APESTNet with Mask R-CNN for Liver Tumor Segmentation and Classification
Abstract
Simple Summary
Abstract
1. Introduction
- Three steps are only included in this study such as pre-processing, segmentation, and classification.
- Histogram Equalization and medium filtering are used for the improvement of the input images.
- Enhanced Mask R-CNN is used to segment the liver tumor from the pre-processed images. The research work introduces multistage optimization for deep learning segmentation networks. The study used a multi-optimization training system by utilizing stochastic gradient descent and adaptive moment estimation (Adam) with preprocessed CT images in enhanced Mask-RCNN.
- APESTNet is introduced in this study for classification. Overfitting issues in the Swin Transformer model are prevented by introducing Adversarial propagation in the classifier.
2. Related Works
3. Materials and Methods
3.1. Dataset and Implementation
3.2. Image Pre-Processing
Histogram Equalization
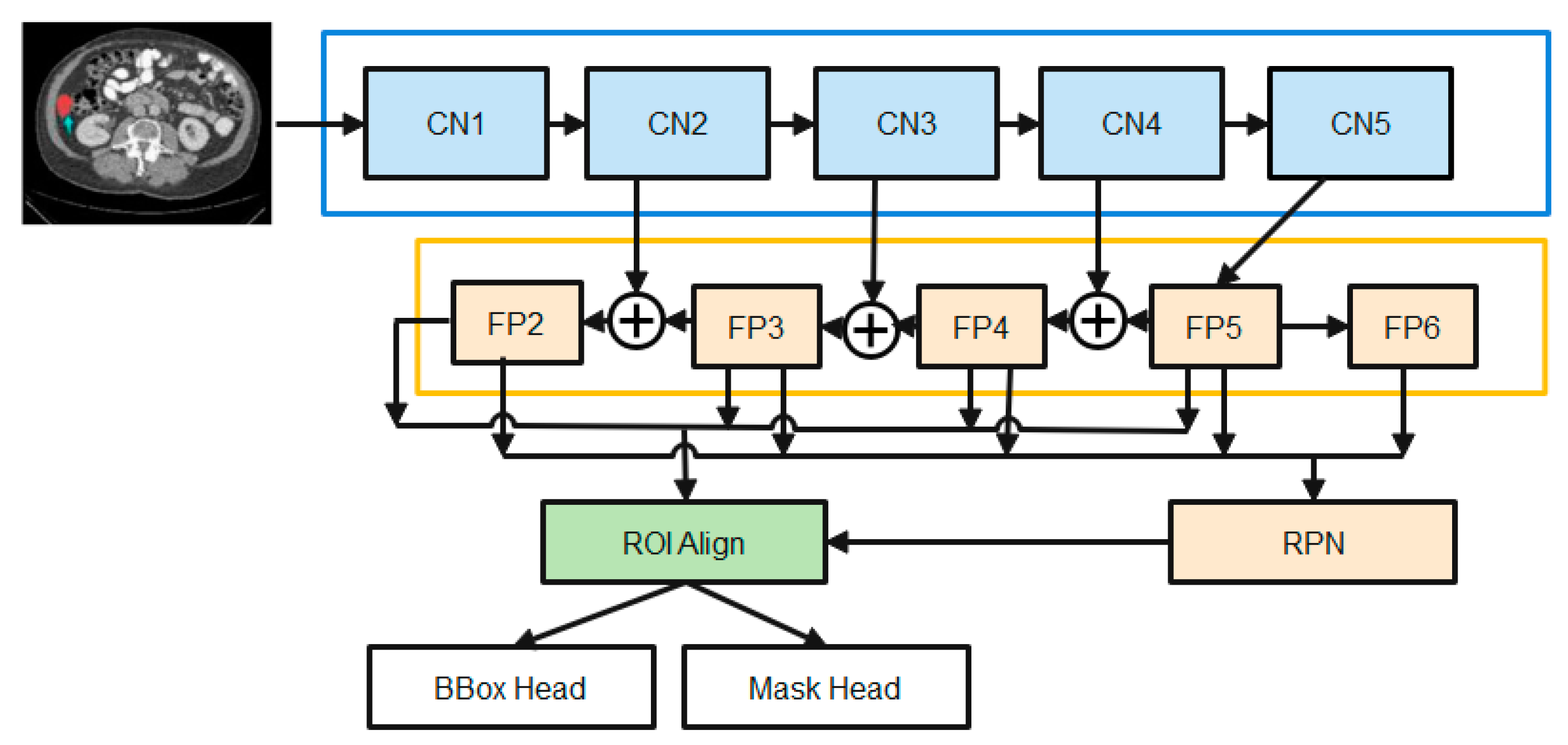
3.3. Segmentation Using Enhanced M-RCNN
- (1)
- The Backbone converts the incoming image into a raw feature map. Here, we employ a variant of ResNet-50-based on the design.
- (2)
- The Neck joins the Spine to the Head. The original feature map is refined and rearranged. It has a vertical corridor and horizontal branches. To produce a feature pyramid map of the same size as the raw feature map, the top-bottom route is used. Convolutional add operations between two parallel pathways’ corresponding levels characterize lateral linkages.
- (3)
- Third, the Dense Head can be used to perform dense placements of feature maps. The RPN examines each area and makes the best guess as to whether or not an object is present. The RPN’s main benefit is that it doesn’t need to look at the real image itself. Through the use of a fixed number of anchor boxes, the network performs a rapid scan of the feature map.
- (4)
- ROIHead (BBoxHead, MaskHead): Using ROIAlign, extract features that affect the ROI from different feature maps while maintaining their precise spatial placements. This section takes in ROI features and then predicts task-related outcomes based on those features. At this point, you’ll be doing two things at once:
- In the detection branch, the location of the bounding box (BBoxHead) is identified and classified for intervertebral disc (IVD) detection.
- In the segmentation node, the FCN created the IVD image segmentation. b. MaskHead.
Loss Functions
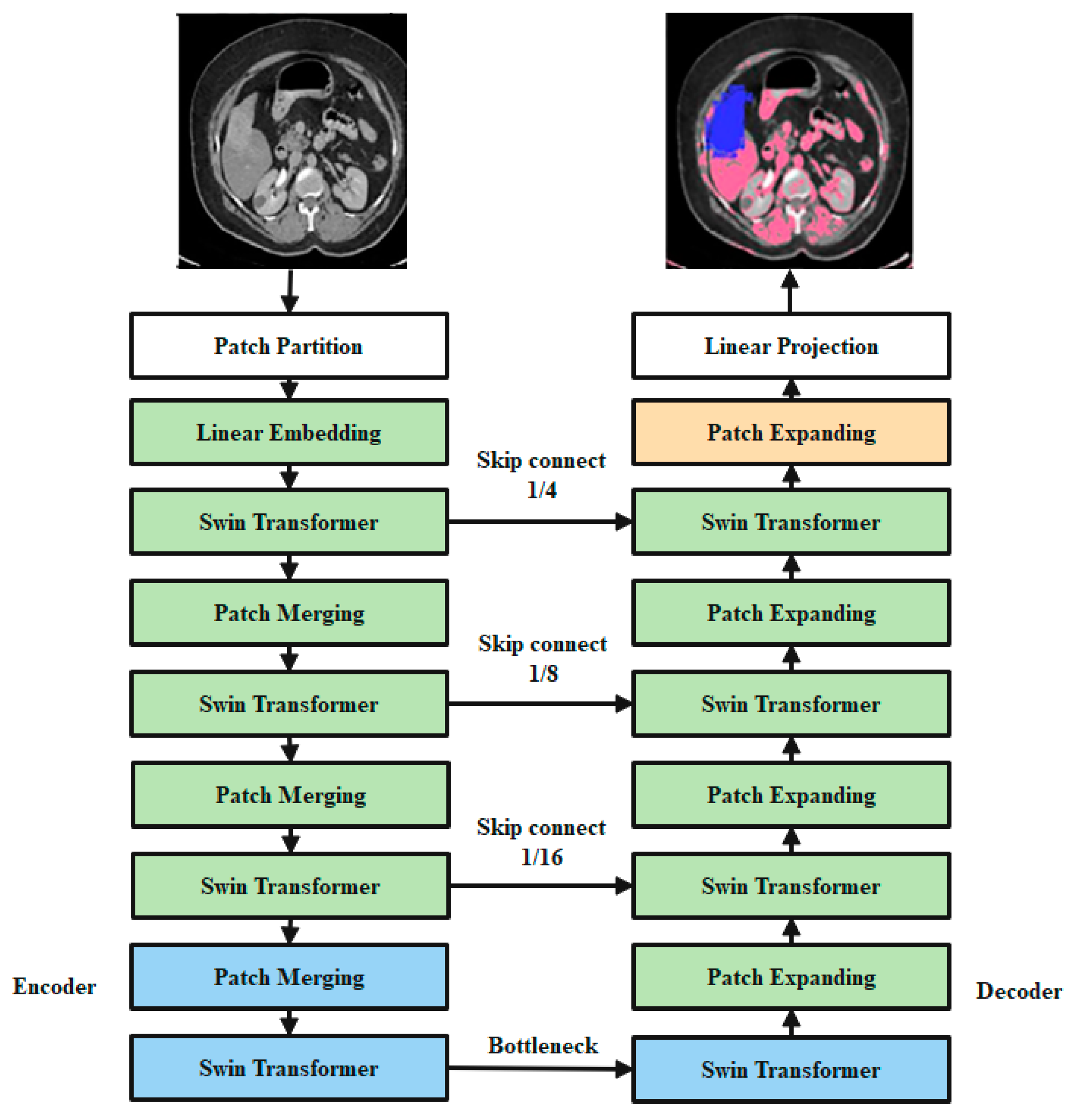
3.4. Classification
3.4.1. Patch Embedding
3.4.2. Patch Merging
3.4.3. Mask
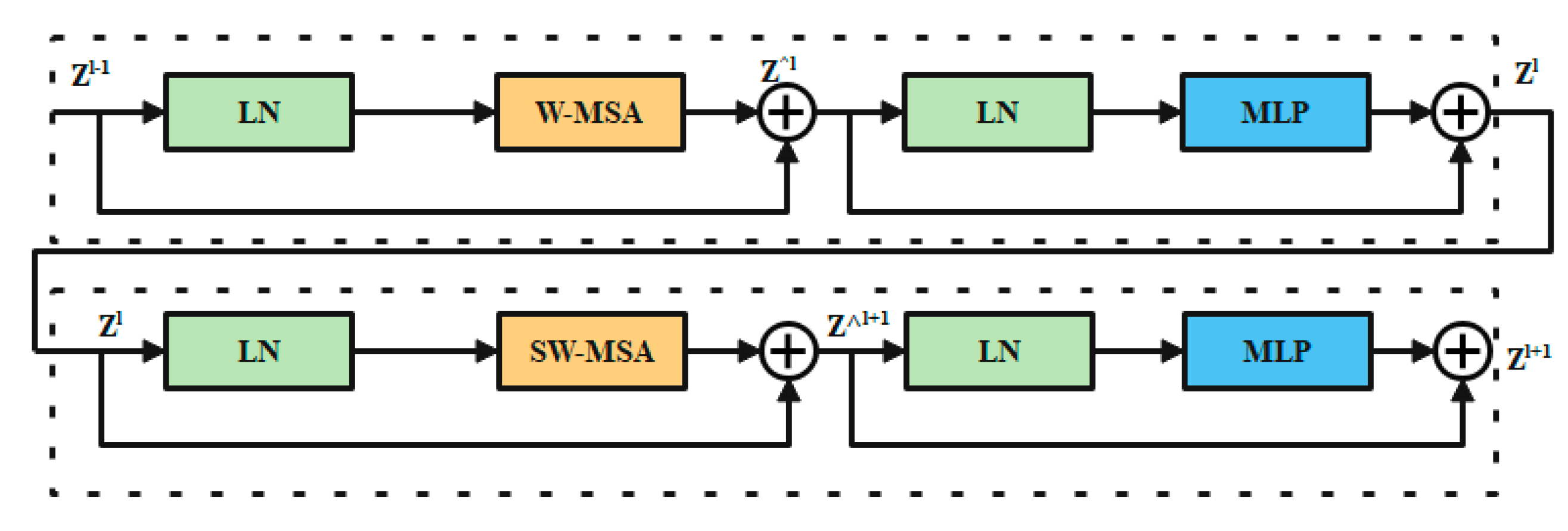
3.4.4. Swin Transformer Block
3.4.5. Encoder
3.4.6. Bottleneck
3.4.7. Decoder
3.4.8. Skip Connection
4. Results and Discussion
4.1. Segmentation Results
Evaluation Metrics for Segmentation
| Case Num | VOE | ASD (mm) | MSD (mm) | RVD | Dice |
|---|---|---|---|---|---|
| 1 | 0.103 | 1.568 | 28.453 | −0.029 | 0.955 |
| 2 | 0.089 | 1.471 | 34.951 | −0.033 | 0.963 |
| 3 | 0.100 | 1.382 | 31.259 | −0.049 | 0.956 |
| 4 | 0.097 | 1.494 | 25.494 | −0.048 | 0.968 |
| 5 | 0.114 | 1.797 | 28.315 | −0.040 | 0.949 |
| 6 | 0.107 | 1.933 | 29.756 | −0.038 | 0.953 |
| 7 | 0.094 | 1.229 | 30.657 | 0.034 | 0.960 |
| 8 | 0.073 | 0.955 | 39.421 | 0.043 | 0.961 |
| 9 | 0.086 | 1.673 | 34.598 | 0.036 | 0.954 |
| 10 | 0.090 | 1.863 | 28.534 | 0.040 | 0.952 |
| Avg | 0.095 | 1.544 | 29.144 | −0.0084 | 0.957 |
4.2. Classification Results
4.2.1. Performance Measure for Classification
- Accuracy: On test samples, accuracy is referred to as “accuracy.”
- Precision: In the context of predictive value, precision refers to a positive value and is the ratio of genuine positive models to the total number of false positive samples.
- Recall: Classifier performance can be evaluated using this metric. Alternatively known as Sensitivity or True Positive Rate, which describes an organization model, Recall discards a positive prediction if it’s not accurate.
- F1: Classification is an example of a machine learning task for which this measure is well-known. It is the arithmetic mean of the estimates’ precision and recall.
4.2.2. Validation Analysis Using 70% of Training Data–30% of Testing Data
4.2.3. Validation Analysis Using 60% of Training Data–40% of Testing Data
| Model | Accuracy | Precision | Recall | F1 |
|---|---|---|---|---|
| CNN-RNN [30] | 81.10 | 99.41 | 75.91 | 86.72 |
| Ga-CNN [31] | 87.70 | 99.41 | 85.21 | 91.82 |
| GAN-R-CNN [32] | 92.50 | 99.82 | 90.98 | 95.27 |
| SAR-U-Net [33] | 92.90 | 99.78 | 91.52 | 95.41 |
| HCNN [34] | 92.50 | 99.63 | 91.38 | 95.18 |
| RF [35] | 92.70 | 99.90 | 91.93 | 95.32 |
| CNN [36] | 93.27 | 99.91 | 92.47 | 95.63 |
| Proposed APESTNet | 94.32 | 99.95 | 93.24 | 96.02 |
5. Limitation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [CrossRef]
- Damhorst, G.L.; Murtagh, M.; Rodriguez, W.R.; Bashir, R. Microfluidics and nanotechnology for detection of global infectious diseases. Proc. IEEE 2015, 103, 150–160. [Google Scholar] [CrossRef]
- Murakami, T.; Imai, Y.; Okada, M.; Hyodo, T.; Lee, W.-J.; Kim, M.-J.; Kim, T.; Choi, B.I. Ultrasonography, computed tomography and magnetic resonance imaging of hepatocellular carcinoma: Toward improved treatment decisions. Oncology 2011, 81, 86–99. [Google Scholar] [CrossRef]
- Ranjbarzadeh, R.; Saadi, S.B. Automated liver and tumor segmentation based on concave and convex points using fuzzy c-means and mean shift clustering. Measurement 2020, 150, 107086. [Google Scholar] [CrossRef]
- Hou, W.; Toh, T.B.; Abdullah, L.N.; Yvonne, T.W.Z.; Lee, K.J.; Guenther, I.; Chow, E.K.-H. Nanodiamond– manganese dual mode MRI contrast agents for enhanced liver tumor detection. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Dogantekin, A.; Ozyurt, F.; Avcı, E.; Koc, M. A novel approach for liver image classification: PH-C-ELM. Measurement 2019, 137, 332–338. [Google Scholar] [CrossRef]
- Nasiri, N.; Foruzan, A.H.; Chen, Y.-W. Integration of a knowledge-based constraint into generative models with applications in semi-automatic segmentation of liver tumors. Biomed Signal Process. Control 2020, 57, 101725. [Google Scholar] [CrossRef]
- Lebre, M.-A.; Vacavant, A.; Grand-Brochier, M.; Rositi, H.; Abergel, A.; Chabrot, P.; Magnin, B. Automatic segmentation methods for liver and hepatic vessels from CT and MRI volumes, applied to the Couinaud scheme. Comput. Biol. Med. 2019, 110, 42–51. [Google Scholar] [CrossRef]
- Xu, L.; Zhu, Y.; Zhang, Y.; Yang, H. Liver segmentation based on region growing and level set active contour model with new signed pressure force function. Optik 2020, 202, 163705. [Google Scholar] [CrossRef]
- Patil, D.D.; Deore, S.G. Medical image segmentation: A review. Int. J. Comput. Sci. Mob. Comput. 2013, 2, 22–27. [Google Scholar]
- Cheng, D.; Guot, Y.; Zhang, Y. A novel image segmentation approach based onneutrosophic set and improved fuzzy Cmeans algorithm. New Mat. Nat. Comput. 2011, 7, 155–171. [Google Scholar] [CrossRef]
- Budak, U.; Guo, Y.; Tanyildizi, E.; Şengür, A. Cascaded deep convolutional encoder-decoder neural networks for efficient liver tumor segmentation. Med. Hypotheses 2020, 134, 109431. [Google Scholar] [CrossRef]
- Baazaoui, A.; Barhoumi, W.; Ahmed, A.; Zagrouba, E. Semiautomated segmentation of single and multiple Tumors in liver CT images using entropy-based fuzzy region growing. IRBM 2017, 38, 98–108. [Google Scholar] [CrossRef]
- Tanaka, O.; Kojima, T.; Ohbora, A.; Makita, C.; Taniguchi, T.; Ono, K.; Matsuo, M.; Nagata, Y. Scores of Child-Pugh Classification Impact Overall Survival after Stereotactic Body Radiation Therapy for Primary and Metastatic Liver tumors. J. Clin. Exp. Hepatol. 2019, 10, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Perell, K.; Vincent, M.; Vainer, B.; Petersen, B.L.; Federspiel, B.; Møller, A.K.; Madsen, M.; Hansen, N.R.; Friis-Hansen, L.; Nielsen, F.C.; et al. Development and validation of a micro RNA based diagnostic assay for primary tumor site classification of liver core biopsies. Mol. Oncol. 2015, 9, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wu, J.; Ren, X. The study and application of the improved region growing algorithm for liver segmentation. Opt. —Int. J. Light Electron Opt. 2014, 125, 2142–2147. [Google Scholar] [CrossRef]
- Li, X.; Huang, C.; Jia, F. Automatic Liver Segmentation Using Statistical Prior Models and Free-form Deformation. In Medical Computer Vision: Algorithms for Big Data; Springer: Cham, Switzerland, 2014; Volume 8848, pp. 181–188. [Google Scholar]
- Shi, C.; Cheng, Y.; Wang, J.; Wang, Y.; Mori, M.; Tamura, S. Low-rank and sparse decomposition based shape model and probabilistic atlas for automatic pathological organ segmentation. Med. Image Anal. 2017, 38, 30–49. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, Y.; Guo, C.; Wang, Y.; Tamura, S. Shape–intensity prior level set combining probabilistic atlas and probability map constrains for automatic liver segmentation from abdominal CT images. Int. J. Comput. Assist. Radiol. Surg. 2016, 11, 817–826. [Google Scholar] [CrossRef]
- Krizhevsky, A.; Sutskever, I.; Hinton, G.E. Imagenet classification with deep convolutional neural networks. In Proceedings of the 25th International Conference on Neural Information Processing Systems, Lake Tahoe, NV, USA, 3–6 December 2012; pp. 1097–1105. [Google Scholar]
- He, K.; Zhang, X.; Ren, S.; Sun, J. Deep residual learning for image recognition. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Las Vegas, NV, USA, 27–30 June 2016; pp. 770–778. Available online: https://ieeexplore.ieee.org/document/7780459 (accessed on 29 November 2022).
- Ren, S.; He, K.; Girshick, R.; Sun, J. Faster r-cnn: Towards real-time object detection with region proposal networks. In Proceedings of the 28th International Conference on Neural Information Processing Systems, Montreal, QC, Canada, 7–12 December 2015; pp. 91–99. [Google Scholar]
- Long, J.; Shelhamer, E.; Darrell, T. Fully convolutional networks for semantic segmentation. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, IEEE Computer Society, Boston, MA, USA, 7–12 June 2015; pp. 3431–3440. [Google Scholar]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional Networks for Biomedical Image Segmentation. In Medical Image Computing and Computer-Assisted Intervention—MICCAI 2015; Springer: Cham, Switzerland, 2015; pp. 234–241. [Google Scholar]
- Milletari, F.; Navab, N.; Ahmadi, S.-A. V-Net: Fully Convolutional Neural Networks for Volumetric Medical Image Segmentation. In Proceedings of the 2016 Fourth International Conference on 3D Vision, Stanford, CA, USA, 25–28 October 2016; pp. 565–571. [Google Scholar]
- Rida, I.; Al-Maadeed, N.; Al-Maadeed, S.; Bakshi, S. Automatic liver and lesion segmentation in CT using cascaded fully convolutional neural networks and 3 D conditional random fields. In Proceedings of the Medical Image Computing and Computer-Assisted Intervention (MICCAI 2016), Athens, Greece, 17–21 October 2016; pp. 415–423. [Google Scholar]
- Kaluva, K.C.; Khened, M.; Kori, A.; Krishnamurthi, G. 2d-densely connected convolution neural networks for automatic liver and tumor segmentation. arXiv 2018, arXiv:1802.02182. [Google Scholar]
- Li, X.; Chen, H.; Dou, Q.; Fu, C.W.; Heng, P.A. H-DenseU-Net: Hybrid densely connected U-Net for liver and liver tumor segmentation from CT volumes. arXiv 2017, arXiv:1709.07330. [Google Scholar]
- Han, X. Automatic liver lesion segmentation using a deep convolutional neural network method. arXiv 2017, arXiv:1704.07239. [Google Scholar]
- Rela, M.; Nagaraja Rao, S.; Ramana Reddy, P. Optimized segmentation and classification for liver tumor segmentation and classification using opposition-based spotted hyena optimization. Int. J. Imaging Syst. Technol. 2021, 31, 627–656. [Google Scholar] [CrossRef]
- Ahmad, M.; Qadri, S.F.; Qadri, S.; Saeed, I.A.; Zareen, S.S.; Iqbal, Z.; Alabrah, A.; Alaghbari, H.M.; Rahman, M.; Md, S. A Lightweight Convolutional Neural Network Model for Liver Segmentation in Medical Diagnosis. Comput. Intell. Neurosci. 2022, 2022, 9956983. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Chen, X.; Lai, C.; Zhu, Y.; Yang, H.; Du, Y. Automatic Liver Segmentation in CT Images with Enhanced GAN and Mask Region-Based CNN Architectures. BioMed Res. Int. 2021, 2021, 9956983. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lv, P.; Wang, H.; Shi, C. SAR-U-Net: Squeeze-and-excitation block and atrous spatial pyramid pooling based residual U-Net for automatic liver segmentation in Computed Tomography. Comput. Methods Programs Biomed. 2021, 208, 106268. [Google Scholar] [CrossRef] [PubMed]
- Saha Roy, S.; Roy, S.; Mukherjee, P.; Halder Roy, A. An automated liver tumour segmentation and classification model by deep learning based approaches. Comput. Methods Biomech. Biomed. Eng. Imaging Vis. 2022, 1–13. [Google Scholar] [CrossRef]
- Hussain, M.; Saher, N.; Qadri, S. Computer Vision Approach for Liver Tumor Classification Using CT Dataset. Appl. Artif. Intell. 2022, 36, 2055395. [Google Scholar] [CrossRef]
- Kaur, A.; Chauhan, A.P.S.; Aggarwal, A.K. An automated slice sorting technique for multi-slice computed tomography liver cancer images using convolutional network. Expert Syst. Appl. 2021, 186, 115686. [Google Scholar] [CrossRef]
- Jeong, J.G.; Choi, S.; Kim, Y.J.; Lee, W.S.; Kim, K.G. Deep 3 D attention CLSTM U-Net based automated liver segmentation and volumetry for the liver transplantation in abdominal CT volumes. Sci. Rep. 2022, 12, 6370. [Google Scholar] [CrossRef]
- Militello, C.; Rundo, L.; Dimarco, M.; Orlando, A.; Woitek, R.; D’Angelo, I.; Russo, G.; Bartolotta, T.V. 3 D DCE-MRI radiomic analysis for malignant lesion prediction in breast cancer patients. Acad. Radiol. 2022, 29, 830–840. [Google Scholar] [CrossRef]
- Dorothy, R.; Joany, R.M.; Rathish, J.; Santhana Prabha, S.; Rajendran, S.; Joseph, S.T. Image enhancement by histogram equalization. Int. J. Nano Corros. Sci. Eng. 2015, 2, 21–30. [Google Scholar]
- Zhu, Y.; Cheng, H. An improved median filtering algorithm for image noise reduction. Phys Procedia. 2012, 25, 609–616. [Google Scholar] [CrossRef]
- Liu, Z.; Lin, Y.; Cao, Y.; Hu, H.; Wei, Y.; Zhang, Z.; Lin, S.; Guo, B. Swin transformer: Hierarchical vision transformer using shifted windows. In Proceedings of the IEEE/CVF International Conference on Computer Vision, Montreal, QC, Canada, 10–17 October 2021. [Google Scholar]
- Hu, H.; Gu, J.; Zhang, Z.; Dai, J.; Wei, Y. Relation networks for object detection. In Proceedings of the 2018 IEEE/CVF Conference on Computer Vision and Pattern Recognition, Salt Lake City, UT, USA, 18–23 June 2018; pp. 3588–3597. [Google Scholar]
- Hu, H.; Zhang, Z.; Xie, Z.; Lin, S. Local relation networks for image recognition. In Proceedings of the 2019 IEEE/CVF International Conference on Computer Vision (ICCV), Seoul, Republic of Korea, 27 October–2 November 2019; pp. 3463–3472. [Google Scholar]
- Touvron, H.; Cord, M.; Sablayrolles, A.; Synnaeve, G.; J’egou, H. Going deeper with image transformers. arXiv 2021, arXiv:2103.172392021. [Google Scholar]
- Xie, Q.; Luong, M.-T.; Hovy, E.; Le, Q.V. Self-training with noisy student improves imagenet classication. In Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR), Seattle, WA, USA, 13–19 June 2020. [Google Scholar]
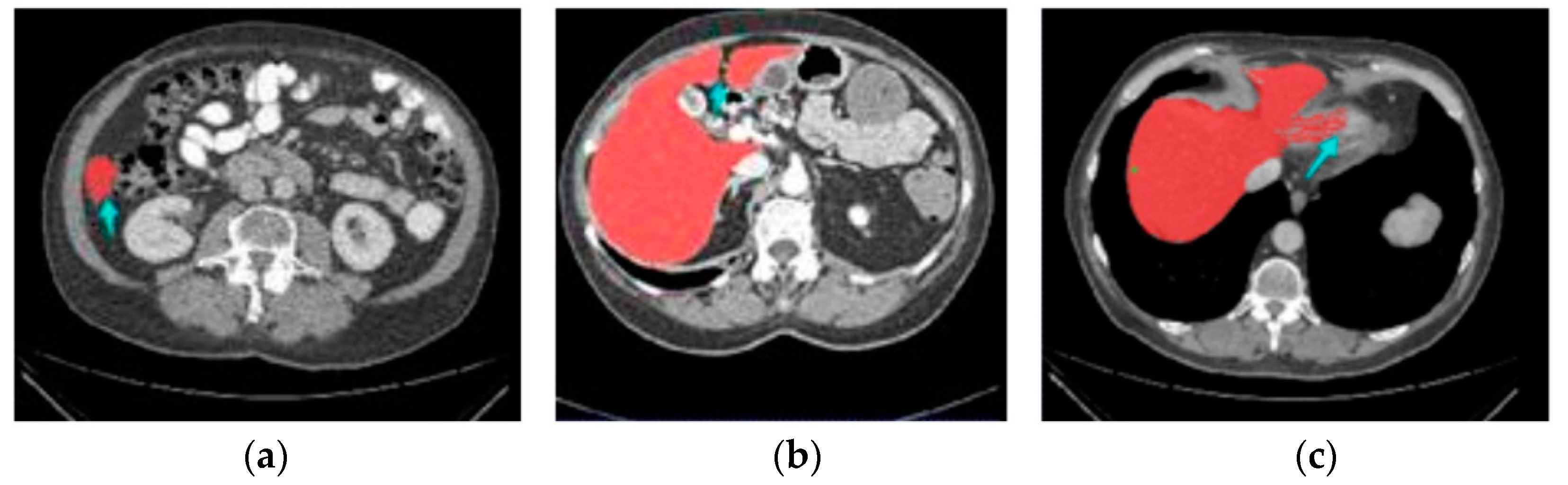
| Methods | Dice (%) | VOE (%) | RVD (%) | ASD (mm) | MSD (mm) |
|---|---|---|---|---|---|
| Adaptive Thresholding [30] | 95.60 ± 3.41 | 8.23 ± 5.89 | −2.38 ± 2.16 | 2.19 ± 0.38 | 36.69 ± 1.45 |
| Ga-CNN [31] | 96.94 ± 1.78 | 5.31 ± 3.48 | −0.54 ± 2.24 | 1.95 ± 0.34 | 30.66 ± 2.03 |
| GAN Mask R-CNN [32] | 96.66 ± 2.19 | 6.38 ± 3.93 | −1.29 ± 3.58 | 1.80 ± 0.38 | 28.30 ± 2.05 |
| Proposed Enhanced M-RCNN model | 97.31 ± 1.49 | 5.37 ± 3.27 | −1.08 ± 2.06 | 1.85 ± 0.30 | 27.45 ± 1.89 |
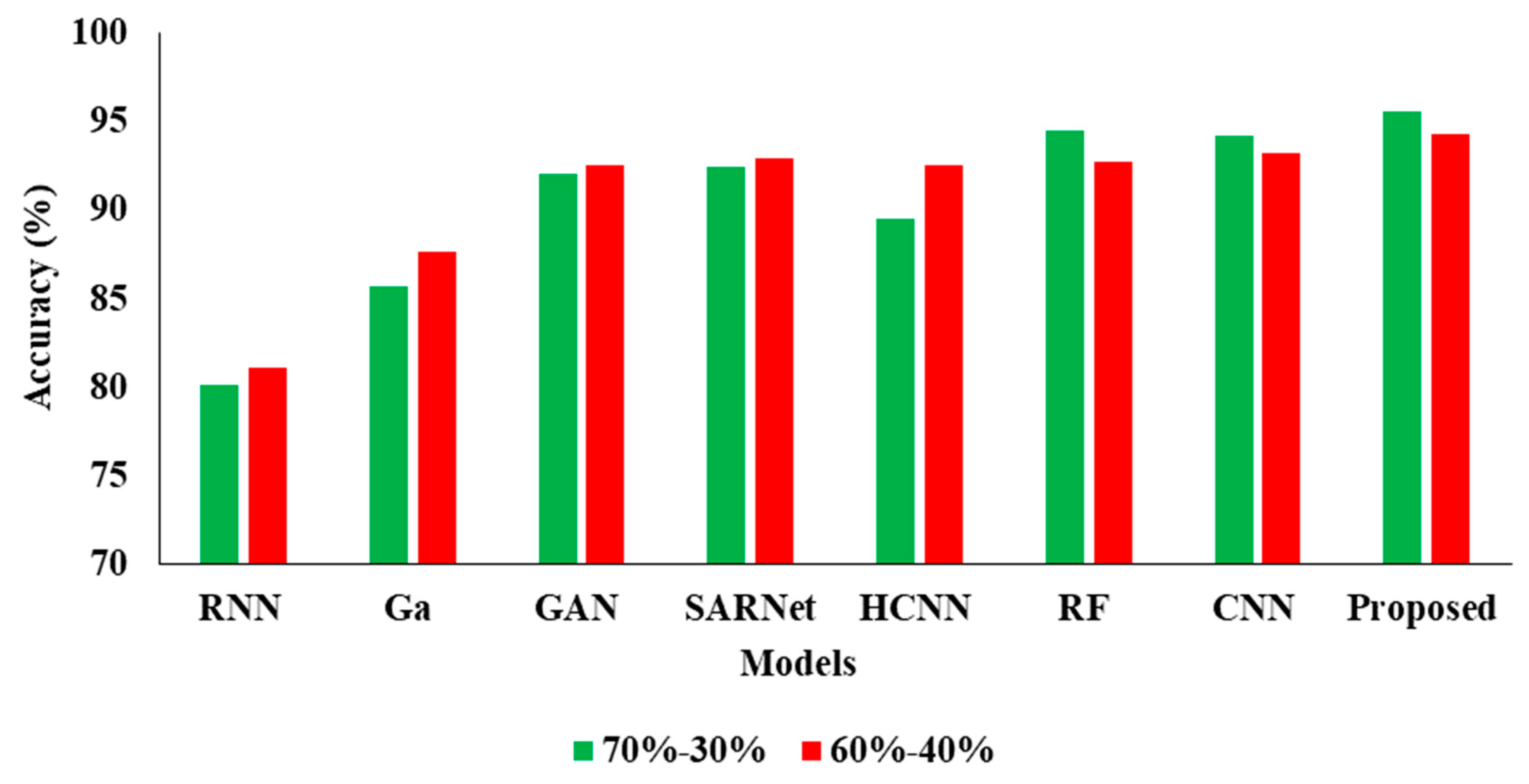
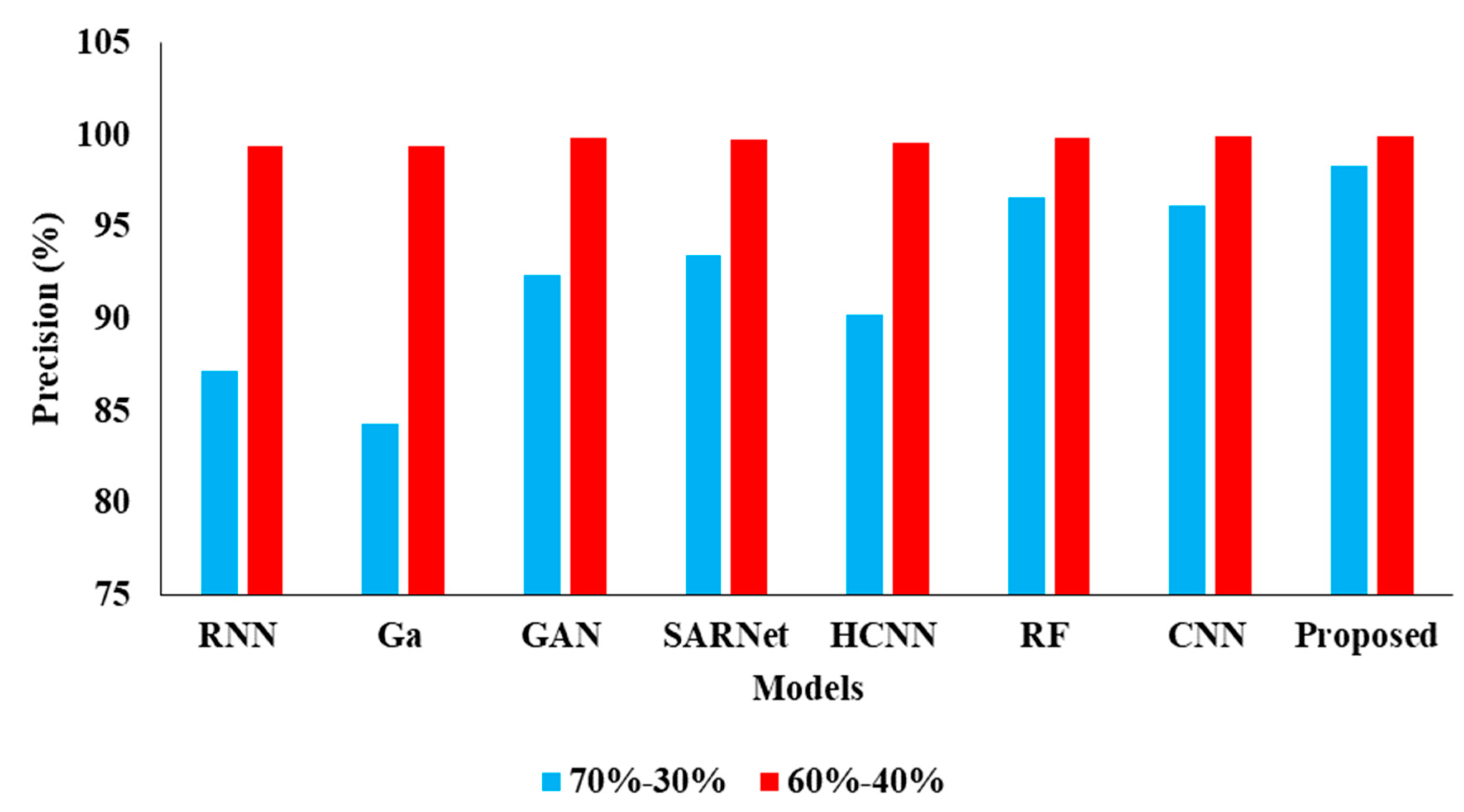
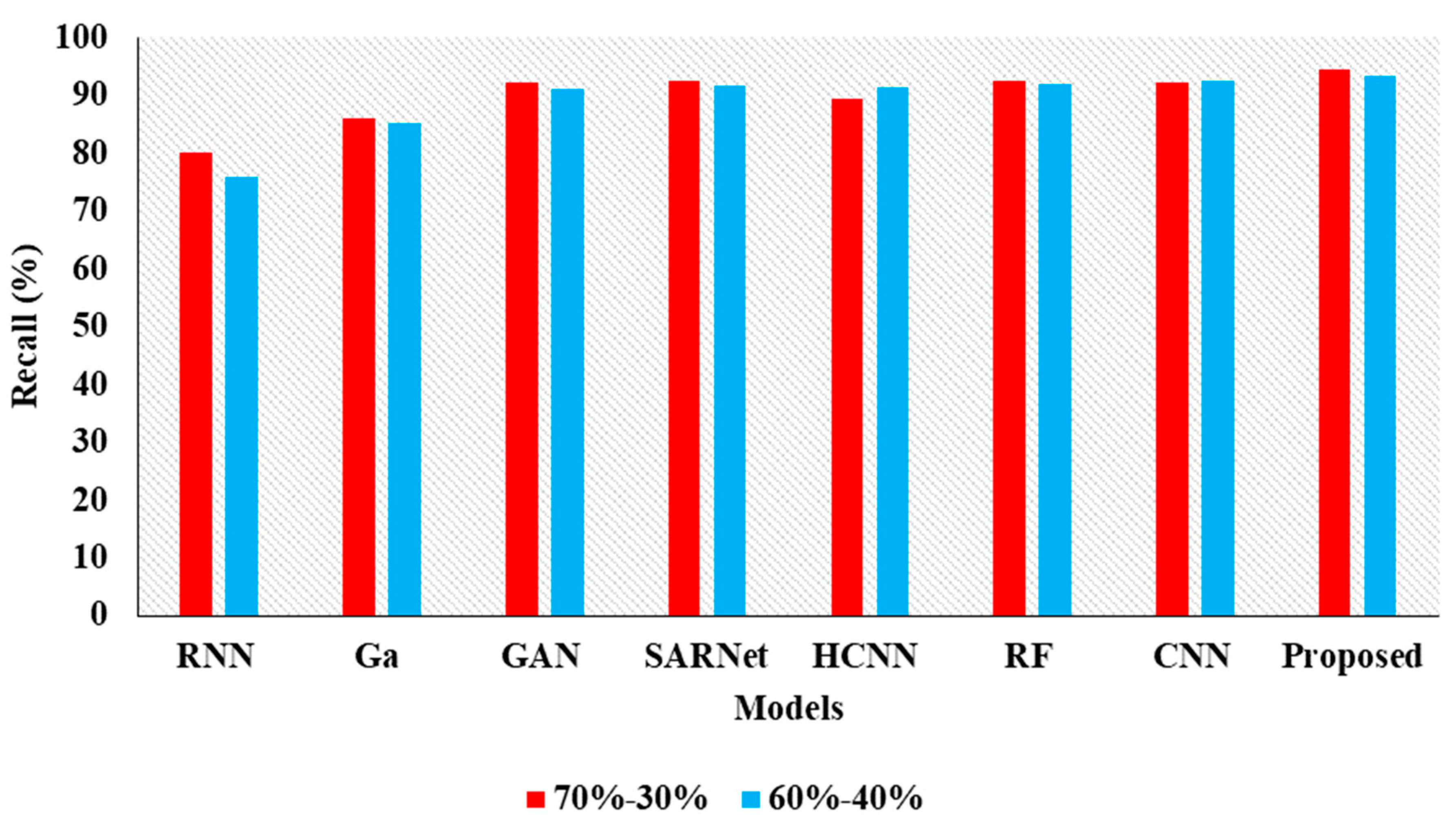
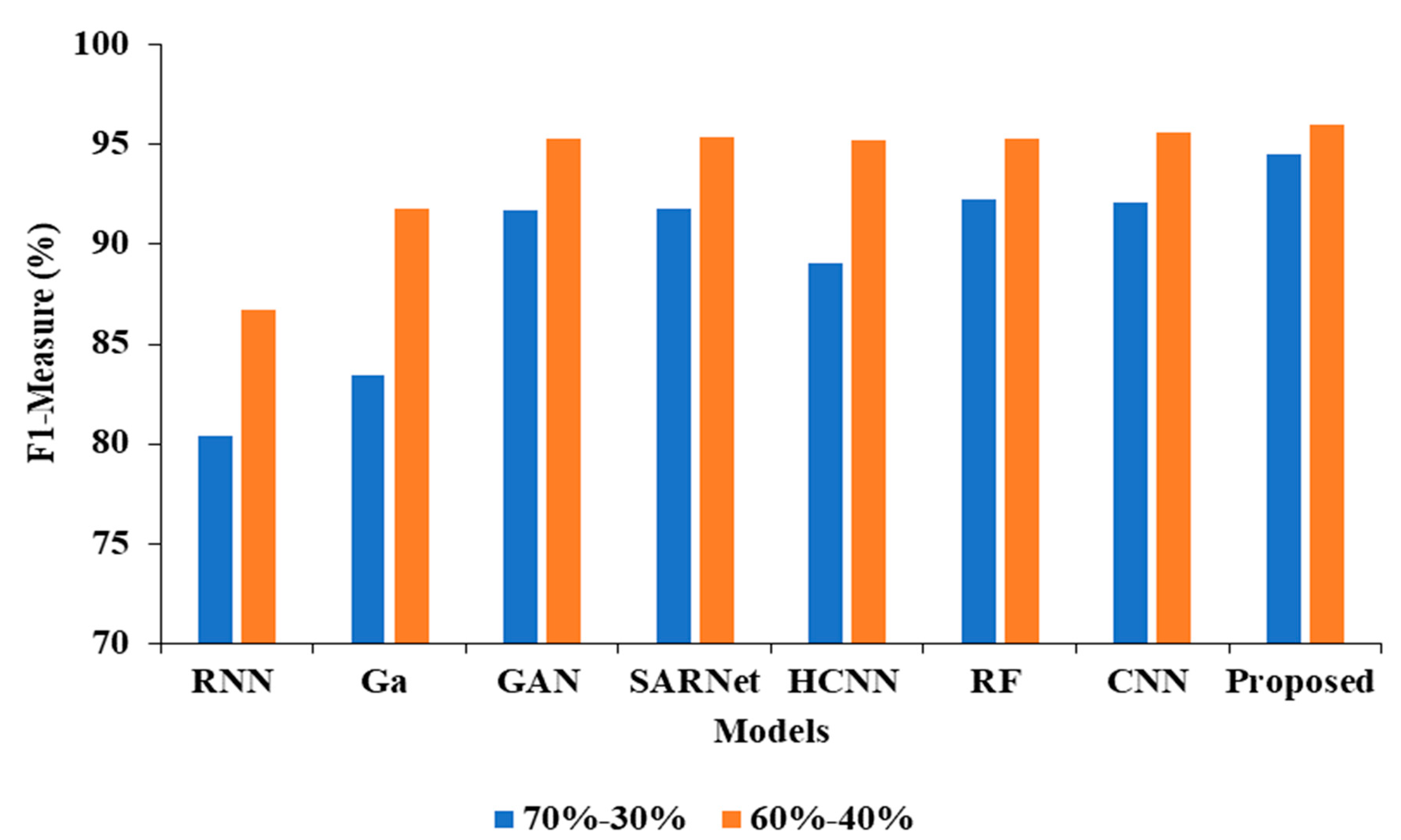
| Model | Accuracy | Precision | Recall | F1 |
|---|---|---|---|---|
| CNN-RNN [30] | 80.10 | 87.21 | 80.15 | 80.43 |
| Ga-CNN [31] | 85.71 | 84.32 | 85.93 | 83.45 |
| GAN-R-CNN [32] | 92.10 | 92.43 | 92.15 | 91.68 |
| SAR-U-Net [33] | 92.46 | 93.48 | 92.44 | 91.81 |
| HCNN [34] | 89.52 | 90.21 | 89.54 | 89.03 |
| RF [35] | 94.53 | 96.61 | 92.52 | 92.24 |
| CNN [36] | 94.16 | 96.17 | 92.32 | 92.10 |
| Proposed APESTNet | 95.62 | 98.32 | 94.62 | 94.53 |
| Model | Training Time (s) | Testing Time (s) | Execution Time (s) |
|---|---|---|---|
| CNN-RNN [30] | 630.01 | 69.17 | 0.01233 |
| Ga-CNN [31] | 450.53 | 67.38 | 0.01369 |
| GAN-R-CNN [32] | 543.21 | 65.89 | 0.01369 |
| SAR-U-Net [33] | 577.66 | 63.71 | 0.01657 |
| CNN [34] | 480.23 | 60.41 | 0. 01309 |
| RF [35] | 583.42 | 59.78 | 0. 01297 |
| CNN [36] | 423.17 | 59.192 | 0. 01289 |
| Proposed APESTNet | 390.33 | 57.621 | 0.01128 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balasubramanian, P.K.; Lai, W.-C.; Seng, G.H.; C, K.; Selvaraj, J. APESTNet with Mask R-CNN for Liver Tumor Segmentation and Classification. Cancers 2023, 15, 330. https://doi.org/10.3390/cancers15020330
Balasubramanian PK, Lai W-C, Seng GH, C K, Selvaraj J. APESTNet with Mask R-CNN for Liver Tumor Segmentation and Classification. Cancers. 2023; 15(2):330. https://doi.org/10.3390/cancers15020330
Chicago/Turabian StyleBalasubramanian, Prabhu Kavin, Wen-Cheng Lai, Gan Hong Seng, Kavitha C, and Jeeva Selvaraj. 2023. "APESTNet with Mask R-CNN for Liver Tumor Segmentation and Classification" Cancers 15, no. 2: 330. https://doi.org/10.3390/cancers15020330
APA StyleBalasubramanian, P. K., Lai, W.-C., Seng, G. H., C, K., & Selvaraj, J. (2023). APESTNet with Mask R-CNN for Liver Tumor Segmentation and Classification. Cancers, 15(2), 330. https://doi.org/10.3390/cancers15020330





