Eugenia jambolana (Java Plum) Fruit Extract Exhibits Anti-Cancer Activity against Early Stage Human HCT-116 Colon Cancer Cells and Colon Cancer Stem Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Evaluation of the Bioactive Compound Profile in JPE
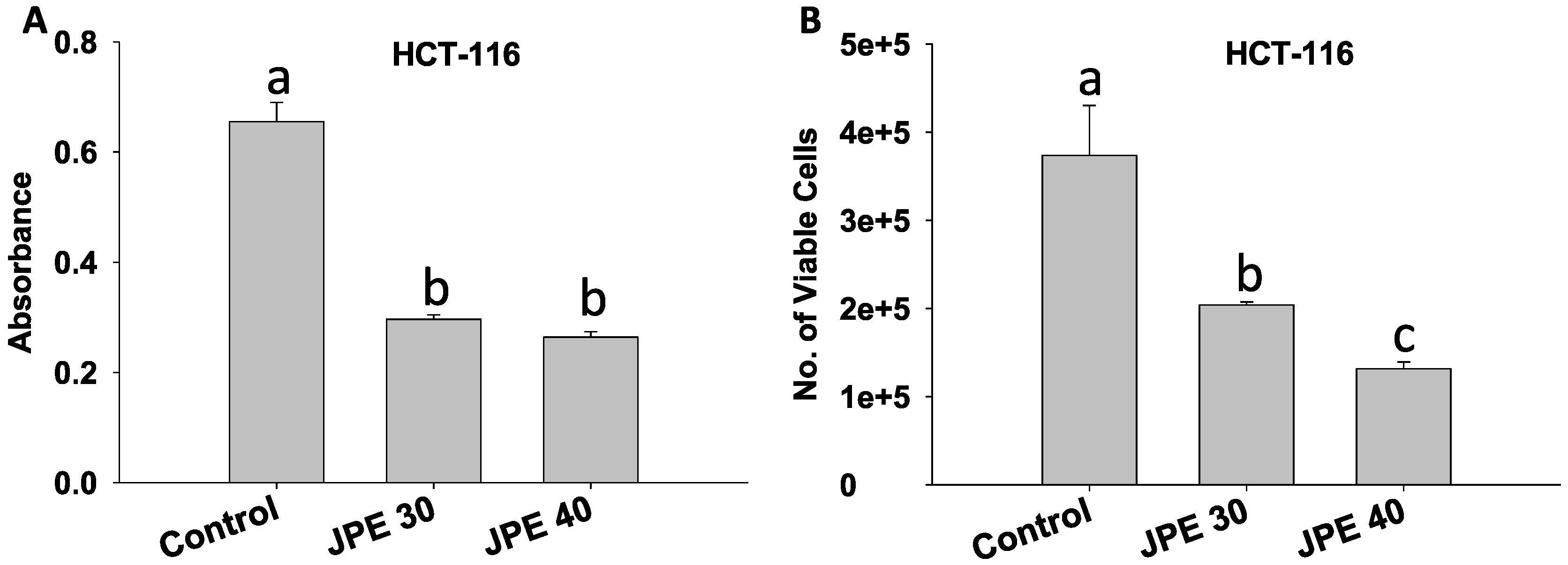
2.2. JPE Suppressed Proliferation in HCT-116 Cells
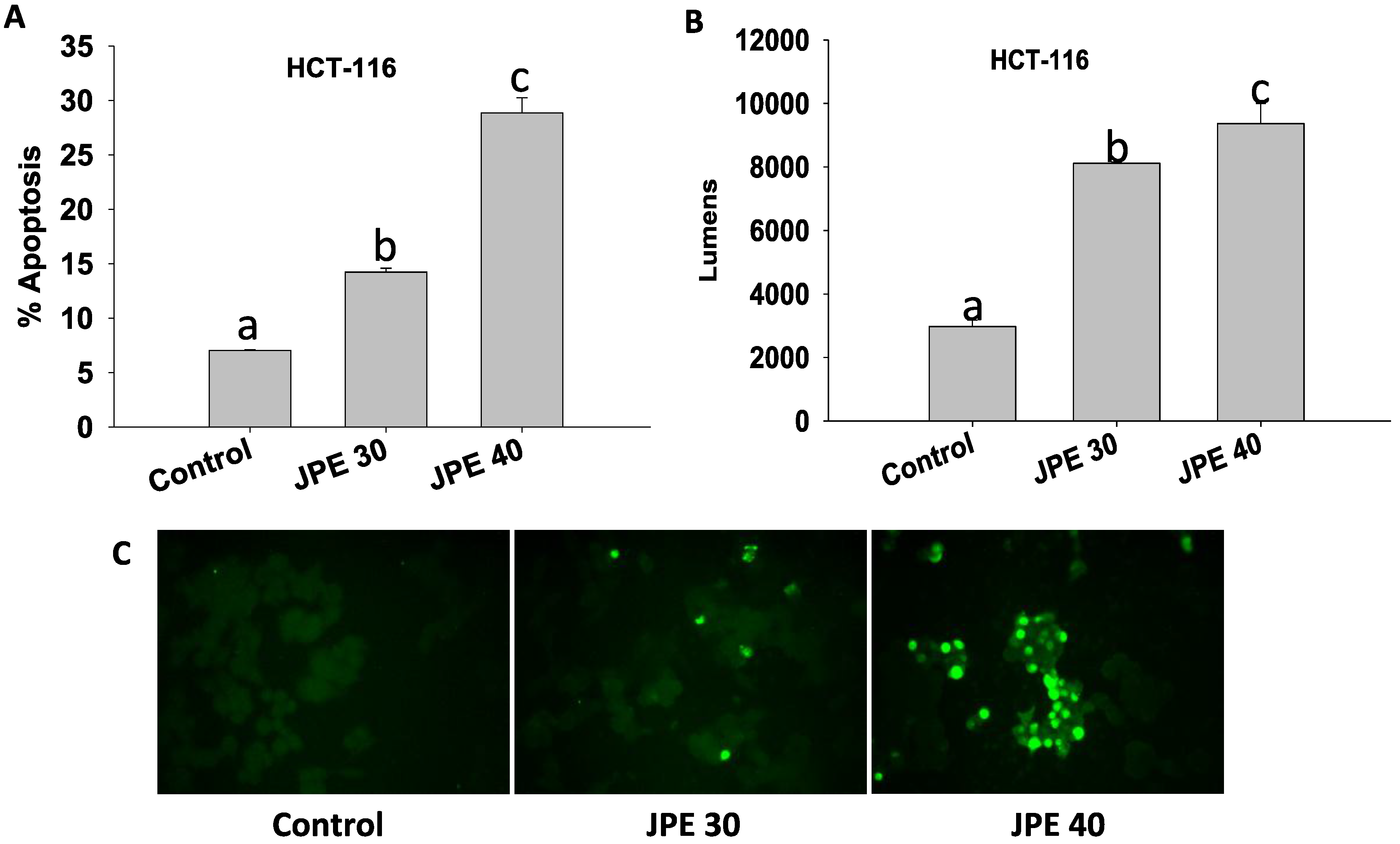
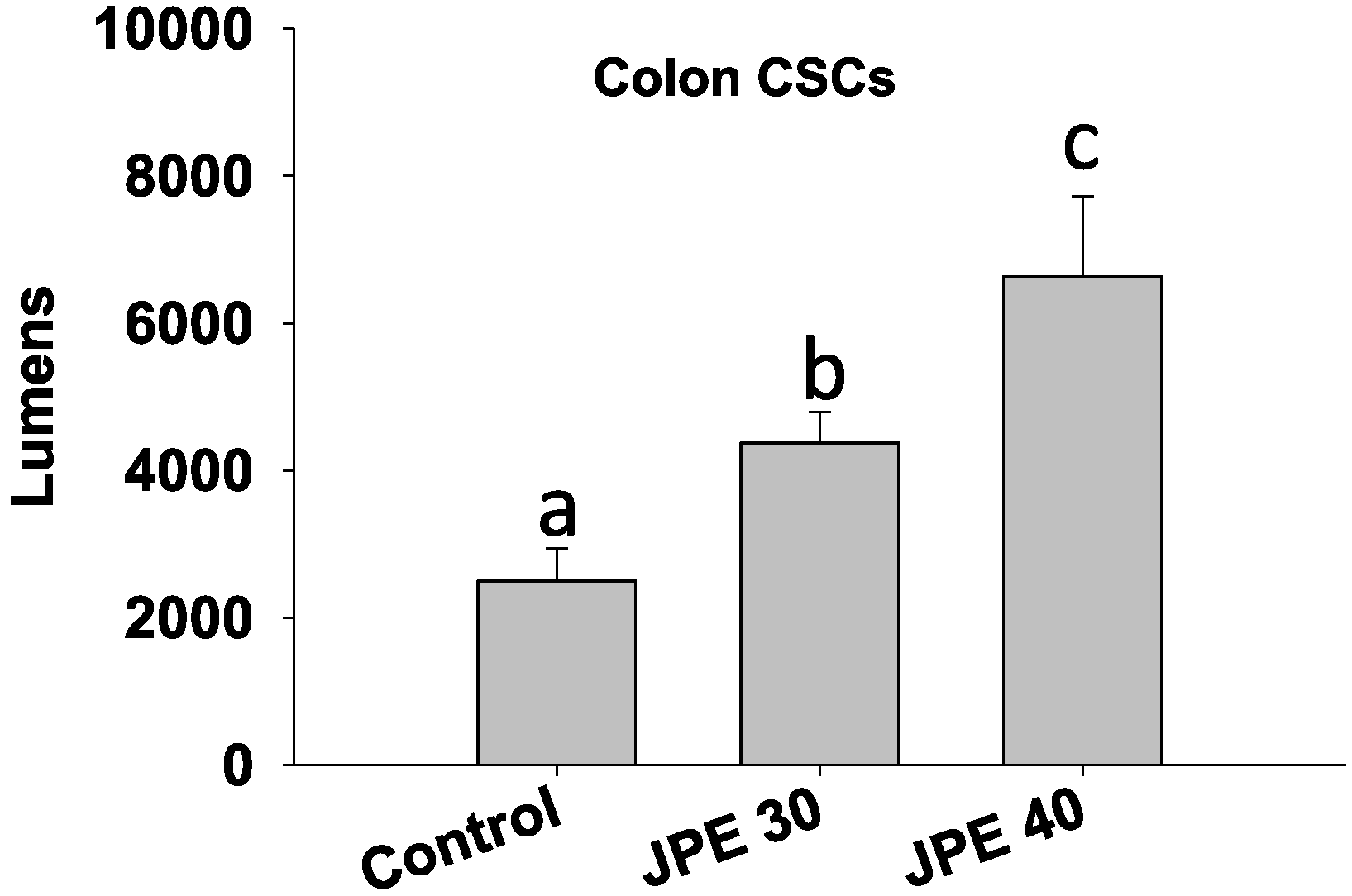
2.3. JPE Induced Apoptosis in HCT-116 Cells and Colon CSCs
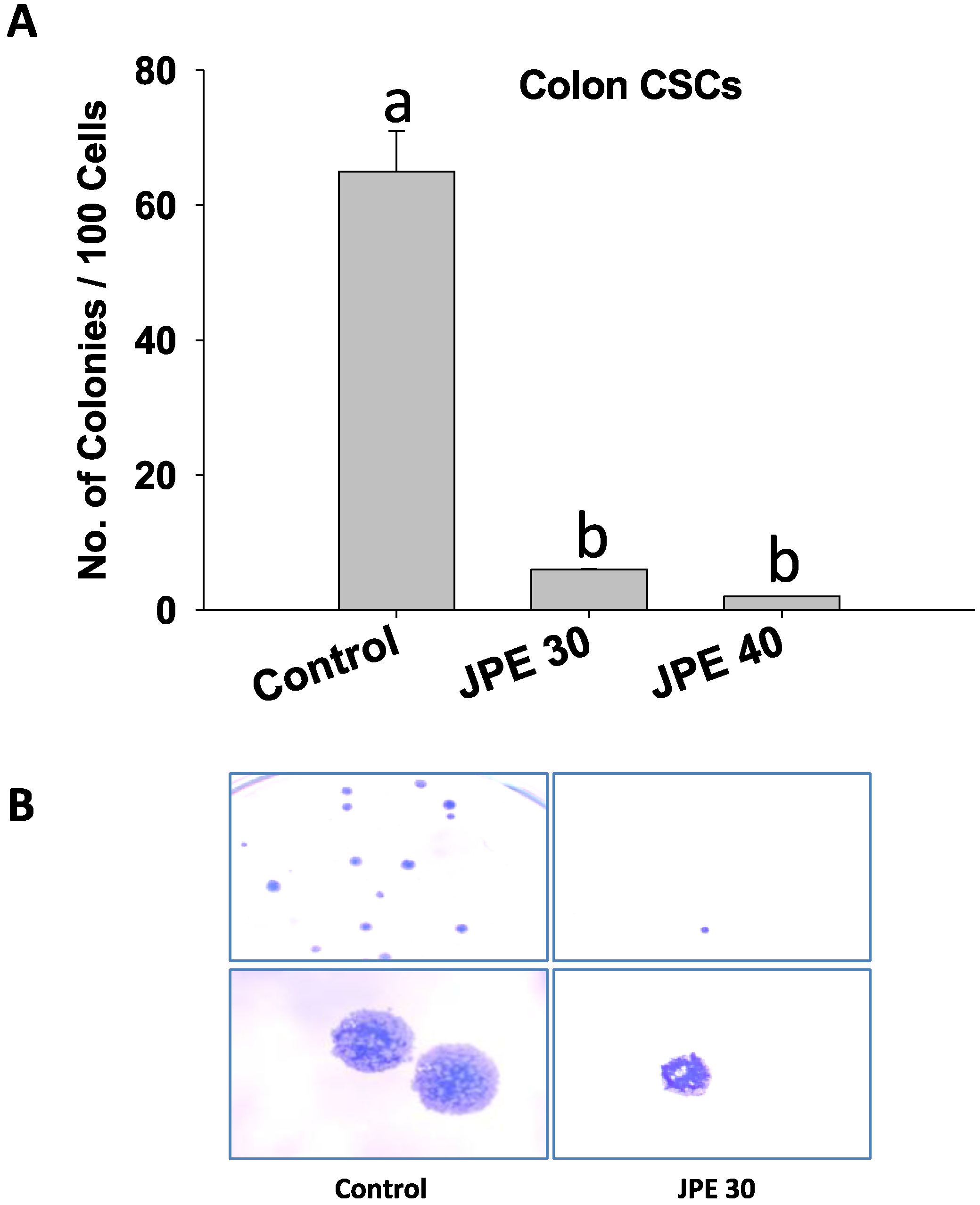
2.4. JPE Suppressed Colony Formation in Colon CSCs
3. Materials and Methods
3.1. Extraction and Purification of Anthocyanins from Java Plum
3.2. Chemicals
3.3. High Performance Liquid Chromatography Mass Spectrometry (HPLC-MS) Analysis
3.4. Cell Lines
3.5. Cell Viability
3.5.1. MTT Assay
3.5.2. Cell Counting
3.6. Apoptosis
3.6.1. Caspase Glo 3/7 Assay
3.6.2. TUNEL Assay
3.7. Colony Formation Assay
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, S.D.; Bertagnolli, M.M. Molecular origins of cancer: Molecular basis of colorectal cancer. N. Engl. J. Med. 2009, 361, 2449–2460. [Google Scholar] [CrossRef] [PubMed]
- Reya, T.; Morrison, S.J.; Clarke, M.F.; Weissman, I.L. Stem cells, cancer, and cancer stem cells. Nature 2001, 414, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Todaro, M.; Francipane, M.G.; Medema, J.P.; Stassi, G. Colon cancer stem cells: Promise of targeted therapy. Gastroenterology 2010, 138, 2151–2162. [Google Scholar] [CrossRef] [PubMed]
- Oh, P.S.; Patel, V.B.; Sanders, M.A.; Kanwar, S.S.; Yu, Y.; Nautiyal, J.; Patel, B.B.; Majumdar, A.P. Schlafen-3 decreases cancer stem cell marker expression and autocrine/juxtacrine signaling in folfox-resistant colon cancer cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Rosen, J.M. Stem cells in the etiology and treatment of cancer. Curr. Opin. Genet. Dev. 2006, 16, 60–64. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Colorectal cancer facts & figures 2014-2016. Available online: http://www.cancer.org/Research/CancerFactsStatistics/colorectal-cancer-facts-and-figures-2014-2016 (page 2; accessed on 25 January 2016).
- Aune, D.; Lau, R.; Chan, D.S.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Nonlinear reduction in risk for colorectal cancer by fruit and vegetable intake based on meta-analysis of prospective studies. Gastroenterology 2011, 141, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Boffetta, P.; Couto, E.; Wichmann, J.; Ferrari, P.; Trichopoulos, D.; Bueno-de-Mesquita, H.B.; Van Duijnhoven, F.J.; Büchner, F.L.; Key, T.; Boeing, H. Fruit and vegetable intake and overall cancer risk in the european prospective investigation into cancer and nutrition (EPIC). J. Natl. Cancer Inst. 2010, 102, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.; Favot, L.; Matz, R.; Lugnier, C.; Andriantsitohaina, R. Delphinidin inhibits endothelial cell proliferation and cell cycle progression through a transient activation of erk-1/-2. Biochem. Pharmacol. 2003, 65, 669–675. [Google Scholar] [CrossRef]
- Wang, L.S.; Stoner, G.D. Anthocyanins and their role in cancer prevention. Cancer Lett. 2008, 269, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Thomasset, S.; Berry, D.P.; Cai, H.; West, K.; Marczylo, T.H.; Marsden, D.; Brown, K.; Dennison, A.; Garcea, G.; Miller, A. Pilot study of oral anthocyanins for colorectal cancer chemoprevention. Cancer Prev. Res. 2009, 2, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Madiwale, G.P.; Reddivari, L.; Stone, M.; Holm, D.G.; Vanamala, J. Combined effects of storage and processing on the bioactive compounds and pro-apoptotic properties of color-fleshed potatoes in human colon cancer cells. J. Agric. Food Chem. 2012, 60, 11088–11096. [Google Scholar] [CrossRef] [PubMed]
- Stewart, B.; Wild, C. World cancer report 2014; International Agency for Research on Cancer of World Health Organization: Lyon, France, 2014. [Google Scholar]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2015. CA Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Veigas, J.M.; Narayan, M.S.; Laxman, P.M.; Neelwarne, B. Chemical nature, stability and bioefficacies of anthocyanins from fruit peel of syzygium cumini skeels. Food Chem. 2007, 105, 619–627. [Google Scholar] [CrossRef]
- Benherlal, P.S.; Arumughan, C. Chemical composition and in vitro antioxidant studies on syzygium cumini fruit. J. Sci. Food Agric. 2007, 87, 2560–2569. [Google Scholar] [CrossRef] [PubMed]
- Sagrawat, H.; Mann, A.; Kharya, M. Pharmacological potential of Eugenia jambolana: A review. Pharmacogn Mag. 2006, 2, 96–105. [Google Scholar]
- Helmstädter, A. Syzygium cumini (L.) skeels (Myrtaceae) against diabetes––125 years of research. Die Pharmazie 2008, 63, 91–101. [Google Scholar] [PubMed]
- Ayyanar, M.; Subash-Babu, P. Syzygium cumini (L.) skeels: A review of its phytochemical constituents and traditional uses. Asian Pac. J. Trop. Biomed. 2012, 2, 240–246. [Google Scholar] [CrossRef]
- De Brito, E.S.; de Araujo, M.C.P.; Alves, R.E.; Carkeet, C.; Clevidence, B.A.; Novotny, J.A. Anthocyanins present in selected tropical fruits: Acerola, jambolão, jussara, and guajiru. J. Agric. Food Chem. 2007, 55, 9389–9394. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Adams, L.S.; Chen, S.; Killian, C.; Ahmed, A.; Seeram, N.P. Eugenia jambolana lam. Berry extract inhibits growth and induces apoptosis of human breast cancer but not non-tumorigenic breast cells. J. Agric. Food Chem. 2009, 57, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Nazif, N.M. The anthocyanin components and cytotoxic activity of Syzygium cumini (L.) fruits growing in Egypt. Nat. Prod. Sci. 2007, 13, 135–139. [Google Scholar]
- Li, L.; Adams, L.S.; Chen, S.; Killian, C.; Ahmed, A.; Seeram, N.P. Eugenia jambolana lam. Berry extract inhibits growth and induces apoptosis of human breast cancer but not non-tumorigenic breast cells. J. Agric. Food Chem. 2009, 57, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.S.; Arnold, M.; Huang, Y.W.; Sardo, C.; Seguin, C.; Martin, E.; Huang, T.H.; Riedl, K.; Schwartz, S.; Frankel, W.; et al. Modulation of genetic and epigenetic biomarkers of colorectal cancer in humans by black raspberries: A phase I pilot study. Clin. Cancer Res. 2011, 17, 598–610. [Google Scholar] [CrossRef] [PubMed]
- Galati, G.; O'Brien, P.J. Potential toxicity of flavonoids and other dietary phenolics: Significance for their chemopreventive and anticancer properties. Free Radic. Biol. Med. 2004, 37, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Vanamala, J.; Radhakrishnan, S.; Reddivari, L.; Bhat, V.B.; Ptitsyn, A. Resveratrol suppresses human colon cancer cell proliferation and induces apoptosis via targeting the pentose phosphate and the talin-fak signaling pathways-a proteomic approach. Proteome Sci. 2011. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Vanamala, J.; Reddivari, L.; Radhakrishnan, S.; Tarver, C. Resveratrol suppresses IGF-1 induced human colon cancer cell proliferation and elevates apoptosis via suppression of IGF-1R/Wnt and activation of p53 signaling pathways. BMC Cancer 2010. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.M.; Afaq, F.; Khan, N.; Mukhtar, H. Delphinidin, an anthocyanidin in pigmented fruits and vegetables, induces apoptosis and cell cycle arrest in human colon cancer HCT116 cells. Mol. Carcinog. 2009, 48, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Zhao, C.; Schoene, N.; Guisti, M.M.; Moyer, M.P.; Magnuson, B.A. Anthocyanin-rich extract from Aronia meloncarpa E. Induces a cell cycle block in colon cancer but not normal colonic cells. Nutr. Cancer 2003, 46, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Akoh, C.C.; Fischer, J.; Krewer, G. Effect of anthocyanin fractions from selected cultivars of Georgia-grown blueberries on apoptosis and phase II enzymes. J. Agric. Food Chem. 2007, 55, 3180–3185. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Kanwar, S.S.; Patel, B.B.; Nautiyal, J.; Sarkar, F.H.; Majumdar, A.P. Elimination of colon cancer stem-like cells by the combination of curcumin and folfox. Transl. Oncol. 2009, 2, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Todaro, M.; Alea, M.P.; Di Stefano, A.B.; Cammareri, P.; Vermeulen, L.; Iovino, F.; Tripodo, C.; Russo, A.; Gulotta, G.; Medema, J.P. Colon cancer stem cells dictate tumor growth and resist cell death by production of interleukin-4. Cell Stem Cell 2007, 1, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, S.; Reddivari, L.; Sclafani, R.; Das, U.N.; Vanamala, J. Resveratrol potentiates grape seed extract induced human colon cancer cell apoptosis. Front. Biosci. 2011, 3, 1509–1523. [Google Scholar] [CrossRef]
- Massey, A.R.; Reddivari, L.; Vanamala, J. The dermal layer of sweet sorghum (Sorghum bicolor) stalk, a byproduct of biofuel production and source of unique 3-deoxyanthocyanidins, has more antiproliferative and proapoptotic activity than the pith in p53 variants of HCT116 and colon cancer stem cells. J. Agric. Food Chem. 2014, 62, 3150–3159. [Google Scholar]
- Apel, A.; Herr, I.; Schwarz, H.; Rodemann, H.P.; Mayer, A. Blocked autophagy sensitizes resistant carcinoma cells to radiation therapy. Cancer Res. 2008, 68, 1485–1494. [Google Scholar] [CrossRef] [PubMed]
- Matés, J.M.; Segura, J.A.; Alonso, F.J.; Márquez, J. Anticancer antioxidant regulatory functions of phytochemicals. Curr. Med. Chem. 2011, 18, 2315–2338. [Google Scholar] [CrossRef] [PubMed]
- Charepalli, V.; Reddivari, L.; Radhakrishnan, S.; Vadde, R.; Agarwal, R.; Vanamala, K.P.J. Anthocyanin-containing purple-fleshed potatoes suppress colon tumorigenesis via elimination of colon cancer stem cells. J. Nutr. Biochem. 2015, 26, 1641–1649. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Giusti, M.M.; Malik, M.; Moyer, M.P.; Magnuson, B.A. Effects of commercial anthocyanin-rich extracts on colonic cancer and nontumorigenic colonic cell growth. J. Agric. Food Chem. 2004, 52, 6122–6128. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130, 2073S–2085S. [Google Scholar] [PubMed]
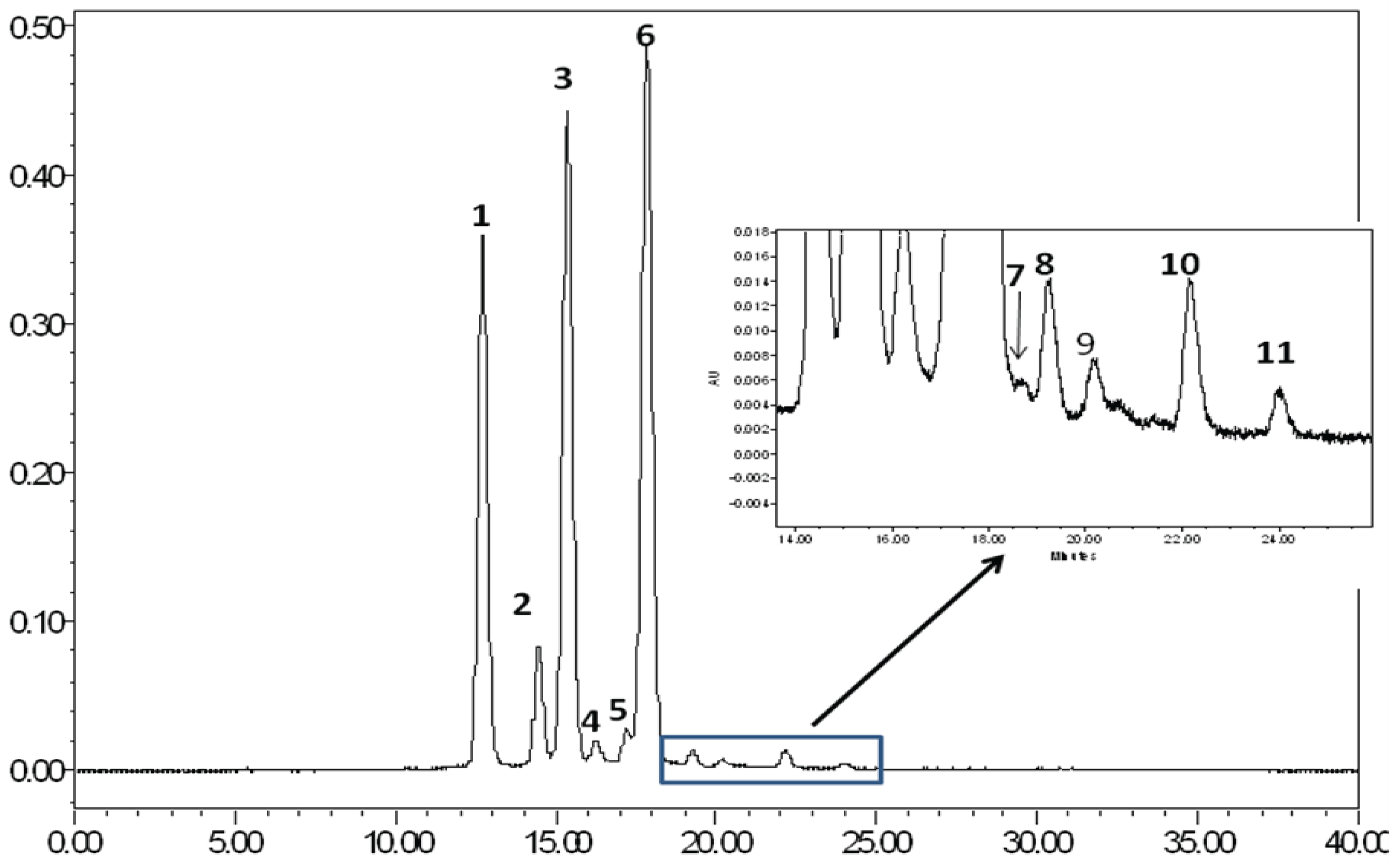
| Peak | Anthocyanin | RT | [M]+ | ESI-PI(m/z) |
|---|---|---|---|---|
| 1 | Delphinidin-3,5-diglucoside | 12.68 | 627 | 465 [M-162]+; 303 [M-162-162]+ |
| 2 | Cyanidin-3,5-diglucoside | 14.44 | 611 | 449 [M-162]+; 287 [M-162-162]+ |
| 3 | Petunidin-3,5-diglucoside | 15.33 | 641 | 479 [M-162]+; 317 [M-162-162]+ |
| 4 | Delphinidin-3-glucoside | 16.22 | 449 | 287 [M-162]+ |
| 5 | Peonidin-3,5-diglucoside | 17.17 | 625 | 463 [M-162]+; 301 [M-162-162]+ |
| 6 | Malvidin-3,5-diglucoside | 17.83 | 655 | 493[M-162]+; 331 [M-162-162]+ |
| 7 | Cyanidin-3-glucoside | 18.74 | 449 | 287 [M-162]+ |
| 8 | Petunidin-3-glucoside | 19.26 | 479 | 317 [M-162]+ |
| 9 | Unknown | 20.28 | - | - |
| 10 | Malvidin-3-glucoside | 22.14 | 493 | 331 [M-162]+ |
| 11 | Unknown | 24.02 | - | - |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Charepalli, V.; Reddivari, L.; Vadde, R.; Walia, S.; Radhakrishnan, S.; Vanamala, J.K.P. Eugenia jambolana (Java Plum) Fruit Extract Exhibits Anti-Cancer Activity against Early Stage Human HCT-116 Colon Cancer Cells and Colon Cancer Stem Cells. Cancers 2016, 8, 29. https://doi.org/10.3390/cancers8030029
Charepalli V, Reddivari L, Vadde R, Walia S, Radhakrishnan S, Vanamala JKP. Eugenia jambolana (Java Plum) Fruit Extract Exhibits Anti-Cancer Activity against Early Stage Human HCT-116 Colon Cancer Cells and Colon Cancer Stem Cells. Cancers. 2016; 8(3):29. https://doi.org/10.3390/cancers8030029
Chicago/Turabian StyleCharepalli, Venkata, Lavanya Reddivari, Ramakrishna Vadde, Suresh Walia, Sridhar Radhakrishnan, and Jairam K. P Vanamala. 2016. "Eugenia jambolana (Java Plum) Fruit Extract Exhibits Anti-Cancer Activity against Early Stage Human HCT-116 Colon Cancer Cells and Colon Cancer Stem Cells" Cancers 8, no. 3: 29. https://doi.org/10.3390/cancers8030029
APA StyleCharepalli, V., Reddivari, L., Vadde, R., Walia, S., Radhakrishnan, S., & Vanamala, J. K. P. (2016). Eugenia jambolana (Java Plum) Fruit Extract Exhibits Anti-Cancer Activity against Early Stage Human HCT-116 Colon Cancer Cells and Colon Cancer Stem Cells. Cancers, 8(3), 29. https://doi.org/10.3390/cancers8030029





