Global Impact of Monoclonal Antibodies (mAbs) in Children: A Focus on Anti-GD2
Simple Summary
Abstract
1. Introduction
2. Monoclonal Antibodies and Pediatric Cancer
2.1. Monoclonal Antibodies in Pediatric Hematological Malignancies
2.2. Monoclonal Antibodies Specifically Developed for Pediatric Solid Tumors
Anti-GD2 Monoclonal Antibodies
2.3. Monoclonal Antibodies Repurposed for Pediatric Solid Tumors
3. Monoclonal Antibodies for Childhood Cancer: Current Limitations and Future Strategies
3.1. Biological Limitations
3.1.1. Paucity of Clinically Relevant Targets
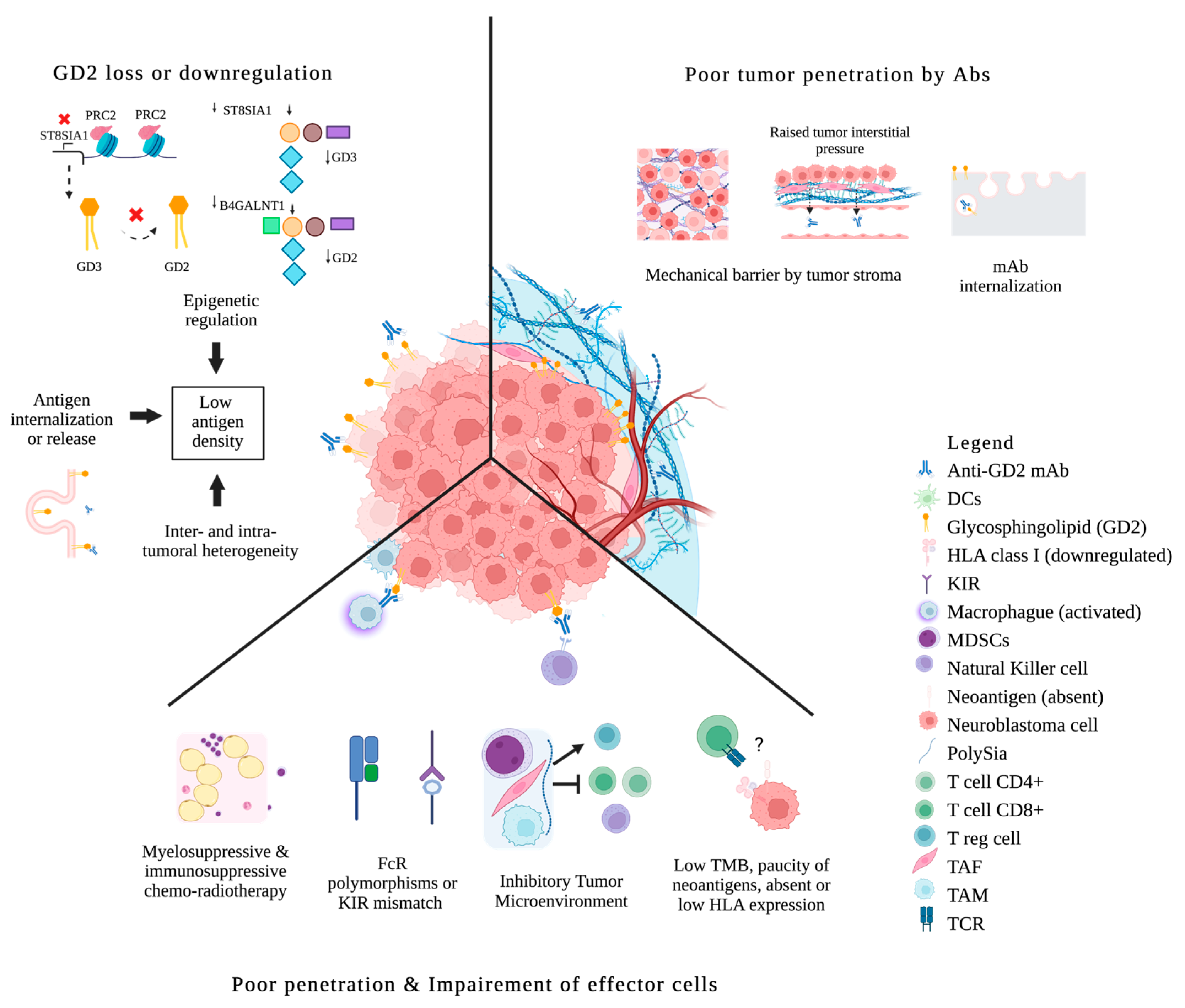
3.1.2. Antigen Loss or Downregulation under Immune Pressure
3.1.3. Poor Tumor Penetration
Drug Conjugates
Radio-Immuno-Conjugates
Drug Delivery Platforms
3.1.4. Insufficient or Impaired Effector Functions
| Mechanisms of Anti-GD2 Resistance | Strategies to Overcome Them |
|---|---|
| Antigen loss or downregulation by epigenetic modulation | EZH2 inhibition [113,114,115] |
| Poor tumor penetration | Increased payload: Antibody-drug-conjugates [121] Radio-immunotherapy conjugates [122,123] Drug delivery platforms [124] |
| Impaired effector functions | Fc engineering [120,137,138] Engaging T-cells by bispecific antibodies [139,140,141,142] Co-administration with certain cytokines [136] or immuno-conjugates [136,147,148] Co-administration with granulocyte-macrophage colony-stimulating factor GM-CSF [50,53,133,134] Early administration of antibody within the cytotoxic therapeutic plan [150] GD2 conjugated vaccines [152,153] |
3.1.5. Lack of Biomarkers to Predict Response and Survival
3.2. Difficult Integration into the Standards of Care
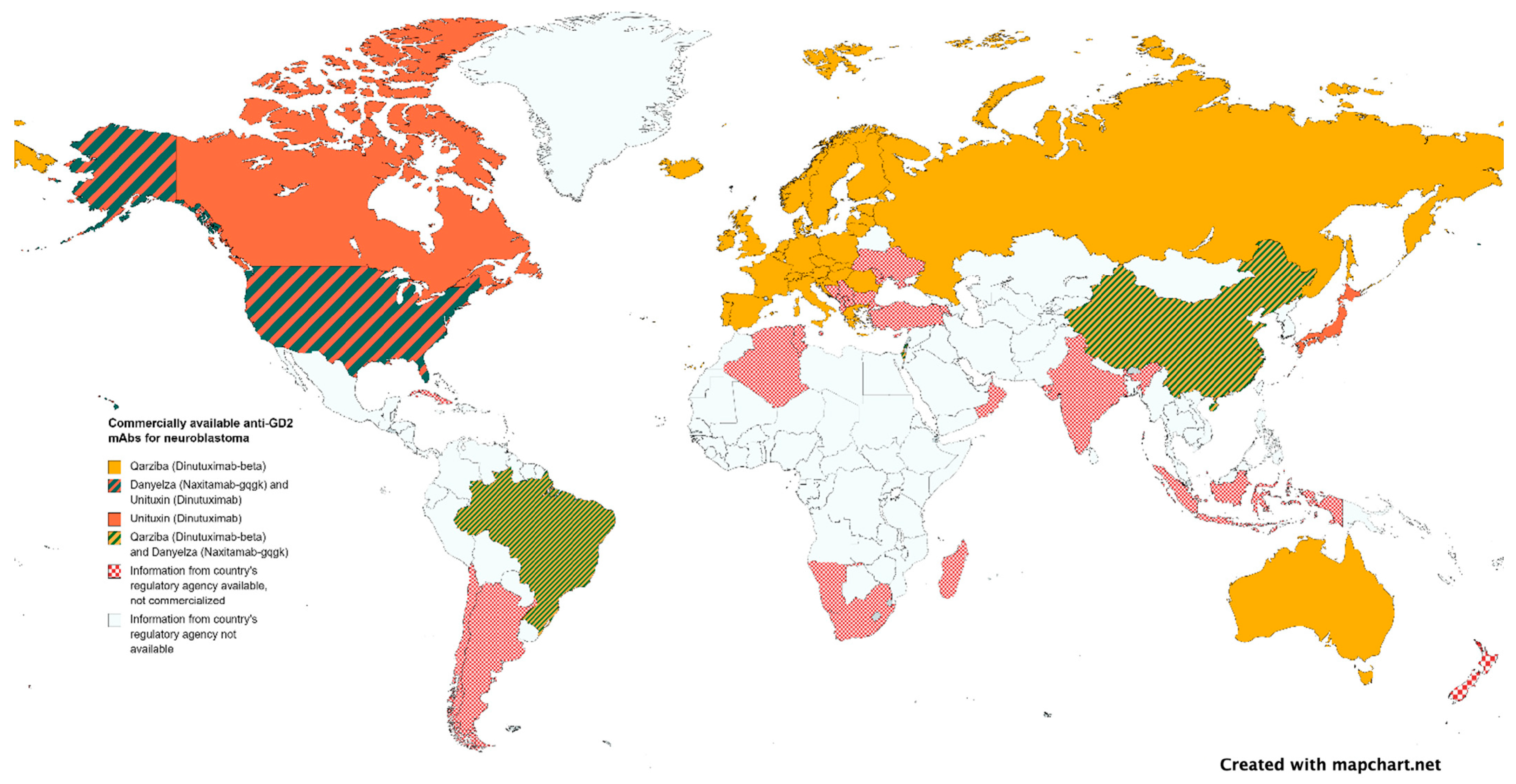
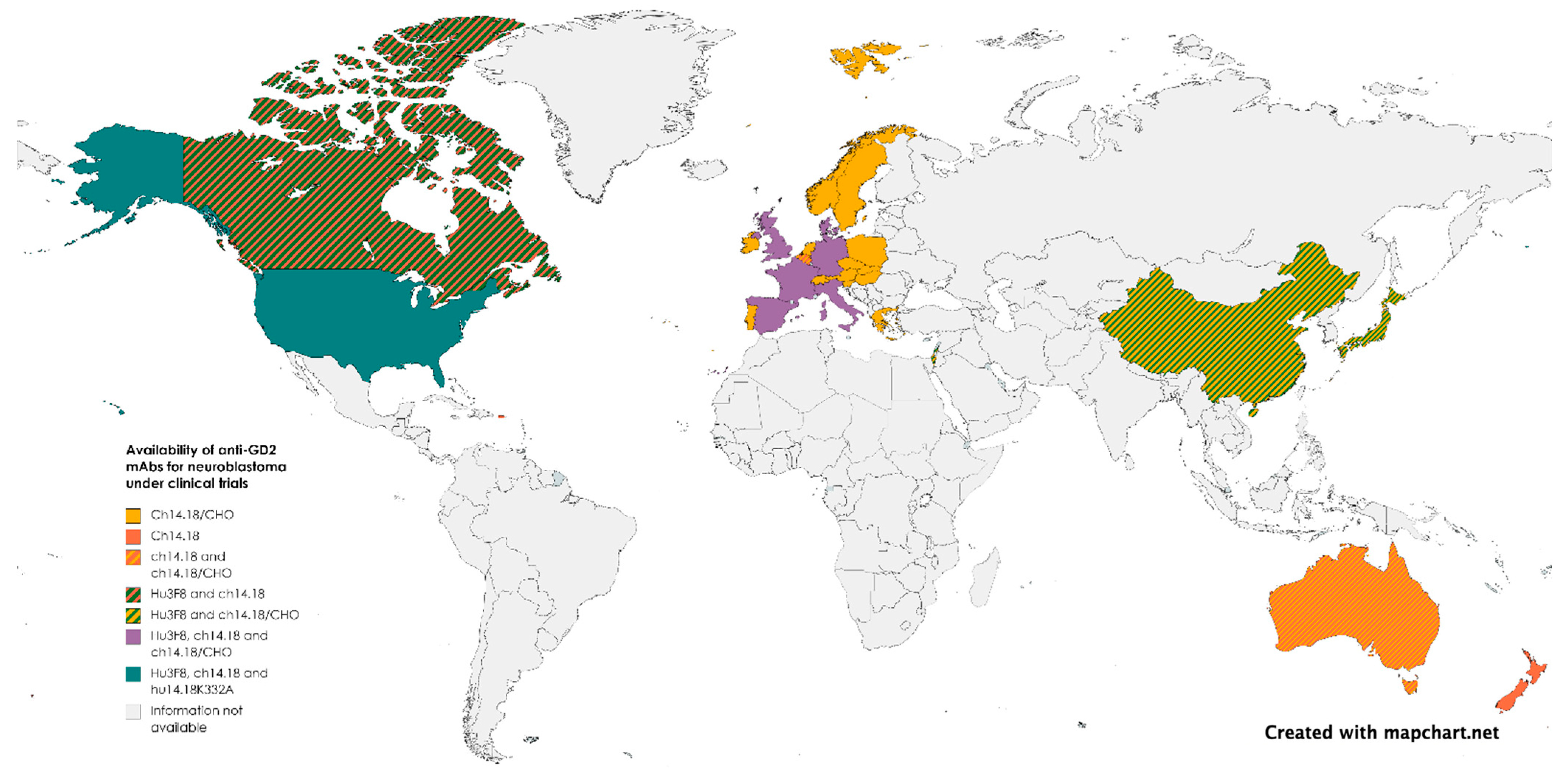
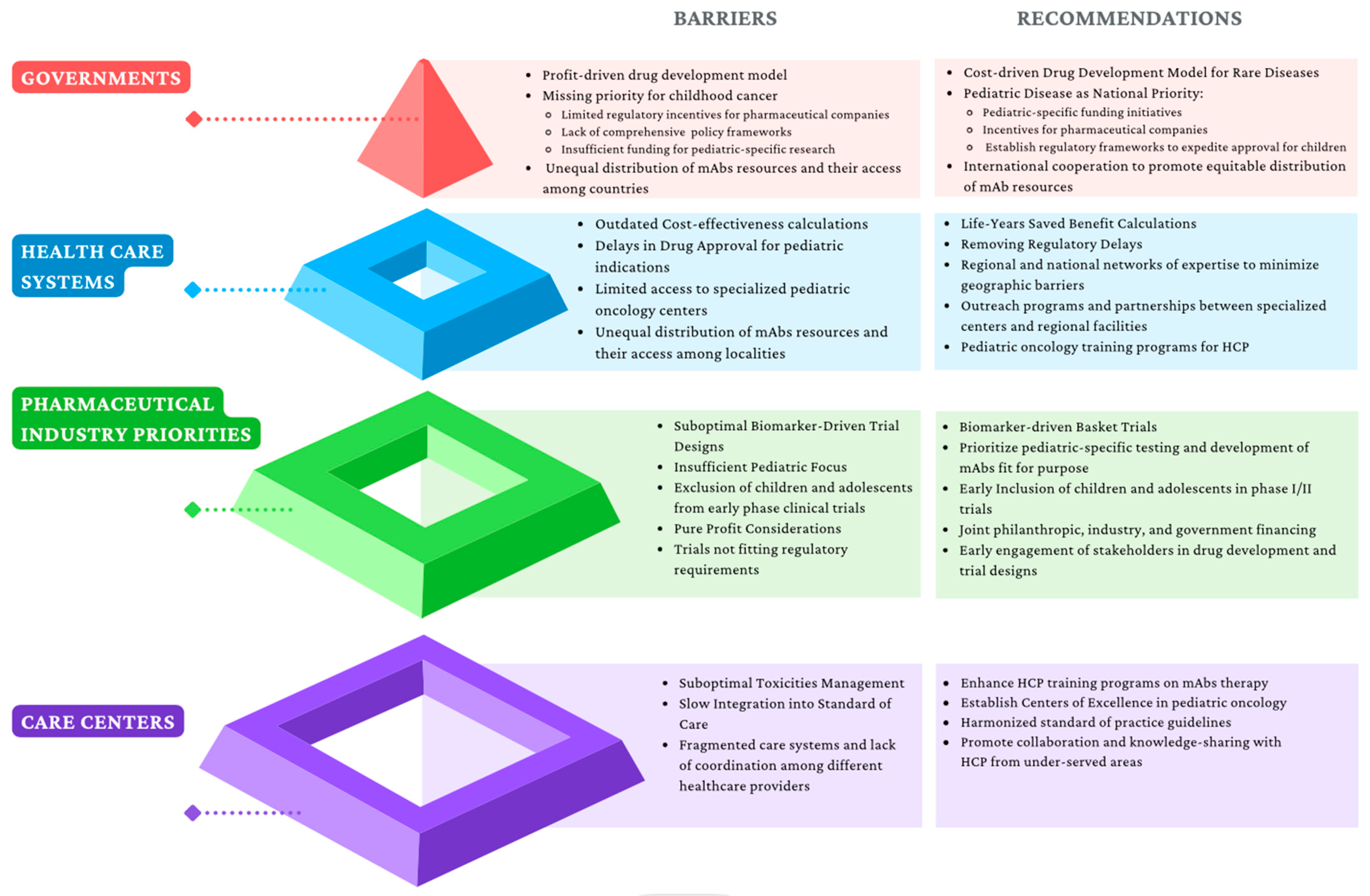
3.3. Commercialization, Regulation, and Political Limitations
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Busch, W. Aus Der Sitzung Der Medicinischen Section Vom 13 November 1867. Berl. Klin. Wochenschr. 1868, 5, 137. [Google Scholar]
- McCarthy, E.F. The Toxins of William B. Coley and the Treatment of Bone and Soft-Tissue Sarcomas. Iowa Orthop. J. 2006, 26, 154–158. [Google Scholar]
- Buss, N.A.P.S.; Henderson, S.J.; McFarlane, M.; Shenton, J.M.; De Haan, L. Monoclonal Antibody Therapeutics: History and Future. Curr. Opin. Pharmacol. 2012, 12, 615–622. [Google Scholar] [CrossRef]
- Liu, J.K.H. The History of Monoclonal Antibody Development—Progress, Remaining Challenges and Future Innovations. Ann. Med. Surg. (Lond.) 2014, 3, 113–116. [Google Scholar] [CrossRef]
- Köhler, G.; Milstein, C. Continuous Cultures of Fused Cells Secreting Antibody of Predefined Specificity. Nature 1975, 256, 495–497. [Google Scholar] [CrossRef]
- Ekstrand, B.C.; Lucas, J.B.; Horwitz, S.M.; Fan, Z.; Breslin, S.; Hoppe, R.T.; Natkunam, Y.; Bartlett, N.L.; Horning, S.J. Rituximab in Lymphocyte-Predominant Hodgkin Disease: Results of a Phase 2 Trial. Blood 2003, 101, 4285–4289. [Google Scholar] [CrossRef]
- Mullard, A. FDA Approves 100th Monoclonal Antibody Product; NLM (Medline): Bethesda, MD, USA, 2021; Volume 20, pp. 491–495. [Google Scholar]
- Cheung, N.K.; Lazarus, H.; Miraldi, F.D.; Abramowsky, C.R.; Kallick, S.; Saarinen, U.M.; Spitzer, T.; Strandjord, S.E.; Coccia, P.F.; Berger, N.A. Ganglioside GD2 Specific Monoclonal Antibody 3F8: A Phase I Study in Patients with Neuroblastoma and Malignant Melanoma. J. Clin. Oncol. 1987, 5, 1430–1440. [Google Scholar] [CrossRef]
- Cheung, N.-K.V.; Kushner, B.H.; Cheung, I.Y.; Kramer, K.; Canete, A.; Gerald, W.; Bonilla, M.A.; Finn, R.; Yeh, S.J.; Larson, S.M. Anti-GD 2 Antibody Treatment of Minimal Residual Stage 4 Neuroblastoma Diagnosed at More Than 1 Year of Age. J. Clin. Oncol. 1998, 16, 3053–3060. [Google Scholar] [CrossRef]
- Cheung, N.K.V.; Cheung, I.Y.; Kushner, B.H.; Ostrovnaya, I.; Chamberlain, E.; Kramer, K.; Modak, S. Murine Anti-GD2 Monoclonal Antibody 3F8 Combined with Granulocyte- Macrophage Colony-Stimulating Factor and 13-Cis-Retinoic Acid in High-Risk Patients with Stage 4 Neuroblastoma in First Remission. J. Clin. Oncol. 2012, 30, 3264–3270. [Google Scholar] [CrossRef]
- Maude, S.L.; Laetsch, T.W.; Buechner, J.; Rives, S.; Boyer, M.; Bittencourt, H.; Bader, P.; Verneris, M.R.; Stefanski, H.E.; Myers, G.D.; et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. N. Engl. J. Med. 2018, 378, 439–448. [Google Scholar] [CrossRef]
- June, C.H.; O’Connor, R.S.; Kawalekar, O.U.; Ghassemi, S.; Milone, M.C. CAR T Cell Immunotherapy for Human Cancer. Science 2018, 359, 1361–1365. [Google Scholar] [CrossRef]
- Martin-Mateos, M.A. Monoclonal Antibodies in Pediatrics: Use in Prevention and Treatment. Allergol. Immunopathol. (Madr.) 2007, 35, 145–150. [Google Scholar] [CrossRef][Green Version]
- Gonzales, T.; Bergamasco, A.; Cristarella, T.; Goyer, C.; Wojdyla, M.; Oladapo, A.; Sawicky, J.; Yee, J.; Moride, Y. Effectiveness and Safety of Palivizumab for the Prevention of Serious Lower Respiratory Tract Infection Caused by Respiratory Syncytial Virus: A Systematic Review. Am. J. Perinatol. 2023. [Google Scholar] [CrossRef]
- Orders, M. An EUA for Sotrovimab for Treatment of COVID-19. Med. Lett. Drugs Ther. 2021, 63, 97–98. [Google Scholar]
- Casirivimab and Imdevimab (REGEN-COV) for Post-Exposure Prophylaxis of COVID-19. Med. Lett. Drugs Ther. 2021, 63, 130–131.
- Orders, M. An EUA for Bamlanivimab and Etesevimab for COVID-19. Med. Lett. Drugs Ther. 2021, 63, 49–51. [Google Scholar]
- Gatta, G.; Botta, L.; Rossi, S.; Aareleid, T.; Bielska-Lasota, M.; Clavel, J.; Dimitrova, N.; Jakab, Z.; Kaatsch, P.; Lacour, B.; et al. Childhood Cancer Survival in Europe 1999-2007: Results of EUROCARE-5—A Population-Based Study. Lancet Oncol. 2014, 15, 35–47. [Google Scholar] [CrossRef]
- Lam, C.G.; Howard, S.C.; Bouffet, E.; Pritchard-Jones, K. Science and Health for All Children with Cancer. Science 2019, 363, 1182–1186. [Google Scholar] [CrossRef]
- Shin, H.; Dudley, W.N.; Bhakta, N.; Horan, M.R.; Wang, Z.; Bartlett, T.R.; Srivastava, D.; Yasui, Y.; Baker, J.N.; Robison, L.L.; et al. Associations of Symptom Clusters and Health Outcomes in Adult Survivors of Childhood Cancer: A Report from the St Jude Lifetime Cohort Study. J. Clin. Oncol. 2023, 41, 497–507. [Google Scholar] [CrossRef]
- Huang, M.A.; Krishnadas, D.K.; Lucas, K.G. Pediatric Cancer Immunotherapy. J. Immunol. Res. 2015, 2015, 675269. [Google Scholar] [CrossRef]
- Minard-Colin, V.; Aupérin, A.; Pillon, M.; Burke, G.A.A.; Barkauskas, D.A.; Wheatley, K.; Delgado, R.F.; Alexander, S.; Uyttebroeck, A.; Bollard, C.M.; et al. Rituximab for High-Risk, Mature B-Cell Non-Hodgkin’s Lymphoma in Children. N. Engl. J. Med. 2020, 382, 2207–2219. [Google Scholar] [CrossRef] [PubMed]
- Grillo-Lopez, A.; White, C.; Dallaire, B.; Varns, C.; Shen, C.; Wei, A.; Leonard, J.; McClure, A.; Weaver, R.; Cairelli, S.; et al. Rituximab The First Monoclonal Antibody Approved for the Treatment of Lymphoma. Curr. Pharm. Biotechnol. 2005, 1, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zwaan, C.M. Gemtuzumab Ozogamicin: First Clinical Experiences in Children with Relapsed/Refractory Acute Myeloid Leukemia Treated on Compassionate-Use Basis. Blood 2003, 101, 3868–3871. [Google Scholar] [CrossRef] [PubMed]
- Gamis, A.S.; Alonzo, T.A.; Meshinchi, S.; Sung, L.; Gerbing, R.B.; Raimondi, S.C.; Hirsch, B.A.; Kahwash, S.B.; Heerema-McKenney, A.; Winter, L.; et al. Gemtuzumab Ozogamicin in Children and Adolescents with de Novo Acute Myeloid Leukemia Improves Event-Free Survival by Reducing Relapse Risk: Results from the Randomized Phase III Children’s Oncology Group Trial AAML0531. J. Clin. Oncol. 2014, 32, 3021–3032. [Google Scholar] [CrossRef]
- Food and Drug Administration FDA Approves Gemtuzumab Ozogamicin for CD33-Positive AML. Available online: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-gemtuzumab-ozogamicin-cd33-positive-aml-pediatric-patients (accessed on 17 April 2023).
- Castellino, S.M.; Pei, Q.; Parsons, S.K.; Hodgson, D.; McCarten, K.; Horton, T.; Cho, S.; Wu, Y.; Punnett, A.; Dave, H.; et al. Brentuximab Vedotin with Chemotherapy in Pediatric High-Risk Hodgkin’s Lymphoma. N. Engl. J. Med. 2022, 387, 1649–1660. [Google Scholar] [CrossRef]
- Brown, P.A.; Ji, L.; Xu, X.; Devidas, M.; Hogan, L.; Borowitz, M.J.; Raetz, E.A.; Zugmaier, G.; Sharon, E.; Gore, L.; et al. A Randomized Phase 3 Trial of Blinatumomab Vs. Chemotherapy As Post-Reinduction Therapy in High and Intermediate Risk (HR/IR) First Relapse of B-Acute Lymphoblastic Leukemia (B-ALL) in Children and Adolescents/Young Adults (AYAs) Demonstrates Superior Eff. Blood 2019, 134, LBA-1. [Google Scholar] [CrossRef]
- Perkins, S.M.; Shinohara, E.T.; DeWees, T.; Frangoul, H. Outcome for Children with Metastatic Solid Tumors over the Last Four Decades. PLoS ONE 2014, 9, e100396. [Google Scholar] [CrossRef]
- Haghiri, S.; Fayech, C.; Mansouri, I.; Dufour, C.; Pasqualini, C.; Bolle, S.; Rivollet, S.; Dumas, A.; Boumaraf, A.; Belhout, A.; et al. Long-Term Follow-up of High-Risk Neuroblastoma Survivors Treated with High-Dose Chemotherapy and Stem Cell Transplantation Rescue. Bone Marrow Transplant. 2021, 56, 1984–1997. [Google Scholar] [CrossRef]
- Bier, H.; Hoffmann, T.; Haas, I.; van Lierop, A. Anti-(Epidermal Growth Factor) Receptor Monoclonal Antibodies for the Induction of Antibody-Dependent Cell-Mediated Cytotoxicity against Squamous Cell Carcinoma Lines of the Head and Neck. Cancer Immunol. Immunother. 1998, 46, 167–173. [Google Scholar] [CrossRef]
- Rimawi, M.F.; Schiff, R.; Osborne, C.K. Targeting HER2 for the Treatment of Breast Cancer. Annu. Rev. Med. 2015, 66, 111–128. [Google Scholar] [CrossRef]
- Tung, I.; Sahu, A. Immune Checkpoint Inhibitor in First-Line Treatment of Metastatic Renal Cell Carcinoma: A Review of Current Evidence and Future Directions. Front. Oncol. 2021, 11, 707214. [Google Scholar] [CrossRef]
- Peng, T.; Lin, H.; Tsai, F.; Wu, T. Immune Checkpoint Inhibitors for First-line Treatment of Advanced Non-small-cell Lung Cancer: A Systematic Review and Network Meta-analysis. Thorac. Cancer 2021, 12, 2873–2885. [Google Scholar] [CrossRef]
- Seifert, H. Über Ein Weiteres Hirntumorcharakteristisches Gangliosid. Klin. Wochenschr. 1966, 44, 469–470. [Google Scholar] [CrossRef] [PubMed]
- Alistair Lammie, G.; Cheung, N.V.; Gerald, W.; Rosenblum, M.; Cordon-cardo, C. Ganglioside GD 2 Expression in the Human Nervous System and in Neuroblastomas-An Immunohistochemical Study. Int. J. Oncol. 1993, 3, 909–915. [Google Scholar]
- Wu, Z.-L.; Schwartz, E.; Seeger, R.; Ladisch3, S. Expression of GD2Ganglioside by Untreated Primary Human Neuroblastomas1. Cancer Res. 1986, 46, 440–443. [Google Scholar] [PubMed]
- Roth, M.; Linkowski, M.; Tarim, J.; Piperdi, S.; Sowers, R.; Geller, D.; Gill, J.; Gorlick, R. Ganglioside GD2 as a Therapeutic Target for Antibody-Mediated Therapy in Patients with Osteosarcoma. Cancer 2014, 120, 548–554. [Google Scholar] [CrossRef]
- Poon, V.I.; Roth, M.; Piperdi, S.; Geller, D.; Gill, J.; Rudzinski, E.R.; Hawkins, D.S.; Gorlick, R. Ganglioside GD2 Expression Is Maintained upon Recurrence in Patients with Osteosarcoma. Clin. Sarcoma Res. 2015, 5, 4. [Google Scholar] [CrossRef]
- Heiner, J.P.; Miraldi, F.; Kallick, S.; Makley, J.; Neely, J.; Smith-Mensah, W.H.; Cheung, N.K. Localization of GD2-SpecificMonoclonal Antibody 3F8 in Human Osteosarcoma1. Cancer Res. 1987, 47, 5377–5381. [Google Scholar]
- Shibuya, H.; Hamamura, K.; Hotta, H.; Matsumoto, Y.; Nishida, Y.; Hattori, H.; Furukawa, K.; Ueda, M.; Furukawa, K. Enhancement of Malignant Properties of Human Osteosarcoma Cells with Disialyl Gangliosides GD2/GD3. Cancer Sci. 2012, 103, 1656–1664. [Google Scholar] [CrossRef]
- Wikstrand, C.J.; Fredman, P.; Svennerholm, L.; Humphrey, P.A.; Bigner, S.H.; Bigner, D.D. Monoclonal Antibodies to Malignant Human Gliomas. Mol. Chem. Neuropathol. 1992, 17, 137–146. [Google Scholar] [CrossRef]
- Mennel, H.D.; Bosslet, K.; Geissel, H.; Bauer, B.L. Immunohistochemically Visualized Localisation of Gangliosides Glac2 (GD3) and Gtri2 (GD2) in Cells of Human Intracranial Tumors. Exp. Toxicol. Pathol. 2000, 52, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Shinoura, N.; Dohi, T.; Kondo, T.; Yoshioka, M.; Takakura, K.; Oshima, M. Ganglioside Composition and Its Relation to Clinical Data in Brain Tumors. Neurosurgery 1992, 31, 541–549. [Google Scholar] [CrossRef]
- Nazha, B.; Inal, C.; Owonikoko, T.K. Disialoganglioside GD2 Expression in Solid Tumors and Role as a Target for Cancer Therapy. Front. Oncol. 2020, 10, 1000. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.R.; Cordon-Cardo, C.; Houghton, A.N.; Cheung, N.-K.V.; Brennan, M.F. Expression of Disialogangliosides GO2 and GD3 on Human Soft Tissue Sarcomas. Cancer 1992, 70, 633–638. [Google Scholar] [CrossRef]
- Cheung, N.-K.K.V.; Guo, H.; Hu, J.; Tassev, D.V.; Cheung, I.Y. Humanizing Murine IgG3 Anti-GD2 Antibody M3F8 Substantially Improves Antibody-Dependent Cell-Mediated Cytotoxicity While Retaining Targeting in Vivo. Oncoimmunology 2012, 1, 477–486. [Google Scholar] [CrossRef]
- Saito, M.; Yu, R.K.; Cheung, N.-K.V. Ganglioside GD2 Specificity of Monoclonal Antibodies to Human Neuroblastoma Cell. Biochem. Biophys. Res. Commun. 1985, 127, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cheung, N.K.; Saarinen, U.M.; Neely, J.E.; Landmeier, B.; Donovan, D.; Coccia, P.F. Monoclonal Antibodies to a Glycolipid Antigen on Human Neuroblastoma Cells. Cancer Res. 1985, 45, 2642–2649. [Google Scholar]
- Kushner, B.H.; Cheung, I.Y.; Modak, S.; Basu, E.M.; Roberts, S.S.; Cheung, N.-K. Humanized 3F8 Anti-G2 Monoclonal Antibody Dosing with Granulocyte-Macrophage Colony-Stimulating Factor in Patients With Resistant Neuroblastoma. JAMA Oncol. 2018, 4, 1729. [Google Scholar] [CrossRef]
- Markham, A. Naxitamab: First Approval. Drugs 2021, 81, 291–296. [Google Scholar] [CrossRef]
- Dhillon, S. Dinutuximab: First Global Approval. Drugs 2015, 75, 923–927. [Google Scholar] [CrossRef]
- Yu, A.L.; Gilman, A.L.; Ozkaynak, M.F.; London, W.B.; Kreissman, S.G.; Chen, H.X.; Smith, M.; Anderson, B.; Villablanca, J.G.; Matthay, K.K.; et al. Anti-GD2 Antibody with GM-CSF, Interleukin-2, and Isotretinoin for Neuroblastoma. N. Engl. J. Med. 2010, 363, 1324–1334. [Google Scholar] [CrossRef]
- Ladenstein, R.; Pötschger, U.; Valteau-Couanet, D.; Luksch, R.; Castel, V.; Yaniv, I.; Laureys, G.; Brock, P.; Michon, J.M.; Owens, C.; et al. Interleukin 2 with Anti-GD2 Antibody Ch14.18/CHO (Dinutuximab Beta) in Patients with High-Risk Neuroblastoma (HR-NBL1/SIOPEN): A Multicentre, Randomised, Phase 3 Trial. Lancet Oncol. 2018, 19, 1617–1629. [Google Scholar] [CrossRef]
- Mora, J.; Bear, M.; Chan, G.; Morgenstern, D.A.; Nysom, K.; Tornøe, K.; Sørensen, P.S.; Kushner, B. 891P Naxitamab Treatment for Relapsed or Refractory High-Risk Neuroblastoma: Outcomes from the First Prespecified Analyses of the Pivotal 201 Trial. Ann. Oncol. 2022, 33, S956. [Google Scholar] [CrossRef]
- Navid, F.; Santana, V.M.; Barfield, R.C. Anti-GD2 Antibody Therapy for GD2-Expressing Tumors. Curr. Cancer Drug. Targets 2010, 10, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Dobrenkov, K.; Ostrovnaya, I.; Gu, J.; Cheung, I.Y.; Cheung, N.K.V. Oncotargets GD2 and GD3 Are Highly Expressed in Sarcomas of Children, Adolescents, and Young Adults. Pediatr. Blood Cancer 2016, 63, 1780–1785. [Google Scholar] [CrossRef] [PubMed]
- Hingorani, P.; Krailo, M.; Buxton, A.; Hutson, P.; Sondel, P.M.; Diccianni, M.; Yu, A.; Morris, C.D.; Womer, R.B.; Crompton, B.; et al. Phase 2 Study of Anti-Disialoganglioside Antibody, Dinutuximab, in Combination with GM-CSF in Patients with Recurrent Osteosarcoma: A Report from the Children’s Oncology Group. Eur. J. Cancer 2022, 172, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.Y.; Panzer, J.; Maris, J.M.; Castañeda, A.; Gomez-Chiari, M.; Mora, J. Transverse Myelitis as an Unexpected Complication Following Treatment with Dinutuximab in Pediatric Patients with High-Risk Neuroblastoma: A Case Series. Pediatr. Blood Cancer 2018, 65, e26732. [Google Scholar] [CrossRef]
- Kushner, B.H.; Modak, S.; Basu, E.M.; Roberts, S.S.; Kramer, K.; Cheung, N.K.V. Posterior Reversible Encephalopathy Syndrome in Neuroblastoma Patients Receiving Anti-GD2 3F8 Monoclonal Antibody. Cancer 2013, 119, 2789–2795. [Google Scholar] [CrossRef]
- Yu, A.L.; Gilman, A.L.; Ozkaynak, M.F.; Naranjo, A.; Diccianni, M.B.; Gan, J.; Hank, J.A.; Batova, A.; London, W.B.; Tenney, S.C.; et al. Long-Term Follow-up of a Phase III Study of Ch14.18 (Dinutuximab) + Cytokine Immunotherapy in Children with High-Risk Neuroblastoma: COG Study ANBL0032. Clin. Cancer Res. 2021, 27, 2179–2189. [Google Scholar] [CrossRef]
- Wang, S.; Zhong, G.; Wang, X.; Yu, F.; Weng, D.; Wang, X.; Lin, J. Prognostic Significance of the Expression of HER Family Members in Primary Osteosarcoma. Oncol. Lett. 2018, 16, 2185–2194. [Google Scholar] [CrossRef]
- Ebb, D.; Meyers, P.; Grier, H.; Bernstein, M.; Gorlick, R.; Lipshultz, S.E.; Krailo, M.; Devidas, M.; Barkauskas, D.A.; Siegal, G.P.; et al. Phase II Trial of Trastuzumab in Combination with Cytotoxic Chemotherapy for Treatment of Metastatic Osteosarcoma with Human Epidermal Growth Factor Receptor 2 Overexpression: A Report from the Children’s Oncology Group. J. Clin. Oncol. 2012, 30, 2545–2551. [Google Scholar] [CrossRef]
- Lee, V.; Murphy, A.; Le, D.T.; Diaz, L.A. Mismatch Repair Deficiency and Response to Immune Checkpoint Blockade. Oncologist 2016, 21, 1200–1211. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.L.; Fox, E.; Merchant, M.S.; Reid, J.M.; Kudgus, R.A.; Liu, X.; Minard, C.G.; Voss, S.; Berg, S.L.; Weigel, B.J.; et al. Nivolumab in Children and Young Adults with Relapsed or Refractory Solid Tumours or Lymphoma (ADVL1412): A Multicentre, Open-Label, Single-Arm, Phase 1–2 Trial. Lancet Oncol. 2020, 21, 541–550. [Google Scholar] [CrossRef]
- Geoerger, B.; Kang, H.J.; Yalon-Oren, M.; Marshall, L.V.; Vezina, C.; Pappo, A.S.; Laetsch, T.W.; Petrilli, A.S.; Ebinger, M.; Toporski, J.; et al. KEYNOTE-051: An Update on the Phase 2 Results of Pembrolizumab (Pembro) in Pediatric Patients (Pts) with Advanced Melanoma or a PD-L1–Positive Advanced, Relapsed or Refractory Solid Tumor or Lymphoma. J. Clin. Oncol. 2018, 36, 10525. [Google Scholar] [CrossRef]
- Tawbi, H.A.; Burgess, M.; Bolejack, V.; Van Tine, B.A.; Schuetze, S.M.; Hu, J.; D’Angelo, S.; Attia, S.; Riedel, R.F.; Priebat, D.A.; et al. Pembrolizumab in Advanced Soft-Tissue Sarcoma and Bone Sarcoma (SARC028): A Multicentre, Two-Cohort, Single-Arm, Open-Label, Phase 2 Trial. Lancet Oncol. 2017, 18, 1493–1501. [Google Scholar] [CrossRef]
- Naqash, A.R.; O’Sullivan Coyne, G.H.; Moore, N.; Sharon, E.; Takebe, N.; Fino, K.K.; Ferry-Galow, K.V.; Hu, J.S.; Van Tine, B.A.; Burgess, M.A.; et al. Phase II Study of Atezolizumab in Advanced Alveolar Soft Part Sarcoma (ASPS). J. Clin. Oncol. 2021, 39, 11519. [Google Scholar] [CrossRef]
- Modak, S.; Kramer, K.; Gultekin, S.H.; Guo, H.F.; Cheung, N.K. Monoclonal Antibody 8H9 Targets a Novel Cell Surface Antigen Expressed by a Wide Spectrum of Human Solid Tumors. Cancer Res. 2001, 61, 4048–4054. [Google Scholar]
- Loo, D.; Alderson, R.F.; Chen, F.Z.; Huang, L.; Zhang, W.; Gorlatov, S.; Burke, S.; Ciccarone, V.; Li, H.; Yang, Y.; et al. Development of an Fc-Enhanced Anti–B7-H3 Monoclonal Antibody with Potent Antitumor Activity. Clin. Cancer Res. 2012, 18, 3834–3845. [Google Scholar] [CrossRef]
- Kramer, K.; Cheung, N.-K.V.; Humm, J.; DiResta, G.; Arbit, E.; Larson, S.; Finn, R.; Rosenblum, M.; Nguyen, H.; Gonzalez, G.; et al. Pharmacokinetics and Acute Toxicology of Intraventricular 131I-Monoclonal Antibody Targeting Disialoganglioside in Non-Human Primates. J. Neurooncol. 1997, 35, 101–112. [Google Scholar] [CrossRef]
- Kramer, K.; Pandit-Taskar, N.; Kushner, B.H.; Zanzonico, P.; Humm, J.L.; Tomlinson, U.; Donzelli, M.; Wolden, S.L.; Haque, S.; Dunkel, I.; et al. Phase 1 Study of Intraventricular 131I-Omburtamab Targeting B7H3 (CD276)-Expressing CNS Malignancies. J. Hematol. Oncol. 2022, 15, 165. [Google Scholar] [CrossRef]
- Pandit-Taskar, N.; Zanzonico, P.B.; Kramer, K.; Grkovski, M.; Fung, E.K.; Shi, W.; Zhang, Z.; Lyashchenko, S.K.; Fung, A.M.; Pentlow, K.S.; et al. Biodistribution and Dosimetry of Intraventricularly Administered 124 I-Omburtamab in Patients with Metastatic Leptomeningeal Tumors. J. Nucl. Med. 2019, 60, 1794–1801. [Google Scholar] [CrossRef] [PubMed]
- Souweidane, M.M.; Kramer, K.; Pandit-Taskar, N.; Zhou, Z.; Haque, S.; Zanzonico, P.; Carrasquillo, J.A.; Lyashchenko, S.K.; Thakur, S.B.; Donzelli, M.; et al. Convection-Enhanced Delivery for Diffuse Intrinsic Pontine Glioma: A Single-Centre, Dose-Escalation, Phase 1 Trial. Lancet Oncol. 2018, 19, 1040–1050. [Google Scholar] [CrossRef] [PubMed]
- Kramer, K.; Kushner, B.H.; Modak, S.; Pandit-Taskar, N.; Tomlinson, U.; Wolden, S.L.; Zanzonico, P.; John, H.L.; Haque, S.; Souweidane, M.M.; et al. A Curative Approach to Central Nervous System Metastases of Neuroblastoma. Pediatr. Blood Cancer 2019, 66, e27989. [Google Scholar] [CrossRef]
- Modak, S.; Zanzonico, P.; Grkovski, M.; Slotkin, E.K.; Carrasquillo, J.A.; Lyashchenko, S.K.; Lewis, J.S.; Cheung, I.Y.; Heaton, T.; LaQuaglia, M.P.; et al. B7H3-Directed Intraperitoneal Radioimmunotherapy With Radioiodinated Omburtamab for Desmoplastic Small Round Cell Tumor and Other Peritoneal Tumors: Results of a Phase I Study. J. Clin. Oncol. 2020, 38, 4283–4291. [Google Scholar] [CrossRef]
- Desantes, K.; Maris, J.M.; McDowell, K.; Mackall, C.; Shankar, S.; Vasselli, J.; Chen, F.; Loo, D.; Moore, P.A.; Wigginton, J.M.; et al. A Phase 1, Open-Label, Dose Escalation Study of Enoblituzumab (MGA271) in Pediatric Patients with B7-H3-Expressing Relapsed or Refractory Solid Tumors. J. Clin. Oncol. 2017, 35, TPS2596. [Google Scholar] [CrossRef]
- Mascarenhas, L.; Chi, Y.-Y.; Hingorani, P.; Anderson, J.R.; Lyden, E.R.; Rodeberg, D.A.; Indelicato, D.J.; Kao, S.C.; Dasgupta, R.; Spunt, S.L.; et al. Randomized Phase II Trial of Bevacizumab or Temsirolimus in Combination with Chemotherapy for First Relapse Rhabdomyosarcoma: A Report from the Children’s Oncology Group. J. Clin. Oncol. 2019, 37, 2866–2874. [Google Scholar] [CrossRef]
- Ferrari, A.; Merks, J.H.M.; Chisholm, J.C.; Orbach, D.; Brennan, B.; Gallego, S.; van Noesel, M.M.; McHugh, K.; van Rijn, R.R.; Gaze, M.N.; et al. Outcomes of Metastatic Non-Rhabdomyosarcoma Soft Tissue Sarcomas (NRSTS) Treated within the BERNIE Study: A Randomised, Phase II Study Evaluating the Addition of Bevacizumab to Chemotherapy. Eur. J. Cancer 2020, 130, 72–80. [Google Scholar] [CrossRef]
- Narayana, A.; Kunnakkat, S.; Chacko-Mathew, J.; Gardner, S.; Karajannis, M.; Raza, S.; Wisoff, J.; Weiner, H.; Harter, D.; Allen, J. Bevacizumab in Recurrent High-Grade Pediatric Gliomas. Neuro Oncol. 2010, 12, 985–990. [Google Scholar] [CrossRef]
- Packer, R.J.; Jakacki, R.; Horn, M.; Rood, B.; Vezina, G.; MacDonald, T.; Fisher, M.J.; Cohen, B. Objective Response of Multiply Recurrent Low-Grade Gliomas to Bevacizumab and Irinotecan. Pediatr. Blood Cancer 2009, 52, 791–795. [Google Scholar] [CrossRef]
- Moreno, L.; Moroz, V.; Owens, C.; Valteau-Couanet, D.; Gambart, M.; Castel, V.; van Eijkelenburg, N.; Castellano, A.; Nysom, K.; Gerber, N.; et al. Bevacizumab for Children with Relapsed & Refractory High-Risk Neuroblastoma (RR-HRNB): Results of the BEACON-Neuroblastoma Randomized Phase II Trial—A European ITCC-SIOPEN Trial. Ann. Oncol. 2019, 30, v901. [Google Scholar] [CrossRef]
- Trippett, T.M.; Herzog, C.; Whitlock, J.A.; Wolff, J.; Kuttesch, J.; Bagatell, R.; Hunger, S.P.; Boklan, J.; Smith, A.A.; Arceci, R.J.; et al. Phase I and Pharmacokinetic Study of Cetuximab and Irinotecan in Children with Refractory Solid Tumors: A Study of the Pediatric Oncology Experimental Therapeutic Investigators’ Consortium. J. Clin. Oncol. 2009, 27, 5102–5108. [Google Scholar] [CrossRef] [PubMed]
- Rajappa, P.; Krass, J.; Riina, H.A.; Boockvar, J.A.; Greenfield, J.P. Super-Selective Basilar Artery Infusion of Bevacizumab and Cetuximab for Multiply Recurrent Pediatric Ependymoma. Interv. Neuroradiol. 2011, 17, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Macy, M.E.; Kieran, M.W.; Chi, S.N.; Cohen, K.J.; MacDonald, T.J.; Smith, A.A.; Etzl, M.M.; Kuei, M.C.; Donson, A.M.; Gore, L.; et al. A Pediatric Trial of Radiation/Cetuximab Followed by Irinotecan/Cetuximab in Newly Diagnosed Diffuse Pontine Gliomas and High-Grade Astrocytomas: A Pediatric Oncology Experimental Therapeutics Investigators’ Consortium Study. Pediatr. Blood Cancer 2017, 64, e26621. [Google Scholar] [CrossRef] [PubMed]
- McCrea, H.J.; Ivanidze, J.; O’Connor, A.; Hersh, E.H.; Boockvar, J.A.; Gobin, Y.P.; Knopman, J.; Greenfield, J.P. Intraarterial Delivery of Bevacizumab and Cetuximab Utilizing Blood-Brain Barrier Disruption in Children with High-Grade Glioma and Diffuse Intrinsic Pontine Glioma: Results of a Phase I Trial. J. Neurosurg. Pediatr. 2021, 28, 371–379. [Google Scholar] [CrossRef]
- Bailey, K.; Cost, C.; Davis, I.; Glade-Bender, J.; Grohar, P.; Houghton, P.; Isakoff, M.; Stewart, E.; Laack, N.; Yustein, J.; et al. Emerging Novel Agents for Patients with Advanced Ewing Sarcoma: A Report from the Children’s Oncology Group (COG) New Agents for Ewing Sarcoma Task Force [Version 1; Peer Review: 3 Approved]. F1000Research 2019, 8, 493. [Google Scholar] [CrossRef]
- Gajdosik, Z. Racotumomab—A Novel Anti-Idiotype Monoclonal Antibody Vaccine for the Treatment of Cancer. Drugs Today 2014, 50, 301-7. [Google Scholar] [CrossRef]
- Cacciavillano, W.; Sampor, C.; Venier, C.; Gabri, M.R.; de Dávila, M.T.G.; Galluzzo, M.L.; Guthmann, M.D.; Fainboim, L.; Alonso, D.F.; Chantada, G.L. A Phase I Study of the Anti-Idiotype Vaccine Racotumomab in Neuroblastoma and Other Pediatric Refractory Malignancies. Pediatr. Blood Cancer 2015, 62, 2120–2124. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Terry, R.L.; Meyran, D.; Ziegler, D.S.; Haber, M.; Ekert, P.G.; Trapani, J.A.; Neeson, P.J. Immune Profiling of Pediatric Solid Tumors. J. Clin. Investig. 2020, 130, 3391–3402. [Google Scholar] [CrossRef]
- Wienke, J.; Dierselhuis, M.P.; Tytgat, G.A.M.; Künkele, A.; Nierkens, S.; Molenaar, J.J. The Immune Landscape of Neuroblastoma: Challenges and Opportunities for Novel Therapeutic Strategies in Pediatric Oncology. Eur. J. Cancer 2021, 144, 123–150. [Google Scholar] [CrossRef]
- Mardanpour, K.; Rahbar, M.; Mardanpour, S.; Mardanpour, N.; Rezaei, M. CD8+ T-Cell Lymphocytes Infiltration Predict Clinical Outcomes in Wilms’ Tumor. Tumor Biol. 2020, 42, 101042832097597. [Google Scholar] [CrossRef] [PubMed]
- Boldrini, R.; De Pasquale, M.D.; Melaiu, O.; Chierici, M.; Jurman, G.; Benedetti, M.C.; Salfi, N.C.; Castellano, A.; Collini, P.; Furlanello, C.; et al. Tumor-Infiltrating T Cells and PD-L1 Expression in Childhood Malignant Extracranial Germ-Cell Tumors. Oncoimmunology 2019, 8, e1542245. [Google Scholar] [CrossRef] [PubMed]
- Plant, A.S.; Koyama, S.; Sinai, C.; Solomon, I.H.; Griffin, G.K.; Ligon, K.L.; Bandopadhayay, P.; Betensky, R.; Emerson, R.; Dranoff, G.; et al. Immunophenotyping of Pediatric Brain Tumors: Correlating Immune Infiltrate with Histology, Mutational Load, and Survival and Assessing Clonal T Cell Response. J. Neurooncol. 2018, 137, 269–278. [Google Scholar] [CrossRef]
- Cornel, A.M.; Mimpen, I.L.; Nierkens, S. MHC Class I Downregulation in Cancer: Underlying Mechanisms and Potential Targets for Cancer Immunotherapy. Cancers 2020, 12, 1760. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lage, M.; Torres-Ruiz, R.; Puig-Serra, P.; Moreno-Gaona, P.; Martin, M.C.; Moya, F.J.; Quintana-Bustamante, O.; Garcia-Silva, S.; Carcaboso, A.M.; Petazzi, P.; et al. In Vivo CRISPR/Cas9 Targeting of Fusion Oncogenes for Selective Elimination of Cancer Cells. Nat. Commun. 2020, 11, 5060. [Google Scholar] [CrossRef] [PubMed]
- Ledford, H. CRISPR Cancer Trial Success Paves the Way for Personalized Treatments. Nature 2022, 611, 433–434. [Google Scholar] [CrossRef]
- Tian, Z.; Liang, G.; Cui, K.; Liang, Y.; Wang, Q.; Lv, S.; Cheng, X.; Zhang, L. Insight Into the Prospects for RNAi Therapy of Cancer. Front. Pharmacol. 2021, 12, 644718. [Google Scholar] [CrossRef]
- Kermi, C.; Lau, L.; Asadi Shahmirzadi, A.; Classon, M. Disrupting Mechanisms That Regulate Genomic Repeat Elements to Combat Cancer and Drug Resistance. Front. Cell Dev. Biol. 2022, 10, 826461. [Google Scholar] [CrossRef]
- Saunders, L.R.; Bankovich, A.J.; Anderson, W.C.; Aujay, M.A.; Bheddah, S.; Black, K.; Desai, R.; Escarpe, P.A.; Hampl, J.; Laysang, A.; et al. A DLL3-Targeted Antibody-Drug Conjugate Eradicates High-Grade Pulmonary Neuroendocrine Tumor-Initiating Cells in Vivo. Sci. Transl. Med. 2015, 7, 302ra136. [Google Scholar] [CrossRef]
- Phung, Y.; Gao, W.; Man, Y.-G.; Nagata, S.; Ho, M. High-Affinity Monoclonal Antibodies to Cell Surface Tumor Antigen Glypican-3 Generated through a Combination of Peptide Immunization and Flow Cytometry Screening. MAbs 2012, 4, 592–599. [Google Scholar] [CrossRef]
- Weidle, U.H.; Eggle, D.; Klostermann, S. L1-CAM as a Target for Treatment of Cancer with Monoclonal Antibodies. Anticancer. Res. 2009, 29, 4919. [Google Scholar] [PubMed]
- Kendsersky, N.M.; Lindsay, J.; Kolb, E.A.; Smith, M.A.; Teicher, B.A.; Erickson, S.W.; Earley, E.J.; Mosse, Y.P.; Martinez, D.; Pogoriler, J.; et al. The B7-H3–Targeting Antibody–Drug Conjugate M276-SL-PBD Is Potently Effective Against Pediatric Cancer Preclinical Solid Tumor Models. Clin. Cancer Res. 2021, 27, 2938–2946. [Google Scholar] [CrossRef] [PubMed]
- Raskov, H.; Orhan, A.; Christensen, J.P.; Gögenur, I. Cytotoxic CD8+ T Cells in Cancer and Cancer Immunotherapy. Br. J. Cancer 2021, 124, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Dong, Z.; Wang, X.; Cheng, W.; Smith, D.L.; Song, J.Y.; Aldoss, I.; Forman, S.J.; Kwak, L.W. Overcoming CD19 Antigen Loss in B-Cell Malignancies with CAR T Cells Targeting BAFF-R. Blood 2019, 134, 3871. [Google Scholar] [CrossRef]
- Miyake, K.; Karasuyama, H. The Role of Trogocytosis in the Modulation of Immune Cell Functions. Cells 2021, 10, 1255. [Google Scholar] [CrossRef] [PubMed]
- Gröbner, S.N.; Worst, B.C.; Weischenfeldt, J.; Buchhalter, I.; Kleinheinz, K.; Rudneva, V.A.; Johann, P.D.; Balasubramanian, G.P.; Segura-Wang, M.; Brabetz, S.; et al. The Landscape of Genomic Alterations across Childhood Cancers. Nature 2018, 555, 321–327. [Google Scholar] [CrossRef]
- Duman, B.B.; Şahin, B.; Ergin, M.; Guvenc, B. Loss of CD20 Antigen Expression after Rituximab Therapy of CD20 Positive B Cell Lymphoma (Diffuse Large B Cell Extranodal Marginal Zone Lymphoma Combination): A Case Report and Review of the Literature. Med. Oncol. 2012, 29, 1223–1226. [Google Scholar] [CrossRef]
- Kramer, K.; Gerald, W.L.; Kushner, B.H.; Larson, S.M.; Hameed, M.; Cheune, N.-K.V. Disialoganglioside GD2 Loss Following Monoclonal Antibody Therapy Is Rare in Neuroblastoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 1998, 4, 2135–2139. [Google Scholar] [CrossRef]
- Schumacher-Kuckelkorn, R.; Hero, B.; Ernestus, K.; Berthold, F. Lacking Immunocytological GD2 Expression in Neuroblastoma: Report of Three Cases. Pediatr. Blood Cancer 2005, 45, 195–201. [Google Scholar] [CrossRef]
- Sengupta, S.; Das, S.; Crespo, A.C.; Cornel, A.M.; Patel, A.G.; Mahadevan, N.R.; Campisi, M.; Ali, A.K.; Sharma, B.; Rowe, J.H.; et al. Mesenchymal and Adrenergic Cell Lineage States in Neuroblastoma Possess Distinct Immunogenic Phenotypes. Nat. Cancer 2022, 3, 1228–1246. [Google Scholar] [CrossRef]
- Mabe, N.W.; Huang, M.; Dalton, G.N.; Alexe, G.; Schaefer, D.A.; Geraghty, A.C.; Robichaud, A.L.; Conway, A.S.; Khalid, D.; Mader, M.M.; et al. Transition to a Mesenchymal State in Neuroblastoma Confers Resistance to Anti-GD2 Antibody via Reduced Expression of ST8SIA1. Nat. Cancer 2022, 3, 976–993. [Google Scholar] [CrossRef] [PubMed]
- Kailayangiri, S.; Altvater, B.; Lesch, S.; Balbach, S.; Göttlich, C.; Kühnemundt, J.; Mikesch, J.H.; Schelhaas, S.; Jamitzky, S.; Meltzer, J.; et al. EZH2 Inhibition in Ewing Sarcoma Upregulates G D2 Expression for Targeting with Gene-Modified T Cells. Mol. Ther. 2019, 27, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Sha, Y.; Han, L.; Sun, B.; Zhao, Q. Identification of a Glycosyltransferase Signature for Predicting Prognosis and Immune Microenvironment in Neuroblastoma. Front. Cell Dev. Biol. 2022, 9, 769580. [Google Scholar] [CrossRef] [PubMed]
- Tibbetts, R.; Yeo, K.K.; Muthugounder, S.; Lee, M.-H.; Jung, C.; Porras-Corredor, T.; Sheard, M.A.; Asgharzadeh, S. Anti-Disialoganglioside Antibody Internalization by Neuroblastoma Cells as a Mechanism of Immunotherapy Resistance. Cancer Immunol. Immunother. 2022, 71, 153–164. [Google Scholar] [CrossRef]
- Saga, T.; Neumann, R.D.; Heya, T.; Sato, J.; Kinuya, S.; Le, N.; Paik, C.H.; Weinstein, J.N. Targeting Cancer Micrometastases with Monoclonal Antibodies: A Binding-Site Barrier. Proc. Natl. Acad. Sci. USA 1995, 92, 8999–9003. [Google Scholar] [CrossRef]
- Cruz, E.; Kayser, V. Monoclonal Antibody Therapy of Solid Tumors: Clinical Limitations and Novel Strategies to Enhance Treatment Efficacy. Biologics 2019, 13, 33–51. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Guo, H.; Cheung, I.Y.; Cheung, N.K.V. Antitumor Efficacy of Anti-GD2 IgG1 Is Enhanced by Fc Glyco-Engineering. Cancer Immunol. Res. 2016, 4, 631–638. [Google Scholar] [CrossRef]
- Fu, Z.; Li, S.; Han, S.; Shi, C.; Zhang, Y. Antibody Drug Conjugate: The “Biological Missile” for Targeted Cancer Therapy. Signal. Transduct. Target. Ther. 2022, 7, 93. [Google Scholar] [CrossRef]
- Kalinovsky, D.V.; Kibardin, A.V.; Kholodenko, I.V.; Svirshchevskaya, E.V.; Doronin, I.I.; Konovalova, M.V.; Grechikhina, M.V.; Rozov, F.N.; Larin, S.S.; Deyev, S.M.; et al. Therapeutic Efficacy of Antibody-Drug Conjugates Targeting GD2-Positive Tumors. J. Immunother. Cancer 2022, 10, e004646. [Google Scholar] [CrossRef]
- Kramer, K.; Humm, J.L.; Souweidane, M.M.; Zanzonico, P.B.; Dunkel, I.J.; Gerald, W.L.; Khakoo, Y.; Yeh, S.D.; Yeung, H.W.; Finn, R.D.; et al. Phase I Study of Targeted Radioimmunotherapy for Leptomeningeal Cancers Using Intra-Ommaya 131-I-3F8. J. Clin. Oncol. 2007, 25, 5465–5470. [Google Scholar] [CrossRef]
- Kramer, K.; Pandit-Taskar, N.; Humm, J.L.; Zanzonico, P.B.; Haque, S.; Dunkel, I.J.; Wolden, S.L.; Donzelli, M.; Goldman, D.A.; Lewis, J.S.; et al. A Phase II Study of Radioimmunotherapy with Intraventricular 131 I-3F8 for Medulloblastoma. Pediatr. Blood Cancer 2018, 65, e26754. [Google Scholar] [CrossRef] [PubMed]
- Larson, S.M.; Carrasquillo, J.A.; Cheung, N.-K.V.; Press, O.W. Radioimmunotherapy of Human Tumours. Nat. Rev. Cancer 2015, 15, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Santich, B.H.; Cheal, S.M.; Ahmed, M.; McDevitt, M.R.; Ouerfelli, O.; Yang, G.; Veach, D.R.; Fung, E.K.; Patel, M.; Burnes Vargas, D.; et al. A Self-Assembling and Disassembling (SADA) Bispecific Antibody (BsAb) Platform for Curative Two-Step Pretargeted Radioimmunotherapy. Clin. Cancer Res. 2021, 27, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Tsao, C.-Y.; Sabbatino, F.; Cheung, N.-K.V.; Chi-Feng Hsu, J.; Villani, V.; Wang, X.; Ferrone, S. Anti-Proliferative and pro-Apoptotic Activity of GD2 Ganglioside-Specific Monoclonal Antibody 3F8 in Human Melanoma Cells. Oncoimmunology 2015, 4, e1023975. [Google Scholar] [CrossRef] [PubMed]
- Adams, G.P.; Weiner, L.M. Monoclonal Antibody Therapy of Cancer. Nat. Biotechnol. 2005, 23, 1147–1157. [Google Scholar] [CrossRef]
- Braster, R.; O’Toole, T.; van Egmond, M. Myeloid Cells as Effector Cells for Monoclonal Antibody Therapy of Cancer. Methods 2014, 65, 28–37. [Google Scholar] [CrossRef]
- Clynes, R.A.; Towers, T.L.; Presta, L.G.; Ravetch, J.V. Inhibitory Fc Receptors Modulate in Vivo Cytoxicity against Tumor Targets. Nat. Med. 2000, 6, 443–446. [Google Scholar] [CrossRef]
- Capuano, C.; Romanelli, M.; Pighi, C.; Cimino, G.; Rago, A.; Molfetta, R.; Paolini, R.; Santoni, A.; Galandrini, R. Anti-CD20 Therapy Acts via FcγRIIIA to Diminish Responsiveness of Human Natural Killer Cells. Cancer Res. 2015, 75, 4097–4108. [Google Scholar] [CrossRef]
- Dahal, L.N.; Roghanian, A.; Beers, S.A.; Cragg, M.S. FcγR Requirements Leading to Successful Immunotherapy. Immunol. Rev. 2015, 268, 104–122. [Google Scholar] [CrossRef]
- Kushner, B.; Cheung, N. GM-CSF Enhances 3F8 Monoclonal Antibody-Dependent Cellular Cytotoxicity against Human Melanoma and Neuroblastoma. Blood 1989, 73, 1936–1941. [Google Scholar] [CrossRef]
- Ozkaynak, M.F.; Gilman, A.L.; London, W.B.; Naranjo, A.; Diccianni, M.B.; Tenney, S.C.; Smith, M.; Messer, K.S.; Seeger, R.; Reynolds, C.P.; et al. A Comprehensive Safety Trial of Chimeric Antibody 14.18 with GM-CSF, IL-2, and Isotretinoin in High-Risk Neuroblastoma Patients Following Myeloablative Therapy: Children’s Oncology Group Study ANBL0931. Front. Immunol. 2018, 9, 1355. [Google Scholar] [CrossRef] [PubMed]
- Ladenstein, R.L.; Poetschger, U.; Valteau-Couanet, D.; Gray, J.; Luksch, R.; Balwierz, W.; Castel, V.; Ash, S.; Popovic, M.B.; Laureys, G.; et al. Randomization of Dose-Reduced Subcutaneous Interleukin-2 (ScIL2) in Maintenance Immunotherapy (IT) with Anti-GD2 Antibody Dinutuximab Beta (DB) Long-Term Infusion (LTI) in Front–Line High-Risk Neuroblastoma Patients: Early Results from the HR-NBL1/SIOPEN Trial. J. Clin. Oncol. 2019, 37, 10013. [Google Scholar] [CrossRef]
- Nguyen, R.; Zhang, X.; Sun, M.; Abbas, S.; Seibert, C.; Kelly, M.C.; Shern, J.F.; Thiele, C.J. Anti-GD2 Antibodies Conjugated to IL15 and IL21 Mediate Potent Antitumor Cytotoxicity against Neuroblastoma. Clin. Cancer Res. 2022, 28, 3785–3796. [Google Scholar] [CrossRef] [PubMed]
- Shinkawa, T.; Nakamura, K.; Yamane, N.; Shoji-Hosaka, E.; Kanda, Y.; Sakurada, M.; Uchida, K.; Anazawa, H.; Satoh, M.; Yamasaki, M.; et al. The Absence of Fucose but Not the Presence of Galactose or Bisecting N-Acetylglucosamine of Human IgG1 Complex-Type Oligosaccharides Shows the Critical Role of Enhancing Antibody-Dependent Cellular Cytotoxicity. J. Biol. Chem. 2003, 278, 3466–3473. [Google Scholar] [CrossRef] [PubMed]
- Oganesyan, V.; Damschroder, M.M.; Leach, W.; Wu, H.; Dall’Acqua, W.F. Structural Characterization of a Mutated, ADCC-Enhanced Human Fc Fragment. Mol. Immunol. 2008, 45, 1872–1882. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Cotton, M.; Cheung, N.K.V. Bispecific Antibodies for the Treatment of Neuroblastoma. Pharmacol. Ther. 2022, 237, 108241. [Google Scholar] [CrossRef]
- Aldoss, I.; Bargou, R.C.; Nagorsen, D.; Friberg, G.R.; Baeuerle, P.A.; Forman, S.J. Redirecting T Cells to Eradicate B-Cell Acute Lymphoblastic Leukemia: Bispecific T-Cell Engagers and Chimeric Antigen Receptors. Leukemia 2017, 31, 777–787. [Google Scholar] [CrossRef]
- Algeri, M.; Del Bufalo, F.; Galaverna, F.; Locatelli, F. Current and Future Role of Bispecific T-Cell Engagers in Pediatric Acute Lymphoblastic Leukemia. Expert. Rev. Hematol. 2018, 11, 945–956. [Google Scholar] [CrossRef]
- Ribera, J.-M.; Genescà, E.; Ribera, J. Bispecific T-Cell Engaging Antibodies in B-Cell Precursor Acute Lymphoblastic Leukemias: Focus on Blinatumomab. Ther. Adv. Hematol. 2020, 11, 2040620720919632. [Google Scholar] [CrossRef]
- Lopez-Albaitero, A.; Xu, H.; Guo, H.; Wang, L.; Wu, Z.; Tran, H.; Chandarlapaty, S.; Scaltriti, M.; Janjigian, Y.; de Stanchina, E.; et al. Overcoming Resistance to HER2-Targeted Therapy with a Novel HER2/CD3 Bispecific Antibody. Oncoimmunology 2017, 6, e1267891. [Google Scholar] [CrossRef]
- Xu, H.; Cheng, M.; Guo, H.; Chen, Y.; Huse, M.; Cheung, N.-K. V Retargeting T Cells to GD2 Pentasaccharide on Human Tumors Using Bispecific Humanized Antibody. Cancer Immunol. Res. 2015, 3, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Yankelevich, M.; Kondadasula, S.V.; Thakur, A.; Buck, S.; Cheung, N.K.V.; Lum, L.G. Anti-CD3×anti-GD2 Bispecific Antibody Redirects T-Cell Cytolytic Activity to Neuroblastoma Targets. Pediatr. Blood Cancer 2012, 59, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, A.; Kwan, B.H.; Opel, C.F.; Navaratna, T.; Wittrup, K.D. Antigen Specificity Can Be Irrelevant to Immunocytokine Efficacy and Biodistribution. Proc. Natl. Acad. Sci. USA 2015, 112, 3320–3325. [Google Scholar] [CrossRef] [PubMed]
- Shusterman, S.; London, W.B.; Gillies, S.D.; Hank, J.A.; Voss, S.D.; Seeger, R.C.; Reynolds, C.P.; Kimball, J.; Albertini, M.R.; Wagner, B.; et al. Antitumor Activity of Hu14.18-IL2 in Patients with Relapsed/Refractory Neuroblastoma: A Children’s Oncology Group (COG) Phase II Study. J. Clin. Oncol. 2010, 28, 4969–4975. [Google Scholar] [CrossRef]
- Nguyen, R.; Moustaki, A.; Norrie, J.L.; Brown, S.; Akers, W.J.; Shirinifard, A.; Dyer, M.A. Interleukin-15 Enhances Anti-GD2 Antibody-Mediated Cytotoxicity in an Orthotopic PDX Model of Neuroblastoma. Clin. Cancer Res. 2019, 25, 7554–7564. [Google Scholar] [CrossRef]
- Cheung, N.-K.V.; Cañete, A.; Cheung, I.Y.; Ye, J.-N.; Liu, C. Disialoganglioside g d 2 anti-idiotypic monoclonal antibodies. Int. J. Cancer 1993, 54, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Furman, W.L.; McCarville, B.; Shulkin, B.L.; Davidoff, A.; Krasin, M.; Hsu, C.W.; Pan, H.; Wu, J.; Brennan, R.; Bishop, M.W.; et al. Improved Outcome in Children with Newly Diagnosed High-Risk Neuroblastoma Treated with Chemoimmunotherapy: Updated Results of a Phase II Study Using Hu14.18K322A. J. Clin. Oncol. 2021, 40, 335–344. [Google Scholar] [CrossRef]
- Park, J.R.; Scott, J.R.; Stewart, C.F.; London, W.B.; Naranjo, A.; Santana, V.M.; Shaw, P.J.; Cohn, S.L.; Matthay, K.K. Pilot Induction Regimen Incorporating Pharmacokinetically Guided Topotecan for Treatment of Newly Diagnosed High-Risk Neuroblastoma: A Children’s Oncology Group Study. J. Clin. Oncol. 2011, 29, 4351–4357. [Google Scholar] [CrossRef]
- Kushner, B.H.; Cheung, I.Y.; Modak, S.; Kramer, K.; Ragupathi, G.; Cheung, N.K.V. Phase i Trial of a Bivalent Gangliosides Vaccine in Combination with β-Glucan for High-Risk Neuroblastoma in Second or Later Remission. Clin. Cancer Res. 2014, 20, 1375–1382. [Google Scholar] [CrossRef]
- Cheung, I.Y.; Cheung, N.K.V.; Modak, S.; Mauguen, A.; Feng, Y.; Basu, E.; Roberts, S.S.; Ragupathi, G.; Kushner, B.H. Survival Impact of Anti-GD2 Antibody Response in a Phase II Ganglioside Vaccine Trial Among Patients With High-Risk Neuroblastoma With Prior Disease Progression. J. Clin. Oncol. 2021, 39, 215–226. [Google Scholar] [CrossRef]
- Cheung, N.-K.V.; Sowers, R.; Vickers, A.J.; Cheung, I.Y.; Kushner, B.H.; Gorlick, R. FCGR2A Polymorphism Is Correlated With Clinical Outcome After Immunotherapy of Neuroblastoma With Anti-GD2 Antibody and Granulocyte Macrophage Colony-Stimulating Factor. J. Clin. Oncol. 2006, 24, 2885–2890. [Google Scholar] [CrossRef] [PubMed]
- Forlenza, C.J.; Boudreau, J.E.; Zheng, J.; Le Luduec, J.-B.; Chamberlain, E.; Heller, G.; Cheung, N.-K.V.; Hsu, K.C. KIR3DL1 Allelic Polymorphism and HLA-B Epitopes Modulate Response to Anti-GD2 Monoclonal Antibody in Patients with Neuroblastoma. J. Clin. Oncol. 2016, 34, 2443–2451. [Google Scholar] [CrossRef]
- Cheung, N.-K.K.V.; Ostrovnaya, I.; Kuk, D.; Cheung, I.Y. Bone Marrow Minimal Residual Disease Was an Early Response Marker and a Consistent Independent Predictor of Survival After Anti-GD2 Immunotherapy. J. Clin. Oncol. 2015, 33, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, J.; Schwenck, J.; Maurer, A.; Przybille, M.; Sonanini, D.; Reischl, G.; Wehrmüller, J.E.; Quintanilla-Martinez, L.; Gillies, S.D.; Krueger, M.A.; et al. Translational ImmunoPET Imaging Using a Radiolabeled GD2-Specific Antibody in Neuroblastoma. Theranostics 2022, 12, 5615–5630. [Google Scholar] [CrossRef] [PubMed]
- Trautwein, N.F.; Reischl, G.; Seitz, C.; Dittmann, H.; Seith, F.; Scheuermann, S.; Feuchtinger, T.; Dombrowski, F.; Handgretinger, R.; Fuchs, J.; et al. First in Human PET/MRI Imaging of in Vivo GD2 Expression in Osteosarcoma. J. Nucl. Med. 2023, 64, 337–338. [Google Scholar] [CrossRef] [PubMed]
- Butch, E.R.; Mead, P.E.; Diaz, V.A.; Tillman, H.; Stewart, E.; Mishra, J.K.; Kim, J.; Bahrami, A.; Dearling, J.L.J.; Packard, A.B.; et al. Positron Emission Tomography Detects In Vivo Expression of Disialoganglioside GD2 in Mouse Models of Primary and Metastatic Osteosarcoma. Cancer Res. 2019, 79, 3112–3124. [Google Scholar] [CrossRef] [PubMed]
- Balis, F.M.; Busch, C.M.; Desai, A.V.; Hibbitts, E.; Naranjo, A.; Bagatell, R.; Irwin, M.; Fox, E. The Ganglioside GD2 as a Circulating Tumor Biomarker for Neuroblastoma. Pediatr. Blood Cancer 2020, 67, e28031. [Google Scholar] [CrossRef] [PubMed]
- Busch, C.M.; Desai, A.V.; Moorthy, G.S.; Fox, E.; Balis, F.M. A Validated HPLC-MS/MS Method for Estimating the Concentration of the Ganglioside, GD2, in Human Plasma or Serum. J. Chromatogr. B 2018, 1102–1103, 60–65. [Google Scholar] [CrossRef]
- Mody, R.; Naranjo, A.; Van Ryn, C.; Yu, A.L.; London, W.B.; Shulkin, B.L.; Parisi, M.T.; Servaes, S.E.N.; Diccianni, M.B.; Sondel, P.M.; et al. Irinotecan–Temozolomide with Temsirolimus or Dinutuximab in Children with Refractory or Relapsed Neuroblastoma (COG ANBL1221): An Open-Label, Randomised, Phase 2 Trial. Lancet Oncol. 2017, 18, 946–957. [Google Scholar] [CrossRef]
- Modak, S.; Kushner, B.H.; Mauguen, A.; Castañeda, A.; Varo, A.; Gorostegui, M.; Muñoz, J.P.; Santa-Maria, V.; Basu, E.M.; Iglesias Cardenas, F.; et al. Naxitamab-Based Chemoimmunotherapy for Resistant High-Risk Neuroblastoma: Results of “HITS” Phase II Study. J. Clin. Oncol. 2022, 40, 10028. [Google Scholar] [CrossRef]
- Gray, J.; Moreno, L.; Weston, R.; Barone, G.; Rubio, A.; Makin, G.; Vaidya, S.; Ng, A.; Castel, V.; Nysom, K.; et al. BEACON-Immuno: Results of the Dinutuximab Beta (DB) Randomization of the BEACON-Neuroblastoma Phase 2 Trial-A European Innovative Therapies for Children with Cancer) Trial. J. Clin. Oncol. 2022, 40, 10002. [Google Scholar] [CrossRef]
- Mora, J.; Chan, G.C.; Morgenstern, D.A.; Nysom, K.; Bear, M.K.; Tornøe, K.; Kushner, B.H. Outpatient Administration of Naxitamab in Combination with Granulocyte-macrophage Colony-stimulating Factor in Patients with Refractory and/or Relapsed High-risk Neuroblastoma: Management of Adverse Events. Cancer Rep. 2023, 6, e1627. [Google Scholar] [CrossRef] [PubMed]
- Castañeda, A.; Gorostegui, M.; Miralles, S.L.; Chamizo, A.; Patiño, S.C.; Flores, M.A.; Garraus, M.; Lazaro, J.J.; Santa-Maria, V.; Varo, A.; et al. How We Approach the Treatment of Patients with High-Risk Neuroblastoma with Naxitamab: Experience from the Hospital Sant Joan de Déu in Barcelona, Spain. ESMO Open. 2022, 7, 100462. [Google Scholar] [CrossRef] [PubMed]
- Navid, F.; Sondel, P.M.; Barfield, R.; Shulkin, B.L.; Kaufman, R.A.; Allay, J.A.; Gan, J.; Hutson, P.; Seo, S.; Kim, K.; et al. Phase I Trial of a Novel Anti-GD2 Monoclonal Antibody, Hu14.18K322A, Designed to Decrease Toxicity in Children With Refractory or Recurrent Neuroblastoma. J. Clin. Oncol. 2014, 32, 1445–1452. [Google Scholar] [CrossRef]
- Varo, A.; Castañeda, A.; Chamorro, S.; Muñoz, J.P.; Gorostegui, M.; Celma, M.S.; Lopez, S.; Simao, M.; Perez-Jaume, S.; Mora, J. Novel Infusion Strategy Reduces Severe Adverse Events Caused by the Anti-GD2 Monoclonal Antibody Naxitamab. Front. Oncol. 2023, 13, 1164949. [Google Scholar] [CrossRef] [PubMed]
- Majzner, R.G.; Ramakrishna, S.; Yeom, K.W.; Patel, S.; Chinnasamy, H.; Schultz, L.M.; Richards, R.M.; Jiang, L.; Barsan, V.; Mancusi, R.; et al. GD2-CAR T Cell Therapy for H3K27M-Mutated Diffuse Midline Gliomas. Nature 2022, 603, 934–941. [Google Scholar] [CrossRef]
- Del Bufalo, F.; De Angelis, B.; Caruana, I.; Del Baldo, G.; De Ioris, M.A.; Serra, A.; Mastronuzzi, A.; Cefalo, M.G.; Pagliara, D.; Amicucci, M.; et al. GD2-CART01 for Relapsed or Refractory High-Risk Neuroblastoma. N. Engl. J. Med. 2023, 388, 1284–1295. [Google Scholar] [CrossRef]
- Assessing National Capacity for the Prevention and Control of Noncommunicable Diseases: Report of the 2019 Global Survey. Available online: https://www.who.int/publications/i/item/9789240002319 (accessed on 17 April 2023).
- Schoot, R.A.; Otth, M.A.; Frederix, G.W.J.; Leufkens, H.G.M.; Vassal, G. Market Access to New Anticancer Medicines for Children and Adolescents with Cancer in Europe. Eur. J. Cancer 2022, 165, 146–153. [Google Scholar] [CrossRef]
- CADTH Reimbursement Recommendation Dinutuximab (Unituxin) 2. Available online: https://www.cadth.ca/dinutuximab (accessed on 17 April 2023).
- Petrou, S. Methodological Challenges Surrounding QALY Estimation for Paediatric Economic Evaluation. Cost. Eff. Resour. Alloc. 2022, 20, 10. [Google Scholar] [CrossRef]
- Mora, J. Autologous Stem-Cell Transplantation for High-Risk Neuroblastoma: Historical and Critical Review. Cancers 2022, 14, 2572. [Google Scholar] [CrossRef]
- Bird, N.; Scobie, N.; Palmer, A.; Ludwinski, D. To Transplant, or Not to Transplant? That Is the Question. A Patient Advocate Evaluation of Autologous Stem Cell Transplant in Neuroblastoma. Pediatr. Blood Cancer 2022, 69, e29663. [Google Scholar] [CrossRef] [PubMed]
- Elgundi, Z.; Reslan, M.; Cruz, E.; Sifniotis, V.; Kayser, V. The State-of-Play and Future of Antibody Therapeutics. Adv. Drug. Deliv. Rev. 2017, 122, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Laetsch, T.W.; DuBois, S.G.; Glade Bender, J.; Macy, M.E.; Moreno, L. Opportunities and Challenges in Drug Development for Pediatric Cancers. Cancer Discov. 2021, 11, 545–559. [Google Scholar] [CrossRef] [PubMed]
- Pritchard-Jones, K.; Pieters, R.; Reaman, G.H.; Hjorth, L.; Downie, P.; Calaminus, G.; Naafs-Wilstra, M.C.; Steliarova-Foucher, E. Sustaining Innovation and Improvement in the Treatment of Childhood Cancer: Lessons from High-Income Countries. Lancet Oncol. 2013, 14, e95–e103. [Google Scholar] [CrossRef]
| MAb | FDA Approval for Adults | FDA Approval for Children | EMA Approval for Adults | EMA Approval for Children | Median Time Gap (Years) |
|---|---|---|---|---|---|
| Rituximab | 1997 | 2021 | 1998 | 2020 | 23 |
| Breuntuximab vedotin | 2011 | 2022 | NA | NA | 11 |
| Blinatumomab | 2014 | 2018 | 2015 | 2016 | 2.5 |
| Gemtuzumab ozogamycin | 2017 1 | 2017 | 2017 2 | 2017 | 0 |
| Inotuzumab | 2017 | NA | 2017 | NA | NA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larrosa, C.; Mora, J.; Cheung, N.-K. Global Impact of Monoclonal Antibodies (mAbs) in Children: A Focus on Anti-GD2. Cancers 2023, 15, 3729. https://doi.org/10.3390/cancers15143729
Larrosa C, Mora J, Cheung N-K. Global Impact of Monoclonal Antibodies (mAbs) in Children: A Focus on Anti-GD2. Cancers. 2023; 15(14):3729. https://doi.org/10.3390/cancers15143729
Chicago/Turabian StyleLarrosa, Cristina, Jaume Mora, and Nai-Kong Cheung. 2023. "Global Impact of Monoclonal Antibodies (mAbs) in Children: A Focus on Anti-GD2" Cancers 15, no. 14: 3729. https://doi.org/10.3390/cancers15143729
APA StyleLarrosa, C., Mora, J., & Cheung, N.-K. (2023). Global Impact of Monoclonal Antibodies (mAbs) in Children: A Focus on Anti-GD2. Cancers, 15(14), 3729. https://doi.org/10.3390/cancers15143729








