The Impact of PSMA-PET on Oncologic Control in Prostate Cancer Patients Who Experienced PSA Persistence or Recurrence
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. PSMA-PET Procedure and Interpretation Criteria
2.3. Patients’ Management and Treatments
2.4. Outcomes Measurements
2.5. Statistical Analyses
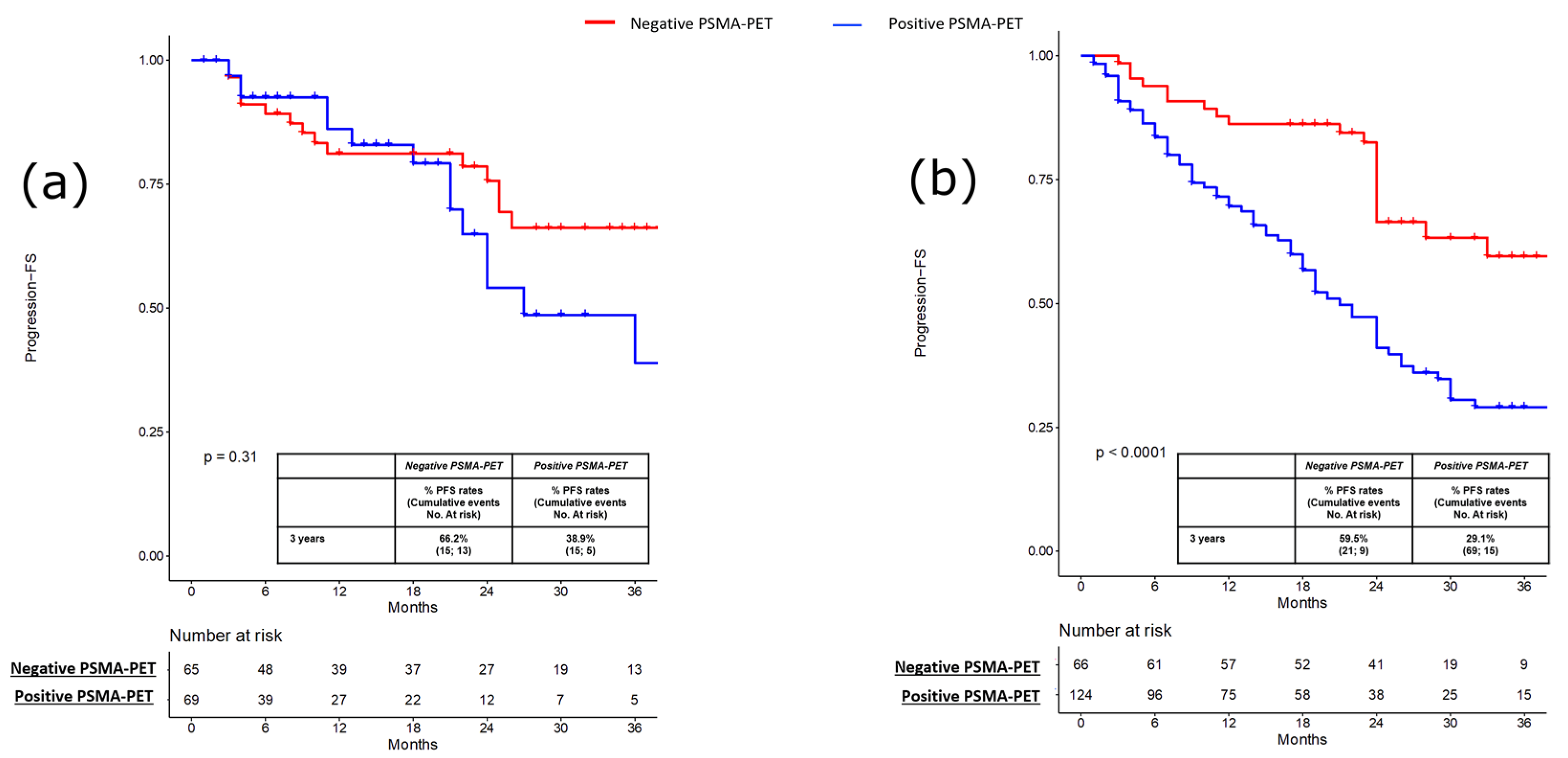
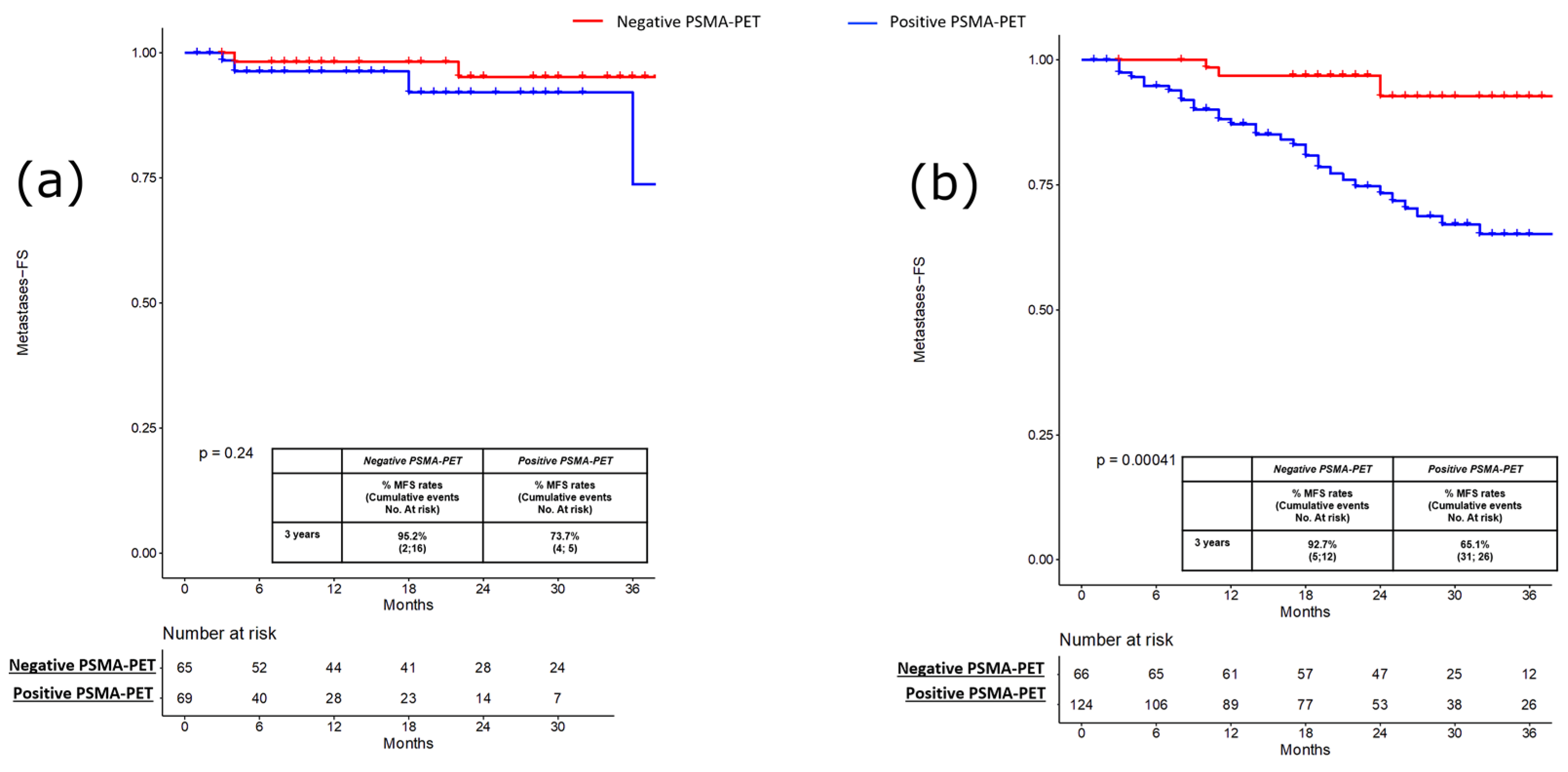
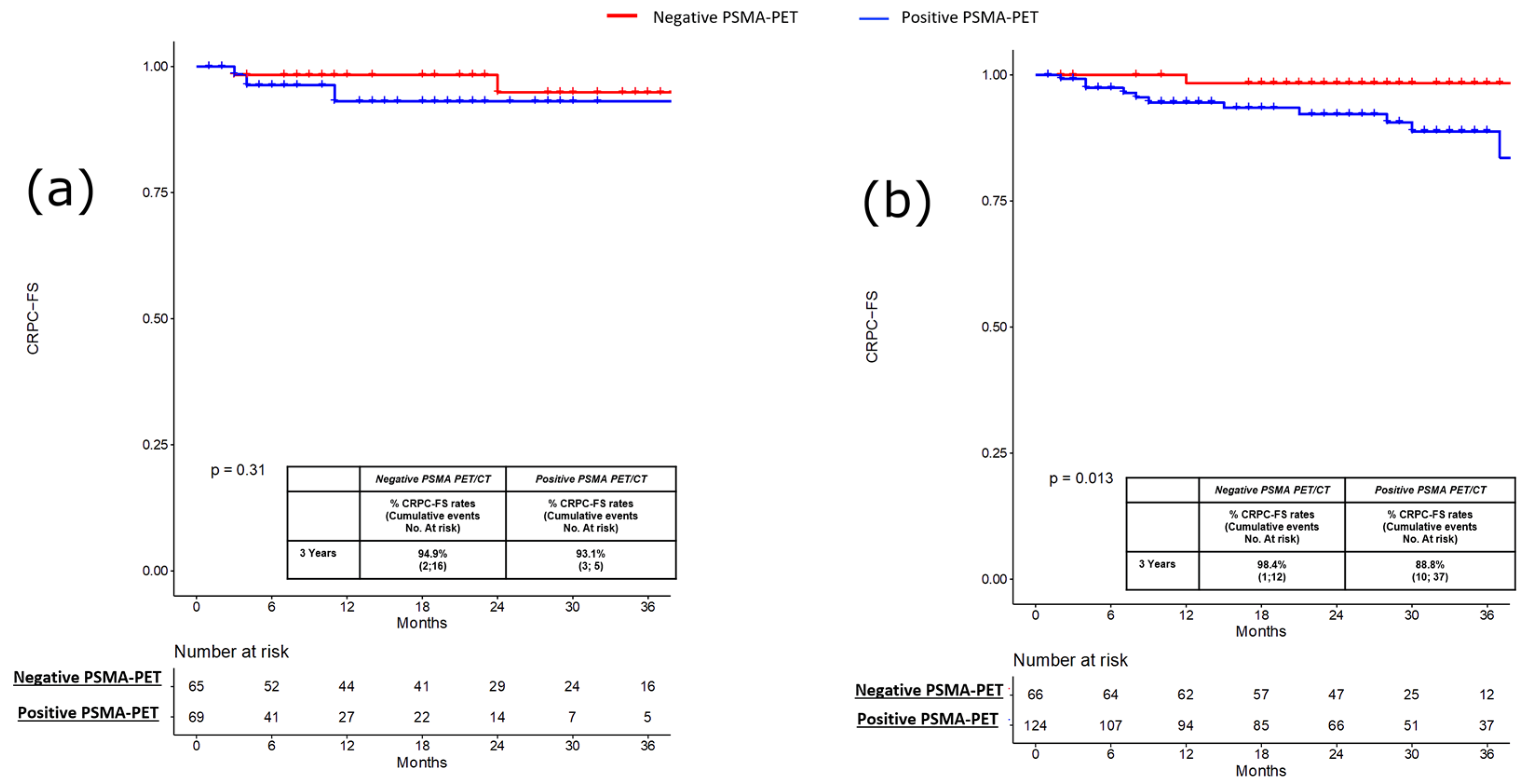
3. Results
3.1. Pre-Salvage Setting (n = 134)
3.2. Pre-Salvage Setting (n = 190)
4. Discussion
5. Limitation
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mottet, N.; van den Bergh, R.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2021, 79, 243–262. [Google Scholar] [CrossRef] [PubMed]
- Ost, P.; Reynders, D.; Decaestecker, K.; Fonteyne, V.; Lumen, N.; De Bruycker, A.; Lambert, B.; Delrue, L.; Bultijnck, R.; Claeys, T.; et al. Surveillance or Metastasis-Directed Therapy for Oligometastatic Prostate Cancer Recurrence: A Prospective, Randomized, Multicenter Phase II Trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 446–453. [Google Scholar] [CrossRef]
- Gundem, G.; Van Loo, P.; Kremeyer, B.; Alexandrov, L.B.; Tubio, J.M.; Papaemmanuil, E.; Brewer, D.S.; Kallio, H.M.; Högnäs, G.; Annala, M. The evolutionary history of lethal metastatic prostate cancer. Nature 2015, 520, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.; Shi, W.Y.; Deek, M.; Radwan, N.; Lim, S.J.; Antonarakis, E.S.; Rowe, S.P.; Ross, A.E.; Gorin, M.A.; Deville, C.; et al. Outcomes of Observation vs. Stereotactic Ablative Radiation for Oligometastatic Prostate Cancer: The ORIOLE Phase 2 Randomized Clinical Trial. JAMA Oncol. 2020, 6, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Calais, J.; Fendler, W.P.; Herrmann, K.; Eiber, M.; Ceci, F. Comparison of (68)Ga-PSMA-11 and (18)F-Fluciclovine PET/CT in a Case Series of 10 Patients with Prostate Cancer Recurrence. J. Nucl. Med. 2018, 59, 789–794. [Google Scholar] [CrossRef]
- Perera, M.; Papa, N.; Christidis, D.; Wetherell, D.; Hofman, M.S.; Murphy, D.G.; Bolton, D.; Lawrentschuk, N. Sensitivity, Specificity, and Predictors of Positive (68)Ga-Prostate-specific Membrane Antigen Positron Emission Tomography in Advanced Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. 2016, 70, 926–937. [Google Scholar] [CrossRef]
- Farolfi, A.; Ceci, F.; Castellucci, P.; Graziani, T.; Siepe, G.; Lambertini, A.; Schiavina, R.; Lodi, F.; Morganti, A.G.; Fanti, S. (68)Ga-PSMA-11 PET/CT in prostate cancer patients with biochemical recurrence after radical prostatectomy and PSA <0.5 ng/ml. Efficacy and impact on treatment strategy. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 11–19. [Google Scholar] [CrossRef]
- Deandreis, D.; Guarneri, A.; Ceci, F.; Lillaz, B.; Bartoncini, S.; Oderda, M.; Nicolotti, D.G.; Pilati, E.; Passera, R.; Zitella, A.; et al. (68)Ga-PSMA-11 PET/CT in recurrent hormone-sensitive prostate cancer (HSPC): A prospective single-centre study in patients eligible for salvage therapy. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2804–2815. [Google Scholar] [CrossRef]
- Bianchi, L.; Schiavina, R.; Borghesi, M.; Ceci, F.; Angiolini, A.; Chessa, F.; Droghetti, M.; Bertaccini, A.; Manferrari, F.; Marcelli, E.; et al. How does 68Ga-prostate-specific membrane antigen positron emission tomography/computed tomography impact the management of patients with prostate cancer recurrence after surgery? Int. J. Urol. 2019, 26, 804–811. [Google Scholar] [CrossRef]
- Calais, J.; Fendler, W.P.; Eiber, M.; Gartmann, J.; Chu, F.-I.; Nickols, N.G.; Reiter, R.E.; Rettig, M.B.; Marks, L.S.; Ahlering, T.E.; et al. Impact of (68)Ga-PSMA-11 PET/CT on the Management of Prostate Cancer Patients with Biochemical Recurrence. J. Nucl. Med. 2018, 59, 434–441. [Google Scholar] [CrossRef]
- Ceci, F.; Bianchi, L.; Borghesi, M.; Polverari, G.; Farolfi, A.; Briganti, A.; Schiavina, R.; Brunocilla, E.; Castellucci, P.; Fanti, S. Prediction nomogram for 68Ga-PSMA-11 PET/CT in different clinical settings of PSA failure after radical treatment for prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, L.; Castellucci, P.; Farolfi, A.; Droghetti, M.; Artigas, C.; Leite, J.; Corona, P.; Shagera, Q.A.; Moreira, R.; González, C.; et al. Multicenter External Validation of a Nomogram for Predicting Positive Prostate-specific Membrane Antigen/Positron Emission Tomography Scan in Patients with Prostate Cancer Recurrence. Eur. Urol. Oncol. 2021, 21, S2588–S9311. [Google Scholar] [CrossRef] [PubMed]
- Rauscher, I.; Düwel, C.; Haller, B.; Rischpler, C.; Heck, M.M.; Gschwend, J.E.; Schwaiger, M.; Maurer, T.; Eiber, M. Efficacy, Predictive Factors, and Prediction Nomograms for (68)Ga-labeled Prostate-specific Membrane Antigen-ligand Positron-emission Tomography/Computed Tomography in Early Biochemical Recurrent Prostate Cancer After Radical Prostatectomy. Eur Urol. 2018, 73, 656–661. [Google Scholar] [CrossRef]
- Calais, J.; Armstrong, W.R.; Kishan, A.U.; Booker, K.M.; Hope, T.A.; Fendler, W.P.; Elashoff, D.; Nickols, N.G.; Czernin, J. Update from PSMA-SRT Trial NCT03582774: A Randomized Phase 3 Imaging Trial of Prostate-specific Membrane Antigen Positron Emission Tomography for Salvage Radiation Therapy for Prostate Cancer Recurrence Powered for Clinical Outcome. Eur. Urol. Focus 2021, 7, 238–240. [Google Scholar] [CrossRef] [PubMed]
- Fanti, S.; Minozzi, S.; Morigi, J.J.; Giesel, F.; Ceci, F.; Uprimny, C.; Hofman, M.; Eiber, M.; Schwarzenbock, S.; Castellucci, P.; et al. Development of standardized image interpretation for 68Ga-PSMA PET/CT to detect prostate cancer recurrent lesions. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1622–1635. [Google Scholar] [CrossRef]
- Fendler, W.P.; Eiber, M.; Beheshti, M.; Bomanji, J.; Ceci, F.; Cho, S.; Giesel, F.; Haberkorn, U.; Hope, T.A.; Kopka, K.; et al. (68)Ga-PSMA PET/CT: Joint EANM and SNMMI procedure guideline for prostate cancer imaging: Version 1.0. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1014–1024. [Google Scholar] [CrossRef] [PubMed]
- Ceci, F.; Oprea-Lager, D.E.; Emmett, L.; Adam, J.A.; Bomanji, J.; Czernin, J.; Eiber, M.; Haberkorn, U.; Hofman, M.S.; Hope, T.A.; et al. E-PSMA: The EANM standardized reporting guidelines v1.0 for PSMA-PET. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1626–1638. [Google Scholar] [CrossRef]
- Radwan, N.; Phillips, R.; Ross, A.; Rowe, S.P.; Gorin, M.A.; Antonarakis, E.S.; Deville, C.; Greco, S.; Denmeade, S.; Paller, C.; et al. A phase II randomized trial of Observation versus stereotactic ablative RadiatIon for OLigometastatic prostate CancEr (ORIOLE). BMC Cancer 2017, 17, 453. [Google Scholar] [CrossRef]
- Ceci, F.; Rovera, G.; Iorio, G.C.; Guarneri, A.; Chiofalo, V.; Passera, R.; Oderda, M.; Dall’Armellina, S.; Liberini, V.; Grimaldi, S.; et al. Event-free survival after (68) Ga-PSMA-11 PET/CT in recurrent hormone-sensitive prostate cancer (HSPC) patients eligible for salvage therapy. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 3257–3268. [Google Scholar] [CrossRef]
- Meijer, D.; Eppinga, W.S.; Mohede, R.M.; Vanneste, B.G.; Meijnen, P.; Meijer, O.W.; Daniels, L.A.; Bergh, R.C.V.D.; Lont, A.P.; Ettema, R.H.; et al. Prostate-specific Membrane Antigen Positron Emission Tomography/Computed Tomography Is Associated with Improved Oncological Outcome in Men Treated with Salvage Radiation Therapy for Biochemically Recurrent Prostate Cancer. Eur. Urol. Oncol. 2022, 5, 146–152. [Google Scholar] [CrossRef]
- Zaorsky, N.G.; Calais, J.; Fanti, S.; Tilki, D.; Dorff, T.; Spratt, D.E.; Kishan, A.U. Salvage therapy for prostate cancer after radical prostatectomy. Nat. Rev. Urol. 2021, 18, 643–668. [Google Scholar] [CrossRef] [PubMed]
- Hölscher, T.; Baumann, M.; Kotzerke, J.; Zöphel, K.; Paulsen, F.; Müller, A.-C.; Zips, D.; Koi, L.; Thomas, C.; Löck, S.; et al. Toxicity and Efficacy of Local Ablative, Image-guided Radiotherapy in Gallium-68 Prostate-specific Membrane Antigen Targeted Positron Emission Tomography-staged, Castration-sensitive Oligometastatic Prostate Cancer: The OLI-P Phase 2 Clinical Trial. Eur. Urol. Oncol. 2022, 5, 44–51. [Google Scholar] [CrossRef] [PubMed]
- De Bruycker, A.; Spiessens, A.; Dirix, P.; Koutsouvelis, N.; Semac, I.; Liefhooghe, N.; Gomez-Iturriaga, A.; Everaerts, W.; Otte, F.; Papachristofilou, A.; et al. PEACE V—Salvage Treatment of OligoRecurrent nodal prostate cancer Metastases (STORM): A study protocol for a randomized controlled phase II trial. BMC Cancer 2020, 20, 406. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M.J.; Chatfield, M.D.; Hruby, G.; Nandurkar, R.; Roach, P.; Watts, J.A.; Cusick, T.; Kneebone, A.; Eade, T.; Ho, B.; et al. Event-free survival after radical prostatectomy according to prostate-specific membrane antigen-positron emission tomography and European Association of Urology biochemical recurrence risk groups. BJU Int. 2022, 130, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Fendler, W.P.; Calais, J.; Eiber, M.; Flavell, R.R.; Mishoe, A.; Feng, F.Y.; Nguyen, H.G.; Reiter, R.E.; Rettig, M.B.; Okamoto, S.; et al. Assessment of 68Ga-PSMA-11 PET Accuracy in Localizing Recurrent Prostate Cancer: A Prospective Single-Arm Clinical Trial. JAMA Oncol. 2019, 5, 856–863. [Google Scholar] [CrossRef] [PubMed]
| Overall | Pre-Salvage Setting, n = 134 (41.4%) | Post-Salvage Setting, n = 190 (58.6%) | |||||
|---|---|---|---|---|---|---|---|
| Positive PSMA-PET | Negative PSMA-PET | p Value | Positive PSMA-PET | Negative PSMA-PET | p Value | ||
| Patients, n (%) | 324 (100) | 69 (52) | 65 (48) | - | 124 (65) | 66 (35) | - |
| Age Median (IQR) | 63 (57–68) | 64 (59–70) | 63 (56–68) | 0.4 | 62 (56–66) | 65 (59–70) | 0.02 |
| PSA level at RP (ng/mL) Median (IQR) | 8.34 (5.51–12.88) | 7.45 (6–13.22) | 8.54 (4.33–12) | 0.4 | 7.34 (2.33–13.85) | 7.07 (4.85–9.97) | 0.8 |
| pT stage, n (%) pT2 pT3a pT3b-pT4 | 102 (31.5) 108 (33.3) 114 (35.2) | 12 (17.4) 23 (33.3) 34 (49.3) | 31 (47.7) 17 (26.2) 17 (26.2) | 0.01 | 40 (32.3) 41 (33.1) 43 (34.7) | 19 (28.8) 27 (40.9) 20 (30.3) | 0.6 |
| pN stage, n (%) pNx pN0 pN1 | 69 (21.3) 178 (54.9) 77 (23.8) | 9 (13) 34 (49.3) 26 (37.7) | 15 (23.1) 38 (58.5) 12 (18.5) | 0.03 | 30 (24.2) 64 (51.6) 30 (24.2) | 15 (22.7) 42 (63.6) 9 (13.6) | 0.2 |
| Pathologic ISUP group, n (%) ISUP 1–3 ISUP 4–5 | 157 (48) 167 (52) | 24 (34.8) 45 (65.2) | 45 (69.2) 20 (30.8) | ≤0.001 | 58 (46.8) 66 (53.2) | 30 (45.5) 36 (54.5) | 0.9 |
| Adjuvant Radiotherapy, n (%) Yes No | 88 (27.2) 236 (72.8) | 50 (72.5) 19 (7.5) | 51 (78.5) 14 (21.5) | 0.4 | 38 (30.6) 86 (69.4) | 17 (25.8) 49 (4.2) | 0.5 |
| PSA level at PET PSMA, ng/mL Median (IQR) | 0.5 (0.28–1.2) | 0.8 (0.31–1.99) | 0.33 (0.25–0.56) | ≤0.001 | 0.66 (0.31–1.45) | 0.48 (0.28–1) | 0.06 |
| Overall | Pre-Salvage Setting, n = 134 (41.4%) | Post-Salvage Setting, n = 190 (58.6%) | |||||
|---|---|---|---|---|---|---|---|
| Positive PSMA-PET | Negative PSMA-PET | p Value | Positive PSMA-PET | Negative PSMA-PET | p Value | ||
| Patients, n (%) | 324 (100) | 69 (51) | 65 (49) | - | 124 | 66 | - |
| Treatment performed after PSMA-PET, n (%) Prostate bed RT/whole pelvis RT sLND Lymph node SRBT Bone SRBT sLND + SBRT Pelvic RT + bone SBRT Cyberknife Cryotherapy Metastases resection ADT ADT + ARTA/Chemo Observation | 88 (27.2) 45 (13.9) 9 (2.8) 18 (5.6) 6 (1.9) 19 (5.9) 2 (0.6) 4 (1.2) 1 (0.3) 69 (21.3) 4 (1.2) 59 (18.2) | 18 (26.1) 7 (10.1) 1 (1.4) 4 (5.8) 3 (4.3) 17 (24.6) 0 (0) 0 (0) 0 (0) 8 (11.6) 1 (1.4) 10 (14.5) | 26 (40) 1 (1.5) 0 (0) 0 (0) 0 (0) 0 (0) 0 (0) 0 (0) 0 (0) 14 (21.5) 0 (0) 24 (36.9) | ≤0.001 | 26 (21) 37 (29.8) 8 (6.5) 14 (11.3) 3 (2.4) 2 (1.6) 2 (1.6) 4 (3.2) 1 (0.8) 15 (12.1) 3 (2.4) 9 (7.3) | 18 (27.3) 0 (0) 0 (0) 0 (0) 0 (0) 0 (0) 0 (0) 0 (0) 0 (0) 32 (48.5) 0 (0) 16 (24.2) | ≤0.001 |
| PSA at recurrence after PSMA-PET, ng/mL Median (IQR) | 0.85 (0.43–1.95) | 0.85 (0.23–1.85 | 0.58 (0.17–2.9) | 0.9 | 0.98 (0.5–2.54) | 1.17 (0.48–1.88) | 0.5 |
| Time to PSA recurrence, months Median (IQR) | 9 (3–23) | 11.5 (5–24) | 6 (3–25) | 0.9 | 8 (3–17) | 14 (4–24) | 0.3 |
| Metastatic recurrence, n (%) Yes No | 47 (14.5) 277 (85.5) | 4 (5.8) 65 (94.2) | 3 (4.6) 62 (95.4) | 0.8 | 35 (28.2) 89 (71.8) | 5 (7.6) 61 (92.4) | 0.001 |
| Time to metastatic recurrence, months Median (IQR) | 21 (8–32) | 11 (3–32) | 22 (8–35) | ≤0.001 | 17 (8–25) | 24 (11–34) | 0.003 |
| CRPC, n (%) Yes No | 26 (8) 298 (92) | 4 (5.8) 65 (94.2) | 3 (4.6) 62 (95.4) | 0.8 | 18 (14.5) 106 (85.5) | 1 (1.5) 65 (85.5) | 0.004 |
| Time to CRPC, months Median (IQR) | 23 (9–33) | 7.5 (3–37) | 22 (8–36) | 0.001 | 29 (8–40) | 27 (26–33) | 0.3 |
| Overall Mortality, n (%) Yes No | 9 (2.8) 315 (97.2) | 0 69 (100) | 1 (1.5) 64 (98.5) | 0.3 | 7 (95.6) 117 (94.4) | 1 (1.5) 65 (85.5) | 0.2 |
| Cancer specific mortality, n (%) Yes No | 3 (0.9) 321 (99.1) | 0 69 (100) | 0 65 (100) | - | 3 (2.4) 121 (97.6) | 0 66 (100) | 0.2 |
| Follow up (months) from PSMA-PET Median (IQR) | 23 (10–34) | 8 (3–21) | 23 (9–37) | ≤0.001 | 27 (15–40) | 27 (23–33) | 0.6 |
| Follow up (months) from RP Median (IQR) | 62 (30–108) | 35 (16–74) | 63 (45–100) | ≤0.001 | 68 (0–118) | 74 (50–141) | 0.02 |
| Variables | Progression | Metastasis | ||
|---|---|---|---|---|
| HR (95% C.I.) | p Value | HR (95% C.I.) | p Value | |
| Age (years) | 0.97 (0.95–0.99) | 0.04 | - | - |
| Clinical setting for PSMA-PET Pre-salvage setting Post-salvage setting | 1.0 (Ref) 1.31 (0.86–1.98) | 0.2 | 1.0 (Ref) 1.40 (0.93–2.10) | 0.1 |
| Pathologic stage pT2 pT3a pT3b-pT4 | 1.0 (Ref) 1.47 (0.90–2.39) 1.84 (1.10–3.09) | 0.1 0.02 | 1.0 (Ref) 1.49 (0.93–2.41) 2.03 (1.26–3.26) | 0.09 0.003 |
| Pathologic ISUP group ISUP 1– 3ISUP 4–5 | 1.0 (Ref) 0.92 (0.60–1.46) | 0.7 | 1.0 (Ref) 0.87 (0.93–1.28) | 0.5 |
| pN stage, n (%) pNx/pN0 pN1 | (Ref) 1.04 (0.66–1.65) | 0.8 | - | - |
| ADT at salvage treatment | 0.58 (0.40–0.85) | 0.005 | 0.54 (0.37–0.78) | 0.001 |
| PSA at PSMA-PET (ng/mL) | 0.99 (0.97–1-02) | 0.4 | - | - |
| PSMA-PET result Negative Positive | 1.0 (Ref) 2.15 (1.42–3.25) | <0.001 | 1.0 (Ref) 2.37 (1.60–3.50) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bianchi, L.; Ceci, F.; Costa, F.; Balestrazzi, E.; Droghetti, M.; Piazza, P.; Pissavini, A.; Mei, R.; Farolfi, A.; Castellucci, P.; et al. The Impact of PSMA-PET on Oncologic Control in Prostate Cancer Patients Who Experienced PSA Persistence or Recurrence. Cancers 2023, 15, 247. https://doi.org/10.3390/cancers15010247
Bianchi L, Ceci F, Costa F, Balestrazzi E, Droghetti M, Piazza P, Pissavini A, Mei R, Farolfi A, Castellucci P, et al. The Impact of PSMA-PET on Oncologic Control in Prostate Cancer Patients Who Experienced PSA Persistence or Recurrence. Cancers. 2023; 15(1):247. https://doi.org/10.3390/cancers15010247
Chicago/Turabian StyleBianchi, Lorenzo, Francesco Ceci, Francesco Costa, Eleonora Balestrazzi, Matteo Droghetti, Pietro Piazza, Alessandro Pissavini, Riccardo Mei, Andrea Farolfi, Paolo Castellucci, and et al. 2023. "The Impact of PSMA-PET on Oncologic Control in Prostate Cancer Patients Who Experienced PSA Persistence or Recurrence" Cancers 15, no. 1: 247. https://doi.org/10.3390/cancers15010247
APA StyleBianchi, L., Ceci, F., Costa, F., Balestrazzi, E., Droghetti, M., Piazza, P., Pissavini, A., Mei, R., Farolfi, A., Castellucci, P., Puliatti, S., Larcher, A., Gandaglia, G., Robesti, D., Mottrie, A., Briganti, A., Morganti, A. G., Fanti, S., Montorsi, F., ... Brunocilla, E. (2023). The Impact of PSMA-PET on Oncologic Control in Prostate Cancer Patients Who Experienced PSA Persistence or Recurrence. Cancers, 15(1), 247. https://doi.org/10.3390/cancers15010247









