Stem Cell Growth Factor-β as a Predictive Biomarker of Response to Chemotherapy and Prognosis in Patients with Advanced-Stage Hepatocellular Carcinoma: A Retrospective Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Patient Selection and Classification
2.3. Quantification of Serum Cytokines
2.4. Therapy Response Analysis
2.5. Survival Analysis
2.6. Statistical Analysis
3. Results
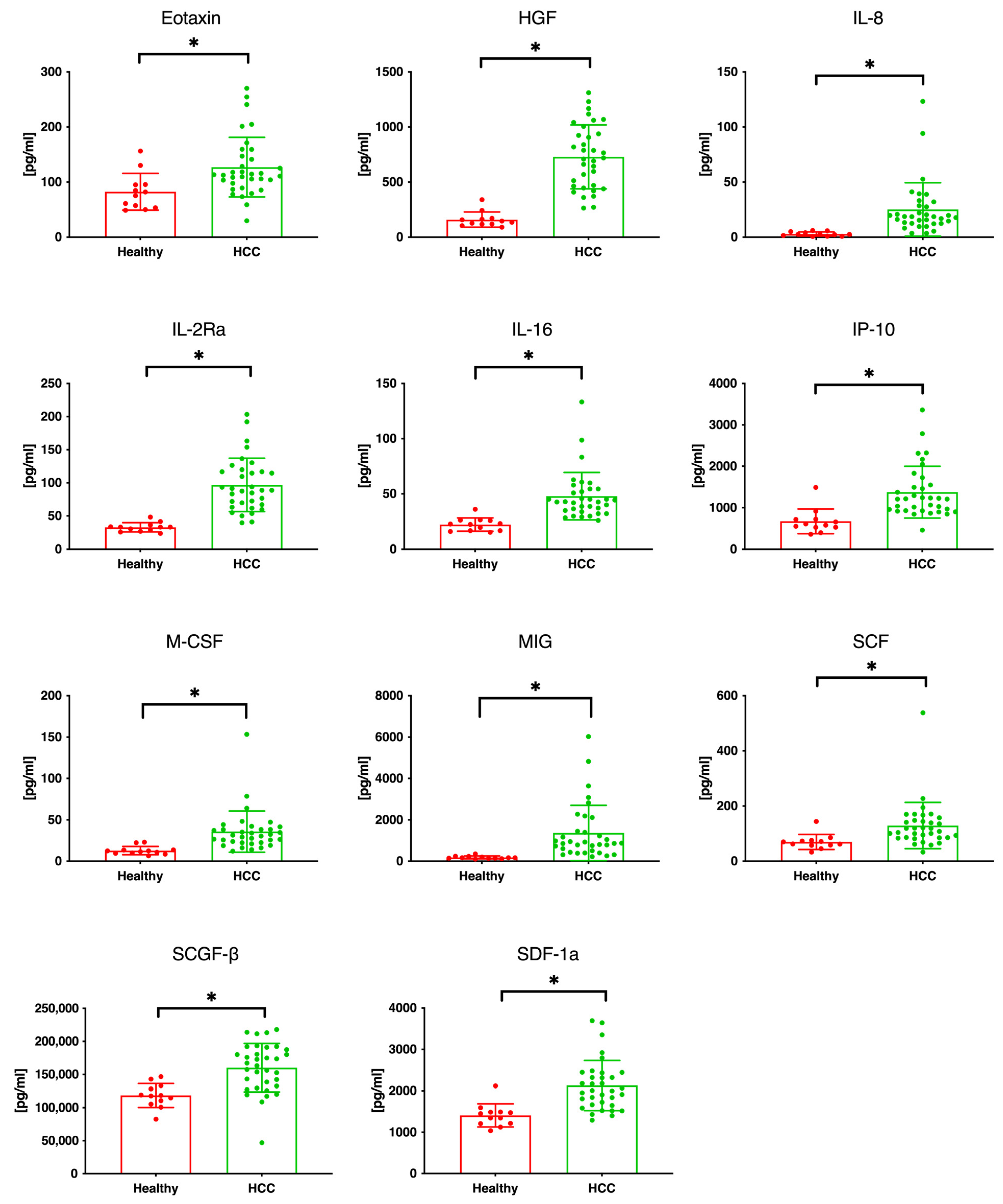
3.1. Serum Cytokine and Chemokine Levels in Patients with HCC
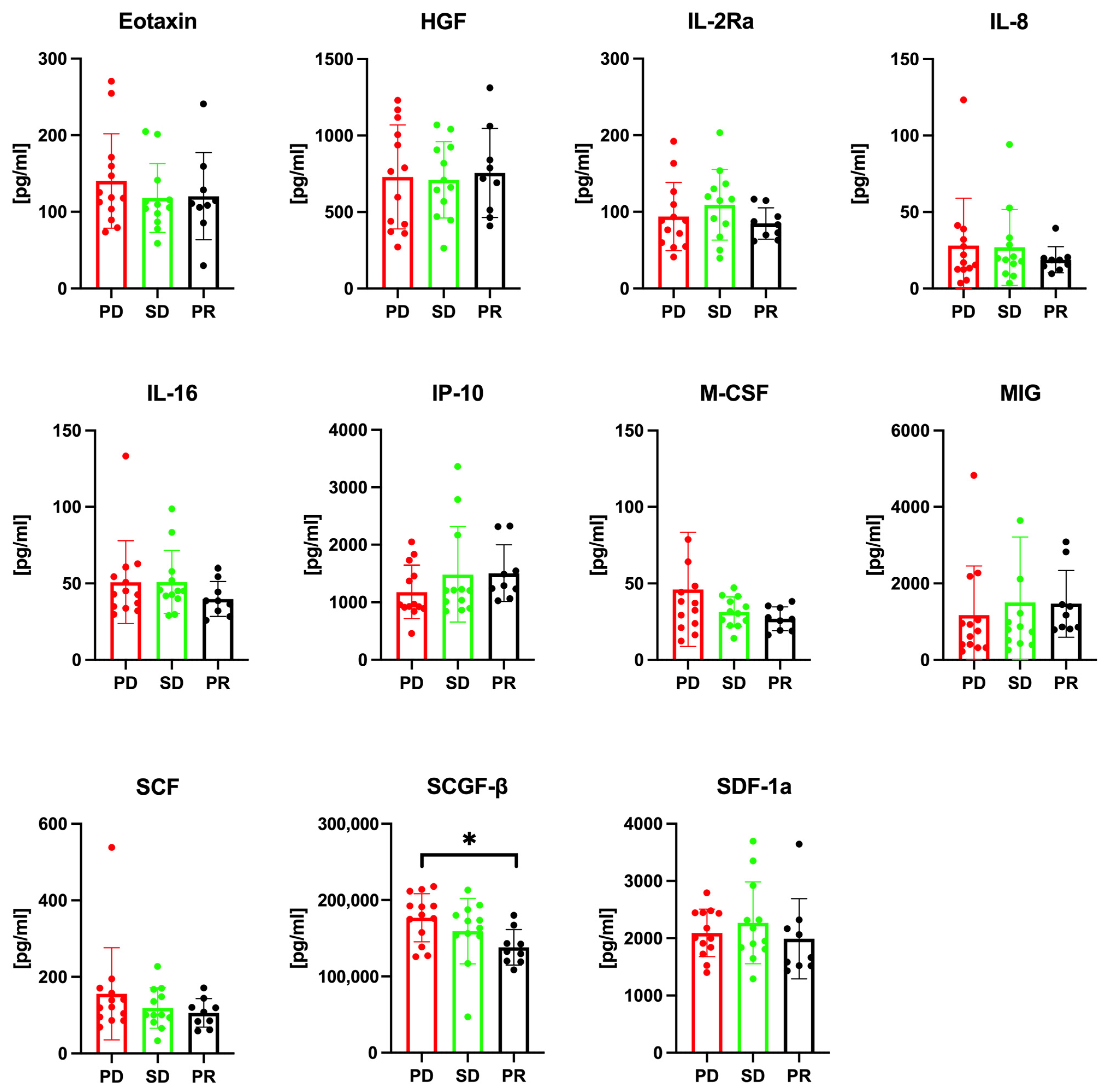
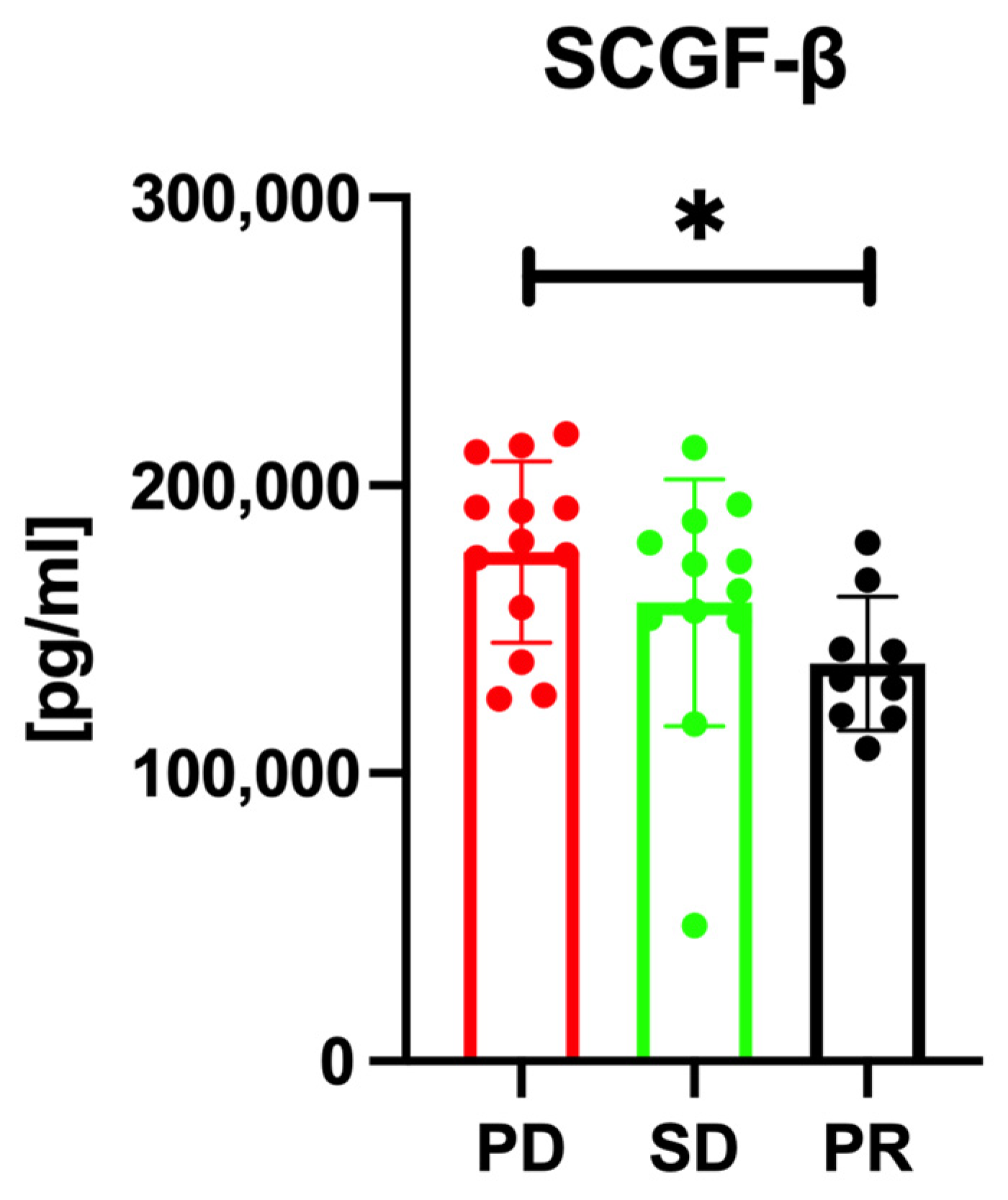
3.2. Serum SCGF-β Levels and Treatment Response Prediction
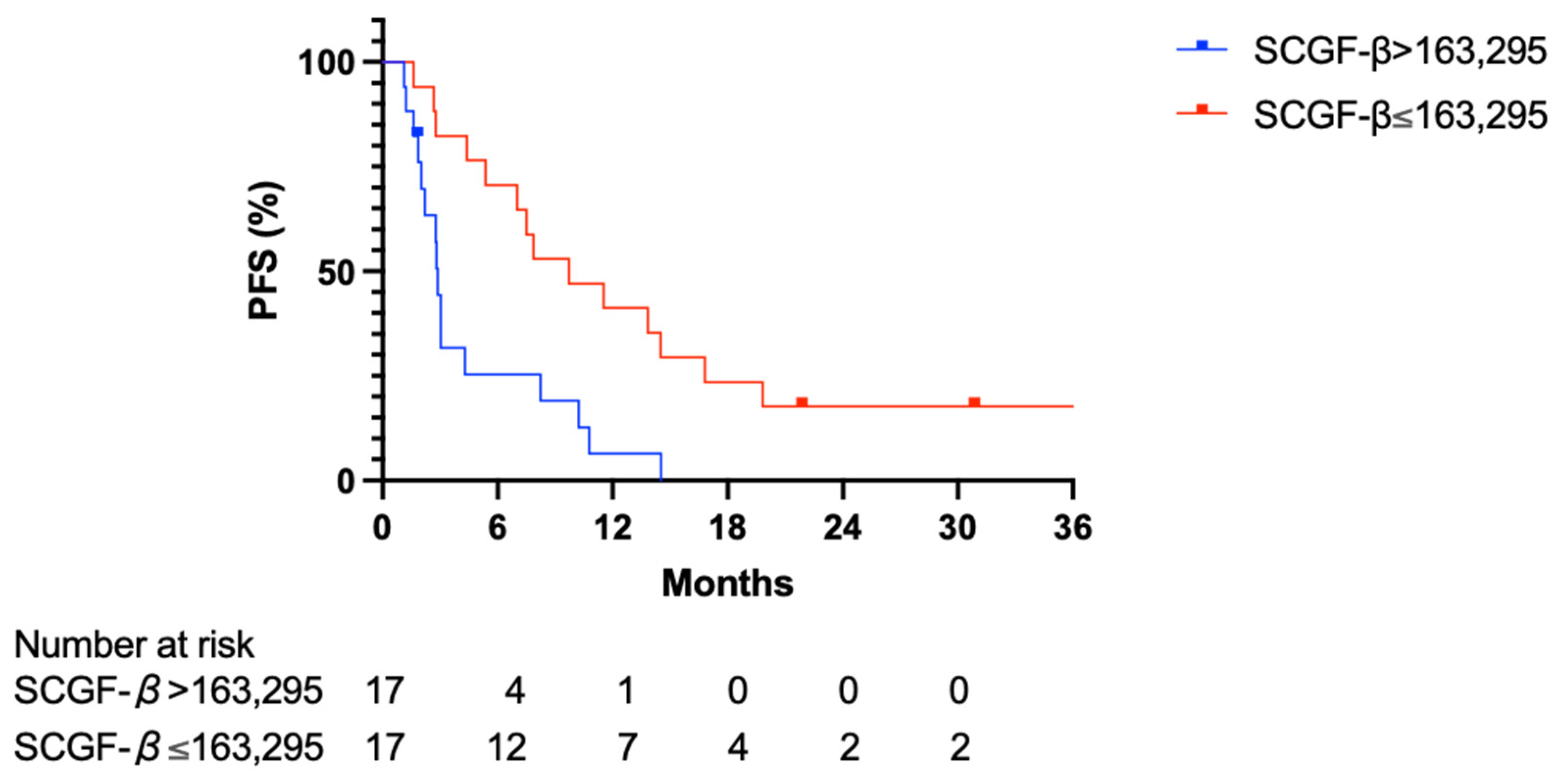
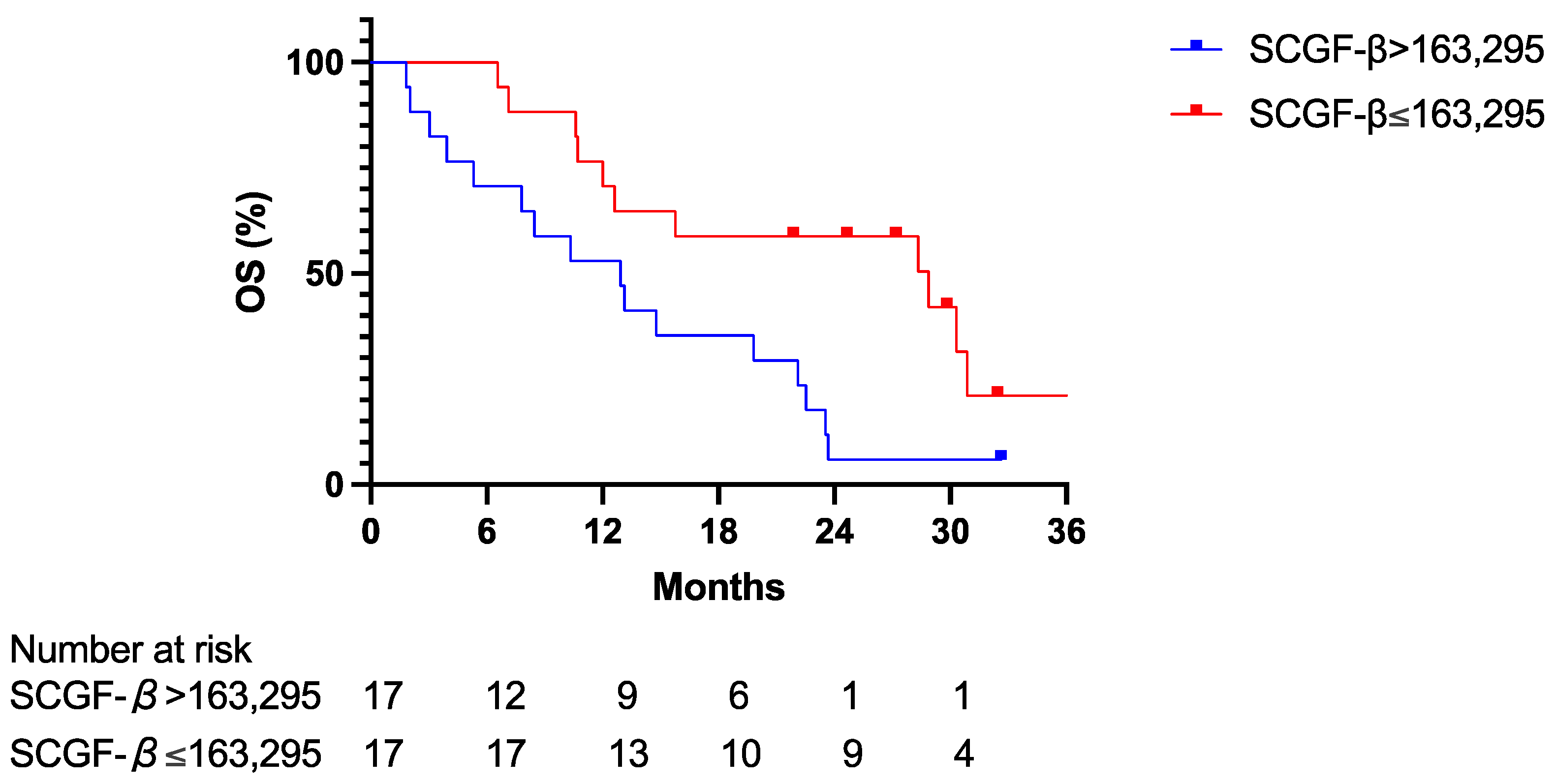
3.3. Predictive Value of Serum SCGF-β Level
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Zucman-Rossi, J.; Pikarsky, E.; Sangro, B.; Schwartz, M.; Sherman, M.; Gores, G. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2016, 2, 16018. [Google Scholar] [CrossRef]
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef] [PubMed]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Llovet, J.M.; Peña, C.E.; Lathia, C.D.; Shan, M.; Meinhardt, G.; Bruix, J.; SHARP Investigators Study Group. Plasma biomarkers as predictors of outcome in patients with advanced hepatocellular carcinoma. Clin. Cancer Res. 2012, 18, 2290–2300. [Google Scholar] [CrossRef]
- Jang, J.W.; Oh, B.S.; Kwon, J.H.; You, C.R.; Chung, K.W.; Kay, C.S.; Jung, H.S. Serum interleukin-6 and C-reactive protein as a prognostic indicator in hepatocellular carcinoma. Cytokine 2012, 60, 686–693. [Google Scholar] [CrossRef]
- Tangkijvanich, P.; Thong-Ngam, D.; Mhachai, V.; Theamboonlers, A.; Poovorawan, Y. Role of serum interleukin-18 as a prognostic factor in patients with hepatocellular carcinoma. World J. Gastroenterol. 2007, 13, 4345–4349. [Google Scholar] [CrossRef]
- Hiraoka, A.; Yano, K.I.; Kagami, N.; Takeshige, K.; Mio, H.; Anazawa, H.; Sugimoto, S. Stem cell growth factor: In situ hybridization analysis on the gene expression, molecular characterization and in vitro proliferative activity of a recombinant preparation on primitive hematopoietic progenitor cells. Hematol. J. 2001, 2, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Mego, M.; Cholujova, D.; Minarik, G.; Sedlackova, T.; Gronesova, P.; Karaba, M.; Benca, J.; Cingelova, S.; Cierna, Z.; Manasova, D.; et al. CXCR4-SDF-1 interaction potentially mediates trafficking of circulating tumor cells in primary breast cancer. BMC Cancer 2016, 16, 127. [Google Scholar] [CrossRef]
- Levina, V.; Marrangoni, A.M.; DeMarco, R.; Gorelik, E.; Lokshin, A.E. Drug-selected human lung cancer stem cells: Cytokine network, tumorigenic and metastatic properties. PLoS ONE 2008, 3, e3077. [Google Scholar] [CrossRef] [PubMed]
- Sukowati, C.H.; Patti, R.; Pascut, D.; Ladju, R.B.; Tarchi, P.; Zanotta, N.; Comar, M.; Tiribelli, C.; Crocè, L.S. Serum stem cell growth factor beta for the prediction of therapy response in hepatocellular carcinoma. BioMed Res. Int. 2018, 2018, 6435482. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Goldschmidt, I.; Chichelnitskiy, E.; Rübsamen, N.; Jaeger, V.K.; Karch, A.; D’Antiga, L.; Di Giorgio, A.; Nicastro, E.; Kelly, D.A.; McLin, V.; et al. Diagnosing acute cellular rejection after paediatric liver transplantation-is there room for interleukin profiles? Children 2023, 10, 128. [Google Scholar] [CrossRef]
- Hiraoka, A.; Sugimura, A.; Seki, T.; Nagasawa, T.; Ohta, N.; Shimonishi, M.; Hagiya, M.; Shimizu, S. Cloning, expression, and characterization of a cDNA encoding a novel human growth factor for primitive hematopoietic progenitor cells. Proc. Natl. Acad. Sci. USA 1997, 94, 7577–7582. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A. Leukemia cell lines require self-secreted stem cell growth factor (SCGF) for their proliferation. Leuk. Res. 2008, 32, 1623–1625. [Google Scholar] [CrossRef]
- Jiang, J.; Ye, F.; Yang, X.; Zong, C.; Gao, L.; Yang, Y.; Zhao, Q.; Han, Z.; Wei, L. Peri-tumor associated fibroblasts promote intrahepatic metastasis of hepatocellular carcinoma by recruiting cancer stem cells. Cancer Lett. 2017, 404, 19–28. [Google Scholar] [CrossRef]
- Bhojwani, D.; Kang, H.; Moskowitz, N.P.; Min, D.J.; Lee, H.; Potter, J.W.; Davidson, G.; Willman, C.L.; Borowitz, M.J.; Belitskaya-Levy, I.; et al. Biologic pathways associated with relapse in childhood acute lymphoblastic leukemia: A Children’s Oncology Group study. Blood 2006, 108, 711–717. [Google Scholar] [CrossRef]
- Svetlovska, D.; Miskovska, V.; Cholujova, D.; Gronesova, P.; Cingelova, S.; Chovanec, M.; Sycova-Mila, Z.; Obertova, J.; Palacka, P.; Rajec, J.; et al. Plasma cytokines correlated with disease characteristics, progression-free survival, and overall survival in testicular germ-cell tumor patients. Clin. Genitourin. Cancer 2017, 15, 411–416.e2. [Google Scholar] [CrossRef]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Ferrara, N.; Hillan, K.J.; Gerber, H.P.; Novotny, W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat. Rev. Drug Discov. 2004, 3, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Abe, R.; Fujita, Y.; Ando, S.; Inokuma, D.; Shimizu, H. Mesenchymal stem cells are recruited into wounded skin and contribute to wound repair by transdifferentiation into multiple skin cell type. J. Immunol. 2008, 180, 2581–2587. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P.; Jain, R.K. Molecular mechanisms and clinical applications of angiogenesis. Nature 2011, 473, 298–307. [Google Scholar] [CrossRef] [PubMed]
| PD | SD | PR | Total | p-Value | |
|---|---|---|---|---|---|
| Patient characteristics | |||||
| Male/Female | 11/1 | 6/6 | 8/2 | 25/9 | ns |
| Age (years) | 64 ± 10 | 71 ± 13 | 74 ± 6 | 69 ± 11 | ns |
| Etiologies | 12 | 12 | 9 | 34 | |
| HBV | 4 | 2 | 0 | 6 | ns |
| HCV | 4 | 3 | 4 | 11 | ns |
| Alcohol | 3 | 4 | 1 | 8 | ns |
| NASH | 0 | 1 | 2 | 3 | ns |
| IPH | 0 | 1 | 0 | 1 | ns |
| Unknown | 1 | 1 | 3 | 5 | ns |
| Clinical characteristics | |||||
| CTP class A/B | 10/2 | 11/1 | 10/0 | 31/3 | ns |
| ALBI grade 1/2a/2b | 6/2/4 | 2/6/4 | 3/3/4 | 11/11/12 | ns |
| Maximum tumor diameter, cm, median (range) | 4.0 (1.1–15.6) | 4.8 (1.5–13.5) | 5.0 (1.8–10.2) | 4.5 (1.1–15.6) | ns |
| Number of tumors, median (range) | 4 (2–9) | 4 (2–13) | 4 (2–10) | 4 (2–13) | ns |
| BCLC stage B/C | 2/11 | 3/9 | 2/7 | 7/27 | ns |
| MVI | 2 | 3 | 2 | 7 | ns |
| Extra-hepatic metastases | 10 | 7 | 2 | 19 | ns |
| γ-GTP (IU/L) | 191 ± 177.1 | 142.2 ± 168.1 | 160.0 ± 177.2 | 165.5 ± 175.3 | ns |
| Albumin (g/dL) | 3.8 ± 0.5 | 3.6 ± 0.3 | 3.2 ± 0.4 | 3.7 ± 0.4 | ns |
| T-Bil (mg/dL) | 0.96 ± 0.80 | 0.95 ± 0.50 | 0.83 ± 0.38 | 0.92 ± 0.62 | ns |
| Sodium (mmol/L) | 140.6 ± 1.8 | 139.8 ± 2.0 | 139.3 ± 4.4 | 140.0 ± 2.8 | ns |
| Cre (mg/dL) | 0.85 ± 0.16 | 0.78 ± 0.19 | 0.81 ± 0.16 | 0.81 ± 0.18 | ns |
| WBC (103/μL) | 5.9 ± 1.4 | 5.6 ± 2.0 | 5.8 ± 2.6 | 5.8 ± 2.0 | ns |
| Hb (g/dL) | 12.7 ± 1.9 | 12.5 ± 1.8 | 12.5 ± 1.9 | 12.6 ± 1.8 | ns |
| Plt (104/μL) | 21.1 ± 7.4 | 14.8 ± 5.9 | 15.3 ± 6.8 | 17.3 ± 7.3 | ns |
| PT-INR | 1.01 ± 0.1 | 1.06 ± 0.1 | 1.03 ± 0.1 | 1.04 ± 0.1 | ns |
| AFP (ng/mL) | 43,499.6 ± 98,693.4 | 913.7 ± 1632.0 | 1042 ± 2137.9 | 17,230.5 ± 64,448.4 | ns |
| PIVKA-II (mAU/mL) | 8693.2 ± 15,344.7 | 9664.7 ± 17,505.5 | 4937 ± 10,934.0 | 8041.8 ± 15,280.0 | ns |
| Variable | Category/Unit | High SCGF-β Level (n = 17) | Low SCGF-β Level (n = 17) | p-Value | Odds Ratio (OR) | 95% CI for OR |
|---|---|---|---|---|---|---|
| Sex | Male vs. Female | 14 (82.4%) | 11 (64.7%) | 0.4384 | 2.545 | 0.5473 to 10.66 |
| Maximum tumor diameter | cm | 6.8 ± 4.8 | 10.2 ± 2.9 | 0.4280 | - | - |
| Number of tumors | Count | 5.0 ± 2.7 | 4.7 ± 3.1 | 0.4624 | - | - |
| MVI | Present vs. Absent | 4 (23.5%) | 3 (17.6%) | >0.9999 | 1.436 | 0.3289 to 6.498 |
| Extrahepatic metastases | Present vs. Absent | 8 (47.1%) | 8 (47.1%) | >0.9999 | 1 | 0.2629 to 3.803 |
| Age | Years | 65.6 ± 10.9 | 73.0 ± 10.8 | 0.0219 | - | - |
| Child-Pugh | B vs. A | 1 (5.9%) | 2 (11.8%) | >0.9999 | 0.4688 | 0.0307 to 4.445 |
| AFP | ng/mL | 15,706 ± 58,207 | 18,755 ± 73,712 | 0.1309 | - | - |
| PIVKA-II | mAU/mL | 9197 ± 15,487 | 6887 ± 15,920 | 0.4332 | - | - |
| Variable | Coefficient (β) | Standard Error | 95% Confidence Interval | Hazard Ratio (Exp [β]) | p-Value |
|---|---|---|---|---|---|
| Sex (Male) | 0.3309 | 0.5644 | −0.7720 to 1.474 | 1.392 | 0.123 |
| Maximum tumor diameter | 0.2121 | 0.077 | 0.06030 to 0.3664 | 1.236 | 0.004 |
| Number of tumors | 0.06756 | 0.08408 | −0.1032 to 0.2313 | 1.07 | 0.431 |
| Microvascular invasion (MVI [+]) | −1.128 | 0.6639 | −2.564 to 0.1000 | 0.3237 | 0.081 |
| Extrahepatic metastases ([−]) | 0.6563 | 0.6935 | −0.6543 to 2.093 | 1.928 | 0.324 |
| Age | 0.01476 | 0.02541 | −0.03408 to 0.06639 | 1.015 | 0.535 |
| SCGF-β [High] | 1.563 | 0.5073 | 0.6020 to 2.616 | 4.774 | 0.002 |
| Child-Pugh [B] | 3.632 | 0.9318 | 1.756 to 5.516 | 37.79 | <0.001 |
| AFP | 9.5 × 10−6 | 4.37 × 10−6 | 5.67 × 10−7 to 1.819 × 10−5 | 1 | 0.047 |
| PIVKA-II | −2.62 × 10−5 | 1.74 × 10−5 | −6.42 × 10−5 to 5.32 × 10−6 | 1 | 0.101 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kimura, M.; Nishikawa, K.; Imamura, J.; Kimura, K. Stem Cell Growth Factor-β as a Predictive Biomarker of Response to Chemotherapy and Prognosis in Patients with Advanced-Stage Hepatocellular Carcinoma: A Retrospective Study. Cancers 2024, 16, 320. https://doi.org/10.3390/cancers16020320
Kimura M, Nishikawa K, Imamura J, Kimura K. Stem Cell Growth Factor-β as a Predictive Biomarker of Response to Chemotherapy and Prognosis in Patients with Advanced-Stage Hepatocellular Carcinoma: A Retrospective Study. Cancers. 2024; 16(2):320. https://doi.org/10.3390/cancers16020320
Chicago/Turabian StyleKimura, Masamichi, Koji Nishikawa, Jun Imamura, and Kiminori Kimura. 2024. "Stem Cell Growth Factor-β as a Predictive Biomarker of Response to Chemotherapy and Prognosis in Patients with Advanced-Stage Hepatocellular Carcinoma: A Retrospective Study" Cancers 16, no. 2: 320. https://doi.org/10.3390/cancers16020320
APA StyleKimura, M., Nishikawa, K., Imamura, J., & Kimura, K. (2024). Stem Cell Growth Factor-β as a Predictive Biomarker of Response to Chemotherapy and Prognosis in Patients with Advanced-Stage Hepatocellular Carcinoma: A Retrospective Study. Cancers, 16(2), 320. https://doi.org/10.3390/cancers16020320






