The Impact of an Incidental Dose on Axillary Tumor Control and Toxicity in Localized Breast Cancer: A Retrospective Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Ethical Approval
2.2. Patients
2.3. Contouring and Planning
2.4. Statistical Analysis
3. Results
Dosimetric Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dominici, L.S.; Morrow, M.; Mittendorf, E.; Bellon, J.; King, T.A. Trends and Controversies in Multi-Disciplinary Care of the Breast Cancer Patient. Curr. Probl. Surg. 2016, 53, 559–595. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Donker, M.; Slaets, L.; van Tienhoven, G.; Rutgers, E.J.T. Axillary lymph node dissection versus axillary radiotherapy in patients with a positive sentinel node: The AMAROS trial. Ned. Tijdschr. Geneeskd. 2015, 159, A9302. [Google Scholar] [PubMed]
- Li, C.Z.; Zhang, P.; Li, R.W.; Wu, C.T.; Zhang, X.P.; Zhu, H.C. Axillary Lymph Node Dissection versus Sentinel Lymph Node Biopsy Alone for Early Breast Cancer with Sentinel Node Metastasis: A Meta-Analysis. Eur. J. Surg. Oncol. 2015, 41, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.E.; Ballman, K.V.; McCall, L.; Beitsch, P.D.; Brennan, M.B.; Kelemen, P.R.; Ollila, D.W.; Hansen, N.M.; Whitworth, P.W.; Blumencranz, P.W.; et al. Effect of Axillary Dissection vs No Axillary Dissection on 10-Year Overall Survival among Women with Invasive Breast Cancer and Sentinel Node Metastasis: The ACOSOG Z0011 (Alliance) Randomized Clinical Trial. JAMA 2017, 318, 918–926. [Google Scholar] [CrossRef]
- Rastogi, K.; Sharma, S.; Gupta, S.; Agarwal, N.; Bhaskar, S.; Jain, S. Dosimetric Comparison of IMRT versus 3DCRT for Post-Mastectomy Chest Wall Irradiation. Radiat. Oncol. J. 2018, 36, 71–78. [Google Scholar] [CrossRef]
- Liu, H.; Chen, X.; He, Z.; Li, J. Evaluation of 3D-CRT, IMRT and VMAT Radiotherapy Plans for Left Breast Cancer Based on Clinical Dosimetric Study. Comput. Med. Imaging Graph. 2016, 54, 1–5. [Google Scholar] [CrossRef]
- Hong, L.; Hunt, M.; Chui, C.; Spirou, S.; Forster, K.; Lee, H.; Yahalom, J.; Kutcher, G.J.; McCormick, B. Intensity-Modulated Tangential Beam Irradiation of the Intact Breast. Int. J. Radiat. Oncol. Biol. Phys. 1999, 44, 1155–1164. [Google Scholar] [CrossRef]
- Warren, L.E.G.; Miller, C.L.; Horick, N.; Skolny, M.N.; Jammallo, L.S.; Sadek, B.T.; Shenouda, M.N.; O’Toole, J.A.; MacDonald, S.M.; Specht, M.C.; et al. The Impact of Radiation Therapy on the Risk of Lymphedema after Treatment for Breast Cancer: A Prospective Cohort Study. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 565–571. [Google Scholar] [CrossRef]
- Mukesh, M.B.; Barnett, G.C.; Wilkinson, J.S.; Moody, A.M.; Wilson, C.; Dorling, L.; Chan Wah Hak, C.; Qian, W.; Twyman, N.; Burnet, N.G.; et al. Randomized Controlled Trial of Intensity-Modulated Radiotherapy for Early Breast Cancer: 5-Year Results Confirm Superior Overall Cosmesis. J. Clin. Oncol. 2013, 31, 4488–4495. [Google Scholar] [CrossRef]
- Gonzalez, V.J.; Buchholz, D.J.; Langen, K.M.; Olivera, G.H.; Chauhan, B.; Meeks, S.L.; Ruchala, K.J.; Haimerl, J.; Lu, W.; Kupelian, P.A. Evaluation of Two Tomotherapy-Based Techniques for the Delivery of Whole-Breast Intensity-Modulated Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 284–290. [Google Scholar] [CrossRef]
- Reynders, T.; Tournel, K.; De Coninck, P.; Heymann, S.; Vinh-Hung, V.; Van Parijs, H.; Duchateau, M.; Linthout, N.; Gevaert, T.; Verellen, D.; et al. Dosimetric Assessment of Static and Helical TomoTherapy in the Clinical Implementation of Breast Cancer Treatments. Radiother. Oncol. 2009, 93, 71–79. [Google Scholar] [CrossRef]
- Ashikaga, T.; Krag, D.N.; Land, S.R.; Julian, T.B.; Anderson, S.J.; Brown, A.M.; Skelly, J.M.; Harlow, S.P.; Weaver, D.L.; Mamounas, E.P.; et al. Morbidity Results from the NSABP B-32 Trial Comparing Sentinel Lymph Node Dissection versus Axillary Dissection. J. Surg. Oncol. 2010, 102, 111–118. [Google Scholar] [CrossRef]
- Gross, J.P.; Sachdev, S.; Helenowski, I.B.; Lipps, D.; Hayes, J.P.; Donnelly, E.D.; Strauss, J.B. Radiation Therapy Field Design and Lymphedema Risk after Regional Nodal Irradiation for Breast Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 71–78. [Google Scholar] [CrossRef]
- Hayes, S.B.; Freedman, G.M.; Li, T.; Anderson, P.R.; Ross, E. Does Axillary Boost Increase Lymphedema Compared with Supraclavicular Radiation Alone after Breast Conservation? Int. J. Radiat. Oncol. Biol. Phys. 2008, 72, 1449–1455. [Google Scholar] [CrossRef]
- Abbassi, L.M.; Arsène-Henry, A.; Amessis, M.; Kirova, Y.M. Radiation Dose to the Low Axilla in Patients Treated for Early-Stage Breast Cancer by Locoregional Intensity-Modulated Radiotherapy (IMRT). Cancer Radiother. 2021; in press. [Google Scholar] [CrossRef]
- Schmitt, M.; Pin, Y.; Pflumio, C.; Mathelin, C.; Pivot, X.; Noel, G. Incidental Axillary Dose Delivery to Axillary Lymph Node Levels I-III by Different Techniques of Whole-Breast Irradiation: A Systematic Literature Review. Strahlenther. Onkol. 2021, 197, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.; Chambrelant, I.; Pflumio, I.; Hild, C.; Petit, T.; Noël, G. A retrospective analysis of survival and prognostic factors of axillary recurrence of breast cancer. Cancer Radiother. 2022; Under review. [Google Scholar]
- Offersen, B.V.; Boersma, L.J.; Kirkove, C.; Hol, S.; Aznar, M.C.; Biete Sola, A.; Kirova, Y.M.; Pignol, J.-P.; Remouchamps, V.; Verhoeven, K.; et al. ESTRO Consensus Guideline on Target Volume Delineation for Elective Radiation Therapy of Early Stage Breast Cancer. Radiother. Oncol. 2015, 114, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Hennequin, C. Le Rapport Alpha/Bêta Revisité à L’heure de L’hypofractionnement. Available online: https://www.em-consulte.com/article/837910/le-rapport-alphabeta-revisite-a-l-heure-de-l-hypof (accessed on 8 September 2021).
- Borm, K.J.; Oechsner, M.; Düsberg, M.; Buschner, G.; Weber, W.; Combs, S.E.; Duma, M.-N. Irradiation of Regional Lymph Node Areas in Breast Cancer—Dose Evaluation According to the Z0011, AMAROS, EORTC 10981-22023 and MA-20 Field Design. Radiother. Oncol. 2020, 142, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Reznik, J.; Cicchetti, M.G.; Degaspe, B.; Fitzgerald, T.J. Analysis of Axillary Coverage during Tangential Radiation Therapy to the Breast. Int. J. Radiat. Oncol. Biol. Phys. 2005, 61, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Belkacemi, Y.; Bigorie, V.; Pan, Q.; Bouaita, R.; Pigneur, F.; Itti, E.; Badaoui, H.; Assaf, E.; Caillet, P.; Calitchi, E.; et al. Breast Radiotherapy (RT) Using Tangential Fields (TgF): A Prospective Evaluation of the Dose Distribution in the Sentinel Lymph Node (SLN) Area as Determined Intraoperatively by Clip Placement. Ann. Surg. Oncol. 2014, 21, 3758–3765. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, J.; Kim, S.-W.; Son, S.H. Dosimetric Evaluation of Incidental Irradiation to the Axilla during Whole Breast Radiotherapy for Patients with Left-Sided Early Breast Cancer in the IMRT Era. Medicine 2016, 95, e4036. [Google Scholar] [CrossRef] [PubMed]
- Alço, G.; Iğdem, S.I.; Ercan, T.; Dinçer, M.; Şentürk, R.; Atilla, S.; Oral Zengin, F.; Okkan, S. Coverage of Axillary Lymph Nodes with High Tangential Fields in Breast Radiotherapy. Br. J. Radiol. 2010, 83, 1072–1076. [Google Scholar] [CrossRef] [PubMed]
- Li, X.A.; Tai, A.; Arthur, D.W.; Buchholz, T.A.; Macdonald, S.; Marks, L.B.; Moran, J.M.; Pierce, L.J.; Rabinovitch, R.; Taghian, A.; et al. Variability of Target and Normal Structure Delineation for Breast Cancer Radiotherapy: An RTOG Multi-Institutional and Multiobserver Study. Int. J. Radiat. Oncol. Biol. Phys. 2009, 73, 944–951. [Google Scholar] [CrossRef]
- Fleissig, A.; Fallowfield, L.J.; Langridge, C.I.; Johnson, L.; Newcombe, R.G.; Dixon, J.M.; Kissin, M.; Mansel, R.E. Post-Operative Arm Morbidity and Quality of Life. Results of the ALMANAC Randomised Trial Comparing Sentinel Node Biopsy with Standard Axillary Treatment in the Management of Patients with Early Breast Cancer. Breast Cancer Res. Treat. 2006, 95, 279–293. [Google Scholar] [CrossRef]
- van Wely, B.J.; Teerenstra, S.; Schinagl, D.A.X.; Aufenacker, T.J.; de Wilt, J.H.W.; Strobbe, L.J.A. Systematic Review of the Effect of External Beam Radiation Therapy to the Breast on Axillary Recurrence after Negative Sentinel Lymph Node Biopsy. Br. J. Surg. 2011, 98, 326–333. [Google Scholar] [CrossRef]
- Fisher, B.; Anderson, S.; Bryant, J.; Margolese, R.G.; Deutsch, M.; Fisher, E.R.; Jeong, J.-H.; Wolmark, N. Twenty-Year Follow-up of a Randomized Trial Comparing Total Mastectomy, Lumpectomy, and Lumpectomy plus Irradiation for the Treatment of Invasive Breast Cancer. N. Engl. J. Med. 2002, 347, 1233–1241. [Google Scholar] [CrossRef]

| Characteristics | Mean (SD) | Median (Q25–75) | Min | Max | n |
|---|---|---|---|---|---|
| Age at diagnosis (years) | 61.2 (11.6) | 62.0 (52.0; 70.0) | 33.0 | 85.0 | 171 |
| Distance between the upper beam boundary and humeral head (cm) | 3.12 (1.21) | 3.00 (2.30; 4.03) | 0.500 | 5.70 | 148 |
| Average heart dose (Gy) | 1.51 (1.87) | 0.720 (0.440; 1.85) | 0.120 | 10.2 | 158 |
| Total dose (Gy) | 60.9 (8.30) | 66.0 (50.0; 66.0) | 20.0 | 66.0 | 171 |
| Follow-up time (month) | 40.7 (20.3) | 38.0 (29.0; 48.0) | 2.00 | 123 | 171 |
| Fractionation | 28.8 (6.53) | 33.0 (19.0; 33.0) | 5.00 | 33.0 | 171 |
| Body mass index | 26.9 (5.56) | 25.7 (22.4; 31.2) | 16.0 | 49.4 | 170 |
| Ki 67 (%) | 19.5 (17.9) | 15.0 (8.00; 21.5) | 2.00 | 80.0 | 166 |
| Number of sentinel lymph nodes removed | 2.40 (1.60) | 2.00 (1.00; 3.00) | 0 | 9.00 | 169 |
| Size (mm) | 14.6 (7.71) | 13.0 (9.50; 19.0) | 1.00 | 45.0 | 163 |
| Target breast volume | 778.4 (448) | 686.3 (453.6; 1040.4) | 119.0 | 2439.0 | 163 |
| Target parietal volume | 135.2 (37.6) | 127.3 (120.3; 139.8) | 95.0 | 219.6 | 8 |
| Ln1 vol | 148 (44.2) | 142 (111; 176) | 67.7 | 281 | 171 |
| Ln1 Dmax | 52.8 (14.1) | 53.4 (47.9; 62.8) | 15.3 | 69.9 | 171 |
| Ln1 Dmean | 24.5 (12.1) | 24.7 (14.2; 33.5) | 0.607 | 57.8 | 171 |
| Ln2 vol | 33.8 (12.4) | 31.3 (26.1; 38.0) | 14.7 | 85.6 | 171 |
| Ln2 Dmax | 30.3 (18.4) | 36.2 (15.0; 46.3) | 0.200 | 61.7 | 171 |
| Ln2 Dmean | 8.05 (10.6) | 3.70 (1.25; 10.3) | 0.100 | 50.5 | 171 |
| Ln3 vol | 36.5 (70.7) | 29.1 (24.8; 35.8) | 13.4 | 946 | 171 |
| Ln3 Dmax | 12.4 (16.4) | 3.40 (1.30; 18.5) | 0.100 | 53.7 | 171 |
| Ln3 Dmean | 4.45 (11.5) | 0.800 (0.405; 1.85) | 0 | 50.7 | 171 |
| Characteristics | n (%) | |
|---|---|---|
| Lymph node recurrence | No | 165 (98%) |
| Yes | 3 (1.8%) | |
| Adjuvant chemotherapy | No | 136 (80%) |
| Yes | 35 (20%) | |
| Neoadjuvant chemotherapy | No | 162 (95%) |
| Yes | 9 (5.3%) | |
| Conservative surgery | No | 8 (4.7%) |
| Yes | 163 (95%) | |
| Sentinel lymph node | No | 2 (1.2%) |
| Yes | 169 (99%) | |
| Scarff–Bloom–Richardson grade | 1 | 64 (39%) |
| 2 | 70 (42%) | |
| 3 | 31 (19%) | |
| HER | Negative | 159 (94%) |
| Positive | 10 (5.9%) | |
| Histology | Invasive ductal carcinoma | 155 (88%) |
| Invasive lobular carcinoma | 14 (8.2%) | |
| Medullary carcinoma | 1 (0.59%) | |
| Triple negative | No | 157 (92%) |
| Yes | 14 (8.2%) | |
| Yes | 150 (88%) | |
| Laterality | Right | 100 (58%) |
| Left | 71 (42%) | |
| Lymphoedema | No | 164 (99%) |
| Yes | 2 (1.2%) | |
| Normofractionation | No | 44 (26%) |
| Yes | 127 (74%) | |
| Estrogen receptor positivity | Yes | 153 (89%) |
| No | 17 (9.9%) | |
| NA | 1 (0.58%) | |
| Regional node irradiation | Yes | 8 (4.7%) |
| No | 163 (95%) | |
| Radiation technique | 3DRT | 117 (68%) |
| HT | 30 (18%) | |
| IMRT | 24 (14%) | |
| Standard tangential | No | 30 (20%) |
| Yes | 119 (80%) | |
| Variable | Correlation Coefficient (95% CI) | n | p | Test | Correlation Coefficient |
|---|---|---|---|---|---|
| Ln1 Dmax | 0.0714 (−0.0795; 0.219) | 171 | 0.35 | Pearson | - |
| Ln1 Dmean | 0.306 (0.164; 0.436) | 171 | <0.001 | Pearson | - |
| Ln1 vol | 0.480 (0.356; 0.588) | 171 | <0.001 | Pearson | - |
| Ln2 Dmax | 0.276 (0.131; 0.409) | 171 | <0.001 | Pearson | - |
| Ln2 Dmean | 0.117 (−0.0338; 0.262) | 171 | 0.13 | Pearson | - |
| Ln2 vol | 0.220 (0.0720; 0.358) | 171 | <0.01 | Pearson | - |
| Ln3 Dmax | - | 171 | <0.001 | Spearman | 0.363 |
| Ln3 Dmean | −0.0713 (−0.219; 0.0796) | 171 | 0.35 | Pearson | - |
| Ln3 vol | 0.143 (−0.00736; 0.287) | 171 | 0.062 | Pearson | - |
| Variable | HT (n = 30) | ST (n = 119) | n | p |
|---|---|---|---|---|
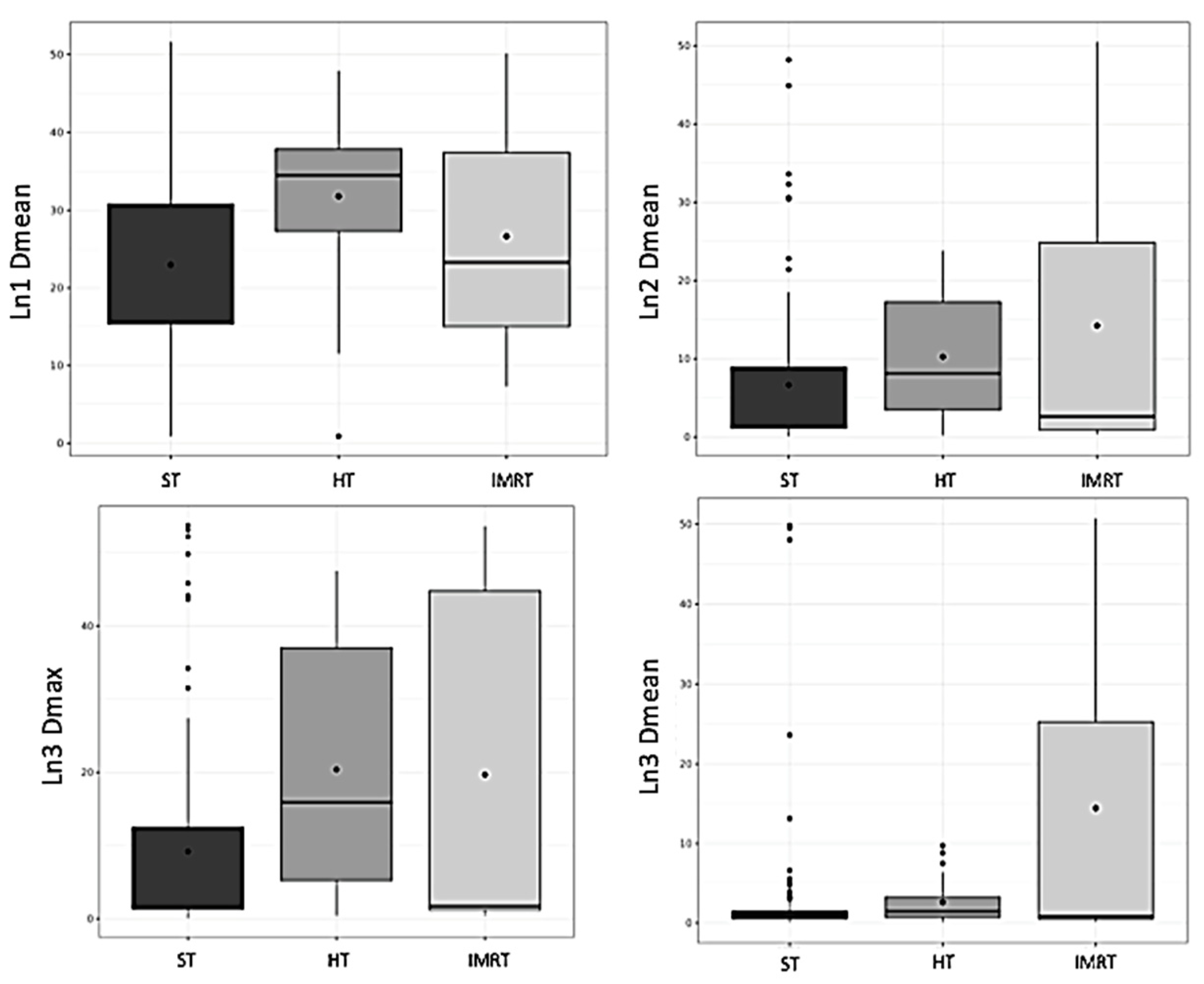
| Ln1 Dmax, median | 52.0 (46.8; 62.0) | 53.6 (6.0; 63.2) | 149 | 0.59 |
| Ln1 Dmean, median | 34.3 (27.0; 37.9) | 23.2 (13.1; 30.2) | 149 | <0.001 |
| Ln2 Dmax, median | 41.9 (33.1; 47.1) | 34.1 (11.8; 46.0) | 149 | 0.066 |
| Ln2 Dmean, median | 8.10 (3.05; 14.5) | 3.25 (1.00; 8.82) | 149 | <0.01 |
| Ln3 Dmax, median | 15.9 (3.76; 36.9) | 2.40 (1.10; 12.7) | 149 | <0.001 |
| Ln3 Dmean, median | 1.45 (0.602; 3.00) | 0.700 (0.400; 1.30) | 149 | <0.01 |
| Variable | Supraclavicular Irradiation | n | p | |
|---|---|---|---|---|
| Yes (n = 8) | No (n = 163) | |||
| Ln1 Dmean (Gy) | 38.6 (30.5; 43.6) | 24.6 (13.6; 32.6) | 171 | <0.01 |
| Ln2 Dmean (Gy) | 35.9 (28.6; 44.6) | 3.30 (1.15; 9.40) | 171 | <0.001 |
| Ln3 Dmean (Gy) | 49.5 (47.6; 49.7) | 0.700 (0.403; 1.50) | 171 | <0.001 |
| Variable | AMAROS | ACOSOG | Retrospective Study | ||
|---|---|---|---|---|---|
| ST | HT | ST | HT | ||
| Ln1 Dmean (Gy) | 52.6 ± 6.5 | 42.2 ± 13.8 | 48.4 ± 4.1 | 23.2 (3.1; 30.2) | 34.3 (27.0; 37.9) |
| Ln2 Dmean (Gy) | 49.4 ± 3.7 | 35.6 ± 17.6 | 47.5 ± 3.9 | 3.25 (1.00; 8.82) | 8.10 (3.05; 14.5) |
| Ln3 Dmean (Gy) | 47.3 ± 1.7 | 12.0 ± 12.8 | 44.7 ± 5.6 | 0.700 (0.400; 1.30) | 1.45 (0.602; 3.00) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmitt, M.; Chambrelant, I.; Hong Chheang, P.; Pflumio, C.; Hild, C.; Petit, T.; Noël, G. The Impact of an Incidental Dose on Axillary Tumor Control and Toxicity in Localized Breast Cancer: A Retrospective Analysis. Cancers 2022, 14, 807. https://doi.org/10.3390/cancers14030807
Schmitt M, Chambrelant I, Hong Chheang P, Pflumio C, Hild C, Petit T, Noël G. The Impact of an Incidental Dose on Axillary Tumor Control and Toxicity in Localized Breast Cancer: A Retrospective Analysis. Cancers. 2022; 14(3):807. https://doi.org/10.3390/cancers14030807
Chicago/Turabian StyleSchmitt, Martin, Isabelle Chambrelant, Parigna Hong Chheang, Carole Pflumio, Carole Hild, Thierry Petit, and Georges Noël. 2022. "The Impact of an Incidental Dose on Axillary Tumor Control and Toxicity in Localized Breast Cancer: A Retrospective Analysis" Cancers 14, no. 3: 807. https://doi.org/10.3390/cancers14030807
APA StyleSchmitt, M., Chambrelant, I., Hong Chheang, P., Pflumio, C., Hild, C., Petit, T., & Noël, G. (2022). The Impact of an Incidental Dose on Axillary Tumor Control and Toxicity in Localized Breast Cancer: A Retrospective Analysis. Cancers, 14(3), 807. https://doi.org/10.3390/cancers14030807






