Treatment Algorithm for Cancerous Wounds: A Systematic Review
Abstract
:Simple Summary
Abstract
1. Introduction
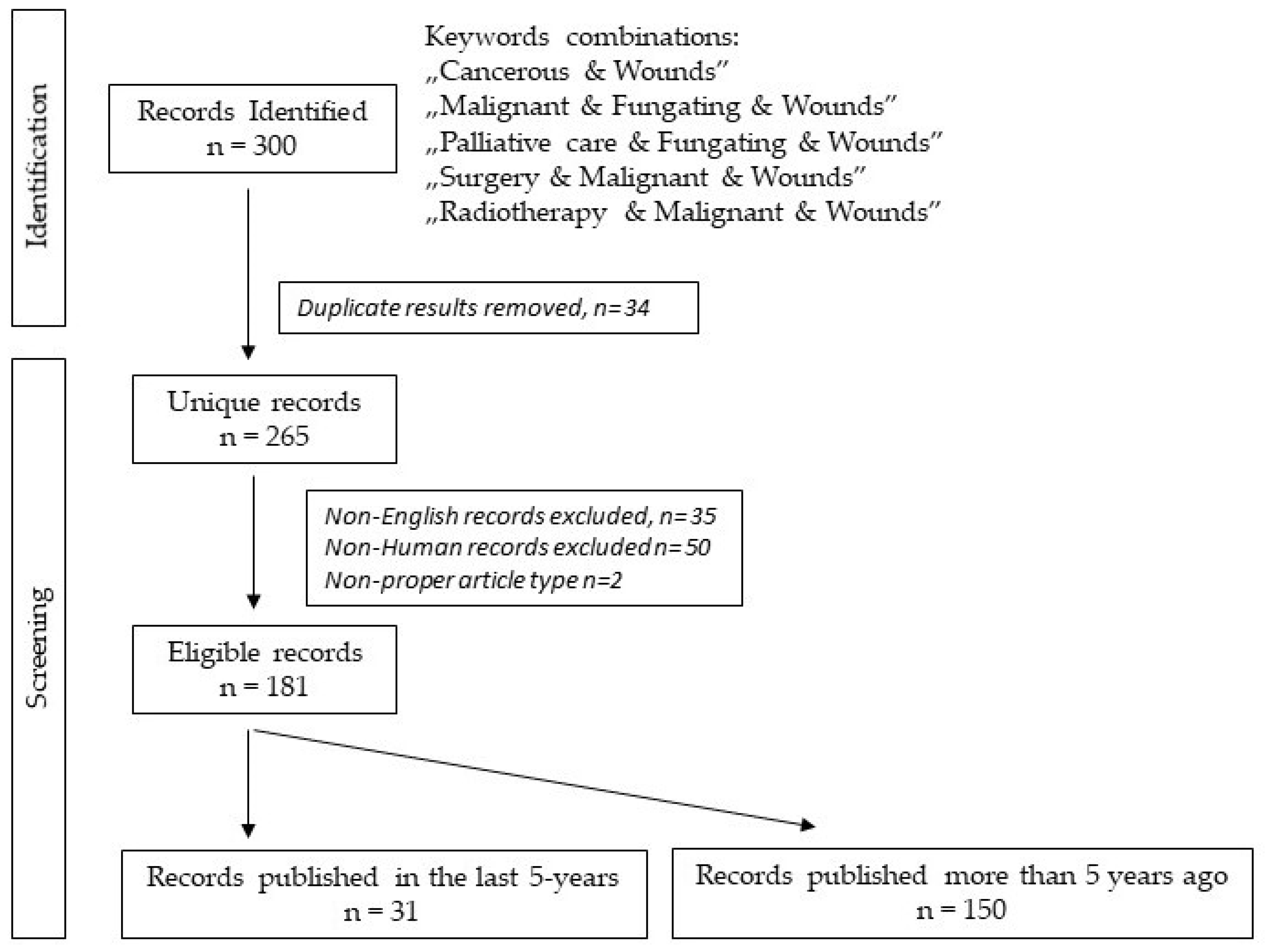
2. Materials and Methods
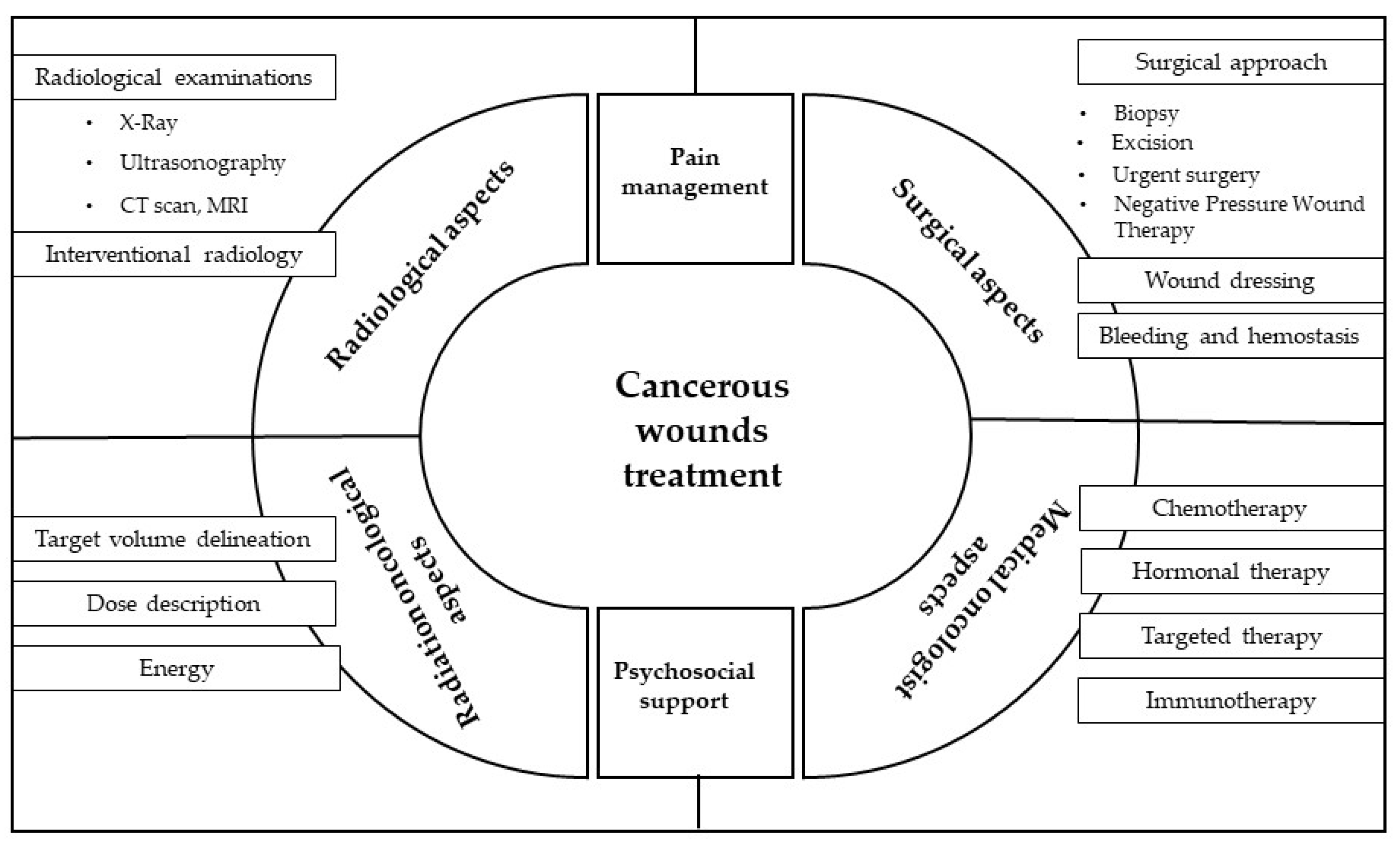
3. Results
3.1. Grading of Cancerous Wound, Measurement Tools
3.2. Radiological Aspects
3.2.1. Radiological Examinations
- X-ray can be informative in case of cancerous wound if there is suspicion of fistula formation or perforation. Free air is visualized on X-ray in such condition. X-ray is also diagnostic to evaluate bone involvement and consecutive osteomyelitis.
- Ultrasonography is useful to detect free air, free fluid. With its help the point for drainage can be marked.
- CT scan, MRI are sophisticated radiological methods. Use them in the end-of-life treatment only if the result changes the therapeutic decision and prolongs a meaningful life.
3.2.2. Interventional Radiology Can Be Useful in Case of Severe Bleeding
3.3. Surgical Aspects
3.3.1. Surgical Approach Is Not the First Chosen Method to Treat Malignant Fungating Wounds, since It Has Limited Role in Managing Cancerous Wounds
- Biopsy has to be taken in case on unknown histology or when progression develops during systemic treatment and the new biopsy data could change the therapeutic algorithm, i.e., new mutation with therapeutical consequence.
- Excision is rare in case of cancerous wound. When such wounds appear and infiltrating the skin the tumorous condition requires systemic treatment, furthermore R0 resection is highly unrealistic. In certain case tumor size may allow for performance in toto excision.
- Urgent surgery in rare cases would be inevitable. Acute surgical indications are bleeding, ileus (palliative deviation needed), septic condition (requires even amputation) [10], myelon compression, etc. The mortality rate is very high.
- Negative Pressure Wound Therapy (−100 to −125 mmHg pressure continuously for 24 h) is not routinely advised in malignant fungating wound as it influences the lymphatic drainage and may transfer tumorous cells into the circulation [5]. From the other hand, it may alleviate procedure decreasing symptoms and improving quality of life in end term phase of the disease, therefore in selected cases can be used in palliative care [5,11,12].
3.3.2. Wound Dressing
- The most important component of cancerous wound care is to select the appropriate wound dressing, since many aspects can influence our decision.
- Type of dressing.Antimicrobial effect of dressing is useful due to accompanying infection. Antimicrobial properties of the silver dressings are very effective to reduce malignant fungating wound discharge and malodor, respectively [5,13,14]. However, it is to be considered, that radiotherapy is not compatible with silver contain bandages as it influences the ionizing radiation, mainly enhances unwanted side effects. Other antimicrobial bandages were tested in randomized studies to find effective agents. Honey coated foam dressing has the same effect as silver containing bandage [5,14,15]. However, authors do not recommend honey as it cannot be standardized treatment agent.Hydrocolloid foam bandages can ease the pain and help the probable healing circumstances with its moisturizing effect meanwhile it absorbs massive exudation as well.Calcium alginate compress can minimize the bleeding [16].
- Time of dressing.Proper time for wound dressing change must be implemented the patient’s daily routine together with bathing or hygienic procedures. In case of sever condition, pain prevention is obligate. It is recommended to minimize the frequency of dressing change according to the wound quality.
- Topicals for hemostasis and antibiotics develop good effect in reduction of malodor and discharge:Polyhexamethylene biguanide (PHMB 0.2%) has the same result as metronidazole. Both significantly reduce wound odor by day eight [17].6% miltefostine solution is less useful in malignant wounds. It was applied in breast cancer patients with the aim of slowing progression [18].Arsenic trioxide with hemostyptic effect is an easily applicable and cheap agent for topical use [19].Green tea extract as an essential traditional phytotherapy has antimicrobial effect and also suppresses malodor [19,20].Oxymetazoline, an alpha adrenoreceptor agonist has vasoconstrictor sympathomimetic and decongestant effect [21].Etamsylate is a sulfonic acid derivative with vascular protective and hemostatic effect can be directly applied into the wound.Charcoal as absorbent blocks malodor [5].
3.3.3. Bleeding and Hemostasis
3.4. Radiation Oncological Aspects
3.4.1. Target Volume Delineation
3.4.2. Dose Description
3.4.3. Energy
3.5. Systemic Therapy from a Medical Oncologist’s Perspective
3.5.1. Chemotherapy
3.5.2. Hormonal Therapy
3.5.3. Targeted Therapy
3.5.4. Immunotherapy
3.6. Pain Management Aspects
3.6.1. Pain Mechanism
3.6.2. Analgesia
- Pharmacological treatment can be local and systemic. When removing the dressing, in addition to saline hydrating, it may be necessary to use local anesthetics, preferably in the form of cream, gel or hydrogel, which is applied for approximately 20 min before dressing. Although lidocaine molecule changes its polarity in an inflammatory (acidic) environment and penetration the cell membrane is more difficult, a Cochrane systematic review found adequate evidence supporting use of a combination of lidocaine and prilocaine (Eutectic Mixture of Local Anesthetics, EMLA) cream for dressing pain [29]. Topically applied non-steroidal anti-inflammatory drugs (ibuprofen foam, diclofenac gel) have also been shown to be effective, although in both cases the statistical efficacy is lower than expected, NNT (number needed to treat) was 6 [29]. Topical ketamine, amitriptyline, morphine, methadone, buprenorphine, aspirin, capsaicin (0.025 to 0.075%), clonidine 0.1%, and menthol have all been described to reduce wound pain, but evidence regarding safety and efficacy are weak or lacking [28]. In systemic pain treatment, due to the presence of inflammatory pathomechanism, the use of non-steroidal anti-inflammatory drug is logical but may slightly enhance bleeding. The inflammatory and myofascial pain components respond poorly to opiates. In the latter case, infiltration of the affected muscles with local anesthetic, gentle massage and gymnastics can help. In case of a neuropathic pathomechanism, the opiate response can be predicted by simple drug tests, however, adjuvant analgesics are most likely required. Since the statistical efficacy of each agent in neuropathic pain is low [30,31], it is advisable to select the first adjuvant so that it can be combined with others [32]. The patient’s age, comorbidities, and medications should take into account when select these agents. A fully detailed discussion of the issue is beyond the scope of this study, but some specific aspects are worth mentioning. The first choice should be tricyclic antidepressants (NNT 3.5). Duloxetine and gabapentinoids are also effective, though to a lesser extent than NNT (6.4 for duloxetine; 7.1 for gabapentin and 7.7 for pregabalin) [30,33]. Many other drugs have been investigated, but the evidence of efficacy is sometimes questionable [30,31]. Drugs with strong serotonergic activity (SNRIs, clomipramine) may slightly increase bleeding. The edematous side effect of gabapentinoids based on vasodilation is well known. Whether this may have a harmful role to increase the amount of wound exudate is not yet clear. Carbamazepine is a strong inductor in the CYP system, interacts significantly with many drugs (NSAIDs, tramadol, fentanyl, oxycodone, methadone) and may increase bleeding. Valproate inhibits the metabolism of most NSAIDs and may slightly increase bleeding. Systemic lidocaine and ketamine have been frequently used in palliative care for the treatment of neuropathic pain in the last decade, but there is no consensus on the route of administration and applied doses. Efficacy data are also conflicting [34,35,36,37,38,39]. Preventing dressing pain fast acting opioids (transmucosal or parenteral fentanyl, sublingual methadone), parenteral or nasal ketamine and nitrous oxide may help [35,40,41,42].
- Complementary procedures (relaxation, aromatherapy, music therapy, meditation, etc.) can help reducing anticipation of anxiety and pain, thus inhibit central pain processes [43].
- Interventional treatments: intrathecal drug administration (opioids, local anesthetics, ziconotide, baclofen, clonidin) may be necessary in a small percentage of patients who are unresponsive to conventional therapies [33].
- Neuroablative procedure (chemical or thermal) may also be a therapeutic modality in refractory cases. In case of thoracic or abdominal wall infiltration, use of an easy-to-perform intercostal neurolytic blockade should be considered. In case of perineal tumors, if the patient has a stool and urinary deviation or a catheter, sacral neurolytic blockade may give significant help [44,45].
3.6.3. Pruritus and Skin Care
3.7. Psychosocial Aspect
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mortimer, P. Skin problems in palliative care: Medical aspects. In Oxford Textbook of Palliative Medicine, 3rd ed.; Doyle, D., Hanks, G., Macdonald, N., Eds.; Oxford Medical Publications: Oxford, UK, 1993; pp. 384–395. [Google Scholar]
- Grocott, P. The palliative management of fungating malignant wounds. J. Wound Care 1995, 4, 240–242. [Google Scholar] [CrossRef] [PubMed]
- Tilley, C.; Lipson, J.; Ramos, M. Palliative Wound Care for Malignant Fungating Wounds: Holistic Considerations at End-of-Life. Nurs. Clin. N. Am. 2016, 51, 513–531. [Google Scholar] [CrossRef] [PubMed]
- Lo, S.F.; Hu, W.Y.; Hayter, M.; Chang, S.C.; Hsu, M.Y.; Wu, L.Y. Experiences of living with a malignant fungating wound: A qualitative study. J. Clin. Nurs. 2008, 17, 2699–2708. [Google Scholar] [CrossRef]
- Tsichlakidou, A.; Govina, O.; Vasilopoulos, G.; Kavga, A.; Vastardi, M.; Kalemikerakis, I. Intervention for symptom management in patients with malignant fungating wounds—A systematic review. J. Balk. Union Oncol. 2019, 24, 1301–1308. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372. [Google Scholar] [CrossRef]
- Atkinson, T.M.; Ryan, S.J.; Bennett, A.V.; Stover, A.M.; Saracino, R.M.; Rogak, L.J.; Jewell, S.T.; Matsoukas, K.; Li, Y.; Basch, E. The association between clinician-based common terminology criteria for adverse events (CTCAE) and patient-reported outcomes (PRO): A systematic review. Support. Care Cancer 2016, 24, 3669–3676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacCarthy-Morrogh, L.; Martin, P. The hallmarks of cancer are also the hallmarks of wound healing. Sci. Signal. 2020, 13, eaay8690. [Google Scholar] [CrossRef]
- Chrisman, C.A. Care of chronic wounds in palliative care and end-of-life patients. Int. Wound J. 2010, 7, 214–235. [Google Scholar] [CrossRef]
- Parsons, C.M.; Pimiento, J.M.; Cheong, D.; Marzban, S.S.; Gonzalez, R.J.; Johnson, D.; Letson, G.D.; Zager, J.S. The role of radical amputations for extremity tumors: A single institution experience and review of the literature. J. Surg. Oncol. 2012, 105, 149–155. [Google Scholar] [CrossRef] [Green Version]
- Riot, S.; de Bonnecaze, G.; Garrido, I.; Ferron, G.; Grolleau, J.L.; Chaput, B. Is the use of negative pressure wound therapy for a malignant wound legitimate in a palliative context? “The concept of NPWT ad vitam”: A case series. Palliat. Med. 2015, 29, 470–473. [Google Scholar] [CrossRef]
- Cai, S.S.; Gowda, A.U.; Alexander, R.H.; Silverman, R.P.; Goldberg, N.H.; Rasko, Y.M. Use of negative pressure wound therapy on malignant wounds—A case report and review of literature. Int. Wound J. 2017, 14, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Kalemikerakis, J.; Vardaki, Z.; Fouka, G.; Vlachou, E.; Gkovina, U.; Kosma, E.; Dionyssopoulos, A. Comparison of foam dressings with silver versus foam dressings without silver in the care of malodorous malignant fungating wounds. J. Balk. Union Oncol. 2012, 17, 560. [Google Scholar]
- Lund-Nielsen, B.; Adamsen, L.; Kolmos, H.J.; Rørth, M.; Tolver, A.; Gottrup, F. The effect of honey-coated bandages compared with silver-coated bandages on treatment of malignant wounds—A randomized study. Wound Repair Regen. 2011, 19, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Lund-Nielsen, B.; Adamsen, L.; Gottrup, F.; Rorth, M.; Tolver, A.; Jorn Kolmos, H. Qualitative bacteriology in malignant wounds--a prospective, randomized, clinical study to compare the effect of honey and silver dressings. Ostomy Wound Manag. 2011, 57, 28–36. [Google Scholar]
- Firmino, F.; Villela-Castro, D.L.; Dos Santos, J.; de Gouveia Santos, V.L. Topical management of bleeding from malignant wounds caused by breast cancer: A systematic review. J. Pain Symptom Manag. 2021, 61, 1278–1286. [Google Scholar] [CrossRef]
- Villela-Castro, D.L.; de Gouveia Santos, V.L.; Woo, K. Polyhexanide versus metronidazole for odor management in malignant (fungating) wounds: A double-blinded, randomized, clinical trial. J. Wound Ostomy Cont. Nurs. 2018, 45, 413–418. [Google Scholar] [CrossRef]
- Adderley, U.J.; Holt, I.G. Topical agents and dressings for fungating wounds. Cochrane Database Syst. Rev. 2014, 5, 1–25. [Google Scholar] [CrossRef] [Green Version]
- da Costa Santos, C.M.; de Mattos Pimenta, C.A.; Nobre, M.R. A systematic review of topical treatments to control the odor of malignant fungating wounds. J. Pain Symptom Manag. 2010, 39, 1065–1076. [Google Scholar] [CrossRef]
- Wiese, F.; Kutschan, S.; Doerfler, J.; Mathies, V.; Buentzel, J.; Buentzel, J.; Huebner, J. Green tea and green tea extract in oncological treatment: A systematic review. Int. J. Vitam. Nutr. Res. 2021, 1–13. [Google Scholar] [CrossRef]
- Kratz, A.; Danon, A. Controlling bleeding from superficial wounds by the use of topical alpha adrenoreceptor agonists spray. A randomized, masked, controlled study. Injury 2004, 35, 1096–1101. [Google Scholar] [CrossRef]
- Marks, L.B.; Ten Haken, R.K.; Martel, M.K. Guest editor’s introduction to QUANTEC: A users guide. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, S1–S2. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Zhou, C. The past, present and future of immunotherapy against tumor. Transl. Lung Cancer Res. 2015, 4, 253–264. [Google Scholar] [PubMed]
- Tamai, N.; Mugita, Y.; Ikeda, M.; Sanada, H. The relationship between malignant wound status and pain in breast cancer patients. Eur. J. Oncol. Nurs. 2016, 24, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Probst, S.; Arber, A.; Faithful, S. Malignant fungating wounds: A survey of nurses’ clinical practice in Switzerland. Eur. J. Oncol. Nurs. 2009, 13, 295–298. [Google Scholar] [CrossRef]
- Price, P.; Fogh, K.; Glynn, C.; Krasner, D.L.; Osterbrink, J.; Sibbald, R.G. Managing painful chronic wounds: The Wound Pain Management Model. Int. Wound J. 2007, 4, 4–15. [Google Scholar] [CrossRef]
- Løhre, E.T.; Klepstad, P.; Bennett, M.I.; Brunelli, C.; Caraceni, A.; Fainsinger, R.L.; Knudsen, A.K.; Mercadante, S.; Sjøgren, P.; Kaasa, S. European Association for Palliative Care Research Network. From “breakthrough” to “episodic” cancer pain? A European Association for Palliative Care Research Network Expert Delphi Survey toward a common terminology and classification of transient cancer pain exacerbations. J. Pain Symptom Manag. 2016, 51, 1013–1019. [Google Scholar] [CrossRef] [Green Version]
- Woo, K.Y.; Krasner, D.L.; Kennedy, B.; Wardle, D.; Moir, O. Palliative wound care management strategies for palliative patients and their circles of care. Adv. Ski. Wound Care 2015, 28, 130–140. [Google Scholar] [CrossRef] [Green Version]
- Briggs, M.; Nelson, E.A.; Martyn-St James, M. Topical agents or dressings for pain in venous leg ulcers. Cochrane Database Syst. Rev. 2012, 11, 1–41. [Google Scholar] [CrossRef]
- Finnerup, N.B.; Attal, N.; Haroutounian, S.; McNicol, E.; Baron, R.; Dworkin, R.H.; Gilron, I.; Haanpää, M.; Hansson, P.; Jensen, T.S.; et al. Pharmacotherapy for neuropathic pain in adults: A systematic review and meta-analysis. Lancet Neurol. 2015, 14, 162–173. [Google Scholar] [CrossRef] [Green Version]
- Di Stefano, G.; Di Lionardo, A.; Di Pietro, G.; Cruccu, G.; Truini, A. Pharmacotherapeutic options for managing neuropathic pain: A systematic review and meta-analysis. Pain Res. Manag. 2021, 2021, 6656863. [Google Scholar] [CrossRef]
- Afonso, A.S.; Carnaval, T.; Cés, S.V. Combination therapy for neuropathic pain. A systematic review. J. Clin. Med. 2021, 10, 3533. [Google Scholar] [CrossRef] [PubMed]
- Fallon, M.; Giusti, R.; Aielli, F.; Hoskin, P.; Rolke, R.; Sharma, M.; Ripamonti, C.I. Management of cancer pain in adult patients: ESMO Clinical Practice Guidelines. Ann. Oncol. 2018, 29, 166–191. [Google Scholar] [CrossRef] [PubMed]
- Rupniewska-Ładyko, A. Ketamine—A unique adjuvant drug in pain treatment. Palliat. Med. Pract. 2020, 14, 188–193. [Google Scholar] [CrossRef]
- Page, N.; Nirabhawane, V. Intranasal Ketamine for the Management of Incidental Pain during Wound Dressing in Cancer Patients: A Pilot Study. Indian J. Palliat. Care 2018, 24, 58–60. [Google Scholar] [CrossRef]
- Oh, D.; Haffey, P.; Patel, A.; Gulati, A. Intravenous Ketamine for Cancer Pain Management Including Flares During the COVID-19 Pandemic: A Retrospective Study. Pain Med. 2021, 22, 1642–1650. [Google Scholar] [CrossRef]
- Kumar, A.; Kohli, A. Comeback of ketamine: Resurfacing facts and dispelling myths. Korean J. Anesthesiol. 2021, 74, 103–114. [Google Scholar] [CrossRef]
- Gupta, H.; Patel, A.; Eswani, Z.; Moore, P.; Steib, M.; Lee, C.; Kaye, A.D. Role of Intravenous Lidocaine Infusion in the Treatment of Peripheral Neuropathy. Orthop. Rev. 2021, 13, 1–13. [Google Scholar] [CrossRef]
- Moulin, D.E.; Morley-Forster, P.K.; Pirani, Z.; Rohfritsch, C.; Stitt, L. Intravenous lidocaine in the management of chronic peripheral neuropathic pain: A randomized-controlled trial. Can. J. Anesth. 2019, 66, 820–827. [Google Scholar] [CrossRef]
- Liu, Q.; Gao, L.L.; Dai, Y.L.; Li, Y.X.; Wang, Y.; Bai, C.F.; Mu, G.X.; Chai, X.M.; Han, W.J.; Zhou, L.J.; et al. Nitrous oxide/oxygen mixture for analgesia in adult cancer patients with breakthrough pain: A randomized, double-blind controlled trial. Eur. J. Pain 2018, 22, 492–500. [Google Scholar] [CrossRef] [Green Version]
- Hagen, N.A.; Fisher, K.; Stiles, C. Sublingual methadone for the management of cancer-related breakthrough pain: A pilot study. J. Palliat. Med. 2007, 10, 331–337. [Google Scholar] [CrossRef]
- Simon, S.M.; Schwartzberg, L.S. A review of rapid-onset opioids for breakthrough pain in patients with cancer. J. Opioid Manag. 2014, 10, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Woo, K.Y. Meeting the challenges of wound-associated pain: Anticipatory pain, anxiety, stress, and wound healing. Ostomy Wound Manag. 2008, 54, 10–12. [Google Scholar]
- Choi, E.J.; Choi, Y.M.; Jang, E.J.; Kim, J.Y.; Kim, T.K.; Kim, K.H. Neural ablation and regeneration in pain practice. Korean J. Pain 2016, 29, 3. [Google Scholar] [CrossRef]
- Waldman, S.D. Atlas of Interventional Pain Management, 4th ed.; Elsevier: Philadelphia, PA, USA, 2015; pp. 336–349, 576–586. [Google Scholar]
- Maida, V.; Ennis, M.; Kuzienisky, C.; Trozzolo, L. Symptoms associated with malignant wounds: A prospective case study. J. Pain Symptom Manag. 2009, 37, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Cevikbas, F.; Lerner, E.A. Physiology and pathophysiology of itch. Physiol. Rev. 2020, 100, 945–982. [Google Scholar] [CrossRef]
- Parnell, L.K.S. Itching for knowledge about wound and scar pruritus. Wounds 2018, 30, 17–26. [Google Scholar]
- Huguen, J.; Brenaut, E.; Clerc, C.J.; Poizeau, F.; Marcorelles, P.; Quereux, G.; Dupuy, A.; Misery, L. Comparison of characteristics of neuropathic and non-neuropathic pruritus to develop a tool for the diagnosis of neuropathic pruritus: The NP5. Front. Med. 2019, 6, 79. [Google Scholar] [CrossRef]
- Andrade, A.; Kuah, C.Y.; Martin-Lopez, J.E.; Chua, S.; Shpadaruk, V.; Sanclemente, G.; Franco, J.V. Interventions for chronic pruritus of unknown origin. Cochrane Database Syst. Rev. 2020, 1, 1–71. [Google Scholar] [CrossRef]
- Gibson, S.; Green, J. Review of patients’ experiences with fungating wounds and associated quality of life. J. Wound Care 2013, 22, 265–275. [Google Scholar] [CrossRef] [Green Version]
- Bolton, L. Evidence Corner: Evidence-based Care for Malignant Wounds. Wounds 2016, 28, 214–216. [Google Scholar]
- Ramasubbu, D.A.; Smith, V.; Hayden, F.; Cronin, P. Systemic antibiotics for treating malignant wounds. Cochrane Database Syst. Rev. 2017, 8, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, D.; Roberge, D.; Touati, N.; Maunsell, E.; Berbiche, D. Effects of interdisciplinary teamwork on patient-reported experience of cancer care. BMC Health Serv. Res. 2017, 17, 218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Furka, A.; Simkó, C.; Kostyál, L.; Szabó, I.; Valikovics, A.; Fekete, G.; Tornyi, I.; Oross, E.; Révész, J. Treatment Algorithm for Cancerous Wounds: A Systematic Review. Cancers 2022, 14, 1203. https://doi.org/10.3390/cancers14051203
Furka A, Simkó C, Kostyál L, Szabó I, Valikovics A, Fekete G, Tornyi I, Oross E, Révész J. Treatment Algorithm for Cancerous Wounds: A Systematic Review. Cancers. 2022; 14(5):1203. https://doi.org/10.3390/cancers14051203
Chicago/Turabian StyleFurka, Andrea, Csaba Simkó, László Kostyál, Imre Szabó, Anikó Valikovics, Gábor Fekete, Ilona Tornyi, Endre Oross, and János Révész. 2022. "Treatment Algorithm for Cancerous Wounds: A Systematic Review" Cancers 14, no. 5: 1203. https://doi.org/10.3390/cancers14051203
APA StyleFurka, A., Simkó, C., Kostyál, L., Szabó, I., Valikovics, A., Fekete, G., Tornyi, I., Oross, E., & Révész, J. (2022). Treatment Algorithm for Cancerous Wounds: A Systematic Review. Cancers, 14(5), 1203. https://doi.org/10.3390/cancers14051203





