Gender Differences in Sinonasal Cancer Incidence: Data from the Italian Registry
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
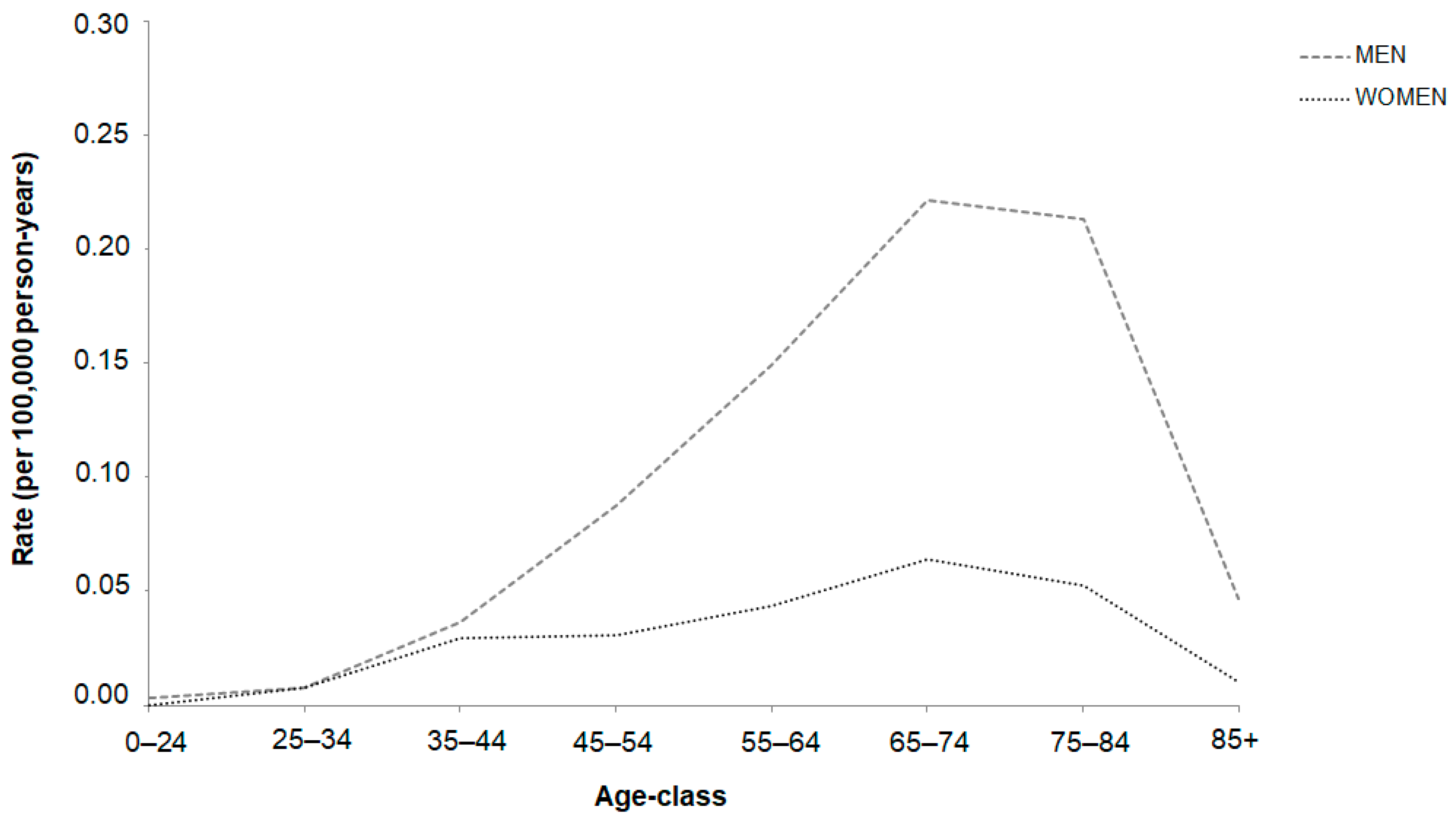
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Agency for Research on Cancer (IARC). List of Classifications by Cancer Sites with Sufficient or Limited Evidence in Humans; IARC Monographs; IARC: Lyon, France, 2023; Volume 1–133, Available online: https://monographs.iarc.who.int/agents-classified-by-the-iarc/ (accessed on 3 May 2024).
- Rushton, L.; Hutchings, S.J.; Fortunato, L.; Young, C.; Evans, G.S.; Brown, T.; Bevan, R.; Slack, R.; Holmes, P.; Bagga, S.; et al. Occupational cancer burden in Great Britain. Br. J. Cancer 2012, 107 (Suppl. 1), S3–S7. [Google Scholar] [CrossRef] [PubMed]
- Collatuzzo, G.; Turati, F.; Malvezzi, M.; Negri, E.; La Vecchia, C.; Boffetta, P. Attributable Fraction of Cancer Related to Occupational Exposure in Italy. Cancers 2023, 15, 2234. [Google Scholar] [CrossRef] [PubMed]
- Bishop, J.A.; Guo, T.W.; Smith, D.F.; Wang, H.; Ogawa, T.; Pai, S.I.; Westra, W.H. Human papillomavirus-related carcinomas of the sinonasal tract. Am. J. Surg. Pathol. 2013, 37, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.S., Jr. Sinonasal Squamous Cell Carcinoma: A Review with Emphasis on Emerging Histologic Subtypes and the Role of Human Papillomavirus. Head Neck Pathol. 2016, 10, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Kılıç, S.; Kılıç, S.S.; Kim, E.S.; Baredes, S.; Mahmoud, O.; Gray, S.T.; Eloy, J.A. Significance of human papillomavirus positivity in sinonasal squamous cell carcinoma. Int. Forum Allergy Rhinol. 2017, 7, 980–989. [Google Scholar] [CrossRef]
- Dutta, R.; Dubal, P.M.; Svider, P.F.; Liu, J.K.; Baredes, S.; Eloy, J.A. Sinonasal malignancies: A population-based analysis of site-specific incidence and survival. Laryngoscope 2015, 125, 2491–2497. [Google Scholar] [CrossRef]
- Turner, J.H.; Reh, D.D. Incidence and survival in patients with sinonasal cancer: A historical analysis of population-based data. Head Neck 2012, 34, 877–885. [Google Scholar] [CrossRef]
- Gore, M.R. Survival in sinonasal and middle ear malignancies: A population-based study using the SEER 1973-2015 database. BMC Ear Nose Throat Disord. 2018, 19, 13. [Google Scholar] [CrossRef]
- Low, C.M.; Balakrishnan, K.; Smith, B.M.; Stokken, J.K.; O’Brien, E.K.; Van Gompel, J.J.; Rowan, N.R.; Choby, G. Sinonasal adenocarcinoma: Population-based analysis of demographic and socioeconomic disparities. Head Neck 2021, 43, 2946–2953. [Google Scholar] [CrossRef]
- Consonni, D.; Stella, S.; Denaro, N.; Binazzi, A.; Dallari, B.; Rugarli, S.; Borello, F.; Coviello, E.; Mensi, C. Survival of Patients with Sinonasal Cancers in a Population-Based Registry, Lombardy, Italy, 2008–2023. Cancers 2024, 16, 896. [Google Scholar] [CrossRef]
- Llorente, J.L.; López, F.; Suárez, C.; Hermsen, M.A. Sinonasal carcinoma: Clinical, pathological, genetic and therapeutic advances. Nature reviews. Clin. Oncol. 2014, 11, 460–472. [Google Scholar] [CrossRef] [PubMed]
- Gatta, G.; Capocaccia, R.; Botta, L. Descriptive epidemiology of the head and neck cancers in old patients. Front. Oncol. 2023, 13, 1102236. [Google Scholar] [CrossRef]
- Unsal, A.A.; Kılıç, S.; Dubal, P.M.; Baredes, S.; Eloy, J.A.; EUROCARE-5 Working Group. A population-based comparison of European and North American sinonasal cancer survival. Auris Nasus Larynx 2018, 45, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Park, J.O.; Nam, I.C.; Kim, C.S.; Park, S.J.; Lee, D.H.; Kim, H.B.; Han, K.D.; Joo, Y.H. Sex Differences in the Prevalence of Head and Neck Cancers: A 10-Year Follow-Up Study of 10 Million Healthy People. Cancers 2022, 14, 2521. [Google Scholar] [CrossRef] [PubMed]
- El-Naggar, A.K.; Chan, J.K.C.; Grandis, J.R.; Takata, T.; Slootweg, P.J. WHO Classification of Head and Neck Tumours. In WHO/IARC Classification of Tumours, 4th ed.; International Agency for Research on Cancer (IARC): Lyon, France, 2017; Volume 9, pp. 11–60. [Google Scholar]
- Italian Institute of Statistics (ISTAT). Classification ATECO (Classification of Economic Activity). Available online: https://www.istat.it/it/archivio/17888 (accessed on 3 May 2024).
- Binazzi, A.; Miligi, L.; Giovannetti, L.; Piro, S.; Franchi, A.; Mensi, C.; Calisti, R.; Galli, P.; Camagni, A.; Romeo, E.; et al. ReNaTuNS. Sorveglianza Epidemiologica dei Tumori Naso-Sinusali. Manuale Operativo; INAIL Press: Milan, Italy, 2020; Available online: https://www.inail.it/cs/internet/comunicazione/pubblicazioni/catalogo-generale/pubbl-renatuns-sorv-epid-tumorinaso-sinusali-manuale.html (accessed on 3 May 2024).
- Vera, R.; Juan-Vidal, O.; Safont-Aguilera, M.J.; de la Peña, F.A.; Del Alba, A.G. Sex differences in the diagnosis, treatment and prognosis of cancer: The rationale for an individualised approach. Clin. Transl. Oncol. 2023, 25, 2069–2076. [Google Scholar] [CrossRef] [PubMed]
- Global Burden of Disease Cancer Collaboration; Fitzmaurice, C.; Akinyemiju, T.F.; Al Lami, F.H.; Alam, T.; Alizadeh-Navaei, R.; Allen, C.; Alsharif, U.; Alvis-Guzman, N.; Amini, E.; et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived with Disability, and Disability-Adjusted Life-Years for 29 Cancer Groups, 1990 to 2016: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2018, 4, 1553–1568. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.D.; Chiu, T.J.; Chen, W.C.; Wang, C.S. Sex Differences in Otolaryngology: Focus on the Emerging Role of Estrogens in Inflammatory and Pro-Resolving Responses. Int. J. Mol. Sci. 2021, 22, 8768. [Google Scholar] [CrossRef]
- Zhu, Y.; Shao, X.; Wang, X.; Liu, L.; Liang, H. Sex disparities in cancer. Cancer Lett. 2019, 466, 35–38. [Google Scholar] [CrossRef]
- AIRTUM (Italian Association of Cancer Registries). Epithelial tumors of head and neck. Italian Cancer Figures—Report 2015: The Burden of Rare Cancers in Italy. Epidemiol. Prev. 2016, 40 (Suppl. 2), 1–120. Available online: http://www.registri-tumori.it/PDF/AIRTUM2016/TUMORIRARI/AIRTUM_RARI_S001_headandneck.pdf (accessed on 3 May 2024).
- Yang, T.H.; Xirasagar, S.; Cheng, Y.F.; Chen, C.S.; Chang, W.P.; Lin, H.C. Trends in the incidence of head and neck cancer: A nationwide population-based study. Oral Oncol. 2023, 140, 106391. [Google Scholar] [CrossRef]
- Sjöstedt, S.; Jensen, D.H.; Jakobsen, K.K.; Grønhøj, C.; Geneser, C.; Karnov, K.; Specht, L.; Agander, T.K.; von Buchwald, C. Incidence and survival in sinonasal carcinoma: A Danish population-based, nationwide study from 1980 to 2014. Acta Oncol. 2018, 57, 1152–1158. [Google Scholar] [CrossRef] [PubMed]
- Gatta, G.; Capocaccia, R.; Botta, L.; Mallone, S.; De Angelis, R.; Ardanaz, E.; Comber, H.; Dimitrova, N.; Leinonen, M.K.; Siesling, S.; et al. Burden and centralised treatment in Europe of rare tumours: Results of RARECAREnet—A population-based study. Lancet Oncol. 2017, 18, 1022–1039. [Google Scholar] [CrossRef] [PubMed]
- Elliot, A.; Jangard, M.; Marklund, L.; Håkansson, N.; Dickman, P.; Hammarstedt-Nordenvall, L.; Stjärne, P. Sinonasal malignancies in Sweden 1960-2010; a nationwide study of the Swedish population. Rhinology 2015, 53, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Youlden, D.R.; Cramb, S.M.; Peters, S.; Porceddu, S.V.; Møller, H.; Fritschi, L.; Baade, P.D. International comparisons of the incidence and mortality of sinonasal cancer. Cancer Epidemiol. 2013, 37, 770–779. [Google Scholar] [CrossRef]
- Kuijpens, J.H.; Louwman, M.W.; Peters, R.; Janssens, G.O.; Burdorf, A.L.; Coebergh, J.W. Trends in sinonasal cancer in The Netherlands: More squamous cell cancer, less adenocarcinoma. Eur. J. Cancer 2012, 48, 2369–2374. [Google Scholar] [CrossRef] [PubMed]
- Bracigliano, A.; Tatangelo, F.; Perri, F.; Di Lorenzo, G.; Tafuto, R.; Ottaiano, A.; Clemente, O.; Barretta, M.L.; Losito, N.S.; Santorsola, M.; et al. Malignant Sinonasal Tumors: Update on Histological and Clinical Management. Curr. Oncol. 2021, 28, 2420–2438. [Google Scholar] [CrossRef] [PubMed]
- d’Errico, A.; Pasian, S.; Baratti, A.; Zanelli, R.; Alfonzo, S.; Gilardi, L.; Beatrice, F.; Bena, A.; Costa, G. A case-control study on occupational risk factors for sino-nasal cancer. Occup. Environ. Med. 2009, 66, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Elgart, K.; Faden, D.L. Sinonasal Squamous Cell Carcinoma: Etiology, Pathogenesis, and the Role of Human Papilloma Virus. Curr. Otorhinolaryngol. Rep. 2020, 8, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Chang Sing Pang, K.J.W.; Mur, T.; Collins, L.; Rao, S.R.; Faden, D.L. Human Papillomavirus in Sinonasal Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis. Cancers 2020, 13, 45. [Google Scholar] [CrossRef]
- Choussy, O.; Ferron, C.; Védrine, P.O.; Toussaint, B.; Liétin, B.; Marandas, P.; Babin, E.; De Raucourt, D.; Reyt, E.; Cosmidis, A.; et al. Adenocarcinoma of Ethmoid: A GETTEC retrospective multicenter study of 418 cases. Laryngoscope 2008, 118, 437–443. [Google Scholar] [CrossRef]
- ’t Mannetje, A.; Kogevinas, M.; Luce, D.; Demers, P.A.; Bégin, D.; Bolm-Audorff, U.; Comba, P.; Hardell, L.; Hayes, R.B.; Leclerc, A.; et al. Sinonasal cancer, occupation, and tobacco smoking in European women and men. Am. J. Ind. Med. 1999, 36, 101–107. [Google Scholar] [CrossRef]
- Capper, J.W.R. Fifty years of woodworkers’ nasal adenocarcinoma in High Wycombe. J. Laryngol. Otol. 2022, 136, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Gallet, P.; Nguyen, D.T.; Russel, A.; Jankowski, R.; Vigouroux, C.; Rumeau, C. Intestinal and non-intestinal nasal cavity adenocarcinoma: Impact of wood dust exposure. Eur. Ann. Otorhinolaryngol. Head. Neck Dis. 2018, 135, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Bonzini, M.; Battaglia, P.; Parassoni, D.; Casa, M.; Facchinetti, N.; Turri-Zanoni, M.; Borchini, R.; Castelnuovo, P.; Ferrario, M.M. Prevalence of occupational hazards in patients with different types of epithelial sinonasal cancers. Rhinology 2013, 51, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Blot, W.J.; Chow, W.H.; McLaughlin, J.K. Wood dust and nasal cancer risk. A review of the evidence from North America. J. Occup. Environ. Med. 1997, 39, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Cantu, G.; Solero, C.L.; Mariani, L.; Lo Vullo, S.; Riccio, S.; Colombo, S.; Pompilio, M.; Perrone, F.; Formillo, P.; Quattrone, P. Intestinal type adenocarcinoma of the ethmoid sinus in wood and leather workers: A retrospective study of 153 cases. Head Neck 2011, 33, 535–542. [Google Scholar] [CrossRef] [PubMed]
- d‘Errico, A.; Zajacova, J.; Cacciatore, A.; Alfonzo, S.; Beatrice, F.; Ricceri, F.; Valente, G. Exposure to occupational hazards and risk of sinonasal epithelial cancer: Results from an extended Italian case-control study. Occup. Environ. Med. 2020, 28, 106738. [Google Scholar] [CrossRef] [PubMed]
- Greiser, E.M.; Greiser, K.H.; Ahrens, W.; Hagen, R.; Lazszig, R.; Maier, H.; Schick, B.; Zenner, H.P. Risk factors for nasal malignancies in German men: The South-German Nasal cancer study. BMC Cancer 2012, 12, 506. [Google Scholar] [CrossRef]
- Russo, A.L.; Adams, J.A.; Weyman, E.A.; Busse, P.M.; Goldberg, S.I.; Varvares, M.; Deschler, D.D.; Lin, D.T.; Delaney, T.F.; Chan, A.W. Long-Term Outcomes After Proton Beam Therapy for Sinonasal Squamous Cell Carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 368–376. [Google Scholar] [CrossRef]
- Cogliano, V.J.; Baan, R.; Straif, K.; Grosse, Y.; Lauby-Secretan, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Freeman, C.; et al. Preventable exposures associated with human cancers. J. Natl. Cancer Inst. 2011, 103, 1827–1839. [Google Scholar] [CrossRef]
- Binazzi, A.; Corfiati, M.; Di Marzio, D.; Cacciatore, A.M.; Zajacovà, J.; Mensi, C.; Galli, P.; Miligi, L.; Calisti, R.; Romeo, E.; et al. Sinonasal cancer in the Italian national surveillance system: Epidemiology, occupation, and public health implications. Am. J. Ind. Med. 2018, 61, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Binazzi, A.; Ferrante, P.; Marinaccio, A. Occupational exposure and sinonasal cancer: A systematic review and meta-analysis. BMC Cancer 2015, 15, 49. [Google Scholar] [CrossRef] [PubMed]
- Massari, S.; Malpassuti, V.C.; Binazzi, A.; Paris, L.; Gariazzo, C.; Marinaccio, A. Occupational Mortality Matrix: A Tool for Epidemiological Assessment of Work-Related Risk Based on Current Data Sources. Int. J. Environ. Res. Public Health 2022, 19, 5652. [Google Scholar] [CrossRef] [PubMed]


| Variable | Men N (%) | Women N (%) |
|---|---|---|
| Sex | 2073 (73) | 778 (27) |
| Age, years (mean ± SD) | 67 ± 12.4 | 66.0 ± 15.2 |
| Age class | ||
| 0–44 | 104 (5) | 82 (11) |
| 45–54 | 230 (11) | 92 (12) |
| 0–54 | 334 (16) | 174 (22) |
| 55–64 | 460 (22) | 142 (18) |
| 65–74 | 639 (31) | 195 (25) |
| 75+ | 640 (31) | 267 (34) |
| 55+ | 1739 (84) | 604 (78) |
| Topography (ICD-X codes) | ||
| Nasal cavities (C30.0) | 1204 (50) | 452 (50) |
| Maxillary sinus (C31.0) | 420 (17) | 220 (24) |
| Ethmoidal sinus (C31.1) | 583 (24) | 146 (16) |
| Frontal sinus (C31.2) | 48 (2) | 14 (2) |
| Sphenoidal sinus (C31.3) | 113 (5) | 51 (6) |
| Overlapping lesion of accessory sinuses (C31.8) | 19 (1) | 8 (1) |
| Accessory sinus, unspecified (31.9) | 32 (1) | 10 (1) |
| Histology (a) | ||
| Adenocarcinomas | ||
| ITAC | 678 (33) | 74 (10) |
| Non-ITAC | 93 (5) | 42 (6) |
| AD NOS | 160 (8) | 26 (3) |
| SGC | 112 (6) | 108 (14) |
| Squamous cell carcinomas | ||
| SCC | 730 (36) | 376 (50) |
| Neuroendocrine carcinomas | ||
| NEC | 43 (2) | 21 (3) |
| Other epithelial neoplasms | ||
| SNC NOS | 108 (5) | 49 (6) |
| SNUC | 114 (6) | 57 (8) |
| Malignant Neoplasm | 16 (1) | 9 (1) |
| Variable | Incidence Rate | IRR (95% CI) | |
|---|---|---|---|
| Men | Women | ||
| Age-class | |||
| 0–24 | - | - | - |
| 25–34 | 0.01 | 0.01 | 0.98 (0.28–3.37) |
| 35–44 | 0.04 | 0.03 | 1.22 (0.70–2.12) |
| 45–54 | 0.09 | 0.03 | 2.83 (1.82–4.39) |
| 55–64 | 0.15 | 0.04 | 3.55 (2.44–5.19) |
| 65–74 | 0.22 | 0.06 | 3.42 (2.54–4.62) |
| 75+ | 0.26 | 0.06 | 4.05 (3.11–5.27) |
| Topography (most represented) | |||
| Nasal cavities (C30.0) | 0.51 | 0.15 | 3.41 (2.82–4.13) |
| Maxillary sinus (C31.0) | 0.16 | 0.06 | 2.47 (1.82–3.35) |
| Ethmoid sinus (C31.1) | 0.18 | 0.04 | 4.01 (2.86–5.63) |
| Histology (main groups) | |||
| Adenocarcinoma | 0.35 | 0.07 | 5.00 (3.83–6.53) |
| Squamous cell carcinoma | 0.30 | 0.13 | 2.31 (1.85–2.88) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Binazzi, A.; di Marzio, D.; Mensi, C.; Consonni, D.; Miligi, L.; Piro, S.; Zajacovà, J.; Sorasio, D.; Galli, P.; Camagni, A.; et al. Gender Differences in Sinonasal Cancer Incidence: Data from the Italian Registry. Cancers 2024, 16, 2053. https://doi.org/10.3390/cancers16112053
Binazzi A, di Marzio D, Mensi C, Consonni D, Miligi L, Piro S, Zajacovà J, Sorasio D, Galli P, Camagni A, et al. Gender Differences in Sinonasal Cancer Incidence: Data from the Italian Registry. Cancers. 2024; 16(11):2053. https://doi.org/10.3390/cancers16112053
Chicago/Turabian StyleBinazzi, Alessandra, Davide di Marzio, Carolina Mensi, Dario Consonni, Lucia Miligi, Sara Piro, Jana Zajacovà, Denise Sorasio, Paolo Galli, Angela Camagni, and et al. 2024. "Gender Differences in Sinonasal Cancer Incidence: Data from the Italian Registry" Cancers 16, no. 11: 2053. https://doi.org/10.3390/cancers16112053
APA StyleBinazzi, A., di Marzio, D., Mensi, C., Consonni, D., Miligi, L., Piro, S., Zajacovà, J., Sorasio, D., Galli, P., Camagni, A., Calisti, R., Massacesi, S., Cozzi, I., Balestri, A., Murano, S., Fedeli, U., Comiati, V., Eccher, S., Lattanzio, S., ... ReNaTuNS Working Group. (2024). Gender Differences in Sinonasal Cancer Incidence: Data from the Italian Registry. Cancers, 16(11), 2053. https://doi.org/10.3390/cancers16112053









