The Gut Microbiome’s Impact on the Pathogenesis and Treatment of Gastric Cancer—An Updated Literature Review
Simple Summary
Abstract
1. Introduction
2. Role of Gut Microbiota and Gastric Carcinogenesis
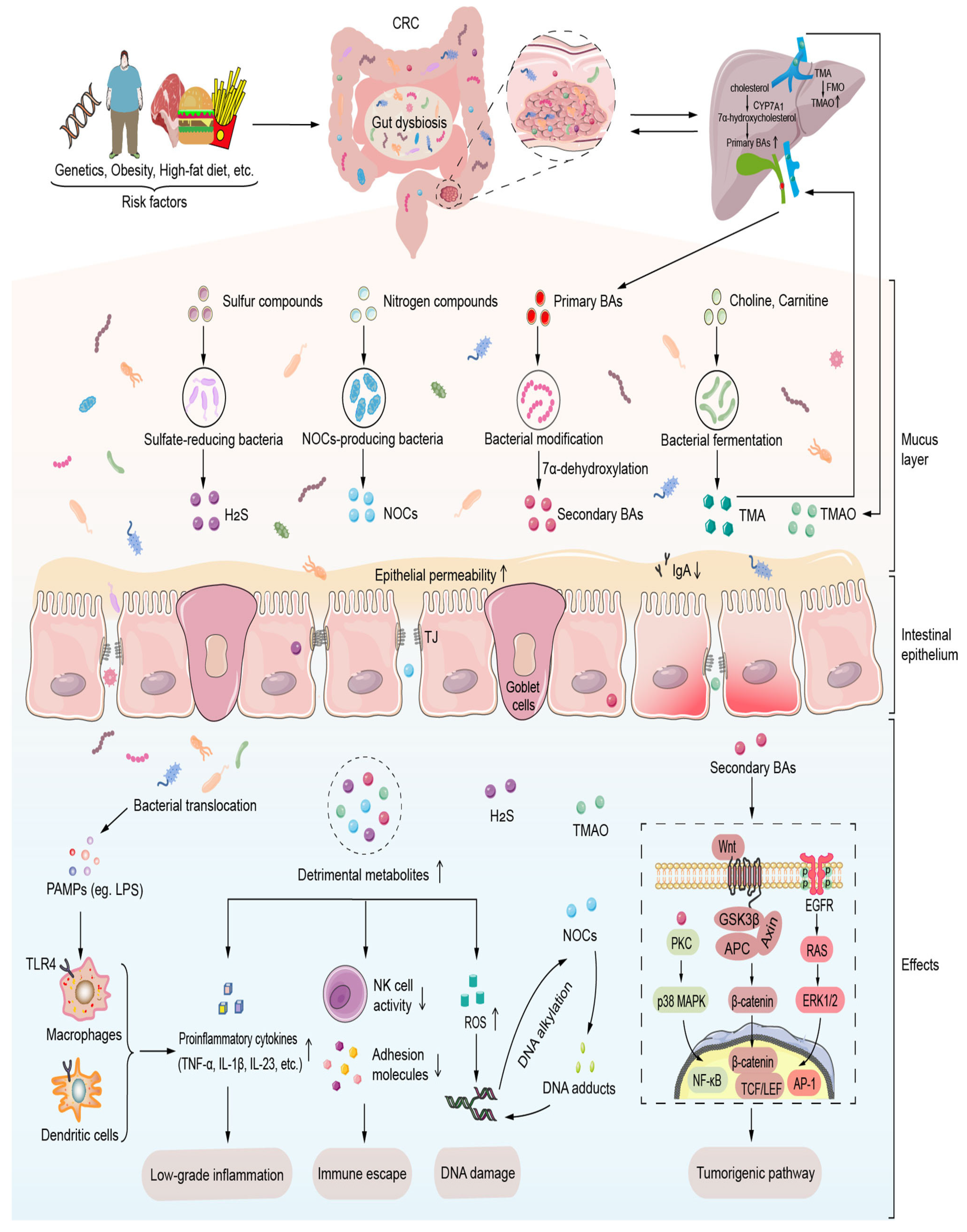
2.1. Microbial Dysbiosis in Gastric Cancer
2.2. Inflammatory Pathways and Immune Modulation
2.3. Carcinogenic Metabolites
2.4. Host–Microbiota Gene Interactions
2.5. Animal Model Evidence
3. Impact of Helicobacter pylori and Beyond
H. pylori and Gastric Carcinogenesis
4. Microbiota-Driven Inflammation and Cancer Progression
5. Post-Helicobacter pylori Eradication and Gastric Cancer Risk
6. Impact of Chemotherapy on the Gut Microbiome
7. Emerging Therapeutic Perspectives
| Intervention | Type of Study | Patient Population | Key Findings | Trial Phase | Evidence Level | Notes |
|---|---|---|---|---|---|---|
| Probiotics (Lactobacillus, multi-strain) [93] | Meta-analyses of RCTs | Chronic gastritis, peptic ulcer, dyspepsia, and some with failed eradication | Modest increase in H. pylori eradication rates, reduced side effects; heterogeneous data, mostly Asian studies | Meta-analyses (RCT) | Evidence-based (hypothesis-generating) | Not routine standard care in North America due to heterogeneity and lack of high-quality RCTs |
| Probiotics (L. acidophilus, B. lactis) [94] | RCT | H. pylori with premalignant intestinal metaplasia post-eradication | Higher regression of intestinal metaplasia (IM) (37.5% vs. 11.5%, OR 4.60, p = 0.025); miR-185 levels correlated with response | Phase II RCT | Investigational | Mechanistic support via COX-2/β-catenin pathway regulation; no current role in established gastric cancer |
| Prebiotics [95] | Review/Conceptual | General (in H. pylori infection context) | Promote growth of beneficial bacteria; no direct clinical trial data mentioned | Pre-clinical | Investigational | Potential to restore microbial balance; clinical trials needed |
| Synbiotics (probiotic + prebiotic) [97] | Meta-analyses of RCTs | H. pylori eradication adjunct | Possible increased eradication and decreased adverse events; statistically insignificant due to limited data | Meta-analyses (RCT) | Investigational | More RCTs needed to confirm effects |
| Fecal microbiota transplantation (FMT) [98] | RCT | H. pylori patients after bismuth quadruple therapy | No effect on eradication rates; alleviated short-term GI symptoms; microbiota returned to baseline by 10 weeks | Phase II RCT | Investigational | Small sample (30 pts); no acceleration of microbiota restoration |
| FMT (allogenic vs. autologous) [99] | RCT | Metastatic HER-2-negative gastric adenocarcinoma | Improved disease control rate (DCR), overall survival (OS), and progression-free survival (PFS) with allogenic FMT | Phase II RCT | Investigational | Primary satiety endpoint not met; promising microbiota engraftment; larger studies needed |
8. Future Directions and Research Gaps
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yang, Q.; Xu, D.; Yang, Y.; Lu, S.; Wang, D.; Wang, L. Global, Regional, and National Burden of Gastric Cancer in Adolescents and Young Adults, 1990-2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Am. J. Gastroenterol. 2024, 119, 454–467. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Collatuzzo, G.; Cortez Lainez, J.; Pelucchi, C.; Negri, E.; Bonzi, R.; Palli, D.; Ferraroni, M.; Zhang, Z.-F.; Yu, G.-P.; Lunet, N.; et al. The association between dietary fiber intake and gastric cancer: A pooled analysis of 11 case–control studies. Eur. J. Nutr. 2024, 63, 1857–1865. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Park, J.Y.; Camargo, M.C.; Lunet, N.; Forman, D.; Soerjomataram, I. Is gastric cancer becoming a rare disease? A global assessment of predicted incidence trends to 2035. Gut 2020, 69, 823–829. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Qi, J.; Li, M.; Wang, L.; Hu, Y.; Liu, W.; Long, Z.; Zhou, Z.; Yin, P.; Zhou, M. National and subnational trends in cancer burden in China, 2005–2020: An analysis of national mortality surveillance data. Lancet Public Health 2023, 8, e943–e955. [Google Scholar] [CrossRef] [PubMed]
- GBD 2017 Stomach Cancer Collaborators. The global, regional, and national burden of stomach cancer in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 42–54. [Google Scholar] [CrossRef]
- Li, Y.; Hahn, A.I.; Laszkowska, M.; Jiang, F.; Zauber, A.G.; Leung, W.K. Global burden of young-onset gastric cancer: A systematic trend analysis of the global burden of disease study 2019. Gastric Cancer 2024, 27, 684–700. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, L.; Dong, Q.; Wang, Y.; Li, X.; Li, C.; Li, F.; Zhang, J. Global trends and risk factors in gastric cancer: A comprehensive analysis of the Global Burden of Disease Study 2021 and multi-omics data. Int. J. Med. Sci. 2025, 22, 341–356. [Google Scholar] [CrossRef]
- Li, R.; Yang, T.; Dong, Z.; Gao, Y.; Li, N.; Song, T.; Sun, J.; Chen, Y. Factors influencing the incidence of early gastric cancer: A bayesian network analysis. BMC Gastroenterol. 2025, 25, 194. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, Y.; Hahn, A.I.; Laszkowska, M.; Jiang, F.; Zauber, A.G.; Leung, W.K. Clinicopathological characteristics and risk factors of young-onset gastric carcinoma: A systematic review and meta-analysis. Clin. Transl. Gastroenterol. 2024, 15, e00714. [Google Scholar] [CrossRef]
- Kawai, S.; Wang, C.; Lin, Y.; Sasakabe, T.; Okuda, M.; Kikuchi, S. Lifetime incidence risk for gastric cancer in the Helicobacter pylori-infected and uninfected population in Japan: A Monte Carlo simulation study. Int. J. Cancer 2022, 150, 18–27. [Google Scholar] [CrossRef]
- Trinh, T.T.K.; Lee, K.; Oh, J.-K.; Suh, M.; Jun, J.K.; Choi, K.S. Cluster of lifestyle risk factors for stomach cancer and screening behaviors among Korean adults. Sci. Rep. 2023, 13, 17503. [Google Scholar] [CrossRef]
- Huang, D.; Song, M.; Abe, S.K.; Rahman, M.S.; Islam, M.R.; Saito, E.; De la Torre, K.; Sawada, N.; Tamakoshi, A.; Shu, X.O.; et al. Family history and gastric cancer incidence and mortality in Asia: A pooled analysis of more than half a million participants. Gastric Cancer 2024, 27, 701–713. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Smyth, E.C.; Nilsson, M.; Grabsch, H.I.; van Grieken, N.C.T.; Lordick, F. Gastric cancer. Lancet 2020, 396, 635–648. [Google Scholar] [CrossRef]
- Choi, H.G.; Chun, W.; Jung, K.H. Association between gastric cancer and the family history of gastric cancer: A cross-sectional study using Korean Genome and Epidemiology Study data. Eur. J. Cancer Prev. 2022, 31, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Mabe, K.; Inoue, K.; Kamada, T.; Kato, K.; Kato, M.; Haruma, K. Endoscopic screening for gastric cancer in Japan: Current status and future perspectives. Dig. Endosc. 2022, 34, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Li, H.; Li, N.; Shi, J.F.; Li, J.; Chen, H.D.; Yu, Y.W.; Qin, C.; Ren, J.S.; Chen, W.Q.; et al. Risk factors for gastric cancer: A large-scale, population-based case-control study. Chin. Med. J. 2021, 134, 1952–1958. [Google Scholar] [CrossRef]
- Zeng, R.; Gou, H.; Lau, H.C.H.; Yu, J. Stomach microbiota in gastric cancer development and clinical implications. Gut 2024, 73, 2062–2073. [Google Scholar] [CrossRef]
- Yu, C.; Su, Z.; Li, Y.; Li, Y.; Liu, K.; Chu, F.; Liu, T.; Chen, R.; Ding, X. Dysbiosis of gut microbiota is associated with gastric carcinogenesis in rats. Biomed. Pharmacother. 2020, 126, 110036. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Tian, C.; Zou, Z.; Jin, M.; Liu, H. Gastrointestinal microbiota in gastric cancer: Potential mechanisms and clinical applications—A literature review. Cancers 2024, 16, 3547. [Google Scholar] [CrossRef]
- Schwabe, R.F.; Jobin, C. The microbiome and cancer. Nat. Rev. Cancer 2013, 13, 800–812. [Google Scholar] [CrossRef]
- Nasr, R.; Shamseddine, A.; Mukherji, D.; Nassar, F.; Temraz, S. The crosstalk between microbiome and immune response in gastric cancer. Int. J. Mol. Sci. 2020, 21, 6586. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; He, Y.; Zhang, X.; Fu, B.; Song, Z.; Wang, L.; Fu, R.; Lu, X.; Xing, J.; Lv, J.; et al. Sustained exposure to Helicobacter pylori induces immune tolerance by desensitizing TLR6. Gastric Cancer 2024, 27, 324–342. [Google Scholar] [CrossRef]
- Marasco, G.; Colecchia, L.; Salvi, D.; Bruni, A.; Capelli, C.; Dajti, E.; Barbaro, M.R.; Cremon, C.; Stanghellini, V.; Barbara, G. The role of microbiota in upper gastrointestinal cancers. Cancers 2025, 17, 1719. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; An, Y.; Qin, X.; Wu, X.; Wang, X.; Hou, H.; Song, X.; Liu, T.; Wang, B.; Huang, X.; et al. Gut microbiota-derived metabolites in colorectal cancer: The bad and the challenges. Front. Oncol. 2021, 11, 739648. [Google Scholar] [CrossRef]
- Lei, C.; Xu, Y.; Zhang, S.; Huang, C.; Qin, J. The role of microbiota in gastric cancer: A comprehensive review. Helicobacter 2024, 29, e13071. [Google Scholar] [CrossRef]
- Liu, C.; Fu, L.; Wang, Y.; Yang, W. Influence of the gut microbiota on immune cell interactions and cancer treatment. J. Transl. Med. 2024, 22, 939. [Google Scholar] [CrossRef] [PubMed]
- Wroblewski, L.E.; Peek, R.M., Jr. Helicobacter pylori, cancer, and the gastric microbiota. Adv. Exp. Med. Biol. 2016, 908, 393–408. [Google Scholar]
- Liatsos, C.; Papaefthymiou, A.; Kyriakos, N.; Galanopoulos, M.; Doulberis, M.; Giakoumis, M.; Petridou, E.; Mavrogiannis, C.; Rokkas, T.; Kountouras, J. Helicobacter pylori, gastric microbiota and gastric cancer relationship: Unrolling the tangle. World J. Gastrointest. Oncol. 2022, 14, 959–972. [Google Scholar] [CrossRef]
- Guo, Y.; Cao, X.S.; Zhou, M.G.; Yu, B. Gastric microbiota in gastric cancer: Different roles of Helicobacter pylori and other microbes. Front. Cell. Infect. Microbiol. 2023, 12, 1105811. [Google Scholar] [CrossRef]
- Santos, M.L.C.; de Brito, B.B.; da Silva, F.A.F.; Sampaio, M.M.; Marques, H.S.; Oliveira e Silva, N.; de Magalhães Queiroz, D.M.; de Melo, F.F. Helicobacter pylori infection: Beyond gastric manifestations. World J. Gastroenterol. 2020, 26, 4076–4093. [Google Scholar] [CrossRef]
- Kalisperati, P.; Spanou, E.; Pateras, I.S.; Korkolopoulou, P.; Varvarigou, A.; Karavokyros, I.; Gorgoulis, V.G.; Vlachoyiannopoulos, P.G.; Sougioultzis, S. Inflammation, DNA damage, Helicobacter pylori and gastric tumorigenesis. Front. Genet. 2017, 8, 20. [Google Scholar] [CrossRef]
- Salvatori, S.; Marafini, I.; Laudisi, F.; Monteleone, G.; Stolfi, C. Helicobacter pylori and gastric cancer: Pathogenetic mechanisms. Int. J. Mol. Sci. 2023, 24, 2895. [Google Scholar] [CrossRef]
- Ohnishi, N.; Yuasa, H.; Tanaka, S.; Sawa, H.; Miura, M.; Matsui, A.; Higashi, H.; Musashi, M.; Iwabuchi, K.; Suzuki, M.; et al. Transgenic expression of Helicobacter pylori CagA induces gastrointestinal and hematopoietic neoplasms in mouse. Proc. Natl. Acad. Sci. USA 2008, 105, 1003–1008. [Google Scholar] [CrossRef]
- Palframan, S.L.; Kwok, T.; Gabriel, K. Vacuolating cytotoxin A (VacA), a key toxin for Helicobacter pylori pathogenesis. Front. Cell. Infect. Microbiol. 2012, 2, 92. [Google Scholar] [CrossRef]
- Yousefi, B.; Mohammadlou, M.; Abdollahi, M.; Salek Farrokhi, A.; Karbalaei, M.; Keikha, M.; Kokhaei, P.; Valizadeh, S.; Rezaiemanesh, A.; Arabkari, V.; et al. Epigenetic changes in gastric cancer induction by Helicobacter pylori. J. Cell Physiol. 2019, 234, 21770–21784. [Google Scholar] [CrossRef]
- Ferreira, R.M.; Pereira-Marques, J.; Pinto-Ribeiro, I.; Costa, J.L.; Carneiro, F.; Machado, J.C.; Figueiredo, C. Gastric microbial community profiling reveals a dysbiotic cancer-associated microbiota. Gut 2018, 67, 226–236. [Google Scholar] [CrossRef]
- Pădureanu, V.; Dop, D.; Caragea, D.C.; Rădulescu, D.; Pădureanu, R.; Forțofoiu, M.C. Cardiovascular and neurological diseases and association with Helicobacter pylori infection: An overview. Diagnostics 2024, 14, 1781. [Google Scholar] [CrossRef] [PubMed]
- Magen, E.; Delgado, J.S. Helicobacter pylori and skin autoimmune diseases. World J. Gastroenterol. 2014, 20, 1510–1516. [Google Scholar] [CrossRef] [PubMed]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef]
- Vargas, A.; Robinson, B.L.; Houston, K.; Vilela Sangay, A.R.; Saadeh, M.; D’Souza, S.; Johnson, D.A. Gut microbiota-derived metabolites and chronic inflammatory diseases. Explor. Med. 2025, 6, 1001275. [Google Scholar] [CrossRef]
- He, Y.; Huang, J.; Li, Q.; Xia, W.; Zhang, C.; Liu, Z.; Xiao, J.; Yi, Z.; Deng, H.; Xiao, Z.; et al. Gut microbiota and tumor immune escape: A new perspective for improving tumor immunotherapy. Cancers 2022, 14, 5317. [Google Scholar] [CrossRef]
- Fasano, A. The intestinal epithelial barrier: The physical barrier that prevents bacterial translocation and gut inflammation. In Physical and Biological Barriers at the Interface Between the Gut Microbiome and the Immune System; The Microbiomes of Humans, Animals, Plants, and the Environment; Falcone, M., Antonini Cencicchio, M., Eds.; Springer Nature Switzerland: Cham, Switzerland, 2025; Volume 5. [Google Scholar]
- Shemtov, S.J.; Emani, R.; Bielska, O.; Covarrubias, A.J.; Verdin, E.; Andersen, J.K.; Winer, D.A. The intestinal immune system and gut barrier function in obesity and ageing. FEBS J. 2023, 290, 4163–4186. [Google Scholar] [CrossRef] [PubMed]
- Selvakumar, B.; Sekar, P.; Samsudin, A.R. Intestinal macrophages in pathogenesis and treatment of gut leakage: Current strategies and future perspectives. J. Leukoc. Biol. 2024, 115, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wu, M. Pattern recognition receptors in health and diseases. Signal Transduct. Target. Ther. 2021, 6, 291. [Google Scholar] [CrossRef]
- Di Lorenzo, A.; Bolli, E.; Tarone, L.; Cavallo, F.; Conti, L. Toll-like receptor 2 at the crossroad between cancer cells, the immune system, and the microbiota. Int. J. Mol. Sci. 2020, 21, 9418. [Google Scholar] [CrossRef]
- Lu, Q.; Ding, H.; Li, W. Role of Toll-like receptors in microbiota-associated gastrointestinal cancer metastasis. J. Cancer Res. Ther. 2013, 9 (Suppl. S3), S142–S149. [Google Scholar] [PubMed]
- Li, Z.; Xiong, W.; Liang, Z. Critical role of the gut microbiota in immune responses and cancer immunotherapy. J. Hematol. Oncol. 2024, 17, 33. [Google Scholar] [CrossRef]
- Prasad, S.; Singh, S.; Menge, S.; Mohapatra, I.; Kim, S.; Helland, L. Gut redox and microbiome: Charting the roadmap to T-cell regulation. Front. Immunol. 2024, 15, 1387903. [Google Scholar] [CrossRef]
- Bernstein, H.; Bernstein, C.; Payne, C.M.; Beard, S.E.; Schneider, J. Bile acids as carcinogens in human gastrointestinal cancers. Mutat. Res. 2005, 589, 47–65. [Google Scholar] [CrossRef] [PubMed]
- Attene-Ramos, M.S.; Wagner, E.D.; Plewa, M.J.; Gaskins, H.R. Evidence that hydrogen sulfide is a genotoxic agent. Mol. Cancer Res. 2006, 4, 9–14. [Google Scholar] [CrossRef]
- Fung, K.Y.C.; Cosgrove, L.; Lockett, T.; Head, R.; Topping, D.L. A review of the potential mechanisms for the lowering of colorectal oncogenesis by butyrate. Br. J. Nutr. 2012, 108, 820–831. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhang, D.Y.; Li, D.; Zhou, Y.; Wang, H.; Lin, J. Human gastric microbiota analysis of refractory Helicobacter pylori infection. Sci. Rep. 2024, 14, 15619. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.S.; Leung, W.K. Risk of gastric cancer development after eradication of Helicobacter pylori. World J. Gastrointest. Oncol. 2018, 10, 115–123. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shin, C.M.; Kim, N.; Park, J.H.; Lee, D.H. Changes in gastric corpus microbiota with age and after Helicobacter pylori eradication: A long-term follow-up study. Front. Microbiol. 2021, 11, 621879. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ge, Y.; Wang, X.; Guo, Y.; Yan, J.; Abuduwaili, A.; Aximujiang, K.; Yan, J.; Wu, M. Correction to: Gut microbiota influence tumor development and alter interactions with the human immune system. J. Exp. Clin. Cancer Res. 2021, 40, 334. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sitkin, S.; Lazebnik, L. Gastrointestinal microbiome and Helicobacter pylori: Eradicate, leave it as it is, or take a personalized benefit-risk approach? World J. Gastroenterol. 2022, 28, 766–774. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sharma, S.; Shukla, G.; Singh, A.K.; Ghosh, M.; Jha, N.K. Probiotics and cancer: Mechanistic insights and organ-specific evidence. Crit. Rev. Food Sci. Nutr. 2022, 62, 5301–5320. [Google Scholar]
- Kim, Y.; Kim, G.; Kim, S.; Lee, J.; Lee, H.; Park, J.; Lee, S.; Kim, H.; Lee, Y.; Lee, K.; et al. Fecal microbiota transplantation improves anti-PD-1 inhibitor efficacy in unresectable or metastatic solid cancers refractory to anti-PD-1 inhibitor. Cell Host Microbe 2024, 32, 1380–1393.e9. [Google Scholar] [CrossRef]
- Pérez Escriva, P.; Correia Tavares Bernardino, C.; Letellier, E. De-coding the complex role of microbial metabolites in cancer. Cell Rep. 2025, 44, 115358. [Google Scholar] [CrossRef]
- Serrano, C.; Harris, P.R.; Smith, P.D.; Bimczok, D. Interactions between Helicobacter pylori and the gastric microbiome: Impact on gastric homeostasis and disease. Curr. Opin. Physiol. 2021, 21, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Hou, K.; Wu, Z.-X.; Chen, X.-Y.; Wang, J.-Q.; Zhang, D.; Xiao, C.; Dan, Z.; Koya, J.B.; Wei, L.; Li, J.; et al. Microbiota in health and diseases. Signal Transduct. Target. Ther. 2022, 7, 135. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.J.; Kim, C.G.; Lee, J.Y.; Kim, Y.I.; Kook, M.C.; Park, B.; Joo, J. Family history of gastric cancer and Helicobacter pylori treatment. N. Engl. J. Med. 2020, 382, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.Y.; Rugge, M.; Genta, R.M. Diagnosis: Gastric intestinal metaplasia—What to do next? Curr. Opin. Gastroenterol. 2019, 35, 535–543. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ford, A.C.; Yuan, Y.; Park, J.Y.; Forman, D.; Moayyedi, P. Eradication therapy to prevent gastric cancer in Helicobacter pylori-positive individuals: Systematic review and meta-analysis of randomized controlled trials and observational studies. Gastroenterology 2025, 169, 261–276. [Google Scholar] [CrossRef] [PubMed]
- Blair, V.R.; McLeod, M.; Carneiro, F.; Coit, D.G.; D’Addario, J.L.; van Dieren, J.M.; Caldas, C.; Oliveira, C.; Huntsman, D.; Kaurah, P.; et al. Hereditary diffuse gastric cancer: Updated clinical practice guidelines. Lancet Oncol. 2020, 21, e386–e397. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jaroenlapnopparat, A.; Bhatia, K.; Coban, S. Inflammation and gastric cancer. Diseases 2022, 10, 35. [Google Scholar] [CrossRef]
- He, P.; Li, X.; Zou, D.; Tang, F.; Chen, H.; Li, Y. Environmental factors inducing gastric cancer: Insights into risk and prevention strategies. Discov. Oncol. 2025, 16, 25. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wiklund, A.-K.; Santoni, G.; Yan, J.; Radkiewicz, C.; Xie, S.; Birgisson, H.; Ness-Jensen, E.; von Euler-Chelpin, M.; Kauppila, J.H.; Lagergren, J. Risk of gastric adenocarcinoma after eradication of Helicobacter pylori. Gastroenterology 2025, 169, 244–250. [Google Scholar] [CrossRef]
- Sugano, K.; Tack, J.; Kuipers, E.J.; Graham, D.Y.; El-Omar, E.M.; Miura, S.; Malfertheiner, P.; Asaka, M.; Bazzoli, F.; Malagelada, J.-R.; et al. faculty members of Kyoto Global Consensus Conference. Kyoto global consensus report on Helicobacter pylori gastritis. Gut 2015, 64, 1353–1367. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gawron, A.J.; Shah, S.C.; Altayar, O.; Davitkov, P.; Morgan, D.; Turner, K.; Mustafa, R.A. AGA technical review on gastric intestinal metaplasia—Natural history and clinical outcomes. Gastroenterology 2020, 158, 705–731.e5. [Google Scholar] [CrossRef] [PubMed]
- Khazaaleh, S.; Alomari, M.; Ur Rashid, M.; Castaneda, D.; Castro, F.J. Gastric intestinal metaplasia and gastric cancer prevention: Watchful waiting. Clevel. Clin. J. Med. 2024, 91, 33–39. [Google Scholar] [CrossRef]
- Shiotani, A.; Haruma, K.; Graham, D.Y. Metachronous gastric cancer after successful Helicobacter pylori eradication. World J. Gastroenterol. 2014, 20, 11552–11559. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dinis-Ribeiro, M.; Kuipers, E.J. How to manage a patient with gastric intestinal metaplasia: An international perspective. Gastroenterology 2020, 158, 1534–1537. [Google Scholar] [CrossRef] [PubMed]
- Botezatu, A.; Farcas, R.A.; Grad, S.; Dumitrașcu, D.L.; Bodrug, N.; Rugge, M. Assessing gastric cancer risk using the OLGA and OLGIM systems in Republic of Moldova. Front. Med. 2025, 12, 1563889. [Google Scholar] [CrossRef] [PubMed]
- Oh, A.; Rustgi, S.D.; Hur, C.; In, H. Cost-effectiveness of serum pepsinogen as a gastric cancer targeted screening strategy in the United States. Gastro Hep Adv. 2025, 4, 100564. [Google Scholar] [CrossRef] [PubMed]
- Teshima, H.; Takigawa, H.; Kotachi, T.; Tsuboi, A.; Tanaka, H.; Yamashita, K.; Kishida, Y.; Urabe, Y.; Kuwai, T.; Ishikawa, A.; et al. A proton pump inhibitor independently elevates gastrin levels as a marker for metachronous gastric cancer after endoscopic submucosal dissection. J. Clin. Med. 2024, 13, 6599. [Google Scholar] [CrossRef]
- Murphy, G.; Abnet, C.C.; Choo-Wosoba, H.; Vogtmann, E.; Weinstein, S.J.; Taylor, P.R.; Männistö, S.; Albanes, D.; Dawsey, S.M.; Rehfeld, J.F.; et al. Serum gastrin and cholecystokinin are associated with subsequent development of gastric cancer in a prospective cohort of Finnish smokers. Int. J. Epidemiol. 2017, 46, 914–923. [Google Scholar] [CrossRef]
- Ali, H.; Muzammil, M.A.; Dahiya, D.S.; Ali, F.; Yasin, S.; Hanif, W.; Gangwani, M.K.; Aziz, M.; Khalaf, M.; Basuli, D.; et al. Artificial intelligence in gastrointestinal endoscopy: A comprehensive review. Ann. Gastroenterol. 2024, 37, 133–141. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, G.H.; Liang, P.S.; Bang, S.J.; Hwang, J.H. Screening and surveillance for gastric cancer in the United States: Is it needed? Gastrointest. Endosc. 2016, 84, 18–28. [Google Scholar] [CrossRef]
- Zhang, T.; Tang, X. Chemoprevention Strategies for Precancerous Gastric Lesions Beyond Helicobacter pylori Eradication. QJM Mon. J. Assoc. Physicians 2025, hcaf030. [Google Scholar] [CrossRef]
- Teshima, H.; Kotachi, T.; Kuwai, T.; Tsuboi, A.; Tanaka, H.; Yamashita, K.; Takigawa, H.; Kishida, Y.; Urabe, Y.; Oka, S. Clinicopathologic features of early gastric cancer after Helicobacter pylori eradication in Japanese patients: Comparative study between early (<10 years) and late (>10 years) onset. Cancers 2024, 16, 3154. [Google Scholar] [CrossRef]
- Zhao, L.-Y.; Mei, J.-X.; Yu, G.; Lei, L.; Zhang, W.-H.; Liu, K.; Chen, X.-L.; Kołat, D.; Yang, K.; Hu, J.-K. Role of the gut microbiota in anticancer therapy: From molecular mechanisms to clinical applications. Signal Transduct. Target. Ther. 2023, 8, 201. [Google Scholar] [CrossRef]
- Yang, S.; Hao, S.; Ye, H.; Zhang, X. Crosstalk between gut microbiota and cancer chemotherapy: Current status and trends. Discover Oncol. 2024, 15, 833. [Google Scholar] [CrossRef]
- Papanicolas, L.E.; Sims, S.K.; Taylor, S.L.; Miller, S.J.; Karapetis, C.S.; Wesselingh, S.L.; Gordon, D.L.; Rogers, G.B. Conventional myelosuppressive chemotherapy for non-haematological malignancy disrupts the intestinal microbiome. BMC Cancer 2021, 21, 591. [Google Scholar] [CrossRef] [PubMed]
- Žukauskaitė, K.; Baušys, B.; Horvath, A.; Sabaliauskaitė, R.; Šeštokaitė, A.; Mlynska, A.; Jarmalaitė, S.; Stadlbauer, V.; Baušys, R.; Baušys, A. Gut microbiome changes after neoadjuvant chemotherapy and surgery in patients with gastric cancer. Cancers 2024, 16, 4074. [Google Scholar] [CrossRef]
- Xu, Q.; Gao, J.; Zhao, R.; Li, H.; Cui, H.; Yuan, Z.; Ren, H.; Cao, B.; Wei, B. Akkermansia muciniphila-derived pentadecanoic acid enhances oxaliplatin sensitivity in gastric cancer by modulating glycolysis. Pharmacol. Res. 2024, 206, 107278. [Google Scholar] [CrossRef]
- Lin, H.Y.; Chen, C.Y.; Lin, T.C.; Yeh, L.F.; Hsieh, W.C.; Gao, S.; Burnouf, P.A.; Chen, B.M.; Hsieh, T.J.; Dashnyam, P.; et al. Entropy-driven binding of gut bacterial β-glucuronidase inhibitors ameliorates irinotecan-induced toxicity. Commun. Biol. 2021, 4, 280. [Google Scholar] [CrossRef]
- Feng, J.; Gao, M.; Zhao, C.; Yang, J.; Gao, H.; Lu, X.; Ju, R.; Zhang, X.; Zhang, Y. Oral administration of probiotics reduces chemotherapy-induced diarrhoea and oral mucositis: A systematic review and meta-analysis. Front. Nutr. 2022, 9, 823288. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Cao, C.; Ren, Y.; Weng, S.; Liu, L.; Guo, C.; Wang, L.; Han, X.; Ren, J.; Liu, Z. Antitumor effects of fecal microbiota transplantation: Implications for microbiome modulation in cancer treatment. Front. Immunol. 2022, 13, 949490. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.G.; Koh, J.Y.; Shin, S.J.; Shin, J.H.; Hong, M.; Chung, H.C.; Rha, S.Y.; Kim, H.S.; Lee, C.K.; Lee, J.H.; et al. Prior antibiotic administration disrupts anti-PD-1 responses in advanced gastric cancer by altering the gut microbiome and systemic immune response. Cell Rep. Med. 2023, 4, 101251. [Google Scholar] [CrossRef]
- Marashi, A.; Hasany, S.; Moghimi, S.; Kiani, R.; Mehran Asl, S.; Dareghlou, Y.A.; Lorestani, P.; Varmazyar, S.; Jafari, F.; Ataeian, S.; et al. Targeting gut-microbiota for gastric cancer treatment: A systematic review. Front. Med. 2024, 11, 1412709. [Google Scholar] [CrossRef]
- Chey, W.D.; Howden, C.W.; Moss, S.F.; Morgan, D.R.; Greer, K.B.; Grover, S.; Shah, S.C. ACG Clinical Guideline: Treatment of Helicobacter pylori Infection. Am. J. Gastroenterol. 2024, 119, 1730–1753. [Google Scholar] [CrossRef]
- Yang, Y.J.; Wu, C.T.; Cheng, H.C.; Chen, W.Y.; Tseng, J.T.; Chang, W.L.; Sheu, B.S. Probiotics ameliorate H. pylori-associated gastric β-catenin and COX-2 carcinogenesis signaling by regulating miR-185. J. Biomed. Sci. 2025, 32, 55. [Google Scholar] [CrossRef]
- Wilson, B.; Whelan, K. Prebiotic inulin-type fructans and galacto-oligosaccharides: Definition, specificity, function, and application in gastrointestinal disorders. J. Gastroenterol. Hepatol. 2017, 32 (Suppl. S1), 64–68. [Google Scholar] [CrossRef]
- Khursheed, R.; Gulati, M.; Wadhwa, S.; Vishwas, S.; Sharma, D.S.; Corrie, L.; Alam, A.; Alnasser, S.M.; Aba Alkhayl, F.F.; Parveen, Z.; et al. Multifaceted role of synbiotics as nutraceuticals, therapeutics and carrier for drug delivery. Chem. Biol. Interact. 2022, 368, 110223. [Google Scholar] [CrossRef]
- Pourmasoumi, M.; Najafgholizadeh, A.; Hadi, A.; Mansour-Ghanaei, F.; Joukar, F. The effect of synbiotics in improving Helicobacter pylori eradication: A systematic review and meta-analysis. Complement. Ther. Med. 2019, 43, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.T.; Zhang, Y.; Wang, X.W.; Zou, P.Y.; Zhao, Z.; Mei, H.; Liu, Y.X.; Su, N.Y.; Zhu, Y.J.; Wang, B.; et al. Long-term effects of fecal microbiota transplantation on gut microbiota after Helicobacter pylori eradication with bismuth quadruple therapy: A randomized controlled trial. Helicobacter 2024, 29, e13079. [Google Scholar] [CrossRef] [PubMed]
- de Clercq, N.C.; van den Ende, T.; Prodan, A.; Hemke, R.; Davids, M.; Pedersen, H.K.; Nielsen, H.B.; Groen, A.K.; de Vos, W.M.; Nieuwdorp, M.; et al. Fecal Microbiota Transplantation from Overweight or Obese Donors in Cachectic Patients with Advanced Gastroesophageal Cancer: A Randomized, Double-blind, Placebo-Controlled, Phase II Study. Clin. Cancer Res. 2021, 27, 3784–3792. [Google Scholar] [CrossRef]
- Chen, Z.; Jin, D.; Hu, J.; Guan, D.; Bai, Q.; Gou, Y. Microbiota and gastric cancer: From molecular mechanisms to therapeutic strategies. Front. Cell Infect. Microbiol. 2025, 15, 1563061. [Google Scholar] [CrossRef] [PubMed]
- Zaramella, A.; Arcidiacono, D.; Duci, M.; Benna, C.; Pucciarelli, S.; Fantin, A.; Rosato, A.; De Re, V.; Cannizzaro, R.; Fassan, M.; et al. Predictive Value of a Gastric Microbiota Dysbiosis Test for Stratifying Cancer Risk in Atrophic Gastritis Patients. Nutrients 2024, 17, 142. [Google Scholar] [CrossRef]
- Liu, C.; Ng, S.K.; Ding, Y.; Lin, Y.; Liu, W.; Wong, S.H.; Sung, J.J.; Yu, J. Meta-analysis of mucosal microbiota reveals universal microbial signatures and dysbiosis in gastric carcinogenesis. Oncogene 2022, 41, 3599–3610. [Google Scholar] [CrossRef] [PubMed]
- Coker, O.O.; Dai, Z.; Nie, Y.; Zhao, G.; Cao, L.; Nakatsu, G.; Wu, W.K.K.; Wong, S.H.; Chen, Z.; Sung, J.J.Y.; et al. Mucosal microbiome dysbiosis in gastric carcinogenesis. Gut 2018, 67, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Han, W.; Wang, N.; Han, M.; Ban, M.; Dai, J.; Dong, Y.; Sun, T.; Xu, J. The role of microbiota in the development and treatment of gastric cancer. Front. Oncol. 2023, 13, 1224669. [Google Scholar] [CrossRef]
- Wizenty, J.; Sigal, M. Helicobacter pylori, microbiota and gastric cancer—Principles of microorganism-driven carcinogenesis. Nat. Rev. Gastroenterol. Hepatol. 2025, 22, 296–313. [Google Scholar] [CrossRef]
- Elghannam, M.T.; Hassanien, M.H. Gut microbiome and gastric cancer: Microbial interactions and therapeutic potential. Gut Pathog. 2025, 17, 29. [Google Scholar] [CrossRef]
- Elsalem, L.; Jum’ah, A.A.; Alfaqih, M.A.; Aloudat, O. The bacterial microbiota of gastrointestinal cancers: Role in cancer pathogenesis and therapeutic perspectives. Clin. Exp. Gastroenterol. 2020, 13, 151–185. [Google Scholar] [CrossRef] [PubMed]
| Microbial Genus/Group | Role in Gastric Cancer Pathogenesis | Clinical Relevance |
|---|---|---|
| H. pylori [40,53,54,55,56] | Dominates gastric microbiota; triggers Correa’s cascade; induces chronic inflammation, DNA damage, and epigenetic alterations, driving tumorigenesis. | Primary carcinogenic driver; eradication reduces gastric cancer risk but may cause dysbiosis, impacting long-term outcomes. |
| Firmicutes (e.g., Streptococcus, Lactobacillus) [53,54,57] | Reduced abundance during H. pylori infection; some species are linked to precancerous lesions and sustained inflammation. | Changes in abundance may signal premalignant conditions; some strains are considered for probiotic therapy. |
| Bacteroidetes [53,57] | Decreased post-H. pylori infection and eradication; plays role in maintaining microbial balance and gut homeostasis. | Loss linked to dysbiosis and gastrointestinal symptoms after eradication therapy. |
| Actinobacteria (e.g., Bifidobacterium, Actinomyces) [53,54,57] | Decreased after eradication therapy; protective role in gut homeostasis; implicated in precancerous lesion development. | Protective taxa that may be targeted to restore balance post-eradication; potential probiotic candidates. |
| Proteobacteria (e.g., Enterobacteriaceae, Escherichia-Shigella) [54,57] | Transiently increases after eradication; associated with dysbiosis and pro-inflammatory states. | Increased abundance linked to mucosal inflammation and potential carcinogenic microenvironment. |
| Clostridium [50] | Produces genotoxic secondary bile acids (e.g., deoxycholic acid) that induce DNA damage and promote tumor growth. | Potential biomarker for colorectal and gastric tumorigenesis; possible target for metabolite-based therapies. |
| Hydrogen sulfide-producing bacteria [51] | Generate hydrogen sulfide, impairing DNA repair mechanisms and facilitating mutagenesis. | Metabolic byproducts contribute to carcinogenesis; inhibitors may reduce genotoxic effects. |
| Short-chain fatty acid producers (e.g., butyrate producers) [52] | Usually anti-inflammatory, but under hypoxic or oncogenic conditions, it may promote tumor cell proliferation. | Complex role; modulation may have therapeutic potential depending on tumor microenvironment context. |
| Streptococcus, Prevotella, Lactobacillus [54,57] | Associated with gastric mucosal changes resembling precancerous conditions post-H. pylori eradication. | May serve as markers for monitoring premalignant progression; some species have been explored as probiotics. |
| Bacillus [54] | Increased abundance correlates with progression from atrophic gastritis to gastric cancer. | Included in predictive microbial biomarker panels for neoplastic progression risk stratification. |
| Parvimonas, Peptostreptococcus, Rothia, Granulicatella [57] | Implicated in the development of precancerous gastric lesions. | Potential contributors to carcinogenesis; further study needed for clinical application as biomarkers or targets. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohamed, A.S.; Bhuju, R.; Martinez, E.; Basta, M.; Deyab, A.; Mansour, C.; Tejada, D.; Deshpande, V.; Elias, S.; Nagesh, V.K. The Gut Microbiome’s Impact on the Pathogenesis and Treatment of Gastric Cancer—An Updated Literature Review. Cancers 2025, 17, 2795. https://doi.org/10.3390/cancers17172795
Mohamed AS, Bhuju R, Martinez E, Basta M, Deyab A, Mansour C, Tejada D, Deshpande V, Elias S, Nagesh VK. The Gut Microbiome’s Impact on the Pathogenesis and Treatment of Gastric Cancer—An Updated Literature Review. Cancers. 2025; 17(17):2795. https://doi.org/10.3390/cancers17172795
Chicago/Turabian StyleMohamed, Ahmed S., Ruchi Bhuju, Emelyn Martinez, Marina Basta, Ashrakat Deyab, Charlene Mansour, Daniel Tejada, Vishal Deshpande, Sameh Elias, and Vignesh Krishnan Nagesh. 2025. "The Gut Microbiome’s Impact on the Pathogenesis and Treatment of Gastric Cancer—An Updated Literature Review" Cancers 17, no. 17: 2795. https://doi.org/10.3390/cancers17172795
APA StyleMohamed, A. S., Bhuju, R., Martinez, E., Basta, M., Deyab, A., Mansour, C., Tejada, D., Deshpande, V., Elias, S., & Nagesh, V. K. (2025). The Gut Microbiome’s Impact on the Pathogenesis and Treatment of Gastric Cancer—An Updated Literature Review. Cancers, 17(17), 2795. https://doi.org/10.3390/cancers17172795







