Role of IL-17A and IL-17RA in Prostate Cancer with Lymph Nodes Metastasis: Expression Patterns and Clinical Significance
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients Selection
2.2. Tissue Microarrays (TMAs) and Immunohistochemical (IHC) Staining
2.3. Statistical Analysis
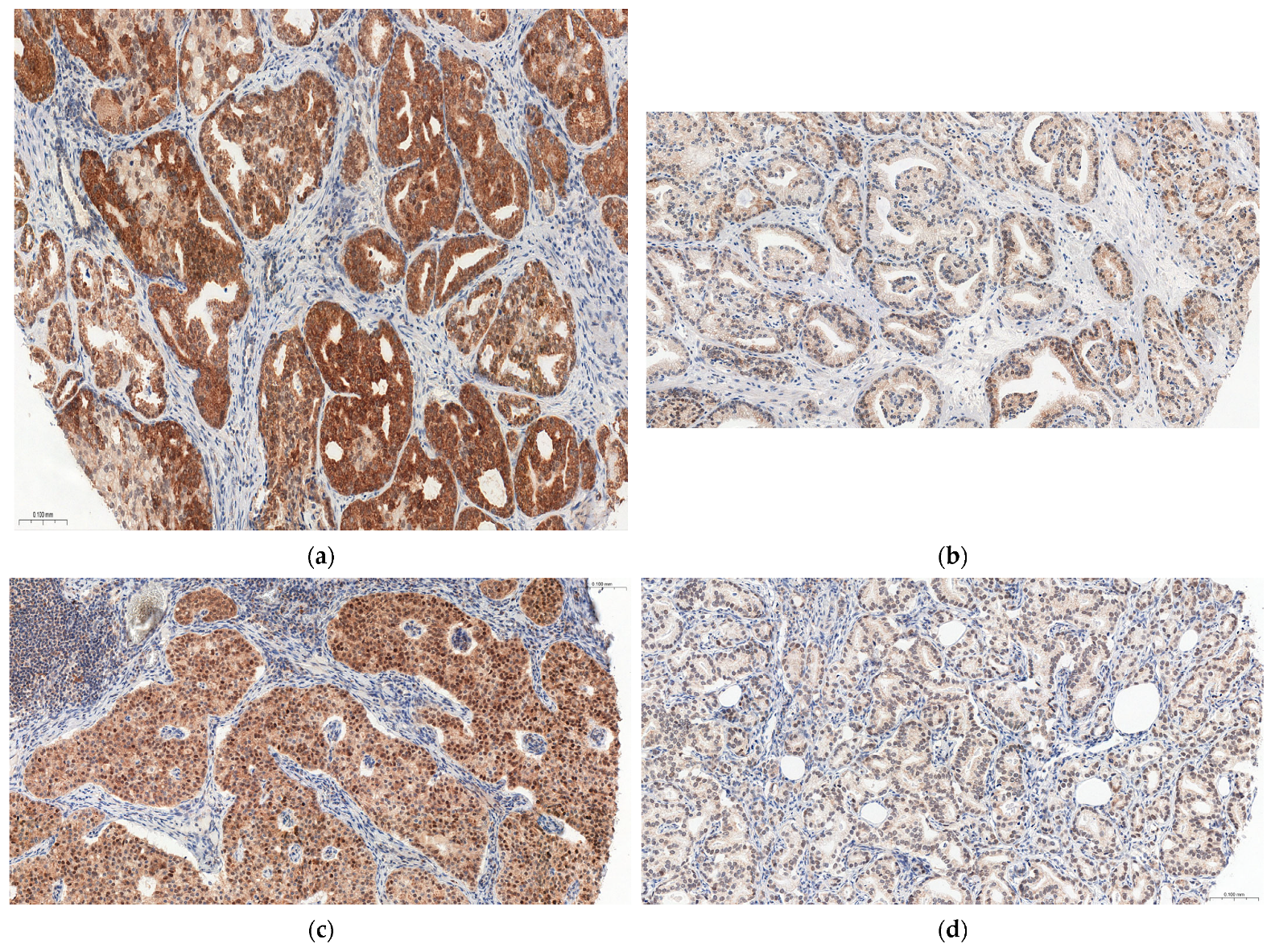
3. Results
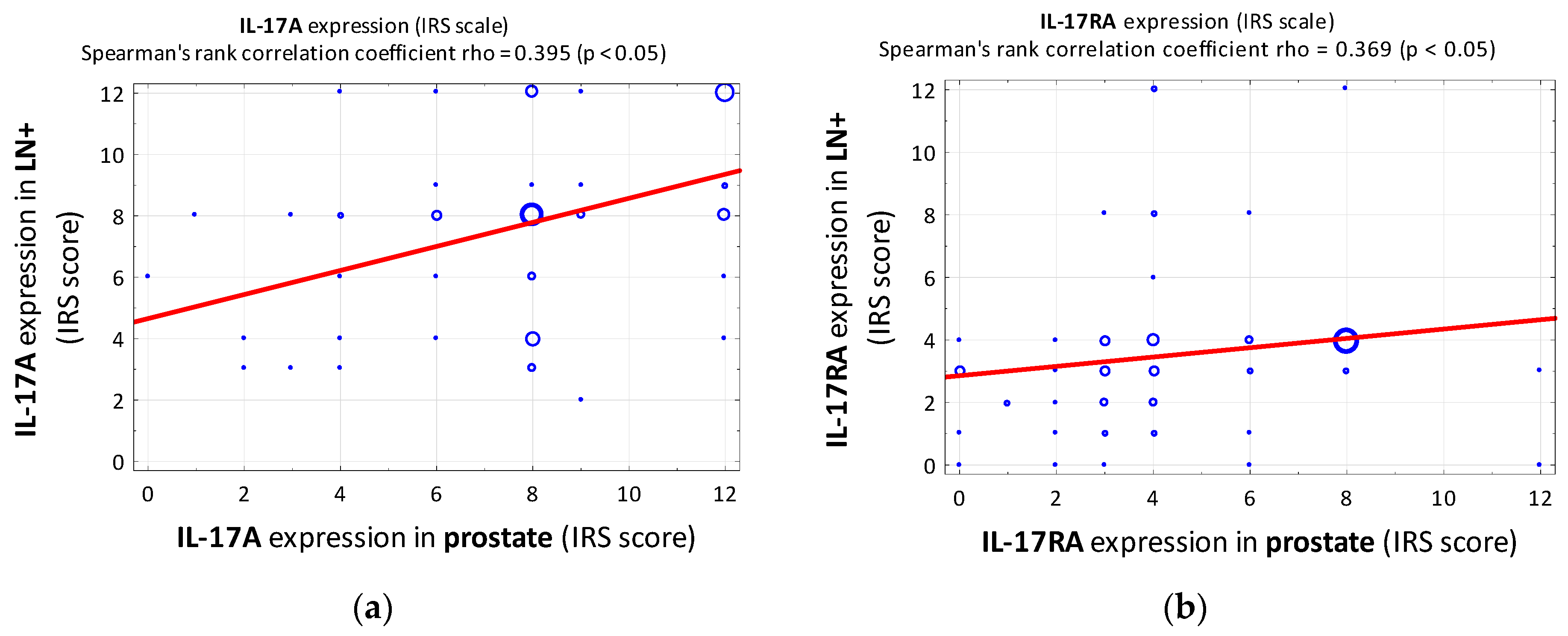
3.1. IL-17A
3.2. IL-17RA
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| EAU | European Association of Urology |
| ECE | extracapsular extension |
| EMT | epithelial-to-mesenchymal transition |
| GGG ISUP | International Society of Urological Pathology grade (group) system |
| HE | hematoxylin and eosin |
| IHC | immunohistochemistry |
| IL | interleukin |
| IRS | immunoreactive scale |
| LN+ | metastatic/positive lymph node |
| LVI | lymphovascular invasion |
| MMP7 | matrix metalloproteinase 7 |
| MSKCC | Memorial Sloan Kettering Cancer Center |
| NVI | neurovascular invasion |
| PCa | prostate cancer |
| PIN | prostatic intraepithelial neoplasia |
| PSA | prostate-specific antigen |
| pT | pathological tumor stage |
| RP | radical prostatectomy |
| TMA | tissue microarray |
| TURP | transurethral resection of the prostate |
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer Incidence and Mortality Worldwide: Sources, Methods and Major Patterns in GLOBOCAN 2012: Globocan 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Rawla, P. Epidemiology of Prostate Cancer. World J. Oncol. 2019, 10, 63–89. [Google Scholar] [CrossRef] [PubMed]
- Van Baelen, A.; Mottet, N.; Spahn, M.; Briganti, A.; Gontero, P.; Joniau, S. Sense and Nonsense of an Extended Pelvic Lymph Node Dissection in Prostate Cancer. Adv. Urol. 2012, 2012, 983058. [Google Scholar] [CrossRef]
- Allaf, M.E.; Palapattu, G.S.; Trock, B.J.; Carter, H.B.; Walsh, P.C. Anatomical Extent of Lymph Node Dissection: Impact on Men with Clinically Localized Prostate Cancer. J. Urol. 2004, 172, 1840–1844. [Google Scholar] [CrossRef]
- Stabile, A.; Pellegrino, A.; Mazzone, E.; Cannoletta, D.; de Angelis, M.; Barletta, F.; Scuderi, S.; Cucchiara, V.; Gandaglia, G.; Raggi, D.; et al. Can Negative Prostate-Specific Membrane Antigen Positron Emission Tomography/Computed Tomography Avoid the Need for Pelvic Lymph Node Dissection in Newly Diagnosed Prostate Cancer Patients? A Systematic Review and Meta-Analysis with Backup Histology as Reference Standard. Eur. Urol. Oncol. 2022, 5, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Hövels, A.M.; Heesakkers, R.A.M.; Adang, E.M.; Jager, G.J.; Strum, S.; Hoogeveen, Y.L.; Severens, J.L.; Barentsz, J.O. The Diagnostic Accuracy of CT and MRI in the Staging of Pelvic Lymph Nodes in Patients with Prostate Cancer: A Meta-Analysis. Clin. Radiol. 2008, 63, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Fossati, N.; Willemse, P.-P.M.; Van den Broeck, T.; van den Bergh, R.C.N.; Yuan, C.Y.; Briers, E.; Bellmunt, J.; Bolla, M.; Cornford, P.; De Santis, M.; et al. The Benefits and Harms of Different Extents of Lymph Node Dissection During Radical Prostatectomy for Prostate Cancer: A Systematic Review. Eur. Urol. 2017, 72, 84–109. [Google Scholar] [CrossRef]
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2021, 79, 243–262. [Google Scholar] [CrossRef]
- Ohshima, H.; Bartsch, H. Chronic Infections and Inflammatory Processes as Cancer Risk Factors: Possible Role of Nitric Oxide in Carcinogenesis. Mutat. Res. 1994, 305, 253–264. [Google Scholar] [CrossRef]
- Lonkar, P.; Dedon, P.C. Reactive Species and DNA Damage in Chronic Inflammation: Reconciling Chemical Mechanisms and Biological Fates. Int. J. Cancer 2011, 128, 1999–2009. [Google Scholar] [CrossRef]
- Schetter, A.J.; Heegaard, N.H.H.; Harris, C.C. Inflammation and Cancer: Interweaving MicroRNA, Free Radical, Cytokine and P53 Pathways. Carcinogenesis 2010, 31, 37–49. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Coussens, L.M.; Werb, Z. Inflammation and Cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Jung, G.; Kim, J.K.; Kim, H.; Lee, J.; Hong, S.K. The Association between Prostatitis and Risk of Prostate Cancer: A National Health Insurance Database Study. World J. Urol. 2022, 40, 2781–2787. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Li, J.; Yunxia, Z.; Zhu, H.; Liu, J.; Pumill, C. The Role of Prostatitis in Prostate Cancer: Meta-Analysis. PLoS ONE 2013, 8, e85179. [Google Scholar] [CrossRef] [PubMed]
- Sutcliffe, S.; Giovannucci, E.; De Marzo, A.M.; Leitzmann, M.F.; Willett, W.C.; Platz, E.A. Gonorrhea, Syphilis, Clinical Prostatitis, and the Risk of Prostate Cancer. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2160–2166. [Google Scholar] [CrossRef]
- Gurel, B.; Lucia, M.S.; Thompson, I.M.; Goodman, P.J.; Tangen, C.M.; Kristal, A.R.; Parnes, H.L.; Hoque, A.; Lippman, S.M.; Sutcliffe, S.; et al. Chronic Inflammation in Benign Prostate Tissue Is Associated with High-Grade Prostate Cancer in the Placebo Arm of the Prostate Cancer Prevention Trial. Cancer Epidemiol. Biomark. Prev. 2014, 23, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Bardia, A.; Platz, E.A.; Yegnasubramanian, S.; De Marzo, A.M.; Nelson, W.G. Anti-Inflammatory Drugs, Antioxidants, and Prostate Cancer Prevention. Curr. Opin. Pharmacol. 2009, 9, 419–426. [Google Scholar] [CrossRef][Green Version]
- Thapa, D.; Ghosh, R. Antioxidants for Prostate Cancer Chemoprevention: Challenges and Opportunities. Biochem. Pharmacol. 2012, 83, 1319–1330. [Google Scholar] [CrossRef]
- Kiełb, P.; Kowalczyk, K.; Gurwin, A.; Nowak, Ł.; Krajewski, W.; Sosnowski, R.; Szydełko, T.; Małkiewicz, B. Novel Histopathological Biomarkers in Prostate Cancer: Implications and Perspectives. Biomedicines 2023, 11, 1552. [Google Scholar] [CrossRef]
- Onishi, R.M.; Gaffen, S.L. Interleukin-17 and Its Target Genes: Mechanisms of Interleukin-17 Function in Disease. Immunology 2010, 129, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Liu, S.; Parajuli, K.R.; Zhang, W.; Zhang, K.; Mo, Z.; Liu, J.; Chen, Z.; Yang, S.; Wang, A.R.; et al. Interleukin-17 Promotes Prostate Cancer via MMP7-Induced Epithelial-to-Mesenchymal Transition. Oncogene 2017, 36, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Pappu, R.; Ramirez-Carrozzi, V.; Sambandam, A. The Interleukin-17 Cytokine Family: Critical Players in Host Defence and Inflammatory Diseases. Immunology 2011, 134, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Steiner, G.E.; Newman, M.E.; Paikl, D.; Stix, U.; Memaran-Dagda, N.; Lee, C.; Marberger, M.J. Expression and Function of Pro-Inflammatory Interleukin IL-17 and IL-17 Receptor in Normal, Benign Hyperplastic, and Malignant Prostate. Prostate 2003, 56, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Haudenschild, D.; Moseley, T.; Rose, L.; Reddi, A.H. Soluble and Transmembrane Isoforms of Novel Interleukin-17 Receptor-like Protein by RNA Splicing and Expression in Prostate Cancer. J. Biol. Chem. 2002, 277, 4309–4316. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhao, X.; Sun, X.; Li, Y.; Wang, Z.; Jiang, J.; Han, H.; Shen, W.; Corrigan, C.J.; Sun, Y. Expression of IL-17A, E, and F and Their Receptors in Human Prostatic Cancer: Comparison with Benign Prostatic Hyperplasia. Prostate 2015, 75, 1844–1856. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, S.; Ge, D.; Zhang, Q.; Xue, Y.; Xiong, Z.; Abdel-Mageed, A.B.; Myers, L.; Hill, S.M.; Rowan, B.G.; et al. Interleukin-17 Promotes Formation and Growth of Prostate Adenocarcinoma in Mouse Models. Cancer Res. 2012, 72, 2589–2599. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, S.; Zhang, Q.; Xiong, Z.; Wang, A.R.; Myers, L.; Melamed, J.; Tang, W.W.; You, Z. Interleukin-17 Promotes Development of Castration-Resistant Prostate Cancer Potentially through Creating an Immunotolerant and pro-Angiogenic Tumor Microenvironment. Prostate 2014, 74, 869–879. [Google Scholar] [CrossRef]
- Cunningham, D.; Zhang, Q.; Liu, S.; Parajuli, K.R.; Nie, Q.; Ma, L.; Zhang, A.; Chen, Z.; You, Z. Interleukin-17 Promotes Metastasis in an Immunocompetent Orthotopic Mouse Model of Prostate Cancer. Am. J. Clin. Exp. Urol. 2018, 6, 114–122. [Google Scholar]
- Janiczek, M.; Szylberg, Ł.; Antosik, P.; Kasperska, A.; Marszałek, A. Expression Levels of IL-17A, IL-17F, IL-17RA, and IL-17RC in Prostate Cancer with Taking into Account the Histological Grade According to Gleason Scale in Comparison to Benign Prostatic Hyperplasia: In Search of New Therapeutic Options. J. Immunol. Res. 2020, 2020, 4910595. [Google Scholar] [CrossRef]
- Remmele, W.; Stegner, H.E. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar]
- Kaemmerer, D.; Peter, L.; Lupp, A.; Schulz, S.; Sänger, J.; Baum, R.P.; Prasad, V.; Hommann, M. Comparing of IRS and Her2 as Immunohistochemical Scoring Schemes in Gastroenteropancreatic Neuroendocrine Tumors. Int. J. Clin. Exp. Pathol. 2012, 5, 187–194. [Google Scholar]
- Moussa, A.S.; Li, J.; Soriano, M.; Klein, E.A.; Dong, F.; Jones, J.S. Prostate Biopsy Clinical and Pathological Variables That Predict Significant Grading Changes in Patients with Intermediate and High Grade Prostate Cancer. BJU Int. 2009, 103, 43–48. [Google Scholar] [CrossRef]
- Hyun, Y.S.; Han, D.S.; Lee, A.R.; Eun, C.S.; Youn, J.; Kim, H.-Y. Role of IL-17A in the Development of Colitis-Associated Cancer. Carcinogenesis 2012, 33, 931–936. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yi, T.; Zhang, W.; Pardoll, D.M.; Yu, H. IL-17 Enhances Tumor Development in Carcinogen-Induced Skin Cancer. Cancer Res. 2010, 70, 10112–10120. [Google Scholar] [CrossRef]
- Jarocki, M.; Karska, J.; Kowalski, S.; Kiełb, P.; Nowak, Ł.; Krajewski, W.; Saczko, J.; Kulbacka, J.; Szydełko, T.; Małkiewicz, B. Interleukin 17 and Its Involvement in Renal Cell Carcinoma. J. Clin. Med. 2022, 11, 4973. [Google Scholar] [CrossRef] [PubMed]
- Benatar, T.; Cao, M.Y.; Lee, Y.; Li, H.; Feng, N.; Gu, X.; Lee, V.; Jin, H.; Wang, M.; Der, S.; et al. Virulizin Induces Production of IL-17E to Enhance Antitumor Activity by Recruitment of Eosinophils into Tumors. Cancer Immunol. Immunother. 2008, 57, 1757–1769. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2017, 9, 7204–7218. [Google Scholar] [CrossRef]
- Berraondo, P.; Sanmamed, M.F.; Ochoa, M.C.; Etxeberria, I.; Aznar, M.A.; Pérez-Gracia, J.L.; Rodríguez-Ruiz, M.E.; Ponz-Sarvise, M.; Castañón, E.; Melero, I. Cytokines in Clinical Cancer Immunotherapy. Br. J. Cancer 2019, 120, 6–15. [Google Scholar] [CrossRef]
- Locy, H.; de Mey, S.; de Mey, W.; De Ridder, M.; Thielemans, K.; Maenhout, S.K. Immunomodulation of the Tumor Microenvironment: Turn Foe Into Friend. Front. Immunol. 2018, 9, 2909. [Google Scholar] [CrossRef]
- Chen, K.; Kolls, J.K. Interluekin-17A (IL17A). Gene 2017, 614, 8–14. [Google Scholar] [CrossRef]
- Yan, R.; Dai, W.; Mao, Y.; Yu, G.; Li, W.; Shu, M.; Xu, B. Melittin Inhibits Tumor Cell Migration and Enhances Cisplatin Sensitivity by Suppressing IL-17 Signaling Pathway Gene LCN2 in Castration-Resistant Prostate Cancer. Prostate 2023, 1–16. [Google Scholar] [CrossRef]
- McDowell, K.L.; Begley, L.A.; Mor-Vaknin, N.; Markovitz, D.M.; Macoska, J.A. Leukocytic Promotion of Prostate Cellular Proliferation. Prostate 2010, 70, 377–389. [Google Scholar] [CrossRef] [PubMed]
- De Marzo, A.M.; Platz, E.A.; Sutcliffe, S.; Xu, J.; Grönberg, H.; Drake, C.G.; Nakai, Y.; Isaacs, W.B.; Nelson, W.G. Inflammation in Prostate Carcinogenesis. Nat. Rev. Cancer 2007, 7, 256–269. [Google Scholar] [CrossRef] [PubMed]
- Bardan, R.; Dumache, R.; Dema, A.; Cumpanas, A.; Bucuras, V. The Role of Prostatic Inflammation Biomarkers in the Diagnosis of Prostate Diseases. Clin. Biochem. 2014, 47, 909–915. [Google Scholar] [CrossRef]
- Stark, T.; Livas, L.; Kyprianou, N. Inflammation in Prostate Cancer Progression and Therapeutic Targeting. Transl. Androl. Urol. 2015, 4, 455–463. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, Q.; Chen, C.; Ge, D.; Qu, Y.; Chen, R.; Fan, Y.-M.; Li, N.; Tang, W.W.; Zhang, W.; et al. Hyperinsulinemia Enhances Interleukin-17-Induced Inflammation to Promote Prostate Cancer Development in Obese Mice through Inhibiting Glycogen Synthase Kinase 3-Mediated Phosphorylation and Degradation of Interleukin-17 Receptor. Oncotarget 2016, 7, 13651–13666. [Google Scholar] [CrossRef]
- Khanna, D.; Khanna, S.; Khanna, P.; Kahar, P.; Patel, B.M. Obesity: A Chronic Low-Grade Inflammation and Its Markers. Cureus 2022, 14, e22711. [Google Scholar] [CrossRef]
- Ellulu, M.S.; Patimah, I.; Khaza’ai, H.; Rahmat, A.; Abed, Y. Obesity and Inflammation: The Linking Mechanism and the Complications. Arch. Med. Sci. 2017, 13, 851–863. [Google Scholar] [CrossRef]
- Cohen, E.; Margalit, I.; Shochat, T.; Goldberg, E.; Krause, I. Markers of Chronic Inflammation in Overweight and Obese Individuals and the Role of Gender: A Cross-Sectional Study of a Large Cohort. J. Inflamm. Res. 2021, 14, 567–573. [Google Scholar] [CrossRef]
- Briganti, A.; Larcher, A.; Abdollah, F.; Capitanio, U.; Gallina, A.; Suardi, N.; Bianchi, M.; Sun, M.; Freschi, M.; Salonia, A.; et al. Updated Nomogram Predicting Lymph Node Invasion in Patients with Prostate Cancer Undergoing Extended Pelvic Lymph Node Dissection: The Essential Importance of Percentage of Positive Cores. Eur. Urol. 2012, 61, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Gandaglia, G.; Fossati, N.; Zaffuto, E.; Bandini, M.; Dell’Oglio, P.; Bravi, C.A.; Fallara, G.; Pellegrino, F.; Nocera, L.; Karakiewicz, P.I.; et al. Development and Internal Validation of a Novel Model to Identify the Candidates for Extended Pelvic Lymph Node Dissection in Prostate Cancer. Eur. Urol. 2017, 72, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Cimino, S.; Reale, G.; Castelli, T.; Favilla, V.; Giardina, R.; Russo, G.I.; Privitera, S.; Morgia, G. Comparison between Briganti, Partin and MSKCC Tools in Predicting Positive Lymph Nodes in Prostate Cancer: A Systematic Review and Meta-Analysis. Scand. J. Urol. 2017, 51, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Małkiewicz, B.; Ptaszkowski, K.; Knecht, K.; Gurwin, A.; Wilk, K.; Kiełb, P.; Dudek, K.; Zdrojowy, R. External Validation of the Briganti Nomogram to Predict Lymph Node Invasion in Prostate Cancer-Setting a New Threshold Value. Life 2021, 11, 479. [Google Scholar] [CrossRef] [PubMed]
- Gandaglia, G.; Ploussard, G.; Valerio, M.; Mattei, A.; Fiori, C.; Fossati, N.; Stabile, A.; Beauval, J.-B.; Malavaud, B.; Roumiguié, M.; et al. A Novel Nomogram to Identify Candidates for Extended Pelvic Lymph Node Dissection Among Patients with Clinically Localized Prostate Cancer Diagnosed with Magnetic Resonance Imaging-Targeted and Systematic Biopsies. Eur. Urol. 2019, 75, 506–514. [Google Scholar] [CrossRef]
- Suardi, N.; Ficarra, V.; Willemsen, P.; De Wil, P.; Gallina, A.; De Naeyer, G.; Schatteman, P.; Montorsi, F.; Carpentier, P.; Mottrie, A. Long-Term Biochemical Recurrence Rates After Robot-Assisted Radical Prostatectomy: Analysis of a Single-Center Series of Patients With a Minimum Follow-up of 5 Years. Urology 2012, 79, 133–138. [Google Scholar] [CrossRef]
- Detre, S.; Saclani Jotti, G.; Dowsett, M. A “Quickscore” Method for Immunohistochemical Semiquantitation: Validation for Oestrogen Receptor in Breast Carcinomas. J. Clin. Pathol. 1995, 48, 876–878. [Google Scholar] [CrossRef]
- Fedchenko, N.; Reifenrath, J. Different Approaches for Interpretation and Reporting of Immunohistochemistry Analysis Results in the Bone Tissue—A Review. Diagn. Pathol. 2014, 9, 221. [Google Scholar] [CrossRef]

| Immunoreactive Scale (IRS) | |||
| A—Percentage of Positive Cancer Cells | B—Staining Intensity | ||
| Score | Score | ||
| 0 | no cells with positive reaction | 0 | no color reaction |
| 1 | <10% cells with positive reaction | 1 | mild reaction |
| 2 | 10–50% cells with positive reaction | 2 | moderate reaction |
| 3 | 51–80% cells with positive reaction | 3 | intense reaction |
| 4 | >80% cells with positive reaction | ||
| IRS SCORE (A X B): 0–12 points | |||
| Final score | Level of expression | ||
| 1–7 | Low expression | ||
| 8–12 | High expression | ||
| Variable | Statistics |
|---|---|
| General characteristics of patients | |
| Age (years): | |
| M ± SD | 64.9 ± 5.5 |
| BMI (kg/m2): | |
| M ± SD | 28.1 ± 3.7 |
| Preoperative PSA (ng/mL): | |
| Me (Q1; Q3) | 19.8 (12; 36.1) |
| EAU risk group, n (%): | |
| Low-risk | 1 (1.3) |
| Intermediate-risk | 8 (10.4) |
| High-risk | 38 (49.3) |
| High-risk locally advanced | 30 (39) |
| Clinicopathological parameters | |
| pT, n (%): | |
| 2a | 1 (1.3) |
| 2c | 9 (11.7) |
| 3a | 14 (18.2) |
| 3b | 53 (68.8) |
| Postoperative Gleason, n (%): | |
| 3 + 3 | 1 (1.3) |
| 3 + 4 | 10 (13) |
| 3 + 5 | 4 (5.2) |
| 4 + 3 | 19 (24.7) |
| 4 + 4 | 3 (3.9) |
| 4 + 5 | 29 (37.6) |
| 5 + 3 | 2 (2.6) |
| 5 + 4 | 8 (10.4) |
| 5 + 5 | 1 (1.3) |
| Postoperative GGG ISUP, n (%): | |
| 1 | 1 (1.3) |
| 2 | 10 (13) |
| 3 | 19 (24.7) |
| 4 | 9 (11.7) |
| 5 | 38 (48.3) |
| Extracapsular extension of prostate, n (%): | |
| Yes | 66 (85.7) |
| No | 11 (14.3) |
| Extracapsular extension of lymph node, n (%): | |
| Yes | 19 (24.7) |
| No | 58 (75.3) |
| Resection margin, n (%): | |
| Positive | 54 (70.1) |
| Negative | 23 (29.9) |
| Neurovascular invasion, n (%): | |
| Yes | 70 (90.9) |
| No | 1 (1.3) |
| No data | 6 (7.8) |
| Lymphovascular invasion, n (%): | |
| Yes | 57 (74) |
| No | 15 (19.5) |
| No data | 5 (6.5) |
| Affected lymph nodes (%): | |
| Me (Q1; Q3) | 12.5 (8.3; 27.3) |
| Radical procedure, n (%): | |
| Yes | 36 (46.7) |
| No | 41 (53.3) |
| Expression (IRS Scale) | ||||||
|---|---|---|---|---|---|---|
| IL-17A | IL-17RA | |||||
| Prostate | Metastatic Lymph Node | p-Value | Prostate | Metastatic Lymph Node | p-Value | |
| A—Percentage of positively stained cancer cells (score) | 0.634 | 0.271 | ||||
| Me (Q1; Q3) | 4 [3; 4] | 4 [3; 4] | 3 [3; 4] | 3 [2; 4] | ||
| Min–Max | 0–4 | 2–4 | 0–4 | 0–4 | ||
| B—Intensity of staining (score) | 0.446 | 0.112 | ||||
| Me (Q1; Q3) | 2 [2; 3] | 2 [2; 3] | 1 [1; 2] | 1 [1; 1] | ||
| Min–Max | 0–3 | 1–3 | 0–3 | 0–3 | ||
| IRS score (A × B) | 0.415 | 0.009 | ||||
| Me (Q1; Q3) | 8 [6; 12] | 8 [6; 9] | 4 [3; 6] | 3 [2; 4] | ||
| Min–Max | 0–12 | 2–12 | 0–12 | 0–12 | ||
| Expression level: | 0.308 | 0.012 | ||||
| Low expression (1–7 score), n (%) | 19 (25) | 25 (32.5) | 52 (74.3) | 65 (90.3) | ||
| High expression (8–12 score), n (%) | 57 (75) | 52 (67.5) | 18 (25.7) | 7 (9.7) | ||
| IL-17A | IL-17RA | |||||||
|---|---|---|---|---|---|---|---|---|
| Prostate | Metastatic Lymph Node | Prostate | Metastatic Lymph Node | |||||
| rho | p | rho | p | rho | p | rho | p | |
| Preoperative PSA (ng/mL) | 0.033 | 0.778 | 0.027 | 0.813 | 0.057 | 0.623 | 0.100 | 0.387 |
| Affected lymph nodes (%) | −0.007 | 0.952 | 0.312 | 0.006 | 0.100 | 0.385 | 0.144 | 0.211 |
| Age (years) | −0.037 | 0.752 | −0.037 | 0.749 | 0.047 | 0.682 | 0.019 | 0.873 |
| BMI (kg/m2) | 0.251 | 0.028 | 0.013 | 0.912 | 0.096 | 0.404 | 0.079 | 0.494 |
| EAU risk group | 0.159 | 0.168 | 0.376 | 0.001 | 0.051 | 0.660 | 0.229 | 0.045 |
| Postoperative GGG ISUP | −0.020 | 0.862 | 0.146 | 0.205 | −0.111 | 0.335 | −0.023 | 0.841 |
| IL-17A Expression Level (IRS Score-Based) | |||||||
|---|---|---|---|---|---|---|---|
| Variables | Expression of IL-17A in PROSTATE | Expression of IL-17A in METASTATIC LYMPH NODE | |||||
| Level of Expression | p-Value | Level of Expression | p-Value | ||||
| Low (N = 19) | High (N = 57) | Low (N = 25) | High (N = 52) | ||||
| n (%) | n (%) | n (%) | n (%) | ||||
| pT | 3a and 3b | 15 (79.0%) | 51 (89.5%) | 0.257 | 20 (80.0%) | 47 (90.4%) | 0.279 |
| 2a and 2c | 4 (21.0%) | 6 (10.5%) | 5 (20.0%) | 5 (9.6%) | |||
| ECE of prostate | Yes | 16 (84.2%) | 49 (86.0%) | 1.000 | 18 (72.0%) | 48 (92.3%) | 0.033 |
| No | 3 (15.8%) | 8 (14.0%) | 7 (28.0%) | 4 (7.7%) | |||
| Resection margin | Positive | 11 (57.9%) | 42 (73.7%) | 0.251 | 18 (72.0%) | 36 (69.2%) | 0.986 |
| Negative | 8 (42.1%) | 15 (26.3%) | 7 (28.0%) | 16 (30.8%) | |||
| ECE of lymph node | Yes | 6 (31.6%) | 13 (22.8%) | 0.543 | 3 (12.0%) | 16 (30.8%) | 0.094 |
| No | 13 (68.4%) | 44 (77.2%) | 22 (88.0%) | 36 (69.2%) | |||
| NVI | Yes | 16 (100.0%) | 53 (98.2%) | 1.000 | 20 (95.2%) | 50 (100.0%) | 0.296 |
| No | 0 (0.0%) | 1 (1.8%) | 1 (4.8%) | 0 (0.0%) | |||
| LVI | Yes | 12 (80.0%) | 44 (78.6%) | 1.000 | 16 (69.6%) | 41 (83.7%) | 0.288 |
| No | 3 (20.0%) | 12 (21.4%) | 7 (30.4%) | 8 (16.3%) | |||
| Radical procedure | Yes | 7 (43.8%) | 28 (58.3%) | 0.389 | 12 (57.1%) | 24 (54.5%) | 0.944 |
| No | 9 (56.3%) | 20 (41.7%) | 9 (42.9%) | 20 (45.5%) | |||
| Expression of IL-17A in metastatic lymph node | Low | 8 (42.1%) | 16 (28.1%) | 0.287 | XX | XX | XX |
| High | 11 (57.9%) | 41 (71.9%) | XX | XX | |||
| Expression of IL-17A in prostate | Low | XX | XX | XX | 8 (33.3%) | 11 (21.2%) | 0.178 |
| High | XX | XX | 16 (66.7%) | 41 (78.8%) | |||
| IL-17RA Expression Level (IRS Score-Based) | |||||||
|---|---|---|---|---|---|---|---|
| Variables | Expression of IL-17RA in PROSTATE | Expression of IL-17RA in METASTATIC LYMPH NODE | |||||
| Level of Expression | p-Value | Level of Expression | p-Value | ||||
| Low (N = 52) | High (N = 18) | Low (N = 65) | High (N = 7) | ||||
| n (%) | n (%) | n (%) | n (%) | ||||
| pT | 3a and 3b | 45 (86.5%) | 16 (88.9%) | 1.000 | 57 (87.7%) | 6 (85.7%) | 1.000 |
| 2a and 2c | 7 (13.5%) | 2 (11.1%) | 8 (12.3%) | 1 (14.3%) | |||
| ECE of prostate | Yes | 47 (90.4%) | 14 (77.8%) | 0.222 | 58 (89.2%) | 5 (71.4%) | 0.209 |
| No | 5 (9.6%) | 4 (22.2%) | 7 (10.8%) | 2 (28.6%) | |||
| Resection margin | Positive | 37 (71.2%) | 13 (72.2%) | 1.000 | 45 (69.2%) | 6 (85.7%) | 0.665 |
| Negative | 15 (28.8%) | 5 (27.8%) | 20 (30.8%) | 1 (14.3%) | |||
| ECE of lymph node | Yes | 13 (25.0%) | 3 (16.7%) | 0.745 | 13 (20.0%) | 5 (71.4%) | 0.009 |
| No | 39 (75.0%) | 15 (83.3%) | 52 (80.0%) | 2 (28.6%) | |||
| NVI | Yes | 45 (97.8%) | 18 (100.0%) | 1.000 | 59 (100.0%) | 7 (100.0%) | 1.000 |
| No | 1 (2.2%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |||
| LVI | Yes | 40 (81.6%) | 12 (70.6%) | 0.491 | 48 (80.0%) | 6 (85.7%) | 1.000 |
| No | 9 (18.4%) | 5 (29.4%) | 12 (20.0%) | 1 (14.3%) | |||
| Radical procedure | Yes | 25 (58.1%) | 8 (50.0%) | 0.769 | 31 (57.4%) | 2 (33.3%) | 0.394 |
| No | 18 (41.9%) | 8 (50.0%) | 23 (42.6%) | 4 (66.7%) | |||
| Expression of IL-17RA in metastatic lymph node | Low | 43 (87.8%) | 16 (94.1%) | 0.667 | XX | XX | XX |
| High | 6 (12.2%) | 1 (5.9%) | XX | XX | |||
| Expression of IL-17RA in prostate | Low | XX | XX | XX | 43 (72.9%) | 6 (85.7%) | 0.667 |
| High | XX | XX | 16 (27.1%) | 1 (14.3%) | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kiełb, P.; Kaczorowski, M.; Kowalczyk, K.; Piotrowska, A.; Nowak, Ł.; Krajewski, W.; Chorbińska, J.; Dudek, K.; Dzięgiel, P.; Hałoń, A.; et al. Role of IL-17A and IL-17RA in Prostate Cancer with Lymph Nodes Metastasis: Expression Patterns and Clinical Significance. Cancers 2023, 15, 4578. https://doi.org/10.3390/cancers15184578
Kiełb P, Kaczorowski M, Kowalczyk K, Piotrowska A, Nowak Ł, Krajewski W, Chorbińska J, Dudek K, Dzięgiel P, Hałoń A, et al. Role of IL-17A and IL-17RA in Prostate Cancer with Lymph Nodes Metastasis: Expression Patterns and Clinical Significance. Cancers. 2023; 15(18):4578. https://doi.org/10.3390/cancers15184578
Chicago/Turabian StyleKiełb, Paweł, Maciej Kaczorowski, Kamil Kowalczyk, Aleksandra Piotrowska, Łukasz Nowak, Wojciech Krajewski, Joanna Chorbińska, Krzysztof Dudek, Piotr Dzięgiel, Agnieszka Hałoń, and et al. 2023. "Role of IL-17A and IL-17RA in Prostate Cancer with Lymph Nodes Metastasis: Expression Patterns and Clinical Significance" Cancers 15, no. 18: 4578. https://doi.org/10.3390/cancers15184578
APA StyleKiełb, P., Kaczorowski, M., Kowalczyk, K., Piotrowska, A., Nowak, Ł., Krajewski, W., Chorbińska, J., Dudek, K., Dzięgiel, P., Hałoń, A., Szydełko, T., & Małkiewicz, B. (2023). Role of IL-17A and IL-17RA in Prostate Cancer with Lymph Nodes Metastasis: Expression Patterns and Clinical Significance. Cancers, 15(18), 4578. https://doi.org/10.3390/cancers15184578








