Inter-Breath-Hold Geometric and Dosimetric Variations in Organs at Risk during Pancreatic Stereotactic Body Radiotherapy: Implications for Adaptive Radiation Therapy
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Radiotherapy Planning
2.3. OAR Dose Variability
2.4. Statistical Analysis
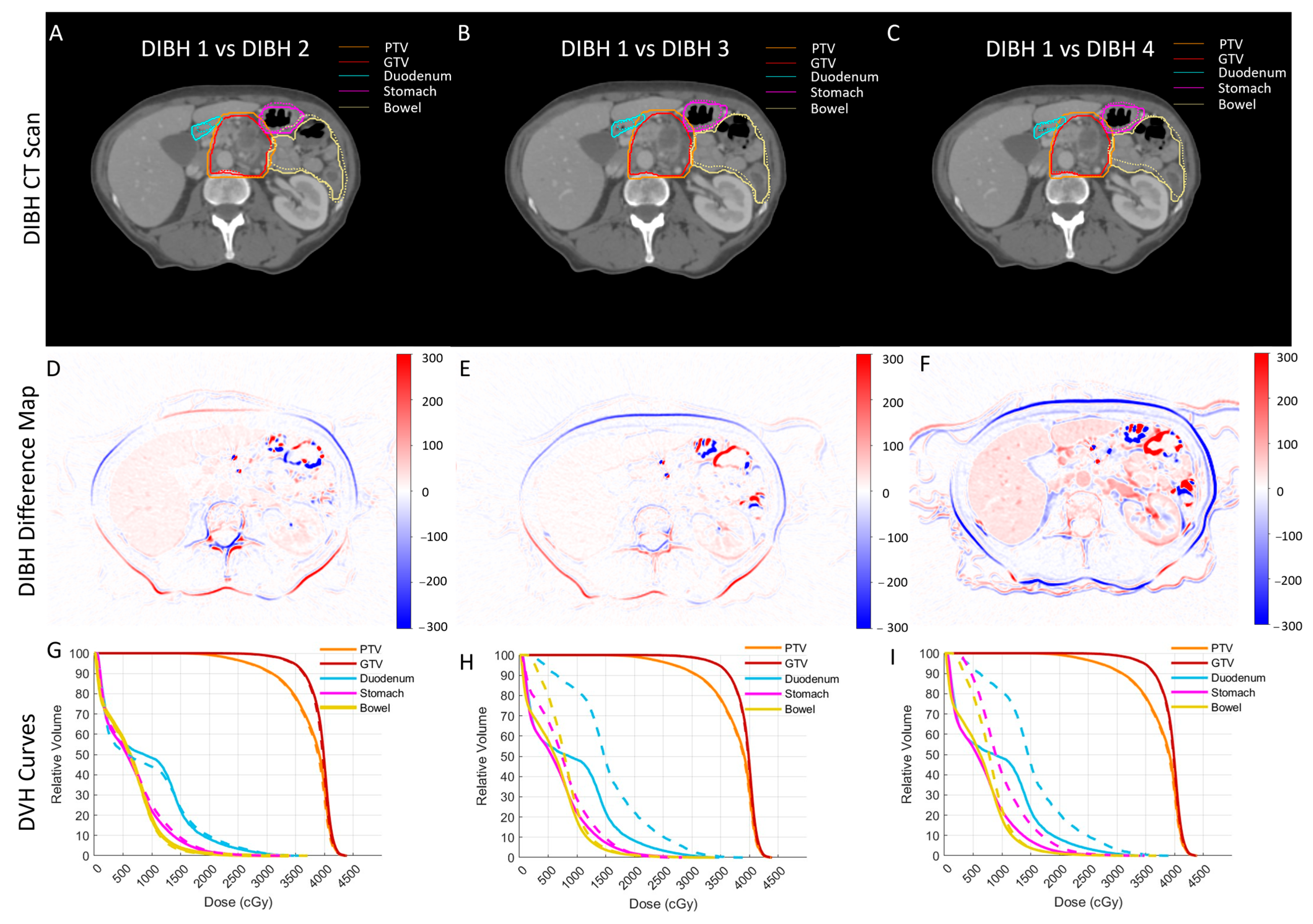
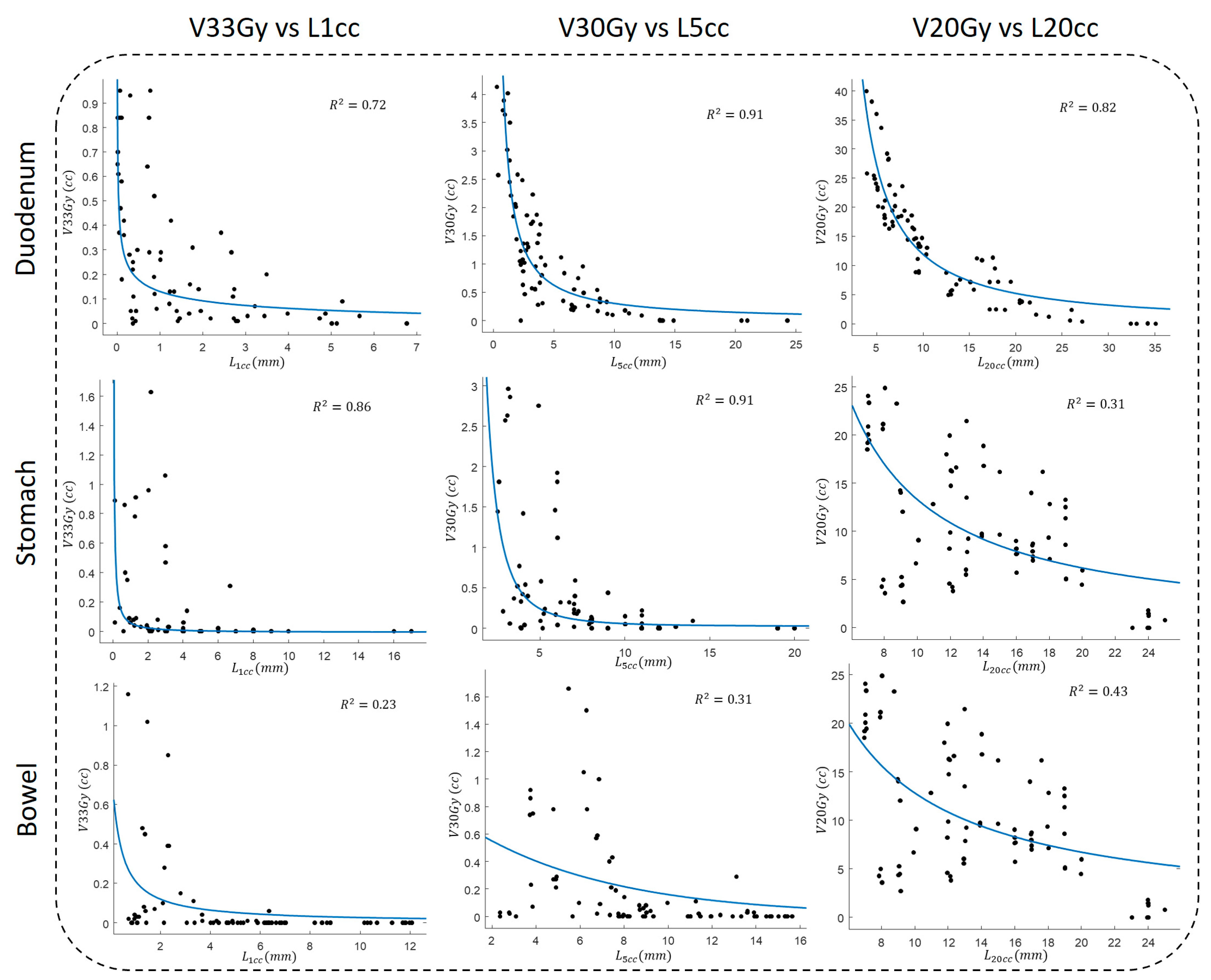
3. Results
3.1. Radiotherapy Planning Results
3.2. Variability Analysis
3.3. OVH–Dose Correlation Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef]
- Rudra, S.; Narang, A.K.; Pawlik, T.M.; Wang, H.; Jaffee, E.M.; Zheng, L.; Le, D.T.; Cosgrove, D.; Hruban, R.H.; Fishman, E.K. Evaluation of Predictive Variables in Locally Advanced Pancreatic Adenocarcinoma Patients Receiving Definitive Chemoradiation. Pract. Radiat. Oncol. 2012, 2, 77–85. [Google Scholar] [CrossRef][Green Version]
- Dholakia, A.S.; Hacker-Prietz, A.; Wild, A.T.; Raman, S.P.; Wood, L.D.; Huang, P.; Laheru, D.A.; Zheng, L.; De Jesus-Acosta, A.; Le, D.T. Resection of Borderline Resectable Pancreatic Cancer after Neoadjuvant Chemoradiation Does Not Depend on Improved Radiographic Appearance of Tumor–Vessel Relationships. J. Radiat. Oncol. 2013, 2, 413–425. [Google Scholar] [CrossRef]
- Sener, S.F.; Fremgen, A.; Menck, H.R.; Winchester, D.P. Pancreatic Cancer: A Report of Treatment and Survival Trends for 100,313 Patients Diagnosed from 1985–1995, Using the National Cancer Database. J. Am. Coll. Surg. 1999, 189, 1–7. [Google Scholar] [CrossRef]
- Herman, J.M.; Swartz, M.J.; Hsu, C.C.; Winter, J.; Pawlik, T.M.; Sugar, E.; Robinson, R.; Laheru, D.A.; Jaffee, E.; Hruban, R.H. Analysis of Fluorouracil-Based Adjuvant Chemotherapy and Radiation after Pancreaticoduodenectomy for Ductal Adenocarcinoma of the Pancreas: Results of a Large, Prospectively Collected Database at the Johns Hopkins Hospital. J. Clin. Oncol. 2008, 26, 3503. [Google Scholar] [CrossRef]
- Iacobuzio-Donahue, C.A.; Fu, B.; Yachida, S.; Luo, M.; Abe, H.; Henderson, C.M.; Vilardell, F.; Wang, Z.; Keller, J.W.; Banerjee, P. DPC4 Gene Status of the Primary Carcinoma Correlates with Patterns of Failure in Patients with Pancreatic Cancer. J. Clin. Oncol. 2009, 27, 1806. [Google Scholar] [CrossRef]
- Shin, S.H.; Kim, H.J.; Hwang, D.W.; Lee, J.H.; Song, K.B.; Jun, E.; Shim, I.K.; Hong, S.M.; Kim, H.J.; Park, K.M.; et al. The DPC4/SMAD4 Genetic Status Determines Recurrence Patterns and Treatment Outcomes in Resected Pancreatic Ductal Adenocarcinoma: A Prospective Cohort Study. Oncotarget 2017, 8, 17945–17959. [Google Scholar] [CrossRef]
- Rao, A.D.; Feng, Z.; Shin, E.J.; He, J.; Waters, K.M.; Coquia, S.; DeJong, R.; Rosati, L.M.; Su, L.; Li, D.; et al. A Novel Absorbable Radiopaque Hydrogel Spacer to Separate the Head of the Pancreas and Duodenum in Radiation Therapy for Pancreatic Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2017, 99, 1111–1120. [Google Scholar] [CrossRef]
- Hooshangnejad, H. Precision Medicine for Application of Hydrogel Spacer in Image-Guided Radiation Therapy; Johns Hopkins University: Baltimore, MD, USA, 2023. [Google Scholar]
- Hooshangnejad, H.; Youssefian, S.; Guest, J.K.; Ding, K. FEMOSSA: Patient-Specific Finite Element Simulation of the Prostate–Rectum Spacer Placement, a Predictive Model for Prostate Cancer Radiotherapy. Med. Phys. 2021, 48, 3438–3452. [Google Scholar] [CrossRef]
- Hooshangnejad, H.; Han-Oh, S.; Shin, E.J.; Narang, A.; Rao, A.D.; Lee, J.; McNutt, T.; Hu, C.; Wong, J.; Ding, K. Demonstrating the Benefits of Corrective Intra-Operative Feedback in Improving the Quality of Duodenal Hydrogel Spacer Placement. Med. Phys. 2022, 49, 4794–4803. [Google Scholar] [CrossRef]
- Hooshangnejad, H.; Youssefian, S.; Narang, A.; Shin, E.J.; Rao, A.D.; Han-Oh, S.; McNutt, T.; Lee, J.; Hu, C.; Wong, J.; et al. Finite Element-Based Personalized Simulation of Duodenal Hydrogel Spacer: Spacer Location Dependent Duodenal Sparing and a Decision Support System for Spacer-Enabled Pancreatic Cancer Radiation Therapy. Front. Oncol. 2022, 12, 833231. [Google Scholar] [CrossRef]
- Ben-Josef, E.; Shields, A.F.; Vaishampayan, U.; Vaitkevicius, V.; El-Rayes, B.F.; McDermott, P.; Burmeister, J.; Bossenberger, T.; Philip, P.A. Intensity-Modulated Radiotherapy (IMRT) and Concurrent Capecitabine for Pancreatic Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 454–459. [Google Scholar] [CrossRef]
- Li, J.; Ng, J.; Allendorf, J.; Saif, M.W. Locally Advanced Pancreatic Adenocarcinoma: Are We Making Progress? Highlights from the “2011 ASCO Annual Meeting”. Chicago, IL, USA; June 3-7, 2011. JOP 2011, 12, 347–350. [Google Scholar]
- Machtay, M.; Paulus, R.; Moughan, J.; Komaki, R.; Bradley, J.E.; Choy, H.; Albain, K.; Movsas, B.; Sause, W.T.; Curran, W.J. Defining Local-Regional Control and Its Importance in Locally Advanced Non-Small Cell Lung Carcinoma. J. Thorac. Oncol. 2012, 7, 716–722. [Google Scholar] [CrossRef]
- Berger, A.C.; Garcia Jr, M.; Hoffman, J.P.; Regine, W.F.; Abrams, R.A.; Safran, H.; Konski, A.; Benson 3rd, A.B.; MacDonald, J.; Willett, C.G. Postresection CA 19-9 Predicts Overall Survival in Patients with Pancreatic Cancer Treated with Adjuvant Chemoradiation: A Prospective Validation by RTOG 9704. J. Clin. Oncol. 2008, 26, 5918–5922. [Google Scholar] [CrossRef]
- Bruynzeel, A.M.E.; Lagerwaard, F.J. The Role of Biological Dose-Escalation for Pancreatic Cancer. Clin. Transl. Radiat. Oncol. 2019, 18, 128–130. [Google Scholar] [CrossRef]
- Zaorsky, N.G.; Lehrer, E.J.; Handorf, E.; Meyer, J.E. Dose Escalation in Stereotactic Body Radiation Therapy for Pancreatic Cancer: A Meta-Analysis. Am J. Clin. Oncol. 2019, 42, 46–55. [Google Scholar] [CrossRef]
- Zhu, X.; Cao, Y.; Su, T.; Zhu, X.; Ju, X.; Zhao, X.; Jiang, L.; Ye, Y.; Cao, F.; Qing, S.; et al. Failure Patterns and Outcomes of Dose Escalation of Stereotactic Body Radiotherapy for Locally Advanced Pancreatic Cancer: A Multicenter Cohort Study. Ther. Adv. Med. Oncol. 2020, 12, 1758835920977155. [Google Scholar] [CrossRef]
- Rudra, S.; Jiang, N.; Rosenberg, S.A.; Olsen, J.R.; Roach, M.C.; Wan, L.; Portelance, L.; Mellon, E.A.; Bruynzeel, A.; Lagerwaard, F.; et al. Using Adaptive Magnetic Resonance Image-Guided Radiation Therapy for Treatment of Inoperable Pancreatic Cancer. Cancer Med. 2019, 8, 2123–2132. [Google Scholar] [CrossRef]
- Kerdsirichairat, T.; Narang, A.K.; Thompson, E.; Kim, S.-H.; Rao, A.; Ding, K.; Shin, E.J. Feasibility of Using Hydrogel Spacers for Borderline-Resectable and Locally Advanced Pancreatic Tumors. Gastroenterology 2019, 157, 933–935. [Google Scholar] [CrossRef]
- Rao, A.D.; Shin, E.J.; Meyer, J.; Thompson, E.L.; Fu, W.; Hu, C.; Fishman, E.K.; Weiss, M.; Wolfgang, C.; Burkhart, R.; et al. Evaluation of a Novel Absorbable Radiopaque Hydrogel in Patients Undergoing Image Guided Radiation Therapy for Borderline Resectable and Locally Advanced Pancreatic Adenocarcinoma. Pract. Radiat. Oncol. 2020, 10, e508–e513. [Google Scholar] [CrossRef] [PubMed]
- Artinyan, A.; Soriano, P.A.; Prendergast, C.; Low, T.; Ellenhorn, J.D.I.; Kim, J. The Anatomic Location of Pancreatic Cancer Is a Prognostic Factor for Survival. HPB 2008, 10, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Ravanini, G.D.A.G.; De Almeida, S.B.; Benevenuto, D.S.; Da Silva, A.D.C.F.; De Lima, F.N.R.; Nascimento, B.E. Robotic Modified Appleby: Case Report, Technical Aspects and Video. Int. J. Surg. Case Rep. 2022, 95, 107266. [Google Scholar] [CrossRef]
- Kensen, C.M.; Janssen, T.M.; Betgen, A.; Wiersema, L.; Peters, F.P.; Remeijer, P.; Marijnen, C.A.M.; van der Heide, U.A. Effect of Intrafraction Adaptation on PTV Margins for MRI Guided Online Adaptive Radiotherapy for Rectal Cancer. Radiat. Oncol. 2022, 17, 110. [Google Scholar] [CrossRef]
- Hooshangnejad, H.; Han, D.; Feng, Z.; Dong, L.; Sun, E.; Du, K.; Ding, K. Systematic Study of the Iodinated Rectal Hydrogel Spacer Material Discrepancy on Accuracy of Proton Dosimetry. J. Appl. Clin. Med. Phys. 2022, 23, e13774. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, W.D.; Naqvi, S.A.; Cedric, X.Y. Real-Time Intra-Fraction-Motion Tracking Using the Treatment Couch: A Feasibility Study. Phys. Med. Biol. 2005, 50, 4021. [Google Scholar] [CrossRef]
- Yi, B.Y.; Han-Oh, S.; Lerma, F.; Berman, B.L.; Yu, C. Real-time Tumor Tracking with Preprogrammed Dynamic Multileaf-collimator Motion and Adaptive Dose-rate Regulation. Med. Phys. 2008, 35, 3955–3962. [Google Scholar] [CrossRef]
- Keall, P.J.; Nguyen, D.T.; O’Brien, R.; Caillet, V.; Hewson, E.; Poulsen, P.R.; Bromley, R.; Bell, L.; Eade, T.; Kneebone, A. The First Clinical Implementation of Real-Time Image-Guided Adaptive Radiotherapy Using a Standard Linear Accelerator. Radiother. Oncol. 2018, 127, 6–11. [Google Scholar] [CrossRef]
- Ohara, K.; Okumura, T.; Akisada, M.; Inada, T.; Mori, T.; Yokota, H.; Calaguas, M.J.B. Irradiation Synchronized with Respiration Gate. Int. J. Radiat. Oncol. Biol. Phys. 1989, 17, 853–857. [Google Scholar] [CrossRef]
- Hanley, J.; Debois, M.M.; Mah, D.; Mageras, G.S.; Raben, A.; Rosenzweig, K.; Mychalczak, B.; Schwartz, L.H.; Gloeggler, P.J.; Lutz, W. Deep Inspiration Breath-Hold Technique for Lung Tumors: The Potential Value of Target Immobilization and Reduced Lung Density in Dose Escalation. Int. J. Radiat. Oncol. Biol. Phys. 1999, 45, 603–611. [Google Scholar] [CrossRef]
- Han-Oh, S.; Hill, C.; Kang-Hsin Wang, K.; Ding, K.; Wright, J.L.; Alcorn, S.; Meyer, J.; Herman, J.; Narang, A. Geometric Reproducibility of Fiducial Markers and Efficacy of a Patient-Specific Margin Design Using Deep Inspiration Breath Hold for Stereotactic Body Radiation Therapy for Pancreatic Cancer. Adv. Radiat. Oncol. 2021, 6, 100655. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.S.; Han-Oh, S.; Cheng, Z.; Wang, K.K.-H.; Meyer, J.J.; Herman, J.M.; Narang, A.K. Fiducial-Based Image-Guided SBRT for Pancreatic Adenocarcinoma: Does Inter-and Intra-Fraction Treatment Variation Warrant Adaptive Therapy? Radiat. Oncol. 2021, 16, 53. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.J.; Martin, D.; Whyte, R.; Hai, J.; Ozhasoglu, C.; Le, Q.-T. The Effectiveness of Breath-Holding to Stabilize Lung and Pancreas Tumors during Radiosurgery. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Teboh, R.F.; Srinivasan, S.; Ng, S.P.; Aliru, M.L.; Herman, J.M. Setup Management for Stereotactic Body Radiation Therapy of Patients with Pancreatic Cancer Treated via the Breath-Hold Technique. Pract. Radiat. Oncol. 2020, 10, e280–e289. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Rao, A.D.; Cheng, Z.; Shin, E.J.; Moore, J.; Su, L.; Kim, S.-H.; Wong, J.; Narang, A.; Herman, J.M.; et al. Dose Prediction Model for Duodenum Sparing with a Biodegradable Hydrogel Spacer for Pancreatic Cancer Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 651–659. [Google Scholar] [CrossRef]
- Yang, Y.; Ford, E.C.; Wu, B.; Pinkawa, M.; van Triest, B.; Campbell, P.; Song, D.Y.; McNutt, T.R. An Overlap-Volume-Histogram Based Method for Rectal Dose Prediction and Automated Treatment Planning in the External Beam Prostate Radiotherapy Following Hydrogel Injection. Med. Phys. 2013, 40, 11709. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Pang, D.; Lei, S.; Gatti, J.; Tong, M.; McNutt, T.; Kole, T.; Dritschilo, A.; Collins, S. Improved Robotic Stereotactic Body Radiation Therapy Plan Quality and Planning Efficacy for Organ-Confined Prostate Cancer Utilizing Overlap-Volume Histogram-Driven Planning Methodology. Radiother. Oncol. 2014, 112, 221–226. [Google Scholar] [CrossRef]
- Wu, B.; Ricchetti, F.; Sanguineti, G.; Kazhdan, M.; Simari, P.; Jacques, R.; Taylor, R.; McNutt, T. Data-Driven Approach to Generating Achievable Dose-Volume Histogram Objectives in Intensity-Modulated Radiotherapy Planning. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; McNutt, T.; Zahurak, M.; Simari, P.; Pang, D.; Taylor, R.; Sanguineti, G. Fully Automated Simultaneous Integrated Boosted-Intensity Modulated Radiation Therapy Treatment Planning Is Feasible for Head-and-Neck Cancer: A Prospective Clinical Study. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, e647–e653. [Google Scholar] [CrossRef]
- Hooshangnejad, H.; Chen, Q.; Feng, X.; Zhang, R.; Farjam, R.; Voong, K.R.; Hales, R.K.; Du, Y.; Jia, X.; Ding, K. DAART: A Deep Learning Platform for Deeply Accelerated Adaptive Radiation Therapy for Lung Cancer. Front. Oncol. 2023, 13, 1201679. [Google Scholar] [CrossRef]
- Lens, E.; van der Horst, A.; Versteijne, E.; Bel, A.; van Tienhoven, G. Considerable Pancreatic Tumor Motion during Breath-Holding. Acta Oncol. 2016, 55, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Jayachandran, P.; Minn, A.Y.; Van Dam, J.; Norton, J.A.; Koong, A.C.; Chang, D.T. Interfractional Uncertainty in the Treatment of Pancreatic Cancer with Radiation. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Hooshangnejad, H.; Chen, Q.; Feng, X.; Zhang, R.; Ding, K. DeepPERFECT: Novel Deep Learning CT Synthesis Method for Expeditious Pancreatic Cancer Radiotherapy. Cancers 2023, 15, 3061. [Google Scholar] [CrossRef]
- Kawaguchi, H.; Demizu, Y.; Mukumoto, N.; Ishihara, T.; Miyawaki, D.; Komatsu, S.; Akasaka, H.; Shinoto, M.; Shioyama, Y.; Nakamura, K.; et al. Efficacy of Spacers in Radiation Therapy for Locally Advanced Pancreatic Cancer: A Planning Study. Anticancer Res. 2021, 41, 503–508. [Google Scholar] [CrossRef]
- Kim, S.-H.; Ding, K.; Rao, A.; He, J.; Bhutani, M.S.; Herman, J.M.; Narang, A.; Shin, E.J. EUS-Guided Hydrogel Microparticle Injection in a Cadaveric Model. J. Appl. Clin. Med. Phys. 2021, 22, 83–91. [Google Scholar] [CrossRef]
- Huang, X.; Hooshangnejad, H.; China, D.; Feng, Z.; Lee, J.; Bell, M.A.L.; Ding, K. Ultrasound Imaging with Flexible Array Transducer for Pancreatic Cancer Radiation Therapy. Cancers 2023, 15, 3294. [Google Scholar] [CrossRef]
- Winkel, D.; Bol, G.H.; Kroon, P.S.; van Asselen, B.; Hackett, S.S.; Werensteijn-Honingh, A.M.; Intven, M.P.W.; Eppinga, W.S.C.; Tijssen, R.H.N.; Kerkmeijer, L.G.W. Adaptive Radiotherapy: The Elekta Unity MR-Linac Concept. Clin. Transl. Radiat. Oncol. 2019, 18, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.A.; Paulson, E.S.; van der Heide, U.A.; Fuller, C.D.; Raaymakers, B.W.; Lagendijk, J.J.W.; Li, X.A.; Jaffray, D.A.; Dawson, L.A.; Erickson, B. The Transformation of Radiation Oncology Using Real-Time Magnetic Resonance Guidance: A Review. Eur. J. Cancer 2019, 122, 42–52. [Google Scholar] [CrossRef]
- Acharya, S.; Fischer-Valuck, B.W.; Kashani, R.; Parikh, P.; Yang, D.; Zhao, T.; Green, O.; Wooten, O.; Li, H.H.; Hu, Y. Online Magnetic Resonance Image Guided Adaptive Radiation Therapy: First Clinical Applications. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 394–403. [Google Scholar] [CrossRef]
- De Mol Van Otterloo, S.R.; Christodouleas, J.P.; Blezer, E.L.A.; Akhiat, H.; Brown, K.; Choudhury, A.; Eggert, D.; Erickson, B.A.; Daamen, L.A.; Faivre-Finn, C. Patterns of Care, Tolerability, and Safety of the First Cohort of Patients Treated on a Novel High-Field MR-Linac Within the MOMENTUM Study: Initial Results from a Prospective Multi-Institutional Registry. Int. J. Radiat. Oncol. Biol. Phys. 2021, 111, 867–875. [Google Scholar] [CrossRef]
- Krishnan, S.; Chadha, A.S.; Suh, Y.; Chen, H.-C.; Rao, A.; Das, P.; Minsky, B.D.; Mahmood, U.; Delclos, M.E.; Sawakuchi, G.O.; et al. Focal Radiation Therapy Dose Escalation Improves Overall Survival in Locally Advanced Pancreatic Cancer Patients Receiving Induction Chemotherapy and Consolidative Chemoradiation. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Bryant, J.M.; Weygand, J.; Keit, E.; Cruz-Chamorro, R.; Sandoval, M.L.; Oraiqat, I.M.; Andreozzi, J.; Redler, G.; Latifi, K.; Feygelman, V. Stereotactic Magnetic Resonance-Guided Adaptive and Non-Adaptive Radiotherapy on Combination MR-Linear Accelerators: Current Practice and Future Directions. Cancers 2023, 15, 2081. [Google Scholar] [CrossRef] [PubMed]
- Tchelebi, L.T.; Zaorsky, N.G.; Rosenberg, J.C.; Sharma, N.K.; Tuanquin, L.C.; Mackley, H.B.; Ellis, R.J. Reducing the Toxicity of Radiotherapy for Pancreatic Cancer with Magnetic Resonance-Guided Radiotherapy. Toxicol. Sci. 2020, 175, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Bohoudi, O.; Bruynzeel, A.M.E.; Senan, S.; Cuijpers, J.P.; Slotman, B.J.; Lagerwaard, F.J.; Palacios, M.A. Fast and Robust Online Adaptive Planning in Stereotactic MR-Guided Adaptive Radiation Therapy (SMART) for Pancreatic Cancer. Radiother. Oncol. 2017, 125, 439–444. [Google Scholar] [CrossRef]
- Henke, L.; Kashani, R.; Yang, D.; Zhao, T.; Green, O.; Olsen, L.; Rodriguez, V.; Wooten, H.O.; Li, H.H.; Hu, Y. Simulated Online Adaptive Magnetic Resonance–Guided Stereotactic Body Radiation Therapy for the Treatment of Oligometastatic Disease of the Abdomen and Central Thorax: Characterization of Potential Advantages. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 1078–1086. [Google Scholar] [CrossRef]
- Feldman, A.M.; Modh, A.; Glide-Hurst, C.; Chetty, I.J.; Movsas, B. Real-Time Magnetic Resonance-Guided Liver Stereotactic Body Radiation Therapy: An Institutional Report Using a Magnetic Resonance-Linac System. Cureus 2019, 11, e5774. [Google Scholar] [CrossRef]
- Rosenberg, S.A.; Henke, L.E.; Shaverdian, N.; Mittauer, K.; Wojcieszynski, A.P.; Hullett, C.R.; Kamrava, M.; Lamb, J.; Cao, M.; Green, O.L. A Multi-Institutional Experience of MR-Guided Liver Stereotactic Body Radiation Therapy. Adv. Radiat. Oncol. 2019, 4, 142–149. [Google Scholar] [CrossRef]
- Boldrini, L.; Cellini, F.; Manfrida, S.; Chiloiro, G.; Teodoli, S.; Cusumano, D.; Fionda, B.; Mattiucci, G.C.; De Gaetano, A.M.; Azario, L. Use of Indirect Target Gating in Magnetic Resonance-Guided Liver Stereotactic Body Radiotherapy: Case Report of an Oligometastatic Patient. Cureus 2018, 10, e2292. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hooshangnejad, H.; Miles, D.; Hill, C.; Narang, A.; Ding, K.; Han-Oh, S. Inter-Breath-Hold Geometric and Dosimetric Variations in Organs at Risk during Pancreatic Stereotactic Body Radiotherapy: Implications for Adaptive Radiation Therapy. Cancers 2023, 15, 4332. https://doi.org/10.3390/cancers15174332
Hooshangnejad H, Miles D, Hill C, Narang A, Ding K, Han-Oh S. Inter-Breath-Hold Geometric and Dosimetric Variations in Organs at Risk during Pancreatic Stereotactic Body Radiotherapy: Implications for Adaptive Radiation Therapy. Cancers. 2023; 15(17):4332. https://doi.org/10.3390/cancers15174332
Chicago/Turabian StyleHooshangnejad, Hamed, Devin Miles, Colin Hill, Amol Narang, Kai Ding, and Sarah Han-Oh. 2023. "Inter-Breath-Hold Geometric and Dosimetric Variations in Organs at Risk during Pancreatic Stereotactic Body Radiotherapy: Implications for Adaptive Radiation Therapy" Cancers 15, no. 17: 4332. https://doi.org/10.3390/cancers15174332
APA StyleHooshangnejad, H., Miles, D., Hill, C., Narang, A., Ding, K., & Han-Oh, S. (2023). Inter-Breath-Hold Geometric and Dosimetric Variations in Organs at Risk during Pancreatic Stereotactic Body Radiotherapy: Implications for Adaptive Radiation Therapy. Cancers, 15(17), 4332. https://doi.org/10.3390/cancers15174332









