Thymoma and Thymic Carcinoma: Surgical Resection and Multidisciplinary Treatment
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
3. Surgical Treatment for Thymic Tumor
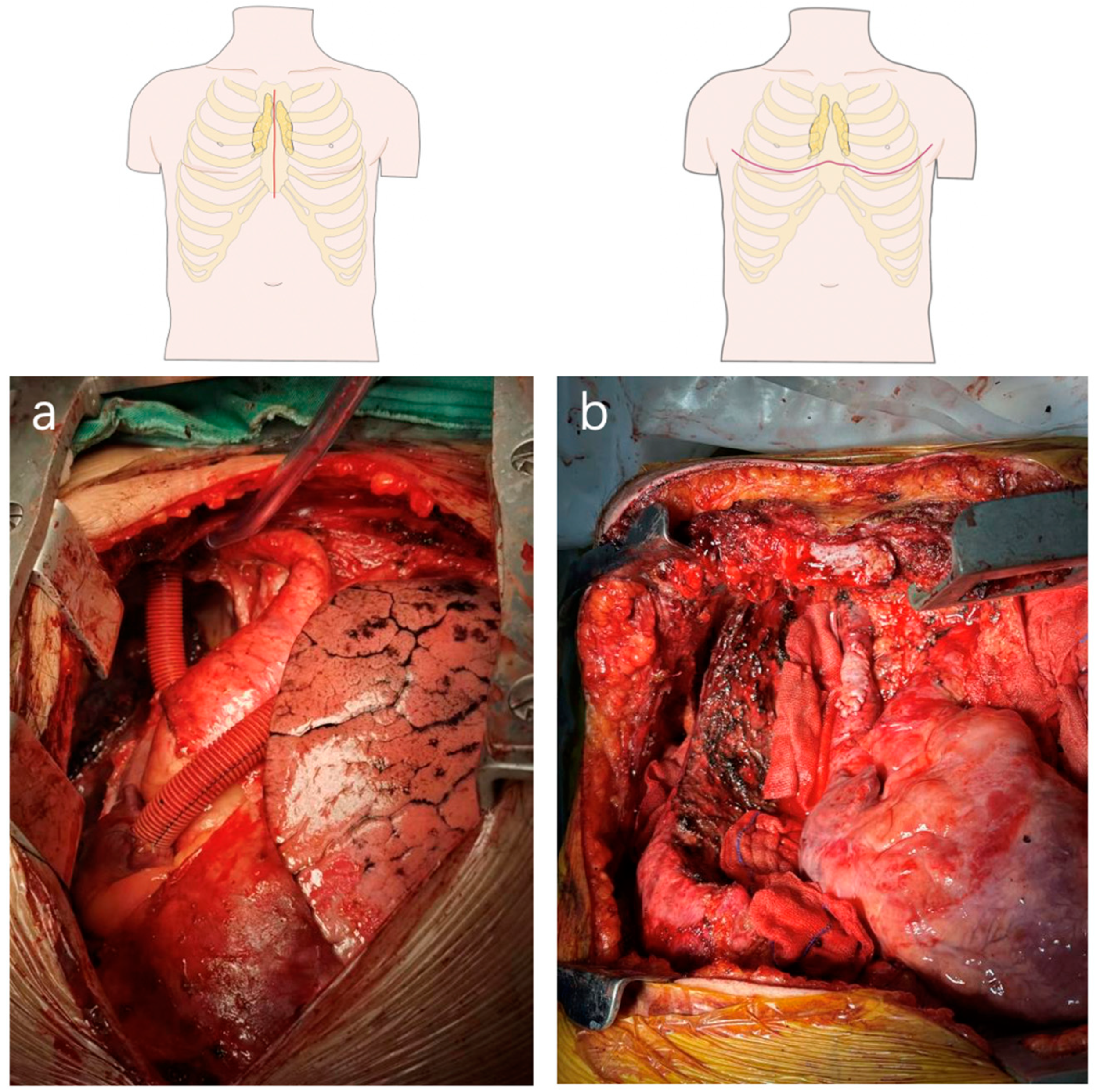
3.1. Open Surgery
Large Vessel Replacement/CPB or ECMO Assisted Surgery (Agg)
- Lung hilum(s) is suspected to be involved;
- SVC replacement at a very high level (almost bifurcation of IJV and RSV);
- Synchronous lung cancer surgery requiring standard mediastinal lymphadenectomy;
- Huge tumor impossible to be exposed through median sternotomy;
- Tumor suspicious of invading sternum;
- History of sternotomy.
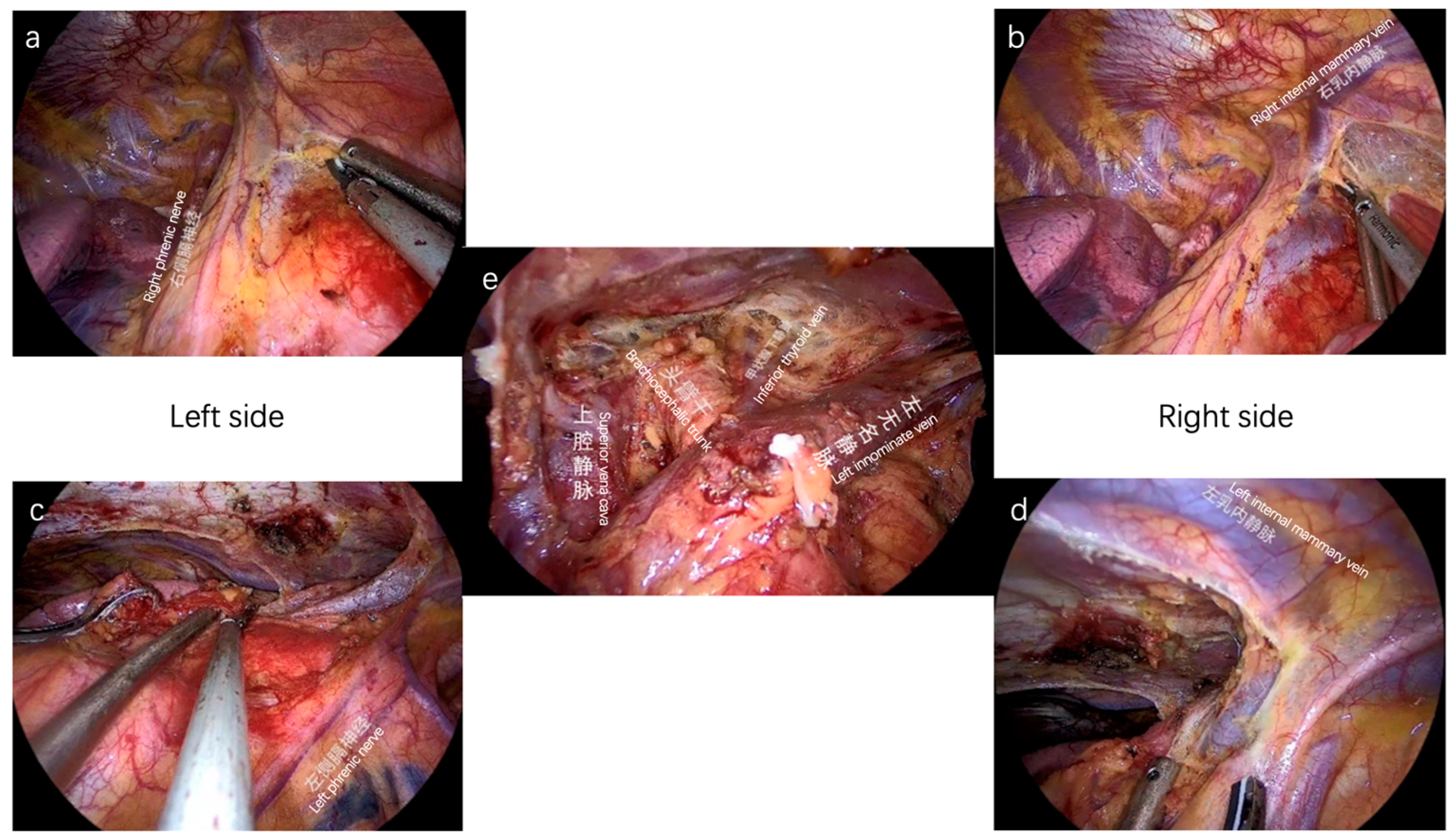
3.2. Minimally Invasive Surgery: New Era and New Challenges
3.2.1. Subxiphoid Approach for Better Exposure
3.2.2. Double Elevation of Sternum for Stage I–III Patients
4. Debatable Issues about Surgery for Thymic Tumors
4.1. Resectability: Where Is the Border?
4.2. Minimally Invasive Surgery for Locally Advanced Thymic Tumors
4.3. Lymphadenectomy in Thymic Tumors
4.4. Debulking Surgery
- Thymoma has a much better prognosis due to its slow growth and low rate of distant metastasis;
- DS may reduce tumor size, reducing the harm caused by later radiation therapy to nearby tissue [60];
- Preference for DS leads to more investigation of potentially resectable thymoma.
4.5. Salvage (Rescue) Surgery (SS)
5. The Role of a Multidisciplinary Team in Surgical Decision Making
5.1. Oncology Therapy
5.2. Radiation Therapy
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Engels, E.A. Epidemiology of thymoma and associated malignancies. J. Thorac. Oncol. 2010, 5, S260–S265. [Google Scholar] [CrossRef]
- Fang, W.; Fu, J.; Shen, Y.; Wei, Y.; Tan, L.; Zhang, P.; Han, Y.; Chen, C.; Zhang, R.; Li, Y.; et al. Management of thymic tumors-consensus based on the Chinese Alliance for Research in Thymomas Multi-institutional retrospective studies. J. Thorac. Dis. 2016, 8, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Shi, J.; Fan, L.; Hu, D.; Yang, J.; Zhao, H. Surgical treatment of thymoma: An 11-year experience with 761 patients. Eur. J. Cardiothorac. Surg. 2016, 49, 1144–1149. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Rizk, N.P.; Travis, W.D.; Riely, G.J.; Park, B.J.; Bains, M.S.; Dycoco, J.; Flores, R.M.; Downey, R.J.; Rusch, V.W. Comparison of patterns of relapse in thymic carcinoma and thymoma. J. Thorac. Cardiovasc. Surg. 2009, 138, 26–31. [Google Scholar] [CrossRef]
- Masaoka, A. Staging system of thymoma. J. Thorac. Oncol. 2010, 5, S304–S312. [Google Scholar] [CrossRef]
- Detterbeck, F.; Youssef, S.; Ruffini, E.; Okumura, M. A review of prognostic factors in thymic malignancies. J. Thorac. Oncol. 2011, 6, S1698–S1704. [Google Scholar] [CrossRef] [PubMed]
- Detterbeck, F.C.; Zeeshan, A. Thymoma: Current diagnosis and treatment. Chin. Med. J. 2013, 126, 2186–2191. [Google Scholar]
- Conforti, F.; Pala, L.; Giaccone, G.; De Pas, T. Thymic epithelial tumors: From biology to treatment. Cancer Treat. Rev. 2020, 86, 102014. [Google Scholar] [CrossRef]
- Rajan, A.; Zhao, C. Deciphering the biology of thymic epithelial tumors. Mediastinum 2019, 3, 36. [Google Scholar] [CrossRef]
- Detterbeck, F.C.; Parsons, A.M. Thymic tumors. Ann. Thorac. Surg. 2004, 77, 1860–1869. [Google Scholar] [CrossRef]
- Litvak, A.M.; Woo, K.; Hayes, S.; Huang, J.; Rimner, A.; Sima, C.S.; Moreira, A.L.; Tsukazan, M.; Riely, G.J. Clinical characteristics and outcomes for patients with thymic carcinoma: Evaluation of Masaoka staging. J. Thorac. Oncol. 2014, 9, 1810–1815. [Google Scholar] [CrossRef] [PubMed]
- Marx, A.; Chan, J.K.C.; Chalabreysse, L.; Dacic, S.; Detterbeck, F.; French, C.A.; Hornick, J.L.; Inagaki, H.; Jain, D.; Lazar, A.J.; et al. The 2021 WHO Classification of Tumors of the Thymus and Mediastinum: What Is New in Thymic Epithelial, Germ Cell, and Mesenchymal Tumors? J. Thorac. Oncol. 2022, 17, 200–213. [Google Scholar] [CrossRef]
- Detterbeck, F.C.; Nicholson, A.G.; Kondo, K.; Van Schil, P.; Moran, C. The Masaoka-Koga stage classification for thymic malignancies: Clarification and definition of terms. J. Thorac. Oncol. 2011, 6, S1710–S1716. [Google Scholar] [CrossRef]
- Di Crescenzo, V.G.; Napolitano, F.; Panico, C.; Di Crescenzo, R.M.; Zeppa, P.; Vatrella, A.; Laperuta, P. Surgical approach in thymectomy: Our experience and review of the literature. Int. J. Surg. Case Rep. 2017, 39, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C.; Frih, H.; Pasquet, F.; Kerever, S.; Jamilloux, Y.; Tronc, F.; Guibert, B.; Isaac, S.; Devouassoux, M.; Chalabreysse, L.; et al. Thymoma associated with autoimmune diseases: 85 cases and literature review. Autoimmun. Rev. 2016, 15, 82–92. [Google Scholar] [CrossRef]
- Gilhus, N.E. Myasthenia Gravis. N. Engl. J. Med. 2016, 375, 2570–2581. [Google Scholar] [CrossRef]
- Aprile, V.; Korasidis, S.; Bacchin, D.; Petralli, G.; Petrini, I.; Ricciardi, R.; Ambrogi, M.C.; Lucchi, M. Thymectomy in Myasthenic Patients with Thymoma: Killing Two Birds with One Stone. Ann. Thorac. Surg. 2021, 112, 1782–1789. [Google Scholar] [CrossRef]
- Roden, A.C.; Ahmad, U.; Cardillo, G.; Girard, N.; Jain, D.; Marom, E.M.; Marx, A.; Moreira, A.L.; Nicholson, A.G.; Rajan, A.; et al. Thymic Carcinomas-A Concise Multidisciplinary Update on Recent Developments From the Thymic Carcinoma Working Group of the International Thymic Malignancy Interest Group. J. Thorac. Oncol. 2022, 17, 637–650. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Simon-Keller, K.; Belharazem-Vitacolonnna, D.; Bohnenberger, H.; Kriegsmann, M.; Kriegsmann, K.; Hamilton, G.; Graeter, T.; Preissler, G.; Ott, G.; et al. A Tuft Cell-Like Signature Is Highly Prevalent in Thymic Squamous Cell Carcinoma and Delineates New Molecular Subsets Among the Major Lung Cancer Histotypes. J. Thorac. Oncol. 2021, 16, 1003–1016. [Google Scholar] [CrossRef]
- Ruffini, E.; Filosso, P.L.; Guerrera, F.; Lausi, P.; Lyberis, P.; Oliaro, A. Optimal surgical approach to thymic malignancies: New trends challenging old dogmas. Lung Cancer 2018, 118, 161–170. [Google Scholar] [CrossRef]
- Friedant, A.J.; Handorf, E.A.; Su, S.; Scott, W.J. Minimally Invasive versus Open Thymectomy for Thymic Malignancies: Systematic Review and Meta-Analysis. J. Thorac. Oncol. 2016, 11, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Chiappetta, M.; Grossi, U.; Sperduti, I.; Margaritora, S.; Marulli, G.; Fiorelli, A.; Sandri, A.; Mizuno, T.; Cusumano, G.; Hamaji, M.; et al. Which Is the Best Treatment in Recurrent Thymoma? A Systematic Review and Meta-Analysis. Cancers 2021, 13, 1559. [Google Scholar] [CrossRef]
- Kondo, K.; Monden, Y. Therapy for thymic epithelial tumors: A clinical study of 1320 patients from Japan. Ann. Thorac. Surg. 2003, 76, 878–884, discussion 884–875. [Google Scholar] [CrossRef] [PubMed]
- Hamaji, M.; Allen, M.S.; Cassivi, S.D.; Nichols, F.C., 3rd; Wigle, D.A.; Deschamps, C.; Shen, K.R. The role of surgical management in recurrent thymic tumors. Ann. Thorac. Surg. 2012, 94, 247–254, discussion 254. [Google Scholar] [CrossRef] [PubMed]
- Agatsuma, H.; Yoshida, K.; Yoshino, I.; Okumura, M.; Higashiyama, M.; Suzuki, K.; Tsuchida, M.; Usuda, J.; Niwa, H. Video-Assisted Thoracic Surgery Thymectomy Versus Sternotomy Thymectomy in Patients With Thymoma. Ann. Thorac. Surg. 2017, 104, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, K.E.; Kreaden, U.S.; Hebert, A.E.; Eaton, D.; Redmond, K.C. A systematic review of robotic versus open and video assisted thoracoscopic surgery (VATS) approaches for thymectomy. Ann. Cardiothorac. Surg. 2019, 8, 174–193. [Google Scholar] [CrossRef]
- Rea, F.; Marulli, G.; Girardi, R.; Bortolotti, L.; Favaretto, A.; Galligioni, A.; Sartori, F. Long-term survival and prognostic factors in thymic epithelial tumours. Eur. J. Cardiothorac. Surg. 2004, 26, 412–418. [Google Scholar] [CrossRef]
- Yuan, Z.Y.; Gao, S.G.; Mu, J.W.; Xue, Q.; Mao, Y.S.; Wang, D.L.; Zhao, J.; Gao, Y.S.; Huang, J.F.; He, J. Long-term outcomes of 307 patients after complete thymoma resection. Chin. J. Cancer 2017, 36, 46. [Google Scholar] [CrossRef]
- Tateishi, Y.; Horita, N.; Namkoong, H.; Enomoto, T.; Takeda, A.; Kaneko, T. Postoperative Radiotherapy for Completely Resected Masaoka/Masaoka-Koga Stage II/III Thymoma Improves Overall Survival: An Updated Meta-Analysis of 4746 Patients. J. Thorac. Oncol. 2021, 16, 677–685. [Google Scholar] [CrossRef]
- Li, F.; Ismail, M.; Elsner, A.; Uluk, D.; Bauer, G.; Meisel, A.; Rueckert, J.C. Surgical Techniques for Myasthenia Gravis: Robotic-Assisted Thoracoscopic Surgery. Thorac. Surg. Clin. 2019, 29, 177–186. [Google Scholar] [CrossRef]
- Sehitogullari, A.; Nasir, A.; Anbar, R.; Erdem, K.; Bilgin, C. Comparison of perioperative outcomes of videothoracoscopy and robotic surgical techniques in thymoma. Asian J. Surg. 2020, 43, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Li, J.; Li, J.; Che, G. Robot-assisted thoracic surgery versus video-assisted thoracic surgery for treatment of patients with thymoma: A systematic review and meta-analysis. Thorac. Cancer 2022, 13, 151–161. [Google Scholar] [CrossRef]
- Toker, A.; Sonett, J.; Zielinski, M.; Rea, F.; Tomulescu, V.; Detterbeck, F.C. Standard terms, definitions, and policies for minimally invasive resection of thymoma. J. Thorac. Oncol. 2011, 6, S1739–S1742. [Google Scholar] [CrossRef]
- Aramini, B.; Fan, J. Technique for Myasthenia Gravis: Subxiphoid Approach. Thorac. Surg. Clin. 2019, 29, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Zielinski, M.; Rybak, M.; Solarczyk-Bombik, K.; Wilkojc, M.; Czajkowski, W.; Kosinski, S.; Fryzlewicz, E.; Nabialek, T.; Pankowski, J. Subxiphoid thymectomy-technical variants. Video-Assist. Thorac. 2017, 2, 9. [Google Scholar] [CrossRef]
- Xu, H.; Liu, D.; Li, Y.; Yang, L.; Wang, F.; Wang, W.; Zhang, L. The Outcomes of Subxiphoid Thoracoscopic Versus Video-Assisted Thoracic Surgery for Thymic Diseases. J. Laparoendosc. Adv. Surg. Tech. 2020, 30, 508–513. [Google Scholar] [CrossRef]
- Zielinski, M.; Czajkowski, W.; Gwozdz, P.; Nabialek, T.; Szlubowski, A.; Pankowski, J. Resection of thymomas with use of the new minimally-invasive technique of extended thymectomy performed through the subxiphoid-right video-thoracoscopic approach with double elevation of the sternum. Eur. J. Cardiothorac. Surg. 2013, 44, e113–e119, discussion e119. [Google Scholar] [CrossRef]
- Pennathur, A.; Qureshi, I.; Schuchert, M.J.; Dhupar, R.; Ferson, P.F.; Gooding, W.E.; Christie, N.A.; Gilbert, S.; Shende, M.; Awais, O.; et al. Comparison of surgical techniques for early-stage thymoma: Feasibility of minimally invasive thymectomy and comparison with open resection. J. Thorac. Cardiovasc. Surg. 2011, 141, 694–701. [Google Scholar] [CrossRef]
- Tseng, Y.C.; Hsieh, C.C.; Huang, H.Y.; Huang, C.S.; Hsu, W.H.; Huang, B.S.; Huang, M.H.; Hsu, H.S. Is thymectomy necessary in nonmyasthenic patients with early thymoma? J. Thorac. Oncol. 2013, 8, 952–958. [Google Scholar] [CrossRef]
- Ye, B.; Tantai, J.C.; Ge, X.X.; Li, W.; Feng, J.; Cheng, M.; Shi, J.X.; Zhao, H. Surgical techniques for early-stage thymoma: Video-assisted thoracoscopic thymectomy versus transsternal thymectomy. J. Thorac. Cardiovasc. Surg. 2014, 147, 1599–1603. [Google Scholar] [CrossRef]
- Tagawa, T.; Yamasaki, N.; Tsuchiya, T.; Miyazaki, T.; Morino, S.; Akamine, S.; Nagayasu, T. Thoracoscopic versus transsternal resection for early stage thymoma: Long-term outcomes. Surg. Today 2014, 44, 2275–2280. [Google Scholar] [CrossRef]
- Liu, T.J.; Lin, M.W.; Hsieh, M.S.; Kao, M.W.; Chen, K.C.; Chang, C.C.; Kuo, S.W.; Huang, P.M.; Hsu, H.H.; Chen, J.S.; et al. Video-assisted thoracoscopic surgical thymectomy to treat early thymoma: A comparison with the conventional transsternal approach. Ann. Surg. Oncol. 2014, 21, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Marulli, G.; Rea, F.; Melfi, F.; Schmid, T.A.; Ismail, M.; Fanucchi, O.; Augustin, F.; Swierzy, M.; Di Chiara, F.; Mussi, A.; et al. Robot-aided thoracoscopic thymectomy for early-stage thymoma: A multicenter European study. J. Thorac. Cardiovasc. Surg. 2012, 144, 1125–1130. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.T.; Mao, T.; Chen, W.H.; Fang, W. Comparison of video-assisted thoracoscopic surgery and median sternotomy approaches for thymic tumor resections at a single institution. Surg. Laparosc. Endosc. Percutan Tech. 2015, 25, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Whitson, B.A.; Andrade, R.S.; Mitiek, M.O.; D’Cunha, J.; Maddaus, M.A. Thoracoscopic thymectomy: Technical pearls to a 21st century approach. J. Thorac. Dis. 2013, 5, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Burt, B.M.; Nguyen, D.; Groth, S.S.; Palivela, N.; Ripley, R.T.; Makris, K.I.; Farjah, F.; Cornwell, L.; Massarweh, N.N. Utilization of Minimally Invasive Thymectomy and Margin-Negative Resection for Early-Stage Thymoma. Ann. Thorac. Surg. 2019, 108, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Aramini, B.; Xu, H.; Fan, J. Thymectomy with angioplasty through a thoracoscopic subxiphoid approach with double elevation of the sternum in Masaoka stage III thymoma. JTCVS Tech. 2021, 8, 208–210. [Google Scholar] [CrossRef]
- Jiang, J.H.; Gao, J.; Zhang, Y.; Wang, H.; Tan, L.J.; Ding, J.Y. Modified Subxiphoid Thoracoscopic Thymectomy for Locally Invasive Thymoma. Ann. Thorac. Surg. 2021, 112, 1095–1100. [Google Scholar] [CrossRef]
- Wang, X.; Aramini, B.; Zhu, Y.; Jiang, G.; Fan, J. Management of bleeding complications during thymectomy by subxiphoid approach with double elevation of the sternum: A case report. Mediastinum 2021, 5, 10. [Google Scholar] [CrossRef]
- Maurizi, G.; Poggi, C.; D’Andrilli, A.; Vanni, C.; Ciccone, A.M.; Ibrahim, M.; Andreetti, C.; Tierno, S.M.; Venuta, F.; Rendina, E.A. Superior Vena Cava Replacement for Thymic Malignancies. Ann. Thorac. Surg. 2019, 107, 386–392. [Google Scholar] [CrossRef]
- Kuno, H.; Funaki, S.; Kimura, K.; Shimamura, K.; Kin, K.; Kuratani, T.; Sawa, Y.; Shintani, Y. Complete resection of local advanced thymic carcinoma with total aortic arch replacement after chemotherapy: A case report. Surg. Case Rep. 2019, 5, 198. [Google Scholar] [CrossRef] [PubMed]
- Hirono, M.; Nonaka, M.; Himuro, N.; Tomita, Y.; Kataoka, D.; Kadokura, M. Two cases of thymoma with pulmonary metastasis: A case report. World J. Surg. Oncol. 2014, 12, 114. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.D. Pleuropneumonectomy for the treatment of Masaoka stage IVA thymoma. Ann. Thorac. Surg. 2006, 82, 1234–1239. [Google Scholar] [CrossRef]
- Aoki, K.; Izumi, Y.; Watanabe, W.; Shimizu, Y.; Osada, H.; Honda, N.; Itoh, T.; Nakayama, M. Generation of ventilation/perfusion ratio map in surgical patients by dual-energy CT after xenon inhalation and intravenous contrast media. J. Cardiothorac. Surg. 2018, 13, 43. [Google Scholar] [CrossRef]
- Falkson, C.B.; Bezjak, A.; Darling, G.; Gregg, R.; Malthaner, R.; Maziak, D.E.; Yu, E.; Smith, C.A.; McNair, S.; Ung, Y.C.; et al. The management of thymoma: A systematic review and practice guideline. J. Thorac. Oncol. 2009, 4, 911–919. [Google Scholar] [CrossRef]
- Hayes, S.A.; Huang, J.; Golia Pernicka, J.; Cunningham, J.; Zheng, J.; Moskowitz, C.S.; Ginsberg, M.S. Radiographic Predictors of Resectability in Thymic Carcinoma. Ann. Thorac. Surg. 2018, 106, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Feng, J.; Ji, C.; Xiang, Y. Minimally invasive thymectomy for locally advanced recurrent thymoma. J. Vis. Surg. 2016, 2, 58. [Google Scholar] [CrossRef]
- Brascia, D.; De Palma, A.; Schiavone, M.; De Iaco, G.; Signore, F.; Panza, T.; Sampietro, D.; Di Milo, G.; Valentini, M.; Pisconti, S.; et al. Lymph Nodes Involvement and Lymphadenectomy in Thymic Tumors: Tentative Answers for Unsolved Questions. Cancers 2021, 13, 85. [Google Scholar] [CrossRef]
- Hamaji, M.; Kojima, F.; Omasa, M.; Sozu, T.; Sato, T.; Chen, F.; Sonobe, M.; Date, H. A meta-analysis of debulking surgery versus surgical biopsy for unresectable thymoma. Eur. J. Cardiothorac. Surg. 2015, 47, 602–607. [Google Scholar] [CrossRef]
- Attaran, S.; McCormack, D.; Pilling, J.; Harrison-Phipps, K. Which stages of thymoma benefit from adjuvant chemotherapy post-thymectomy? Interact. Cardiovasc. Thorac. Surg. 2012, 15, 273–275. [Google Scholar] [CrossRef]
- Petrella, F.; Leo, F.; Veronesi, G.; Solli, P.; Borri, A.; Galetta, D.; Gasparri, R.; Lembo, R.; Radice, D.; Scanagatta, P.; et al. “Salvage” surgery for primary mediastinal malignancies: Is it worthwhile? J. Thorac. Oncol. 2008, 3, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Lucchi, M.; Ambrogi, M.C.; Duranti, L.; Basolo, F.; Fontanini, G.; Angeletti, C.A.; Mussi, A. Advanced stage thymomas and thymic carcinomas: Results of multimodality treatments. Ann. Thorac. Surg. 2005, 79, 1840–1844. [Google Scholar] [CrossRef] [PubMed]
- Girard, N.; Ruffini, E.; Marx, A.; Faivre-Finn, C.; Peters, S.; Committee, E.G. Thymic epithelial tumours: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2015, 26 (Suppl. 5), v40–v55. [Google Scholar] [CrossRef] [PubMed]
- Attaran, S.; Acharya, M.; Anderson, J.R.; Punjabi, P.P. Does surgical debulking for advanced stages of thymoma improve survival? Interact.Cardiovasc. Thorac. Surg. 2012, 15, 494–497. [Google Scholar] [CrossRef] [PubMed]
- Ruffini, E.; Detterbeck, F.; Van Raemdonck, D.; Rocco, G.; Thomas, P.; Weder, W.; Brunelli, A.; Evangelista, A.; Venuta, F.; European Association of Thoracic Surgeons Thymic Working Group. Tumours of the thymus: A cohort study of prognostic factors from the European Society of Thoracic Surgeons database. Eur. J. Cardiothorac. Surg. 2014, 46, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Z.; Chen, Y.; Tan, L.; Zeng, Z.; Ding, J.; Du, S. Induction Strategy for Locally Advanced Thymoma. Front. Oncol. 2021, 11, 704220. [Google Scholar] [CrossRef] [PubMed]
- Padda, S.K.; Riess, J.W.; Schwartz, E.J.; Tian, L.; Kohrt, H.E.; Neal, J.W.; West, R.B.; Wakelee, H.A. Diffuse high intensity PD-L1 staining in thymic epithelial tumors. J. Thorac. Oncol. 2015, 10, 500–508. [Google Scholar] [CrossRef]
- Yokoyama, S.; Miyoshi, H.; Nishi, T.; Hashiguchi, T.; Mitsuoka, M.; Takamori, S.; Akagi, Y.; Kakuma, T.; Ohshima, K. Clinicopathologic and Prognostic Implications of Programmed Death Ligand 1 Expression in Thymoma. Ann. Thorac. Surg. 2016, 101, 1361–1369. [Google Scholar] [CrossRef]
- Katsuya, Y.; Fujita, Y.; Horinouchi, H.; Ohe, Y.; Watanabe, S.; Tsuta, K. Immunohistochemical status of PD-L1 in thymoma and thymic carcinoma. Lung Cancer 2015, 88, 154–159. [Google Scholar] [CrossRef]
- He, Y.; Ramesh, A.; Gusev, Y.; Bhuvaneshwar, K.; Giaccone, G. Molecular predictors of response to pembrolizumab in thymic carcinoma. Cell Rep. Med. 2021, 2, 100392. [Google Scholar] [CrossRef]
- Cho, J.; Kim, H.S.; Ku, B.M.; Choi, Y.L.; Cristescu, R.; Han, J.; Sun, J.M.; Lee, S.H.; Ahn, J.S.; Park, K.; et al. Pembrolizumab for Patients with Refractory or Relapsed Thymic Epithelial Tumor: An Open-Label Phase II Trial. J. Clin. Oncol. 2019, 37, 2162–2170. [Google Scholar] [CrossRef] [PubMed]
- Giaccone, G.; Kim, C.; Thompson, J.; McGuire, C.; Kallakury, B.; Chahine, J.J.; Manning, M.; Mogg, R.; Blumenschein, W.M.; Tan, M.T.; et al. Pembrolizumab in patients with thymic carcinoma: A single-arm, single-centre, phase 2 study. Lancet Oncol. 2018, 19, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Ballman, M.; Zhao, C.; McAdams, M.J.; Rajan, A. Immunotherapy for Management of Thymic Epithelial Tumors: A Double-Edged Sword. Cancers 2022, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Giaccone, G.; Kim, C. Durable Response in Patients with Thymic Carcinoma Treated with Pembrolizumab after Prolonged Follow-Up. J. Thorac. Oncol. 2021, 16, 483–485. [Google Scholar] [CrossRef]
- Ettinger, D.S.; Riely, G.J.; Akerley, W.; Borghaei, H.; Chang, A.C.; Cheney, R.T.; Chirieac, L.R.; D’Amico, T.A.; Demmy, T.L.; Govindan, R.; et al. Thymomas and thymic carcinomas: Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2013, 11, 562–576. [Google Scholar] [CrossRef]
- Sato, J.; Satouchi, M.; Itoh, S.; Okuma, Y.; Niho, S.; Mizugaki, H.; Murakami, H.; Fujisaka, Y.; Kozuki, T.; Nakamura, K.; et al. Lenvatinib in patients with advanced or metastatic thymic carcinoma (REMORA): A multicentre, phase 2 trial. Lancet Oncol. 2020, 21, 843–850. [Google Scholar] [CrossRef]
- Masaoutis, C.; Palamaris, K.; Kokkali, S.; Levidou, G.; Theocharis, S. Unraveling the Immune Microenvironment of Thymic Epithelial Tumors: Implications for Autoimmunity and Treatment. Int. J. Mol. Sci. 2022, 23, 7864. [Google Scholar] [CrossRef]
- Xin, Z.; Lin, M.; Hao, Z.; Chen, D.; Chen, Y.; Chen, X.; Xu, X.; Li, J.; Wu, D.; Chai, Y.; et al. The immune landscape of human thymic epithelial tumors. Nat. Commun. 2022, 13, 5463. [Google Scholar] [CrossRef]
- Nishii, Y.; Furuhashi, K.; Ito, K.; Sakaguchi, T.; Suzuki, Y.; Fujiwara, K.; Yasuma, T.; Kobayashi, T.; D’Alessandro-Gabazza, C.N.; Gabazza, E.C.; et al. Good Response of Advanced Thymic Carcinoma with Low PD-L1 Expression to Chemotherapy plus Pembrolizumab as First-Line Therapy and to Pembrolizumab as Maintenance Therapy: A Case Report. Pharmaceuticals 2022, 15, 889. [Google Scholar] [CrossRef]
- Conforti, F.; Zucali, P.A.; Pala, L.; Catania, C.; Bagnardi, V.; Sala, I.; Della Vigna, P.; Perrino, M.; Zagami, P.; Corti, C.; et al. Avelumab plus axitinib in unresectable or metastatic type B3 thymomas and thymic carcinomas (CAVEATT): A single-arm, multicentre, phase 2 trial. Lancet Oncol. 2022, 23, 1287–1296. [Google Scholar] [CrossRef]
- Remon, J.; Girard, N.; Novello, S.; de Castro, J.; Bigay-Game, L.; Bernabe, R.; Greillier, L.; Mosquera, J.; Cousin, S.; Juan, O.; et al. PECATI: A Multicentric, Open-Label, Single-Arm Phase II Study to Evaluate the Efficacy and Safety of Pembrolizumab and Lenvatinib in Pretreated B3-Thymoma and Thymic Carcinoma Patients. Clin. Lung Cancer 2022, 23, e243–e246. [Google Scholar] [CrossRef] [PubMed]
- Aprile, V.; Bacchin, D.; Korasidis, S.; Nesti, A.; Marrama, E.; Ricciardi, R.; Petrini, I.; Ambrogi, M.C.; Paladini, P.; Lucchi, M. Surgical treatment of pleural recurrence of thymoma: Is hyperthermic intrathoracic chemotherapy worthwhile? Interact. Cardiovasc. Thorac. Surg. 2020, 30, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Gomez, D.; Komaki, R.; Yu, J.; Ikushima, H.; Bezjak, A. Radiation therapy definitions and reporting guidelines for thymic malignancies. J. Thorac. Oncol. 2011, 6, S1743–S1748. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.J.; Kim, H.J.; Wu, H.G. Role of Postoperative Radiotherapy in Nonlocalized Thymoma: Propensity-Matched Analysis of Surveillance, Epidemiology, and End Results Database. J. Thorac. Oncol. 2015, 10, 1357–1363. [Google Scholar] [CrossRef]
- Liu, J.; Govindarajan, A.; Williams, T.M.; Kim, J.; Erhunmwunsee, L.; Raz, D.; Massarelli, E.; Salgia, R.; Chen, Y.J.; Amini, A. An Updated Review on Radiation Treatment Management in Thymus Cancers. Clin. Lung Cancer 2022, 23, 561–570. [Google Scholar] [CrossRef]
- Drevet, G.; Collaud, S.; Tronc, F.; Girard, N.; Maury, J.M. Optimal management of thymic malignancies: Current perspectives. Cancer Manag. Res. 2019, 11, 6803–6814. [Google Scholar] [CrossRef]

| Methods | Pros | Cons |
|---|---|---|
| Unilateral Transthoracic VATS Thymectomy | May be consistent with the surgical habits of thoracic surgeons | Pain, Injury Insufficient exposure |
| Transcervical VATS Thymectomy | Less injury | Difficult to resect completely |
| Conventional subxiphoid VATS Thymectomy | Less pain Better exposure | Insufficient space under the sternum |
| Subxiphoid VATS Thymectomy with Double elevation of sternum | Raising the sternum sufficiently | Require special equipment |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Lin, D.; Aramini, B.; Yang, F.; Chen, X.; Wang, X.; Wu, L.; Huang, W.; Fan, J. Thymoma and Thymic Carcinoma: Surgical Resection and Multidisciplinary Treatment. Cancers 2023, 15, 1953. https://doi.org/10.3390/cancers15071953
Zhang Y, Lin D, Aramini B, Yang F, Chen X, Wang X, Wu L, Huang W, Fan J. Thymoma and Thymic Carcinoma: Surgical Resection and Multidisciplinary Treatment. Cancers. 2023; 15(7):1953. https://doi.org/10.3390/cancers15071953
Chicago/Turabian StyleZhang, Yue, Dong Lin, Beatrice Aramini, Fu Yang, Xi Chen, Xing Wang, Liang Wu, Wei Huang, and Jiang Fan. 2023. "Thymoma and Thymic Carcinoma: Surgical Resection and Multidisciplinary Treatment" Cancers 15, no. 7: 1953. https://doi.org/10.3390/cancers15071953
APA StyleZhang, Y., Lin, D., Aramini, B., Yang, F., Chen, X., Wang, X., Wu, L., Huang, W., & Fan, J. (2023). Thymoma and Thymic Carcinoma: Surgical Resection and Multidisciplinary Treatment. Cancers, 15(7), 1953. https://doi.org/10.3390/cancers15071953







