Artificial Diets with Selective Restriction of Amino Acids and Very Low Levels of Lipids Induce Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Drugs and Reagents
2.2. Cell Culture
2.3. In Vitro Experiments
2.4. Animals
2.5. In Vivo Cancer Models
2.6. Artificial Diet Preparation and Composition
2.7. Statistical Analysis
3. Results
3.1. Amino Acid Restriction Induces Selective Anticancer Activity in Breast Cancer Cells In Vitro

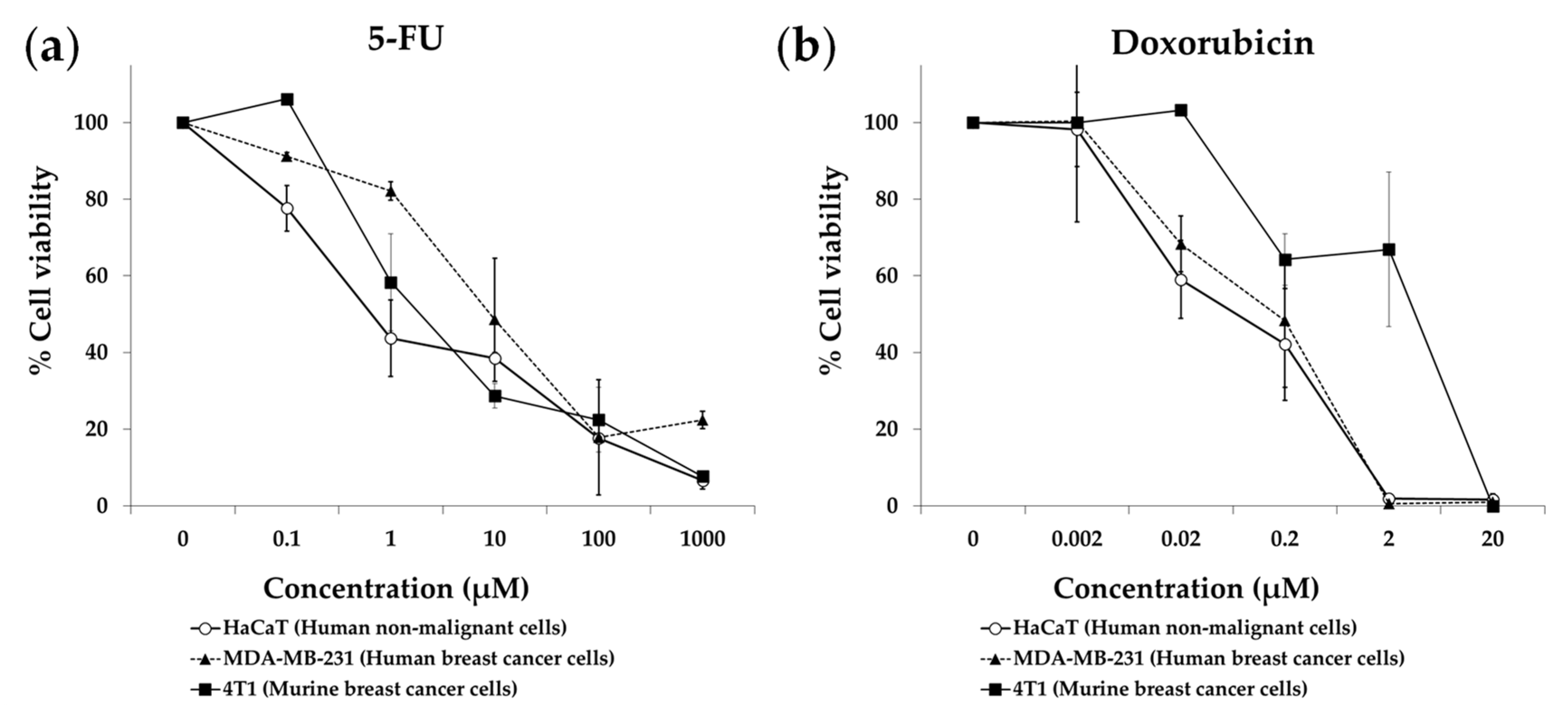
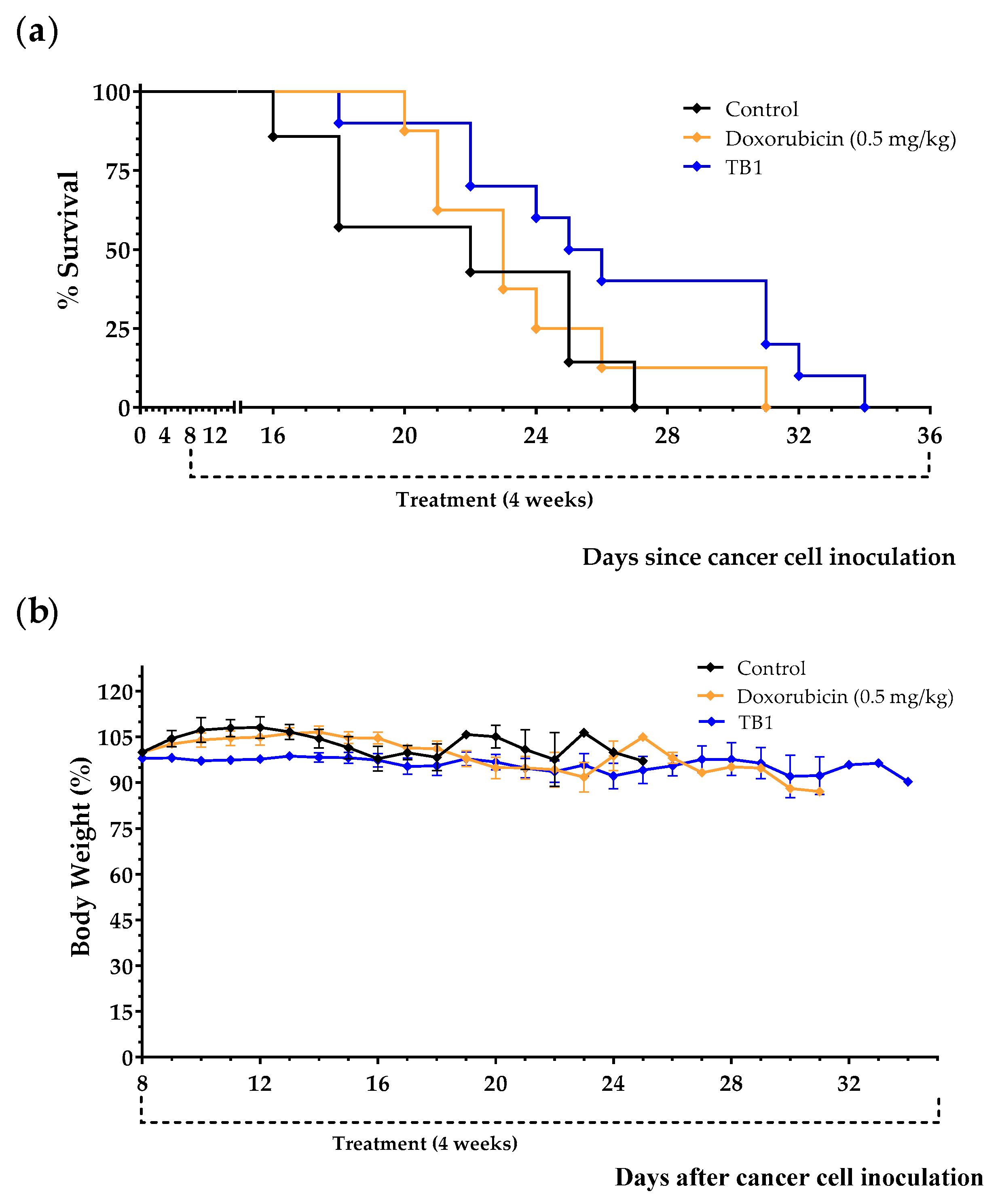
3.2. Anticancer Activity of an Artificial Diet Lacking 10 NEAAs in Mice with Metastatic Triple-Negative Breast Cancer
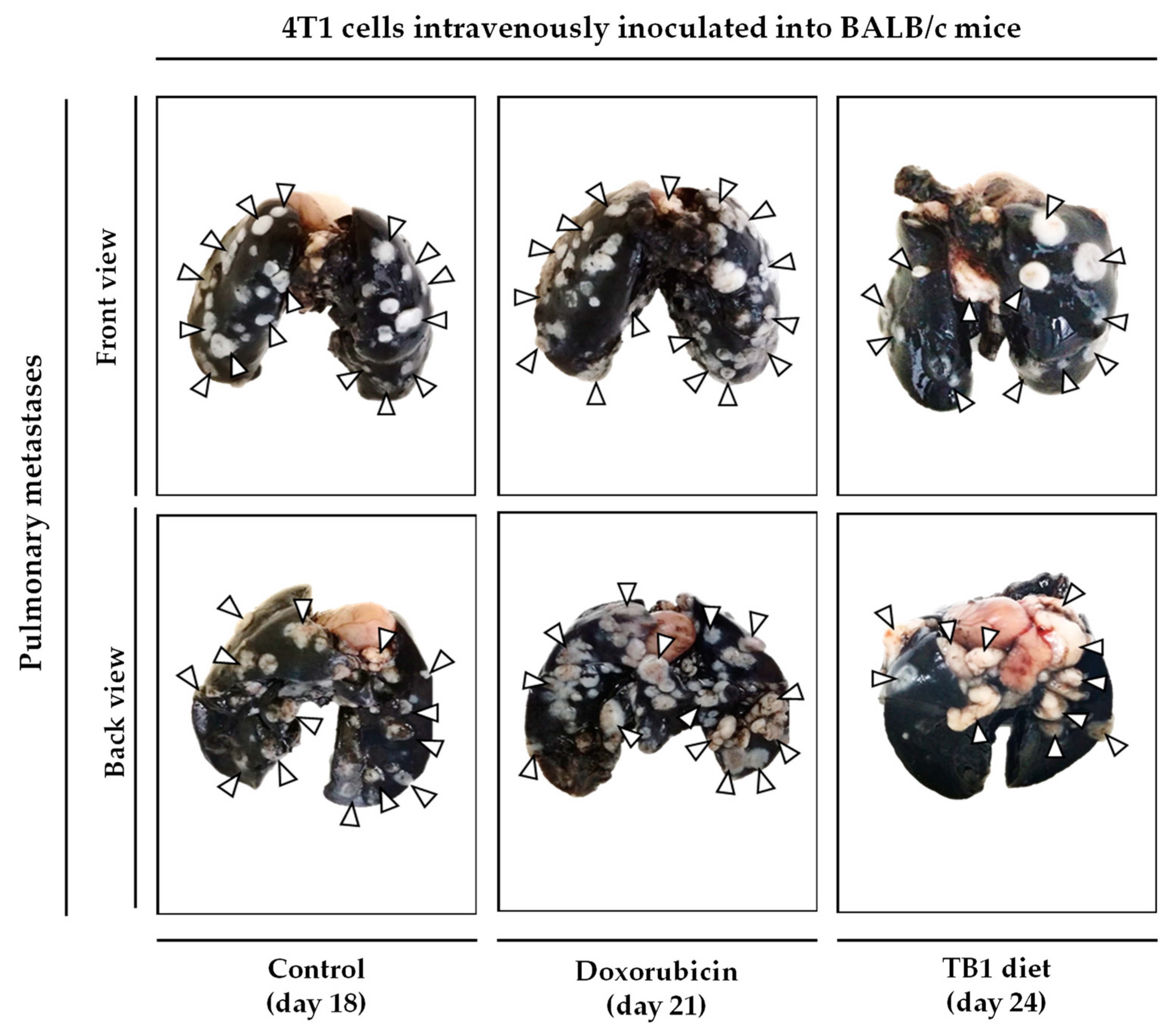
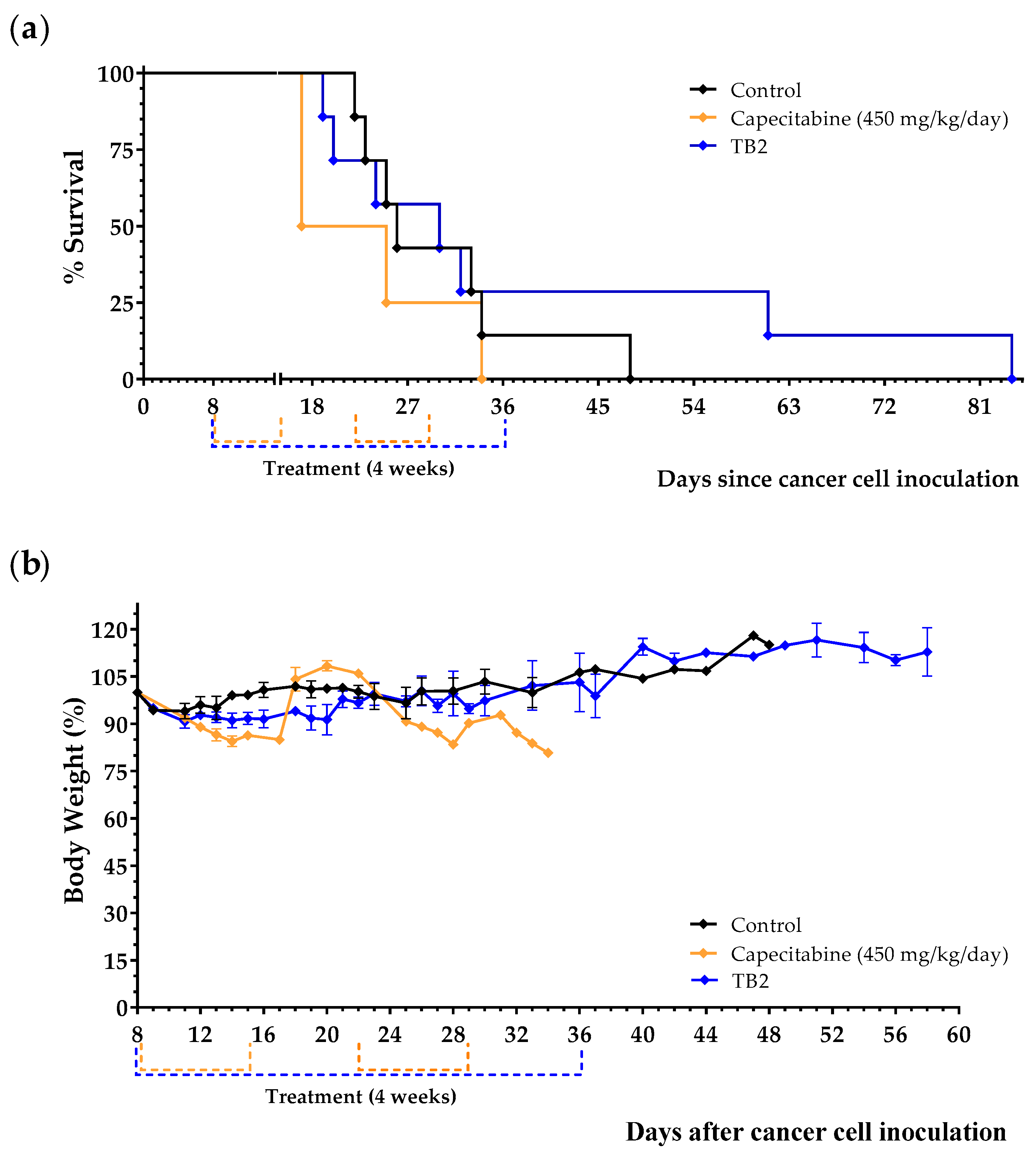
3.3. An Artificial Diet without 10 NEAAs and with 1% Lipids Induces Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer
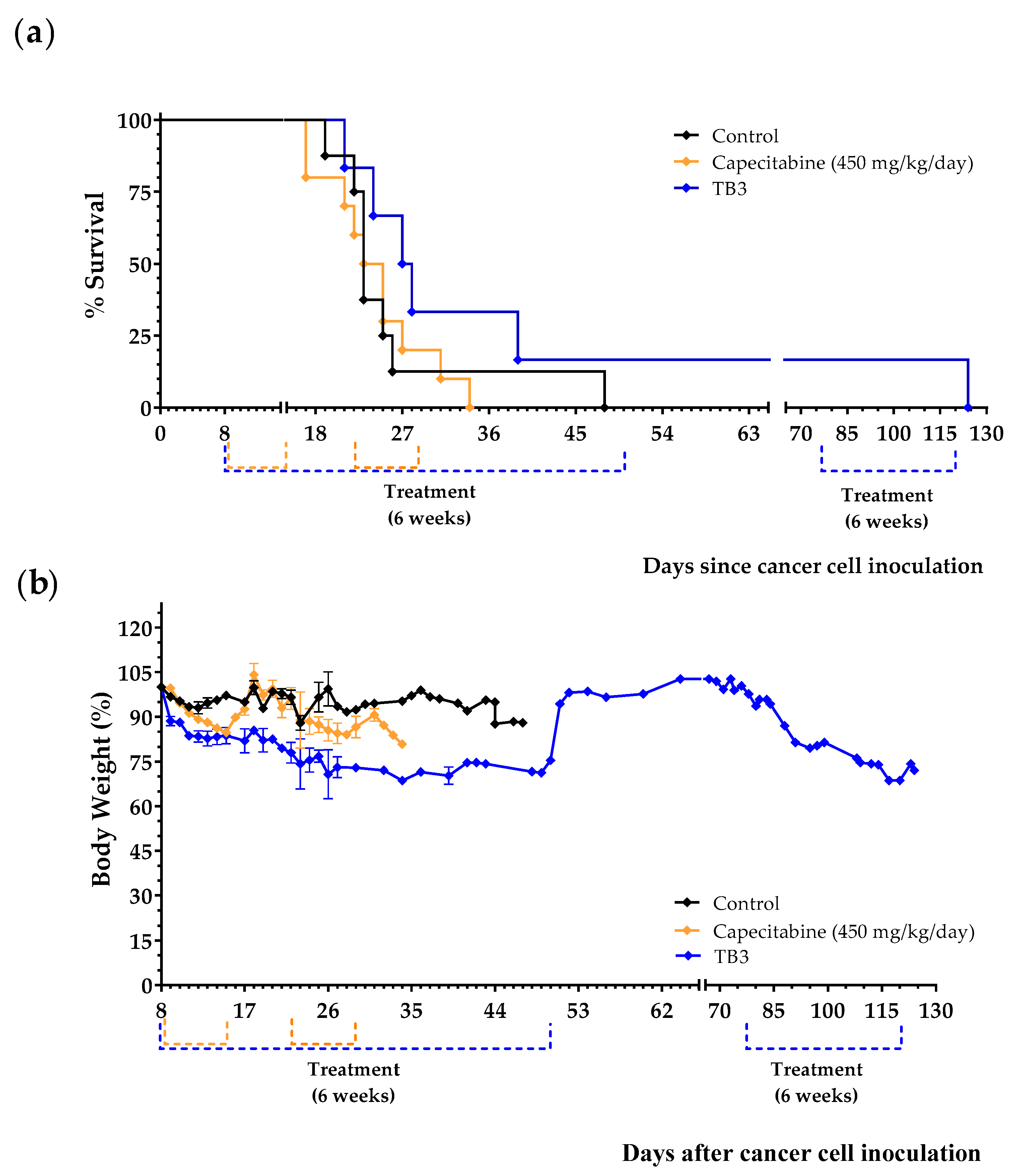
3.4. An Artificial Diet without 10 NEAAs, with Reduced Levels of EAAs, and with 1% Lipids (Diet TB3) Induces Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer
3.5. Diet TB3 Induces Anticancer Activity in Mice with Other Types of Metastatic Cancers
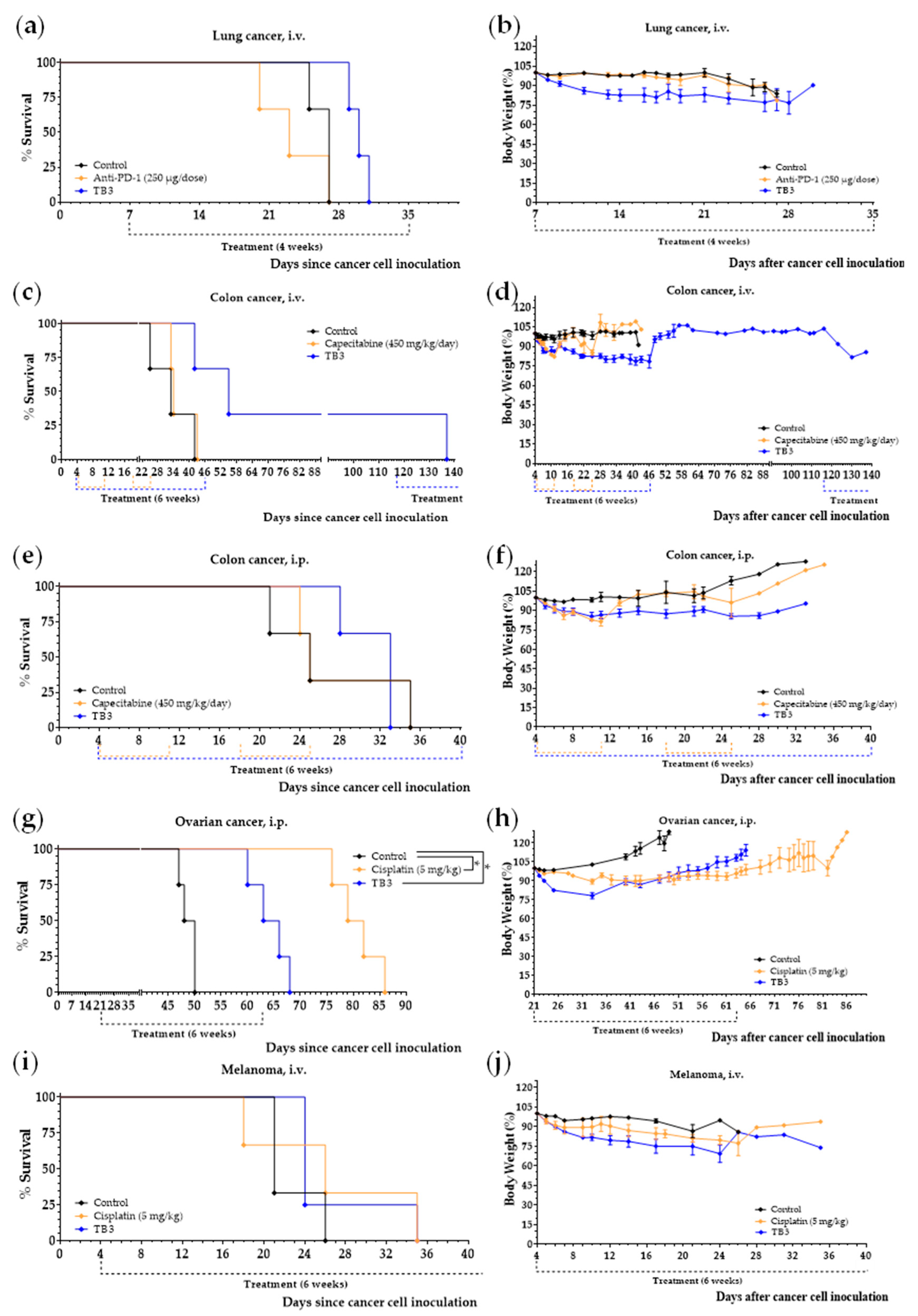

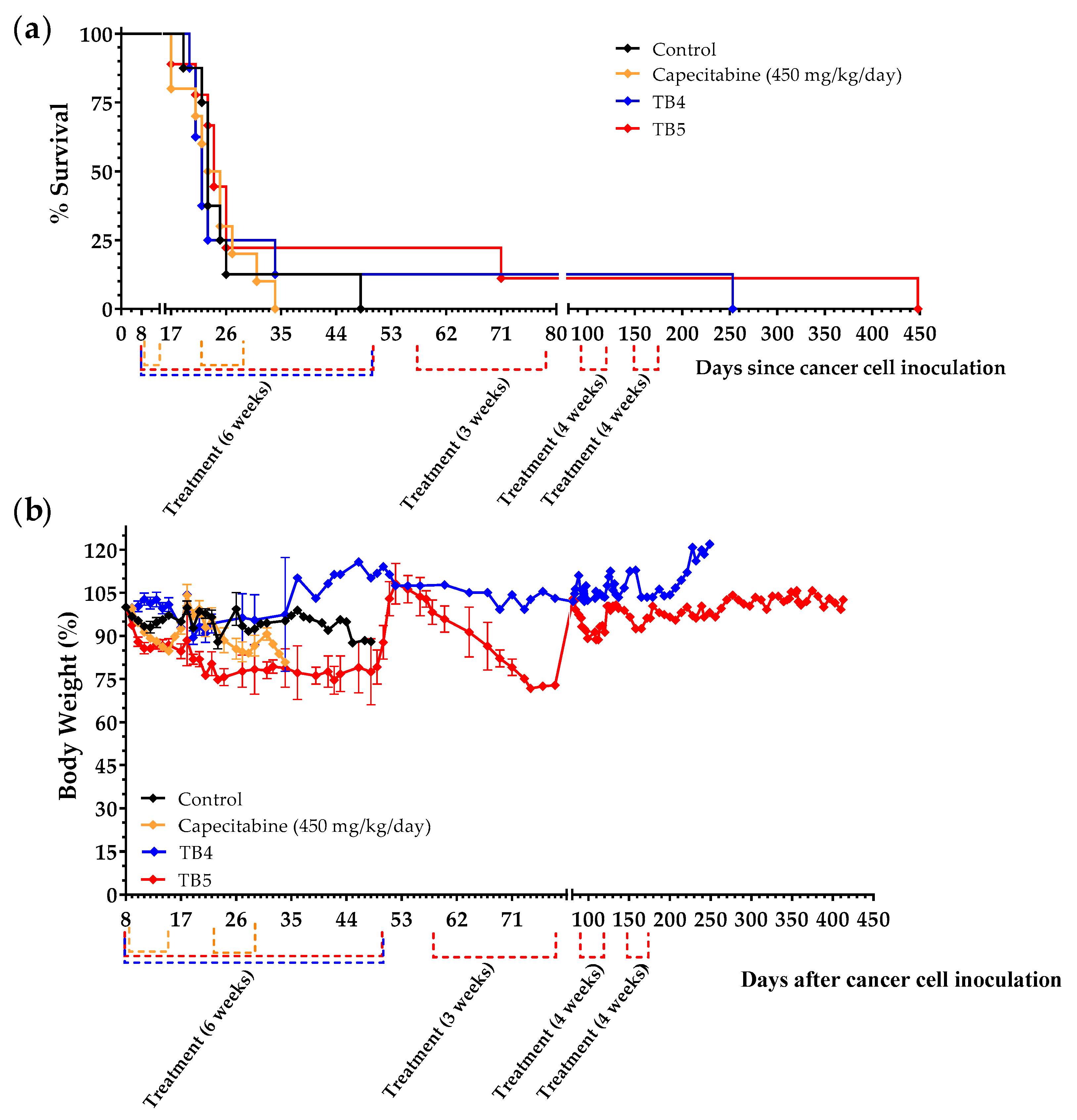
3.6. Diets TB4 and TB5 (Artificial Diets with 6% Casein and 1% Lipids) Induce Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. Surveillance, Epidemiology, and End Results Program-Breast Cancer Survival Statistics. 2022. Available online: https://seer.cancer.gov/statistics-network/explorer/application.html?site=55&data_type=4&graph_type=5&compareBy=stage&chk_stage_104=104&chk_stage_105=105&chk_stage_106=106&chk_stage_107=107&series=9&sex=3&race=1&age_range=1&advopt_precision=1&advopt_show_ (accessed on 14 October 2022).
- Li, X.; Yang, J.; Peng, L.; Sahin, A.A.; Huo, L.; Ward, K.C.; O’Regan, R.; Torres, M.A.; Meisel, J.L. Triple-Negative Breast Cancer Has Worse Overall Survival and Cause-Specific Survival than Non-Triple-Negative Breast Cancer. Breast Cancer Res. Treat. 2017, 161, 279–287. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. NCCN Guidelines Version 4.2022 Breast Cancer. 2022. Available online: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1419 (accessed on 14 October 2022).
- Tutt, A.N.J.; Garber, J.E.; Kaufman, B.; Viale, G.; Fumagalli, D.; Rastogi, P.; Gelber, R.D.; de Azambuja, E.; Fielding, A.; Balmaña, J.; et al. Adjuvant Olaparib for Patients with BRCA1-or BRCA2-Mutated Breast Cancer. N. Engl. J. Med. 2021, 384, 2394–2405. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Angulo, A.M.; Timms, K.M.; Liu, S.; Chen, H.; Litton, J.K.; Potter, J.; Lanchbury, J.S.; Stemke-Hale, K.; Hennessy, B.T.; Arun, B.K.; et al. Incidence and Outcome of BRCA Mutations in Unselected Patients with Triple Receptor-Negative Breast Cancer. Clin. Cancer Res. 2011, 17, 1082–1089. [Google Scholar] [CrossRef]
- Cortes, J.; Rugo, H.S.; Cescon, D.W.; Im, S.-A.; Yusof, M.M.; Gallardo, C.; Lipatov, O.; Barrios, C.H.; Perez-Garcia, J.; Iwata, H.; et al. Pembrolizumab plus Chemotherapy in Advanced Triple-Negative Breast Cancer. N. Engl. J. Med. 2022, 387, 217–226. [Google Scholar] [CrossRef]
- Gong, Y.; Ji, P.; Yang, Y.S.; Xie, S.; Yu, T.J.; Xiao, Y.; Jin, M.L.; Ma, D.; Guo, L.W.; Pei, Y.C.; et al. Metabolic-Pathway-Based Subtyping of Triple-Negative Breast Cancer Reveals Potential Therapeutic Targets. Cell Metab. 2021, 33, 51–64.e9. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, Q.; Dong, C. Metabolic Reprogramming in Triple-Negative Breast Cancer. Cancer Biol. Med. 2020, 17, 44–59. [Google Scholar] [CrossRef]
- Sun, X.; Wang, M.; Wang, M.; Yu, X.; Guo, J.; Sun, T.; Li, X.; Yao, L.; Dong, H.; Xu, Y. Metabolic Reprogramming in Triple-Negative Breast Cancer. Front. Oncol. 2020, 10, 428. [Google Scholar] [CrossRef]
- Martínez-Reyes, I.; Chandel, N.S. Cancer Metabolism: Looking Forward. Nat. Rev. Cancer 2021, 21, 669–680. [Google Scholar] [CrossRef]
- Tajan, M.; Vousden, K.H. Dietary Approaches to Cancer Therapy. Cancer Cell 2020, 37, 767–785. [Google Scholar] [CrossRef]
- Timmerman, L.A.; Holton, T.; Yuneva, M.; Louie, R.J.; Padró, M.; Daemen, A.; Hu, M.; Chan, D.A.; Ethier, S.P.; van’t Veer, L.J.; et al. Glutamine Sensitivity Analysis Identifies the XCT Antiporter as a Common Triple-Negative Breast Tumor Therapeutic Target. Cancer Cell 2013, 24, 450–465. [Google Scholar] [CrossRef]
- Lanzardo, S.; Conti, L.; Rooke, R.; Ruiu, R.; Accart, N.; Bolli, E.; Arigoni, M.; Macagno, M.; Barrera, G.; Pizzimenti, S.; et al. Immunotargeting of Antigen XCT Attenuates Stem-like Cell Behavior and Metastatic Progression in Breast Cancer. Cancer Res. 2016, 76, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Gross, M.I.; Demo, S.D.; Dennison, J.B.; Chen, L.; Chernov-Rogan, T.; Goyal, B.; Janes, J.R.; Laidig, G.J.; Lewis, E.R.; Li, J.; et al. Antitumor Activity of the Glutaminase Inhibitor CB-839 in Triple-Negative Breast Cancer. Mol. Cancer Ther. 2014, 13, 890–901. [Google Scholar] [CrossRef] [PubMed]
- Scott, G.K.; Yau, C.; Becker, B.C.; Khateeb, S.; Mahoney, S.; Jensen, M.B.; Hann, B.; Cowen, B.J.; Pegan, S.D.; Benz, C.C. Targeting Mitochondrial Proline Dehydrogenase with a Suicide Inhibitor to Exploit Synthetic Lethal Interactions with P53 Upregulation and Glutaminase Inhibition. Mol. Cancer Ther. 2019, 18, 1374–1385. [Google Scholar] [CrossRef] [PubMed]
- Elia, I.; Broekaert, D.; Christen, S.; Boon, R.; Radaelli, E.; Orth, M.F.; Verfaillie, C.; Grünewald, T.G.P.; Fendt, S.M. Proline Metabolism Supports Metastasis Formation and Could Be Inhibited to Selectively Target Metastasizing Cancer Cells. Nat. Commun. 2017, 8, 15267. [Google Scholar] [CrossRef]
- Sun, J.; Nagel, R.; Zaal, E.A.; Ugalde, A.P.; Han, R.; Proost, N.; Song, J.; Pataskar, A.; Burylo, A.; Fu, H.; et al. SLC 1A3 Contributes to L-asparaginase Resistance in Solid Tumors. EMBO J. 2019, 38, e102147. [Google Scholar] [CrossRef]
- Knott, S.R.V.; Wagenblast, E.; Khan, S.; Kim, S.Y.; Soto, M.; Wagner, M.; Turgeon, M.O.; Fish, L.; Erard, N.; Gable, A.L.; et al. Asparagine Bioavailability Governs Metastasis in a Model of Breast Cancer. Nature 2018, 554, 378–381. [Google Scholar] [CrossRef]
- Krall, A.S.; Mullen, P.J.; Surjono, F.; Momcilovic, M.; Schmid, E.W.; Halbrook, C.J.; Thambundit, A.; Mittelman, S.D.; Lyssiotis, C.A.; Shackelford, D.B.; et al. Asparagine Couples Mitochondrial Respiration to ATF4 Activity and Tumor Growth. Cell Metab. 2021, 33, 1013–1026.e6. [Google Scholar] [CrossRef] [PubMed]
- Qiu, F.; Chen, Y.R.; Liu, X.; Chu, C.Y.; Shen, L.J.; Xu, J.; Gaur, S.; Forman, H.J.; Zhang, H.; Zheng, S.; et al. Cancer: Arginine Starvation Impairs Mitochondrial Respiratory Function in ASS1-Deficient Breast Cancer Cells. Sci. Signal. 2014, 7, ra31. [Google Scholar] [CrossRef]
- Cramer, S.L.; Saha, A.; Liu, J.; Tadi, S.; Tiziani, S.; Yan, W.; Triplett, K.; Lamb, C.; Alters, S.E.; Rowlinson, S.; et al. Systemic Depletion of L-Cyst(e)Ine with Cyst(e)Inase Increases Reactive Oxygen Species and Suppresses Tumor Growth. Nat. Med. 2017, 23, 120–127. [Google Scholar] [CrossRef]
- Yao, S.; Janku, F.; Koenig, K.; Tsimberidou, A.M.; Piha-Paul, S.A.; Shi, N.; Stewart, J.; Johnston, A.; Bomalaski, J.; Meric-Bernstam, F.; et al. Phase 1 Trial of ADI-PEG 20 and Liposomal Doxorubicin in Patients with Metastatic Solid Tumors. Cancer Med. 2022, 11, 340–347. [Google Scholar] [CrossRef]
- Strekalova, E.; Malin, D.; Good, D.M.; Cryns, V.L. Methionine Deprivation Induces a Targetable Vulnerability in Triple-Negative Breast Cancer Cells by Enhancing TRAIL Receptor-2 Expression. Clin. Cancer Res. 2015, 21, 2780–2791. [Google Scholar] [CrossRef]
- Strekalova, E.; Malin, D.; Rajanala, H.; Cryns, V.L. Preclinical Breast Cancer Models to Investigate Metabolic Priming by Methionine Restriction. Methods Mol. Biol. 2019, 1866, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.; Kim, J.H.; Lee, E.; Jang, Y.J.; Son, J.E.; Kwon, J.Y.; Lim, T.; Kim, S.; Park, J.H.Y.; Kim, J.-E.; et al. Methionine Deprivation Suppresses Triple-Negative Breast Cancer Metastasis in Vitro and in Vivo. Oncotarget 2016, 7, 67223–67234. [Google Scholar] [CrossRef] [PubMed]
- Hens, J.R.; Sinha, I.; Perodin, F.; Cooper, T.; Sinha, R.; Plummer, J.; Perrone, C.E.; Orentreich, D. Methionine-Restricted Diet Inhibits Growth of MCF10AT1-Derived Mammary Tumors by Increasing Cell Cycle Inhibitors in Athymic Nude Mice. BMC Cancer 2016, 16, 349. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.-T.T.; Qi, Y.; Wang, Y.-C.C.; Chi, K.K.; Chung, Y.; Ouyang, C.; Chen, Y.-R.Y.-H.Y.H.R.; Oh, M.E.; Sheng, X.; Tang, Y.; et al. Arginine Starvation Kills Tumor Cells through Aspartate Exhaustion and Mitochondrial Dysfunction. Commun. Biol. 2018, 1, 178. [Google Scholar] [CrossRef]
- Sullivan, M.R.; Mattaini, K.R.; Dennstedt, E.A.; Nguyen, A.A.; Sivanand, S.; Reilly, M.F.; Meeth, K.; Muir, A.; Darnell, A.M.; Bosenberg, M.W.; et al. Increased Serine Synthesis Provides an Advantage for Tumors Arising in Tissues Where Serine Levels Are Limiting. Cell Metab. 2019, 29, 1410–1421.e4. [Google Scholar] [CrossRef] [PubMed]
- Broadfield, L.A.; Pane, A.A.; Talebi, A.; Swinnen, J.V.; Fendt, S.M. Lipid Metabolism in Cancer: New Perspectives and Emerging Mechanisms. Dev. Cell 2021, 56, 1363–1393. [Google Scholar] [CrossRef] [PubMed]
- Hoy, A.J.; Nagarajan, S.R.; Butler, L.M. Tumour Fatty Acid Metabolism in the Context of Therapy Resistance and Obesity. Nat. Rev. Cancer 2021, 21, 753–766. [Google Scholar] [CrossRef]
- Wang, D.; Dubois, R.N. Eicosanoids and Cancer. Nat. Rev. Cancer 2010, 10, 181–193. [Google Scholar] [CrossRef]
- Pascual, G.; Avgustinova, A.; Mejetta, S.; Martín, M.; Castellanos, A.; Attolini, C.S.O.; Berenguer, A.; Prats, N.; Toll, A.; Hueto, J.A.; et al. Targeting Metastasis-Initiating Cells through the Fatty Acid Receptor CD36. Nature 2017, 541, 41–45. [Google Scholar] [CrossRef]
- Incio, J.; Tam, J.; Rahbari, N.N.; Suboj, P.; McManus, D.T.; Chin, S.M.; Vardam, T.D.; Batista, A.; Babykutty, S.; Jung, K.; et al. PlGF/VEGFR-1 Signaling Promotes Macrophage Polarization and Accelerated Tumor Progression in Obesity. Clin. Cancer Res. 2016, 22, 2993–3004. [Google Scholar] [CrossRef] [PubMed]
- Incio, J.; Liu, H.; Suboj, P.; Chin, S.M.; Chen, I.X.; Pinter, M.; Ng, M.R.; Nia, H.T.; Grahovac, J.; Kao, S.; et al. Obesity-Induced Inflammation and Desmoplasia Promote Pancreatic Cancer Progression and Resistance to Chemotherapy. Cancer Discov. 2016, 6, 852–869. [Google Scholar] [CrossRef] [PubMed]
- Philip, B.; Roland, C.L.; Daniluk, J.; Liu, Y.; Chatterjee, D.; Gomez, S.B.; Ji, B.; Huang, H.; Wang, H.; Fleming, J.B.; et al. A High-Fat Diet Activates Oncogenic Kras and COX2 to Induce Development of Pancreatic Ductal Adenocarcinoma in Mice. Gastroenterology 2013, 145, 1449–1458. [Google Scholar] [CrossRef] [PubMed]
- Malvi, P.; Chaube, B.; Singh, S.V.; Mohammad, N.; Pandey, V.; Vijayakumar, M.V.; Radhakrishnan, R.M.; Vanuopadath, M.; Nair, S.S.; Nair, B.G.; et al. Weight Control Interventions Improve Therapeutic Efficacy of Dacarbazine in Melanoma by Reversing Obesity-Induced Drug Resistance. Cancer Metab. 2016, 4, 21. [Google Scholar] [CrossRef]
- Xia, S.; Lin, R.; Jin, L.; Zhao, L.; Kang, H.-B.; Pan, Y.; Liu, S.; Qian, G.; Qian, Z.; Konstantakou, E.; et al. Prevention of Dietary-Fat-Fueled Ketogenesis Attenuates BRAF V600E Tumor Growth. Cell Metab. 2017, 25, 358–373. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Vijayakumar, M.V.; Ajay, A.K.; Malvi, P.; Bhat, M.K. Diet-Induced Obesity Increases Melanoma Progression: Involvement of Cav-1 and FASN. Int. J. Cancer 2012, 130, 497–508. [Google Scholar] [CrossRef]
- Labbé, D.P.; Zadra, G.; Yang, M.; Reyes, J.M.; Lin, C.Y.; Cacciatore, S.; Ebot, E.M.; Creech, A.L.; Giunchi, F.; Fiorentino, M.; et al. High-Fat Diet Fuels Prostate Cancer Progression by Rewiring the Metabolome and Amplifying the MYC Program. Nat. Commun. 2019, 10, 4358. [Google Scholar] [CrossRef]
- Tang, F.-Y.; Pai, M.-H.; Chiang, E.-P.I. Consumption of High-Fat Diet Induces Tumor Progression and Epithelial–Mesenchymal Transition of Colorectal Cancer in a Mouse Xenograft Model. J. Nutr. Biochem. 2012, 23, 1302–1313. [Google Scholar] [CrossRef]
- Ngo, T.H.; Barnard, R.J.; Anton, T.; Tran, C.; Elashoff, D.; Heber, D.; Freedland, S.J.; Aronson, W.J. Effect of Isocaloric Low-Fat Diet on Prostate Cancer Xenograft Progression to Androgen Independence. Cancer Res. 2004, 64, 1252–1254. [Google Scholar] [CrossRef]
- Ngo, T.H.; Barnard, R.J.; Cohen, P.; Freedland, S.; Tran, C.; DeGregorio, F.; Elshimali, Y.I.; Heber, D.; Aronson, W.J. Effect of Isocaloric Low-Fat Diet on Human LAPC-4 Prostate Cancer Xenografts in Severe Combined Immunodeficient Mice and the Insulin-like Growth Factor Axis. Clin. Cancer Res. 2003, 9, 2734–2743. [Google Scholar] [PubMed]
- Kobayashi, N.; Barnard, R.J.; Said, J.; Hong-Gonzalez, J.; Corman, D.M.; Ku, M.; Doan, N.B.; Gui, D.; Elashoff, D.; Cohen, P.; et al. Effect of Low-Fat Diet on Development of Prostate Cancer and Akt Phosphorylation in the Hi-Myc Transgenic Mouse Model. Cancer Res. 2008, 68, 3066–3073. [Google Scholar] [CrossRef]
- Sundaram, S.; Yan, L. High-Fat Diet Enhances Mammary Tumorigenesis and Pulmonary Metastasis and Alters Inflammatory and Angiogenic Profiles in MMTV-PyMT Mice. Anticancer Res. 2016, 36, 6279–6287. [Google Scholar] [CrossRef]
- Evangelista, G.C.M.; Salvador, P.A.; Soares, S.M.A.; Barros, L.R.C.; da Xavier, F.H.C.; Abdo, L.M.; Gualberto, A.C.M.; Macedo, G.C.; Clavijo-Salomon, M.A.; Gameiro, J. 4T1 Mammary Carcinoma Colonization of Metastatic Niches Is Accelerated by Obesity. Front. Oncol. 2019, 9, 685. [Google Scholar] [CrossRef] [PubMed]
- Bousquenaud, M.; Fico, F.; Solinas, G.; Rüegg, C.; Santamaria-Martínez, A. Obesity Promotes the Expansion of Metastasis-Initiating Cells in Breast Cancer. Breast Cancer Res. 2018, 20, 104. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Chang, C. High Fat Diet-Induced Breast Cancer Model in Rat. Bio-Protoc 2016, 6, e1852. [Google Scholar] [CrossRef]
- Chlebowski, R.T.; Aragaki, A.K.; Anderson, G.L.; Thomson, C.A.; Manson, J.A.E.; Simon, M.S.; Howard, B.V.; Rohan, T.E.; Snetselar, L.; Lane, D.; et al. Low-Fat Dietary Pattern and Breast Cancer Mortality in the Women’s Health Initiative Randomized Controlled Trial. Proc. J. Clin. Oncol. 2017, 35, 2919–2926. [Google Scholar] [CrossRef]
- Chlebowski, R.T.; Blackburn, G.L.; Thomson, C.A.; Nixon, D.W.; Shapiro, A.; Hoy, M.K.; Goodman, M.T.; Giuliano, A.E.; Karanja, N.; McAndrew, P.; et al. Dietary Fat Reduction and Breast Cancer Outcome: Interim Efficacy Results from the Women’s Intervention Nutrition Study. J. Natl. Cancer Inst. 2006, 98, 1767–1776. [Google Scholar] [CrossRef]
- Jiménez-Alonso, J.; Guillén-Mancina, E.; Calderón-Montaño, J.; Jiménez-González, V.; Díaz-Ortega, P.; Burgos-Morón, E.; López-Lázaro, M. Artificial Diets Based on Selective Amino Acid Restriction versus Capecitabine in Mice with Metastatic Colon Cancer. Nutrients 2022, 14, 3378. [Google Scholar] [CrossRef]
- Calderón-Montaño, J.M.; Guillén-Mancina, E.; Jiménez-Alonso, J.J.; Jiménez-González, V.; Burgos-Morón, E.; Mate, A.; Pérez-Guerrero, M.C.; López-Lázaro, M. Manipulation of Amino Acid Levels with Artificial Diets Induces a Marked Anticancer Activity in Mice with Renal Cell Carcinoma. Int. J. Mol. Sci. 2022, 23, 16132. [Google Scholar] [CrossRef]
- Boukamp, P.; Petrussevska, R.T.; Breitkreutz, D.; Hornung, J.; Markham, A.; Fusenig, N.E. Normal Keratinization in a Spontaneously Immortalized Aneuploid Human Keratinocyte Cell Line. J. Cell Biol. 1988, 106, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Walton, J.; Blagih, J.; Ennis, D.; Leung, E.; Dowson, S.; Farquharson, M.; Tookman, L.A.; Orange, C.; Athineos, D.; Mason, S.; et al. CRISPR/Cas9-Mediated Trp53 and Brca2 Knockout to Generate Improved Murine Models of Ovarian High-Grade Serous Carcinoma. Cancer Res. 2016, 76, 6118–6129. [Google Scholar] [CrossRef] [PubMed]
- Aslakson, C.J.; Miller2, F.R. Selective Events in the Metastatic Process Defined by Analysis of the Sequential Dissemination of Subpopulations of a Mouse Mammary Tumor. Cancer Res. 1992, 52, 1399–1405. [Google Scholar] [PubMed]
- Liu, Y.; Wang, L.; Liu, J.; Xie, X.; Hu, H.; Luo, F. Anticancer Effects of ACT001 via NF-ΚB Suppression in Murine Triple-Negative Breast Cancer Cell Line 4T1. Cancer Manag. Res. 2020, 12, 5131. [Google Scholar] [CrossRef] [PubMed]
- Corbett, T.H.; Griswold Jr, D.P.; Roberts, B.J.; Peckham, J.C.; Schabel Jr, F.M. Tumor Induction Relationships in Development of Transplantable Cancers of the Colon in Mice for Chemotherapy Assays, with a Note on Carcinogen Structure. Cancer Res. 1975, 35, 2434–2439. [Google Scholar] [PubMed]
- Wang, L.; Hu, X.; Xu, Y.; Liu, Z. Arsenic Trioxide Inhibits Lung Metastasis of Mouse Colon Cancer via Reducing the Infiltration of Regulatory T Cells. Tumour Biol. 2016, 37, 15165–15173. [Google Scholar] [CrossRef]
- Burikhanov, R.; Hebbar, N.; Noothi, S.K.; Shukla, N.; Sledziona, J.; Araujo, N.; Kudrimoti, M.; Wang, Q.J.; Watt, D.S.; Welch, D.R.; et al. Chloroquine-Inducible Par-4 Secretion Is Essential for Tumor Cell Apoptosis and Inhibition of Metastasis. Cell Rep. 2017, 18, 508–519. [Google Scholar] [CrossRef]
- Zhao, M.; Suetsugu, A.; Ma, H.; Zhang, L.; Liu, F.; Zhang, Y.; Tran, B.; Hoffman, R.M. Efficacy against Lung Metastasis with a Tumor-Targeting Mutant of Salmonella Typhimurium in Immunocompetent Mice. Cell Cycle 2012, 11, 187. [Google Scholar] [CrossRef]
- Yan, J.; Wang, Z.Y.; Yang, H.Z.; Liu, H.Z.; Mi, S.; Lv, X.X.; Fu, X.M.; Yan, H.M.; Zhang, X.W.; Zhan, Q.M.; et al. Timing Is Critical for an Effective Anti-Metastatic Immunotherapy: The Decisive Role of IFNγ/STAT1-Mediated Activation of Autophagy. PLoS ONE 2011, 6, e24705. [Google Scholar] [CrossRef]
- Calderón-Montaño, J.M.; Burgos-Morón, E.; López-Lázaro, M. The in Vivo Antitumor Activity of Cardiac Glycosides in Mice Xenografted with Human Cancer Cells Is Probably an Experimental Artifact. Oncogene 2014, 33, 2947–2948. [Google Scholar] [CrossRef]
- Efeyan, A.; Comb, W.C.; Sabatini, D.M. Nutrient-Sensing Mechanisms and Pathways. Nature 2015, 517, 302–310. [Google Scholar] [CrossRef] [PubMed]
- López-Lázaro, M. Selective Amino Acid Restriction Therapy (SAART): A Non- Pharmacological Strategy against All Types of Cancer Cells. Oncoscience 2015, 2, 857. [Google Scholar] [CrossRef] [PubMed]
- Magtanong, L.; Ko, P.J.; To, M.; Cao, J.Y.; Forcina, G.C.; Tarangelo, A.; Ward, C.C.; Cho, K.; Patti, G.J.; Nomura, D.K.; et al. Exogenous Monounsaturated Fatty Acids Promote a Ferroptosis-Resistant Cell State. Cell Chem. Biol. 2019, 26, 420–432.e9. [Google Scholar] [CrossRef]
- Zhao, G.; Tan, Y.; Cardenas, H.; Vayngart, D.; Wang, Y.; Huang, H.; Keathley, R.; Wei, J.J.; Ferreira, C.R.; Orsulic, S.; et al. Ovarian Cancer Cell Fate Regulation by the Dynamics between Saturated and Unsaturated Fatty Acids. Proc. Natl. Acad. Sci. USA 2022, 119, e2203480119. [Google Scholar] [CrossRef] [PubMed]
- Staaf, J.; Glodzik, D.; Bosch, A.; Vallon-Christersson, J.; Reuterswärd, C.; Häkkinen, J.; Degasperi, A.; Amarante, T.D.; Saal, L.H.; Hegardt, C.; et al. Whole-Genome Sequencing of Triple-Negative Breast Cancers in a Population-Based Clinical Study. Nat. Med. 2019, 25, 1526–1533. [Google Scholar] [CrossRef]
- López-Lázaro, M. Two Preclinical Tests to Evaluate Anticancer Activity and to Help Validate Drug Candidates for Clinical Trials. Oncoscience 2015, 2, 91–98. [Google Scholar] [CrossRef] [PubMed]
- López-Lázaro, M. A Simple and Reliable Approach for Assessing Anticancer Activity in Vitro. Curr. Med. Chem. 2015, 22, 1324–1334. [Google Scholar] [CrossRef]
- Lieu, E.L.; Nguyen, T.; Rhyne, S.; Kim, J. Amino Acids in Cancer. Exp. Mol. Med. 2020, 52, 15–30. [Google Scholar] [CrossRef]
- Medić, B.; Stojanović, M.; Stimec, B.V.; Divac, N.; Vujović, K.S.; Stojanović, R.; Čolović, M.; Krstić, D.; Prostran, M. Lithium-Pharmacological and Toxicological Aspects: The Current State of the Art. Curr. Med. Chem. 2018, 27, 337–351. [Google Scholar] [CrossRef]
- Kissin, I.; Vlassakov, K.V. Pharmacology of General Anesthetics: Quantitative History of Research Attractiveness. Anesth. Analg. 2021, 132, 1486–1488. [Google Scholar] [CrossRef]
- Scott, L.; Lamb, J.; Smith, S.; Wheatley, D.N. Single amino acid (arginine) deprivation: Rapid and selective death of cultured transformed and malignant cells. Br. J. Cancer 2000, 83, 800–810. [Google Scholar] [CrossRef] [PubMed]
| Amino Acid (g/L) | M0 | M1 |
|---|---|---|
| Leucine (Leu) | 528 | 528 |
| Isoleucine (Ile) | 96 | 96 |
| Lysine (Lys) | 240 | 240 |
| Valine (Val) | 240 | 240 |
| Phenylalanine (Phe) | 192 | 192 |
| Threonine (Thr) | 160 | 160 |
| Histidine (His) | 80 | 80 |
| Methionine (Met) | 48 | 48 |
| Tryptophan (Trp) | 16 | 16 |
| Glutamine (Gln) | 1000 | 1000 |
| Glycine (Gly) | 200 | - |
| Aspartate (Asp) | 20 | - |
| Alanine (Ala) | 20 | - |
| Arginine (Arg) | 100 | - |
| Serine (Ser) | 40 | - |
| Cysteine (Cys) | 60 | - |
| Asparagine (Asn) | 50 | - |
| Proline (Pro) | 20 | - |
| Glutamate (Glu) | 20 | - |
| Tyrosine (Tyr) | 100 | - |
| Artificial Diet | TB1 | TB2 | TB3 | TB4 | TB5 |
|---|---|---|---|---|---|
| Dietary Constituents | |||||
| Casein | - | - | - | 6.00 | 6.00 |
| Glutamine (Gln) | 6.00 | 6.00 | 6.00 | 5.00 | 5.00 |
| Leucine (Leu) | 6.00 | 6.00 | 6.00 | - | 5.00 |
| Methionine (Met) | 0.60 | 0.60 | 0.17 | - | - |
| Phenylalanine (Phe) | 2.16 | 2.16 | 0.61 | - | - |
| Histidine (His) | 0.85 | 0.85 | 0.24 | - | - |
| Lysine (Lys) | 2.64 | 2.64 | 0.72 | - | - |
| Threonine (Thr) | 1.80 | 1.80 | 0.56 | - | - |
| Isoleucine (Ile) | 1.07 | 1.07 | 0.30 | - | - |
| Valine (Val) | 2.64 | 2.64 | 0.72 | - | - |
| Tryptophan (Trp) | 0.24 | 0.24 | 0.07 | - | - |
| Cystine (CySS) | - | - | - | - | - |
| Arginine (Arg) | - | - | - | - | - |
| Glycine (Gly) | - | - | - | - | - |
| Serine (Ser) | - | - | - | - | - |
| Tyrosine (Tyr) | - | - | - | - | - |
| Alanine (Ala) | - | - | - | - | - |
| Aspartate (Asp) | - | - | - | - | - |
| Proline (Pro) | - | - | - | - | - |
| Asparagine (Asn) | - | - | - | - | - |
| Glutamate (Glu) | - | - | - | - | - |
| Olive oil | 14.00 | 1.00 | - | - | - |
| Coconut oil | - | - | 1.00 | 1.00 | - |
| Salmon oil | - | - | - | - | 1.00 |
| Choline | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 |
| Vitamin Mix | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Mineral Mix | 3.50 | 3.50 | 3.50 | 3.50 | 3.50 |
| Sucrose | 15.00 | 15.00 | 15.00 | 15.00 | 15.00 |
| Cellulose | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 |
| Corn starch | 37.25 | 50.25 | 58.86 | 63.25 | 58.25 |
| Total (g or %) | 100 | 100 | 100 | 100 | 100 |
| Treatment | n | Survival Time (Mean ± SEM; Days) | Survival Improvement vs. Control (Days) | p Value vs. Control |
|---|---|---|---|---|
| Control | 7 | 21.6 ± 1.6 | - | - |
| Doxorubicin (0.5 mg/kg) | 8 | 23.6 ± 1.3 | +2.1 | 0.4838 |
| Diet TB1 | 10 | 26.5 ± 1.7 | +4.9 | 0.0837 |
| Treatment | n | Survival Time (Mean ± SEM; Days) | Survival Improvement vs. Control (Days) | p Value vs. Control |
|---|---|---|---|---|
| Control | 7 | 30.1 ± 3.5 | - | - |
| Capecitabine (450 mg/kg/day) | 4 | 23.3 ± 4.0 | −6.9 | 0.2349 |
| Diet TB2 | 7 | 38.6 ± 9.3 | +8.4 | 0.9496 |
| Control | Diet TB3 | Capecitabine (450 mg/kg/Day) | Anti-PD-1 (250 µg/Dose) | Cisplatin (5 mg/kg) | ||
|---|---|---|---|---|---|---|
| Breast cancer | Survival time (Mean ± SEM; days) | 26.1 ± 3.2 | 43.8 ± 16.2 | 24.2 ± 1.7 | - | - |
| Survival improvement vs. Control (days) | - | +17.7 | −1.9 | - | - | |
| n | 8 | 6 | 10 | - | - | |
| Lung cancer | Survival time (Mean ± SEM; days) | 26.3 ± 0.7 | 30.0 ± 0.6 | - | 23.3 ± 2.0 | - |
| Survival improvement vs. Control (days) | - | +3.7 | - | −3 | - | |
| n | 3 | 3 | - | 3 | - | |
| Ovarian cancer | Survival time (Mean ± SEM; days) | 48.8 ± 0.8 | 64.3 ± 1.8 | - | - | 80.8 ± 2.1 |
| Survival improvement vs. Control (days) | - | +15.5 | - | - | +32.0 | |
| n | 4 | 4 | - | - | 4 | |
| Melanoma | Survival time (Mean ± SEM; days) | 22.7 ± 01.7 | 26.8 ± 2.8 | - | - | 26.3 ± 4.9 |
| Survival improvement vs. Control (days) | - | +4.1 | - | - | +3.7 | |
| n | 3 | 3 | - | - | 4 | |
| Colon cancer (i.v.) | Survival time (Mean ± SEM; days) | 33.3 ± 4.9 | 78.0 ± 29.7 | 36.7 ± 3.2 | - | - |
| Survival improvement vs. Control (days) | - | +44.7 | +3.3 | - | - | |
| n | 3 | 3 | 3 | - | - | |
| Colon cancer (i.p.) | Survival time (Mean ± SEM; days) | 27.0 ± 4.2 | 31.3 ± 1.7 | 28.0 ± 3.5 | - | - |
| Survival improvement vs. Control (days) | - | +4.3 | +1.0 | - | - | |
| n | 3 | 3 | 3 | - | - | |
| Total | Survival time (Mean ± SEM; days) | 30.5 ± 2.2 | 45.4 ± 6.4 | 27.3 ± 1.8 | 23.3 ± 2.0 | 57.4 ± 11.2 |
| Survival improvement vs. Control (days) | - | +14.9 | −3.3 | −7.2 | +26.9 | |
| n | 24 | 23 | 16 | 3 | 7 |
| Treatment | n | Survival Time (Mean ± SEM; Days) | Survival Improvement vs. Control (Days) | p Value vs. Control |
|---|---|---|---|---|
| Control | 8 | 26.1 ± 3.2 | - | - |
| Capecitabine (450 mg/kg/day) | 10 | 24.2 ± 1.7 | −1.9 | 1.0000 |
| Diet TB4 | 8 | 52.0 ± 28.8 | +25.9 | 0.4256 |
| Diet TB5 | 9 | 75.6 ± 46.9 | +49.4 | 0.4678 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guillén-Mancina, E.; Jiménez-Alonso, J.J.; Calderón-Montaño, J.M.; Jiménez-González, V.; Díaz-Ortega, P.; Burgos-Morón, E.; López-Lázaro, M. Artificial Diets with Selective Restriction of Amino Acids and Very Low Levels of Lipids Induce Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer. Cancers 2023, 15, 1540. https://doi.org/10.3390/cancers15051540
Guillén-Mancina E, Jiménez-Alonso JJ, Calderón-Montaño JM, Jiménez-González V, Díaz-Ortega P, Burgos-Morón E, López-Lázaro M. Artificial Diets with Selective Restriction of Amino Acids and Very Low Levels of Lipids Induce Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer. Cancers. 2023; 15(5):1540. https://doi.org/10.3390/cancers15051540
Chicago/Turabian StyleGuillén-Mancina, Emilio, Julio José Jiménez-Alonso, José Manuel Calderón-Montaño, Víctor Jiménez-González, Patricia Díaz-Ortega, Estefanía Burgos-Morón, and Miguel López-Lázaro. 2023. "Artificial Diets with Selective Restriction of Amino Acids and Very Low Levels of Lipids Induce Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer" Cancers 15, no. 5: 1540. https://doi.org/10.3390/cancers15051540
APA StyleGuillén-Mancina, E., Jiménez-Alonso, J. J., Calderón-Montaño, J. M., Jiménez-González, V., Díaz-Ortega, P., Burgos-Morón, E., & López-Lázaro, M. (2023). Artificial Diets with Selective Restriction of Amino Acids and Very Low Levels of Lipids Induce Anticancer Activity in Mice with Metastatic Triple-Negative Breast Cancer. Cancers, 15(5), 1540. https://doi.org/10.3390/cancers15051540








