CD19 CAR-T Outcomes in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Retrospective Cohort Study from the Calabria Referral Center in Southern Italy
Simple Summary
Abstract
1. Introduction
2. Patients and Methods
3. Results
3.1. Baseline Population Characteristics
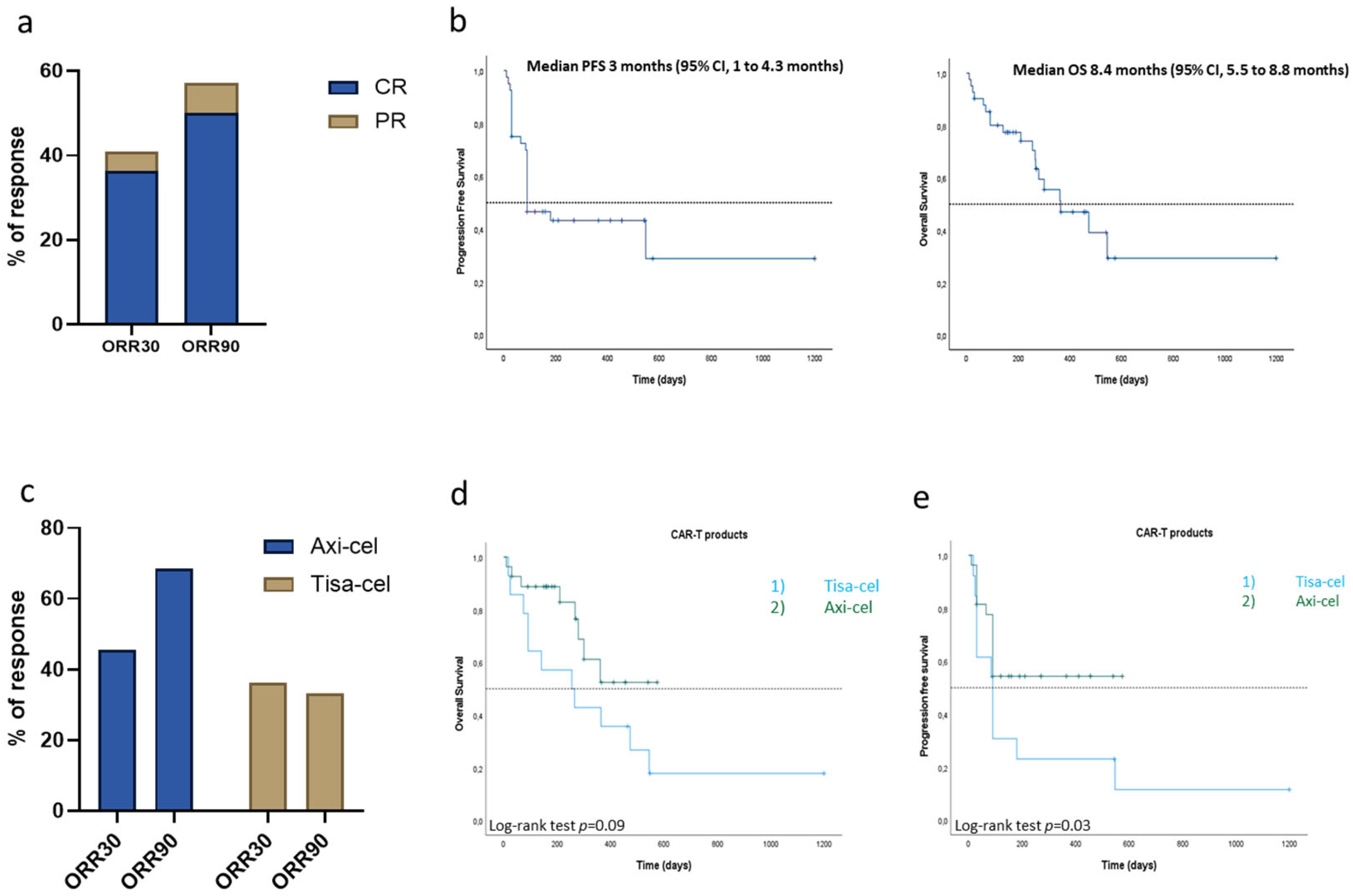
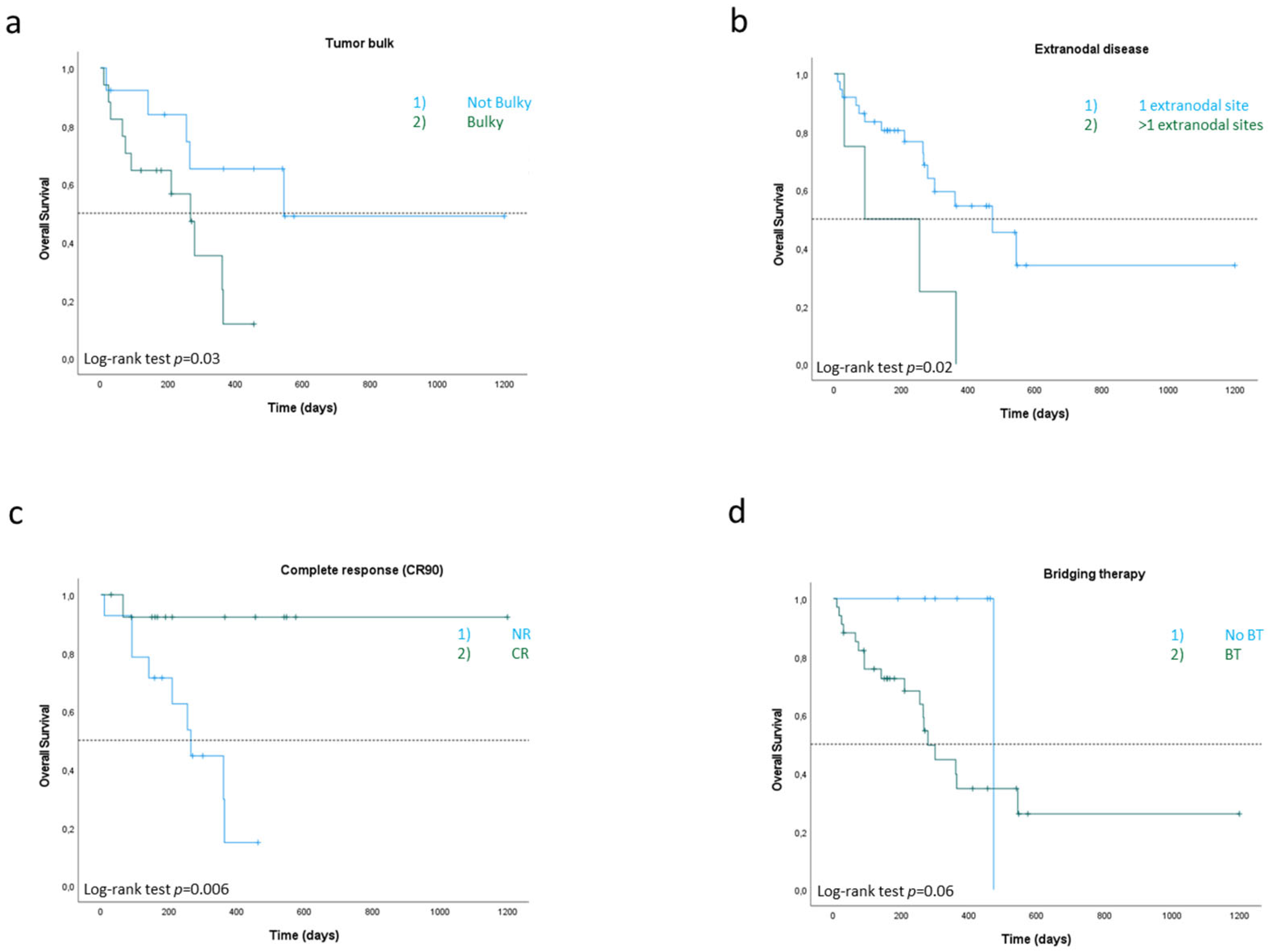
3.2. Efficacy and Outcomes
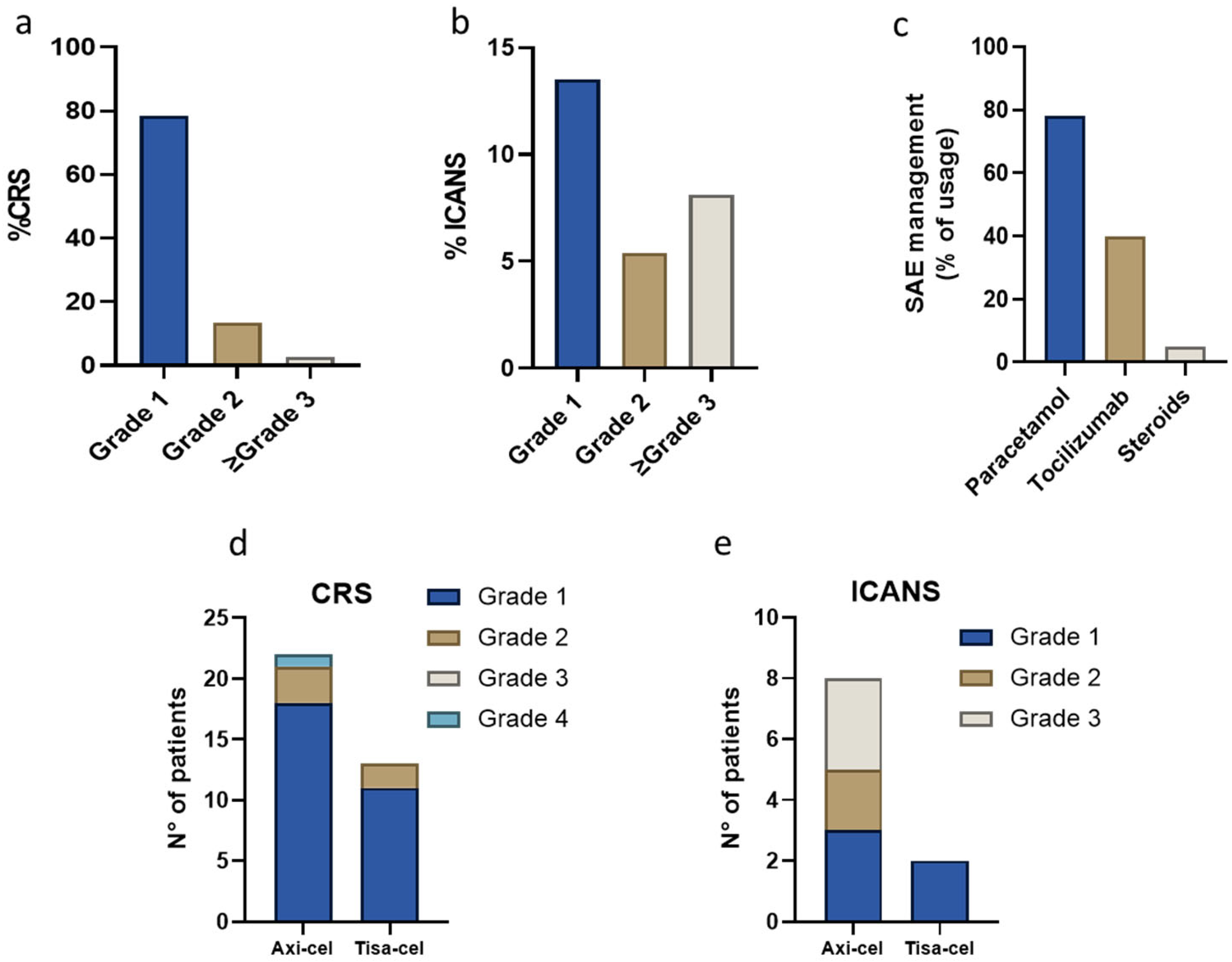
3.3. Safety
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DLBCL | Diffuse Large B-cell Lymphoma |
| CAR-T | Chimeric Antigen Receptor T cell |
| ORR | Overall Response Rate |
| PFS | Progression-Free Survival |
| OS | Overall Survival |
| CRS | Cytokine Release Syndrome |
| ICANS | Immune-Cell-Associated Neurotoxicity |
| ECOG | Eastern Cooperative Oncology Group |
| AIFA | Agenzia Italiana del Farmaco |
References
- Coiffier, B.; Lepage, E.; Brière, J.; Herbrecht, R.; Tilly, H.; Bouabdallah, R.; Morel, P.; Van Den Neste, E.; Salles, G.; Gaulard, P.; et al. CHOP Chemotherapy plus Rituximab Compared with CHOP Alone in Elderly Patients with Diffuse Large-B-Cell Lymphoma. N. Engl. J. Med. 2002, 346, 235–242. [Google Scholar] [CrossRef]
- Mohammed, R.; Milne, A.; Kayani, K.; Ojha, U. How the discovery of rituximab impacted the treatment of B-cell non-Hodgkin’s lymphomas. J. Blood Med. 2019, 10, 71–84. [Google Scholar] [CrossRef]
- Coiffier, B.; Thieblemont, C.; Van Den Neste, E.; Lepeu, G.; Plantier, I.; Castaigne, S.; Lefort, S.; Marit, G.; Macro, M.; Sebban, C.; et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: A study by the Groupe d’Etudes des Lymphomes de l’Adulte. Blood 2010, 116, 2040–2045. [Google Scholar] [CrossRef]
- Philip, T.; Guglielmi, C.; Hagenbeek, A.; Somers, R.; Van Der Lelie, H.; Bron, D.; Sonneveld, P.; Gisselbrecht, C.; Cahn, J.-Y.; Harousseau, J.-L.; et al. Autologous Bone Marrow Transplantation as Compared with Salvage Chemotherapy in Relapses of Chemotherapy-Sensitive Non-Hodgkin’s Lymphoma. N. Engl. J. Med. 1995, 333, 1540–1545. [Google Scholar] [CrossRef]
- Feugier, P.; Van Hoof, A.; Sebban, C.; Solal-Celigny, P.; Bouabdallah, R.; Fermé, C.; Christian, B.; Lepage, E.; Tilly, H.; Morschhauser, F.; et al. Long-Term Results of the R-CHOP Study in the Treatment of Elderly Patients with Diffuse Large B-Cell Lymphoma: A Study by the Groupe d’Etude des Lymphomes de l’Adulte. J. Clin. Oncol. 2005, 23, 4117–4126. [Google Scholar] [CrossRef]
- Crump, M.; Neelapu, S.S.; Farooq, U.; Van Den Neste, E.; Kuruvilla, J.; Westin, J.; Link, B.K.; Hay, A.; Cerhan, J.R.; Zhu, L.; et al. Outcomes in refractory diffuse large B-cell lymphoma: Results from the international SCHOLAR-1 study. Blood 2017, 130, 1800–1808. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Korsos, V.; Palomba, M.L. Chimeric antigen receptor T-cell therapy for aggressive B-cell lymphomas. Front. Oncol. 2024, 14, 1394057. [Google Scholar] [CrossRef] [PubMed]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; Lin, Y.; et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N. Engl. J. Med. 2017, 377, 2531–2544. [Google Scholar] [CrossRef] [PubMed]
- Schuster, S.J.; Bishop, M.R.; Tam, C.S.; Waller, E.K.; Borchmann, P.; McGuirk, J.P.; Jäger, U.; Jaglowski, S.; Andreadis, C.; Westin, J.R.; et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N. Engl. J. Med. 2019, 380, 45–56. [Google Scholar] [CrossRef]
- Locke, F.L.; Miklos, D.B.; Jacobson, C.A.; Perales, M.-A.; Kersten, M.-J.; Oluwole, O.O.; Ghobadi, A.; Rapoport, A.P.; McGuirk, J.; Pagel, J.M.; et al. Axicabtagene Ciloleucel as Second-Line Therapy for Large B-Cell Lymphoma. N. Engl. J. Med. 2022, 386, 640–654. [Google Scholar] [CrossRef]
- Westin, J.R.; Oluwole, O.O.; Kersten, M.J.; Miklos, D.B.; Perales, M.A.; Ghobadi, A.; Rapoport, A.P.; Sureda, A.; Jacobson, C.A.; Farooq, U.; et al. Survival with Axicabtagene Ciloleucel in Large B-Cell Lymphoma. N. Engl. J. Med. 2023, 389, 148–157. [Google Scholar] [CrossRef]
- The Lancet Haematology. CAR T-cell therapy: Navigating real-world challenges beyond clinical trials. Lancet Haematol. 2025, 12, e231. [Google Scholar] [CrossRef] [PubMed]
- Jommi, C.; Bramanti, S.; Pani, M.; Ghirardini, A.; Santoro, A. CAR T-Cell Therapies in Italy: Patient Access Barriers and Recommendations for Health System Solutions. Front. Pharmacol. 2022, 13, 915342. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Prev. Med. 2007, 45, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Bishop, M.R.; Dickinson, M.; Purtill, D.; Barba, P.; Santoro, A.; Hamad, N.; Kato, K.; Sureda, A.; Greil, R.; Thieblemont, C.; et al. Second-Line Tisagenlecleucel or Standard Care in Aggressive B-Cell Lymphoma. N. Engl. J. Med. 2022, 386, 629–639. [Google Scholar] [CrossRef]
- Canelo-Vilaseca, M.; Sabbah, M.; Di Blasi, R.; Cristinelli, C.; Sureda, A.; Caillat-Zucman, S.; Thieblemont, C. Lymphodepletion chemotherapy in chimeric antigen receptor-engineered T (CAR-T) cell therapy in lymphoma. Bone Marrow Transplant. 2025, 60, 559–567. [Google Scholar] [CrossRef]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A. Recommendations for initial evaluation, stag-ing, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef]
- Delgado, A.; Guddati, A.K. Clinical endpoints in oncology-a primer. Am. J. Cancer Res. 2021, 11, 1121–1131. [Google Scholar]
- Lee, D.W.; Santomasso, B.D.; Locke, F.L.; Ghobadi, A.; Turtle, C.J.; Brudno, J.N.; Maus, M.V.; Park, J.H.; Mead, E.; Pavletic, S.; et al. ASTCT Consensus Grading for Cytokine Release Syndrome and Neurologic Toxicity Associated with Immune Effector Cells. Biol. Blood Marrow Transplant. 2019, 25, 625–638. [Google Scholar] [CrossRef]
- Freeman, G.H.; Halton, J.H. Note on an Exact Treatment of Contingency, Goodness of Fit and Other Problems of Significance. Biometrika 1951, 38, 141–149. [Google Scholar] [CrossRef]
- Andrade, C. Mean Difference, Standardized Mean Difference (SMD), and Their Use in Meta-Analysis: As Simple as It Gets. J. Clin. Psychiatry 2020, 81, 20f13681. [Google Scholar] [CrossRef] [PubMed]
- Casadei, B.; Argnani, L.; Guadagnuolo, S.; Pellegrini, C.; Stefoni, V.; Broccoli, A.; Nanni, L.; Morigi, A.; Lolli, G.; Guarino, M.; et al. Real World Evidence of CAR T-Cell Therapies for the Treatment of Relapsed/Refractory B-Cell Non-Hodgkin Lymphoma: A Monocentric Experience. Cancers 2021, 13, 4789. [Google Scholar] [CrossRef] [PubMed]
- Iacoboni, G.; Villacampa, G.; Martinez-Cibrian, N.; Bailén, R.; Corral, L.L.; Sanchez, J.M.; Guerreiro, M.; Caballero, A.C.; Mussetti, A.; Sancho, J.; et al. Real-world evidence of tisagenlecleucel for the treatment of relapsed or refractory large B-cell lymphoma. Cancer Med. 2021, 10, 3214–3223. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, C.A.; Munoz, J.; Sun, F.; Kanters, S.; Limbrick-Oldfield, E.H.; Spooner, C.; Mignone, K.; Ayuk, F.; Sanderson, R.; Whitmore, J.; et al. Real-World Outcomes with Chimeric Antigen Receptor T Cell Therapies in Large B Cell Lymphoma: A Systematic Review and Meta-Analysis. Biol. Blood Marrow Transplant. 2023, 30, 77.e1–77.e15. [Google Scholar] [CrossRef]
- Bethge, W.A.; Martus, P.; Schmitt, M.; Holtick, U.; Subklewe, M.; von Tresckow, B.; Ayuk, F.; Wagner-Drouet, E.M.; Wulf, G.G.; Marks, R.; et al. GLA/DRST real-world outcome analysis of CAR-T cell therapies for large B-cell lymphoma in Germany. Blood 2022, 140, 349–358. [Google Scholar] [CrossRef]
- Cusatis, R.; Litovich, C.; Feng, Z.; Allbee-Johnson, M.; Kapfhammer, M.; Mattila, D.; Akinola, I.; Phelan, R.; Broglie, L.; Auletta, J.J.; et al. Current Trends and Outcomes in Cellular Therapy Activity in the United States, Including Prospective Patient-Reported Outcomes Data Collection in the Center for International Blood and Marrow Transplant Research Registry. Biol. Blood Marrow Transplant. 2024, 30, 917.e1–917.e12. [Google Scholar] [CrossRef]
- Cappell, K.M.; Kochenderfer, J.N. Long-term outcomes following CAR T cell therapy: What we know so far. Nat. Rev. Clin. Oncol. 2023, 20, 359–371. [Google Scholar] [CrossRef]
- Ahmed, N.; Wesson, W.; Lutfi, F.; Porter, D.L.; Bachanova, V.; Nastoupil, L.J.; Perales, M.-A.; Maziarz, R.T.; Brower, J.; Shah, G.L.; et al. Optimizing the post-CAR T monitoring period in recipients of axicabtagene ciloleucel, tisagenlecleucel, and lisocabtagene maraleucel. Blood Adv. 2024, 8, 5346–5354. [Google Scholar] [CrossRef]
- Bücklein, V.; Perez, A.; Rejeski, K.; Iacoboni, G.; Jurinovic, V.; Holtick, U.; Penack, O.; Kharboutli, S.; Blumenberg, V.; Ackermann, J.; et al. Inferior Outcomes of EU Versus US Patients Treated with CD19 CAR-T for Relapsed/Refractory Large B-cell Lymphoma: Association with Differences in Tumor Burden, Systemic Inflammation, Bridging Therapy Utilization, and CAR-T Product Use. HemaSphere 2023, 7, e907. [Google Scholar] [CrossRef]
- Locke, F.L.; Siddiqi, T.; Jacobson, C.A.; Nikiforow, S.; Ahmed, S.; Miklos, D.B.; Lin, Y.; Lunning, M.A.; Hill, B.T.; Ghobadi, A.; et al. Impact of vein-to-vein time in patients with R/R LBCL treated with axicabtagene ciloleucel. Blood Adv. 2025, 9, 2663–2676. [Google Scholar] [CrossRef]
- Kwon, M.; Iacoboni, G.; Reguera, J.L.; Corral, L.L.; Morales, R.H.; Ortiz-Maldonado, V.; Guerreiro, M.; Caballero, A.C.; Domínguez, M.L.G.; Pina, J.M.S.; et al. Axicabtagene ciloleucel compared to tisagenlecleucel for the treatment of aggressive B-cell lymphoma. Haematologica 2022, 108, 110–121. [Google Scholar] [CrossRef]
- Liao, C.; Zeng, L.; Lu, S.; Zheng, S.; Guo, B.; Ke, Q.; Wang, M.; Sun, J.; Rong, C.; He, S.; et al. Comparison of the Efficacy and Safety of Axi-Cel and Tisa-Cel Based on Meta-Analysis. J. Cancer 2024, 15, 5729–5741. [Google Scholar] [CrossRef] [PubMed]
- Galtier, J.; Mesguich, C.; Sesques, P.; Dupont, V.; Bachy, E.; Di Blasi, R.; Thieblemont, C.; Gastinne, T.; Cartron, G.; Brisou, G.; et al. Outcomes of patients with relapsed or refractory primary mediastinal B-cell lymphoma treated with anti-CD19 CAR-T cells: CARTHYM, a study from the French national DESCAR-T registry. Hemasphere 2025, 9, e70091. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, J.L.; Ramdhanny, A.; Munir, M.; Rimmalapudi, S.; Epperla, N. Association of CAR-T approval on outcomes in patients with diffuse large B-cell lymphoma at the population level in the United States. Biomark. Res. 2025, 13, 64. [Google Scholar] [CrossRef] [PubMed]
- St-Pierre, F.; Bhatta, S.; Doukas, P.G.; Jenkin, M.; Annunzio, K.; Rojek, A.E.; Gibson, A.; Tiger, Y.K.; McCall, B.; Alhamad, K.; et al. Impact of extranodal involvement at CAR T-cell therapy on outcomes in patients with relapsed or refractory large B-cell lymphoma—Results from a multicenter cohort study. Blood Cancer J. 2025, 15, 110. [Google Scholar] [CrossRef]
- Schubert, M.-L.; Schmitt, M.; Wang, L.; Ramos, C.; Jordan, K.; Müller-Tidow, C.; Dreger, P. Side-effect management of chimeric antigen receptor (CAR) T-cell therapy. Ann. Oncol. 2021, 32, 34–48. [Google Scholar] [CrossRef]
- Dahunsi, D.; Eleanya, C.; Akintunde, A.; Oluwole, O. Prolonged Cytopenia with CAR-T Cell Therapy and Management Recommendations. Clin. Hematol. Int. 2025, 7, 47–54. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Guo, Q.; Zhang, Y.; Liu, A. Analysis and Investigation of Bioinformatics and Epigenetics Reveal the Underlying Mechanisms by which FLOT2 Modulates the Progression of Diffuse Large B-cell Lymphoma. Discov. Med. 2024, 36, 621–631. [Google Scholar] [CrossRef]
- Ning, F.; Wang, H.; Liang, Z.; Lan, J. TNFAIP3 Overexpression Inhibits Diffuse Large B-Cell Lymphoma Progression by Promoting Autophagy through TLR4/MyD88/NF-κB Signaling Pathway. Discov. Med. 2024, 36, 1627–1640. [Google Scholar] [CrossRef]
- Scholler, N.; Perbost, R.; Locke, F.L.; Jain, M.D.; Turcan, S.; Danan, C.; Chang, E.C.; Neelapu, S.S.; Miklos, D.B.; Jacobson, C.A.; et al. Tumor immune contexture is a determinant of anti-CD19 CAR T cell efficacy in large B cell lymphoma. Nat. Med. 2022, 28, 1872–1882. [Google Scholar] [CrossRef]
- Sehgal, A.; Hoda, D.; Riedell, P.A.; Ghosh, N.; Hamadani, M.; Hildebrandt, G.C.; Godwin, J.E.; Reagan, P.M.; Wagner-Johnston, N.; Essell, J.; et al. Lisocabtagene maraleucel as second-line therapy in adults with relapsed or refractory large B-cell lymphoma who were not intended for haematopoietic stem cell transplantation (PILOT): An open-label, phase 2 study. Lancet Oncol. 2022, 23, 1066–1077. [Google Scholar] [CrossRef]
- Shahid, S.; Prockop, S.E.; Flynn, G.C.; Mauguen, A.; White, C.O.; Bieler, J.; McAvoy, D.; Hosszu, K.; Cancio, M.I.; Jakubowski, A.A.; et al. Allogeneic off-the-shelf CAR T-cell therapy for relapsed or refractory B-cell malignancies. Blood Adv. 2025, 9, 1644–1657. [Google Scholar] [CrossRef]
| Demographic and Clinical Characteristics | n | % |
|---|---|---|
| Sex | ||
| Female | 9 | 22 |
| male | 32 | 78 |
| Age | ||
| Median, y | 66 | |
| <70 y | 34 | 82.9 |
| >70 y | 7 | 17.1 |
| Performance Status (ECOG) | ||
| 0–1 | 39 | 4.9 |
| >1 | 2 | 95.1 |
| Disease involvement at relapse | ||
| Bulky disease | 17 | 56.7 |
| Bone marrow involvement | 4 | 9.8 |
| >1 extranodal sites | 4 | 9.8 |
| Disease type | ||
| Primary DLBCL | 34 | 82.9 |
| Transformed DLBCL | 7 | 17.1 |
| Status of disease before apheresis | ||
| Controlled disease | 20 | 52.2 |
| Active disease | 21 | 48.8 |
| Prior stem-cell transplant | 10 | 24.4 |
| Number previous line of therapy | ||
| 1 | 10 | 24.4 |
| 2 | 23 | 56.1 |
| 3 | 5 | 12.2 |
| 4 | 3 | 7.3 |
| Primary refractory disease | 25 | 61 |
| Bridging therapy | ||
| Immuno-chemo | 19 | 48.7 |
| Chemo | 8 | 19.5 |
| IMiDs | 1 | 2.5 |
| RT | 3 | 7.3 |
| Steroids | 3 | 7.3 |
| None | 7 | 17.1 |
| CAR-T product infused | ||
| Axi-cel | 27 | 65.8 |
| Tisa-cel | 14 | 34.2 |
| Line of treatment | ||
| Second line | 10 | 37 |
| From third line | 17 | 63 |
| Variables | % | p-Value |
|---|---|---|
| Diagnosis | 0.06 | |
| De novo | 65.2 | |
| Transformed | 20 | |
| Sex | 0.144 | |
| Male | 83.3 | |
| Female | 50 | |
| CAR-T product | 0.08 | |
| Axi-cel | 68.4 | |
| Tisa-cel | 33.3 | |
| Age | 0.161 | |
| <70 y | 62.5 | |
| >70 y | 25.5 | |
| Performance status (ECOG) | 0.240 | |
| 0–1 | 59.2 | |
| >1 | 0 | |
| Disease involvement at relapse | ||
| Bulky | 0.466 | |
| Yes | 70 | |
| No | 54.5 | |
| Extranodal sites | 0.376 | |
| 0 | 60 | |
| >1 | 33.3 | |
| Line of therapy (LOT) | 0.277 | |
| II line | 50 | |
| from III line | 75 | |
| Bridging therapy | 0.755 | |
| Immuno-chemo | 50 | |
| None | 58.3 |
| Variables | p-Value | HR | 95% CI | |
|---|---|---|---|---|
| Lower | Upper | |||
| Sex | ||||
| Male | 0.121 | 0.311 | 0.071 | 1.359 |
| Age | 0.312 | 0.982 | 0.949 | 1.017 |
| <40 years | 0.309 | 1.896 | 0.419 | 8.577 |
| 40–49 years | 0.052 | 4.749 | 1.251 | 18.031 |
| 50–59 years | 0.378 | 0.570 | 0.164 | 1.987 |
| 60–69 years | 0.761 | 0.870 | 0.347 | 2.181 |
| >70 years | 0.659 | 0.756 | 0.218 | 0.768 |
| Tumor bulk | 0.044 | 3.322 | 1.032 | 10.711 |
| >1 Extranodal sites | 0.031 | 3.462 | 1.123 | 10.674 |
| Transformed | 0.374 | 1.608 | 0.565 | 4.678 |
| ECOG > 1 | 0.572 | 1.432 | 3.422 | 6.919 |
| Treatment | ||||
| Bridging therapy | 0.096 | 5.578 | 0.738 | 42.134 |
| Second line | 0.838 | 0.854 | 0.186 | 3.910 |
| ORR30 | 0.005 | 0.053 | 0.007 | 0.419 |
| ORR90 | 0.011 | 0.125 | 0.025 | 0.653 |
| Baseline high CRP (>3 mg/dL) | 0.462 | 0.682 | 0.246 | 1.893 |
| Baseline high ferritine (>650 ng/mL) | 0.370 | 0.647 | 0.250 | 1.675 |
| Safety Analysis | n | % |
|---|---|---|
| Cytokine release syndrome/CRS | ||
| Grade 1 | 29 | 78.5 |
| Grade 2 | 5 | 13.5 |
| Grade ≥ 3 | 1 | 2.7 |
| Neurotoxicity/ICANS | ||
| Grade 1 | 5 | 13.5 |
| Grade 2 | 2 | 5.4 |
| Grade ≥ 3 | 3 | 8.1 |
| ICU admission < 30 days post-infusion | 0 | 0 |
| Therapy given for CRS/ICANS treatment | ||
| Paracetamol | 27 | 77.1 |
| Corticosteroids | 2 | 4.8 |
| Tocilizumab | 23 | 56 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caracciolo, D.; Canale, F.A.; Naso, V.; Alati, C.; Marafioti, V.; Porto, G.; Tedesco, L.; Pensabene, G.; Martino, E.A.; Allegra, A.; et al. CD19 CAR-T Outcomes in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Retrospective Cohort Study from the Calabria Referral Center in Southern Italy. Cancers 2025, 17, 2796. https://doi.org/10.3390/cancers17172796
Caracciolo D, Canale FA, Naso V, Alati C, Marafioti V, Porto G, Tedesco L, Pensabene G, Martino EA, Allegra A, et al. CD19 CAR-T Outcomes in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Retrospective Cohort Study from the Calabria Referral Center in Southern Italy. Cancers. 2025; 17(17):2796. https://doi.org/10.3390/cancers17172796
Chicago/Turabian StyleCaracciolo, Daniele, Filippo Antonio Canale, Virginia Naso, Caterina Alati, Violetta Marafioti, Gaetana Porto, Ludovica Tedesco, Giulia Pensabene, Enrica Antonia Martino, Alessandro Allegra, and et al. 2025. "CD19 CAR-T Outcomes in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Retrospective Cohort Study from the Calabria Referral Center in Southern Italy" Cancers 17, no. 17: 2796. https://doi.org/10.3390/cancers17172796
APA StyleCaracciolo, D., Canale, F. A., Naso, V., Alati, C., Marafioti, V., Porto, G., Tedesco, L., Pensabene, G., Martino, E. A., Allegra, A., Gerace, D. G., Cimminiello, M., Gentile, M., Tagliaferri, P., Tassone, P., & Martino, M. (2025). CD19 CAR-T Outcomes in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Retrospective Cohort Study from the Calabria Referral Center in Southern Italy. Cancers, 17(17), 2796. https://doi.org/10.3390/cancers17172796








