Epithelial–Mesenchymal Transition in Osteosarcoma as a Key Driver of Pulmonary Metastasis
Simple Summary
Abstract
1. Introduction
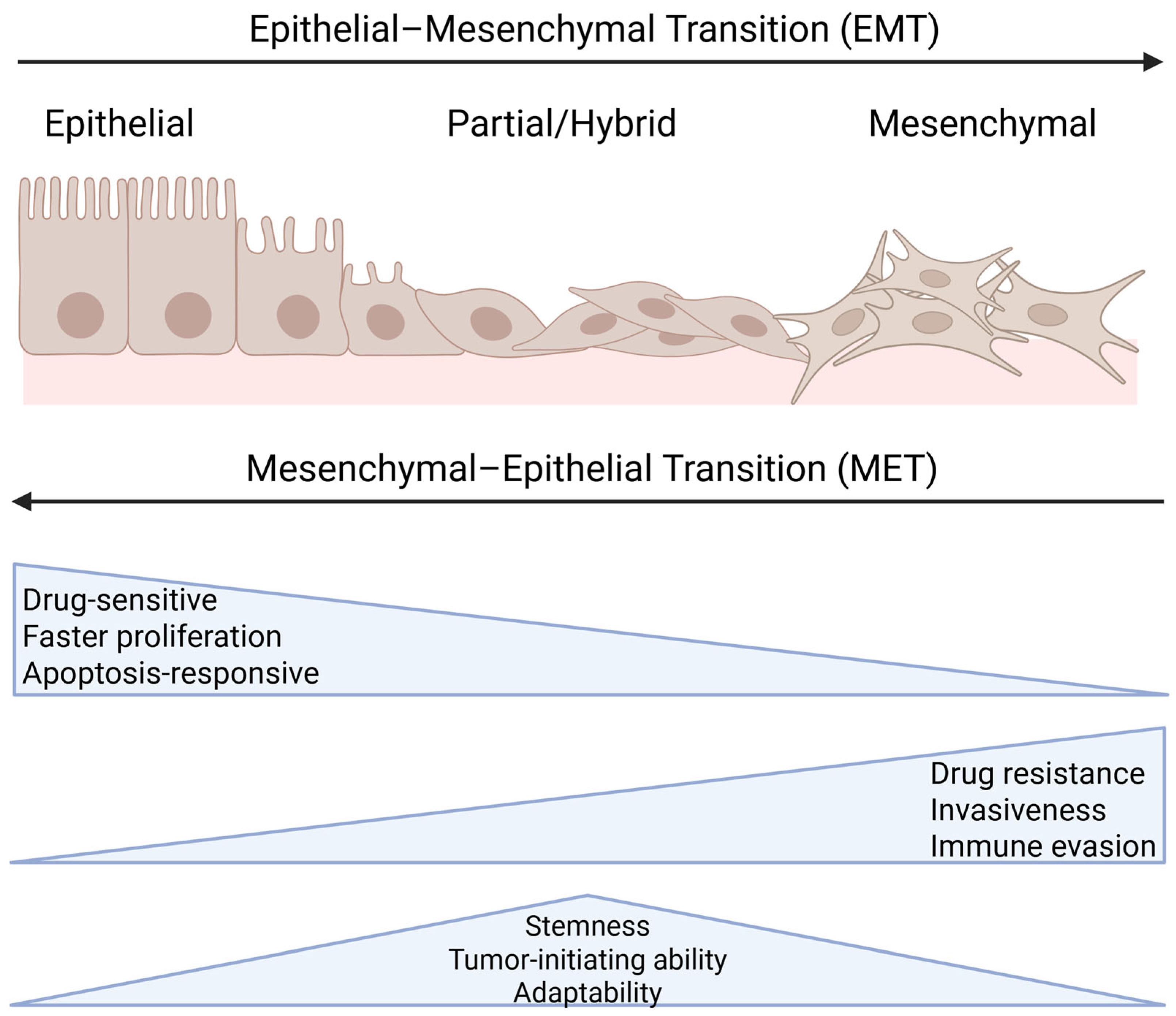
2. EMT and Its Role in Cancer Progression
3. EMT-Related Signaling Pathways in Cancer
3.1. TGF-β Signaling Pathway
3.2. MAPK Signaling Pathway
3.3. Notch Signaling Pathway
3.4. STAT3 Signaling Pathway
3.5. PI3/Akt Signaling Pathway
3.6. Wnt/β-Catenin Signaling Pathway
4. EMT in OS
5. Regulation of EMT in OS: Key Signaling Pathways
5.1. TGF-β Signaling Pathway
5.2. MAPK Signaling Pathway
5.3. Notch Signaling Pathway
5.4. STAT3 Signaling Pathway
5.5. PI3/Akt Signaling Pathway
5.6. Wnt/β-Catenin Signaling Pathway
6. The Interplay Between EMT and the Tumor Microenvironment in OS
7. EMT-Targeting Non-Coding RNAs as Therapeutic Tools
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| OS | Osteosarcoma |
| EMT | Epithelial–Mesenchymal Transition |
| TGF-β | Transforming Growth Factor-beta |
| RTK | Receptor Tyrosine Kinase |
| MAPK | Mitogen-Activated Protein Kinase |
| PI3K | Phosphoinositide 3-Kinase |
| AKT | Protein Kinase B |
| mTOR | Mechanistic Target of Rapamycin |
| miRNA | MicroRNA |
| lncRNA | Long Non-Coding RNA |
| circRNA | Circular RNA |
| ceRNA | Competing Endogenous RNA |
| TF | Transcription Factor |
| ncRNA | Non-Coding RNA |
References
- Belayneh, R.; Fourman, M.S.; Bhogal, S.; Weiss, K.R. Update on osteosarcoma. Curr. Oncol. Rep. 2021, 23, 71. [Google Scholar] [CrossRef]
- Beird, H.C.; Bielack, S.S.; Flanagan, A.M.; Gill, J.; Heymann, D.; Janeway, K.A.; Livingston, J.A.; Roberts, R.D.; Strauss, S.J.; Gorlick, R. Osteosarcoma. Nat. Rev. Dis. Prim. 2022, 8, 77. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Sunkara, R.R.; Parmar, M.Y.; Shaikh, S.; Waghmare, S.K. EMT imparts cancer stemness and plasticity: New perspectives and therapeutic potential. Front. Biosci. 2021, 26, 238–265. [Google Scholar] [CrossRef] [PubMed]
- Strauss, S.J.; Frezza, A.M.; Abecassis, N.; Bajpai, J.; Bauer, S.; Biagini, R.; Bielack, S.; Blay, J.Y.; Bolle, S.; Bonvalot, S.; et al. Bone sarcomas: ESMO-EURACAN-GENTURIS-ERN PaedCan Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2021, 32, 1520–1536. [Google Scholar] [CrossRef]
- Rothzerg, E.; Pfaff, A.L.; Koks, S. Innovative approaches for treatment of osteosarcoma. Exp. Biol. Med. 2022, 247, 310–316. [Google Scholar] [CrossRef]
- Babaei, G.; Aziz, S.G.G.; Jaghi, N.Z.Z. EMT, Cancer stem cells and autophagy; The three main axes of metastasis. Biomed. Pharmacother. 2021, 133, 110909. [Google Scholar] [CrossRef]
- Mittal, V. Epithelial mesenchymal transition in tumor metastasis. Annu. Rev. Pathol. 2018, 13, 395–412. [Google Scholar] [CrossRef]
- Genna, A.; Vanwynsberghe, A.M.; Villard, A.V.; Pottier, C.; Ancel, J.; Polette, M.; Gilles, C. EMT-associated heterogeneity in circulating tumor cells: Sticky friends on the road to metastasis. Cancers 2020, 12, 1632. [Google Scholar] [CrossRef]
- Hinton, K.; Kirk, A.; Paul, P.; Persad, S. Regulation of the epithelial to mesenchymal transition in osteosarcoma. Biomolecules 2023, 13, 398. [Google Scholar] [CrossRef]
- Pastushenko, I.; Mauri, F.; Song, Y.; de Cock, F.; Meeusen, B.; Swedlund, B.; Impens, F.; Van Haver, D.; Opitz, M.; Thery, M.; et al. Fat1 deletion promotes hybrid EMT state, tumour stemness and metastasis. Nature 2021, 589, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Brabletz, S.; Schuhwerk, H.; Brabletz, T.; Stemmler, M.P. Dynamic EMT: A multi-tool for tumor progression. EMBO J. 2021, 40, e108647. [Google Scholar] [CrossRef]
- Fontana, R.; Mestre-Farrera, A.; Yang, J. Update on epithelial-mesenchymal plasticity in cancer progression. Annu. Rev. Pathol. Mech. Dis. 2024, 19, 133–156. [Google Scholar] [CrossRef]
- Brabletz, T.; Jung, A.; Spaderna, S.; Hlubek, F.; Kirchner, T. Opinion: Migrating cancer stem cells—An integrated concept of malignant tumour progression. Nat. Rev. Cancer 2005, 5, 744–749. [Google Scholar] [CrossRef]
- Mani, S.A.; Guo, W.; Liao, M.J.; Eaton, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008, 133, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Burk, U.; Schubert, J.; Wellner, U.; Schmalhofer, O.; Vincan, E.; Spaderna, S.; Brabletz, T. A reciprocal repression between ZEB1 and members of the miR-200 family promotes EMT and invasion in cancer cells. EMBO Rep. 2008, 9, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Gregory, P.A.; Bert, A.G.; Paterson, E.L.; Barry, S.C.; Tsykin, A.; Farshid, G.; Vadas, M.A.; Khew-Goodall, Y.; Goodall, G.J. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat. Cell Biol. 2008, 10, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Bardia, A.; Wittner, B.S.; Stott, S.L.; Smas, M.E.; Ting, D.T.; Isakoff, S.J.; Ciciliano, J.C.; Wells, M.N.; Shah, A.M.; et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 2013, 339, 580–584. [Google Scholar] [CrossRef]
- Zheng, X.; Carstens, J.L.; Kim, J.; Scheible, M.; Kaye, J.; Sugimoto, H.; Wu, C.C.; LeBleu, V.S.; Kalluri, R. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature 2015, 527, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Fuxe, J.; Vincent, T.; Garcia de Herreros, A. Transcriptional crosstalk between TGF-β and stem cell pathways in tumor cell invasion: Role of EMT promoting Smad complexes. Cell Cycle 2010, 9, 2363–2374. [Google Scholar] [CrossRef]
- Pallasch, F.B.; Schumacher, U. Angiotensin inhibition, TGF-β and EMT in cancer. Cancers 2020, 12, 2785. [Google Scholar] [CrossRef]
- Lee, J.H.; Massagué, J. TGF-β in developmental and fibrogenic EMTs. Semin. Cancer Biol. 2022, 86, 136–145. [Google Scholar] [CrossRef]
- Inui, N.; Sakai, S.; Kitagawa, M. Molecular pathogenesis of pulmonary fibrosis, with focus on pathways related to TGF-β and the Ubiquitin-Proteasome pathway. Int. J. Mol. Sci. 2021, 22, 6107. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Lamouille, S.; Derynck, R. TGF-beta-induced epithelial to mesenchymal transition. Cell Res. 2009, 19, 156–172. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Sui, X.; Weng, L.; Liu, Y. SNAIL1: Linking tumor metastasis to immune evasion. Front. Immunol. 2021, 12, 724200. [Google Scholar] [CrossRef]
- Luo, K. Signaling cross talk between TGF-β/Smad and other signaling pathways. Cold Spring Harb. Perspect. Biol. 2017, 9, a022137. [Google Scholar] [CrossRef]
- Derynck, R.; Muthusamy, B.P.; Saeteurn, K.Y. Signaling pathway cooperation in TGF-β-induced epithelial-mesenchymal transition. Curr. Opin. Cell Biol. 2014, 31, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, Y.Y.; Chen, Y.; Wang, J.; Wang, Q.; Lu, H. TGF-β signaling and resistance to cancer therapy. Front. Cell Dev. Biol. 2021, 9, 786728. [Google Scholar] [CrossRef]
- Guo, Y.J.; Pan, W.W.; Liu, S.B.; Shen, Z.F.; Xu, Y.; Hu, L.L. ERK/MAPK signalling pathway and tumorigenesis. Exp. Ther. Med. 2020, 19, 1997–2007. [Google Scholar] [CrossRef]
- Su, J.; Morgani, S.M.; David, C.J.; Wang, Q.; Er, E.E.; Huang, Y.H.; Basnet, H.; Zou, Y.; Shu, W.; Soni, R.K.; et al. TGF-β orchestrates fibrogenic and developmental EMTs via the RAS effector RREB1. Nature 2020, 577, 566–571. [Google Scholar] [CrossRef]
- Lee, J.H.; Basnet, H.; Sanchez-Rivera, F.; Wang, Z.; Li, L.; Massague, J. Mechanistic basis for TGF-β-induced fibrogenic EMTs in metastasis. Cancer Res. 2023, 83, PR004. [Google Scholar]
- Lee, J.H.; Sánchez-Rivera, F.J.; He, L.; Basnet, H.; Chen, F.X.; Spina, E.; Li, L.; Torner, C.; Chan, J.E.; Yarlagadda, D.V.K.; et al. TGF-β and RAS jointly unmask primed enhancers to drive metastasis. Cell 2024, 187, 6182–6199.e29. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Y.; Kong, D.; Sarkar, F.H. The role of Notch signaling pathway in epithelial-mesenchymal transition (EMT) during development and tumor aggressiveness. Curr. Drug Targets 2010, 11, 745–751. [Google Scholar] [CrossRef]
- Zavadil, J.; Cermak, L.; Soto-Nieves, N.; Böttinger, E.P. Integration of TGF-beta/Smad and Jagged1/Notch signalling in epithelial-to-mesenchymal transition. EMBO J. 2004, 23, 1155–1165. [Google Scholar] [CrossRef] [PubMed]
- Jin, W. Role of JAK/STAT3 signaling in the regulation of metastasis, the transition of cancer stem cells, and chemoresistance of cancer by epithelial-mesenchymal transition. Cells 2020, 9, 217. [Google Scholar] [CrossRef]
- Sun, S.; Jin, S.; Guo, R. Role of STAT3 in Resistance of Non-small Cell Lung Cancer. Chin. J. Lung Cancer 2019, 22, 457–463. [Google Scholar] [CrossRef]
- Gong, L.; Wu, Z.; Zhou, Q. The roles of signal transducer and activator of transcription 3 in tumor metastasis. Chin. J. Lung Cancer 2010, 13, 980–984. [Google Scholar] [CrossRef]
- Loh, C.Y.; Chai, J.Y.; Tang, T.F.; Wong, W.F.; Sethi, G.; Shanmugam, M.K.; Chong, P.P.; Looi, C.Y. The E-cadherin and N-cadherin switch in epithelial-to-mesenchymal transition: Signaling, therapeutic implications, and challenges. Cells 2019, 8, 1118. [Google Scholar] [CrossRef]
- Maharati, A.; Moghbeli, M. PI3K/AKT signaling pathway as a critical regulator of epithelial-mesenchymal transition in colorectal tumor cells. Cell Commun. Signal. 2023, 21, 201. [Google Scholar] [CrossRef]
- Worby, C.A.; Dixon, J.E. Pten. Annu. Rev. Biochem. 2014, 83, 641–669. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Chen, J.; He, L.; Stiles, B.L. PTEN: Tumor suppressor and metabolic regulator. Front. Endocrinol. 2018, 9, 338. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Yang, Z.; Lu, N. A new role for the PI3K/Akt signaling pathway in the epithelial-mesenchymal transition. Cell Adhes. Migr. 2015, 9, 317–324. [Google Scholar] [CrossRef]
- Xue, W.; Yang, L.; Chen, C.; Ashrafizadeh, M.; Tian, Y.; Sun, R. Wnt/β-catenin-driven EMT regulation in human cancers. Cell. Mol. Life Sci. 2024, 81, 79. [Google Scholar] [CrossRef]
- Xi, Y.; Chen, Y. Wnt signaling pathway: Implications for therapy in lung cancer and bone metastasis. Cancer Lett. 2014, 353, 8–16. [Google Scholar] [CrossRef]
- Breuer, E.K.; Fukushiro-Lopes, D.; Dalheim, A.; Burnette, M.; Zartman, J.; Kaja, S.; Wells, C.; Campo, L.; Curtis, K.J.; Romero-Moreno, R.; et al. Potassium channel activity controls breast cancer metastasis by affecting β-catenin signaling. Cell Death Dis. 2019, 10, 180. [Google Scholar] [CrossRef]
- Katoh, M.; Nakagama, H. FGF receptors: Cancer biology and therapeutics. Med. Res. Rev. 2014, 34, 280–300. [Google Scholar] [CrossRef]
- Yoshida, G.J. Regulation of heterogeneous cancer-associated fibroblasts: The molecular pathology of activated signaling pathways. J. Exp. Clin. Cancer Res. 2020, 39, 112. [Google Scholar] [CrossRef] [PubMed]
- Pastushenko, I.; Blanpain, C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 2019, 29, 212–226. [Google Scholar] [CrossRef] [PubMed]
- Rho, S.B.; Byun, H.J.; Kim, B.R.; Lee, C.H. Snail promotes cancer cell proliferation via its interaction with the BIRC3. Biomol. Ther. 2022, 30, 380–388. [Google Scholar] [CrossRef]
- Yang, D.; Liu, J.; Qian, H.; Zhuang, Q. Cancer-associated fibroblasts: From basic science to anticancer therapy. Exp. Mol. Med. 2023, 55, 1322–1332. [Google Scholar] [CrossRef]
- De Las Rivas, J.; Brozovic, A.; Izraely, S.; Casas-Pais, A.; Witz, I.P.; Figueroa, A. Cancer drug resistance induced by EMT: Novel therapeutic strategies. Arch. Toxicol. 2021, 95, 2279–2297. [Google Scholar] [CrossRef] [PubMed]
- Ge, R.; Huang, G.M. Targeting transforming growth factor beta signaling in metastatic osteosarcoma. J. Bone Oncol. 2023, 43, 100513. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liu, M. miR-522 stimulates TGF-β/Smad signaling pathway and promotes osteosarcoma tumorigenesis by targeting PPM1A. J. Cell. Biochem. 2019, 120, 18425–18434. [Google Scholar] [CrossRef]
- Zhang, D.; Han, S.; Pan, X.; Li, H.; Zhao, H.; Gao, X.; Wang, S. EFEMP1 binds to STEAP1 to promote osteosarcoma proliferation and invasion via the Wnt/β-catenin and TGF-β/Smad2/3 signal pathways. J. Bone Oncol. 2022, 37, 100458. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Gao, J.; Zheng, L.; Liu, S.; Ye, L.; Lai, H.; Pan, B.; Pan, W.; Lou, C.; Chen, Z.; et al. TGF-β inhibitor RepSox suppresses osteosarcoma via the JNK/Smad3 signaling pathway. Int. J. Oncol. 2021, 59, 84. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Wang, D.; Zhang, L.; Yang, P.; Wang, J.; Liu, Q.; Yan, F.; Lin, F. The TGFβ-miR-499a-SHKBP1 pathway induces resistance to EGFR inhibitors in osteosarcoma cancer stem cell-like cells. J. Exp. Clin. Cancer Res. 2019, 38, 226. [Google Scholar] [CrossRef]
- Sun, Y.; Jiang, X.; Lu, Y.; Zhu, J.; Yu, L.; Ma, B.; Zhang, Q. Oridonin prevents epithelial-mesenchymal transition and TGF-β1-induced epithelial-mesenchymal transition by inhibiting TGF-β1/Smad2/3 in osteosarcoma. Chem. Biol. Interact. 2018, 296, 57–64. [Google Scholar] [CrossRef]
- Jiang, X.; Zhang, Z.; Song, C.; Deng, H.; Yang, R.; Zhou, L.; Sun, Y.; Zhang, Q. Glaucocalyxin A reverses EMT and TGF-β1-induced EMT by inhibiting TGF-β1/Smad2/3 signaling pathway in osteosarcoma. Chem. Biol. Interact. 2019, 307, 158–166. [Google Scholar] [CrossRef]
- Liu, F.; Wang, K.; Zhang, L.; Yang, Y.L. Bone morphogenetic protein and activin membrane-bound inhibitor suppress bone cancer progression in MG63 and SAOS cells via regulation of the TGF-β-induced EMT signaling pathway. Oncol. Lett. 2018, 16, 5113–5121. [Google Scholar] [CrossRef]
- Xie, C.; Liu, S.; Wu, B.; Zhao, Y.; Chen, B.; Guo, J.; Qiu, S.; Cao, Y.M. miR-19 promotes cell proliferation, invasion, migration, and EMT by inhibiting SPRED2-mediated autophagy in osteosarcoma cells. Cell Transplant. 2020, 29, 963689720962460. [Google Scholar] [CrossRef]
- Gao, G.; Tian, Z.; Zhu, H.Y.; Ouyang, X.Y. miRNA-133b targets FGFR1 and presents multiple tumor suppressor activities in osteosarcoma. Cancer Cell Int. 2018, 18, 210. [Google Scholar] [CrossRef]
- Fan, M.K.; Zhang, G.C.; Chen, W.; Qi, L.L.; Xie, M.F.; Zhang, Y.Y.; Wang, L.; Zhang, Q. Siglec-15 promotes tumor progression in osteosarcoma via DUSP1/MAPK pathway. Front. Oncol. 2021, 11, 710689. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.M.; Park, B.S.; Kang, H.K.; Park, H.R.; Yu, S.B.; Kim, I.R. Delphinidin induces apoptosis and inhibits epithelial-to-mesenchymal transition via the ERK/p38 MAPK-signaling pathway in human osteosarcoma cell lines. Environ. Toxicol. 2018, 33, 640–649. [Google Scholar] [CrossRef]
- Yuan, X.H.; Zhang, P.; Yu, T.T.; Huang, H.K.; Zhang, L.L.; Yang, C.M.; Tan, T.; Yang, S.D.; Luo, X.J.; Luo, J.Y. Lycorine inhibits tumor growth of human osteosarcoma cells by blocking Wnt/β-catenin, ERK1/2/MAPK and PI3K/AKT signaling pathway. Am. J. Transl. Res. 2020, 12, 5381–5398. [Google Scholar]
- Huang, H.; Nie, C.; Qin, X.; Zhou, J.; Zhang, L. Diosgenin inhibits the epithelial-mesenchymal transition initiation in osteosarcoma cells via the p38MAPK signaling pathway. Oncol. Lett. 2019, 18, 4278–4287. [Google Scholar] [CrossRef] [PubMed]
- Niu, N.K.; Wang, Z.L.; Pan, S.T.; Ding, H.Q.; Au, G.H.T.; He, Z.X.; Zhou, Z.W.; Xiao, G.; Yang, Y.X.; Zhang, X.; et al. Pro-apoptotic and pro-autophagic effects of the Aurora kinase A inhibitor alisertib (MLN8237) on human osteosarcoma U-2 OS and MG-63 cells through the activation of mitochondria-mediated pathway and inhibition of p38 MAPK/PI3K/Akt/mTOR signaling pathway. Drug Des. Dev. Ther. 2015, 9, 1555–1584. [Google Scholar] [CrossRef]
- Yang, J.; Guo, W.; Wang, L.; Yu, L.; Mei, H.; Fang, S.; Chen, A.; Liu, Y.; Xia, K.; Liu, G. Notch signaling is important for epithelial-mesenchymal transition induced by low concentrations of doxorubicin in osteosarcoma cell lines. Oncol. Lett. 2017, 13, 2260–2268. [Google Scholar] [CrossRef][Green Version]
- Dai, G.; Liu, G.; Zheng, D.; Song, Q. Inhibition of the Notch signaling pathway attenuates progression of cell motility, metastasis, and epithelial-to-mesenchymal transition-like phenomena induced by low concentrations of cisplatin in osteosarcoma. Eur. J. Pharmacol. 2021, 899, 174058. [Google Scholar] [CrossRef]
- Su, H.; Zhu, G.; Rong, X.; Zhou, Y.; Jiang, P.; Chen, P. Upregulation of ATG4A promotes osteosarcoma cell epithelial-to-mesenchymal transition through the Notch signaling pathway. Int. J. Clin. Exp. Pathol. 2017, 10, 7975–7982. [Google Scholar]
- Liang, G.; Duan, C.; He, J.; Shi, L. Spindle and kinetochore-related complex subunit 3 has a protumour function in osteosarcoma by activating the Notch pathway. Toxicol. Appl. Pharmacol. 2024, 483, 116826. [Google Scholar] [CrossRef]
- Hu, Y.Y.; Zheng, M.H.; Zhang, R.; Liang, Y.M.; Han, H. Notch signaling pathway and cancer metastasis. Adv. Exp. Med. Biol. 2012, 727, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Tao, Y.; Xiao, S.; Li, X.; Fang, K.; Wen, J.; He, P.; Zeng, M. LncRNA KIAA0087 suppresses the progression of osteosarcoma by mediating the SOCS1/JAK2/STAT3 signaling pathway. Exp. Mol. Med. 2023, 55, 831–843. [Google Scholar] [CrossRef]
- Zhang, T.; Li, S.; Li, J.; Yin, F.; Hua, Y.; Wang, Z.; Wang, H.; Zuo, D.; Xu, J.; Cai, Z. Pectolinarigenin acts as a potential anti-osteosarcoma agent via mediating SHP-1/JAK2/STAT3 signaling. Biomed. Pharmacother. 2022, 153, 113323. [Google Scholar] [CrossRef]
- Chen, Y.; Li, J.; Xiao, J.K.; Xiao, L.; Xu, B.W.; Li, C. The lncRNA NEAT1 promotes the epithelial-mesenchymal transition and metastasis of osteosarcoma cells by sponging miR-483 to upregulate STAT3 expression. Cancer Cell Int. 2021, 21, 90. [Google Scholar] [CrossRef]
- Han, Y.; Guo, W.; Ren, T.; Huang, Y.; Wang, S.; Liu, K.; Zheng, B.; Yang, K.; Zhang, H.; Liang, X. Tumor-associated macrophages promote lung metastasis and induce epithelial-mesenchymal transition in osteosarcoma by activating the COX-2/STAT3 axis. Cancer Lett. 2019, 440, 116–125. [Google Scholar] [CrossRef]
- Hu, Y.; Luo, X.; Zhou, J.; Chen, S.; Gong, M.; Deng, Y.; Zhang, H. Piperlongumine inhibits the progression of osteosarcoma by downregulating the SOCS3/JAK2/STAT3 pathway via miR-30d-5p. Life Sci. 2021, 277, 119501. [Google Scholar] [CrossRef]
- Zheng, B.; Ren, T.; Huang, Y.; Guo, W. Apatinib inhibits migration and invasion as well as PD-L1 expression in osteosarcoma by targeting STAT3. Biochem. Biophys. Res. Commun. 2018, 495, 1695–1701. [Google Scholar] [CrossRef] [PubMed]
- Kong, G.; Jiang, Y.; Sun, X.; Cao, Z.; Zhang, G.; Zhao, Z.; Zhao, Y.; Yu, Q.; Cheng, G. Irisin reverses the IL-6 induced epithelial-mesenchymal transition in osteosarcoma cell migration and invasion through the STAT3/Snail signaling pathway. Oncol. Rep. 2017, 38, 2647–2656. [Google Scholar] [CrossRef] [PubMed]
- Sadrkhanloo, M.; Paskeh, M.D.A.; Hashemi, M.; Raesi, R.; Bahonar, A.; Nakhaee, Z.; Entezari, M.; Beig Goharrizi, M.A.S.; Salimimoghadam, S.; Ren, J.; et al. New emerging targets in osteosarcoma therapy: PTEN and PI3K/Akt crosstalk in carcinogenesis. Pathol. Res. Prim. 2023, 251, 154902. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Wei, R.; Zhou, J.; Wu, K.; Li, J. Emerging roles of long non-coding RNAs in osteosarcoma. Front. Mol. Biosci. 2024, 11, 1327459. [Google Scholar] [CrossRef]
- Ye, F.; Tian, L.; Zhou, Q.; Feng, D. LncRNA FER1L4 induces apoptosis and suppresses EMT and the activation of PI3K/AKT pathway in osteosarcoma cells via inhibiting miR-18a-5p to promote SOCS5. Gene 2019, 721, 144093. [Google Scholar] [CrossRef]
- Tong, C.J.; Deng, Q.C.; Ou, D.J.; Long, X.; Liu, H.; Huang, K. LncRNA RUSC1-AS1 promotes osteosarcoma progression through regulating the miR-340-5p and PI3K/AKT pathway. Aging 2021, 13, 20116–20130. [Google Scholar] [CrossRef]
- Ma, L.; Zhang, L.; Guo, A.; Liu, L.C.; Yu, F.; Diao, N.; Xu, C.; Wang, D. Overexpression of FER1L4 promotes the apoptosis and suppresses epithelial-mesenchymal transition and stemness markers via activating PI3K/AKT signaling pathway in osteosarcoma cells. Pathol. Res. Prim. 2019, 215, 152412. [Google Scholar] [CrossRef]
- Gao, L.F.; Jia, S.; Zhang, Q.M.; Xia, Y.F.; Li, C.J.; Li, Y.H. MicroRNA-802 promotes the progression of osteosarcoma through targeting p27 and activating PI3K/AKT pathway. Clin. Transl. Oncol. 2022, 24, 266–275. [Google Scholar] [CrossRef]
- Huang, Y.; Xu, Y.Q.; Feng, S.Y.; Zhang, X.; Ni, J.D. LncRNA TDRG1 promotes proliferation, invasion and epithelial-mesenchymal transformation of osteosarcoma through PI3K/AKT signal pathway. Cancer Manag. Res. 2020, 12, 4531–4540. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Zou, M.; Shen, T.; Duan, S. Dysfunction of miR-802 in tumors. J. Clin. Lab. Anal. 2021, 35, e23989. [Google Scholar] [CrossRef]
- Fan, H.P.; Wang, S.Y.; Shi, Y.Y.; Sun, J. MicroRNA-340-5p inhibits the malignant phenotypes of osteosarcoma by directly targeting NRF2 and deactivating the PI3K/AKT pathway. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 3661–3669. [Google Scholar] [CrossRef]
- Yao, P.; Ni, Y.; Liu, C. Long Non-Coding RNA 691 regulated PTEN/PI3K/AKT signaling pathway in osteosarcoma through miRNA-9-5p. OncoTargets Ther. 2020, 13, 4597–4606. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Jin, D.; Zhuo, Z.; Zhang, B.; Chen, K. MiR-1224–5p Activates Autophagy, Cell Invasion and Inhibits Epithelial-to-Mesenchymal Transition in Osteosarcoma Cells by Directly Targeting PLK1 through PI3K/AKT/mTOR Signaling Pathway. OncoTargets Ther. 2020, 13, 11807–11818. [Google Scholar] [CrossRef]
- Cui, Y.; Dong, Y.Y. ZCCHC12 promotes the progression of osteosarcoma via PI3K/AKT pathway. Aging 2022, 14, 7505–7516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Liu, H.; Wang, W.; Xu, G.; Yin, C.; Wang, S. STEAP2 promotes osteosarcoma progression by inducing epithelial-mesenchymal transition via the PI3K/AKT/mTOR signaling pathway and is regulated by EFEMP2. Cancer Biol. Ther. 2022, 23, 1–16. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, S.; Chen, J.; Liu, H.; Lu, J.; Jiang, H.; Huang, A.; Chen, Y. Fibulin-4 promotes osteosarcoma invasion and metastasis by inducing epithelial to mesenchymal transition via the PI3K/Akt/mTOR pathway. Int. J. Oncol. 2017, 50, 1513–1530. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Huang, G.; Zhong, X.; Yang, Y.; Ye, J. N6-(2-hydroxyethyl)-adenosine (HEA) exhibits antitumor activity for osteosarcoma progression by regulating IGF1 signaling. Fitoterapia 2025, 180, 106319. [Google Scholar] [CrossRef]
- Liu, G.; An, L.; Zhang, H.; Du, P.; Sheng, Y. Activation of CXCL6/CXCR1/2 axis promotes the growth and metastasis of osteosarcoma cells in vitro and in vivo. Front. Pharmacol. 2019, 10, 307. [Google Scholar] [CrossRef]
- Han, G.D.; Dai, J.; Hui, H.X.; Zhu, J. ALOX5AP suppresses osteosarcoma progression via Wnt/β-catenin/EMT pathway and associates with clinical prognosis and immune infiltration. J. Orthop. Surg. Res. 2023, 18, 446. [Google Scholar] [CrossRef]
- Liang, K.; Liao, L.; Liu, Q.; Ouyang, Q.; Jia, L.; Wu, G. microRNA-377-3p inhibits osteosarcoma progression by targeting CUL1 and regulating Wnt/β-catenin signaling pathway. Clin. Transl. Oncol. 2021, 23, 2350–2357. [Google Scholar] [CrossRef]
- Kong, H.; Yu, W.; Chen, Z.; Li, H.; Ye, G.; Hong, J.; Xie, Z.; Chen, K.; Wu, Y.; Shen, H. CCR9 initiates epithelial-mesenchymal transition by activating Wnt/β-catenin pathways to promote osteosarcoma metastasis. Cancer Cell Int. 2021, 21, 648. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Zhou, T.; Chen, H.; Li, C.; Jiang, Z.; Lao, L.; Kahn, S.A.; Duarte, M.E.L.; Zhao, J.; Daubs, M.D.; et al. Bone morphogenetic protein-2 promotes osteosarcoma growth by promoting epithelial-mesenchymal transition (EMT) through the Wnt/β-catenin signaling pathway. J. Orthop. Res. 2019, 37, 1638–1648. [Google Scholar] [CrossRef]
- Bai, S.; Li, Y.; Wang, Y.; Zhou, G.; Liu, C.; Xiong, W.; Chen, J. Long non-coding RNA MINCR regulates the growth and metastasis of human osteosarcoma cells via Wnt/β-catenin signaling pathway. Acta Biochim. Pol. 2022, 69, 551–557. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, P. lncRNA-CASC15 promotes osteosarcoma proliferation and metastasis by regulating epithelial-mesenchymal transition via the Wnt/β-catenin signaling pathway. Oncol. Rep. 2021, 45, 76. [Google Scholar] [CrossRef]
- Li, K.; Yu, H.; Zhao, C.; Li, J.; Tan, R.; Chen, L. Down-regulation of PRR11 affects the proliferation, migration and invasion of osteosarcoma by inhibiting the Wnt/β-catenin pathway. J. Cancer 2021, 12, 6656–6664. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Zheng, D.; Pi, W.; Qiu, Y.; Xia, K.; Guo, W. Isoquercitrin restrains the proliferation and promotes apoptosis of human osteosarcoma cells by inhibiting the Wnt/β-catenin pathway. J. Bone Oncol. 2023, 38, 100468. [Google Scholar] [CrossRef]
- Mao, X.; Jin, Y.; Feng, T.; Wang, H.; Liu, D.; Zhou, Z.; Yan, Q.; Yang, H.; Yang, J.; Yang, J.; et al. Ginsenoside Rg3 inhibits the growth of osteosarcoma and attenuates metastasis through the Wnt/β-catenin and EMT signaling pathway. Evid.-Based Complement. Altern. Med. 2020, 2020, 6065124. [Google Scholar] [CrossRef]
- He, G.; Nie, J.J.; Liu, X.; Ding, Z.; Luo, P.; Liu, Y.; Zhang, B.W.; Wang, R.; Liu, X.; Hai, Y.; et al. Zinc oxide nanoparticles inhibit osteosarcoma metastasis by downregulating β-catenin via HIF-1α/BNIP3/LC3B-mediated mitophagy pathway. Bioact. Mater. 2023, 19, 690–702. [Google Scholar] [CrossRef]
- Taki, M.; Abiko, K.; Ukita, M.; Murakami, R.; Yamanoi, K.; Yamaguchi, K.; Hamanishi, J.; Baba, T.; Matsumura, N.; Mandai, M. Tumor immune microenvironment during epithelial-mesenchymal transition. Clin. Cancer Res. 2021, 27, 4669–4679. [Google Scholar] [CrossRef]
- Huang, X.; Wang, L.; Guo, H.; Zhang, W.; Shao, Z. Single-cell transcriptomics reveals the regulative roles of cancer associated fibroblasts in tumor immune microenvironment of recurrent osteosarcoma. Theranostics 2022, 12, 5877–5887. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Li, J.; Gao, S.; Han, Y.; Han, X.; Wu, Y.; Bi, J.; Xu, M.; Bi, W. Sulfatinib, a novel multi-targeted tyrosine kinase inhibitor of FGFR1, CSF1R, and VEGFR1-3, suppresses osteosarcoma proliferation and invasion via dual role in tumor cells and tumor microenvironment. Front. Oncol. 2023, 13, 1158857. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Ding, G.; Jiang, W.; Fan, X.; Zhu, L. Effect of tumor-associated macrophages on lncRNA PURPL/miR-363/PDZD2 axis in osteosarcoma cells. Cell Death Discov. 2021, 7, 307. [Google Scholar] [CrossRef]
- Hou, C.; Lu, M.; Lei, Z.; Dai, S.; Chen, W.; Du, S.; Jin, Q.; Zhou, Z.; Li, H. HMGB1 positive feedback loop between cancer cells and tumor-associated macrophages promotes osteosarcoma migration and invasion. Lab. Investig. 2023, 103, 100054. [Google Scholar] [CrossRef]
- Cheng, Z.; Wang, L.; Wu, C.; Huang, L.; Ruan, Y.; Xue, W. Tumor-derived exosomes induced M2 macrophage polarization and promoted the metastasis of osteosarcoma cells through Tim-3. Arch. Med. Res. 2021, 52, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liang, X.; Liang, H.; Wang, B. SENP1/HIF-1α feedback loop modulates hypoxia-induced cell proliferation, invasion, and EMT in human osteosarcoma cells. J. Cell. Biochem. 2018, 119, 1819–1826. [Google Scholar] [CrossRef]
- Zeng, X.; Liu, S.; Yang, H.; Jia, M.; Liu, W.; Zhu, W. Synergistic anti-tumour activity of ginsenoside Rg3 and doxorubicin on proliferation, metastasis and angiogenesis in osteosarcoma by modulating mTOR/HIF-1α/VEGF and EMT signalling pathways. J. Pharm. Pharmacol. 2023, 75, 1405–1417. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, H.; Liu, M.; Lin, F.; Hua, J. Resveratrol abrogates the effects of hypoxia on cell proliferation, invasion and EMT in osteosarcoma cells through downregulation of the HIF-1α protein. Mol. Med. Rep. 2015, 11, 1975–1981. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, Y.; Zou, J.; Yan, L.; Du, W.; Zhang, Y.; Sun, H.; Lu, P.; Geng, S.; Gu, R.; et al. Tetrahydrocurcumin induces mesenchymal-epithelial transition and suppresses angiogenesis by targeting HIF-1α and autophagy in human osteosarcoma. Oncotarget 2017, 8, 91134–91149. [Google Scholar] [CrossRef]
- Zhu, S.T.; Wang, X.; Wang, J.Y.; Xi, G.H.; Liu, Y. Downregulation of miR-22 contributes to epithelial-mesenchymal transition in osteosarcoma by targeting Twist1. Front. Oncol. 2020, 10, 406. [Google Scholar] [CrossRef]
- Zhao, X.; Xu, Y.; Sun, X.; Ma, Y.; Zhang, Y.; Wang, Y.; Guan, H.; Jia, Z.; Li, Y.; Wang, Y. miR-17-5p promotes proliferation and epithelial-mesenchymal transition in human osteosarcoma cells by targeting SRC kinase signaling inhibitor 1. J. Cell. Biochem. 2019, 120, 5495–5504. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, W.; Mao, J.; Xu, Z.; Fan, M. miR-186-5p functions as a tumor suppressor in human osteosarcoma by targeting FOXK1. Cell. Physiol. Biochem. 2019, 52, 553–564. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, M.; Chen, Q.; Zhang, Q. Downregulation of microRNA-145 promotes epithelial-mesenchymal transition via regulating Snail in osteosarcoma. Cancer Gene Ther. 2017, 24, 83–88. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, H.; Liu, W.; Fang, L.; Qian, Z.; Kong, R.; Zhang, Q.; Li, J.; Cao, X. miR-766-3p targeting BCL9L suppressed tumorigenesis, epithelial-mesenchymal transition, and metastasis through the β-catenin signaling pathway in osteosarcoma cells. Front. Cell Dev. Biol. 2020, 8, 594135. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, J.; Meng, F.; Zhu, H.; Yan, H.; Guo, Y.; Zhang, S. MicroRNA-93 promotes the tumorigenesis of osteosarcoma by targeting TIMP2. Biosci. Rep. 2019, 39, BSR20191237. [Google Scholar] [CrossRef]
- Yu, X.; Wang, X.; Xu, F.; Zhang, X.; Wang, M.; Zhou, R.; Sun, Z.; Pan, X.; Feng, L.; Zhang, W.; et al. Mir-615-3p promotes osteosarcoma progression via the SESN2/AMPK/mTOR pathway. Cancer Cell Int. 2024, 24, 411. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Guo, D.; Cao, Z.; Xiao, L.; Wang, G. Inhibitory effect of MicroRNA-107 on osteosarcoma malignancy through regulation of Wnt/β-catenin signaling in vitro. Cancer Investig. 2018, 36, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Lin, J.; He, L.; Huang, J.; Liu, Q. TNF-α/miR-155 axis induces the transformation of osteosarcoma cancer stem cells independent of TP53INP1. Gene 2020, 726, 144224. [Google Scholar] [CrossRef]
- Xu, M.; Jin, H.; Xu, C.X.; Sun, B.; Song, Z.G.; Bi, W.Z.; Wang, Y. miR-382 inhibits osteosarcoma metastasis and relapse by targeting Y box-binding protein 1. Mol. Ther. 2015, 23, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhou, W.; Yang, Z.; Li, J.; Jin, Y. miR-185-5p represses cells growth and metastasis of osteosarcoma via targeting cathepsin E. Int. J. Toxicol. 2022, 41, 115–125. [Google Scholar] [CrossRef]
- Waresijiang, N.; Sun, J.; Abuduaini, R.; Jiang, T.; Zhou, W.; Yuan, H. The downregulation of miR-125a-5p functions as a tumor suppressor by directly targeting MMP-11 in osteosarcoma. Mol. Med. Rep. 2016, 13, 4859–4864. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sun, N.; Zhang, Z.; Zhou, Y.; Liu, H.; Zhou, X.; Zhang, Y.; Zhao, Y. Overexpression pattern of miR-301b in osteosarcoma and its relevance with osteosarcoma cellular behaviors via modulating SNX10. Biochem. Genet. 2023, 61, 87–100. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, L.; Zhang, X.; Xing, C.; Liu, R.; Zhang, F. MiR-196a promoted cell migration, invasion and the epithelial-mesenchymal transition by targeting HOXA5 in osteosarcoma. Cancer Biomark. 2020, 29, 291–298. [Google Scholar] [CrossRef]
- Wang, X.; Li, C.; Yao, W.; Tian, Z.; Liu, Z.; Ge, H. MicroRNA-761 suppresses tumor progression in osteosarcoma via negatively regulating ALDH1B1. Life Sci. 2020, 262, 118544. [Google Scholar] [CrossRef]
- Wang, D.; Bao, F.; Teng, Y.; Li, Q.; Li, J. MicroRNA-506-3p initiates mesenchymal-to-epithelial transition and suppresses autophagy in osteosarcoma cells by directly targeting SPHK1. Biosci. Biotechnol. Biochem. 2019, 83, 836–844. [Google Scholar] [CrossRef]
- Sun, L.; Wang, P.; Zhang, Z.; Zhang, K.; Xu, Z.; Li, S.; Mao, J. MicroRNA-615 functions as a tumor suppressor in osteosarcoma through the suppression of HK2. Oncol. Lett. 2020, 20, 226. [Google Scholar] [CrossRef]
- Sun, L.; Liu, M.; Luan, S.; Shi, Y.; Wang, Q. MicroRNA-744 promotes carcinogenesis in osteosarcoma through targeting LATS2. Oncol. Lett. 2019, 18, 2523–2529. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.K.; Guo, Y.H. MiR-139-5p suppresses osteosarcoma cell growth and invasion through regulating DNMT1. Biochem. Biophys. Res. Commun. 2018, 503, 459–466. [Google Scholar] [CrossRef]
- Shen, S.; Huang, K.; Wu, Y.; Ma, Y.; Wang, J.; Qin, F.; Ma, J. A miR-135b-TAZ positive feedback loop promotes epithelial-mesenchymal transition (EMT) and tumorigenesis in osteosarcoma. Cancer Lett. 2017, 407, 32–44. [Google Scholar] [CrossRef]
- Raimondi, L.; Gallo, A.; Cuscino, N.; De Luca, A.; Costa, V.; Carina, V.; Bellavia, D.; Bulati, M.; Alessandro, R.; Fini, M.; et al. Potential anti-metastatic role of the novel miR-CT3 in tumor angiogenesis and osteosarcoma invasion. Int. J. Mol. Sci. 2022, 23, 705. [Google Scholar] [CrossRef]
- Mo, J.; Zheng, T.; Lei, L.; Dai, P.; Liu, J.; He, H.; Shi, J.; Chen, X.; Guo, T.; Yuan, B.; et al. MicroRNA-1253 suppresses cell proliferation migration and invasion of osteosarcoma by targeting MMP9. Technol. Cancer Res. Treat. 2021, 20, 1533033821995278. [Google Scholar] [CrossRef]
- Lv, S.; Guan, M. miRNA-1284, a regulator of HMGB1, inhibits cell proliferation and migration in osteosarcoma. Biosci. Rep. 2018, 38, BSR20171675. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, J.; Xing, C.; Wei, S.; Guo, N.; Wang, Y. miR-486 inhibited osteosarcoma cells invasion and epithelial-mesenchymal transition by targeting PIM1. Cancer Biomark. 2018, 23, 269–277. [Google Scholar] [CrossRef]
- Liu, X.; Liang, Z.; Gao, K.; Li, H.; Zhao, G.; Wang, S.; Fang, J. MicroRNA-128 inhibits EMT of human osteosarcoma cells by directly targeting integrin α2. Tumor Biol. 2016, 37, 7951–7957. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, B.; Duan, A.; Shen, K.; Zhang, Q.; Tang, X.; Wei, Y.; Tang, J.; Zhang, S. Exosomal transfer of miR-769-5p promotes osteosarcoma proliferation and metastasis by targeting DUSP16. Cancer Cell Int. 2021, 21, 541. [Google Scholar] [CrossRef]
- Liu, R.; Shen, L.; Qu, N.; Zhao, X.; Wang, J.; Geng, J. MiR-19a promotes migration and invasion by targeting RHOB in osteosarcoma. OncoTargets Ther. 2019, 12, 7801–7808. [Google Scholar] [CrossRef]
- Li, G.W.; Yan, X. Lower miR-630 expression predicts poor prognosis of osteosarcoma and promotes cell proliferation, migration and invasion by targeting PSMC2. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 1915–1925. [Google Scholar] [CrossRef]
- Jin, C.; Feng, Y.; Ni, Y.; Shan, Z. MicroRNA-610 suppresses osteosarcoma oncogenicity via targeting TWIST1 expression. Oncotarget 2017, 8, 56174–56184. [Google Scholar] [CrossRef][Green Version]
- Hu, Y.; Liang, D.; Chen, X.; Chen, L.; Bai, J.; Li, H.; Yin, C.; Zhong, W. MiR-671-5p negatively regulates SMAD3 to inhibit migration and invasion of osteosarcoma cells. J. South. Med. Univ. 2021, 41, 1562–1568. [Google Scholar] [CrossRef]
- He, F.; Fang, L.; Yin, Q. miR-363 acts as a tumor suppressor in osteosarcoma cells by inhibiting PDZD2. Oncol. Rep. 2019, 41, 2729–2738. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Zhang, J.; Pang, J.; He, S.; Li, G.; Chong, Y.; Li, C.; Jiao, Z.; Zhang, S.; Shao, M. MicroRNA-503 represses epithelial-mesenchymal transition and inhibits metastasis of osteosarcoma by targeting c-myb. Tumor Biol. 2016, 37, 9181–9187. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Zhang, N.; Liu, S.; Pang, Z.; Chen, Z. By targeting TRAF6, miR-140-3p inhibits TGF-β1-induced human osteosarcoma epithelial-to-mesenchymal transition, migration, and invasion. Biotechnol. Lett. 2020, 42, 2123–2133. [Google Scholar] [CrossRef]
- Gong, H.L.; Tao, Y.; Mao, X.Z.; Song, D.Y.; You, D.; Ni, J.D. MicroRNA-29a suppresses the invasion and migration of osteosarcoma cells by regulating the SOCS1/NF-κB signalling pathway through negatively targeting DNMT3B. Int. J. Mol. Med. 2019, 44, 1219–1232. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Tang, Y.; Wang, J.; Guo, Z. MicroRNA-181c suppresses the biological progression of osteosarcoma via targeting SMAD7 and regulating transforming growth factor-β (TGF-β) signaling pathway. Med. Sci. Monit. 2019, 25, 4801–4810. [Google Scholar] [CrossRef]
- Feng, T.; Zhu, Z.; Jin, Y.; Wang, H.; Mao, X.; Liu, D.; Li, Y.; Lu, L.; Zuo, G. The microRNA-708-5p/ZEB1/EMT axis mediates the metastatic potential of osteosarcoma. Oncol. Rep. 2020, 43, 491–502. [Google Scholar] [CrossRef]
- Duan, N.; Hu, X.; Yang, X.; Cheng, H.; Zhang, W. MicroRNA-370 directly targets FOXM1 to inhibit cell growth and metastasis in osteosarcoma cells. Int. J. Clin. Exp. Pathol. 2015, 8, 10250–10260. [Google Scholar]
- Dong, C.; Du, Q.; Wang, Z.; Wang, Y.; Wu, S.; Wang, A. MicroRNA-665 suppressed the invasion and metastasis of osteosarcoma by directly inhibiting RAB23. Am. J. Transl. Res. 2016, 8, 4975–4981. [Google Scholar] [PubMed]
- Deng, Y.; Luan, F.; Zeng, L.; Zhang, Y.; Ma, K. MiR-429 suppresses the progression and metastasis of osteosarcoma by targeting ZEB1. Excli J. 2017, 16, 618–627. [Google Scholar] [CrossRef]
- Cheng, G.; Xu, D.; Chu, K.; Cao, Z.; Sun, X.; Yang, Y. The effects of MiR-214-3p and Irisin/FNDC5 on the biological behavior of osteosarcoma cells. Cancer Biother. Radiopharm. 2020, 35, 92–100. [Google Scholar] [CrossRef]
- Chen, Z.; Zhao, G.; Zhang, Y.; Ma, Y.; Ding, Y.; Xu, N. MiR-199b-5p promotes malignant progression of osteosarcoma by regulating HER2. J. Buon 2018, 23, 1816–1824. [Google Scholar]
- Chen, J.; Yan, D.; Wu, W.; Zhu, J.; Ye, W.; Shu, Q. MicroRNA-130a promotes the metastasis and epithelial-mesenchymal transition of osteosarcoma by targeting PTEN. Oncol. Rep. 2016, 35, 3285–3292. [Google Scholar] [CrossRef]
- Cao, R.; Shao, J.; Hu, Y.; Wang, L.; Li, Z.; Sun, G.; Gao, X. microRNA-338-3p inhibits proliferation, migration, invasion, and EMT in osteosarcoma cells by targeting activator of 90 kDa heat shock protein ATPase homolog 1. Cancer Cell Int. 2018, 18, 49. [Google Scholar] [CrossRef]
- Cao, J.; Han, X.; Qi, X.; Jin, X.; Li, X. TUG1 promotes osteosarcoma tumorigenesis by upregulating EZH2 expression via miR-144-3p. Int. J. Oncol. 2017, 51, 1115–1123. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Ge, S.; Li, M. MiR-451a promotes cell growth, migration and EMT in osteosarcoma by regulating YTHDC1-mediated m6A methylation to activate the AKT/mTOR signaling pathway. J. Bone Oncol. 2022, 33, 100412. [Google Scholar] [CrossRef]
- Bi, W.; Yang, M.; Xing, P.; Huang, T. MicroRNA miR-331-3p suppresses osteosarcoma progression via the Bcl-2/Bax and Wnt/β-catenin signaling pathways and the epithelial-mesenchymal transition by targeting N-acetylglucosaminyltransferase I (MGAT1). Bioengineered 2022, 13, 14159–14174. [Google Scholar] [CrossRef]
- Zhu, S.; Liu, Y.; Wang, X.; Wang, J.; Xi, G. lncRNA SNHG10 promotes the proliferation and invasion of osteosarcoma via Wnt/β-catenin signaling. Mol. Ther. Nucleic Acids 2020, 22, 957–970. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, X.; Yang, H. LINC00612 functions as a ceRNA for miR-214-5p to promote the proliferation and invasion of osteosarcoma in vitro and in vivo. Exp. Cell Res. 2020, 392, 112012. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, Z.; Zhou, Z.; Xu, X.; Yang, H. Silencing of long non-coding RNA LINC00607 prevents tumor proliferation of osteosarcoma by acting as a sponge of miR-607 to downregulate E2F6. Front. Oncol. 2020, 10, 584452. [Google Scholar] [CrossRef]
- Zheng, H.L.; Yang, R.Z.; Xu, W.N.; Liu, T.; Chen, P.B.; Zheng, X.F.; Li, B.; Jiang, L.S.; Jiang, S.D. Characterization of LncRNA SNHG22 as a protector of NKIRAS2 through miR-4492 binding in osteosarcoma. Aging 2020, 12, 18571–18587. [Google Scholar] [CrossRef]
- Zheng, C.; Li, R.; Zheng, S.; Fang, H.; Xu, M.; Zhong, L. The knockdown of lncRNA DLGAP1-AS2 suppresses osteosarcoma progression by inhibiting aerobic glycolysis via the miR-451a/HK2 axis. Cancer Sci. 2023, 114, 4747–4762. [Google Scholar] [CrossRef]
- Zhao, W.; Li, L. SP1-induced upregulation of long non-coding RNA HCP5 promotes the development of osteosarcoma. Pathol. Res. Prim. 2019, 215, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Wang, Y.; Hou, W.; Ding, X.; Wang, W. Long non-coding RNA MALAT1 promotes cell proliferation, migration and invasion by targeting miR-590-3p in osteosarcoma. Exp. Ther. Med. 2022, 24, 672. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chen, R. LINC01140 regulates osteosarcoma proliferation and invasion by targeting the miR-139-5p/HOXA9 axis. Biochem. Biophys. Rep. 2022, 31, 101301. [Google Scholar] [CrossRef]
- Zhang, Q.; Tang, X.; Zhou, Y.; Chen, X.; Peng, K.; Jiang, R.; Liu, Z.; Song, X.; Xia, H. LINC01060 knockdown inhibits osteosarcoma cell malignant behaviors in vitro and tumor growth and metastasis in vivo through the PI3K/Akt signaling. Cancer Biol. Ther. 2023, 24, 2198904. [Google Scholar] [CrossRef]
- Zhang, L.; Song, J.; Xu, X.; Sun, D.; Huang, H.; Chen, Y.; Zhang, T. Silencing long non-coding RNA linc00689 suppresses the growth and invasion of osteosarcoma cells by targeting miR-129-5p/NUSAP1. Int. J. Exp. Pathol. 2025, 106, e12524. [Google Scholar] [CrossRef]
- Zhang, H.; Lin, J.; Chen, J.; Gu, W.; Mao, Y.; Wang, H.; Zhang, Y.; Liu, W. DDX11-AS1 contributes to osteosarcoma progression via stabilizing DDX11. Life Sci. 2020, 254, 117392. [Google Scholar] [CrossRef] [PubMed]
- Ye, K.; Wang, S.; Zhang, H.; Han, H.; Ma, B.; Nan, W. Long noncoding RNA GAS5 suppresses cell growth and epithelial-mesenchymal transition in osteosarcoma by regulating the miR-221/ARHI pathway. J. Cell. Biochem. 2017, 118, 4772–4781. [Google Scholar] [CrossRef]
- Yao, W.; Hou, J.; Liu, G.; Wu, F.; Yan, Q.; Guo, L.; Wang, C. LncRNA CBR3-AS1 promotes osteosarcoma progression through the network of miR-140-5p/DDX54-NUCKS1-mTOR signaling pathway. Mol. Ther. Oncolytics 2022, 25, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Shan, Z.; Zhou, X.; Peng, L.; Zhi, C.; Chai, J.; Liu, H.; Yang, J.; Zhang, Z. Knockdown of lncRNA GHET1 inhibits osteosarcoma cells proliferation, invasion, migration and EMT in vitro and in vivo. Cancer Biomark. 2018, 23, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zou, Y.; Wu, J.; Chen, B.; Luo, C.; Chen, X.; Shen, H.; Luo, L. The long noncoding RNA ZEB2-AS1 contributes to proliferation and epithelial-to-mesenchymal transition of osteosarcoma. Cancer Biother. Radiopharm. 2023, 38, 596–603. [Google Scholar] [CrossRef]
- Xu, J.; Ding, R.; Xu, Y. Effects of long non-coding RNA SPRY4-IT1 on osteosarcoma cell biological behavior. Am. J. Transl. Res. 2016, 8, 5330–5337. [Google Scholar]
- Xu, B.; Jin, X.; Yang, T.; Zhang, Y.; Liu, S.; Wu, L.; Ying, H.; Wang, Z. Upregulated lncRNA THRIL/TNF-α signals promote cell growth and predict poor clinical outcomes of osteosarcoma. OncoTargets Ther. 2020, 13, 119–129. [Google Scholar] [CrossRef]
- Xiao, X.; Liu, M.; Xie, S.; Liu, C.; Huang, X.; Huang, X. Long non-coding HOXA-AS3 contributes to osteosarcoma progression through the miR-1286/TEAD1 axis. J. Orthop. Surg. Res. 2023, 18, 730. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, Z.; Zhang, S.; Li, Z.; Li, D.; Yang, S.; Zhang, H.; Zeng, X.; Liu, J. LncRNA FAL1 is a negative prognostic biomarker and exhibits pro-oncogenic function in osteosarcoma. J. Cell. Biochem. 2018, 119, 8481–8489. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, R.; Cheng, G.; Xu, R.; Han, X. Long non-coding RNA HOXA-AS2 promotes migration and invasion by acting as a ceRNA of miR-520c-3p in osteosarcoma cells. Cell Cycle 2018, 17, 1637–1648. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhou, J.; Zhang, Y.; Hu, T.; Sun, Y. Long non-coding RNA HCG11 aggravates osteosarcoma carcinogenesis via regulating the microRNA-579/MMP13 axis. Int. J. Gen. Med. 2020, 13, 1685–1695. [Google Scholar] [CrossRef]
- Wang, L. ELF1-activated FOXD3-AS1 promotes the migration, invasion and EMT of osteosarcoma cells via sponging miR-296-5p to upregulate ZCCHC3. J. Bone Oncol. 2021, 26, 100335. [Google Scholar] [CrossRef] [PubMed]
- Wan, D.; Qu, Y.; Zhang, L.; Ai, S.; Cheng, L. The lncRNA LINC00691Functions as a ceRNA for miRNA-1256 to suppress osteosarcoma by regulating the expression of ST5. OncoTargets Ther. 2020, 13, 13171–13181. [Google Scholar] [CrossRef]
- Tang, Y.; Ji, F. lncRNA HOTTIP facilitates osteosarcoma cell migration, invasion and epithelial-mesenchymal transition by forming a positive feedback loop with c-Myc. Oncol. Lett. 2019, 18, 1649–1656. [Google Scholar] [CrossRef]
- Tang, N.; Chen, Y.; Su, Y.; Zhang, S.; Huang, T. The role of disulfidptosis-associated LncRNA-LINC01137 in Osteosarcoma Biology and its regulatory effects on macrophage polarization. Funct. Integr. Genom. 2024, 24, 219. [Google Scholar] [CrossRef]
- Sun, Y.; Jia, X.; Wang, M.; Deng, Y. Long noncoding RNA MIR31HG abrogates the availability of tumor suppressor microRNA-361 for the growth of osteosarcoma. Cancer Manag. Res. 2019, 11, 8055–8064. [Google Scholar] [CrossRef]
- Sun, M.X.; An, H.Y.; Sun, Y.B.; Sun, Y.B.; Bai, B. LncRNA EBLN3P attributes methotrexate resistance in osteosarcoma cells through miR-200a-3p/O-GlcNAc transferase pathway. J. Orthop. Surg. Res. 2022, 17, 557. [Google Scholar] [CrossRef]
- Shi, D.; Wu, F.; Mu, S.; Hu, B.; Zhong, B.; Gao, F.; Qing, X.; Liu, J.; Zhang, Z.; Shao, Z. LncRNA AFAP1-AS1 promotes tumorigenesis and epithelial-mesenchymal transition of osteosarcoma through RhoC/ROCK1/p38MAPK/Twist1 signaling pathway. J. Exp. Clin. Cancer Res. 2019, 38, 375. [Google Scholar] [CrossRef] [PubMed]
- Saraswat, S.K.; Mahmood, B.S.; Ajila, F.; Kareem, D.S.; Alwan, M.; Athab, Z.H.; Shaier, J.B.; Hosseinifard, S.R. Deciphering the oncogenic landscape: Unveiling the molecular machinery and clinical significance of LncRNA TMPO-AS1 in human cancers. Pathol. Res. Prim. 2024, 255, 155190. [Google Scholar] [CrossRef]
- Ning, Y.; Bai, Z. DSCAM-AS1 accelerates cell proliferation and migration in osteosarcoma through miR-186-5p/GPRC5A signaling. Cancer Biomark. 2021, 30, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Zhang, Z.; Chen, L.; Wang, X.; Zhang, Q.; Liu, S. Silencing of the long non-coding RNA TTN-AS1 attenuates the malignant progression of osteosarcoma cells by regulating the miR-16-1-3p/TFAP4 axis. Front. Oncol. 2021, 11, 652835. [Google Scholar] [CrossRef]
- Ma, H.Z.; Wang, J.; Shi, J.; Zhang, W.; Zhou, D.S. LncRNA LINC00467 contributes to osteosarcoma growth and metastasis through regulating HMGA1 by directly targeting miR-217. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 5933–5945. [Google Scholar] [CrossRef]
- Ma, H.; Su, R.; Feng, H.; Guo, Y.; Su, G. Long noncoding RNA UCA1 promotes osteosarcoma metastasis through CREB1-mediated epithelial-mesenchymal transition and activating PI3K/AKT/mTOR pathway. J. Bone Oncol. 2019, 16, 100228. [Google Scholar] [CrossRef]
- Luo, Y.; Tao, H.; Jin, L.; Xiang, W.; Guo, W. CDKN2B-AS1 exerts oncogenic role in osteosarcoma by promoting cell proliferation and epithelial to mesenchymal transition. Cancer Biother. Radiopharm. 2020, 35, 58–65. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Zhang, J.; Ma, J.; Xu, X.; Wang, Y.; Zhou, Z.; Jiang, D.; Shen, S.; Ding, Y.; et al. Silencing of HuR inhibits osteosarcoma cell epithelial-mesenchymal transition via AGO2 in association with long non-coding RNA XIST. Front. Oncol. 2021, 11, 601982. [Google Scholar] [CrossRef]
- Liu, X.; Wang, M.; Zhang, L.; Huang, L. LncRNA ZFAS1 contributes to osteosarcoma progression via miR-520b and miR-520e-mediated inhibition of RHOC signaling. Clinics 2023, 78, 100143. [Google Scholar] [CrossRef]
- Liu, X.; Wang, H.; Tao, G.L.; Chu, T.B.; Wang, Y.X.; Liu, L. LncRNA-TMPO-AS1 promotes apoptosis of osteosarcoma cells by targeting miR-329 and regulating E2F1. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 11006–11015. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, Q.; Shen, K.; Li, K.; Chang, J.; Li, H.; Duan, A.; Zhang, S.; Huang, Y. Long noncoding RNA LINC00909 induces epithelial-mesenchymal transition and contributes to osteosarcoma tumorigenesis and metastasis. J. Oncol. 2022, 2022, 8660965. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zheng, M.; Wang, X.; Gao, Y.; Gu, Q. LncRNA NR_136400 suppresses cell proliferation and invasion by acting as a ceRNA of TUSC5 that is modulated by miR-8081 in Osteosarcoma. Front. Pharmacol. 2020, 11, 641. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Tang, Y.; Xing, W.; Dong, W.; Wang, Z. LncRNA, CRNDE promotes osteosarcoma cell proliferation, invasion and migration by regulating Notch1 signaling and epithelial-mesenchymal transition. Exp. Mol. Pathol. 2018, 104, 19–25. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Y.; Sun, F.; Zhang, G.; Pan, X.A.; Zhou, Q. Long noncoding RNA PCGEM1 facilitates tumor growth and metastasis of osteosarcoma by sponging miR-433-3p and targeting OMA1. Orthop. Surg. 2023, 15, 1060–1071. [Google Scholar] [CrossRef]
- Li, J.; Yuan, X.; Ma, C.; Li, J.; Qu, G.; Yu, B.; Cai, F.; Peng, Y.; Liu, L.; Zeng, D.; et al. LncRNA LBX2-AS1 impacts osteosarcoma sensitivity to JQ-1 by sequestering miR-597-3p away from BRD4. Front. Oncol. 2023, 13, 1139588. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Fu, S.; Wang, W. Long non-coding RNA HOTAIR promotes human osteosarcoma proliferation, migration through activation of the Wnt/b-catenin signaling pathway. J. Oncol. 2023, 2023, 9667920. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Ren, P.; Wang, Z. Long non-coding RNA Ftx promotes osteosarcoma progression via the epithelial to mesenchymal transition mechanism and is associated with poor prognosis in patients with osteosarcoma. Int. J. Clin. Exp. Pathol. 2018, 11, 4503–4511. [Google Scholar]
- Jiang, Z.; Jiang, C.; Fang, J. Up-regulated lnc-SNHG1 contributes to osteosarcoma progression through sequestration of miR-577 and activation of WNT2B/Wnt/β-catenin pathway. Biochem. Biophys. Res. Commun. 2018, 495, 238–245. [Google Scholar] [CrossRef]
- Jiang, Y.; Luo, Y. LINC01354 promotes osteosarcoma cell invasion by up-regulating integrin β1. Arch. Med. Res. 2020, 51, 115–123. [Google Scholar] [CrossRef]
- Jiang, R.; Zhang, Z.; Zhong, Z.; Zhang, C. Long-non-coding RNA RUSC1-AS1 accelerates osteosarcoma development by miR-101-3p-mediated Notch1 signalling pathway. J. Bone Oncol. 2021, 30, 100382. [Google Scholar] [CrossRef]
- Han, G.; Guo, Q.; Ma, N.; Bi, W.; Xu, M.; Jia, J.; Wang, W. LncRNA BCRT1 facilitates osteosarcoma progression via regulating miR-1303/FGF7 axis. Aging 2021, 13, 15501–15510. [Google Scholar] [CrossRef]
- Gui, D.; Cao, H. Long non-coding RNA CDKN2B-AS1 promotes osteosarcoma by increasing the expression of MAP3K3 via sponging miR-4458. In Vitro Cell. Dev. Biol. Anim. 2020, 56, 24–33. [Google Scholar] [CrossRef]
- Gu, Z.; Wu, S.; Wang, J.; Zhao, S. Long non-coding RNA LINC01419 mediates miR-519a-3p/PDRG1 axis to promote cell progression in osteosarcoma. Cancer Cell Int. 2020, 20, 147. [Google Scholar] [CrossRef]
- Gao, S.; Guo, J.; Li, F.; Zhang, K.; Zhang, Y.; Zhang, Y.; Guo, Y. Long non-coding RNA lncTCF7 predicts poor prognosis and promotes tumor metastasis in osteosarcoma. Int. J. Clin. Exp. Pathol. 2017, 10, 10918–10925. [Google Scholar] [PubMed]
- Ding, Q.; Mo, F.; Cai, X.; Zhang, W.; Wang, J.; Yang, S.; Liu, X. LncRNA CRNDE is activated by SP1 and promotes osteosarcoma proliferation, invasion, and epithelial-mesenchymal transition via Wnt/β-catenin signaling pathway. J. Cell. Biochem. 2020, 121, 3358–3371. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, F.; Zhang, Z.; Sun, F.; Wang, M. Long noncoding RNA SNHG7 promotes the tumor growth and epithelial-to-mesenchymal transition via regulation of miR-34a signals in osteosarcoma. Cancer Biother. Radiopharm. 2018, 33, 365–372. [Google Scholar] [CrossRef]
- Cai, L.; Lv, J.; Zhang, Y.; Li, J.; Wang, Y.; Yang, H. The lncRNA HNF1A-AS1 is a negative prognostic factor and promotes tumorigenesis in osteosarcoma. J. Cell. Mol. Med. 2017, 21, 2654–2662. [Google Scholar] [CrossRef]
- Cai, F.; Liu, L.; Bo, Y.; Yan, W.; Tao, X.; Peng, Y.; Zhang, Z.; Liao, Q.; Yi, Y. LncRNA RPARP-AS1 promotes the progression of osteosarcoma cells through regulating lipid metabolism. BMC Cancer 2024, 24, 166. [Google Scholar] [CrossRef]
- Bu, J.; Guo, R.; Xu, X.Z.; Luo, Y.; Liu, J.F. LncRNA SNHG16 promotes epithelial-mesenchymal transition by upregulating ITGA6 through miR-488 inhibition in osteosarcoma. J. Bone Oncol. 2021, 27, 100348. [Google Scholar] [CrossRef]
- Bai, J.; Zhang, X.; Jiang, F.; Shan, H.; Gao, X.; Bo, L.; Zhang, Y. A feedback loop of LINC00665 and the Wnt signaling pathway expedites osteosarcoma cell proliferation, invasion, and epithelial-mesenchymal transition. Orthop. Surg. 2023, 15, 286–300. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhou, Q.; Shen, W. Circ-FOXM1 promotes the proliferation, migration and EMT process of osteosarcoma cells through FOXM1-mediated Wnt pathway activation. J. Orthop. Surg. Res. 2022, 17, 344. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Li, H.; Wu, C.; Li, J. Circ_0021087 acts as a miR-184 sponge and represses gastric cancer progression by adsorbing miR-184 and elevating FOSB expression. Eur. J. Clin. Investig. 2021, 51, e13605. [Google Scholar] [CrossRef]
- Yang, J.; Hu, Z.; Ru, X.; He, M.; Hu, Z.; Qin, X.; Xiao, S.; Liu, D.; Huang, H.; Wei, Q. Hsa_circ_0002005 aggravates osteosarcoma by increasing cell proliferation, migration, and invasion. Gene 2025, 942, 149221. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Yang, T.; Wang, Z.; Zhang, Y.; Liu, S.; Shen, M. CircRNA CDR1as/miR-7 signals promote tumor growth of osteosarcoma with a potential therapeutic and diagnostic value. Cancer Manag. Res. 2018, 10, 4871–4880. [Google Scholar] [CrossRef]
- Wu, H.; Li, W.; Zhu, S.; Zhang, D.; Zhang, M. Circular RNA circUBAP2 regulates proliferation and invasion of osteosarcoma cells through miR-641/YAP1 axis. Cancer Cell Int. 2020, 20, 223. [Google Scholar] [CrossRef]
- Wang, Z.; Deng, M.; Chen, L.; Wang, W.; Liu, G.; Liu, D.; Han, Z.; Zhou, Y. Circular RNA Circ-03955 Promotes Epithelial-Mesenchymal Transition in Osteosarcoma by Regulating miR-3662/Metadherin Pathway. Front. Oncol. 2020, 10, 545460. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Long, Q.; Zhang, W.; Zeng, D.; Hu, B.; Liu, S.; Li, C. Circular RNA expression profile identifies circMGEA5 as a novel metastasis-promoting factor and potential biomarker in osteosarcoma. J. Biochem. Mol. Toxicol. 2023, 37, e23286. [Google Scholar] [CrossRef]
- Gao, Y.; Ma, H.; Gao, Y.; Tao, K.; Fu, L.; Ren, R.; Hu, X.; Kou, M.; Chen, B.; Shi, J.; et al. CircRNA Circ_0001721 promotes the progression of osteosarcoma through miR-372-3p/MAPK7 axis. Cancer Manag. Res. 2020, 12, 8287–8302. [Google Scholar] [CrossRef]
- Feng, Z.H.; Zheng, L.; Yao, T.; Tao, S.Y.; Wei, X.A.; Zheng, Z.Y.; Zheng, B.J.; Zhang, X.Y.; Huang, B.; Liu, J.H.; et al. EIF4A3-induced circular RNA PRKAR1B promotes osteosarcoma progression by miR-361-3p-mediated induction of FZD4 expression. Cell Death Dis. 2021, 12, 1025. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Zhou, H.; Rong, W. Circular RNA_0078767 upregulates Kruppel-like factor 9 expression by targeting microRNA-889, thereby inhibiting the progression of osteosarcoma. Bioengineered 2022, 13, 14313–14328. [Google Scholar] [CrossRef]
- Chen, J.; Liu, G.; Wu, Y.; Ma, J.; Wu, H.; Xie, Z.; Chen, S.; Yang, Y.; Wang, S.; Shen, P.; et al. CircMYO10 promotes osteosarcoma progression by regulating miR-370-3p/RUVBL1 axis to enhance the transcriptional activity of β-catenin/LEF1 complex via effects on chromatin remodeling. Mol. Cancer 2019, 18, 150. [Google Scholar] [CrossRef] [PubMed]



| Non-Coding RNA | Type | Regulatory Function on EMT | Target Genes/Pathways | Therapeutic Potential | Delivery Strategy | References |
|---|---|---|---|---|---|---|
| MiR-19 | miRNA | Promotes | SPRED2, ERK/MAPK, Autophagy | Enhances OS proliferation, invasion, migration, and EMT | miRNA inhibitors | PMID: 33023313 [59] |
| MiR-135b | miRNA | Promotes | TAZ, Hippo pathway, EMT markers | Targeting miR-135b may suppress OS proliferation, EMT, and metastasis | miRNA inhibitors | PMID: 28823959 [133] |
| MiR-486 | miRNA | Suppresses | PIM1, EMT markers | Targeting miR-486 may inhibit OS invasion and EMT | miRNA mimics | PMID: 30103304 [137] |
| MiR-429 | miRNA | Suppresses | ZEB1, EMT markers (E-cadherin, Vimentin, N-Cadherin, Snail) | Targeting miR-429 may inhibit OS progression and metastasis | miRNA mimics | PMID: 28694763 [152] |
| MiR-128 | miRNA | Suppresses | ITGA2, EMT markers | Targeting miR-128 may inhibit OS migration, invasion, and EMT | miRNA mimics | PMID: 26700675 [138] |
| MiR-22 | miRNA | Suppresses | Twist1, EMT markers | Targeting miR-22 may inhibit OS EMT and progression | miRNA mimics | PMID: 32391253 [114] |
| MiR-155 | miRNA | Promotes | TNF-α, ERK signaling, CSC markers (CD24, CD90, CD133) | Targeting miR-155 may inhibit OS CSC transformation and EMT | miRNA inhibitors | PMID: 31669646 [122] |
| Non-Coding RNA | Type | Regulatory Function on EMT | Target Genes/Pathways | Therapeutic Potential | Delivery Strategy | References |
|---|---|---|---|---|---|---|
| ZEB2-AS1 | lncRNA | Promotes | ZEB2-AS1 pathway | Promotes OS proliferation, EMT, migration, and invasion | Knockdown | PMID: 33085924 [174] |
| HOTAIR | lncRNA | Suppresses | LPR5, Wnt/β-catenin signaling | Suppresses OS migration, invasion, and proliferation | Overexpression | PMID: 36816362 [202] |
| MALAT1 | lncRNA | Promotes | miR-590-3p | Promotes OS proliferation, migration, invasion, and EMT | Knockdown | PMID: 36277152 [166] |
| KIAA0087 | lncRNA | Suppresses | miR-411-3p/SOCS1/JAK2/STAT3 | Inhibits OS growth, metastasis, and EMT | Overexpression | PMID: 37009803 [71] |
| TUG1 | lncRNA | Promotes | miR-144-3p/EZH2/Wnt/β-catenin | Promotes OS tumorigenesis, migration, and EMT | Knockdown | PMID: 28902349 [157] |
| FER1L4 | lncRNA | Suppresses | miR-18a-5p/SOCS5, PI3K/AKT | Induces apoptosis, inhibits EMT and PI3K/AKT activation | Overexpression | PMID: 31473323 [80] |
| CRNDE | lncRNA | Promotes | Wnt/β-catenin pathway | Enhances OS proliferation, invasion, and EMT | Knockdown | PMID: 31898343 [211] |
| Non-Coding RNA | Type | Regulatory Function on EMT | Target Genes/Pathways | Therapeutic Potential | Delivery Strategy | References |
|---|---|---|---|---|---|---|
| Circ_0001721 | circRNA | Promotes | miR-372-3p/MAPK7 | Enhances OS progression, migration, invasion, and EMT | Knockdown | PMID: 32982424 [224] |
| Circ-FOXM1 | circRNA | Promotes | miR-320a/miR-320b/FOXM1/Wnt | Enhances OS proliferation, migration, invasion, and EMT | Knockdown | PMID: 35799265 [217] |
| Circ-CDR1as | circRNA | Promotes | miR-7/EGFR/CCNE1/PI3KCD/RAF1 | Enhances OS proliferation, migration, invasion, and EMT | Knockdown | PMID: 30425578 [220] |
| CircMYO10 | circRNA | Promotes | miR-370-3p/RUVBL1/Wnt/β-catenin | Enhances OS proliferation, chromatin remodeling, and EMT | Knockdown | PMID: 31665067 [227] |
| CircMGEA5 | circRNA | Promotes | miR-153-3p/miR-8084/ZEB1/Snail | Enhances OS metastasis, EMT, and invasion | Knockdown | PMID: 36564929 [223] |
| CircPRKAR1B | circRNA | Promotes | miR-361-3p/FZD4/Wnt | Enhances OS proliferation, migration, invasion, and EMT | Knockdown | PMID: 34716310 [225] |
| Circ_0078767 | circRNA | Suppresses | miR-889/KLF9 | Inhibits OS proliferation, migration, invasion, and EMT | Overexpression | PMID: 35758280 [226] |
| CircUBAP2 | circRNA | Promotes | miR-641/YAP1 | Enhances OS proliferation, invasion, and EMT | Knockdown | PMID: 32528231 [221] |
| Circ_0021087 | circRNA | Suppresses | miR-184/FOSB | Inhibits OS proliferation, migration, invasion, and EMT | Overexpression | PMID: 34076278 [218] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, F.; Ando, K.; Takemura, Y.; Park, T.-H.; Yayama, T.; Imai, S. Epithelial–Mesenchymal Transition in Osteosarcoma as a Key Driver of Pulmonary Metastasis. Cancers 2025, 17, 2922. https://doi.org/10.3390/cancers17172922
Luo F, Ando K, Takemura Y, Park T-H, Yayama T, Imai S. Epithelial–Mesenchymal Transition in Osteosarcoma as a Key Driver of Pulmonary Metastasis. Cancers. 2025; 17(17):2922. https://doi.org/10.3390/cancers17172922
Chicago/Turabian StyleLuo, Fangcheng, Kosei Ando, Yoshinori Takemura, Tae-Hwi Park, Takafumi Yayama, and Shinji Imai. 2025. "Epithelial–Mesenchymal Transition in Osteosarcoma as a Key Driver of Pulmonary Metastasis" Cancers 17, no. 17: 2922. https://doi.org/10.3390/cancers17172922
APA StyleLuo, F., Ando, K., Takemura, Y., Park, T.-H., Yayama, T., & Imai, S. (2025). Epithelial–Mesenchymal Transition in Osteosarcoma as a Key Driver of Pulmonary Metastasis. Cancers, 17(17), 2922. https://doi.org/10.3390/cancers17172922







