RETRACTED: Mechanisms and Therapeutic Implications of Cell Death Induction by Indole Compounds
Abstract
1. Introduction
2. Indole-3-carbinol (I3C) and its Dimer 3,3′-Diindolylmethane (DIM)
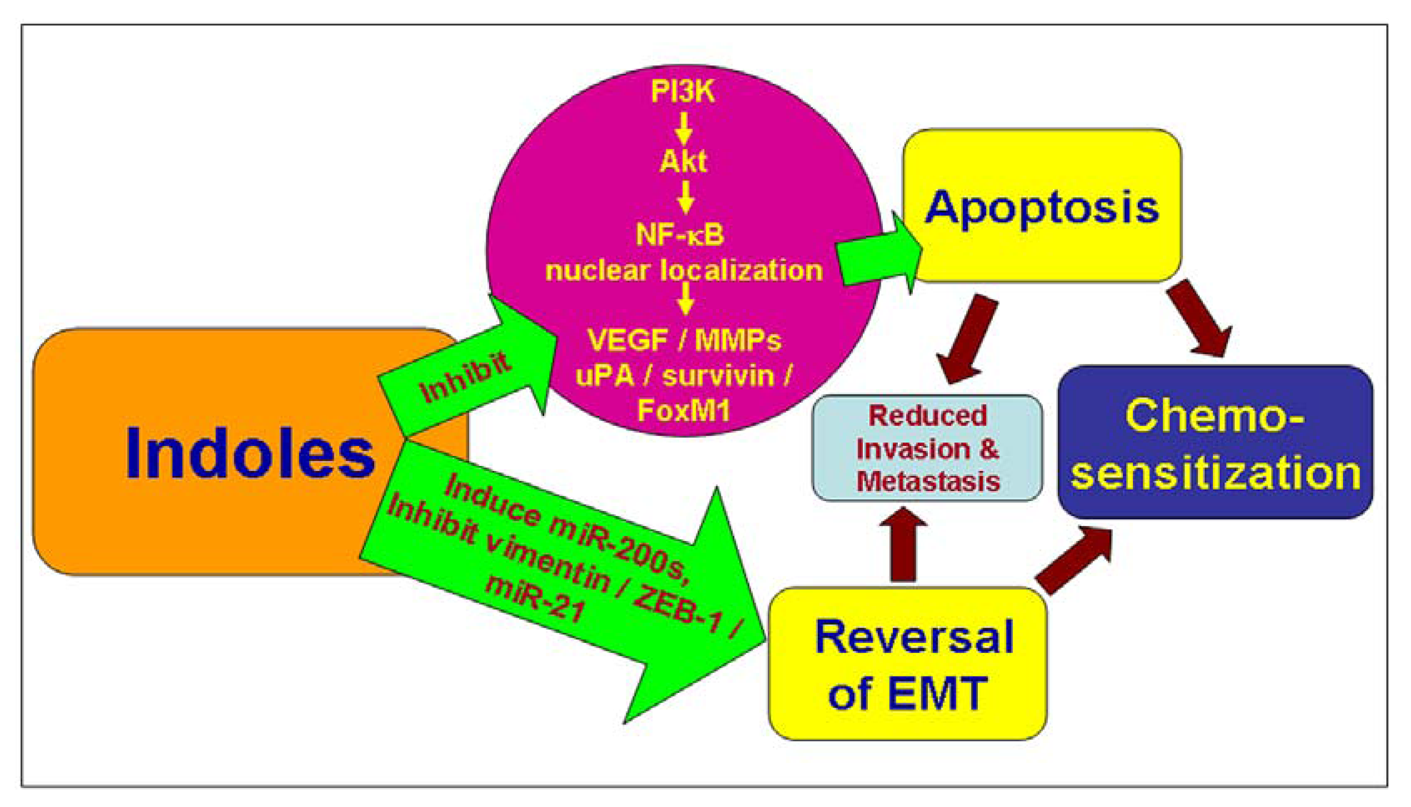
3. Induction of Cell Death by Indoles
3.1. Modulation of Pro-/Anti-Apoptotic Factors by Indoles
3.2. Inhibition of NF-κB Signaling by Indoles
3.3. Down-Regulation of Survivin by Indoles
4. Inhibition of Invasion and Metastases by Indoles
5. Cancer Therapy: The Problem of Drug-Resistance
6. Chemosensitization by Indole Compounds
7. Indole Compounds in Combinational Therapy
8. Conclusions and Perspective
Acknowledgements
References
- Boone, C.W.; Kelloff, G.J.; Malone, W.E. Identification of candidate cancer chemopreventive agents and their evaluation in animal models and human clinical trials: A review. Cancer Res. 1990, 50, 2–9. [Google Scholar]
- Ahmad, A.; Sakr, W.A.; Rahman, K.M.W. Novel targets for detection of cancer and their modulation by chemopreventive natural compounds. Front. Biosci. 2011, in press. [Google Scholar]
- Heber, D.; Bowerman, S. Applying science to changing dietary patterns. J. Nutr. 2001, 131, 3078S–3081S. [Google Scholar]
- Steinmetz, K.A.; Potter, J.D. Vegetables, fruit, and cancer prevention: A review. J. Am. Diet. Assoc. 1996, 96, 1027–1039. [Google Scholar]
- Michaud, D.S.; Spiegelman, D.; Clinton, S.K.; Rimm, E.B.; Willett, W.C.; Giovannucci, E.L. Fruit and vegetable intake and incidence of bladder cancer in a male prospective cohort. J. Natl. Cancer Inst. 1999, 91, 605–613. [Google Scholar]
- Terry, P.; Wolk, A.; Persson, I.; Magnusson, C. Brassica vegetables and breast cancer risk. JAMA 2001, 285, 2975–2977. [Google Scholar]
- van Poppel, G.; Verhoeven, D.T.; Verhagen, H.; Goldbohm, R.A. Brassica vegetables and cancer prevention. Epidemiology and mechanisms. Adv. Exp. Med. Biol. 1999, 472, 159–168. [Google Scholar]
- Verhagen, H.; Poulsen, H.E.; Loft, S.; van Poppel, G.; Willems, M.I.; van Bladeren, P.J. Reduction of oxidative DNA-damage in humans by brussels sprouts. Carcinogenesis 1995, 16, 969–970. [Google Scholar]
- Keck, A.S.; Finley, J.W. Cruciferous vegetables: Cancer protective mechanisms of glucosinolate hydrolysis products and selenium. Integr. Cancer Ther. 2004, 3, 5–12. [Google Scholar]
- McDanell, R.; McLean, A.E.; Hanley, A.B.; Heaney, R.K.; Fenwick, G.R. The effect of feeding brassica vegetables and intact glucosinolates on mixed-function-oxidase activity in the livers and intestines of rats. Food Chem. Toxicol. 1989, 27, 289–293. [Google Scholar]
- Oganesian, A.; Hendricks, J.D.; Williams, D.E. Long term dietary indole-3-carbinol inhibits diethylnitrosamine-initiated hepatocarcinogenesis in the infant mouse model. Cancer Lett. 1997, 118, 87–94. [Google Scholar]
- Jin, L.; Qi, M.; Chen, D.Z.; Anderson, A.; Yang, G.Y.; Arbeit, J.M.; Auborn, K.J. Indole-3-carbinol prevents cervical cancer in human papilloma virus type 16 (HPV16) transgenic mice. Cancer Res. 1999, 59, 3991–3997. [Google Scholar]
- He, Y.H.; Friesen, M.D.; Ruch, R.J.; Schut, H.A. Indole-3-carbinol as a chemopreventive agent in 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) carcinogenesis: Inhibition of PhIP-DNA adduct formation, acceleration of PhIP metabolism, and induction of cytochrome P450 in female F344 rats. Food Chem. Toxicol. 2000, 38, 15–23. [Google Scholar]
- Kojima, T.; Tanaka, T.; Mori, H. Chemoprevention of spontaneous endometrial cancer in female Donryu rats by dietary indole-3-carbinol. Cancer Res. 1994, 54, 1446–1449. [Google Scholar]
- Wong, G.Y.; Bradlow, L.; Sepkovic, D.; Mehl, S.; Mailman, J.; Osborne, M.P. Dose-ranging study of indole-3-carbinol for breast cancer prevention. J. Cell Biochem. Suppl. 1997. [Google Scholar]
- Yuan, F.; Chen, D.Z.; Liu, K.; Sepkovic, D.W.; Bradlow, H.L.; Auborn, K. Anti-estrogenic activities of indole-3-carbinol in cervical cells: Implication for prevention of cervical cancer. Anticancer Res. 1999, 19, 1673–1680. [Google Scholar]
- Verhoeven, D.T.; Verhagen, H.; Goldbohm, R.A.; van den Brandt, P.A.; van Poppel, G. A review of mechanisms underlying anticarcinogenicity by brassica vegetables. Chem. Biol. Interact. 1997, 103, 79–129. [Google Scholar]
- Shertzer, H.G.; Senft, A.P. The micronutrient indole-3-carbinol: Implications for disease and chemoprevention. Drug Metabol. Drug Interact. 2000, 17, 159–188. [Google Scholar]
- Grose, K.R.; Bjeldanes, L.F. Oligomerization of indole-3-carbinol in aqueous acid. Chem. Res. Toxicol. 1992, 5, 188–193. [Google Scholar]
- Stresser, D.M.; Blanchard, A.P.; Turner, S.D.; Erve, J.C.; Dandeneau, A.A.; Miller, V.P.; Crespi, C.L. Substrate-dependent modulation of CYP3A4 catalytic activity: Analysis of 27 test compounds with four fluorometric substrates. Drug Metab Dispos. 2000, 28, 1440–1448. [Google Scholar]
- Anderton, M.J.; Manson, M.M.; Verschoyle, R.D.; Gescher, A.; Lamb, J.H.; Farmer, P.B.; Steward, W.P.; Williams, M.L. Pharmacokinetics and tissue disposition of indole-3-carbinol and its acid condensation products after oral administration to mice. Clin. Cancer Res. 2004, 10, 5233–5241. [Google Scholar]
- Cotter, T.G. Apoptosis and cancer: The genesis of a research field. Nat Rev Cancer 2009, 9, 501–507. [Google Scholar]
- Sarkar, F.H.; Li, Y. Harnessing the fruits of nature for the development of multi-targeted cancer therapeutics. Cancer Treat. Rev. 2009, 35, 597–607. [Google Scholar]
- Sarkar, F.H.; Li, Y.; Wang, Z.; Kong, D. Cellular signaling perturbation by natural products. Cell Signal. 2009, 21, 1541–1547. [Google Scholar]
- Moiseeva, E.P.; Almeida, G.M.; Jones, G.D.; Manson, M.M. Extended treatment with physiologic concentrations of dietary phytochemicals results in altered gene expression, reduced growth, and apoptosis of cancer cells. Mol. Cancer Ther. 2007, 6, 3071–3079. [Google Scholar]
- Cover, C.M.; Hsieh, S.J.; Tran, S.H.; Hallden, G.; Kim, G.S.; Bjeldanes, L.F.; Firestone, G.L. Indole-3-carbinol inhibits the expression of cyclin-dependent kinase-6 and induces a G1 cell cycle arrest of human breast cancer cells independent of estrogen receptor signaling. J. Biol. Chem. 1998, 273, 3838–3847. [Google Scholar]
- Hong, C.; Firestone, G.L.; Bjeldanes, L.F. Bcl-2 family-mediated apoptotic effects of 3,3′-diindolylmethane (DIM) in human breast cancer cells. Biochem. Pharmacol. 2002, 63, 1085–1097. [Google Scholar]
- Hong, C.; Kim, H.A.; Firestone, G.L.; Bjeldanes, L.F. 3,3′-Diindolylmethane (DIM) induces a G(1) cell cycle arrest in human breast cancer cells that is accompanied by Sp1-mediated activation of p21(WAF1/CIP1) expression. Carcinogenesis 2002, 23, 1297–1305. [Google Scholar]
- Chinni, S.R.; Li, Y.; Upadhyay, S.; Koppolu, P.K.; Sarkar, F.H. Indole-3-carbinol (I3C) induced cell growth inhibition, G1 cell cycle arrest and apoptosis in prostate cancer cells. Oncogene 2001, 20, 2927–2936. [Google Scholar]
- Rahman, K.M.; Aranha, O.; Glazyrin, A.; Chinni, S.R.; Sarkar, F.H. Translocation of Bax to mitochondria induces apoptotic cell death in indole-3-carbinol (I3C) treated breast cancer cells. Oncogene 2000, 19, 5764–5771. [Google Scholar]
- Rahman, K.M.; Aranha, O.; Sarkar, F.H. Indole-3-carbinol (I3C) induces apoptosis in tumorigenic but not in nontumorigenic breast epithelial cells. Nutr. Cancer 2003, 45, 101–112. [Google Scholar]
- Rahman, K.M.; Li, Y.; Sarkar, F.H. Inactivation of akt and NF-kappaB play important roles during indole-3-carbinol-induced apoptosis in breast cancer cells. Nutr. Cancer 2004, 48, 84–94. [Google Scholar]
- Rahman, K.W.; Sarkar, F.H. Inhibition of nuclear translocation of nuclear factor-{kappa}B contributes to 3,3′-diindolylmethane-induced apoptosis in breast cancer cells. Cancer Res. 2005, 65, 364–371. [Google Scholar]
- Rahman, K.W.; Li, Y.; Wang, Z.; Sarkar, S.H.; Sarkar, F.H. Gene expression profiling revealed survivin as a target of 3,3′-diindolylmethane-induced cell growth inhibition and apoptosis in breast cancer cells. Cancer Res. 2006, 66, 4952–4960. [Google Scholar]
- Stewart, Z.A.; Westfall, M.D.; Pietenpol, J.A. Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol. Sci. 2003, 24, 139–145. [Google Scholar]
- Cover, C.M.; Hsieh, S.J.; Cram, E.J.; Hong, C.; Riby, J.E.; Bjeldanes, L.F.; Firestone, G.L. Indole-3-carbinol and tamoxifen cooperate to arrest the cell cycle of MCF-7 human breast cancer cells. Cancer Res. 1999, 59, 1244–1251. [Google Scholar]
- Stresser, D.M.; Williams, D.E.; Griffin, D.A.; Bailey, G.S. Mechanisms of tumor modulation by indole-3-carbinol. Disposition and excretion in male Fischer 344 rats. Drug Metab Dispos. 1995, 23, 965–975. [Google Scholar]
- Ge, X.; Fares, F.A.; Yannai, S. Induction of apoptosis in MCF-7 cells by indole-3-carbinol is independent of p53 and bax. Anticancer Res. 1999, 19, 3199–3203. [Google Scholar]
- Bradlow, H.L.; Sepkovic, D.W.; Telang, N.T.; Osborne, M.P. Multifunctional aspects of the action of indole-3-carbinol as an antitumor agent. Ann. N. Y. Acad. Sci. 1999, 889, 204–213. [Google Scholar]
- Chen, D.Z.; Qi, M.; Auborn, K.J.; Carter, T.H. Indole-3-carbinol and diindolylmethane induce apoptosis of human cervical cancer cells and in murine HPV16-transgenic preneoplastic cervical epithelium. J. Nutr. 2001, 131, 3294–3302. [Google Scholar]
- Howells, L.M.; Gallacher-Horley, B.; Houghton, C.E.; Manson, M.M.; Hudson, E.A. Indole-3-carbinol inhibits protein kinase B/Akt and induces apoptosis in the human breast tumor cell line MDA MB468 but not in the nontumorigenic HBL100 line. Mol. Cancer Ther. 2002, 1, 1161–1172. [Google Scholar]
- Rahman, K.M.; Sarkar, F.H. Steroid hormone mimics: Molecular mechanisms of cell growth and apoptosis in normal and malignant mammary epithelial cells. J. Steroid Biochem. Mol. Biol. 2002, 80, 191–201. [Google Scholar]
- Ali, S.; Varghese, L.; Pereira, L.; Tulunay-Ugur, O.E.; Kucuk, O.; Carey, T.E.; Wolf, G.T.; Sarkar, F.H. Sensitization of squamous cell carcinoma to cisplatin induced killing by natural agents. Cancer Lett. 2009, 278, 201–209. [Google Scholar]
- Chen, Y.; Xu, J.; Jhala, N.; Pawar, P.; Zhu, Z.B.; Ma, L.; Byon, C.H.; McDonald, J.M. Fas-mediated apoptosis in cholangiocarcinoma cells is enhanced by 3,3′-diindolylmethane through inhibition of AKT signaling and FLICE-like inhibitory protein. Am. J. Pathol. 2006, 169, 1833–1842. [Google Scholar]
- Pappa, G.; Lichtenberg, M.; Iori, R.; Barillari, J.; Bartsch, H.; Gerhauser, C. Comparison of growth inhibition profiles and mechanisms of apoptosis induction in human colon cancer cell lines by isothiocyanates and indoles from Brassicaceae. Mutat. Res. 2006, 599, 76–87. [Google Scholar]
- Kim, E.J.; Park, S.Y.; Shin, H.K.; Kwon, D.Y.; Surh, Y.J.; Park, J.H. Activation of caspase-8 contributes to 3,3′-Diindolylmethane-induced apoptosis in colon cancer cells. J. Nutr. 2007, 137, 31–36. [Google Scholar]
- Frydoonfar, H.R.; McGrath, D.R.; Spigelman, A.D. Inhibition of proliferation of a colon cancer cell line by indole-3-carbinol. Colorectal Dis. 2002, 4, 205–207. [Google Scholar]
- Frydoonfar, H.R.; McGrath, D.R.; Spigelman, A.D. The effect of indole-3-carbinol and sulforaphane on a prostate cancer cell line. ANZ J. Surg. 2003, 73, 154–156. [Google Scholar]
- Suzui, M.; Inamine, M.; Kaneshiro, T.; Morioka, T.; Yoshimi, N.; Suzuki, R.; Kohno, H.; Tanaka, T. Indole-3-carbinol inhibits the growth of human colon carcinoma cells but enhances the tumor multiplicity and volume of azoxymethane-induced rat colon carcinogenesis. Int. J. Oncol. 2005, 27, 1391–1399. [Google Scholar]
- Savino, J.A., III; Evans, J.F.; Rabinowitz, D.; Auborn, K.J.; Carter, T.H. Multiple, disparate roles for calcium signaling in apoptosis of human prostate and cervical cancer cells exposed to diindolylmethane. Mol. Cancer Ther. 2006, 5, 556–563. [Google Scholar]
- Kandala, P.K.; Srivastava, S.K. Activation of checkpoint kinase 2 by 3,3′-diindolylmethane is required for causing G2/M cell cycle arrest in human ovarian cancer cells. Mol. Pharmacol. 2010, 78, 297–309. [Google Scholar]
- Abdelrahim, M.; Newman, K.; Vanderlaag, K.; Samudio, I.; Safe, S. 3,3′-diindolylmethane (DIM) and its derivatives induce apoptosis in pancreatic cancer cells through endoplasmic reticulum stress-dependent upregulation of DR5. Carcinogenesis 2006, 27, 717–728. [Google Scholar]
- Banerjee, S.; Wang, Z.; Kong, D.; Sarkar, F.H. 3,3′-Diindolylmethane enhances chemosensitivity of multiple chemotherapeutic agents in pancreatic cancer. Cancer Res. 2009, 69, 5592–5600. [Google Scholar]
- Le, H.T.; Schaldach, C.M.; Firestone, G.L.; Bjeldanes, L.F. Plant-derived 3,3′-Diindolylmethane is a strong androgen antagonist in human prostate cancer cells. J. Biol. Chem. 2003, 278, 21136–21145. [Google Scholar]
- Nachshon-Kedmi, M.; Fares, F.A.; Yannai, S. Therapeutic activity of 3,3′-diindolylmethane on prostate cancer in an in vivo model. Prostate 2004, 61, 153–160. [Google Scholar]
- Sarkar, F.H.; Li, Y. Indole-3-carbinol and prostate cancer. J. Nutr. 2004, 134 (12 Suppl.), 3493S–3498S. [Google Scholar]
- Cho, H.J.; Park, S.Y.; Kim, E.J.; Kim, J.K.; Park, J.H. 3,3′-Diindolylmethane inhibits prostate cancer development in the transgenic adenocarcinoma mouse prostate model. Mol. Carcinog. 2011, 50, 100–112. [Google Scholar]
- Wang, Z.; Yu, B.W.; Rahman, K.M.; Ahmad, F.; Sarkar, F.H. Induction of growth arrest and apoptosis in human breast cancer cells by 3,3-diindolylmethane is associated with induction and nuclear localization of p27kip. Mol. Cancer Ther. 2008, 7, 341–349. [Google Scholar]
- Sarkar, F.H.; Li, Y.; Wang, Z.; Kong, D. NF-kappaB signaling pathway and its therapeutic implications in human diseases. Int. Rev Immunol. 2008, 27, 293–319. [Google Scholar]
- Kong, D.; Li, Y.; Wang, Z.; Banerjee, S.; Sarkar, F.H. Inhibition of angiogenesis and invasion by 3,3′-diindolylmethane is mediated by the nuclear factor-kappaB downstream target genes MMP-9 and uPA that regulated bioavailability of vascular endothelial growth factor in prostate cancer. Cancer Res. 2007, 67, 3310–3319. [Google Scholar]
- Bhuiyan, M.M.; Li, Y.; Banerjee, S.; Ahmed, F.; Wang, Z.; Ali, S.; Sarkar, F.H. Down-regulation of androgen receptor by 3,3′-diindolylmethane contributes to inhibition of cell proliferation and induction of apoptosis in both hormone-sensitive LNCaP and insensitive C4-2B prostate cancer cells. Cancer Res. 2006, 66, 10064–10072. [Google Scholar]
- Ahmad, A.; Sakr, W.A.; Rahman, K.M.W. Role of Nuclear Factor-kappa B Signaling in Anticancer Properties of Indole Compounds. J. Exp. Clin. Med. 2011, 3, 55–62. [Google Scholar]
- Rahman, K.M.; Banerjee, S.; Ali, S.; Ahmad, A.; Wang, Z.; Kong, D.; Sakr, W.A. 3,3′-Diindolylmethane enhances taxotere-induced apoptosis in hormone-refractory prostate cancer cells through survivin down-regulation. Cancer Res. 2009, 69, 4468–4475. [Google Scholar]
- Bhatnagar, N.; Li, X.; Chen, Y.; Zhou, X.; Garrett, S.H.; Guo, B. 3,3′-diindolylmethane enhances the efficacy of butyrate in colon cancer prevention through down-regulation of survivin. Cancer Prev. Res. (Phila. PA) 2009, 2, 581–589. [Google Scholar]
- Ahmad, A.; Kong, D.; Sarkar, S.H.; Wang, Z.; Banerjee, S.; Sarkar, F.H. Inactivation of uPA and its receptor uPAR by 3,3′-diindolylmethane (DIM) leads to the inhibition of prostate cancer cell growth and migration. J. Cell Biochem. 2009, 107, 516–527. [Google Scholar]
- Ahmad, A.; Kong, D.; Wang, Z.; Sarkar, S.H.; Banerjee, S.; Sarkar, F.H. Down-regulation of uPA and uPAR by 3,3′-diindolylmethane contributes to the inhibition of cell growth and migration of breast cancer cells. J. Cell Biochem. 2009, 108, 916–925. [Google Scholar]
- Li, Y.; Wang, Z.; Kong, D.; Murthy, S.; Dou, Q.P.; Sheng, S.; Reddy, G.P.; Sarkar, F.H. Regulation of FOXO3a/beta-catenin/GSK-3beta signaling by 3,3′-diindolylmethane contributes to inhibition of cell proliferation and induction of apoptosis in prostate cancer cells. J. Biol. Chem. 2007, 282, 21542–21550. [Google Scholar]
- Khwaja, F.S.; Wynne, S.; Posey, I.; Djakiew, D. 3,3′-diindolylmethane induction of p75NTR-dependent cell death via the p38 mitogen-activated protein kinase pathway in prostate cancer cells. Cancer Prev. Res. (Phila PA) 2009, 2, 566–571. [Google Scholar]
- Lei, P.; Abdelrahim, M.; Cho, S.D.; Liu, X.; Safe, S. Structure-dependent activation of endoplasmic reticulum stress-mediated apoptosis in pancreatic cancer by 1,1-bis(3′-indoly)-1-(p-substituted phenyl)methanes. Mol. Cancer Ther. 2008, 7, 3363–3372. [Google Scholar]
- Guo, J.; Chintharlapalli, S.; Lee, S.O.; Cho, S.D.; Lei, P.; Papineni, S.; Safe, S. Peroxisome proliferator-activated receptor gamma-dependent activity of indole ring-substituted 1,1-bis(3′-indolyl)-1-(p-biphenyl)methanes in cancer cells. Cancer Chemother. Pharmacol. 2010, 66, 141–150. [Google Scholar]
- Salomons, G.S.; Brady, H.J.; Verwijs-Janssen, M.; Van Den Berg, J.D.; Hart, A.A.; Van Den, B.H.; Behrendt, H.; Hahlen, K.; Smets, L.A. The Bax alpha:Bcl-2 ratio modulates the response to dexamethasone in leukaemic cells and is highly variable in childhood acute leukaemia. Int. J. Cancer 1997, 71, 959–965. [Google Scholar]
- Kim, D.S.; Jeong, Y.M.; Moon, S.I.; Kim, S.Y.; Kwon, S.B.; Park, E.S.; Youn, S.W.; Park, K.C. Indole-3-carbinol enhances ultraviolet B-induced apoptosis by sensitizing human melanoma cells. Cell Mol. Life Sci. 2006, 63, 2661–2668. [Google Scholar]
- Karin, M. The IkappaB kinase-a bridge between inflammation and cancer. Cell Res. 2008, 18, 334–342. [Google Scholar]
- Karin, M.; Greten, F.R. NF-kappaB: Linking inflammation and immunity to cancer development and progression. Nat. Rev. Immunol. 2005, 5, 749–759. [Google Scholar]
- Karin, M. Nuclear factor-kappaB in cancer development and progression. Nature 2006, 441, 431–436. [Google Scholar]
- Haefner, B. NF-kappa B: Arresting a major culprit in cancer. Drug Discov. Today 2002, 7, 653–663. [Google Scholar]
- Wang, Z.; Banerjee, S.; Li, Y.; Rahman, K.M.; Zhang, Y.; Sarkar, F.H. Down-regulation of notch-1 inhibits invasion by inactivation of nuclear factor-kappaB, vascular endothelial growth factor, and matrix metalloproteinase-9 in pancreatic cancer cells. Cancer Res. 2006, 66, 2778–2784. [Google Scholar]
- Wang, Z.; Sengupta, R.; Banerjee, S.; Li, Y.; Zhang, Y.; Rahman, K.M.; Aboukameel, A.; Mohammad, R.; Majumdar, A.P.; Abbruzzese, J.L.; Sarkar, F.H. Epidermal growth factor receptor-related protein inhibits cell growth and invasion in pancreatic cancer. Cancer Res. 2006, 66, 7653–7660. [Google Scholar]
- Ahmad, A.; Banerjee, S.; Wang, Z.; Kong, D.; Majumdar, A.P.N.; Sarkar, F.H. Aging and Inflammation: Etiological culprits of Cancer. Curr. Aging Sci. 2009, 2, 174–186. [Google Scholar]
- Wu, K.; Bonavida, B. The activated NF-kappaB-Snail-RKIP circuitry in cancer regulates both the metastatic cascade and resistance to apoptosis by cytotoxic drugs. Crit. Rev. Immunol. 2009, 29, 241–254. [Google Scholar]
- Li, Y.; Chinni, S.R.; Sarkar, F.H. Selective growth regulatory and pro-apoptotic effects of DIM is mediated by AKT and NF-kappaB pathways in prostate cancer cells. Front Biosci. 2005, 10, 236–243. [Google Scholar]
- Garikapaty, V.P.; Ashok, B.T.; Tadi, K.; Mittelman, A.; Tiwari, R.K. Synthetic dimer of indole-3-carbinol: Second generation diet derived anti-cancer agent in hormone sensitive prostate cancer. Prostate 2006, 66, 453–462. [Google Scholar]
- Garikapaty, V.P.; Ashok, B.T.; Tadi, K.; Mittelman, A.; Tiwari, R.K. 3,3′-Diindolylmethane downregulates pro-survival pathway in hormone independent prostate cancer. Biochem. Biophys. Res. Commun. 2006, 340, 718–725. [Google Scholar]
- Garcia, M.G.; Alaniz, L.D.; Cordo Russo, R.I.; Alvarez, E.; Hajos, S.E. PI3K/Akt inhibition modulates multidrug resistance and activates NF-kappaB in murine lymphoma cell lines. Leuk. Res. 2009, 33, 288–296. [Google Scholar]
- Li, Y.; VandenBoom, T.G.; Wang, Z.; Kong, D.; Ali, S.; Philip, P.A.; Sarkar, F.H. miR-146a suppresses invasion of pancreatic cancer cells. Cancer Res. 2010, 70, 1486–1495. [Google Scholar]
- Takada, Y.; Andreeff, M.; Aggarwal, B.B. Indole-3-carbinol suppresses NF-kappaB and IkappaBalpha kinase activation, causing inhibition of expression of NF-kappaB-regulated antiapoptotic and metastatic gene products and enhancement of apoptosis in myeloid and leukemia cells. Blood 2005, 106, 641–649. [Google Scholar]
- Kim, E.J.; Park, H.; Kim, J.; Park, J.H. 3,3′-diindolylmethane suppresses 12-O-tetradecanoylphorbol-13-acetate-induced inflammation and tumor promotion in mouse skin via the downregulation of inflammatory mediators. Mol. Carcinog. 2010, 49, 672–683. [Google Scholar]
- Altieri, D.C. Survivin, cancer networks and pathway-directed drug discovery. Nat. Rev. Cancer 2008, 8, 61–70. [Google Scholar]
- Andersen, M.H.; Svane, I.M.; Becker, J.C.; Straten, P.T. The universal character of the tumor-associated antigen survivin. Clin. Cancer Res. 2007, 13, 5991–5994. [Google Scholar]
- Mita, A.C.; Mita, M.M.; Nawrocki, S.T.; Giles, F.J. Survivin: Key regulator of mitosis and apoptosis and novel target for cancer therapeutics. Clin. Cancer Res. 2008, 14, 5000–5005. [Google Scholar]
- Pennati, M.; Folini, M.; Zaffaroni, N. Targeting survivin in cancer therapy: Fulfilled promises and open questions. Carcinogenesis 2007, 28, 1133–1139. [Google Scholar]
- Pennati, M.; Folini, M.; Zaffaroni, N. Targeting survivin in cancer therapy. Expert Opin. Ther. Targets 2008, 12, 463–476. [Google Scholar]
- Sreevalsan, S.; Jutooru, I.; Chadalapaka, G.; Walker, M.; Safe, S. 1,1-Bis(3′-indolyl)-1-(p-bromophenyl) methane and related compounds repress survivin and decrease gamma-radiation-induced survivin in colon and pancreatic cancer cells. Int. J. Oncol. 2009, 35, 1191–1199. [Google Scholar]
- Meng, Q.; Yuan, F.; Goldberg, I.D.; Rosen, E.M.; Auborn, K.; Fan, S. Indole-3-carbinol is a negative regulator of estrogen receptor-alpha signaling in human tumor cells. J. Nutr. 2000, 130, 2927–2931. [Google Scholar]
- Meng, Q.; Goldberg, I.D.; Rosen, E.M.; Fan, S. Inhibitory effects of Indole-3-carbinol on invasion and migration in human breast cancer cells. Breast Cancer Res. Treat. 2000, 63, 147–152. [Google Scholar]
- Meng, Q.; Qi, M.; Chen, D.Z.; Yuan, R.; Goldberg, I.D.; Rosen, E.M.; Auborn, K.; Fan, S. Suppression of breast cancer invasion and migration by indole-3-carbinol: Associated with up-regulation of BRCA1 and E-cadherin/catenin complexes. J. Mol. Med. 2000, 78, 155–165. [Google Scholar]
- Kong, D.; Banerjee, S.; Huang, W.; Li, Y.; Wang, Z.; Kim, H.R.; Sarkar, F.H. Mammalian target of rapamycin repression by 3,3′-diindolylmethane inhibits invasion and angiogenesis in platelet-derived growth factor-D-overexpressing PC3 cells. Cancer Res. 2008, 68, 1927–1934. [Google Scholar]
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat. Med. 1995, 1, 27–31. [Google Scholar]
- Hanahan, D.; Christofori, G.; Naik, P.; Arbeit, J. Transgenic mouse models of tumour angiogenesis: The angiogenic switch, its molecular controls, and prospects for preclinical therapeutic models. Eur. J. Cancer 1996, 32A, 2386–2393. [Google Scholar]
- Chang, X.; Tou, J.C.; Hong, C.; Kim, H.A.; Riby, J.E.; Firestone, G.L.; Bjeldanes, L.F. 3,3′-Diindolylmethane inhibits angiogenesis and the growth of transplantable human breast carcinoma in athymic mice. Carcinogenesis 2005, 26, 771–778. [Google Scholar]
- Rahman, K.M.; Sarkar, F.H.; Banerjee, S.; Wang, Z.; Liao, D.J.; Hong, X.; Sarkar, N.H. Therapeutic intervention of experimental breast cancer bone metastasis by indole-3-carbinol in SCID-human mouse model. Mol. Cancer Ther. 2006, 5, 2747–2756. [Google Scholar]
- Hsu, E.L.; Chen, N.; Westbrook, A.; Wang, F.; Zhang, R.; Taylor, R.T.; Hankinson, O. CXCR4 and CXCL12 down-regulation: a novel mechanism for the chemoprotection of 3,3′-diindolylmethane for breast and ovarian cancers. Cancer Lett. 2008, 265, 113–123. [Google Scholar]
- Hsu, E.L.; Chen, N.; Westbrook, A.; Wang, F.; Zhang, R.; Taylor, R.T.; Hankinson, O. Modulation of CXCR4, CXCL12, and Tumor Cell Invasion Potential In vitro by Phytochemicals. J. Oncol. 2009, 2009, 491985. [Google Scholar]
- Kim, E.J.; Shin, M.; Park, H.; Hong, J.E.; Shin, H.K.; Kim, J.; Kwon, D.Y.; Park, J.H. Oral Administration of 3,3′-Diindolylmethane Inhibits Lung Metastasis of 4T1 Murine Mammary Carcinoma Cells in BALB/c Mice. J. Nutr. 2009, 139, 2373–2379. [Google Scholar]
- Szakacs, G.; Paterson, J.K.; Ludwig, J.A.; Booth-Genthe, C.; Gottesman, M.M. Targeting multidrug resistance in cancer. Nat. Rev. Drug Discov. 2006, 5, 219–234. [Google Scholar]
- Borst, P.; Jonkers, J.; Rottenberg, S. What makes tumors multidrug resistant? Cell Cycle 2007, 6, 2782–2787. [Google Scholar]
- Gottesman, M.M. Mechanisms of cancer drug resistance. Annu. Rev. Med. 2002, 53, 615–627. [Google Scholar]
- Katsman, A.; Umezawa, K.; Bonavida, B. Chemosensitization and immunosensitization of resistant cancer cells to apoptosis and inhibition of metastasis by the specific NF-kappaB inhibitor DHMEQ. Curr. Pharm. Des 2009, 15, 792–808. [Google Scholar]
- Christensen, J.G.; LeBlanc, G.A. Reversal of multidrug resistance in vivo by dietary administration of the phytochemical indole-3-carbinol. Cancer Res. 1996, 56, 574–581. [Google Scholar]
- Ahmad, A.; Wang, Z.; Kong, D.; Ali, S.; Li, Y.; Banerjee, S.; Ali, R.; Sarkar, F.H. FoxM1 down-regulation leads to inhibition of proliferation, migration and invasion of breast cancer cells through the modulation of extra-cellular matrix degrading factors. Breast Cancer Res Treat. 2010, 122, 337–346. [Google Scholar]
- Ahmad, A.; Ali, S.; Wang, Z.; Ali, A.S.; Sethi, S.; Sakr, W.A.; Raz, A.; Rahman, K.W. 3,3′-diindolylmethane enhances taxotere-induced growth inhibition of breast cancer cells through down-regulation of FoxM1. Int. J. Cancer 2011. [Google Scholar] [CrossRef]
- Rahman, K.M.; Ali, S.; Aboukameel, A.; Sarkar, S.H.; Wang, Z.; Philip, P.A.; Sakr, W.A.; Raz, A. Inactivation of NF-kappaB by 3,3′-diindolylmethane contributes to increased apoptosis induced by chemotherapeutic agent in breast cancer cells. Mol. Cancer Ther. 2007, 6, 2757–2765. [Google Scholar]
- Ali, S.; Banerjee, S.; Ahmad, A.; El-Rayes, B.F.; Philip, P.A.; Sarkar, F.H. Apoptosis-inducing effect of erlotinib is potentiated by 3,3′-diindolylmethane in vitro and in vivo using an orthotopic model of pancreatic cancer. Mol. Cancer Ther. 2008, 7, 1708–1719. [Google Scholar]
- Ahmad, A.; Sakr, W.A.; Rahman, K.M. Anticancer properties of indole compounds: Mechanism of apoptosis induction and role in chemotherapy. Curr. Drug Targets 2010, 11, 652–666. [Google Scholar]
- McGuire, K.P.; Ngoubilly, N.; Neavyn, M.; Lanza-Jacoby, S. 3,3′-diindolylmethane and paclitaxel act synergistically to promote apoptosis in HER2/Neu human breast cancer cells. J. Surg. Res. 2006, 132, 208–213. [Google Scholar]
- Malejka-Giganti, D.; Parkin, D.R.; Bennett, K.K.; Lu, Y.; Decker, R.W.; Niehans, G.A.; Bliss, R.L. Suppression of mammary gland carcinogenesis by post-initiation treatment of rats with tamoxifen or indole-3-carbinol or their combination. Eur. J. Cancer Prev. 2007, 16, 130–141. [Google Scholar]
- Lyn-Cook, B.D.; Mohammed, S.I.; Davis, C.; Word, B.; Haefele, A.; Wang, H.; Hammons, G. Gender differences in gemcitabine (Gemzar) efficacy in cancer cells: Effect of indole-3-carbinol. Anticancer Res. 2010, 30, 4907–4913. [Google Scholar]
- Dashwood, R.H. Indole-3-carbinol: Anticarcinogen or tumor promoter in brassica vegetables? Chem. Biol. Interact. 1998, 110, 1–5. [Google Scholar]
- Malejka-Giganti, D.; Niehans, G.A.; Reichert, M.A.; Bliss, R.L. Post-initiation treatment of rats with indole-3-carbinol or beta-naphthoflavone does not suppress 7, 12-dimethylbenz[a]anthracene-induced mammary gland carcinogenesis. Cancer Lett. 2000, 160, 209–218. [Google Scholar]
- Kang, J.S.; Kim, D.J.; Ahn, B.; Nam, K.T.; Kim, K.S.; Choi, M.; Jang, D.D. Post-initiation treatment of Indole-3-carbinol did not suppress N-methyl-N-nitrosourea induced mammary carcinogenesis in rats. Cancer Lett. 2001, 169, 147–154. [Google Scholar]
- Bailey, G.S.; Hendricks, J.D.; Shelton, D.W.; Nixon, J.E.; Pawlowski, N.E. Enhancement of carcinogenesis by the natural anticarcinogen indole-3-carbinol. J. Natl. Cancer Inst. 1987, 78, 931–934. [Google Scholar]
- Dashwood, R.H.; Fong, A.T.; Williams, D.E.; Hendricks, J.D.; Bailey, G.S. Promotion of aflatoxin B1 carcinogenesis by the natural tumor modulator indole-3-carbinol: Influence of dose, duration, and intermittent exposure on indole-3-carbinol promotional potency. Cancer Res. 1991, 51, 2362–2365. [Google Scholar]
- Oganesian, A.; Hendricks, J.D.; Pereira, C.B.; Orner, G.A.; Bailey, G.S.; Williams, D.E. Potency of dietary indole-3-carbinol as a promoter of aflatoxin B1-initiated hepatocarcinogenesis: Results from a 9000 animal tumor study. Carcinogenesis 1999, 20, 453–458. [Google Scholar]
- Yoshida, M.; Katashima, S.; Ando, J.; Tanaka, T.; Uematsu, F.; Nakae, D.; Maekawa, A. Dietary indole-3-carbinol promotes endometrial adenocarcinoma development in rats initiated with N-ethyl-N′-nitro-N-nitrosoguanidine, with induction of cytochrome P450s in the liver and consequent modulation of estrogen metabolism. Carcinogenesis 2004, 25, 2257–2264. [Google Scholar]
- Shimamoto, K.; Dewa, Y.; Ishii, Y.; Kemmochi, S.; Taniai, E.; Hayashi, H.; Imaoka, M.; Morita, R.; Kuwata, K.; Suzuki, K.; Shibutani, M.; Mitsumori, K. Indole-3-carbinol enhances oxidative stress responses resulting in the induction of preneoplastic liver cell lesions in partially hepatectomized rats initiated with diethylnitrosamine. Toxicology 2011, 283, 109–117. [Google Scholar]
- Ahmad, A.; Farhan, A.S.; Singh, S.; Hadi, S.M. DNA breakage by resveratrol and Cu(II): Reaction mechanism and bacteriophage inactivation. Cancer Lett. 2000, 154, 29–37. [Google Scholar]
- Hadi, S.M.; Asad, S.F.; Singh, S.; Ahmad, A. Putative mechanism for anticancer and apoptosis-inducing properties of plant-derived polyphenolic compounds. IUBMB Life 2000, 50, 167–171. [Google Scholar]
- Ullah, M.F.; Ahmad, A.; Zubair, H.; Khan, H.Y.; Wang, Z.; Sarkar, F.H.; Hadi, S.M. Soy isoflavone genistein induces cell death in breast cancer cells through mobilization of endogenous copper ions and generation of reactive oxygen species. Mol. Nutr. Food Res. 2011, 55, 553–559. [Google Scholar]
- Siddiqui, I.A.; Adhami, V.M.; Ahmad, N.; Mukhtar, H. Nanochemoprevention: sustained release of bioactive food components for cancer prevention. Nutr. Cancer 2010, 62, 883–890. [Google Scholar]
- Safe, S.; Papineni, S.; Chintharlapalli, S. Cancer chemotherapy with indole-3-carbinol, bis(3′-indolyl)methane and synthetic analogs. Cancer Lett. 2008, 269, 326–338. [Google Scholar]
- Li, Y.; VandenBoom, T.G.; Kong, D.; Wang, Z.; Ali, S.; Philip, P.A.; Sarkar, F.H. Up-regulation of miR-200 and let-7 by natural agents leads to the reversal of epithelial-to-mesenchymal transition in gemcitabine-resistant pancreatic cancer cells. Cancer Res. 2009, 69, 6704–6712. [Google Scholar]
- Wang, Z.; Li, Y.; Ahmad, A.; Banerjee, S.; Azmi, A.S.; Kong, D.; Sarkar, F.H. Pancreatic cancer: Understanding and overcoming chemoresistance. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 27–33. [Google Scholar]
- Wang, Z.; Li, Y.; Ahmad, A.; Azmi, A.S.; Kong, D.; Banerjee, S.; Sarkar, F.H. Targeting miRNAs involved in cancer stem cell and EMT regulation: An emerging concept in overcoming drug resistance. Drug Resist. Updat. 2010, 13, 109–118. [Google Scholar]
- Bao, B.; Wang, Z.; Ali, S.; Kong, D.; Banerjee, S.; Ahmad, A.; Li, Y.; Azmi, A.S.; Miele, L.; Sarkar, F.H. Over-expression of FoxM1 leads to epithelial-mesenchymal transition and cancer stem cell phenotype in pancreatic cancer cells. J. Cell Biochem. 2011. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ahmad, A.; Sakr, W.A.; Rahman, K.W. RETRACTED: Mechanisms and Therapeutic Implications of Cell Death Induction by Indole Compounds. Cancers 2011, 3, 2955-2974. https://doi.org/10.3390/cancers3032955
Ahmad A, Sakr WA, Rahman KW. RETRACTED: Mechanisms and Therapeutic Implications of Cell Death Induction by Indole Compounds. Cancers. 2011; 3(3):2955-2974. https://doi.org/10.3390/cancers3032955
Chicago/Turabian StyleAhmad, Aamir, Wael A. Sakr, and KM Wahidur Rahman. 2011. "RETRACTED: Mechanisms and Therapeutic Implications of Cell Death Induction by Indole Compounds" Cancers 3, no. 3: 2955-2974. https://doi.org/10.3390/cancers3032955
APA StyleAhmad, A., Sakr, W. A., & Rahman, K. W. (2011). RETRACTED: Mechanisms and Therapeutic Implications of Cell Death Induction by Indole Compounds. Cancers, 3(3), 2955-2974. https://doi.org/10.3390/cancers3032955




