Immunogenicity of COVID-19 Vaccinations in Hematological Patients: 6-Month Follow-Up and Evaluation of a 3rd Vaccination
Abstract
:Simple Summary
Abstract
1. Introduction
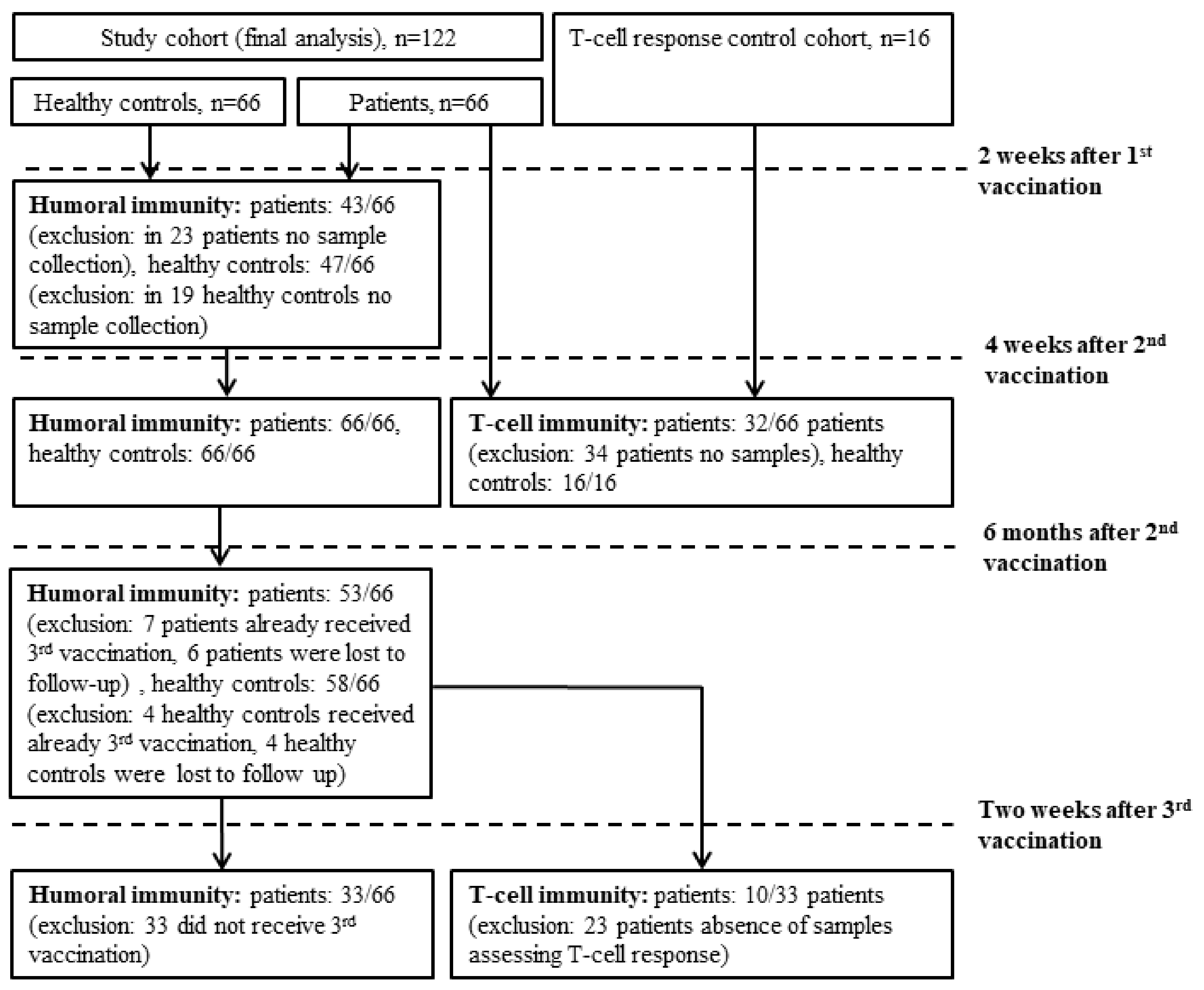
2. Materials and Methods
2.1. Assessment of Humoral Immunity
2.2. Assessment of Cellular Immunity
2.3. Statistical Analysis
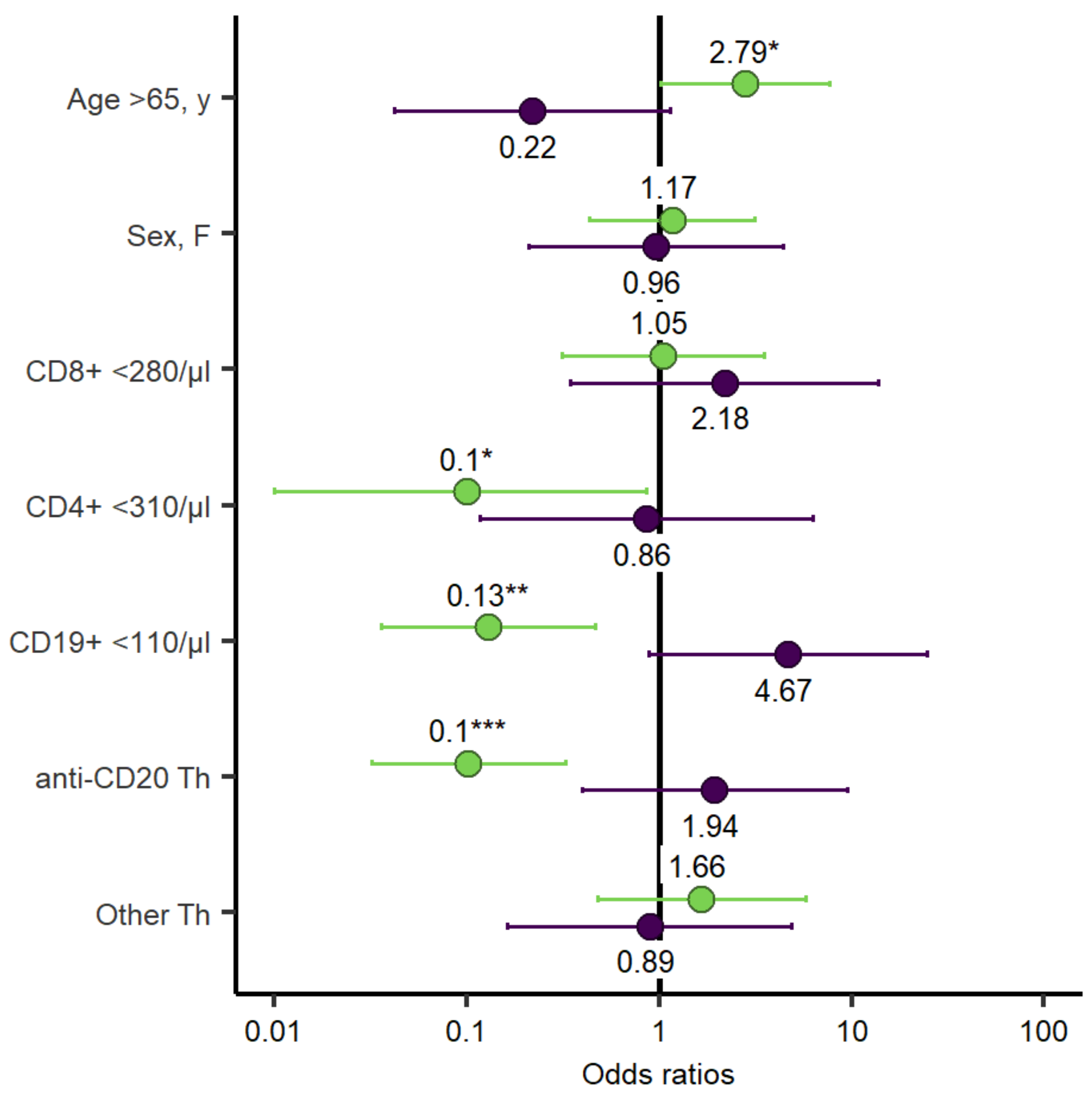
3. Results Multivariate Analysis
3.1. Study Participants Characteristics
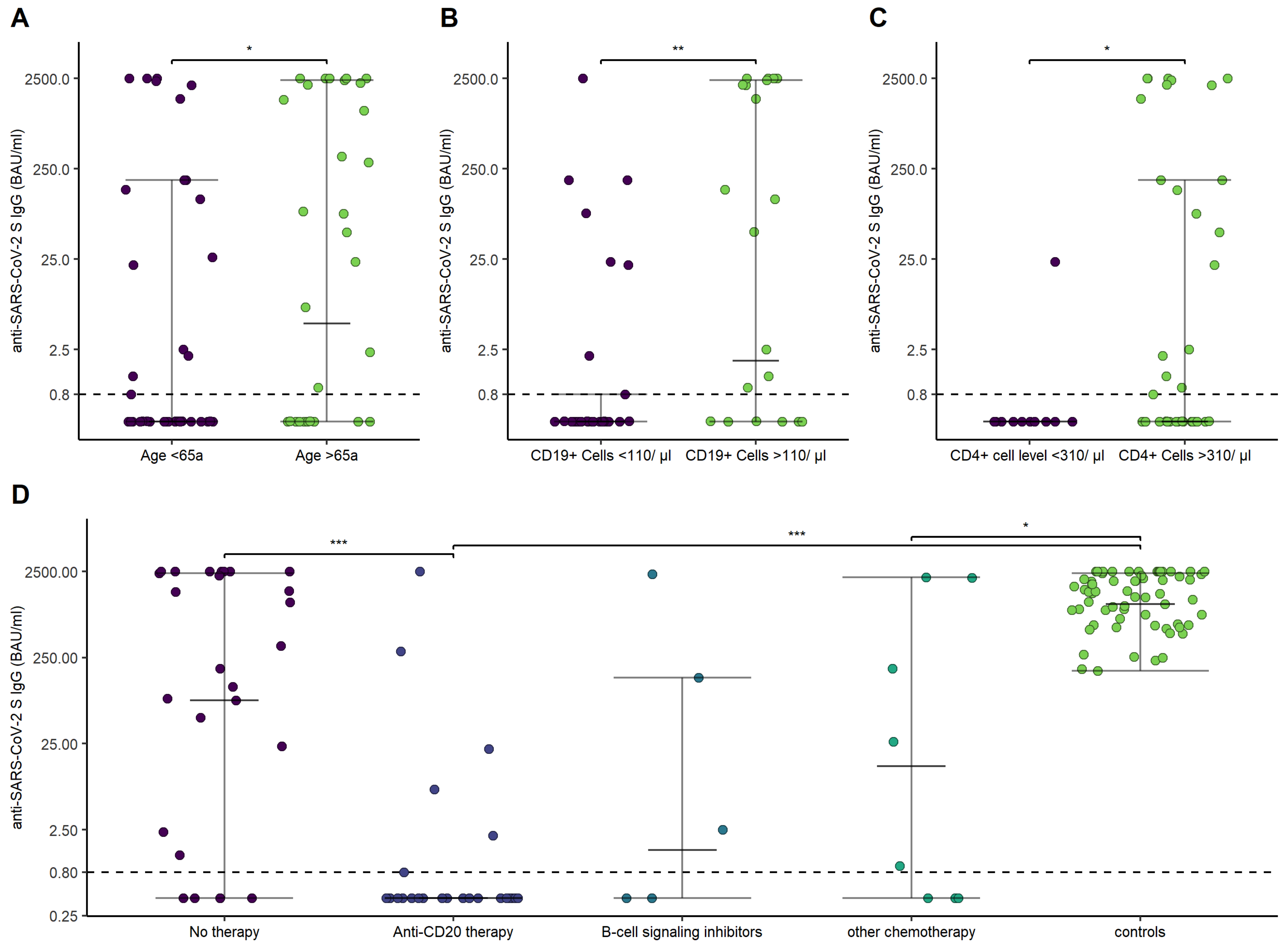
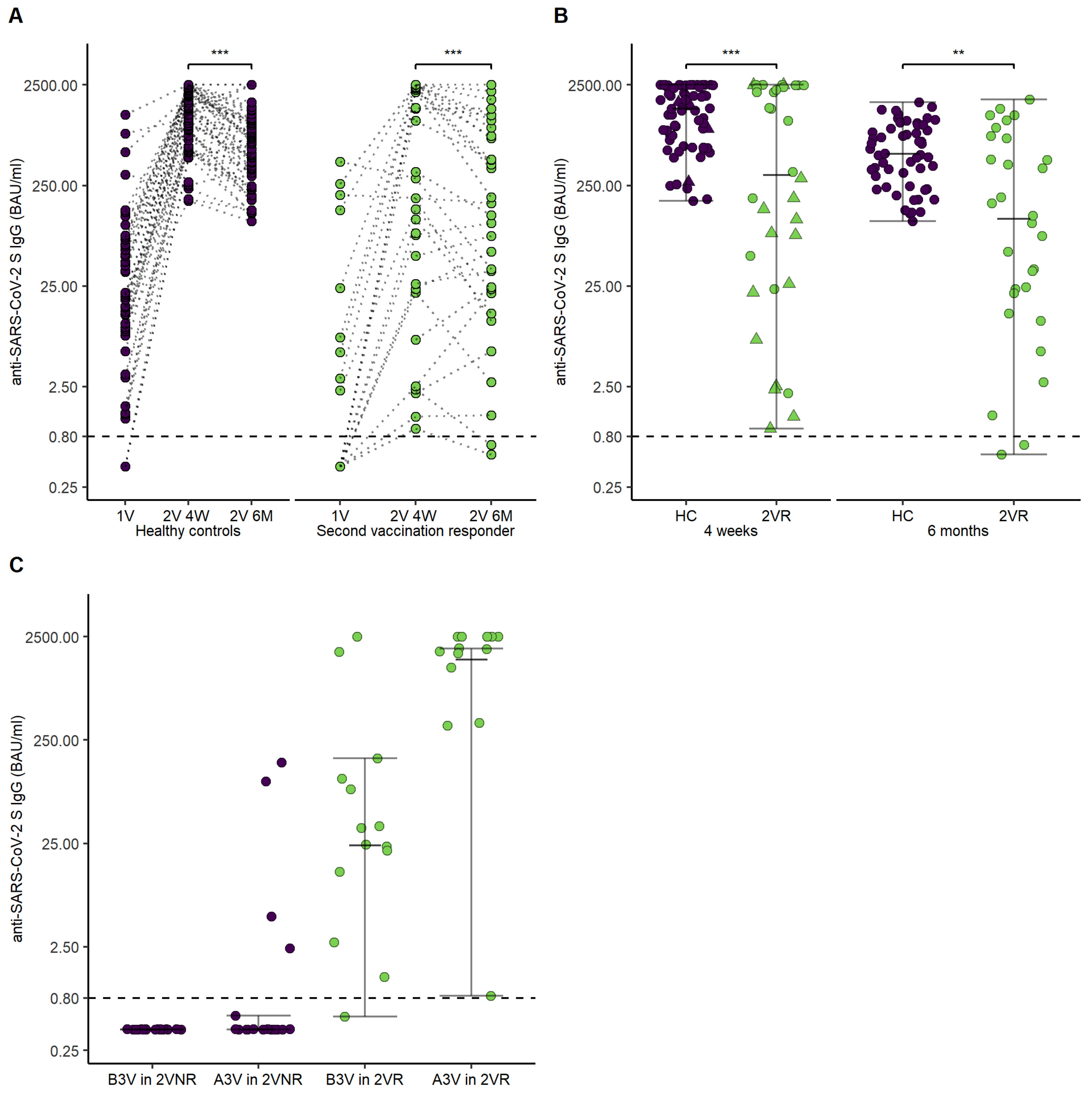
3.2. Humoral Response to mRNA SARS-CoV-2 Vaccination
3.3. Cellular Response to mRNA SARS-CoV-2 Vaccination
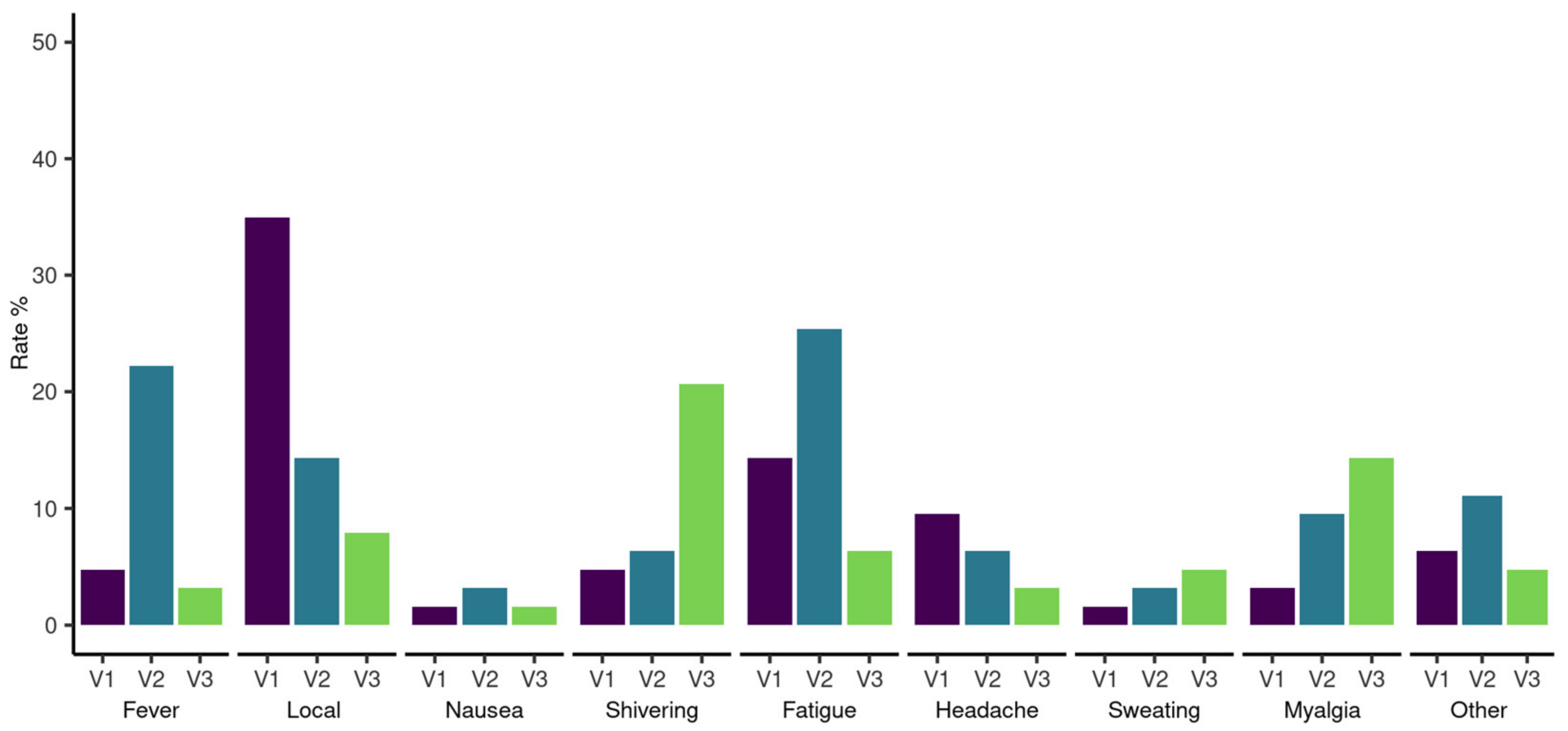
3.4. Adverse Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eastin, C.; Eastin, T. Clinical Characteristics of Coronavirus Disease 2019 in China. J. Emerg. Med. 2020, 58, 711–712. [Google Scholar] [CrossRef]
- Vijenthira, A.; Gong, I.Y.; Fox, T.A.; Booth, S.; Cook, G.; Fattizzo, B.; Martín-Moro, F.; Razanamahery, J.; Riches, J.C.; Zwicker, J.; et al. Outcomes of patients with hematologic malignancies and COVID-19: A systematic review and meta-analysis of 3377 patients. Blood 2020, 136, 2881–2892. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Sun, C.; Pleyer, C.; Wiestner, A. COVID-19 vaccines for patients with haematological conditions. Lancet Haematol. 2021, 8, e312–e314. [Google Scholar] [CrossRef]
- Herishanu, Y.; Avivi, I.; Aharon, A.; Shefer, G.; Levi, S.; Bronstein, Y.; Morales, M.; Ziv, T.; Shorer Arbel, Y.; Scarfò, L.; et al. Efficacy of the BNT162b2 mRNA COVID-19 vaccine in patients with chronic lymphocytic leukemia. Blood 2021, 137, 3165–3173. [Google Scholar] [CrossRef]
- Agha, M.; Blake, M.; Chilleo, C.; Wells, A.; Haidar, G. Suboptimal Response to Coronavirus Disease 2019 Messenger RNA Vaccines in Patients With Hematologic Malignancies: A Need for Vigilance in the Postmasking Era. Open Forum Infect. Dis. 2021, 8, ofab353. [Google Scholar] [CrossRef]
- Terpos, E.; Trougakos, I.P.; Gavriatopoulou, M.; Papassotiriou, I.; Sklirou, A.D.; Ntanasis-Stathopoulos, I.; Papanagnou, E.D.; Fotiou, D.; Kastritis, E.; Dimopoulos, M.A. Low neutralizing antibody responses against SARS-CoV-2 in older patients with myeloma after the first BNT162b2 vaccine dose. Blood 2021, 137, 3674–3676. [Google Scholar] [CrossRef]
- Blixt, L.; Bogdanovic, G.; Buggert, M.; Gao, Y.; Hober, S.; Healy, K.; Johansson, H.; Kjellander, C.; Mravinacova, S.; Muschiol, S.; et al. COVID-19 in patients with chronic lymphocytic leukemia: Clinical outcome and B- and T-cell immunity during 13 months in consecutive patients. Leukemia 2021, 36, 476–481. [Google Scholar] [CrossRef]
- Bange, E.M.; Han, N.A.; Wileyto, P.; Kim, J.Y.; Gouma, S.; Robinson, J.; Greenplate, A.R.; Hwee, M.A.; Porterfield, F.; Owoyemi, O.; et al. CD8+ T cells contribute to survival in patients with COVID-19 and hematologic cancer. Nat. Med. 2021, 27, 1280–1289. [Google Scholar] [CrossRef]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; et al. Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell 2020, 181, 1489–1501.e15. [Google Scholar] [CrossRef]
- Malard, F.; Gaugler, B.; Gozlan, J.; Bouquet, L.; Fofana, D.; Siblany, L.; Eshagh, D.; Adotevi, O.; Laheurte, C.; Ricard, L.; et al. Weak immunogenicity of SARS-CoV-2 vaccine in patients with hematologic malignancies. Blood Cancer J. 2021, 11, 142. [Google Scholar] [CrossRef]
- Dispinseri, S.; Secchi, M.; Pirillo, M.F.; Tolazzi, M.; Borghi, M.; Brigatti, C.; De Angelis, M.L.; Baratella, M.; Bazzigaluppi, E.; Venturi, G.; et al. Neutralizing antibody responses to SARS-CoV-2 in symptomatic COVID-19 is persistent and critical for survival. Nat. Commun. 2021, 12, 2670. [Google Scholar] [CrossRef]
- Goldberg, Y.; Mandel, M.; Bar-On, Y.M.; Bodenheimer, O.; Freedman, L.; Haas, E.J.; Milo, R.; Alroy-Preis, S.; Ash, N.; Huppert, A. Waning Immunity after the BNT162b2 Vaccine in Israel. N. Engl. J. Med. 2021, 385, e85. [Google Scholar] [CrossRef]
- Garcia-Beltran, W.F.; Lam, E.C.; St. Denis, K.; Nitido, A.D.; Garcia, Z.H.; Hauser, B.M.; Feldman, J.; Pavlovic, M.N.; Gregory, D.J.; Poznansky, M.C.; et al. Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell 2021, 184, 2372. [Google Scholar] [CrossRef]
- Mair, M.J.; Berger, J.M.; Mitterer, M.; Gansterer, M.; Bathke, A.C.; Trutschnig, W.; Berghoff, A.S.; Perkmann, T.; Haslacher, H.; Lamm, W.W.; et al. Third dose of SARS-CoV-2 vaccination in hemato-oncological patients and health care workers: Immune responses and adverse events—A retrospective cohort study. Eur. J. Cancer 2022, 165, 184. [Google Scholar] [CrossRef]
- Herishanu, Y.; Rahav, G.; Levi, S.; Braester, A.; Itchaki, G.; Bairey, O.; Dally, N.; Shvidel, L.; Ziv-Baran, T.; Polliack, A.; et al. Efficacy of a third BNT162b2 mRNA COVID-19 vaccine dose in patients with CLL who failed standard 2-dose vaccination. Blood 2022, 139, 678. [Google Scholar] [CrossRef]
- Higgins, V.; Fabros, A.; Kulasingam, V. Quantitative measurement of Anti-SARS-CoV-2 Antibodies: Analytical and clinical evaluation. J. Clin. Microbiol. 2021, 59, e03149-20. [Google Scholar] [CrossRef]
- Simon, D.; Tascilar, K.; Fagni, F.; Krönke, G.; Kleyer, A.; Meder, C.; Atreya, R.; Leppkes, M.; Kremer, A.E.; Ramming, A.; et al. SARS-CoV-2 vaccination responses in untreated, conventionally treated and anticytokine-treated patients with immune-mediated inflammatory diseases. Ann. Rheum. Dis. 2021, 80, 1312–1316. [Google Scholar] [CrossRef]
- Lumley, S.F.; O’Donnell, D.; Stoesser, N.E.; Matthews, P.C.; Howarth, A.; Hatch, S.B.; Marsden, B.D.; Cox, S.; James, T.; Warren, F.; et al. Antibody Status and Incidence of SARS-CoV-2 Infection in Health Care Workers. N. Engl. J. Med. 2021, 384, 533–540. [Google Scholar] [CrossRef]
- Roeker, L.E.; Knorr, D.A.; Thompson, M.C.; Nivar, M.; Lebowitz, S.; Peters, N.; Deonarine, I.; Momotaj, S.; Sharan, S.; Chanlatte, V.; et al. COVID-19 vaccine efficacy in patients with chronic lymphocytic leukemia. Leukemia 2021, 35, 2703–2705. [Google Scholar] [CrossRef] [PubMed]
- Deepak, P.; Kim, W.; Paley, M.A.; Yang, M.; Carvidi, A.B.; Demissie, E.G.; El-Qunni, A.A.; Haile, A.; Huang, K.; Kinnett, B.; et al. Effect of immunosuppression on the immunogenicity of mrna vaccines to SARS-CoV-2 a prospective cohort study. Ann. Intern. Med. 2021, 174, 1572–1585. [Google Scholar] [CrossRef] [PubMed]
- Re, D.; Barrière, J.; Chamorey, E.; Delforge, M.; Gastaud, L.; Petit, E.; Chaminade, A.; Verrière, B.; Peyrade, F. Low rate of seroconversion after mRNA anti-SARS-CoV-2 vaccination in patients with hematological malignancies. Leuk. Lymphoma 2021, 62, 3308–3310. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, F.; Pimpinelli, F.; Giannarelli, D.; Ronchetti, L.; Papa, E.; Falcucci, P.; Pontone, M.; Di Domenico, E.G.; di Martino, S.; Laquintana, V.; et al. Impact of anti-CD20 monoclonal antibodies on serologic response to BNT162b2 vaccine in B-cell Non-Hodgkin’s lymphomas. Leukemia 2021, 36, 588–590. [Google Scholar] [CrossRef]
- Painter, M.M.; Mathew, D.; Goel, R.R.; Apostolidis, S.A.; Pattekar, A.; Kuthuru, O.; Baxter, A.E.; Herati, R.S.; Oldridge, D.A.; Gouma, S.; et al. Rapid induction of antigen-specific CD4+ T cells is associated with coordinated humoral and cellular immunity to SARS-CoV-2 mRNA vaccination. Immunity 2021, 54, 2133–2142.e3. [Google Scholar] [CrossRef]
- Tober-Lau, P.; Schwarz, T.; Vanshylla, K.; Hillus, D.; Gruell, H.; Suttorp, N.; Landgraf, I.; Kappert, K.; Seybold, J.; Drosten, C.; et al. Long-term immunogenicity of BNT162b2 vaccination in older people and younger health-care workers. Lancet Respir. Med. 2021, 9, e104–e105. [Google Scholar] [CrossRef]
- Kalimuddin, S.; Tham, C.Y.; Qui, M.; de Alwis, R.; Sim, J.X.; Lim, J.M.; Tan, H.C.; Syenina, A.; Zhang, S.L.; Le Bert, N.; et al. Early T cell and binding antibody responses are associated with COVID-19 RNA vaccine efficacy onset. Med 2021, 2, 682–688.e4. [Google Scholar] [CrossRef]
- Brudno, J.N.; Kochenderfer, J.N. Chimeric antigen receptor T-cell therapies for lymphoma. Nat. Rev. Clin. Oncol. 2017, 15, 31–46. [Google Scholar] [CrossRef]
- Hall, V.G.; Ferreira, V.H.; Ku, T.; Ierullo, M.; Majchrzak-Kita, B.; Chaparro, C.; Selzner, N.; Schiff, J.; McDonald, M.; Tomlinson, G.; et al. Randomized Trial of a Third Dose of mRNA-1273 Vaccine in Transplant Recipients. N. Engl. J. Med. 2021, 385, 1244–1246. [Google Scholar] [CrossRef]
- Kamar, N.; Abravanel, F.; Marion, O.; Couat, C.; Izopet, J.; Bello, A. Del Three Doses of an mRNA Covid-19 Vaccine in Solid-Organ Transplant Recipients. N. Engl. J. Med. 2021, 385, 661–662. [Google Scholar] [CrossRef]
- Re, D.; Seitz-Polski, B.; Brglez, V.; Carles, M.; Graça, D.; Benzaken, S.; Liguori, S.; Zahreddine, K.; Delforge, M.; Bailly-Maitre, B.; et al. Humoral and cellular responses after a third dose of SARS-CoV-2 BNT162b2 vaccine in patients with lymphoid malignancies. Nat. Commun. 2022, 13, 864. [Google Scholar] [CrossRef]
- Fendler, A.; Shepherd, S.T.C.; Au, L.; Wilkinson, K.A.; Wu, M.; Schmitt, A.M.; Tippu, Z.; Farag, S.; Rogiers, A.; Harvey, R.; et al. Immune responses following third COVID-19 vaccination are reduced in patients with hematological malignancies compared to patients with solid cancer. Cancer Cell 2022, 40, 114. [Google Scholar] [CrossRef]

| Baseline Demographics | Patients (n = 66) | |
|---|---|---|
| Age, median (Q1–Q3), years | 62 (50–69) | |
| Females, n (%) | 26 (39.4) | |
| Male, n (%) | 40 (60.6) | |
| Hematological malignancies, n (%) | ||
| Lymphoid malignancies | ||
| Chronic lymphocytic leukemia | 16 (24.2) | |
| Indolent NHL | 20 (30.3) | |
| Aggressive NHL | 16 (24.2) | |
| Multiple myeloma | 6 (9.1) | |
| B-lymphoblastic leukemia | 1 (1.5) | |
| Hodgkin lymphoma | 1 (1.5) | |
| Langerhans cell histiocytosis | 1 (1.5) | |
| Myeloid malignancies | ||
| Acute myeloid leukemia | 2 (30.3) | |
| Myeloproliferative neoplasms | 3 (45.5) | |
| Disease/treatment status, n (%) | ||
| On-therapy response | 35 (53) | |
| On-therapy partial response | 7 (10.6) | |
| In remission stable | 10 (15.2) | |
| Awaiting therapy | 14 (21.2) | |
| Treatment, n (%) | ||
| Anti-CD-20 < 1 year | 27 (40.9) | |
| B-cell signalling inhibitors | 5 (7.6) | |
| Other chemotherapy | 8 (12.29) | |
| No treatment | 26 (39.4) | |
| Bone marrow transplantation | ||
| Autologous stem cell transplantation | 5 (7.6) | |
| Allogenic stem cell transplantation | 4 (6.1) | |
| Anti-CD19 CAR T-cells | 6 (9.1) | |
| Vaccine, n (%) | ||
| mRNA-1273 | 35 (53) | |
| BNT162b2 | 31 (47) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schubert, L.; Koblischke, M.; Schneider, L.; Porpaczy, E.; Winkler, F.; Jaeger, U.; Blüml, S.; Haslacher, H.; Burgmann, H.; Aberle, J.H.; et al. Immunogenicity of COVID-19 Vaccinations in Hematological Patients: 6-Month Follow-Up and Evaluation of a 3rd Vaccination. Cancers 2022, 14, 1962. https://doi.org/10.3390/cancers14081962
Schubert L, Koblischke M, Schneider L, Porpaczy E, Winkler F, Jaeger U, Blüml S, Haslacher H, Burgmann H, Aberle JH, et al. Immunogenicity of COVID-19 Vaccinations in Hematological Patients: 6-Month Follow-Up and Evaluation of a 3rd Vaccination. Cancers. 2022; 14(8):1962. https://doi.org/10.3390/cancers14081962
Chicago/Turabian StyleSchubert, Lorenz, Maximilian Koblischke, Lisa Schneider, Edit Porpaczy, Florian Winkler, Ulrich Jaeger, Stephan Blüml, Helmuth Haslacher, Heinz Burgmann, Judith H. Aberle, and et al. 2022. "Immunogenicity of COVID-19 Vaccinations in Hematological Patients: 6-Month Follow-Up and Evaluation of a 3rd Vaccination" Cancers 14, no. 8: 1962. https://doi.org/10.3390/cancers14081962
APA StyleSchubert, L., Koblischke, M., Schneider, L., Porpaczy, E., Winkler, F., Jaeger, U., Blüml, S., Haslacher, H., Burgmann, H., Aberle, J. H., Winkler, S., & Tobudic, S. (2022). Immunogenicity of COVID-19 Vaccinations in Hematological Patients: 6-Month Follow-Up and Evaluation of a 3rd Vaccination. Cancers, 14(8), 1962. https://doi.org/10.3390/cancers14081962






