VISTA in Soft Tissue Sarcomas: A Perspective for Immunotherapy?
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Histopathology and Tissue Microarray Construction
2.3. VISTA Immunohistochemistry
2.4. TILs, CD3, PD-1 and PD-L1
2.5. Statistical Analysis
3. Results
3.1. Patients
3.2. VISTA Expression in STS and Histopathological Subtypes
3.3. VISTA Expression Is Associated with TILs, PD-1, PD-L1 and Grading
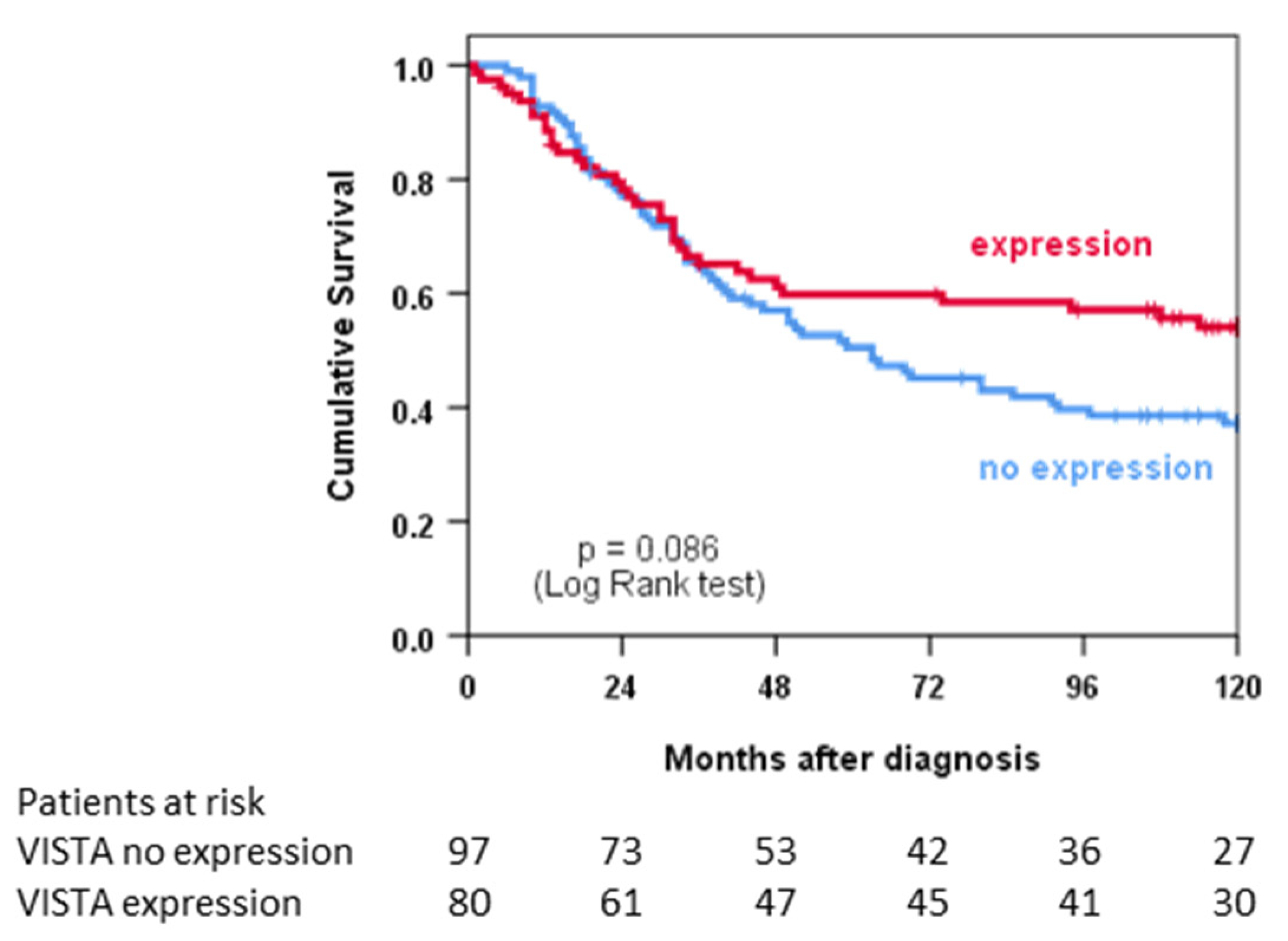
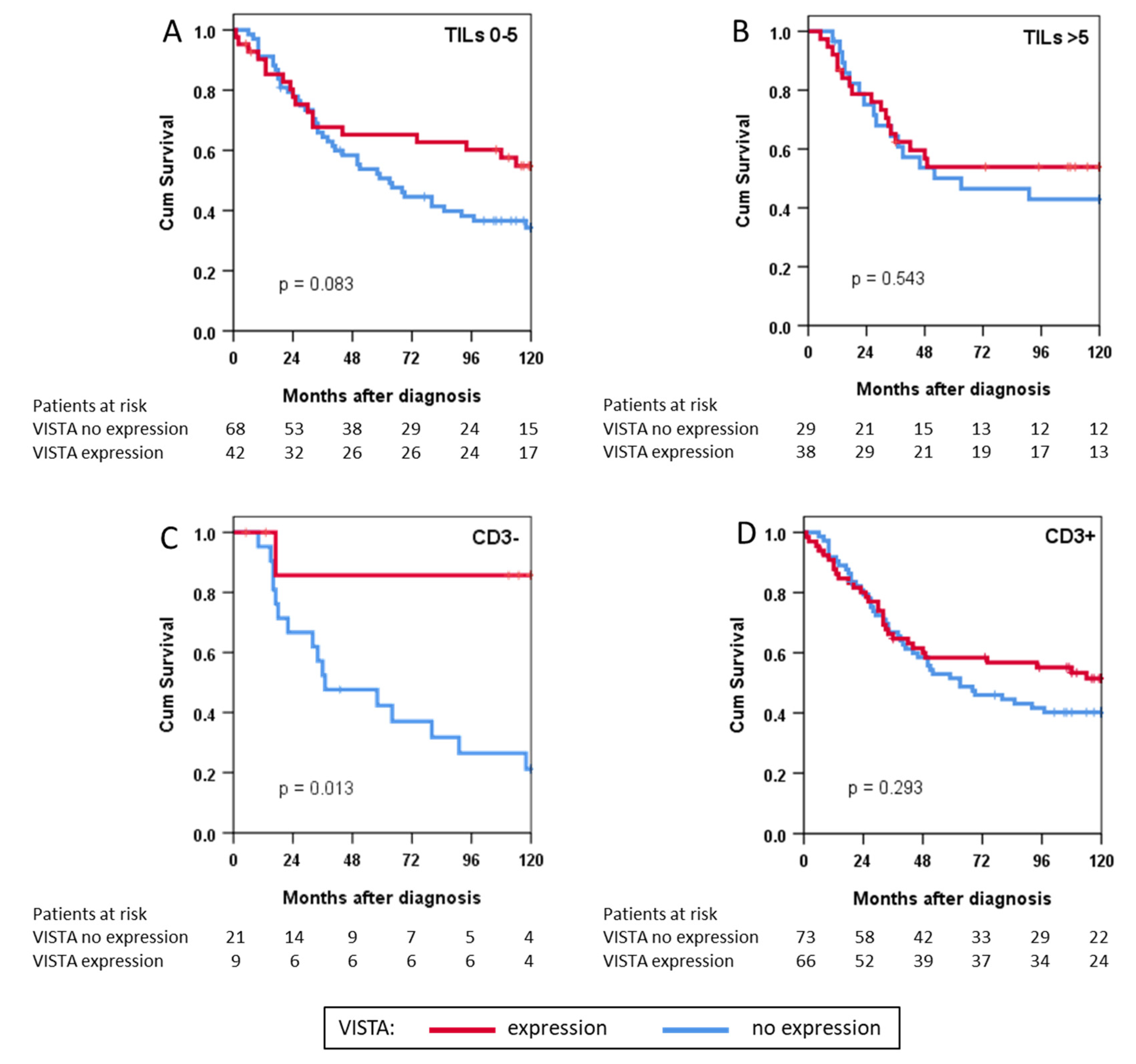
3.4. VISTA Expression and Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ratan, R.; Patel, S.R. Chemotherapy for soft tissue sarcoma. Cancer 2016, 122, 2952–2960. [Google Scholar] [CrossRef] [Green Version]
- Gronchi, A.; Ferrari, S.; Quagliuolo, V.; Broto, J.M.; Pousa, A.L.; Grignani, G.; Basso, U.; Blay, J.Y.; Tendero, O.; Beveridge, R.D.; et al. Histotype-tailored neoadjuvant chemotherapy versus standard chemotherapy in patients with high-risk soft-tissue sarcomas (ISG-STS 1001): An international, open-label, randomised, controlled, phase 3, multicentre trial. Lancet Oncol. 2017, 18, 812–822. [Google Scholar] [CrossRef]
- Hoang, N.T.; Acevedo, L.A.; Mann, M.J.; Tolani, B. A review of soft-tissue sarcomas: Translation of biological advances into treatment measures. Cancer Manag. Res. 2018, 10, 1089–1114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klemen, N.D.; Kelly, C.M.; Bartlett, E.K. The emerging role of immunotherapy for the treatment of sarcoma. J. Surg. Oncol. 2021, 123, 730–738. [Google Scholar] [CrossRef] [PubMed]
- Dufresne, A.; Lesluyes, T.; Ménétrier-Caux, C.; Brahmi, M.; Darbo, E.; Toulmonde, M.; Italiano, A.; Mir, O.; Le Cesne, A.; Le Guellec, S.; et al. Specific immune landscapes and immune checkpoint expressions in histotypes and molecular subtypes of sarcoma. OncoImmunology 2020, 9, 1792036. [Google Scholar] [CrossRef] [PubMed]
- Lines, J.L.; Pantazi, E.; Mak, J.; Sempere, L.F.; Wang, L.; O’Connell, S.; Ceeraz, S.; Suriawinata, A.A.; Yan, S.; Ernstoff, M.S.; et al. VISTA Is an Immune Checkpoint Molecule for Human T Cells. Cancer Res. 2014, 74, 1924–1932. [Google Scholar] [CrossRef] [Green Version]
- Issels, R.D.; Lindner, L.H.; Verweij, J.; Wust, P.; Reichardt, P.; Schem, B.C.; Abdel-Rahman, S.; Daugaard, S.; Salat, C.; Wendtner, C.M.; et al. Neo-adjuvant chemotherapy alone or with regional hyperthermia for localised high-risk soft-tissue sarcoma: A randomised phase 3 multicentre study. Lancet Oncol. 2010, 11, 561–570. [Google Scholar] [CrossRef] [Green Version]
- Knösel, T.; Emde, A.; Schluns, K.; Chen, Y.; Jurchott, K.; Krause, M.; Dietel, M.; Petersen, I. Immunoprofiles of 11 biomarkers using tissue microarrays identify prognostic subgroups in colorectal cancer. Neoplasia 2005, 7, 741–747. [Google Scholar] [CrossRef] [Green Version]
- Orth, M.F.; Buecklein, V.L.; Kampmann, E.; Subklewe, M.; Noessner, E.; Cidre-Aranaz, F.; Romero-Perez, L.; Wehweck, F.S.; Lindner, L.; Issels, R.; et al. A comparative view on the expression patterns of PD-L1 and PD-1 in soft tissue sarcomas. Cancer Immunol. Immunother. CII 2020, 69, 1353–1362. [Google Scholar] [CrossRef]
- Hmeljak, J.; Sanchez-Vega, F.; Hoadley, K.A.; Shih, J.; Stewart, C.; Heiman, D.; Tarpey, P.; Danilova, L.; Drill, E.; Gibb, E.A.; et al. Integrative Molecular Characterization of Malignant Pleural Mesothelioma. Cancer Discov. 2018, 8, 1548–1565. [Google Scholar] [CrossRef] [Green Version]
- Mansfield, A.S.; Murphy, S.J.; Peikert, T.; Yi, E.S.; Vasmatzis, G.; Wigle, D.A.; Aubry, M.C. Heterogeneity of Programmed Cell Death Ligand 1 Expression in Multifocal Lung Cancer. Clin. Cancer Res. 2016, 22, 2177–2182. [Google Scholar] [CrossRef] [Green Version]
- McLaughlin, J.; Han, G.; Schalper, K.A.; Carvajal-Hausdorf, D.; Pelekanou, V.; Rehman, J.; Velcheti, V.; Herbst, R.; Lorusso, P.; Rimm, D.L. Quantitative Assessment of the Heterogeneity of PD-L1 Expression in Non–Small-Cell Lung Cancer. JAMA Oncol. 2016, 2, 46. [Google Scholar] [CrossRef] [PubMed]
- Coindre, J.-M. Grading of Soft Tissue Sarcomas: Review and Update. Arch. Pathol. Lab. Med. 2006, 130, 1448–1453. [Google Scholar] [CrossRef]
- Issels, R.D.; Noessner, E.; Lindner, L.H.; Schmidt, M.; Albertsmeier, M.; Blay, J.Y.; Stutz, E.; Xu, Y.; Buecklein, V.; Altendorf-Hofmann, A.; et al. Immune infiltrates in patients with localised high-risk soft tissue sarcoma treated with neoadjuvant chemotherapy without or with regional hyperthermia: A translational research program of the EORTC 62961-ESHO 95 randomised clinical trial. Eur. J. Cancer 2021, 158, 123–132. [Google Scholar] [CrossRef]
- Huang, X.; Zhang, X.; Li, E.; Zhang, G.; Wang, X.; Tang, T.; Bai, X.; Liang, T. VISTA: An immune regulatory protein checking tumor and immune cells in cancer immunotherapy. J. Hematol. Oncol. 2020, 13, 83. [Google Scholar] [CrossRef] [PubMed]
- Blando, J.; Sharma, A.; Higa, M.G.; Zhao, H.; Vence, L.; Yadav, S.S.; Kim, J.; Sepulveda, A.M.; Sharp, M.; Maitra, A.; et al. Comparison of immune infiltrates in melanoma and pancreatic cancer highlights VISTA as a potential target in pancreatic cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 1692–1697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zong, L.; Zhou, Y.; Zhang, M.; Chen, J.; Xiang, Y. VISTA expression is associated with a favorable prognosis in patients with high-grade serous ovarian cancer. Cancer Immunol. Immunother. CII 2020, 69, 33–42. [Google Scholar] [CrossRef] [Green Version]
- Loeser, H.; Kraemer, M.; Gebauer, F.; Bruns, C.; Schröder, W.; Zander, T.; Persa, O.-D.; Alakus, H.; Hoelscher, A.; Buettner, R.; et al. The expression of the immune checkpoint regulator VISTA correlates with improved overall survival in pT1/2 tumor stages in esophageal adenocarcinoma. Oncoimmunology 2019, 8, e1581546. [Google Scholar] [CrossRef]
- Böger, C.; Behrens, H.-M.; Krüger, S.; Röcken, C. The novel negative checkpoint regulator VISTA is expressed in gastric carcinoma and associated with PD-L1/PD-1: A future perspective for a combined gastric cancer therapy? Oncoimmunology 2017, 6, e1293215. [Google Scholar] [CrossRef]
- Zhang, M.; Pang, H.-J.; Zhao, W.; Li, Y.-F.; Yan, L.-X.; Dong, Z.-Y.; He, X.-F. VISTA expression associated with CD8 confers a favorable immune microenvironment and better overall survival in hepatocellular carcinoma. BMC Cancer 2018, 18, 511. [Google Scholar] [CrossRef]
- Mulati, K.; Hamanishi, J.; Matsumura, N.; Chamoto, K.; Mise, N.; Abiko, K.; Baba, T.; Yamaguchi, K.; Horikawa, N.; Murakami, R.; et al. VISTA expressed in tumour cells regulates T cell function. Br. J. Cancer 2019, 120, 115–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, H.-X.; Gao, Y.; Fu, J.-C.; Zhou, Q.-H.; Wang, X.-X.; Bai, B.; Li, P.-F.; Huang, C.; Rong, Q.-X.; Ping, L.-Q.; et al. VISTA and PD-L1 synergistically predict poor prognosis in patients with extranodal natural killer/T-cell lymphoma. OncoImmunology 2021, 10, 1907059. [Google Scholar] [CrossRef] [PubMed]
- Tagliamento, M.; Agostinetto, E.; Borea, R.; Brandão, M.; Poggio, F.; Addeo, A.; Lambertini, M. VISTA: A Promising Target for Cancer Immunotherapy? ImmunoTargets Ther. 2021, 10, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Ward, J.F.; Pettaway, C.A.; Shi, L.Z.; Subudhi, S.K.; Vence, L.M.; Zhao, H.; Chen, J.; Chen, H.; Efstathiou, E.; et al. VISTA is an inhibitory immune checkpoint that is increased after ipilimumab therapy in patients with prostate cancer. Nat. Med. 2017, 23, 551–555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kakavand, H.; Jackett, L.A.; Menzies, A.M.; Gide, T.N.; Carlino, M.S.; Saw, R.P.M.; Thompson, J.F.; Wilmott, J.S.; Long, G.V.; Scolyer, R.A. Negative immune checkpoint regulation by VISTA: A mechanism of acquired resistance to anti-PD-1 therapy in metastatic melanoma patients. Mod. Pathol. 2017, 30, 1666–1676. [Google Scholar] [CrossRef]
- Wu, L.; Deng, W.-W.; Huang, C.-F.; Bu, L.-L.; Yu, G.-T.; Mao, L.; Zhang, W.-F.; Liu, B.; Sun, Z.-J. Expression of VISTA correlated with immunosuppression and synergised with CD8 to predict survival in human oral squamous cell carcinoma. Cancer Immunol. Immunother. CII 2017, 66, 627–636. [Google Scholar] [CrossRef]
- Signorelli, D.; Giannatempo, P.; Grazia, G.; Aiello, M.M.; Bertolini, F.; Mirabile, A.; Buti, S.; Vasile, E.; Scotti, V.; Pisapia, P.; et al. Patients Selection for Immunotherapy in Solid Tumors: Overcome the Naïve Vision of a Single Biomarker. BioMed Res. Int. 2019, 2019, 9056417. [Google Scholar] [CrossRef] [Green Version]


| Variation | n | % | |
| Total | 213 | 100 | |
| Sex | Male | 105 | 49 |
| Female | 108 | 51 | |
| Histological subtype | UPS | 77 | 36 |
| Leiomyosarcoma | 47 | 22 | |
| Synovial sarcoma | 27 | 13 | |
| Dedifferentiated liposarcoma | 34 | 16 | |
| Myxoid liposarcoma | 9 | 4 | |
| Angiosarcoma | 8 | 4 | |
| MPNST | 11 | 5 | |
| Location | Extremities | 71 | 33 |
| Retroperitoneal | 45 | 21 | |
| Abdominal/visceral | 35 | 17 | |
| Trunk | 55 | 26 | |
| Other | 7 | 3 | |
| Grading | Intermediate (G2) | 102 | 48 |
| High (G3) | 111 | 52 | |
| Size | <50 mm | 17 | 8 |
| 50–79 mm | 48 | 22 | |
| 80–120 mm | 62 | 29 | |
| >120 mm | 63 | 30 | |
| Missing | 23 | 11 | |
| Primary tumour/ recurrence | Primary tumour | 195 | 92 |
| Recurrence | 18 | 8 | |
| Metastatic disease | M0 | 195 | 92 |
| M1 | 18 | 8 | |
| Surgical margins | R0/R1 | 186 | 87 |
| R2 or no resection | 27 | 13 | |
| Radiotherapy | Done | 168 | 79 |
| Not done | 45 | 21 | |
| Regional hyperthermia | Done | 164 | 77 |
| Not done | 49 | 23 | |
| Chemotherapy | Neoadjuvant/perioperative | 213 | 100 |
| Adjuvant only | 0 | 0 | |
| Variation | VISTA Expression | TILs | |||||||||||
| Histological | 0 | 1 | 2 | 3 | Positive | Positive | Total | ||||||
| Subtype | n | % | n | % | n | % | n | % | n | % | n | % | n |
| UPS | 38 | 49% | 31 | 40% | 7 | 9% | 1 | 1% | 39 | 51% | 37 | 48% | 77 |
| Leiomyosarcoma | 25 | 53% | 21 | 45% | 1 | 2% | 0 | 0% | 22 | 47% | 16 | 34% | 47 |
| Synovial sarcoma | 14 | 52% | 13 | 48% | 0 | 0% | 0 | 0% | 13 | 48% | 3 | 11% | 27 |
| Liposarcoma | 32 | 74% | 10 | 23% | 1 | 2% | 0 | 0% | 11 | 26% | 17 | 40% | 43 |
| DDLPS | 25 | 74% | 8 | 24% | 1 | 3% | 0 | 0% | 9 | 26% | 15 | 44% | 34 |
| Myxoid | 7 | 78% | 2 | 22% | 0 | 0% | 0 | 0% | 2 | 22% | 2 | 22% | 9 |
| Angiosarcoma | 3 | 38% | 4 | 50% | 1 | 13% | 0 | 0% | 5 | 63% | 4 | 50% | 8 |
| MPNST | 5 | 45% | 6 | 55% | 0 | 0% | 0 | 0% | 6 | 55% | 3 | 27% | 11 |
| Total | 117 | 55% | 85 | 40% | 10 | 5% | 1 | 0% | 96 | 45% | 80 | 38% | 213 |
| Variation | VISTA | ||||||
| Total | Expression | No Expression | p | ||||
| TILs | 0–5 | 133 | 52 | 39% | 81 | 61% | 0.033 |
| ≥ 6 | 80 | 44 | 55% | 36 | 45% | ||
| Total | 213 | 96 | 45% | 117 | 55% | ||
| CD3 | No expression | 34 | 9 | 26% | 25 | 74% | 0.023 |
| Expression | 170 | 82 | 48% | 88 | 52% | ||
| Total | 204 | 91 | 45% | 113 | 55% | ||
| PD-1 | 0–3 | 145 | 58 | 40% | 87 | 60% | 0.046 |
| ≥ 4 | 61 | 34 | 56% | 27 | 44% | ||
| Total | 206 | 92 | 45% | 114 | 55% | ||
| PD-L1 | No expression | 171 | 73 | 43% | 98 | 57% | 0.031 |
| Expression | 31 | 20 | 65% | 11 | 35% | ||
| Total | 202 | 93 | 46% | 109 | 54% | ||
| Metastasis | M0 | 195 | 85 | 44% | 110 | 56% | 0.215 |
| M1 | 18 | 11 | 61% | 7 | 39% | ||
| Total | 213 | 96 | 45% | 117 | 55% | ||
| Location | Extremities | 73 | 37 | 51% | 36 | 49% | 0.249 |
| Non-extremities | 140 | 59 | 42% | 81 | 58% | ||
| Total | 213 | 96 | 45% | 117 | 55% | ||
| Grade | Intermediate | 102 | 37 | 36% | 65 | 64% | 0.019 |
| High | 111 | 59 | 53% | 52 | 47% | ||
| Total | 213 | 96 | 45% | 117 | 55% | ||
| Variation | HR (95% CI) | p-Value | |
|---|---|---|---|
| Age | Increase by 1 year | 1.015 (0.999–1.031) | 0.059 |
| Sex | Female (vs. male) | 0.800 (0.525–1.218) | 0.298 |
| VISTA | No expression (vs. expression) | 1.014 (1.014–2.363) | 0.043 |
| TILs | ≥6 (vs. 0–5) | 1.125 (0.726–1.743) | 0.597 |
| CD3 | No expression (vs. expression) | 0.982 (0.525–1.838) | 0.956 |
| PD-1 | ≥4 (vs. 0–3) | 1.104 (0.696–1.750) | 0.674 |
| PD-L1 | No expression (vs. expression) | 0.890 (0.488–1.622) | 0.703 |
| Size | ≥8 cm (vs. <8 cm) | 1.358 (0.866–2.129) | 0.183 |
| Metastasis | M1 (vs. M0) | 1.986 (1.008–3.912) | 0.047 |
| Grade | Grade 1/2 (vs. Grade 3) | 1.446 (0.937–2.231) | 0.095 |
| Surgical outcome | R2/not resected (vs. R0/1) | 5.936 (3.390–10.393) | <0.001 |
| Histotype | UPS (Reference) | 0.075 | |
| Leiomyosarcoma | 1.246 (0.727–2.135) | 0.424 | |
| Synovial sarcoma | 1.012 (0.474–2.159) | 0.975 | |
| Liposarcoma | 0.800 (0.450–1.423) | 0.447 | |
| Angiosarcoma | 3.992 (1.099–14.497) | 0.035 | |
| MPNST | 2.461 (1.010–5.995) | 0.047 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albertsmeier, M.; Altendorf-Hofmann, A.; Lindner, L.H.; Issels, R.D.; Kampmann, E.; Dürr, H.-R.; Angele, M.K.; Klauschen, F.; Werner, J.; Jungbluth, A.A.; et al. VISTA in Soft Tissue Sarcomas: A Perspective for Immunotherapy? Cancers 2022, 14, 1006. https://doi.org/10.3390/cancers14041006
Albertsmeier M, Altendorf-Hofmann A, Lindner LH, Issels RD, Kampmann E, Dürr H-R, Angele MK, Klauschen F, Werner J, Jungbluth AA, et al. VISTA in Soft Tissue Sarcomas: A Perspective for Immunotherapy? Cancers. 2022; 14(4):1006. https://doi.org/10.3390/cancers14041006
Chicago/Turabian StyleAlbertsmeier, Markus, Annelore Altendorf-Hofmann, Lars H. Lindner, Rolf D. Issels, Eric Kampmann, Hans-Roland Dürr, Martin K. Angele, Frederick Klauschen, Jens Werner, Achim A. Jungbluth, and et al. 2022. "VISTA in Soft Tissue Sarcomas: A Perspective for Immunotherapy?" Cancers 14, no. 4: 1006. https://doi.org/10.3390/cancers14041006
APA StyleAlbertsmeier, M., Altendorf-Hofmann, A., Lindner, L. H., Issels, R. D., Kampmann, E., Dürr, H.-R., Angele, M. K., Klauschen, F., Werner, J., Jungbluth, A. A., & Knösel, T. (2022). VISTA in Soft Tissue Sarcomas: A Perspective for Immunotherapy? Cancers, 14(4), 1006. https://doi.org/10.3390/cancers14041006






