Disturbing the Redox Balance Using Buthionine Sulfoximine Radiosensitized Somatostatin Receptor-2 Expressing Pre-Clinical Models to Peptide Receptor Radionuclide Therapy with 177Lu-DOTATATE
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Cell Culture
2.2. Production of 177Lu-DOTATATE
2.3. In Vitro Treatments
2.4. Cell Survival Assay
2.5. Animals and Treatments
2.6. Ex Vivo Biodistribution Studies and Time-Activity Curves
2.7. Glutathione Quantification
2.8. In Vivo Imaging and 18F-FDG PET/CT Images Analysis
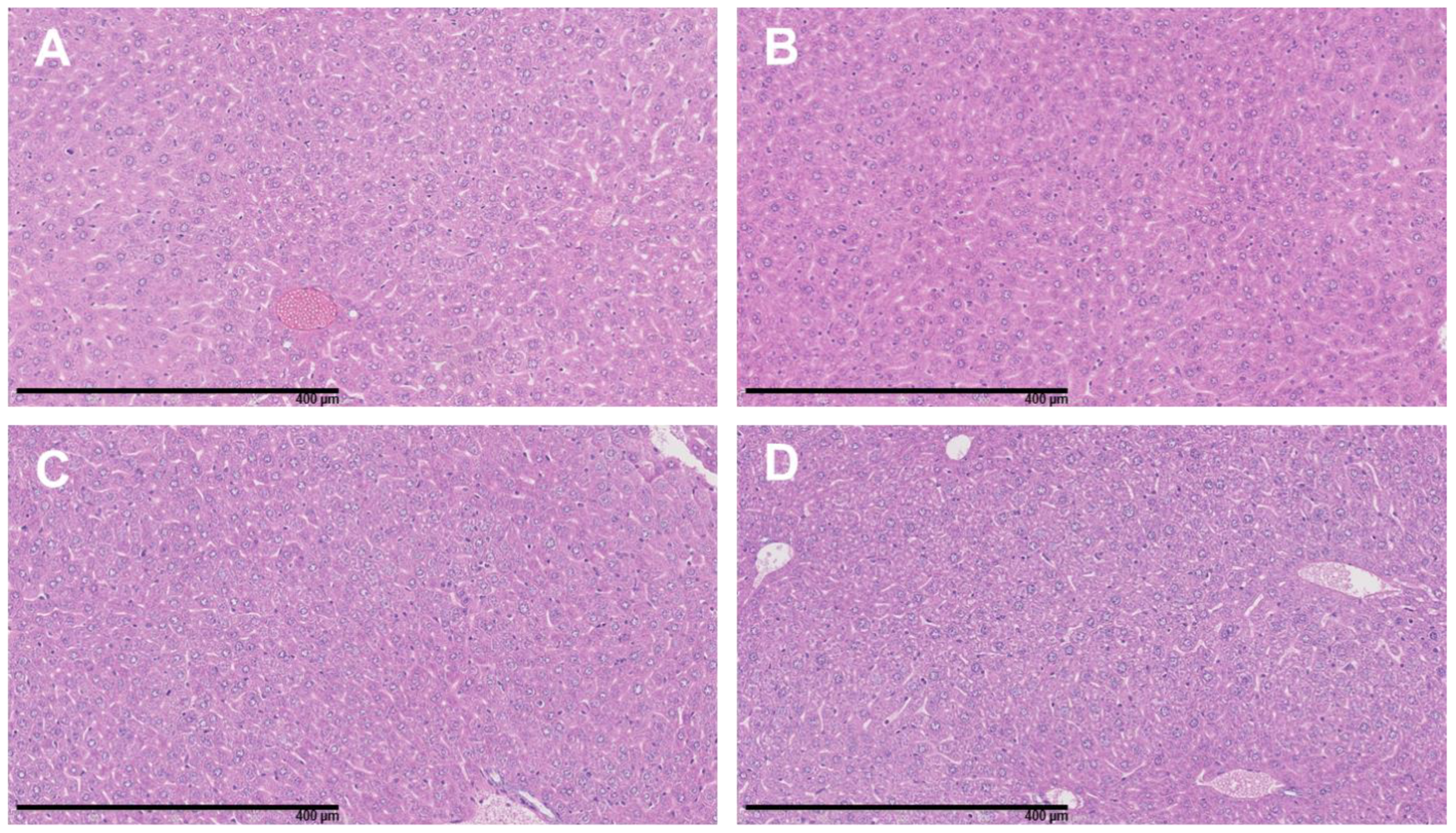
2.9. Ex Vivo Tissue Preparation and Hematoxylin and Eosin (H & E) Staining
2.10. Bone Marrow Assessment
- Bone marrow smears and cytological assessment.
- Bone marrow apoptosis assessment.
2.11. Statistical Analyses
3. Results
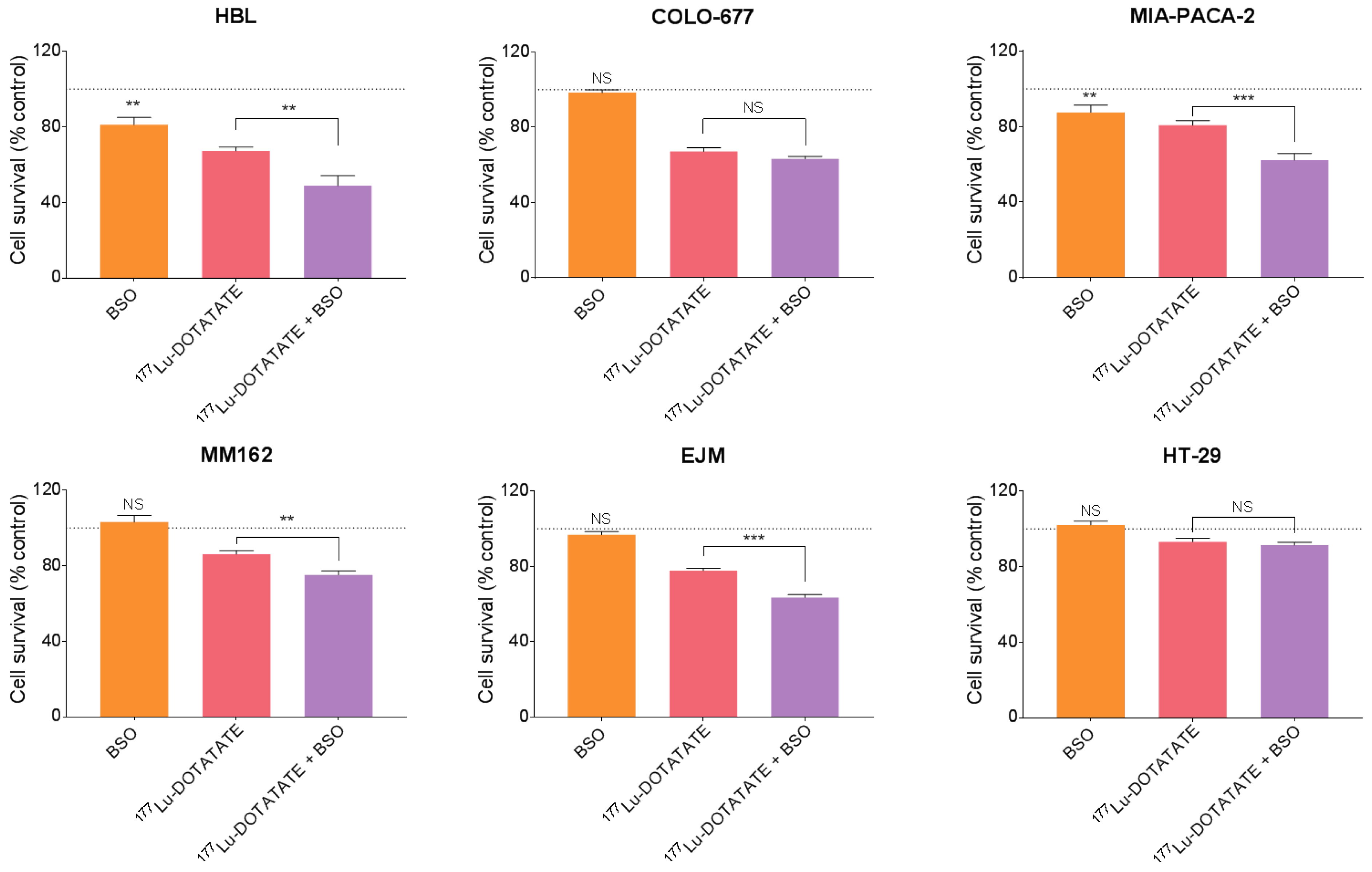
3.1. BSO-Radiosensitized SSTR-Positive Human Cancer Cell Lines to 177Lu-DOTATATE
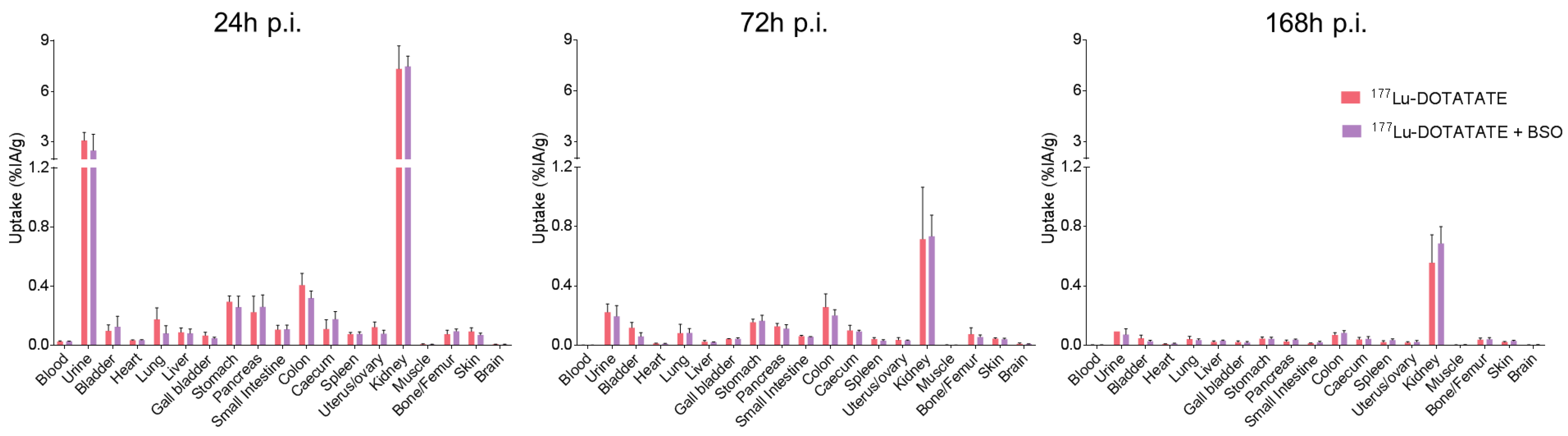
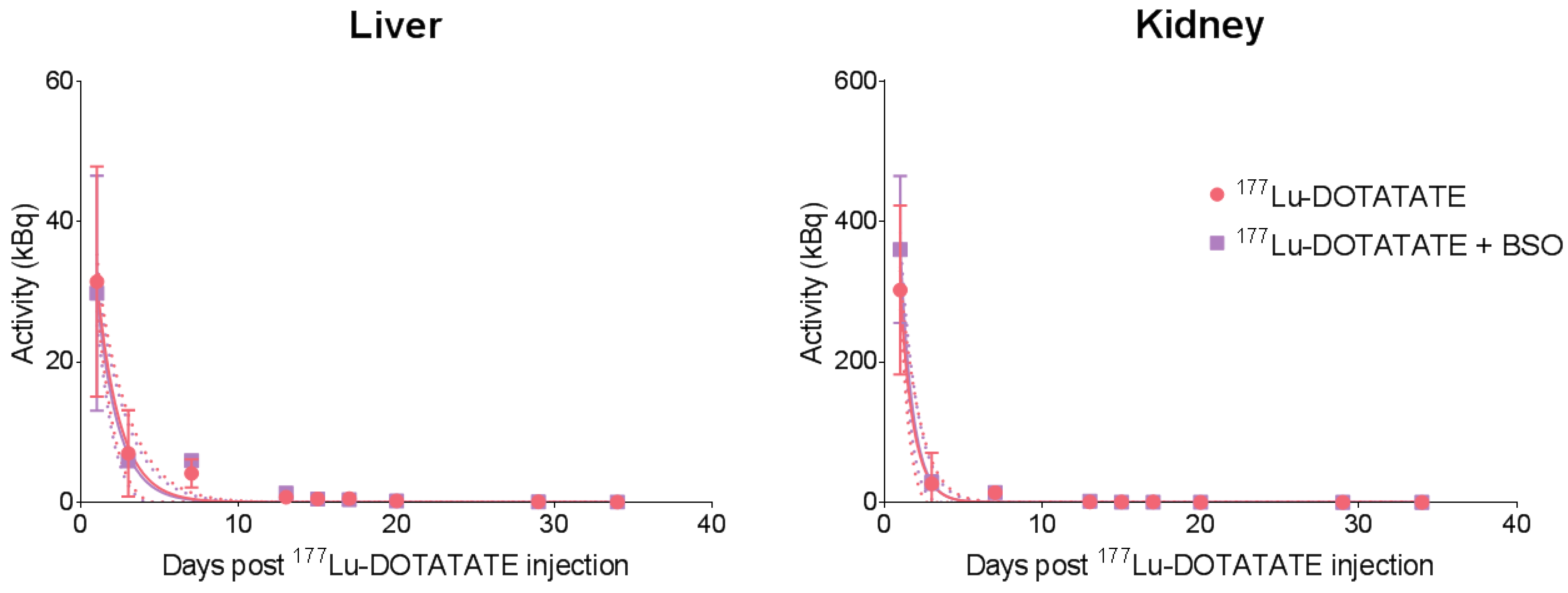
3.2. BSO Did Not Influence 177Lu-DOTATATE Biodistribution nor Pharmacokinetics In Vivo
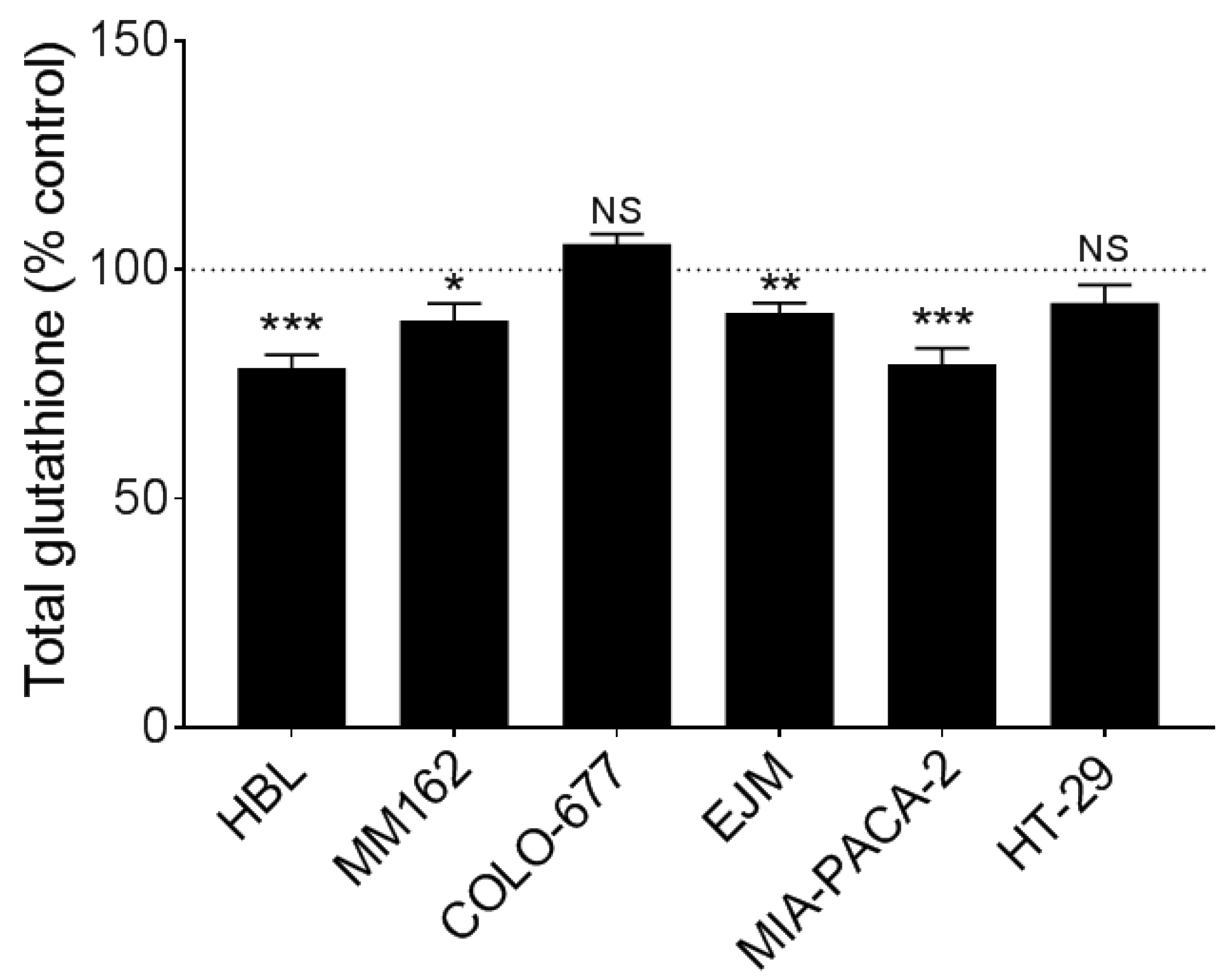
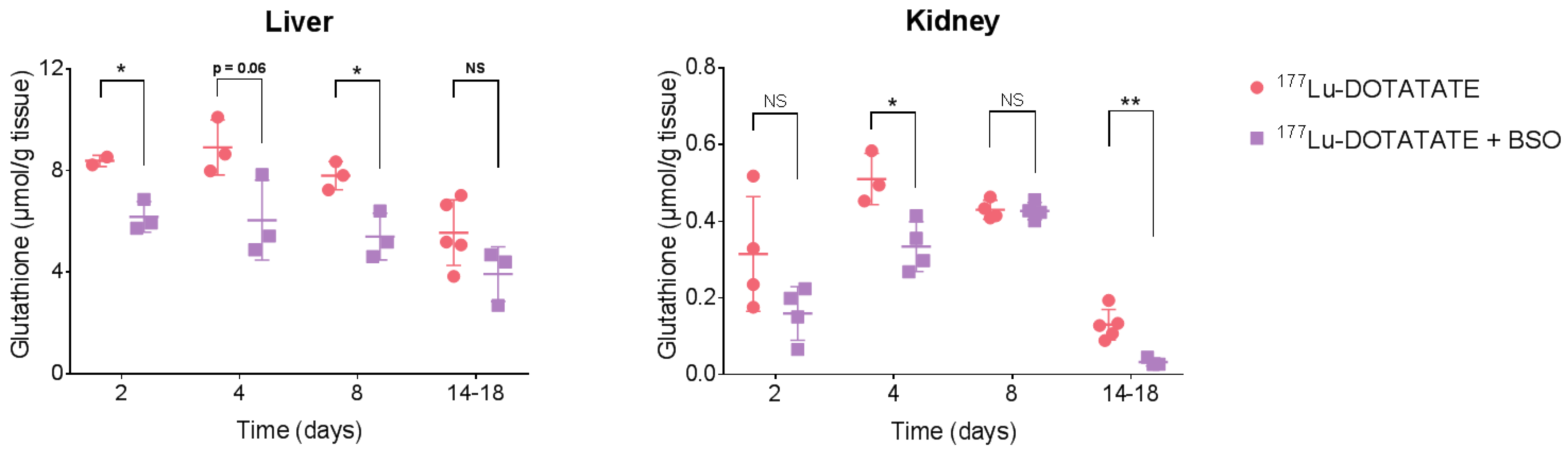
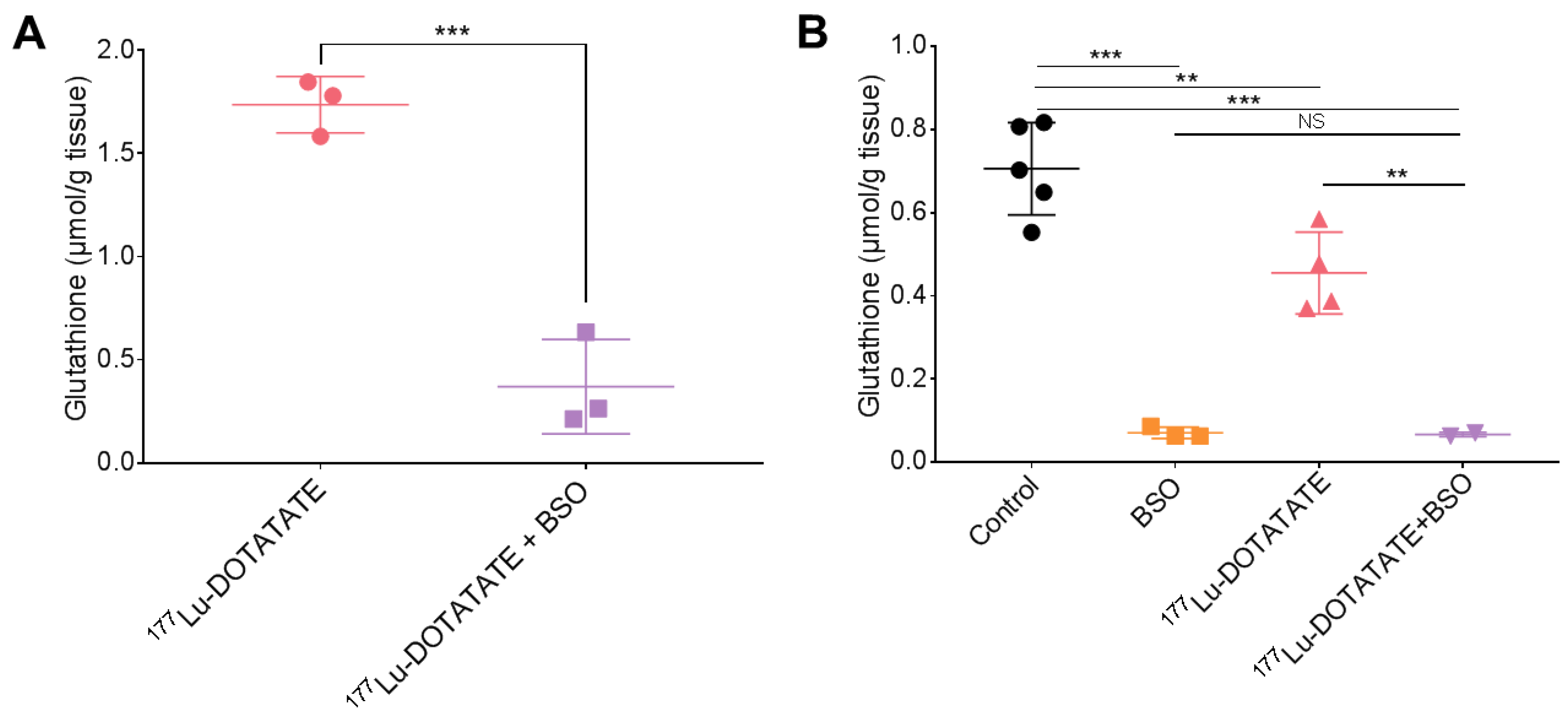
3.3. BSO Reduced Glutathione Levels in the Liver, Kidneys and Tumour
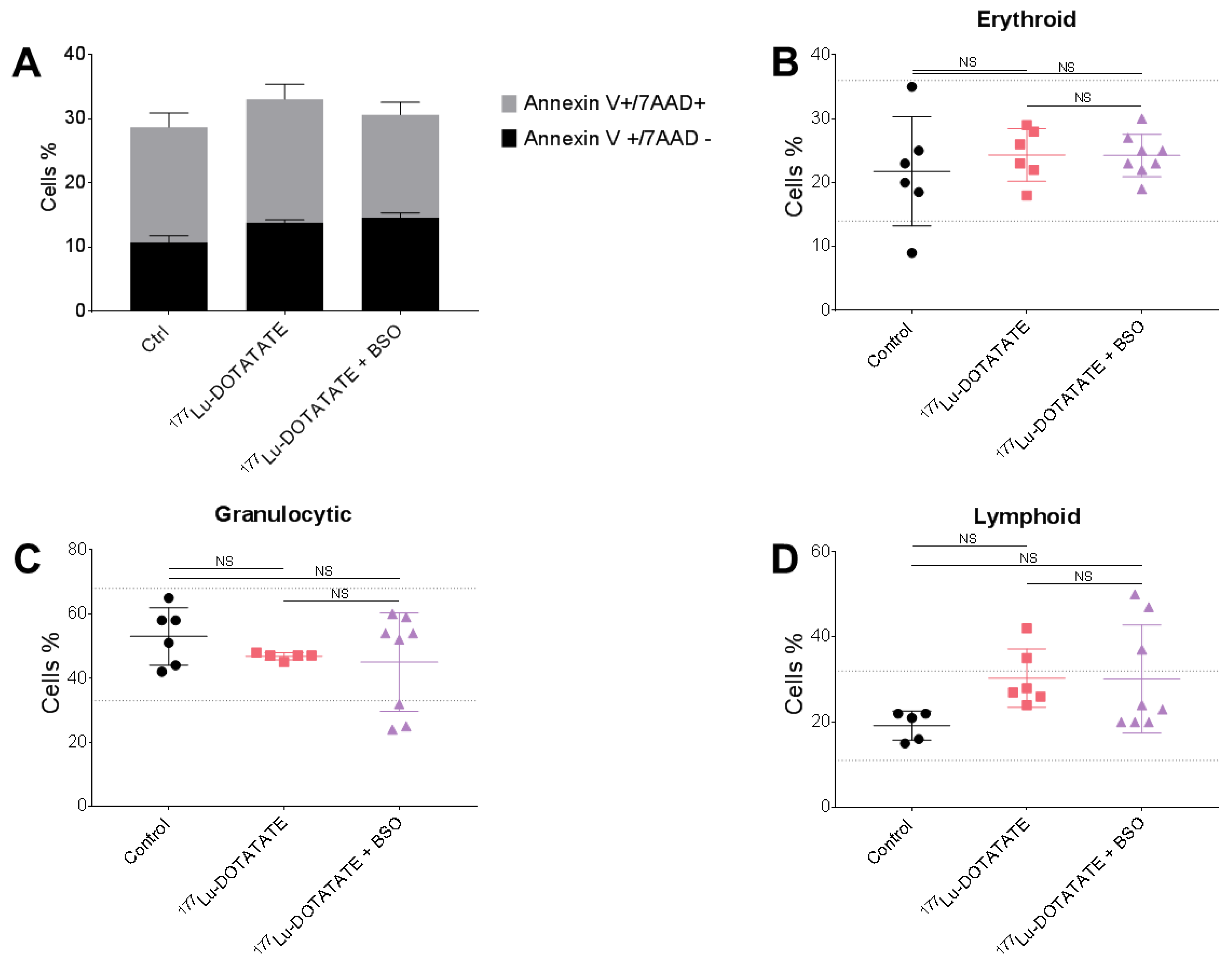
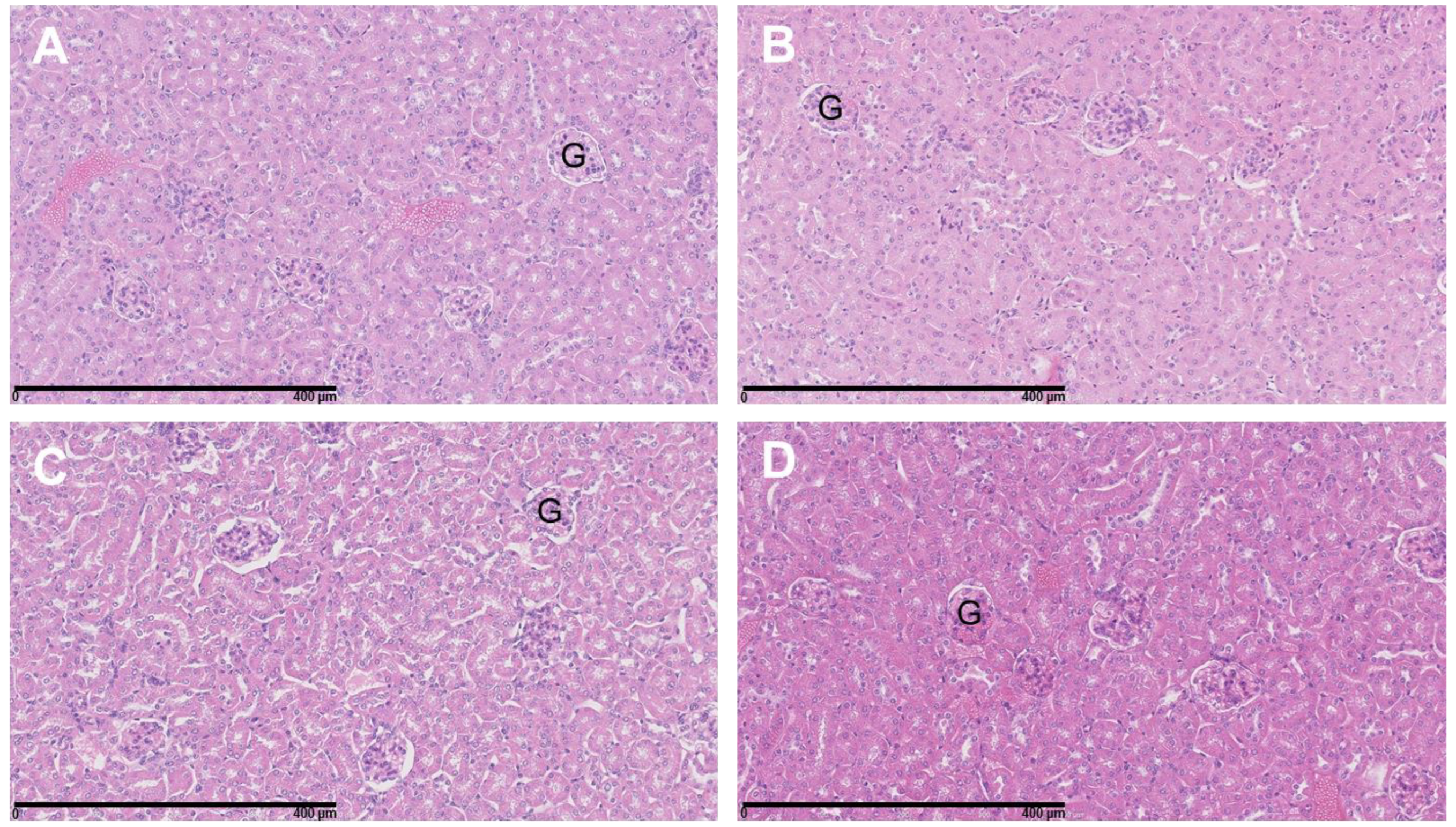
3.4. The Combination of 177Lu-DOTATATE with BSO Did Not Result in Signs of Additional Hemato-, Nephro- or Hepatotoxicity
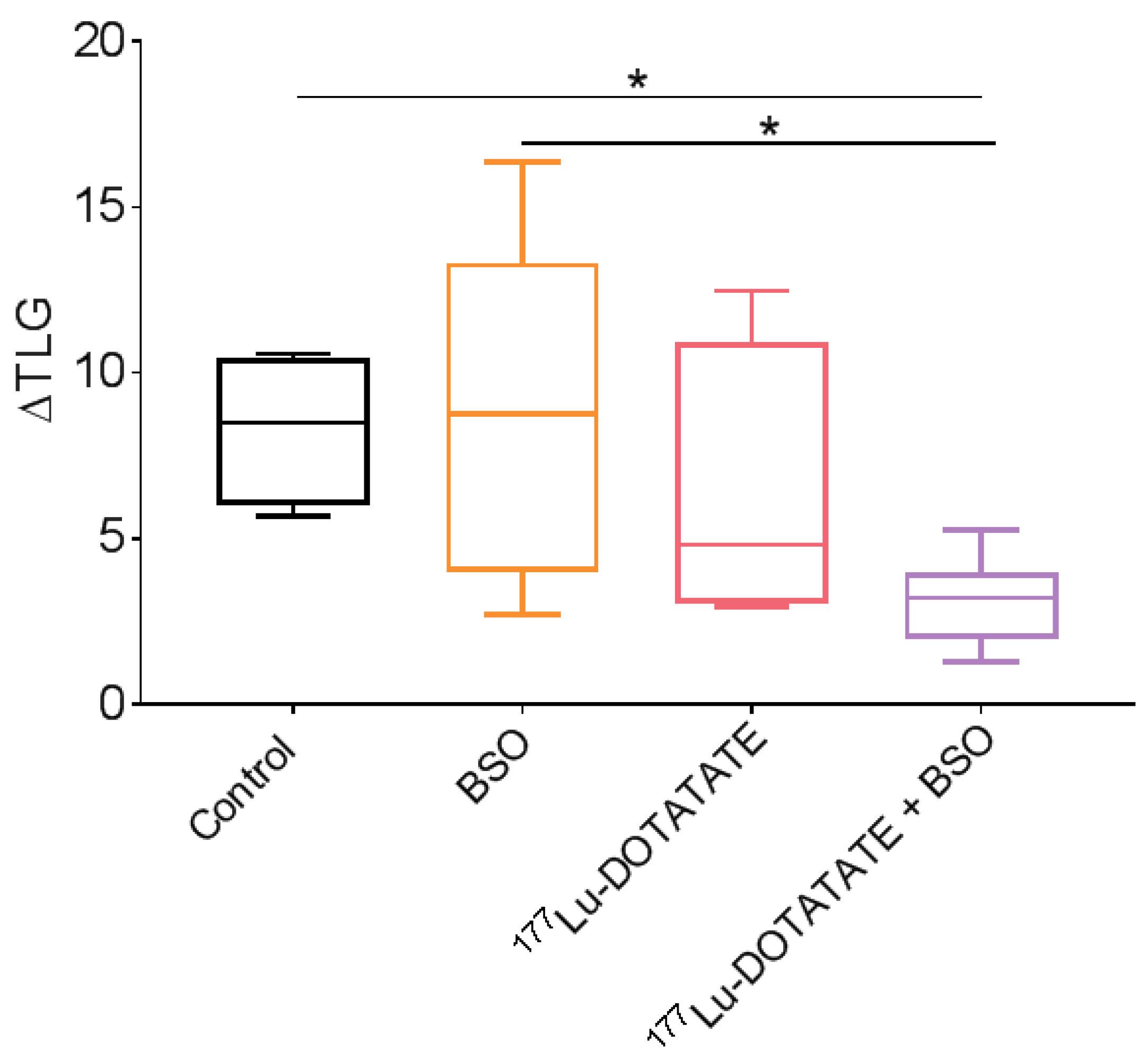
3.5. The Combination of 177Lu-DOTATATE with BSO Reduced Tumour Growth
3.6. The combination of 177Lu-DOTATATE with BSO Reduced 18F-FDG Metabolic Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Strosberg, J.; Wolin, E.; Chasen, B.; Kulke, M.; Bushnell, D.; Caplin, M.; Baum, R.P.; Kunz, P.; Hobday, T.; Hendifar, A.; et al. Health-Related Quality of Life in Patients With Progressive Midgut Neuroendocrine Tumors Treated With 177Lu-Dotatate in the Phase III NETTER-1 Trial. J. Clin. Oncol. 2018, 36, 2578–2584. [Google Scholar] [CrossRef]
- Brabander, T.; van der Zwan, W.A.; Teunissen, J.J.; Kam, B.L.; Feelders, R.A.; de Herder, W.W.; van Eijck, C.H.; Franssen, G.J.; Krenning, E.P.; Kwekkeboom, D.J. Long-Term Efficacy, Survival, and Safety of [177Lu-DOTA0,Tyr3]octreotate in Patients with Gastroenteropancreatic and Bronchial Neuroendocrine Tumors. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 4617–4624. [Google Scholar] [CrossRef] [PubMed]
- Sistani, G.; Sutherland, D.E.K.; Mujoomdar, A.; Wiseman, D.P.; Khatami, A.; Tsvetkova, E.; Reid, R.H.; Laidley, D.T. Efficacy of 177Lu-Dotatate Induction and Maintenance Therapy of Various Types of Neuroendocrine Tumors: A Phase II Registry Study. Curr. Oncol. 2020, 28, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Pouget, J.-P.; Konijnenberg, M.; Eberlein, U.; Glatting, G.; Gabina, P.M.; Herrmann, K.; Holm, S.; Strigari, L.; van Leeuwen, F.W.B.; Lassmann, M. An EANM position paper on advancing radiobiology for shaping the future of nuclear medicine. Eur. J. Nucl. Med. Mol. Imaging 2022, 50, 242–246. [Google Scholar] [CrossRef]
- Chan, T.G.; O’Neill, E.; Habjan, C.; Cornelissen, B. Combination Strategies to Improve Targeted Radionuclide Therapy. J. Nucl. Med. 2020, 61, 1544–1552. [Google Scholar] [CrossRef]
- Adant, S.; Shah, G.M.; Beauregard, J.-M. Combination treatments to enhance peptide receptor radionuclide therapy of neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 907–921. [Google Scholar] [CrossRef]
- Brabander, T.; Nonnekens, J.; Hofland, J. The next generation of peptide receptor radionuclide therapy. Endocr. Relat. Cancer 2019, 26, C7–C11. [Google Scholar] [CrossRef]
- Bidakhvidi, N.A.; Goffin, K.; Dekervel, J.; Baete, K.; Nackaerts, K.; Clement, P.; Van Cutsem, E.; Verslype, C.; Deroose, C.M. Peptide Receptor Radionuclide Therapy Targeting the Somatostatin Receptor: Basic Principles, Clinical Applications and Optimization Strategies. Cancers 2021, 14, 129. [Google Scholar] [CrossRef]
- Nonnekens, J.; Van Kranenburg, M.; Beerens, C.E.; Suker, M.; Doukas, M.; Van Eijck, C.H.; De Jong, M.; Van Gent, D.C. Potentiation of Peptide Receptor Radionuclide Therapy by the PARP Inhibitor Olaparib. Theranostics 2016, 6, 1821–1832. [Google Scholar] [CrossRef]
- Feijtel, D.; Reuvers, T.G.A.; Serooskerken, C.v.T.-V.; de Ridder, C.M.A.; Stuurman, D.C.; de Blois, E.; Verkaik, N.S.; de Bruijn, P.; Koolen, S.L.W.; de Jong, M.; et al. In Vivo Efficacy Testing of Peptide Receptor Radionuclide Therapy Radiosensitization Using Olaparib. Cancers 2023, 15, 915. [Google Scholar] [CrossRef]
- Cullinane, C.; Waldeck, K.; Kirby, L.; Rogers, B.E.; Eu, P.; Tothill, R.W.; Hicks, R.J. Enhancing the anti-tumour activity of 177 Lu-DOTA-octreotate radionuclide therapy in somatostatin receptor-2 expressing tumour models by targeting PARP. Sci. Rep. 2020, 10, 10196. [Google Scholar] [CrossRef] [PubMed]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative Stress and Antioxidant Defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef]
- Delbart, W.; Ghanem, G.E.; Karfis, I.; Flamen, P.; Wimana, Z. Investigating intrinsic radiosensitivity biomarkers to peptide receptor radionuclide therapy with [177Lu]Lu-DOTATATE in a panel of cancer cell lines. Nucl. Med. Biol. 2021, 96–97, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Colvin, O.M.; Friedman, H.S.; Gamcsik, M.P.; Fenselau, C.; Hilton, J. Role of glutathione in cellular resistance to alkylating agents. Adv. Enzyme Regul. 1993, 33, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Du, S.; Du, Y.; Ren, J.; Ying, G.; Yan, Z. Glutathione reductase mediates drug resistance in glioblastoma cells by regulating redox homeostasis. J. Neurochem. 2018, 144, 93–104. [Google Scholar] [CrossRef]
- Andrews, P.A.; Schiefer, M.A.; Murphy, M.P.; Howell, S.B. Enhanced potentiation of cisplatin cytotoxicity in human ovarian carcinoma cells by prolonged glutathione depletion. Chem. Biol. Interact. 1988, 65, 51–58. [Google Scholar] [CrossRef]
- Iida, M.; Doi, H.; Asamoto, S.; Sugiyama, H.; Sakagami, H.; Kuribayashi, N.; Takeda, M.; Okamura, Y.; Matsumoto, K. Effect of glutathione-modulating compounds on platinum compounds-induced cytotoxicity in human glioma cell lines. Anticancer Res. 1999, 19, 5383–5384. [Google Scholar]
- McGuirk, S.; Audet-Delage, Y.; Annis, M.G.; Xue, Y.; Vernier, M.; Zhao, K.; St-Louis, C.; Minarrieta, L.; Patten, D.A.; Morin, G.; et al. Resistance to different anthracycline chemotherapeutics elicits distinct and actionable primary metabolic dependencies in breast cancer. eLife 2021, 10, e65150. [Google Scholar] [CrossRef]
- Fojo, T.; Bates, S. Strategies for reversing drug resistance. Oncogene 2003, 22, 7512–7523. [Google Scholar] [CrossRef] [PubMed]
- Vanhoefer, U.; Cao, S.; Minderman, H.; Toth, K.; Skenderis, B.S.; Slovak, M.L.; Rustum, Y.M. d,l-buthionine-(S,R)-sulfoximine potentiates in vivo the therapeutic efficacy of doxorubicin against multidrug resistance protein-expressing tumors. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 1996, 2, 1961–1968. [Google Scholar]
- Griffith, O.W.; Meister, A. Potent and specific inhibition of glutathione synthesis by buthionine sulfoximine (S-n-butyl homocysteine sulfoximine). J. Biol. Chem. 1979, 254, 7558–7560. [Google Scholar] [CrossRef]
- Griffith, O.W. Mechanism of action, metabolism, and toxicity of buthionine sulfoximine and its higher homologs, potent inhibitors of glutathione synthesis. J. Biol. Chem. 1982, 257, 13704–13712. [Google Scholar] [CrossRef] [PubMed]
- Bailey, H.H. l-S,R-buthionine sulfoximine: Historical development and clinical issues. Chem. Biol. Interact. 1998, 111–112, 239–254. [Google Scholar] [CrossRef]
- Dorr, R.T.; Liddil, J.D.; Soble, M.J. Cytotoxic effects of glutathione synthesis inhibition by L-buthionine-(SR)-sulfoximine on human and murine tumor cells. Investig. New Drugs 1986, 4, 305–313. [Google Scholar] [CrossRef]
- Tagde, A.; Singh, H.; Kang, M.H.; Reynolds, C.P. The glutathione synthesis inhibitor buthionine sulfoximine synergistically enhanced melphalan activity against preclinical models of multiple myeloma. Blood Cancer J. 2014, 4, e229. [Google Scholar] [CrossRef] [PubMed]
- Georgii-Hemming, P.; Strömberg, T.; Janson, E.T.; Stridsberg, M.; Wiklund, H.J.; Nilsson, K. The somatostatin analog octreotide inhibits growth of interleukin-6 (IL-6)-dependent and IL-6-independent human multiple myeloma cell lines. Blood 1999, 93, 1724–1731. [Google Scholar] [CrossRef]
- Kerros, C.; Cavey, T.; Sola, B.; Jauzac, P.; Allouche, S. Somatostatin and opioid receptors do not regulate proliferation or apoptosis of the human multiple myeloma U266 cells. J. Exp. Clin. Cancer Res. 2009, 28, 77. [Google Scholar] [CrossRef]
- Sharma, P.; Dhull, V.S.; Suman, S.K.C.; Bal, C.; Malhotra, A.; Kumar, R. 68Ga-DOTANOC somatostatin receptor PET-CT imaging in multiple myeloma. Clin. Nucl. Med. 2014, 39, 374–375. [Google Scholar] [CrossRef]
- Agool, A.; Slart, R.H.J.A.; Dierckx, R.A.J.O.; Kluin, P.M.; Visser, L.; Jager, P.L.; Vellenga, E. Somatostatin receptor scintigraphy might be useful for detecting skeleton abnormalities in patients with multiple myeloma and plasmacytoma. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 124–130. [Google Scholar] [CrossRef]
- Sonmezoglu, K.; Vatankulu, B.; Elverdi, T.; Akyel, R.; Erkan, M.E.; Halac, M.; Ocak, M.; Demirci, E.; Aydin, Y. The role of 68Ga-DOTA-TATE PET/CT scanning in the evaluation of patients with multiple myeloma: Preliminary results. Nucl. Med. Commun. 2017, 38, 76–83. [Google Scholar] [CrossRef]
- Helgebostad, R.; Revheim, M.-E.; Johnsrud, K.; Amlie, K.; Alavi, A.; Connelly, J.P. Clinical Applications of Somatostatin Receptor (Agonist) PET Tracers beyond Neuroendocrine Tumors. Diagnostics 2022, 12, 528. [Google Scholar] [CrossRef] [PubMed]
- Zimmerer, R.M.; Korn, P.; Demougin, P.; Kampmann, A.; Kokemüller, H.; Eckardt, A.M.; Gellrich, N.-C.; Tavassol, F. Functional features of cancer stem cells in melanoma cell lines. Cancer Cell Int. 2013, 13, 78. [Google Scholar] [CrossRef]
- Krayem, M.; Journe, F.; Wiedig, M.; Morandini, R.; Sales, F.; Awada, A.; Ghanem, G. Prominent role of cyclic adenosine monophosphate signalling pathway in the sensitivity of WTBRAF/WTNRAS melanoma cells to vemurafenib. Eur. J. Cancer 2014, 50, 1310–1320. [Google Scholar] [CrossRef]
- Rambow, F.; Rogiers, A.; Marin-Bejar, O.; Aibar, S.; Femel, J.; Dewaele, M.; Karras, P.; Brown, D.; Chang, Y.H.; Debiec-Rychter, M.; et al. Toward Minimal Residual Disease-Directed Therapy in Melanoma. Cell 2018, 174, 843–855.e19. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.P. The changing paradigm of tumour response to irradiation. Br. J. Radiol. 2017, 90, 20160474. [Google Scholar] [CrossRef]
- Marin, G.; Vanderlinden, B.; Karfis, I.; Guiot, T.; Wimana, Z.; Reynaert, N.; Vandenberghe, S.; Flamen, P. A dosimetry procedure for organs-at-risk in 177Lu peptide receptor radionuclide therapy of patients with neuroendocrine tumours. Phys. Med. 2018, 56, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Sagisaka, H.; Arakawa, S.; Shibaya, Y.; Watanabe, M.; Igarashi, I.; Tanaka, K.; Totsuka, S.; Takasaki, W.; Manabe, S. A novel model of continuous depletion of glutathione in mice treated with L-buthionine (S,R)-sulfoximine. J. Toxicol. Sci. 2003, 28, 455–469. [Google Scholar] [CrossRef]
- Tomayko, M.M.; Reynolds, C.P. Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother. Pharmacol. 1989, 24, 148–154. [Google Scholar] [CrossRef]
- Van Hoy, M.; Larson, M.; Nunez, V.; Martzall, A.; Leong, L.; Foreman, O.; Katavolos, P. Optimized method for murine bone marrow collection, fixation and staining for microscopic evaluation. J. Histotechnol. 2016, 39, 30–34. [Google Scholar] [CrossRef]
- Reagan, W.J.; Irizarry-Rovira, A.; Poitout-Belissent, F.; Bolliger, A.P.; Ramaiah, S.K.; Travlos, G.; Walker, D.; Bounous, D.; Walter, G.; Bone Marrow Working Group of ASVCP/STP. Best practices for evaluation of bone marrow in nonclinical toxicity studies. Toxicol. Pathol. 2011, 39, 435–448. [Google Scholar] [CrossRef]
- Kramer, R.A.; Soble, M.; Howes, A.E.; Montoya, V.P. The effect of glutathione (GSH) depletion in vivo by buthionine sulfoximine (BSO) on the radiosensitization of SR 2508. Int. J. Radiat. Oncol. 1989, 16, 1325–1329. [Google Scholar] [CrossRef]
- Sun, J.D.; Sha Ragsdale, S.; Benson, J.M.; Henderson, R.F. Effects of the long-term depletion of reduced glutathione in mice administered l-buthionine-S,R-sulfoximine. Fundam. Appl. Toxicol. 1985, 5, 913–919. [Google Scholar] [CrossRef]
- Villablanca, J.G.; Volchenboum, S.L.; Cho, H.; Kang, M.H.; Cohn, S.L.; Anderson, C.P.; Marachelian, A.; Groshen, S.; Tsao-Wei, D.; Matthay, K.K.; et al. A Phase I New Approaches to Neuroblastoma Therapy Study of Buthionine Sulfoximine and Melphalan With Autologous Stem Cells for Recurrent/Refractory High-Risk Neuroblastoma. Pediatr. Blood Cancer 2016, 63, 1349–1356. [Google Scholar] [CrossRef]
- Bailey, H.H.; Mulcahy, R.T.; Tutsch, K.D.; Arzoomanian, R.Z.; Alberti, D.; Tombes, M.B.; Wilding, G.; Pomplun, M.; Spriggs, D.R. Phase I clinical trial of intravenous L-buthionine sulfoximine and melphalan: An attempt at modulation of glutathione. J. Clin. Oncol. 1994, 12, 194–205. [Google Scholar] [CrossRef]
- O’Dwyer, P.J.; Hamilton, T.C.; LaCreta, F.P.; Gallo, J.M.; Kilpatrick, D.; Halbherr, T.; Brennan, J.; Bookman, M.A.; Hoffman, J.; Young, R.C.; et al. Phase I trial of buthionine sulfoximine in combination with melphalan in patients with cancer. J. Clin. Oncol. 1996, 14, 249–256. [Google Scholar] [CrossRef]
- Bailey, H.H.; Ripple, G.; Tutsch, K.D.; Arzoomanian, R.Z.; Alberti, D.; Feierabend, C.; Mahvi, D.; Schink, J.; Pomplun, M.; Mulcahy, R.T.; et al. Phase I Study of Continuous-Infusion l—S,R-Buthionine Sulfoximine With Intravenous Melphalan. JNCI J. Natl. Cancer Inst. 1997, 89, 1789–1796. [Google Scholar] [CrossRef]
- Schmitt, A.; Bernhardt, P.; Nilsson, O.; Ahlman, H.; Kölby, L.; Schmitt, J.; Forssel-Aronsson, E. Biodistribution and Dosimetry of 177Lu-Labeled [DOTA0,Tyr3]Octreotate in Male Nude Mice with Human Small Cell Lung Cancer. Cancer Biother. Radiopharm. 2003, 18, 593–599. [Google Scholar] [CrossRef]
- Dalmo, J.; Rudqvist, N.; Spetz, J.; Laverman, P.; Nilsson, O.; Ahlman, H.; Forssell-Aronsson, E. Biodistribution of 177Lu-octreotate and 111In-minigastrin in female nude mice transplanted with human medullary thyroid carcinoma GOT2. Oncol. Rep. 2012, 27, 174–181. [Google Scholar] [CrossRef]
- Bodei, L.; Kidd, M.; Paganelli, G.; Grana, C.M.; Drozdov, I.; Cremonesi, M.; Lepensky, C.; Kwekkeboom, D.J.; Baum, R.P.; Krenning, E.P.; et al. Long-term tolerability of PRRT in 807 patients with neuroendocrine tumours: The value and limitations of clinical factors. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 5–19. [Google Scholar] [CrossRef]
- Bergsma, H.; Konijnenberg, M.W.; Kam, B.L.R.; Teunissen, J.J.M.; Kooij, P.P.; de Herder, W.W.; Franssen, G.J.H.; van Eijck, C.H.J.; Krenning, E.P.; Kwekkeboom, D.J. Subacute haematotoxicity after PRRT with 177Lu-DOTA-octreotate: Prognostic factors, incidence and course. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 453–463. [Google Scholar] [CrossRef]
- Griffith, O.W.; Meister, A. Glutathione: Interorgan translocation, turnover, and metabolism. Proc. Natl. Acad. Sci. USA 1979, 76, 5606–5610. [Google Scholar] [CrossRef]
- Neuschwander-Tetri, B.A.; Rozin, T. Diurnal variability of cysteine and glutathione content in the pancreas and liver of the mouse. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1996, 114, 91–95. [Google Scholar] [CrossRef]
- Skapek, S.X.; Vandellen, A.F.; McMahon, D.P.; Postels, D.G.; Griffith, O.W.; Bigner, D.D.; Friedman, H.S. Melphalan-induced toxicity in nude mice following pretreatment with buthionine sulfoximine. Cancer Chemother. Pharmacol. 1991, 28, 15–21. [Google Scholar] [CrossRef]
- Yang, M.; Büsche, G.; Ganser, A.; Li, Z. Morphology and quantitative composition of hematopoietic cells in murine bone marrow and spleen of healthy subjects. Ann. Hematol. 2013, 92, 587–594. [Google Scholar] [CrossRef]
- Svensson, J.; Mölne, J.; Forssell-Aronsson, E.; Konijnenberg, M.; Bernhardt, P. Nephrotoxicity profiles and threshold dose values for [177Lu]-DOTATATE in nude mice. Nucl. Med. Biol. 2012, 39, 756–762. [Google Scholar] [CrossRef]
- Capello, A.; Krenning, E.; Bernard, B.; Reubi, J.-C.; Breeman, W.; de Jong, M. 111In-labelled somatostatin analogues in a rat tumour model: Somatostatin receptor status and effects of peptide receptor radionuclide therapy. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 1288–1295. [Google Scholar] [CrossRef]
- de Jong, M.; Breeman, W.A.; Bernard, B.F.; Bakker, W.H.; Schaar, M.; van Gameren, A.; Bugaj, J.E.; Erion, J.; Schmidt, M.; Srinivasan, A.; et al. [177Lu-DOTA0,Tyr3]octreotate for somatostatin receptor-targeted radionuclide therapy. Int. J. Cancer 2001, 92, 628–633. [Google Scholar] [CrossRef]
- Smith, A.C.; Liao, J.T.; Page, J.G.; Wientjes, M.G.; Grieshaber, C.K. Pharmacokinetics of buthionine sulfoximine (NSC 326231) and its effect on melphalan-induced toxicity in mice. Cancer Res. 1989, 49, 5385–5391. [Google Scholar]


| HBL | MM162 | COLO-677 | EJM | MIA-PACA-2 | HT-29 |
|---|---|---|---|---|---|
| 0.89 | 0.83 | NA | 0.85 | 0.88 | NA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delbart, W.; Marin, G.; Stamatopoulos, B.; de Wind, R.; Sirtaine, N.; Demetter, P.; Vercruyssen, M.; Woff, E.; Karfis, I.; Ghanem, G.E.; et al. Disturbing the Redox Balance Using Buthionine Sulfoximine Radiosensitized Somatostatin Receptor-2 Expressing Pre-Clinical Models to Peptide Receptor Radionuclide Therapy with 177Lu-DOTATATE. Cancers 2023, 15, 2332. https://doi.org/10.3390/cancers15082332
Delbart W, Marin G, Stamatopoulos B, de Wind R, Sirtaine N, Demetter P, Vercruyssen M, Woff E, Karfis I, Ghanem GE, et al. Disturbing the Redox Balance Using Buthionine Sulfoximine Radiosensitized Somatostatin Receptor-2 Expressing Pre-Clinical Models to Peptide Receptor Radionuclide Therapy with 177Lu-DOTATATE. Cancers. 2023; 15(8):2332. https://doi.org/10.3390/cancers15082332
Chicago/Turabian StyleDelbart, Wendy, Gwennaëlle Marin, Basile Stamatopoulos, Roland de Wind, Nicolas Sirtaine, Pieter Demetter, Marie Vercruyssen, Erwin Woff, Ioannis Karfis, Ghanem E. Ghanem, and et al. 2023. "Disturbing the Redox Balance Using Buthionine Sulfoximine Radiosensitized Somatostatin Receptor-2 Expressing Pre-Clinical Models to Peptide Receptor Radionuclide Therapy with 177Lu-DOTATATE" Cancers 15, no. 8: 2332. https://doi.org/10.3390/cancers15082332
APA StyleDelbart, W., Marin, G., Stamatopoulos, B., de Wind, R., Sirtaine, N., Demetter, P., Vercruyssen, M., Woff, E., Karfis, I., Ghanem, G. E., Flamen, P., & Wimana, Z. (2023). Disturbing the Redox Balance Using Buthionine Sulfoximine Radiosensitized Somatostatin Receptor-2 Expressing Pre-Clinical Models to Peptide Receptor Radionuclide Therapy with 177Lu-DOTATATE. Cancers, 15(8), 2332. https://doi.org/10.3390/cancers15082332






