Decreased Lipid Phosphate Phosphatase 1/3 and Increased Lipid Phosphate Phosphatase 2 Expression in the Human Breast Cancer Tumor Microenvironment Promotes Tumor Progression and Immune System Evasion
Abstract
Simple Summary
Abstract
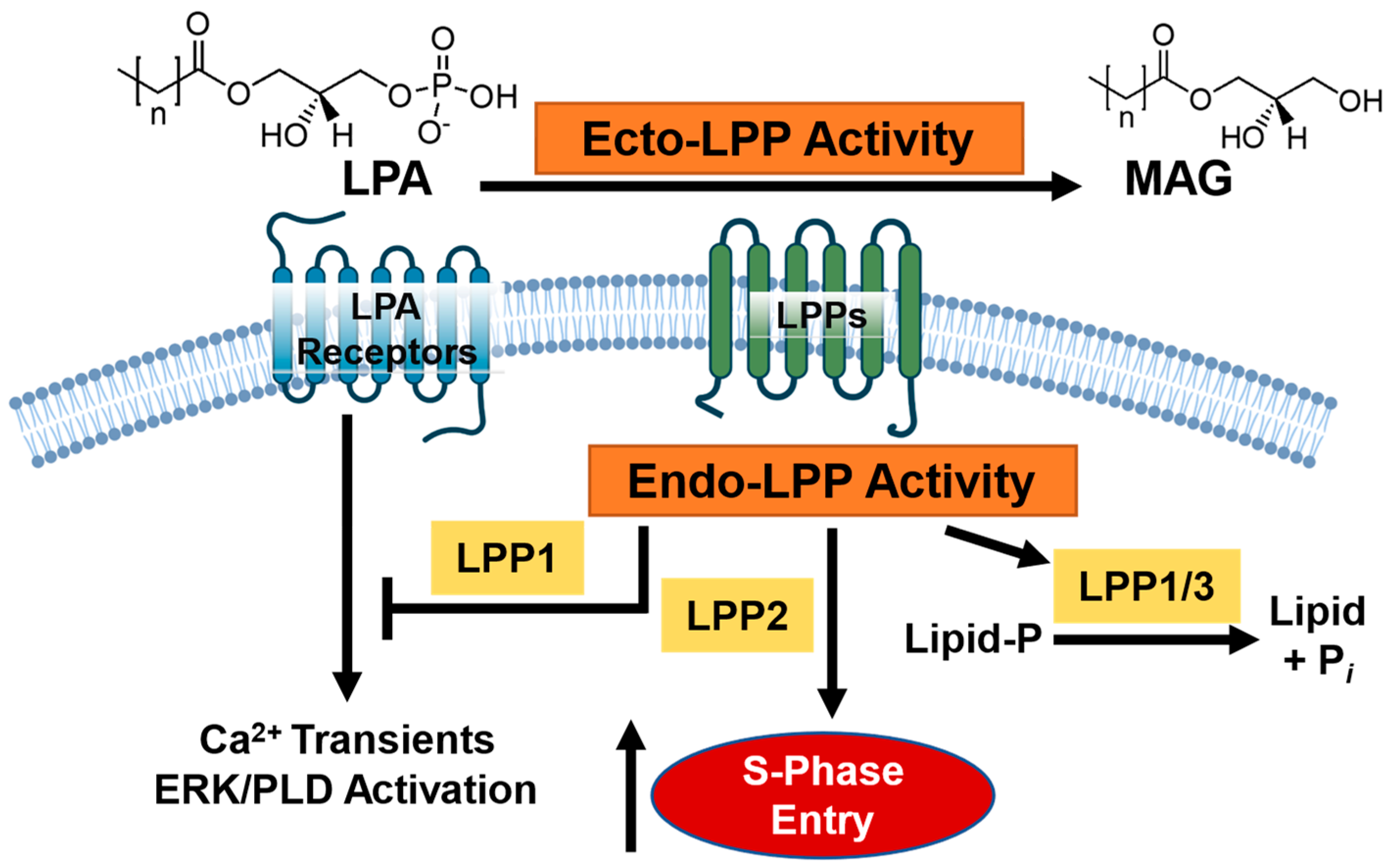
1. Introduction
2. Materials and Methods
2.1. Data Acquisition
2.2. Gene Set Enrichment Analysis
2.3. Other Scores
2.4. Statistical Anlayses
3. Results
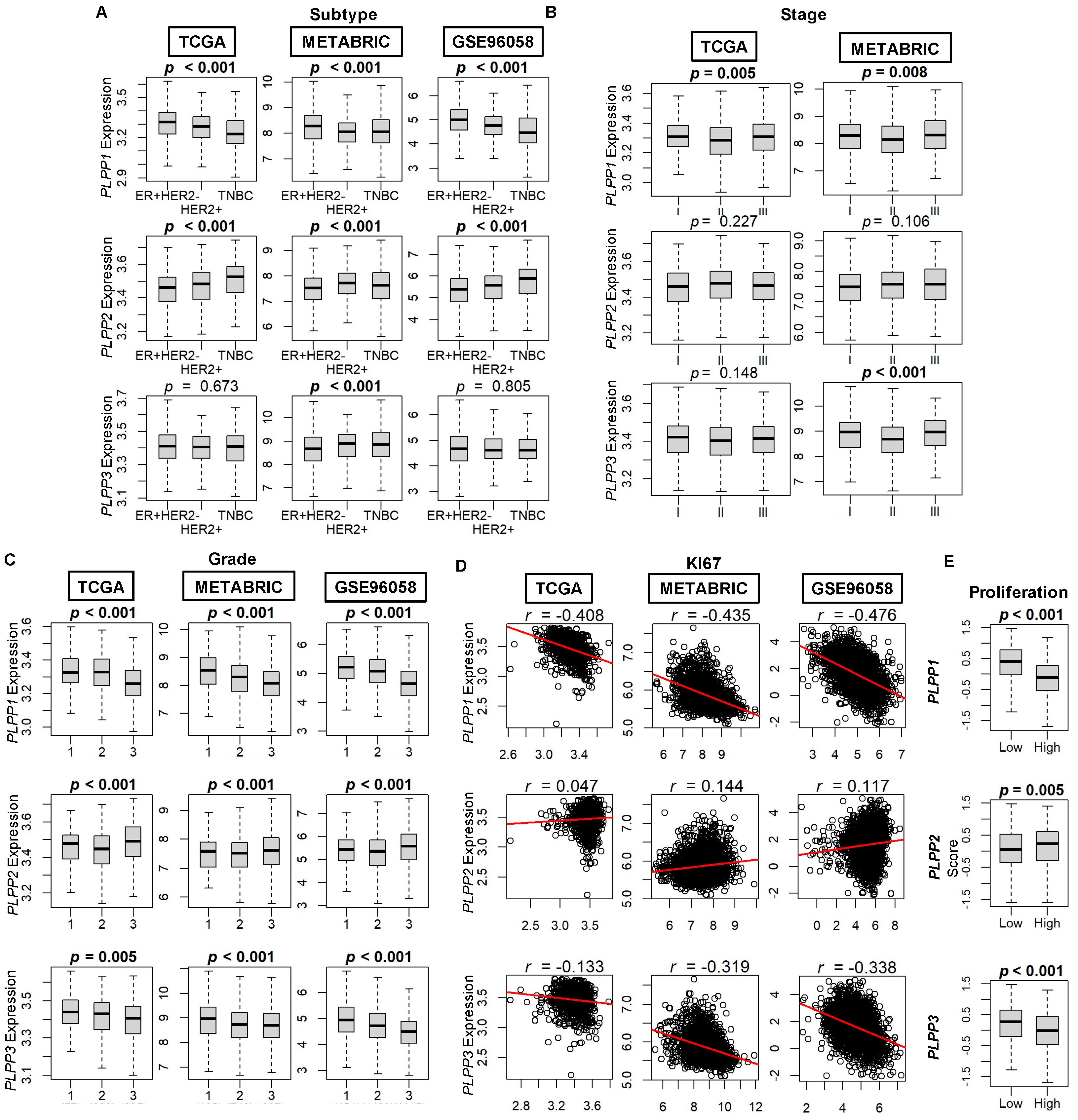
3.1. High LPP1 and LPP3 Gene Expression and Low LPP2 Gene Expression Correlate to a Less Aggressive Breast Cancer Phenotype
3.2. LPP1- and LPP3-Mediated Gene Set Enrichment Patterns Favor a Mixed Pro- and Anti-Cancer Phenotype, whereas LPP2 Patterns Favor Cell Cycling Progression Pathways
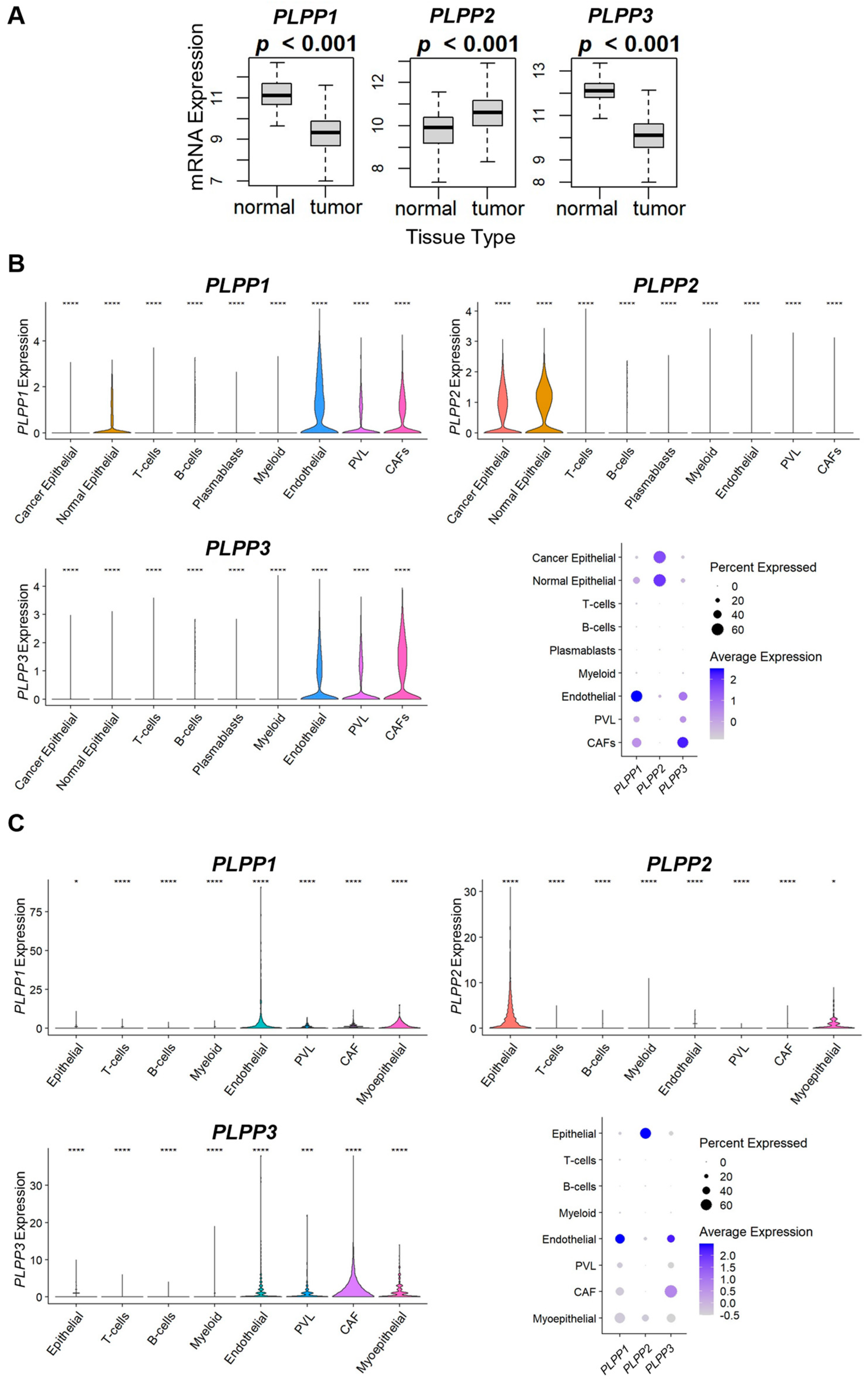
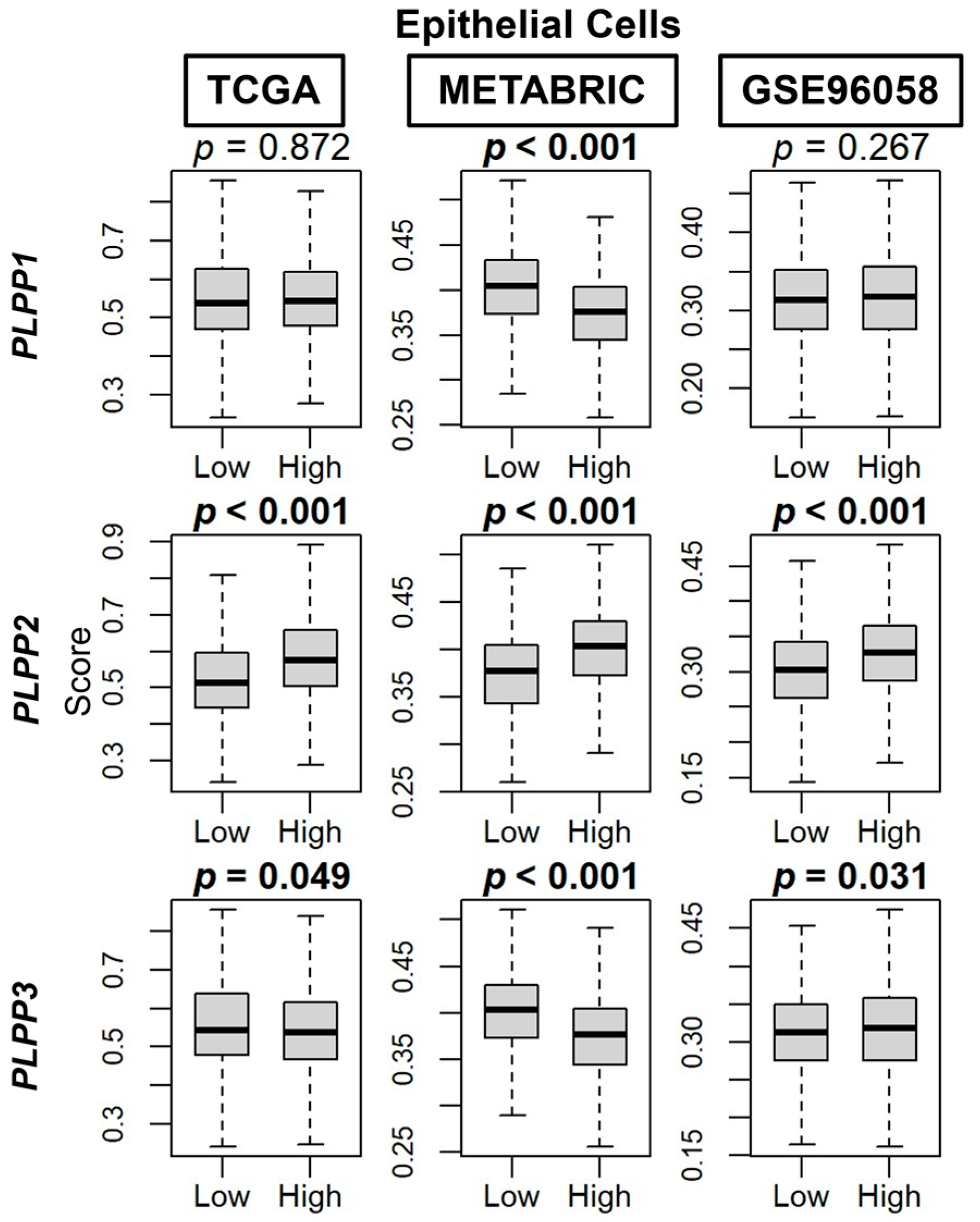
3.3. LPP1 and LPP3 Are Predominantly Expressed in Tumor Stroma, While LPP2 Is Expressed in Cancer Cells, and a Low Tumor LPP1/3 and High LPP2 Expression Pattern Correlates to Immune System Evasion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Braunstein, L.Z.; Taghian, A.G.; Niemierko, A.; Salama, L.; Capuco, A.; Bellon, J.R.; Wong, J.S.; Punglia, R.S.; MacDonald, S.M.; Harris, J.R. Breast-cancer subtype, age, and lymph node status as predictors of local recurrence following breast-conserving therapy. Breast Cancer Res. Treat. 2017, 161, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhu, Y.; Liu, X.; Liao, X.; He, J.; Niu, L. The clinicopathological features and survival outcomes of patients with different metastatic sites in stage iv breast cancer. BMC Cancer 2019, 19, 1091. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. U.S. Cancer Statistics Female Breast Cancer Stat Bite; US Department of Health and Human Services: Washington, DC, USA, 2022.
- Benesch, M.G.K.; Tang, X.; Brindley, D.N. Autotaxin and breast cancer: Towards overcoming treatment barriers and sequelae. Cancers 2020, 12, 374. [Google Scholar] [CrossRef] [PubMed]
- Lainetti, P.F.; Leis-Filho, A.F.; Laufer-Amorim, R.; Battazza, A.; Fonseca-Alves, C.E. Mechanisms of resistance to chemotherapy in breast cancer and possible targets in drug delivery systems. Pharmaceutics 2020, 12, 1193. [Google Scholar] [CrossRef]
- Benesch, M.G.K.; Yang, Z.; Tang, X.; Meng, G.; Brindley, D.N. Lysophosphatidate signaling: The tumor microenvironment’s new nemesis. Trends Cancer 2017, 3, 748–752. [Google Scholar] [CrossRef]
- Brindley, D.N.; Tang, X.; Meng, G.; Benesch, M.G.K. Role of adipose tissue-derived autotaxin, lysophosphatidate signaling, and inflammation in the progression and treatment of breast cancer. Int. J. Mol. Sci. 2020, 21, 5938. [Google Scholar] [CrossRef]
- Tang, X.; Benesch, M.G.; Brindley, D.N. Lipid phosphate phosphatases and their roles in mammalian physiology and pathology. J. Lipid Res. 2015, 56, 2048–2060. [Google Scholar] [CrossRef]
- Morris, A.J.; Smyth, S.S. Lipid phosphate phosphatases: More than one way to put the brakes on lpa signaling? J. Lipid Res. 2014, 55, 2195–2197. [Google Scholar] [CrossRef]
- Kok, B.P.; Venkatraman, G.; Capatos, D.; Brindley, D.N. Unlike two peas in a pod: Lipid phosphate phosphatases and phosphatidate phosphatases. Chem. Rev. 2012, 112, 5121–5146. [Google Scholar] [CrossRef]
- Jasinska, R.; Zhang, Q.X.; Pilquil, C.; Singh, I.; Xu, J.; Dewald, J.; Dillon, D.A.; Berthiaume, L.G.; Carman, G.M.; Waggoner, D.W.; et al. Lipid phosphate phosphohydrolase-1 degrades exogenous glycerolipid and sphingolipid phosphate esters. Biochem. J. 1999, 340 Pt 3, 677–686. [Google Scholar] [CrossRef]
- Benesch, M.G.K.; Ko, Y.M.; McMullen, T.P.W.; Brindley, D.N. Autotaxin in the crosshairs: Taking aim at cancer and other inflammatory conditions. FEBS Lett. 2014, 588, 2712–2727. [Google Scholar] [CrossRef] [PubMed]
- Benesch, M.G.; Tang, X.; Venkatraman, G.; Bekele, R.T.; Brindley, D.N. Recent advances in targeting the autotaxin-lysophosphatidate-lipid phosphate phosphatase axis in vivo. J. Biomed. Res. 2016, 30, 272–284. [Google Scholar] [PubMed]
- Lee, S.C.; Fujiwara, Y.; Tigyi, G.J. Uncovering unique roles of lpa receptors in the tumor microenvironment. Recept. Clin. Investig. 2015, 2, e440. [Google Scholar]
- Tang, X.; Benesch, M.G.; Dewald, J.; Zhao, Y.Y.; Patwardhan, N.; Santos, W.L.; Curtis, J.M.; McMullen, T.P.; Brindley, D.N. Lipid phosphate phosphatase-1 expression in cancer cells attenuates tumor growth and metastasis in mice. J. Lipid Res. 2014, 55, 2389–2400. [Google Scholar] [CrossRef]
- Nakayama, J.; Raines, T.A.; Lynch, K.R.; Slack-Davis, J.K. Decreased peritoneal ovarian cancer growth in mice lacking expression of lipid phosphate phosphohydrolase 1. PLoS ONE 2015, 10, e0120071. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; McMullen, T.P.W.; Brindley, D.N. Increasing the low lipid phosphate phosphatase 1 activity in breast cancer cells decreases transcription by ap-1 and expressions of matrix metalloproteinases and cyclin d1/d3. Theranostics 2019, 9, 6129–6142. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, J.M.; Funes, J.M.; Henderson, S.; Wild, L.; Carey, N.; Boshoff, C. Genomics screen in transformed stem cells reveals rnaseh2a, ppap2c, and adarb1 as putative anticancer drug targets. Mol. Cancer Ther. 2009, 8, 249–260. [Google Scholar] [CrossRef]
- Morris, K.E.; Schang, L.M.; Brindley, D.N. Lipid phosphate phosphatase-2 activity regulates s-phase entry of the cell cycle in rat2 fibroblasts. J. Biol. Chem. 2006, 281, 9297–9306. [Google Scholar] [CrossRef]
- Tang, X.; Cromwell, C.R.; Liu, R.; Godbout, R.; Hubbard, B.P.; McMullen, T.P.W.; Brindley, D.N. Lipid phosphate phosphatase-2 promotes tumor growth through increased c-myc expression. Theranostics 2022, 12, 5675–5690. [Google Scholar] [CrossRef]
- Pilquil, C.; Dewald, J.; Cherney, A.; Gorshkova, I.; Tigyi, G.; English, D.; Natarajan, V.; Brindley, D.N. Lipid phosphate phosphatase-1 regulates lysophosphatidate-induced fibroblast migration by controlling phospholipase d2-dependent phosphatidate generation. J. Biol. Chem. 2006, 281, 38418–38429. [Google Scholar] [CrossRef]
- Pyne, S.; Kong, K.C.; Darroch, P.I. Lysophosphatidic acid and sphingosine 1-phosphate biology: The role of lipid phosphate phosphatases. Semin. Cell Dev. Biol. 2004, 15, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Samadi, N.; Bekele, R.; Capatos, D.; Venkatraman, G.; Sariahmetoglu, M.; Brindley, D.N. Regulation of lysophosphatidate signaling by autotaxin and lipid phosphate phosphatases with respect to tumor progression, angiogenesis, metastasis and chemo-resistance. Biochimie 2011, 93, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Zhao, Y.Y.; Dewald, J.; Curtis, J.M.; Brindley, D.N. Tetracyclines increase lipid phosphate phosphatase expression on plasma membranes and turnover of plasma lysophosphatidate. J. Lipid Res. 2016, 57, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Tanyi, J.L.; Hasegawa, Y.; Lapushin, R.; Morris, A.J.; Wolf, J.K.; Berchuck, A.; Lu, K.; Smith, D.I.; Kalli, K.; Hartmann, L.C.; et al. ;Role of decreased levels of lipid phosphate phosphatase-1 in accumulation of lysophosphatidic acid in ovarian cancer. Clin. Cancer Res. 2003, 9, 3534–3545. [Google Scholar] [PubMed]
- Tomsig, J.L.; Snyder, A.H.; Berdyshev, E.V.; Skobeleva, A.; Mataya, C.; Natarajan, V.; Brindley, D.N.; Lynch, K.R. Lipid phosphate phosphohydrolase type 1 (lpp1) degrades extracellular lysophosphatidic acid in vivo. Biochem. J. 2009, 419, 611–618. [Google Scholar] [CrossRef]
- Palmer, S.M.; Snyder, L.; Todd, J.L.; Soule, B.; Christian, R.; Anstrom, K.; Luo, Y.; Gagnon, R.; Rosen, G. Randomized, double-blind, placebo-controlled, phase 2 trial of bms-986020, a lysophosphatidic acid receptor antagonist for the treatment of idiopathic pulmonary fibrosis. Chest 2018, 154, 1061–1069. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. A Study to Assess an Atx Inhibitor (ioa-289) in Patients with Metastatic Pancreatic Cancer. National Institute of Health. 2022. Available online: https://clinicaltrials.gov/ct2/show/ (accessed on 1 February 2023).
- Helmer, E.; Willson, A.; Brearley, C.; Westerhof, M.; Delage, S.; Shaw, I.; Cooke, R.; Sidhu, S. Pharmacokinetics and metabolism of ziritaxestat (glpg1690) in healthy male volunteers following intravenous and oral administration. Clin. Pharmacol. Drug Dev. 2022, 11, 246–256. [Google Scholar] [CrossRef]
- Banerjee, S.; Lee, S.; Norman, D.D.; Tigyi, G.J. Designing dual inhibitors of autotaxin-lpar gpcr axis. Molecules 2022, 27, 5487. [Google Scholar] [CrossRef]
- Meduri, B.; Pujar, G.V.; Durai Ananda Kumar, T.; Akshatha, H.S.; Sethu, A.K.; Singh, M.; Kanagarla, A.; Mathew, B. Lysophosphatidic acid (lpa) receptor modulators: Structural features and recent development. Eur. J. Med. Chem. 2021, 222, 113574. [Google Scholar] [CrossRef]
- Wu, R.; Yu, I.; Tokumaru, Y.; Asaoka, M.; Oshi, M.; Yan, L.; Okuda, S.; Ishikawa, T.; Takabe, K. Elevated bile acid metabolism and microbiome are associated with suppressed cell proliferation and better survival in breast cancer. Am. J. Cancer Res. 2022, 12, 5271–5285. [Google Scholar]
- Oshi, M.; Tokumaru, Y.; Benesch, M.G.; Sugito, N.; Wu, R.; Yan, L.; Yamada, A.; Chishima, T.; Ishikawa, T.; Endo, I.; et al. High mir-99b expression is associated with cell proliferation and worse patient outcomes in breast cancer. Am. J. Cancer Res. 2022, 12, 4840–4852. [Google Scholar] [PubMed]
- Lonsdale, J.; Thomas, J.; Salvatore, M.; Phillips, R.; Lo, E.; Shad, S.; Hasz, R.; Walters, G.; Garcia, F.; Young, N.; et al. The genotype-tissue expression (gtex) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Z.; Al-Eryani, G.; Roden, D.L.; Junankar, S.; Harvey, K.; Andersson, A.; Thennavan, A.; Wang, C.; Torpy, J.R.; Bartonicek, N.; et al. A single-cell and spatially resolved atlas of human breast cancers. Nat. Genet. 2021, 53, 1334–1347. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Z.; Roden, D.L.; Wang, C.; Holliday, H.; Harvey, K.; Cazet, A.S.; Murphy, K.J.; Pereira, B.; Al-Eryani, G.; Bartonicek, N.; et al. Stromal cell diversity associated with immune evasion in human triple-negative breast cancer. EMBO J. 2020, 39, e104063. [Google Scholar] [CrossRef]
- Liberzon, A.; Birger, C.; Thorvaldsdóttir, H.; Ghandi, M.; Mesirov, J.P.; Tamayo, P. The molecular signatures database (msigdb) hallmark gene set collection. Cell Syst. 2015, 1, 417–425. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef]
- Aran, D.; Hu, Z.; Butte, A.J. Xcell: Digitally portraying the tissue cellular heterogeneity landscape. Genome. Biol. 2017, 18, 220. [Google Scholar] [CrossRef] [PubMed]
- Tokumaru, Y.; Oshi, M.; Murthy, V.; Tian, W.; Yan, L.; Angarita, F.A.; Nagahashi, M.; Matsuhashi, N.; Futamura, M.; Yoshida, K.; et al. Low intratumoral genetic neutrophil-to-lymphocyte ratio (nlr) is associated with favorable tumor immune microenvironment and with survival in triple negative breast cancer (tnbc). Am. J. Cancer Res. 2021, 11, 5743–5755. [Google Scholar] [PubMed]
- Chouliaras, K.; Oshi, M.; Asaoka, M.; Tokumaru, Y.; Khoury, T.; Endo, I.; Ishikawa, T.; Takabe, K. Increased intratumor heterogeneity, angiogenesis and epithelial to mesenchymal transition pathways in metaplastic breast cancer. Am. J. Cancer Res. 2021, 11, 4408–4420. [Google Scholar]
- Le, L.; Tokumaru, Y.; Oshi, M.; Asaoka, M.; Yan, L.; Endo, I.; Ishikawa, T.; Futamura, M.; Yoshida, K.; Takabe, K. Th2 cell infiltrations predict neoadjuvant chemotherapy response of estrogen receptor-positive breast cancer. Gland Surg. 2021, 10, 154–165. [Google Scholar] [CrossRef]
- Oshi, M.; Asaoka, M.; Tokumaru, Y.; Angarita, F.A.; Yan, L.; Matsuyama, R.; Zsiros, E.; Ishikawa, T.; Endo, I.; Takabe, K. Abundance of regulatory t cell (treg) as a predictive biomarker for neoadjuvant chemotherapy in triple-negative breast cancer. Cancers 2020, 12, 3038. [Google Scholar] [CrossRef] [PubMed]
- Thorsson, V.; Gibbs, D.L.; Brown, S.D.; Wolf, D.; Bortone, D.S.; Ou Yang, T.H.; Porta-Pardo, E.; Gao, G.F.; Plaisier, C.L.; Eddy, J.A.; et al. The immune landscape of cancer. Immunity 2018, 48, 812–830. [Google Scholar] [CrossRef] [PubMed]
- Wakiyama, H.; Masuda, T.; Motomura, Y.; Hu, Q.; Tobo, T.; Eguchi, H.; Sakamoto, K.; Hirakawa, M.; Honda, H.; Mimori, K. Cytolytic activity (cyt) score is a prognostic biomarker reflecting host immune status in hepatocellular carcinoma (hcc). Anticancer Res. 2018, 38, 6631–6638. [Google Scholar] [CrossRef]
- Tanyi, J.L.; Morris, A.J.; Wolf, J.K.; Fang, X.; Hasegawa, Y.; Lapushin, R.; Auersperg, N.; Sigal, Y.J.; Newman, R.A.; Felix, E.A.; et al. The human lipid phosphate phosphatase-3 decreases the growth, survival, and tumorigenesis of ovarian cancer cells: Validation of the lysophosphatidic acid signaling cascade as a target for therapy in ovarian cancer. Cancer Res. 2003, 63, 1073–1082. [Google Scholar] [PubMed]
- Escalante-Alcalde, D.; Hernandez, L.; Le Stunff, H.; Maeda, R.; Lee, H.S.; Jr Gang, C.; Sciorra, V.A.; Daar, I.; Spiegel, S.; Morris, A.J.; et al. The lipid phosphatase lpp3 regulates extra-embryonic vasculogenesis and axis patterning. Development 2003, 130, 4623–4637. [Google Scholar] [CrossRef]
- Zhang, N.; Sundberg, J.P.; Gridley, T. Mice mutant for ppap2c, a homolog of the germ cell migration regulator wunen, are viable and fertile. Genesis 2000, 27, 137–140. [Google Scholar] [CrossRef]
- Meng, G.; Tang, X.; Yang, Z.; Zhao, Y.; Curtis, J.M.; McMullen, T.P.W.; Brindley, D.N. Dexamethasone decreases the autotaxin-lysophosphatidate-inflammatory axis in adipose tissue: Implications for the metabolic syndrome and breast cancer. FASEB J. 2019, 33, 1899–1910. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benesch, M.G.K.; Wu, R.; Tang, X.; Brindley, D.N.; Ishikawa, T.; Takabe, K. Decreased Lipid Phosphate Phosphatase 1/3 and Increased Lipid Phosphate Phosphatase 2 Expression in the Human Breast Cancer Tumor Microenvironment Promotes Tumor Progression and Immune System Evasion. Cancers 2023, 15, 2299. https://doi.org/10.3390/cancers15082299
Benesch MGK, Wu R, Tang X, Brindley DN, Ishikawa T, Takabe K. Decreased Lipid Phosphate Phosphatase 1/3 and Increased Lipid Phosphate Phosphatase 2 Expression in the Human Breast Cancer Tumor Microenvironment Promotes Tumor Progression and Immune System Evasion. Cancers. 2023; 15(8):2299. https://doi.org/10.3390/cancers15082299
Chicago/Turabian StyleBenesch, Matthew G. K., Rongrong Wu, Xiaoyun Tang, David N. Brindley, Takashi Ishikawa, and Kazuaki Takabe. 2023. "Decreased Lipid Phosphate Phosphatase 1/3 and Increased Lipid Phosphate Phosphatase 2 Expression in the Human Breast Cancer Tumor Microenvironment Promotes Tumor Progression and Immune System Evasion" Cancers 15, no. 8: 2299. https://doi.org/10.3390/cancers15082299
APA StyleBenesch, M. G. K., Wu, R., Tang, X., Brindley, D. N., Ishikawa, T., & Takabe, K. (2023). Decreased Lipid Phosphate Phosphatase 1/3 and Increased Lipid Phosphate Phosphatase 2 Expression in the Human Breast Cancer Tumor Microenvironment Promotes Tumor Progression and Immune System Evasion. Cancers, 15(8), 2299. https://doi.org/10.3390/cancers15082299








