Chloride Channels and Transporters: Roles beyond Classical Cellular Homeostatic pH or Ion Balance in Cancers
Abstract
Simple Summary
Abstract
1. Chloride Transport
2. Membrane-Associated Components of Chloride Movement
2.1. Transmembrane Member 16A
2.2. TMEM206
2.3. Calcium-Activated Chloride Channel Regulators
2.4. Chloride Intracellular Channels
2.5. Voltage-Gated Chloride Channels
2.6. Cystic Fibrosis Transmembrane Conductance Regulator
2.7. Voltage-Dependent Anion Channels
2.8. Volume-Regulated Anion Channel
2.9. Chloride-Bicarbonate Exchangers
3. Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Stauber, T.; Jentsch, T.J. Chloride in vesicular trafficking and function. Annu. Rev. Physiol. 2013, 75, 453–477. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Li, H.; Chen, Y.; Li, P.; Gao, L.; Zheng, Y.; Sun, Y.; Chen, J.; Qian, X. Expression of anoctamin 1 is associated with advanced tumor stage in patients with non-small cell lung cancer and predicts recurrence after surgery. Clin. Transl. Oncol. 2017, 19, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Ardeleanu, C.; Arsene, D.; Hinescu, M.; Andrei, F.; Gutu, D.; Luca, L.; Popescu, L.M. Pancreatic expression of DOG1 a novel gastrointestinal stromal tumor (GIST) biomarker. Appl. Immunohistochem. Mol. Morphol. 2009, 17, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Lu, M.; Liu, B.G.; Huang, Y.; Wang, K.W. Inhibition of Ca2+-activated Cl− channel ANO1/TMEM16A expression suppresses tumor growth and invasiveness in human prostate carcinoma. Cancer Lett. 2012, 326, 41–51. [Google Scholar] [CrossRef]
- Ubby, I.; Bussani, E.; Colonna, A.; Stacul, G.; Locatelli, M.; Scudieri, P.; Galietta, L.; Pagani, F. TMEM16A alternative splicing coordination in breast cancer. Mol. Cancer 2013, 12, 75. [Google Scholar] [CrossRef]
- Foda, A.M.; Mohamed, M.A. Aberrant expressions of c-KIT and DOG-1 in mucinous and nonmucinous colorectal carcinomas and relation to clinicopathologic features and prognosis. Ann. Diagn. Pathol. 2015, 19, 335–340. [Google Scholar] [CrossRef]
- Liu, F.; Cao, Q.H.; Lu, D.J.; Luo, B.; Lu, X.F.; Luo, R.C.; Wang, X.G. TMEM16A overexpression contributes to tumor invasion and poor prognosis of human gastric cancer through TGF-beta signaling. Oncotarget 2015, 6, 11585–11599. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Y.; Ren, Y.G.; Kang, L.; Zhang, L.H. Transmembrane protein with unknown function 16A overexpression promotes glioma formation through the nuclear factor-.B signaling pathway. Mol. Med. Rep. 2014, 9, 1068–1074. [Google Scholar] [CrossRef]
- Lee, Y.S.; Lee, J.K.; Bae, Y.; Lee, B.S.; Kim, E.; Cho, C.H.; Ryoo, K.; Yoo, J.; Kim, C.H.; Yi, G.S.; et al. Suppression of 14-3-3 gamma-mediated surface expression of ANO1 inhibits cancer progression of glioblastoma cells. Sci. Rep. 2016, 6, 26413. [Google Scholar] [CrossRef]
- Shang, L.; Hao, J.J.; Zhao, X.K.; He, J.Z.; Shi, Z.Z.; Liu, H.J.; Wu, L.F.; Jiang, Y.Y.; Shi, F.; Yang, H.; et al. ANO1 protein as a potential biomarker for esophageal cancer prognosis and precancerous lesion development prediction. Oncotarget 2016, 7, 24374–24382. [Google Scholar] [CrossRef]
- Jia, L.H.; Liu, W.; Guan, L.Z.; Lu, M.; Wang, K.W. Inhibition of calcium-activated chloride channel ANO1/TMEM16A suppresses tumor growth and invasion in human lung cancer. PLoS ONE 2015, 10, e0136584. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Yang, J.H.; Chen, H.W.; Ma, B.; Pan, K.M.; Su, C.K.; Xu, F.F.; Zhang, J.H. Knockdown of TMEM16A suppressed MAPK and inhibited cell proliferation and migration in hepatocellular carcinoma. Oncotargets Ther. 2016, 9, 325–333. [Google Scholar] [CrossRef]
- Jung, I.; Gurzu, S.; Turdean, S.; Ciortea, D.; Sahlean, D.I.; Golea, M.; Bara, T. Relationship of endothelial area with VEGF-A, COX-2, maspin, c-KIT, and DOG-1 immunoreactivity in liposarcomas versus non-lipomatous soft tissue tumors. Int. J. Clin. Exp. Pathol. 2015, 8, 1776–1782. [Google Scholar] [PubMed]
- Sah, S.P.; McCluggage, W.G. DOG1 immunoreactivity in uterine leiomyosarcomas. J. Clin. Pathol. 2013, 66, 40–43. [Google Scholar] [CrossRef]
- Abd Raboh, N.M.; Hakim, S.A. Diagnostic role of DOG1 and p63 immunohistochemistry in salivary gland carcinomas. Int. J. Clin. Exp. Pathol. 2015, 8, 9214–9222. [Google Scholar]
- Akpalo, H.; Lange, C.; Zustin, J. Discovered on gastrointestinal stromal tumour 1 (DOG1): A useful immunohistochemical marker for diagnosing chondroblastoma. Histopathology 2012, 60, 1099–1106. [Google Scholar] [CrossRef]
- Wang, H.; Zou, L.; Ma, K.; Yu, J.; Wu, H.; Wei, M.; Xiao, Q. Cell-specific mechanisms of TMEM16A Ca2+-activated chloride channel in cancer. Mol. Cancer 2017, 16, 152. [Google Scholar] [CrossRef]
- Kunzelmann, K.; Ousingsawat, J.; Benedetto, R.; Cabrita, I.; Schreiber, R. Contribution of anoctamins to cell survival and cell death. Cancers 2019, 11, 382. [Google Scholar] [CrossRef]
- Hu, C.; Zhang, R.G.; Jiang, D.P. TMEM16A as a potential biomarker in the diagnosis and prognosis of lung cancer. Arch. Iran. Med. 2019, 22, 32–38. [Google Scholar]
- Zhang, C.T.; Liu, J.X.; Han, Z.Y.; Cui, X.; Peng, D.T.; Xing, Y.F. Inhibition of TMEM16A suppresses growth and induces apoptosis in hepatocellular carcinoma. Int. J. Clin. Oncol. 2020, 25, 1145–1154. [Google Scholar] [CrossRef]
- Mokutani, Y.; Uemura, M.; Munakata, K.; Okuzaki, D.; Haraguchi, N.; Takahashi, H.; Nishimura, J.; Hata, T.; Murata, K.; Takemasa, I.; et al. Down-regulation of microRNA-132 is associated with poor prognosis of colorectal cancer. Ann. Surg. Oncol. 2016, 23, S599–S608. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Song, J.; Ouyang, Y.; Han, Q.; Chen, W.; Zhao, X.; Xie, Y.; Chen, Y.; Yuan, W.; Fan, C. Advances in Roles of miR-132 in the nervous system. Front. Pharmacol. 2017, 8, 770. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Tan, C.; Tang, C.; Ren, G.; Xiang, T.; Qiu, Z.; Liu, R.; Wu, Z. Epigenetic repression of miR-132 expression by the hepatitis B virus x protein in hepatitis B virus-related hepatocellular carcinoma. Cell. Signal. 2013, 25, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Spitzner, M.; Martins, J.R.; Soria, R.B.; Ousingsawat, J.; Scheidt, K.; Schreiber, R.; Kunzelmann, K. Eag1 and Bestrophin 1 are up-regulated in fast-growing colonic cancer cells. J. Biol. Chem. 2008, 283, 7421–7428. [Google Scholar] [CrossRef]
- Bharill, S.; Fu, Z.; Palty, R.; Isacoff, E.Y. Stoichiometry and specific assembly of Best ion channels. Proc. Natl. Acad. Sci. USA 2014, 111, 6491–6496. [Google Scholar] [CrossRef]
- Dam, V.S.; Boedtkjer, D.M.; Aalkjaer, C.; Matchkov, V. The bestrophin- and TMEM16A-associated Ca2+ -activated Cl− channels in vascular smooth muscles. Channels 2014, 8, 361–369. [Google Scholar] [CrossRef]
- Yoshimoto, S.; Matsuda, M.; Kato, K.; Jimi, E.; Takeuchi, H.; Nakano, S.; Kajioka, S.; Matsuzaki, E.; Hirofuji, T.; Inoue, R.; et al. Volume-regulated chloride channel regulates cell proliferation and is involved in the possible interaction between TMEM16A and LRRC8A in human metastatic oral squamous cell carcinoma cells. Eur. J. Pharmacol. 2021, 895, 173881. [Google Scholar] [CrossRef]
- Marsey, L.L.; Winpenny, J.P. Bestrophin expression and function in the human pancreatic duct cell line, CFPAC-1. J. Physiol. 2009, 587, 2211–2224. [Google Scholar] [CrossRef]
- Kunzelmann, K.; Kongsuphol, P.; Aldehni, F.; Tian, Y.; Ousingsawat, J.; Warth, R.; Schreiber, R. Bestrophin and TMEM16-Ca2+ activated Cl− channels with different functions. Cell Calcium 2009, 46, 233–241. [Google Scholar] [CrossRef]
- Barro-Soria, R.; Aldehni, F.; Almaca, J.; Witzgall, R.; Schreiber, R.; Kunzelmann, K. ER-localized bestrophin 1 activates Ca2+-dependent ion channels TMEM16A and SK4 possibly by acting as a counterion channel. Pflug. Arch. Eur. J. Phys. 2010, 459, 485–497. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, H.; Huang, D.; Qi, J.; Xu, J.; Gao, H.; Du, X.; Gamper, N.; Zhang, H. Characterization of the effects of Cl(-) channel modulators on TMEM16A and bestrophin-1 Ca2+ activated Cl− channels. Pflug. Arch. 2015, 467, 1417–1430. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.B.; Zhu, D.H.; Zhang, X.P.; Zhang, Y.; Zhou, J.P.; Dong, M. TMEM206 promotes the malignancy of colorectal cancer cells by interacting with AKT and extracellular signal-regulated kinase signaling pathways. J. Cell. Physiol. 2019, 234, 10888–10898. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, S.Y.; Yang, X.; Wang, Y.Q.; Cheng, Y.X. TMEM206 is a potential prognostic marker of hepatocellular carcinoma. Oncol. Lett. 2020, 20, 174. [Google Scholar] [CrossRef] [PubMed]
- Qiang, Y.Y.; Li, C.Z.; Sun, R.; Zheng, L.S.; Peng, L.X.; Yang, J.P.; Meng, D.F.; Lang, Y.H.; Mei, Y.; Xie, P.; et al. Along with its favorable prognostic role, CLCA2 inhibits growth and metastasis of nasopharyngeal carcinoma cells via inhibition of FAK/ERK signaling. J. Exp. Clin. Cancer Res. 2018, 37, 34. [Google Scholar] [CrossRef]
- Chen, H.; Liu, Y.; Jiang, C.D.; Chen, Y.M.; Li, H.; Liu, Q.A. Calcium-activated chloride channel A4 (CLCA4) plays inhibitory roles in invasion and migration through suppressing epithelial-mesenchymal transition via PI3K/AKT signaling in colorectal cancer. Med. Sci. Monit. 2019, 25, 4176–4185. [Google Scholar] [CrossRef]
- Walia, V.; Ding, M.; Kumar, S.; Nie, D.; Premkumar, L.S.; Elble, R.C. hCLCA2 is a p53-inducible inhibitor of breast cancer cell proliferation. Cancer Res. 2009, 69, 6624–6632. [Google Scholar] [CrossRef]
- Yu, Y.; Walia, V.; Elble, R.C. Loss of CLCA4 promotes epithelial-to-mesenchymal transition in breast cancer cells. PLoS ONE 2013, 8, e83943. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Cao, L.; Liu, B.; McCaig, C.D.; Pu, J. The transition from proliferation to differentiation in colorectal cancer is regulated by the calcium activated chloride channel A1. PLoS ONE 2013, 8, e60861. [Google Scholar] [CrossRef]
- Wang, W.; Wan, M.H.; Liao, D.J.; Peng, G.L.; Xu, X.; Yin, W.Q.; Guo, G.X.; Jiang, F.N.; Zhong, W.D.; He, J.X. Identification of potent chloride intracellular channel protein 1 inhibitors from traditional chinese medicine through structure-based virtual screening and molecular dynamics analysis. Biomed. Res. Int. 2017, 2017, 4751780. [Google Scholar] [CrossRef]
- Wang, W.; Xu, X.; Wang, W.J.; Shao, W.L.; Li, L.P.; Yin, W.Q.; Xiu, L.C.; Mo, M.C.; Zhao, J.; He, Q.Y.; et al. The expression and clinical significance of CLIC1 and HSP27 in lung adenocarcinoma. Tumor Biol. 2011, 32, 1199–1208. [Google Scholar] [CrossRef]
- Schlesinger, P.H.; Blair, H.C.; Teitelbaum, S.L.; Edwards, J.C. Characterization of the osteoclast ruffled border chloride channel and its role in bone resorption. J. Biol. Chem. 1997, 272, 18636–18643. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, S.M.; Mazzanti, M.; Tonini, R.; Qiu, M.R.; Warton, K.; Musgrove, E.A.; Campbell, T.J.; Breit, S.N. The nuclear chloride ion channel NCC27 is involved in regulation of the cell cycle. J. Physiol. 2000, 529, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.H.; Zhao, C.; Wu, H.; Wang, W.; Liu, X.M. CLIC1 protein A candidate prognostic biomarker for malignant-transformed hydatidiform Moles. Int. J. Gynecol. Cancer 2011, 21, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.F.; Chen, J.Q.; Wang, Z.; Liu, J.L.; Li, B.P. Function of chloride intracellular channel 1 in gastric cancer cells. World J. Gastroenterol. 2012, 18, 3070–3080. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Zhang, C.; Yu, P.W.; Tang, B.; Liu, T.; Cui, H.; Xu, J.H. Regulation of colon cancer cell migration and invasion by CLIC1-mediated RVD. Mol. Cell. Biochem. 2012, 365, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.D.; Guan, Y.B.; Jia, Y.Y.; Meng, Q.J.; Yang, J.J. Chloride intracellular channel 1 regulates prostate cancer cell proliferation and migration through the MAPK/ERK pathway. Cancer Biother. Radiopharm. 2014, 29, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Zeng, Y.; Liu, T.; Zhang, C.; Yu, P.W.; Hao, Y.X.; Luo, H.X.; Liu, G. Chloride intracellular channel 1 regulates colon cancer cell migration and invasion through ROS/ERK pathway. World J. Gastroenterol. 2014, 20, 2071–2078. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.C.; Li, M.L.; Wu, X.S.; Zhang, L.; Wu, W.G.; Ding, Q.; Weng, H.; Wang, X.A.; Liu, Y.B. CLIC1 overexpression is associated with poor prognosis in gallbladder cancer. Tumor Biol. 2015, 36, 193–198. [Google Scholar] [CrossRef]
- Chen, C.D.; Wang, C.S.; Huang, Y.H.; Chien, K.Y.; Liang, Y.; Chen, W.J.; Lin, K.H. Overexpression of CLIC1 in human gastric carcinoma and its clinicopathological significance. Proteomics 2007, 7, 155–167. [Google Scholar] [CrossRef]
- Nesiu, A.; Cimpean, A.M.; Ceausu, R.A.; Adile, A.; Ioiart, I.; Porta, C.; Mazzanti, M.; Camerota, T.C.; Raica, M. Intracellular chloride ion channel protein-1 expression in clear cell renal cell carcinoma. Cancer Genomics Proteomics 2019, 16, 299–307. [Google Scholar] [CrossRef]
- Setti, M.; Savalli, N.; Osti, D.; Richichi, C.; Angelini, M.; Brescia, P.; Fornasari, L.; Carro, M.S.; Mazzanti, M.; Pelicci, G. Functional role of CLIC1 ion channel in glioblastoma-derived stem/progenitor cells. J. Natl. Cancer Inst. 2013, 105, 1644–1655. [Google Scholar] [CrossRef] [PubMed]
- Peretti, M.; Raciti, F.M.; Carlini, V.; Verduci, I.; Sertic, S.; Barozzi, S.; Garre, M.; Pattarozzi, A.; Daga, A.; Barbieri, F.; et al. Mutual influence of ROS, pH, and CLIC1 membrane protein in the regulation of G1-S phase progression in human glioblastoma stem cells. Mol. Cancer Ther. 2018, 17, 2451–2461. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.S.; Chao, C.C.; Su, T.L.; Yeh, S.H.; Chen, D.S.; Chen, C.T.; Chen, P.J.; Jou, Y.S. Diverse cellular transformation capability of overexpressed genes in human hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 2004, 315, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.M.; Lin, S.H.; Yu, M.C.; Hsieh, S.Y. CLIC1 recruits PIP5K1A/C to induce cell-matrix adhesions for tumor metastasis. J. Clin. Investig. 2021, 131. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A.; Pollard, J.W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer 2009, 9, 239–252. [Google Scholar] [CrossRef]
- Law, A.Y.; Wong, C.K. Stanniocalcin-2 promotes epithelial-mesenchymal transition and invasiveness in hypoxic human ovarian cancer cells. Exp. Cell Res. 2010, 316, 3425–3434. [Google Scholar] [CrossRef]
- Kokura, S.; Yoshida, N.; Imamoto, E.; Ueda, M.; Ishikawa, T.; Uchiyama, K.; Kuchide, M.; Naito, Y.; Okanoue, T.; Yoshikawa, T. Anoxia/reoxygenation down-regulates the expression of E-cadherin in human colon cancer cell lines. Cancer Lett. 2004, 211, 79–87. [Google Scholar] [CrossRef]
- Jia, N.N.; Dong, S.L.; Zhao, G.; Gao, H.; Li, X.Q.; Zhang, H.H. CLIC1 overexpression is associated with poor prognosis in pancreatic ductal adenocarcinomas. J. Cancer Res. Ther. 2016, 12, 892–896. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, X.M.; Yin, Z.Y.; Zhao, W.X.; Zhou, J.Y.; Zhao, B.X.; Liu, P.G. Chloride intracellular channel 1 is overexpression in hepatic tumor and correlates with a poor prognosis. APMIS 2013, 121, 1047–1053. [Google Scholar] [CrossRef]
- Lu, J.H.; Dong, Q.; Zhang, B.T.; Wang, X.F.; Ye, B.; Zhang, F.; Song, X.L.; Gao, G.F.; Mu, J.S.; Wang, Z.; et al. Chloride intracellular channel 1 (CLIC1) is activated and functions as an oncogene in pancreatic cancer. Med. Oncol. 2015, 32, 171. [Google Scholar] [CrossRef]
- Zheng, D.L.; Huang, Q.L.; Zhou, F.; Huang, Q.J.; Lin, J.Y.; Lin, X. PA28 ss regulates cell invasion of gastric cancer via modulating the expression of chloride intracellular channel 1. J. Cell Biochem. 2012, 113, 1537–1546. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.W.; Chen, L.X.; Xu, B.; Lijing, W.J.; Li, H.Z.; Guo, J.; Li, W.D.; Nie, S.H.; Jacob, T.J.C.; Wang, L.W. Suppression of ClC-3 channel expression reduces migration of nasopharyngeal carcinoma cells. Biochem. Pharmacol. 2008, 75, 1706–1716. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.P.; Cui, Y.Y.; You, Q.; Lu, Y.X.; Zhang, J.F. MicroRNA-124 negatively regulates chloride intracellular channel 1 to suppress the migration and invasion of liver cancer cells. Oncol. Rep. 2019, 42, 1380–1390. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Cheng, W.; Peng, C.; Liu, Y.; Jiang, B. Decreased expression of hsa-miR-372 predicts poor prognosis in patients with gallbladder cancer by affecting chloride intracellular channel 1. Mol. Med. Rep. 2017, 16, 7848–7854. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, M.L.; Song, M.Y.; Chen, W.J.; Mao, J.; Song, L.; Wei, Y.Y.; Huang, Y.H.; Tang, J.W. Clic1 plays a role in mouse hepatocarcinoma via modulating Annexin A7 and Gelsolin in vitro and in vivo. Biomed. Pharmacother. 2015, 69, 416–419. [Google Scholar] [CrossRef]
- Barbieri, F.; Wurth, R.; Pattarozzi, A.; Verduci, I.; Mazzola, C.; Cattaneo, M.G.; Tonelli, M.; Solari, A.; Bajetto, A.; Daga, A.; et al. Inhibition of chloride intracellular channel 1 (CLIC1) as biguanide class-effect to impair human glioblastoma stem cell viability. Front. Pharmacol. 2018, 9, 899. [Google Scholar] [CrossRef]
- Dozynkiewicz, M.A.; Jamieson, N.B.; MacPherson, I.; Grindlay, J.; van den Berghe, P.V.E.; von Thun, A.; Morton, J.P.; Gourley, C.; Timpson, P.; Nixon, C.; et al. Rab25 and CLIC3 collaborate to promote integrin recycling from late endosomes/lysosomes and drive cancer progression. Dev. Cell 2012, 22, 131–145. [Google Scholar] [CrossRef]
- Zou, Q.; Yang, Z.L.; Li, D.Q.; Liu, Z.R.; Yuan, Y. Association of chloride intracellular channel 4 and Indian hedgehog proteins with survival of patients with pancreatic ductal adenocarcinoma. Int. J. Exp. Pathol. 2016, 97, 422–429. [Google Scholar] [CrossRef]
- Ueno, Y.; Ozaki, S.; Umakoshi, A.; Yano, H.; Choudhury, M.E.; Abe, N.; Sumida, Y.; Kuwabara, J.; Uchida, R.; Islam, A.; et al. Chloride intracellular channel protein 2 in cancer and non-cancer human tissues: Relationship with tight junctions. Tissue Barriers 2019, 7, e1593775. [Google Scholar] [CrossRef]
- Lemonnier, L.; Shuba, Y.; Crepin, A.; Roudbaraki, M.; Slomianny, C.; Mauroy, B.; Nilius, B.; Prevarskaya, N.; Skryma, R. Bcl-2-dependent modulation of swelling-activated Cl- current and ClC-3 expression in human prostate cancer epithelial cells. Cancer Res. 2004, 64, 4841–4848. [Google Scholar] [CrossRef]
- Weylandt, K.H.; Nebrig, M.; Jansen-Rosseck, N.; Amey, J.S.; Carmena, D.; Wiedenmann, B.; Higgins, C.F.; Sardini, A. CIC-3 expression enhances etoposide resistance by increasing acidification of the late endocytic compartment. Mol. Cancer Ther. 2008, 7, 2261. [Google Scholar] [CrossRef]
- Sontheimer, H. An unexpected role for ion channels in brain tumor metastasis. Exp. Biol. Med. 2008, 233, 779–791. [Google Scholar] [CrossRef]
- Cheng, W.; Zheng, S.T.; Li, L.; Zhou, Q.; Zhu, H.P.; Hu, J.; Luo, H.B. Chloride channel 3 (CIC-3) predicts the tumor size in hepatocarcinoma. Acta Histochem. 2019, 121, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Mao, J.W.; Wang, L.W.; Zhu, L.Y.; Li, H.Z.; Wang, W.Z.; Jin, X.B.; Zhu, J.Y.; Chen, L.X. ClC-3 chloride channels are essential for cell proliferation and cell cycle progression in nasopharyngeal carcinoma cells. Acta Biochim. Biophys. Sin. 2010, 42, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Fodde, R.; Brabletz, T. Wnt/beta-catenin signaling in cancer stemness and malignant behavior. Curr. Opin. Cell Biol. 2007, 19, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.C.; Guan, H.Y.; Fang, L.S.; Yang, Y.; Zhu, X.; Yuan, J.; Wu, J.H.; Li, M.F. MicroRNA-374a activates Wnt/beta-catenin signaling to promote breast cancer metastasis. J. Clin. Investig. 2013, 123, 566–579. [Google Scholar] [CrossRef]
- Mu, H.L.; Mu, L.J.; Gao, J.F. Suppression of CLC-3 reduces the proliferation, invasion and migration of colorectal cancer through Wnt/beta-catenin signaling pathway. Biochem. Biophys. Res. Commun. 2020, 533, 1240–1246. [Google Scholar] [CrossRef]
- Hughes-Davies, L.; Caldas, C.; Wishart, G.C. Tamoxifen: The drug that came in from the cold. Br. J. Cancer 2009, 101, 875–878. [Google Scholar] [CrossRef]
- Mao, J.W.; Xu, B.; Li, H.Z.; Chen, L.X.; Jin, X.B.; Zhu, J.Y.; Wang, W.Z.; Zhu, L.Y.; Zuo, W.H.; Chen, W.Q.; et al. Lack of association between stretch-activated and volume-activated Cl− currents in hepatocellular carcinoma cells. J. Cell. Physiol. 2011, 226, 1176–1185. [Google Scholar] [CrossRef]
- Mao, J.W.; Yuan, J.; Wang, L.W.; Zhang, H.F.; Jin, X.B.; Zhu, J.Y.; Li, H.Z.; Xu, B.; Chen, L.X. Tamoxifen inhibits migration of estrogen receptor-negative hepatocellular carcinoma cells by blocking the swelling-activated chloride current. J. Cell. Physiol. 2013, 228, 991–1001. [Google Scholar] [CrossRef]
- Mohammad-Panah, R.; Ackerley, C.; Rommens, J.; Choudhury, M.; Wang, Y.C.; Bear, C.E. The chloride channel ClC-4 co-localizes with cystic fibrosis transmembrane conductance regulator and may mediate chloride flux across the apical membrane of intestinal epithelia. J. Biol. Chem. 2002, 277, 566–574. [Google Scholar] [CrossRef]
- Ishiguro, T.; Avila, H.; Lin, S.Y.; Nakamura, T.; Yamamoto, M.; Boyd, D.D. Gene trapping identifies chloride channel 4 as a novel inducer of colon cancer cell migration, invasion and metastases. Br. J. Cancer 2010, 102, 774–782. [Google Scholar] [CrossRef][Green Version]
- Gatenby, R.A.; Gillies, R.J. Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 2004, 4, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Montcourrier, P.; Mangeat, P.H.; Valembois, C.; Salazar, G.; Sahuquet, A.; Duperray, C.; Rochefort, H. Characterization of very acidic phagosomes in breast-cancer cells and their association with invasion. J. Cell Sci. 1994, 107, 2381–2391. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Lin, L.B.; Yong, M.; Dong, X.J.; Yu, T.H.; Hu, L.N. Adenovirus-mediated overexpression of cystic fibrosis transmembrane conductance regulator enhances invasiveness and motility of serous ovarian cancer cells. Mol. Med. Rep. 2016, 13, 265–272. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Peng, X.; Wu, Z.; Yu, L.; Li, J.K.; Xu, W.M.; Chan, H.C.; Zhang, Y.; Hu, L.N. Overexpression of cystic fibrosis transmembrane conductance regulator (CFTR) is associated with human cervical cancer malignancy, progression and prognosis. Gynecol. Oncol. 2012, 125, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Tu, Z.W.; Chen, Q.; Zhang, J.T.; Jiang, X.H.; Xia, Y.F.; Chan, H.C. CFTR is a potential marker for nasopharyngeal carcinoma prognosis and metastasis. Oncotarget 2016, 7, 76955–76965. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, C.; Song, C.; Li, J.X.; Sun, Q. CFTR functions as a tumor suppressor and is regulated by DNA methylation in colorectal cancer. Cancer Manag. Res. 2020, 12, 4261–4270. [Google Scholar] [CrossRef]
- Zhang, J.T.; Jiang, X.H.; Xie, C.; Cheng, H.; Dong, J.D.; Wang, Y.; Fok, K.L.; Zhang, X.H.; Sun, T.T.; Tsang, L.L.; et al. Downregulation of CFTR promotes epithelial-to-mesenchymal transition and is associated with poor prognosis of breast cancer. BBA Mol. Cell Res. 2013, 1833, 2961–2969. [Google Scholar] [CrossRef]
- Li, J.; Zhang, J.T.; Jiang, X.H.; Shi, X.S.; Shen, J.F.; Feng, F.L.; Chen, J.Y.; Liu, G.H.; He, P.; Jiang, J.H.; et al. The cystic fibrosis transmembrane conductance regulator as a biomarker in non-small cell lung cancer. Int. J. Oncol. 2015, 46, 2107–2115. [Google Scholar] [CrossRef]
- Xia, X.; Wang, J.; Liu, Y.; Yue, M. Lower cystic fibrosis transmembrane conductance regulator (CFTR) promotes the proliferation and migration of endometrial carcinoma. Med. Sci. Monit. 2017, 23, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Jiang, X.H.; Zhang, J.T.; Sun, T.T.; Dong, J.D.; Sanders, A.J.; Diao, R.Y.; Wang, Y.; Fok, K.L.; Tsang, L.L.; et al. CFTR suppresses tumor progression through miR-193b targeting urokinase plasminogen activator (uPA) in prostate cancer. Oncogene 2013, 32, 2282–2291. [Google Scholar] [CrossRef] [PubMed]
- Than, B.L.N.; Linnekamp, J.F.; Starr, T.K.; Largaespada, D.A.; Rod, A.; Zhang, Y.; Bruner, V.; Abrahante, J.; Schumann, A.; Luczak, T.; et al. CFTR is a tumor suppressor gene in murine and human intestinal cancer. Oncogene 2016, 35, 4191–4199. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Li, H.; Liu, Y.; Jiang, L. Knockdown of CFTR enhances sensitivity of prostate cancer cells to cisplatin via inhibition of autophagy. Neoplasma 2017, 64, 709–717. [Google Scholar] [CrossRef]
- Li, H.; Ma, N.X.; Wang, J.; Wang, Y.; Yuan, C.; Wu, J.; Luo, M.H.; Yang, J.L.; Chen, J.; Shi, J.; et al. Nicotine induces progressive properties of lung adenocarcinoma A549 cells by inhibiting cystic fibrosis transmembrane conductance regulator (CFTR) expression and plasma membrane localization. Technol. Cancer Res. Treat. 2018, 17, 1533033818809984. [Google Scholar] [CrossRef]
- Jozwiak, P.; Ciesielski, P.; Forma, E.; Kozal, K.; Wojcik-Krowiranda, K.; Cwonda, L.; Bienkiewicz, A.; Brys, M.; Krzeslak, A. Expression of voltage-dependent anion channels in endometrial cancer and its potential prognostic significance. Tumour Biol. 2020, 42, 1010428320951057. [Google Scholar] [CrossRef]
- Yang, G.; Zhou, D.; Li, J.; Wang, W.; Zhong, W.; Fan, W.; Yu, M.; Cheng, H. VDAC1 is regulated by BRD4 and contributes to JQ1 resistance in breast cancer. Oncol. Lett. 2019, 18, 2340–2347. [Google Scholar] [CrossRef]
- Liu, X.; He, B.; Xu, T.; Pan, Y.; Hu, X.; Chen, X.; Wang, S. MiR-490-3p functions as a tumor suppressor by inhibiting oncogene VDAC1 expression in colorectal cancer. J. Cancer 2018, 9, 1218–1230. [Google Scholar] [CrossRef]
- Ko, J.H.; Gu, W.; Lim, I.; Zhou, T.; Bang, H. Expression profiling of mitochondrial voltage-dependent anion channel-1 associated genes predicts recurrence-free survival in human carcinomas. PLoS ONE 2014, 9, e110094. [Google Scholar] [CrossRef]
- Arif, T.; Vasilkovsky, L.; Refaely, Y.; Konson, A.; Shoshan-Barmatz, V. Silencing VDAC1 expression by siRNA inhibits cancer cell proliferation and tumor growth in vivo. Mol. Ther. Nucleic Acids 2014, 3, e159. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, T.; Zhao, W.; Xu, L.; Yang, Y.; Liao, Q.; Zhao, Y. A single talent immunogenic membrane antigen and novel prognostic predictor: Voltage-dependent anion channel 1 (VDAC1) in pancreatic cancer. Sci. Rep. 2016, 6, 33648. [Google Scholar] [CrossRef]
- Wang, F.R.; Qiang, Y.; Zhu, L.R.; Jiang, Y.S.; Wang, Y.D.; Shao, X.; Yin, L.; Chen, J.H.; Chen, Z. MicroRNA-7 downregulates the oncogene VDAC1 to influence hepatocellular carcinoma proliferation and metastasis. Tumor Biol. 2016, 37, 10235–10246. [Google Scholar] [CrossRef]
- Maldonado, E.N.; Lemasters, J.J. Warburg revisited: Regulation of mitochondrial metabolism by voltage-dependent anion channels in cancer cells. J. Pharmacol. Exp. Ther. 2012, 342, 637–641. [Google Scholar] [CrossRef]
- Shoshan-Barmatz, V.; De Pinto, V.; Zweckstetter, M.; Raviv, Z.; Keinan, N.; Arbel, N. VDAC, a multi-functional mitochondrial protein regulating cell life and death. Mol. Asp. Med. 2010, 31, 227–285. [Google Scholar] [CrossRef] [PubMed]
- Koren, I.; Raviv, Z.; Shoshan-Barmatz, V. Downregulation of voltage-dependent anion channel-1 expression by RNA interference prevents cancer cell growth in vivo. Cancer Biol. Ther. 2010, 9, 1046–1052. [Google Scholar] [CrossRef] [PubMed]
- Simamura, E.; Hirai, K.; Shimada, H.; Koyama, J.; Niwa, Y.; Shimizu, S. Furanonaphthoquinones cause apoptosis of cancer cells by inducing the production of reactive oxygen species by the mitochondrial voltage-dependent anion channel. Cancer Biol. Ther. 2006, 5, 1523–1529. [Google Scholar] [CrossRef] [PubMed]
- Pernemalm, M.; De Petris, L.; Branca, R.M.; Forshed, J.; Kanter, L.; Soria, J.C.; Girard, P.; Validire, P.; Pawitan, Y.; van den Oord, J.; et al. Quantitative proteomics profiling of primary lung adenocarcinoma tumors reveals functional perturbations in tumor metabolism. J. Proteome Res. 2013, 12, 3934–3943. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.Y.; Liu, M.; Li, X.; Tang, H. miR-490-3p modulates cell growth and epithelial to mesenchymal transition of hepatocellular carcinoma cells by targeting endoplasmic reticulum-Golgi intermediate compartment protein 3 (ERGIC3). J. Biol. Chem. 2013, 288, 4035–4047. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Liu, Y.; Gao, Q.; Han, Y.; Zhang, G.; Xu, S.; Cheng, K.; Zou, W. miR-490-3p inhibits the growth and invasiveness in triple-negative breast cancer by repressing the expression of TNKS2. Gene 2016, 593, 41–47. [Google Scholar] [CrossRef]
- Maldonado, E.N.; Sheldon, K.L.; DeHart, D.N.; Patnaik, J.; Manevich, Y.; Townsend, D.M.; Bezrukov, S.M.; Rostovtseva, T.K.; Lemasters, J.J. Voltage-dependent anion channels modulate mitochondrial metabolism in cancer cells: Regulation by free tubulin and erastin. J. Biol. Chem. 2013, 288, 11920–11929. [Google Scholar] [CrossRef]
- Zhou, K.; Yao, Y.L.; He, Z.C.; Chen, C.; Zhang, X.N.; Yang, K.D.; Liu, Y.Q.; Liu, Q.; Fu, W.J.; Chen, Y.P.; et al. VDAC2 interacts with PFKP to regulate glucose metabolism and phenotypic reprogramming of glioma stem cells. Cell Death Dis. 2018, 9, 988. [Google Scholar] [CrossRef]
- Verkman, A.S.; Galietta, L.J.V. Chloride transport modulators as drug candidates. Am. J. Physiol. Cell Physiol. 2021, 321, C932–C946. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, L.; Zhu, L.; Nie, S.; Zhang, J.; Zhong, P.; Cai, B.; Luo, H.; Jacob, T.J. Cell cycle-dependent expression of volume-activated chloride currents in nasopharyngeal carcinoma cells. Am. J. Physiol. Cell Physiol. 2002, 283, C1313–C1323. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.W.; Chen, L.X.; Zhu, L.Y.; Rawle, M.; Nie, S.H.; Zhang, J.; Ping, Z.; Cai, K.R.; Jacob, T.J.C. Regulatory volume decrease is actively modulated during the cell cycle. J. Cell. Physiol. 2002, 193, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.X.; Zhu, L.Y.; Jacob, T.J.; Wang, L.W. Roles of volume-activated Cl- currents and regulatory volume decrease in the cell cycle and proliferation in nasopharyngeal carcinoma cells. Cell Prolif. 2007, 40, 253–267. [Google Scholar] [CrossRef]
- Kurashima, K.; Shiozaki, A.; Kudou, M.; Shimizu, H.; Arita, T.; Kosuga, T.; Konishi, H.; Komatsu, S.; Kubota, T.; Fujiwara, H.; et al. LRRC8A influences the growth of gastric cancer cells via the p53 signaling pathway. Gastric Cancer 2021, 24, 1063–1075. [Google Scholar] [CrossRef]
- Wong, R.; Chen, W.; Zhong, X.; Rutka, J.T.; Feng, Z.P.; Sun, H.S. Swelling-induced chloride current in glioblastoma proliferation, migration, and invasion. J. Cell. Physiol. 2018, 233, 363–370. [Google Scholar] [CrossRef]
- Lu, P.; Ding, Q.; Li, X.; Ji, X.; Li, L.; Fan, Y.; Xia, Y.; Tian, D.; Liu, M. SWELL1 promotes cell growth and metastasis of hepatocellular carcinoma in vitro and in vivo. EBioMedicine 2019, 48, 100–116. [Google Scholar] [CrossRef]
- Min, X.J.; Li, H.; Hou, S.C.; He, W.; Liu, J.; Hu, B.; Wang, J. Dysfunction of volume-sensitive chloride channels contributes to cisplatin resistance in human lung adenocarcinoma cells. Exp. Biol. Med. 2011, 236, 483–491. [Google Scholar] [CrossRef]
- Sorensen, B.H.; Dam, C.S.; Sturup, S.; Lambert, I.H. Dual role of LRRC8A-containing transporters on cisplatin resistance in human ovarian cancer cells. J. Inorg. Biochem. 2016, 160, 287–295. [Google Scholar] [CrossRef]
- Planells-Cases, R.; Lutter, D.; Guyader, C.; Gerhards, N.M.; Ullrich, F.; Elger, D.A.; Kucukosmanoglu, A.; Xu, G.; Voss, F.K.; Reincke, S.M.; et al. Subunit composition of VRAC channels determines substrate specificity and cellular resistance to Pt-based anti-cancer drugs. EMBO J. 2015, 34, 2993–3008. [Google Scholar] [CrossRef] [PubMed]
- Mueller, T.J.; Morrison, M. Detection of a variant of protein 3, the major transmembrane protein of the human erythrocyte. J. Biol. Chem. 1977, 252, 6573–6576. [Google Scholar] [CrossRef]
- Demuth, D.R.; Showe, L.C.; Ballantine, M.; Palumbo, A.; Fraser, P.J.; Cioe, L.; Rovera, G.; Curtis, P.J. Cloning and structural characterization of a human non-erythroid band 3-like protein. EMBO J. 1986, 5, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Kopito, R.R.; Lee, B.S.; Simmons, D.M.; Lindsey, A.E.; Morgans, C.W.; Schneider, K. Regulation of intracellular pH by a neuronal homolog of the erythrocyte anion exchanger. Cell 1989, 59, 927–937. [Google Scholar] [CrossRef]
- Tsuganezawa, H.; Kobayashi, K.; Iyori, M.; Araki, T.; Koizumi, A.; Watanabe, S.I.; Kaneko, A.; Fukao, T.; Monkawa, T.; Yoshida, T.; et al. A new member of the HCO 3-transporter superfamily is an apical anion exchanger of beta-intercalated cells in the kidney. J. Biol. Chem. 2001, 276, 8180–8189. [Google Scholar] [CrossRef]
- Shen, W.W.; Wu, J.; Cai, L.; Liu, B.Y.; Gao, Y.; Chen, G.Q.; Fu, G.H. Expression of anion exchanger 1 sequestrates p16 in the cytoplasm in gastric and colonic adenocarcinoma. Neoplasia 2007, 9, 812–819. [Google Scholar] [CrossRef]
- Xu, W.Q.; Song, L.J.; Liu, Q.; Zhao, L.; Zheng, L.; Yan, Z.W.; Fu, G.H. Expression of anion exchanger 1 is associated with tumor progress in human gastric cancer. J. Cancer Res. Clin. 2009, 135, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Suo, W.H.; Zhang, N.; Wu, P.P.; Zhao, L.; Song, L.J.; Shen, W.W.; Zheng, L.; Tao, J.; Long, X.D.; Fu, G.H. Anti-tumour effects of small interfering RNA targeting anion exchanger 1 in experimental gastric cancer. Br. J. Pharmacol. 2012, 165, 135–147. [Google Scholar] [CrossRef][Green Version]
- Wu, J.; Zhang, Y.C.; Suo, W.H.; Liu, X.B.; Shen, W.W.; Tian, H.; Fu, G.H. Induction of anion exchanger-1 translation and its opposite roles in the carcinogenesis of gastric cancer cells and differentiation of K562 cells. Oncogene 2010, 29, 1987–1996. [Google Scholar] [CrossRef]
- Shiozaki, A.; Kudou, M.; Ichikawa, D.; Shimizu, H.; Arita, T.; Kosuga, T.; Konishi, H.; Komatsu, S.; Fujiwara, H.; Okamoto, K.; et al. Expression and role of anion exchanger 1 in esophageal squamous cell carcinoma. Oncotarget 2017, 8, 17921–17935. [Google Scholar] [CrossRef]
- Wu, T.T.; Hsieh, Y.H.; Wu, C.C.; Tsai, J.H.; Hsieh, Y.S.; Huang, C.Y.; Liu, J.Y. Overexpression of anion exchanger 2 in human hepatocellular carcinoma. Chin. J. Physiol. 2006, 49, 192–198. [Google Scholar] [PubMed]
- Song, L.J.; Liu, R.J.; Zeng, Z.; Alper, S.L.; Cui, H.J.; Lu, Y.; Zheng, L.; Yan, Z.W.; Fu, G.H. Gastrin inhibits a novel, pathological colon cancer signaling pathway involving EGR1, AE2, and P-ERK. J. Mol. Med. 2012, 90, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhao, L.; Yang, Y.; Tian, H.; Suo, W.H.; Yan, M.; Fu, G.H. EGR1 is critical for gastrin-dependent upregulation of anion exchanger 2 in gastric cancer cells. FEBS J. 2013, 280, 174–183. [Google Scholar] [CrossRef]
- Yang, Y.; Wu, P.P.; Wu, J.; Shen, W.W.; Wu, Y.L.; Fu, A.F.; Zheng, L.; Jin, X.L.; Fu, G.H. Expression of anion exchanger 2 in human gastric cancer. Exp. Oncol. 2008, 30, 81–87. [Google Scholar]
- Hwang, J.M.; Kao, S.H.; Hsieh, Y.H.; Li, K.L.; Wang, P.H.; Hsu, L.S.; Liu, J.Y. Reduction of anion exchanger 2 expression induces apoptosis of human hepatocellular carcinoma cells. Mol. Cell. Biochem. 2009, 327, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.J.; Lu, R.; Song, Y.N.; Zhu, J.Y.; Xia, W.; Zhang, M.; Shao, Z.Y.; Huang, Y.; Zhou, Y.; Zhang, H.; et al. Knockdown of anion exchanger 2 suppressed the growth of ovarian cancer cells via mTOR/p70S6K1 signaling. Sci. Rep. 2017, 7, 6362. [Google Scholar] [CrossRef] [PubMed]
- Khosrowabadi, E.; Rivinoja, A.; Risteli, M.; Tuomisto, A.; Salo, T.; Makinen, M.J.; Kellokumpu, S. SLC4A2 anion exchanger promotes tumour cell malignancy via enhancing net acid efflux across golgi membranes. Cell. Mol. Life Sci. 2021, 78, 6283–6304. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.; Shin, D.M.; Hong, J.H. Drug repurposing as an antitumor agent: Disulfiram-mediated carbonic anhydrase 12 and anion exchanger 2 modulation to inhibit cancer cell migration. Molecules 2019, 24, 3409. [Google Scholar] [CrossRef] [PubMed]
- Cordat, E.; Reithmeier, R.A. Structure, function, and trafficking of SLC4 and SLC26 anion transporters. Curr. Top. Membr. 2014, 73, 1–67. [Google Scholar] [CrossRef]
- Ko, S.B.; Shcheynikov, N.; Choi, J.Y.; Luo, X.; Ishibashi, K.; Thomas, P.J.; Kim, J.Y.; Kim, K.H.; Lee, M.G.; Naruse, S.; et al. A molecular mechanism for aberrant CFTR-dependent HCO3− transport in cystic fibrosis. EMBO J. 2002, 21, 5662–5672. [Google Scholar] [CrossRef] [PubMed]
- Di Stadio, C.S.; Altieri, F.; Miselli, G.; Elce, A.; Severino, V.; Chambery, A.; Quagliariello, V.; Villano, V.; de Dominicis, G.; Rippa, E.; et al. AMP18 interacts with the anion exchanger SLC26A3 and enhances its expression in gastric cancer cells. Biochimie 2016, 121, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Huang, Y.; Chen, L.; Guo, L.; Wang, L.; Li, M.; Liang, Y. Up-regulation of SLC26A6 in hepatocellular carcinoma and its diagnostic and prognostic significance. Crit. Rev. Eukaryot. Gene Expr. 2021, 31, 79–94. [Google Scholar] [CrossRef] [PubMed]
- Gururaja Rao, S.; Patel, N.J.; Singh, H. Intracellular chloride channels: Novel biomarkers in diseases. Front. Physiol. 2020, 11, 96. [Google Scholar] [CrossRef] [PubMed]
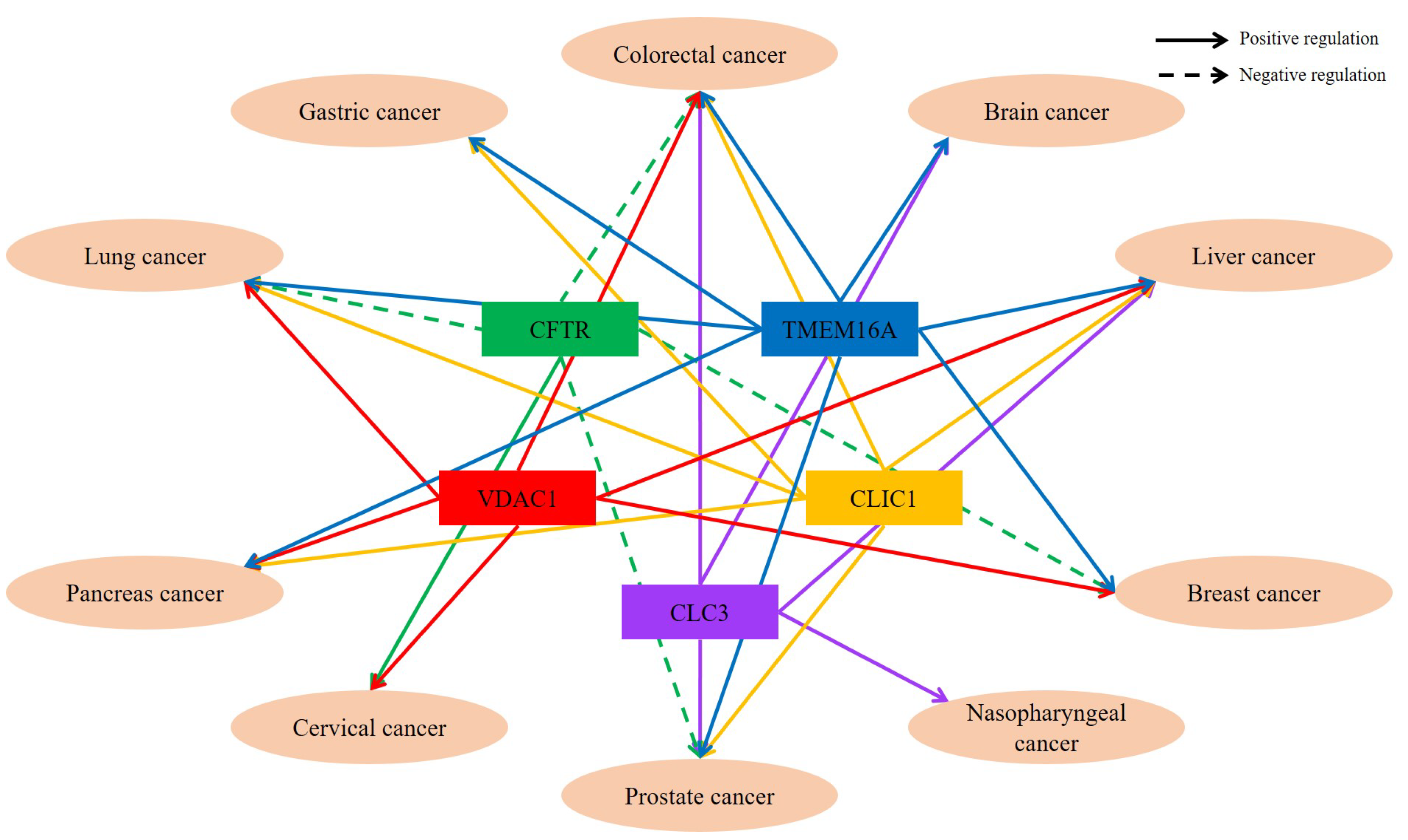
| Diseases | Chloride Channels/Transporters | Function | Reference |
|---|---|---|---|
| Lung cancer | TMEM16A | Cell proliferation and development to malignant tumor Positive correlation between TMEM16A and EGFR | [2,11,19] |
| TMEM206 | Enhanced cellular migration, invasion, and proliferation | [32] | |
| CLIC1 | Enhanced cellular migration and invasion | [40] | |
| CFTR | Inhibited various cancer-related processes, such as EMT | [90,95] | |
| VDAC1 | Cell growth, proliferation, migration, and invasion | [100,107] | |
| Pancreatic cancer | TMEM16A | Cell proliferation and development to malignant tumor | [3] |
| CLIC1 | Cell proliferation, growth, and invasiveness | [58,60] | |
| CLIC4 | Proposed metastatic marker | [68] | |
| VDAC1 | Cell growth, invasion, and migration | [101] | |
| Hepatic cancer | TMEM16A | Cell proliferation and development to malignant tumor | [12,20] |
| CLIC1 | Promoted cancer cell survival, proliferation, and invasion | [53,54,59,63,65] | |
| CLIC2 | Correlated with the tight junction | [69] | |
| CLC3 | Related to tumor size and prognosis marker Cell volume regulation | [73,79,80] | |
| VDAC1 | Regulated cell growth and EMT Cell growth, proliferation, migration, and invasion | [102,108] | |
| VDAC3 | Regulation of mitochondrial membrane potential | [103] | |
| AE2 | Inhibited proliferation and viability of cancer cells | [131,135] | |
| SLC26A6 | Diagnostic or prognostic biomarker for cancer | [142] | |
| Nasopharyngeal cancer | CLCA2 | Negatively regulate cancer development Regulate FAK/ERK signaling | [34] |
| CLC3 | Correlation between cell volume regulation and cancer development and metastasis | [62,74] | |
| CFTR | Enhanced cell migration and invasion Regulate EMT | [87] | |
| VRAC | Regulates proliferation | [115] | |
| Breast cancer | TMEM16A | Cell proliferation and development to malignant tumor | [5] |
| CLCA2 | Negatively regulate cancer development | [36] | |
| CLCA4 | Negatively regulate cancer development Decreased EMT | [37] | |
| CFTR | Inhibited various cancer-related processes, such as EMT | [89] | |
| VDAC1 | Promoted cell proliferation | [97] | |
| Colorectal cancer | TMEM16A | Cell proliferation and development to malignant tumor | [6,21] |
| Bestrophin-1 | Involved in cellular proliferation | [24] | |
| CLCA1 | Negatively regulate cancer development | [38] | |
| CLCA4 | Negatively regulate cancer development Decreased EMT | [35] | |
| CLIC1 | Enhanced cellular migration and invasion | [45,47] | |
| CLC3 | Regulate cell viability, proliferation, and metastasis | [77] | |
| CLC4 | Regulate invasion, migration, and pH of cytosolic and intracellular organelles | [81,82] | |
| CFTR | Decreased cell proliferation, migration, and invasion | [88] | |
| VDAC1 | Regulate apoptosis, proliferation, migration, and invasion | [98] | |
| AE2 | Contributed to progression of cancer Promote tumor cell malignancy Responsible for elevated Golgi resting pH | [132,137] | |
| Prostate cancer | TMEM16A | Cell proliferation and development to malignant tumor | [4] |
| CLIC1 | Promoted cancer cell survival, proliferation, and invasion | [46] | |
| CLC3 | Correlation between cell volume regulation and cancer development/ metastasis | [70] | |
| CFTR | Inhibited various cancer-related processes, such as EMT Enhanced chemo-resistance | [92,94] | |
| Gallbladder cancer | CLIC1 | Enhanced cellular migration and invasion | [48,64] |
| Gastric cancer | TMEM16A | Cell proliferation and development to malignant tumor | [7] |
| CLIC1 | Enhanced cellular migration and invasion | [44,49,61] | |
| AE1 | Involved in release of p16INK4A and cell growth Positive correlation with cancer size and metastasis | [126,127,128,129] | |
| AE2 | Contributed to progression of cancer | [133,134] | |
| SLC26A3 | Interact with AMP18 | [141] | |
| VRAC | Regulates proliferation | [116] | |
| Brain tumor | TMEM16A | Cell proliferation and development to malignant tumor | [8,9] |
| CLC3 | Enhanced cell invasion | [72] | |
| VDAC2 | Regulator for the metabolic reprogramming | [111] | |
| Clear cell renal cell carcinoma | CLIC1 | Cell invasion | [50] |
| Neuroendocrine tumor | CLC3 | Enhanced resistance to anticancer drug | [71] |
| Esophageal cancer | TMEM16A | Cell proliferation and development to malignant tumor | [10] |
| AE1 | Related with tumor progression through the crosstalk with MAPK and hedgehog signaling pathways | [130] | |
| Liposarcoma | TMEM16A | Cell proliferation and development to malignant tumor | [13] |
| Leiomyosarcoma | TMEM16A | Cell proliferation and development to malignant tumor | [14] |
| Salivary gland cancer | TMEM16A | Cell proliferation and development to malignant tumor | [15] |
| Chondroblastoma | TMEM16A | Cell proliferation and development to malignant tumor | [16] |
| Hydatidiform moles | CLIC1 | Enhanced cellular migration and invasion | [43] |
| Ovarian cancer cells | CLIC3 | Co-localized with active integrin α5β1 Cell migration and invasion | [67] |
| CFTR | Enhanced cell invasion and migration | [85] | |
| AE2 | Involved in tumorigenesis through activation of the mTOR/p70S6K1 pathway | [136] | |
| Cervical cancer | CFTR | Enhanced cell invasion and migration | [86] |
| VDAC1 | Regulate progression of tumorigenesis | [105] | |
| Endometrial cancer | CFTR | Inhibited various cancer-related processes, such as EMT | [91] |
| VDAC1 | Pore-forming role in mitochondrial membrane Cell growth, proliferation, migration, and invasion | [96] | |
| Oral squamous cell carcinoma | Bestrophin-1 | Involved in proliferation | [27] |
| VRAC | Regulates proliferation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.J.; Lee, P.C.-W.; Hong, J.H. Chloride Channels and Transporters: Roles beyond Classical Cellular Homeostatic pH or Ion Balance in Cancers. Cancers 2022, 14, 856. https://doi.org/10.3390/cancers14040856
Kim HJ, Lee PC-W, Hong JH. Chloride Channels and Transporters: Roles beyond Classical Cellular Homeostatic pH or Ion Balance in Cancers. Cancers. 2022; 14(4):856. https://doi.org/10.3390/cancers14040856
Chicago/Turabian StyleKim, Hyeong Jae, Peter Chang-Whan Lee, and Jeong Hee Hong. 2022. "Chloride Channels and Transporters: Roles beyond Classical Cellular Homeostatic pH or Ion Balance in Cancers" Cancers 14, no. 4: 856. https://doi.org/10.3390/cancers14040856
APA StyleKim, H. J., Lee, P. C.-W., & Hong, J. H. (2022). Chloride Channels and Transporters: Roles beyond Classical Cellular Homeostatic pH or Ion Balance in Cancers. Cancers, 14(4), 856. https://doi.org/10.3390/cancers14040856







