Survival in Elderly Ovarian Cancer Remains Challenging in the Nordic Countries
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
3. Results
3.1. Case Numbers and Diagnostic Ages
3.2. Relative Survival
3.3. Age-Specific Mortality
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ledermann, J.A.; Matias-Guiu, X.; Amant, F.; Concin, N.; Davidson, B.; Fotopoulou, C.; González-Martin, A.; Gourley, C.; Leary, A.; Lorusso, D.; et al. ESGO-ESMO-ESP consensus conference recommendations on ovarian cancer: Pathology and molecular biology and early, advanced and recurrent disease. Ann. Oncol. 2024, 35, 248–266. [Google Scholar] [CrossRef] [PubMed]
- Tichanek, F.; Försti, A.; Hemminki, O.; Hemminki, A.; Hemminki, K. Survival, Incidence, and Mortality Trends in Female Cancers in the Nordic Countries. Obstet. Gynecol. Int. 2023, 2023, 6909414. [Google Scholar] [CrossRef] [PubMed]
- González-Martín, A.; Harter, P.; Leary, A.; Lorusso, D.; Miller, R.E.; Pothuri, B.; Ray-Coquard, I.; Tan, D.S.P.; Bellet, E.; Oaknin, A.; et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2023, 34, 833–848. [Google Scholar] [CrossRef] [PubMed]
- Hemminki, K.; Sundquist, K.; Sundquist, J.; Försti, A.; Hemminki, A.; Li, X. Familial Risks and Proportions Describing Population Landscape of Familial Cancer. Cancers 2021, 13, 4385. [Google Scholar] [CrossRef] [PubMed]
- Ledermann, J.A.; Raja, F.A.; Fotopoulou, C.; Gonzalez-Martin, A.; Colombo, N.; Sessa, C. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2013, 24 (Suppl. 6), vi24–vi32. [Google Scholar] [CrossRef] [PubMed]
- Hemminki, J.; Försti, A.; Hemminki, A.; Hemminki, K. Survival trends in solid cancers in the Nordic countries through 50 years. Eur. J. Cancer 2022, 175, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, M.; Sonke, G.S.; Van de Vijver, K.K.; van der Aa, M.A.; Kruitwagen, R. No improvement in long-term survival for epithelial ovarian cancer patients: A population-based study between 1989 and 2014 in the Netherlands. Eur. J. Cancer 2018, 88, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Allemani, C.; Matsuda, T.; Di Carlo, V.; Harewood, R.; Matz, M.; Nikšić, M.; Bonaventure, A.; Valkov, M.; Johnson, C.J.; Estève, J.; et al. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 2018, 391, 1023–1075. [Google Scholar] [CrossRef] [PubMed]
- Yun, B.S.; Park, E.H.; Ha, J.; Lee, J.Y.; Lee, K.H.; Lee, T.S.; Lee, K.J.; Kim, Y.J.; Jung, K.W.; Roh, J.W. Incidence and survival of gynecologic cancer including cervical, uterine, ovarian, vaginal, vulvar cancer and gestational trophoblastic neoplasia in Korea, 1999–2019: Korea Central Cancer Registry. Obstet. Gynecol. Sci. 2023, 66, 545–561. [Google Scholar] [CrossRef]
- Pukkala, E.; Engholm, G.; Højsgaard, S.L.K.; Storm, H.; Khan, S.; Lambe, M.; Pettersson, D.; Ólafsdóttir, E.; Tryggvadóttir, L.; Hakanen, T.; et al. Nordic Cancer Registries—An overview of their procedures and data comparability. Acta Oncol. 2018, 57, 440–455. [Google Scholar] [CrossRef]
- Engholm, G.; Ferlay, J.; Christensen, N.; Bray, F.; Gjerstorff, M.L.; Klint, A.; Køtlum, J.E.; Olafsdóttir, E.; Pukkala, E.; Storm, H.H. NORDCAN—A Nordic tool for cancer information, planning, quality control and research. Acta Oncol. 2010, 49, 725–736. [Google Scholar] [CrossRef]
- Larønningen, S.; Arvidsson, G.; Bray, F.; Engholm, G.; Ervik, M.; Guðmundsdóttir, E.M.; Gulbrandsen, J.; Hansen, H.L.; Hansen, H.M.; Johannesen, T.B.; et al. NORDCAN: Cancer Incidence, Mortality, Prevalence and Survival in the Nordic Countries, Version 9.3. Association of the Nordic Cancer Registries. Cancer Registry of Norway, 2023. Available online: https://nordcan.iarc.fr/en/database (accessed on 12 April 2024).
- Storm, H.H.; Klint, A.; Tryggvadóttir, L.; Gislum, M.; Engholm, G.; Bray, F.; Hakulinen, T. Trends in the survival of patients diagnosed with malignant neoplasms of lymphoid, haematopoietic, and related tissue in the Nordic countries 1964–2003 followed up to the end of 2006. Acta Oncol. 2010, 49, 694–712. [Google Scholar] [CrossRef] [PubMed]
- Engholm, G.; Gislum, M.; Bray, F.; Hakulinen, T. Trends in the survival of patients diagnosed with cancer in the Nordic countries 1964–2003 followed up to the end of 2006. Material and methods. Acta Oncol. 2010, 49, 545–560. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, F.E.; Ekman, S.; Johansson, A.L.V.; Engholm, G.; Birgisson, H.; Ólafsdóttir, E.J.; Mørch, L.S.; Johannesen, T.B.; Andersson, T.M.; Pettersson, D.; et al. Trends in cancer survival in the Nordic countries 1990–2016: The NORDCAN survival studies. Acta Oncol. 2020, 59, 1266–1274. [Google Scholar] [CrossRef] [PubMed]
- Tichanek, F.; Försti, A.; Liska, V.; Hemminki, A.; Hemminki, K. Survival in Colon, Rectal and Small Intestinal Cancers in the Nordic Countries through a Half Century. Cancers 2023, 15, 991. [Google Scholar] [CrossRef]
- Bürkner, P. An R package for Bayesian multilevel models using Stan. J. Stat. Softw. 2017, 80, 1–28. [Google Scholar] [CrossRef]
- Bürkner, P. Advanced Bayesian multilevel modeling with the R package (brms). R J. 2018, 10, 395–411. [Google Scholar] [CrossRef]
- Carpenter, B.; Gelman, A.; Hoffman, M.D.; Lee, D.; Goodrich, B.; Betancourt, M.; Brubaker, M.; Guo, J.; Li, P.; Riddell, A. Stan: A probabilistic programming language. J. Stat. Softw. 2017, 76, 1–32. [Google Scholar] [CrossRef]
- Leandersson, P.; Hogberg, T.; Dickman, P.W.; Malander, S.; Borgfeldt, C. Incidence and survival of epithelial ovarian, fallopian tube, peritoneal, and undesignated abdominal/pelvic cancers in Sweden 1960–2014: A population-based cohort study. BMC Cancer 2021, 21, 465. [Google Scholar] [CrossRef]
- Berek, J.S.; Hacker, N.F.; Lagasse, L.D. Recent progress in the treatment of epithelial ovarian malignancy. West. J. Med. 1982, 137, 273–277. [Google Scholar]
- Fader, A.N.; Rose, P.G. Role of surgery in ovarian carcinoma. J. Clin. Oncol. 2007, 25, 2873–2883. [Google Scholar] [CrossRef] [PubMed]
- Fortner, R.T.; Trewin-Nybråten, C.B.; Paulsen, T.; Langseth, H. Characterization of ovarian cancer survival by histotype and stage: A nationwide study in Norway. Int. J. Cancer 2023, 153, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Mallen, A.; Todd, S.; Robertson, S.E.; Kim, J.; Sehovic, M.; Wenham, R.M.; Extermann, M.; Chon, H.S. Impact of age, comorbidity, and treatment characteristics on survival in older women with advanced high grade epithelial ovarian cancer. Gynecol. Oncol. 2021, 161, 693–699. [Google Scholar] [CrossRef]
- Akeson, M.; Zetterqvist, B.M.; Holmberg, E.; Horvath, G. Improved survival with clinical guidelines? Evaluation of a quality register linked to clinical guidelines for ovarian cancer in the western health care region in Sweden between 1 September 1993 and 1 June 1998. Acta Obstet. Gynecol. Scand. 2005, 84, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
- Aronson, S.L.; Lopez-Yurda, M.; Koole, S.N.; Schagen van Leeuwen, J.H.; Schreuder, H.W.R.; Hermans, R.H.M.; de Hingh, I.H.J.T.; van Gent, M.D.J.M.; Arts, H.J.G.; van Ham, M.A.P.C.; et al. Cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy in patients with advanced ovarian cancer (OVHIPEC-1): Final survival analysis of a randomised, controlled, phase 3 trial. Lancet Oncol. 2023, 24, 1109–1118. [Google Scholar] [CrossRef]

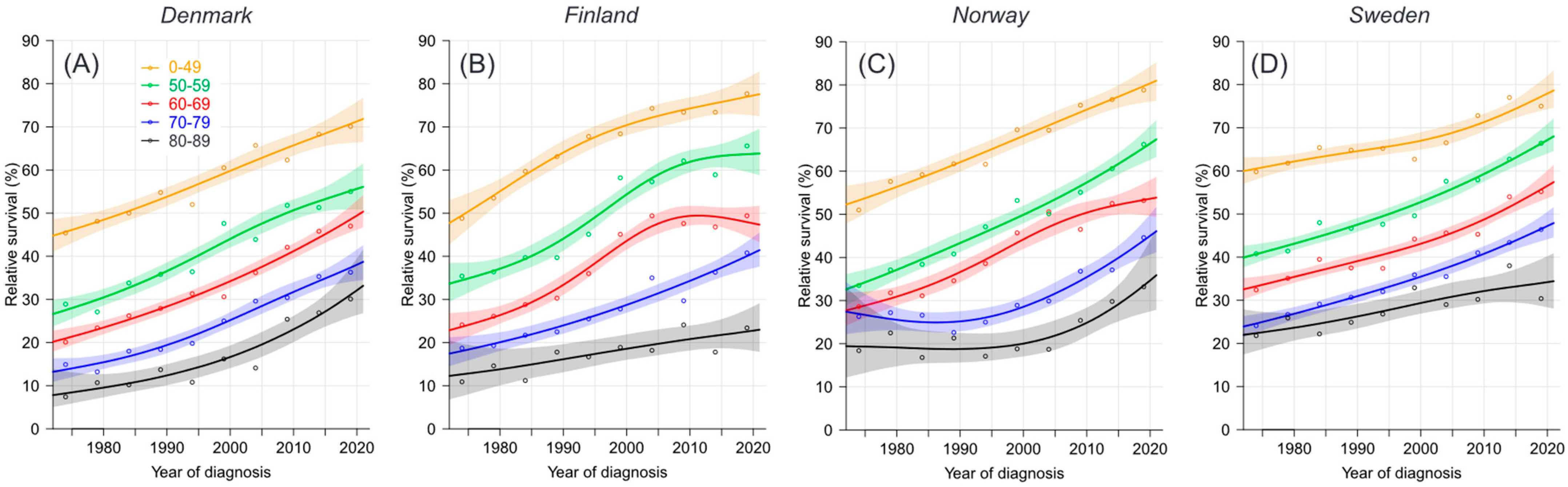
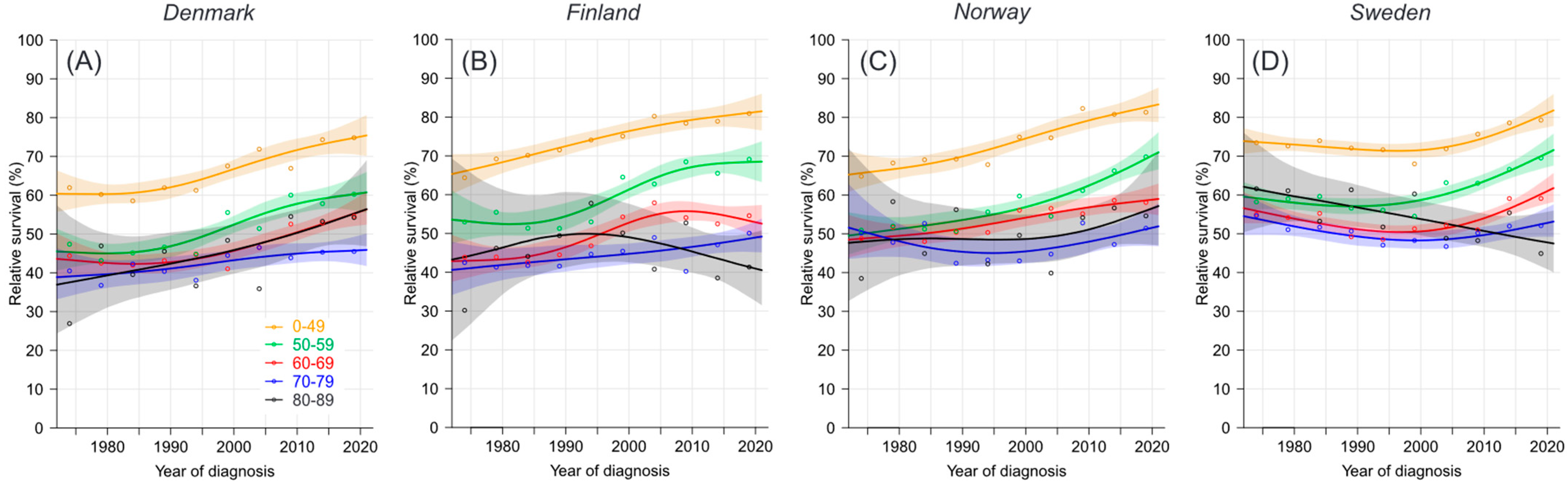
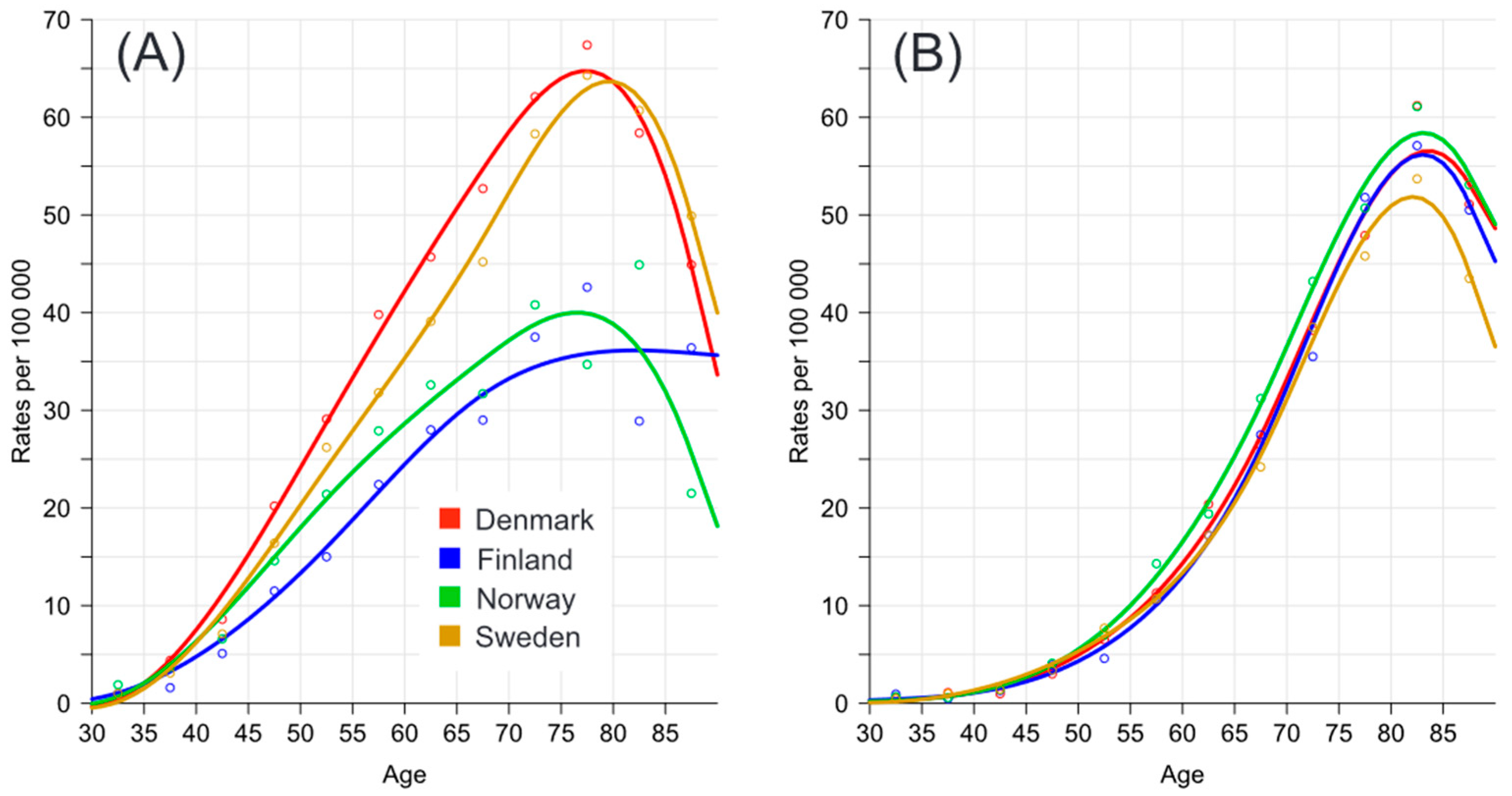
| Denmark | ||||||||||
| Age group | 1972–1976 | 1977–1981 | 1982–1986 | 1987–1991 | 1992–1996 | 1997–2001 | 2002–2006 | 2007–2011 | 2012–2016 | 2017–2021 |
| 0–49 | 61.9 [57.1–67.2] | 60.2 [55.3–65.5] | 58.5 [53.8–63.7] | 61.9 [57.8–66.4] | 61.2 [56.8–66.1] | 67.5 [63.3–72.1] | 71.9 [67.2–76.9] | 66.9 [61.9–72.4] | 74.3 [69.4–79.6] | 74.8 [69.8–80.2] * |
| 50–59 | 47.4 [42.5–52.8] | 43.2 [38.6–48.2] | 45.1 [40.6–50.2] | 46.6 [42.1–51.6] | 44.8 [40.7–49.4] | 55.5 [51.7–59.7] | 51.4 [47.2–55.9] | 60.0 [55.6–64.8] | 57.8 [53.1–63.0] | 60.2 [55.4–65.5]* |
| 60–69 | 44.4 [38.8–50.7] | 42.5 [37.6–48.0] | 41.9 [37.6–46.7] | 43.1 [38.6–48.1] | 44.7 [40.5–49.2] | 41.0 [36.9–45.6] | 46.5 [42.4–50.9] | 52.6 [48.7–56.7] | 52.8 [48.9–57.0] | 54.6 [50.7–58.8] |
| 70–79 | 40.5 [33.0–49.7] | 36.8 [30.3–44.6] | 42.3 [36.1–49.5] | 40.4 [34.3–47.5] | 38.1 [32.7–44.3] | 44.5 [39.2–50.5] | 46.5 [41.3–52.4] | 43.9 [39.1–49.2] | 45.3 [41.1–49.9] | 45.5 [41.4–50.0] * |
| 80–89 | 26.9 [13.0–55.7] | 46.9 [30.4–72.3] | 39.5 [25.6–61.1] | 45.5 [32.0–64.7] | 36.6 [24.1–55.7] | 48.4 [34.9–67.0] | 35.9 [25.4–50.7] | 54.5 [44.0–67.6] | 53.3 [43.1–65.8] | 54.2 [44.1–66.7] |
| Age stand | 45.9 [42.7–49.3] | 45.1 [42.3–48.1] | 45.6 [43.0–48.5] | 46.9 [44.3–49.5] | 45.4 [42.9–47.9] | 49.9 [47.5–52.4] | 50.6 [48.1–53.1] | 54.1 [51.7–56.6] | 54.7 [52.4–57.1] | 55.8 [53.5–58.3]* |
| Finland | ||||||||||
| Age group | 1972–1976 | 1977–1981 | 1982–1986 | 1987–1991 | 1992–1996 | 1997–2001 | 2002–2006 | 2007–2011 | 2012–2016 | 2017–2021 |
| 0–49 | 64.4 [58.6–70.8] | 69.2 [63.7–75.1] | 70.2 [65.2–75.5] | 71.5 [67.0–76.4] | 74.1 [70.1–78.4] | 75.1 [70.8–79.6] | 80.2 [76.1–84.6] | 78.4 [73.7–83.5] | 78.9 [74.1–84.0] | 80.9 [76.2–85.9] * |
| 50–59 | 53.0 [47.0–59.7] | 55.5 [49.6–62.0] | 51.4 [46.1–57.2] | 51.3 [46.0–57.1] | 53.0 [48.1–58.4] | 64.5 [60.3–69.0] | 62.8 [58.8–67.0] | 68.5 [64.2–73.0] | 65.5 [60.9–70.5] | 69.1 [64.4–74.2] * |
| 60–69 | 43.9 [38.0–50.7] | 43.9 [38.4–50.2] | 42.6 [37.4–48.6] | 44.5 [39.7–49.8] | 46.8 [42.5–51.6] | 54.3 [49.8–59.1] | 57.9 [53.8–62.3] | 54.1 [50.3–58.2] | 52.5 [48.6–56.6] | 54.6 [50.7–58.7] |
| 70–79 | 42.5 [34.0–53.2] | 41.3 [33.9–50.3] | 41.7 [34.8–50.0] | 41.6 [35.3–49.0] | 44.7 [38.4–52.0] | 45.4 [39.7–52.0] | 49.0 [43.7–54.8] | 40.2 [35.4–45.7] | 47.1 [42.5–52.1] | 50.1 [45.6–55.0] |
| 80–89 | 30.2 [11.3–80.6] | 46.2 [29.1–73.2] | 44.1 [26.6–73.1] | 49.4 [33.8–72.4] | 57.8 [41.0–81.5] | 50.1 [36.3–69.2] | 40.8 [30.5–54.6] | 52.7 [42.3–65.7] | 38.5 [29.8–49.8] | 41.3 [32.3–52.9] |
| Age stand | 47.8 [43.6–52.3] | 50.2 [46.8–53.8] | 48.9 [45.9–52.1] | 50.2 [47.3–53.2] | 52.8 [50.0–55.7] | 57.1 [54.6–59.8] | 58.2 [56.0–60.6] | 56.4 [54.3–58.6] | 55.9 [53.7–58.1] | 58.3 [56.1–60.6] * |
| Norway | ||||||||||
| Age group | 1972–1976 | 1977–1981 | 1982–1986 | 1987–1991 | 1992–1996 | 1997–2001 | 2002–2006 | 2007–2011 | 2012–2016 | 2017–2021 |
| 0–49 | 64.8 [59.3–70.8] | 68.2 [63.3–73.6] | 69.1 [64.3–74.2] | 69.2 [64.8–74.0] | 67.8 [63.4–72.6] | 74.9 [70.8–79.3] | 74.7 [70.3–79.4] | 82.3 [78.0–86.8] | 80.8 [76.4–85.4] | 81.3 [77.0–85.9] * |
| 50–59 | 50.9 [45.2–57.4] | 52.0 [46.6–57.9] | 51.2 [46.2–56.8] | 50.6 [45.4–56.5] | 55.6 [50.8–60.9] | 59.7 [55.3–64.4] | 54.5 [50.3–59.1] | 61.2 [56.8–65.8] | 66.2 [62.0–70.8] | 69.8 [65.5–74.5] * |
| 60–69 | 48.8 [42.8–55.7] | 51.7 [46.2–57.8] | 48.0 [43.1–53.5] | 50.4 [45.5–55.8] | 50.3 [45.3–55.9] | 56.1 [51.2–61.5] | 56.5 [52.0–61.5] | 55.2 [51.1–59.5] | 58.6 [54.6–62.8] | 58.1 [54.1–62.4] |
| 70–79 | 50.2 [41.5–60.7] | 47.8 [40.3–56.7] | 52.7 [45.5–60.9] | 42.4 [36.2–49.6] | 43.3 [37.5–49.9] | 43.0 [38.0–48.7] | 44.8 [39.2–51.1] | 52.8 [47.0–59.3] | 47.3 [42.0–53.1] | 51.4 [46.5–56.9] |
| 80–89 | 38.5 [21.7–68.2] | 58.3 [38.6–88.0] | 44.9 [29.8–67.6] | 56.2 [42.8–73.8] | 42.2 [30.1–59.2] | 49.6 [36.9–66.7] | 39.9 [31.0–51.3] | 54.2 [43.6–67.3] | 56.7 [46.1–69.7] | 54.6 [43.9–68.0] |
| Age stand | 50.9 [46.9–55.1] | 54.1 [50.4–58.0] | 53.1 [50.0–56.5] | 52.3 [49.4–55.3] | 51.7 [49.2–54.3] | 55.6 [53.1–58.3] | 54.3 [51.9–56.9] | 59.5 [57.1–62.0] | 59.8 [57.4–62.3] | 61.1 [58.7–63.7] * |
| Sweden | ||||||||||
| Age group | 1972–1976 | 1977–1981 | 1982–1986 | 1987–1991 | 1992–1996 | 1997–2001 | 2002–2006 | 2007–2011 | 2012–2016 | 2017–2021 |
| 0–49 | 73.5 [70.4–76.7] | 72.6 [69.5–75.9] | 74.0 [70.8–77.3] | 72.1 [68.8–75.6] | 71.6 [68.2–75.2] | 68.0 [64.4–71.8] | 71.9 [68.3–75.7] | 75.7 [71.9–79.7] | 78.6 [74.9–82.4] | 79.3 [75.6–83.1] |
| 50–59 | 58.2 [55.0–61.6] | 59.0 [55.6–62.6] | 59.6 [56.2–63.2] | 56.6 [53.0–60.5] | 56.1 [52.6–59.8] | 54.5 [51.3–58.0] | 63.2 [59.8–66.7] | 63.0 [59.3–66.9] | 66.6 [62.9–70.4] | 69.5 [65.8–73.4] * |
| 60–69 | 55.0 [51.3–59.0] | 54.2 [50.7–57.8] | 55.2 [52.2–58.4] | 49.2 [46.0–52.7] | 48.5 [45.1–52.2] | 51.3 [47.8–55.0] | 52.2 [48.9–55.6] | 51.0 [47.9–54.3] | 59.1 [56.0–62.3] | 59.1 [55.8–62.6] |
| 70–79 | 54.6 [49.4–60.5] | 51.1 [46.3–56.3] | 51.7 [47.3–56.5] | 50.7 [46.5–55.2] | 47.1 [43.3–51.1] | 48.3 [44.4–52.4] | 46.7 [42.4–51.4] | 50.2 [46.2–54.5] | 52.0 [48.1–56.2] | 52.1 [48.4–56.0] |
| 80–89 | 61.6 [47.8–79.3] | 61.1 [48.5–76.9] | 53.2 [42.1–67.3] | 61.3 [50.5–74.5] | 51.7 [42.1–63.6] | 60.3 [50.5–71.8] | 48.8 [41.1–57.9] | 48.2 [40.4–57.6] | 55.4 [46.6–65.9] | 44.9 [36.8–54.8] |
| Age stand | 59.4 [57.0–61.9] | 58.2 [55.8–60.6] | 58.2 [56.1–60.3] | 55.7 [53.7–57.8] | 53.3 [51.4–55.3] | 54.3 [52.4–56.3] | 55.1 [53.2–57.1] | 56.2 [54.2–58.2] | 60.8 [58.7–62.9] | 59.9 [58.0–61.8] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hemminki, K.; Zitricky, F.; Försti, A.; Hemminki, A. Survival in Elderly Ovarian Cancer Remains Challenging in the Nordic Countries. Cancers 2024, 16, 2198. https://doi.org/10.3390/cancers16122198
Hemminki K, Zitricky F, Försti A, Hemminki A. Survival in Elderly Ovarian Cancer Remains Challenging in the Nordic Countries. Cancers. 2024; 16(12):2198. https://doi.org/10.3390/cancers16122198
Chicago/Turabian StyleHemminki, Kari, Frantisek Zitricky, Asta Försti, and Akseli Hemminki. 2024. "Survival in Elderly Ovarian Cancer Remains Challenging in the Nordic Countries" Cancers 16, no. 12: 2198. https://doi.org/10.3390/cancers16122198
APA StyleHemminki, K., Zitricky, F., Försti, A., & Hemminki, A. (2024). Survival in Elderly Ovarian Cancer Remains Challenging in the Nordic Countries. Cancers, 16(12), 2198. https://doi.org/10.3390/cancers16122198








