1. Introduction
An epidemic of infectious disease occurs when a virus population undergoes genetic mutations or new species of viruses are introduced into the host population, and host immunity to that change in the virus population is suddenly reduced below a certain threshold. Hence, epidemic modeling should take into account not only the population dynamics of the host population, but also those of the viruses. In traditional epidemiological models, differential equations are used to capture the dynamic evolution of different classes of host populations. In particular, susceptible (S) is the class of people who are not infected; infected (I) is the class of people who have the disease; and removed or recovered (R) represents the quarantined or immune population. The commonly used SIR model [
1,
2] is used to describe the population migrations between these three classes of models. In order to capture the interdependencies between a virus and host populations, we established a system framework that combines the SIR model with evolutionary models that describe virus mutations.
In this work, we studied an influenza epidemic in urban populations. We analyzed the evolutionary model for virus mutations together with SIR models for the evolution of susceptible, infected, and recovered subpopulations. Over the time, individuals from these subpopulations randomly interact with each other and change their state. We consider the epidemic process as a dynamic process of changing states from susceptible individuals to the infected and finally to the recovered. The influenza epidemic is a fast-spreading process, involving a large part of the total population. Hence, one key concern is protecting a population during the annual epidemic season. There exist methods of prevention that reduce the sickness rate to protect populations, and medical measures (pharmacological products, quarantine policies, etc.) that reduce the number of the infected in the population.
Another aspect of the influenza epidemic is that different strains of influenza viruses can spread in the population during each epidemic season. As it was shown in previous studies, influenza viruses have many modifications and can quickly transform every season. Thus, by including a simulation of the mutation process into the SIR model, we can preliminarily forecast treatment strategies. In this work, we used evolutionary-game tools to describe the evolution of the influenza virus because this approach allows us to estimate the survivability of virus modifications and to determine the strongest modification. Thereby, we can use treatment strategies more effectively.
More specifically, in our work, we assume that the virus has two types with different strains and fitness functions. Both types of viruses spread in urban populations, and, hence, during an epidemic, different parts of population are infected. In our model, we split infected subpopulations into two subgroups and considered a modified SIR model. Therefore, the epidemic process in urban population depends on changes in the virus population.
In our work, we formulated the SIR model under the mechanism of a virus mutation that affects a human population and considered the minimization of treatment costs and the number of infected in both subpopulations to reduce the speed of epidemics. This complex problem is formulated as an optimal-control problem, and the virus mutation is described by replicator dynamics.
The paper is organized as follows. In
Section 2, we discuss related work to our model.
Section 3 presents the evolutionary model of viruses. In
Section 4, we establish the epidemic model for urban populations. In
Section 5, we use Pontryagin’s maximum principle to find the optimal control and present structural results of the optimal-control problem. In
Section 6, we used a numerical simulation to illustrate our results. The paper is concluded in
Section 7.
2. Related Works
The recent literature has seen a surge of interest in using optimal control and game-theoretic methods to study disease control of influenza for public health [
3,
4,
5]. This research problem can be traced back to Reference [
6], where an SIR type of mathematical framework was proposed to study epidemic spread in a homogeneous population. It provides a deterministic dynamical system model as the mean field approximation of the underlying stochastic evolution of the host subpopulations. In Reference [
7], a control problem was formulated for a model of the carrier-borne epidemic model, and it was shown that the optimal-control effort switches at maximum once between the maximum and the minimum control effort. In Reference [
8], many variants of optimal-control models of SIR epidemics were investigated for the application of medical vaccination and health-promotion campaigns. In References [
9,
10], a dynamic SIR epidemic model was used to identify the optimal vaccination-policy mixes for flu season.
Epidemic models have also been used in computer science and engineering to describe the temporal evolution of worm propagation in computer networks [
11,
12,
13]. Methods such as stochastic system analysis and optimal control have been applied to provide insights on the spread of epidemics, as well as disease-control policies for protecting a population with quarantine and removal. In Reference [
14], sequential hypothesis testing was adopted to detect worm-epidemic propagation over the Internet under a stochastic density-dependent Markov jump process propagation model. In References [
15,
16], optimal-control methods were used to study the class of epidemic models in mobile wireless networks, and Pontryagin’s maximum principle was used to quantify the damage that malware could inflict on the network by deploying optimum decision rules.
Game-theoretic approaches were also used to analyze the strategic interactions between malicious worms and the system under attack. In Reference [
17], a virus-protection game was proposed based on two-state epidemic models for
N nodes and the characterization of the equilibrium focus on the steady state of the response. In Reference [
18], static- and dynamic-game frameworks were used to design equilibrium-revocation strategies for defending sensor networks from node-capturing and cloning attacks. It was shown that the
non-zero-sum differential game framework was equivalent to a zero-sum differential game between a team of
N attackers and the system. In References [
19,
20] the SIR model was combined with a game-theoretical approach to define the optimal medical approach: vaccination or treatment of seasonal influenza.
In many modern studies, different modifications of SIR models were used to estimate the behavior of infectious diseases, such as Ebola and Severe acute respiratory syndrome (SARS) [
21].
Different from past works, this paper considers a coupled system framework composed of the SIR epidemic model and the evolutionary dynamic model for virus mutations. This framework is motivated by the fact that the epidemic spread of a virus can facilitate virus mutations, strengthening its virulence, which, in turn, expedites the spread and worsens epidemics. The SIR epidemic model with virus mutations can capture this complex interactions between virus and host, and allows us to explain more complex phenomena through analysis.
3. Evolutionary Model of Virus Mutations
Infectious diseases, such as influenza and SARS, are urgent public health problems in modern urban environments. Influenza spreads faster, especially in large urban populations, and affects people’s lifestyle and working facilities. The occurrence of epidemics depends on many factors, such as the size of the human population, and the virus strain and virulence, and it has become important to use effective tools to reduce their influence on human populations [
22,
23,
24]. A mathematical model of virus infections in a population can be used to study the factors that influence epidemic growth to improve existing treatments and evaluate new effective prevention measures and treatment. In earlier research in the literature, it was shown that, during epidemic season, the influenza virus can mutate, and several types of the influenza virus circulate in the human population. Different mutations of the influenza virus affect human beings with different intensities, and epidemics evolve depending on virus type and intensity. Hence, the evolution of virus mutations should be taken into account when the SIR model is used to model influenza epidemics.
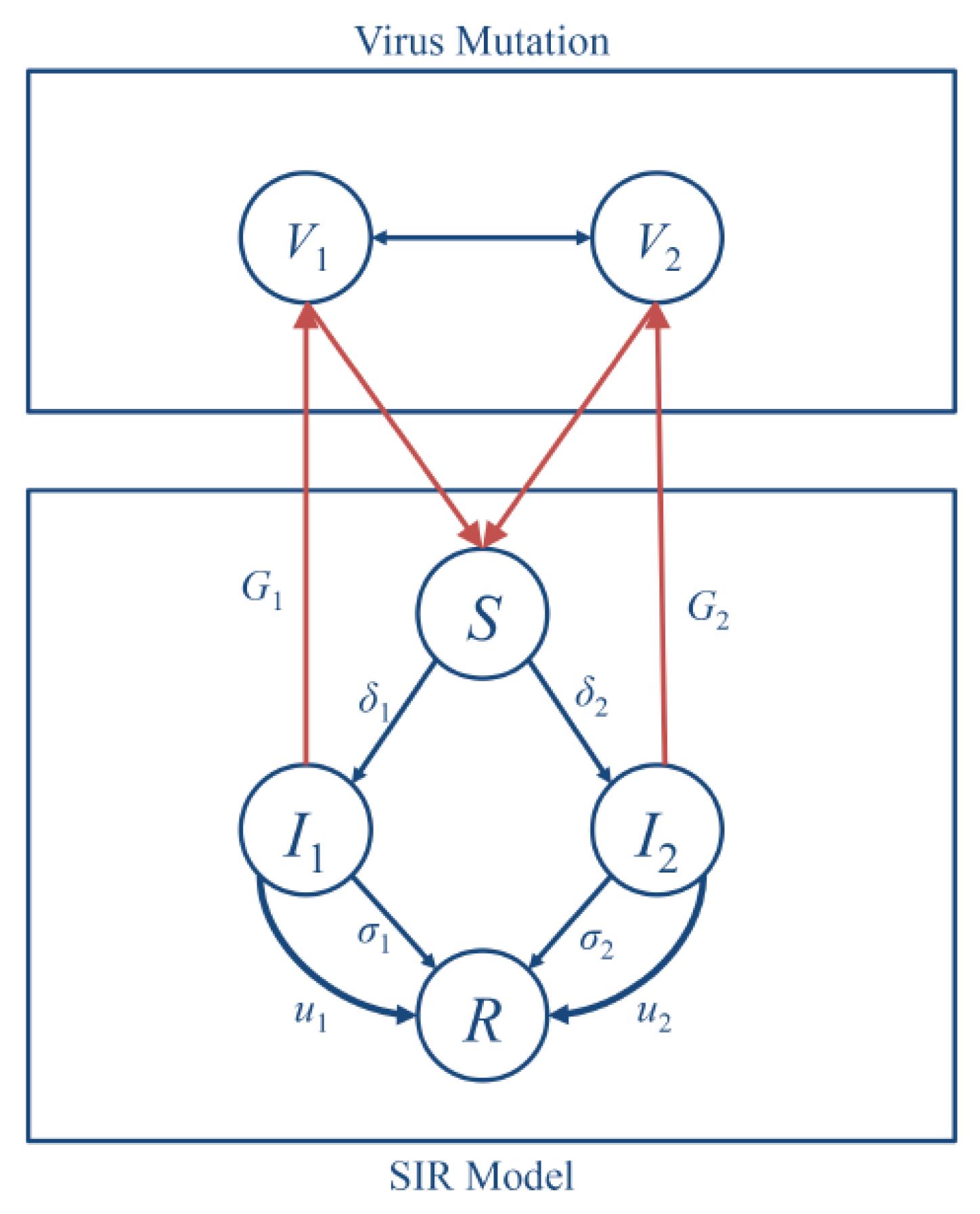
In this work, we coupled together two dynamic processes, i.e., the evolution of virus mutations and the epidemic process in human populations, as one dynamical system. The corresponding scheme of the system is illustrated in
Figure 1. At the top level, two different types of the influenza virus compete to infect the host to continue their life cycles, and thereby lead to the spread of epidemics in the human population. The total population would contain several infected subpopulations that correspond to different virus types. On the bottom level, the human population is divided into subpopulations: the susceptible (S), the infected (I), and the recovered (R).
Spreading of viruses can be controlled using prevention measures such as medical treatment or isolation of infected individuals of population. Thus, at this level, we considered the SIR model with those control parameters.
At the top level of coupled dynamical system, we used evolutionary dynamics to describe the mutations within the virus population. We first describe the interactions between two virus types using an evolutionary-game model, for which we defined pure strategies, fitness, and rule of changes in a population. In the game, two types of viruses compete for human organisms, and, depending on the strength of the virus, one type can survive or vanish from the virus population.
We assume that the virus has two types or strains, denoted by and , and without loss of generality, we assume that dominates virus . The fitness of virus type in the population is , which depends on the survivability of the virus among its infected population (e.g., human beings). The life cycle of viruses requires a host organism, and occupation of such an organism leads to energy costs. Hence, virus payoff is composed of two components: one is the utility of occupying the host organism, and the other is the cost, i.e., energy costs, , , , , Utility of occupation , as well as energy costs , are the functions of population state and, hence, mixed strategies over set are also dependent on the population states. Here, a mixed strategy is defined as the fraction of corresponding virus types circulating in a population. Thereby, the state of the virus population is defined as value , where .
Depending on virus strength, the number of people infected by different virus types is different. We used two flows of epidemic processes in a human population to describe our model and the population. We used to denote the population state for the subpopulation infected by the Type 1 virus, and is the state for the subpopulation infected by the Type 2 virus. Both viruses spread over the entire human population, and the interactions between two viruses when attacking the same human organism are described with the following four scenarios:
if virus meets virus , then utility for both is equal to . The virus incurs energy costs with probability if it cannot occupy a host organism, and achieves utility of with probability if it succeeds in occupation.
if virus competes with virus , then virus obtains utility of 0, and obtains a payoff of .
if virus meets , then both viruses obtain a payoff of .
The above four competition cases between the two types of viruses are summarized in the following matrix representation:
| | | |
| | |
| | . |
According to the theory of evolutionary games [
25], we compared the payoff of the
i-th pure strategy with the average payoff of the total population. If the difference was positive, then the number of individuals using this pure strategy increases, or otherwise decreases. The average payoff of population
is defined as
where
is a vector with
i-th element being one and 0 otherwise, indicating the
i-th pure strategy;
u is a continuous function. The payoff of the
i-th pure strategy is defined by
where
A is the payoff matrix of the current symmetric game, and
is a fraction of virus
[
26].
To describe the evolution of the virus, we need to use a system of differential equations. In the current work, we focused on replicator dynamics [
27] to describe changes of states in virus populations.
where
is the time-scaling factor. Since the mutation process in a virus population and the epidemic process in a human population may develop with different speeds (e.g., virus can mutate faster than spread in human populations),
can be used to describe such difference in the time scale of the two dynamics.
The stationary state of System of differential Equation (
1) leads to a symmetric Nash equilibrium [
26,
27]. Therefore, depending on parameters
and
, the game has two asymmetric Nash equilibria
,
corresponding to the strategies in which all populations are types
and
, respectively; and one symmetric Nash equilibrium
, where
,
, here
,
. The symmetric case is more interesting since both virus types have influence on the human population. By using the evolutionary scenario of virus behaviors, we can predict which type of virus will prevail in a human population. Hence, this knowledge can correct treatment therapy and, at the same time, decrease the total cost of epidemics.
4. Epidemic Process for an Urban Population
Consider a total urban population of size N with two types of viruses circulating in the population during epidemic season. The human population is divided into four groups: the susceptible, the infected by virus , the infected by virus , and the recovered. The susceptible are a subpopulation of human beings that are not infected by viruses but could be infected by one or both types of viruses, and they do not have immunity to them. We assume that, in human populations, two types of viruses coexist at the same time. Human organisms can be occupied by both types of viruses, and, hence, this leads to competition between viruses for the host. Depending on virus strength, we observed that the number of people infected by virus i or by virus j can be different, and people infected by virus or belong to the infected subpopulation. The recovered subpopulation consists of people recovered from being infected. The mixing of urban populations allows viruses to spread quickly, and each person in the population is assumed to be in contact with others with equal probability. Hence, when an infected individual interacts with a susceptible one, the virus spreading is made possible. A virus with higher virulence, by our assumption, has a higher probability of success in spreading when interaction occurs between an infected individual and a susceptible individual.
4.1. Epidemic Dynamics
We modeled a virus spreading in an urban population using the epidemiological SIR model, where a system of differential equations is used to describe the fraction of the urban population as a function of time. Then, at time t, , , , correspond to fractions of the population who are susceptible, infected by virus , infected by virus , and recovered, respectively; for all t, condition is justified. Define , , , as portions of the susceptible, the infected and the recovered in the population. At the beginning of epidemic process , most people in the population belong to the Susceptible subpopulation, a small group in the total population is infected, and the other people are in the recovered subpopulation. Hence, the initial states are: , , ,
We extended the simple SIR model introduced by References [
6,
28] to describe the situation with two virus types:
where
are the infection rates for virus
,
, We can interpret self-recovery rate
for virus
or
for virus
, which shows the probability that infected nodes from subgroups
or
are recovered from the infection without causing any costs to our system. Infection rate is defined as the product of infection transmissibility, i.e., the probability of infection being transmitted during contact:
where
Here
determine the virulence of the particular virus or the ability to infect a susceptible host.
In this work, changes in the virus population influence the parameters of the SIR model; therefore, the number of infected is a function of corresponding virus subpopulation
,
. We let
be linear and
take the following form:
Then, the SIR model can be rewritten as follows:
In the model above, the infection rate is captured in the evolution of the mutation process in the urban population. Medical treatment or quarantine isolation reduces the number of infected individuals in the urban population. These prevention measures can be interpreted as control parameters in the system defined as
; here,
are fractions of the infected that are quarantined or under intensive medical treatment:
,
.
4.2. Objective Function
In this work, we minimize overall cost in time interval . At any given t, the following costs exist in the system: , infected costs; , benefit rate; , costs for medical treatments (i.e., quarantine or removal) that help reduce the epidemic spread; , , , costs and benefit for invective and recovered at the end of the epidemic. Here, functions are non-decreasing and twice-differentiable, convex functions, i.e., , for , is non-decreasing and differentiable function and , is twice-differentiable and increasing function in such as , when . We show the structure of the optimal control strategies for the general case of costs functions. Hence, in particular cases this conditions will be satisfied for any functions with the same properties.
The cost for the aggregated system is given by
and the optimal-control problem is to minimize the cost, i.e.,
To simplify the analysis, we consider the case where
.
6. Numerical Simulation
In this section, we present numerical simulations to corroborate our results. Consider a city with population
100,000 people, where two viruses, of different strengths, spread (
and
). At time moment
, half of the population are susceptible to the infection, i.e.,
. The initial Recovered subpopulation is
. We set
of population as infected by virus
, and
of the population are infected by virus
, i.e.,
and
. The epidemic lasted for 45 days. We assumed that, in this experiment, the self-recovery rates were equal to
and
. During the epidemic, people from the infected population incur infected costs and, hence, we defined cost functions as
,
; the benefit rate from the recovered subpopulation was
. In
Section 5, we have shown that medical-treatment cost functions to describe the value of treatment can be chosen as concave or strictly convex. In our simulations, or concave cost functions, we used
, and
; for convex cost functions, we used
and
. The costs here were measured in the same monetary units (m.u.), which could be US dollars, Chinese RMB, or Euros, depending on the context.
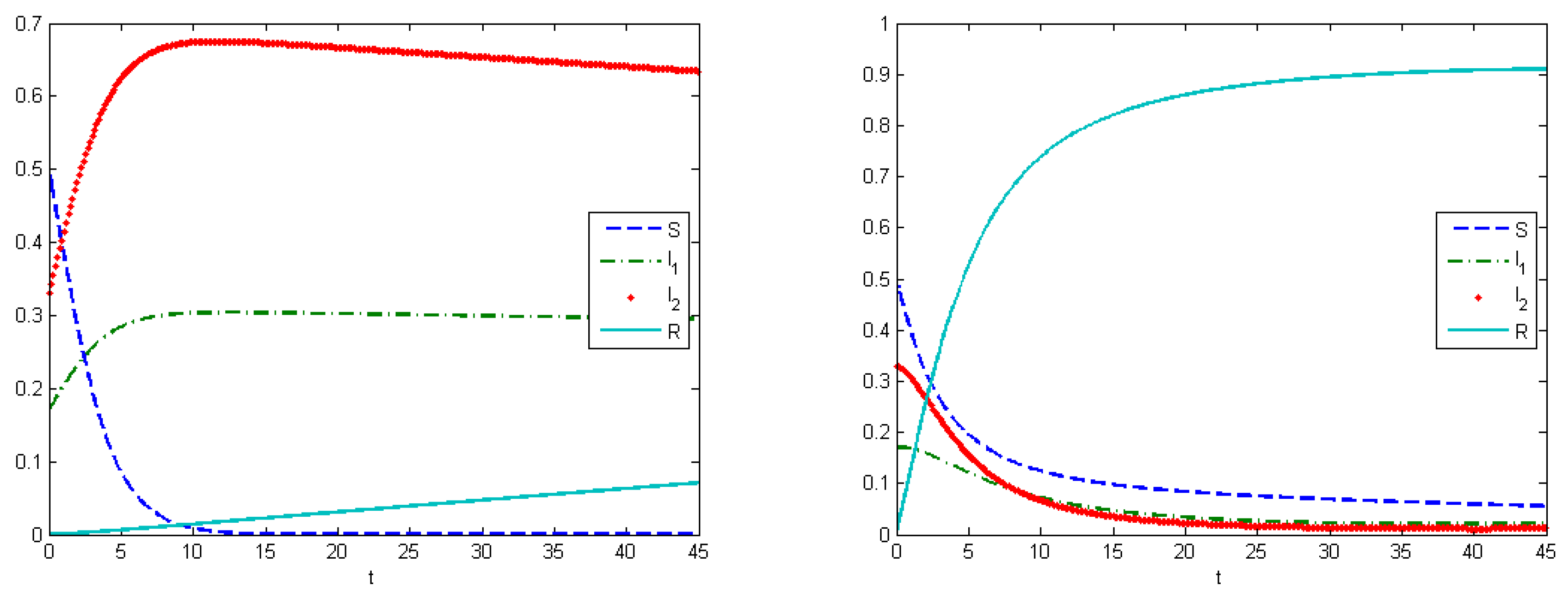
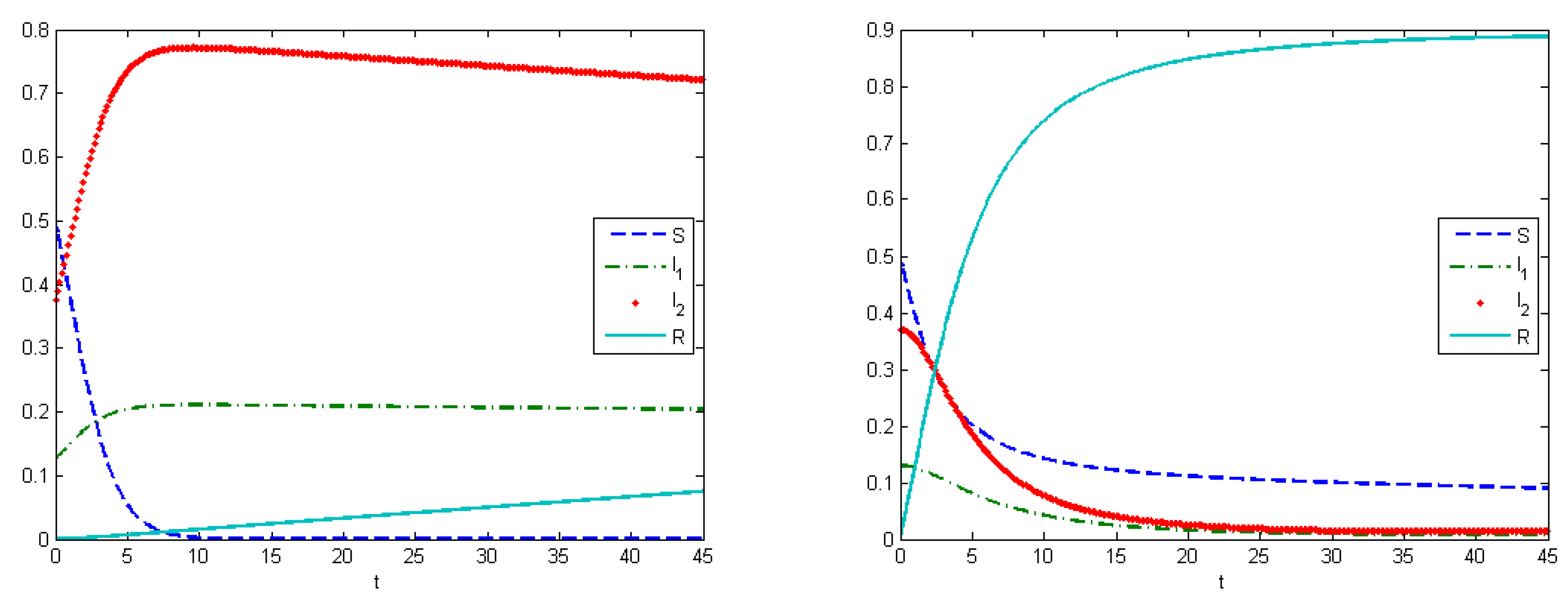
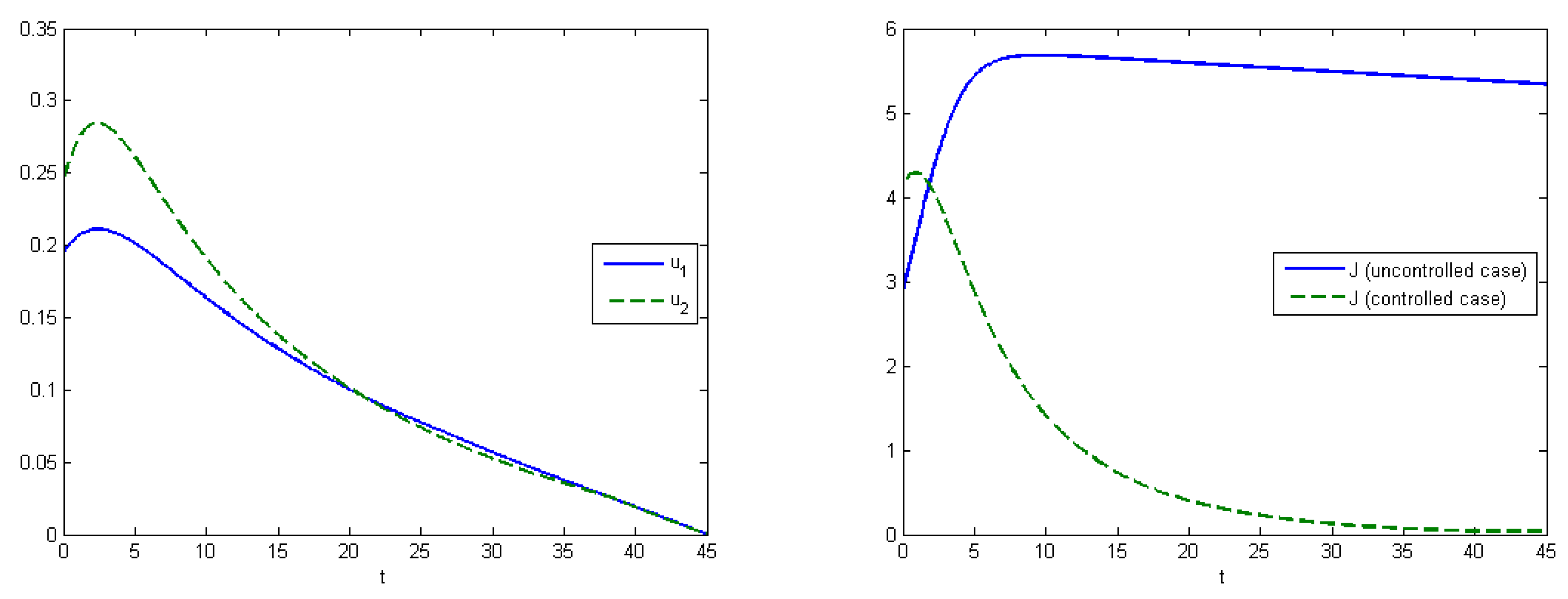
The first experiment shows the behavior of the system in the absence of treatment (control). As a control strategy, we used the medical treatment of the infected host, and the convex form of cost functions, i.e.,
and
. After simulations, we obtained that the maximum amount of replicas in the case without applying treatment were
at
, and
at
(
Figure 2). From Experiment 1, we can see that, in the absence of treatment in the end of epidemic period, our population had the following distribution of Infected hosts:
are infected by first type of virus and
are infected by the second type of virus. As we have self-recovery rate
and
, a fraction of the Recovered is
. In the case when we applied the control, the fraction of infected hosts was zero at the end of interval
, and the fraction of the recovered was
. There were also some susceptible nodes (
) that were not affected by the epidemic. Comparing the controlled and uncontrolled case, the aggregated system costs were:
in the uncontrolled model and
in the controlled model (
Figure 3).
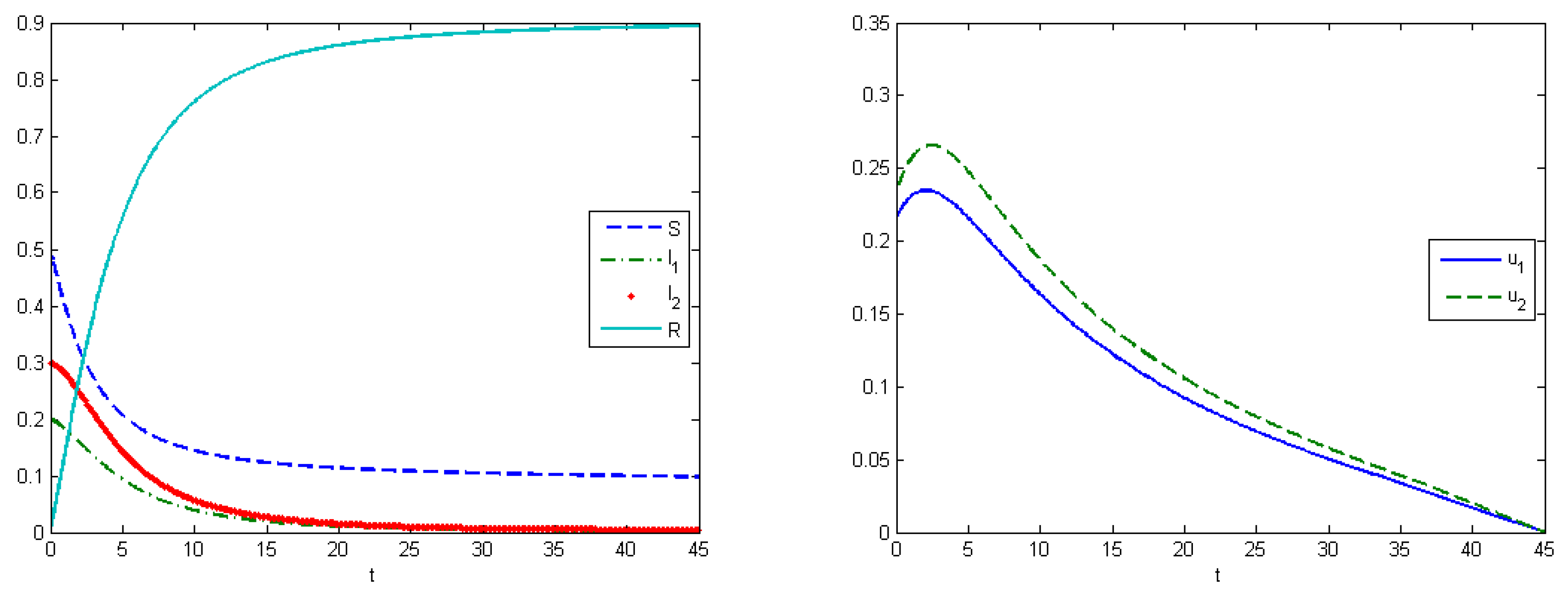
Experiment 2 represents the SIR model with virus mutation. Infection rates indicate the speed of viruses spreading in the population. In our work, it means that we take the competition between viruses for the host into account. Here, we supposed that the stronger virus captures more hosts. The strength of the virus depends on the infection rates, which change under a mutation process. In this experiment, infection rates changed by the formula for . Evolutionary dynamics allow us to estimate how viruses evolve in an urban population, and to calculate the Nash equilibrium. The Nash equilibrium shows the expected proportions of infected populations after a certain amount of time. This estimation can be useful when building forecasts and tactics to prevent an epidemic. We can allocate available resources in such way that the aggregated costs of the system are minimized. Using the SIR model, we illustrate how the system develops under various types of control.
Here, we have utility of occupation functions as
and
, and energy-cost functions are
and
. According to these data, we observed that the Nash equilibrium was equal to
in the case when we did not apply any treatment.
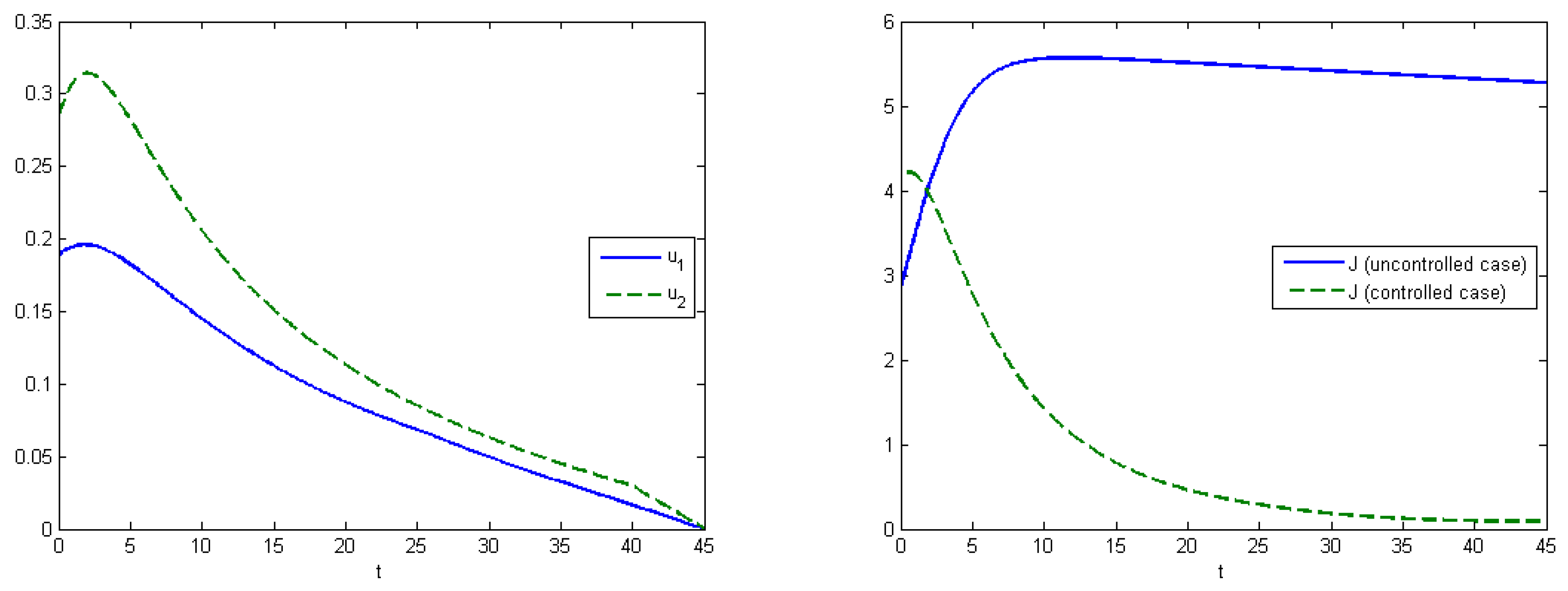
Figure 4 (left) shows the behavior of the system in the uncontrolled case, where the maximum population of
was
at
and the maximum population of
was
at
. By applying optimal treatment strategies, we observed that the maximal values for both types of viruses were equal to their initial state (
and
). The structure of the optimal control in the case with the virus mutation is shown in
Figure 5 (left). Comparison of aggregated costs is presented on
Figure 5 (right). The aggregated system cost was
in the uncontrolled model and
in the controlled model.
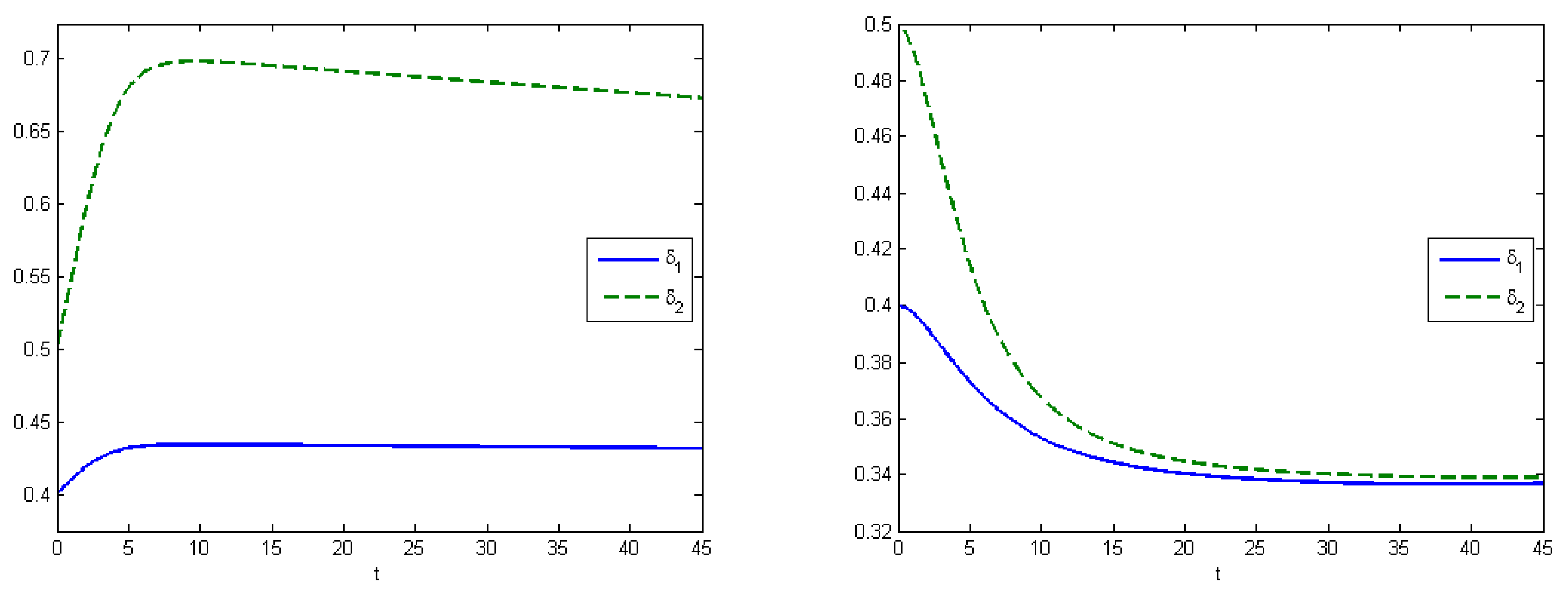
Experiment 3 shows the SIR model with the virus mutation (
Figure 6). Utility-of-occupation functions were
and
, and energy-cost functions were
and
. The expected proportions of infected subpopulations were equal to the Nash equilibrium
, according to simulations we received that the difference between parameters
and
influences the equilibrium state. From Equation (
1), we have that the fraction of the stronger virus is higher if the difference is smaller.
Figure 7 demonstrates the change of the infection rates of Experiments 2 and 3. Under the application of optimal control, we have that the maximum values for both types of viruses are equal to their initial state (
and
).
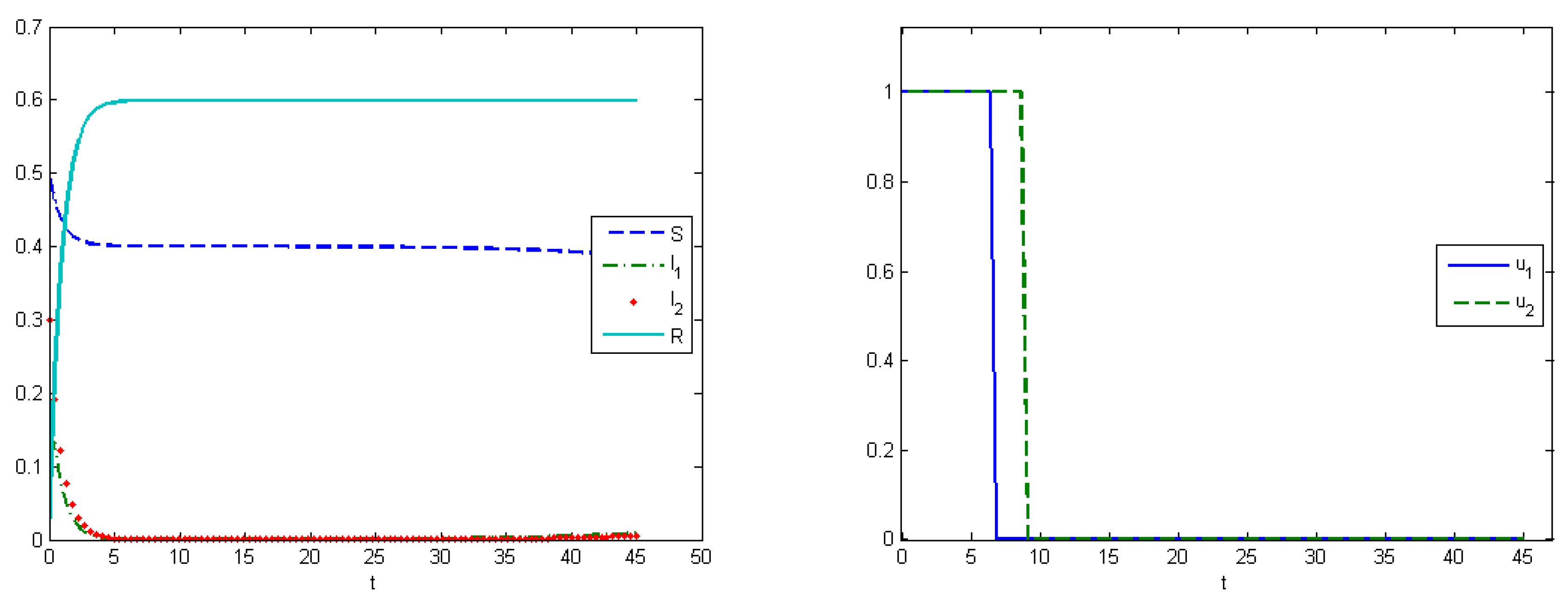
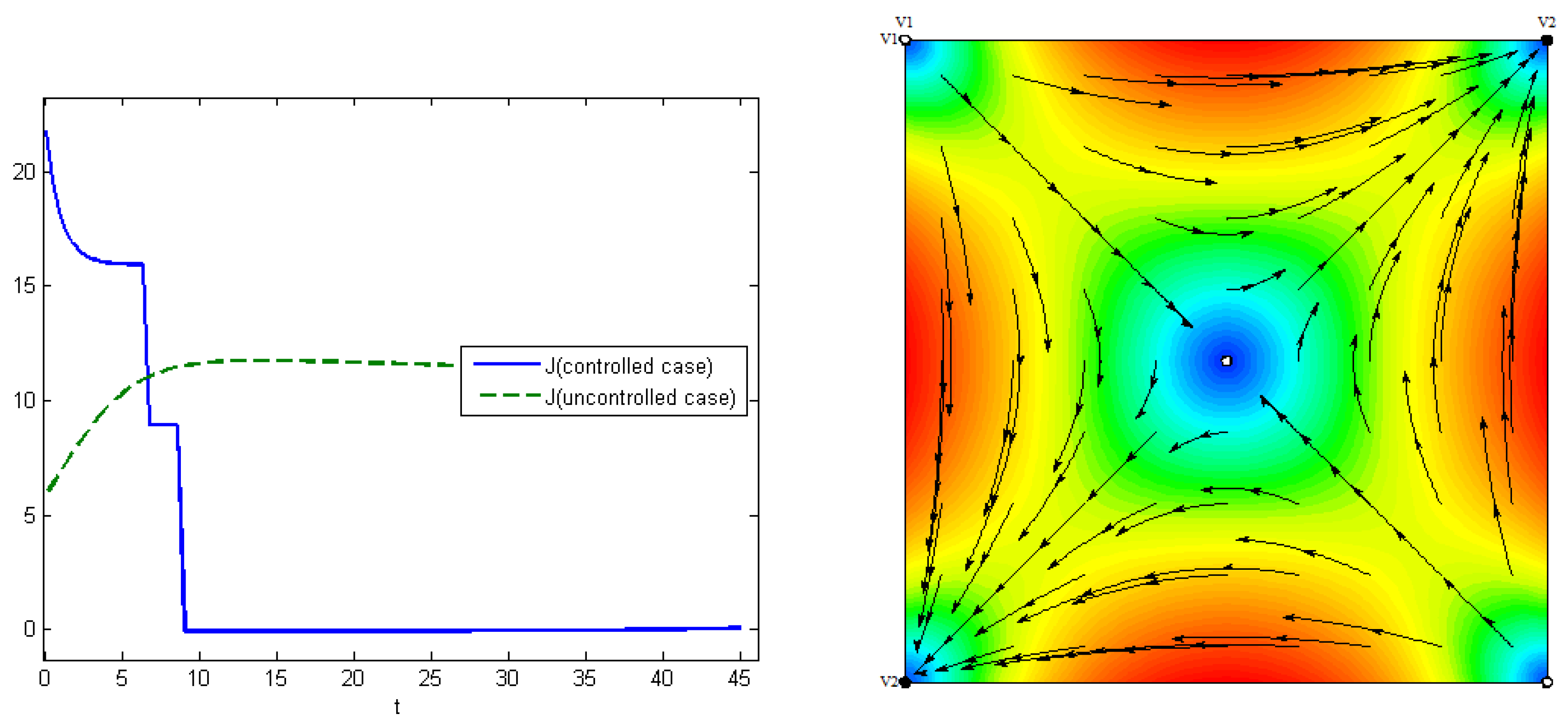
The fourth experiment describes the case when the costs for medical treatments are concave functions. In this case, we have proven in
Section 5 that the optimal control has a “bang-bang” structure, i.e.,
Figure 8 (right). The control for the first type of virus is turned off on the seventh day, and, for the second type, on the ninth day. At the end of interval
, the proportion of the recovered hosts was
, while the remaining population was still susceptible to infection. The comparisons of the aggregated costs are presented on
Figure 9 (left). Aggregated system cost was
in the uncontrolled model and
in the controlled model.
Figure 9 (right) demonstrates the evolution of viruses over time in human populations. Here, we can see that there are three stationary states corresponding to three Nash equilibria, and that the convergence of solution trajectories of ODE (
1) depends on the initial states.