Expression Analysis of JCAD and IL-33 in Gingival Cancer Tumor Angiogenesis
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Data and Specimens
2.2. Antibodies
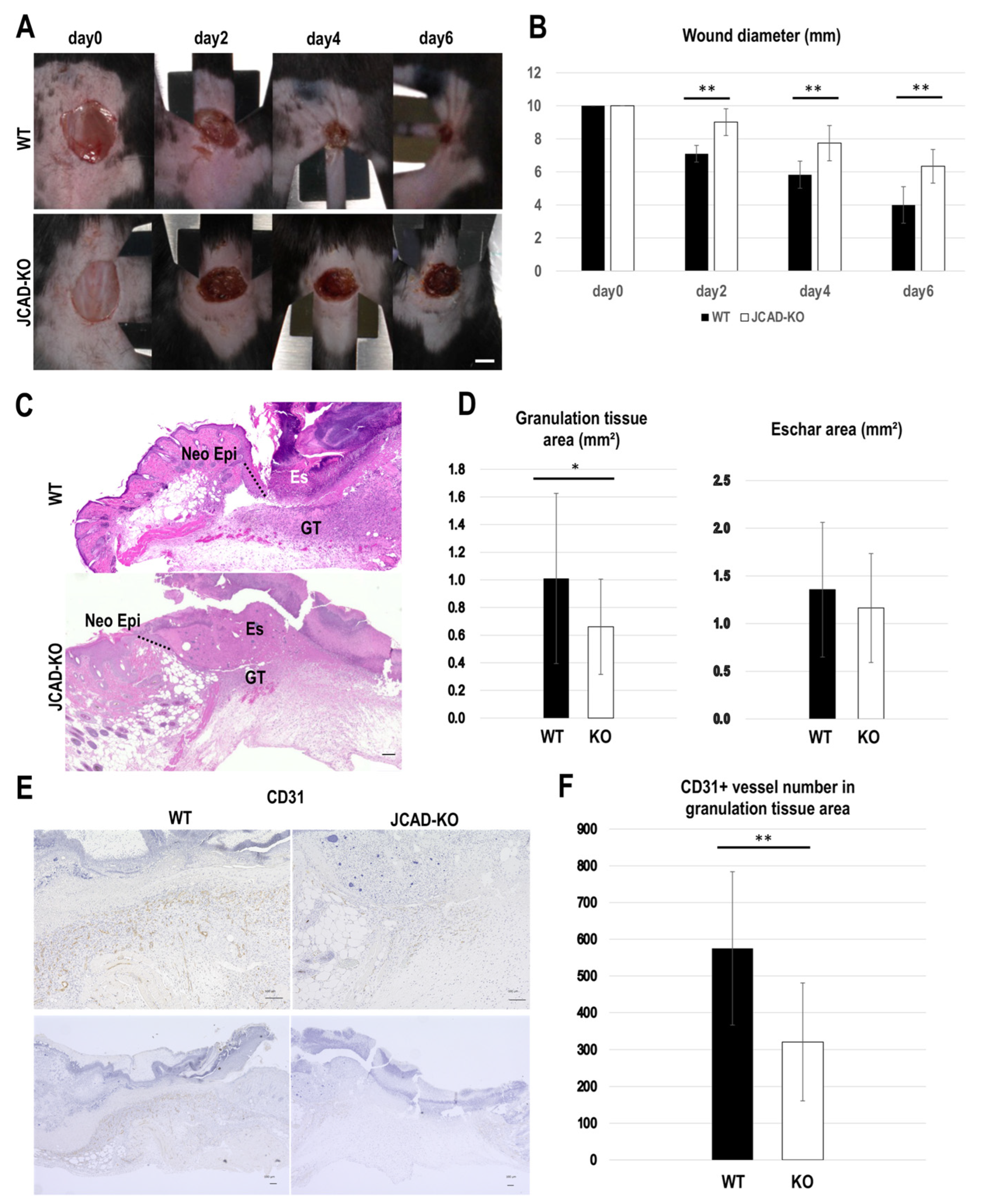
2.3. Wound Healing Assay in JCAD-KO Mice
2.4. IHC Analysis of the Tumor Microvascular Density in Human Mandibular Gingival SCC
2.5. Cell Culture and Quantification of Cell Numbers With and Without IL-33 Intranuclear Expression Following JCAD Knockdown in HUVECs
2.6. Statistical Analysis
3. Results
3.1. JCAD Deficiency Delays Wound Healing via Suppression of Angiogenesis
3.2. JCAD and IL-33 Expression as Pathological Angiogenesis Markers in Human Mandibular Gingival Cancer
3.3. JCAD and IL-33 Expression as Pathological Angiogenesis Markers in Human Metastatic Lymph Nodes
3.4. JCAD Knockdown Reduces the Effect of Tumor Necrosis Factor (TNF)-α on IL-33 Localization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sarode, G.; Maniyar, N.; Sarode, S.C.; Jafer, M.; Patil, S.; Awan, K.H. Epidemiologic aspects of oral cancer. Dis. Mon. 2020, 66, 100988. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Wang, Z.; Xu, M.; Li, B.; Huang, Z.; Qin, S.; Nice, E.C.; Tang, J.; Huang, C. Oral squamous cell carcinomas: State of the field and emerging directions. Int. J. Oral Sci. 2023, 15, 44. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Vassilakopoulou, M.; Psyrri, A.; Argiris, A. Targeting angiogenesis in head and neck cancer. Oral Oncol. 2015, 51, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Akashi, M.; Higashi, T.; Masuda, S.; Komori, T.; Furuse, M. A coronary artery disease-associated gene product, JCAD/KIAA1462, is a novel component of endothelial cell-cell junctions. Biochem. Biophys. Res. Commun. 2011, 413, 224–229. [Google Scholar] [CrossRef]
- Hara, T.; Monguchi, T.; Iwamoto, N.; Akashi, M.; Mori, K.; Oshita, T.; Okano, M.; Toh, R.; Irino, Y.; Shinohara, M.; et al. Targeted Disruption of JCAD (Junctional Protein Associated With Coronary Artery Disease)/KIAA1462, a Coronary Artery Disease-Associated Gene Product, Inhibits Angiogenic Processes In Vitro and In Vivo. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1667–1673. [Google Scholar] [CrossRef] [PubMed]
- Shigeoka, M.; Arimoto, S.; Akashi, M. JCAD expression and localization in human blood endothelial cells. Heliyon 2020, 6, e05121. [Google Scholar] [CrossRef]
- Bark, R.; Mercke, C.; Munck-Wikland, E.; Wisniewski, N.A.; Hammarstedt-Nordenvall, L. Cancer of the gingiva. Eur. Arch. Otorhinolaryngol. 2016, 273, 1335–1345. [Google Scholar] [CrossRef]
- Ma, Y.; Yu, Y.; Yin, Y.; Wang, L.; Yang, H.; Luo, S.; Zheng, Q.; Pan, Y.; Zhang, D. Potential role of epithelial-mesenchymal transition induced by periodontal pathogens in oral cancer. J. Cell Mol. Med. 2023, 28, e18064. [Google Scholar] [CrossRef]
- Okuyama, K.; Yanamoto, S. Oral Bacterial Contributions to Gingival Carcinogenesis and Progression. Cancer Prev. Res. 2023, 16, 199–209. [Google Scholar] [CrossRef]
- Liu, A.; Hayashi, M.; Ohsugi, Y.; Katagiri, S.; Akira, S.; Iwata, T.; Nakashima, T. The IL-33/ST2 axis is protective against acute inflammation during the course of periodontitis. Nat. Commun. 2024, 15, 2707. [Google Scholar] [CrossRef]
- Köseoğlu, S.; Hatipoğlu, M.; Sağlam, M.; Enhoş, Ş.; Esen, H.H. Interleukin-33 could play an important role in the pathogenesis of periodontitis. J. Periodontal Res. 2015, 50, 525–534. [Google Scholar] [CrossRef]
- Cayrol, C.; Girard, J.P. Interleukin-33 (IL-33): A critical review of its biology and the mechanisms involved in its release as a potent extracellular cytokine. Cytokine 2022, 156, 155891. [Google Scholar] [CrossRef] [PubMed]
- Moussion, C.; Ortega, N.; Girard, J.P. The IL-1-like cytokine IL-33 is constitutively expressed in the nucleus of endothelial cells and epithelial cells in vivo: A novel ’alarmin’? PLoS ONE 2008, 3, e3331. [Google Scholar] [CrossRef]
- Küchler, A.M.; Pollheimer, J.; Balogh, J.; Sponheim, J.; Manley, L.; Sorensen, D.R.; De Angelis, P.M.; Scott, H.; Haraldsen, G. Nuclear interleukin-33 is generally expressed in resting endothelium but rapidly lost upon angiogenic or proinflammatory activation. Am. J. Pathol. 2008, 173, 1229–1242. [Google Scholar] [CrossRef]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumors, 8th ed.; Wiley Blackwell: Malden, MA, USA, 2017. [Google Scholar]
- American Joint Committee on Cancer. Cancer Staging Manual, 8th ed.; Springer: New York, NY, USA, 2018. [Google Scholar]
- Kimura, S.; Nanbu, U.; Noguchi, H.; Harada, Y.; Kumamoto, K.; Sasaguri, Y.; Nakayama, T. Macrophage CCL22 expression in the tumor microenvironment and implications for survival in patients with squamous cell carcinoma of the tongue. J. Oral Pathol. Med. 2019, 48, 677–685. [Google Scholar] [CrossRef]
- Suraya, R.; Nagano, T.; Yumura, M.; Hara, T.; Akashi, M.; Yamamoto, M.; Tachihara, M.; Nishimura, Y.; Kobayashi, K. Loss of JCAD/KIAA1462 Protects the Lung from Acute and Chronic Consequences of Chronic Obstructive Pulmonary Disease. Int. J. Mol. Sci. 2024, 25, 9492. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Yamaguchi, Y.; Itami, S.; Yoshikawa, K.; Tabata, Y.; Matsumoto, K.; Nakamura, T. Neutralization of hepatocyte growth factor leads to retarded cutaneous wound healing associated with decreased neovascularization and granulation tissue formation. J. Investig. Dermatol. 2003, 120, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Qiang, L.; Yang, S.; Cui, Y.H.; He, Y.Y. Keratinocyte autophagy enables the activation of keratinocytes and fibroblastsand facilitates wound healing. Autophagy 2021, 17, 2128–2143. [Google Scholar] [CrossRef]
- Thandavarayan, R.A.; Garikipati, V.N.; Joladarashi, D.; Suresh Babu, S.; Jeyabal, P.; Verma, S.K.; Mackie, A.R.; Khan, M.; Arumugam, S.; Watanabe, K.; et al. Sirtuin-6 deficiency exacerbates diabetes-induced impairment of wound healing. Exp. Dermatol. 2015, 24, 773–778. [Google Scholar] [CrossRef]
- Anannamcharoen, S.; Boonya-Ussadorn, C.; Nimmanon, T. Tissue Microvessel Density as a Potential Predictive Marker for Vascular Invasion in Colorectal Cancer. Asian Pac. J. Cancer Prev. 2023, 24, 4097–4102. [Google Scholar] [CrossRef]
- Marioni, G.; D’Alessandro, E.; Giacomelli, L.; Staffieri, A. CD105 is a marker of tumour vasculature and a potential target for the treatment of head and neck squamous cell carcinoma. J. Oral Pathol. Med. 2010, 39, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Takase, Y.; Kai, K.; Masuda, M.; Akashi, M.; Tokunaga, O. Endoglin (CD105) expression and angiogenesis status in small cell lung cancer. Pathol. Res. Pract. 2010, 206, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Van Itallie, C.M.; Aponte, A.; Tietgens, A.J.; Gucek, M.; Fredriksson, K.; Anderson, J.M. The N and C termini of ZO-1 are surrounded by distinct proteins and functional protein networks. J. Biol. Chem. 2013, 288, 13775–13788. [Google Scholar] [CrossRef]
- Liberale, L.; Puspitasari, Y.M.; Ministrini, S.; Akhmedov, A.; Kraler, S.; Bonetti, N.R.; Beer, G.; Vukolic, A.; Bongiovanni, D.; Han, J.; et al. JCAD promotes arterial thrombosis through PI3K/Akt modulation: A translational study. Eur. Heart J. 2023, 44, 1818–1833. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.; Zhao, L.; Li, T.; Li, Y. Mechanosensitive JCAD: Emerged as a promising target for atherosclerosis. Acta Biochim. Biophys. Sin. 2020, 52, 580–581. [Google Scholar] [CrossRef]
- Xu, S.; Xu, Y.; Liu, P.; Zhang, S.; Liu, H.; Slavin, S.; Kumar, S.; Koroleva, M.; Luo, J.; Wu, X.; et al. The novel coronary artery disease risk gene JCAD/KIAA1462 promotes endothelial dysfunction and atherosclerosis. Eur. Heart J. 2019, 40, 2398–2408. [Google Scholar] [CrossRef]
- Ye, J.; Li, T.S.; Xu, G.; Zhao, Y.M.; Zhang, N.P.; Fan, J.; Wu, J. JCAD Promotes Progression of Nonalcoholic Steatohepatitis to Liver Cancer by Inhibiting LATS2 Kinase Activity. Cancer Res. 2017, 77, 5287–5300. [Google Scholar] [CrossRef]
- Jones, P.D.; Kaiser, M.A.; Ghaderi Najafabadi, M.; Koplev, S.; Zhao, Y.; Douglas, G.; Kyriakou, T.; Andrews, S.; Rajmohan, R.; Watkins, H.; et al. JCAD, a Gene at the 10p11 Coronary Artery Disease Locus, Regulates Hippo Signaling in Endothelial Cells. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 1711–1722. [Google Scholar] [CrossRef]
- Hooglugt, A.; van der Stoel, M.M.; Boon, R.A.; Huveneers, S. Endothelial YAP/TAZ Signaling in Angiogenesis and Tumor Vasculature. Front. Oncol. 2021, 10, 612802. [Google Scholar] [CrossRef]
- Mia, M.M.; Cibi, D.M.; Ghani, S.A.B.A.; Singh, A.; Tee, N.; Sivakumar, V.; Bogireddi, H.; Cook, S.A.; Mao, J.; Singh, M.K. Loss of Yap/Taz in cardiac fibroblasts attenuates adverse remodelling and improves cardiac function. Cardiovasc. Res. 2022, 118, 1785–1804. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.Z.; Yang, C.C.; Shueng, P.W.; Wang, Y.J.; Huang, C.S.; Chao, Y.C.; Chen, C.H.; Lin, C.W. Obesity-mediated upregulation of the YAP/IL33 signaling axis promotes aggressiveness and induces an immunosuppressive tumor microenvironment in breast cancer. J. Cell Physiol. 2023, 238, 992–1005. [Google Scholar] [CrossRef]
- Lou, J.; Zhang, B.; Cai, J.; Zhang, L.; Zhao, Y.; Zhao, Z. Diabetes exacerbates periodontitis by disrupting IL-33-mediated interaction between periodontal ligament fibroblasts and macrophages. Int. Immunopharmacol. 2025, 147, 113896. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, H.; Li, Y.; Xia, D.; Yang, L.; Ma, Y.; Li, H. The role of YAP/TAZ activity in cancer metabolic reprogramming. Mol. Cancer 2018, 17, 134. [Google Scholar] [CrossRef]
- Li, M.; van Esch, B.C.; Henricks, P.A.; Garssen, J.; Folkerts, G. IL-33 Is Involved in the Anti-Inflammatory Effects of Butyrate and Propionate on TNFα-Activated Endothelial Cells. Int. J. Mol. Sci. 2021, 22, 2447. [Google Scholar] [CrossRef]
- Wang, M.; Gao, M.; Yi, Z. Biological effects of IL-33/ST2 axis on oral diseases: Autoimmune diseases and periodontal diseases. Int. Immunopharmacol. 2023, 122, 110524. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.M. Role of IL-33 in inflammation and disease. J. Inflamm. 2011, 8, 22. [Google Scholar] [CrossRef]
- Sundlisaeter, E.; Edelmann, R.J.; Hol, J.; Sponheim, J.; Küchler, A.M.; Weiss, M.; Udalova, I.A.; Midwood, K.S.; Kasprzycka, M.; Haraldsen, G. The alarmin IL-33 is a notch target in quiescent endothelial cells. Am. J. Pathol. 2012, 181, 1099–1111. [Google Scholar] [CrossRef]
- Zhao, M.; He, Y.; Zhu, N.; Song, Y.; Hu, Q.; Wang, Z.; Ni, Y.; Ding, L. IL-33/ST2 signaling promotes constitutive and inductive PD-L1 expression and immune escape in oral squamous cell carcinoma. Br. J. Cancer 2023, 128, 833–843. [Google Scholar] [CrossRef]
- Chen, S.F.; Nieh, S.; Jao, S.W.; Wu, M.Z.; Liu, C.L.; Chang, Y.C.; Lin, Y.S. The paracrine effect of cancer-associated fibroblast-induced interleukin-33 regulates the invasiveness of head and neck squamous cell carcinoma. J. Pathol. 2013, 231, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, K.; Yagi-Nakanishi, S.; Nakanishi, Y.; Kondo, S.; Tsuji, A.; Endo, K.; Wakisaka, N.; Murono, S.; Yoshizaki, T. Expression of interleukin-33 is correlated with poor prognosis of patients with squamous cell carcinoma of the tongue. Auris Nasus Larynx 2014, 41, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Pichery, M.; Mirey, E.; Mercier, P.; Lefrancais, E.; Dujardin, A.; Ortega, N.; Girard, J.P. Endogenous IL-33 is highly expressed in mouse epithelial barrier tissues, lymphoid organs, brain, embryos, and inflamed tissues: In situ analysis using a novel Il-33-LacZ gene trap reporter strain. J. Immunol. 2012, 188, 3488–3495. [Google Scholar] [CrossRef]
- Smith, B.N.; Mishra, R.; Billet, S.; Placencio-Hickok, V.R.; Kim, M.; Zhang, L.; Duong, F.; Madhav, A.; Scher, K.; Moldawer, N.; et al. Antagonizing CD105 and androgen receptor to target stromal-epithelial interactions for clinical benefit. Mol. Ther. 2023, 31, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Wang, L.; Cui, C.; Chen, H.; Zeng, W.; Li, X. MicroRNA-19a-3p inhibits endothelial dysfunction in atherosclerosis by targeting JCAD. BMC Cardiovasc. Disord. 2024, 24, 394. [Google Scholar] [CrossRef] [PubMed]



| No. | Age | Sex | cTNM | pT | pN | Clinical Presentation | Histological Differentiation | Tumor Thickness (mm) | DOI a (mm) | YK Classification b | Lymphatic Invasion | Vascular Invasion | Perineural Invasion | Prognosis |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 76 | F | T1N0M0 | 2 | 0 | Exophytic | Well | 2.1 | 0.8 | 3 | No | No | No | NED c |
| 2 | 71 | M | T2N0M0 | 2 | 0 | Endophytic | Moderate | 9 | 10 | 3 | No | Yes | No | NED |
| 3 | 86 | F | T4aN0M0 | 4a | 2b | Endophytic | Poor | 16 | 16 | 4C | No | No | Yes | Death of local failure |
| 4 | 77 | F | T1N0M0 | 2 | 0 | Endophytic | Well | 5 | 7 | 3 | No | No | No | NED |
| 5 | 73 | M | T4bN2bM0 | 4a | 2c | Endophytic | Well | 24 | 24 | 3 | No | No | Yes | Death of local and regional failure |
| 6 | 66 | F | T4aN0M0 | 4a | 1 | Endophytic | Moderate | 10 | 9 | 3 | No | No | No | NED |
| 7 | 55 | F | Tis | 2 | 0 | Endophytic | Moderate | 2.8 | 2.8 | 3 | No | No | No | NED |
| 8 | 41 | M | T4bN3bM0 | 4b | 2a | Endophytic | Well | 26 | 20 | 4C | No | Yes | Yes | Death of local failure |
| 9 | 61 | M | T4aN1M0 | 3 | 3b | Endophytic | Moderate | 15 | 13 | 3 | Yes | No | No | NED |
| 10 | 61 | M | T4aN1M0 | 3 | 0 | Endophytic | Well | 20 | 20 | 3 | No | No | No | NED |
| No. a | Age | Sex | Metastatic Lymph Node Diameter (mm) | cTNM | pT | pN | Extranodal Extension | Number of Metastatic Lymph Nodes | Metastasis Site (Level) | Prognosis |
|---|---|---|---|---|---|---|---|---|---|---|
| 3 | 86 | F | 8 | T4aN0M0 | 4a | 2b | No | 2 | 2a | Death of local failure |
| 11 | 75 | M | 3 | T4aN0M0 | 3 | 1 | No | 1 | 1b | NED b |
| 6 | 66 | F | 3 | T4aN0M0 | 4a | 1 | No | 1 | 2a | NED |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shirai, T.; Kakei, Y.; Muraki, Y.; Nagano, T.; Suraya, R.; Uryu, K.; Takeda, D.; Shigeoka, M.; Kimoto, A.; Hasegawa, T.; et al. Expression Analysis of JCAD and IL-33 in Gingival Cancer Tumor Angiogenesis. Cancers 2025, 17, 3732. https://doi.org/10.3390/cancers17233732
Shirai T, Kakei Y, Muraki Y, Nagano T, Suraya R, Uryu K, Takeda D, Shigeoka M, Kimoto A, Hasegawa T, et al. Expression Analysis of JCAD and IL-33 in Gingival Cancer Tumor Angiogenesis. Cancers. 2025; 17(23):3732. https://doi.org/10.3390/cancers17233732
Chicago/Turabian StyleShirai, Tatsuya, Yasumasa Kakei, Yumi Muraki, Tatsuya Nagano, Ratoe Suraya, Kaito Uryu, Daisuke Takeda, Manabu Shigeoka, Akira Kimoto, Takumi Hasegawa, and et al. 2025. "Expression Analysis of JCAD and IL-33 in Gingival Cancer Tumor Angiogenesis" Cancers 17, no. 23: 3732. https://doi.org/10.3390/cancers17233732
APA StyleShirai, T., Kakei, Y., Muraki, Y., Nagano, T., Suraya, R., Uryu, K., Takeda, D., Shigeoka, M., Kimoto, A., Hasegawa, T., Hara, T., Emoto, N., & Akashi, M. (2025). Expression Analysis of JCAD and IL-33 in Gingival Cancer Tumor Angiogenesis. Cancers, 17(23), 3732. https://doi.org/10.3390/cancers17233732






