Potential Correlation between Changes in Serum FGF21 Levels and Lenvatinib-Induced Appetite Loss in Patients with Unresectable Hepatocellular Carcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Study Design
2.2. Analysis of FGF21
2.3. Lenvatinib Treatment Protocol
2.4. Statistical Analysis
3. Results
3.1. Patient Enrollment and Baseline Characteristics
3.2. Occurrence Rate of Appetite Loss during Lenvatinib Treatment and Comparison between Patients with or without Grade ≥ 2 Appetite Loss
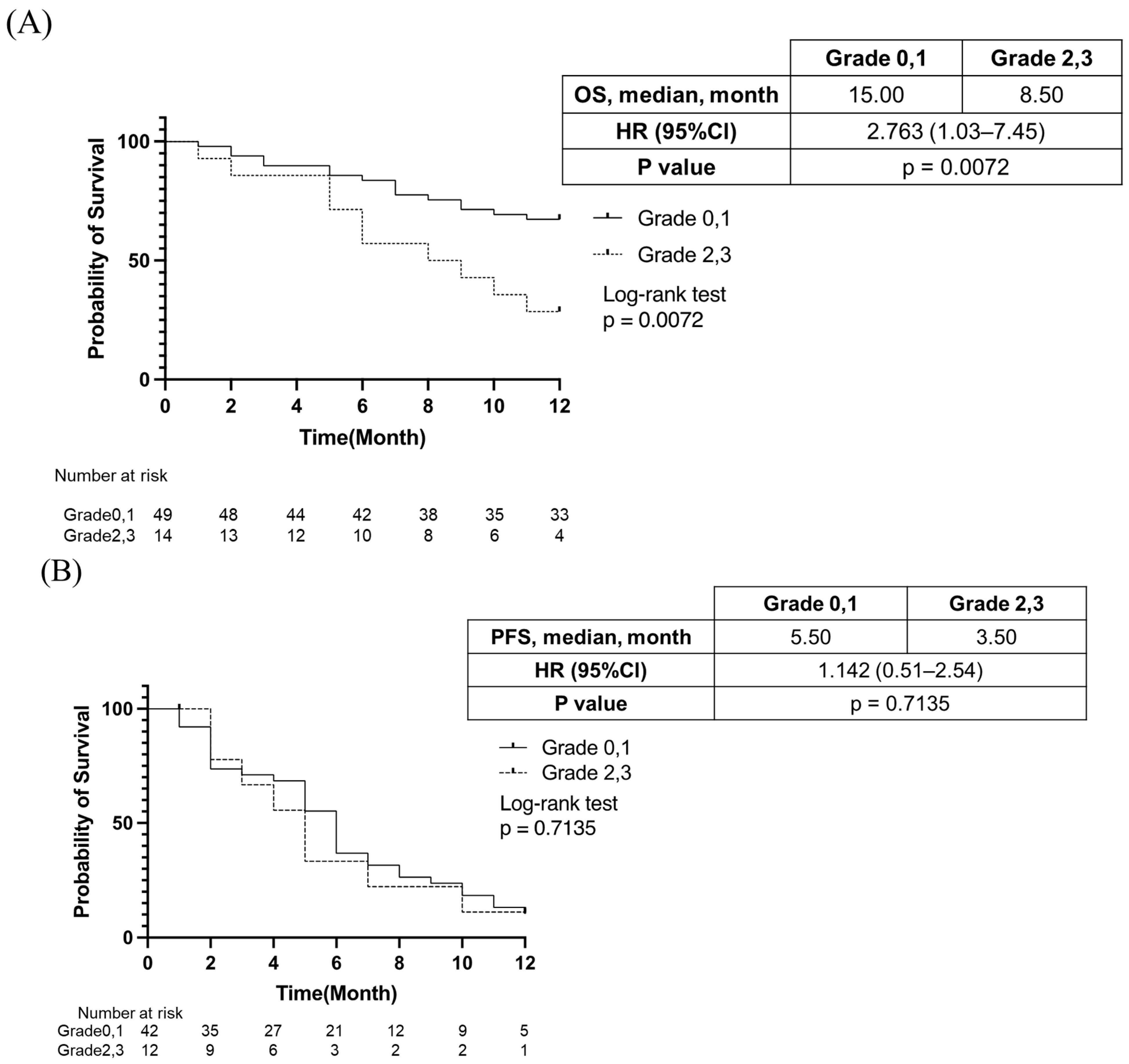
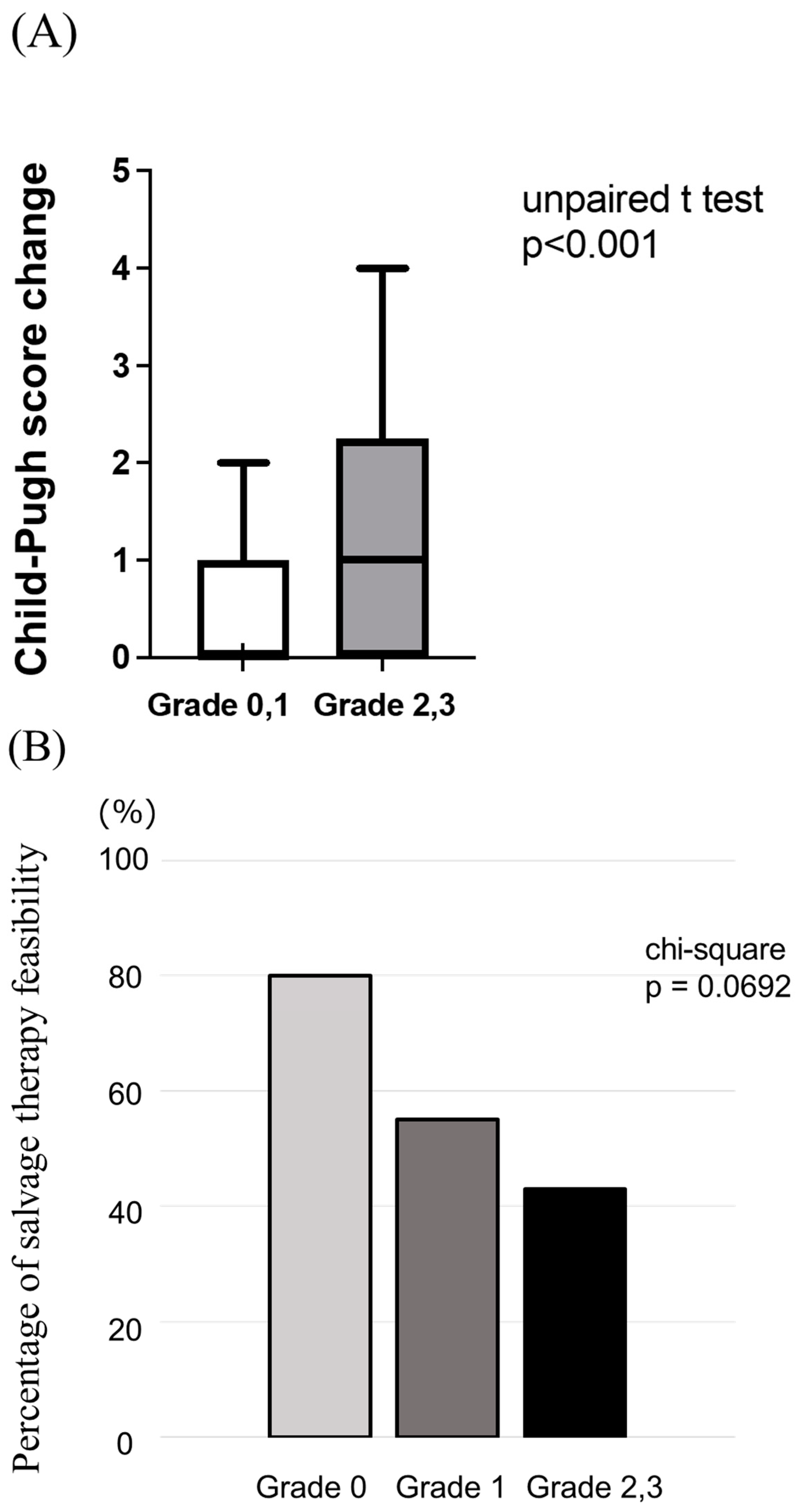
3.3. Impact of Grade ≥ 2 Appetite Loss on Treatment Response, Prognosis, and Liver Functional Reserve
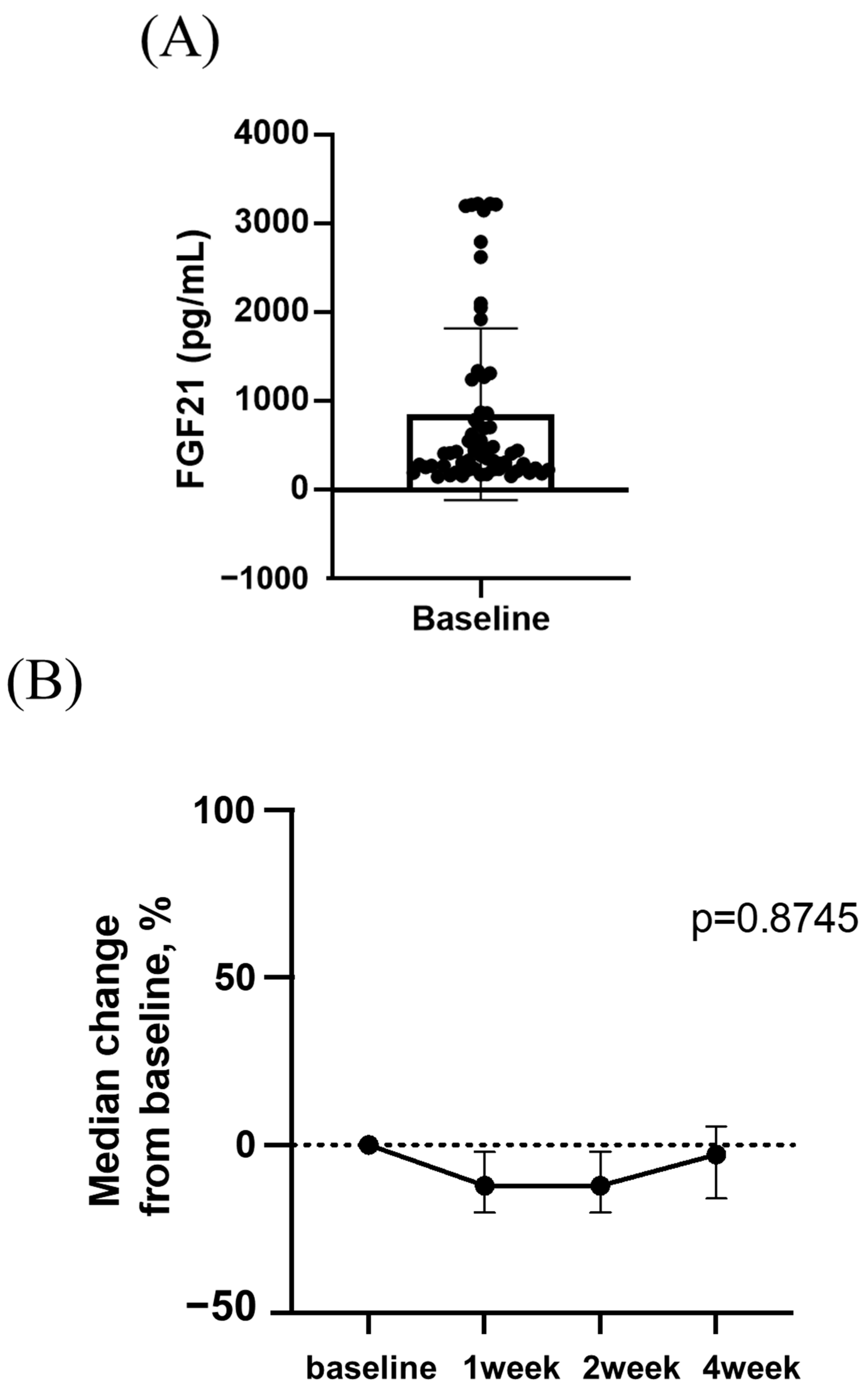
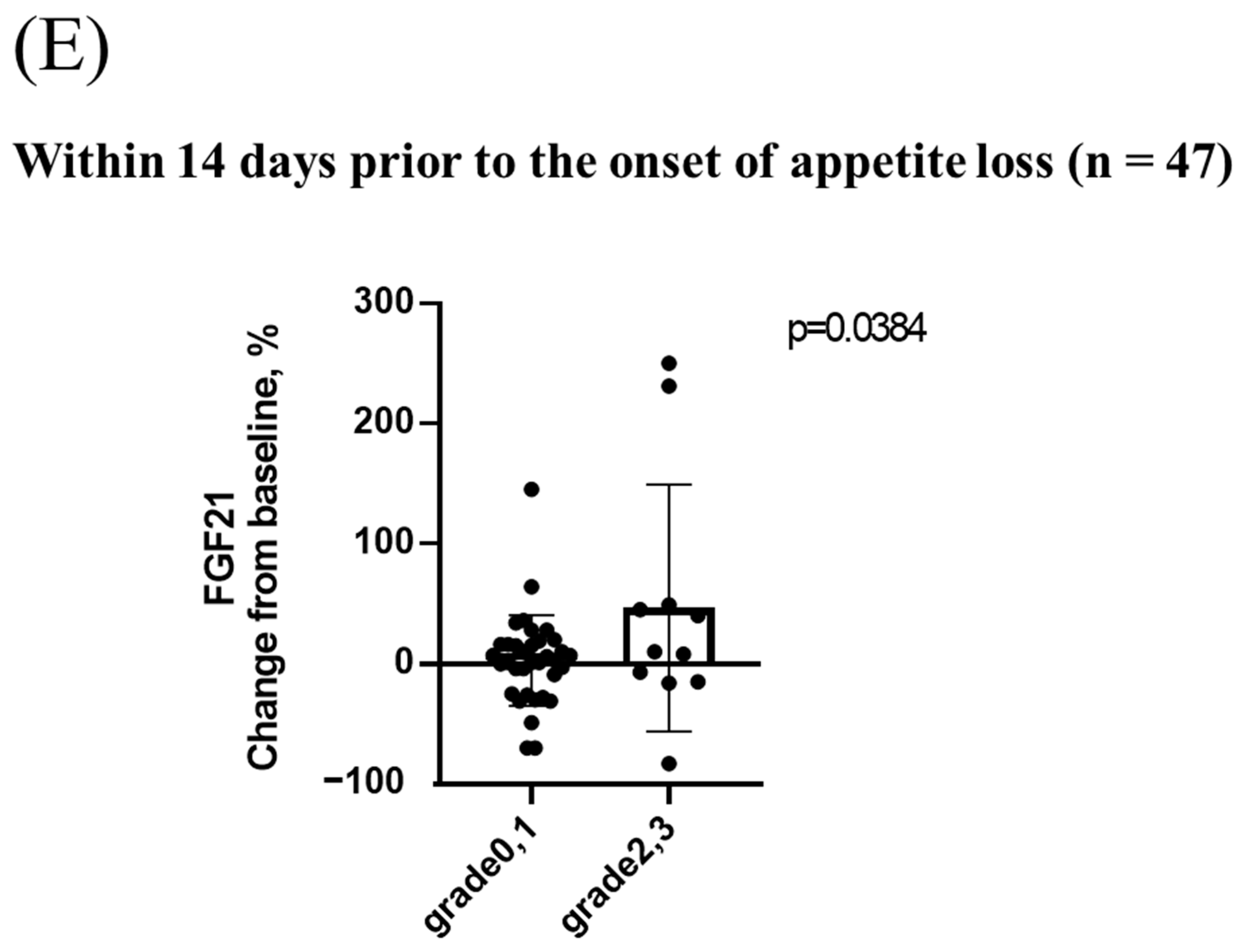
3.4. Changes in Serum FGF21 Levels during Lenvatinib Treatment and the Relationship between Changes in Serum FGF21 Levels and Appetite Loss
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Llovet, J.M.; Burroughs, A.; Bruix, J. Hepatocellular carcinoma. Lancet 2003, 362, 1907–1917. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular carcinoma. Nat. Rev. Dis. Prim. 2021, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Kudo, M.; Tsuchiya, K.; Kato, N.; Hagihara, A.; Numata, K.; Aikata, H.; Inaba, Y.; Kondo, S.; Motomura, K.; Furuse, J.; et al. Cabozantinib in Japanese patients with advanced hepatocellular carcinoma: A phase 2 multicenter study. J. Gastroenterol. 2021, 56, 181–190. [Google Scholar] [CrossRef]
- Zhu, A.X.; Kang, Y.K.; Yen, C.J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased alpha-fetoprotein concentrations (REACH-2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Casadei-Gardini, A.; Rimini, M.; Kudo, M.; Shimose, S.; Tada, T.; Suda, G.; Goh, M.J.; Jefremow, A.; Scartozzi, M.; Cabibbo, G.; et al. Real life study of lenvatinib therapy for hepatocellular carcinoma: RELEVANT study. Liver Cancer 2022, 11, 527–539. [Google Scholar] [CrossRef]
- Hosoda, S.; Suda, G.; Sho, T.; Ogawa, K.; Kimura, M.; Yang, Z.; Yoshida, S.; Kubo, A.; Tokuchi, Y.; Kitagataya, T.; et al. Low baseline CXCL9 predicts early progressive disease in unresectable HCC with atezolizumab plus bevacizumab treatment. Liver Cancer 2022. [Google Scholar] [CrossRef]
- Yang, Z.; Suda, G.; Maehara, O.; Ohara, M.; Yoda, T.; Sasaki, T.; Kohya, R.; Yoshida, S.; Hosoda, S.; Tokuchi, Y.; et al. Changes in serum growth factors during resistance to atezolizumab plus bevacizumab treatment in patients with unresectable hepatocellular carcinoma. Cancers 2023, 15, 593. [Google Scholar] [CrossRef]
- Yang, Z.; Suda, G.; Maehara, O.; Ohara, M.; Yoshida, S.; Hosoda, S.; Kimura, M.; Kubo, A.; Tokuchi, Y.; Fu, Q.; et al. Changes in serum growth factors during lenvatinib predict the post progressive survival in patients with unresectable hepatocellular carcinoma. Cancers 2022, 14, 232. [Google Scholar] [CrossRef] [PubMed]
- Sho, T.; Suda, G.; Ogawa, K.; Kimura, M.; Kubo, A.; Tokuchi, Y.; Kitagataya, T.; Maehara, O.; Ohnishi, S.; Shigesawa, T.; et al. Early response and safety of atezolizumab plus bevacizumab for unresectable hepatocellular carcinoma in patients who do not meet IMbrave150 eligibility criteria. Hepatol. Res. 2021, 51, 979–989. [Google Scholar] [CrossRef] [PubMed]
- Sho, T.; Suda, G.; Ogawa, K.; Shigesawa, T.; Suzuki, K.; Nakamura, A.; Ohara, M.; Umemura, M.; Kawagishi, N.; Natsuizaka, M.; et al. Lenvatinib in patients with unresectable hepatocellular carcinoma who do not meet the REFLECT trial eligibility criteria. Hepatol. Res. 2020, 50, 966–977. [Google Scholar] [CrossRef] [PubMed]
- Gordan, J.D.; Kennedy, E.B.; Abou-Alfa, G.K.; Beg, M.S.; Brower, S.T.; Gade, T.P.; Goff, L.; Gupta, S.; Guy, J.; Harris, W.P.; et al. Systemic therapy for advanced hepatocellular carcinoma: ASCO guideline. J. Clin. Oncol. 2020, 38, 4317–4345. [Google Scholar] [CrossRef]
- Shigesawa, T.; Maehara, O.; Suda, G.; Natsuizaka, M.; Kimura, M.; Shimazaki, T.; Yamamoto, K.; Yamada, R.; Kitagataya, T.; Nakamura, A.; et al. Lenvatinib suppresses cancer stem-like cells in HCC by inhibiting FGFR 1-3 signaling, but not FGFR4 signaling. Carcinogenesis 2021, 42, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Kariyama, K.; Takaguchi, K.; Itobayashi, E.; Shimada, N.; Tajiri, K.; Tsuji, K.; Ishikawa, T.; Ochi, H.; et al. Therapeutic potential of lenvatinib for unresectable hepatocellular carcinoma in clinical practice: Multicenter analysis. Hepatol. Res. 2019, 49, 111–117. [Google Scholar] [CrossRef]
- Shimose, S.; Iwamoto, H.; Niizeki, T.; Shirono, T.; Noda, Y.; Kamachi, N.; Okamura, S.; Nakano, M.; Suga, H.; Kuromatsu, R.; et al. Clinical significance of adverse events for patients with unresectable hepatocellular carcinoma treated with lenvatinib: A multicenter retrospective study. Cancers 2020, 12, 1867. [Google Scholar] [CrossRef]
- Finn, R.S.; Kudo, M.; Cheng, A.L.; Wyrwicz, L.; Ngan, R.K.C.; Blanc, J.F.; Baron, A.D.; Vogel, A.; Ikeda, M.; Piscaglia, F.; et al. Pharmacodynamic biomarkers predictive of survival benefit with lenvatinib in unresectable hepatocellular carcinoma: From the Phase III REFLECT study. Clin. Cancer Res. 2021, 27, 4848–4858. [Google Scholar] [CrossRef]
- Tillman, E.J.; Rolph, T. FGF21: An emerging therapeutic target for non-alcoholic steatohepatitis and related metabolic diseases. Front. Endocrinol. 2020, 11, 601290. [Google Scholar] [CrossRef]
- Flippo, K.H.; Potthoff, M.J. Metabolic messengers: FGF21. Nat. Metab. 2021, 3, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Santoso, P.; Nakata, M.; Shiizaki, K.; Boyang, Z.; Parmila, K.; Otgon-Uul, Z.; Hashimoto, K.; Satoh, T.; Mori, M.; Kuro, O.M.; et al. Fibroblast growth factor 21, assisted by elevated glucose, activates paraventricular nucleus NUCB2/Nesfatin-1 neurons to produce satiety under fed states. Sci. Rep. 2017, 7, 45819. [Google Scholar] [CrossRef] [PubMed]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Shigesawa, T.; Suda, G.; Kimura, M.; Shimazaki, T.; Maehara, O.; Yamada, R.; Kitagataya, T.; Suzuki, K.; Nakamura, A.; Ohara, M.; et al. Baseline angiopoietin-2 and FGF19 levels predict treatment response in patients receiving multikinase inhibitors for hepatocellular carcinoma. JGH Open 2020, 4, 880–888. [Google Scholar] [CrossRef]
- von Holstein-Rathlou, S.; Gillum, M.P. Fibroblast growth factor 21: An endocrine inhibitor of sugar and alcohol appetite. J. Physiol. 2019, 597, 3539–3548. [Google Scholar] [CrossRef]
- Franz, K.; Ost, M.; Otten, L.; Herpich, C.; Coleman, V.; Endres, A.S.; Klaus, S.; Muller-Werdan, U.; Norman, K. Higher serum levels of fibroblast growth factor 21 in old patients with cachexia. Nutrition 2019, 63–64, 81–86. [Google Scholar] [CrossRef]
- Morimoto, T.; Machii, K.; Matsumoto, H.; Takai, S. Web Questionnaire Survey on Appetite Loss and Weight Loss Associated with Cancer Cachexia Japanese Evidence for Patients of Cancer Cachexia(J-EPOCC)—The understanding of cancer cachexia. Gan To Kagaku Ryoho 2020, 47, 1075–1080. [Google Scholar]
- Solheim, T.S.; Laird, B.J.A.; Balstad, T.R.; Bye, A.; Stene, G.; Baracos, V.; Strasser, F.; Griffiths, G.; Maddocks, M.; Fallon, M.; et al. Cancer cachexia: Rationale for the MENAC (Multimodal-Exercise, Nutrition and Anti-inflammatory medication for Cachexia) trial. BMJ Support. Palliat. Care 2018, 8, 258–265. [Google Scholar] [CrossRef]
- Galman, C.; Lundasen, T.; Kharitonenkov, A.; Bina, H.A.; Eriksson, M.; Hafstrom, I.; Dahlin, M.; Amark, P.; Angelin, B.; Rudling, M. The circulating metabolic regulator FGF21 is induced by prolonged fasting and PPARalpha activation in man. Cell Metab. 2008, 8, 169–174. [Google Scholar] [CrossRef]
- Hughes, S.E. Differential expression of the fibroblast growth factor receptor (FGFR) multigene family in normal human adult tissues. J. Histochem. Cytochem. 1997, 45, 1005–1019. [Google Scholar] [CrossRef]
- Lin, X.; Li, G.; He, X.; Ma, X.; Zhang, K.; Zhang, H.; Zeng, G.; Wang, Z. FGF21 inhibits apolipoprotein(a) expression in HepG2 cells via the FGFR1-ERK1/2-Elk-1 pathway. Mol. Cell. Biochem. 2014, 393, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lei, T.; Huang, J.F.; Wang, S.B.; Zhou, L.L.; Yang, Z.Q.; Chen, X.D. The link between fibroblast growth factor 21 and sterol regulatory element binding protein 1c during lipogenesis in hepatocytes. Mol. Cell. Endocrinol. 2011, 342, 41–47. [Google Scholar] [CrossRef]
- Schlumberger, M.; Tahara, M.; Wirth, L.J.; Robinson, B.; Brose, M.S.; Elisei, R.; Habra, M.A.; Newbold, K.; Shah, M.H.; Hoff, A.O.; et al. Lenvatinib versus placebo in radioiodine-refractory thyroid cancer. N. Engl. J. Med. 2015, 372, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.H.; Lee, C.H.; Makker, V.; Rasco, D.; Dutcus, C.E.; Wu, J.; Stepan, D.E.; Shumaker, R.C.; Motzer, R.J. Phase IB/II Trial of lenvatinib plus pembrolizumab in patients with advanced renal cell carcinoma, endometrial cancer, and other selected advanced solid tumors. J. Clin. Oncol. 2020, 38, 1154–1163. [Google Scholar] [CrossRef] [PubMed]
- Oost, L.J.; Kustermann, M.; Armani, A.; Blaauw, B.; Romanello, V. Fibroblast growth factor 21 controls mitophagy and muscle mass. J. Cachexia Sarcopenia Muscle 2019, 10, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Suda, G.; Ohara, M.; Fu, Q.; Yang, Z.; Hosoda, S.; Kimura, M.; Akinori, K.; Tokuchi, Y.; Yamada, R.; et al. Frequency and characteristics of overestimated renal function in Japanese patients with chronic liver disease and its relation to sarcopenia. Nutrients 2021, 13, 2415. [Google Scholar] [CrossRef]
- Ohara, M.; Suda, G.; Kimura, M.; Maehara, O.; Shimazaki, T.; Shigesawa, T.; Suzuki, K.; Nakamura, A.; Kawagishi, N.; Nakai, M.; et al. Analysis of the optimal psoas muscle mass index cut-off values, as measured by computed tomography, for the diagnosis of loss of skeletal muscle mass in Japanese people. Hepatol. Res. 2020, 50, 715–725. [Google Scholar] [CrossRef]
- Yoshida, S.; Suda, G.; Ohara, M.; Kimura, M.; Yang, Z.; Maehara, O.; Fu, Q.; Hosoda, S.; Akinori, K.; Tokuchi, Y.; et al. Overestimated renal function in patients with liver cirrhosis predicts poor prognosis. Hepatol. Res. 2022, 52, 603–613. [Google Scholar] [CrossRef]


| Age (years), median (range) | 70 (45–88) |
| Sex (Male/Female) | 57/6 |
| BMI (kg/m2) | 24 (17–49) |
| ECOG performance status (0/1) | 47/16 |
| History of diabetes mellitus (0/1) | 29/34 |
| Etiology, n (%) | |
| HBV | 20 (31.7%) |
| HCV | 11 (17.4%) |
| NBNC | 32 (50.7%) |
| Child–Pugh class, n (%) | |
| A | 46 (73.0%) |
| B | 17 (27.0%) |
| BCLC stage, n (%) | |
| A | 1 (1.6%) |
| B | 29 (46.0%) |
| C | 33 (52.4%) |
| TNM classification, n (%) | |
| 2 | 8 (12.7%) |
| 3 | 23 (36.5%) |
| 4A | 19 (30.2%) |
| 4B | 13 (20.6%) |
| Biochemical analysis, median (range) | |
| Platelet, ×104/μL | 16.1 (4.4–51.7) |
| Prothrombin time, % | 88.3 (40.7–117.7) |
| Serum albumin, g/dL | 3.7 (2.6–4.6) |
| AST, IU/L | 38.0 (15.0–303.0) |
| ALT, IU/L | 24.0 (8.0–168.0) |
| Total bilirubin, mg/dL | 0.8 (0.3–3.1) |
| Alpha-fetoprotein, ng/mL | 78.2 (1.6–449,909.0) |
| Alpha-fetoprotein L3, ng/mL | 27.2 (0.5–99.5) |
| Appetite loss | |
| Any grade | 43 (68%) |
| Grade 1 | 29 (46%) |
| Grade 2 | 13 (21%) |
| Grade 3 | 1 (2%) |
| Grade 0, 1 (N = 49, 78%) | Grade 2, 3 (N = 14, 22%) | p Value | |
|---|---|---|---|
| Age (years), median (range) | 70 (45–82) | 72 (54–88) | |
| Sex (Male/Female) | 45/4 | 12/2 | 0.252 |
| BMI (kg/m2) | 24 (17–34) | 25 (18–49) | 0.607 |
| ECOG Performance status (0/1) | 37/12 | 10/4 | 0.136 |
| History of Diabetes mellitus (0/1) | 21/28 | 6/8 | 0.739 |
| Etiology, n (%) | >0.999 | ||
| HBV | 16 | 4 | |
| HCV | 10 | 1 | 0.732 |
| NBNC | 23 | 10 | |
| Child–Pugh class, n (%) | |||
| A | 37 | 9 | |
| B | 12 | 5 | 0.498 |
| BCLC stage, n (%) | |||
| A | 0 | 1 | |
| B | 22 | 7 | 0.732 |
| C | 27 | 6 | |
| TNM classification, n (%) | |||
| 2 | 5 | 2 | |
| 3 | 19 | 3 | 0.144 |
| 4A | 14 | 5 | |
| 4B | 11 | 3 | |
| Biochemical analysis, median (range) | |||
| Total bilirubin, mg/dL | 0.8 (0.3–3.1) | 0.8 (0.4–2.6) | |
| Platelets. ×104/μL | 16.0 (4.4–51.7) | 18.5 (6.5–29.5) | 0.733 |
| Prothrombin time, % | 88.4 (40.7–117.7) | 87.1 (56.5–116.8) | 0.763 |
| Serum albumin, g/dL | 3.8 (2.7–4.6) | 3.7 (2.6–4.6) | 0.792 |
| AST, IU/L | 37.0 (15.0–303.0) | 38.50 (18.0–153.0) | 0.275 |
| ALT, IU/L | 26.0 (8.0–168.0) | 23.5 (13.0–48.0) | 0.573 |
| Alpha-fetoprotein, ng/mL | 92.6 (1.6–449909.0) | 12.5 (2.2–257916.0) | 0.165 |
| ALBI grade | 0.451 | ||
| 1 | 17 | 2 | |
| 2 | 29 | 10 | 0.262 |
| 3 | 3 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kohya, R.; Suda, G.; Ohara, M.; Sasaki, T.; Yoda, T.; Sakurai, N.; Yoshida, S.; Fu, Q.; Yang, Z.; Hosoda, S.; et al. Potential Correlation between Changes in Serum FGF21 Levels and Lenvatinib-Induced Appetite Loss in Patients with Unresectable Hepatocellular Carcinoma. Cancers 2023, 15, 3257. https://doi.org/10.3390/cancers15123257
Kohya R, Suda G, Ohara M, Sasaki T, Yoda T, Sakurai N, Yoshida S, Fu Q, Yang Z, Hosoda S, et al. Potential Correlation between Changes in Serum FGF21 Levels and Lenvatinib-Induced Appetite Loss in Patients with Unresectable Hepatocellular Carcinoma. Cancers. 2023; 15(12):3257. https://doi.org/10.3390/cancers15123257
Chicago/Turabian StyleKohya, Risako, Goki Suda, Masatsugu Ohara, Takashi Sasaki, Tomoka Yoda, Naofumi Sakurai, Sonoe Yoshida, Qingjie Fu, Zijian Yang, Shunichi Hosoda, and et al. 2023. "Potential Correlation between Changes in Serum FGF21 Levels and Lenvatinib-Induced Appetite Loss in Patients with Unresectable Hepatocellular Carcinoma" Cancers 15, no. 12: 3257. https://doi.org/10.3390/cancers15123257
APA StyleKohya, R., Suda, G., Ohara, M., Sasaki, T., Yoda, T., Sakurai, N., Yoshida, S., Fu, Q., Yang, Z., Hosoda, S., Maehara, O., Ohnishi, S., Tokuchi, Y., Kitagataya, T., Suzuki, K., Kawagishi, N., Nakai, M., Sho, T., Natsuizaka, M., ... Sakamoto, N. (2023). Potential Correlation between Changes in Serum FGF21 Levels and Lenvatinib-Induced Appetite Loss in Patients with Unresectable Hepatocellular Carcinoma. Cancers, 15(12), 3257. https://doi.org/10.3390/cancers15123257






