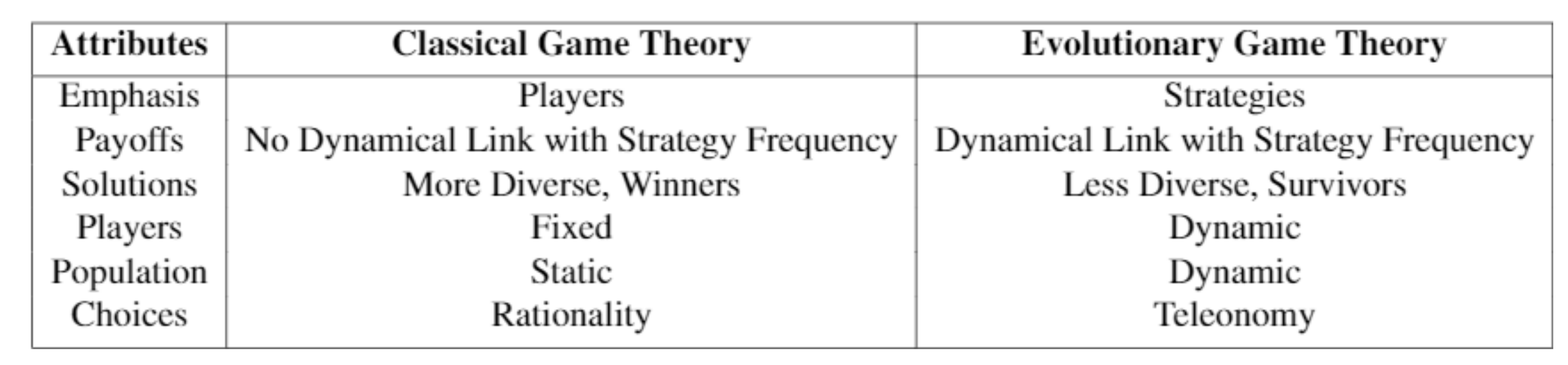
Evolutionary Game Theory: Darwinian Dynamics and the G Function Approach
Abstract
:1. Introduction
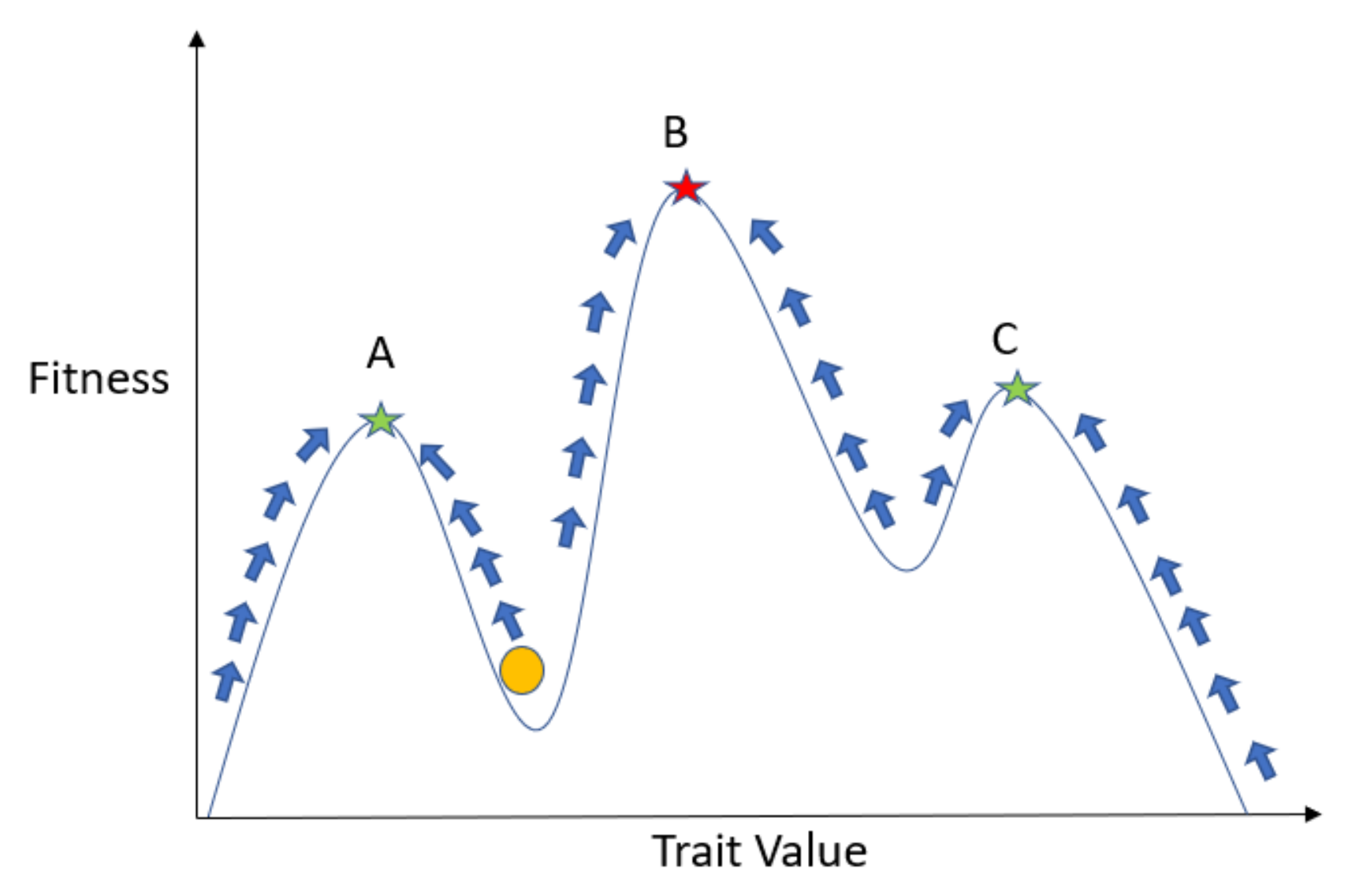
2. Evolutionary Game Theory: Darwinian Dynamics and G Functions
2.1. Introduction to G Functions
2.2. Population and Strategy Dynamics
2.3. G Function Modeling Recipe
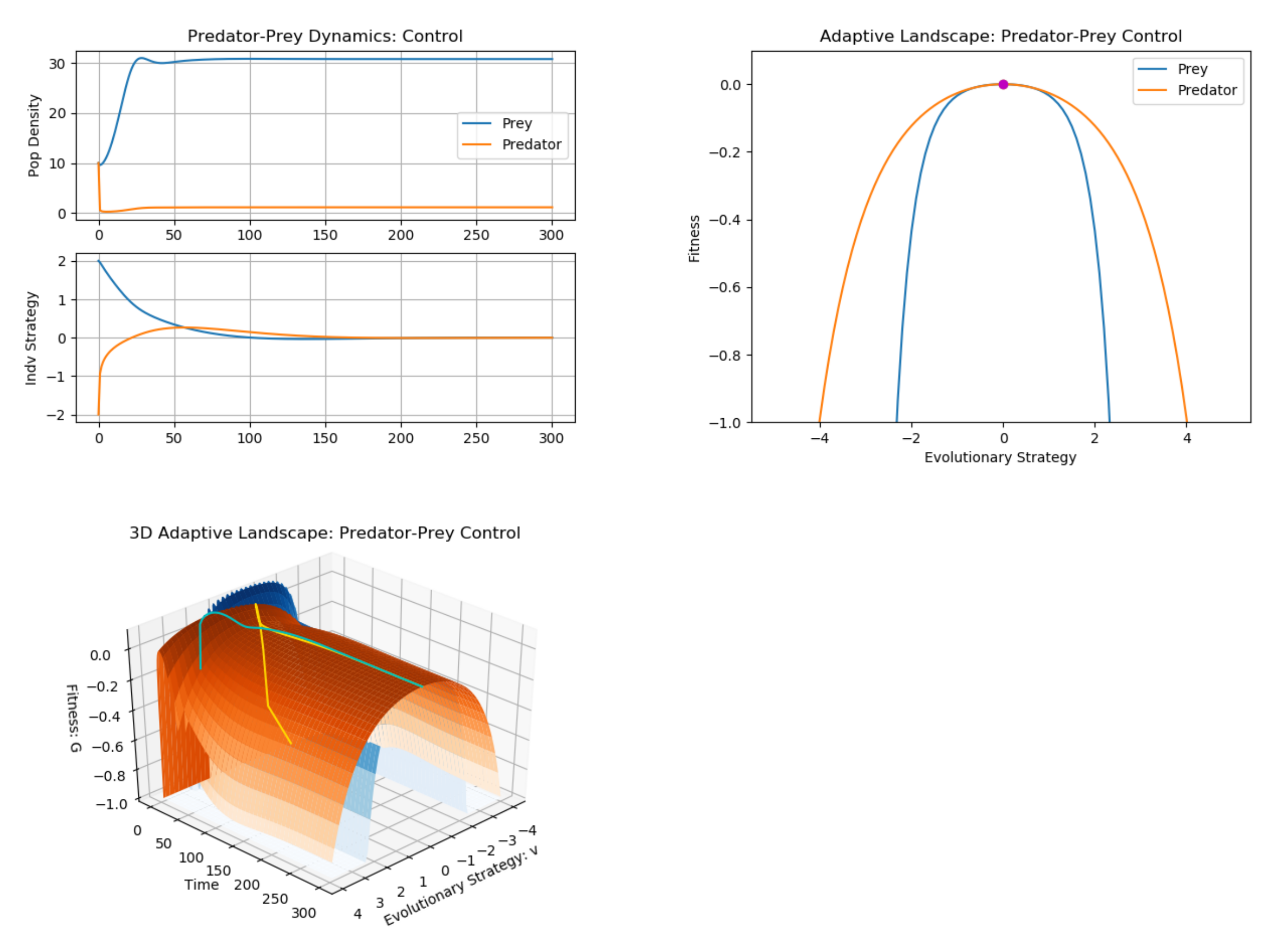
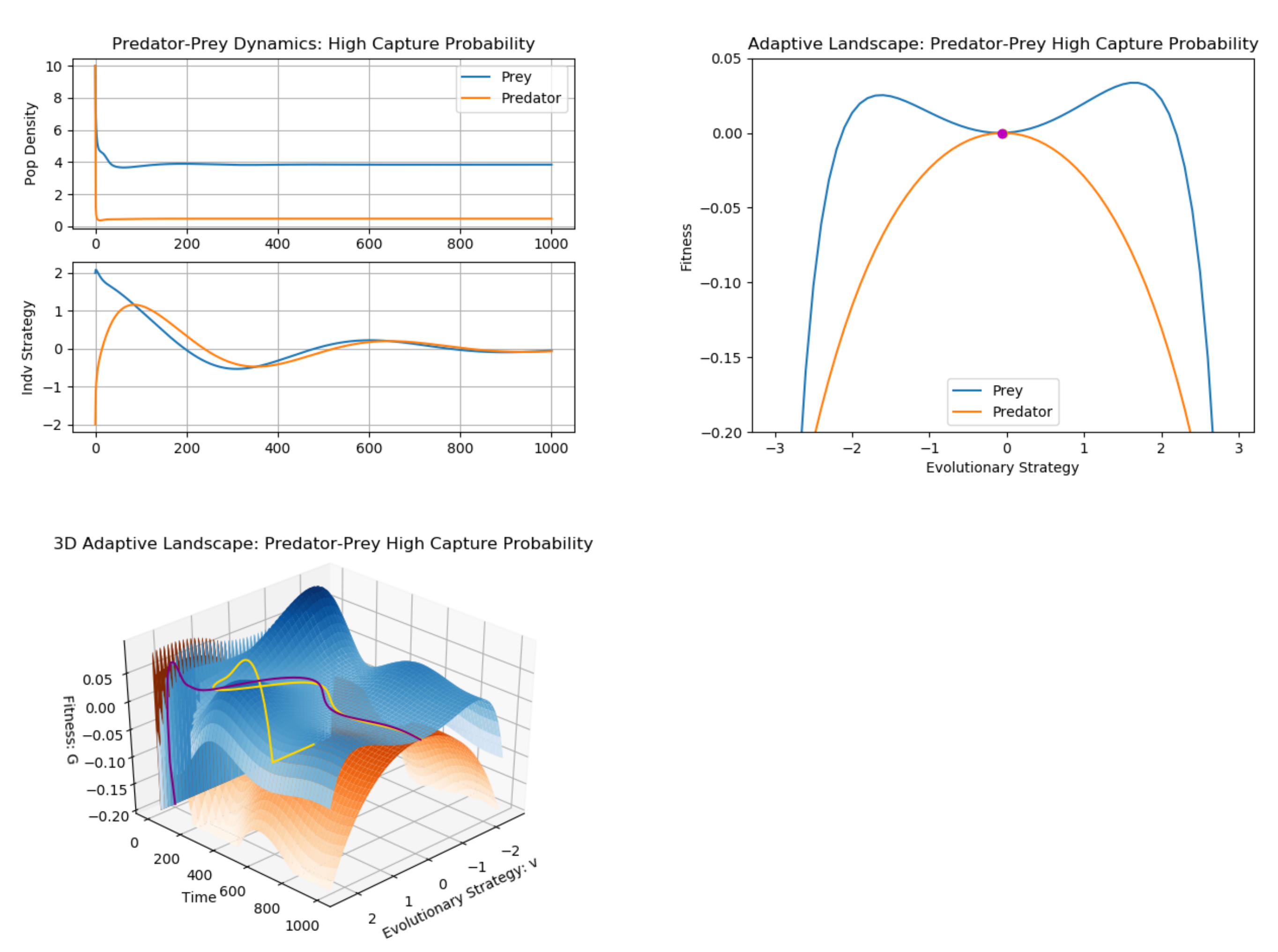
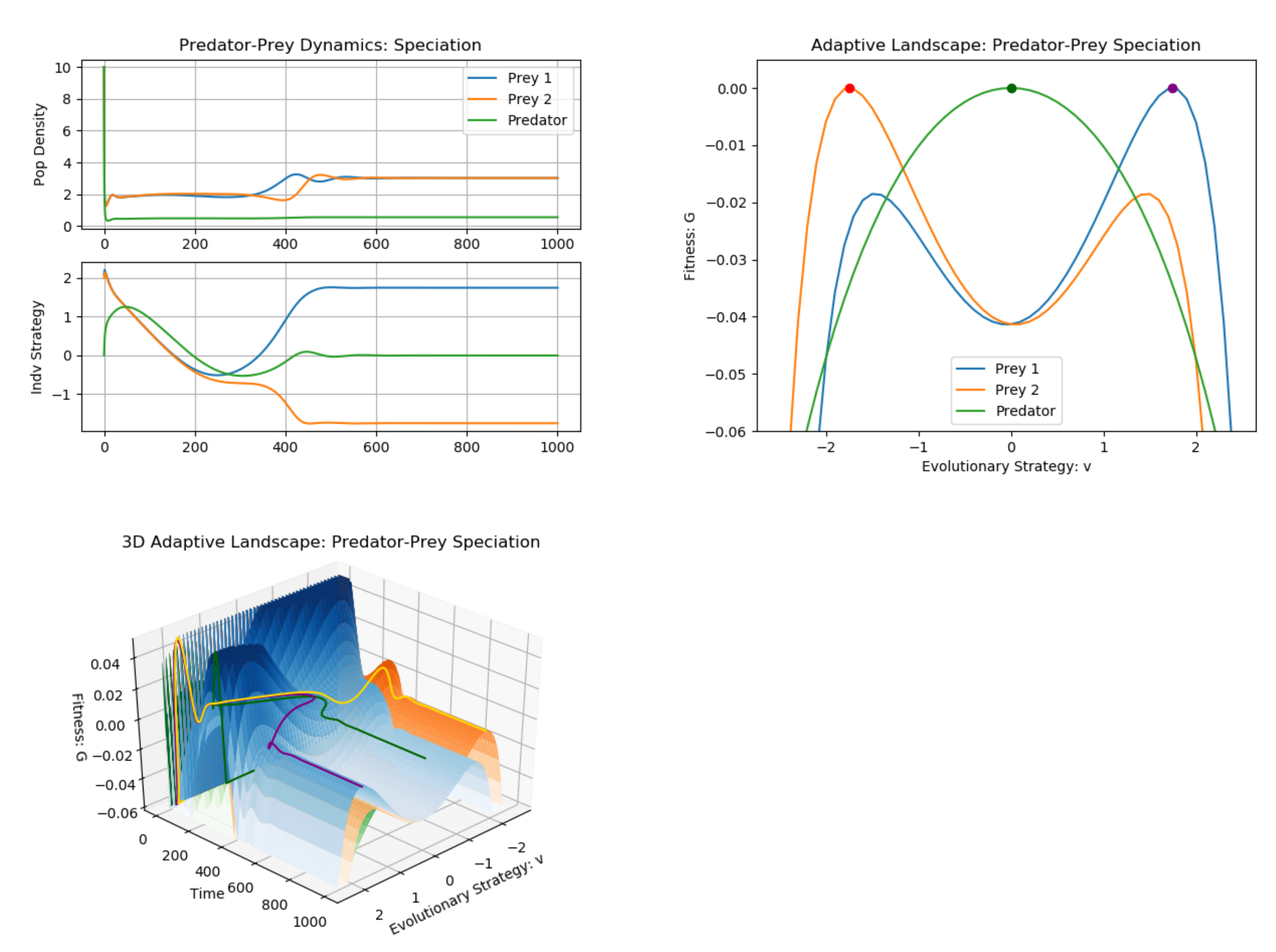
3. Example: Predator–Prey (Multiple G Functions)
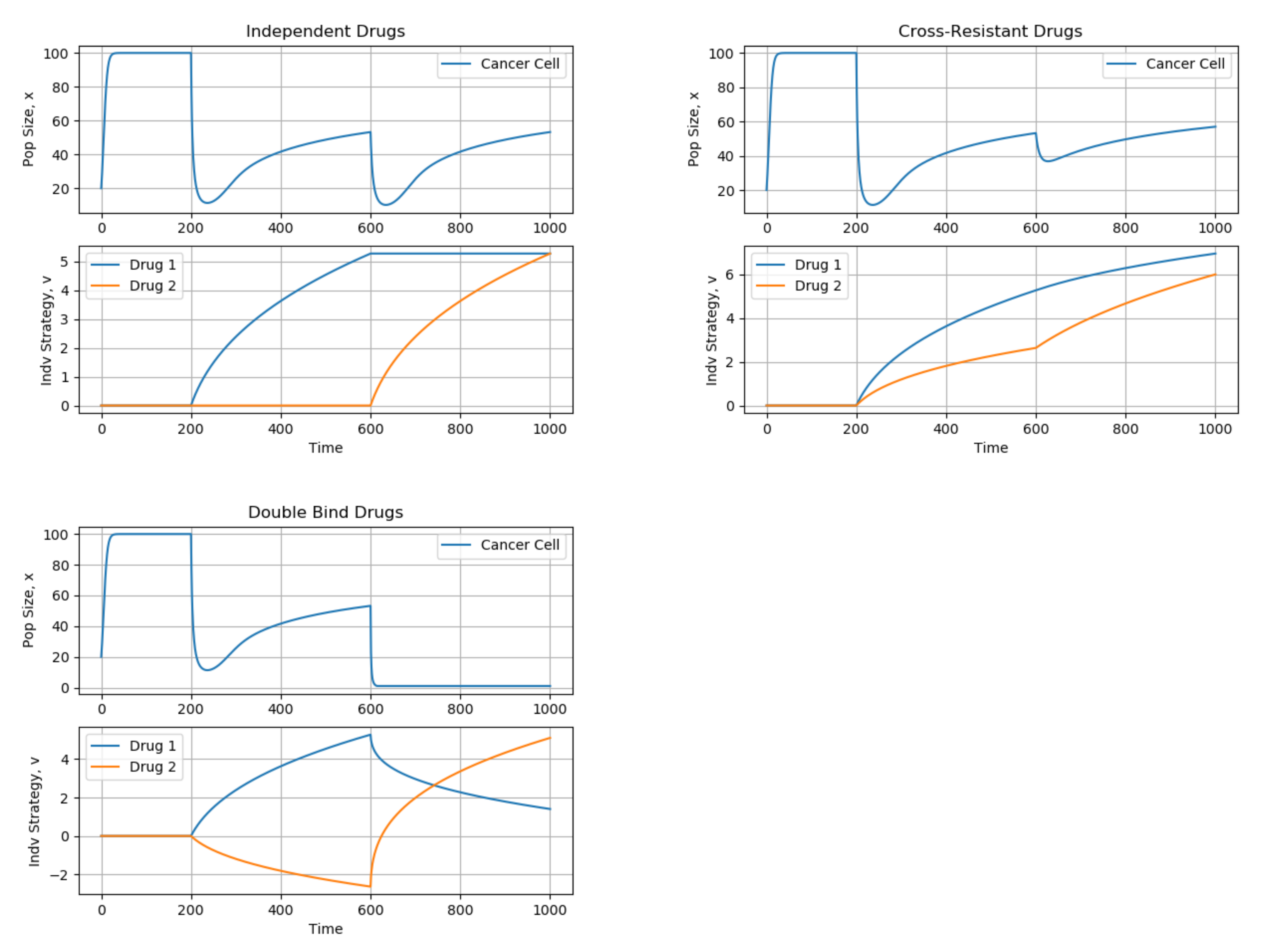
4. Example: Combination Therapy in Cancer (Vector-Valued Strategies)
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Smith, J.M.; Price, G.R. The Logic of Animal Conflict. Nature 1973, 246, 15–18. [Google Scholar] [CrossRef]
- Cohen, Y.; Vincent, T.L.; Brown, J.S. A G-function approach to fitness minima, fitness maxima, evolutionarily stable strategies and adaptive landscapes. Evol. Ecol. Res. 1999, 1, 923–942. [Google Scholar]
- Dieckmann, U.; Law, R. The dynamical theory of coevolution: A derivation from stochastic ecological processes. J. Math. Biol. 1996, 34, 579–612. [Google Scholar] [CrossRef]
- Metz, J.A.J.; de Kovel, C.G. The canonical equation of adaptive dynamics for Mendelian diploids and haplo-diploids. Interface Focus 2013, 3, 20130025. [Google Scholar] [CrossRef] [Green Version]
- Metz, J.A.J.; Staňková, K.; Johansson, J. The canonical equation of adaptive dynamics for life histories: From fitness-returns to selection gradients and Pontryagin’s maximum principle. J. Math. Biol. 2016, 72, 1125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vincent, T.L.; Brown, J.S. Evolutionary Game Theory, Natural Selection, and Darwinian Dynamics; Cambridge University Press: Cambridge, UK, 2005; pp. 1–382. [Google Scholar] [CrossRef]
- Apaloo, J.; Brown, J.S.; Vincent, T.L. Evolutionary game theory: ESS, convergence stability, and NIS. Evol. Ecol. Res. 2009, 11, 489–515. [Google Scholar]
- Mcgill, B.J.; Brown, J.S. Evolutionary Game Theory and Adaptive Dynamics of Continuous Traits. Annu. Rev. Ecol. Evol. Syst. 2007, 38, 403–435. [Google Scholar] [CrossRef] [Green Version]
- Von Neumann, J. Zur Theorie der Gesellschaftsspiele. Math. Ann. 1928, 100, 295–320. [Google Scholar] [CrossRef]
- Basu, K. On the non-existence of a rationality definition for extensive games. Int. J. Game Theory 1990, 19, 33–44. [Google Scholar] [CrossRef]
- Basu, K. The traveler’s dilemma: Paradoxes of rationality in game theory. Am. Econ. Rev. 1994, 84, 391–395. [Google Scholar] [CrossRef]
- Colman, A.M. Cooperation, psychological game theory, and limitations of rationality in social interaction. Behav. Brain Sci. 2003, 26, 139–153. [Google Scholar] [CrossRef] [Green Version]
- Korobkin, R.B.; Ulen, T.S. Law and Behavioral Science: Removing the Rationality Assumption from Law and Economics. Calif. Law Rev. 2000, 88, 1051. [Google Scholar] [CrossRef]
- Bukkuri, A. Optimal Control Analysis of Combined Chemotherapy-Immunotherapy Treatment Regimens in a PKPD Cancer Evolution Model. Biomath 2020, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Blythe, S.P.; Stokes, T.K. Some Consequences of Size-Selective Harvesting on Fitness and on Yield. Math. Med. Biol. 1990, 7, 41–53. [Google Scholar] [CrossRef]
- Law, R.; Grey, D.R. Evolution of yields from populations with age-specific cropping. Evol. Ecol. 1989, 3, 343–359. [Google Scholar] [CrossRef]
- Bierbach, D.; Schulte, M.; Herrmann, N.; Tobler, M.; Stadler, S.; Jung, C.T.; Kunkel, B.; Riesch, R.; Klaus, S.; Ziege, M.; et al. Predator-induced changes of female mating preferences: Innate and experiential effects. BMC Evol. Biol. 2011, 11, 190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dill, L.M. Male mating strategies under predation risk: Do females call the shots? Behav. Ecol. 1999, 10, 452–461. [Google Scholar] [CrossRef]
- Franklin, A.M.; Squires, Z.E.; Stuart-Fox, D. Does predation risk affect mating behavior? An experimental test in dumpling squid (Euprymna tasmanica). PLoS ONE 2014, 9, e115027. [Google Scholar] [CrossRef]
- Ghazy, N.A.; Gotoh, T.; Suzuki, T. Impact of global warming scenarios on life-history traits of Tetranychus evansi (Acari: Tetranychidae). BMC Ecol. 2019, 19, 48. [Google Scholar] [CrossRef] [Green Version]
- Lancaster, L.T.; Morrison, G.; Fitt, R.N. Life history trade-offs, the intensity of competition, and coexistence in novel and evolving communities under climate change. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160046. [Google Scholar] [CrossRef] [Green Version]
- Williams, J.L.; Jacquemyn, H.; Ochocki, B.M.; Brys, R.; Miller, T.E.X. Life history evolution under climate change and its influence on the population dynamics of a long-lived plant. J. Ecol. 2015, 103, 798–808. [Google Scholar] [CrossRef]
- Gause, G. The Struggle for Existence. Ann. Entomol. Soc. Am. 1935, 28, 59. [Google Scholar] [CrossRef]
- Mallet, J. The struggle for existence: How the notion of carrying capacity, K, obscures the links between demography, Darwinian evolution, and speciation. Evol. Ecol. Res. 2012, 14, 627–665. [Google Scholar]
- Turchin, P. Does population ecology have general laws? Oikos 2001, 94, 17–26. [Google Scholar] [CrossRef] [Green Version]
- Vincent, T.L.; Cressman, R. An ESS Maximum Principle for Matrix Games. Theor. Popul. Biol. 2000, 58, 173–186. [Google Scholar] [CrossRef] [PubMed]
- Broom, M.; Rychtar, J. Game-Theoretical Models in Biology; CRC Press: Boca Raton, FL, USA, 2013; pp. 1–520. [Google Scholar]
- Nowak, M. Evolutionary Dynamics: Exploring the Equations of Life; Harvard University Press: Cambridge, MA, USA, 2006; pp. 1–384. [Google Scholar]
- Sandholm, W. Population Games and Evolutionary Dynamics; MIT Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Weibull, J. Evolutionary Game Theory; MIT Press: Cambridge, MA, USA, 1987; pp. 1–265. [Google Scholar]
- Day, T.; Taylor, P.D. Evolutionary dynamics and stability in discrete and continuous games. Evol. Ecol. Res. 2003, 5, 605–613. [Google Scholar]
- Basanta, D.; Gatenby, R.A.; Anderson, A.R. Exploiting evolution to treat drug resistance: Combination therapy and the double bind. Mol. Pharm. 2012, 9, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Wölfl, B.; te Rietmole, H.; Salvioli, M.; Kaznatcheev, A.; Thuijsman, F.; Brown, J.S.; Burgering, B.; Staňková, K. The Contribution of Evolutionary Game Theory to Understanding and Treating Cancer. Dyn. Games Appl. 2021, 1–30. [Google Scholar] [CrossRef]
- Rieppel, O. ’Type’ in morphology and phylogeny. J. Morphol. 2006, 267, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Willmore, K.E. The Body Plan Concept and Its Centrality in Evo-Devo. Evol. Educ. Outreach 2012, 5, 219–230. [Google Scholar] [CrossRef] [Green Version]
- Brown, J.S. Why Darwin would have loved evolutionary game theory. Proc. R. Soc. B Biol. Sci. 2016, 283, 20160847. [Google Scholar] [CrossRef] [Green Version]
- Shubin, N.H. The Implications of “The Bauplan” for Development and Evolution of the Tetrapod Limb. In Developmental Patterning of the Vertebrate Limb; Springer: Boston, MA, USA, 1991; pp. 411–421. [Google Scholar] [CrossRef]
- Wright, S. The Roles of Mutation, Inbreeding, Crossbreeding and Selection in Evolution. In Proceedings of the 6th International Congress of Genetics, Ithaca, NY, USA, 6 December 1932; pp. 356–366. [Google Scholar]
- Roughgarden, J. Resource partitioning among competing species-A coevolutionary approach. Theor. Popul. Biol. 1976, 9, 388–424. [Google Scholar] [CrossRef]
- Abrams, P.A. Alternative Models of Character Displacement and Niche Shift. I. Adaptive Shifts in Resource Use when there is Competition for Nutritionally Nonsubstitutable Resources. Evolution 1987, 41, 651–661. [Google Scholar] [CrossRef]
- Brown, J.; Vincent, T. Coevolution as an Evolutionary Game. Evolution 1987, 41, 66–79. [Google Scholar] [CrossRef] [Green Version]
- Taper, M.L.; Case, T.J. Models of Character Displacement and the Theoretical Robustness of Taxon Cycles. Evolution 1992, 46, 317–333. [Google Scholar] [CrossRef] [Green Version]
- Beatty, J.; Carrera, I. When what had to happen was not bound to happen: History, chance, narrative, evolution. J. Philos. Hist. 2011, 5, 471–495. [Google Scholar] [CrossRef]
- Blount, Z.D.; Lenski, R.E.; Losos, J.B. Contingency and determinism in evolution: Replaying life’s tape. Science 2018, 362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Losos, J.B. Adaptive radiation, ecological opportunity, and evolutionary determinism: American society of naturalists E. O. Wilson award address. Am. Nat. 2010, 175, 623–639. [Google Scholar] [CrossRef]
- Mahler, D.L.; Ingram, T.; Revell, L.J.; Losos, J.B. Exceptional convergence on the macroevolutionary landscape in island lizard radiations. Science 2013, 341, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.C. Evolution: Like any other science it is predictable. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 133–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Powell, S.; Price, S.L.; Kronauer, D.J. Trait evolution is reversible, repeatable, and decoupled in the soldier caste of turtle ants. Proc. Natl. Acad. Sci. USA 2020, 117, 6608–6615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christiansen, F.B. On Conditions for Evolutionary Stability for a Continuously Varying Character. Am. Nat. 1991, 138, 37–50. [Google Scholar] [CrossRef]
- Eshel, I. Evolutionary and continuous stability. J. Theor. Biol. 1983, 103, 99–111. [Google Scholar] [CrossRef]
- Eshel, I. On the changing concept of evolutionary population stability as a reflection of a changing point of view in the quantitative theory of evolution. J. Math. Biol. 1996, 34, 485–510. [Google Scholar] [CrossRef]
- Eshel, I.; Motro, U. Kin selection and strong evolutionary stability of mutual help. Theor. Popul. Biol. 1981, 19, 420–433. [Google Scholar] [CrossRef]
- Vincent, T.L.; Brown, J.S. Stability in an evolutionary game. Theor. Popul. Biol. 1984, 26, 408–427. [Google Scholar] [CrossRef]
- Apaloo, J. Revisiting strategic models of evolution: The concept of neighborhood invader strategies. Theor. Popul. Biol. 1997, 52, 71–77. [Google Scholar] [CrossRef]
- Metz, J.; Geritz, S.; Meszena, G.; Jacobs, F.; Heerwaarden, J.V. Adaptive dynamics: A geometrical study of the consequences of nearly faithful reproduction. Stoch. Spat. Struct. Dyn. Syst. 1996, 45, 183–231. [Google Scholar]
- Brännström, A.; Johansson, J.; von Festenberg, N. The Hitchhiker’s guide to adaptive dynamics. Games 2013, 4, 304–328. [Google Scholar] [CrossRef] [Green Version]
- Diekmann, O. A Beginner’s Guide to Adaptive Dynamics. Math. Model. Popul. Dyn. 2004, 63, 47–86. [Google Scholar]
- Grant, P.R.; Grant, B.R. Unpredictable evolution in a 30-year study of Darwin’s finches. Science 2002, 296, 707–711. [Google Scholar] [CrossRef] [Green Version]
- Geritz, S.A.; Kisdi, E.; Meszéna, G.; Metz, J.A. Evolutionarily singular strategies and the adaptive growth and branching of the evolutionary tree. Evol. Ecol. 1998, 12, 35–57. [Google Scholar] [CrossRef]
- Bomze, I.; Weibull, J. Does Neutral Stability Imply Lyapunov Stability? Games Econ. Behav. 1995, 11, 173–192. [Google Scholar] [CrossRef]
- Hamilton, W.D. Extraordinary sex ratios. Science 1967, 156, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Geritz, S.A.; Metz, J.A.; Kisdi, E.; Meszéna, G. Dynamics of adaptation and evolutionary branching. Phys. Rev. Lett. 1997, 78, 2024–2027. [Google Scholar] [CrossRef] [Green Version]
- Cunningham, J.J.; Gatenby, R.A.; Brown, J.S. Evolutionary Dynamics in Cancer Therapy. Mol. Pharm. 2011, 8, 2094–2100. [Google Scholar] [CrossRef] [PubMed]
- Vincent, T.L.; Vincent, T.L. Using the ESS Maximum Principle to Explore Root-shoot Allocation, Competition and Coexistence. J. Theor. Biol. 1996, 180, 111–120. [Google Scholar] [CrossRef] [Green Version]
- Bukkuri, A.; Gatenby, R.A.; Brown, J.S. GLUT1 Production in Cancer Cells: A Tragedy of the Commons. 2021; submitted. [Google Scholar]
- Brown, J.S.; Vincent, T.L. Organization of Predator–Prey Communities as an Evolutionary Game. Evolution 1992, 46, 1269–1283. [Google Scholar] [CrossRef] [PubMed]
- Bukkuri, A.; Brown, J. Coevolution, Red Queen Dynamics, Zahavi Handicap, and Double Binds: From Ecology to Cancer. 2021; in preparation. [Google Scholar]
- Moreno-Rueda, G. Disruptive selection by predation offsets stabilizing selection on shell morphology in the land snail Iberus g. gualtieranus. Evol. Ecol. 2009, 23, 463–471. [Google Scholar] [CrossRef]
- Reed, W.L.; Janzen, F.J. Natural selection by avian predators on size and colour of a freshwater snail (Pomacea flagellata). Biol. J. Linn. Soc. 1999, 67, 331–342. [Google Scholar] [CrossRef]
- Gatenby, R.; Brown, J. The evolution and ecology of resistance in cancer therapy. Cold Spring Harb. Perspect. Med. 2018, 8, a033415. [Google Scholar] [CrossRef]
- Strobl, M.A.; West, J.; Viossat, Y.; Damaghi, M.; Robertson-Tessi, M.; Brown, J.S.; Gatenby, R.A.; Maini, P.K.; Anderson, A.R. Turnover modulates the need for a cost of resistance in adaptive therapy. Cancer Res. 2021, 81, 1135–1147. [Google Scholar] [CrossRef]
- Buck, S.A.; Koolen, S.L.; Mathijssen, R.H.; de Wit, R.; van Soest, R.J. Cross-resistance and drug sequence in prostate cancer. Drug Resist. Updat. 2021, 56, 100761. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Mirza, N.; Fricke, I.; Chiappori, A.; Thompson, P.; Williams, N.; Bepler, G.; Simon, G.; Janssen, W.; Lee, J.H.; et al. Combination of p53 cancer vaccine with chemotherapy in patients with extensive stage small cell lung cancer. Clin. Cancer Res. 2006, 12, 878–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amine, K. Multiobjective Simulated Annealing: Principles and Algorithm Variants. Adv. Oper. Res. 2019, 2019, 8134674. [Google Scholar] [CrossRef]
- Guttenberg, N.; Virgo, N.; Penn, A. On the potential for open-endedness in neural networks. Artif. Life 2019, 25, 145–167. [Google Scholar] [CrossRef] [Green Version]
- Suman, B.; Kumar, P. A Survey of Simulated Annealing as a Tool for Single and Multiobjective Optimization. J. Oper. Res. Soc. 2006, 57, 1143–1160. [Google Scholar] [CrossRef]
- Schäfer, F.; Anandkumar, A. Competitive Gradient Descent. Adv. Neural Inf. Process. Syst. 2019, 32, 1–11. [Google Scholar]
- Toro-Zapata, H.D.; Olivar-Tost, G.; Dercole, F. Conditions on the Energy Market Diversification from Adaptive Dynamics. Math. Probl. Eng. 2018, 2018, 9181636. [Google Scholar] [CrossRef]
- Bukkuri, A.; Pienta, K.J.; Amend, S.R.; Hammarlund, E.U.; Brown, J.S. The Contribution of Evolvability to the Eco-Evolutionary Dynamics of Competing Species. 2021; submitted. [Google Scholar]
- Adami, C.; Schossau, J.; Hintze, A. Evolutionary game theory using agent-based methods. Phys. Life Rev. 2016, 19, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Dieckmann, U.; Marrow, P.; Law, R. Evolutionary cycling in predator–prey interactions: Population dynamics and the red queen. J. Theor. Biol. 1995, 176, 91–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dieckmann, U.; Ferrière, R. Adaptive Dynamics and Evolving Biodiversity. In Evolutionary Conservation Biology; Ferrière, R., Dieckmann, U., Couvet, D., Eds.; Cambridge University Press: Cambridge, UK, 2009; pp. 188–224. [Google Scholar] [CrossRef] [Green Version]
- Metz, J.A.; Nisbet, R.M.; Geritz, S.A. How should we define ‘fitness’ for general ecological scenarios? Trends Ecol. Evol. 1992, 7, 198–202. [Google Scholar] [CrossRef]
- Cohen, Y.; Galiano, G. Evolutionary Distributions and Competition by Way of Reaction-Diffusion and by Way of Convolution. Bull. Math. Biol. 2013, 75, 2305–2323. [Google Scholar] [CrossRef] [PubMed]
| Variable | Interpretation |
|---|---|
| G | Per Capita Growth Rate of a Member |
| v | Strategy of Focal Member |
| u | Vector of Strategies of all Members |
| x | Vector of Population Sizes of all Members |
| U | Evolutionary Strategy Set |
| Parameter | Interpretation | Value |
|---|---|---|
| Carrying Capacity | 100 | |
| Maximal Capture Probability | 0.15 | |
| c | Conversion Efficiency: Prey to Predator | 0.25 |
| Intrinsic Proliferation Rate | 0.25 | |
| Evolvability | 0.5 | |
| Range of Resources | 2 | |
| Species Niche Width | 4 | |
| Breadth of Predation | 10 | |
| Evolvability | 0.5 |
| Parameter | Interpretation | Value |
|---|---|---|
| Carrying Capacity | 100 | |
| r | Intrinsic Proliferation Rate | 0.25 |
| Evolvability Variance | 0.5 | |
| Evolvability Covariance | [−0.25, 0, 0.25] | |
| Initial Drug Resistance | 1 | |
| Impact of Drug Resistance on Death | 0.3 | |
| Drug Dosage/Efficacy | 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bukkuri, A.; Brown, J.S. Evolutionary Game Theory: Darwinian Dynamics and the G Function Approach. Games 2021, 12, 72. https://doi.org/10.3390/g12040072
Bukkuri A, Brown JS. Evolutionary Game Theory: Darwinian Dynamics and the G Function Approach. Games. 2021; 12(4):72. https://doi.org/10.3390/g12040072
Chicago/Turabian StyleBukkuri, Anuraag, and Joel S. Brown. 2021. "Evolutionary Game Theory: Darwinian Dynamics and the G Function Approach" Games 12, no. 4: 72. https://doi.org/10.3390/g12040072
APA StyleBukkuri, A., & Brown, J. S. (2021). Evolutionary Game Theory: Darwinian Dynamics and the G Function Approach. Games, 12(4), 72. https://doi.org/10.3390/g12040072






