Abstract
The sonic hedgehog (Shh) signaling pathway is a major regulator of cell differentiation, cell proliferation, and tissue polarity. Aberrant activation of the Shh pathway has been shown in a variety of human cancers, including, basal cell carcinoma, malignant gliomas, medulloblastoma, leukemias, and cancers of the breast, lung, pancreas, and prostate. Tumorigenesis, tumor progression and therapeutic response have all been shown to be impacted by the Shh signaling pathway. Downstream effectors of the Shh pathway include smoothened (SMO) and glioma-associated oncogene homolog (GLI) family of zinc finger transcription factors. Both are regarded as important targets for cancer therapeutics. While most efforts have been devoted towards pharmacologically targeting SMO, developing GLI-targeted approach has its merit because of the fact that GLI proteins can be activated by both Shh ligand-dependent and -independent mechanisms. To date, two SMO inhibitors (LDE225/Sonidegib and GDC-0449/Vismodegib) have received FDA approval for treating basal cell carcinoma while many clinical trials are being conducted to evaluate the efficacy of this exciting class of targeted therapy in a variety of cancers. In this review, we provide an overview of the biology of the Shh pathway and then detail the current landscape of the Shh-SMO-GLI pathway inhibitors including those in preclinical studies and clinical trials.
1. Introduction
The Hedgehog (HH) gene was discovered in 1980 by Nusslein-Volhard and Wieschaus through genetic analysis of the fruit fly Drosophila melanogaster [1]. In the early 1990s, three HH gene homologs were discovered in vertebrates; Sonic Hedgehog (SHH), Indian Hedgehog (IHH), and Desert Hedgehog (DHH) [2,3,4]. DHH and IHH have been shown to play important roles in normal tissue development, including pancreas and testis organogenesis and bone formation [5,6,7,8]. Shh is the most potent of these ligands and is the most widely expressed in adult tissues [9,10]. Shh signaling plays an essential role in embryonic development and is critical for maintenance of tissue polarity. It has been shown that Shh is the dominant oncogenic HH ligand, as ectopic expression of Shh was sufficient to induce basal cell carcinoma in mice [11,12]. The Shh pathway is tightly regulated in most adult tissues but hyperactivation of this pathway is found in many solid tumors [13,14,15,16,17,18,19,20]. Aberrant Shh signaling has been implicated in many human cancers that account for up to 25% of human cancer deaths [21]. Greater understanding of the role of Shh signaling in human cancers has clearly indicated the need for development of anti-cancer therapies targeting the Shh pathway.
1.1. Shh Signaling Pathway Overview
The canonical HH pathway contains several key components, including HH glycoproteins Shh, IHH, and DHH [22]. Upon secretion, Shh glycoproteins bind and inactivate the 12-transmembrane protein Patched1 (PTCH1), which normally inhibits the activity of the 7-transmembrane protein Smoothened (SMO). In the presence of Shh ligand, PTCH1 inhibition of SMO at the primary cilium is abrogated resulting in the nuclear localization of glioma-associated (GLI) transcription factors, which are the terminal effectors of the Shh signaling (Figure 1). PTCH2 receptor shares approximately 54% homology with PTCH1, yet its expression pattern and signaling role in tissue vary significantly from PTCH1. PTCH2 is highly expressed in spermatocytes and helps mediate DHH activity in germ cell development [23]. It has also been shown that in the absence of Shh ligand binding, PTCH2 has a decreased ability to inhibit SMO [24]. In the absence of ligand, Suppressor of Fused (SUFU) negatively regulates the pathway by directly binding to GLI transcription factors and anchoring them in the cytoplasm preventing the activation of GLI target genes [25,26,27]. Cytoplasmic sequestration of GLI transcription factors by SUFU facilitates processing and degradation of GLI proteins, therefore inhibiting Shh pathway signaling [26]. SUFU has also been shown to form a repressor complex leading to interaction with DNA-bound GLI1 and suppression of GLI1-induced gene expression [28]. In vertebrates, there are three GLI transcription factors (GLI1, GLI12 and GLI3). GLI1 is the only full-length transcriptional activator whereas GLI2 and GLI3 act as either a positive or negative regulators as determined by posttranscriptional and posttranslational processing [29,30]. In response to Shh ligand binding, GLI2 accumulates in the primary cilium and drives transcriptional activation, overcoming negative regulation by GLI3 [31]. In addition to regulation by SUFU, GLI1 is also regulated by the kinase Dyrk1. Dyrk1 can potentiate GLI1 activity by phosphorylation at multiple serine/threonine sites that has been shown to induce nuclear accumulation and GLI1-mediated transcription [32]. GLI transcription factors can activate target genes that includes targets involved in HH pathway feedback (e.g., GLI1, PTCH1), proliferation (e.g., Cyclin-D1, MYC), apoptosis (e.g., Bcl-2), angiogenesis (e.g., ANG1/2), epithelial-to-mesenchymal transition (e.g., SNAIL), and stem cell self-renewal (e.g., NANOG, SOX2) [33,34,35].
In addition to the classical (canonical) signaling axis, there are also non-classical (non-canonical) pathways related to Shh signaling. Non-canonical Shh signaling refers to either: (1) activation of signaling from PTCH1/SMO but independent of GLI transcription factors; or (2) activation of GLI transcription factors independent of Shh ligand or PTCH1/SMO. The latter is better studied and multiple pathways have been identified, mostly oncogenic, that can increase GLI activity. GLI transcription factors have been shown to be positively regulated by K-Ras, TGF-β, PI3K-AKT, and PKC-α [36,37,38,39,40,41]. K-Ras, in particular, seems to be a pathway capable of activating GLI1 independent of the Shh pathway as knockdown of SUFU does not affect K-Ras-induced GLI1 [38,40]. Additionally, the GLI proteins have been shown to be negatively regulated by p53, PKA, and PKC-δ [42,43,44,45]. GLI1 transcriptional activity has also been shown to be reduced with p53 overexpression and enhanced with p53 knockdown [44]. Furthermore, p53 has been shown to interact with TAF9 leading to suppression of GLI1 activity [45]. PKA regulation of GLI1 is very specific as PKA directly phosphorylates Thr374 of GLI1, which promotes cytoplasmic localization and reduced activity of GLI1 [43].
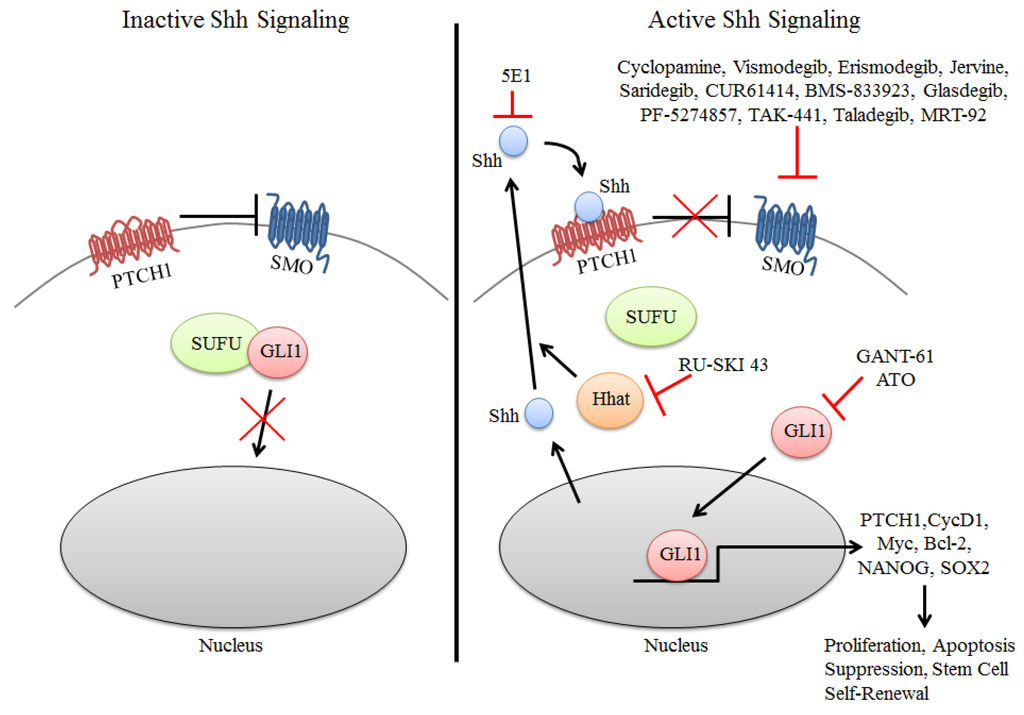
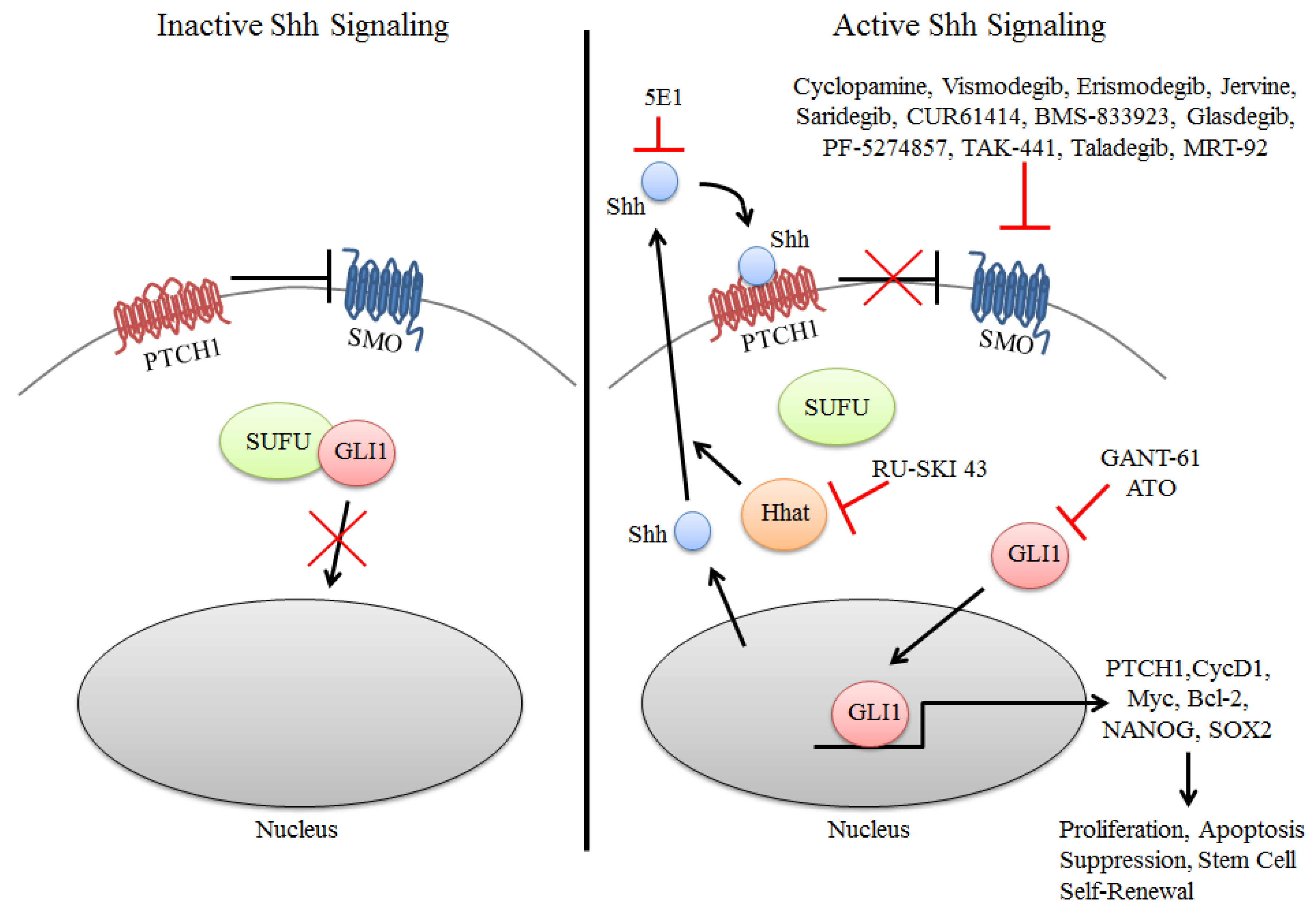
Figure 1.
Inhibition of components of the Shh Pathway in cancer. Inactive signaling (left) occurs in the absence of Shh ligand wherein PTCH1 inhibits SMO resulting in GLI1 sequestration in the cytoplasm by SUFU. In the presence of Shh (right), PTCH1 suppression of SMO is abrogated resulting in the nuclear accumulation of GLI1 and activation of target genes that promote several oncogenic properties to tumor cells. Inhibition of the Shh pathway is primarily directed at inhibition of SMO and GLI1, with many of these compounds in clinical trials for solid cancers. More recently attempts have been made to inhibit the Shh signaling pathway by using the monoclonal antibody 5E1 or the SHHat inhibitor RU-SKI 43 to inhibit SHH directly. SHHat is an O-acyltransferase that catalyzes the palmitoylation of Shh, which is critical to its function.
Figure 1.
Inhibition of components of the Shh Pathway in cancer. Inactive signaling (left) occurs in the absence of Shh ligand wherein PTCH1 inhibits SMO resulting in GLI1 sequestration in the cytoplasm by SUFU. In the presence of Shh (right), PTCH1 suppression of SMO is abrogated resulting in the nuclear accumulation of GLI1 and activation of target genes that promote several oncogenic properties to tumor cells. Inhibition of the Shh pathway is primarily directed at inhibition of SMO and GLI1, with many of these compounds in clinical trials for solid cancers. More recently attempts have been made to inhibit the Shh signaling pathway by using the monoclonal antibody 5E1 or the SHHat inhibitor RU-SKI 43 to inhibit SHH directly. SHHat is an O-acyltransferase that catalyzes the palmitoylation of Shh, which is critical to its function.
1.2. Shh Signaling in Cancer
The role of dysregulated Shh signaling in cancer was first characterized by studies of basal cell nevus syndrome. Basal cell nevus syndrome, also known as Gorlin syndrome, is an autosomal dominant disorder that presents itself with craniofacial and skeletal abnormalities and a notably increased risk of advanced basal cell carcinoma and medulloblastoma [46,47]. The major breakthrough in the understanding of Shh signaling in cancer came from the discovery that mutations in PTCH1 were the cause of Gorlin syndrome suggesting that aberrant Shh pathway activity was responsible for the development of these cancers [48,49]. These findings were reinforced by the discovery of mutations of PTCH1, SMO, and SUFU in a large percentage of spontaneous basal cell carcinomas and medulloblastomas [50,51]. The tumor suppressor role of PTCH1 has been further studied in transgenic mouse models that are heterozygous for a PTCH1 null mutation. These mice showed the critical features of basal cell nevus syndrome, such as development of basal cell carcinomas, medulloblastomas, and rhabdomyosarcomas [48,49,52].
Abnormal Shh signaling is a hallmark of many cancers. It is now understood that somatic mutations in upstream pathway elements such as SMO and PTCH1 do not account for all of the dysregulated Shh signaling observed in tumors. It has been observed in multiple tumor types that Shh pathway dysregulation can also be induced in a ligand-dependent manner through enhanced Shh autocrine or paracrine signaling. This has been reported in a wide variety of cancers including pancreatic, colorectal, metastatic prostate carcinomas, and gliomas [19,53,54,55,56]. Furthermore, studies in epithelial cancers have found that tumor cells secrete Shh ligand in a paracrine fashion that stimulates production of secondary growth factors by stromal cells that drive tumor angiogenesis, tumor cell proliferation, and survival [57].
1.2.1. Shh Signaling in Cancer Stem Cells
The Shh pathway has also been implicated in the regulation and maintenance of cancer stem cells (CSCs). In chronic myeloid leukemia and breast cancer, studies have found that Shh signaling is essential for maintenance of cancer stem cells and inhibition of the Shh pathway results in decreased stem cell propagation and renewal [14,58,59]. Recent studies have suggested that Shh signaling in CSCs in solid tumors is involved in metastatic progression and drives epithelial-mesenchymal transition of CSCs in pancreatic and colorectal cancers [56,60,61], providing a link between Shh signaling in regulation of normal stem cells and its role in cancer stem cell maintenance.
1.2.2. Novel GLI1 Isoform (tGLI1) in Cancer
Recently, a novel alternative splice variant of GLI1 was discovered in our laboratory, termed truncated GLI1 (tGLI1) [62]. tGLI1 is an alternatively spliced, shorter variant of GLI1 that contains an in-frame deletion of 41 amino acids corresponding to entire exon 3 and part of exon 4. Evidence to date indicates that tGLI1 is expressed at high levels in glioblastoma and breast cancer but undetectable or expressed at a low level in normal tissues. We have observed that tGLI1 promotes cell migration, invasion, and angiogenesis by upregulating CD24, HPA1, VEGF-C, and TEM-7 in glioblastoma and breast cancer [54,63,64]. We have also shown that tGLI1 upregulates VEGF-A and VEGFR-2, leading to a distinct autocrine loop that promotes angiogenesis and cell growth in breast cancer [65]. These studies have indicated tGLI1 is a gain-of-function transcription factor compared to GLI1 as it has shown the ability to upregulate several genes that are not GLI1 target genes. Overexpression of tGLI1 results in larger tumor growth and greater tumor angiogenesis compared to tumors overexpressing GLI1 [54,62,63,64]. We have observed tGLI1 expression in a high percentage of breast (78%) and glioblastoma (47%) tumors. Interestingly, tGLI1 is exclusively expressed in cancer cells and tissues as we have not detected tGLI1 in healthy cells or healthy human tissues [66]. Considering the potent oncogenic effects of tGLI1 and its unique expression pattern in cancer cells and tissues, tGLI1 is an optimal therapeutic target although no targeted inhibitors for tGli1 have been developed to date. The potential role tGLI1 plays in the lack of efficacy of inhibitors directed toward the Shh pathway, GLI1-targeted inhibitors in particular, has not been addressed.
Development of therapeutics for the canonical Shh signaling pathway has primarily focused on targeting SMO and GLI1. Natural and synthetic antagonists have been developed for both SMO and GLI1 with many having undergone clinical trials with varying degrees of success. SMO inhibition was first characterized through binding studies of cyclopamine, a natural steroidal alkaloid derived from Veratrum californicum [28,67,68]. Derivatives of cyclopamine have been developed in the hopes of increasing specificity and pharmacological potency. A setback in targeting SMO has been the observation of spontaneous mutations can develop as a response to some SMO inhibitors [69]. GLI1 has also been singled out as a therapeutic target as it is the most characterized GLI transcription factor associated with activation of Shh target genes although the library of GLI1 antagonists is not as extensive as for SMO. Here we review the current and rapidly expanding field of natural and synthetic SMO and GLI small-molecule inhibitors, attempting to provide a basis for future studies and development. We also highlight Shh pathway small-molecule inhibitors currently in preclinical studies and clinical trials.
2. SMO and GLI Inhibitors
2.1. SMO Inhibitors
SMO has been the primary target for the development of Shh-pathway inhibitors. SMO inhibition prevents the downstream activation of GLI transcription factors, leading to suppression of those genes associated with cancer growth and progression. Table 1 indicates the compounds targeting components with the Shh pathway and the clinical status of these compounds.

Table 1.
Small molecule Shh pathway inhibitors in active clinical trials as of January 2016. Data sourced from www.clinicaltrials.gov.
| Compound | Organization | Target | Cancer Type | Clinical Trial | NCT Trial |
|---|---|---|---|---|---|
| GDC-0449 (Vismodegib/Erivedge) | Roche/Genentech/Curis | SMO | Basal Cell Carcinoma - - - - - - - Advanced/Metastatic Basal Cell Carcinoma Basal Cell Nevus Syndrome Medulloblastoma - Recurrent Medulloblastoma Metastatic Castration-Resistant Prostate Cancer Chondrosarcoma Advanced Pancreatic Cancer Metastatic Pancreatic Cancer Myelofibrosis Metastatic Gastric & Esophageal Cancer Advanced Prostate Adenocarcinoma Small-Cell Lung Cancer Keratocystic Odontogenic Tumor Advanced Solid Tumors Acute Myeloid Leukemia Intracranial Meningioma | Phase II | NCT01835626 |
| Phase I | NCT02639117 | ||||
| Phase II | NCT02067104 | ||||
| Phase 0 | NCT01631331 | ||||
| Phase II | NCT01815840 | ||||
| Phase II | NCT01700049 | ||||
| Phase II | NCT01898598 | ||||
| Phase IV | NCT02436408 | ||||
| Phase II | NCT01367665 | ||||
| - | - | ||||
| Phase II | NCT01556009 | ||||
| Phase II | NCT00957229 | ||||
| Phase I/II | NCT01601184 | ||||
| Phase II | NCT01878617 | ||||
| Phase II | NCT01239316 | ||||
| Phase II | NCT00939484 | ||||
| Phase 0 | NCT02115828 | ||||
| - | - | ||||
| - | - | ||||
| Phase II | NCT01267955 | ||||
| Phase II | NCT01195415 | ||||
| - | - | ||||
| Phase I | NCT00878163 | ||||
| Phase II | NCT01088815 | ||||
| Phase I | NCT02593760 | ||||
| Phase II | NCT00982592 | ||||
| - | - | ||||
| Phase I/II | NCT01163084 | ||||
| - | - | ||||
| Phase II | NCT00887159 | ||||
| Phase II | NCT02366312 | ||||
| - | - | ||||
| Phase II | NCT02091141 | ||||
| Phase II | NCT02073838 | ||||
| Phase II | NCT02523014 | ||||
| LDE225 (Erismodegib/Sonidegib/Odomzo®) | Novartis | SMO | Prostate Cancer Castration-Resistant Prostate Cancer Pancreatic Adenocarcinoma Advanced/Metastatic Pancreatic Cancer Refractory Multiple Myeloma Recurrent Ovarian Cancer Triple-Negative Breast Cancer Myeloid Malignancies Basal Cell Carcinoma Advanced/Metastatic Basal Cell Carcinoma - Advanced Solid Tumors - | Phase I | NCT02111187 |
| Phase I | NCT02182622 | ||||
| - | - | ||||
| Phase I/II | NCT01431794 | ||||
| - | - | ||||
| Phase I/II | NCT02358161 | ||||
| - | - | ||||
| Phase II | NCT02086552 | ||||
| - | - | ||||
| Phase I/II | NCT02195973 | ||||
| Phase I | NCT02027376 | ||||
| - | - | ||||
| Phase I | NCT02129101 | ||||
| Phase II | NCT00961896 | ||||
| Phase II | NCT00961896 | ||||
| Phase 0 | NCT02303041 | ||||
| Phase II | NCT01327053 | ||||
| Phase I | NCT01769768 | ||||
| Phase I | NCT01954355 | ||||
| LDE225 (Erismodegib/Sonidegib/Odomzo®) | Novartis | SMO | Advanced Gastroesophageal Adenocarcinoma Small Cell Lung Cancer Myelofibrosis Advanced/Metastatic Hepatocellular Carcinoma Relapsed Medulloblastoma | Phase I | NCT02138929 |
| - | - | ||||
| - | - | ||||
| Phase I | NCT01579929 | ||||
| Phase I/II | NCT01787552 | ||||
| Phase I | NCT02151864 | ||||
| - | - | ||||
| - | - | ||||
| Phase II | NCT01708174 | ||||
| - | - | ||||
| BMS-833923/XL139 | Bristol Myers Squibb/Exelixis | SMO | Basal Cell Nevus Syndrome Chronic Myeloid Leukemia | Phase I | NCT02100371 |
| - | - | ||||
| Phase II | NCT01357655 | ||||
| - | - | ||||
| PF-04449913 (Glasdegib) | Pfizer | SMO | Myelofibrosis Chronic Myelomonocytic Leukemia Myelodysplastic Syndrome | Phase II | NCT02226172 |
| Phase II | NCT01842646 | ||||
| - | - | ||||
| - | - | ||||
| Phase II | NCT01842646 | ||||
| - | - | ||||
| LY2940680 (Taladegib) | Ignyta | SMO | Esophageal Cancer Advanced Solid Tumors | Phase I/II | NCT02530437 |
| Phase I | NCT01919398 | ||||
| - | - | ||||
| IPI-926 (Saridegib) | Infinity | SMO | Advanced Pancreatic Adenocarcinoma | Phase I | NCT01383538 |
| - | - | ||||
| Arsenic Trioxide (ATO) | - | GLI1 | Non-Small-Cell Lung Cancer Small Cell Lung Cancer Acute Myeloid Leukemia Hepatocellular Carcinoma - Malignant Glioma Myelofibrosis Acute Promyelocytic Leukemia - - - - - - - - Chronic Myelogenous Leukemia Acute Myeloid Leukemia Myelodysplastic Syndrome Chronic Myelomonocytic Leukemia | Phase I | NCT02066870 |
| - | - | ||||
| Phase II | NCT01470248 | ||||
| Phase II | NCT01835288 | ||||
| - | - | ||||
| - | - | ||||
| - | - | ||||
| Phase II | NCT02018757 | ||||
| Phase I/II | NCT00275067 | ||||
| Phase I | NCT01014546 | ||||
| Phase III | NCT02339740 | ||||
| Phase II | NCT01404949 | ||||
| Phase II | NCT01409161 | ||||
| Phase III | NCT00378365 | ||||
| Phase IV | NCT01987297 | ||||
| Phase III | NCT00866918 | ||||
| Phase II | NCT00413166 | ||||
| Phase II | NCT00551460 | ||||
| Phase III | NCT00482833 | ||||
| Phase IV | NCT02200978 | ||||
| Phase I | NCT01397734 | ||||
| - | - | ||||
| Phase II | NCT02188706 | ||||
| Phase II | NCT02190695 | ||||
| Phase II | NCT02188706 | ||||
| Phase II | NCT02190695 | ||||
| Phase II | NCT02190695 | ||||
| - | - | ||||
| - | - |
2.1.1. Cyclopamine
Cyclopamine is an alkaloid isolated from V. californicum that showed strong potential to bind to SMO and inhibit the Shh signaling pathway [67,68]. Binding studies using a fluorescent cyclopamine derivative suggested that cyclopamine binds to the heptahelical transmembrane domain of SMO, preventing the necessary conformation shift to activate SMO [70,71]. Cyclopamine has been shown to inhibit tumor growth and proliferation in many mouse xenograft models, including human orthotopic glioma, melanoma, colon, pancreatic, and prostate cancers [55,56,60,72,73]. Although it significantly reduced tumor growth in vivo, cyclopamine never reached its therapeutic potential, as it caused many potent side effects, including weight loss, dehydration, and death in mouse models [74,75]. Several cyclopamine derivatives were developed to overcome solubility and stability issues and showed promise in in vitro studies, though in vivo evaluation of these compounds has not been conclusive [76,77,78].
2.1.2. GDC-0449 (Vismodegib/Erivedge)
Vismodegib is a second generation cyclopamine derivative created by Roche/Genentech/Curis that binds directly to SMO to prevent GLI activation [79]. It was approved by the FDA in January 2012 as the first Shh pathway drug approved for treating any cancer. Vismodegib is currently being used to treat adults with metastatic basal cell carcinoma (BCC), or patients with recurrent, locally advanced BCC who are not candidates for surgery or radiation therapy [80]. In a subsequent clinical trial for metastatic BCC, vismodegib treatment resulted in tumor regression; however, after three months, a novel SMO mutation was discovered in the tumor tissue and the treatment progress ceased [81,82]. Recently, several other novel SMO mutations have been uncovered that may also play a role in vismodegib resistance [83]. Tumor stroma interactions may also play a role in the ineffectiveness of Shh pathway inhibition. Preclinical data showed that SMO inhibition could deplete the tumor stroma; however, a Phase Ib/II trial comparing gemcitabine in combination with vismodegib to gemcitabine with placebo for pancreatic ductal adenocarcinoma (PDAC) was terminated early because it did not meet the criteria for futility [84,85]. A follow-up study showed that while SMO inhibition in a genetic mouse model of PDAC resulted in decreased tumor stroma, the tumor itself was more aggressive, more highly vascularized, and more poorly differentiated, resulting in lower survival rates than controls [86]. It was later shown that the preclinical data for the original vismodegib trial was generated from mice that had developed large PDAC tumors, which did not allow for evaluation of long-term treatment effects, due to the short survival time of the mice [85]. Currently, vismodegib, as a monotherapy and in combination with other chemotherapeutics, is being studied in a long list of clinical trials in a wide array of cancers, including medulloblastoma, small cell lung cancer, metastatic pancreatic cancer, metastatic prostate cancer, intracranial meningioma, recurrent glioblastoma, and acute myeloid leukemia (Table 1).
2.1.3. LDE-225 (Erismodegib/Sonidegib/Odomzo)
Erismodegib (also known as sonidegib and Odomzo®, East Hanover, NJ, USA) is an orally bioavailable SMO antagonist created by Novartis that has been shown to induce cell cycle arrest and apoptosis in a variety of cancer cell lines [87]. It has been effective in decreasing the epithelial-mesenchymal transition and invasive potential of several cancer types, including glioblastoma, prostate cancer, and renal cell carcinoma, implying that it can affect both tumor epithelial cells and cancer stem cells [88,89,90]. In July 2015, it was approved by the FDA to treat adult patients with locally advanced BCC that has recurred following surgery or radiation therapy, or those who are not candidates for surgery or radiation therapy. Upon this approval, LDE-225 became the second HH pathway inhibitor receiving FDA approval to treat cancer patients. Several Phase I/II trial for erismodegib as a monotherapy and in combination are underway, treating both solid tumors and hematological malignancies (Table 1).
2.1.4. IPI-926 (Saridegib)
Saridegib came about as a result of chemical modification of the base structure of cyclopamine, and was determined to have inhibitory activity on the HH signaling pathway by its ability to suppress tumor growth of B837Tx medulloblastoma allografts in mice. Daily administration of 40 mg/kg led to tumor regression, with no recurrence after 30 days of treatment [91]. Saridegib has also been shown to decrease tumor growth in lung and chondrosarcoma xenografts [92,93]. Interestingly, saridegib did not affect epithelial tumor cells; however, when co-administered with gemcitabine in a mouse model of pancreatic ductal carcinoma, a marked increase in gemcitabine tumor delivery was recorded as a result of increased angiogenesis [85]. One Phase I clinical trial with saridegib is currently underway (Table 1).
2.1.5. CUR61414
High throughput screening of over 100,000 compounds on a GLI-luciferase reporter assay in mouse C3H10T1/2 cells yielded CUR6414. Competition studies with cyclopamine and an SMO agonist showed that CUR6414 directly binds to SMO and prevents its activation [94]. In a PTCH+/− mouse embryonic BCC model, CUR6414 was able to prevent the formation and promote the regression of spontaneous and UV-induced basaloid lesions without affecting normal basal keratinocytes. The lesions also showed increased tumor cell apoptosis [95]. A Phase I clinical trial for treatment of basal-cell carcinomas was stopped due to the failure of the drug to penetrate human skin [96].
2.1.6. BMS-833923/XL139
BMS-833923, a Bristol-Myers Squibb/Exelexis product, was discovered to bind SMO by competition studies against a fluorescent-tagged cyclopamine, BODPIY-cyclopamine. It decreased GLI1 and PTCH1 mRNA expression in vitro and inhibited proliferation of several cancer cell lines [97,98,99]. In vivo studies showed that BMS-833923 reduced medulloblastoma, pancreatic carcinoma, and human cholangiocarcinoma xenograft growth in mice [97,98]. Phase I and Phase II trials including multiple tumor types are currently underway (Table 1).
2.1.7. PF-04449913 (Glasdegib)
Glasdegib is a newly marketed product from Pfizer that has been shown to bind and inhibit SMO [100]. Studies have shown that glasdegib abrogates leukemia-initiation potential and leukemia stem cell dormancy in chronic myeloid leukemia (CML) and acute myeloid leukemia (AML) cells, most likely due to downstream GLI2 inhibition [101,102]. Several Phase II trials for treating hematological malignancies are currently underway (Table 1).
2.1.8. PF-5274857
Pfizer concurrently discovered PF-5274857 to be a selective and potent SMO antagonist with the ability to penetrate the blood-brain barrier. In vitro, PF-5274857 was able to specifically bind to SMO and reduce GLI1 gene expression in MEF cells [103]. The ability of PF-5274857 to penetrate the blood-brain barrier made it a prime candidate for treatment of brain tumors and brain metastases driven by aberrant HH signaling. In PTCH+/− p53+/− medulloblastoma allograft mouse models and primary medulloblastoma mice, PF-5274857 inhibited tumor growth and reduced GLI1, GLI2, PTCH1, and PTCH2 gene expression with no significant side effects [103]. This makes it an ideal candidate for clinical trials in brain malignancies. No clinical trials to verify the effectiveness of PF-5274857 in humans are currently underway.
2.1.9. TAK-441
Ohashi et al. first reported TAK-441 as a highly potent and orally bioavailable SMO inhibitor in 2012. In NIH3T3 cells transfected with GLI-luciferase reporter construct, reporter activity was significantly reduced in response to TAK-441 treatment [104]. TAK-441 inhibited growth in PTCH+/− p53+/− medulloblastoma allografts models in mice and was shown to significantly reduce GLI1 mRNA expression in PAN-04 pancreatic tumor xenografts in mice [104,105]. Studies have also shown that in LNCaP prostate cancer xenografts mouse models, TAK-441 delayed castration-resistant progression of the disease and significantly suppressed GLI1, GLI2, and PTCH1 gene expression. Upon androgen starvation, LNCaP cells, an androgen-sensitive prostate cancer cell line, upregulate Shh expression, but are normally insensitive to SMO antagonists. These data suggest that TAK-441 was able to inhibit the Shh pathway by suppressing Shh paracrine signaling [106]. TAK-441 has also been suggested to be effective in inhibiting Vismodegib-resistant SMO D473H mutant. In several different assays, TAK-441 was shown to have equal affinity for wild-type and mutated SMO, while Vismodegib and cyclopamine showed reduced affinity for the D473H mutant suggesting that TAK-441 may be clinically relevant to treating Vismodegib-resistant, Shh pathway-driven cancers [107]. There are no currently active clinical trials looking at treating any type of cancer with TAK-441.
2.1.10. LY2940680 (Taladegib)
Taladegib is an experimental SMO antagonist developed by Lilly USA that binds directly to SMO and potently inhibits HH signaling in DAOY and C3H10T1/2 cells [108]. Wang et al. found that taladegib binds to the extracellular end of the transmembrane-helix bundle of SMO, inhibiting propagation of HH signaling [109]. In PTCH+/− p53−/− transgenic mice, taladegib reduced proliferation of spontaneously developed medulloblastoma and induced Caspase-3 activity signifying increased apoptosis. Taladegib also suppressed HH-mediated gene expression in subcutaneous xenograft tumor stroma and potently inhibited tumor growth. Importantly, taladegib was also shown to inhibit the activity of vismodegib-resistant SMO mutant (D473H) emphasizing its clinical potential [108]. Currently, taladegib is being tested in Phase I and Phase II trials for advanced solid tumors and esophageal cancers (Table 1).
2.1.11. MRT-92
MRT-92, developed by Hoch et al. at the Neuroscience Paris-Saclay Institute in the Centre National de la Recherche Scientifique, was very recently reported to have anti-SMO activity by blocking several overlapping sites of the SMO transmembrane domain [110]. It displayed subnanomolar antagonistic activity against SMO, blocking SAG (SMO agonist)-induced trafficking of SMO at the primary cilium and SAG-induced differentiation of C3H10T1/2 cells. Hoch et al. also found that MRT-92 maintained similar pharmacological characteristics when binding to vismodegib-resistant SMO mutant [110]. Together these data indicate that MRT-92 is a very strong candidate for clinical trials, as its binding site overlaps those of many previous generation SMO antagonists, allowing for stronger binding affinity at a lower drug concentration. MRT-92 is not currently being tested in any clinical trials.
2.1.12. Jervine
Jervine is a natural alkaloid isolated from the corn lily Veratrum californicum. It is a teratogen that inhibits the Shh-mediated response of chick neural plate cells and has also been shown to inhibit the growth of liver HepG2 and human colon carcinoma HT29 cell lines [67,111]. Jervine is able to inhibit HH signaling by binding SMO and preventing its conversion to an active state, leading to the accumulation of inactivated SMO in the primary cilium [112]. Clinical applications of jervine were never pursued due to its teratogenic side effects.
2.2. GLI Inhibitors
GLI transcription factors are the terminal effectors of the Shh-SMO signaling pathway and can also be activated independent of Shh and SMO by other important molecular pathways. Activation of GLI1 and GLI12 leads to upregulation of many pro-proliferative, pro-survival and pro-angiogenic genes, leading to tumor growth and therapeutic resistance [113]. Here we discuss inhibitors specifically targeting GLI transcription factors.
2.2.1. GANTs
GLI antagonists, or GANTs, were discovered at the National Cancer Institute in a GLI-luciferase reporter assay screen in HEK293 cells [114]. GANT-58 and GANT-61 were both discovered to inhibit GLI-mediated gene activation, though GANT-61 showed more specificity towards GLI proteins and more effectively reduced GLI1 and GLI2 DNA-binding ability. GANT-61 has shown potent inhibition of GLI1 and GLI2 in many cancer cell lines, including rhabdomyosarcoma, osteosarcoma, neuroblastoma, and ovarian cancer [115,116,117,118]. In a human prostate cancer xenograft model in mice, GANT-61 reduced tumor growth and proliferation and strongly reduced expression of PTCH1 mRNA [114]. No clinical trials are currently ongoing using GANT-61 to treat any type of cancer.
2.2.2. Arsenic Trioxide (ATO)
Arsenic trioxide is an FDA-approved inhibitor of GLI1 and GLI2 transcription factors. It has been approved for treatment of acute promyelocytic leukemia [119]. ATO directly binds to GLI1 and GLI2, inhibiting activity and decreasing expression of canonical Shh-GLI genes [119,120]. Kim et al. found that ATO reduces the stability of GLI2 transcription factor, preventing its accumulation in the primary cilium in response to Shh signaling [121]. ATO has also been shown to increase apoptosis, reduce tumor cell growth, and decrease expression of Shh target genes in vitro and in vivo in osteosarcoma, acute promyelocytic leukemia, malignant pleural mesothelioma, malignant rhabdosarcoma, prostate, and colon cancer cell lines and xenograft models [122,123,124,125,126,127]. Studies have shown that ATO also reduced the viability of pancreatic cancer stem cells and prostate cancer-initiating cells, underlining its effectiveness in killing of tumor epithelial cells and tumor-initiating cells [122,128]. Arsenic trioxide is currently in several clinical trials ranging from Phase I to Phase IV for both solid tumors and hematological malignancies (Table 1).
2.3. Shh Inhibitors
SHH is the most potent of the three Hedgehog ligands [10]. Consequently, targeting Shh has come under consideration to inhibit cancers with dysregulated Shh pathway activation. While these therapeutic approaches have not reached the clinic, they have been shown to successfully inhibit the Shh pathway.
2.3.1. RU-SKI 43
Following translation and synthesis of the Shh protein, the signal peptide is cleaved resulting in a 19-kDa product. During the final steps of SHH synthesis, the enzyme SHHat, a membrane-bound O-acyltransferase, catalyzes the attachment of palmitate to SHH [129,130]. Palmitoylation is critical to the potency of Shh as SHHat knockout mice showed developmental defects that mirrored Shh knockout mice [131]. As such, SHHat has become a target to inhibit the efficacy of SHH signaling. A screen for inhibitors of SHHat has resulted in RU-SKI 43, which inhibits SHHat activity and consequently inhibits Shh signaling in cultured cells [132]. Knockdown of SHHat or exposure to RU-SKI 43 reduced proliferation and anchorage-independent growth of breast cancer cells [133]. This approach was further shown to inhibit pancreatic tumor growth in animal models [134]. This approach has significant promise but has not yet reached human trials.
2.3.2. Shh Monoclonal Antibody 5E1
Inhibition of Shh with antibody targeting has also been attempted. The Shh monoclonal antibody 5E1 has been shown to inhibit growth of medulloblastoma in mouse models [135]. Furthermore, animals receiving 5E1 shown a reduced tumor proliferation index, increased tumor cell apoptosis, and had better survival compared to cyclopamine-treated mice [135]. The 5E1 antibody has also been shown to reduce growth of pancreatic tumors in mice [136]. Targeting Shh via monoclonal antibodies has also not reached human trials.
2.4. Therapeutic Targets Regulated by tGLI1
Clinical efficacy of SMO-targeted therapies has been mixed while GLI-targeted treatments are still in preclinical testing. Clearly, there is a need to deepen the biological understanding of tumors with aberrant SMO and GLI activities. To this end, our lab identified a truncated, gain-of-function isoform of the GLI1 transcription factor, tGLI1, that is only present in cancerous cells and is undetectable in normal cells [62], making it an ideal drug target. We have shown that tGLI1 is a gain-of-function GLI1 that has a higher propensity than GLI1 to induce aggressive cancer phenotypes in both breast cancer and glioblastoma, including, growth, invasion, migration, and angiogenesis [54,62,63,64,65]. These observations point to the need to pharmacologically target tGLI1.
Currently, there is no means to specifically inhibit tGLI1. However, several tGLI1 target genes can be targeted using already developed agents, including, CD24, VEGF-A, VEGFR2, HPA1 and TEM7. Studies have attempted to target CD24 with a monoclonal antibody (mAb) in several cancer models, including lung, ovarian, colorectal, and pancreatic cancer [137,138]. These studies showed decreased proliferation, motility, and tumorigenicity of cancer cell lines in mouse models, and one study showed specifically that combination therapy of CD24 mAb and gemcitabine strongly potentiated its anti-cancer efficacy in a mouse lung cancer model [138]. Several steps towards marketing a CD24 mAb as a cancer therapeutic have been made. A Phase I clinical trial (NCT02650895) studying the safety profile of a CD24 mAb in healthy adults was recently completed, though no results have been published, and a patent was filed for the use of a CD24 inhibitor to treat neoplastic conditions [139]. CD24 remains a viable target for targeting the HH-tGLI1 pathway and further refinement of immunotherapies could prove to be beneficial in treating tumors with tGLI1 expression.
Antiangiogenic therapies have been established as a new focus for cancer drug development, as increased tumor vascularization is indicative of aggressive cancer phenotypes and required for growth and metastasis [140]. Heparanase, also recognized as HPSE or HPA1, is another tGLI1 target gene that has been more extensively researched as an antiangiogenic, anti-cancer drug target [141]. Several classes of heparanase inhibitors have been studied, the most characterized of which include the heparan sulfate mimetics PI-88 and PG545. Researchers have shown in a myriad of cancer models that PI-88 and PG545 potently inhibit tumor angiogenesis by binding the active site of heparanase, and can be used in combination with other chemotherapeutics or as an adjuvant therapy [142,143,144,145]. PI-88 has been tested in multiple Phase I, II, and II trials spanning solid tumors and hematological malignancies, while PG545 is currently being tested in a Phase I clinical trial for advanced solid tumors (NCT02042781). PI-88 was fast-tracked for FDA approval in 2007 for treatment of post-resection hepatocellular carcinoma. Roneparstat, a heparin mimetic, has also been shown to have strong anti-cancer therapeutic effects and is currently in a Phase I trial for multiple myeloma (NCT01764880) [146].
TEM7, also termed PLXDC1, has been used as a prognostic marker for resectable gastric and colorectal cancers. As currently understood, TEM7 does not appear to be a viable therapeutic target, though it retains usefulness as a key prognostic marker for progression, invasion, and metastasis in several types of solid tumors [147,148]. There are no clinical trials currently testing experimental therapeutics targeting TEM7.
VEGF-A and VEGFRs are the most widely characterized targets for antiangiogenic therapeutics and these drugs have shown a varying range of effectiveness across tumor types [149]. There are currently two FDA-approved immunotherapies for targeting VEGF-A and VEGFR interactions. Bevacuzimab, trade name Avastin, is a humanized monoclonal antibody that inhibits VEGF-A by direct binding [150]. It is currently approved as either first-line monotherapy or in combination for metastatic colorectal cancer, non-small cell lung cancer, metastatic renal cell carcinoma, glioblastoma, and a variety of other advanced solid tumors [151]. In 2011, the FDA revoked its approval for the use of bevacuzimab in treating breast cancer, citing a lack of advantage in survival rates, no improvement in quality of life, and significant side effects [152]. Currently, over 500 active clinical trials are using bevacuzimab, both as a monotherapy and in combination, to treat a variety of solid tumors. Ziv-aflibercept, trade name ZALTRAP, is the other FDA-approved immunotherapy for targeting VEGF-A, though its mechanism of action is slightly different from bevacuzimab. ZALTRAP is a recombinant fusion protein that acts as a decoy VEGF receptor, binding to VEGF-A and preventing it from interacting with VEGFR-1 and VEGFR-2 [153]. It was approved by the FDA in 2012 for use in combination therapy with several other chemotherapeutics for treatment of metastatic colorectal cancer that has progressed following oxaliplatin treatment [154]. There are currently a multitude of active clinical trials testing ZALTRAP in a variety of solid tumors.
Our laboratory has shown that in addition to increasing VEGF-A expression, tGLI1 also increases expression of VEGF-C. The library of compounds for targeting VEGF-C is not as extensively developed as the one for VEGF-A, although there is one immunotherapy showing promising results in clinical trials. Circadian Technologies created a human monoclonal antibody named AGX-100 that is currently undergoing a first-in-human Phase I clinical trial both as a monotherapy and in combination with bevacuzimab for the treatment of metastatic solid tumors (NCT01514123) [155].
We have shown that tGLI1 is also able to upregulate transcription of VEGFR2, creating a powerful autocrine loop with VEGF-A [65]. VEGFR2 has long been a key target in the development of antiangiogenesis drugs as binding of VEGF-A to VEGFR2 accounts for the majority of the pro-angiogenic signals as observed in mouse models [156]. There are currently nine FDA-approved drugs for targeting VEGFR2, eight of which show multi-kinase inhibitory activity (Table 2).
In addition to targeting tGLI1 downstream target genes, inhibiting tGLI1 synthesis and targeting its transcription co-regulators could also inhibit tGLI1 leading to tumor cell kill. Unfortunately, the splicing events leading to tGLI1 synthesis are still unknown. Also unknown is whether tGLI1 requires transcription co-factors for its gain-of-function transcriptional functions. GLI1 has been shown to cooperate with transcription co-factors to activate target gene expression, including SMAD4, PCAF, and SAP18 [157,158]. Furthermore, whether tGLI1 is subjected to regulation by non-canonical pathways has not been investigated. Ideally, tGLI1 activity can be blocked by inhibiting the non-classical pathways that activate tGLI1 activity. Filling these knowledge gaps of tGLI1 will help with developing strategies to target tGLI1-driven tumors.

Table 2.
FDA-approved inhibitors targeting VEGFR2.
| Name | Company | Molecular Target(s) | Clinical Indications |
|---|---|---|---|
| Sorafenib (Nexavar) | Bayer Onyx Pharmaceuticals | VEGFR1-3 [159] PDGFRβ Raf | Unresectable Hepatocellular Carcinoma [160] Renal Cell Carcinoma [161] Radiation-Resistant Thyroid Cancer [162] |
| Sunitinib (Sutent) | Pfizer | VEGFR2 [163] PDGFRβ [164] Flt3 [164] | Renal Cell Carcinoma [165] Imatinib-Resistant Gastrointestinal Stroma Tumors [166] |
| Pazopanib (Votrient) | GlaxoSmithKline | VEGFR1-3 [167] PDGFR FGFR1-3 EGFR c-Kit | Metastatic Renal Cell Carcinoma [168] Metastatic Soft Tissue Sarcoma [169] |
| Regorafenib (Stivarga) | Bayer | VEGFR1-3 [170] PDGFRβ FGFR-1 KIT RET B-Raf | Metastatic Colorectal Cancer [171] Metastatic Gastrointestinal Stromal Tumors [172] |
| Vandetanib (Caprelsa) | Genzyme | VEGFR2-3 [173] EGFR RET | Metastatic Medullary Thyroid Cancer [174] |
| Cabozantinib (Cometriq) | Exelixis | VEGFR2 [175] c-MET RET | Medullary Thyroid Cancer [176] |
| Lenvatinib (Lenvima) | Eisai Inc | VEGFR1-3 [177] FGFR1-4 PDGFRα c-Kit RET | Radiation-Resistant Differentiated Thyroid Cancer [178] |
| Axitinib(Inlyta) | Pfizer | VEGFR1-3 [179] PDGFRα,β c-Kit | Renal Cell Carcinoma [180] |
| Ramucirumab (Cyramza) | Eli Lilly | VEGFR2 [181] | Gastric Adenocarcinoma [182] Gastro-Esophageal Junction Adenocarcinoma [182] Metastatic Non-Small-Cell Lung Cancer [183] |
3. Conclusions
The Shh signaling pathway is highly complex that has been shown to play important roles in promoting oncogenesis, tumor growth and progression, and tumor drug resistance. Therefore, several components of the Shh pathway (Shh, SMO, and GLI1/2) are viable therapeutic targets for anti-cancer therapies. Since SMO and GLI1/2 can be activated by Shh-independent stimuli, targeting them potentially target the transduction of a number of important molecular pathways. Single agent SMO inhibitors has already been found to be relatively effective in treating some malignancies in preclinical experiments and two of these orally active agents have received FDA approval for use in treating advanced or metastatic BCC. The sheer number of clinical trials using SMO inhibitors highlights the importance of pharmacological targeting of SMO in cancer. However, these inhibitors also showed limited efficacy against a number of cancers. Therefore, further advances must also be made to understand the mechanisms of resistance to small molecule SMO inhibitors and how to overcome them. Case reports from medulloblastoma patients treated with vismodegib indicate two specific mutations that occur in response to treatment, both presenting on the target SMO [69]. The SMO G497W mutation results in a partial blocking of the drug entry site while the SMO D437H mutation totally disrupts hydrogen bond stability in the binding site [184]. Further development of drugs such as MRT-92, which binds multiple sites on SMO and shows activity against the SMO D473H mutant, is crucial to overcoming treatment-induced resistance [110]. Given the facts that tGLI1 is expressed in a tumor-specific fashion and behaves as a more potent transcription regulator compared to GLI1, an important task is warranted to target tGLI1-driven malignancies directly or indirectly by targeting its upstream regulators and downstream targets.
Acknowledgments
The study is supported by NIH grants, R01 NS087169 (HWL), R01 NS087169-3S1 (HWL), P30 CA012197, and T32 CA079448 (RLC).
Author Contributions
T.K.R., R.L.C., and H.W.L. developed the concept and contributed to the writing of the manuscript. S.Q. and M.C. provided important clinical inputs to the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Nusslein-Volhard, C.; Wieschaus, E. Mutations affecting segment number and polarity in drosophila. Nature 1980, 287, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Echelard, Y.; Epstein, D.J.; St-Jacques, B.; Shen, L.; Mohler, J.; McMahon, J.A.; McMahon, A.P. Sonic hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell 1993, 75, 1417–1430. [Google Scholar] [CrossRef]
- Krauss, S.; Concordet, J.P.; Ingham, P.W. A functionally conserved homolog of the drosophila segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 1993, 75, 1431–1444. [Google Scholar] [CrossRef]
- Roelink, H.; Augsburger, A.; Heemskerk, J.; Korzh, V.; Norlin, S.; Ruiz i Altaba, A.; Tanabe, Y.; Placzek, M.; Edlund, T.; Jessell, T.M.; et al. Floor plate and motor neuron induction by vhh-1, a vertebrate homolog of hedgehog expressed by the notochord. Cell 1994, 76, 761–775. [Google Scholar] [CrossRef]
- Hebrok, M.; Kim, S.K.; St Jacques, B.; McMahon, A.P.; Melton, D.A. Regulation of pancreas development by hedgehog signaling. Development 2000, 127, 4905–4913. [Google Scholar] [PubMed]
- Kawahira, H.; Ma, N.H.; Tzanakakis, E.S.; McMahon, A.P.; Chuang, P.-T.; Hebrok, M. Combined activities of hedgehog signaling inhibitors regulate pancreas development. Development 2003, 130, 4871–4879. [Google Scholar] [CrossRef] [PubMed]
- St-Jacques, B.; Hammerschmidt, M.; McMahon, A.P. Indian hedgehog signaling regulates proliferation and differentiation of chondrocytes and is essential for bone formation. Genes Dev. 1999, 13, 2072–2086. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.H.; Whoriskey, W.; Capel, B. Desert Hedgehog/Patched 1 signaling specifies fetal Leydig cell fate in testis organogenesis. Genes Dev. 2002, 16, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Ingham, P.W.; McMahon, A.P. Hedgehog signaling in animal development: Paradigms and principles. Genes Dev. 2001, 15, 3059–3087. [Google Scholar] [CrossRef] [PubMed]
- Pathi, S.; Pagan-Westphal, S.; Baker, D.P.; Garber, E.A.; Rayhorn, P.; Bumcrot, D.; Tabin, C.J.; Blake Pepinsky, R.; Williams, K.P. Comparative biological responses to human sonic, Indian, and desert hedgehog. Mech. Dev. 2001, 106, 107–117. [Google Scholar] [CrossRef]
- Fan, H.; Oro, A.E.; Scott, M.P.; Khavari, P.A. Induction of basal cell carcinoma features in transgenic human skin expressing sonic hedgehog. Nat. Med. 1997, 3, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Oro, A.E.; Higgins, K.M.; Hu, Z.; Bonifas, J.M.; Epstein, E.H.; Scott, M.P. Basal cell carcinomas in mice overexpressing sonic hedgehog. Science 1997, 276, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Berman, D.M.; Karhadkar, S.S.; Hallahan, A.R.; Pritchard, J.I.; Eberhart, C.G.; Watkins, D.N.; Chen, J.K.; Cooper, M.K.; Taipale, J.; Olson, J.M.; et al. Medulloblastoma growth inhibition by hedgehog pathway blockade. Science 2002, 297, 1559–1561. [Google Scholar] [CrossRef] [PubMed]
- Dierks, C.; Beigi, R.; Guo, G.R.; Zirlik, K.; Stegert, M.R.; Manley, P.; Trussell, C.; Schmitt-Graeff, A.; Landwerlin, K.; Veelken, H.; et al. Expansion of Bcr-Abl-positive leukemic stem cells is dependent on Hedgehog pathway activation. Cancer Cell 2008, 14, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Kubo, M. Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res. 2004, 64, 6071–6074. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, P.; Hernández, A.M.; Stecca, B.; Kahler, A.J.; DeGueme, A.M.; Barrett, A.; Beyna, M.; Datta, M.W.; Datta, S.; Ruiz i Altaba, A. Inhibition of prostate cancer proliferation by interference with SONIC HEDGEHOG-GLI1 signaling. Proc. Natl. Acad. Sci. USA 2004, 101, 12561–12566. [Google Scholar] [CrossRef] [PubMed]
- Thayer, S.P.; di Magliano, M.P.; Heiser, P.W.; Nielsen, C.M.; Roberts, D.J.; Lauwers, G.Y.; Qi, Y.P.; Gysin, S.; Fernández-del Castillo, C.; Yajnik, V.; et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 2003, 425, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Tojo, M.; Kiyosawa, H.; Iwatsuki, K.; Kaneko, F. Expression of a sonic hedgehog signal transducer, hedgehog-interacting protein, by human basal cell carcinoma. Br. J. Dermatol. 2002, 146, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Watkins, D.N.; Berman, D.M.; Burkholder, S.G.; Wang, B.; Beachy, P.A.; Baylin, S.B. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 2003, 422, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Goetz, J.A.; Singh, S.; Ogden, S.K.; Petty, W.J.; Black, C.C.; Memoli, V.A.; Dmitrovsky, E.; Robbins, D.J. Frequent requirement of hedgehog signaling in non-small cell lung carcinoma. Oncogene 2006, 26, 1046–1055. [Google Scholar] [CrossRef] [PubMed]
- Lum, L.; Beachy, P.A. The hedgehog response network: Sensors, switches, and routers. Science 2004, 304, 1755–1759. [Google Scholar] [CrossRef] [PubMed]
- Varjosalo, M.; Taipale, J. Hedgehog: Functions and mechanisms. Genes Dev. 2008, 22, 2454–2472. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, D.; Stone, D.M.; Brush, J.; Ryan, A.; Armanini, M.; Frantz, G.; Rosenthal, A.; de Sauvage, F.J. Characterization of two patched receptors for the vertebrate hedgehog protein family. Proc. Natl. Acad. Sci. USA 1998, 95, 13630–13634. [Google Scholar] [CrossRef] [PubMed]
- Rahnama, F.; Toftgård, R.; Zaphiropoulos, P.G. Distinct roles of PTCH2 splice variants in hedgehog signalling. Biochem. J. 2004, 378, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.Y.; Bishop, J.M. Suppressor of fused represses Gli-mediated transcription by recruiting the SAP18-mSin3 corepressor complex. Proc. Natl. Acad. Sci. USA 2002, 99, 5442–5447. [Google Scholar] [CrossRef] [PubMed]
- Kogerman, P.; Grimm, T.; Kogerman, L.; Krause, D.; Unden, A.B.; Sandstedt, B.; Toftgard, R.; Zaphiropoulos, P.G. Mammalian suppressor-of-fused modulates nuclear-cytoplasmic shuttling of Gli-1. Nat. Cell. Biol. 1999, 1, 312–319. [Google Scholar] [PubMed]
- Paces-Fessy, M.; Boucher, D.; Petit, E.; Paute-Briand, S.; Blanchet-Tournier, M.-F. The negative regulator of Gli, suppressor of fused (Sufu), interacts with SAP18, Galectin3 and other nuclear proteins. Biochem. J. 2004, 378, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.K.; Taipale, J.; Cooper, M.K.; Beachy, P.A. Inhibition of hedgehog signaling by direct binding of cyclopamine to smoothened service inhibition of hedgehog signaling by direct binding of cyclopamine to smoothened. Genes Dev. 2002, 2743–2748. [Google Scholar] [CrossRef] [PubMed]
- Ruiz i Altaba, A. Catching a Gli-mpse of Hedgehog. Cell 1997, 90, 193–196. [Google Scholar] [CrossRef]
- Sasaki, H.; Nishizaki, Y.; Hui, C.; Nakafuku, M.; Kondoh, H. Regulation of Gli2 and Gli3 activities by an amino-terminal repression domain: Implication of Gli2 and Gli3 as primary mediators of Shh signaling. Development (Cambridge, England) 1999, 126, 3915–3924. [Google Scholar]
- Kim, J.; Kato, M.; Beachy, P.A. Gli2 trafficking links hedgehog-dependent activation of smoothened in the primary cilium to transcriptional activation in the nucleus. Proc. Natl. Acad. Sci. USA 2009, 106, 21666–21671. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Maye, P.; Kogerman, P.; Tejedor, F.J.; Toftgard, R.; Xie, W.; Wu, G.; Wu, D. Regulation of Gli1 transcriptional activity in the nucleus by Dyrk1. J. Biol. Chem. 2002, 277, 35156–35161. [Google Scholar] [CrossRef] [PubMed]
- Hui, C.-C.; Angers, S. Gli proteins in development and disease. Annu. Rev. Cell Dev. Biol. 2011, 27, 513–537. [Google Scholar] [CrossRef] [PubMed]
- Scales, S.J.; de Sauvage, F.J. Mechanisms of hedgehog pathway activation in cancer and implications for therapy. Trends Pharmacol. Sci. 2009, 30, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Stecca, B.; Ruiz, I.A.A. Context-dependent regulation of the Gli code in cancer by hedgehog and non-hedgehog signals. J. Mol. Cell Biol. 2010, 2, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.T.; Vanderbilt, D.B.; Lin, C.C.; Martin, K.H.; Brundage, K.M.; Ruppert, J.M. SOX9 inhibits beta-TrCP-mediated protein degradation to promote nuclear GLI1 expression and cancer stem cell properties. J. Cell Sci. 2015, 128, 1123–1138. [Google Scholar] [CrossRef] [PubMed]
- Ke, Z.; Caiping, S.; Qing, Z.; Xiaojing, W. Sonic hedgehog-GLI1 signals promote epithelial-mesenchymal transition in ovarian cancer by mediating PI3K/AKT pathway. Med. Oncol. 2015, 32, 368. [Google Scholar] [CrossRef] [PubMed]
- Rajurkar, M.; De Jesus-Monge, W.E.; Driscoll, D.R.; Appleman, V.A.; Huang, H.; Cotton, J.L.; Klimstra, D.S.; Zhu, L.J.; Simin, K.; Xu, L.; et al. The activity of GLI transcription factors is essential for Kras-induced pancreatic tumorigenesis. Proc. Natl. Acad. Sci. USA 2012, 109, E1038–E1047. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, B.; Lu, Y.; Teng, K.Y.; Nuovo, G.; Li, X.; Shapiro, C.L.; Majumder, S. Hedgehog signaling is a novel therapeutic target in tamoxifen-resistant breast cancer aberrantly activated by PI3K/AKT pathway. Cancer Res. 2012, 72, 5048–5059. [Google Scholar] [CrossRef] [PubMed]
- Seto, M.; Ohta, M.; Asaoka, Y.; Ikenoue, T.; Tada, M.; Miyabayashi, K.; Mohri, D.; Tanaka, Y.; Ijichi, H.; Tateishi, K.; et al. Regulation of the hedgehog signaling by the mitogen-activated protein kinase cascade in gastric cancer. Mol. Carcinog. 2009, 48, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhu, G.; Huang, J.; Li, L.; Du, Y.; Gao, Y.; Wu, D.; Wang, X.; Hsieh, J.T.; He, D.; et al. Non-canonical GLI1/2 activation by PI3K/AKT signaling in renal cell carcinoma: A novel potential therapeutic target. Cancer Lett. 2016, 370, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Makinodan, E.; Marneros, A.G. Protein kinase a activation inhibits oncogenic sonic hedgehog signalling and suppresses basal cell carcinoma of the skin. Exp. Dermatol. 2012, 21, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Sheng, T.; Chi, S.; Zhang, X.; Xie, J. Regulation of GLI1 localization by the camp/protein kinase a signaling axis through a site near the nuclear localization signal. J. Biol. Chem. 2006, 281, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Stecca, B.; Ruiz i Altaba, A. A GLI1-p53 inhibitory loop controls neural stem cell and tumour cell numbers. EMBO J. 2009, 28, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.W.; Lamm, M.; Iannaccone, S.; Higashiyama, N.; Leong, K.F.; Iannaccone, P.; Walterhouse, D. p53 modulates the activity of the GLI1 oncogene through interactions with the shared coactivator TAF9. DNA Repair (Amst.) 2015, 34, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Gorlin, R.J.; Goltz, R.W. Multiple nevoid basal-cell epithelioma, jaw cysts and bifid rib. N. Engl. J. Med. 1960, 262, 908–912. [Google Scholar] [CrossRef] [PubMed]
- Kimonis, V.E.; Goldstein, A.M.; Pastakia, B.; Yang, M.L.; Kase, R.; DiGiovanna, J.J.; Bale, A.E.; Bale, S.J. Clinical manifestations in 105 persons with nevoid basal cell carcinoma syndrome. Am. J. Med. Genet. 1997, 69, 299–308. [Google Scholar] [CrossRef]
- Aszterbaum, M.; Rothman, A.; Johnson, R.L.; Fisher, M.; Xie, J.; Bonifas, J.M.; Zhang, X.; Scott, M.P.; Epstein, E.H. Identification of mutations in the human patched gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. 1998, 110, 885–888. [Google Scholar] [CrossRef] [PubMed]
- Hahn, H.; Wicking, C.; Zaphiropoulos, P.G.; Gailani, M.R.; Shanley, S.; Chidambaram, A.; Vorechovsky, I.; Holmberg, E.; Unden, A.B.; Gillies, S.; et al. Mutations of the human homolog of drosophila patched in the nevoid basal cell carcinoma syndrome. Cell 2015, 85, 841–851. [Google Scholar] [CrossRef]
- Epstein, E.H. Basal cell carcinomas: Attack of the hedgehog. Nat. Rev. Cancer 2008, 8, 743–754. [Google Scholar] [CrossRef] [PubMed]
- Kool, M.; Koster, J.; Bunt, J.; Hasselt, N.E.; Lakeman, A.; van Sluis, P.; Troost, D.; Meeteren, N.S.V.; Caron, H.N.; Cloos, J.; et al. Integrated genomics identifies five medulloblastoma subtypes with distinct genetic profiles, pathway signatures and clinicopathological features. PLoS ONE 2008, 3, e3088. [Google Scholar] [CrossRef] [PubMed]
- Goodrich, L.V.; Milenković, L.; Higgins, K.M.; Scott, M.P. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science 1997, 277, 1109–1113. [Google Scholar] [CrossRef] [PubMed]
- Bar, E.E.; Chaudhry, A.; Lin, A.; Fan, X.; Schreck, K.; Matsui, W.; Piccirillo, S.; Vescovi, A.L.; DiMeco, F.; Olivi, A.; et al. Cyclopamine-mediated hedgehog pathway inhibition depletes stem-like cancer cells in glioblastoma. Stem Cells 2007, 25, 2524–2533. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, R.L.; Paw, I.; Zhu, H.; Sirkisoon, S.; Xing, F. The gain-of-function GLI1 transcription factor tGLI1 enhances expression of VEGF-C and TEM7 to promote glioblastoma angiogenesis. 2015, 6, 22653–22665. [Google Scholar] [CrossRef] [PubMed]
- Karhadkar, S.S.; Bova, G.S.; Abdallah, N.; Dhara, S.; Gardner, D.; Maitra, A.; Isaacs, J.T.; Berman, D.M.; Beachy, P.A. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 2004, 431, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Varnat, F.; Duquet, A.; Malerba, M.; Zbinden, M.; Mas, C.; Gervaz, P.; Ruiz i Altaba, A. Human colon cancer epithelial cells harbour active hedgehog-GLI signalling that is essential for tumour growth, recurrence, metastasis and stem cell survival and expansion. EMBO Mol. Med. 2009, 1, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Yauch, R.L.; Gould, S.E.; Scales, S.J.; Tang, T.; Tian, H.; Ahn, C.P.; Marshall, D.; Fu, L.; Januario, T.; Kallop, D.; et al. A paracrine requirement for hedgehog signalling in cancer. Nature 2008, 455, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Dontu, G.; Mantle, I.D.; Patel, S.; Ahn, N.S.; Jackson, K.W.; Suri, P.; Wicha, M.S. Hedgehog signaling and BMI-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res. 2006, 66, 6063–6071. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Chen, A.; Jamieson, C.H.; Fereshteh, M.; Abrahamsson, A.; Blum, J.; Kwon, H.Y.; Kim, J.; Chute, J.P.; Rizzieri, D.; et al. Hedgehog signalling is essential for maintenance of cancer stem cells in myeloid leukaemia. Nature 2009, 458, 776–779. [Google Scholar] [CrossRef] [PubMed]
- Feldmann, G.; Dhara, S.; Fendrich, V.; Bedja, D.; Beaty, R.; Mullendore, M.; Karikari, C.; Alvarez, H.; Iacobuzio-Donahue, C.; Jimeno, A.; et al. Blockade of hedgehog signaling inhibits pancreatic cancer invasion and metastases: A new paradigm for combination therapy in solid cancers. Cancer Res. 2007, 67, 2187–2196. [Google Scholar] [CrossRef]
- Rasheed, Z.A.; Yang, J.; Wang, Q.; Kowalski, J.; Freed, I.; Murter, C.; Hong, S.M.; Koorstra, J.B.; Rajeshkumar, N.V.; He, X.; et al. Prognostic significance of tumorigenic cells with mesenchymal features in pancreatic adenocarcinoma. J. Natl. Cancer Inst. 2010, 102, 340–351. [Google Scholar] [CrossRef]
- Lo, H.W.; Zhu, H.; Cao, X.; Aldrich, A.; Ali-Osman, F. A novel splice variant of GLI1 that promotes glioblastoma cell migration and invasion. Cancer Res. 2009, 69, 6790–6798. [Google Scholar] [CrossRef]
- Cao, X.; Geradts, J.; Dewhirst, M.W.; Lo, H.W. Upregulation of VEGF-A and CD24 gene expression by the tGLI1 transcription factor contributes to the aggressive behavior of breast cancer cells. Oncogene 2012, 31, 104–115. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Carpenter, R.L.; Han, W.; Lo, H.W. The GLI1 splice variant tGLI1 promotes glioblastoma angiogenesis and growth. Cancer Lett. 2014, 343, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Carpenter, R.L.; Lo, H.-W. tGLI1 upregulates expression of VEGFR2 and VEGF-A, leading to a robust VEGF-VEGFR2 autocrine loop and cancer cell growth. Cancer Hallm. 2013, 1, 28–37. [Google Scholar] [CrossRef]
- Carpenter, R.L.; Lo, H.-W. Hedgehog pathway and GLI1 isoforms in human cancer. Discov. Med. 2012, 13, 105–113. [Google Scholar] [PubMed]
- Cooper, M.K.; Porter, J.A.; Young, K.E.; Beachy, P.A. Teratogen-mediated inhibition of target tissue response to Shh signaling. Science (New York) 1998, 280, 1603–1607. [Google Scholar] [CrossRef]
- Incardona, J.P.; Gaffield, W.; Kapur, R.P.; Roelink, H. The teratogenic veratrum alkaloid cyclopamine inhibits sonic hedgehog signal transduction. Development (Cambridge, England) 1998, 125, 3553–3562. [Google Scholar]
- Sharpe, H.J.; Pau, G.; Dijkgraaf, G.J.; Basset-Seguin, N.; Modrusan, Z.; Januario, T.; Tsui, V.; Durham, A.B.; Dlugosz, A.A.; Haverty, P.M.; et al. Genomic analysis of smoothened inhibitor resistance in basal cell carcinoma. Cancer Cell 2015, 27, 327–341. [Google Scholar] [CrossRef] [PubMed]
- Taipale, J.; Chen, J.K.; Cooper, M.K.; Wang, B.; Mann, R.K.; Milenkovic, L.; Scott, M.P.; Beachy, P.A. Effects of oncogenic mutations in smoothened and patched can be reversed by cyclopamine. Nature 2000, 406, 1005–1009. [Google Scholar] [PubMed]
- Zhao, Y.; Tong, C.; Jiang, J. Hedgehog regulates smoothened activity by inducing a conformational switch. Nature 2007, 450, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, P.; Ruiz i Altaba, A. In vivo inhibition of endogenous brain tumors through systemic interference of hedgehog signaling in mice. Mech. Dev. 2005, 122, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Stecca, B.; Mas, C.; Clement, V.; Zbinden, M.; Correa, R.; Piguet, V.; Beermann, F.; Ruiz I Altaba, A. Melanomas require HEDGEHOG-GLI signaling regulated by interactions between GLI1 and the RAS-MEK/AKT pathways. Proc. Natl. Acad. Sci. USA 2007, 104, 5895–5900. [Google Scholar] [CrossRef] [PubMed]
- Hirotsu, M.; Setoguchi, T.; Sasaki, H.; Matsunoshita, Y.; Gao, H.; Nagao, H.; Kunigou, O.; Komiya, S. Smoothened as a new therapeutic target for human osteosarcoma. Mol. Cancer 2010, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Mimeault, M.; Johansson, S.L.; Henichart, J.-P.; Depreux, P.; Batra, S.K. Cytotoxic effects induced by docetaxel, gefitinib, and cyclopamine on side population and nonside population cell fractions from human invasive prostate cancer cells. Mol. Cancer Ther. 2010, 9, 617–630. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Gu, D.; He, M.; Liu, H.; Sheng, T.; Xie, G.; Li, C.X.; Zhang, X.; Wainwright, B.; Garrossian, A.; et al. Tumor shrinkage by cyclopamine tartrate through inhibiting hedgehog signaling. Chin. J. Cancer 2011, 30, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Roy, I.; Anchoori, R.K.; Fazli, S.; Maitra, A.; Beachy, P.A.; Khan, S.R. Targeted inhibition of hedgehog signaling by cyclopamine prodrugs for advanced prostate cancer. Bioorg. Med. Chem. 2008, 16, 2764–2768. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Garrossian, M.; Gardner, D.; Garrossian, A.; Chang, Y.T.; Kim, Y.K.; Chang, C.W.T. Synthesis and anticancer activity studies of cyclopamine derivatives. Bioorg. Med. Chem. Lett. 2008, 18, 1359–1363. [Google Scholar] [CrossRef] [PubMed]
- Robarge, K.D.; Brunton, S.A.; Castanedo, G.M.; Cui, Y.; Dina, M.S.; Goldsmith, R.; Gould, S.E.; Guichert, O.; Gunzner, J.L.; Halladay, J.; et al. GDC-0449—A potent inhibitor of the hedgehog pathway. Bioorg. Med. Chem. Lett. 2009, 19, 5576–5581. [Google Scholar] [CrossRef] [PubMed]
- Sekulic, A.; Migden, M.R.; Oro, A.E.; Dirix, L.; Lewis, K.D.; Hainsworth, J.D.; Solomon, J.A.; Yoo, S.; Arron, S.T.; Friedlander, P.A.; et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N. Engl. J. Med. 2012, 366, 2171–2179. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C.M.; Hann, C.L.; Laterra, J.; Yauch, R.L.; Callahan, C.A.; Fu, L.; Holcomb, T.; Stinson, J.; Gould, S.E.; Coleman, B.; et al. Treatment of medulloblastoma with hedgehog pathway inhibitor GDC-0449. N. Engl. J. Med. 2009, 361, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Yauch, R.L.; Dijkgraaf, G.J.P.; Alicke, B.; Januario, T.; Ahn, C.P.; Holcomb, T.; Pujara, K.; Stinson, J.; Callahan, C.A.; Tang, T.; et al. Smoothened mutation confers resistance to a hedgehog pathway inhibitor in medulloblastoma. Science 2009, 326, 572–574. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, C.; de Sauvage, F.J. Hedgehog fights back: Mechanisms of acquired resistance against smoothened antagonists. Cancer Res. 2011, 71, 5057–5061. [Google Scholar] [CrossRef] [PubMed]
- Catenacci, D.V.T.; Junttila, M.R.; Karrison, T.; Bahary, N.; Horiba, M.N.; Nattam, S.R.; Marsh, R.; Wallace, J.; Kozloff, M.; Rajdev, L.; et al. Randomized phase IB/II study of gemcitabine plus placebo or vismodegib, a hedgehog pathway inhibitor, in patients with metastatic pancreatic cancer. J. Clin. Oncol. 2015, 33, 4284–4292. [Google Scholar] [CrossRef] [PubMed]
- Olive, K.P.; Jacobetz, M.A.; Davidson, C.J.; Gopinathan, A.; McIntyre, D.; Honess, D.; Madhu, B.; Goldgraben, M.A.; Caldwell, M.E.; Allard, D.; et al. Inhibition of hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science (New York) 2009, 324, 1457–1461. [Google Scholar] [CrossRef] [PubMed]
- Rhim, A.D.; Oberstein, P.E.; Thomas, D.H.; Mirek, E.T.; Palermo, C.F.; Sastra, S.A.; Dekleva, E.N.; Saunders, T.; Becerra, C.P.; Tattersall, I.W.; et al. Stromal elements act to restrain, rather than support, pancreatic ductal adenocarcinoma. Cancer Cell 2014, 25, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.; Wu, X.; Jiang, J.; Gao, W.; Wan, Y.; Cheng, D.; Han, D.; Liu, J.; Englund, N.P.; Wang, Y.; et al. Discovery of NVP-LDE225, a potent and selective smoothened antagonist. ACS Med. Chem. Lett. 2010, 1, 130–134. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, C.; Rosa, R.; Marciano, R.; D’Amato, V.; Formisano, L.; Nappi, L.; Raimondo, L.; Di Mauro, C.; Servetto, A.; Fulciniti, F.; et al. Inhibition of hedgehog signalling by NVP-LDE225 (erismodegib) interferes with growth and invasion of human renal cell carcinoma cells. Br. J. Cancer 2014, 111, 1168–1179. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Rodova, M.; Nanta, R.; Meeker, D.; Van Veldhuizen, P.J.; Srivastava, R.K.; Shankar, S. NPV-LDE-225 (Erismodegib) inhibits epithelial mesenchymal transition and self-renewal of glioblastoma initiating cells by regulating miR-21, miR-128, and miR-200. Neuro-Oncology 2013, 15, 691–706. [Google Scholar] [CrossRef] [PubMed]
- Nanta, R.; Kumar, D.; Meeker, D.; Rodova, M.; Van Veldhuizen, P.J.; Shankar, S.; Srivastava, R.K. NVP-LDE-225 (Erismodegib) inhibits epithelial-mesenchymal transition and human prostate cancer stem cell growth in NOD/SCID IL2Rγ null mice by regulating BMI-1 and microRNA-128. Oncogenesis 2013, 2, e42. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.R.; Lescarbeau, A.; Grogan, M.J.; Tan, E.; Lin, G.; Austad, B.C.; Yu, L.-C.; Behnke, M.L.; Nair, S.J.; Hagel, M.; et al. Discovery of a potent and orally active hedgehog pathway antagonist (IPI-926). J. Med. Chem. 2009, 52, 4400–4418. [Google Scholar] [CrossRef] [PubMed]
- Campbell, V.T.; Nadesan, P.P.; Wang, Y.; Whetstone, H.; McGovern, K.; Read, M.; Alman, B.A.; Wunder, J.S. Abstract LB-380: Direct targeting of the Hedgehog pathway in primary chondrosarcoma xenografts with the Smoothened inhibitor IPI-926. Cancer Res. 2011, 71, LB-380. [Google Scholar] [CrossRef]
- Travaglione, V.; Peacock, C.; MacDougall, J.; McGovern, K.; Cushing, J.; Yu, L.C.; Trudeau, M.; Palombella, V.; Adams, J.; Hierman, J.; et al. A novel HH pathway inhibitor, IPI-926, delays recurrence post-chemotherapy in a primary human SCLC xenograft model. Cancer Res. 2008, 68, 4611. [Google Scholar]
- Frank-Kamenetsky, M.; Zhang, X.M.; Bottega, S.; Guicherit, O.; Wichterle, H.; Dudek, H.; Bumcrot, D.; Wang, F.Y.; Jones, S.; Shulok, J.; et al. Small-molecule modulators of hedgehog signaling: Identification and characterization of smoothened agonists and antagonists. J. Biol. 2002, 1, 10. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.A.; Guicherit, O.M.; Zaharian, B.I.; Xu, Y.; Chai, L.; Wichterle, H.; Kon, C.; Gatchalian, C.; Porter, J.A.; Rubin, L.L.; et al. Identification of a small molecule inhibitor of the hedgehog signaling pathway: Effects on basal cell carcinoma-like lesions. Proc. Natl. Acad. Sci. USA 2003, 100, 4616–4621. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.; Tang, J.Y.; Li, D.; Reich, M.; Callahan, C.A.; Fu, L.; Yauch, R.L.; Wang, F.; Kotkow, K.; Chang, K.S.; et al. Targeting superficial or nodular basal cell carcinoma with topically formulated small molecule inhibitor of smoothened. Clin. Can. Res. 2011, 17, 3378–3387. [Google Scholar] [CrossRef] [PubMed]
- Gendreau, S.B.; Hawkins, D.; Ho, C.P.; Lewin, A.; Lin, T.; Merchant, A.; Rowley, R.B.; Wang, Q.; Matsui, W.; Fargnoli, J. Abstract B192: Preclinical characterization of BMS-833923 (XL139), a hedgehog (HH) pathway inhibitor in early clinical development. Mol. Cancer Ther. 2009, 8, B192. [Google Scholar] [CrossRef]
- Riedlinger, D.; Bahra, M.; Boas-Knoop, S.; Lippert, S.; Bradtmöller, M.; Guse, K.; Seehofer, D.; Bova, R.; Sauer, I.M.; Neuhaus, P.; et al. Hedgehog pathway as a potential treatment target in human cholangiocarcinoma. J. Hepato-Biliary-Pancreat. Sci. 2014, 21, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, A.H.; Komatsu, Y.; Kelly, L.A.; Malhotra, U.; Rotoloni, C.; Kosovec, J.E.; Zahoor, H.; Makielski, R.; Bhatt, A.; Hoppo, T.; et al. Smoothened inhibition leads to decreased proliferation and induces apoptosis in esophageal adenocarcinoma cells. Cancer Investig. 2013, 31, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Munchhof, M.J.; Li, Q.; Shavnya, A.; Borzillo, G.V.; Boyden, T.L.; Jones, C.S.; LaGreca, S.D.; Martinez-Alsina, L.; Patel, N.; Pelletier, K.; et al. Discovery of PF-04449913, a potent and orally bioavailable inhibitor of smoothened. ACS Med. Chem. Lett. 2012, 3, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Minami, Y.; Hayakawa, F.; Kiyoi, H.; Sadarangani, A.; Jamieson, C.H.; Naoe, T. Treatment with Hedgehog Inhibitor, PF-04449913, Attenuates Leukemia-Initiation Potential in Acute Myeloid Leukemia Cells. Blood 2013, 122, 1649. [Google Scholar]
- Sadarangani, A.; Pineda, G.; Lennon, K.M.; Chun, H.-J.; Shih, A.; Schairer, A.E.; Court, A.C.; Goff, D.J.; Prashad, S.L.; Geron, I.; et al. GLI2 inhibition abrogates human leukemia stem cell dormancy. J. Transl. Med. 2015, 13, 98. [Google Scholar] [CrossRef] [PubMed]
- Rohner, A.; Spilker, M.E.; Lam, J.L.; Pascual, B.; Bartkowski, D.; Li, Q.J.; Yang, A.H.; Stevens, G.; Xu, M.; Wells, P.A.; et al. Effective targeting of hedgehog signaling in a medulloblastoma model with PF-5274857, a potent and selective smoothened antagonist that penetrates the blood-brain barrier. Mol. Cancer Ther. 2012, 11, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, T.; Oguro, Y.; Tanaka, T.; Shiokawa, Z.; Tanaka, Y.; Shibata, S.; Sato, Y.; Yamakawa, H.; Hattori, H.; Yamamoto, Y.; et al. Discovery of the investigational drug TAK-441, a pyrrolo[3,2-c]pyridine derivative, as a highly potent and orally active hedgehog signaling inhibitor: Modification of the core skeleton for improved solubility. Bioorg. Med. Chem. 2012, 20, 5507–5517. [Google Scholar] [CrossRef] [PubMed]
- Kogame, A.; Tagawa, Y.; Shibata, S.; Tojo, H.; Miyamoto, M.; Tohyama, K.; Kondo, T.; Prakash, S.; Shyu, W.C.; Asahi, S. Pharmacokinetic and pharmacodynamic modeling of hedgehog inhibitor TAK-441 for the inhibition of GLI1 messenger rna expression and antitumor efficacy in xenografted tumor model mice. Drug Metab. Dispos. 2013, 41, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Ibuki, N.; Ghaffari, M.; Pandey, M.; Iu, I.; Fazli, L.; Kashiwagi, M.; Tojo, H.; Nakanishi, O.; Gleave, M.E.; Cox, M.E. TAK-441, a novel investigational smoothened antagonist, delays castration-resistant progression in prostate cancer by disrupting paracrine hedgehog signaling. Int. J. Cancer 2013, 133, 1955–1966. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Shimizu, Y.; Nakashima, K.; Kondo, S.; Ogawa, K.; Sasaki, S.; Matsui, H. Inhibition mechanism exploration of investigational drug TAK-441 as inhibitor against vismodegib-resistant smoothened mutant. Eur. J. Pharmacol. 2014, 723, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Bender, M.H.; Hipskind, P.A.; Capen, A.R.; Cockman, M.; Credille, K.M.; Gao, H.; Bastian, J.A.; Clay, J.M.; Lobb, K.L.; Sall, D.J.; et al. Abstract 2819: Identification and characterization of a novel smoothened antagonist for the treatment of cancer with deregulated hedgehog signaling. Cancer Res. 2011, 71, 2819. [Google Scholar] [CrossRef]
- Wang, C.; Wu, H.; Katritch, V.; Han, G.W.; Huang, X.-P.; Liu, W.; Siu, F.Y.; Roth, B.L.; Cherezov, V.; Stevens, R.C. Structure of the human Smoothened receptor 7TM bound to an antitumor agent. Nature 2013, 497, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Hoch, L.; Faure, H.; Roudaut, H.; Schoenfelder, A.; Mann, A.; Girard, N.; Bihannic, L.; Ayrault, O.; Petricci, E.; Taddei, M.; et al. MRT-92 inhibits hedgehog signaling by blocking overlapping binding sites in the transmembrane domain of the Smoothened receptor. FASEB J. 2015, 29, 1817–1829. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-R.; Kozukue, N.; Han, J.-S.; Park, J.-H.; Chang, E.-Y.; Baek, E.-J.; Chang, J.-S.; Friedman, M. Glycoalkaloids and metabolites inhibit the growth of human colon (HT29) and liver (HEPG2) cancer cells. J. Agric. Food Chem. 2004, 52, 2832–2839. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.W.; Chen, M.-H.; Chuang, P.-T. Smoothened adopts multiple active and inactive conformations capable of trafficking to the primary cilium. PLoS ONE 2009, 4, e5182. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.M.Y.; Curran, T. The hedgehog’s tale: Developing strategies for targeting cancer. Nat. Rev. Cancer 2011, 11, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Lauth, M.; Bergström, Å.; Shimokawa, T.; Toftgård, R. Inhibition of GLI-mediated transcription and tumor cell growth by small-molecule antagonists. Proc. Natl. Acad. Sci. USA 2007, 104, 8455–8460. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Xu, R.; Zeng, C.; Lu, Q.; Huang, D.; Shi, C.; Zhang, W.; Deng, L.; Yan, R.; Rao, H.; et al. Down-regulation of GLI transcription factor leads to the inhibition of migration and invasion of ovarian cancer cells via integrin β4-mediated fak signaling. PLoS ONE 2014, 9, e88386. [Google Scholar] [CrossRef] [PubMed]
- Shahi, M.H.; Holt, R.; Rebhun, R.B. Blocking signaling at the level of GLI regulates downstream gene expression and inhibits proliferation of canine osteosarcoma cells. PLoS ONE 2014, 9, e96593. [Google Scholar]
- Srivastava, R.K.; Kaylani, S.Z.; Edrees, N.; Li, C.; Talwelkar, S.S.; Xu, J.; Palle, K.; Pressey, J.G.; Athar, M. GLI inhibitor GANT-61 diminishes embryonal and alveolar rhabdomyosarcoma growth by inhibiting Shh/AKT-mTOR axis. Oncotarget 2014, 5, 12151–12165. [Google Scholar] [CrossRef] [PubMed]
- Wickström, M.; Dyberg, C.; Shimokawa, T.; Milosevic, J.; Baryawno, N.; Fuskevåg, O.M.; Larsson, R.; Kogner, P.; Zaphiropoulos, P.G.; Johnsen, J.I. Targeting the hedgehog signal transduction pathway at the level of GLI inhibits neuroblastoma cell growth in vitro and in vivo. Int. J. Cancer 2013, 132, 1516–1524. [Google Scholar] [CrossRef] [PubMed]
- List, A.; Beran, M.; DiPersio, J.; Slack, J.; Vey, N.; Rosenfeld, C.S.; Greenberg, P. Opportunities for Trisenox® (arsenic trioxide) in the treatment of myelodysplastic syndromes. Leukemia 2003, 17, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, E.M.; Ringer, L.; Bulut, G.; Sajwan, K.P.; Hall, M.D.; Lee, Y.-C.; Peaceman, D.; Ozdemirli, M.; Rodriguez, O.; Macdonald, T.J.; et al. Arsenic trioxide inhibits human cancer cell growth and tumor development in mice by blocking Hedgehog/GLI pathway. J. Clin. Investig. 2011, 121, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J.J.; Kim, J.; Gardner, D.; Beachy, P.A. Arsenic antagonizes the hedgehog pathway by preventing ciliary accumulation and reducing stability of the GLI2 transcriptional effector. Proc. Natl. Acad. Sci. USA 2010, 107, 13432–13437. [Google Scholar] [CrossRef] [PubMed]
- Bansal, N.; Farley, N.J.; Wu, L.; Lewis, J.; Youssoufian, H.; Bertino, J.R. Darinaparsin inhibits prostate tumor-initiating cells and Du145 xenografts and is an inhibitor of hedgehog signaling. Mol. Cancer Ther. 2015, 14, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Yu, K.; Zhang, L.; Li, Y.; Li, Q.; Yang, Z.; Shen, T.; Duan, L.; Xiong, W.; Wang, W. Synergistic inhibition of colon carcinoma cell growth by Hedgehog-GLI1 inhibitor arsenic trioxide and phosphoinositide 3-kinase inhibitor LY294002. OncoTargets Ther. 2015, 8, 877–883. [Google Scholar]
- Kerl, K.; Moreno, N.; Holsten, T.; Ahlfeld, J.; Mertins, J.; Hotfilder, M.; Kool, M.; Bartelheim, K.; Schleicher, S.; Handgretinger, R.; et al. Arsenic trioxide inhibits tumor cell growth in malignant rhabdoid tumors in vitro and in vivo by targeting overexpressed GLI1. Int. J. Cancer 2014, 135, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Nagano, S.; Nagao, H.; Ishidou, Y.; Yokouchi, M.; Abematsu, M.; Yamamoto, T.; Komiya, S.; Setoguchi, T. Arsenic trioxide prevents osteosarcoma growth by inhibition of GLI transcription via DNA damage accumulation. PLoS ONE 2013, 8, e69466. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Cao, F.; Ye, X.; Zhao, H.; Liu, X.; Li, Y.; Shi, C.; Wang, H.; Zhou, J. Arsenic trioxide inhibits the hedgehog pathway which is aberrantly activated in acute promyelocytic leukemia. Acta Haematol. 2013, 130, 260–267. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Varona-Santos, J.; Singh, S.; Robbins, D.J.; Savaraj, N.; Nguyen, D.M. Targeting of the Hedgehog signal transduction pathway suppresses survival of malignant pleural mesothelioma cells in vitro. J. Thorac. Cardiovasc. Surg. 2014, 147, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Han, J.-B.; Sang, F.; Chang, J.-J.; Hua, Y.-Q.; Shi, W.-D.; Tang, L.-H.; Liu, L.-M. Arsenic trioxide inhibits viability of pancreatic cancer stem cells in culture and in a xenograft model via binding to SHH-GLI. OncoTargets Ther. 2013, 6, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Buglino, J.A.; Resh, M.D. Hhat is a palmitoylacyltransferase with specificity for N-palmitoylation of Sonic Hedgehog. J. Biol. Chem. 2008, 283, 22076–22088. [Google Scholar] [CrossRef] [PubMed]
- Mann, R.K.; Beachy, P.A. Novel lipid modifications of secreted protein signals. Annu. Rev. Biochem. 2004, 73, 891–923. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.H.; Li, Y.J.; Kawakami, T.; Xu, S.M.; Chuang, P.T. Palmitoylation is required for the production of a soluble multimeric hedgehog protein complex and long-range signaling in vertebrates. Genes Dev. 2004, 18, 641–659. [Google Scholar] [CrossRef] [PubMed]
- Petrova, E.; Rios-Esteves, J.; Ouerfelli, O.; Glickman, J.F.; Resh, M.D. Inhibitors of hedgehog acyltransferase block Sonic Hedgehog signaling. Nat. Chem. Biol. 2013, 9, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Matevossian, A.; Resh, M.D. Hedgehog acyltransferase as a target in estrogen receptor positive, HER2 amplified, and tamoxifen resistant breast cancer cells. Mol. Cancer 2015, 14, 72. [Google Scholar] [CrossRef] [PubMed]
- Petrova, E.; Matevossian, A.; Resh, M.D. Hedgehog acyltransferase as a target in pancreatic ductal adenocarcinoma. Oncogene 2015, 34, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Coon, V.; Laukert, T.; Pedone, C.A.; Laterra, J.; Kim, K.J.; Fults, D.W. Molecular therapy targeting Sonic hedgehog and hepatocyte growth factor signaling in a mouse model of medulloblastoma. Mol. Cancer Ther. 2010, 9, 2627–2636. [Google Scholar] [CrossRef] [PubMed]
- Chang, Q.; Foltz, W.D.; Chaudary, N.; Hill, R.P.; Hedley, D.W. Tumor-stroma interaction in orthotopic primary pancreatic cancer xenografts during hedgehog pathway inhibition. Int. J. Cancer 2013, 133, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Aberger, F.; Ruiz i Altaba, A. Context-dependent signal integration by the GLI code: The oncogenic load, pathways, modifiers and implications for cancer therapy. Semin. Cell Dev. Biol. 2014, 33, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Nye, M.D.; Almada, L.L.; Fernandez-Barrena, M.G.; Marks, D.L.; Elsawa, S.F.; Vrabel, A.; Tolosa, E.J.; Ellenrieder, V.; Fernandez-Zapico, M.E. The transcription factor GLI1 interacts with SMAD proteins to modulate transforming growth factor β-induced gene expression in a p300/CREB-binding protein-associated factor (PCAF)-dependent manner. J. Biol. Chem. 2014, 289, 15495–15506. [Google Scholar] [CrossRef] [PubMed]
- Sagiv, E.; Starr, A.; Rozovski, U.; Khosravi, R.; Altevogt, P.; Wang, T.; Arber, N. Targeting CD24 for treatment of colorectal and pancreatic cancer by monoclonal antibodies or small interfering RNA. Cancer Res. 2008, 68, 2803–2812. [Google Scholar] [CrossRef] [PubMed]
- Salnikov, A.V.; Bretz, N.P.; Perne, C.; Hazin, J.; Keller, S.; Fogel, M.; Herr, I.; Schlange, T.; Moldenhauer, G.; Altevogt, P. Antibody targeting of CD24 efficiently retards growth and influences cytokine milieu in experimental carcinomas. Br. J. Cancer 2013, 108, 1449–1459. [Google Scholar] [CrossRef] [PubMed]
- Bretz, N.; Altevogt, P.; Moldenhauer, G.; Salnikov, A.V.; Schlange, T. Particular Uses of CD24 Inhibitors. U.S. Patent EP2574627 A1, 3 April 2013. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Pisano, C.; Vlodavsky, I.; Ilan, N.; Zunino, F. The potential of heparanase as a therapeutic target in cancer. Biochem. Pharmacol. 2014, 89, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Ferro, V.; Dredge, K.; Liu, L.; Hammond, E.; Bytheway, I.; Li, C.; Johnstone, K.; Karoli, T.; Davis, K.; Copeman, E.; et al. PI-88 and novel heparan sulfate mimetics inhibit angiogenesis. Semin. Thromb. Hemost. 2007, 33, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Ferro, V.; Liu, L.; Johnstone, K.D.; Wimmer, N.; Karoli, T.; Handley, P.; Rowley, J.; Dredge, K.; Li, C.P.; Hammond, E.; et al. Discovery of PG545: A highly potent and simultaneous inhibitor of angiogenesis, tumor growth, and metastasis. J. Med. Chem. 2012, 55, 3804–3813. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.J.; Chang, J.; Lee, P.H.; Lin, D.Y.; Wu, C.C.; Jeng, L.B.; Lin, Y.J.; Mok, K.T.; Lee, W.C.; Yeh, H.Z.; et al. Adjuvant heparanase inhibitor PI-88 therapy for hepatocellular carcinoma recurrence. World J. Gastroenterol. 2014, 20, 11384–11393. [Google Scholar] [CrossRef] [PubMed]
- Ostapoff, K.T.; Awasthi, N.; Kutluk Cenik, B.; Hinz, S.; Dredge, K.; Schwarz, R.E.; Brekken, R.A. PG545, an angiogenesis and heparanase inhibitor, reduces primary tumor growth and metastasis in experimental pancreatic cancer. Mol. Cancer Ther. 2013, 12, 1190–1201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ngo, J.A.; Wetzel, M.D.; Marchetti, D. Heparanase mediates a novel mechanism in lapatinib-resistant brain metastatic breast cancer. Neoplasia 2015, 17, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Burgos-Ojeda, D.; McLean, K.; Bai, S.; Pulaski, H.; Gong, Y.; Silva, I.; Skorecki, K.; Tzukerman, M.; Buckanovich, R.J. A novel model for evaluating therapies targeting human tumor vasculature and human cancer stem-like cells. Cancer Res. 2013, 73, 3555–3565. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Z.; Hua, R.; Zhang, J.F.; Zhao, W.Y.; Zhao, E.H.; Tu, L.; Wang, C.J.; Cao, H.; Zhang, Z.G. TEM7 (PLXDC1), a key prognostic predictor for resectable gastric cancer, promotes cancer cell migration and invasion. Am. J. Cancer Res. 2015, 5, 772–781. [Google Scholar] [PubMed]
- Sitohy, B.; Nagy, J.A.; Dvorak, H.F. Anti-VEGF/VEGFR therapy for cancer: Reassessing the target. Cancer Res. 2012, 72, 1909–1914. [Google Scholar] [CrossRef] [PubMed]
- Shih, T.; Lindley, C. Bevacizumab: An angiogenesis inhibitor for the treatment of solid malignancies. Clin. Ther. 2006, 28, 1779–1802. [Google Scholar] [CrossRef] [PubMed]
- Keating, G.M. Bevacizumab: A review of its use in advanced cancer. Drugs 2014, 74, 1891–1925. [Google Scholar] [CrossRef] [PubMed]
- Couzin-Frankel, J.; Ogale, Y. Once on ‘fast track’, avastin now derailed. Science 2011, 333, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Sun, W. Ziv-aflibercept in metastatic colorectal cancer. Biologics 2014, 8, 13–25. [Google Scholar] [PubMed]
- Van Cutsem, E.; Tabernero, J.; Lakomy, R.; Prenen, H.; Prausová, J.; Macarulla, T.; Ruff, P.; van Hazel, G.A.; Moiseyenko, V.; Ferry, D.; et al. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a phase iii randomized trial in patients with metastatic colorectal cancer previously treated with an oxaliplatin-based regimen. J. Clin. Oncol. 2012, 30, 3499–3506. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, M.; Tester, A.; Phelan, D.; Klupacs, R. The novel therapeutic monoclonal antibody VGX-100 neutralises VEGF-C and inhibits tumor growth and metastasis in subcutaneous and orthotopic models of human cancer. Cancer Res. 2011, 71. LB-284. [Google Scholar] [CrossRef]
- Kieran, M.W.; Kalluri, R.; Cho, Y.-J. The VEGF pathway in cancer and disease: Responses, resistance, and the path forward. Cold Spring Harb. Perspect. Med. 2012, 2, a006593. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.M.; Adnane, L.; Newell, P.; Villanueva, A.; Llovet, J.M.; Lynch, M. Preclinical overview of sorafenib, a multikinase inhibitor that targets both raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol. Cancer Ther. 2008, 7, 3129–3140. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.-F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.-L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Escudier, B.; Eisen, T.; Stadler, W.M.; Szczylik, C.; Oudard, S.; Siebels, M.; Negrier, S.; Chevreau, C.; Solska, E.; Desai, A.A.; et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl. J. Med. 2007, 356, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Brose, M.S.; Nutting, C.M.; Jarzab, B.; Elisei, R.; Siena, S.; Bastholt, L.; de la Fouchardiere, C.; Pacini, F.; Paschke, R.; Shong, Y.K.; et al. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: A randomised, double-blind, phase 3 trial. Lancet 2014, 384, 319–328. [Google Scholar] [CrossRef]
- Norton, K.A.; Pandey, N.; Kato, Y.; Artemov, D.; Popel, A. In vitro and in vivo analyses of the effects of sunitinib on endothelial cell-surface vascular endothelial growth factor receptor-2. FASEB J. 2015, 29. [Google Scholar]
- Andrae, N.; Kirches, E.; Hartig, R.; Haase, D.; Keilhoff, G.; Kalinski, T.; Mawrin, C. Sunitinib targets PDGF-receptor and FLT3 and reduces survival and migration of human meningioma cells. Eur. J. Cancer 2012, 48, 1831–1841. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Hutson, T.E.; Tomczak, P.; Michaelson, M.D.; Bukowski, R.M.; Oudard, S.; Negrier, S.; Szczylik, C.; Pili, R.; Bjarnason, G.A.; et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 2009, 27, 3584–3590. [Google Scholar] [CrossRef] [PubMed]
- Demetri, G.D.; van Oosterom, A.T.; Garrett, C.R.; Blackstein, M.E.; Shah, M.H.; Verweij, J.; McArthur, G.; Judson, I.R.; Heinrich, M.C.; Morgan, J.A.; et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: A randomised controlled trial. Lancet 2006, 368, 1329–1338. [Google Scholar] [CrossRef]
- Azad, N.; Ball, D.; Sherman, S.; Rudek, M.; Falchook, G.; Nelkin, B.; Janku, F.; Papadopoulos, N.; O’Conner, A.; Ahmed, S.; et al. Abstract B279: A phase I study determining the safety and tolerability of combination therapy with Pazopanib (P), a VEGFR/PDGFR/Raf inhibitor, and GSK1120212 (Trametinib: T), a MEK inhibitor, in advanced solid tumors with expansion cohorts in advanced differentiated thyroid cancer (DTC), cholangiocarcinoma (CHCA), and soft tissue sarcoma (STS). Mol. Cancer Ther. 2013, 12, B279. [Google Scholar]
- Sternberg, C.N.; Davis, I.D.; Mardiak, J.; Szczylik, C.; Lee, E.; Wagstaff, J.; Barrios, C.H.; Salman, P.; Gladkov, O.A.; Kavina, A.; et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: Results of a randomized phase III trial. J. Clin. Oncol. 2010, 28, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Van der Graaf, W.T.A.; Blay, J.-Y.; Chawla, S.P.; Kim, D.-W.; Bui-Nguyen, B.; Casali, P.G.; Schöffski, P.; Aglietta, M.; Staddon, A.P.; Beppu, Y.; et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2012, 379, 1879–1886. [Google Scholar] [CrossRef]
- Wilhelm, S.M.; Dumas, J.; Adnane, L.; Lynch, M.; Carter, C.A.; Schütz, G.; Thierauch, K.-H.; Zopf, D. Regorafenib (BAY 73-4506): A new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int. J. Cancer 2011, 129, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Grothey, A.; Cutsem, E.V.; Sobrero, A.; Siena, S.; Falcone, A.; Ychou, M.; Humblet, Y.; Bouché, O.; Mineur, L.; Barone, C.; et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (correct): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2013, 381, 303–312. [Google Scholar] [CrossRef]
- Demetri, G.D.; Reichardt, P.; Kang, Y.-K.; Blay, J.-Y.; Rutkowski, P.; Gelderblom, H.; Hohenberger, P.; Leahy, M.; von Mehren, M.; Joensuu, H.; et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2013, 381, 295–302. [Google Scholar] [CrossRef]
- Inoue, K.; Torimura, T.; Nakamura, T.; Iwamoto, H.; Masuda, H.; Abe, M.; Hashimoto, O.; Koga, H.; Ueno, T.; Yano, H.; et al. Vandetanib, an inhibitor of VEGF receptor-2 and EGF receptor, suppresses tumor development and improves prognosis of liver cancer in mice. Clin. Cancer Res. 2012, 18, 3924–3933. [Google Scholar] [CrossRef] [PubMed]
- Wells, S.A.; Robinson, B.G.; Gagel, R.F.; Dralle, H.; Fagin, J.A.; Santoro, M.; Baudin, E.; Elisei, R.; Jarzab, B.; Vasselli, J.R.; et al. Vandetanib in patients with locally advanced or metastatic medullary thyroid cancer: A randomized, double-blind phase III trial. J. Clin. Oncol. 2012, 30, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Yakes, F.M.; Chen, J.; Tan, J.; Yamaguchi, K.; Shi, Y.; Yu, P.; Qian, F.; Chu, F.; Bentzien, F.; Cancilla, B.; et al. Cabozantinib (XL184), a novel met and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol. Cancer Ther. 2011, 10, 2298–2308. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R.; Schlumberger, M.J.; Müller, S.P.; Schöffski, P.; Brose, M.S.; Shah, M.H.; Licitra, L.; Jarzab, B.; Medvedev, V.; Kreissl, M.C.; et al. Cabozantinib in progressive medullary thyroid cancer. J. Clin. Oncol. 2013. [Google Scholar] [CrossRef] [PubMed]
- Tohyama, O.; Matsui, J.; Kodama, K.; Hata-Sugi, N.; Kimura, T.; Okamoto, K.; Minoshima, Y.; Iwata, M.; Funahashi, Y. Antitumor activity of lenvatinib (E7080): An angiogenesis inhibitor that targets multiple receptor tyrosine kinases in preclinical human thyroid cancer models. J. Thyroid Res. 2014, 2014, 638747. [Google Scholar] [CrossRef] [PubMed]
- Schlumberger, M.; Tahara, M.; Wirth, L.J.; Robinson, B.; Brose, M.S.; Elisei, R.; Dutcus, C.E.; de las Heras, B.; Zhu, J.; Habra, M.A.; et al. A phase 3, multicenter, double-blind, placebo-controlled trial of lenvatinib (E7080) in patients with 131I-refractory differentiated thyroid cancer (SELECT). J. Clin. Oncol. 2014, 32, LBA6008. [Google Scholar]
- Gross-Goupil, M.; François, L.; Quivy, A.; Ravaud, A. Axitinib: A review of its safety and efficacy in the treatment of adults with advanced renal cell carcinoma. Clin. Med. Insights. Oncol. 2013, 7, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Escudier, B.; Gore, M. Axitinib for the management of metastatic renal cell carcinoma. Drugs R D 2011, 11, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Krupitskaya, Y.; Wakelee, H.A. Ramucirumab, a fully human mab to the transmembrane signaling tyrosine kinase VEGFR-2 for the potential treatment of cancer. Curr. Opin. Investig. Drugs 2009, 10, 597–605. [Google Scholar] [PubMed]
- Wilke, H.; Muro, K.; Van Cutsem, E.; Oh, S.-C.; Bodoky, G.; Shimada, Y.; Hironaka, S.; Sugimoto, N.; Lipatov, O.; Kim, T.-Y.; et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): A double-blind, randomised phase 3 trial. Lancet Oncol. 2014, 15, 1224–1235. [Google Scholar] [CrossRef]
- Garon, E.B.; Ciuleanu, T.-E.; Arrieta, O.; Prabhash, K.; Syrigos, K.N.; Goksel, T.; Park, K.; Gorbunova, V.; Kowalyszyn, R.D.; Pikiel, J.; et al. Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): A multicentre, double-blind, randomised phase 3 trial. Lancet 2014, 384, 665–673. [Google Scholar] [CrossRef]
- Pricl, S.; Cortelazzi, B.; Dal Col, V.; Marson, D.; Laurini, E.; Fermeglia, M.; Licitra, L.; Pilotti, S.; Bossi, P.; Perrone, F. Smoothened (SMO) receptor mutations dictate resistance to vismodegib in basal cell carcinoma. Mol. Oncol. 2015, 9, 389–397. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
