Racial/Ethnic Differences and Trends in Pathologic Complete Response Following Neoadjuvant Chemotherapy for Breast Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
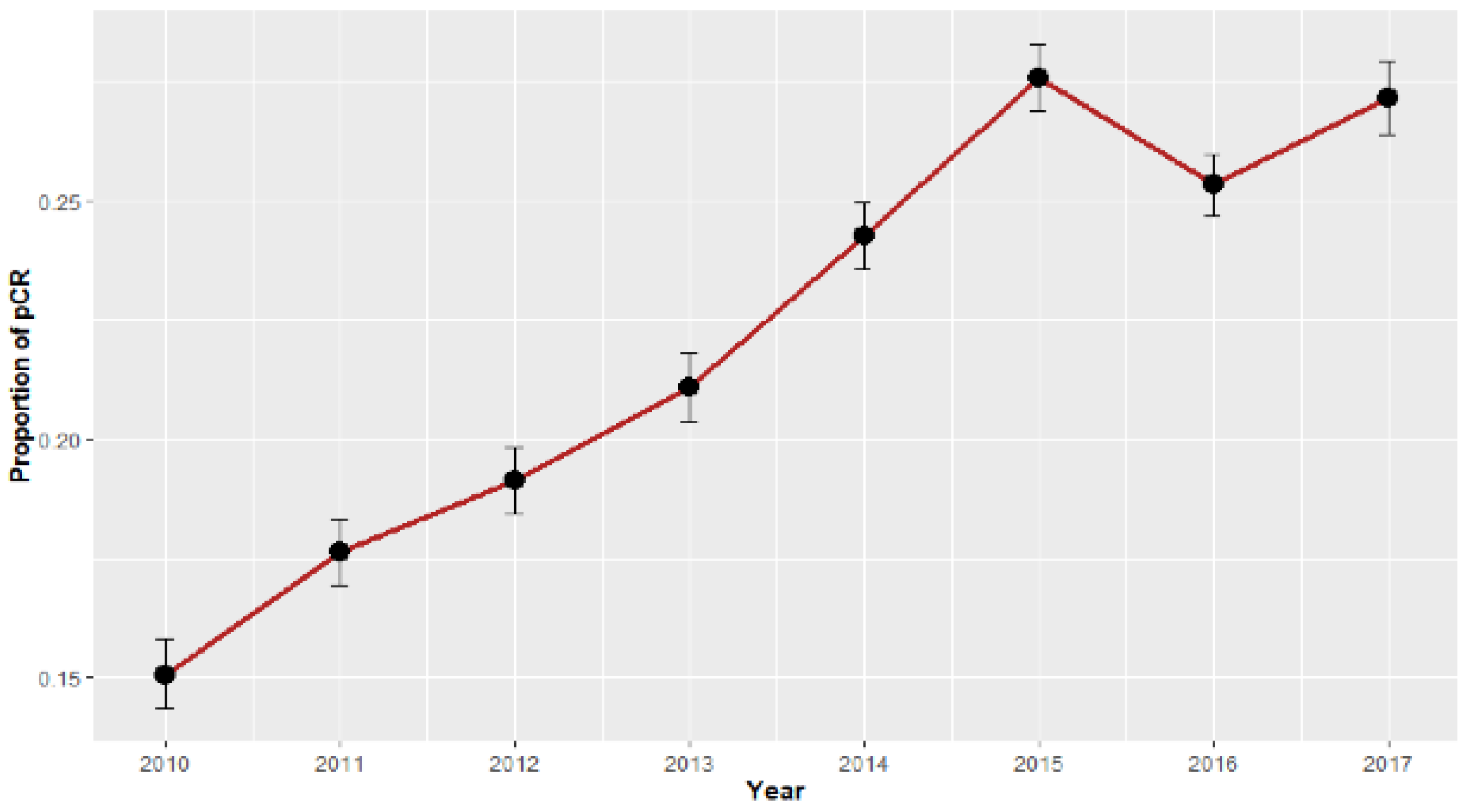
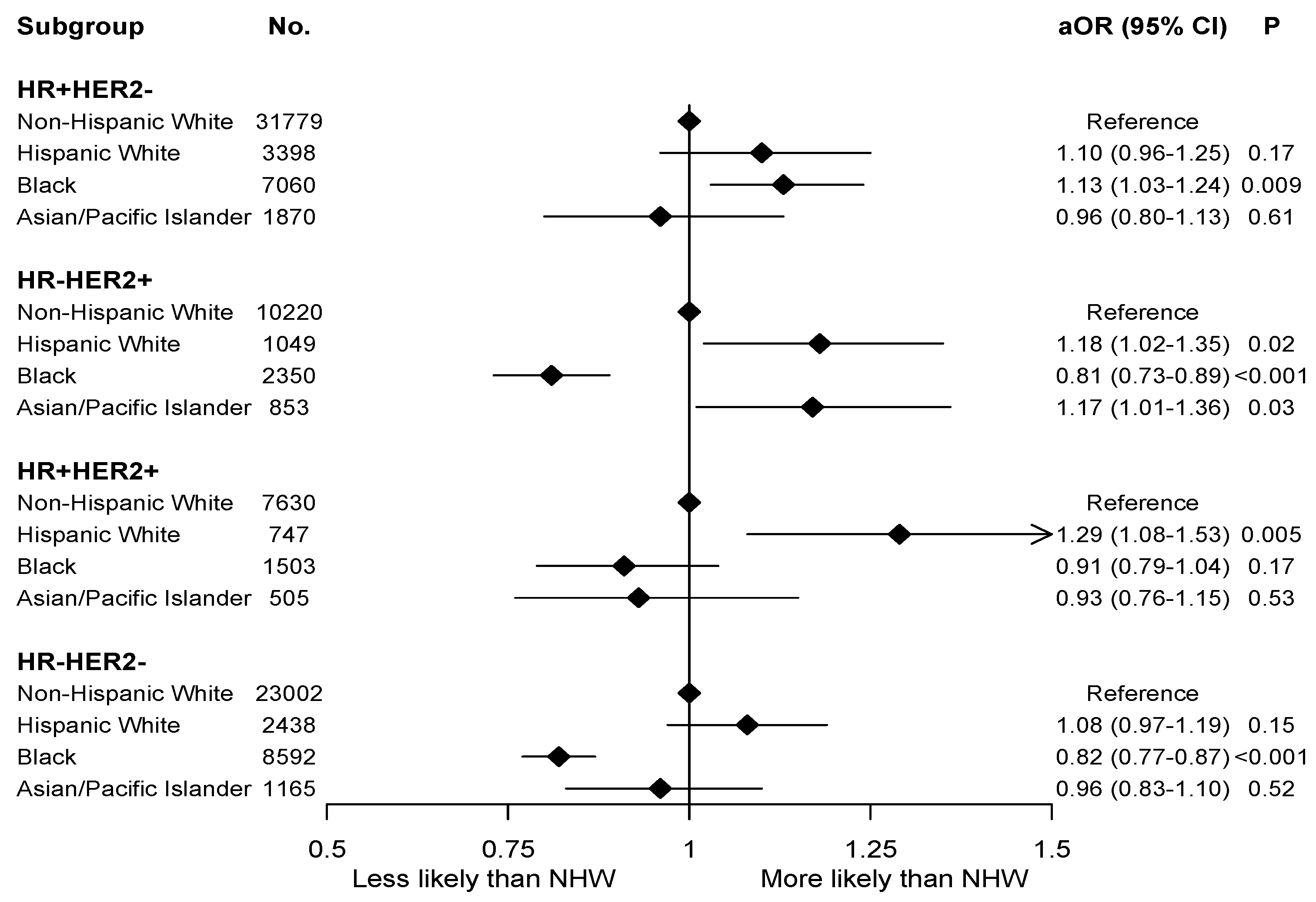
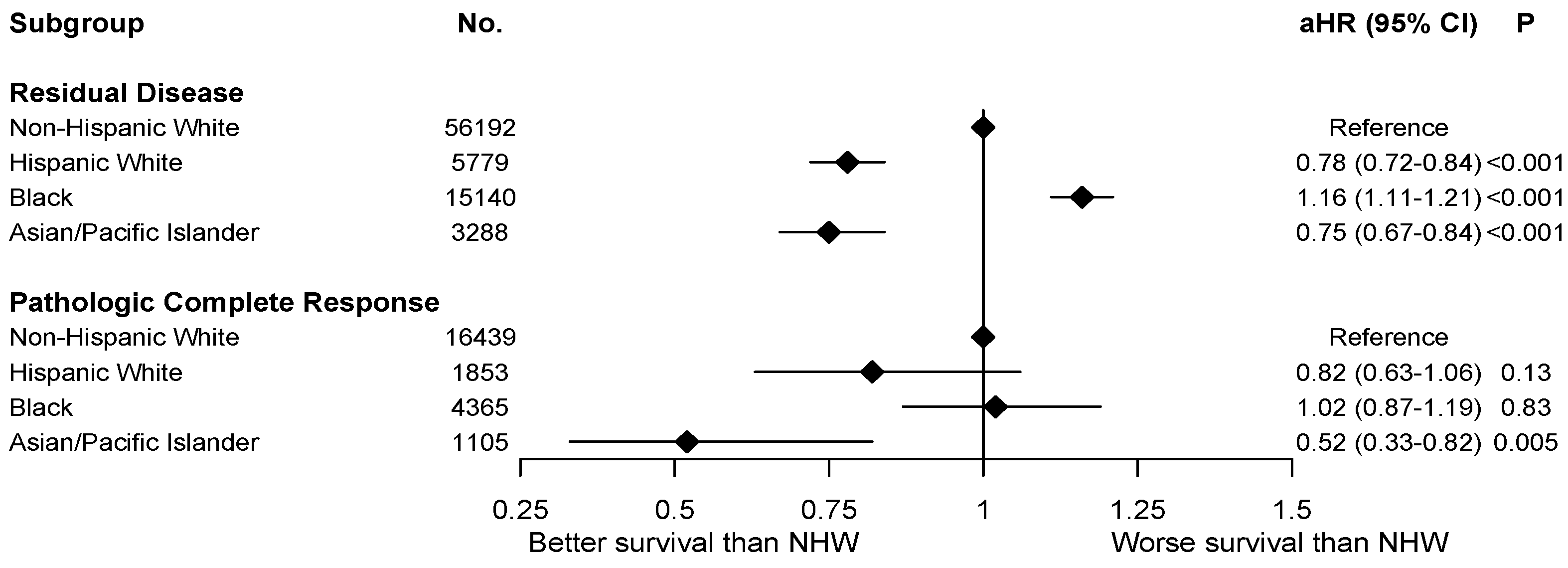
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cortazar, P.; Zhang, L.; Untch, M.; Mehta, K.; Costantino, J.P.; Wolmark, N.; Bonnefoi, H.; Cameron, D.; Gianni, L.; Valagussa, P.; et al. Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. Lancet 2014, 384, 164–172. [Google Scholar] [CrossRef] [Green Version]
- Spring, L.M.; Fell, G.; Arfe, A.; Sharma, C.; Greenup, R.A.; Reynolds, K.L.; Smith, B.L.; Alexander, B.M.; Moy, B.; Isakoff, S.J.; et al. Pathologic Complete Response after Neoadjuvant Chemotherapy and Impact on Breast Cancer Recurrence and Survival: A Comprehensive Meta-analysis. Clin. Cancer Res. 2020, 26, 2838–2848. [Google Scholar] [CrossRef] [PubMed]
- Schmid, P.; Cortes, J.; Pusztai, L.; McArthur, H.; Kümmel, S.; Bergh, J.; Denkert, C.; Park, Y.H.; Hui, R.; Harbeck, N.; et al. Pembrolizumab for Early Triple-Negative Breast Cancer. N. Engl. J. Med. 2020, 382, 810–821. [Google Scholar] [CrossRef]
- van Ramshorst, M.S.; van der Voort, A.; van Werkhoven, E.D.; Mandjes, I.A.; Kemper, I.; Dezentjé, V.O.; Oving, I.M.; Honkoop, A.H.; Tick, L.W.; van de Wouw, A.J.; et al. Neoadjuvant chemotherapy with or without anthracyclines in the presence of dual HER2 blockade for HER2-positive breast cancer (TRAIN-2): A multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2018, 19, 1630–1640. [Google Scholar] [CrossRef]
- Food and Drug Administration. Guidance for Industry: Pathological Complete Response in Neoadjuvant Treatment of High- Risk Early-Stage Breast Cancer: Use as an Endpoint to Support Accelerated Approval; Food and Drug Administration: Silver Spring, MD, USA, 2014. [Google Scholar]
- Sutton, E.J.; Braunstein, L.Z.; El-Tamer, M.B.; Brogi, E.; Hughes, M.; Bryce, Y.; Gluskin, J.S.; Powell, S.; Woosley, A.; Tadros, A.; et al. Accuracy of Magnetic Resonance Imaging–Guided Biopsy to Verify Breast Cancer Pathologic Complete Response After Neoadjuvant Chemotherapy. JAMA Netw. Open 2021, 4, e2034045. [Google Scholar] [CrossRef]
- Mamounas, E.P.; Bandos, H.; White, J.R.; Julian, T.B.; Khan, A.J.; Shaitelman, S.F.; Torres, M.A.; Vicini, F.; Ganz, P.A.; McCloskey, S.A.; et al. NRG Oncology/NSABP B-51/RTOG 1304: Phase III trial to determine if chest wall and regional nodal radiotherapy (CWRNRT) post mastectomy (Mx) or the addition of RNRT to whole breast RT post breast-conserving surgery (BCS) reduces invasive breast cancer recurrence-free interval (IBCR-FI) in patients (pts) with pathologically positive axillary (PPAx) nodes who are ypN0 after neoadjuvant chemotherapy (NC). J. Clin. Oncol. 2019, 37, TPS600. [Google Scholar]
- Albain, K.S.; Unger, J.M.; Crowley, J.J.; Coltman, C.A.; Hershman, D.L. Racial Disparities in Cancer Survival Among Randomized Clinical Trials Patients of the Southwest Oncology Group. JNCI J. Natl. Cancer Inst. 2009, 101, 984–992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hershman, D.L.; Unger, J.M.; Barlow, W.E.; Hutchins, L.F.; Martino, S.; Osborne, C.K.; Livingston, R.B.; Albain, K.S. Treatment Quality and Outcomes of African American Versus White Breast Cancer Patients: Retrospective Analysis of Southwest Oncology Studies S8814/S8897. J. Clin. Oncol. 2009, 27, 2157–2162. [Google Scholar] [CrossRef] [Green Version]
- Newman, L.A.; Griffith, K.A.; Jatoi, I.; Simon, M.S.; Crowe, J.P.; Colditz, G. Meta-Analysis of Survival in African American and White American Patients With Breast Cancer: Ethnicity Compared With Socioeconomic Status. J. Clin. Oncol. 2006, 24, 1342–1349. [Google Scholar] [CrossRef]
- Sparano, J.A.; Wang, M.; Zhao, F.; Stearns, V.; Martino, S.; Ligibel, J.A.; Perez, E.A.; Saphner, T.; Wolff, A.; Sledge, G.W.; et al. Race and Hormone Receptor–Positive Breast Cancer Outcomes in a Randomized Chemotherapy Trial. JNCI J. Natl. Cancer Inst. 2012, 104, 406–414. [Google Scholar] [CrossRef]
- Mac Gregor, M.C.; Litton, J.K.; Chen, H.; Giordano, S.H.; Hudis, C.A.; Wolff, A.; Valero, V.; Hortobagyi, G.N.; Bondy, M.L.; Gonzalez-Angulo, A.M. Pathologic complete response in breast cancer patients receiving anthracycline- and taxane-based neoadjuvant chemotherapy. Cancer 2010, 116, 4168–4177. [Google Scholar] [CrossRef] [Green Version]
- Dawood, S.; Broglio, K.; Kau, S.-W.; Green, M.C.; Giordano, S.H.; Meric-Bernstam, F.; Buchholz, T.A.; Albarracin, C.; Yang, W.T.; Hennessy, B.T.; et al. Triple Receptor–Negative Breast Cancer: The Effect of Race on Response to Primary Systemic Treatment and Survival Outcomes. J. Clin. Oncol. 2009, 27, 220–226. [Google Scholar] [CrossRef] [Green Version]
- Warner, E.T.; Ballman, K.V.; Strand, C.; Boughey, J.C.; Buzdar, A.U.; Carey, L.A.; Sikov, W.M.; Partridge, A.H. Impact of race, ethnicity, and BMI on achievement of pathologic complete response following neoadjuvant chemotherapy for breast cancer: A pooled analysis of four prospective Alliance clinical trials (A151426). Breast Cancer Res. Treat. 2016, 159, 109–118. [Google Scholar] [CrossRef] [Green Version]
- Killelea, B.K.; Yang, V.Q.; Wang, S.-Y.; Hayse, B.; Mougalian, S.; Horowitz, N.R.; Chagpar, A.B.; Pusztai, L.; Lannin, D.R. Racial Differences in the Use and Outcome of Neoadjuvant Chemotherapy for Breast Cancer: Results From the National Cancer Data Base. J. Clin. Oncol. 2015, 33, 4267–4276. [Google Scholar] [CrossRef]
- Gianni, L.; Pienkowski, T.; Im, Y.-H.; Roman, L.; Tseng, L.-M.; Liu, M.-C.; Lluch, A.; Staroslawska, E.; De La Haba-Rodriguez, J.; Im, S.-A.; et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): A randomised multicentre, open-label, phase 2 trial. Lancet Oncol. 2012, 13, 25–32. [Google Scholar] [CrossRef]
- Moon, H.-G.; Im, S.-A.; Han, W.; Oh, D.-Y.; Han, S.-W.; Keam, B.; Park, I.A.; Chang, J.M.; Moon, W.K.; Cho, N.; et al. Estrogen receptor status confers a distinct pattern of response to neoadjuvant chemotherapy: Implications for optimal durations of therapy: Distinct patterns of response according to ER expression. Breast Cancer Res. Treat. 2012, 134, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Ring, A.; Smith, I.; Ashley, S.; Fulford, L.G.; Lakhani, S.R. Oestrogen receptor status, pathological complete response and prognosis in patients receiving neoadjuvant chemotherapy for early breast cancer. Br. J. Cancer 2004, 91, 2012–2017. [Google Scholar] [CrossRef] [PubMed]
- Zambetti, M.; Mansutti, M.; Gómez, P.; Lluch, A.; Dittrich, C.; Zamagni, C.; Ciruelos, E.; Pavesi, L.; Semiglazov, V.; De Benedictis, E.; et al. Pathological complete response rates following different neoadjuvant chemotherapy regimens for operable breast cancer according to ER status, in two parallel, randomized phase II trials with an adaptive study design (ECTO II). Breast Cancer Res. Treat. 2011, 132, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Bear, H.D.; Tang, G.; Rastogi, P.; Geyer, C.E.; Robidoux, A.; Atkins, J.N.; Baez-Diaz, L.; Brufsky, A.M.; Mehta, R.S.; Fehrenbacher, L.; et al. Bevacizumab Added to Neoadjuvant Chemotherapy for Breast Cancer. N. Engl. J. Med. 2012, 366, 310–320. [Google Scholar] [CrossRef] [Green Version]
- Nahleh, Z.; Botrus, G.; Dwivedi, A.; Jennings, M.; Nagy, S.; Tfayli, A. Bevacizumab in the neoadjuvant treatment of human epidermal growth factor receptor 2-negative breast cancer: A meta-analysis of randomized controlled trials. Mol. Clin. Oncol. 2019, 10, 357–365. [Google Scholar] [CrossRef] [Green Version]
- Meng, M.; Wang, H.; Zaorsky, N.G.; Sun, B.; Zhu, L.; Song, Y.; Li, F.; Dong, Y.; Wang, J.; Chen, H.; et al. Risk-adapted stereotactic body radiation therapy for central and ultra-central early-stage inoperable non-small cell lung cancer. Cancer Sci. 2019, 110, 3553–3564. [Google Scholar] [CrossRef]
- Singer, C.F.; Tan, Y.; Fitzal, F.; Steger, G.G.; Egle, D.; Reiner, A.; Rudas, M.; Moinfar, F.; Gruber, C.; Petru, E.; et al. Pathological Complete Response to Neoadjuvant Trastuzumab Is Dependent on HER2/CEP17 Ratio in HER2-Amplified Early Breast Cancer. Clin. Cancer Res. 2017, 23, 3676–3683. [Google Scholar] [CrossRef] [Green Version]
- Gianni, L.; Zambetti, M.; Clark, K.; Baker, J.; Cronin, M.; Wu, J.; Mariani, G.; Rodriguez, J.; Carcangiu, M.; Watson, D.; et al. Gene Expression Profiles in Paraffin-Embedded Core Biopsy Tissue Predict Response to Chemotherapy in Women With Locally Advanced Breast Cancer. J. Clin. Oncol. 2005, 23, 7265–7277. [Google Scholar] [CrossRef]
- Holowatyj, A.N.; Cote, M.L.; Ruterbusch, J.J.; Ghanem, K.; Schwartz, A.G.; Vigneau, F.D.; Gorski, D.H.; Purrington, K.S. Racial Differences in 21-Gene Recurrence Scores Among Patients With Hormone Receptor–Positive, Node-Negative Breast Cancer. J. Clin. Oncol. 2018, 36, 652–658. [Google Scholar] [CrossRef]
- Hoskins, K.F.; Danciu, O.C.; Ko, N.Y.; Calip, G.S. Association of Race/Ethnicity and the 21-Gene Recurrence Score With Breast Cancer–Specific Mortality Among US Women. JAMA Oncol. 2021, 7, 370. [Google Scholar] [CrossRef] [PubMed]
- Kan, Z.; Ding, Y.; Kim, J.; Jung, H.H.; Chung, W.; Lal, S.; Cho, S.; Fernandez-Banet, J.; Lee, S.K.; Kim, S.W.; et al. Multi-omics profiling of younger Asian breast cancers reveals distinctive molecular signatures. Nat. Commun. 2018, 9, 1725. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.W.; Zabidi, M.M.A.; Ng, P.-S.; Meng, M.-Y.; Hasan, S.N.; Sandey, B.; Sammut, S.-J.; Yip, C.-H.; Rajadurai, P.; Rueda, O.M.; et al. The molecular landscape of Asian breast cancers reveals clinically relevant population-specific differences. Nat. Commun. 2020, 11, 6433. [Google Scholar] [CrossRef]
- Yao, S.; Cheng, T.-Y.D.; Elkhanany, A.; Yan, L.; Omilian, A.; I Abrams, S.; Evans, S.; Hong, C.-C.; Qi, Q.; Davis, W.; et al. Breast Tumor Microenvironment in Black Women: A Distinct Signature of CD8+ T-Cell Exhaustion. JNCI: J. Natl. Cancer Inst. 2021, 113, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.X.; Khera, R.; Miccio, J.A.; Jairam, V.; Chang, E.; Yu, J.B.; Park, H.S.; Krumholz, H.M.; Aneja, S. Prevalence of Missing Data in the National Cancer Database and Association With Overall Survival. JAMA Netw. Open 2021, 4, e211793. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, S.J.; Serra, L.M.; Yu, B.; Farrugia, M.K.; Iovoli, A.J.; Yu, H.; Yao, S.; Oladeru, O.T.; Singh, A.K. Racial/Ethnic Differences and Trends in Pathologic Complete Response Following Neoadjuvant Chemotherapy for Breast Cancer. Cancers 2022, 14, 534. https://doi.org/10.3390/cancers14030534
Ma SJ, Serra LM, Yu B, Farrugia MK, Iovoli AJ, Yu H, Yao S, Oladeru OT, Singh AK. Racial/Ethnic Differences and Trends in Pathologic Complete Response Following Neoadjuvant Chemotherapy for Breast Cancer. Cancers. 2022; 14(3):534. https://doi.org/10.3390/cancers14030534
Chicago/Turabian StyleMa, Sung Jun, Lucas M. Serra, Brian Yu, Mark K. Farrugia, Austin J. Iovoli, Han Yu, Song Yao, Oluwadamilola T. Oladeru, and Anurag K. Singh. 2022. "Racial/Ethnic Differences and Trends in Pathologic Complete Response Following Neoadjuvant Chemotherapy for Breast Cancer" Cancers 14, no. 3: 534. https://doi.org/10.3390/cancers14030534
APA StyleMa, S. J., Serra, L. M., Yu, B., Farrugia, M. K., Iovoli, A. J., Yu, H., Yao, S., Oladeru, O. T., & Singh, A. K. (2022). Racial/Ethnic Differences and Trends in Pathologic Complete Response Following Neoadjuvant Chemotherapy for Breast Cancer. Cancers, 14(3), 534. https://doi.org/10.3390/cancers14030534





