The Prognostic Value and Clinical Utility of the 40-Gene Expression Profile (40-GEP) Test in Cutaneous Squamous Cell Carcinoma: Systematic Review and Meta-Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Extraction
2.4. Quality Assessment and Risk of Bias
2.5. Data Analysis
3. Results
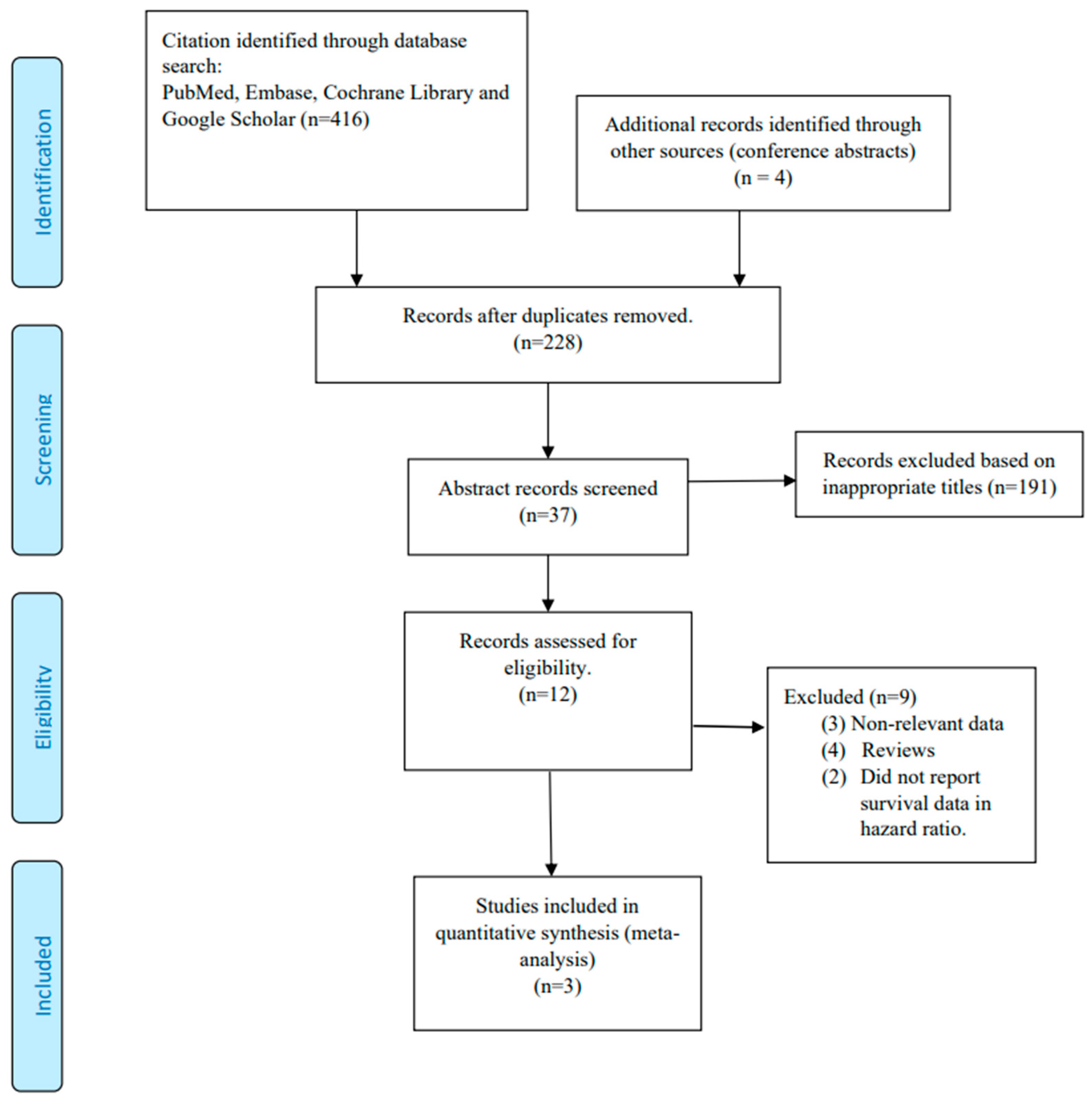
3.1. Study Selection
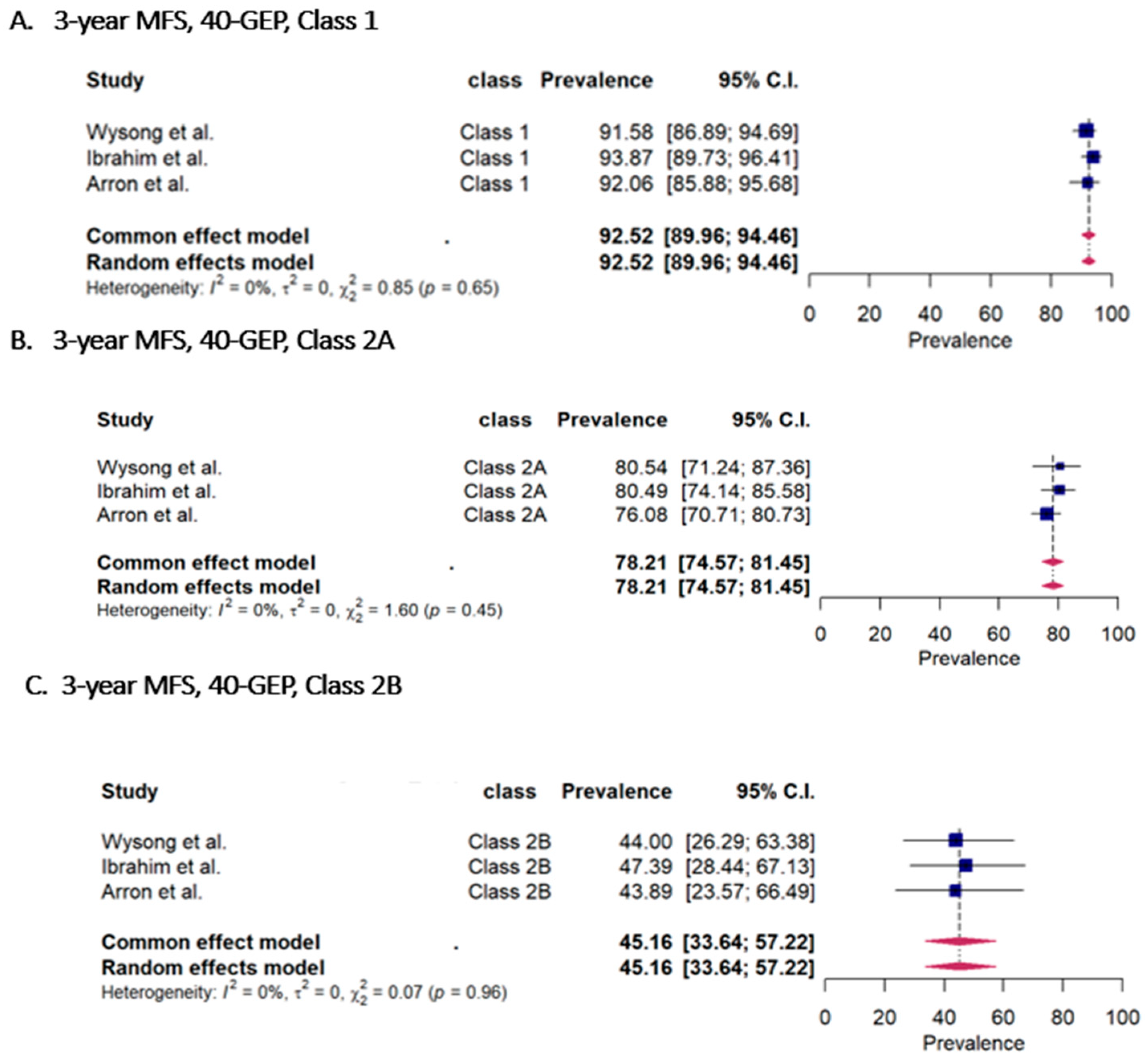
3.2. Metastasis-Free Survival (MFS) Rates
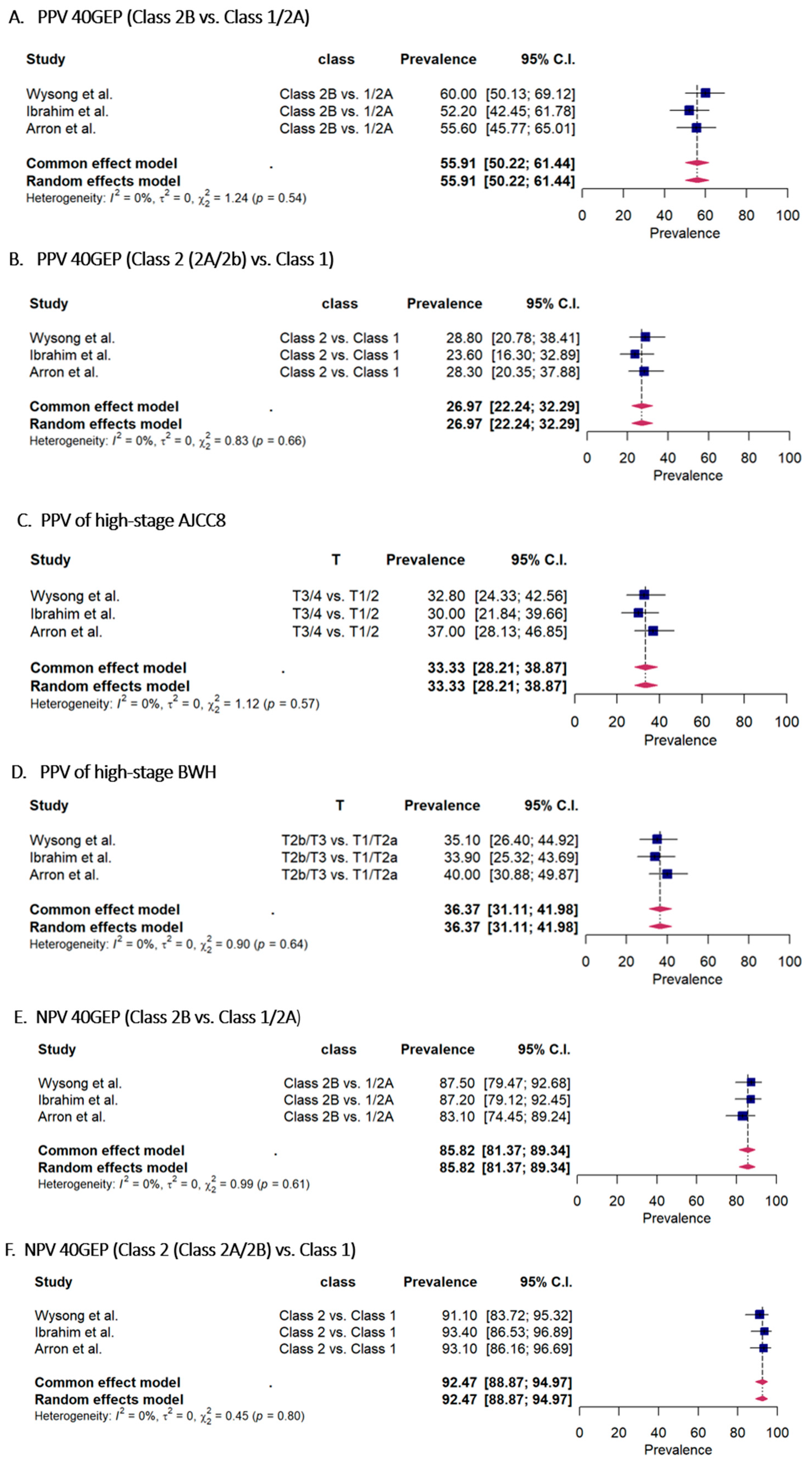
3.3. Accuracy Metric Comparison for Cutaneous Squamous Cell Carcinoma per 40-GEP Class and AJCC8/BWH T Stage
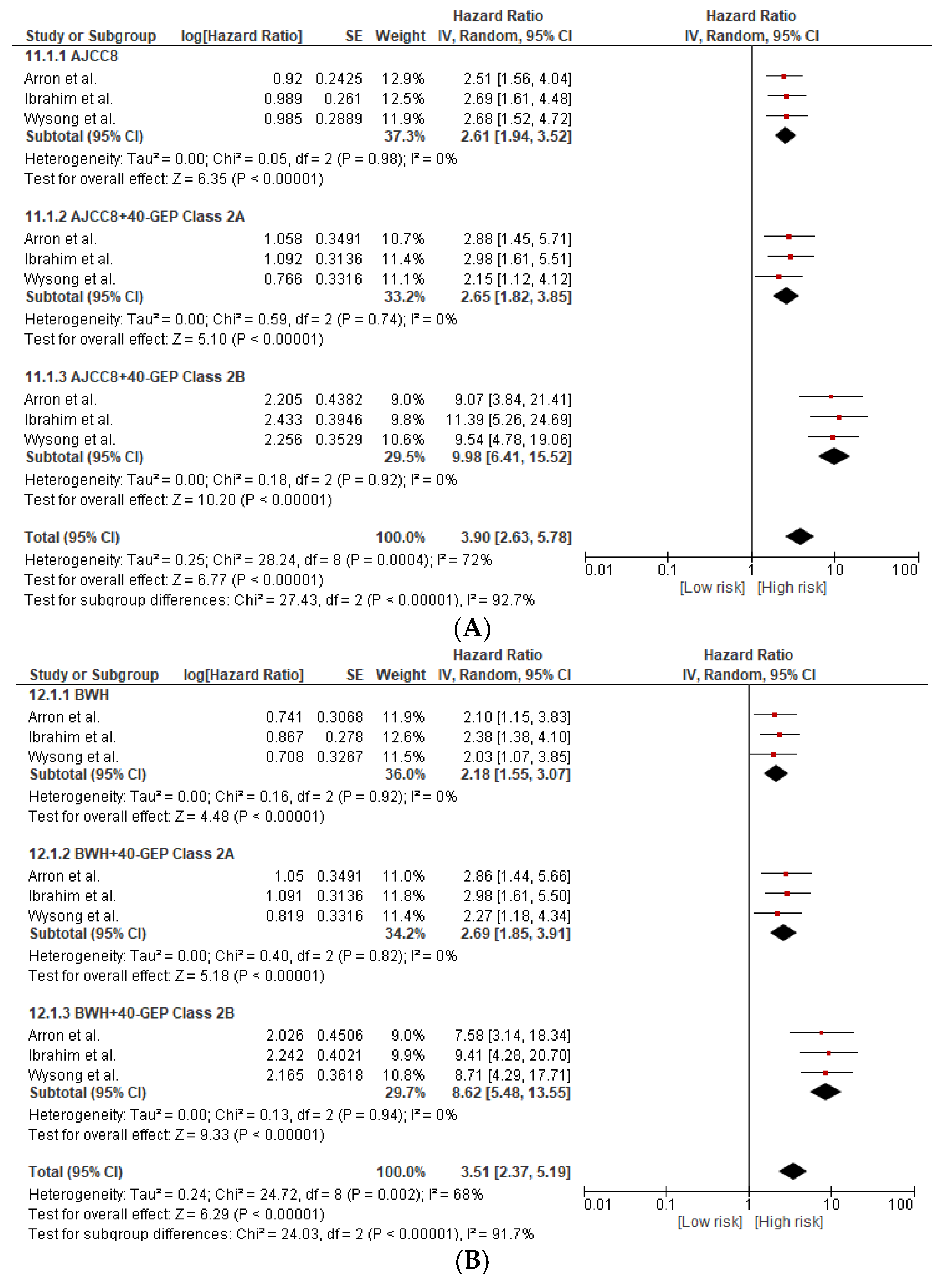
3.4. 40-GEP Prognostic Accuracy Combined with Other Staging Systems (AJCC8 and BWH Tumor Classification)
4. Discussion
4.1. Summary of Evidence
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Karia, P.S.; Han, J.; Schmults, C.D. Cutaneous squamous cell carcinoma: Estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J. Am. Acad. Dermatol. 2013, 68, 957–966. [Google Scholar] [CrossRef]
- Rogers, H.W.; Weinstock, M.A.; Feldman, S.R.; Coldiron, B.M. Incidence Estimate of Nonmelanoma Skin Cancer (Keratinocyte Carcinomas) in the US Population, 2012. JAMA Dermatol. 2015, 151, 1081–1086. [Google Scholar] [CrossRef]
- Schmults, C.D.; Karia, P.S.; Carter, J.B.; Han, J.; Qureshi, A.A. Factors Predictive of Recurrence and Death from Cutaneous Squamous Cell Carcinoma: A 10-Year, Single-Institution Cohort Study. JAMA Dermatol. 2013, 149, 541–547. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. Squamous Cell Skin Cancer, NCCN Guidelines Version 2.2021. 2021. Available online: https://www.nccn.org/professionals/physician_gls/pdf/squamous.pdf (accessed on 21 December 2022).
- Waldman, A.; Schmults, C. Cutaneous Squamous Cell Carcinoma. Hematol. Oncol. Clin. N. Am. 2019, 33, 1–12. [Google Scholar] [CrossRef]
- Thompson, A.K.; Kelley, B.F.; Prokop, L.J.; Murad, M.H.; Baum, C.L. Risk Factors for Cutaneous Squamous Cell Carcinoma Recurrence, Metastasis, and Disease-Specific Death: A Systematic Review and Meta-analysis. JAMA Dermatol. 2016, 152, 419–428. [Google Scholar] [CrossRef]
- Karia, P.S.; Jambusaria-Pahlajani, A.; Harrington, D.P.; Murphy, G.F.; Qureshi, A.A.; Schmults, C.D. Evaluation of American Joint Committee on Cancer, International Union Against Cancer, and Brigham and Women’s Hospital Tumor Staging for Cutaneous Squamous Cell Carcinoma. J. Clin. Oncol. 2014, 32, 327–334. [Google Scholar] [CrossRef]
- Ruiz, E.S.; Karia, P.S.; Besaw, R.; Schmults, C.D. Performance of the American Joint Committee on Cancer Staging Manual, 8th Edition vs the Brigham and Women’s Hospital Tumor Classification System for Cutaneous Squamous Cell Carcinoma. JAMA Dermatol. 2019, 155, 819–825. [Google Scholar] [CrossRef]
- Jambusaria-Pahlajani, A.; Kanetsky, P.A.; Karia, P.S.; Hwang, W.-T.; Gelfand, J.; Whalen, F.M.; Elenitsas, R.; Xu, X.; Schmults, C.D. Evaluation of AJCC Tumor Staging for Cutaneous Squamous Cell Carcinoma and a Proposed Alternative Tumor Staging System. JAMA Dermatol. 2013, 149, 402–410. [Google Scholar] [CrossRef]
- Roscher, I.; Falk, R.S.; Vos, L.; Clausen, O.P.F.; Helsing, P.; Gjersvik, P.; Robsahm, T.E. Validating 4 Staging Systems for Cutaneous Squamous Cell Carcinoma Using Population-Based Data: A Nested Case-Control Study. JAMA Dermatol. 2018, 154, 428–434. [Google Scholar] [CrossRef]
- Cañueto, J.; Burguillo, J.; Moyano-Bueno, D.; Viñolas-Cuadros, A.; Conde-Ferreirós, A.; Corchete-Sánchez, L.A.; Pérez-Losada, J.; Román-Curto, C. Comparing the eighth and the seventh editions of the American Joint Committee on Cancer staging system and the Brigham and Women’s Hospital alternative staging system for cutaneous squamous cell carcinoma: Implications for clinical practice. J. Am. Acad. Dermatol. 2019, 80, 106–113.e2. [Google Scholar] [CrossRef]
- Ibrahim, S.F.; Kasprzak, J.M.; Hall, M.A.; Fitzgerald, A.L.; Siegel, J.J.; Kurley, S.J.; Covington, K.R.; Goldberg, M.S.; Farberg, A.S.; Trotter, S.C.; et al. Enhanced metastatic risk assessment in cutaneous squamous cell carcinoma with the 40-gene expression profile test. Future Oncol. 2022, 18, 833–847. [Google Scholar] [CrossRef]
- Wysong, A.; Newman, J.G.; Covington, K.R.; Kurley, S.; Ibrahim, S.F.; Farberg, A.S.; Bar, A.; Cleaver, N.J.; Somani, A.-K.; Panther, D.; et al. Validation of a 40-gene expression profile test to predict metastatic risk in localized high-risk cutaneous squamous cell carcinoma. J. Am. Acad. Dermatol. 2021, 84, 361–369. [Google Scholar] [CrossRef]
- Farberg, A.S.; Fitzgerald, A.L.; Ibrahim, S.F.; Tolkachjov, S.N.; Soleymani, T.; Douglas, L.M.; Kurley, S.J.; Arron, S.T. Current Methods and Caveats to Risk Factor Assessment in Cutaneous Squamous Cell Carcinoma (cSCC): A Narrative Review. Dermatol. Ther. 2022, 12, 267–284. [Google Scholar] [CrossRef]
- Arron, S.T.; Wysong, A.; Hall, M.A.; Kurley, S.J.; Goldberg, M.S.; Kasprzak, J.M.; Somani, A.; Ibrahim, S.F. Gene expression profiling for metastatic risk in head and neck cutaneous squamous cell carcinoma. Laryngoscope Investig. Otolaryngol. 2022, 7, 135–144. [Google Scholar] [CrossRef]
- Farberg, A.S.; Hall, M.A.; Douglas, L.; Covington, K.R.; Kurley, S.; Cook, R.W.; Dinehart, S.M. Integrating gene expression profiling into NCCN high-risk cutaneous squamous cell carcinoma management recommendations: Impact on patient management. Curr. Med. Res. Opin. 2020, 36, 1301–1307. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement the BMJ. Available online: https://www.bmj.com/content/339/bmj.b2535 (accessed on 8 March 2021).
- Ottawa Hospital Research Institute. Available online: https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 16 May 2021).
- Karia, P.S.; Morgan, F.C.; Califano, J.A.; Schmults, C.D. Comparison of Tumor Classifications for Cutaneous Squamous Cell Carcinoma of the Head and Neck in the 7th vs. 8th Edition of the AJCC Cancer Staging Manual. JAMA Dermatol. 2018, 154, 175–181. [Google Scholar] [CrossRef]
- Yildiz, P.; Aung, P.P.; Milton, D.R.; Hruska, C.; Ivan, D.; Nagarajan, P.; Tetzlaff, M.T.; Curry, J.L.; Torres-Cabala, C.; Prieto, V.G. Measurement of Tumor Thickness in Cutaneous Squamous Cell Carcinomas: Do the Different Methods Provide Better Prognostic Data? Am. J. Dermatopathol. 2020, 42, 337. [Google Scholar] [CrossRef]
- Litchman, G.H.; Fitzgerald, A.L.; Kurley, S.J.; Cook, R.W.; Rigel, D.S. Impact of a prognostic 40-gene expression profiling test on clinical management decisions for high-risk cutaneous squamous cell carcinoma. Curr. Med. Res. Opin. 2020, 36, 1295–1300. [Google Scholar] [CrossRef]
- Hooper, P.B.; Farberg, A.S.; Fitzgerald, A.L.; Siegel, J.J.; Rackley, B.B.; Prasai, A.; Kurley, S.J.; Goldberg, M.S.; Litchman, G.H. Real-World Evidence Shows Clinicians Appropriately Use the Prognostic 40-Gene Expression Profile (40-GEP) Test for High-Risk Cutaneous Squamous Cell Carcinoma (cSCC) Patients. Cancer Investig. 2022, 40, 911–922. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kozlow, J.H.; Mittal, B.; Moyer, J.; Olenecki, T.; Rodgers, P.; Alam, M.; Armstrong, A.; Baum, C.; Bordeaux, J.S.; et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J. Am. Acad. Dermatol. 2018, 78, 560–578. [Google Scholar] [CrossRef]
- Burns, C.; Kubicki, S.; Nguyen, Q.B.; Aboul-Fettouh, N.; Wilmas, K.M.; Chen, O.M.; Doan, H.Q.; Silapunt, S.; Migden, M.R. Advances in Cutaneous Squamous Cell Carcinoma Management. Cancers 2022, 14, 3653. [Google Scholar] [CrossRef]
- Hedberg, M.L.; Berry, C.T.; Moshiri, A.S.; Xiang, Y.; Yeh, C.J.; Attilasoy, C.; Capell, B.C.; Seykora, J.T. Molecular Mechanisms of Cutaneous Squamous Cell Carcinoma. Int. J. Mol. Sci. 2022, 23, 3478. [Google Scholar] [CrossRef]
- Minaei, E.; Mueller, S.A.; Ashford, B.; Thind, A.S.; Mitchell, J.; Perry, J.R.; Genenger, B.; Clark, J.R.; Gupta, R.; Ranson, M. Cancer Progression Gene Expression Profiling Identifies the Urokinase Plasminogen Activator Receptor as a Biomarker of Metastasis in Cutaneous Squamous Cell Carcinoma. Front. Oncol. 2022, 12, 835929. [Google Scholar] [CrossRef]


| Study | Design, Population | Age, y, Median | Male (%) | Immunosuppression (%) | Location in Head and Neck (%) | PNI (%) Present (≥0.1 mm) | Invasion Beyond Subcutaneous Fat (%) | AJCC8 (%) | BWH (%) |
|---|---|---|---|---|---|---|---|---|---|
| Wysong et al. (2020) [13] | Patients with ≥1 high-risk features as defined by NCCN guidelines or by AJCC or BWH staging as being greater than T1. (n = 321) | 70 (34–95) | 73.2% | 23.7% | 66.7% | 2.2% | 13.4% | T1 (62.6%) T2 (18.4%) T3 (16.8%) T4 (2.2%) | T1 (57.9%) T2a (30.5%) T2b (9.4%) T3 (2.2%) |
| Ibrahim et al. (2021) [12] | Patients with ≥1 high-risk features as defined by NCCN guidelines or by AJCC or BWH staging as being greater than T1. (n = 420) | 71 (34–95) | 73.3% | 24.5% | 66.2% | 1.7% | 12.1% | NR | NR |
| Arron et al. (2022) [15] | Patients with ≥1 high-risk features as defined by NCCN guidelines or by AJCC or BWH staging as being greater than T1. (n = 278) | 71 (34–95) | 82.4% | 22.7% | 100% | 2.5% | 13.7% | T1 (57.6%) T2 (23.0%) T3 (15.8%) T4 (3.6%) | T1 (50.7%) T2a (34.9%) T2b (10.8%) T3 (3.6%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masarwy, R.; Shilo, S.; Carmel Neiderman, N.N.; Kampel, L.; Horowitz, G.; Muhanna, N.; Mansour, J. The Prognostic Value and Clinical Utility of the 40-Gene Expression Profile (40-GEP) Test in Cutaneous Squamous Cell Carcinoma: Systematic Review and Meta-Analysis. Cancers 2023, 15, 2456. https://doi.org/10.3390/cancers15092456
Masarwy R, Shilo S, Carmel Neiderman NN, Kampel L, Horowitz G, Muhanna N, Mansour J. The Prognostic Value and Clinical Utility of the 40-Gene Expression Profile (40-GEP) Test in Cutaneous Squamous Cell Carcinoma: Systematic Review and Meta-Analysis. Cancers. 2023; 15(9):2456. https://doi.org/10.3390/cancers15092456
Chicago/Turabian StyleMasarwy, Razan, Shahaf Shilo, Narin Nard Carmel Neiderman, Liyona Kampel, Gilad Horowitz, Nidal Muhanna, and Jobran Mansour. 2023. "The Prognostic Value and Clinical Utility of the 40-Gene Expression Profile (40-GEP) Test in Cutaneous Squamous Cell Carcinoma: Systematic Review and Meta-Analysis" Cancers 15, no. 9: 2456. https://doi.org/10.3390/cancers15092456
APA StyleMasarwy, R., Shilo, S., Carmel Neiderman, N. N., Kampel, L., Horowitz, G., Muhanna, N., & Mansour, J. (2023). The Prognostic Value and Clinical Utility of the 40-Gene Expression Profile (40-GEP) Test in Cutaneous Squamous Cell Carcinoma: Systematic Review and Meta-Analysis. Cancers, 15(9), 2456. https://doi.org/10.3390/cancers15092456








