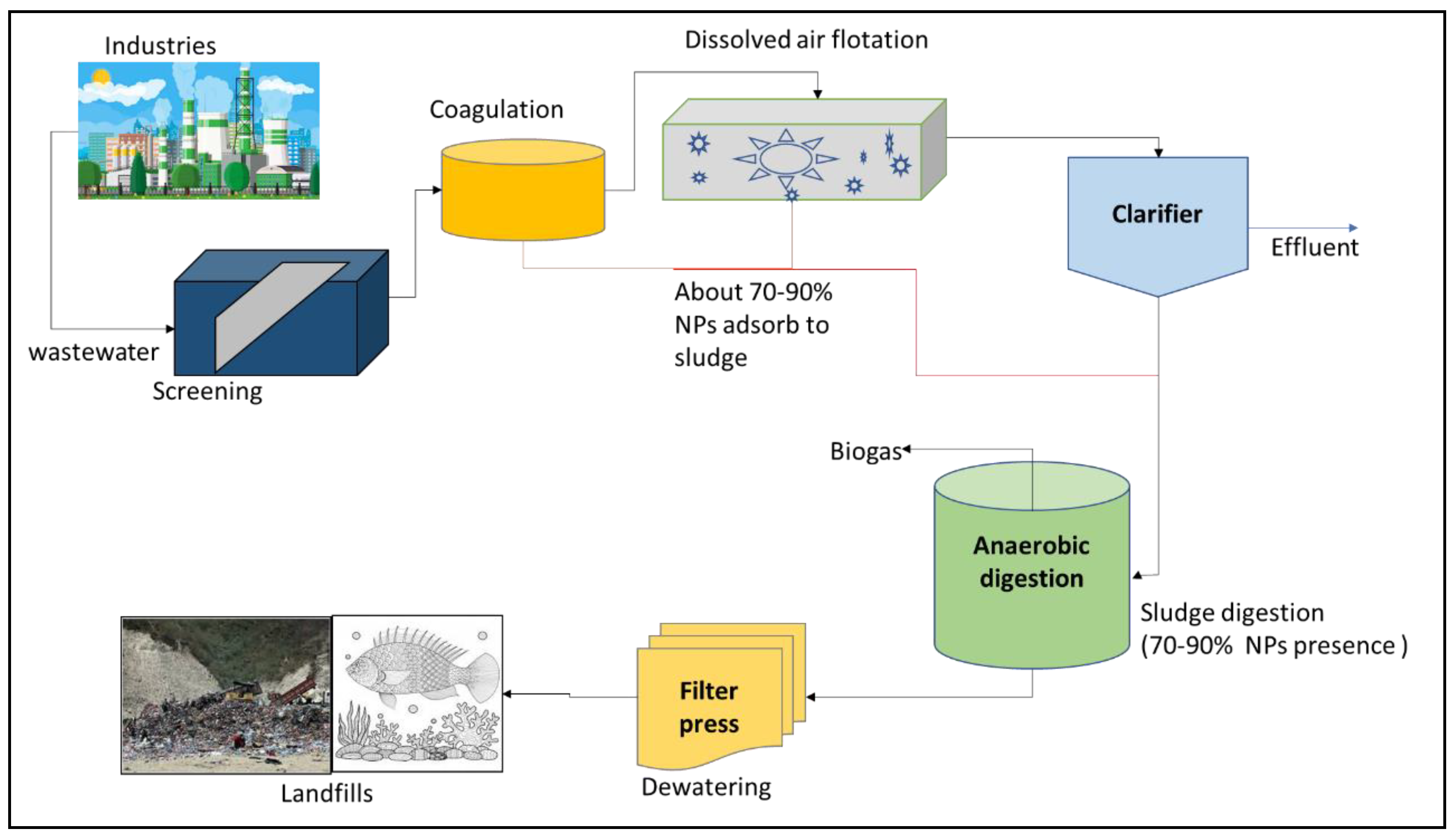
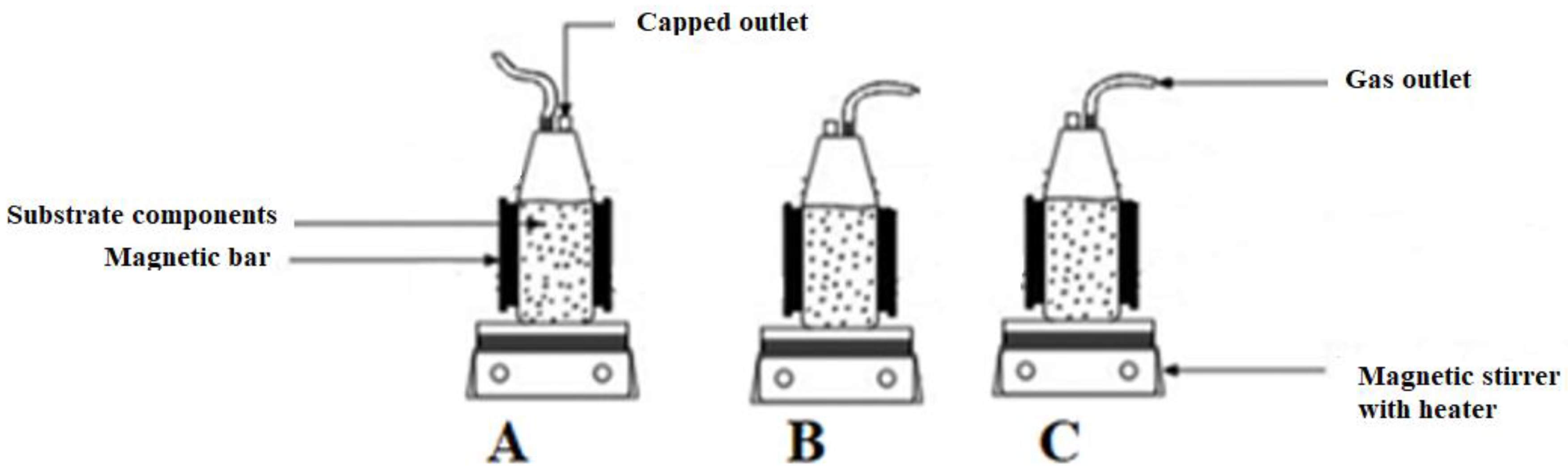
With the rapid development and application of nanotechnology towards green energy [
30,
33], the safety and toxicology of some of these engineered nanomaterials are of environmental concern. In conventional WWTPs, accumulation and adsorption of high amounts of heavy metals or nanomaterials in the sludge might affect the downstream treatment process [
6,
29,
30]. Successively, some of these nanomaterials (iron-based) are likely to be utilized as micronutrients by microbial biomass, whereas others can exhibit an inhibitory impact on the AD process [
27,
28,
29]. Thus, the substrate variation of content in the sludge during the degradation by the microbes can result in different intermediate end products such as sugars, fatty acids, alcohols, carbonic acids and amino acids [
6,
30]. This has led to intensive research on the degradation of sewage sludge with other waste streams by AD processes towards environmental protection, economical and eco-friendly energy [
27,
29]. Nevertheless, this study affirms the presence of heavy metals and nanomaterials in a digester might decrease the AD efficiency [
24,
27,
30]. Thus, the addition of nanoparticles in AD had a synergic effect on the microorganisms for the biogas production [
29].
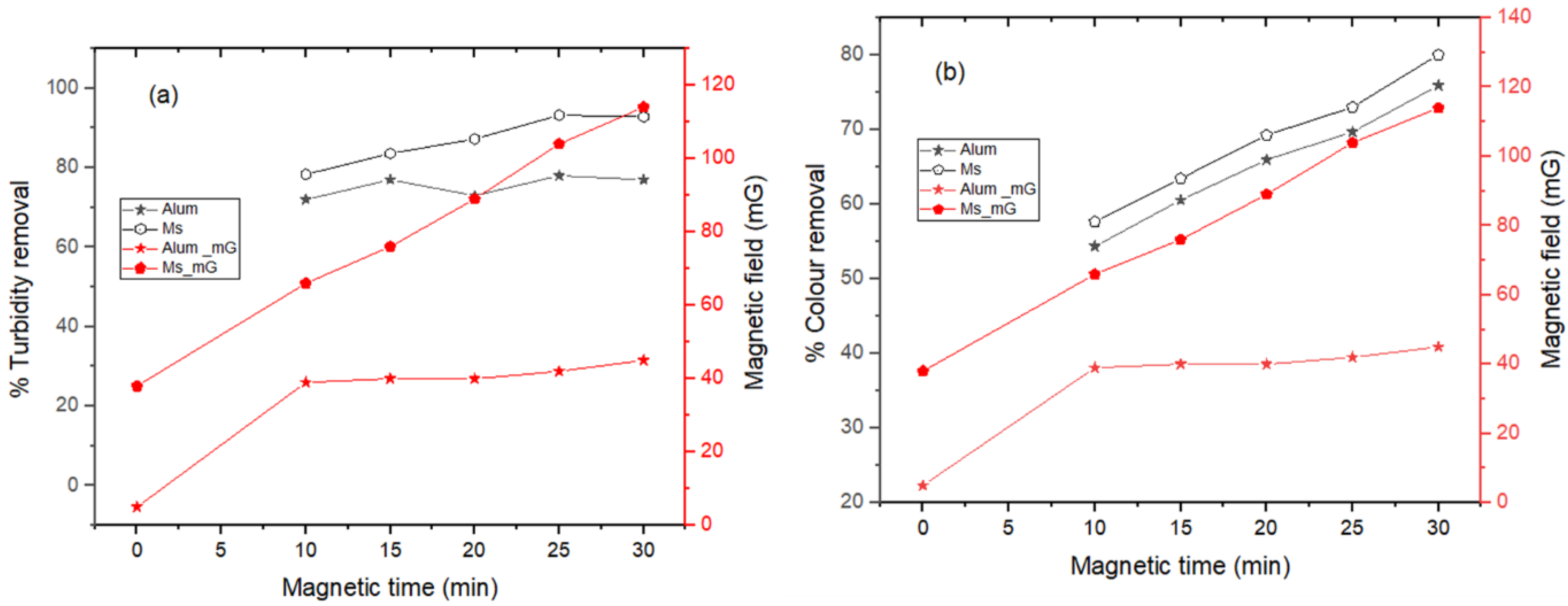
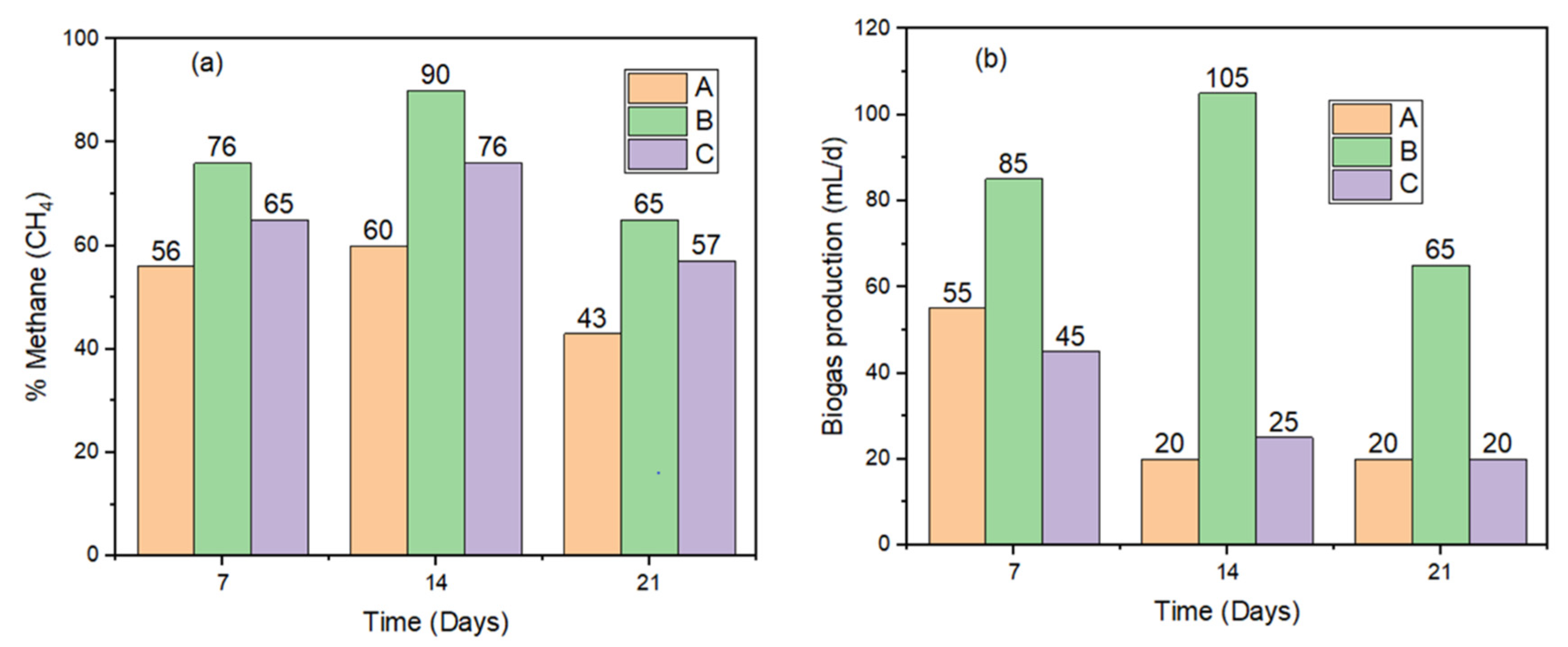
The impacts of the NPs on the setups were ascertained based on the volume of biogas production and methane composition. In
Figure 6 and
Table 4, it is evident that biogas production increased more with time for the setup (B) with Ms as compared to the control (C), and that of set-up (A) was the lowest. The addition of alum to setup (A) contributed to the production of volatile fatty acids and other organic intermediates (SO
4−) [
30,
31,
32]. This mechanism, along with metal toxicity, resulted in methanogenic bacteria inhibition. Additional conversion of the substrate to volatile fatty acid by the acidogenic bacteria also contributed to acidification and system failure in setup (A). Based on
Figure 7 and
Table 4, an enhancement of the biogas volume of production and methane composition was observed when the systems were exposed to external magnetic fields. Thus, the systems under the anaerobic conditions with the magnetic field influenced the growth and multiplicity of the microbes’ activities.
The magnetic field strength of the system reduced the lag phase and increased biogas production and methane composition (
Figure 7). This is in accordance with Zhao et al. [
28] and Zaidi et al. [
30], who reported on the presence of Fe NPs, which provided suitable substrates for methanogenesis enhancement in AD systems. Seyedi et al. [
22] and Biswas [
33] also reported on the effects of trace metals such as iron and their capacity to reduce the lag phase of mixed culture, which caused the improvement in methane composition and the biogas production. Sreekanh and Sahu [
34] reported that NPs uptake may stimulate methanogens by increasing their metabolic intermediates and key enzymatic activities involved in the hydrolysis, acidification, and methanogenesis stages of AD. Here, Fe species acted as an active component, and the catalytic activities were governed by the trivalent and zero-valent (Fe
3+/Fe
0) ions ratio and their exposed surface area [
30,
34,
35]. Sreekanh and Sahu [
34] added 1.4 g of NPs to enhance biogas yield in a portable-food waste digester, while here the waste and environmental conditions of this study are different. Casals et al. [
36] reported that an Fe NPs dose of 0.5–16 mg/L increased cumulative CH
4 production, by reducing the CO
2 via a hydrogenation mechanism. This is in agreement with the current study, which confirms that the Ms increased the CH
4 potential by 20% (
Table 4). Therefore, there are high prospects of Fe NPs to be used as fuel catalysts to reduce CO
2 into CH
4 through the stability of the autotrophic methanogens [
30].
3.3.1. Sludge Surface Complexity and Traced Nanoparticles after Digestion
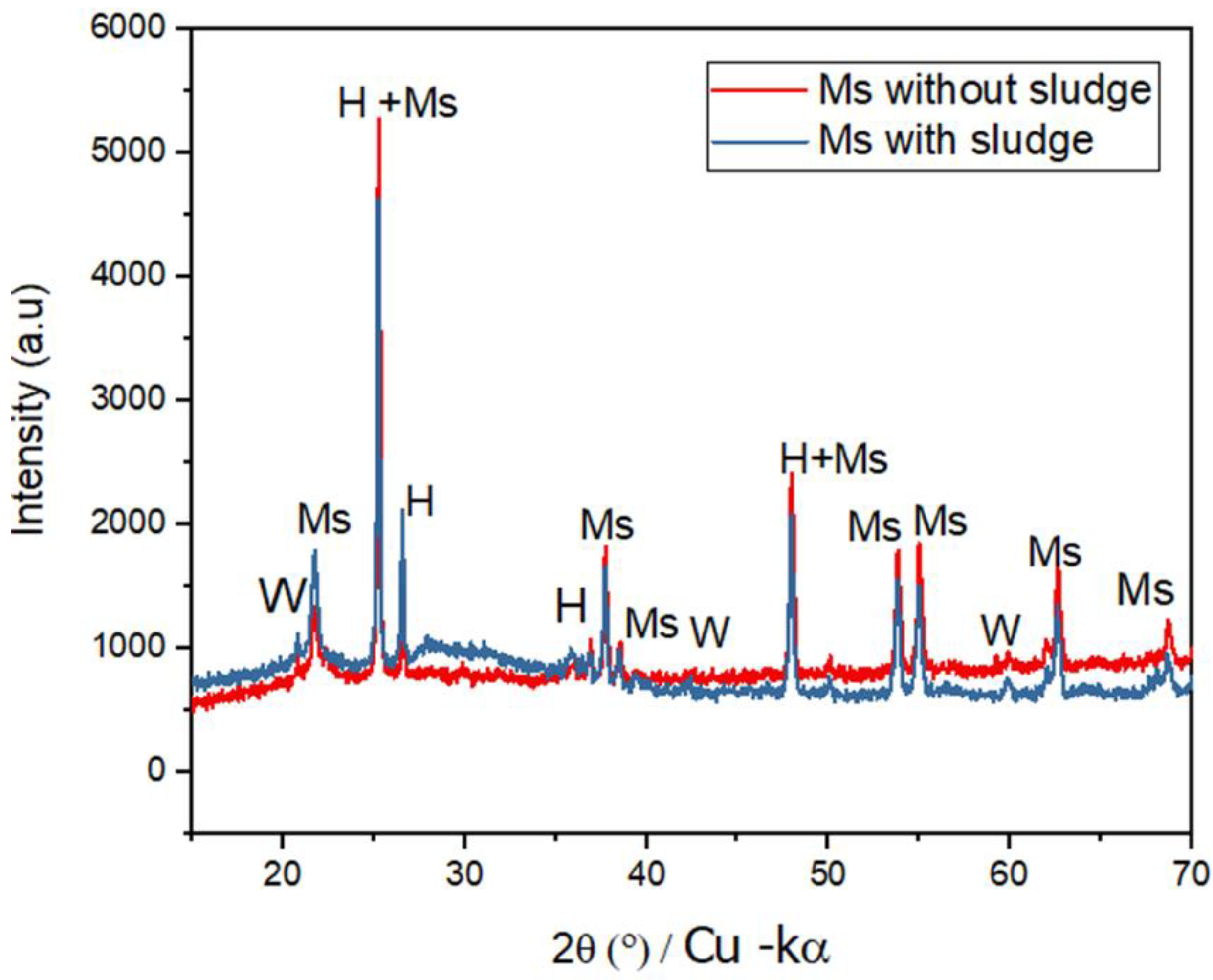
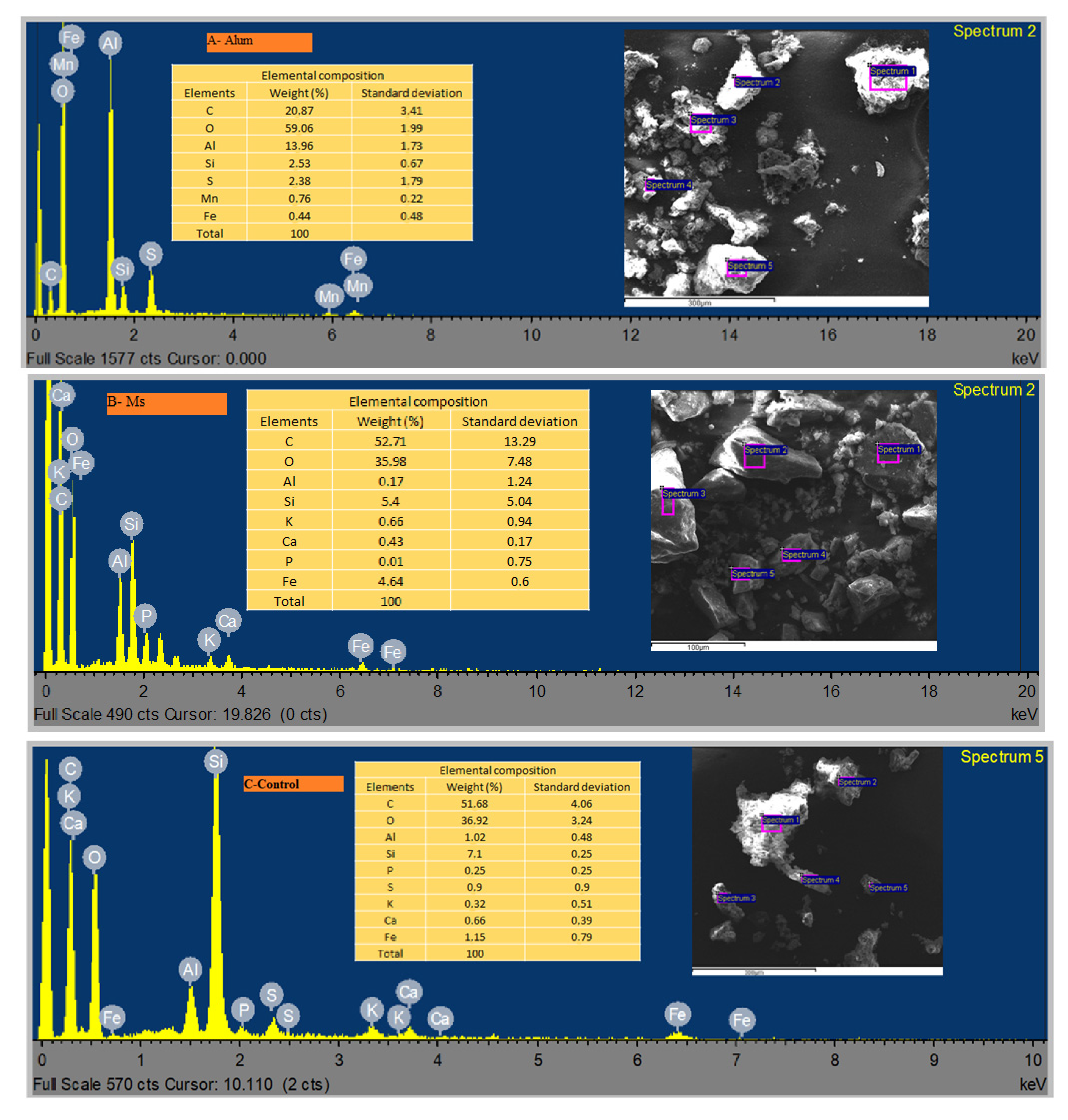
The SEM images (
Figure 8) present the morphology of the activated sludge aggregated with NPs of amorphous shapes with an average diameter less than 10 nm. The addition of the Fe/Al NPs increased the roughness of the activated sludge, with the development of cracks and macrospores. The pores of Ms (
Figure 8B) were larger than those of Alum (
Figure 8A), which is due to the replaceability of the Fe or Al species as ion donors on the surface of the activated sludge. This observation is in agreement with the EDS results (
Figure 9), in which similar peaks for the Fe element between Ms and Alum were observed.
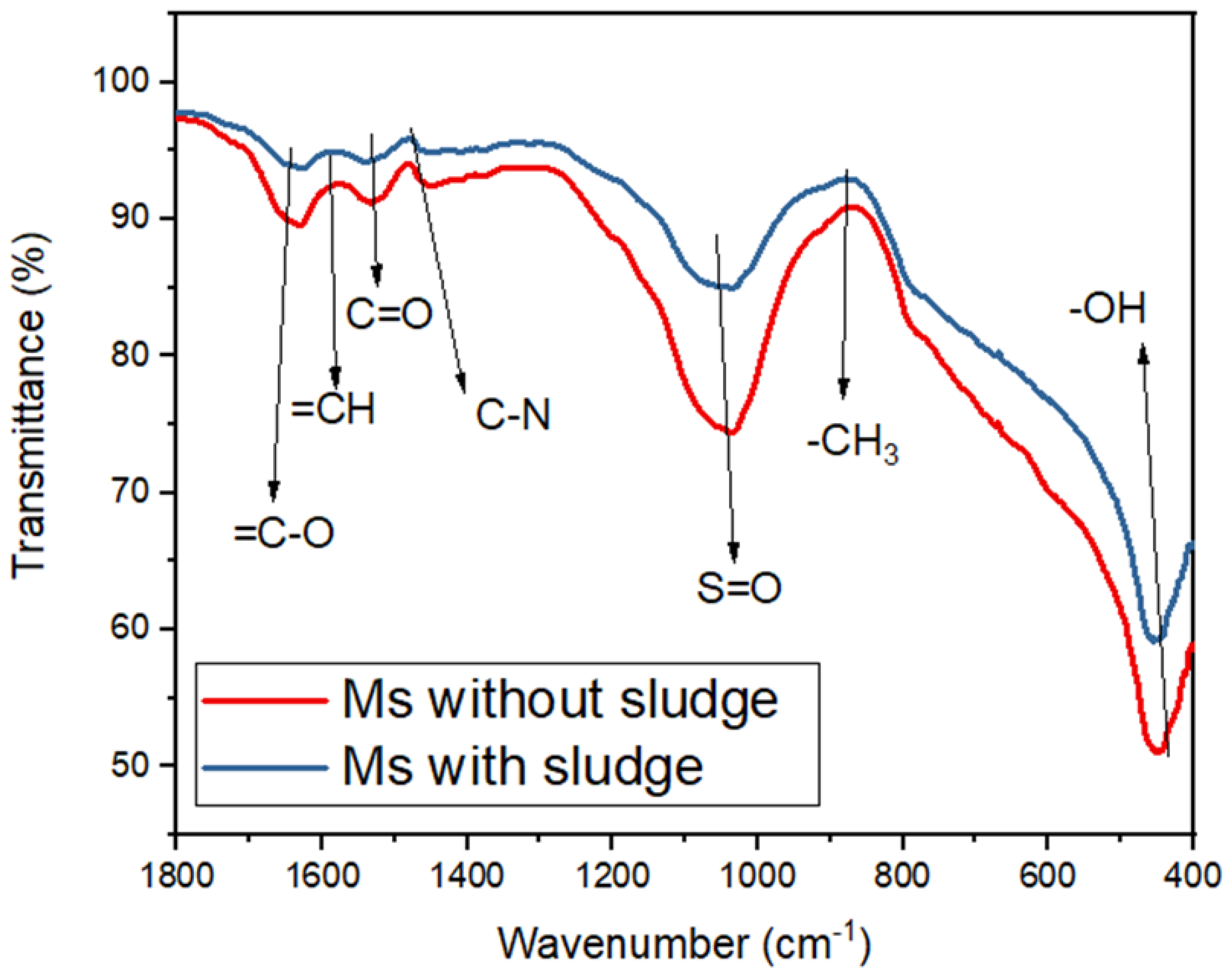
Figure 9 reveals the complexity of the metal ions or organic compounds on the surface of the activated sludge after the complexation reaction and adsorption. This was as a result of the interaction between the electron donors and electron acceptors being bound to the active groups on the activated sludge surface [
31,
33]. The control system (C) had a substrate without nanomaterials and resulted in a low biogas production volume and methane composition. Also, there were some elemental changes in the set-up systems as depicted in the electoral spectrum (
Figure 9), which includes carbonate (C), silicate (Si), sulfur (S), calcium (Ca), potassium (K), phosphorus (P), manganese (Mn), iron (Fe) and aluminum (Al) from the control. The toxicity levels of elementals accumulated in the sludge followed a decreasing order (
Figure 9A) in set-up (A); C > O > Al > Si > S > Mn > Fe;
Figure 9B for setup (B) as C > O > Al > Si > Fe > K > Ca > P and
Figure 9C representing set-up (C) as C > O > Si > Fe > Al > S > Ca > K > P. It can be seen that the low biogas yield (
Table 4) of the alum digester (A) can be attributed to the rapid poisoning of the active microbes in the digester by the SO
4− species (sulfide), which increased the acidity of the system.
Conversely, some of the elementals in the Ms digester (
Figure 10 B) acted as macro-nutrients (Fe, K, Ca, P) for simulating the methanogenic activity, creating active sites and stabilizing surface intermediate species. According to Casals et al., [
36], this can be due to the hydrolysis of soluble proteins and transformational activities of the electron donors, which were driven by redox–proton translocation in a methanogenic species. Furthermore, Zhao et al. [
28] indicated magnetic NPs (Ms), which seems to be non-toxic during long-term contact with bacterial activity and has mild toxicity at the initial stage (
Figure 8 and
Figure 9). However, the magnetic effects of the MBMP system influenced the Ms to enhance the activity of the autotrophic bacteria [
29,
30]. Thus, the propagation of autotrophic bacteria being attached to a carrier with a proton donor is positive for their growth and enrichment [
35,
36].
The two key tracing metal ions (Fe and Al) were assessed based on their distribution in the effluent. These were characterized to understand the possible mechanism of the heavy metal impact order in the supernatant. In
Table 4, it is shown that B—Ms (Al: 8 ± 4 mg/L; Fe: 6 ± 7 mg/L) affected the system positively, followed by C—control (Al: 30 ± 4 mg/L; Fe: 32 ± 3 mg/L), whereas A—Alum (Al: 36 ± 8 mg/L; Fe: 60 ± 16 mg/L) had a negative impact on the system. The Al active sites, besides the primary Fe sites on the surface of activated sludge (
Figure 9C) microspheres, facilitated the creation of Al–peroxide intermediates (Al–OOH), which reduced the apparent activation energy of the decomposition [
34,
35,
36]. The synergistic effect between the Fe and Al sites acted as reaction promoters for most of the metal oxides being used in CO
2 hydrogenation reactions [
34,
35].
As reported, nanomaterials are inhibitory to biodegradation, nitrification and the AD process [
24,
33]. Hence, understanding the complex mechanisms by which these particles interacted with the substrate and the microbes to enhance methanogenesis is very important. Notwithstanding their composition, nanoparticles have high surface area to volume ratios and particle size that can influence the rate of adsorption and biodegradation [
28,
30]. Additional combination of the substrate and the NPs resulted in functional groups containing oxides (Fe
2O
3), which heightened precipitation removal of the contaminants from the effluent [
31,
33]. According to Zhao et al., [
28], the interactions between the Ms and the activated sludge created an anoxic environment, which heterotrophic bacteria prefer, and thereby enhanced their activities. This adds to the advantages of Ms due to the large fraction and highly disperse nature (
Figure 8B) of the Fe species on the surface of the activated sludge (
Figure 9B), making it reachable for the reactants to adsorb.
3.3.2. Biogas Production Kinetics Using First-Order and Modified Gompertz Models
The availability of substrate for metabolism under anaerobic conditions with active microbial community enhanced the rate of biogas production. This mechanism was attributed to the four steps in the AD process: hydrolysis, acidogenesis, acetogenesis and methanogenesis [
30,
33]. The inhibitory effect of the nanoparticles in the AD biogas production was evaluated by the BMP test. To monitor the performance in terms of volume of biogas produced per day, the water displacement method was used. The measuring cylinder used for the gas collection was fixed with butyl rubber at the top to enable injection of the gas for analysis. From
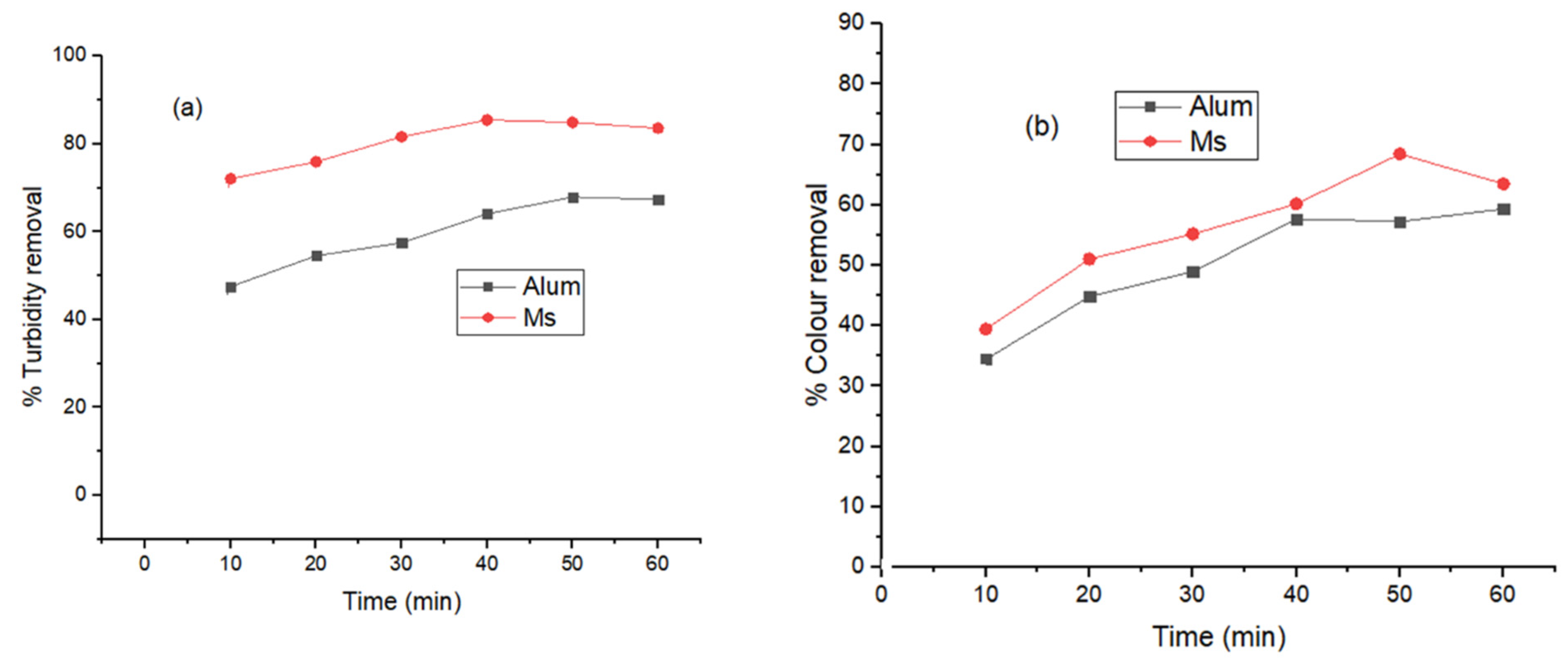
Figure 6, the average daily biogas production recorded was 25, 70 and 32 mL/day respectively for A—alum, B—Ms and C—Control. In
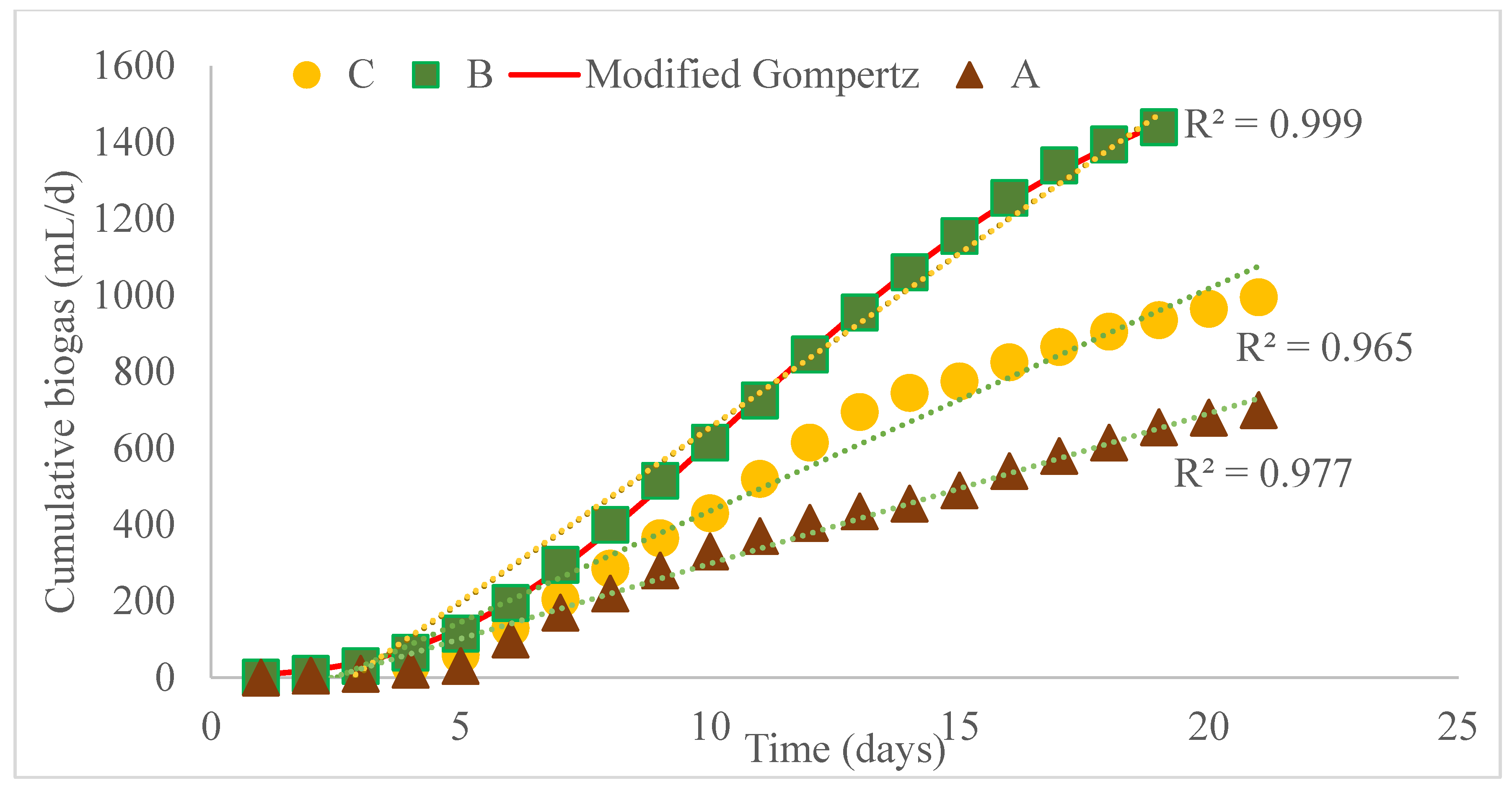
Table 4, organic degradation is also represented as 304 ± 92, 157 ± 8 and 258 ± 72 mg/L of COD respectively for A—alum, B—Ms and C—Control set-ups. The cumulative biogas production data (
Figure 10) obtained was simulated with the first-order kinetic model (5) and modified Gompertz model (6) constants obtained, presented in
Table 3.
Additives of the nanomaterials (Ms) enhanced the kinetics process of the acetotrophic and hydrogenotrophic methanogens in the digester for biogas and methane production. Thus, the electrons released or hydrogen generated by the Ms had a significant influence on the biogas and methane production (
Figure 3). Subsequently, methanogenic reactions under anaerobic conditions were accelerated by the presence of Fe
3+, whereas the Al
3+ retarded or inhibited the methanogens growth rate. The reactions (1 and 3) illustrate the dehydrogenation and hydrogenation pathways of Fe and Al NPs, which served as catalysts to hinder microbial activities or enhanced the degradation of the toxic organic compounds respectively.
From reaction (1), electrons released (Fe) promoted the CH
4 production and by-products like formic acid whose decomposition resulted in a polymerization chain reaction of CO
2 and H
2. Also, the zero-valent Fe
0 can be consumed by the inorganic CO
2 and other complex compounds (sulfide) (2) [
30].
Table 5 presents reported bio-stimulating effects of Al- and Fe-based salts and NPs additives on AD biogas production, which confirms Ms as the most effective nanocatalyst for the bio-hydrogenation and methanogenesis processes [
28,
30,
33]. In
Table 6, Sreekanh and Sahu [
35] reported about 21.3% and 35.7% reduction in the biogas volume as a result of using 100 mg/L of Al- and Fe-based salts for municipality sewage treatment respectively. Although previous studies were on mixed sludge with alum and ferric salts, the current study also corroborates reduction of biogas yield from Alum activated sludge [
35,
36]. Thus, the Al additives promoted the formation of sulfuric acid, and other volatile fatty acids, which were inhibitory to the MBMP process. This retarded the pH level of the AD process to be in acidic medium, thereby affecting the methanogen growth rate and activities for the biogas and methane production [
33,
34,
35]. However, additives of the Ms reduced not only the lag phase but also the time to achieve the highest biogas production (peak).