Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions
Abstract
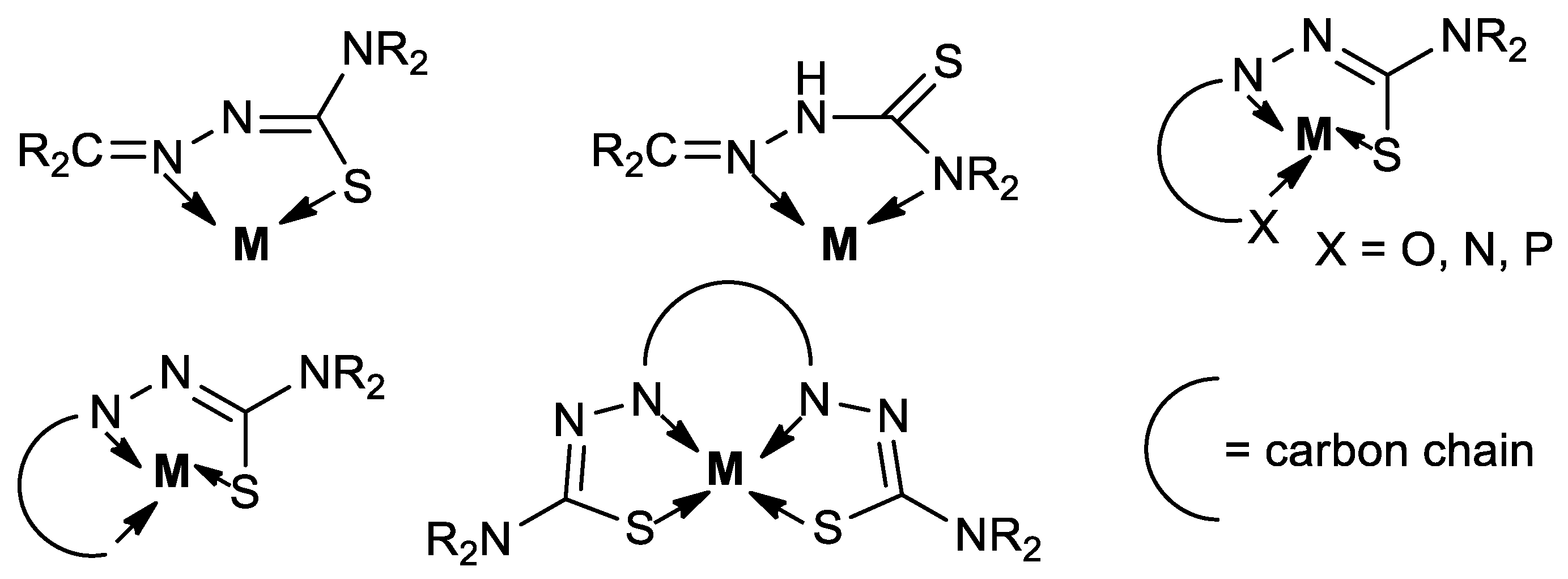
1. Introduction
2. Carbon–Carbon Coupling Reactions
2.1. Mizoroki–Heck Reaction
2.2. Suzuki–Miyaura and Related Reactions
2.3. Sonogashira and Related Reactions
2.4. Kumada–Tamao–Corriu Reaction
3. Carbon–Heteroatom Coupling Reactions
4. Immobilised and Heterogeneous Catalysts
5. Future Prospects
Author Contributions
Funding
Conflicts of Interest
References
- Biffis, A.; Centomo, P.; Del Zotto, A.; Zecca, M. Pd Metal Catalysts for cross-couplings and related reactions in the 21st century: A critical review. Chem. Rev. 2018, 118, 2249–2295. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Alonso, F.; Tyurin, V. The Suzuki-Miyaura reaction after the Nobel prize. Coord. Chem. Rev. 2019, 385, 137–173. [Google Scholar] [CrossRef]
- El-Maiss, J.; Mohy El Dine, T.; Lu, C.-S.; Karamé, I.; Kanj, A.; Polychronopoulou, K.; Shaya, J. Recent advances in metal-catalyzed alkyl–boron (C(sp3)–C(sp2)) Suzuki-Miyaura cross-couplings. Catalysts 2020, 10, 296. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Averin, A.D. New trends in the cross-coupling and other catalytic reactions. Pure Appl. Chem. 2017, 89, 1413–1428. [Google Scholar] [CrossRef]
- Kanwal, I.; Mujahid, A.; Rasool, N.; Rizwan, K.; Malik, A.; Ahmad, G.; Shah, S.A.A.; Rashid, U.; Nasir, N.M. Palladium and copper catalyzed Sonogashira cross coupling an excellent methodology for C-C bond formation over 17 years: A review. Catalysts 2020, 10, 443. [Google Scholar] [CrossRef]
- Choi, J.; Fu, G.C. Transition metal–catalyzed alkyl-alkyl bond formation: Another dimension in cross-coupling chemistry. Science 2017, 356. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S. Recent advances in the Schiff bases and N-heterocyclic carbenes as ligands in the cross-coupling reactions: A comprehensive review. J. Heterocycl. Chem. 2019, 56, 1168–1230. [Google Scholar] [CrossRef]
- Heravi, M.M.; Zadsirjan, V.; Hajiabbasi, P.; Hamidi, H. Advances in Kumada–Tamao–Corriu cross-coupling reaction: An update. Mon. Chem. Chem. Mon. 2019, 150, 535–591. [Google Scholar] [CrossRef]
- Sain, S.; Jain, S.; Srivastava, M.; Vishwakarma, R.; Dwivedi, J. Application of palladium-catalyzed cross-coupling reactions in organic synthesis. Curr. Org. Synth. 2020, 16, 1105–1142. [Google Scholar] [CrossRef]
- Mannepalli, L.K.; Gadipelly, C.; Deshmukh, G.; Likhar, P.; Pottabathula, S. Advances in C-C coupling reactions catalyzed by homogeneous phosphine free palladium catalysts. Bull. Chem. Soc. Jpn. 2020, 93, 355–372. [Google Scholar] [CrossRef]
- Zimmer, M.; Schulte, G.; Luo, X.L.; Crabtree, R.H. Functional-modeling of Ni,Fe Hydrogenases: A nickel-complex in an N,O,S environment. Angew. Chem. Int. Ed. 1991, 30, 193–194. [Google Scholar] [CrossRef]
- Vetter, A.H.; Berkessel, A. Nickel Complex Catalyzed Reduction of Imines. Synthesis (Stuttg) 1995, 1995, 419–422. [Google Scholar] [CrossRef]
- Berkessel, A.; Hermann, G.; Rauch, O.-T.; Büchner, M.; Jacobi, A.; Huttner, G. Preparation and X-ray crystal structure of the first trimeric nickel thiosemicarbazone complex: The first example of oligomerization by both Ni–O–Ni and Ni–S–Ni bridging. Chem. Ber. 1996, 129, 1421–1423. [Google Scholar] [CrossRef]
- Pelagatti, P.; Venturini, A.; Leporati, A.; Carcelli, M.; Costa, M.; Bacchi, A.; Pelizzi, G.; Pelizzi, C. Chemoselective homogeneous hydrogenation of phenylacetylene using thiosemicarbazone and thiobenzoylhydrazone palladium(II) complexes as catalysts. J. Chem. Soc. Dalt. Trans. 1998, 2715–2722. [Google Scholar] [CrossRef]
- Kovala-Demertzi, D.; Yadav, P.N.; Demertzis, M.A.; Jasiski, J.P.; Andreadaki, F.J.; Kostas, I.D. First use of a palladium complex with a thiosemicarbazone ligand as catalyst precursor for the Heck reaction. Tetrahedron Lett. 2004, 45, 2923–2926. [Google Scholar] [CrossRef]
- Kostas, I.D.; Andreadaki, F.J.; Kovala-Demertzi, D.; Christos, P.; Demertzis, M.A. Suzuki–Miyaura cross-coupling reaction of aryl bromides and chlorides with phenylboronic acid under aerobic conditions catalyzed by palladium complexes with thiosemicarbazone ligands. Tetrahedron Lett. 2005, 46, 1967–1970. [Google Scholar] [CrossRef]
- Prajapati, N.P.; Patel, H.D. Novel thiosemicarbazone derivatives and their metal complexes: Recent development. Synth. Commun. 2019, 49, 1–38. [Google Scholar] [CrossRef]
- Shakya, B.; Yadav, P.N. Thiosemicarbazones as potent anticancer agents and their modes of action. Mini Rev. Med. Chem. 2020, 20, 638–661. [Google Scholar] [CrossRef]
- de Oliveira Carneiro Brum, J.; França, T.C.C.; LaPlante, S.R.; Villar, J.D.F. Synthesis and biological activity of hydrazones and derivatives: A review. Mini Rev. Med. Chem. 2020, 20, 342–368. [Google Scholar] [CrossRef]
- Bonaccorso, C.; Marzo, T.; La Mendola, D. Biological applications of thiocarbohydrazones and their Metal complexes: A perspective review. Pharmaceuticals 2019, 13, 4. [Google Scholar] [CrossRef]
- Namiecińska, E.; Sobiesiak, M.; Małecka, M.; Guga, P.; Rozalska, B.; Budzisz, E. Antimicrobial and structural properties of metal ions complexes with thiosemicarbazide motif and related heterocyclic compounds. Curr. Med. Chem. 2019, 26, 664–693. [Google Scholar] [CrossRef]
- Summers, K.L. A structural chemistry perspective on the antimalarial properties of thiosemicarbazone metal complexes. Mini Rev. Med. Chem. 2019, 19, 569–590. [Google Scholar] [CrossRef]
- Beraldo, H.; Gambino, D. The wide pharmacological versatility of semicarbazones, thiosemicarbazones and their metal complexes. Mini Rev. Med. Chem. 2004, 4, 31–39. [Google Scholar] [CrossRef]
- Heffeter, P.; Pape, V.F.S.; Enyedy, É.A.; Keppler, B.K.; Szakacs, G.; Kowol, C.R. Anticancer thiosemicarbazones: Chemical properties, interaction with iron metabolism, and resistance development. Antioxid. Redox Signal. 2019, 30, 1062–1082. [Google Scholar] [CrossRef]
- Mckenzie-Nickson, S.; Bush, A.I.; Barnham, K.J. Bis(thiosemicarbazone) metal complexes as therapeutics for neurodegenerative diseases. Curr. Top. Med. Chem. 2016, 16, 3058–3068. [Google Scholar] [CrossRef]
- Matesanz, A.I.; Caballero, A.B.; Lorenzo, C.; Espargaró, A.; Sabaté, R.; Quiroga, A.G.; Gamez, P. Thiosemicarbazone derivatives as inhibitors of amyloid-β aggregation: Effect of metal coordination. Inorg. Chem. 2020, 59, 6978–6987. [Google Scholar] [CrossRef]
- Brown, O.C.; Baguña Torres, J.; Holt, K.B.; Blower, P.J.; Went, M.J. Copper complexes with dissymmetrically substituted bis(thiosemicarbazone) ligands as a basis for PET radiopharmaceuticals: Control of redox potential and lipophilicity. Dalt. Trans. 2017, 46, 14612–14630. [Google Scholar] [CrossRef]
- Dilworth, J.R.; Hueting, R. Metal complexes of thiosemicarbazones for imaging and therapy. Inorganica Chim. Acta 2012, 389, 3–15. [Google Scholar] [CrossRef]
- Sharma, R.K.; Pandey, A.; Gulati, S.; Adholeya, A. An optimized procedure for preconcentration, determination and on-line recovery of palladium using highly selective diphenyldiketone-monothiosemicarbazone modified silica gel. J. Hazard. Mater. 2012, 209–210, 285–292. [Google Scholar] [CrossRef]
- Bhaskar, R.; Sarveswari, S. Thiocarbohydrazide based Schiff base as a selective colorimetric and fluorescent chemosensor for Hg2+ with “turn-off” fluorescence responses. Chem. Sel. 2020, 5, 4050–4057. [Google Scholar] [CrossRef]
- Udhayakumari, D.; Suganya, S.; Velmathi, S. Thiosemicabazone based fluorescent chemosensor for transition metal ions in aqueous medium. J. Lumin. 2013, 141, 48–52. [Google Scholar] [CrossRef]
- Venkatachalam, T.K.; Bernhardt, P.V.; Pierens, G.K.; Stimson, D.H.R.; Bhalla, R.; Reutens, D.C. Synthesis and characterisation of indium(III) bis-thiosemicarbazone complexes: 18F incorporation for PET imaging. Aust. J. Chem. 2019, 72, 383. [Google Scholar] [CrossRef]
- Cortezon-Tamarit, F.; Sarpaki, S.; Calatayud, D.G.; Mirabello, V.; Pascu, S.I. Applications of “hot” and “cold” bis(thiosemicarbazonato) metal complexes in multimodal imaging. Chem. Rec. 2016, 16, 1380–1397. [Google Scholar] [CrossRef]
- Singh, R.B.; Garg, B.S.; Singh, R.P. Analytical applications of thiosemicarbazones and semicarbazones: A review. Talanta 1978, 25, 619–632. [Google Scholar] [CrossRef]
- Arion, V.B.; Platzer, S.; Rapta, P.; Machata, P.; Breza, M.; Vegh, D.; Dunsch, L.; Telser, J.; Shova, S.; Mac Leod, T.C.O.; et al. Marked stabilization of redox states and enhanced catalytic activity in galactose oxidase models based on transition metal S-methylisothiosemicarbazonates with −SR group in ortho position to the phenolic oxygen. Inorg. Chem. 2013, 52, 7524–7540. [Google Scholar] [CrossRef]
- El-Hendawy, A.M.; Fayed, A.M.; Mostafa, M.R. Complexes of a diacetylmonoxime Schiff base of S-methyldithiocarbazate (H2damsm) with Fe(III), Ru(III)/Ru(II), and V(IV); catalytic activity and X-ray crystal structure of [Fe(Hdamsm)2]NO3·H2O. Transit. Met. Chem. 2011, 36, 351–361. [Google Scholar] [CrossRef]
- Fayed, A.M.; Elsayed, S.A.; El-Hendawy, A.M.; Mostafa, M.R. Complexes of cis-dioxomolybdenum(VI) and oxovanadium(IV) with a tridentate ONS donor ligand: Synthesis, spectroscopic properties, X-ray crystal structure and catalytic activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 129, 293–302. [Google Scholar] [CrossRef]
- Bjelogrlić, S.; Todorović, T.; Bacchi, A.; Zec, M.; Sladić, D.; Srdić-Rajić, T.; Radanović, D.; Radulović, S.; Pelizzi, G.; Anđelković, K. Synthesis, structure and characterization of novel Cd(II) and Zn(II) complexes with the condensation product of 2-formylpyridine and selenosemicarbazide. J. Inorg. Biochem. 2010, 104, 673–682. [Google Scholar] [CrossRef]
- Gligorijević, N.; Todorović, T.; Radulović, S.; Sladić, D.; Filipović, N.; Gođevac, D.; Jeremić, D.; Anđelković, K. Synthesis and characterization of new Pt(II) and Pd(II) complexes with 2-quinolinecarboxaldehyde selenosemicarbazone: Cytotoxic activity evaluation of Cd(II), Zn(II), Ni(II), Pt(II) and Pd(II) complexes with heteroaromatic selenosemicarbazones. Eur. J. Med. Chem. 2009, 44, 1623–1629. [Google Scholar] [CrossRef]
- Kowol, C.R.; Eichinger, R.; Jakupec, M.A.; Galanski, M.; Arion, V.B.; Keppler, B.K. Effect of metal ion complexation and chalcogen donor identity on the antiproliferative activity of 2-acetylpyridine N,N-dimethyl(chalcogen)semicarbazones. J. Inorg. Biochem. 2007, 101, 1946–1957. [Google Scholar] [CrossRef]
- Kowol, C.R.; Reisner, E.; Chiorescu, I.; Arion, V.B.; Galanski, M.; Deubel, D.V.; Keppler, B.K. An electrochemical study of antineoplastic gallium, iron and ruthenium complexes with redox noninnocent α-N-heterocyclic chalcogensemicarbazones. Inorg. Chem. 2008, 47, 11032–11047. [Google Scholar] [CrossRef]
- Molter, A.; Kaluđerović, G.N.; Kommera, H.; Paschke, R.; Langer, T.; Pöttgen, R.; Mohr, F. Synthesis, structures, 119Sn Mössbauer spectroscopic studies and biological activity of some tin(IV) complexes containing pyridyl functionalised selenosemicarbazonato ligands. J. Organomet. Chem. 2012, 701, 80–86. [Google Scholar] [CrossRef]
- Sarhan, A.M.; Elsayed, S.A.; Mashaly, M.M.; El-Hendawy, A.M. Oxovanadium(IV) and ruthenium(II) carbonyl complexes of ONS-donor ligands derived from dehydroacetic acid and dithiocarbazate: Synthesis, characterization, antioxidant activity, DNA binding and in vitro cytotoxicity. Appl. Organomet. Chem. 2019, 33, e4655. [Google Scholar] [CrossRef]
- Shen, H.; Zhu, H.; Song, M.; Tian, Y.; Huang, Y.; Zheng, H.; Cao, R.; Lin, J.; Bi, Z.; Zhong, W. A selenosemicarbazone complex with copper efficiently down-regulates the 90-kDa heat shock protein HSP90AA1 and its client proteins in cancer cells. BMC Cancer 2014, 14, 629. [Google Scholar] [CrossRef]
- Srdić-Rajić, T.; Zec, M.; Todorović, T.; Anđelković, K.; Radulović, S. Non-substituted N-heteroaromatic selenosemicarbazone metal complexes induce apoptosis in cancer cells via activation of mitochondrial pathway. Eur. J. Med. Chem. 2011, 46, 3734–3747. [Google Scholar] [CrossRef]
- Todorović, T.R.; Vukašinović, J.; Portalone, G.; Suleiman, S.; Gligorijević, N.; Bjelogrlić, S.; Jovanović, K.; Radulović, S.; Anđelković, K.; Cassar, A.; et al. (Chalcogen)semicarbazones and their cobalt complexes differentiate HL-60 myeloid leukaemia cells and are cytotoxic towards tumor cell lines. Medchemcomm 2017, 8, 103–111. [Google Scholar] [CrossRef]
- Zec, M.; Srdic-Rajic, T.; Krivokuca, A.; Jankovic, R.; Todorovic, T.; Andelkovic, K.; Radulovic, S. Novel selenosemicarbazone metal complexes exert anti-tumor effect via alternative, caspase-independent necroptotic cell death. Med. Chem. 2014, 10, 759–771. [Google Scholar] [CrossRef]
- Mawat, T.H.; Al-Jeboori, M.J. Synthesis, characterisation, thermal properties and biological activity of coordination compounds of novel selenosemicarbazone ligands. J. Mol. Struct. 2020, 1208, 127876. [Google Scholar] [CrossRef]
- Castle, T.C.; Maurer, R.I.; Sowrey, F.E.; Went, M.J.; Reynolds, C.A.; McInnes, E.J.L.; Blower, P.J. Hypoxia-targeting copper bis(selenosemicarbazone) complexes: Comparison with their sulfur analogues. J. Am. Chem. Soc. 2003, 125, 10040–10049. [Google Scholar] [CrossRef]
- McQuade, P.; Martin, K.E.; Castle, T.C.; Went, M.J.; Blower, P.J.; Welch, M.J.; Lewis, J.S. Investigation into 64Cu-labeled Bis(selenosemicarbazone) and Bis(thiosemicarbazone) complexes as hypoxia imaging agents. Nucl. Med. Biol. 2005, 32, 147–156. [Google Scholar] [CrossRef]
- Dekanski, D.; Todorovic, T.; Mitic, D.; Filipovic, N.; Polovic, N.; Andjelkovic, K. High antioxidative potential and low toxic effects of selenosemicarbazone metal complexes. J. Serbian Chem. Soc. 2013, 78, 1503–1512. [Google Scholar] [CrossRef]
- Djordjevic, I.; Vukasinovic, J.; Todorovic, T.; Filipovic, N.; Rodic, M.; Lolic, A.; Portalone, G.; Zlatovic, M.; Grubisic, S. Synthesis, structures and electronic properties of Co(III) complexes with 2-quinolinecarboxaldehyde thio- and selenosemicarbazone: A combined experimental and theoretical study. J. Serb. Chem. Soc. 2017, 82, 825–839. [Google Scholar] [CrossRef]
- Campbell, M.J.M. Transition metal complexes of thiosemicarbazide and thiosemicarbazones. Coord. Chem. Rev. 1975, 15, 279–319. [Google Scholar] [CrossRef]
- Casas, J.S.; García-Tasende, M.S.; Sordo, J. Main group metal complexes of semicarbazones and thiosemicarbazones. A structural review. Coord. Chem. Rev. 2000, 209, 197–261. [Google Scholar] [CrossRef]
- Padhyé, S.; Kauffman, G.B. Transition metal complexes of semicarbazones and thiosemicarbazones. Coord. Chem. Rev. 1985, 63, 127–160. [Google Scholar] [CrossRef]
- Selander, N.; Szabó, K.J. Catalysis by palladium pincer complexes. Chem. Rev. 2011, 111, 2048–2076. [Google Scholar] [CrossRef]
- Phan, N.T.S.; Van Der Sluys, M.; Jones, C.W. On the nature of the active species in palladium catalyzed Mizoroki–Heck and Suzuki–Miyaura couplings–homogeneous or heterogeneous catalysis, a critical review. Adv. Synth. Catal. 2006, 348, 609–679. [Google Scholar] [CrossRef]
- Anitha, P.; Manikandan, R.; Endo, A.; Hashimoto, T.; Viswanathamurthi, P. Ruthenium(II) complexes containing quinone based ligands: Synthesis, characterization, catalytic applications and DNA interaction. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 99, 174–180. [Google Scholar] [CrossRef]
- Asha, T.M.; Kurup, M.R.P. Synthesis, structural insights and catalytic activity of a dioxidomolybdenum(VI) complex chelated with N4-(3-methoxyphenyl) thiosemicarbazone. Transit. Met. Chem. 2020. [Google Scholar] [CrossRef]
- Hosseini Monfared, H.; Kheirabadi, S.; Asghari Lalami, N.; Mayer, P. Dioxo- and oxovanadium(V) complexes of biomimetic hydrazone ONO and NNS donor ligands: Synthesis, crystal structure and catalytic reactivity. Polyhedron 2011, 30, 1375–1384. [Google Scholar] [CrossRef]
- Islam, M.; Hossain, D.; Mondal, P.; Tuhina, K.; Roy, A.S.; Mondal, S.; Mobarak, M. Synthesis, characterization, and catalytic activity of a polymer-supported copper(II) complex with a thiosemicarbazone ligand. Transit. Met. Chem. 2011, 36, 223–230. [Google Scholar] [CrossRef]
- Raja, G.; Sathya, N.; Jayabalakrishnan, C. Spectroscopic, catalytic, and biological studies on mononuclear ruthenium(II) ONSN chelating thiosemicarbazone complexes. J. Coord. Chem. 2011, 64, 817–831. [Google Scholar] [CrossRef]
- Roy, S.; Saswati; Lima, S.; Dhaka, S.; Maurya, M.R.; Acharyya, R.; Eagle, C.; Dinda, R. Synthesis, structural studies and catalytic activity of a series of dioxidomolybdenum(VI)-thiosemicarbazone complexes. Inorg. Chim. Acta 2018, 474, 134–143. [Google Scholar] [CrossRef]
- Thilagavathi, N.; Manimaran, A.; Padma Priya, N.; Sathya, N.; Jayabalakrishnan, C. Synthesis, spectroscopic, redox, catalytic and antimicrobial properties of some ruthenium(II) Schiff base complexes. Transit. Met. Chem. 2009, 34, 725–732. [Google Scholar] [CrossRef]
- Ulaganatha Raja, M.; Gowri, N.; Ramesh, R. Synthesis, crystal structure and catalytic activity of ruthenium(II) carbonyl complexes containing ONO and ONS donor ligands. Polyhedron 2010, 29, 1175–1181. [Google Scholar] [CrossRef]
- Wang, F.-M. Mononuclear oxovanadium(Iv) complex containing VO(ONS) basic core: Synthesis, structure, thermal gravimetric analysis, and catalytic property. Inorg. Nano Met. Chem. 2017, 47, 1380–1384. [Google Scholar] [CrossRef]
- Maurya, M.R.; Dhaka, S.; Avecilla, F. Synthesis, characterization, reactivity and catalytic activity of dioxidomolybdenum(VI) complexes derived from tribasic ONS donor ligands. Polyhedron 2014, 81, 154–167. [Google Scholar] [CrossRef]
- Maurya, A.; Kesharwani, N.; Kachhap, P.; Mishra, V.K.; Chaudhary, N.; Haldar, C. Polymer-anchored mononuclear and binuclear CuII Schiff-base complexes: Impact of heterogenization on liquid phase catalytic oxidation of a series of alkenes. Appl. Organomet. Chem. 2019, 33. [Google Scholar] [CrossRef]
- Moradi-Shoeili, Z.; Boghaei, D.M.; Amini, M.; Bagherzadeh, M.; Notash, B. New molybdenum(VI) complex with ONS-donor thiosemicarbazone ligand: Preparation, structural characterization, and catalytic applications in olefin epoxidation. Inorg. Chem. Commun. 2013, 27, 26–30. [Google Scholar] [CrossRef]
- Moradi-Shoeili, Z.; Zare, M. The effect of substituents on catalytic performance of bis-thiosemicarbazone Mo(VI) complexes: Synthesis and spectroscopic, electrochemical, and functional properties. Kinet. Catal. 2018, 59, 203–210. [Google Scholar] [CrossRef]
- Muthukumar, M.; Viswanathamurthi, P. Synthesis, spectral characterization and catalytic studies of new ruthenium(II) chalcone thiosemicarbazone complexes. Open Chem. 2010, 8, 229–240. [Google Scholar] [CrossRef]
- Nehar, O.K.; Mahboub, R.; Louhibi, S.; Roisnel, T.; Aissaoui, M. New thiosemicarbazone Schiff base ligands: Synthesis, characterization, catecholase study and hemolytic activity. J. Mol. Struct. 2020, 1204, 127566. [Google Scholar] [CrossRef]
- Ngan, N.K.; Lo, K.M.; Wong, C.S.R. Dinuclear and polynuclear dioxomolybdenum(VI) Schiff base complexes: Synthesis, structural elucidation, spectroscopic characterization, electrochemistry and catalytic property. Polyhedron 2012, 33, 235–251. [Google Scholar] [CrossRef]
- Nirmala, M.; Manikandan, R.; Prakash, G.; Viswanathamurthi, P. Ruthenium(II) complexes of hybrid 8-hydroxyquinoline-thiosemicarbazone ligands: Synthesis, characterization and catalytic applications. Appl. Organomet. Chem. 2014, 28, 18–26. [Google Scholar] [CrossRef]
- Biswas, S.; Sarkar, D.; Roy, P.; Mondal, T.K. Synthesis and characterization of a ruthenium complex with bis(diphenylphosphino)propane and thioether containing ONS donor ligand: Application in transfer hydrogenation of ketones. Polyhedron 2017, 131, 1–7. [Google Scholar] [CrossRef]
- Manikandan, R.; Anitha, P.; Prakash, G.; Vijayan, P.; Viswanathamurthi, P. Synthesis, spectral characterization and crystal structure of Ni(II) pyridoxal thiosemicarbazone complexes and their recyclable catalytic application in the nitroaldol (Henry) reaction in ionic liquid media. Polyhedron 2014, 81, 619–627. [Google Scholar] [CrossRef]
- Raja, N.; Ramesh, R. Mononuclear ruthenium(III) complexes containing chelating thiosemicarbazones: Synthesis, characterization and catalytic property. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 75, 713–718. [Google Scholar] [CrossRef]
- Barber, D.E.; Lu, Z.; Richardson, T.; Crabtree, R.H. Silane alcoholysis by a nickel(II) complex in a N, O, S ligand environment. Inorg. Chem. 1992, 31, 4709–4711. [Google Scholar] [CrossRef]
- Anitha, P.; Manikandan, R.; Vijayan, P.; Anbuselvi, S.; Viswanathamurthi, P. Rhodium(I) complexes containing 9,10-phenanthrenequinone-N-substituted thiosemicarbazone ligands: Synthesis, structure, DFT study and catalytic diastereoselective nitroaldol reaction studies. J. Organomet. Chem. 2015, 791, 244–251. [Google Scholar] [CrossRef]
- Anitha, P.; Viswanathamurthi, P.; Kesavan, D.; Butcher, R.J. Ruthenium(II) 9,10-phenanthrenequinone thiosemicarbazone complexes: Synthesis, characterization, and catalytic activity towards the reduction as well as condensation of nitriles. J. Coord. Chem. 2015, 68, 321–334. [Google Scholar] [CrossRef]
- Manikandan, R.; Viswnathamurthi, P. Coordination behavior of ligand based on NNS and NNO donors with ruthenium(III) complexes and their catalytic and DNA interaction studies. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 97, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Youssef, N.S.; El-Zahany, E.; El-Seidy, A.M.A.; Caselli, A.; Fantauzzi, S.; Cenini, S. Synthesis and characterisation of new Schiff base metal complexes and their use as catalysts for olefin cyclopropanation. Inorg. Chim. Acta 2009, 362, 2006–2014. [Google Scholar] [CrossRef]
- Youssef, N.S.; El-Seidy, A.M.A.; Schiavoni, M.; Castano, B.; Ragaini, F.; Gallo, E.; Caselli, A. Thiosemicarbazone copper complexes as competent catalysts for olefin cyclopropanations. J. Organomet. Chem. 2012, 714, 94–103. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar Rao, G.; Singh, A.K. Organochalcogen ligands and their palladium(II) complexes: Synthesis to catalytic activity for Heck coupling. RSC Adv. 2012, 2, 12552. [Google Scholar] [CrossRef]
- Kumar, A.; Rao, G.K.; Kumar, S.; Singh, A.K. Organosulphur and related ligands in Suzuki–Miyaura C–C coupling. Dalt. Trans. 2013, 42, 5200. [Google Scholar] [CrossRef]
- Kumar, A.; Rao, G.K.; Kumar, S.; Singh, A.K. Formation and role of palladium chalcogenide and other species in Suzuki–Miyaura and Heck C–C coupling reactions catalyzed with palladium(II) complexes of organochalcogen ligands: Realities and speculations. Organometallics 2014, 33, 2921–2943. [Google Scholar] [CrossRef]
- Eremin, D.B.; Ananikov, V.P. Understanding active species in catalytic transformations: From molecular catalysis to nanoparticles, leaching, “cocktails” of catalysts and dynamic systems. Coord. Chem. Rev. 2017, 346, 2–19. [Google Scholar] [CrossRef]
- Polynski, M.V.; Ananikov, V.P. Modeling Key pathways proposed for the formation and evolution of “cocktail”-type systems in Pd-catalyzed reactions involving ArX reagents. ACS Catal. 2019, 9, 3991–4005. [Google Scholar] [CrossRef]
- Ortuño, M.A.; López, N. Reaction mechanisms at the homogeneous–heterogeneous frontier: Insights from first-principles studies on ligand-decorated metal nanoparticles. Catal. Sci. Technol. 2019, 9, 5173–5185. [Google Scholar] [CrossRef]
- Trzeciak, A.M.; Augustyniak, A.W. The role of palladium nanoparticles in catalytic C–C cross-coupling reactions. Coord. Chem. Rev. 2019, 384, 1–20. [Google Scholar] [CrossRef]
- Bourouina, A.; Meille, V.; de Bellefon, C. About solid phase vs. liquid phase in Suzuki-Miyaura reaction. Catalysts 2019, 9, 60. [Google Scholar] [CrossRef]
- Sun, B.; Ning, L.; Zeng, H.C. Confirmation of Suzuki–Miyaura cross-coupling reaction mechanism through synthetic architecture of nanocatalysts. J. Am. Chem. Soc. 2020, 142, 13823–13832. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, S. Heck reaction—State of the art. Catalysts 2017, 7, 267. [Google Scholar] [CrossRef]
- Xie, G.; Chellan, P.; Mao, J.; Chibale, K.; Smith, G.S. Thiosemicarbazone salicylaldiminato-palladium(II)-catalyzed Mizoroki-Heck reactions. Adv. Synth. Catal. 2010, 352, 1641–1647. [Google Scholar] [CrossRef]
- Paul, P.; Datta, S.; Halder, S.; Acharyya, R.; Basuli, F.; Butcher, R.J.; Peng, S.-M.; Lee, G.-H.; Castiñeiras, A.; Drew, M.G.B.; et al. Syntheses, structures and efficient catalysis for C–C coupling of some benzaldehyde thiosemicarbazone complexes of palladium. J. Mol. Catal. A Chem. 2011, 344, 62–73. [Google Scholar] [CrossRef]
- Prabhu, R.N.; Ramesh, R. Catalytic application of dinuclear palladium(II) bis(thiosemicarbazone) complex in the Mizoroki-Heck reaction. Tetrahedron Lett. 2012, 53, 5961–5965. [Google Scholar] [CrossRef]
- Datta, S.; Seth, D.K.; Gangopadhyay, S.; Karmakar, P.; Bhattacharya, S. Nickel complexes of some thiosemicarbazones: Synthesis, structure, catalytic properties and cytotoxicity studies. Inorganica Chim. Acta 2012, 392, 118–130. [Google Scholar] [CrossRef]
- Suganthy, P.K.; Prabhu, R.N.; Sridevi, V.S. Nickel(II) thiosemicarbazone complex catalyzed Mizoroki–Heck reaction. Tetrahedron Lett. 2013, 54, 5695–5698. [Google Scholar] [CrossRef]
- Lima, J.C.; Nascimento, R.D.; Vilarinho, L.M.; Borges, A.P.; Silva, L.H.F.; Souza, J.R.; Dinelli, L.R.; Deflon, V.M.; da Hora Machado, A.E.; Bogado, A.L.; et al. Group 10 metal complexes with a tetradentate thiosemicarbazonate ligand: Synthesis, crystal structures and computational insights into the catalysis for C–C coupling via Mizoroki-Heck reaction. J. Mol. Struct. 2020, 1199, 126997. [Google Scholar] [CrossRef]
- Kostas, I.D.; Heropoulos, G.A.; Kovala-Demertzi, D.; Yadav, P.N.; Jasinski, J.P.; Demertzis, M.A.; Andreadaki, F.J.; Vo-Thanh, G.; Petit, A.; Loupy, A. Microwave-promoted Suzuki–Miyaura cross-coupling of aryl halides with phenylboronic acid under aerobic conditions catalyzed by a new palladium complex with a thiosemicarbazone ligand. Tetrahedron Lett. 2006, 47, 4403–4407. [Google Scholar] [CrossRef]
- Perreux, L.; Loupy, A. A tentative rationalization of microwave effects in organic synthesis according to the reaction medium, and mechanistic considerations. Tetrahedron 2001, 57, 9199–9223. [Google Scholar] [CrossRef]
- Paul, P.; Bhattacharya, S. Organometallic complexes of the platinum metals: Synthesis, structure, and catalytic applications. J. Chem. Sci. 2012, 124, 1165–1173. [Google Scholar] [CrossRef]
- Castiñeiras, A.; Fernandez-Hermida, N.; Garcia-Santos, I.; Gomez-Rodriguez, L. Neutral NiII, PdII and PtII ONS-pincer complexes of 5-acetylbarbituric-4N-dimethylthiosemicarbazone: Synthesis, characterization and properties. Dalt. Trans. 2012, 41, 13486–13495. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, W.A.; Reisinger, C.-P.; Öfele, K.; Broβmer, C.; Beller, M.; Fischer, H. Facile catalytic coupling of aryl bromides with terminal alkynes by phospha-palladacycles. J. Mol. Catal. A Chem. 1996, 108, 51–56. [Google Scholar] [CrossRef]
- Ohff, M.; Ohff, A.; van der Boom, M.E.; Milstein, D. Highly active Pd(II) PCP-type catalysts for the Heck reaction. J. Am. Chem. Soc. 1997, 119, 11687–11688. [Google Scholar] [CrossRef]
- Dutta, J.; Datta, S.; Kumar Seth, D.; Bhattacharya, S. Mixed-ligand benzaldehyde thiosemicarbazone complexes of palladium containing N,O-donor ancillary ligands. Syntheses, structures, and catalytic application in C–C and C–N coupling reactions. RSC Adv. 2012, 2, 11751. [Google Scholar] [CrossRef]
- Dutta, J.; Bhattacharya, S. Controlled interaction of benzaldehyde thiosemicarbazones with palladium: Formation of bis-complexes with cis-geometry and organopalladium complexes, and their catalytic application in C–C and C–N coupling. RSC Adv. 2013, 3, 10707. [Google Scholar] [CrossRef]
- Paul, P.; Sengupta, P.; Bhattacharya, S. Palladium mediated C–H bond activation of thiosemicarbazones: Catalytic application of organopalladium complexes in C–C and C–N coupling reactions. J. Organomet. Chem. 2013, 724, 281–288. [Google Scholar] [CrossRef]
- Yan, H.; Chellan, P.; Li, T.; Mao, J.; Chibale, K.; Smith, G.S. Cyclometallated Pd(II) thiosemicarbazone complexes: New catalyst precursors for Suzuki-coupling reactions. Tetrahedron Lett. 2013, 54, 154–157. [Google Scholar] [CrossRef]
- Pandiarajan, D.; Ramesh, R.; Liu, Y.; Suresh, R. Palladium(II) thiosemicarbazone-catalyzed Suzuki–Miyaura cross-coupling reactions of aryl halides. Inorg. Chem. Commun. 2013, 33, 33–37. [Google Scholar] [CrossRef]
- Verma, P.R.; Mandal, S.; Gupta, P.; Mukhopadhyay, B. Carbohydrate derived thiosemicarbazone and semicarbazone palladium complexes: Homogeneous catalyst for C–C cross coupling reactions. Tetrahedron Lett. 2013, 54, 4914–4917. [Google Scholar] [CrossRef]
- Tenchiu, A.-C.; Ventouri, I.-K.; Ntasi, G.; Palles, D.; Kokotos, G.; Kovala-Demertzi, D.; Kostas, I.D. Synthesis of a palladium complex with a β-D-glucopyranosyl-thiosemicarbazone and its application in the Suzuki–Miyaura coupling of aryl bromides with phenylboronic acid. Inorg. Chim. Acta 2015, 435, 142–146. [Google Scholar] [CrossRef]
- Matsinha, L.C.; Mao, J.; Mapolie, S.F.; Smith, G.S. Water-soluble palladium(II) sulfonated thiosemicarbazone complexes: Facile synthesis and preliminary catalytic studies in the Suzuki-Miyaura cross-coupling reaction in water. Eur. J. Inorg. Chem. 2015, 2015, 4088–4094. [Google Scholar] [CrossRef]
- Baruah, J.; Gogoi, R.; Gogoi, N.; Borah, G. A thiosemicarbazone–palladium(II)–imidazole complex as an efficient pre-catalyst for Suzuki–Miyaura cross-coupling reactions at room temperature in aqueous media. Transit. Met. Chem. 2017, 42, 683–692. [Google Scholar] [CrossRef]
- Dharani, S.; Kalaiarasi, G.; Sindhuja, D.; Lynch, V.M.; Shankar, R.; Karvembu, R.; Prabhakaran, R. Tetranuclear palladacycles of 3-acetyl-7-methoxy-2H-chromen-2-one derived Schiff bases: Efficient catalysts for Suzuki–Miyaura coupling in an aqueous medium. Inorg. Chem. 2019, 58, 8045–8055. [Google Scholar] [CrossRef] [PubMed]
- Bakir, M.; Lawrence, M.W.; Bohari Yamin, M. Novel κ2-Nim,S- and κ4-C,Nim,(μ-S),(μ-S)-coordination of di-2-thienyl ketone thiosemicarbazone (dtktsc). Hydrogen evolution and catalytic properties of palladacyclic [Pd(κ4-C,Nim,(μ-S),(μ-S)-dtktsc-2H)]4. Inorg. Chim. Acta 2020, 507, 119592. [Google Scholar] [CrossRef]
- Thapa, K.; Paul, P.; Bhattacharya, S. A group of diphosphine-thiosemicarbazone complexes of palladium: Efficient precursors for catalytic C–C and C–N coupling reactions. Inorg. Chim. Acta 2019, 486, 232–239. [Google Scholar] [CrossRef]
- Datta, S.; Seth, D.K.; Butcher, R.J.; Bhattacharya, S. Mixed-ligand thiosemicarbazone complexes of nickel: Synthesis, structure and catalytic activity. Inorg. Chim. Acta 2011, 377, 120–128. [Google Scholar] [CrossRef]
- Anitha, P.; Manikandan, R.; Vijayan, P.; Prakash, G.; Viswanathamurthi, P.; Butcher, R.J. Nickel(II) complexes containing ONS donor ligands: Synthesis, characterization, crystal structure and catalytic application towards C–C cross-coupling reactions. J. Chem. Sci. 2015, 127, 597–608. [Google Scholar] [CrossRef]
- Prabhu, R.N.; Ramesh, R. Synthesis and structural characterization of Pd(II) thiosemicarbazonato complex: Catalytic evaluation in synthesis of diaryl ketones from aryl aldehydes and arylboronic acids. Tetrahedron Lett. 2017, 58, 405–409. [Google Scholar] [CrossRef]
- Sonogashira, K.; Tohda, Y.; Hagihara, N. A convenient synthesis of acetylenes: Catalytic substitutions of acetylenic hydrogen with bromoalkenes, iodoarenes and bromopyridines. Tetrahedron Lett. 1975, 16, 4467–4470. [Google Scholar] [CrossRef]
- Prabhu, R.N.; Pal, S. Copper-free Sonogashira reactions catalyzed by a palladium(II) complex bearing pyrenealdehyde thiosemicarbazonate under ambient conditions. Tetrahedron Lett. 2015, 56, 5252–5256. [Google Scholar] [CrossRef]
- Prabhu, R.N.; Ramesh, R. Square-planar Ni(II) thiosemicarbazonato complex as an easily accessible and convenient catalyst for Sonogashira cross-coupling reaction. Tetrahedron Lett. 2016, 57, 4893–4897. [Google Scholar] [CrossRef]
- Lu, L.; Chellan, P.; Smith, G.S.; Zhang, X.; Yan, H.; Mao, J. Thiosemicarbazone salicylaldiminato palladium(II)-catalyzed alkynylation couplings between arylboronic acids and alkynes or alkynyl carboxylic acids. Tetrahedron 2014, 70, 5980–5985. [Google Scholar] [CrossRef]
- Rokade, B.V.; Barker, J.; Guiry, P.J. Development of and recent advances in asymmetric A3 coupling. Chem. Soc. Rev. 2019, 48, 4766–4790. [Google Scholar] [CrossRef]
- Manikandan, R.; Anitha, P.; Viswanathamurthi, P.; Malecki, J.G. Palladium(II) pyridoxal thiosemicarbazone complexes as efficient and recyclable catalyst for the synthesis of propargylamines by a three component coupling reactions in ionic liquids. Polyhedron 2016, 119, 300–306. [Google Scholar] [CrossRef]
- Heravi, M.M.; Hashemi, E.; Nazari, N. Negishi coupling: An easy progress for C–C bond construction in total synthesis. Mol. Divers. 2014, 18, 441–472. [Google Scholar] [CrossRef]
- Muthukumar, M.; Sivakumar, S.; Viswanathamurthi, P.; Karvembu, R.; Prabhakaran, R.; Natarajan, K. Studies on ruthenium(III) chalcone thiosemicarbazone complexes as catalysts for carbon–carbon coupling. J. Coord. Chem. 2010, 63, 296–306. [Google Scholar] [CrossRef]
- Priyarega, S.; Kalaivani, P.; Prabhakaran, R.; Hashimoto, T.; Endo, A.; Natarajan, K. Nickel(II) complexes containing thiosemicarbazone and triphenylphosphine: Synthesis, spectroscopy, crystallography and catalytic activity. J. Mol. Struct. 2011, 1002, 58–62. [Google Scholar] [CrossRef]
- Güveli, Ş.; Agopcan Çınar, S.; Karahan, Ö.; Aviyente, V.; Ülküseven, B. Nickel(II)-PPh3 complexes of S, N-substituted thiosemicarbazones-structure, DFT study, and catalytic efficiency. Eur. J. Inorg. Chem. 2016, 2016, 538–544. [Google Scholar] [CrossRef]
- Dorel, R.; Grugel, C.P.; Haydl, A.M. The Buchwald–Hartwig amination after 25 years. Angew. Chem. Int. Ed. 2019, 58, 17118–17129. [Google Scholar] [CrossRef]
- Heravi, M.M.; Kheilkordi, Z.; Zadsirjan, V.; Heydari, M.; Malmir, M. Buchwald-Hartwig reaction: An overview. J. Organomet. Chem. 2018, 861, 17–104. [Google Scholar] [CrossRef]
- Munir, I.; Zahoor, A.F.; Rasool, N.; Naqvi, S.A.R.; Zia, K.M.; Ahmad, R. Synthetic applications and methodology development of Chan–Lam coupling: A review. Mol. Divers. 2019, 23, 215–259. [Google Scholar] [CrossRef] [PubMed]
- West, M.J.; Fyfe, J.W.B.; Vantourout, J.C.; Watson, A.J.B. Mechanistic development and recent applications of the Chan–Lam amination. Chem. Rev. 2019, 119, 12491–12523. [Google Scholar] [CrossRef]
- Khan, F.; Dlugosch, M.; Liu, X.; Banwell, M.G. The palladium-catalyzed Ullmann cross-coupling reaction: A modern variant on a time-honored process. Acc. Chem. Res. 2018, 51, 1784–1795. [Google Scholar] [CrossRef]
- Jiang, J.; Du, L.; Ding, Y. Aryl-aryl bond formation by Ullmann Reaction: From mechanistic aspects to catalyst. Mini. Rev. Org. Chem. 2020, 17, 26–46. [Google Scholar] [CrossRef]
- Prabhu, R.N.; Ramesh, R. Synthesis and structural characterization of palladium(II) thiosemicarbazone complex: Application to the Buchwald–Hartwig amination reaction. Tetrahedron Lett. 2013, 54, 1120–1124. [Google Scholar] [CrossRef]
- Anitha, P.; Manikandan, R.; Viswanathamurthi, P. Palladium(II) 9,10-phenanthrenequinone N-substituted thiosemicarbazone/semicarbazone complexes as efficient catalysts for N-arylation of imidazole. J. Coord. Chem. 2015, 68, 3537–3550. [Google Scholar] [CrossRef]
- Shan, Y.; Wang, Y.; Jia, X.; Cai, W.; Xiang, J. New, simple, and effective thiosemicarbazide ligand for copper(II)-catalyzed N-arylation of imidazoles. Synth. Commun. 2012, 42, 1192–1199. [Google Scholar] [CrossRef]
- Gogoi, N.; Borah, G.; Gogoi, P.K. Cu(II) complex of phenylthiosemicarbazone: An in situ catalyst for formation of C–N bond between different N-based neucleophiles with arylboronic acids at room temperature. Heteroat. Chem. 2018, 29, e21414. [Google Scholar] [CrossRef]
- Ramachandran, R.; Prakash, G.; Vijayan, P.; Viswanathamurthi, P.; Grzegorz Malecki, J. Synthesis of heteroleptic copper(I) complexes with phosphine-functionalized thiosemicarbazones: An efficient catalyst for regioselective N-alkylation reactions. Inorg. Chim. Acta 2017, 464, 88–93. [Google Scholar] [CrossRef]
- Ramachandran, R.; Prakash, G.; Selvamurugan, S.; Viswanathamurthi, P.; Malecki, J.G.; Ramkumar, V. Efficient and versatile catalysis of N-alkylation of heterocyclic amines with alcohols and one-pot synthesis of 2-aryl substituted benzazoles with newly designed ruthenium(II) complexes of PNS thiosemicarbazones. Dalt. Trans. 2014, 43, 7889–7902. [Google Scholar] [CrossRef]
- Ramachandran, R.; Prakash, G.; Nirmala, M.; Viswanathamurthi, P.; Malecki, J.G. Ruthenium(II) carbonyl complexes designed with arsine and PNO/PNS ligands as catalysts for N-alkylation of amines via hydrogen autotransfer process. J. Organomet. Chem. 2015, 791, 130–140. [Google Scholar] [CrossRef]
- Ramachandran, R.; Prakash, G.; Selvamurugan, S.; Viswanathamurthi, P.; Malecki, J.G.; Linert, W.; Gusev, A. Ruthenium(II) complexes containing a phosphine-functionalized thiosemicarbazone ligand: Synthesis, structures and catalytic C–N bond formation reactions via N-alkylation. RSC Adv. 2015, 5, 11405–11422. [Google Scholar] [CrossRef]
- Ramachandran, R.; Prakash, G.; Viswanathamurthi, P.; Malecki, J.G. Ruthenium(II) complexes containing phosphino hydrazone/thiosemicarbazone ligand: An efficient catalyst for regioselective N-alkylation of amine via borrowing hydrogen methodology. Inorg. Chim. Acta 2018, 477, 122–129. [Google Scholar] [CrossRef]
- Suganthy, P.K.; Prabhu, R.N.; Sridevi, V.S. Palladium(II) thiosemicarbazone complex: Synthesis, structure and application to carbon–oxygen cross-coupling reaction. Inorg. Chem. Commun. 2014, 44, 67–69. [Google Scholar] [CrossRef]
- Hübner, S.; de Vries, J.G.; Farina, V. Why does industry not use immobilized transition metal complexes as catalysts? Adv. Synth. Catal. 2016, 358, 3–25. [Google Scholar] [CrossRef]
- Kovala-Demertzi, D.; Kourkoumelis, N.; Derlat, K.; Michalak, J.; Andreadaki, F.J.; Kostas, I.D. Thiosemicarbazone-derivatised palladium nanoparticles as efficient catalyst for the Suzuki-Miyaura cross-coupling of aryl bromides with phenylboronic acid. Inorg. Chim. Acta 2008, 361, 1562–1565. [Google Scholar] [CrossRef]
- Bakherad, M.; Keivanloo, A.; Bahramian, B.; Jajarmi, S. Synthesis of ynones via recyclable polystyrene-supported palladium(0) complex catalyzed acylation of terminal alkynes with acyl chlorides under copper- and solvent-free conditions. Synlett 2011, 2011, 311–314. [Google Scholar] [CrossRef]
- Sharma, R.K.; Pandey, A.; Gulati, S. Silica-supported palladium complex: An efficient, highly selective and reusable organic–inorganic hybrid catalyst for the synthesis of E-stilbenes. Appl. Catal. A Gen. 2012, 431–432, 33–41. [Google Scholar] [CrossRef]
- Veisi, H.; Metghalchi, Y.; Hekmati, M.; Samadzadeh, S. CuI heterogenized on thiosemicarbazide modified-multi walled carbon nanotubes (thiosemicarbazide-MWCNTs-CuI): Novel heterogeneous and reusable nanocatalyst in the C–N Ullmann coupling reactions. Appl. Organomet. Chem. 2017, 31, e3676. [Google Scholar] [CrossRef]
- Sadjadi, S. Palladium nanoparticles immobilized on cyclodextrin-decorated halloysite nanotubes: Efficient heterogeneous catalyst for promoting copper- and ligand-free Sonogashira reaction in water–ethanol mixture. Appl. Organomet. Chem. 2018, 32, e4211. [Google Scholar] [CrossRef]
- Sadjadi, S.; Hosseinnejad, T.; Malmir, M.; Heravi, M.M. Cu@furfural imine-decorated halloysite as an efficient heterogeneous catalyst for promoting ultrasonic-assisted A3 and KA2 coupling reactions: A combination of experimental and computational study. New J. Chem. 2017, 41, 13935–13951. [Google Scholar] [CrossRef]
- Sadjadi, S.; Heravi, M.M.; Ebrahimizadeh, M. Synthesis of Cu@Fur-SBA-15 as a novel efficient and heterogeneous catalyst for promoting A3-coupling under green and mild reaction conditions. J. Porous Mater. 2018, 25, 779–788. [Google Scholar] [CrossRef]
- Ghodsinia, S.S.E.; Akhlaghinia, B. CuI anchored onto mesoporous SBA-16 functionalized by aminated 3-glycidyloxypropyltrimethoxysilane with thiosemicarbazide (SBA-16/GPTMS-TSC-CuI): A heterogeneous mesostructured catalyst for S-arylation reaction under solvent-free conditions. Green Chem. 2019, 21, 3029–3049. [Google Scholar] [CrossRef]
- Ahmadi, A.; Sedaghat, T.; Azadi, R.; Motamedi, H. Magnetic mesoporous silica nanocomposite functionalized with palladium Schiff base complex: Synthesis, characterization, catalytic efficacy in the Suzuki–Miyaura reaction and α-amylase immobilization. Catal. Lett. 2020, 150, 112–126. [Google Scholar] [CrossRef]



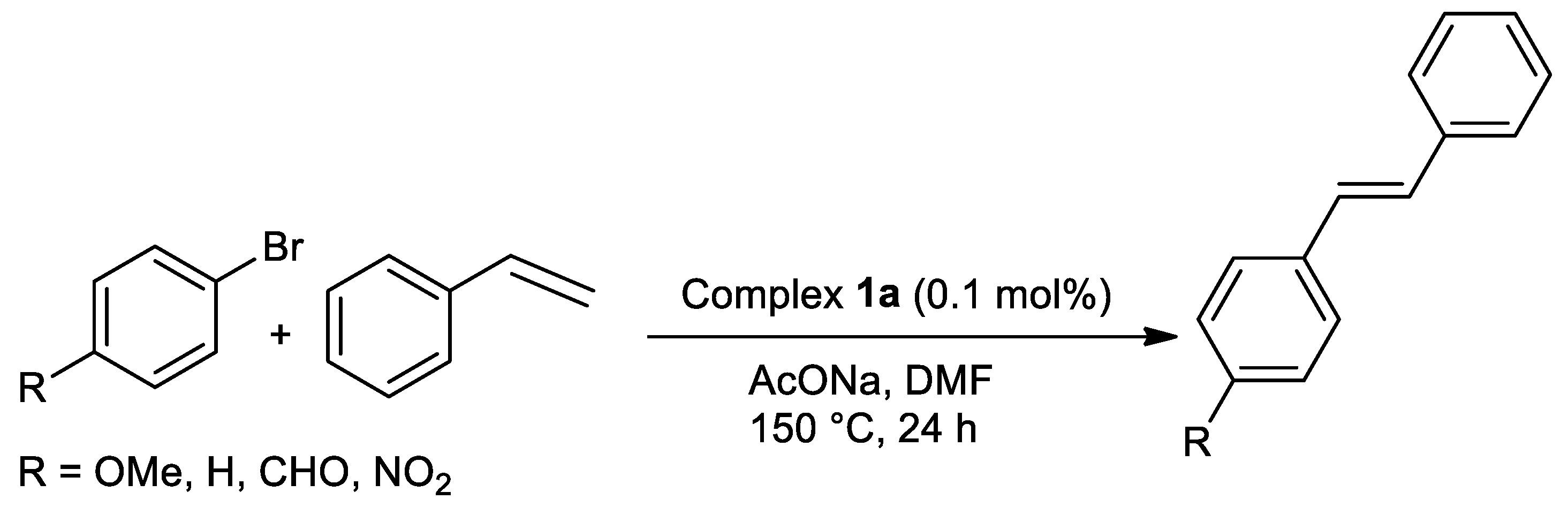

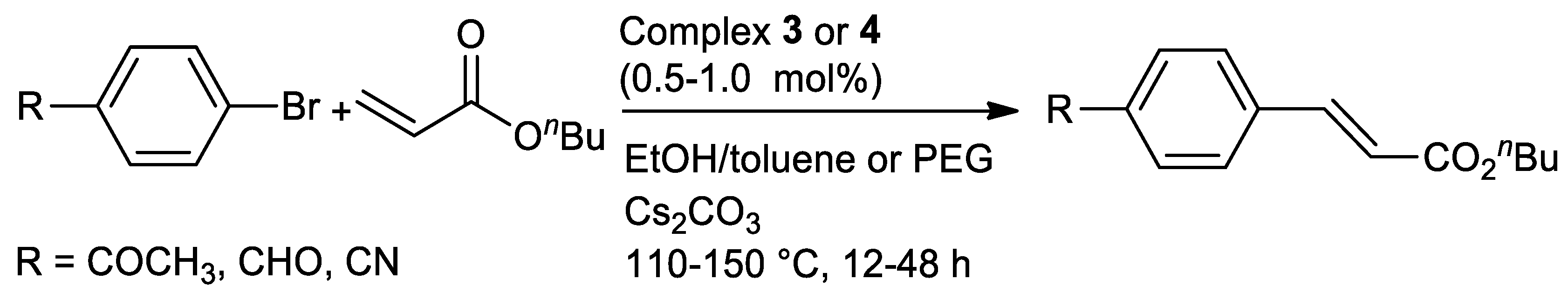
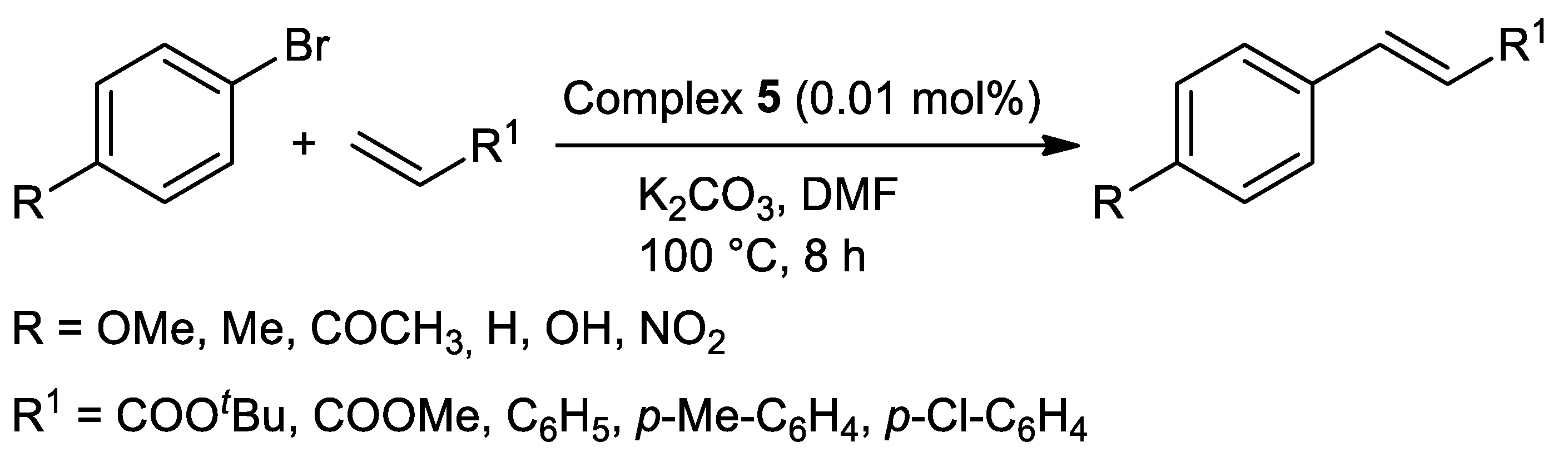


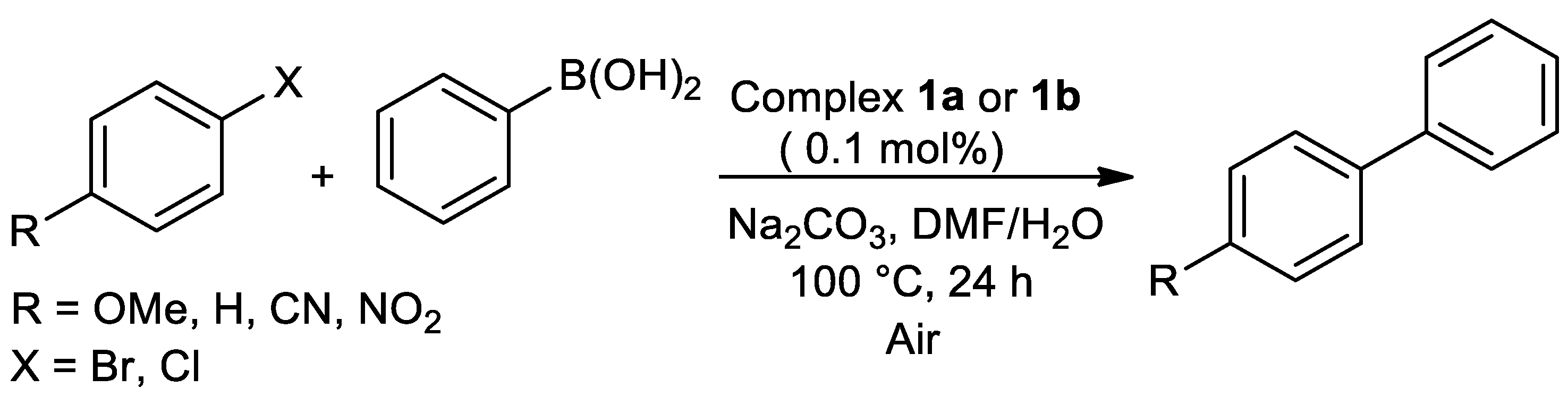

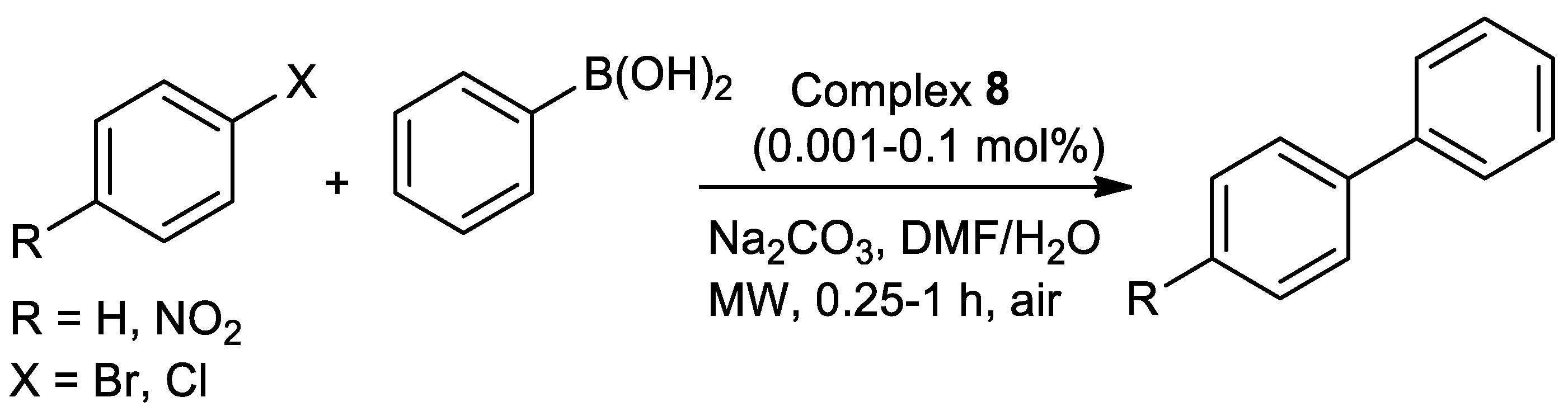


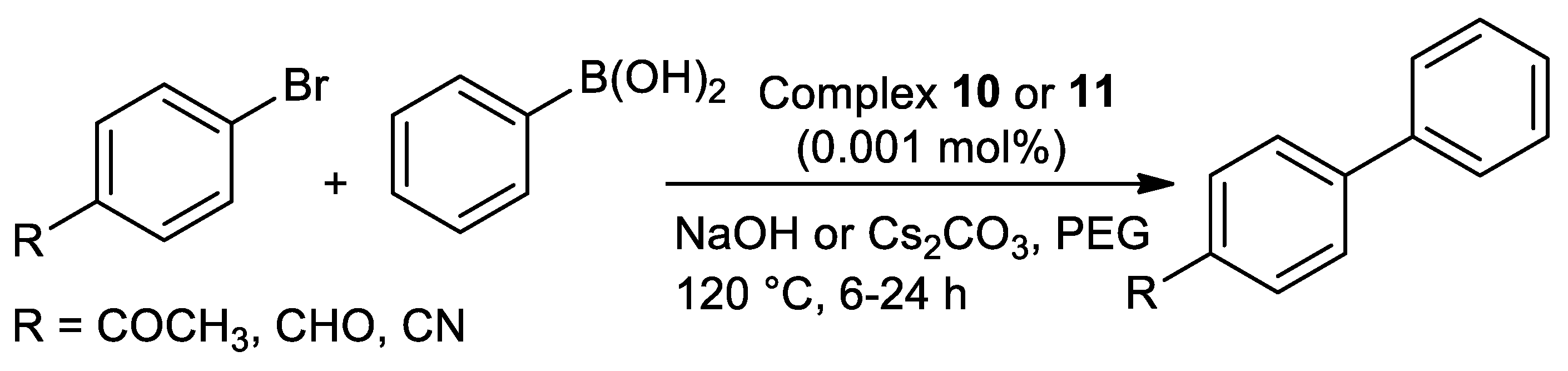
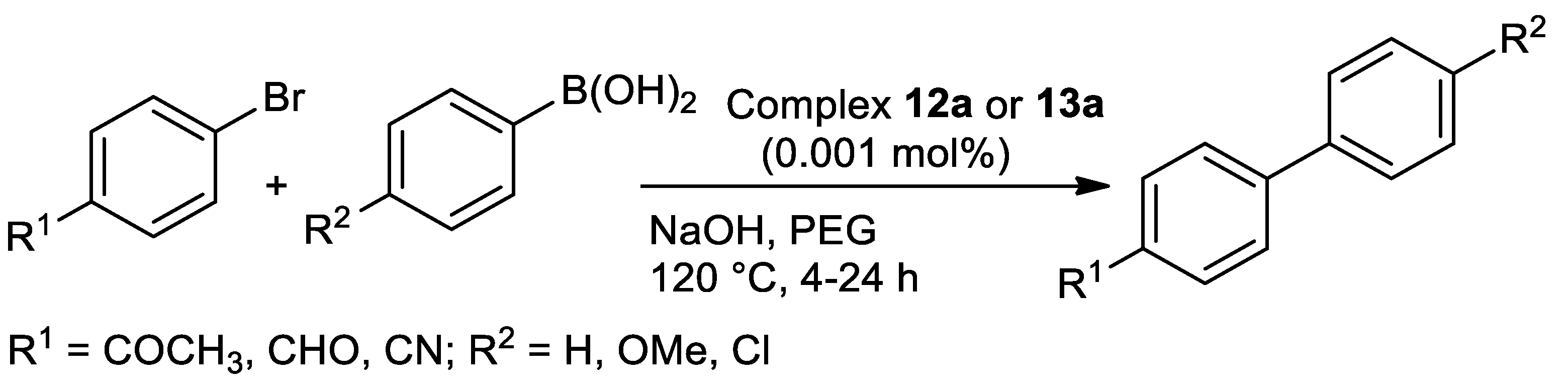




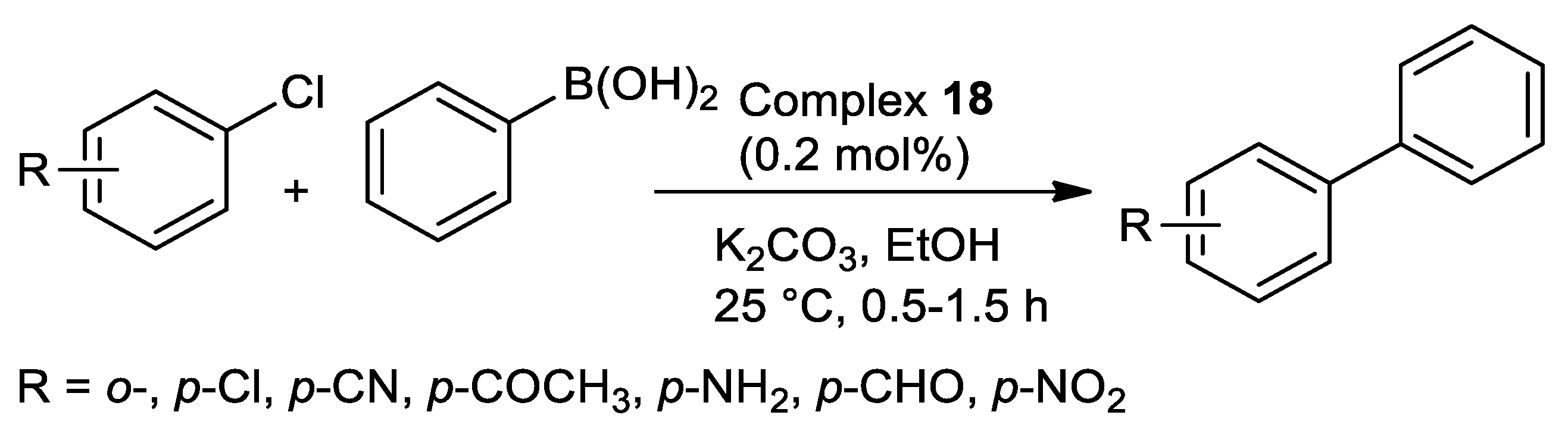

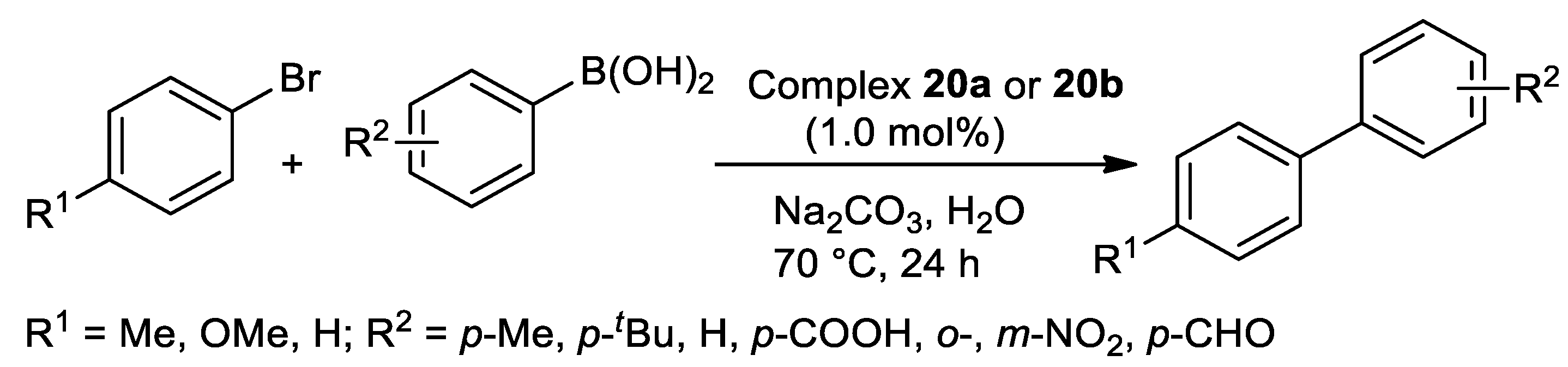

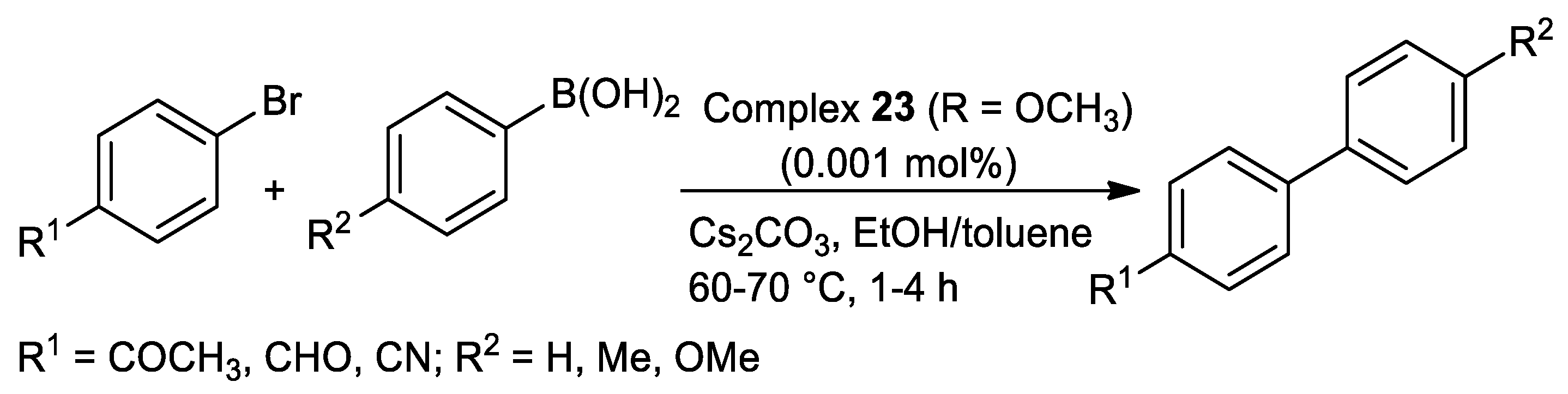




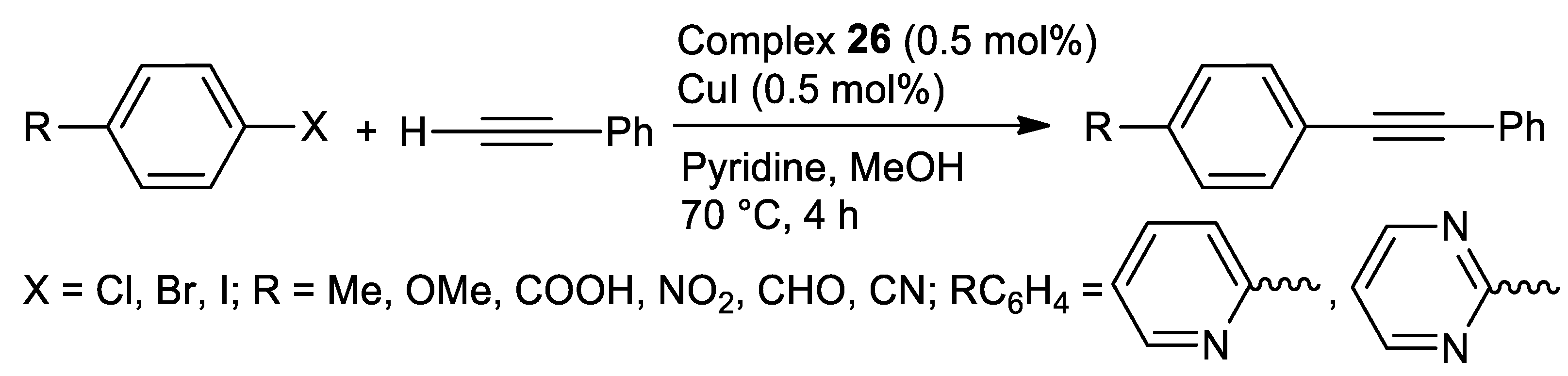


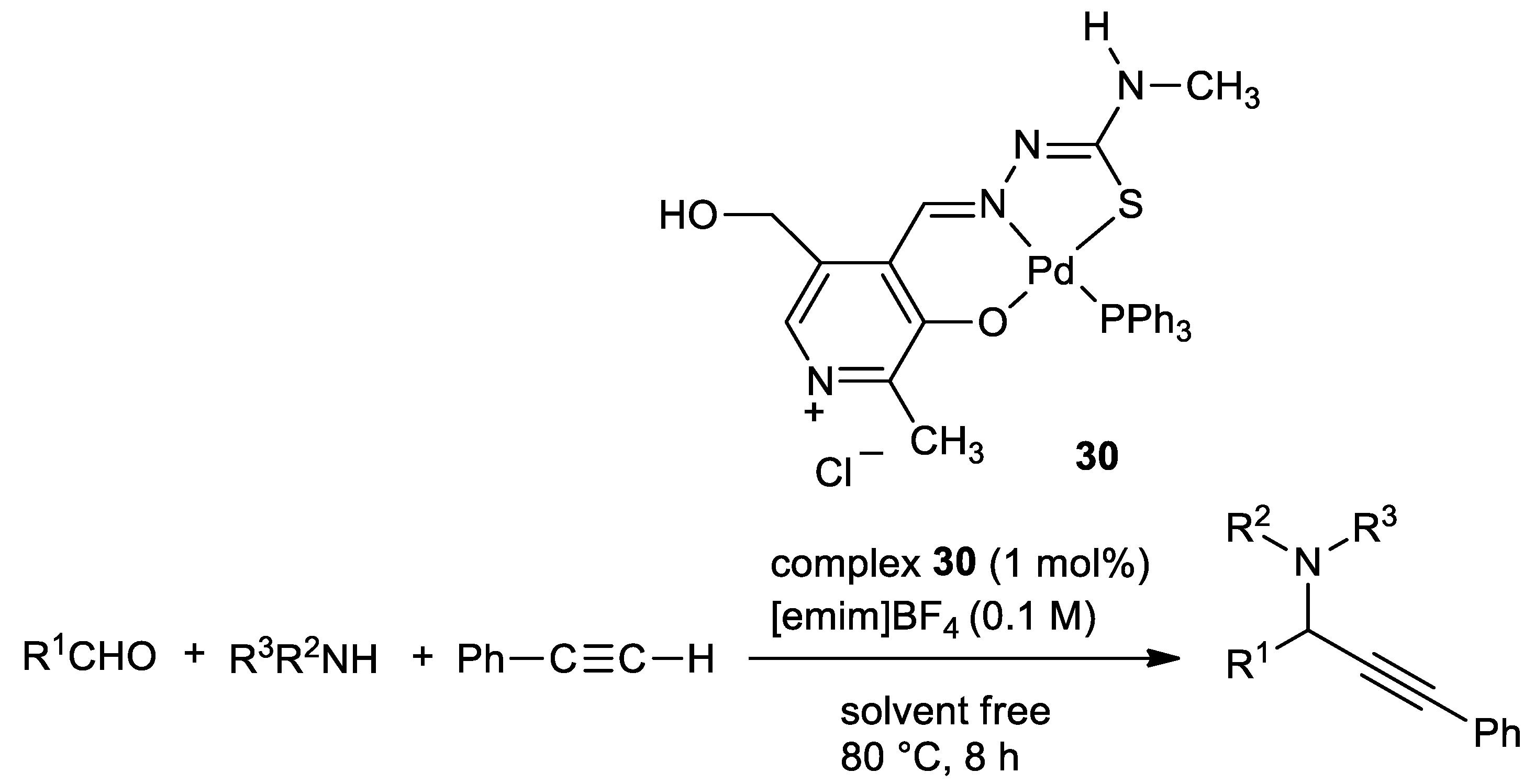

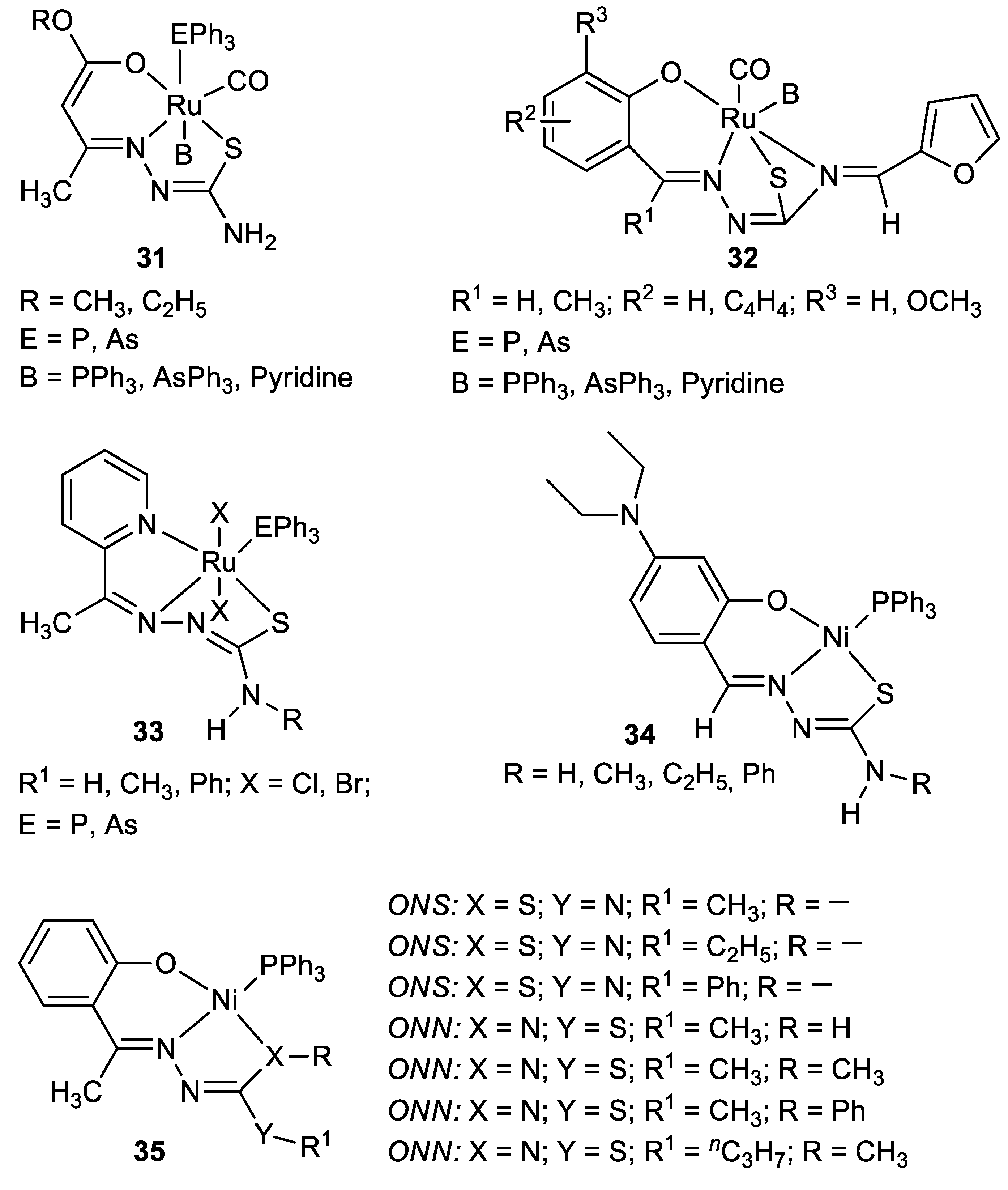


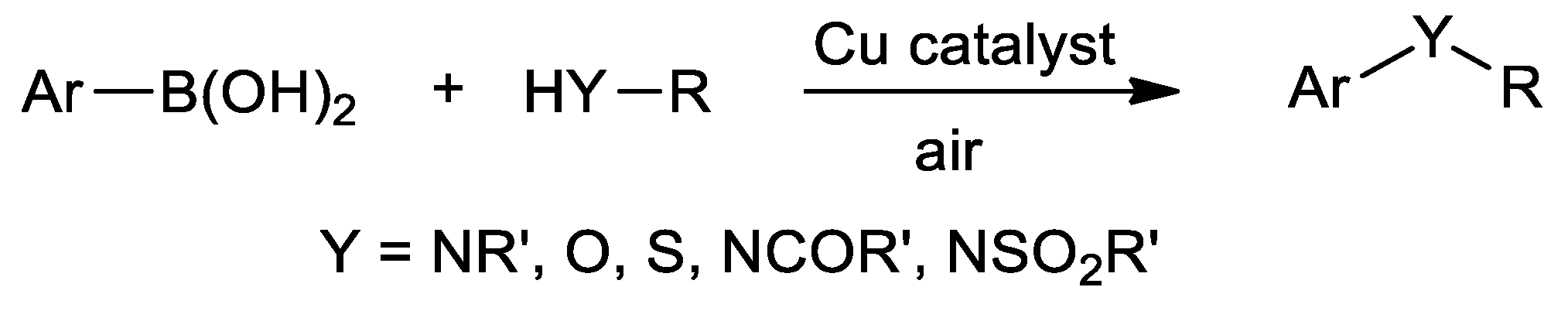

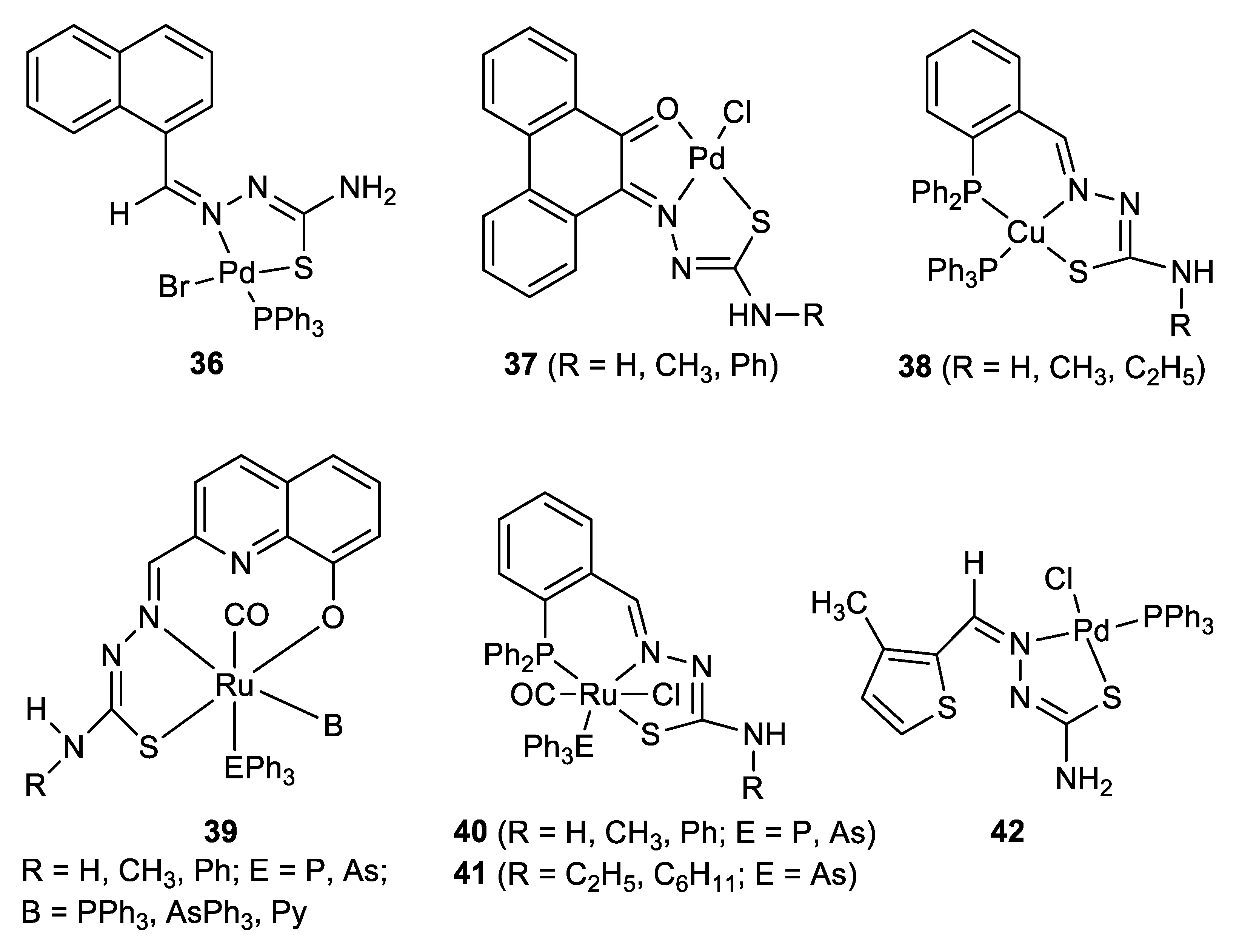










| Metal | T (°C) | Solvent | Time (h) | Ligand 2 | Base | Catalyst (mol%) | Yield (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Pd | 150 | DMF | 24 | O,N,S | NaOAc | 0.1 | 46–95 | [15] |
| Pd | 130–145 | DMF | 24–36 | O,N,S | Na2CO3 | 0.1–1.0 | 50–90 | [94] |
| Pd | 110–150 | EtOH/toluene or PEG | 12–48 | N,S | Cs2CO3 | 0.5–1.0 | 57–80 | [95] |
| Pd | 100 | DMF | 8 | N,S | K2CO3 | 0.01 | 60–97 | [96] |
| Ni | 130 | DMF | 24 | O,N,S | Cs2CO3 | 2.0 | 36–99 | [97] |
| Ni | 110 | DMF | 24 | N,S | K2CO3 | 0.5 | 60–94 | [98] |
| Pd | 120 | DMF | 5–24 | S,N,N,S | Et3N | 3.5 | 67–82 3 | [99] |
| Metal | T (°C) | Solvent | Time (h) | Ligand 2 | Base | Catalyst (mol%) | Yield (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Pd | 100 | DMF/H2O | 24 | O,N,S | Na2CO3 | 0.1 | 40–88 | [16] |
| Pd | 100–157 | DMF/H2O | 0.25–1 | O,N,S | Na2CO3 | 0.001–0.1 | 25–85 | [100] 3 |
| Pd | 25–95 | EtOH/toluene | 9–20 | N,S | NaOH | 0.001 | >99 | [102] |
| Pd | 140 | DMF | 24 | O,N,S | K2CO3 | 2.0 | 46–78 | [103] |
| Pd | 120 | PEG | 6–24 | N,S | NaOH or Cs2CO3 | 0.001 | 100 | [106] |
| Pd | 120 | PEG | 6–24 | N,S | NaOH | 0.001 | 65–100 | [107] |
| Pd | 120 | PEG | 4–24 | C,N,S | NaOH | 0.001 | 71–100 | [107] |
| Pd | 25–95 | EtOH/toluene | 9–20 | N,S | NaOH | 0.001 | 98–100 | [108] |
| Pd | 25–95 | EtOH/toluene | 3–14 | C,N,S | NaOH | 0.001 | 100 | [108] |
| Pd | 130 | DMF | 24–48 | C,N,S | K3PO4 | 0.5 | 31–99 | [109] |
| Pd | reflux | DMF | 3 | O,N,S | K2CO3 | 0.001 | 78–99 | [110] |
| Pd | 25 | EtOH | 0.5–1.5 | N,S | K2CO3 | 0.2 | 76–98 | [111] 4 |
| Pd | 100 | DMF | 24 | N,S | K2CO3 | 0.05 | 60–99 | [112] |
| Pd | 70 | H2O | 24 | O,N,S | Na2CO3 | 1.0 | 25–98 | [113] |
| Pd | 28 | H2O | 2–12 | N,S | K2CO3 | 1.18 | 65–90 | [114] |
| Pd | 60–70 | EtOH/H2O | 1–4 | C,N,S | K2CO3 | 0.125 | 51–99 | [115] |
| Pd | 95 | EtOH/toluene | 6–8 | N,S | Cs2CO3 | 0.001 | 79–100 | [117] |
| Ni | 140 | DMF | 24 | O,N,S | Cs2CO3 | 2.0 | 40–99 | [118] |
| Ni | 90 | DMA | 7 | O,N,S | K2CO3 | 1.0 | 28–64 | [119] |
| Pd | 110 | toluene | 12 | N,S | Cs2CO3 | 5.0 | 62–97 | [120] 5 |
| Metal | T (°C) | Solvent | Time (h) | Ligand 2 | Base | Catalyst (mol%) | Yield (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Pd | 75–110 | EtOH/toluene or PEG | 10–15 | N,S | NaOH | 0.5 | 68–99 | [95] |
| Pd | 80 | Et3N | 8 | N,S | Et3N | 0.5 | 65 | [111] |
| Pd | rt | DMF | 12 | N,S | Et3N | 0.5 | 67–99 | [122] |
| Ni | 70 | MeOH | 4 | O,N,S | pyridine | 0.5 | 55–85 | [119] |
| Ni | 80 | DMF | 8 | N,S | Et3N | 0.1 | 79–99 | [123] |
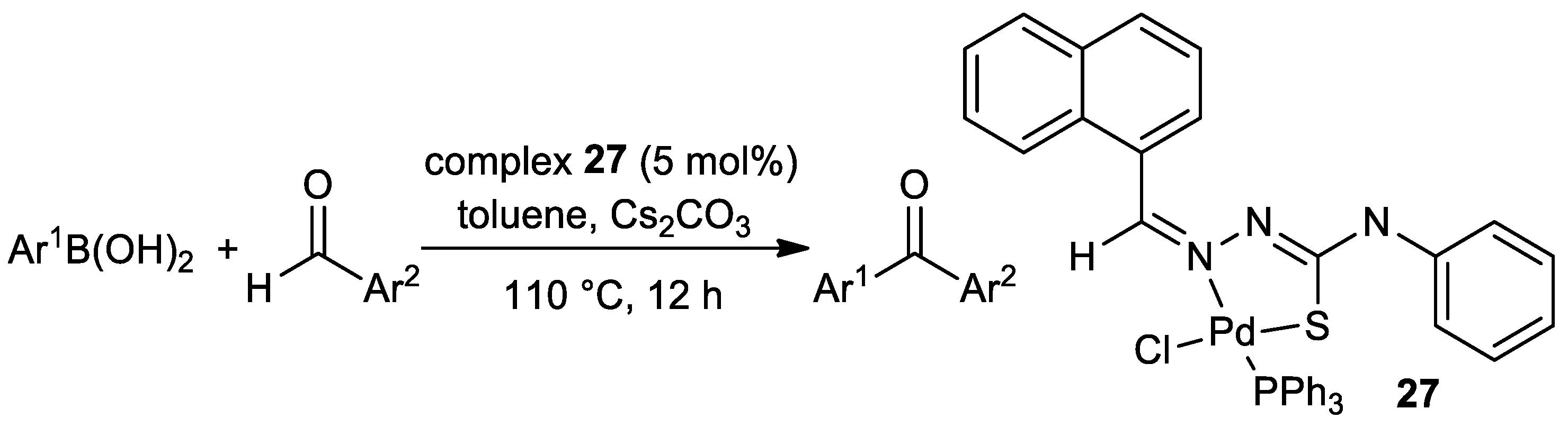
| Pd | 35 | CH2Cl2 | 24 | O,N,S | KOAc | 2.0 | 30–99 | [124] 3 |
| Metal | Substrates | T (°C) | Solvent | Time (h) | Ligand 1 | Base | Catalyst (mol%) | Yield (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Pd | ArBr + 2ary amine | 145 | PEG | 24 | N,S | NaOBut | 1.0 | 50–62 | [106] |
| Pd | ArBr + 1ary/2ary amine | 145 | PEG | 24 | N,S | NaOBut | 0.1 | 100 | [107] |
| Pd | ArBr + 1ary/2ary amine | 145 | PEG | 18 | C,N,S | NaOBut | 0.1 | 100 | [107] |
| Pd | ArBr + aniline | 105 | toluene | 12–18 | C,N,S | NaOBut | 0.01 | 100 | [108] |
| Pd | (het)ArBr + 2ary amine | 100 | 2-BuOH | 24 | N,S | K2CO3 | 0.2 | 77–99 | [137] |
| Pd | (het)ArBr + N-heterocycle | 110 | DMSO | 10 | O,N,S | KOH | 0.75 | 75–90 | [138] |
| Cu | ArBr + N-heterocycle | 110 | DMF | 24 | O,N,S | K2CO3 | 10.0 | 42–56 | [139] |
| Cu | (het)ArB(OH)2 + aniline | r.t. | DMF/H2O | 14–18 | N,S | Et3N | 10.0 | 74–95 | [140] |
| Cu | (het)ArB(OH)2 + N-heterocycle | r.t. | DMF/H2O | 18–24 | N,S | Et3N | 10.0 | 70–94 | [140] |
| Cu | RCH2OH + 1ary amine | 100 | toluene | 12 | P,N,S | KOH | 0.1–0.2 | 89–99 | [141] |
| Ru | RCH2OH + aniline | 100 | none | 6 | O,N,S | KOBut | 1.0 | 61–86 | [74] |
| Ru | RCH2OH + 1ary amine | 100 | toluene | 12–24 | P,N,S | KOH | 0.5–1.0 | 45–99 | [142] |
| Ru | RCH2OH + 1ary amine | 100 | toluene | 12 | P,N,S | KOH | 0.5 | 79–98 | [143] |
| Ru | RCH2OH + 1ary amine | 100 | toluene | 12 | P,N,S | KOH | 0.5 | 59–98 | [144] |
| Ru | RCH2OH + sulfonamide | 120 | toluene | 12 | P,N,S | KOH | 0.5 | 21–99 | [144] |
| Pd | ArBr/ArI + p-cresol | 80 | DMF | 12 | N,S | K2CO3 | 1.0 | 62–94 | [146] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kostas, I.D.; Steele, B.R. Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions. Catalysts 2020, 10, 1107. https://doi.org/10.3390/catal10101107
Kostas ID, Steele BR. Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions. Catalysts. 2020; 10(10):1107. https://doi.org/10.3390/catal10101107
Chicago/Turabian StyleKostas, Ioannis D., and Barry R. Steele. 2020. "Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions" Catalysts 10, no. 10: 1107. https://doi.org/10.3390/catal10101107
APA StyleKostas, I. D., & Steele, B. R. (2020). Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions. Catalysts, 10(10), 1107. https://doi.org/10.3390/catal10101107







