Evolution of Pauson-Khand Reaction: Strategic Applications in Total Syntheses of Architecturally Complex Natural Products (2016–2020)
Abstract
1. Introduction
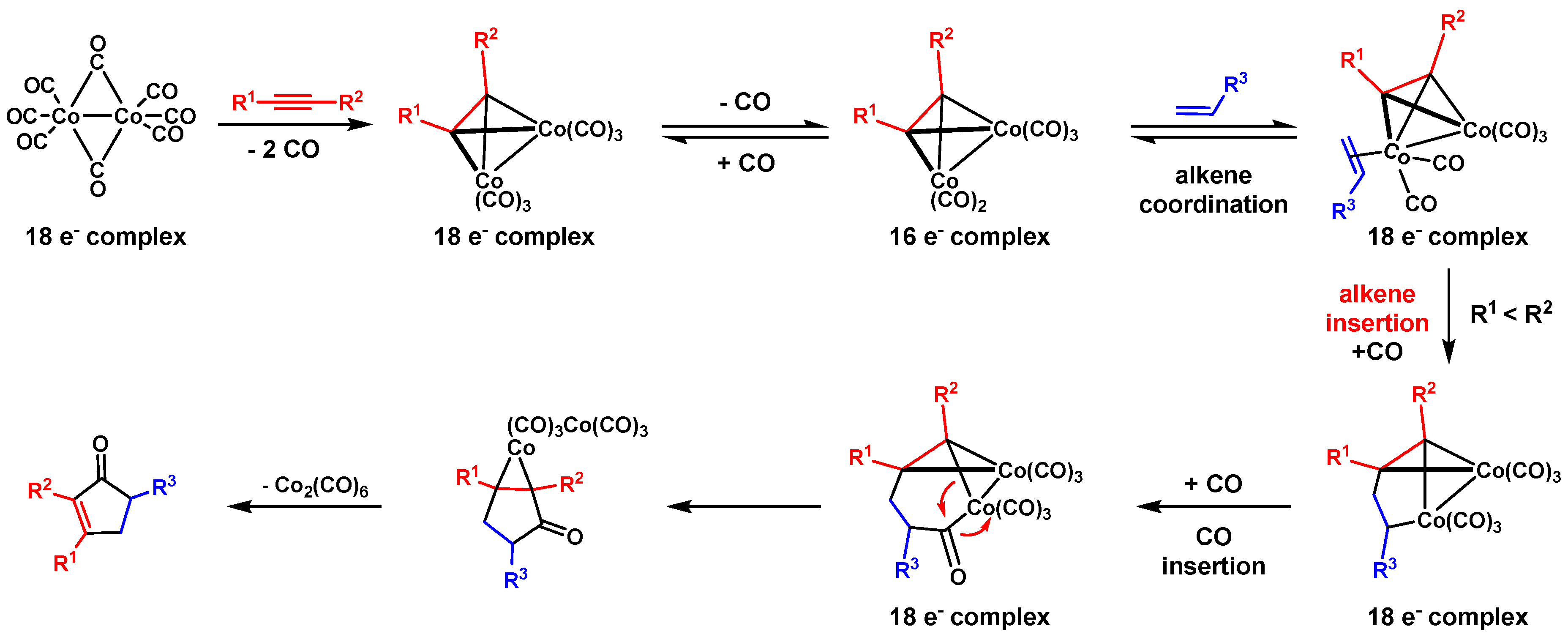
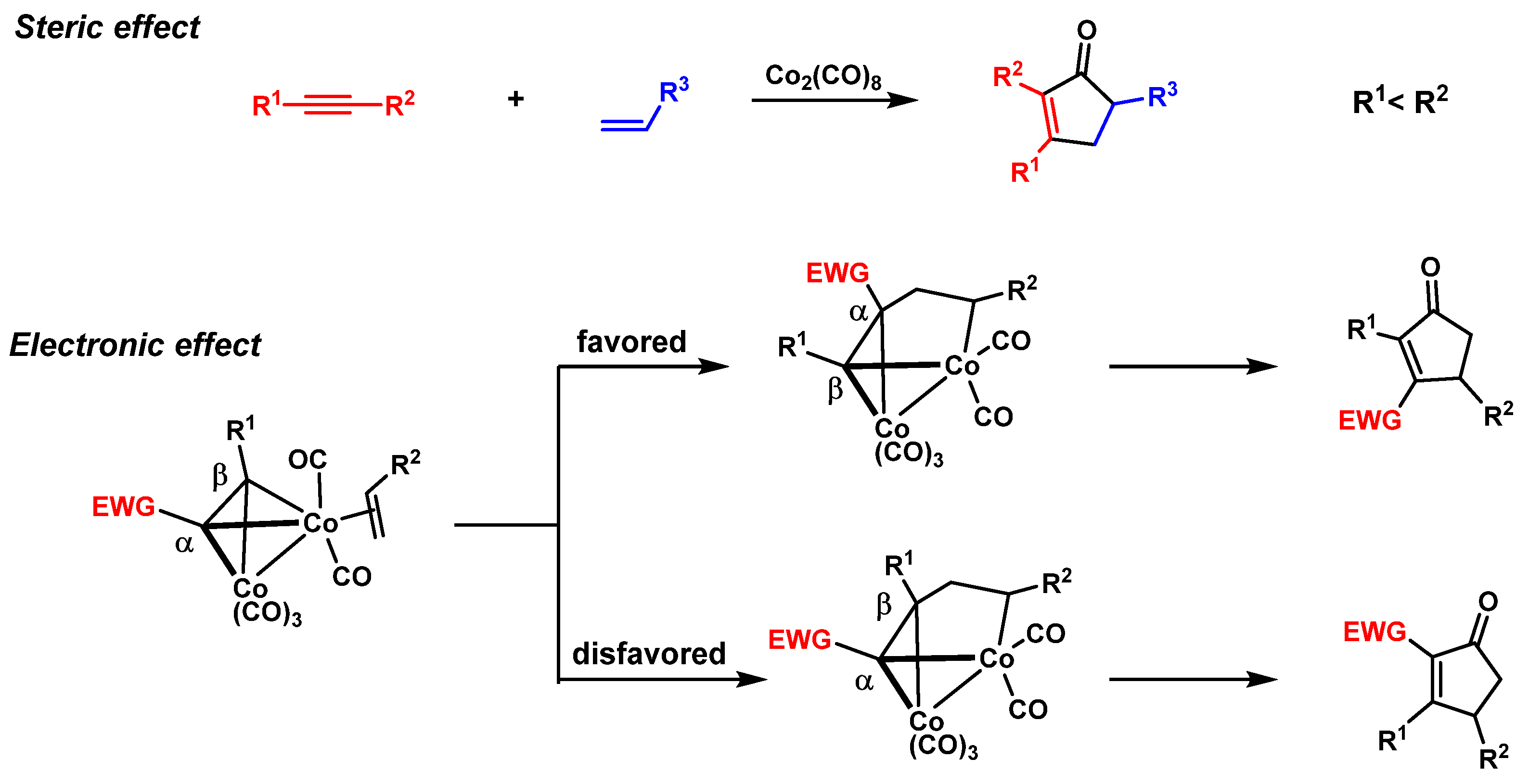
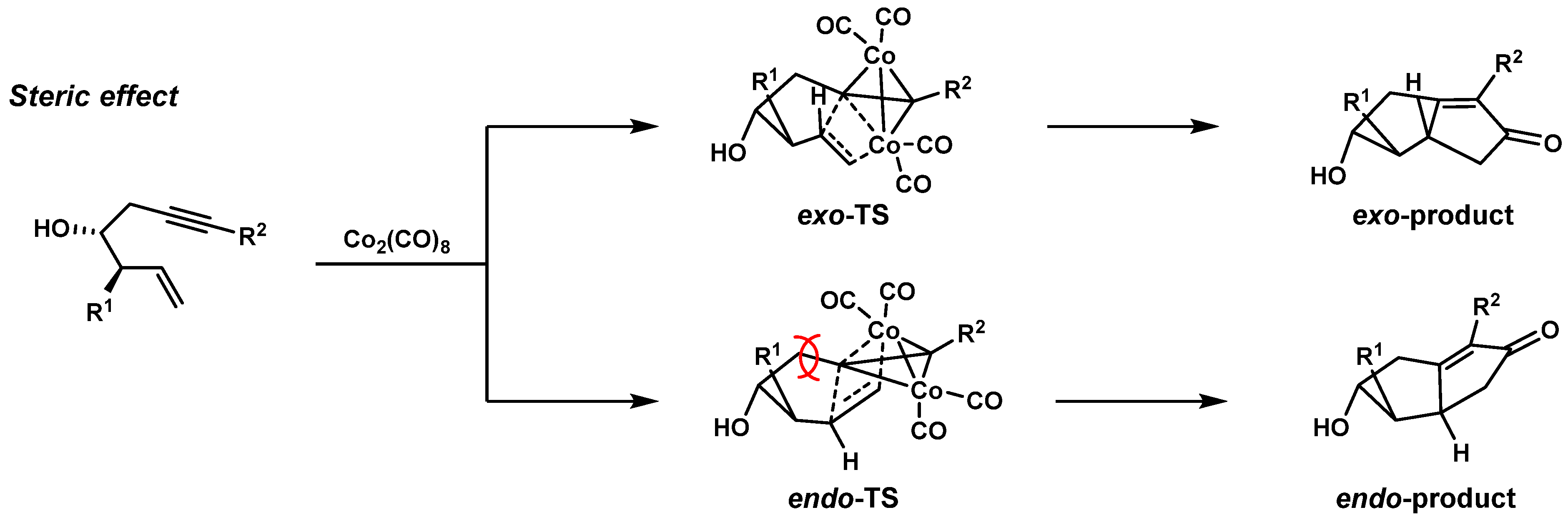
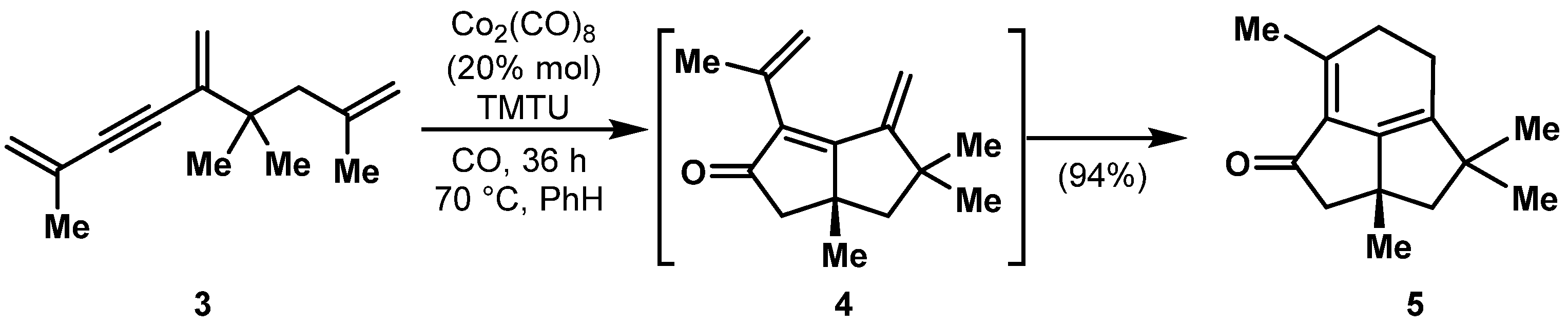
1.1. Classic PK Reaction Catalyzed by Co
1.2. PK Reaction Catalyzed by Rh and Pd
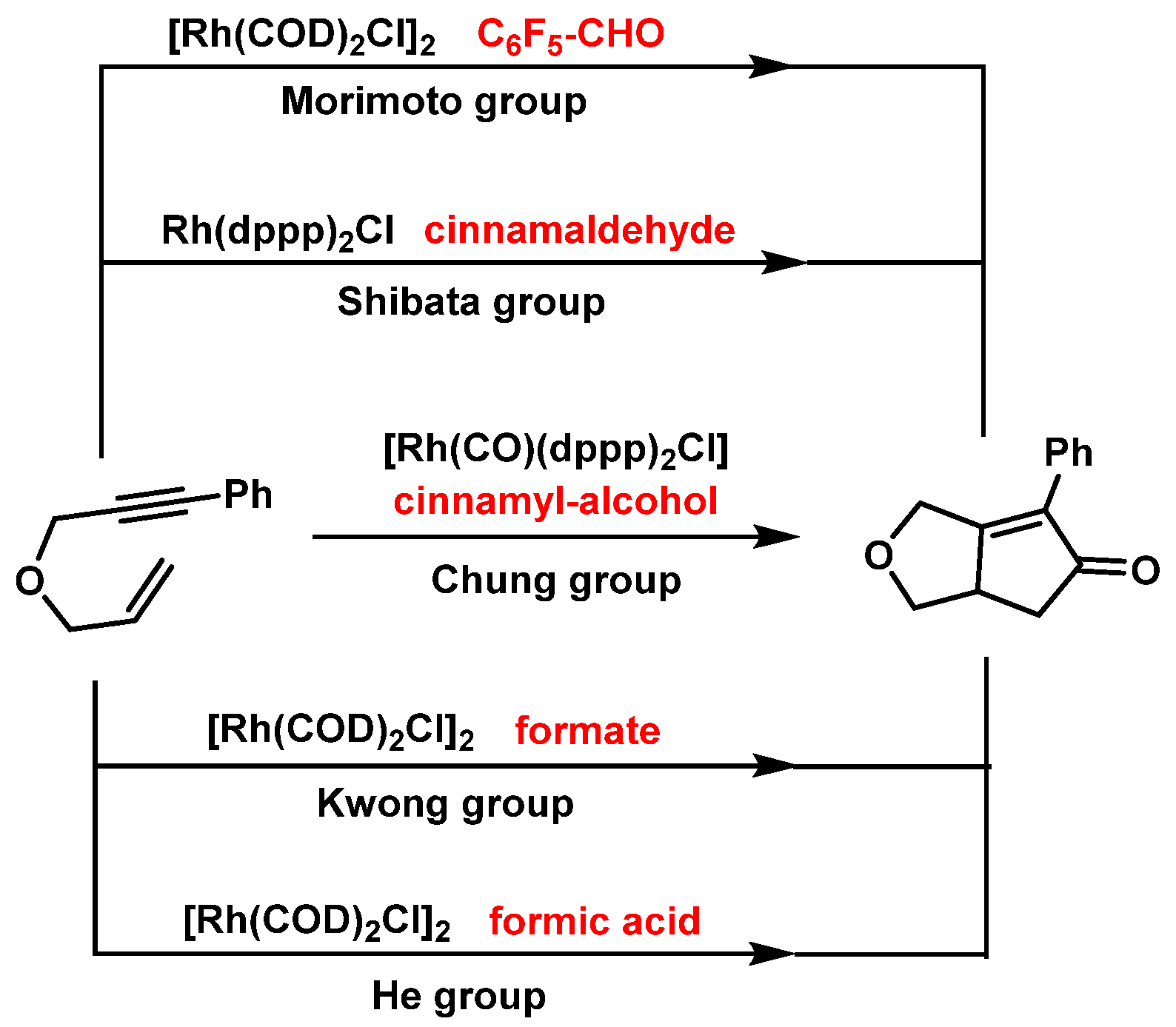
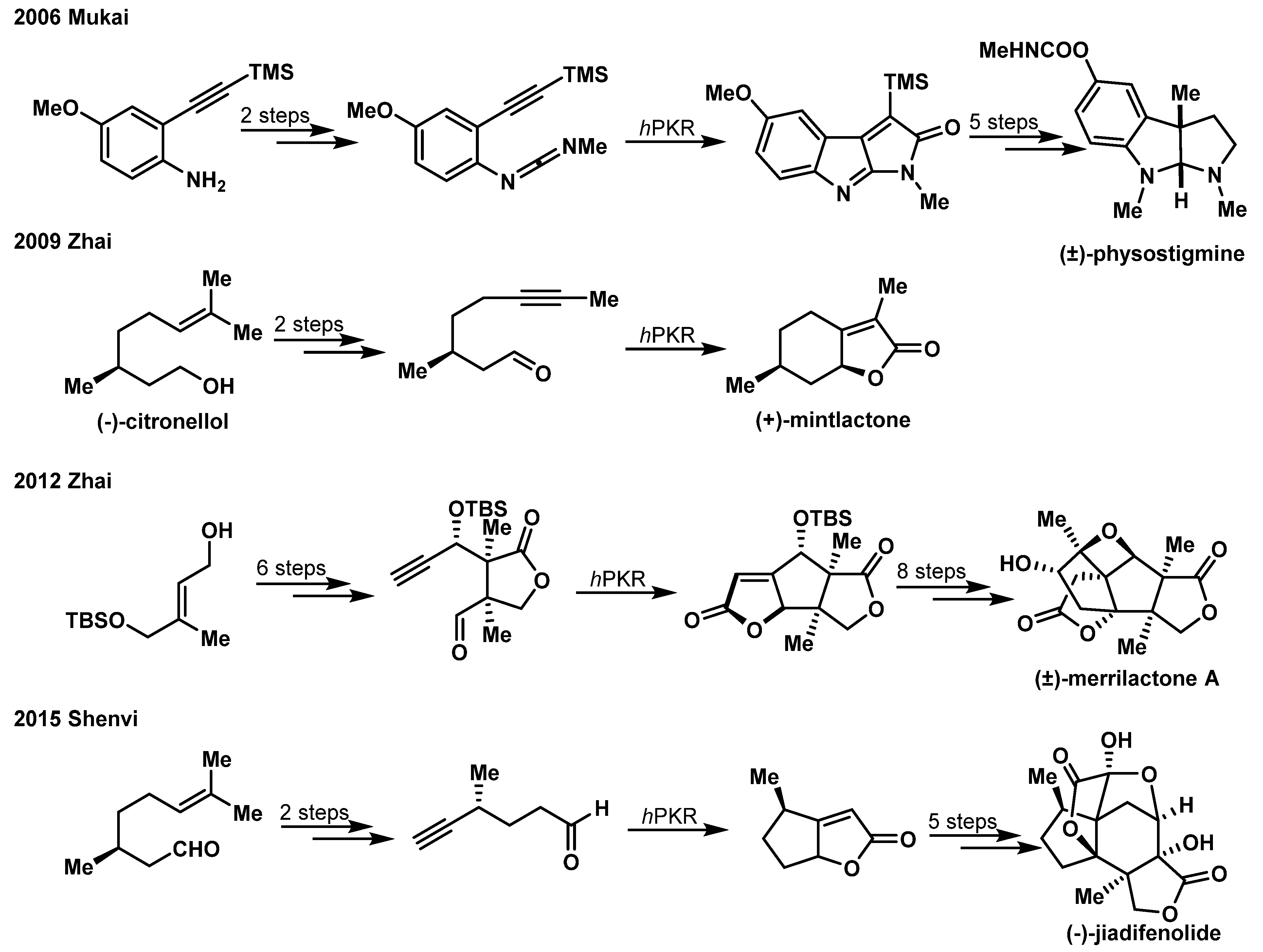
1.3. Hetero-Pauson-Khand Reaction
1.4. Summary
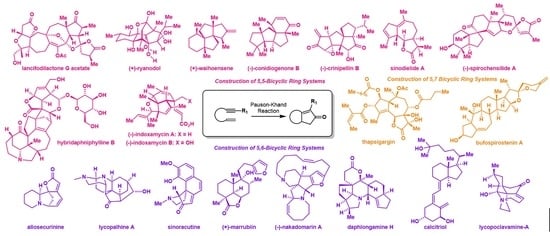
2. Recent Pauson-Khand Reaction Applications in Natural Products Total Syntheses
2.1. Construction of 5,5-Bicyclic Ring Systems
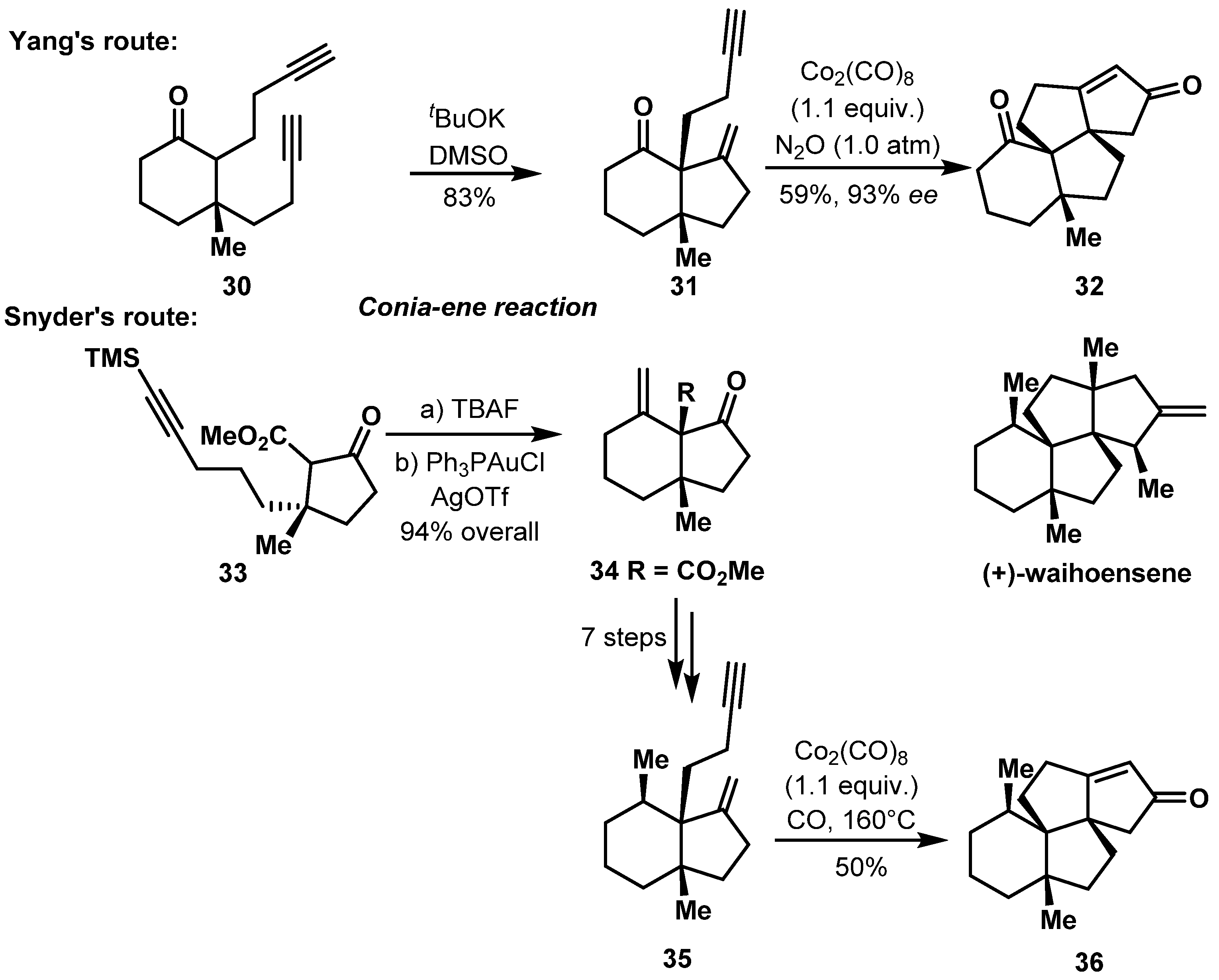
2.2. Construction of 5,6-Bicyclic Ring Systems
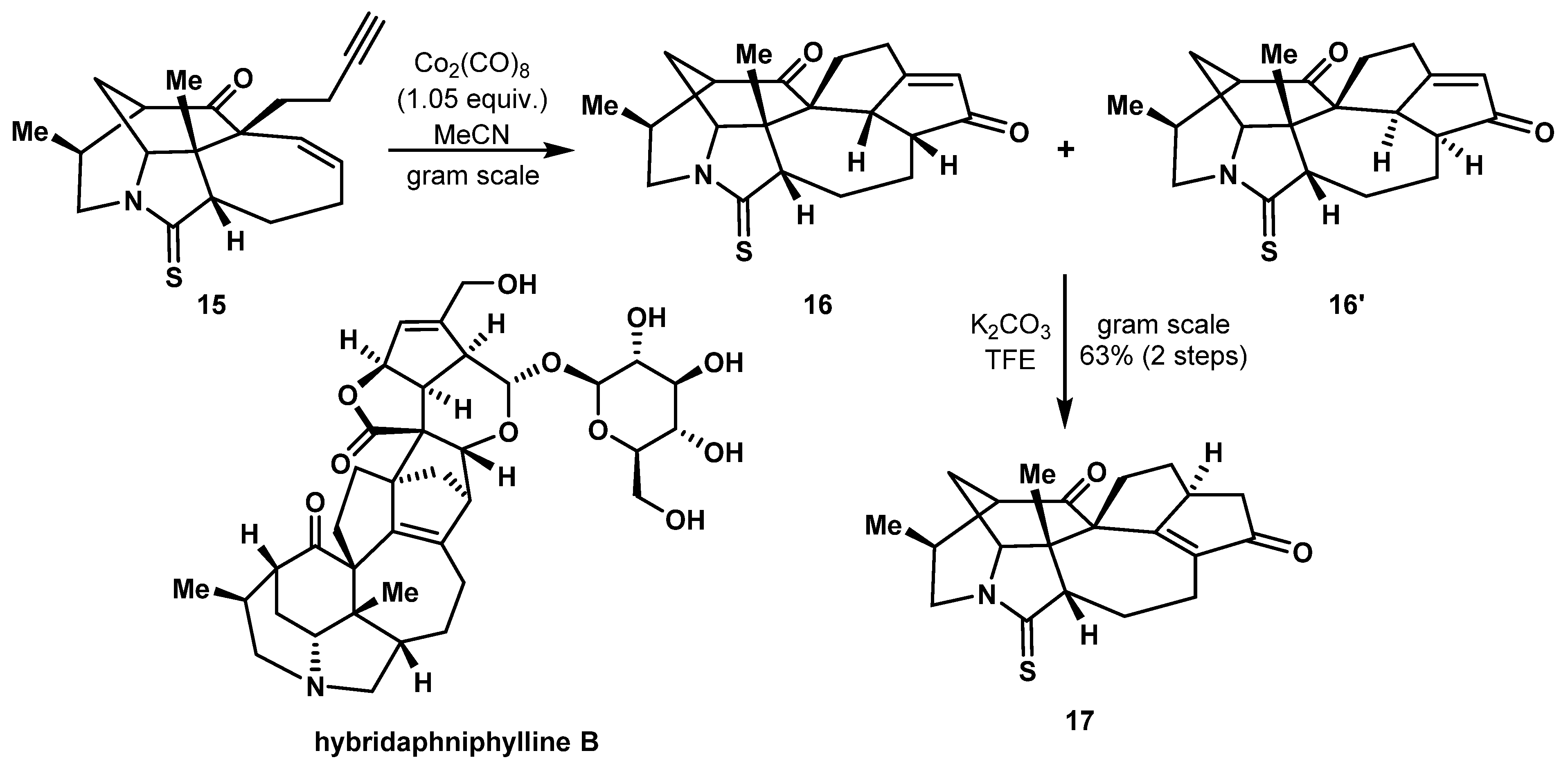
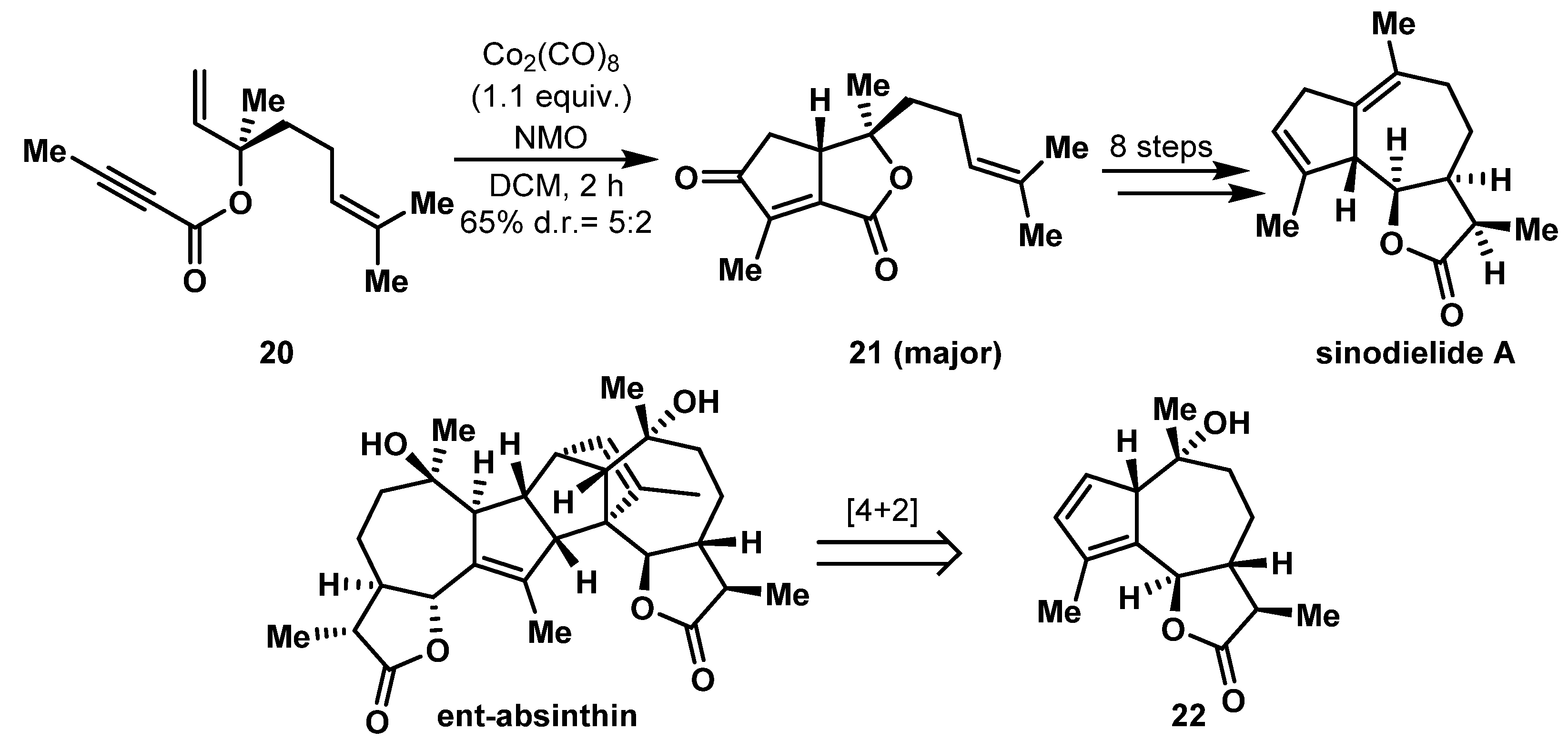
2.3. Construction of 5,7 Bicyclic Ring Systems
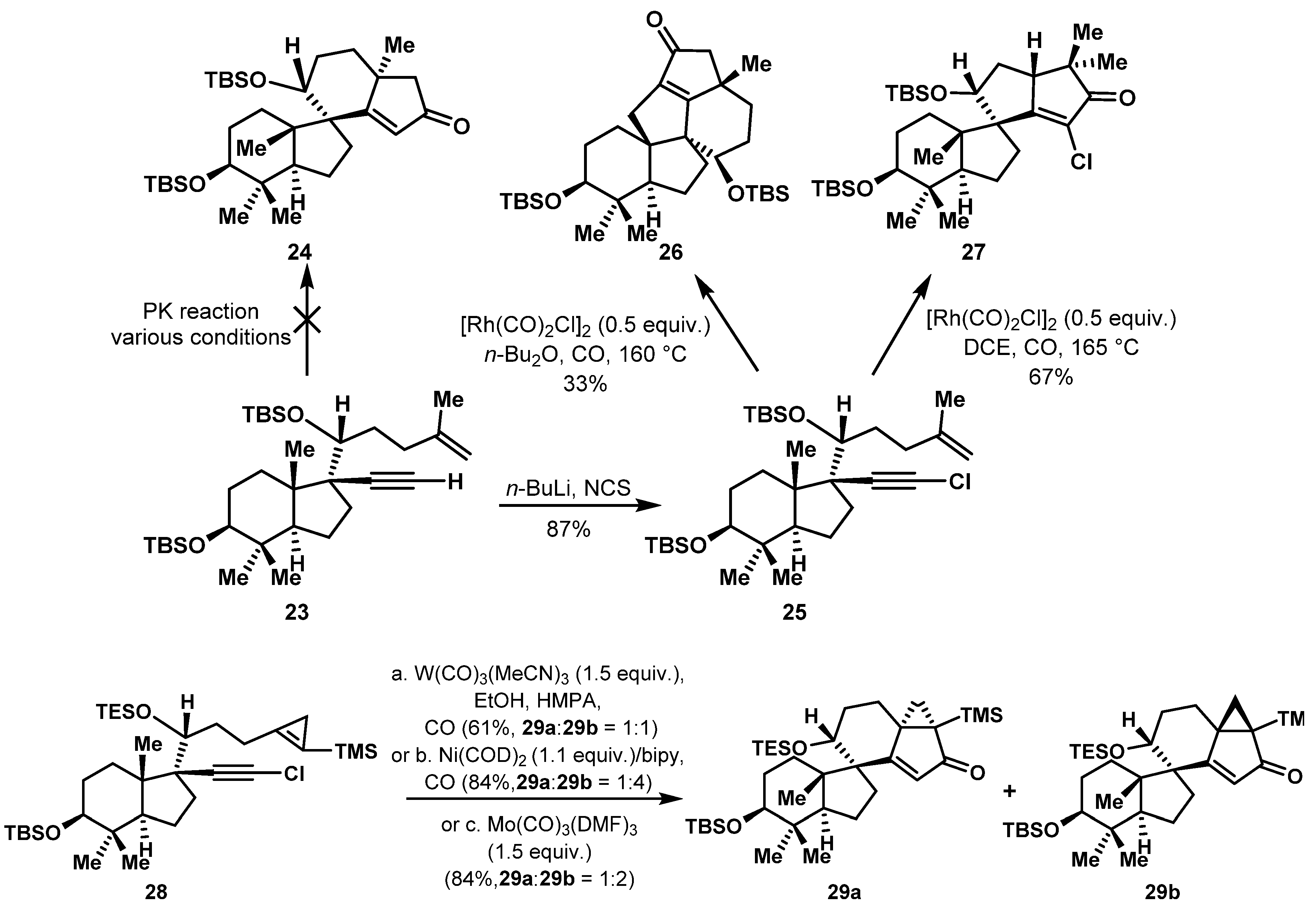
2.4. Construction of Macrocycles
3. Summary and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- D’yakonov, V.A.; Trapeznikova, O.A.; de Meijere, A.; Dzhemilev, U.M. Metal Complex Catalysis in the Synthesis of Spirocarbocycles. Chem. Rev. 2014, 114, 5775–5814. [Google Scholar] [CrossRef] [PubMed]
- Porcheddu, A.; Colacino, E.; De Luca, L.; Delogu, F. Metal-Mediated and Metal-Catalyzed Reactions Under Mechanochemical Conditions. ACS Catal. 2020, 10, 8344–8394. [Google Scholar] [CrossRef]
- Rodríguez, J.; Martínez, C.M. Transition-Metal-Mediated Modification of Biomolecules. Chem.-A Eur. J. 2020, 26, 9792–9813. [Google Scholar] [CrossRef] [PubMed]
- Khand, I.U.; Knox, G.R.; Pauson, P.L.; Watts, W.E.; Foreman, M.I. Organocobalt complexes. II. Reaction of acetylenehexacarbonyl dicobalt complexes, (RC2R1) Co2(CO)6, with norbornene and its derivatives. J. Chem. Soc. Perkin Trans. 1 1973, 9, 977–981. [Google Scholar] [CrossRef]
- Chung, Y.K. Transition metal alkyne complexes: The Pauson–Khand reaction. Coord. Chem. Rev. 1999, 188, 297–314. [Google Scholar] [CrossRef]
- Gibson, S.E.; Stevenazzi, A. The Pauson–Khand Reaction: The Catalytic Age Is Here. Angew. Chem. Int. Ed. 2003, 42, 1800–1810. [Google Scholar] [CrossRef] [PubMed]
- Urgoiti, J.B.; Anorbe, L.; Serrano, L.P.; Dominguez, G.; Castells, L.P. The Pauson–Khand reaction, a powerful synthetic tool for the synthesis of complex molecules. Chem. Soc. Rev. 2004, 33, 32–42. [Google Scholar] [CrossRef]
- Gibson, S.E.; Mainolfi, N. The Intermolecular Pauson–Khand Reaction. Angew. Chem. Int. Ed. 2005, 44, 3022–3037. [Google Scholar] [CrossRef]
- Kitagaki, S.; Inagaki, F.; Mukai, C. [2+2+1] Cyclization of allenes. Chem. Soc. Rev. 2014, 43, 2956–2978. [Google Scholar] [CrossRef]
- Lee, H.-W.; Kwong, F.-Y. A Decade of Advancements in Pauson–Khand-Type Reactions. Eur. J. Org. Chem. 2010, 2010, 789–811. [Google Scholar] [CrossRef]
- Shi, L.; Yang, Z. Exploring the Complexity-Generating Features of the Pauson–Khand Reaction from a Synthetic Perspective. Eur. J. Org. Chem. 2016, 2016, 2356–2368. [Google Scholar] [CrossRef]
- Ricker, J.D.; Geary, L.M. Recent Advances in the Pauson–Khand Reaction. Top. Catal. 2017, 60, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Jeong, N.; Hwang, S.H.; Lee, Y.S.; Chung, Y.K. Catalytic Version of the Intramolecular Pauson-Khand Reaction. J. Am. Chem. Soc. 1994, 116, 3159–3160. [Google Scholar] [CrossRef]
- Hayashi, M.; Hashimoto, Y.; Yamamoto, Y.; Usuki, J.; Saigo, K. Phosphane Sulfide/Octacarbonyldicobalt-Catalyzed Pauson-Khand Reaction under an Atmospheric Pressure of Carbon Monoxide. Angew. Chem. Int. Ed. 2000, 39, 631–632. [Google Scholar] [CrossRef]
- Krafft, M.E.; Boñaga, L.V.R.; Wright, J.A.; Hirosawa, C. Cobalt Carbonyl-Mediated Carbocyclizations of Enynes: Generation of Bicyclooctanones or Monocyclic Alkenes. J. Org. Chem. 2002, 67, 1233–1246. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, L.; Yu, R.; Chen, J.; Yang, Z. CoBr2–TMTU–zinc catalysed-Pauson–Khand reaction. Chem. Commun. 2012, 48, 8183–8185. [Google Scholar] [CrossRef]
- Orgue, S.; Leon, T.; Riera, A.; Verdaguer, X. Asymmetric Intermolecular Cobalt Catalyzed Pauson-Khand Reaction Using a Pstereogenic BisPhosphane. Org. Lett. 2015, 17, 250–253. [Google Scholar] [CrossRef]
- Koga, Y.; Kobayashi, T.; Narasaka, K. Rhodium-Catalyzed Intramolecular Pauson-Khand Reaction. Chem. Lett. 1998, 3, 249–250. [Google Scholar] [CrossRef]
- Jeong, N.; Lee, S.; Sung, B.K. Rhodium(I)-Catalyzed Intramolecular Pauson−Khand Reaction. Organometallics 1998, 17, 3642–3644. [Google Scholar] [CrossRef]
- Fan, B.M.; Xie, J.H.; Li, S.; Tu, Y.Q.; Zhou, Q.L. Rhodium-Catalyzed Asymmetric Pauson–Khand Reaction Using Monophosphoramidite Ligand SIPHOS. Adv. Synth. Catal. 2005, 347, 759–762. [Google Scholar] [CrossRef]
- Jeong, N.; Sung, B.K.; Choi, Y.K. Rhodium(I)-Catalyzed Asymmetric Intramolecular Pauson-Khand-Type Reaction. J. Am. Chem. Soc. 2000, 122, 6771–6772. [Google Scholar] [CrossRef]
- Jeong, N.; Kim, D.H.; Choi, J.H. Desymmetrization of meso-dienyne by asymmetric Pauson–Khand type reaction catalysts. Chem. Commun. 2004, 1134–1135. [Google Scholar] [CrossRef] [PubMed]
- Kwong, F.Y.; Li, Y.M.; Lam, W.H.; Qiu, L.Q.; Lee, H.W.; Yeung, C.H.; Chan, K.S.; Chan, A.S.C. Rhodium-Catalyzed Asymmetric Aqueous Pauson–Khand-Type Reaction. Chem. Eur. J. 2005, 11, 3872–3880. [Google Scholar] [CrossRef]
- Chen, G.Q.; Shi, M. Rhodium-catalyzed tandem Pauson–Khand type reactions of 1,4-enynes tethered by a cyclopropyl group. Chem. Commun. 2013, 49, 698–700. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.J.; Sawyer, J.R.; Evans, P.A.; Baik, M.H. Mechanistic Insight into the Diastereoselective Rhodium-Catalyzed Pauson–Khand Reaction: Role of Coordination Number in Stereocontrol. Angew. Chem. Int. Ed. 2008, 47, 342–345. [Google Scholar] [CrossRef]
- Kondo, T.; Suzuki, N.; Okada, T.; Mitsudo, T. First Ruthenium-Catalyzed Intramolecular Pauson-Khand Reaction. J. Am. Chem. Soc. 1997, 119, 6187–6188. [Google Scholar] [CrossRef]
- Morimoto, T.; Chatani, N.; Fukumoto, Y.; Murai, S. Ru3(CO)12-Catalyzed Cyclocarbonylation of 1,6-Enynes to Bicyclo[3.3.0]Octenones. J. Org. Chem. 1997, 62, 3762–3765. [Google Scholar] [CrossRef]
- Chatani, N.; Morimoto, T.; Fukumoto, Y.; Murai, S. Ru3(CO)12-Catalyzed Cyclocarbonylation of Yne-Aldehydes to Bicyclic α, β-Unsaturated γ-Butyrolactones. J. Am. Chem. Soc. 1998, 120, 5335–5336. [Google Scholar] [CrossRef]
- Miura, H.; Takeuchi, K.; Shishido, T. Intermolecular [2+2+1] Carbonylative Cycloaddition of Aldehydes with Alkynes, and Subsequent Oxidation to γ-Hydroxybutenolides by a Supported Ruthenium Catalyst. Angew. Chem. Int. Ed. 2016, 55, 278–282. [Google Scholar] [CrossRef]
- Kondo, T.; Nomura, M.; Ura, Y.; Wada, K.; Mitsudo, T. Ruthenium-catalyzed [2 + 2 + 1] Cocyclization of Isocyanates, Alkynes, and CO Enables the Rapid Synthesis of Polysubstituted Maleimides. J. Am. Chem. Soc. 2006, 128, 14816–14817. [Google Scholar] [CrossRef]
- Hicks, F.A.; Kablaoui, N.M.; Buchwald, S.L. Scope of the Intramolecular Titanocene-Catalyzed Pauson-Khand Type Reaction. J. Am. Chem. Soc. 1999, 121, 5881–5898. [Google Scholar] [CrossRef]
- Hicks, F.A.; Buchwald, S.L. An intramolecular Titanium-Catalyzed Asymmetric Pauson-Khand Type Reaction. J. Am. Chem. Soc. 1999, 121, 7026–7033. [Google Scholar] [CrossRef]
- Zhao, Z.B.; Ding, Y.; Zhao, G. Bicyclization of Enynes Using the Cp2TiCl2−Mg−BTC System: A Practical Method to Bicyclic Cyclopentenones. J. Org. Chem. 1998, 63, 9285–9291. [Google Scholar] [CrossRef]
- Hicks, F.A.; Kablaoui, N.M.; Buchwald, S.L. Titanocene-Catalyzed Cyclocarbonylation of Enynes to Cyclopentenones. J. Am. Chem. Soc. 1996, 118, 9450–9451. [Google Scholar] [CrossRef]
- Shibata, T.; Takagi, K. Iridium-Chiral Diphosphine Complex Catalyzed Highly Enantioselective Pauson-Khand-Type Reaction. J. Am. Chem. Soc. 2000, 122, 9852–9853. [Google Scholar] [CrossRef]
- Shibata, T.; Toshida, N.; Yamasaki, M.; Maekawa, S.; Takagi, K. Iridium-catalyzed enantioselective Pauson–Khand-type reaction of 1,6-enynes. Tetrahedron 2005, 61, 9974–9979. [Google Scholar] [CrossRef]
- Kwong, F.Y.; Lee, H.W.; Lam, W.H.; Qiu, L.Q.; Chan, A.S.C. Iridium-catalyzed cascade decarbonylation/highly enantioselective Pauson–Khand-type cyclization reactions. Tetrahedron Asymm. 2006, 17, 1238–1252. [Google Scholar] [CrossRef]
- Zhang, M.H.; Buchwald, S.L. A Nickel(0)-Catalyzed Process for the Transformation of Enynes to Bicyclic Cyclopentenones. J. Org. Chem. 1996, 61, 4498–4499. [Google Scholar] [CrossRef]
- Kent, J.L.; Wan, H.H.; Brummond, K.M. A new allenic Pauson-Khand cycloaddition for the preparation of α-methylene cyclopentenones. Tetrahedron Lett. 1995, 36, 2407–2410. [Google Scholar] [CrossRef]
- Adrio, J.; Rivero, M.R.; Carretero, J.C. Mild and Efficient Molybdenum-Mediated Pauson−Khand-Type Reaction. Org. Lett. 2005, 7, 431–434. [Google Scholar] [CrossRef]
- Shibata, T.; Koga, Y.; Narasaka, K. Intra- and Intermolecular Allene–Alkyne Coupling Reactions by the Use of Fe(CO)4(NMe3). Bull. Chem. Soc. Jpn. 1995, 68, 911–919. [Google Scholar] [CrossRef]
- Rutherford, D.T.; Christie, S.D.R. Soluble polymer-supported synthesis of arylpiperazines. Tetrahedron Lett. 1998, 39, 9505–9508. [Google Scholar]
- Park, K.H.; Son, S.U.; Chung, Y.K. Soluble polymer-supported synthesis of arylpiperazines. Chem. Commun. 2003, 1898–1899. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Son, S.U.; Chung, Y.K. Immobilized heterobimetallic Ru/Co nanoparticle-catalyzed Pauson–Khand-type reactions in the presence of pyridylmethyl formate. Chem. Commun. 2008, 2388–2390. [Google Scholar] [CrossRef] [PubMed]
- Khand, I.U.; Pauson, P.L. Organometallic Route to 2,7-Dihydrothiepin-1,1-Dioxides. Heterocycles 1978, 11, 59–67. [Google Scholar]
- Magnus, P.; Principe, L.M. Origins of 1,2-Stereoselectivity and 1,3-Stereoselectivity in Dicobaltoctacarbonyl Alkene Alkyne Cyclizations for the Synthesis of Substituted Bicyclo[3.3.0]Octenones. Tetrahedron Lett. 1985, 26, 4851–4854. [Google Scholar] [CrossRef]
- Magnus, P.; Exon, C.; Albaughrobertson, P. Dicobaltoctacarbonyl Alkyne Complexes as Intermediates in the Synthesis of Bicyclo[3.3.0]Octenones for the Synthesis of Coriolin and Hirsutic Acid. Tetrahedron 1985, 41, 5861–5869. [Google Scholar] [CrossRef]
- Magnus, P.; Principe, L.M.; Slater, M.J. Stereospecific Dicobalt Octacarbonyl Mediated Enyne Cyclization for the Synthesis of the Cytotoxic Sesquiterpene (+/−)-Quadrone. J. Org. Chem. 1987, 52, 1483–1486. [Google Scholar] [CrossRef]
- Labelle, B.E.; Knudsen, M.J.; Olmstead, M.M.; Hope, H.; Yanuck, M.D.; Schore, N.E. Synthesis of 11-Oxatricyclo [5.3.1.02,6] Undecane Derivatives Via Organometallic Cyclizations. J. Org. Chem. 1985, 50, 5215–5222. [Google Scholar] [CrossRef]
- Krafft, M.E.; Romero, R.H.; Scott, I.L. Pauson-Khand Reaction with Electron-Deficient Alkynes. J. Org. Chem. 1992, 57, 5277–5278. [Google Scholar] [CrossRef]
- Robert, F.; Milet, A.; Gimbert, Y.; Konya, D.; Greene, A.E. Regiochemistry in the Pauson-Khand Reaction: Has a Trans Effect Been Overlooked? J. Am. Chem. Soc. 2001, 123, 5396–5400. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Kuwabara, S.; Ando, Y.; Nagata, H.; Nishiyama, H.; Itoh, K. Palladium(0)-Catalyzed Cyclization of Electron-Deficient Enynes and Enediynes. J. Org. Chem. 2004, 69, 6697–6705. [Google Scholar] [CrossRef] [PubMed]
- Pauson, P.L. The Khand Reaction—A Convenient and General-Route to a Wide-Range of Cyclopentenone Derivatives. Tetrahedron 1985, 41, 5855–5860. [Google Scholar] [CrossRef]
- De Bruin, T.J.M.; Milet, A.; Greene, A.E.; Gimbert, Y. Insight into the Reactivity of Olefins in the Pauson-Khand Reaction. J. Org. Chem. 2004, 69, 1075–1080. [Google Scholar] [CrossRef] [PubMed]
- Krafft, M.E. Regiocontrol in the Intermolecular Cobalt-Catalyzed Olefin Acetylene Cyclo-Addition. J. Am. Chem. Soc. 1988, 110, 968–970. [Google Scholar] [CrossRef]
- Krafft, M.E. Steric Control in the Pauson Cyclo-Addition—Further Support for the Proposed Mechanism. Tetrahedron Lett. 1988, 29, 999–1002. [Google Scholar] [CrossRef]
- Sola, J.; Riera, A.; Verdaguer, X.; Maestro, M.A. Phosphine-Substrate Recognition through the C-H⋯O Hydrogen Bond: Application to the Asymmetric Pauson-Khand Reaction. J. Am. Chem. Soc. 2005, 127, 13629–13633. [Google Scholar] [CrossRef]
- Ahmar, M.; Antras, F.; Cazes, B. Pauson-Khand Reaction with Allenic Compounds.1. Synthesis of 4-Alkylidene-2-Cyclopentenones. Tetrahedron Lett. 1995, 36, 4417–4420. [Google Scholar] [CrossRef]
- Ahmar, M.; Chabanis, O.; Gauthier, J.; Cazes, B. Pauson-Khand Reaction with Allenic Compounds II: Reactivity of Functionalized Allenes. Tetrahedron Lett. 1997, 38, 5277–5280. [Google Scholar] [CrossRef]
- Wu, N.; Deng, L.J.; Liu, L.Z.; Liu, Q.; Li, C.C.; Yang, Z. Reverse Regioselectivity in the Palladium(II) Thiourea Catalyzed Intermolecular Pauson-Khand Reaction. Chem. Asian J. 2013, 8, 65–68. [Google Scholar] [CrossRef]
- Smit, W.A.; Gybin, A.S.; Shashkov, A.S.; Strychkov, Y.T.; Kyzmina, L.G.; Mikaelian, G.S.; Caple, R.; Swanson, E.D. New Route to the Synthesis of Polycyclic Compounds Based on a Stepwise Ade-Reaction of Dicobalt Hexacarbonyl Complexes of Conjugated Enynes with a Subsequent Intramolecular Khand-Pauson Type Reaction. Tetrahedron Lett. 1986, 27, 1241–1244. [Google Scholar] [CrossRef]
- Shen, J.K.; Gao, Y.C.; Shi, Q.Z.; Basolo, F. Oxygen Atom Transfer-Reactions to Metal-Carbonyls—Kinetics and Mechanism of Co Substitution of Fe(Co)5, Ru(Co)5, Os(Co)5 in the Presence of (CH3)3NO. Organometallics 1989, 8, 2144–2147. [Google Scholar] [CrossRef]
- Shambayati, S.; Crowe, W.E.; Schreiber, S.L. N-Oxide Promoted Pauson-Khand Cyclizations at Room-Temperature. Tetrahedron Lett. 1990, 31, 5289–5292. [Google Scholar] [CrossRef]
- Gallagher, A.G.; Tian, H.; Torres-Herrera, O.A.; Yin, S.; Xie, A.; Lange, D.M.; Wilson, J.K.; Mueller, L.G.; Gau, M.R.; Carroll, P.J.; et al. Access to Highly Functionalized Cyclopentenones via Diastereoselective Pauson-Khand Reaction of Siloxy-Tethered 1,7-Enynes. Org. Lett. 2019, 21, 8646–8651. [Google Scholar] [CrossRef]
- Hiroi, K.; Watanabe, T.; Kawagishi, R.; Abe, I. Asymmetric Catalytic Pauson-Khand Reactions with Chiral Phosphine Ligands: Dramatic Effects of Substituents in 1,6-Enyne Systems. Tetrahedron Lett. 2000, 41, 891–895. [Google Scholar] [CrossRef]
- Tang, Y.F.; Deng, L.J.; Zhang, Y.D.; Dong, G.B.; Chen, J.H.; Yang, Z. Tetramethyl thiourea/Co2(CO)8-catalyzed Pauson-Khand Reaction under Balloon Pressure of CO. Org. Lett. 2005, 7, 593–595. [Google Scholar] [CrossRef] [PubMed]
- Sugihara, T.; Yamada, M.; Ban, H.; Yamaguchi, M.; Kaneko, C. Rate Enhancement of the Pauson-Khand Reaction by Primary Amines. Angew. Chem. Int. Ed. 1997, 36, 2801–2804. [Google Scholar] [CrossRef]
- Del Valle, C.P.; Milet, A.; Gimbert, Y.; Greene, A.E. Lewis Base Promoters in the Pauson-Khand Reaction: A Different Scenario. Angew. Chem. Int. Ed. 2005, 44, 5717–5719. [Google Scholar] [CrossRef]
- Kobayashi, T.; Koga, Y.; Narasaka, K. The Rhodium-Catalyzed Pauson-Khand Reaction. J. Organomet. Chem. 2001, 624, 73–87. [Google Scholar] [CrossRef]
- Schmid, T.M.; Consiglio, G. Mechanistic and Stereochemical Aspects of the Asymmetric Cyclocarbonylation of 1,6-Enynes with Rhodium Catalysts. Chem. Commun. 2004, 20, 2318–2319. [Google Scholar] [CrossRef]
- Wender, P.A.; Deschamps, N.M.; Williams, T.J. Intermolecular Dienyl Pauson–Khand Reaction. Angew. Chem. Int. Ed. 2004, 43, 3076–3079. [Google Scholar] [CrossRef] [PubMed]
- Wender, P.A.; Croatt, M.P.; Deschamps, N.M. Metal-Catalyzed [2+2+1] Cycloadditions of 1,3-Dienes, Allenes, and CO. Angew. Chem. Int. Ed. 2006, 45, 2459–2462. [Google Scholar] [CrossRef] [PubMed]
- Shibata, T.; Toshida, N.; Takagi, K. Catalytic Pauson-Khand-Type Reaction Using Aldehydes as a CO Source. Org. Lett. 2002, 4, 1619–1621. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Cho, Y.; Chung, Y.K. Rhodium-Catalyzed Pauson-Khand-Type Reaction Using Alcohol as a Source of Carbon Monoxide. Angew. Chem. Int. Ed. 2010, 49, 5138–5141. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Chan, A.S.C.; Kwong, F.Y. Formate as a CO surrogate for cascade processes: Rh-catalyzed cooperative decarbonylation and asymmetric Pauson–Khand-type cyclization reactions. Chem. Commun. 2007, 25, 2633–2635. [Google Scholar] [CrossRef] [PubMed]
- Lang, X.-D.; You, F.; He, X.; Yu, Y.-C.; He, L.-N. Rhodium(I)-catalyzed Pauson Khand-type Reaction Using Formic Acid as a CO Surrogate: An Alternative Approach for Indirect CO2 Utilization. Green Chem. 2019, 21, 509–514. [Google Scholar] [CrossRef]
- Jorgensen, L.; McKerrall, S.J.; Kuttruff, C.A.; Ungeheuer, F.; Felding, J.; Baran, P.S. 14-Step Synthesis of (+)-Ingenol from (+)-3-Carene. Science 2013, 341, 878–882. [Google Scholar] [CrossRef] [PubMed]
- Dai, M.J.; Liang, B.; Wang, C.H.; Chen, J.H.; Yang, Z. Synthesis of a Novel C-2-Symmetric Thiourea and Its Application in the Pd-Catalyzed Cross-Coupling Reactions with Arenediazonium Salts under Aerobic Conditions. Org. Lett. 2004, 6, 221–224. [Google Scholar] [CrossRef]
- Xiong, Z.C.; Wang, N.D.; Dai, M.J.; Li, A.; Chen, J.H.; Yang, Z. Synthesis of Novel Palladacycles and Their Application in Heck and Suzuki Reactions under Aerobic Conditions. Org. Lett. 2004, 6, 3337–3340. [Google Scholar] [CrossRef]
- Liao, Y.; Smith, J.; Fathi, R.; Yang, Z. Novel Pd(II)-Mediated Cascade Carboxylative Annulation to Construct BenzoFuran-3-Carboxylic Acids. Org. Lett. 2005, 7, 2707–2709. [Google Scholar] [CrossRef]
- Deng, L.J.; Liu, J.; Huang, J.Q.; Hu, Y.H.; Chen, M.; Lan, Y.; Chen, J.H.; Lei, A.W.; Yang, Z. Effect of Lithium Chloride on Tuning the Reactivity of Pauson-Khand Reactions Catalyzed by Palladium-Tetramethylthiourea. Synthesis 2007, 2007, 2565–2570. [Google Scholar] [CrossRef]
- Tang, Y.; Deng, L.; Zhang, Y.; Dong, G.; Chen, J.; Yang, Z. Thioureas as Ligands in the Pd-Catalyzed Intramolecular Pauson−Khand Reaction. Org. Lett. 2005, 7, 1657–1659. [Google Scholar] [CrossRef]
- Lan, Y.; Deng, L.J.; Liu, J.; Wang, C.; Wiest, O.; Yang, Z.; Wu, Y.D. On the Mechanism of the Palladium Catalyzed Intramolecular Pauson-Khand-Type Reaction. J. Org. Chem. 2009, 74, 5049–5058. [Google Scholar] [CrossRef] [PubMed]
- Crowe, W.E.; Vu, A.T. Direct Synthesis of Fused, Bicyclic γ-Butyrolactones via Tandem Reductive Cyclization−Carbonylation of Tethered Enals and Enones. J. Am. Chem. Soc. 1996, 118, 1557–1558. [Google Scholar] [CrossRef]
- Mandal, S.K.; Amin, S.R.; Crowe, W.E. γ-Butyrolactone Synthesis via Catalytic Asymmetric Cyclocarbonylation. J. Am. Chem. Soc. 2001, 123, 6457–6458. [Google Scholar] [CrossRef] [PubMed]
- Kablaoui, N.M.; Hicks, F.A.; Buchwald, S.L. Diastereoselective Synthesis of γ-Butyrolactones from Enones Mediated or Catalyzed by a Titanocene Complex. J. Am. Chem. Soc. 1996, 118, 5818–5819. [Google Scholar] [CrossRef]
- Kablaoui, N.M.; Hicks, F.A.; Buchwald, S.L. Titanocene-Catalyzed Cyclocarbonylation of o-Allyl Aryl Ketones to γ-Butyrolactones. J. Am. Chem. Soc. 1997, 119, 4424–4431. [Google Scholar] [CrossRef]
- Chatani, N.; Morimoto, T.; Kamitani, A.; Fukumoto, Y.; Murai, S.J. Ru3(CO)12-catalyzed reaction of yne–imines with carbon monoxide leading to bicyclic α, β-unsaturated lactams. Organomet. Chem. 1999, 579, 177–181. [Google Scholar] [CrossRef]
- Adrio, J.; Carretero, J.C. Butenolide Synthesis by Molybdenum-Mediated Hetero-Pauson−Khand Reaction of Alkynyl Aldehydes. J. Am. Chem. Soc. 2007, 129, 778–779. [Google Scholar] [CrossRef]
- Saito, T.; Sugizaki, K.; Osada, H.; Kutsumura, N.; Otani, T. A Hetero Pauson-Khand Reaction of Ketenimines: A New Synthetic Method for γ-Exomethylene-α, β-unsaturated γ-Lactams. Heterocycles 2010, 80, 207–211. [Google Scholar] [CrossRef]
- Finnegan, D.F.; Snapper, M.L. Formation of Polycyclic Lactones through a Ruthenium-Catalyzed Ring-Closing Metathesis/Hetero-Pauson–Khand Reaction Sequence. J. Org. Chem. 2011, 76, 3644–3653. [Google Scholar] [CrossRef]
- Gao, P.; Xu, P.F.; Zhai, H. Expeditious Construction of (+)-Mintlactone via Intramolecular Hetero-Pauson−Khand Reaction. J. Org. Chem. 2009, 74, 2592–2593. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Gao, P.; Yu, F.; Yang, Y.; Zhu, S.; Zhai, H. Total Synthesis of (±)-Merrilactone A. Angew. Chem. Int. Ed. 2012, 51, 5897–5899. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.-H.; Martinez, M.D.; Shenvi, R.A. An eight-step gram-scale synthesis of (−)-jiadifenolide. Nat. Chem. 2015, 7, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Chirkin, E.; Michel, S.; Porée, F.H. Viability of a [2 + 2 + 1] Hetero-Pauson–Khand Cycloaddition Strategy toward Securinega Alkaloids: Synthesis of the BCD-Ring Core of Securinine and Related Alkaloids. J. Org. Chem. 2015, 80, 6525–6528. [Google Scholar] [CrossRef] [PubMed]
- Mukai, C.; Yoshida, T.; Sorimachi, M.; Odani, A. Co2(CO)8-Catalyzed Intramolecular Hetero-Pauson−Khand Reaction of Alkynecarbodiimide: Synthesis of (±)-Physostigmine. Org. Lett. 2006, 8, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Huang, J.; Qu, Y.; Zhang, W.; Gong, J.; Yang, Z. Total Syntheses of Crinipellins Enabled by Cobalt-Mediated and Palladium-Catalyzed Intramolecular Pauson-Khand Reactions. Angew. Chem. Int. Ed. 2018, 57, 8744–8748. [Google Scholar] [CrossRef]
- Peng, C.; Arya, P.; Zhou, Z.; Snyder, S.A. A Concise Total Synthesis of (+)-Waihoensene Guided by Quaternary Center Analysis. Angew. Chem. Int. Ed. 2020, 59, 13521–13525. [Google Scholar] [CrossRef]
- Qu, Y.; Wang, Z.; Zhang, Z.; Zhang, W.; Huang, J.; Yang, Z. Asymmetric Total Synthesis of (+)-Waihoensene. J. Am. Chem. Soc. 2020, 142, 6511–6515. [Google Scholar] [CrossRef]
- Knudsen, M.J.; Schore, N.E. Synthesis of the Angularly Fused Triquinane Skeleton Via Intramolecular Organometallic Cyclization. J. Org. Chem. 1984, 49, 5025–5026. [Google Scholar] [CrossRef]
- Pallerla, M.K.; Fox, J.M. Enantioselective Synthesis of (−)-Pentalenene. Org. Lett. 2007, 9, 5625–5628. [Google Scholar] [CrossRef] [PubMed]
- Millham, A.B.; Kier, M.J.; Leon, R.M.; Karmakar, R.; Stempel, Z.D.; Micalizio, G.C. A Complementary Process to PausonKhand-Type Annulation Reactions for the Construction of Fully Substituted Cyclopentenones. Org. Lett. 2019, 21, 567–570. [Google Scholar] [CrossRef] [PubMed]
- Bird, R.; Knipe, A.C.; Stirling, C.J.M.J. Intramolecular Reactions. Part X. Transition States in the Cyclisation of N-ω-Halogeno-alkylamines and –sulphonamides. Chem. Soc. Perkin Trans. 2 1973, 9, 1215–1220. [Google Scholar] [CrossRef]
- Wiesner, K.; Valenta, Z.; Findlay, J.A. The structure of ryanodine. Tetrahedron Lett. 1967, 8, 221–223. [Google Scholar] [CrossRef]
- Srivastava, S.N.; Przybylska, M. The molecular structure of ryanodol-p-bromo benzyl ether. Can. J. Chem. 1968, 46, 795–797. [Google Scholar] [CrossRef]
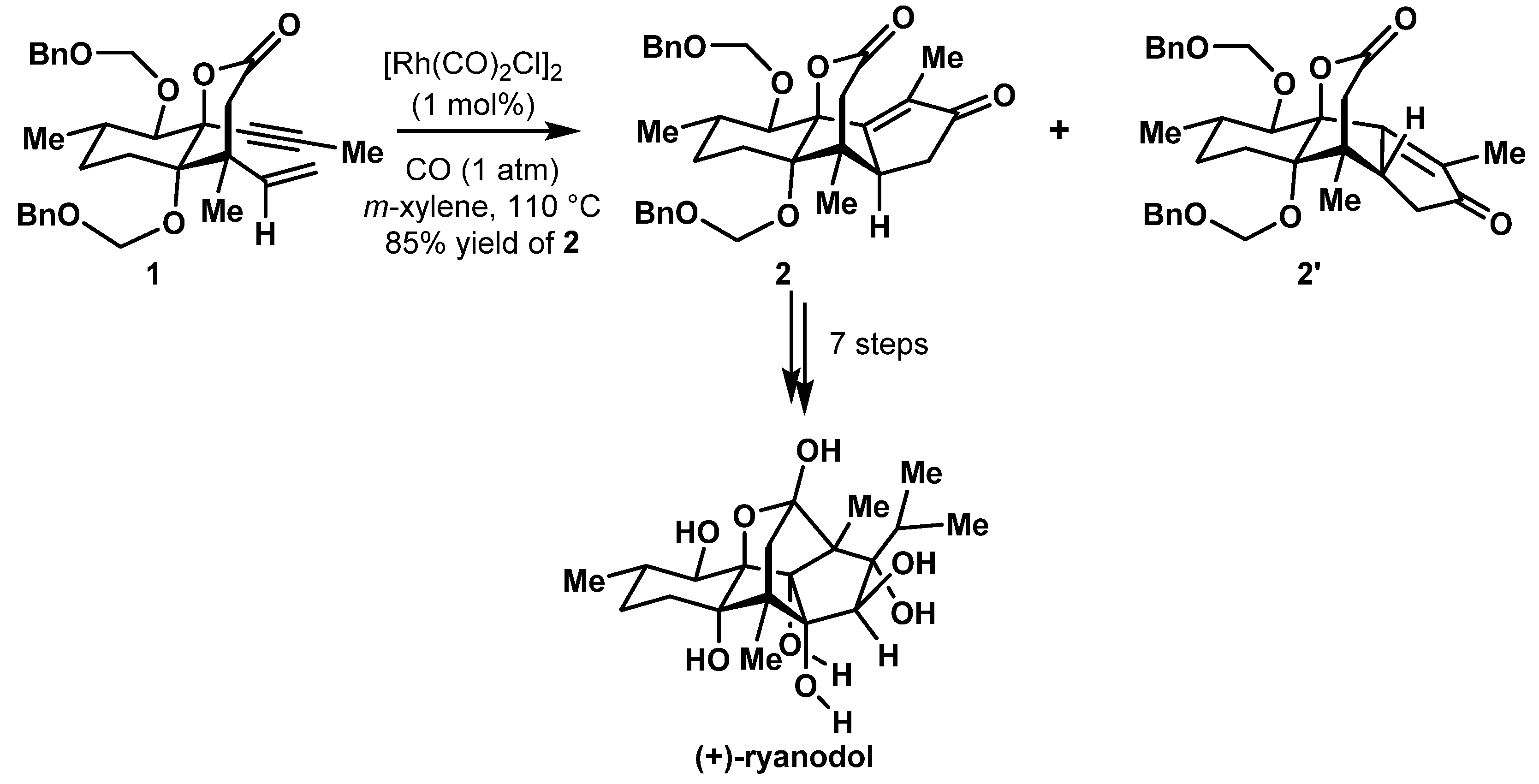
- Chuang, K.V.; Xu, C.; Reisman, S.E. A 15-step synthesis of (+)-ryanodol. Science 2016, 353, 912–915. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Li, Y.; Zhao, D.; He, Y.; Gong, J.; Yang, Z. A Concise Synthesis of Presilphiperfolane Corethrough a Tandem TMTU-Co-Catalyzed Pauson-Khand Reaction and a 6pi Electrocyclization Reaction (TMTU = Tetramethyl Thiourea). Chem. Eur. J. 2017, 23, 1258–1262. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhao, D.; He, Y.; Yang, Z.; Gong, J. Total syntheses of dehydrobotrydienal, dehydrobotrydienol and 10-oxodehydro- dihydrobotrydial. Chin. Chem. Lett. 2019, 30, 1503–1505. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, D.; Zhang, Z.; Tan, X.; Gong, J.; Yang, Z. Synthesis of 4-Desmethyl-rippertenol and 7-Epi-rippertenol via Photo-induced Cyclization of Dienones. CCS Chem. 2020. [Google Scholar] [CrossRef]
- Sun, T.W.; Liu, D.D.; Wang, K.Y.; Tong, B.Q.; Xie, J.X.; Jiang, Y.L.; Li, Y.; Zhang, B.; Liu, Y.F.; Wang, Y.X.; et al. Asymmetric Total Synthesis of Lancifodilactone G Acetate. 1. Diastereoselective Synthesis of CDEFGH Ring System. J. Org. Chem. 2018, 83, 6893–6906. [Google Scholar] [CrossRef]
- Wang, K.Y.; Liu, D.D.; Sun, T.W.; Lu, Y.; Zhang, S.L.; Li, Y.H.; Han, Y.X.; Liu, H.Y.; Peng, C.; Wang, Q.Y.; et al. Asymmetric Total Synthesis of Lancifodilactone G Acetate. 2. Final Phase and Completion of the Total Synthesis. J. Org. Chem. 2018, 83, 6907–6923. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ding, M.; Li, J.; Guo, Z.; Lu, M.; Chen, Y.; Liu, L.; Shen, Y.H.; Li, A. Total Synthesis of Hybridaphniphylline B. J. Am. Chem. Soc. 2018, 140, 4227–4231. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.K.; Lee, B.Y.; Jeong, N.; Hudecek, M.; Pauson, P.L. Promoters for the (alkyne) hexacarbonyldicobalt-based cyclopentenone synthesis. Organometallics 1993, 12, 220–223. [Google Scholar] [CrossRef]
- Hu, N.; Dong, C.; Zhang, C.; Liang, G. Total Synthesis of (−)-Indoxamycins A and B. Angew. Chem. Int. Ed. 2019, 58, 6659–6662. [Google Scholar] [CrossRef]
- Hu, X.; Musacchio, A.J.; Shen, X.; Tao, Y.; Maimone, T.J. Allylative Approaches to the Synthesis of Complex Guaianolide Sesquiterpenes from Apiaceae and Asteraceae. J. Am. Chem. Soc. 2019, 141, 14904–14915. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.T.; Chen, J.H.; Yang, Z. Asymmetric Total Synthesis of (−)-Spirochensilide A. J. Am. Chem. Soc. 2020, 142, 8116–8121. [Google Scholar] [CrossRef]
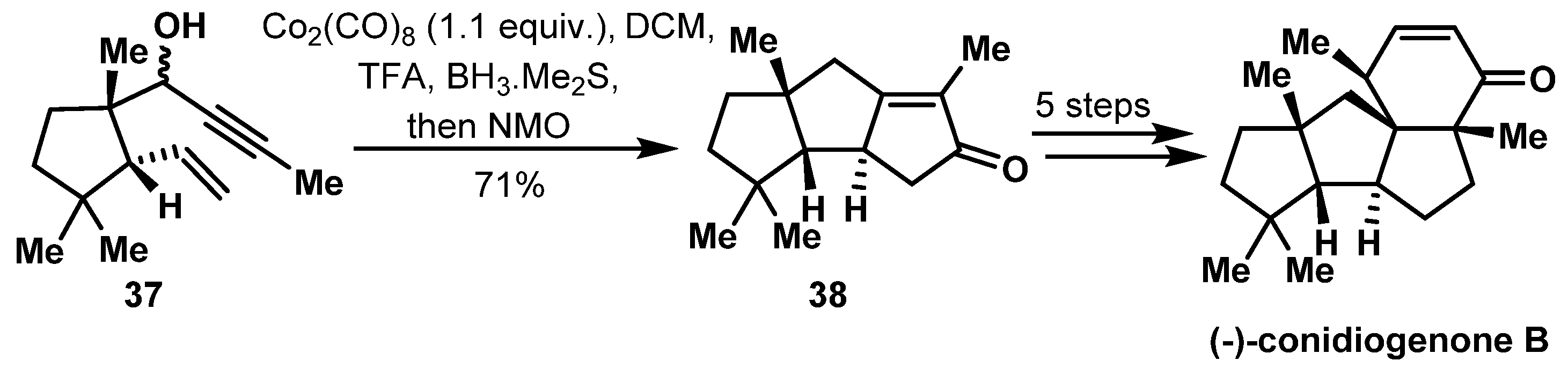
- Xu, B.; Xun, W.; Su, S.; Zhai, H. Total Syntheses of (−)-Conidiogenone B, (−)-Conidiogenone, and (−)-Conidiogenol. Angew. Chem. Int. Ed. 2020, 132, 16617–16621. [Google Scholar]
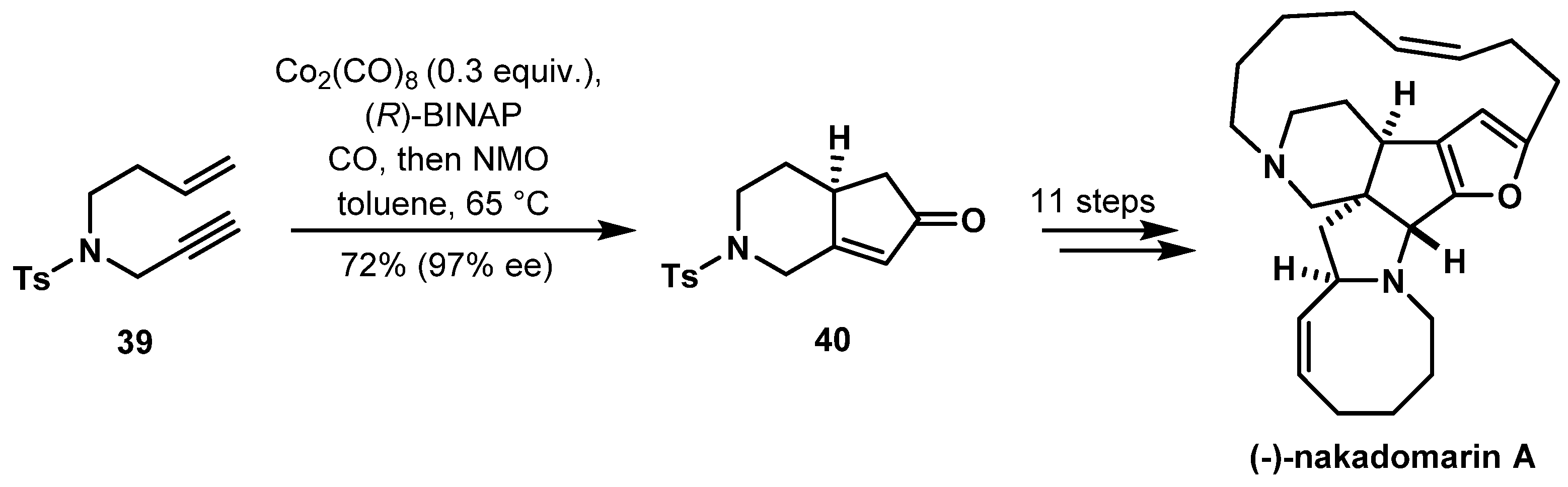
- Clark, J.S.; Xu, C. Total Synthesis of (−)-Nakadomarin A. Angew. Chem. Int. Ed. 2016, 55, 4332–4335. [Google Scholar] [CrossRef] [PubMed]
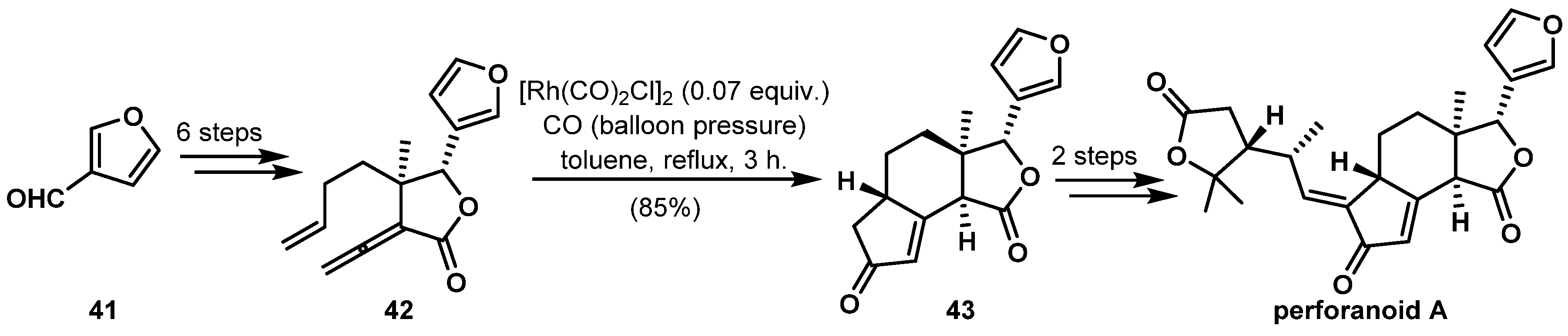
- Lv, C.; Tu, Q.; Gong, J.; Hao, X.; Yang, Z. Asymmetric total synthesis of (−)-perforanoid A. Tetrahedron 2017, 73, 3612–3621. [Google Scholar] [CrossRef]
- Cassayre, J.; Gagosz, F.; Zard, S.Z. A Short Synthesis of (±)-13-Deoxyserratine. Angew. Chem. Int. Ed. 2002, 41, 1783–1785. [Google Scholar] [CrossRef]
- Nakayama, A.; Kogure, N.; Kitajima, M.; Takayama, H. Asymmetric Total Synthesis of a Pentacyclic Lycopodium Alkaloid: Huperzine-Q. Angew. Chem. Int. Ed. 2011, 50, 8025–8028. [Google Scholar] [CrossRef] [PubMed]
- Itoh, N.; Iwata, T.; Sugihara, H.; Inagaki, F.; Mukai, C. Total Syntheses of (±)-Fawcettimine, (±)-Fawcettidine, (±)-Lycoflexine, and (±)-Lycoposerramine-Q. Chem. Eur. J. 2013, 19, 8665–8672. [Google Scholar] [CrossRef]
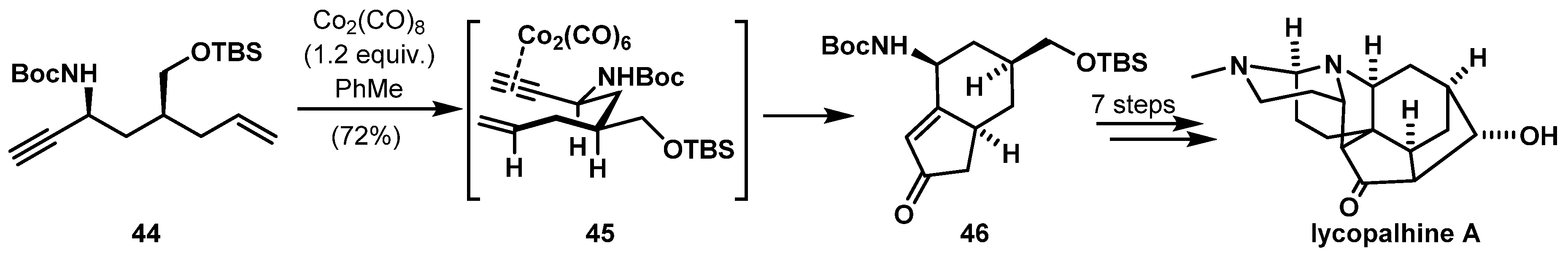
- Williams, B.M.; Trauner, D. Expedient Synthesis of (+)-Lycopalhine A. Angew. Chem. Int. Ed. 2016, 55, 2191–2194. [Google Scholar] [CrossRef]
- Yamakoshi, H.; Sawayama, Y.; Akahori, Y.; Kato, M.; Nakamura, S. Total Syntheses of (+)-Marrubiin and (−)-Marrulibacetal. Org. Lett. 2016, 18, 3430–3433. [Google Scholar] [CrossRef] [PubMed]
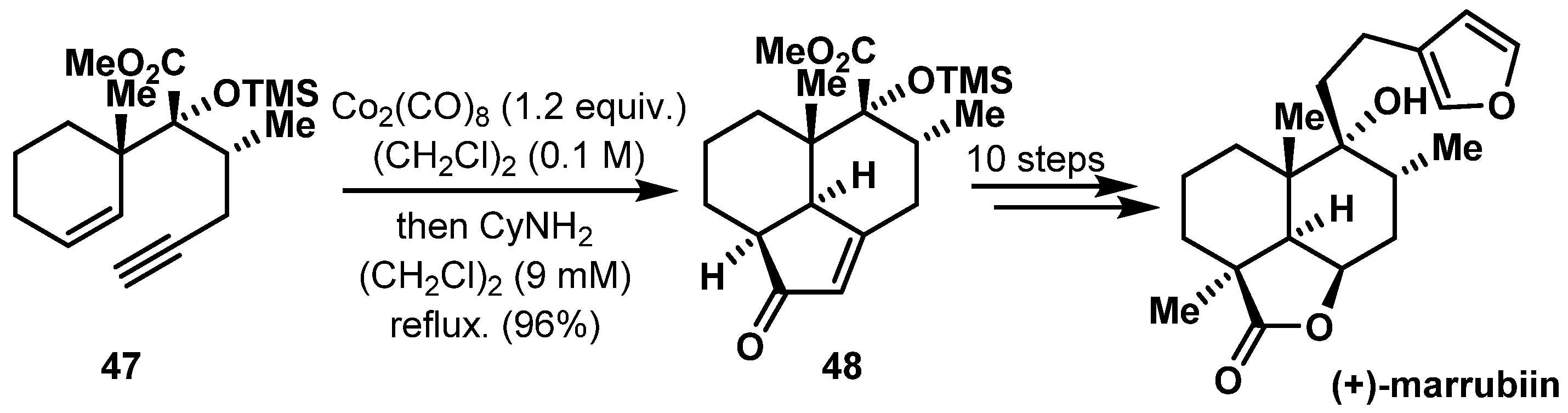
- Sakagami, Y.; Kondo, N.; Sawayama, Y.; Yamakoshi, H.; Nakamura, S. Total syntheses of marrubiin and related labdane diterpene lactones. Molecules 2020, 25, 1610. [Google Scholar] [CrossRef] [PubMed]
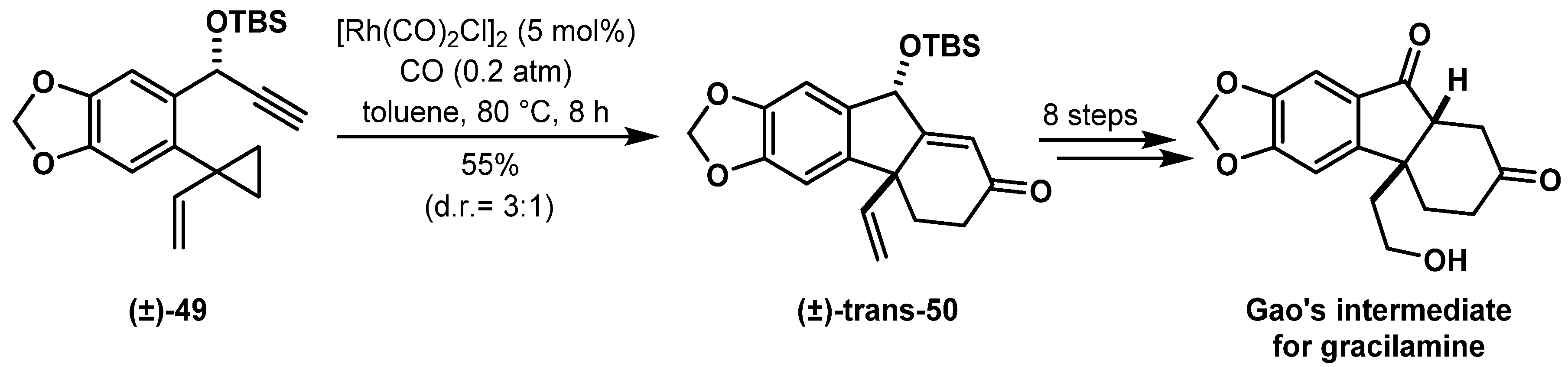
- Bose, S.; Yang, J.; Yu, Z.-X. Formal Synthesis of Gracilamine Using Rh(I)- Catalyzed [3 + 2 + 1] Cycloaddition of 1-Yne-Vinylcyclopropanes and CO. J. Org. Chem. 2016, 81, 6757–6765. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Yang, B.; Cai, S.; Gao, S. Total Synthesis of Gracilamine. Angew. Chem. Int. Ed. 2014, 53, 9539–9543. [Google Scholar] [CrossRef] [PubMed]
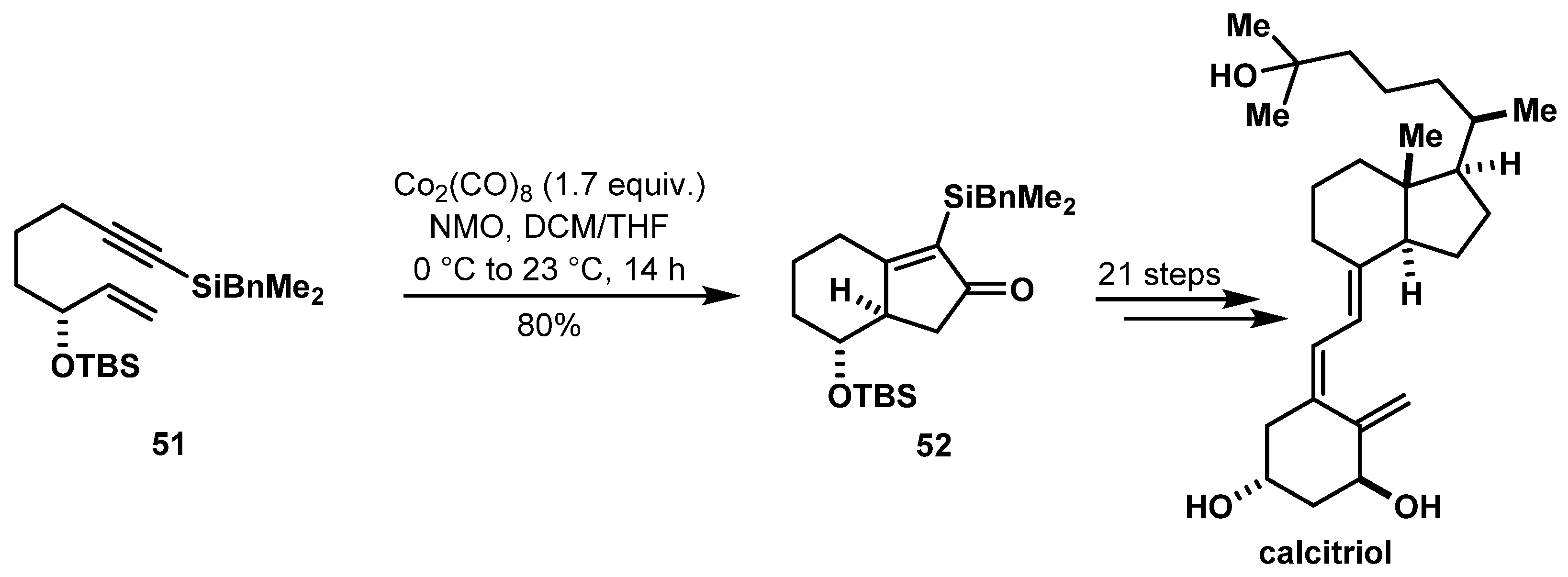
- Lopez-Perez, B.; Maestro, M.A.; Mourino, A. Total synthesis of 1α,25-dihydroxyvitamin D3 (calcitriol) through a Si-assisted allylic substitution. Chem. Commun. 2017, 53, 8144–8147. [Google Scholar] [CrossRef] [PubMed]
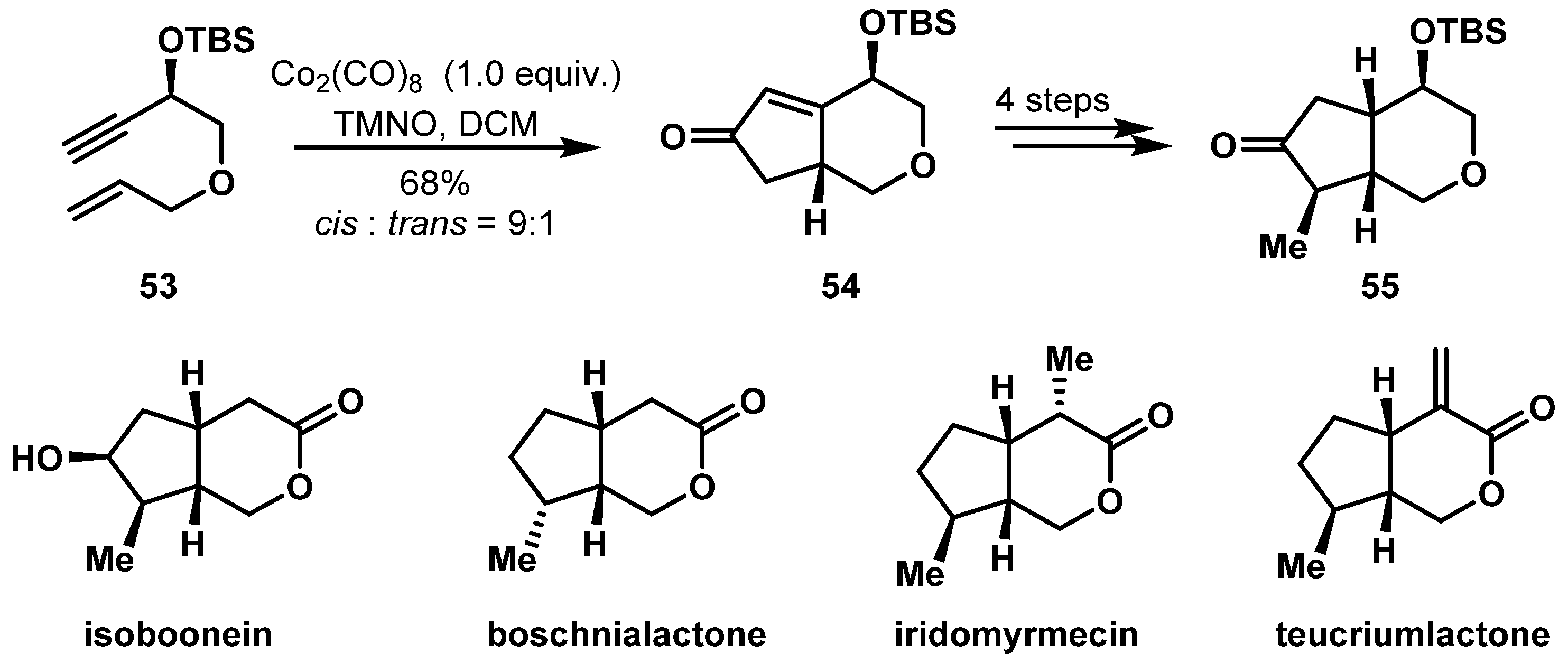
- Salam, A.; Ray, S.; Zaid, M.A.; Kumar, D.; Khan, T. Total syntheses of several iridolactones and the putative structure of noriridoid scholarein A: An intramolecular Pauson–Khand reaction based one-stop synthetic solution. Org. Biomol. Chem. 2019, 17, 6831–6842. [Google Scholar] [CrossRef]
- Kourav, M.S.; Kumar, V.; Kumar, D.; Khan, T. A De Novo Approach for the Stereoselective Synthesis of Cyclopenta[c]pyranone Scaffold Present in Iridoids: Formal Syntheses of Isoboonein, Iridomyrmecin and Isoiridomyrmecin. ChemistrySelect 2018, 3, 5566–5570. [Google Scholar] [CrossRef]
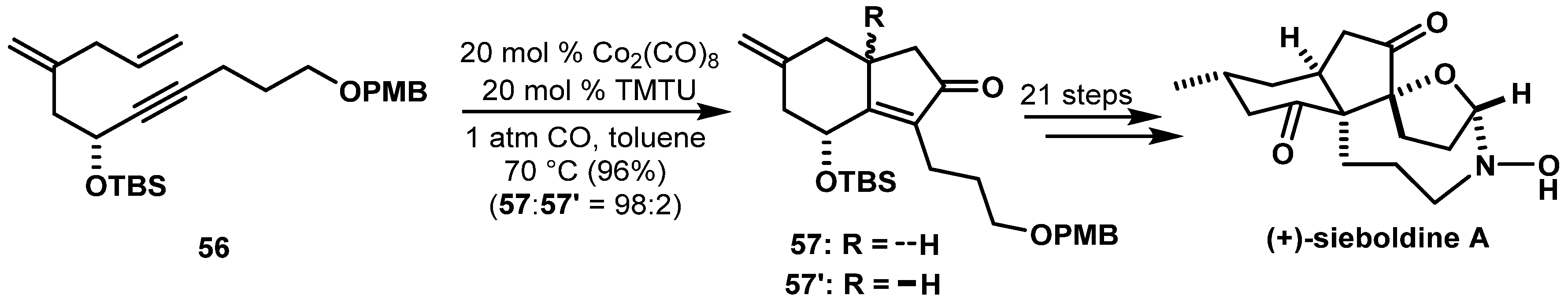
- Hirasawa, Y.; Morita, H.; Shiro, M.; Kobayashi, J. Sieboldine A, a Novel Tetracyclic Alkaloid from Lycopodium sieboldii, Inhibiting Acetylcholinesterase. Org. Lett. 2003, 5, 3991–3993. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Gaber, M.K.; Yasuda, S.; Iida, E.; Mukai, C. Enantioselective Total Synthesis of (+)-Sieboldine A. Org. Lett. 2017, 19, 320–323. [Google Scholar] [CrossRef] [PubMed]
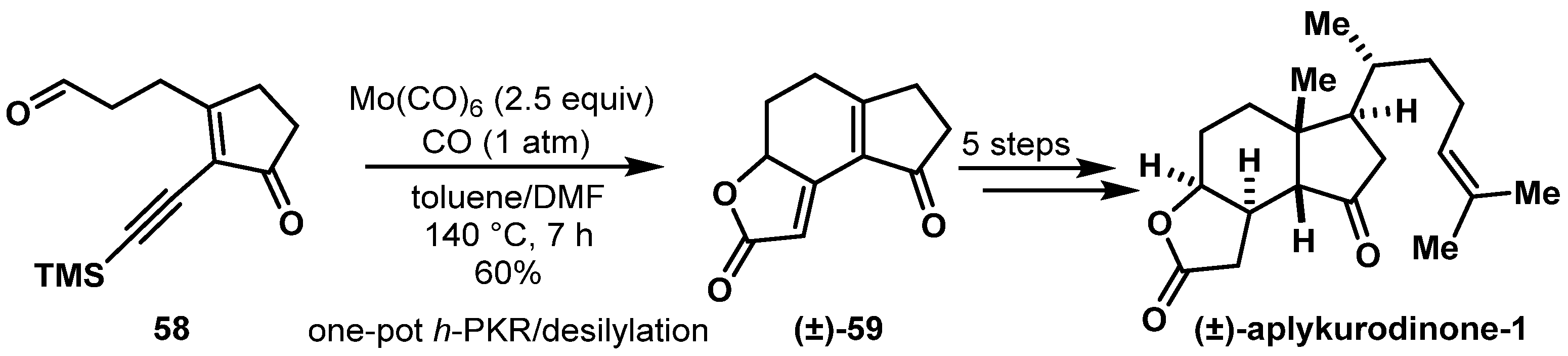
- Tao, C.; Zhang, J.; Chen, X.; Wang, H.; Li, Y.; Cheng, B.; Zhai, H. Formal Synthesis of (+/−)-Aplykurodinone-1 through a Hetero-Pauson-Khand Cycloaddition Approach. Org. Lett. 2017, 19, 1056–1059. [Google Scholar] [CrossRef] [PubMed]
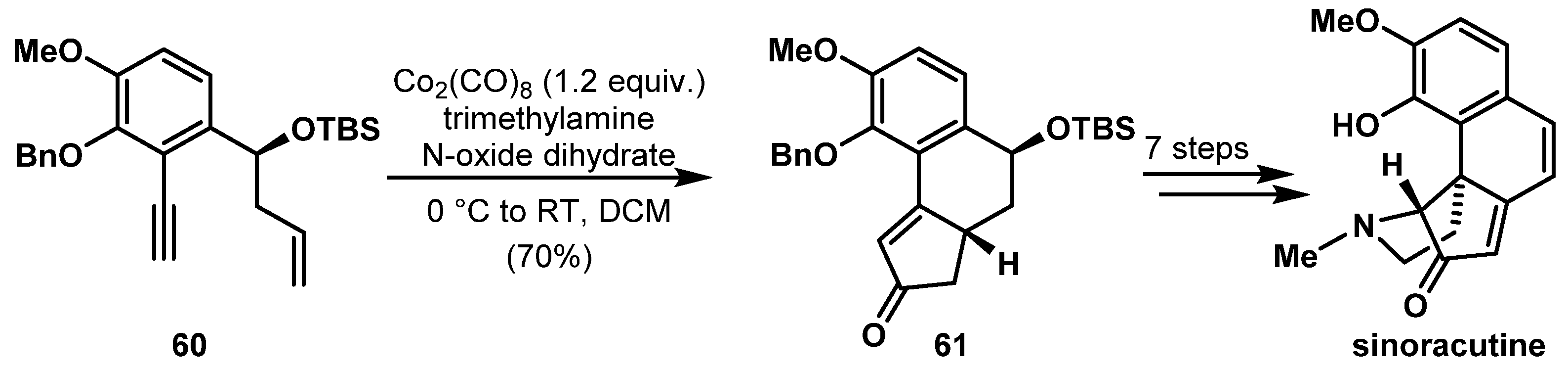
- He, L.; Deng, L.-L.; Mu, S.-Z.; Sun, Q.-Y.; Hao, X.-J.; Zhang, Y.-H. Sinoraculine, the Precursor of the Novel Alkaloid Sinoracutine from Stephania cepharantha Hayata. Helvetica Chimica Acta 2012, 95, 1198–1201. [Google Scholar] [CrossRef]
- Bao, G.-H.; Wang, X.-L.; Tang, X.-C.; Chiu, P.; Qin, G.-W. Sinoracutine, a novel skeletal alkaloid with cell-protective effects from Sinomenium acutum. Tetrahedron Lett. 2009, 50, 4375–4377. [Google Scholar] [CrossRef]
- Volpin, G.; Veprek, N.A.; Bellan, A.B.; Trauner, D. Enantioselective Synthesis and Racemization of (−)-Sinoracutine. Angew. Chem. Int. Ed. 2017, 56, 897–901. [Google Scholar] [CrossRef] [PubMed]
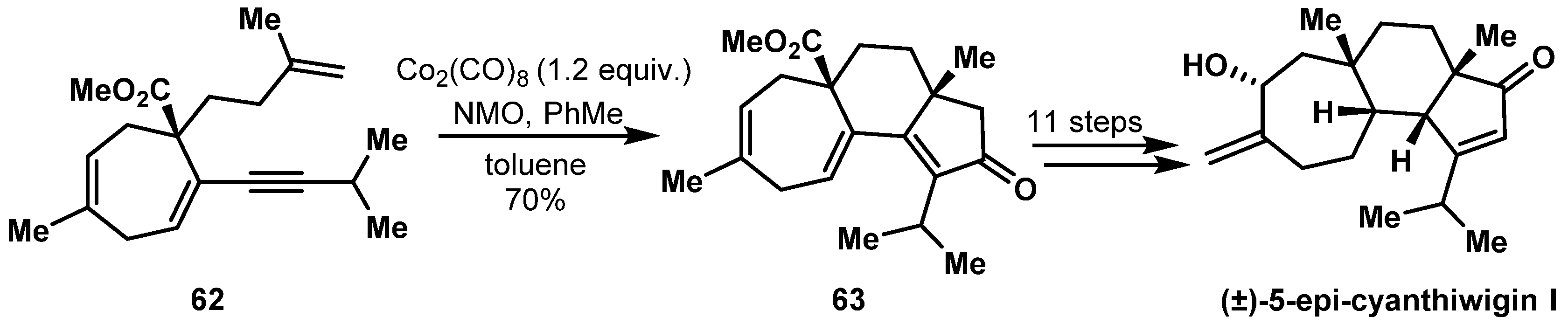
- Sennett, S.H.; Pompeni, S.A.; Wright, A.E. Diterpene Metabolites from Two Chemotypes of the Marine Sponge Myrmekioderma styx. J. Nat. Prod. 1992, 55, 1421–1429. [Google Scholar] [CrossRef]
- Chang, Y.; Shi, L.; Huang, J.; Shi, L.; Zhang, Z.; Hao, H.D.; Gong, J.; Yang, Z. Stereoselective Total Synthesis of (+/−)-5-epi-Cyanthiwigin I via an Intramolecular Pauson-Khand Reaction as the Key Step. Org. Lett. 2018, 20, 2876–2879. [Google Scholar] [CrossRef]
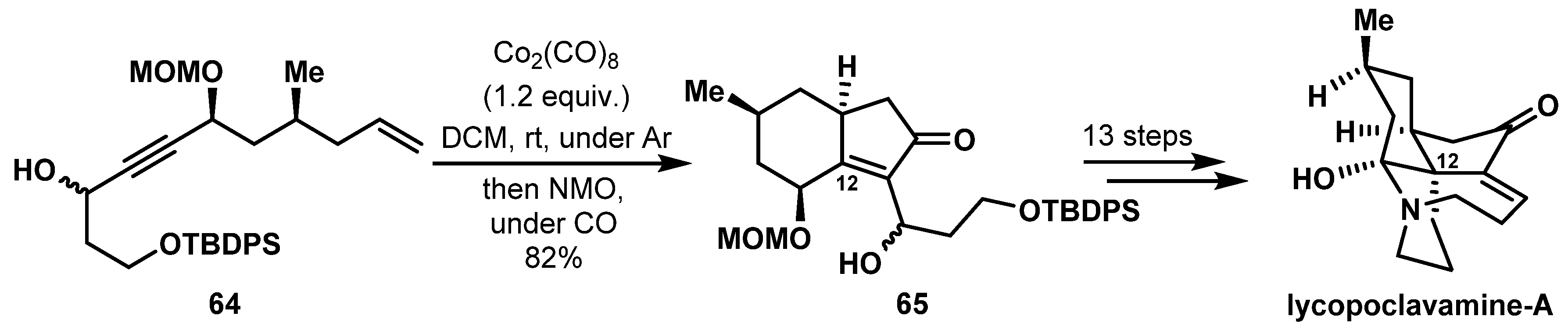
- Kaneko, H.; Takahashi, S.; Kogure, N.; Kitajima, M.; Takayama, H. Asymmetric Total Synthesis of Fawcettimine-Type Lycopodium Alkaloid, Lycopoclavamine-A. J. Org. Chem. 2019, 84, 5645–5654. [Google Scholar] [CrossRef]
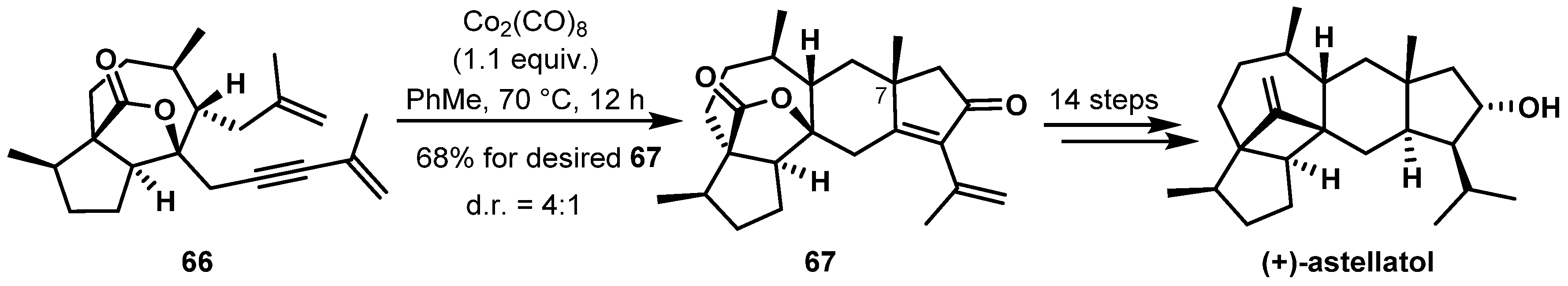
- Sadler, I.H.; Simpson, T.J. The determination by n.m.r. methods of the structure and stereochemistry of astellatol, a new and unusual sesterterpene. J. Chem. Soc. Chem. Commun. 1989, 21, 1602–1604. [Google Scholar] [CrossRef]
- Zhao, N.; Xie, S.; Tian, P.; Tong, R.; Ning, C.; Xu, J. Asymmetric total synthesis of (+)-astellatol and (−)-astellatene. Org. Chem. Front. 2019, 6, 2014–2022. [Google Scholar] [CrossRef]
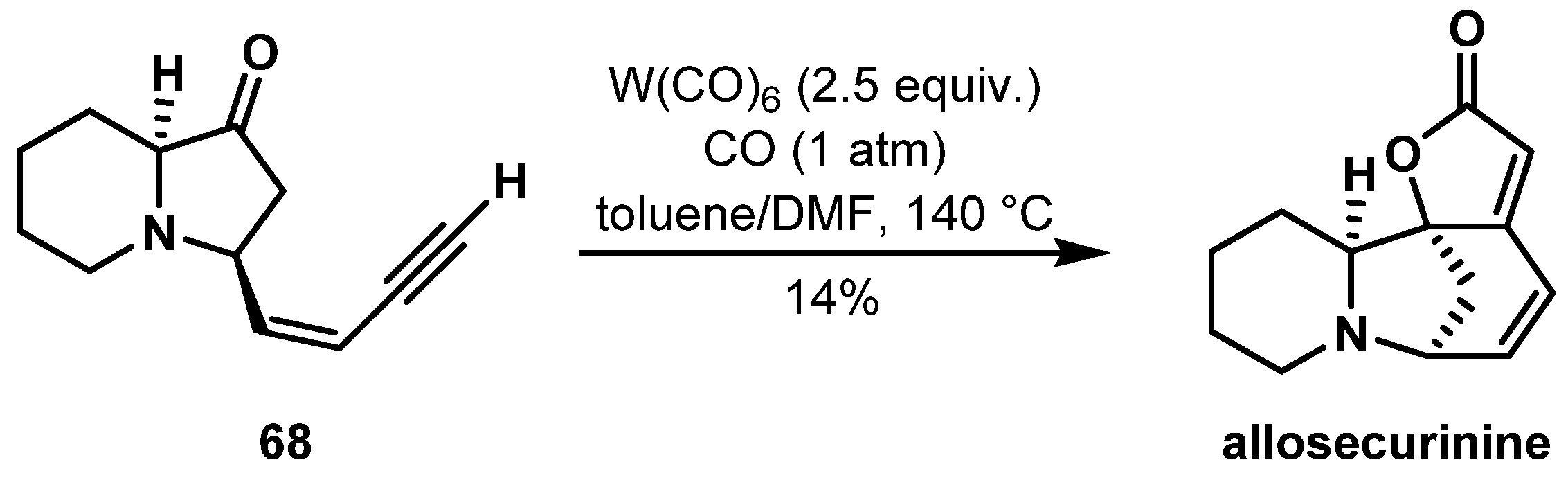
- Chirkin, E.; Bouzidi, C.; Porée, F.H. Tungsten-Promoted Hetero-Pauson–Khand Cycloaddition: Application to the Total Synthesis of (–)-Allosecurinine. Synthesis 2019, 51, 2001–2006. [Google Scholar] [CrossRef]
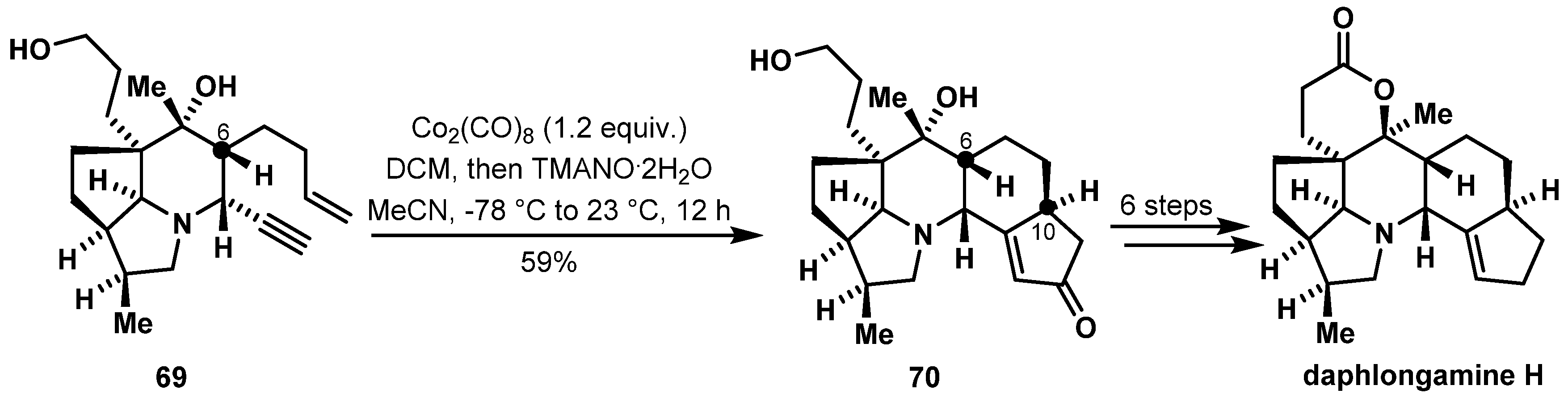
- Hugelshofer, C.L.; Palani, V.; Sarpong, R. Calyciphylline BType Alkaloids: Total Syntheses of (−)-Daphlongamine H and (−)-Isodaphlongamine H. J. Am. Chem. Soc. 2019, 141, 8431–8435. [Google Scholar] [CrossRef]
- Winther, M.; Liu, H.; Sonntag, Y.; Olesen, C.; Le Maire, M.; Soehoel, H.; Olsen, C.E.; Christensen, S.B.; Nissen, P.; MÜller, J.V. Critical Roles of Hydrophobicity and Orientation of Side Chains for Inactivation of Sarcoplasmic Reticulum Ca2+-ATPase with Thapsigargin and Thapsigargin Analogs. J. Biol. Chem. 2010, 285, 28883–28892. [Google Scholar] [CrossRef]
- Skytte, D.M.; MÜller, J.V.; Liu, H.; Nielsen, H.Ø.; Svenningsen, L.E.; Jensen, C.M.; Olsen, C.E.; Christensen, S.B. Elucidation of the Topography of the Thapsigargin Binding Site in the Sarco-endoplasmic Calcium ATPase. Bioorg. Med. Chem. 2010, 18, 5634–5646. [Google Scholar] [CrossRef] [PubMed]
- Denmeade, S.R.; Mhaka, A.M.; Rosen, D.M.; Brennen, W.N.; Dalrymple, S.; Dach, I.; Olesen, C.; Gurel, B.; DeMarzo, A.M.; Wilding, G.; et al. Engineering a Prostate-Specific Membrane Antigen-Activated Tumor Endothelial Cell Prodrug for Cancer Therapy. Sci. Transl. Med. 2012, 4, 140ra86. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.K.; Hutchison, J.J.; Fu, X.; Kunnen, K. Methods of Making Cancer Compositions. WO 2014145035 A1, 18 September 2014. [Google Scholar]
- Zimmermann, T.; Christensen, S.B.; Franzyk, H. Preparation of Enzyme-Activated Thapsigargin Prodrugs by Solid-Phase Synthesis. Molecules 2018, 23, 1463. [Google Scholar] [CrossRef]
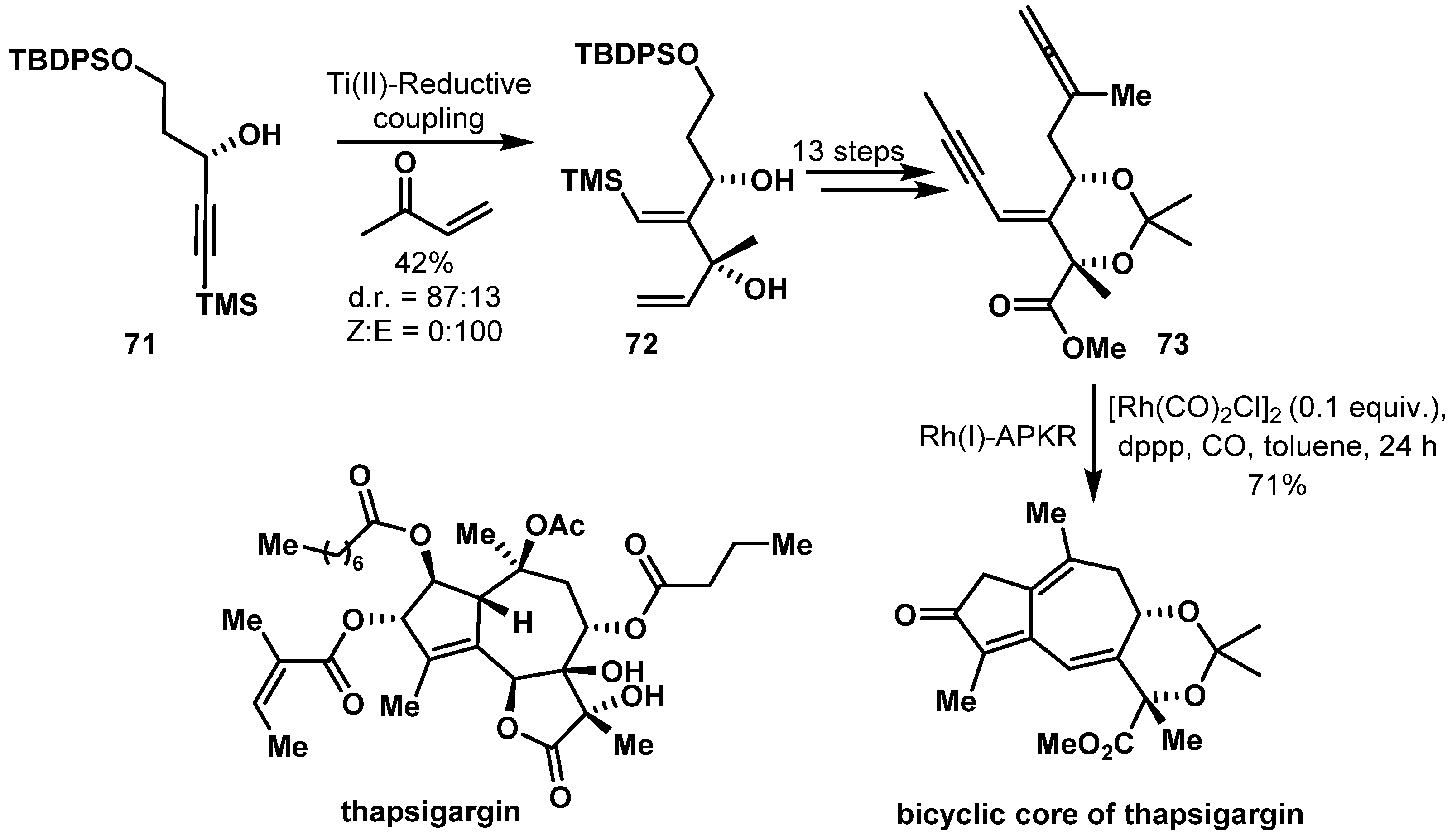
- Sanogo, Y.; Othman, R.B.; Dhambri, S.; Selkti, M.; Jeuken, A.; Prunet, J.; Ferezou, J.P.; Ardisson, J.; Lannou, M.I.; Sorin, G. Ti(II) and Rh(I) Complexes as Reagents toward a Thapsigargin Core. J. Org. Chem. 2019, 84, 5821–5830. [Google Scholar] [CrossRef]
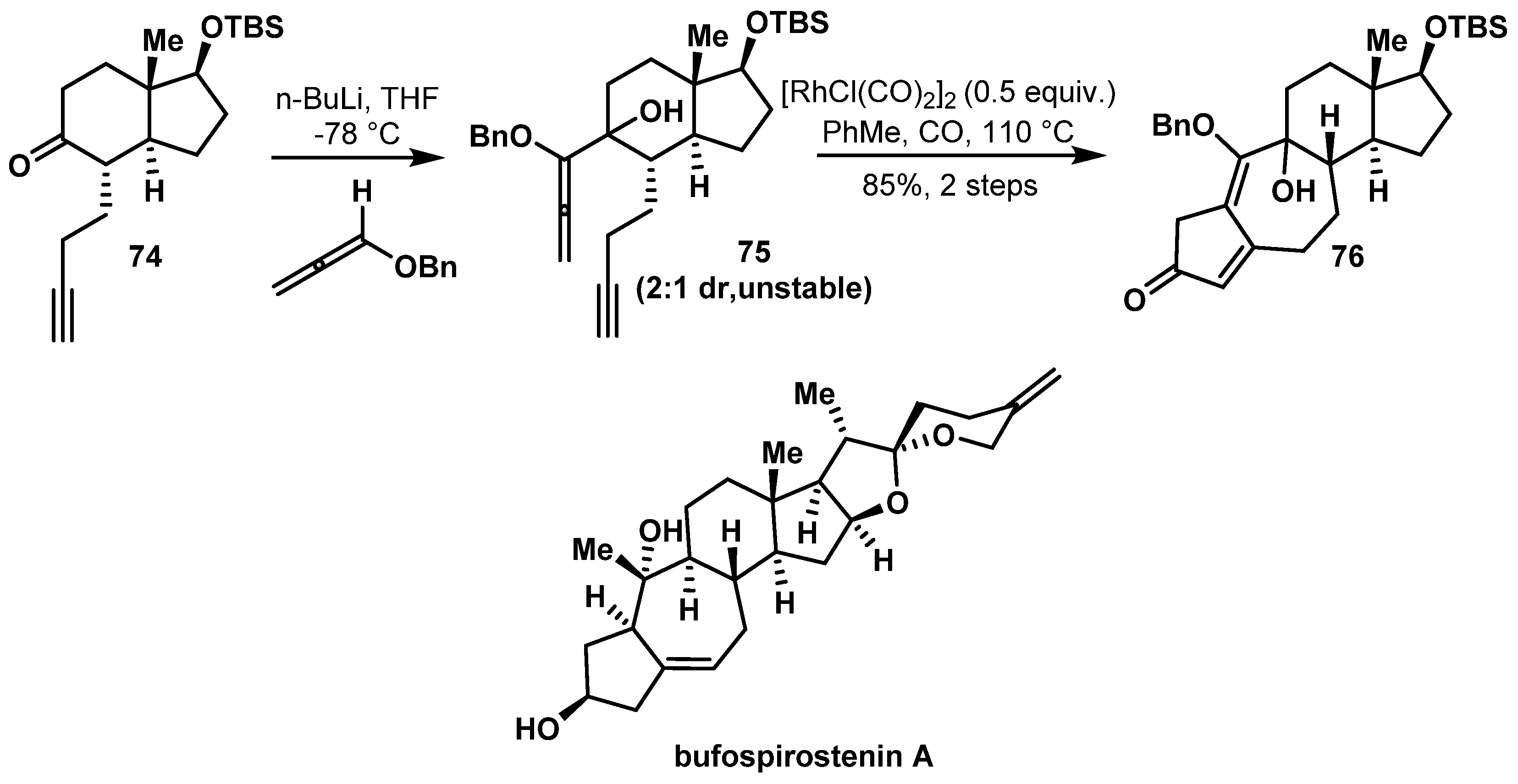
- Tian, H.Y.; Ruan, L.J.; Yu, T.; Zheng, Q.F.; Chen, N.H.; Wu, R.B.; Zhang, X.Q.; Wang, L.; Jiang, R.W.; Ye, W.C. Bufospirostenin A and Bufogargarizin C, Steroids with Rearranged Skeletons from the Toad Bufo Bufo Gargarizans. J. Nat. Prod. 2017, 80, 1182–1186. [Google Scholar] [CrossRef]
- Cheng, M.J.; Zhong, L.P.; Gu, C.C.; Zhu, X.J.; Chen, B.; Liu, J.S.; Wang, L.; Ye, W.C.; Li, C.C. Asymmetric Total Synthesis of Bufospirostenin A. J. Am. Chem. Soc. 2020, 142, 12602–12607. [Google Scholar] [CrossRef]
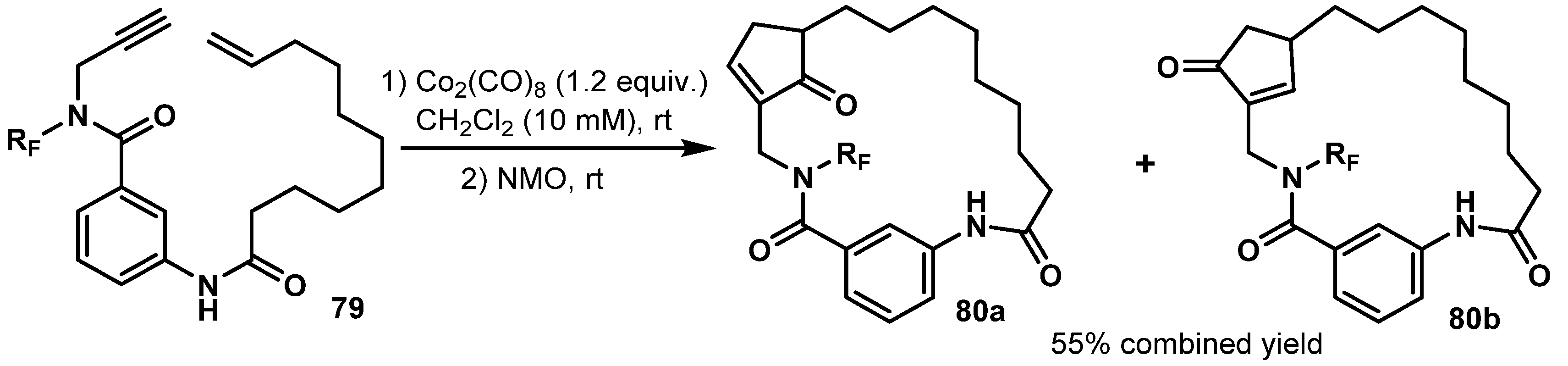
- Closser, K.D.; Quintal, M.M.; Shea, K.M. The Scope and Limitations of Intramolecular Nicholas and Pauson−Khand Reactions for the Synthesis of Tricyclic Oxygen- and Nitrogen-Containing Heterocycles. J. Org. Chem. 2009, 74, 3680–3688. [Google Scholar] [CrossRef] [PubMed]
- Comer, E.; Rohan, E.; Deng, L.; Porco, J.A. An Approach to Skeletal Diversity Using Functional Group Pairing of Multifunctional Scaffolds. Org. Lett. 2007, 9, 2123–2126. [Google Scholar] [CrossRef]
- Nie, F.; Kunciw, D.L.; Wilcke, D.; Stokes, J.E.; Galloway, W.R.; Bartlett, S.; Sore, H.F.; Spring, D.R. A Multidimensional Diversity-Oriented Synthesis Strategy for Structurally Diverse and Complex Macrocycles. Angew. Chem. Int. Ed. 2016, 55, 11139–11143. [Google Scholar] [CrossRef] [PubMed]




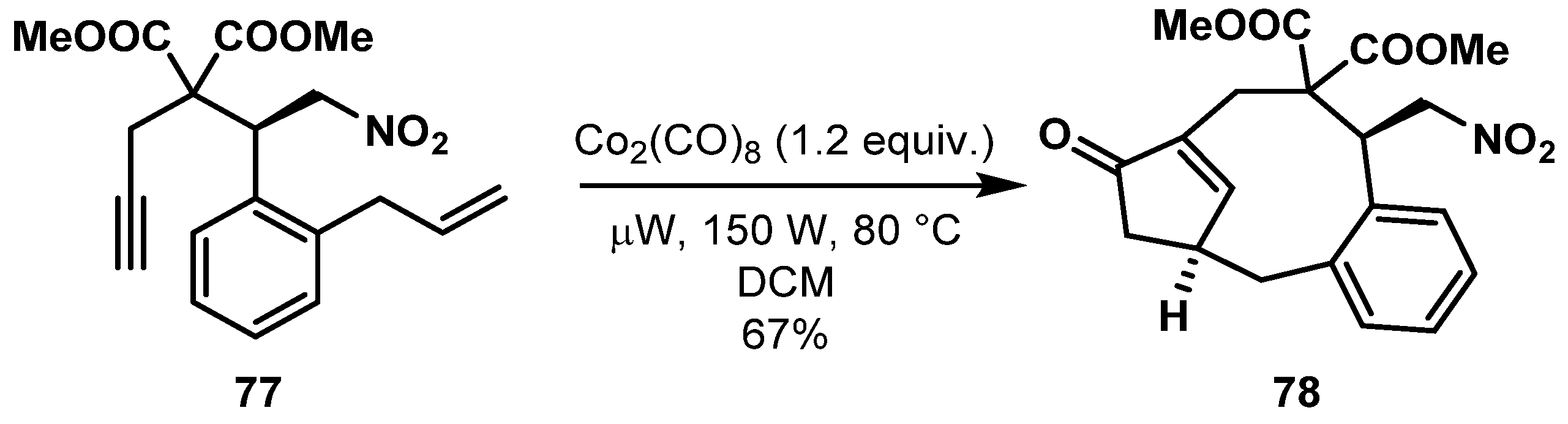
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Jiang, C.; Zheng, N.; Yang, Z.; Shi, L. Evolution of Pauson-Khand Reaction: Strategic Applications in Total Syntheses of Architecturally Complex Natural Products (2016–2020). Catalysts 2020, 10, 1199. https://doi.org/10.3390/catal10101199
Chen S, Jiang C, Zheng N, Yang Z, Shi L. Evolution of Pauson-Khand Reaction: Strategic Applications in Total Syntheses of Architecturally Complex Natural Products (2016–2020). Catalysts. 2020; 10(10):1199. https://doi.org/10.3390/catal10101199
Chicago/Turabian StyleChen, Sijia, Chongguo Jiang, Nan Zheng, Zhen Yang, and Lili Shi. 2020. "Evolution of Pauson-Khand Reaction: Strategic Applications in Total Syntheses of Architecturally Complex Natural Products (2016–2020)" Catalysts 10, no. 10: 1199. https://doi.org/10.3390/catal10101199
APA StyleChen, S., Jiang, C., Zheng, N., Yang, Z., & Shi, L. (2020). Evolution of Pauson-Khand Reaction: Strategic Applications in Total Syntheses of Architecturally Complex Natural Products (2016–2020). Catalysts, 10(10), 1199. https://doi.org/10.3390/catal10101199